Abstract
This paper investigates the impact of deuterated ionic liquids (ILs) on the characterization of mechanical and thermal properties of polymeric films. The polymeric films were synthesized using two types of ionic liquids: 1-N-butyl-3-methylimidazolium chloride ([BMIM]Cl) and 1-methyl-D3-3-butyl-D9-imidazolium chloride ([BMIM-d12]Cl). Six samples were prepared, comprising three concentrations (1 wt%, 3.5 wt%, and 4.5 wt%) of the two ionic liquids. The mechanical properties, stiffness, and hardness, were evaluated using a universal tensile testing machine and a nanoindenter. The thermal properties were assessed using a thermogravimetric analyzer (TGA). The results demonstrated a significant enhancement in the mechanical performance of films formed with deuterated ionic liquids, with stiffness and hardness increasing up to 90 times compared to films synthesized with non-deuterated ionic liquids. Films containing [BMIM-d12]Cl demonstrated a toughness value 7000 times higher than the films without deuterated IL. The results indicate that deuterated ILs produce more thermally stable, stiffer, and tougher films compared to similar compositions of non-deuterated ILs. This improvement highlights the influence of deuterium substitution in ionic liquid structures, likely due to altered intermolecular interactions and increased film cross-linking density.
Introduction
The use of ambient-temperature ionic liquids (ILs) as solvents in the preparation of polymers represents a promising yet relatively underexplored area of materials science, especially when compared to their well-documented roles in other domains of synthetic chemistry. Their unique chemical and physical properties, including negligible vapor pressure, non-flammability, high thermal and electrochemical stability, and tunable viscosity, have positioned them as environmentally friendly alternatives to traditional volatile organic solvents. 1 Ionic liquids are salts in liquid form, typically composed of a bulky organic cation and an inorganic or organic anion. 2 The growing interest in ILs has incited research into their diverse applications, such as electrolytes in batteries, catalytic media, and separation agents.3–7 However, their use as solvents for polymerization reactions remains an emerging field for further exploration.
The intrinsic properties of ILs, such as their high ionic conductivity and ability to dissolve in a wide range of compounds, make them particularly suited for synthesizing conductive polymers and ion-conducting electrolytes. 8 These attributes are crucial in advancing fields such as electronics, energy storage, and bioengineering, where polymeric materials with specific conductivity and mechanical properties are increasingly in demand. Moreover, the ability of ILs to facilitate reactions under mild conditions without requiring high temperatures or pressures aligns with the principles of green chemistry, making them attractive for sustainable industrial processes.9,10
Among the myriad of organic transformations enabled by ILs, carbon-carbon (C-C) bond formation stands out as a cornerstone of modern polymer science. Addition polymerization, a subset of C-C bond formation reactions, is of particular industrial importance. It is widely employed in the production of high-performance polymers used in coatings, adhesives, plastics, and other applications. The incorporation of nanofillers into these polymer matrices further enhances their mechanical, thermal, and functional properties11–17 paving the way for advanced composite materials tailored for specific applications. Despite these advantages, the potential of ILs in polymer science has been overshadowed by their extensive use in catalytic transformations, such as hydrogenation, oxidation, epoxidation, and hydroformylation. 18
Noda and Watanabe 19 demonstrated the feasibility of free radical polymerization of vinyl monomers in ILs to produce conductive polymer electrolyte films. This pioneering study highlighted the advantages of ILs as polymerization media, including their ability to produce transparent, mechanically robust, and highly conductive films. Their work provided a framework for further exploration of ILs in polymer synthesis and set the stage for a deeper investigation into their potential to enhance material properties.
In this study, we seek to expand on the understanding of ILs by exploring the impact of deuterated ionic liquids on the mechanical and thermal properties of polyvinyl alcohol (PVA) polymeric-based films. Deuterated ILs, which contain isotopes of hydrogen with increased neutron count, offer unique physicochemical characteristics that may influence polymer behavior.
20
The isotopic substitution is expected to alter intermolecular interactions within the polymer matrix, potentially leading to enhanced mechanical strength, thermal stability, and durability.
21
By synthesizing and testing six samples of PVA films prepared with varying concentrations of two types of ILs, 1-N-butyl-3-methylimidazolium chloride ([BMIM]Cl) and 1-methyl-D3-3-butyl-D9-imidazolium chloride ([BMIM-d12]Cl), as shown in Figure 1. More specifically, three samples of [BMIM]Cl and three samples of [BMIM-d12]Cl of different weight percentage concentrations (1 wt%, 3.5 wt%, and 4.5 wt%) of both types. This research aims to shed light on the role of IL composition and isotopic effects in modulating polymer properties. 1,3-dialkyl imidazolium cation-based ionic liquids were synthesized by deprotonation of imidazole with potassium Chemical structure of (a)1-N-butyl-3-methylimidazolium chloride ([BMIM]Cl) and (b)1-methyl-D3-3-butyl-D9-imadazolium chloride ILs ([BMIM-d12]Cl).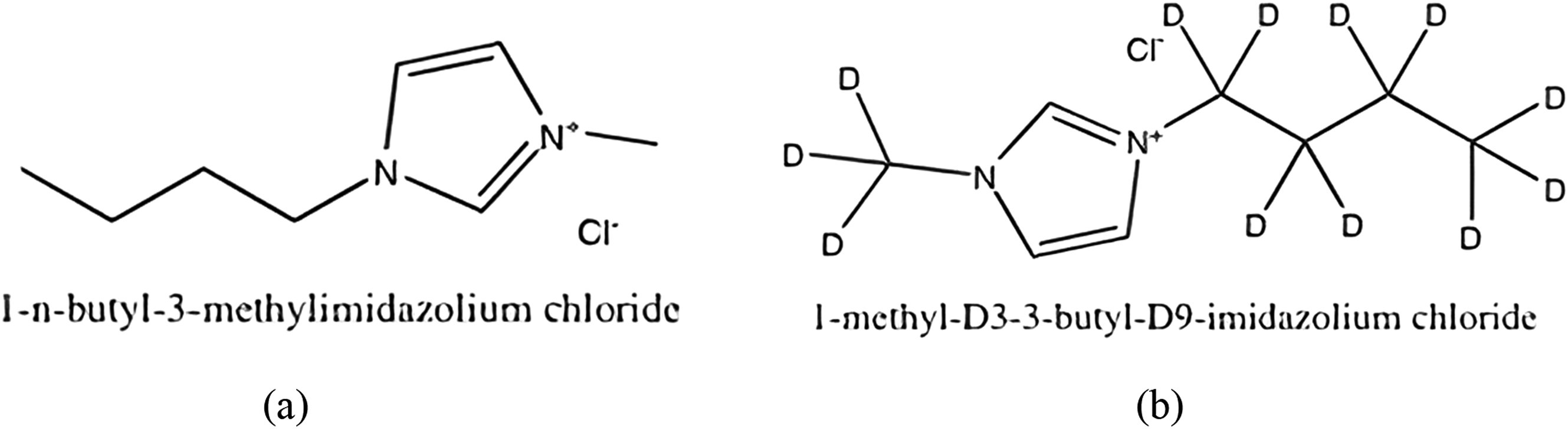
To quantify and assess the mechanical properties, such as stiffness, hardness, and peak stress, of the IL-polymeric films, comprehensive evaluations were conducted using characterization techniques, including universal tensile testing and nanoindentation. The results are expected to reveal critical trends in how deuterated ILs influence polymer formation and stability, contributing to the broader goal of designing next-generation materials for high-performance applications. Beyond mechanical testing, thermal analysis via thermogravimetric analysis (TGA), is utilized to examine the thermal stability and crystallization behavior of the films, offering a holistic view of their material properties.
The significance of this study lies in its potential to bridge the gap between fundamental research on ILs and their practical application in polymer science. By clarifying the effects of deuterated ILs on polymer behavior, this research not only enhances the understanding of IL-polymer interactions but also opens new paths for the development of functionalized polymers for specific industrial needs. Furthermore, the results could enlighten the design of sustainable and efficient polymerization processes, aligning with global efforts to minimize environmental impact while advancing material innovation.
Materials and preparation methods
The two types of ILs used, 1-N-butyl-3-methylimidazolium chloride ([BMIM]Cl) and 1-methyl-D3-3-butyl-D9-imidazolium chloride ([BMIM-d12]Cl), were synthesized in-house to ensure the purity and consistency required for precise material characterization, according to.
22
Each IL was prepared of three different weight-percentage concentrations, 1 wt%, 3.5 wt%, and 4.5 wt%. This resulted in a total of six distinct formulations for comprehensive testing. The polymeric films were fabricated by mixing 20g of polyvinyl alcohol (PVA), a widely used water-soluble polymer known for its excellent film-forming and mechanical properties, with the ILs at the specified concentrations in 8.8 mL of ethanol. The selection of ethanol as the solvent was based on its compatibility with both PVA and the ILs, as well as its role in promoting uniform dispersion of ILs within the polymer matrix. The mixture was thoroughly stirred to ensure homogeneity before being cast into films as illustrated in Figure 2. Preparation method for the polymeric films with ILs.
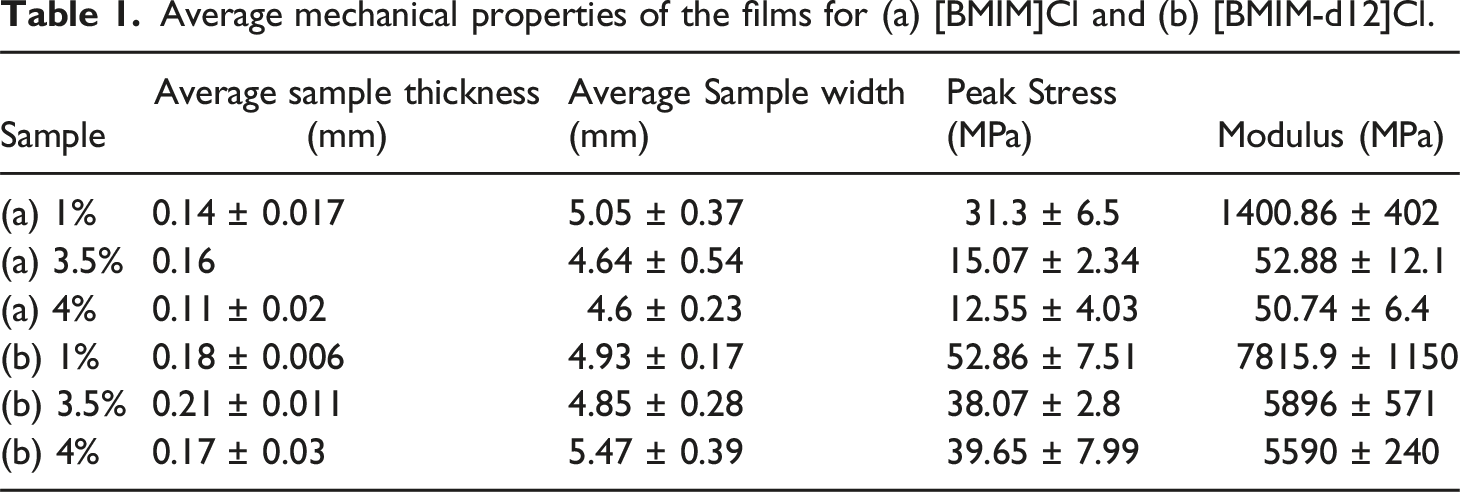
Average mechanical properties of the films for (a) [BMIM]Cl and (b) [BMIM-d12]Cl.
Results and Discussion
Mechanical properties
Figure 3 illustrates the average stress-strain diagrams for all the samples, where (a) represents films containing non-deuterated IL ([BMIM]Cl), and (b) corresponds to those with deuterated IL ([BMIM-d12]Cl). Table 1 provides a quantitative summary of the mechanical properties, including thickness, width, peak stress, and modulus for each composition. Average representations of the stress-strain diagrams for all of the samples. (a) denotes the non-deuterated IL polymeric films of 1-N-butyl-3-methylimidazolium chloride IL ([BMIM]Cl), and (b) denotes deuterated IL polymeric films of 1-methyl-D3-3-butyl-D9-imadazolium chloride IL ([BMIM-d12]Cl).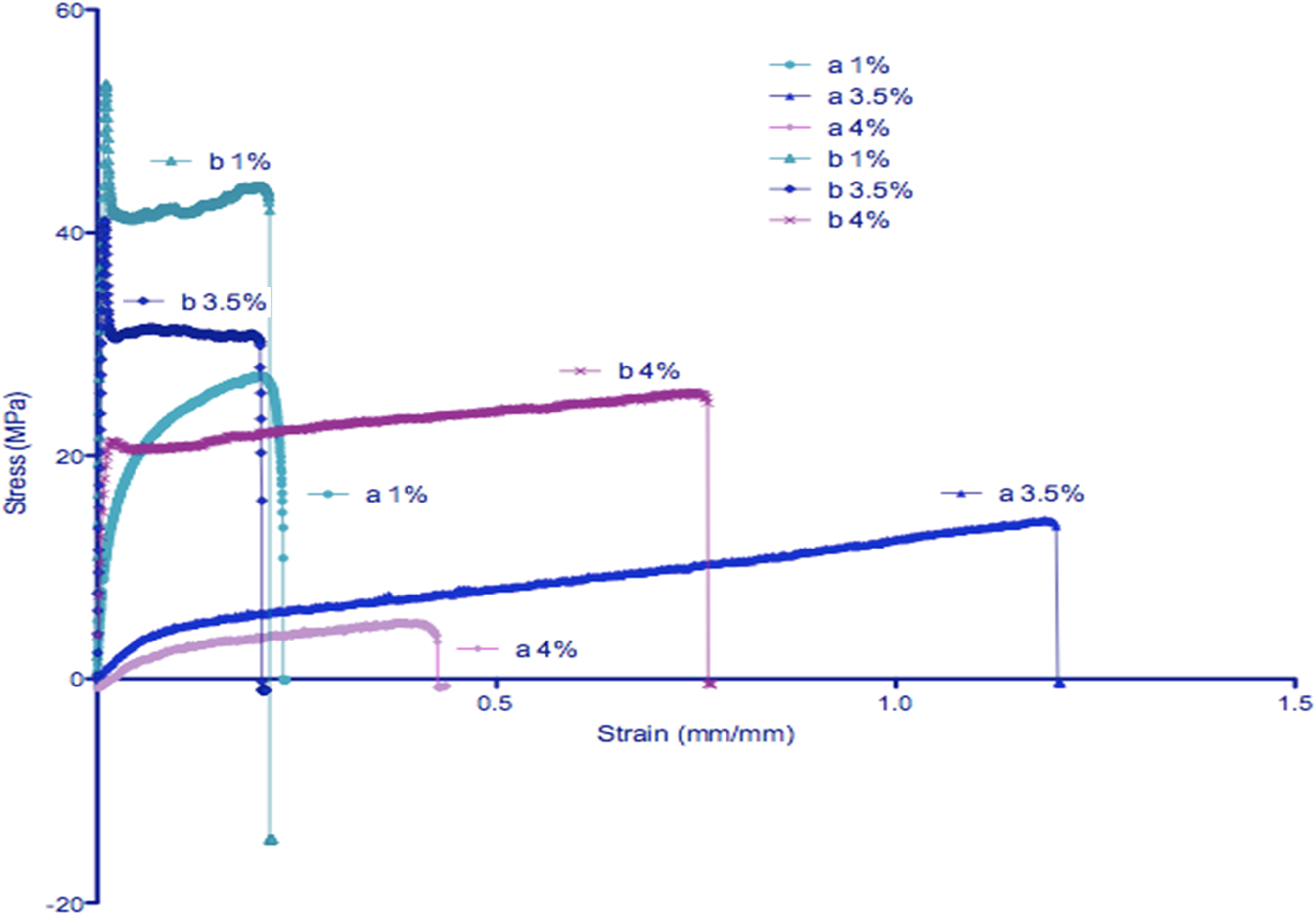
Figure 3 revealed significant differences in the mechanical properties of the films depending on the type of ionic liquid (IL) incorporated. Films with deuterated IL ([BMIM-d12]Cl) exhibited superior mechanical performance compared to those with non-deuterated IL ([BMIM]Cl). Specifically, the modulus of elasticity increased by factors of ∼5.5, ∼111, and ∼111 times for samples containing 1 wt%, 3.5 wt%, and 4 wt% deuterated IL, respectively, relative to their non-deuterated counterparts. Similarly, the peak stress values were enhanced by factors of 1.7, 2.5, and 3.2 times across the same compositions. These substantial improvements suggest that the isotopic substitution within the IL framework contributes to a stiffer and stronger polymer matrix, likely through modified intermolecular interactions and enhanced hydrogen bonding within the polymer-IL composite.
Nanoindentation tests were conducted using the Nano Test 600 (Micro Materials, Inc., Wrexham, UK) to further investigate the films’ mechanical properties at the micro- and nano-scale. This system, which utilizes a pendulum-based depth-sensing mechanism with the sample mounted vertically and the load applied electromagnetically, enables the precise measurement of hardness and modulus. Displacement of the indenter probe was recorded with sub-nanometer resolution, ensuring accurate data collection. Tests were performed on the 3.5 wt% samples for both IL types under maximum loads of 0.25, 0.5, and 1 mN. The loading rate was set to 0.12 mN·s−1 and each test was repeated at least six times. The Berkovich diamond indenter (three-sided pyramidal tip) was employed to achieve consistent results, with sufficient spacing between indentations to eliminate overlapping effects. The instrument’s software corrected all data for thermal drift and instrument compliance.
Mechanical properties obtained for the two 3.5 wt% IL films, where (a) [BMIM]Cl and (b) [BMIM-d12]Cl.
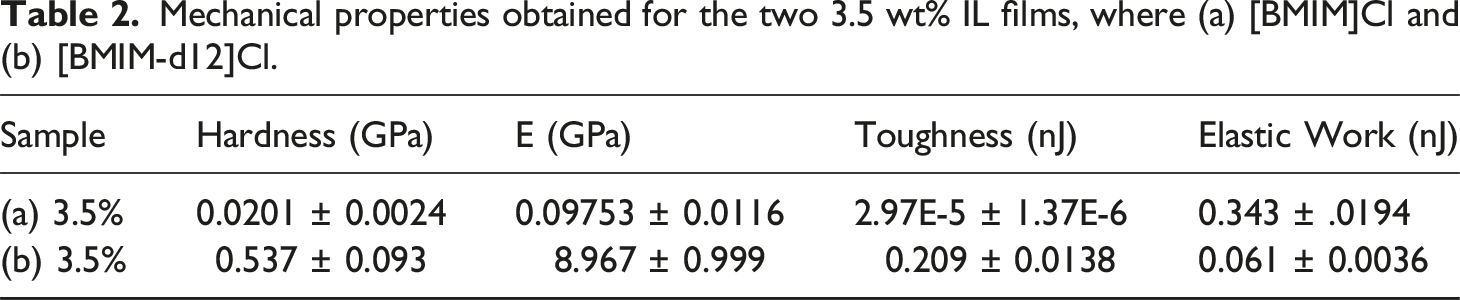
The toughness was quantified through the area under the load-displacement curve. Films containing [BMIM-d12]Cl demonstrated a toughness of 0.209 nJ, a value 7000 times higher than the 2.97 × 10−5 nJ for films without deuterated IL ([BMIM]Cl). This enhancement is indicative of the alteration in the energy-absorbing mechanisms within the films. The deuterated IL significantly enhances the films’ ability to distribute and withstand stress, which can be attributed to its influence on the microstructural arrangement of the polymer matrix. The isotopic substitution may lead to improved load transfer between the IL and polymer chains, minimizing localized deformation and promoting energy dissipation across the entire matrix. As for the elastic work, the results show that non-deuterated IL was higher (0.343 nJ) when compared to deuterated IL (0.061 nJ), about 5.6 times less. This suggests that while the films with [BMIM]Cl are more compliant and store energy elastically, the deuterated films exhibit a predominantly inelastic response under load, contributing to their higher stiffness and toughness. This reduction in elastic work aligns with the premise that deuterium substitution alters the flexibility of the polymer chains, favoring rigidity and strength over elastic recovery. The trade-off between elasticity and stiffness is a common phenomenon in materials engineering and highlights the tunable nature of IL-polymer composites for specific applications.27,28 Therefore, both the bulk mechanical properties and those obtained using a nanoindenter reveal that the stiffness and hardness of the polymeric films using deuterium IL increase more than that which contains no deuterium. The peak stress for the deuterium films is on average doubles its counterpart, this complies with the results obtained in the MTS testing. This enhancement in hardness and stiffness suggests that these materials could be ideal candidates for applications requiring robust, durable films, such as in flexible electronics, advanced coatings, and high-performance membranes in their practical applications in next-generation polymer-based technologies. Furthermore, the ability to tune properties through isotopic substitution provides a powerful tool for customizing materials to meet specific performance criteria.
Thermal analysis
Thermal analysis was conducted using a thermal gravimetric analyzer Q50 (TA Instruments, Waters LLC) to evaluate the thermal stability and phase transition behavior of the polymeric films containing non-deuterated ([BMIM]Cl) and deuterated ([BMIM-d12]Cl) ionic liquids. The modulus of elasticity of film-forming polymer is strongly temperature dependent.
29
The modulus generally decreases as temperature increases. Below the glass-transition temperature (Tg), polymer systems exhibit a glassy solid behavior, while above the Tg they progress through a leathery rubbery state at higher temperatures they exhibit a liquid-like flow behavior. In this study, we tested pieces of the polymeric films of the same thickness on the two ILs of the 3.5 wt% samples. Figure 4 reveals the thermogravimetric analysis (TGA) differences between the two types of films, depicting the changes in melting and crystallization temperatures and the thermal decomposition profiles of the films, thus explaining the impact of deuterium substitution on the thermal behavior of the IL-polymer composites. TGA for films formed by (a)1-N-butyl-3-methylimidazolium chloride and (b)1-methyl-D3-3-butyl-D9-imadazolium chloride ILs.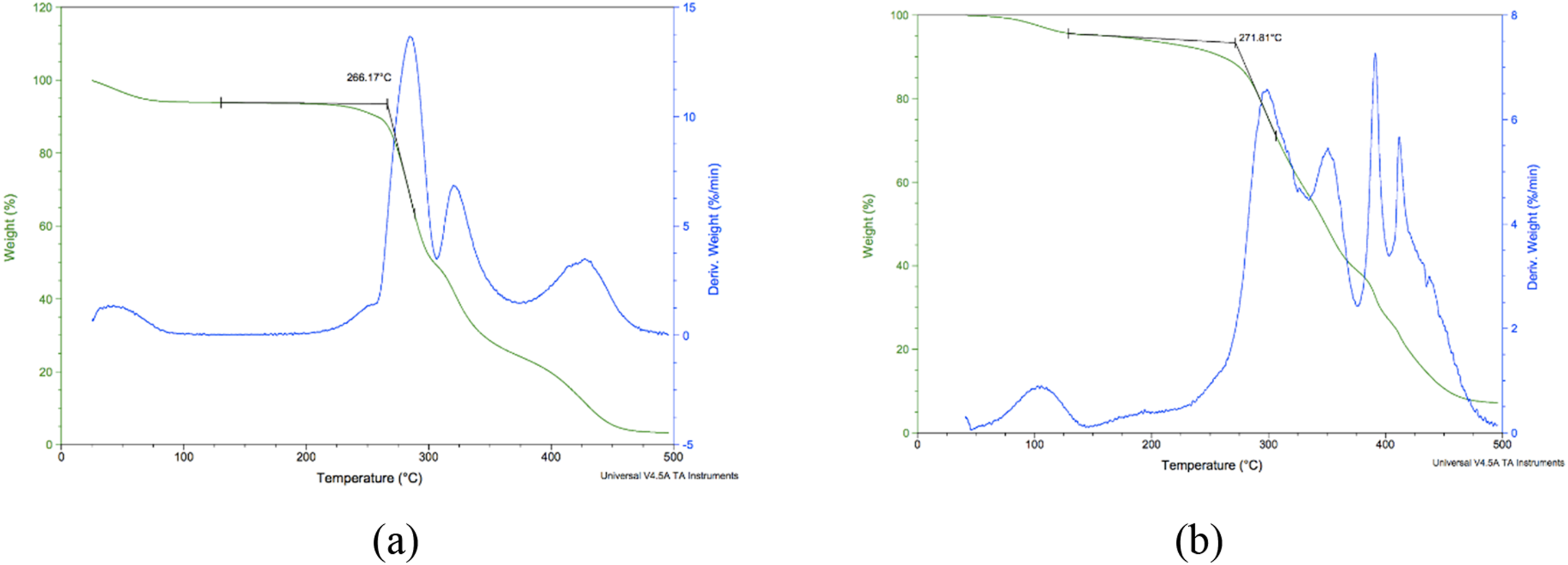
The TGA analysis results show a shift in the first melting peak between films containing deuterated and non-deuterated ILs. The non-deuterated films ([BMIM]Cl) exhibited a melting temperature of 266°C, while those with deuterated IL [BMIM-d12]Cl showed an elevated melting temperature of 271°C. This shift can be attributed to the isotopic substitution in the IL. Deuterium, being heavier than hydrogen, alters the vibrational frequencies of the molecular bonds in the IL, thus resulting in stronger intermolecular forces, such as hydrogen bonding and van der Waals interactions, within the polymer matrix. Consequently, the polymer-IL composite requires more thermal energy to disrupt these interactions, leading to an increased melting temperature. 30
Additionally, the broader melting peaks observed in films with deuterated ILs suggest a more heterogeneous crystalline structure. 31 This could be a result of the altered packing density and distribution of IL molecules within the polymer matrix, influenced by the heavier isotopic substitution.
The crystallization behavior also showed notable differences between the two film types. The [BMIM-d12]Cl films exhibited a crystallization temperature slightly higher than the observed films without deuterated IL [BMIM]Cl. This increase indicates enhanced nucleation and growth of crystalline domains in the presence of the deuterated IL. Deuterium substitution is known to impact the molecular mobility of ILs, reducing their dynamic flexibility. This reduction may facilitate a more orderly alignment of polymer chains during the crystallization process, resulting in a higher crystallization temperature. Such behavior highlights the role of IL composition in modifying the thermal properties of polymer composites. The higher stability and multiple degradation stages observed in deuterated films suggest that these materials undergo a more gradual breakdown, potentially extending their functional lifespan in practical applications. This characteristic is ideal for materials subjected to cyclical thermal loads or prolonged exposure to elevated temperatures.
While both film types displayed excellent thermal stability, the onset of thermal degradation was slightly delayed in films containing deuterated ILs. As indicated earlier, this improved stability can be linked to the stronger intermolecular forces introduced by the deuterium substitution. The shift in decomposition onset suggests that the deuterated ILs contribute to a more robust polymer matrix, capable of withstanding higher thermal stresses before breaking down. Such enhancements in thermal stability are crucial for applications in high-temperature environments, such as electronics, aerospace, and automotive industries.
Conclusions
This study investigated the impact of altering PVA films with deuterated ionic liquids, specifically the mechanical and thermal properties of polymeric films compared to their non-deuterated counterparts. The polymeric films were synthesized using two types of ionic liquids: 1-N-butyl-3-methylimidazolium chloride ([BMIM]Cl) and 1-methyl-D3-3-butyl-D9-imidazolium chloride ([BMIM-d12]Cl) with three weight concentrations (1 wt%, 3.5 wt%, and 4.5 wt%) of the two ionic liquids. Films with deuterated ILs exhibited a significant increase in stiffness and hardness, along with improved thermal stability. These enhancements are attributed to stronger intermolecular interactions facilitated by isotopic substitution. The results revealed a deeper understanding of how isotopic modification in ionic liquids can tailor the mechanical properties of polymeric materials, paving the way for advanced applications in areas requiring enhanced material toughness and durability. The potential of deuterated ILs to develop robust and thermally stable polymeric materials for advanced applications in fields such as aerospace, electronics, and coatings.
Footnotes
Author contributions
Mohammad Y. Al-Haik – Conceptualization, methodology, investigation, validation, resources, writing—original draft preparation, writing—review and editing, funding acquisition: No Funding Available.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
