Abstract
Polyaniline is a remarkable conjugated polymer having valuable structural and physical characteristics. Nevertheless, polyaniline has poor solubility and structural durability which had limited its practical utilization. Consequently, polyaniline has been reinforced with carbonaceous and inorganic nanoparticles to enhance its processability and physical properties, such as electrical/thermal conductivity and thermal/mechanical stability. Accordingly, polyaniline has been applied as a valuable matrix material to form nanocomposites with graphene which is a one atom thick nanosheet of hexagonally arranged carbon atoms. Subsequently, graphene as well as modified graphene forms have been utilized as important nanoadditives for the polyaniline matrix. This state-of-the-art review article has been designed to investigate the fabrication, characteristics, and applications of the polyaniline and graphene/modified graphene based nanomaterials. Potential enhancements in the physical characteristics of the pristine polyaniline upon graphene reinforcement seemed to be reliant upon the matrix-nanofiller interactions, interface formation, and synergetic effects between polyaniline-graphene. Subsequently, practical aspects of the polyaniline/graphene nanocomposites have been discussed regarding their applications in the fields of solar cells, supercapacitors, and sensors. According to the literature analysis, dye sensitized solar cell based on polyaniline/graphene and polyaniline/graphene oxide exhibited superior power conversion efficiencies ∼2.0-7.6 % and open circuit voltage ∼500-800 mV. Supercapacitor electrodes based on polyaniline/graphene oxide and polyaniline/reduced graphene oxide showed significantly high values of specific capacitance and capacitance retention of ∼1100-1900 Fg−1 and ∼80-98 %, respectively, during >5000-6000 cycles. Besides, the polyaniline/graphene oxide depicted superior sensitivity and response time for gas sensors (∼20 % and 50 s, respectively) and humidity sensors (>90 % and 4-7 s, respectively) and low detection limits for biosensors.
Introduction
Polyaniline is an important conjugated polymer having facile synthesis, processability, electrochemical, conducting, and stability properties. 1 Polyaniline has been reinforced with various nanofillers, like nanoparticles, nanofiber, nanospikes, or nanotubes to form high tech nanocomposite. For nanocomposite formation of conjugated polymers, carbon nanofillers, like carbon nanotube, graphene, graphene derivatives, carbon black, etc., have been frequently reported as effective nanoadditives in the literature.2,3 Consequently, various nanocomposites of pristine polyaniline and its blends have been investigated to develop the nanocomposites or hybrid materials. 4 Among carbon nanofillers, graphene and its derivatives have been used as valuable nanofillers for the polyaniline matrix. 5 Similarly, carbon nanotube has been applied as an important nanoadditive for polyaniline to enhance its technical features and applications. 6 Polyaniline has also been combined with carbon black to form the desirable nanomaterials. 7 Consequently, these nanocarbons have capability to develop physical or covalent interactions with the polyaniline matrix to enhance the overall physical property profiles of the nanomaterials. 8 The resulting high performance of the polyaniline/nanocarbons nanocomposite was attributed to the synergistic effects between the matrix and the nanoparticles. Besides, metal oxide, and inorganic nanoparticles have also been applied as useful nanofillers for the polyaniline matrix. 9
Graphene has been identified as one of the most effective nanofillers, for polymers, due to its unique nanostructure, high surface area, and distinct electrical, thermal, and mechanical properties.10,11 Including graphene in polyaniline showed remarkable enhancements in the structural, microstructural, electrochemical, electrical, optical, thermal, mechanical, and physical characteristics of the matrix. 12 In addition to graphene, various modified forms of graphene, like graphene oxide, reduced graphene oxide, and nanoparticle functional graphene/graphene oxide have been used as nanofillers for the polyaniline matrix. 13 Consequently, polyaniline and graphene or modified graphene derived nanomaterials depicted significant applications for photovoltaics, supercapacitors, sensors, and related technological fields.14–16 In due course, the literature surveys so far had reported a number of high performance polyaniline nanomaterials reinforced with graphene oxide/reduced graphene oxide17–19 and sulfur, silver, or manganese dioxide nanoparticle functional graphene20–22 for technical applications. The resulting superior property-performance aspects of these nanocomposites have been attribute to the matrix-nanofiller interactions, compatiblization, and interface formation effects between the polyaniline matrix and graphene/modified graphene nanofillers.23,24
Succinctly, this innovative overview presents the nanocomposite formation potential of polyaniline with graphene and allied nanofillers. In the literature, pristine as well as modified graphene has been used to form the high performance polyaniline nanocomposites. Consequently, these nanomaterials have been studied for their structural, nanostructural, and physical feature investigations. Moreover, the technological applications of polyaniline and graphene based nanocomposites have been explored for the fields of energy and electronics. To the best of the knowledge, this is a revolutionary overview on the polyaniline/graphene nanocomposites conversing the fundamentals, characteristics, and essential technical applications.
Novelty of this review article lies in the systematically outlined topics covering design and features of polyaniline/graphene and polyaniline/graphene derivative nanomaterials as well as focused technical areas of these hybrids. Moreover, the inclusion of specific field literature, valuable discussions, and considerations of future and challenging aspects of polyaniline and graphene derived nanomaterials render this manuscript entirely novel, relative to the reported literature. Hereafter, purpose or objective of this ground-breaking review is to document a deep-seated and up-to-date article on polyaniline/graphene or modified graphene nanocomposite enfolding crucial aspects from fabrication—to—progressive applications. Need of developing such revolutionary state-of-the-art article was analysed due to the lack of comprehensive recent review articles in the field of polyaniline and graphene/modified filled hybrids. Consequently, this all-inclusive overview is planned to throw sufficient light upon contemporary and projected future expansions of polyaniline/graphene nanocomposites which can be helpful for the field scientists/researchers struggling to explore energy and electronics (solar cells, supercapacitors, sensors) related technological possibilities using these high end nanocomposites. Despite the ongoing and anticipated future technical advancements of polyaniline/graphene nanocomposites, there are a number of existing knowledge gaps regarding large scale processing, precise commercial scale models, durability, long life functioning, eco-friendliness, sustainability, recyclability, and economy in this sector. Concisely, all these scenarios point towards an intense need of concentrated future research surveys on polyaniline/graphene nanocomposites to grow the primary lab. scale models—to—industrial prototypes.
Graphene
Graphene, as a unique carbon nanoallotrope, has brought about important revolutions in the nanotechnological fields.25,26 Graphene has one atom thick nanostructure with sp2 hybridized carbon atoms. 27 Since discovery, graphene has been studied for its structure, electron π orbital delocalization, and structural and physical properties. 28 Graphene has been synthesized by numerous bottom up or top down techniques. 29 Among common methods, mechanical exfoliation, chemical vapor deposition, and organic synthesis have been used for the fabrication of graphene. 30 Accordingly, high electron conductivity (200,000 cm2V-1s-1) and Young’s modulus (I TPa) have been reported as the prominent properties of graphene. 31 Due to van der Waals forces, a single layer graphene nanostructure has shown wrinkling tendencies. 32 Consequently, graphene owns numerous technologically beneficial applications in the fields of energy/electronics devices/structures, engineering, defense, textiles, biomedical, and related applied sectors.33–35 An important use of graphene has been observed for nanoreinforcement of the polymeric matrices to form nanocomposites. 36 Subsequently, the resulting polymer/graphene nanomaterials were studied for their structure, interface formation, matrix-nanofiller interactions, etc. For this purpose, polymers, like conducting polymers, rubbers, thermoplasts, and thermosets matrices have been categorized as important matrices for the graphene nanofillers. 37 Numerous facile mixing techniques have been used for synthesizing the polymer/graphene nanocomposites. 38 The resulting graphene based nanomaterials have been studied for wide ranging applications areas ranging from devices to bio medics. 39
Further revolutions in the field of graphene nanomaterials have been brought about through the development of modified forms such as graphene oxide, reduced graphene oxide, functionalized graphene, and so on. For example, graphene oxide has been established as an important graphene derivative with various surface functionalities, like hydroxyl, carboxylic acid, carbonyl, epoxide, ketonic, and several other groups on its surface. 40 This graphene derivative has been frequently prepared using facile and low cost techniques like Hummer’s method. 41 Further reduction of graphene oxide, through thermal, chemical, or electrochemical means, has uncovered another graphene derivative, that is, reduced graphene oxide. Accordingly, reduced graphene oxide revealed important surface properties due to the presence of surface defects and reduction of oxygen groups. Both the graphene oxide and reduced graphene oxide have been recognized for superior surface area, electrical conductivity, thermal conductivity, mechanical stability, and technical solicitations for electrocatalysis, electronics, sensors, and energy devices. 42
Similar to graphene, graphene oxide, reduced graphene oxide, and functional graphene forms have been applied as nonreinforcements to develop high tech polyaniline hybrids. 43 Owing to the presence of hydrophilic functional groups and aromatic ring structure, graphene oxide and reduced graphene oxide can develop better dispersions, interactions, and compatibilization towards the conjugated polymers like polyaniline, relative to pristine graphene. Consequently, graphene oxide and reduced graphene oxide have been observed competent to enhance the heat stability and mechanical robustness of polyaniline, relative to pristine graphene. 44 Furthermore, these graphene derivatives have been reported to develop well-interconnected electron conduction network throughout the polyaniline matrix, so resulting in high electrical conductivity, compared with the graphene filler nanocomposites. Consequently, polyaniline nanohybrids with modified graphene nanofillers depicted technical potential for aerospace, energy/electronic devices, biomedical and allied applied fields. 45
Polyaniline
Polyaniline is one the most widely used conducting polymers. It was discovered in the last century and was primarily known as aniline black. 46 Due to unique conjugated structure, polyaniline had tendency of efficient doping/dedoping. 47 Appropriate fabrication routes and doping techniques have been used to improve the electron conductivity and physical properties of the polyaniline. Consequently, research so far has proposed numerous methods for the formation of polyaniline, including the use of oxidizing agent with aniline monomer for the in situ synthesis of this polymer.48,49 Attempts have also been reported to investigate the mechanism of in situ polymerization of the aniline monomers. 50 During synthesis, polyaniline can be formed in various oxidation states including the emeraldine, leucoemeraldine, or pernigraniline. 51 Emeraldine has been reported as the most stable and highly conducting form of polyaniline. 52 Consequently, electrical conductivity of the polyaniline was seemed to be considerably enhanced through acidic doping processes. Hence, the electron or charge conductivity and reversibly redox behaviours of the polyaniline pointed towards several technological benefits for electronics, sensor, supercapacitor, antistatic coatings, and other technical fields. 48 Polyaniline has been converted to composite or nanocomposite forms to further enhance its practical aspects for applied industrial fields. 53
Polyaniline nanocomposites filled with graphene, graphene oxide or modified graphene
Addition of graphene in the polyaniline matrix caused valuable enhancements in the optical, electronic, and heat stability features of the resulting nanocomposites.54,55 Consequently, the polyaniline/graphene nanocomposites depicted promising attributes for electronics, energy, and related technological devices.
56
In addition to pristine graphene nanostructure, functional or modified graphene has been efficiently used to enhance the physical properties and performance of the polyaniline nanomaterials.
57
In this context, polyaniline has been amalgamated with the sulfonated graphene and used for the technical devices.57–59 The interlinked microstructures of polyaniline and functional graphene seemed to be accountable for efficient electron/charge transport in these nanocomposites.
60
Wang et. al.
61
investigated the electronic structure of polyaniline and polyaniline/graphene nanocomposite using a density functional simulation technique. Accordingly, Figure 1(a) shows a relaxed structure of polyaniline, whereas Figure 1(b) depicts a periodic simulation supercell for the hydrochloric acid doped polyaniline and graphene based nanomaterial. The lattice constants of the supercell were optimized as 2.466 Å. Figure 1(c) illustrates the minimum energy paths in the deprotonation or protonation reactions of the polyaniline. The affinity of polyaniline was observed towards the adsorption of the protonating dopants. Subsequently, the investigation verified that the polyaniline was physically linked to graphene, through van der Waals and charge-transfer interactions, to enhance the electron or charge conductivity of these nanomaterial. Moreover, the electronic excitation and charge transfer between the polymer and nanocarbon were found reliant upon the applied protonation and temperature changes. The structures of (a) PANI with quinoid rings; (b) PANI-G nanocomposite with periodic supercell in simulation; and (c) The minimum energy paths and barriers for the deprotonation reactions of HCl-PANI.
61
PANI = polyaniline; PANI-G = polyaniline-graphene; HCl-PANI = hydrochloric acid-polyaniline. Reproduced with permission from ACS.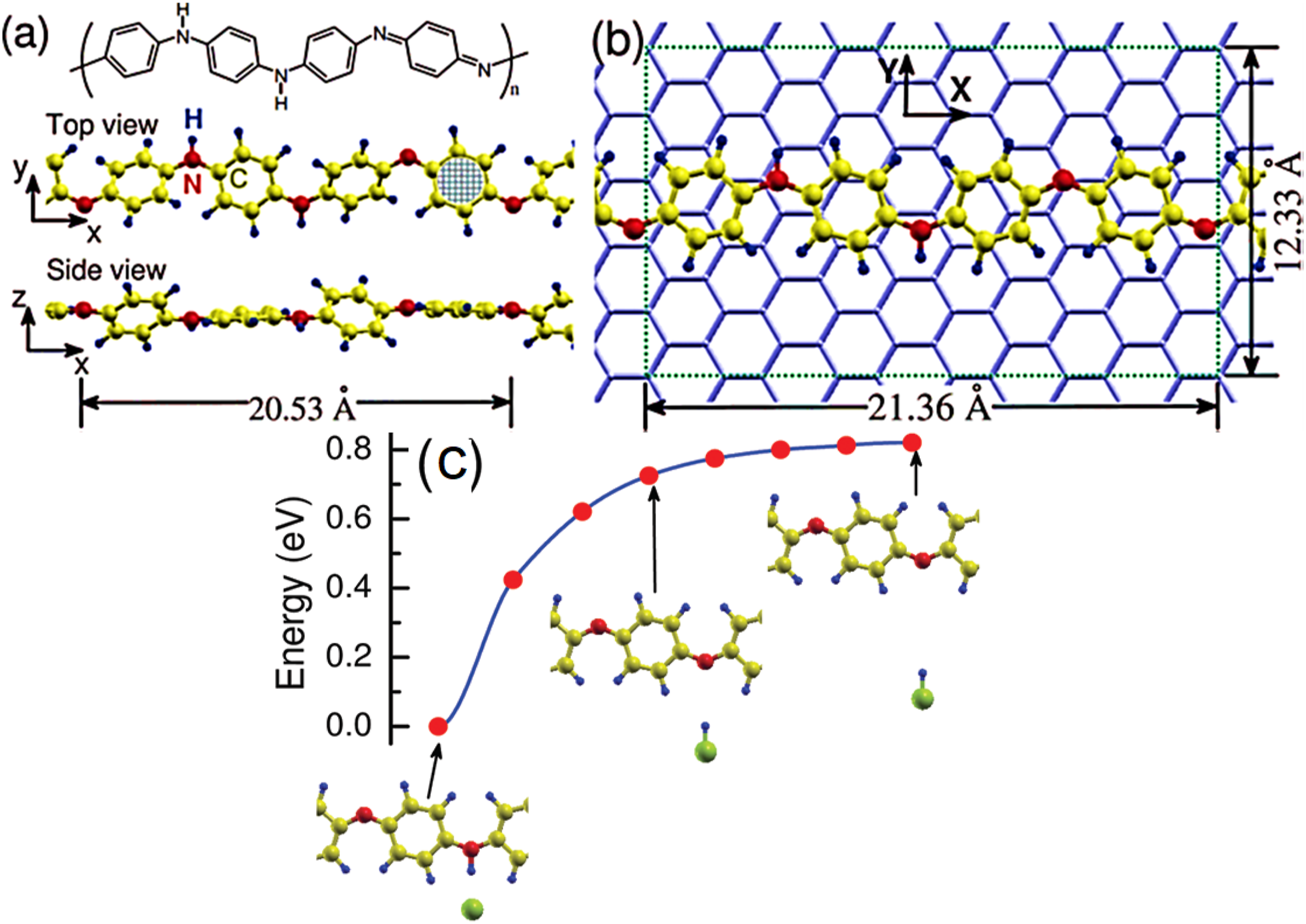
Kumar et. al.
62
formed the polyaniline grafted reduced graphene oxide nanocomposite. Figure 2(a) shows the three step fabrication of the polyaniline grafted reduced graphene oxide. Graphene oxide was formed using the Hummers method and then the graphene oxide was converted to acylated and amine functional graphene oxide using 4-aminophenol compound. Afterwards, the amine functional graphene oxide was finally covalently grafted to polyaniline. (a) Schematic for the preparation of PANI-g-rGO nanocomposite; (b) typical FESEM images of PANI-g-rGO surface; (c) HRTEM image of graphene oxide with inset image of selected-area electron di;raction (SAED) pattern; (d) rGO-NH2 with inset image at higher magnification; and (e) PANI-g-rGO.
62
PANI-g-rGO = polyaniline-graft-graphene oxide; FESEM = field emission scanning electron microscopy; rGO-NH2 = amine functional reduced graphene oxide; GO = graphene oxide; HRTEM = high resolution transmission electron microscopy. Reproduced with permission from ACS.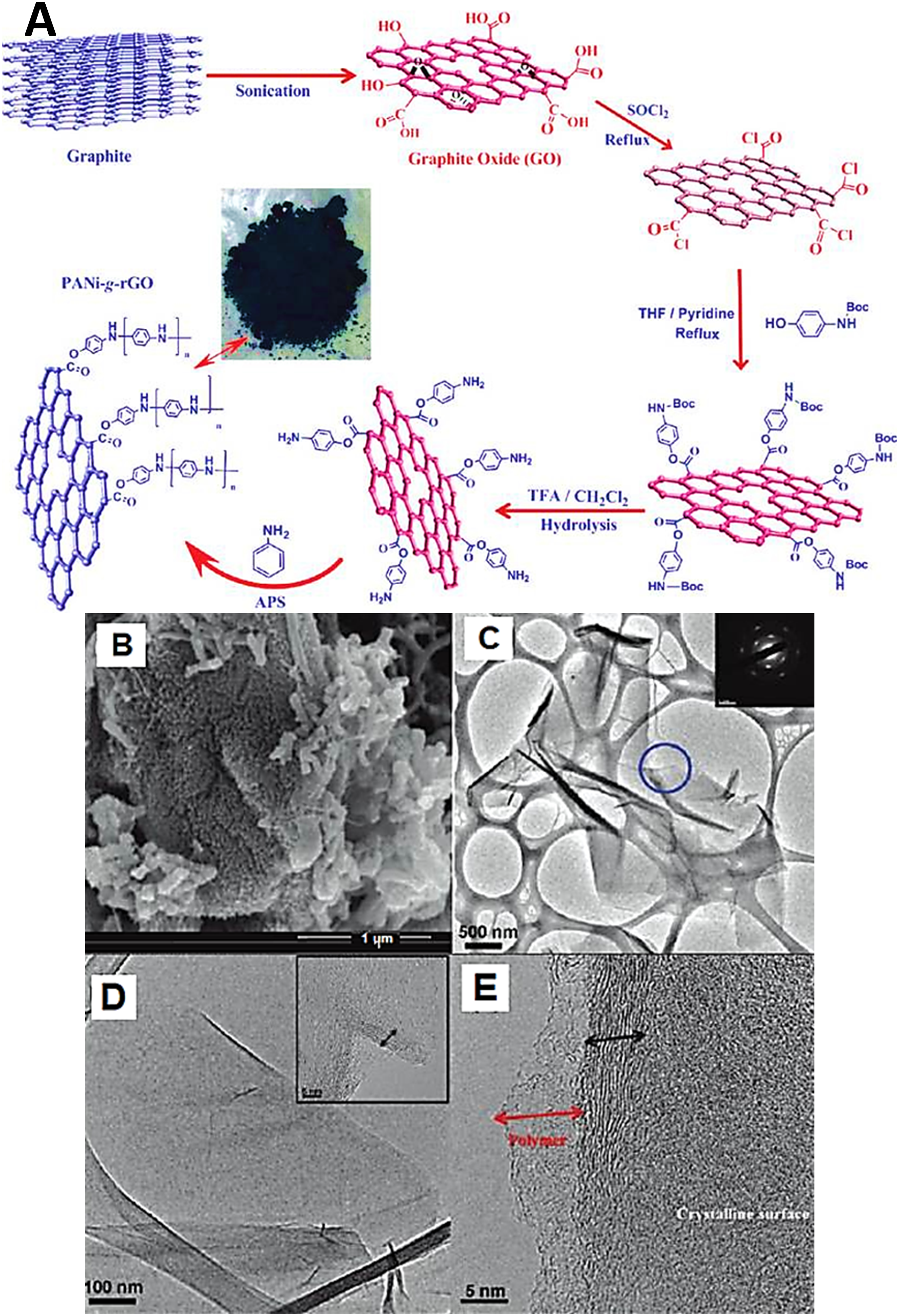
According to field emission scanning electron microscopy, the polyaniline grafted reduced graphene oxide nanocomposite had nanofibrillar microstructure (Figure 2(b)). Graphene flakes were found embedded in the polyaniline matrix due to the formation of interconnected nanostructure. Graphene oxide showed a monolayered transparent nanostructure according to transmission electron microscopy, as shown in Figure 2(c). The microstructures of the amine functional reduced graphene oxide and polyaniline grafted reduced graphene oxide were also studied (Figure 2(d) and (e)). According to the results, the amine functional reduced graphene oxide showed surface alterations, relative to neat graphene sample. In addition, grafting of polyaniline on reduced graphene oxide surface was noticed in the form of polymer layering or wrapping. Due to covalent grafting, homogeneous matrix layering was observed on the nanocarbon nanosheet surface. The electron or charge conductance studies of the as prepared nanocomposites depicted electrical conductivity and specific capacitance of 8.66 Scm−1 and 250 Fg−1, respectively. Fine durability, charge discharge, and working performance were also observed for these nanocomposites. Fan and researchers
63
fabricated the polyaniline and sulfonated graphene derived nanocomposites using an in situ polymerization technique. The doping of polyaniline on the sulfonated graphene surface resulted in the formation of a layered nanostructure.64,65 Herein, benzene sulfonic acid was used to convert the graphene oxide to the sulfonated graphene. Subsequently, the aniline monomer was in situ polymerized on the surface of modified graphene to obtain the nanocomposite. Consequently, structural, thermal, conductivity, and capacitance studies were performed to analyze the properties of these nanocomposites. According to thermogravimetric analysis, decomposition of the polyaniline/sulfonated graphene nanostructure occurred at high temperatures due to reasonable heat stability of the nanomaterials.
66
Due to the formation of fine conducting network, the nanocomposites revealed high capacitance of 478 Fg−1. Similarly, in situ method has been widely investigated in the literature for the synthesis of the polyaniline and graphene based layered or sandwiched nanostructures.67,68 Ameen and colleagues
69
formed the polyaniline and graphene based nanocomposite by an in situ method using ammonium persulfate. Here, polyaniline and graphene developed hydrogen bonding for the formation of a stable nanostructure.
70
Elahi and researchers
71
used in situ polymerization to synthesize the polyaniline and graphene derived nanocomposite. The electrical conductivity of the nanocomposite was found dependent upon the temperature changes. In addition, these nanomateriasl were studied for charge transportation properties. The electron conduction mechanism was also studied and found dependent upon the three dimensional hopping phenomenon.
72
Chauhan and co-workers
73
filled the graphene nanofiller in the polyaniline matrix to attain the nanocomposites. Inclusion of graphene was found to enhance the electroactivity of the polymer matrix.
74
The conduction and capacitance of polyaniline and reduced graphene based nanomaterials were also analysed.75,76 Xiang et. al.
77
developed the polyaniline and exfoliated graphene derived nanocomposites through an in situ polymerization technique. In this regard, the polymer and nanocarbon developed π-π electron delocalization and interaction to form a stable nanostructure. Figure 3(a) shows the formation and charge carrier transportation in the polyaniline and the polyaniline/graphene nanocomposite. Consequently, the polyaniline and graphene nanoplatelet film was stretched for better polymer chain alignment and nanosheet orientations for facilitated electron transfer through the system. Figure 3(b) shows that the deposition of polymer on the graphene surface decreased the electrical conductivity from 200 to 59 Scm−1, relative to neat graphene nanosheet. The decrease in the electron conductivity was observed due to electron hopping between the polymer chains. Pristine graphene nanoplatelet showed higher electron conductivity of 200 Scm−1, whereas neat polyaniline had a lower value of 1.5 Scm−1. (a) Polymerization process of polyaniline without graphene and charge transport mechanism through inter and intrachain hopping; and (b) electrical conduction in polyaniline/graphene nanocomposite (0.03 M and 0.05 M), neat graphene nanoplatelet paper, and neat polyaniline at room temperature.
77
PANI = polyaniline; GNP = graphene nanoplatelet. Reproduced with permission from Elsevier.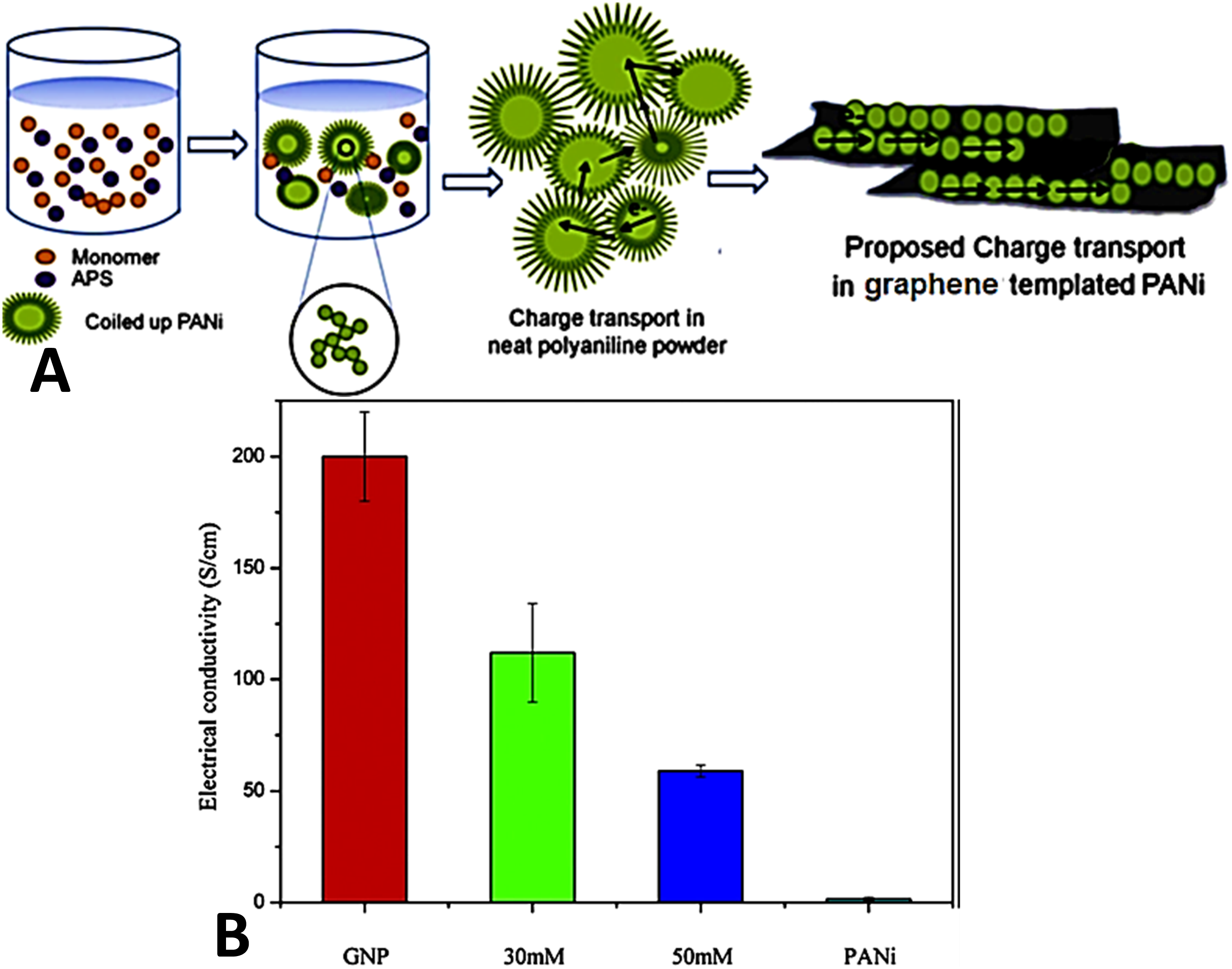
Benchikh et al.
78
reported on the polyaniline and olive stones functional reduced graphene oxide based nanocomposites. The resulting material was used for the formation of supercapacitor electrode. During synthesis, reduced graphene oxide was dispersed in water to form electrostatic self-assembly with the olive stone molecules (Figure 4(a)). (a) The proposed preparation pathway of polyaniline@olive stone-reduced graphene oxide (PANI@OS-rGO) nanomaterial; (b) Ragone plots for electrodes at various current densities; and (c) Nyquist plots for electrode materials.
78
Reproduced with permission from MDPI.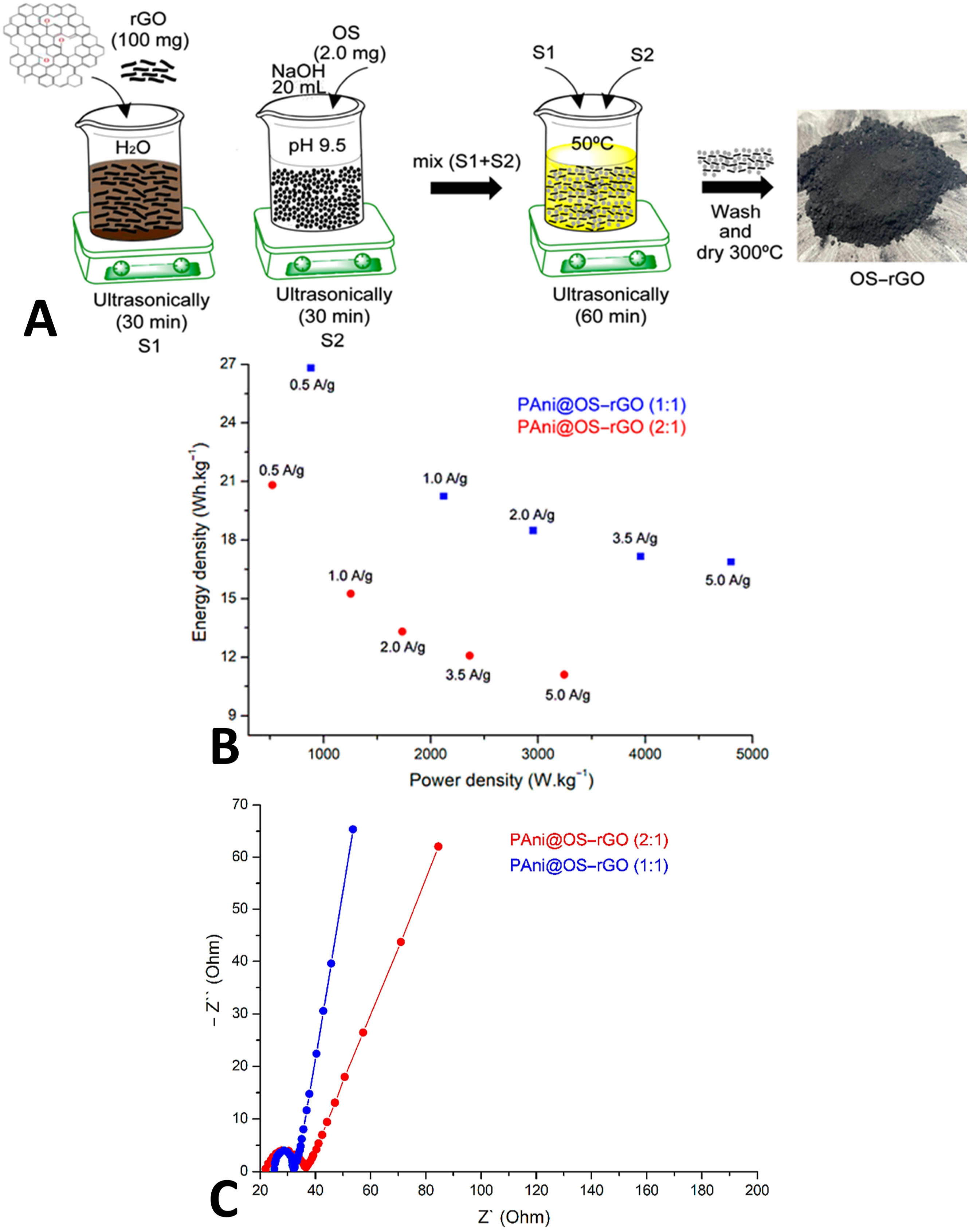
The relationship between the energy density and power density was studied using the Ragon plots and revealed a non linear relationship which became stable at high current density (Figure 4(b)). High energy density behaviour of the nanocomposites was observed due to the formation of interlinked network in the polyaniline/olive stone-reduced graphene oxide. The conducting network formation led to the better charge transfer and Faradaic characteristic in these nanomaterials. The optimally designed electrode had capacitance and energy density of 582.6 Fg−1 and 26.82 Wh·kg−1, respectively at 0.1 Ag−1. The capacity retention of 97% was observed over 3000 cycles for these nanocomposite electrodes. Nyquist plots presents high resistance values of the nanocomposite electrode materials up to 6 Ω (Figure 4(c)). The resistance behaviour was attributed to the electron exchange processes on the electrode surface. Lin et al.
79
developed the polyaniline modified graphene nanocomposites for the lithium ion battery electrodes. For the formation of polyaniline, ammonium persulfate was used as an oxidant for polymerization. Due to in situ polymerization of aniline monomer on the graphene surface, polyaniline showed superior adsorption and interaction properties with the nanofiller. Consequently, the polymer chains formed an interlinked network nanostructure on the graphene surface. Figure 5(a) shows a uniform covering of polyaniline on the porous network structure formed with the graphene nanosheets. Accordingly, Figure 5(b) presents a porous polyaniline-graphene network nanostructure having pore sizes of 10 nm. Furthermore, Figure 5(c) shows the thermal stability profiles of the nanomaterials. Heat stability of the nanocomposite was observed from 100°C to 300°C having 10% weight loss and char yield in the range of 84%–94%, and the weight loss seemed to be continued up to 500°C. (a) SEM images of polyaniline modified graphene (PMGC); (b) higher magnification image of PMGC; (c) Thermogravimetric curves of polyaniline-modified graphene composites; and (d) Impedance of C0A0 and C0.5A0.
79
SEM = scanning electron microscopy. Reproduced with permission from MDPI.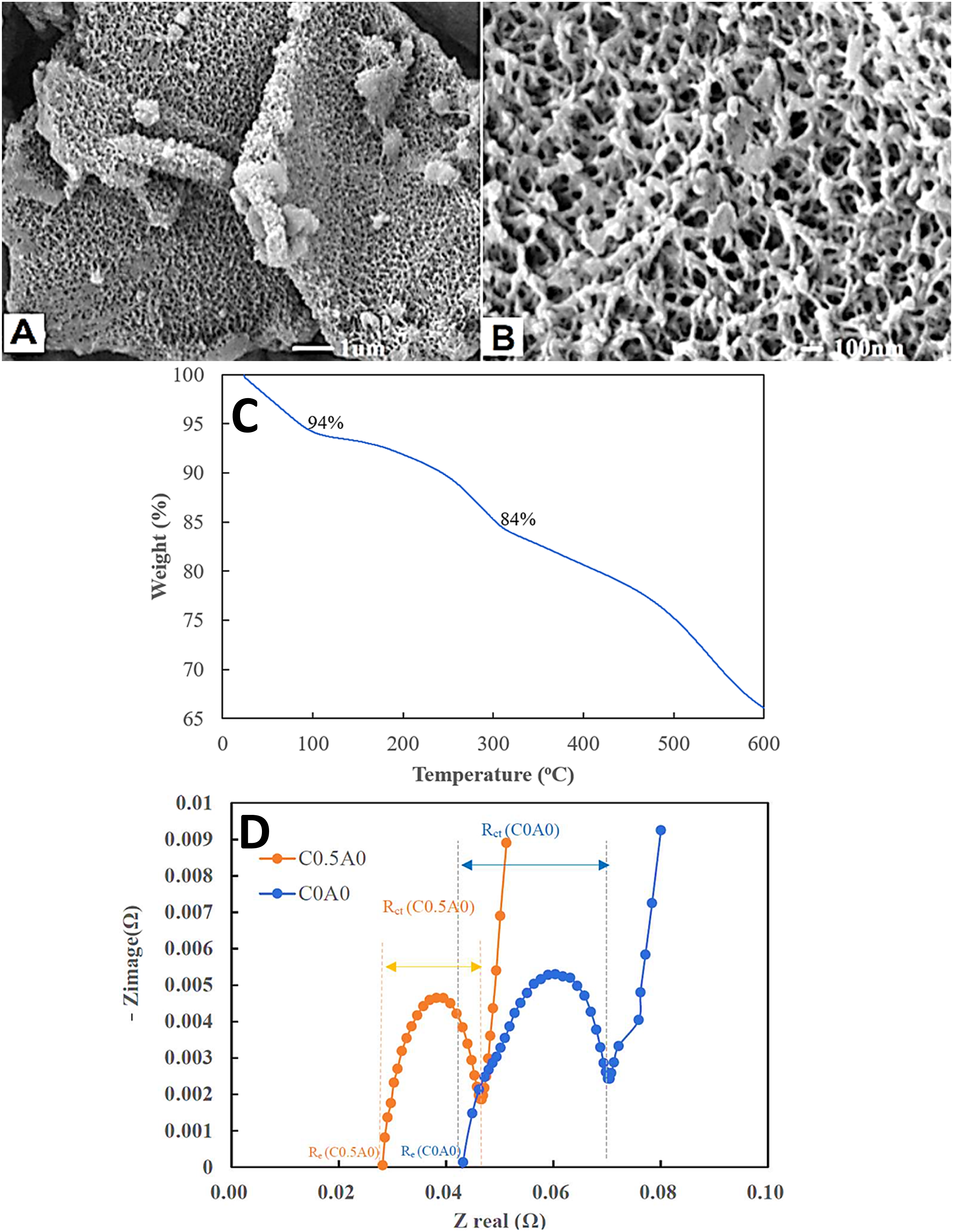
Figure 5(d) illustrates the impedance spectra at 3.6 V Nyquist plots. The real and imaginary impedance parts were plotted at varying angular frequencies. Increasing nanofiller contents were found to reduce the resistance to 0.018 Ω, relative to neat sample with resistance of 0.043 Ω. Including 0.5 % nanofiller reduced the internal resistance of the nanomaterial electrode to facilitate the charge transfer and charge-discharge capacity. Subsequently, the nanomaterial showed potential benefit to store energy in the systems. For better lithium ion battery performance, use of polyaniline and graphene based nanocomposites have been suggested.
Baniasadi
80
formed the conducting polyaniline/graphene nanocomposites. Polyaniline was formed using an in situ emulsion polymerization using sodium dodecyl sulfate. Including up to 1 wt% nanofiller enhanced the electrical conductivity in the range of 2 to 7 S cm−1. Transmission electron microscopy image of graphene shows a transparent monolayer nanostructure (Figure 6(a)). Figure 6(b) depicts the micrographs of a doped polyaniline nanostructure. Consequently, the polyaniline nanoparticles seemed to be deposited in the size range of 10-15 nm. A finely dispersed polyaniline nanostructure was observed for the nanomaterials. Figure 6(c) shows the weight loss behaviour of the nanocomposites with initial weight loss at 110°C, second weight loss in the range of 110 °C–300°C, and subsequent weight loss >300°C. The weight loss at high temperature was observed due to the breakdown of nanocomposite nanostructure. According to differential scanning calorimetry, the endothermic and exothermic peaks around 90 °C–120°C and 150 °C–220°C, respectively, were observed (Figure 6(d)). Due to the moisture contents, endothermic peaks were also observed. Moreover, an exothermic peak of the nanocomposite was observed at high temperature due to the heat stability of the nanocomposite. (a) Transmission electron microscopy image of the graphene nanosheet; (b) scanning electron microscopy micrographs of doped polyaniline; (c) thermogravimetric; and (d) differential scanning calorimetry thermograms of (a) single doped PANI-ES; (b) PANI-EB; (c) binary doped PANI-ES; and (d) polyaniline/graphene nanocomposite.
80
PANI = polyaniline; PANI-ES = Emeraldine salt polyaniline; PANI-EB = Emeraldine base polyaniline. Reproduced with permission from Elsevier.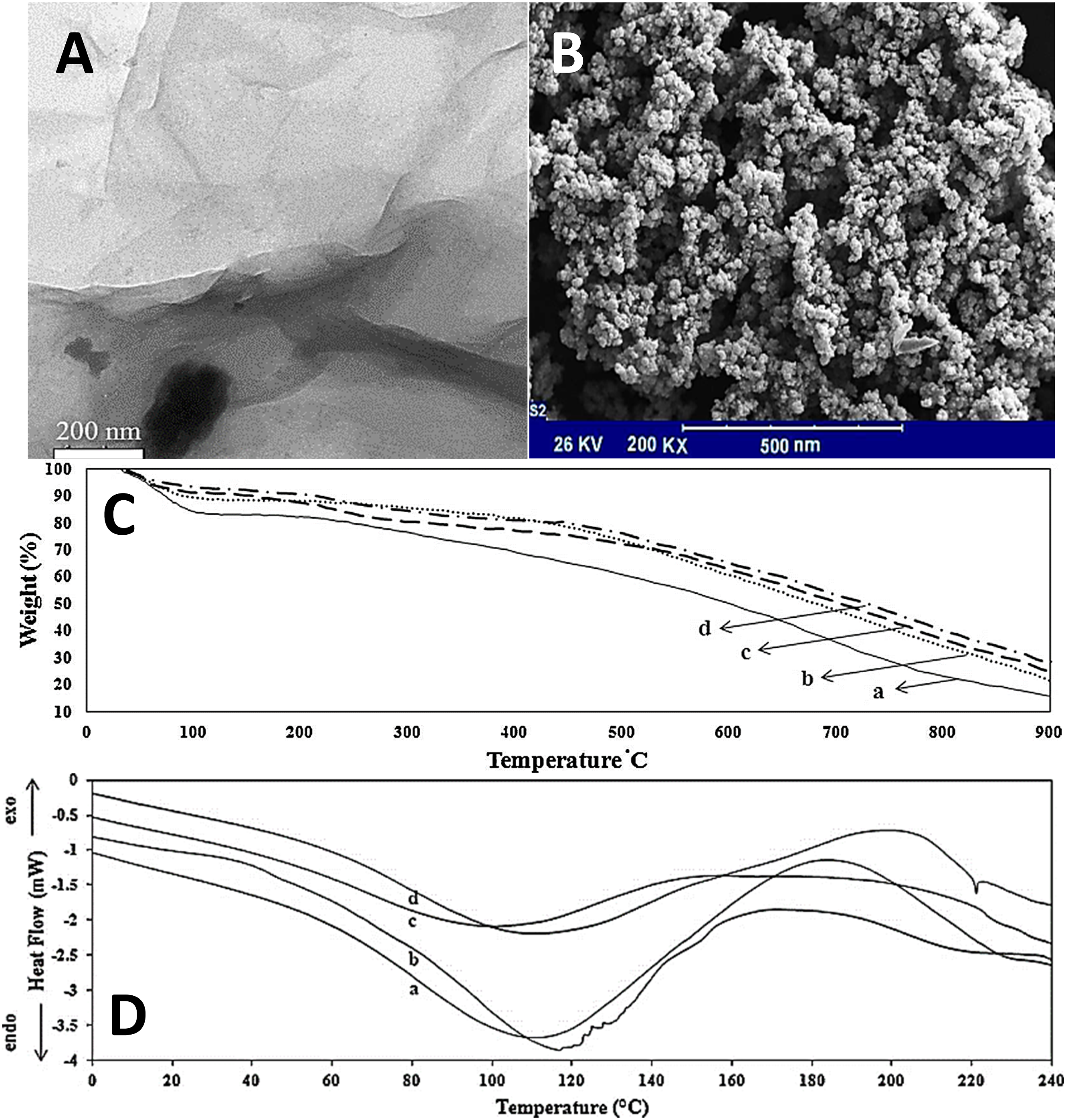
Behera et al.
81
used a one step procedure to form the polyaniline, copper, and bovine serum albumin based nanocomposite and studied their structural and sensing properties. Presence of reduced graphene oxide seemed to enhance the electron transfer between the nanomaterial electrode and the analyte. Field emission and transmission electron microscopy revealed irregular and cubical microstructures with diameter of about 100-200 nm (Figure 7(a)–(d)). (A) (a) FESEM image of PANI-Cu@BSA and TEM images of (b) PANI-Cu@BSA and (c, d) PANI-Cu@BSA/rGO; and (B) linear plot of current versus pH.
81
FESEM = field emission scanning electron microscopy; TEM = transmission electron microscopy; PANI-Cu@BSA/rGO = polyaniline-copper integrated@bovine serum albumin/reduced graphene oxide. Reproduced with permission from MDPI.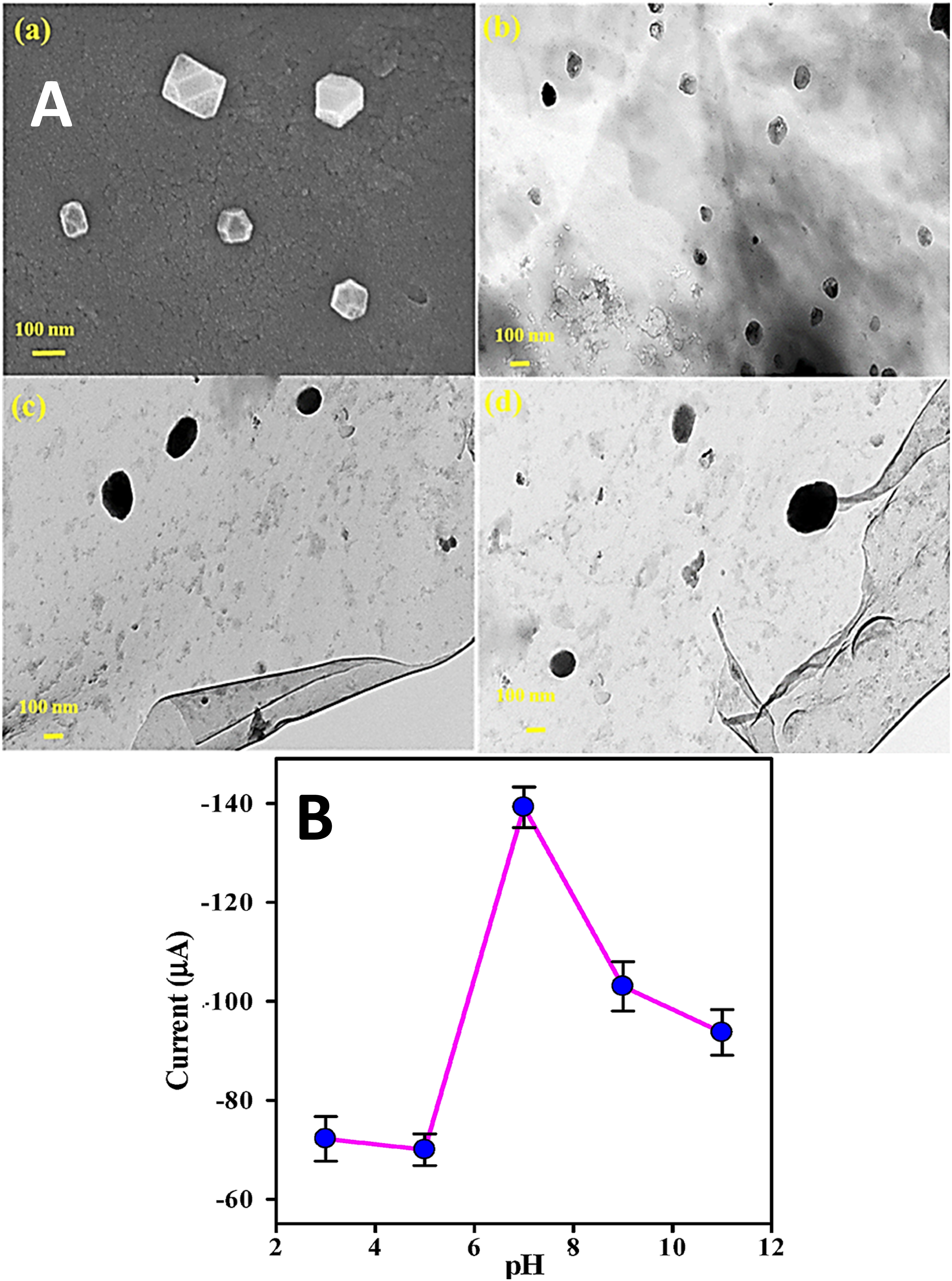
In the polyaniline/copper/bovine serum albumin/reduced graphene oxide nanocomposite, bovine serum albumin formed better miscibility through fine nanoparticle dispersion. Cyclic voltametric studies were carried out to study the effect of pH changes to reduce the bovine serum albumin. From the pH levels from 3 to 7, the current was found to increase, whereas the current was decreased in the pH range from 7 to 11 (Figure 7(b)). At pH 7, the maximum peak current was observed due to facilitated electron movements on the electrode surface. Accordingly, the electrode was used for the electrochemical detection of dimetridazole compound. The senor revealed significantly high sensitivity of about 5.96 μA μM−1 cm−2 at very low detection limit of 1.78 nM. In addition, the nanocomposite sensor had fine stability, repeatability, and selectivity features.
Significance and prospects
Solar cell
Conducting polymer and nanocarbon based nanomaterials have been beneficially applied in various energy and electronics related technological sectors.82,83 Accordingly, the polyaniline has been used in wide ranging fields of solar cells, supercapacitors, sensors, batteries, etc.84–86 Numerous carbon nanoadditives have been reinforced in the polyaniline matrix to form the high performance nanocomposites.
87
In this regard, graphene nanostructures have been effectively applied for the solar cells application.88,89 Unique two dimensional graphene nanosheets revealed superior photovoltaic efficiency.
90
Graphene and derived nanomaterials own low price, durability, high performance, and environmental friendliness for the dye sensitized solar cells. Shih and colleagues
91
used an electrochemical technique to form the polyaniline/graphene nanocomposite films on the fluorine doped tin oxide substrate. The nanocomposites were studied for the structure, thermal, and photovoltaic performance. Graphene nanofiller was capable of developing π-π stacking interactions with the aromatic polyaniline matrix. Figure 8(a) shows the thermogravimetric analysis curved of the polyaniline/graphene nanomaterials. (a) Thermogravimetric plots of polyaniline-graphene (PANI-G) nanocomposite films; and (b) current density of dye sensitized solar cell with platinum (Pt) and nanocomposites.
91
Reproduced with permission from Elsevier.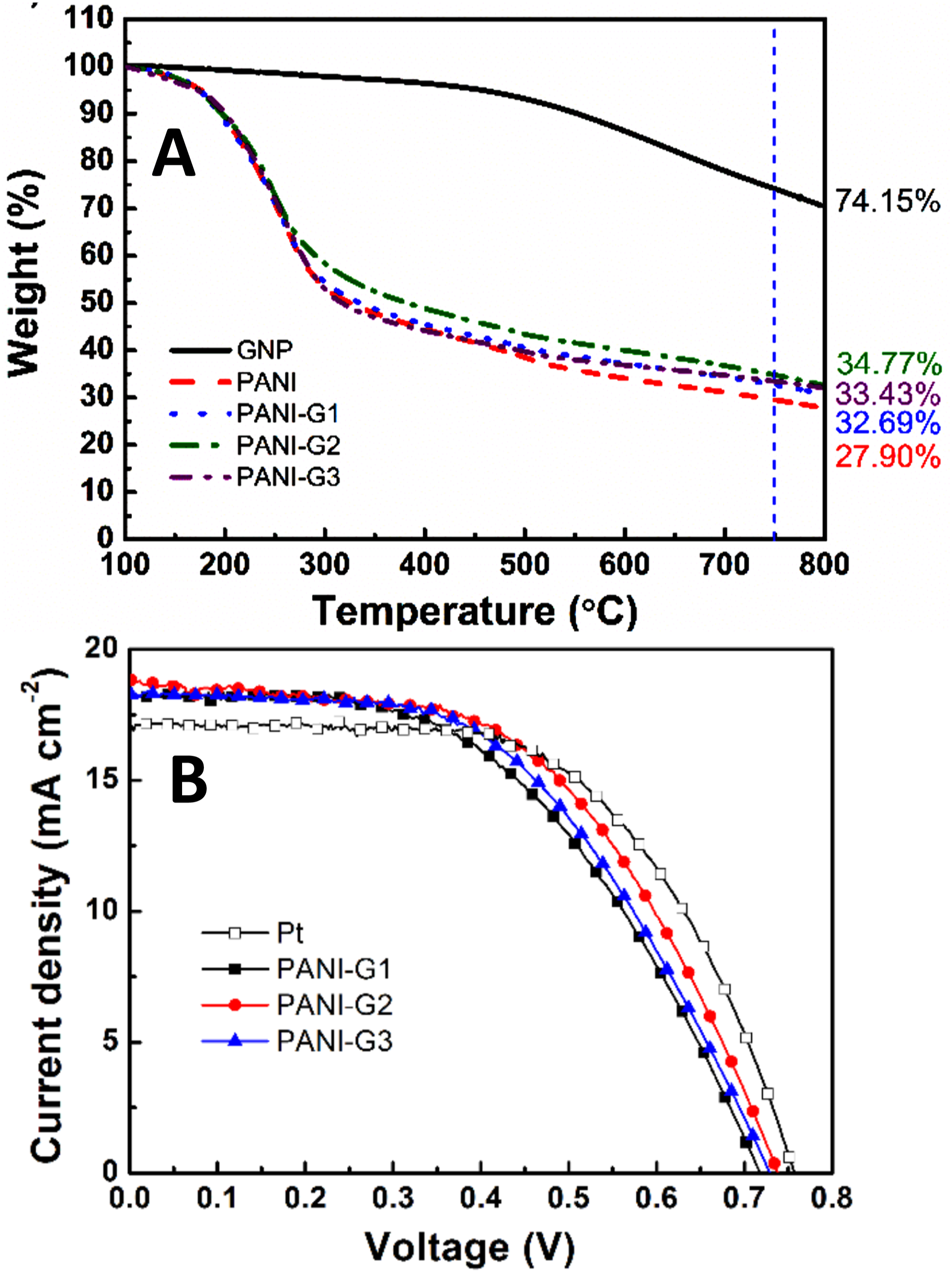
Properties of polyaniline/graphene nanocomposites for dye sensitized solar cell. 91 Voc = open circuit voltage; Jsc = short circuit current. Reproduced with permission from Elsevier.
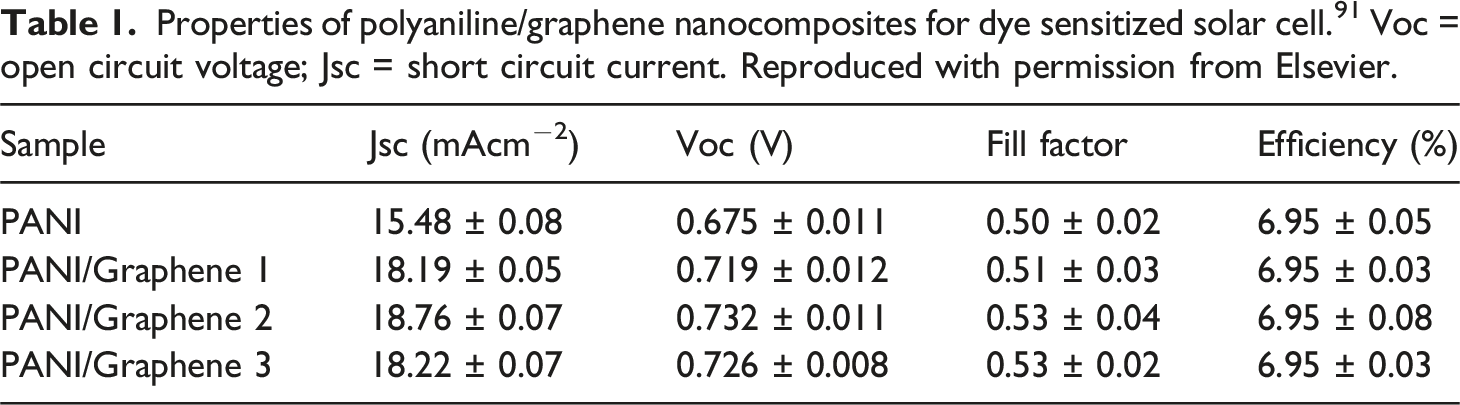
A recent study by Mahmood et al. 92 was observed for the in situ polymerization of aniline monomers with graphene nanosheets to form polyaniline/graphene nanocomposites aiming for the counter electrodes of dye sensitized solar cell. According to the results, adding amounts of graphene from 6 to 15 wt% considerably influenced the electrochemical and photovoltaic performances of dye sensitized solar cells. Herein, a counter electrode based on polyaniline and 9 wt% graphene led to comparable power conversion efficiency of ∼7.5 % and interface resistance of 20.1 Ω, to that of the platinum-based solar cells (power conversion efficiency of 7.6 % and interface resistance of 19.2 Ω). 93 In addition, the polyaniline/graphene nanocomposite based counter electrode depicted current density of 15.6 mA cm−2, open-circuit voltage of 785 mV, and fill factor 0.6, similar to that of the platinum-based solar cell electrode. Another recent research attempt by Saed et al. 94 reported on the polyaniline/graphene oxide and polyaniline/titania nanocomposites as the hole transport layer materials for organic solar cells. The polyaniline/graphene oxide hybrids revealed significantly higher open circuit voltages and power conversion efficiencies of around 520 mV and 2.4 %, respectively, than the values of the polyaniline/titania nanocomposites (310 mV and 1.358%, respectively). Thus, the result depicted better interactions and interfacial network formation between the polyaniline and graphene, relative to polyaniline-titania; therefore, leading to superior solar cell performance. In recent times, Reza et al. 95 prepared the polyaniline/graphene oxide nanocomposites by using plasma surface modification method and applied as counter electrodes for dye-sensitized solar cell. The resulting electrodes had optimum power conversion efficiency of 3.17 %.
Real world applications of conjugated polymer/graphene based solar cells have been perceived in the mobile phone recharging, retro-fitted photovoltaic window screens, and domestic power sources.96,97 Moreover, the polyaniline/graphene nanocomposites have been found practicable as photoabsorbers for interfacial solar steam generation, due to reduced thermal conductivity and durability properties. 98 Such polymer/graphene based photoabsorbers have been found useful due to high solar to thermal efficiency of ∼96 %, under 1 sun (1 sun = 1 kW m−2) illumination.
Supercapacitors
Supercapacitors have been categorized as efficient energy storage devices due to their superior capacitance, power density features, charge-discharge, and life span.99–101 In this concern, high surface area, conducting, and durable nanomaterials have been preferred for the supercapacitor components.
102
Graphene based nanomaterials have been used in supercapacitors owing to their desirably conducting and capacitance characters.103,104 Several combinations of the graphene and derivatives with polyaniline have been reported in literature.105,106 Gao and researchers
107
fabricated the aniline-functional-graphene nanocomposites using the chemical grafting approach. Figure 9 A demonstrates a simple method for the formation of functional nanomaterials. In this route, graphene oxide was reduced through sodium borohydride. During reduction, the surface functional groups of graphene oxide (epoxy, carbonyl, hydroxyl) were reduced.
108
Reduced GO was grafted with the aniline monomers using the p-phenylenediamine in the presence of NaNO2 and aryldiazonium salt.
109
Consequently, the aniline monomer was grafted to the graphene surface and then polymerized. The covalent bonding between the polymer and nanocarbon in turn facilitated the electron and charge transportation to enhance the specific capacitance properties.
110
Transmission electron microscopy images of the aniline functional graphene showed wrinkled and wavy structure having high surface area (Figure 9(b)). The polyaniline and functional graphene nanocomposite revealed the coating of polymer chains on the nanosheet surface (Figure 9(c)). Such nanostructure was accountable for a rapid charge transfer through the nanostructure. (a) Preparation of a-G-PANI nanocomposite; (b) TEM of a-G and (c) a-G-PANI200; (d) specific capacitances of a-G, PANI and a-GPANI200 at different current densities.
107
TEM = transmission electron microscopy; APS = ammonium persulfate; GO = graphene oxide; RGO = reduced graphene oxide; a-G = aniline functionalized graphene; PANI = polyaniline; a-G-PANI = aniline functionalized graphene-polyaniline. Reproduced with permission from Elsevier.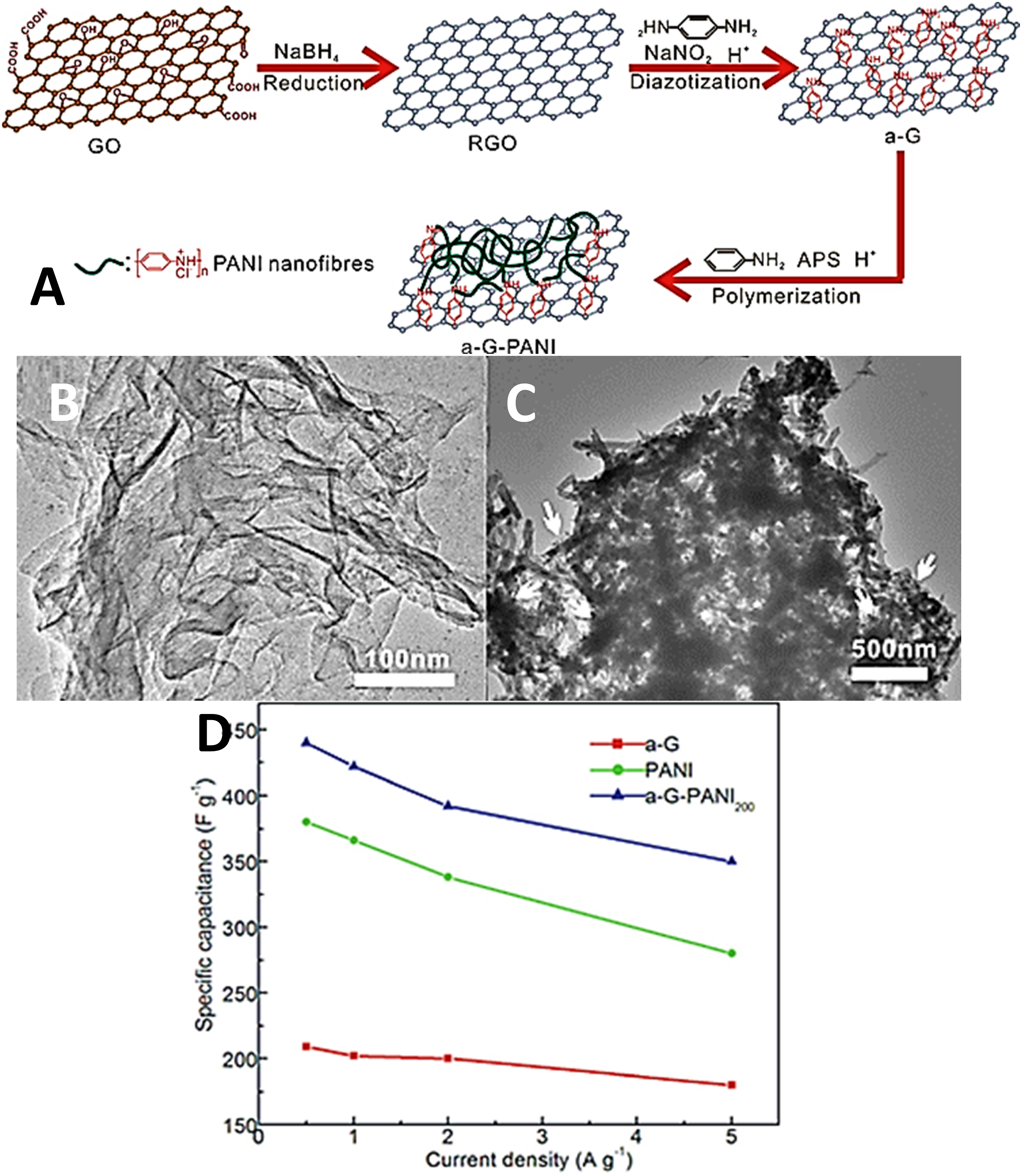
Figure 9(d) depicts the specific capacitance values of the aniline functional graphene, polyaniline, and polyaniline/aniline functional graphene nanocomposites. All the samples displayed a decrease in the specific capacitance with the increasing current density in the range of 0.5-5 Ag−1. The decrease in capacitance values was observed from 143 to 123 Fg−1, 380 to 280 Fg−1, and 440 to 350 F g−1 for functional graphene, polyaniline, and the nanocomposite, respectively. The overall capacitance retention was found in the range of 73%–80%. The highest value of specific capacitance 422 Fg−1 was obtained at 1Ag−1 for the nanocomposite sample. It has been observed that the covalent linking between the polyaniline and graphene led to an enhanced charge transfer through the nanomaterial to enhance the resulting capacitance values.
Recently, Mupit et al. 111 documented the polyaniline/graphene oxide nanocomposites through chemical exfoliation and in situ techniques. The ensuing nanocomposite was used as a supercapacitor electrode and revealed high specific gravimetric capacitance of around 303 Fg−1. Tale et al. 112 designed the supercapacitor electrodes based on polyaniline matrix filled with graphene, graphene oxide, and manganese dioxide nanoparticles. The polyaniline/graphene/graphene oxide/manganese dioxide nanocomposite revealed remarkably high specific capacitance of >1882 Fg−1 and capacitance retention of ∼98 % (6063 cycles) in a symmetric galvanostatic charge/discharge cyclic performance. The superior supercapacitor electrode performance was attributed to the interfacial interactions in the multiphase system. In recent times, Pawar et al. 113 designed the supercapacitor electrodes derived from polyaniline and reduced graphene oxide via chemical bath deposition approach. The resulting polyaniline/reduced graphene oxide nanomaterials had specific capacitance of 1130 Fg−1 along with 82 % capacitance retention (5000 charge-discharge cycles). Moreover, high specific energy density and specific power density of 23 Wh kg−1 and 732 W kg−1, respectively, were observed for the polyaniline/reduced graphene oxide nanocomposite based electrode. The electrochemical features of the nanocomposites suggested them competent for manufacturing advanced asymmetric supercapacitors.
In real life, scientists are continuously struggling to incorporate the polyaniline/graphene nanohybrid designs as the supercapacitor anodes to attain higher storage capacities, cyclic rate, and longevity performance of these electrodes. 114 In addition, recently, graphene derived supercapacitors depicted rapid charging capabilities to store large amount of electricity, relative to traditional commercial superactions. 115 Furthermore, ongoing research on conjugated polymer/graphene derived micro-supercapacitors has predicted the commercially availability, within few years, of these devices for low energy utilizations, like portable computers, laptops, smartphones, etc.116,117
Sensor
An immense scope of graphene and related nanostructures has been observed in the field of electronics.118,119 Graphene addition has been found to enhance the electrical conduction of the matrix due to the formation of the crosslinked network in the matrix.
120
Consequently, graphene has been introduced in the nanocomposite designs to enhance the conduction and sensitivity properties of the resulting nanomaterial. High performance, graphene nanostructures have also been applied in the technical sensing fields like straintronics.121,122 Further research seemed to be desirable in this field to form the advanced electronic devices.
123
The literature reports have been observed for the application of the polyaniline/graphene nanocomposites in the field of sensors.
124
The resulting nanomaterials based devices have been used to sense the gas molecules, moisture, chemicals, etc.125–127 Al-Hartomy et. al.
128
researched on the polyaniline and graphene nanoplatelet based nanomaterials. Structural studies have confirmed the presence of stacking interactions between the polyaniline and delocalized electrons of the graphene derivatives. Consequently, the sensor was used to detect the vapors of benzene and toluene vapors. Moreover, the pristine polyaniline and the graphene filled polyaniline have been investigated for the temperature-dependent conductivity around 30 °C–200°C (Figure 10(a)). The polyaniline and nanocomposite depicted three step variation in conductivity with changing temperature. Increase in the electron conduction of the nanocomposites seemed to be dependent upon the synergetic effects between the polymer and the nanoparticles. Figure 10(b) and (c) shows the change in the material sensitivity towards the toluene and benzene vapours. The non-polar gaseous vapours can interact with the polyaniline/graphene nanoplatelets nanocomposites due to the presence of free electrons in their structures. Subsequently, the active sites in the graphene nanoplatelets caused better chemisorption of these gas molecules. In the nanocomposites having low nanofiller contents, interaction of the polymer chains with the nanocarbons may also decrease, so resulting in the enhanced resistance values. Consequently, the charge transfer and protonation have caused desirable electron conduction in the polyaniline and derived nanomaterials.129,130 Combination of the carbon nanoparticles with the polyaniline matrix has been found to enhance the robustness and electron conduction through the system.
131
In this regard, the effect of graphene dispersion and alignment in the polymer matrices for superior nanocomposite characteristics has been well established in the literature.132,133 Consequently, these characteristics of polyaniline and nanomaterials have been found to enhance the sensing characteristics of the nanomaterials.
134
(a) Variation of conductivity as a function of temperature; (b) variation of sensitivity as a function of toluene gas concentration; and (c) variation of sensitivity as a function of benzene gas concentration.
128
Reproduced with permission from Springer.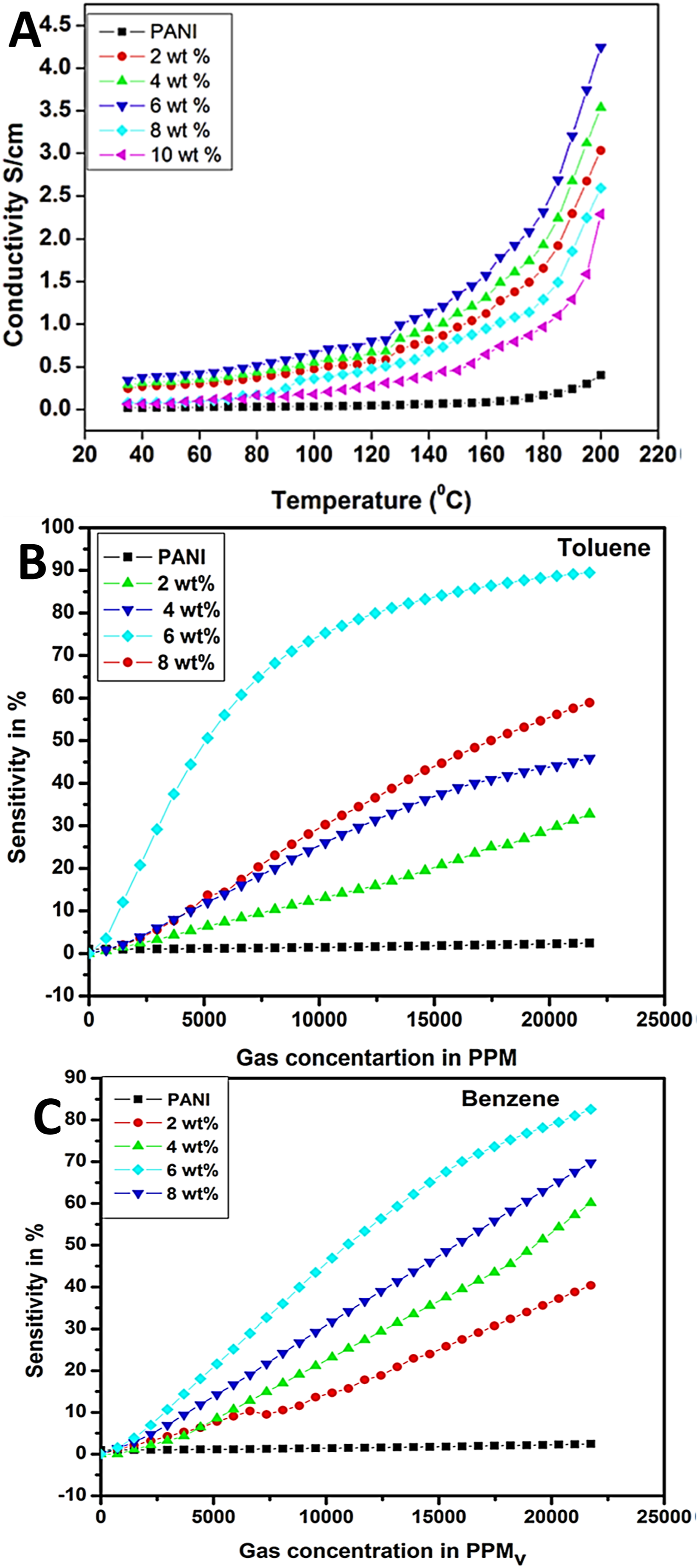
Among recent studies, Mohammed et al. 135 fabricated the silver chloride functional protonated polyaniline and graphene oxide based nanocomposites for carbon monoxide sensing. The protonated polyaniline-silver chloride/graphene oxide hybrid had reasonable sensitivity of ∼20 %, at 130 ppm of carbon monoxide. Moreover, the sensor depicted response and recovery times of around 50 and 39 s, respectively, and had repeated cycling durability properties. In recent years, Chethan et al. 136 formed the polyaniline/graphene oxide nanocomposites by using chemical in situ polymerization and used as an efficient humidity sensor. Including 15 wt% nanofiller in the polyaniline matrix showed a maximum humidity sensing response of >93 %, in the dynamic timing of 4-7 s. Furthermore, Hosseine et al., 137 recently, reported on a multiphase system of polyaniline, nafion, graphene oxide, and magnetite Fe3O4 nanoparticles. The as prepared polyaniline/nafion/graphene oxide/Fe3O4 nanocomposite was used as a biosensor. According to electrochemical studies, the biosensor had a linear range of 102-106 cells mL−1 and detection limit of 5 cells mL−1.
In factual world, ultrasensitive biomedical sensors and humidity sensors based on conjugated polymer and graphene have been developed and used by the medical and profitable companies. 138 These sensors have been found commercially efficient towards sensing the molecules like moisture, DNA, dopamine, nucleotides, and so on. However, one challenging aspect of these sensors is the lack of specificity in compounds detection of samples having more than one analyte.139,140 Besides, a real life application of polyaniline/graphene hybrid has been reported for scalable nanofabrication of field-effect transistor sensor arrays for sensing toxic heavy metals and microbes in domestic water. 141 The scaled-up fabrication of graphene based field-effect transistor sensor arrays showed high frequency impedance, low frequency noise, and high sensitivity, in real practical utilizations.
Challenges and future directions
As surveyed in the preceding sections of this review, polyaniline has shown remarkable enhancements in its physical properties (e.g., electrical/charge conductivity, mechanical stability, thermal constancy, and so on) with the addition of graphene, graphene oxide, reduced graphene oxide, or functional graphene nanoparticles. 142 Consequently, these superior physical attributes have been credited to the mutual π-π stacking interactions, compatible interface formation, and electron donor-acceptor associations between the matrix-nanofiller. 143 However, there is very limited literature available regarding the phenomenon or mechanism behind the synergies between polyaniline/graphene phases. From the literature so far, it can be suggested that there is an intense need of understanding and investigation of the fundamental interaction mechanisms and synergetic effects between polyaniline matrix and graphene nanosheets in the nanocomposite systems. 144
For polyaniline and graphene derived nanomaterials, various structural/property characterization techniques, ranging from microscopy (scanning electron microscopy, transmission electron microscopy) and spectroscopy (infrared spectroscopy, Raman spectroscopy, X-ray photoelectron spectroscopy) to specific physical characteristics analysis techniques for thermal (thermogravimetric analysis and differential scanning calorimetry), mechanical, electrochemical, impedance, electrical conductivity, and sensitivity measurements have been used. 145 These techniques actually provide researchers with the tools essential to comprehend/manipulate nanomaterial characteristics at atomic/molecular levels. Nevertheless, there are certain strengths and limitations associated with the implication of each of these characterization practices for comprehensive understanding of the nanomaterials. For example, scanning electron microscopy and transmission electron microscopy allow high resolution imaging for detailed structural examination (nanoparticle size, shape, orientation, aggregation, etc.); whereas, the spectroscopic methods like infrared spectroscopy and Raman spectroscopy offer insights into the composition and bonding configurations of the polyaniline/graphene nanocomposites. 146 Despite the effectiveness of these characterization methods so far, there are several unaddressed challenges which need to be resolved for precise data interpretation/analysis, like desirable high resolution, throughput, sensitivity, and adoption of standard protocols for these techniques. In this regard, progressions in existing characterization techniques and combination of classical analysis methods by adopting the current technological revolutions, may brought about dynamic insights to unfold the true potential of polyaniline/graphene nanomaterials for promising future technological applications. Henceforth, characterization techniques play a key role in understanding the structure/characteristics and specific practical aspects of the polyaniline/graphene nanocomposites.
Existing fabrication techniques for polyaniline and graphene based nanocomposites mainly include the solution processing, in situ polymerization and allied facile approaches.147,148 Out of these, in situ polymerization has been expansively applied to develop the polyaniline/graphene nanocomposites. 149 In this technique, aniline monomers are usually dispersed in an appropriate solvent (sometimes with surfactants) and then polymerized with the pre-dispersed graphene to form the nanocomposites. In this tactic, aniline monomers are initially adsorbed on the graphene nanosheets and then allowed to polymerize to ensure the fine dispersion and interlinking between the polyaniline-graphene phases. 150 Consequently, the in situ techniques have been deliberated as low price, efficient, as well as environmentally friendly for the formation of polyaniline/graphene nanomaterials. Besides, solution methods have been applied due to low fabrication costs, low energy expenditures, facile processing parameters, and ecological benefits. 151 These methods usually involve the dispersions of preformed polymer and graphene nanoadditives in their respective solvents. Afterwards, solution mixing, solvent evaporation, and casting are generally performed to attain the desired nanomaterial. Polyaniline has been found to adsorbed on the graphene surface by developing interactions, so leading to homogeneous matrix-nanofiller dispersions. 152
Despite the success of the facile lab-scale techniques for polyaniline/graphene hybrids, there is an utmost need for analyzing the possibilities for the scaleup synthesis of these nanomaterials. 153 For scalability, major challenges encountered so far include the achievement of consistent nanoparticle dispersion, matrix-nanofiller interactions, and superior property-performance profiles of the large scale models processed using simple techniques, like in situ polymerization. 154 In other words, parameters of the lab. scale techniques need to carefully controlled and optimized to attain structural homogeneity and resulting high performance of the scalable nanomaterials on commercial level. Herein, utilization of hybrid methods, that is, combination of facile lab. scale approaches, as well as invention/adoption of advanced techniques, like printing, spinning, and coating tactics can be beneficial for the scalability of polyaniline/graphene nanocomposites. 155
Into the bargain, it seems indispensable to point out the environmental concerns related to the synthesis and utilization of the polyaniline/graphene nanomaterials. 156 Perceptibly, the manufacturing, end of life or ingestion, and recycling of these nanocomposites have been found connected to the greenhouse emissions. 157 Generally, 100 % recycling of nanocomposites is not possible, therefore these materials are mostly landfilled at end of life. Subsequently, to avoid the greenhouse emission effects of the polyaniline/graphene hybrids, it has been suggested to analyzed the cradle-to-grave life cycles for better insights into recyclability and sustainability of these nanocomposites.158,159 Recently, cradle-to-grave life cycles assessments appear as handy tools for evaluating the environmental hazards caused by the composites or nanomaterials. 160
Like other polymeric nanocomposites, economical aspects of the production, utilization, and recycling of the polyaniline/graphene nanocomposites need to be analysed.161,162 Particularly, natural/green, sustainable, and non-toxic raw materials or precursors along with the facile room temperature synthesis methods must be practiced for the manufacturing of these hybrids. In this regard, circular economy concept need to be introduced to access the product lifecycle from development, production, use, preservation, and recycling.163,164 Henceforward, inexpensive and environmentally friendly strategies must be adopted for the fabrication, employment, and recycling of the polyaniline/graphene nanocomposites to meet the economical and ecological needs of the modern technological era. 165
Precisely speaking, as stated in the preceding discussions, the literature up till now infers imperative features and applied potential of the polyaniline/graphene nanocomposites.166,167 For this purpose, using graphene derivatives, like graphene oxide or reduced graphene oxide, has been opted as a low cost strategy. 168 Nevertheless, a number of underlying challenges have been discovered regarding the economical and ecological large-scale processing of high performance polyaniline/graphene nanocomposites. 169 Most importantly, only limited research endeavours have been seen so far on the design of commercial scale development of polyaniline and graphene based nanocomposites for real world applications. However, the available reported research indicated an enormous potential forecasts of these nanomaterials for upcoming future nanotechnological breakthroughs. 170 In practical grounds, fabrication of polyaniline/graphene hybrids having consistent microstructures and well defined property-performance profiles, by using predefined processing parameters, seemed to be indispensable for stability, durability, and long-term performance of these nanocomposites in the fields of solar cells, supercapacitors and sensors. For future industrial level designs thorough literature surveys and innovative high-tech design inventions of polyaniline/graphene nanocomposites using advanced strategies need to be focused by the field researchers. Henceforward, comprehensive scrutinization of factors, encounters, opportunities, and techniques projected significant future industrial forecasts for high end polyaniline/graphene hybrids.
Summation
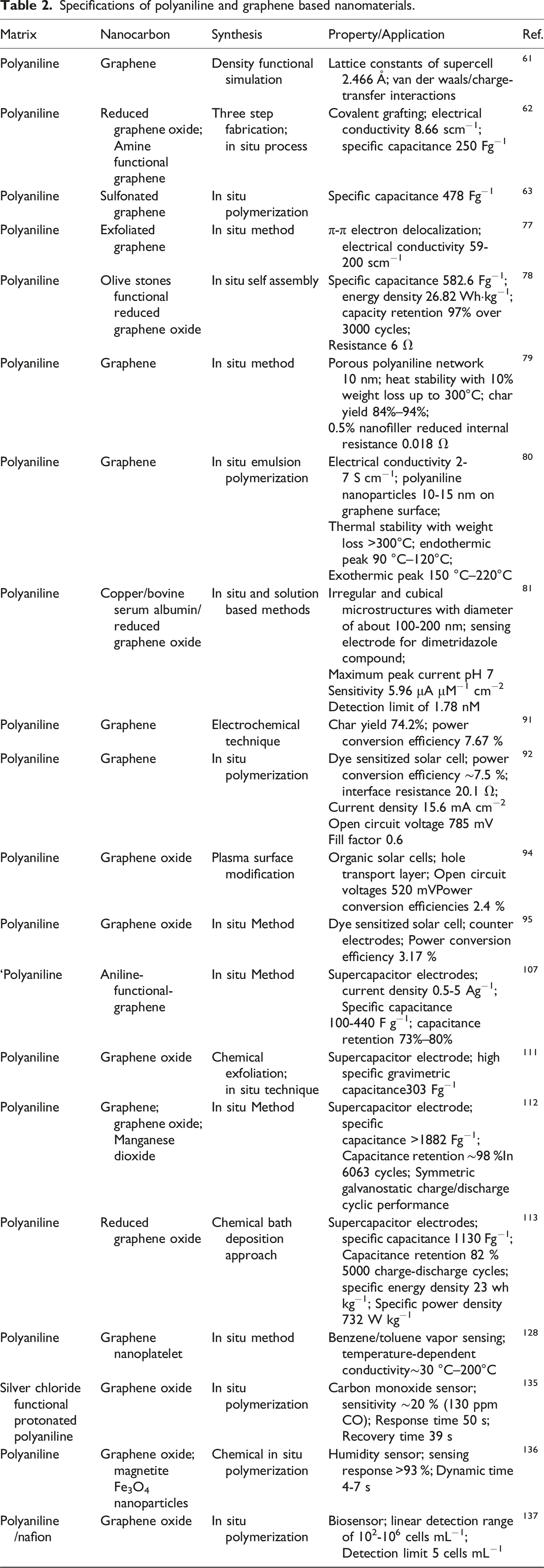
Specifications of polyaniline and graphene based nanomaterials.
An outlook of polyaniline and graphene based nanomaterials.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
