Abstract
Bacterial cellulose (BC) is a natural polymer secreted by microorganisms with excellent tensile strength (TS) and Young’s modulus (YM). However, the elongation at break (EAB) and toughness (TH) of dehydrated BC membranes are inadequate for some special applications. In this study, ionic liquid 1-allyl-3-methylimidazolium chloride (AmimCl) was introduced into BC membranes to fabricate composite membranes. It was found that AmimCl could dissolve the BC nanofibers to a certain extent, and destroy the hydrogen bonds between the protofibrils. Therefore, compared with pure BC membrane, BC/AmimCl composite membranes exhibited improved EAB and TH. In addition, AmimCl had a positive effect on the thermal stability and transparency of the composite membranes. This study shows that ionic liquids have great potential to change the mechanical properties, optical properties and thermal stability of BC membranes.
Introduction
Bacterial cellulose (BC) is a biologically friendly polymer derived from microbial fermentation and consisted of a network of ultrafine fibers, which belongs to the same group of natural cellulose as plant-based cellulose, and both of them share the same molecular formulae.1,2 BC has the advantages of artificially controlled synthesis, biodegradability, high purity, high crystallinity, high degree of polymerization, and non-toxic. 3 It has been widely used in many fields, such as food packaging, cosmetics, and electrical components. 4 In addition to this, compared to plant-based cellulose, BC-derived materials have good biocompatibility and broad application prospects in tissue engineering, wound dressings and various biomedical fields.5–7
Mechanical properties are macroscopic properties of BC membranes, reflecting its response to external forces like tension, compression, bending, and impact in varying environments. The main parameters describing the mechanical properties of BC include tensile strength (TS), Young’s modulus (YM), elongation at break (EAB), and toughness (TH). The TS and YM of BC are mainly determined by the number of effective bonds generated by the inter-fiber interactions and the arrangement of fibers.8,9 The TH of BC is closely related to inter-fiber slippage and fiber-orientation. It has been shown that BC possesses ordinary TS and YM because of the abundance of hydroxyl groups on its surface. 10 The strong hydrogen bonding network of BC also increases the difficulty of hydrogen bond breakage and protofibril reorganization, leading to slippage and inelastic deformation of BC protofibrils, which keep its EAB and TH at a low level. 11 Undehydrated BC wet membrane demonstrates superior EAB and TH. However, the dried film of BC after dehydration possesses more prominent TS and YM, while EAB and TH remain at a lower level.12,13 The difference in mechanical properties between the two states is mainly due to the difference in water content. However, the poor TS and YM as well as high moisture content significantly limited the application of BC in areas, such as high-strength flexible electronic materials, 14 vibrating membranes for microphones and headphones, food packaging materials, 15 water treatment filtration membranes, 16 battery separators. 17 Therefore, improving the mechanical properties of BC after dehydration to meet the needs of more fields has become an urgent problem.
In order to adapt to higher application requirements, several strategies have been developed to modify the mechanical property indexes of BC membranes. Typical methods include chemical modification, polymer reinforcement, nanoparticle reinforcement, and orderly arrangement of fibers. Currently, reinforcements such as graphene oxide18,19 and fluoride 20 are added in situ during the cultivation process of BC membranes, which can significantly increase the toughness and strain capacity of BC membranes. Calcium-modified BC membranes using calcium gluconate as a carbon source has good mechanical properties because calcium gluconate alters the ultrastructure of BC,21,22 which makes it better for applications in different mechanical requirements. However, it is difficult to change the properties of BC membrane by adding inorganic or organic substances during the incubation process, and often comes at the expense of BC yield, and potentially damage BC structure. 20 Chemical modification and exogenous substance compounding can also be used to improve the mechanical properties of BC membranes by altering chemical bonding and adhesion.23,24 In addition, the mechanical properties of BC membranes depend on the ordered arrangement of the fibers to some extent. The movement of the strain can be controlled by changing the static magnetic field 25 and applying a direct current electric field, 26 which in turn regulated the arrangement of the protofibrils for improving the mechanical properties of the BC membrane. While these methods of modifying BC membranes have higher costs and more complicated devices, which have certain limitations.
Ionic liquids, which are molten salt systems composed entirely of ions, have been used as effective plasticizers for cellulosic materials due to their oxidative stability, non-volatility, high polarity, and especially good ability to break the internal hydrogen bonds of cellulose.27–30 In addition, it has been reported that the introduction of ionic liquids could improve the mechanical properties of cellulose membrane.31–33 Inspired by these advantages, in this study, 1-allyl-3-methylimidazolium chloride (AmimCl) were introduced into BC membrane using vacuum filtration, then a hot-pressing process were performed to acquire the dried composite membranes (Figure 1). The morphology and structures were characterized by scanning electron microscopy (SEM) equipped with energy dispersive X-ray spectroscopy (EDS), X-ray diffraction (XRD), and fourier transform infrared spectroscopy (FTIR). The properties including mechanical properties, thermal stability and optical properties were evaluated. Schematic diagram of the composite membrane development.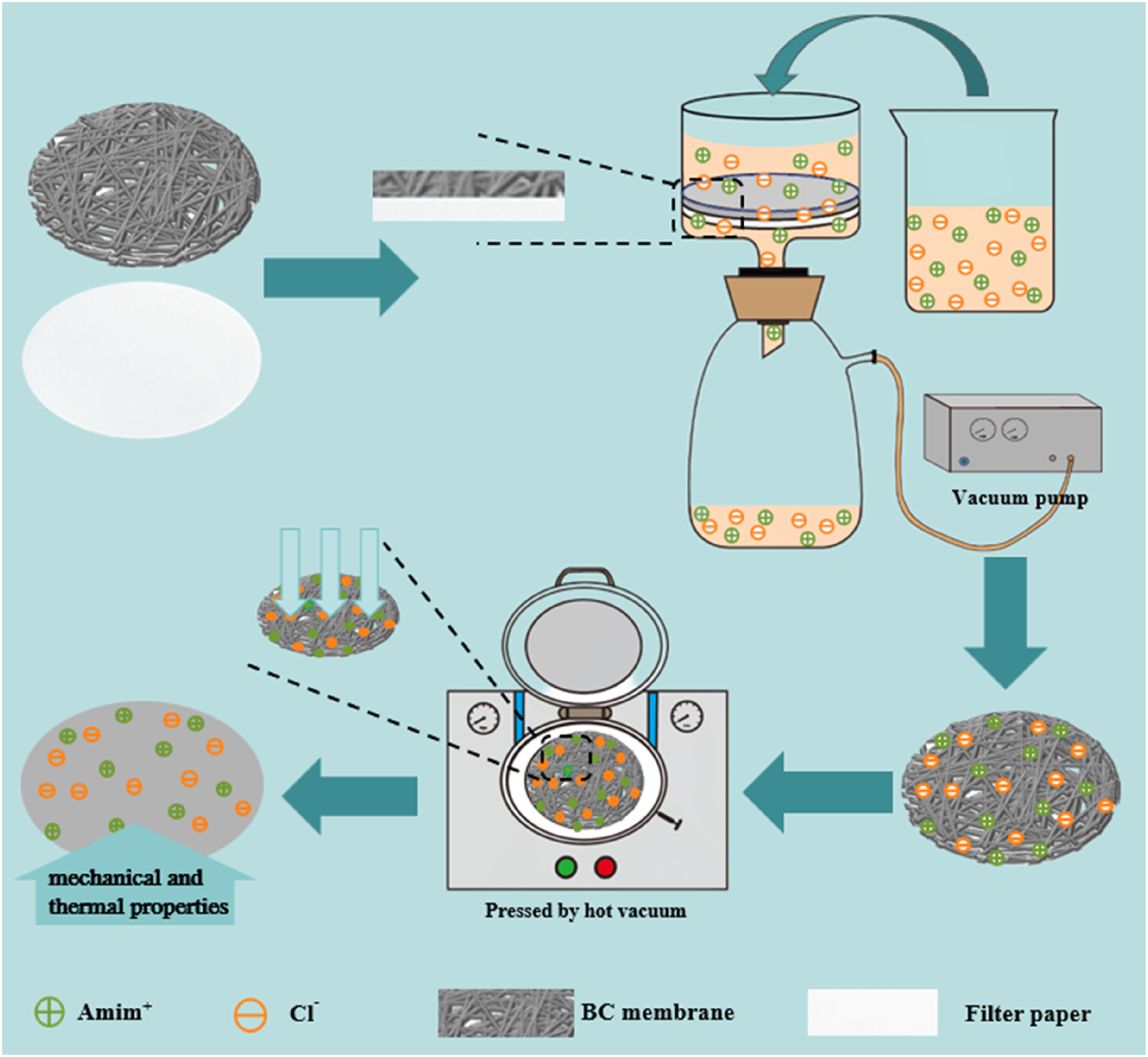
Materials and methods
Materials
Glucose, tryptophan and yeast extract were obtained from Oxoid Ltd (UK). Glacial acetic acid was purchased from Tianjin Chemical Reagent Factory (Tianjin, China). Disodium hydrogen phosphate was supplied by Tianjin Beitianyi Chemical Reagent Factory (Tianjin, China). Sodium hydroxide (NaOH) was purchased from Tianjin Chemical Reagent Factory (Tianjin, China). Sodium chloride (NaCl) was supplied by Shanghai Hutest Laboratory Equipment Co. Ltd (Shanghai, China). 1-allyl-3-methylimidazolium chloride (AmimCl) was purchased from Shanghai Dibao Chemical Technology Co. Ltd (Shanghai, China). Qualitative filter paper purchased from Hangzhou Fuyang North Wood Pulp Paper Co. Ltd (Hangzhou, China). None of the purchased reagents were further processed.
Preparation and purification of BC membranes
BC membranes were prepared by the method described in our previous literature.
34
Briefly, the activated
Preparation of BC/AmimCl composite membranes
After 48 h of fermentation, BC membranes with 2 mm thick obtained. Then, the purified BC membranes were served as filter membrane, and 30 mL of aqueous solutions containing 0.1, 0.5, 1.0, 1.5, 2.0, 2.5% of AmimCl slowly flowed through the BC membrane under vacuum condition, respectively. During the filtration process, a filter paper was placed under the BC membrane to maintain the flatness of the BC membrane. Finally, the wet BC/AmimCl composite membranes were dehydrated by a hot-pressing process using a paper sheet dryer (100°C, 5 min) (RK3A-KWT, Protein Technologies, Austria). The obtained dried BC/AmimCl composite membranes treated by the aqueous solutions containing 0.1, 0.5, 1.0, 1.5, 2.0, 2.5% of AmimCl were denoted as BC/AmimCl0.1, BC/AmimCl0.5, BC/AmimCl1.0, BC/AmimCl1.5, BC/AmimCl2.0, and BC/AmimCl2.5, respectively.
Structural and morphological characterization
The Fourier transform infrared spectrophotometer (IS50, Nicolet, USA) with attenuated total reflectance mode (ATR-FTIR) was used to analyze the chemical composition of dried BC, BC/AmimCl0.5 and BC/AmimCl2.5 membranes. The ATR-FTIR spectra were recorded within the range 400-4000 cm−1 with a resolution of 4 cm−1 and 32 scans. The microstructure and elemental composition of the composite surface were examined using a Field Emission Scanning Electron Microscope (FE-SEM) (Apreo, FEI, USA) equipped with an energy dispersive X-ray (EDS) energy spectrometer. X-ray diffraction (XRD) (Smartlab 9Kw, Rigaku Corporation, Japan) was employed to analyze the crystalline structure of samples, with an angular range of 5∼80°, a speed of 5°/min, and a step sizes of 0.02.
Mechanical properties
The dried composite membranes were cut into 60 mm × 15 mm strips. The mechanical properties of the samples were tested using microcomputer-controlled electronic universal testing machine (CMT4503, Meters Industrial Systems, China). The tensile speed of 5 mm/min, the marking distance L0 = 30 mm, and the initial test force of 0 N were adjusted. Each sample was tested an average of 10 times and data were processed and plotted using origin software.
The tensile strength is expressed as 
The Young’s modulus is expressed as 
Thermogravimetric analysis (TGA)
The TGA of dried BC, BC/AmimCl0.5 and BC/AmimCl2.5 membranes was conducted using the thermogravimetric analyzer (TGA-Q50, TA, USA). In total, 10 mg of dried samples was heated from room temperature to 800°C at a heating rate of 10°C/min under
Transparency analysis
The light transmittance of the membrane samples was performed on a UV spectrophotometer (UV-2550 PC, SHIMADZU, Japan) in the wavelength range of 200-700 nm. The composite membranes with flat surface were cut into strips and firmly attached to the side of a cell. A blank cell was chosen as a control, and each group of samples was tested three times, and the average value was calculated. The transmittance was calculated by the formula:
Results and discussion
Development and structural characterization of BC/AmimCl composite membranes
To prepare BC/AmimCl composite membrane, the wet BC membrane was used as the filter membrane, and the AmimCl aqueous solution flowed slowly through the BC membrane by vacuum filtration, followed by a hot-pressing treatment to remove water.
Then, ATR-FTIR was used to analyze the chemical composition and intermolecular interactions of BC membranes and BC/AmimCl composite membranes. As shown in Figure 2(a), the characteristic peaks at 3367 cm−1, 2904 cm−1, 1741 cm−1, and 1370 cm−1 were observed for the pure BC membrane.35,36 Both BC/AmimCl0.5 and BC/AmimCl2.5 also exhibited characteristic peaks of BC, indicating that the composite membranes still retained the structural features of BC despite the attachment of AmimCl. In addition, the new characteristic peaks at 1550 cm−1 and 1563 cm−1 for BC/AmimCI0.5 and BC/AmimCl2.5 composite membranes belonged to the characteristic peaks of N-H, C=N stretching vibrations in the imidazole cation of AmimCl, respectively. Compared with the BC membrane, the rang 3000-3600 cm−1 corresponding to the -OH stretching vibration became narrower for composite membranes, which can be attributed to the breakdown and rearrangement of hydrogen bonds in BC by AmimCl.31,37 (a) ATR-FTIR and (b) XRD spectra of BC and BC/AmimCl composite membranes.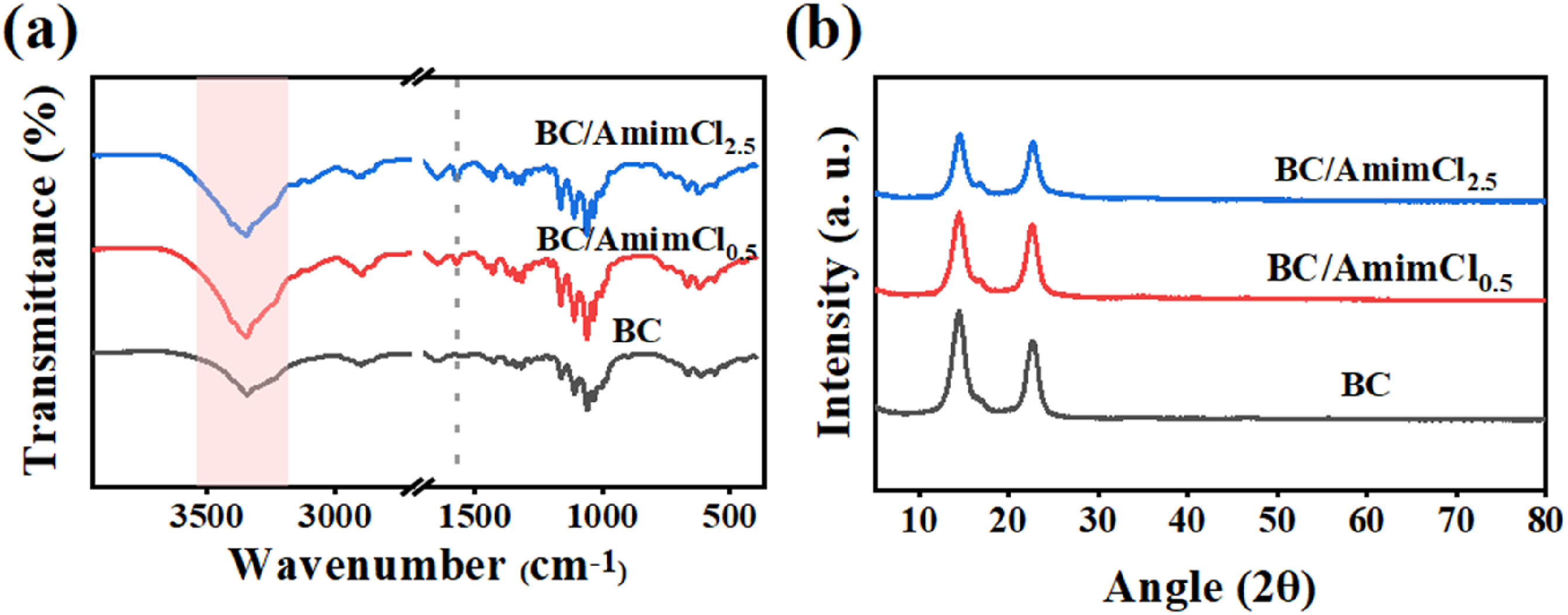
XRD was used to analyze the effect of AmimCl doping on the crystal structure of BC. In Figure 2(b), all samples exhibited identical diffraction peaks at 2
FE-SEM was used to observe the microstructures of BC, BC/AmimCl0.5 and BC/AmimCl2.5 samples. As shown in Figure 3, the pure BC membrane exhibited a dense fiber network. In addition, some small fibers were observed within the fiber network of the BC sample. Compared with BC membrane, the fine fibers decreased in the BC/AmimCl composite membranes. Further analysis of the images revealed that the porosity of the composite membranes decreased after the hot-press treatment (Table S1) and the fibers became denser (Figure 3(d)–(f)). In addition, the average diameter of the fibers in the BC/AmimCl composite membranes decreased compared to the pure BC membranes (Figgure S1). The EDS further confirmed the presence of chlorine (Cl) and nitrogen (N) in the BC/AmimCl0.5 composite membrane (Figure 3(g)–(i)), which constitute AmimCl. In addition, the surface density of the composite membranes was evaluated (Table S1), and the increase in surface density also confirmed the successful adhesion of AmimCl to the fiber membrane. The changes in the parameters associated with the composite membrane itself can be attributed to the solubilizing effect of AmimCl on cellulose. The AmimCl could dissociate into separate Cl− and Amim+ ions, then the free Cl− ion binds to the cellulose hydroxyl proton and the free cation binds to the cellulose hydroxyl oxygen, which in turn breaks the hydrogen bonds in the cellulose, leading to cellulose solubilization.
30
FE-SEM micrographs of the composite membranes of pristine BC, BC/AmimCl0.5 and BC/AmimCl2.5 before (a-c) and after (d-e) hot-press treatment. (g-i) Elemental maps of BC/AmimCl0.5 before hot-press treatment.
Mechanical property
Static uniaxial in-plane tensile tests were conducted to assess the mechanical properties of dried BC/AmimCl membranes with different content of AmimCl, including TS, YM, EAB, and TH. The tensile testing curves were depicted in Figure S2 and all relevant data were summarized in Figure 4. The tensile testing curves of the composite membranes well showed the mechanical information of the membranes under different conditions. The pure BC membrane exhibited TS of 91.81 ± 12.72 Mpa, YM of 3.96 ± 0.59 GPa, EAB of 2.23 ± 0.23%, and TH of 1.24 ± 0.15 MJ/m3. When the concentration of AmimCl was less than 0.1%, the introduction of AmimCl had no obvious effect on the TS of BC membrane. Increasing the concentration of AmimCl to 0.5%, the TS of composite membrane began to decrease. The YM of BC membrane reduced with the increase of AmimCl concentration as the concentration of AmimCl was greater than 0.1%. Compared with TS and YM, the AmimCl exhibited a different effect on the EAB and TH of the composite membranes. The EAB and TH of composite membrane increased with the increasing concentration of AmimCl. The BC/AmimCl2.5 exhibited EAB of 13.75 ± 0.72%, and TH of 4.25 ± 0.15 MJ/m3, which were 6 and 4 times of that of pure BC membrane, respectively. AmimCl as a plasticizer greatly improved the EAB and TH of the composite membranes, but also showed a sacrifice in TS and YM. The mechanical properties of BC/AmimCl composite membranes with different contents of AmimCl. (a) TS, (b) YM, (c) EAB and (d) TH. Error bars show standard deviation with at least 10 repeats.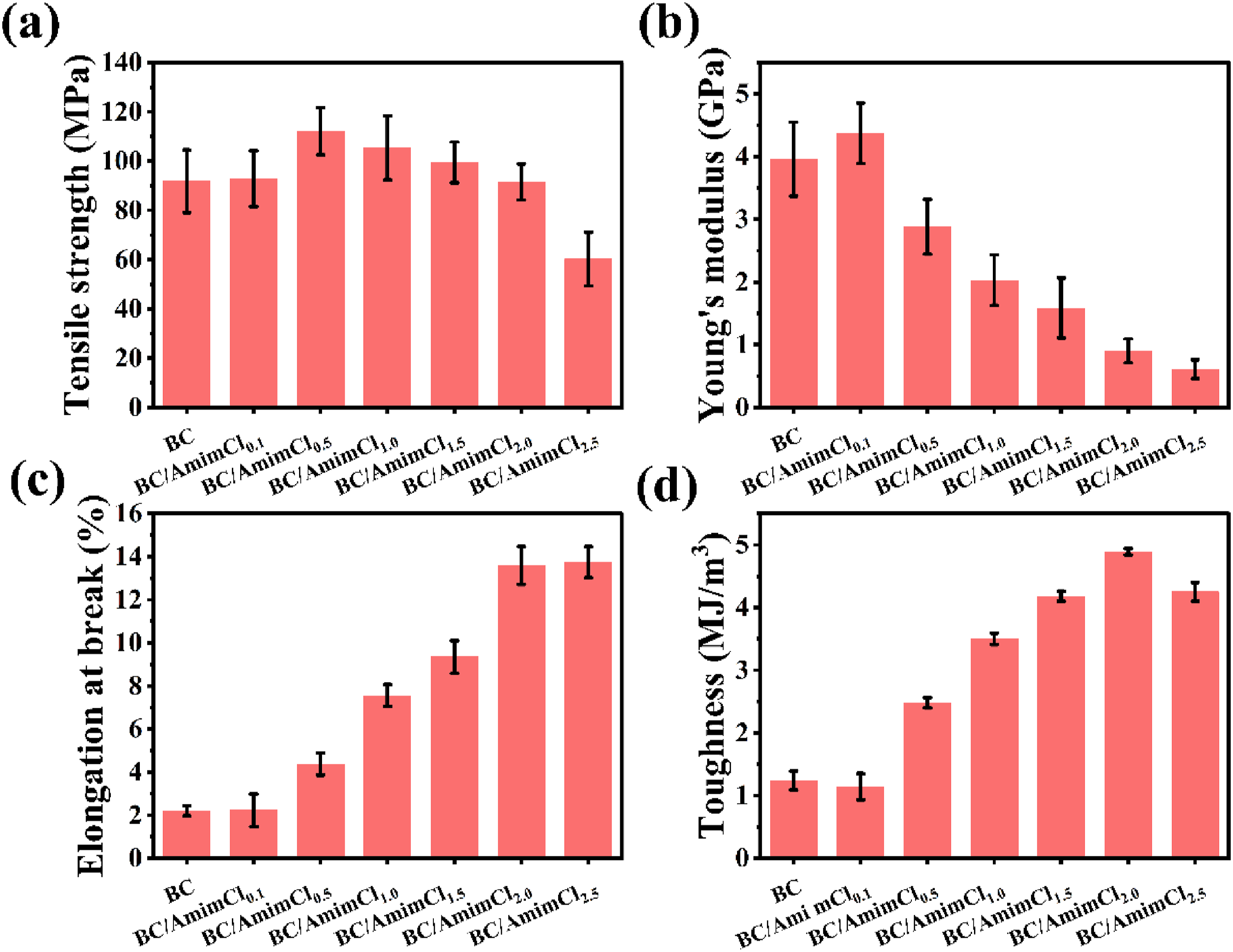
The improvement of EAB and TH of BC/AmimCl composite membranes by low concentration of AmimCl could be attributed to the fact that the ionic liquid AmimCl acted as a plasticizer and solvent for cellulose attached to the BC membranes. The attached AmimCl could disrupt the hydrogen bonding between the BC fibers, softened the rigid BC membrane, and make it easier for single fibers in the BC membrane to migrate and slide.31,42 This allows the fibers to move more freely in the matrix, resulting in better stress dispersion and reduced stress concentration when subjected to external forces. This stress dispersion effect can significantly improve the toughness of the material, so that it is not easy to break when subjected to impact or deformation, thus improving the performance of the BC composite membranes. In addition, the hot-press process resulted in denser interior of the nanomaterials, and fewer voids allowing for the formation of more hydrogen bonds, which enhanced the interactions between the fiber molecules and thus improved the mechanical properties. 31
Thermal properties
The thermal stability of pure BC, BC/AmimCl0.5, and BC/AmimCl2.5 membranes was assessed, and the TGA and derivative TGA curves were shown in Figure 5. All samples exhibited a slight weight loss below 150°C due to the evaporation of water entrapped in membranes even after drying. In addition, the weight loss increased with increase of the AmimCl concentration. This may be due to AmimCl breaking hydrogen bonds and making cellulose structures loose.
37
The major weight loss occurred at 200-400°C, which was the thermal degradation stage of BC, including the exothermic degradation of cellulose and decomposition of glucose units.43,44 In addition, pure BC showed an initial degradation temperature (Ti) of 300°C, higher than Ti of 240°C for BC/AmimCl0.5 and Ti of 220°C for BC/AmimCl2.5. The Ti was markedly decreased after being treated with AmimCl, with higher AmimCl percentage corresponding to lower Ti. The AmimCl would partially dissolve cellulose at high temperature, thus promoting the rapid degradation of BC.30,45 After thermal decomposition, the weight loss was 82.0% for pure BC, 65.8% for BC/AmimCl0.5, and 69.4% for BC/AmimCl2.5. The interpenetration of AmimCl between BC fibers could reduce the weight loss when compared pure BC. The introduction of chlorine by AmimCl could prevent thermal oxidation of cellulose, thereby enhancing the thermal stability of composite membranes.
31
(a) TGA and (b) derivative TGA curves of pure BC, BC/AmimCl0.5 and BC/AmimCl2.5 composite membranes.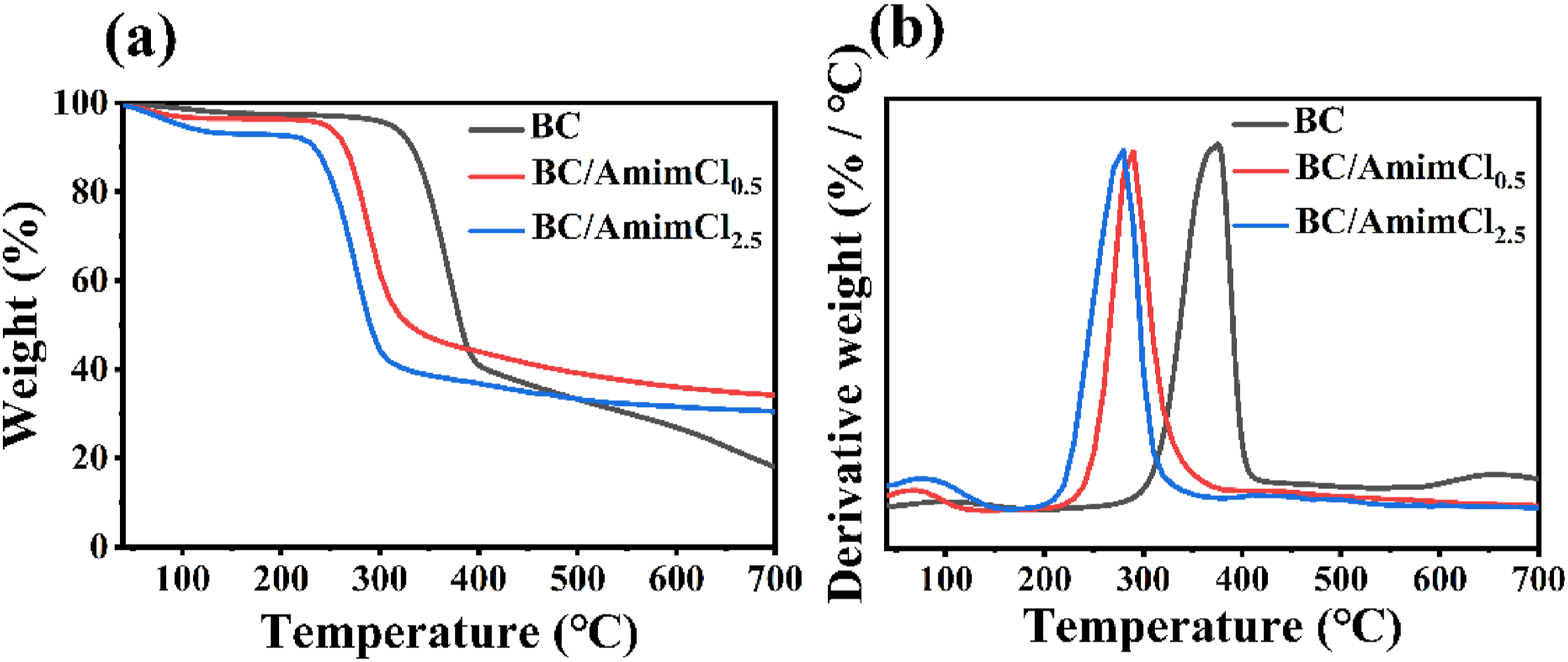
Optical properties
Transparency is an important parameter of dried BC membrane in application. Therefore, the light transmittance analysis of samples was conducted. Figure 6 showed the UV-vis transmittance of BC, BC/mimCl0.5 and BC/AmimCl2.5 membranes. It was found that the transmittance of the BC membrane was higher than that of BC/AmimCl0.5 composite membrane and lower than that of BC/AmimCl2.5 composite membrane in the range of 200-700 nm. The transmittance values of BC, BC/AmimCl0.5, and BC/AmimCl2.5 at 700 nm were 33.5%, 25.7%, and 35.0%, respectively. The results showed that the higher concentration of AmimCl significantly improved the transparency of the composite membranes at wavelengths below 200-700 nm, while the penetration of AmimCl negatively affected the transparency of the composite membrane at wavelengths greater than 700 nm to a certain extent. The higher concentration of AmimCl could destroy the intra and intermolecular hydrogen bonding of the fiber molecules, and make the pores between the fibers of the composite membrane larger than those of the BC membrane, leading to a reduction of light scattering from the composite membrane.30,31 Therefore, the higher penetration of AmimCl could increase the optical transparency of the composite membrane to a certain extent. At low concentration of AmimCl, the attached ionic liquid may be not enough to destroy the structure of the BC membrane, but will lead to the enhancement of light scattering and affect the transparency of the composite membrane. Transparency analysis of BC/AmimCl composite membranes.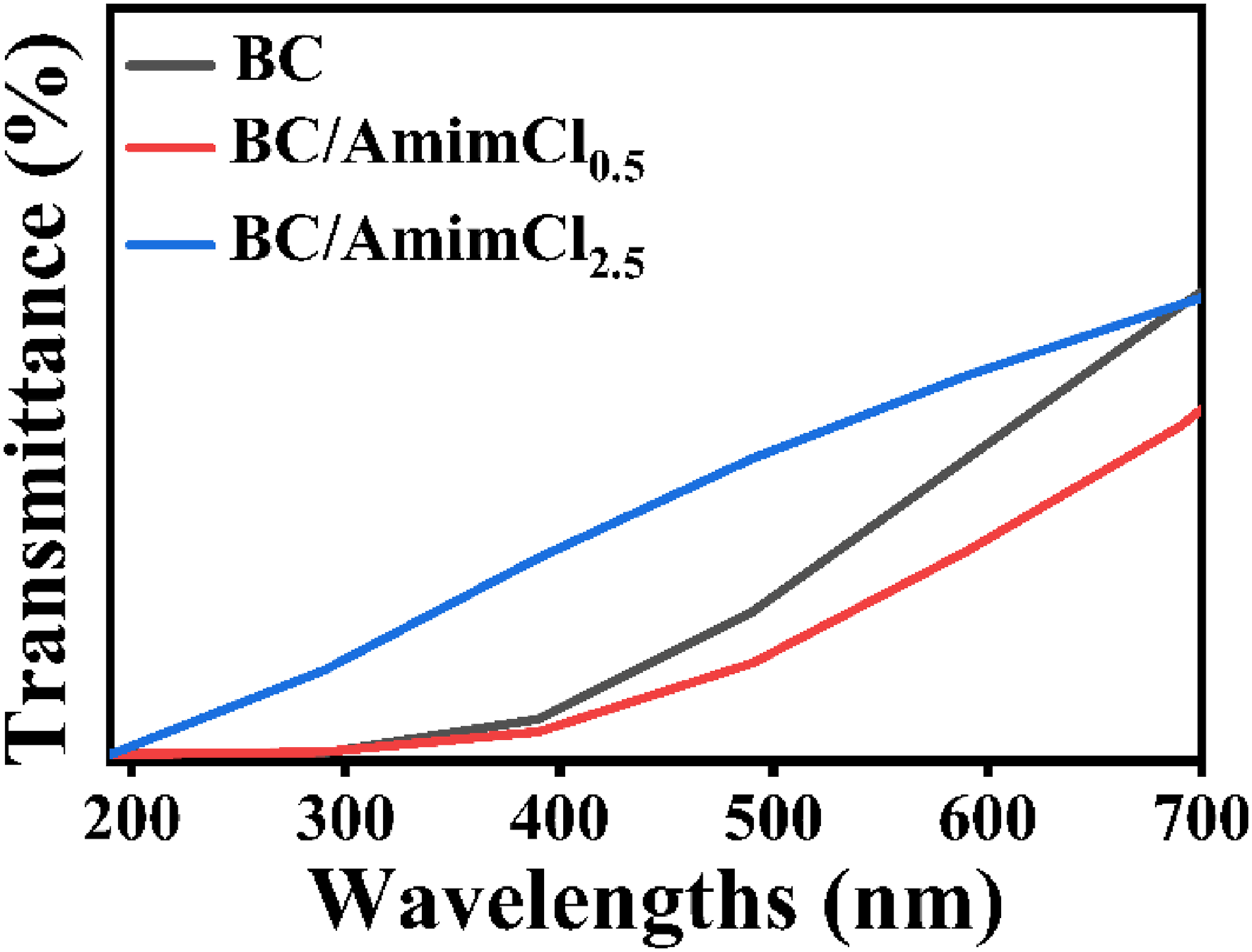
Conclusion
In this study, BC/AmimCl composite membranes were prepared by vacuum filtration and hot-pressing treatment. The effects of composite membranes were characterized through various analyses. The penetration of AmimCl disrupted hydrogen bonding among BC fibers, increased the amorphous phase structure of the membrane, lowered the thermal onset degradation temperature, decreased the final heat loss, and improved transparency. With the increase of AmimCl concentration, TH and EAB were significantly improved, while YM and TS showed a decreasing trend. This is because the strong plasticizing effect of AmimCl on BC, which softens the rigid BC membrane and makes it pliable. Meanwhile, AmimCl can break the hydrogen bonds between the nanofibers, making the BC fibers easy to migrate and slide. Further, hot pressing process will also make the nanofibers more densely arranged. In summary, this paper provides a certain reference value for the improvement of the mechanical properties of BC, but there are still some deficiencies, which should be continued to explore in the subsequent experiments.
Supplemental Material
Supplemental Material - Improved mechanical and thermal properties of bacterial cellulose with the introduction of ionic liquid
Supplemental Material for Improved mechanical and thermal properties of bacterial cellulose with the introduction of ionic liquid by Zhigang Chen, Yunya Wang, Xinrong Li, Xiaozhi Liu, Yanyan Xie and Cheng Zhong in Journal of Thermoplastic Composite Materials
Footnotes
Acknowledgements
The authors thank all students for their research contributions and exceptional enthusiasm in this research direction.
Author contributions
Conceptualization: Yanyan Xie, Cheng Zhong; Methodology: Xinrong Li; Formal analysis and investigation: Yunya Wang, Xiaozhi Liu; Writing-original draft preparation: Zhigang Chen, Xinrong Li; Writing-review and editing: Yanyan Xie, Cheng Zhong; Funding acquisition: Cheng Zhong; Resources: Cheng Zhong; Supervision: Yanyan Xie.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by National Natural Science Foundation of China (grant No. 22278322).
Ethical statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
