Abstract
The current paper aims to study the behavior of Polyamide 6 (PA6) and Polyamide 6 reinforced with 30% of short glass fibers (PA6GF30) under glycol water (GW) mixture used for cars as coolant fluid. Samples were fully immersed in the mixture of GW (20:80) at 50°C, 70°C and 90°C for up to 80 days and periodically weighted. Results gathered revealed the occurrence of substantial changes especially for PA6 samples aged at 90°C. The long term ageing leads to the formation of ester species due to thermo-oxidation as pointed out by Infrared spectroscopy analysis. Moreover, dynamic mechanical analysis measurements showed that glycol water acted as an effective plasticizer in lowering the Tg of the polyamide 6. This plasticizer effect was confirmed by the gain of ductility at early stage of ageing for both materials (PA6 and PA6GF30). Nevertheless, after 80 days of ageing the effect of glycol water ageing is no longer physical in nature and an overall loss of mechanical properties was noticed. Indeed, an embrittlement of PA6 was measured that can be attributed to a decrease of the level of entanglements within the polymer. Nevertheless, for PA6GF30 material, the loss of strength and stiffness was the result of the interfacial debonding between the fibers and the matrix.
Introduction
Owing to their high mechanical strength, ease of manufacturing, low density, and chemical resistance, Polyamides (PAs) have known a widespread use that has percolated in many industrial applications. Thus, they are prominently regarded as a material of choice in many marine structures, offshore applications, and for components where they are in contact with fluids such as fuel lines.1–4 In order to fulfill their functions these materials sustain different conditions (humidity, moisture, heat, ultraviolet radiation and chemicals) that may affect their long term sustainability.5,6 These conditions are generally admitted that they are the most deleterious factors that may be considered as a degradative condition for polymers.7–14 The diversity of results increases the concerns of durability of such materials in different environment conditions.15–20 For PAs, and due to their hygroscopic nature, hygrothermal ageing remains the most studied ageing condition.21–32 This particularity is caused mainly by the presence of the polar functional amide groups in their chemical structure.21,28–30 Thus, they absorb up to 8.5% when they are immersed in water at 23°C.31–33 This water uptake remains the highest one in comparison with poly (ethylene terephthalate) (PET) and poly (butylene terephthalate) (PBT) for the same ageing conditions as reported by Bergeret et al. 2001. 33 This finding confirms the assumption that the diffusion mechanism is greatly dependent on the polymer’s polarity, and the presence of hydrophilic species.14,34–36 Thus, the fluid interacted with polymer via intermolecular reactions14,21 inducing as a consequence reversible and/or irreversible change. 22 The former are physical in nature such as plasticization. This effect leads to an increase in the chain mobility of the polymer inducing therefore a lowering of glass transition temperature.23,37–39 However, after long term ageing these changes turn into chemical ones such as color change,40,41 surface crazing28,40–42 and hydrolysis. 28 All these phenomena may lead to substantial loss in properties and changes in structure 18 affecting afterwards the material’s service life. 22 This loss of the resulting properties especially mechanical ones results from the disruption and the weakness of the H-bonding network that is considered as the main structural characteristic of polyamide materials.43–45
Because it is one of the most important industrial polymers used in automotive industry especially for exterior body components, gears and under-the-hood components, many investigations were conducted so far on PAs ageing.5,18,29,46–49 Currently, because of their contact with ethylene glycol (EG) used for cars as a coolant fluid much attention has been devoted to study their ageing in glycol solution.2,32,50–53 Thus, numerous studies were investigated using many characterization techniques such as Gel Permeation Chromatography (GPC),50,54 Nuclear Magnetic Resonance (NMR), 54 Differential Scanning Calorimetry (DSC)50,54,55; X-ray,55,56 Dynamic Mechanical Analysis (DMA), 55 tensile55–57 Infrared Spectroscopy2,45,49 and Pyrolysis-gas chromatography/mass spectrometry (Py-GCMS) 57 in order to understand the effect of glycol on the bulk properties of studied materials. Nevertheless, and as it is considered as a crucial characteristic of materials, mechanical properties remain the most studied. Thus, it was noted a significant reduction of both stiffness and ultimate strength after conditioning. They reached the lowest values especially after 12 and 20 days (from 60 % to 80%), respectively. Regarding the strain at failure, it increases after 2 days of ageing than it decreases. 56 Hence, at early stage of ageing, the effect is reversible and becomes irreversible after long durations indicating the chemical damage as reported by. 23 This result was recently confirmed by Chekkour et al. 56 who reached to infer the significant and irreversible damages of glycol on PA66 and PA66/GF after 12 days of ageing at 130°C. Thus, a few cracks were observed that evolved to form a large crack network after 20 days of conditioning confirming the severe damage of studied materials.
In the same context, Hong et al. 50 noticed a loss of mechanical properties for Nylon 6,6 when it is repeatedly exposed to ethylene glycol (EG). Moreover, Lee and Kim 57 investigated the effects of monoethylene glycol (MEG) on the degree of hydrolysis of Polyamide 66 (PA66) and Polyamide 66/glass fiber (PA66/GF) composites after ageing at 130°C. According to them hydrolysis occurs for both materials and increases proportionally with the immersion time in MEG leading to a loss of mechanical properties. Regardless the conditioning duration, reinforced polymers remain less sensitive to hygrothermal ageing than unreinforced ones. Thus, the neat polymer undergoes a significant embrittlement after ageing which is not noticed for the reinforced as reported by Chekkour et al. 56 However, and despite the discrepancy of ageing conditions it was agreed that glycol ageing leads to irreversible changes that deteriorate PAs properties.56,57
As previously mentioned, hygrothermal ageing of Polyamide materials in water and in equi-volume mixture (50:50) of glycol/water was studied. Results prove the critical impact of both conditioning environments on the bulk properties of tested materials. Thus, a pronounced damages and subsequent decline in macroscopic properties especially mechanical ones was noticed. This impact was attributed to the significant polymer degradation (hydrolysis). Thence chemical degradation occurs, whatever the ageing bath, because of the combined effect of temperature and ageing duration especially for neat polymer. Nevertheless, for composite material the damage can be occurred even within a short duration of time due to the destruction of the integrity of interfacial adhesion between fibers and polymer as reported by Hong et al. 50
As can be noticed in literature, most relevant papers dealing with glycol ageing previously published are almost at the glycolis temperature or at a range temperature beyond 100°C. That is way the current experimental investigation is conducted. It aims to study the effect of hygrothermal ageing in a commercial mixture of Glycol Water on Polyamide (PA6) and Polyamide 6 reinforced with 30 % of short glass fibers (PA6GF30) properties. This bath is composed of 20% of glycol and 80 of water (20:80 GW) typically used by drivers in Tunisia, as antifreeze and an engine coolant. The ageing conditions chosen were 50°C, 70°C and 90°C for up to 80 days like those adopted in our previous work. 41 These temperatures are closer and beyond glass transition temperature but still lower than the ageing temperatures previously studied by.2,31,56,57 Accordingly, the effect of such conditions on mechanical and chemical structure of PA6 and PA6GF30 will be assessed. First of all we try to point out if the glycolysis process takes place for the adopted conditions. Then, a comparison between the obtained results with those reported by51,52,56 and those obtained in distilled water 41 will be established. Furthermore, and because the stability of the fiber/matrix interface is crucial for material long-term property retention, 58 this work aims to point out the effect of GW mixture on this region at different temperature.
Material and Methods
Materials
The studied materials in this study are a Polyamide 6 and a Polyamide 6 (ALYAMID) reinforced with 30 % of short glass fibers (ALYAMID C2122GF30), respectively. Polyamide 6 is a commercial product supplied by RADICI Chemicals. The glass fibers are from CPIC with an aspect ratio of about 450. (Fiber diameter and length are successively 10 lm and 4.5 mm). Silane was used as coupling agents for the glass fiber. Samples were injected molded using a Battenfeld injection molding machine in accordance with the ISO standard 527–2 type 1A (POLYMERES-SA Tunisia). Prior to testing, samples were dried at 70°C for 24 h and immediately stored in vacuum desiccators containing silica-gel to prevent any moisture.
Ageing Procedure
Ageing was performed at different temperatures; 50°C, 70°C and 90°C for almost 80 days (1920 h) in a mixture of 20% off glycol and 80% of water (GW). The choice of accelerated ageing is to reach result in a short scale of time.
Conditioning was conducted in glass jar containers in air circulating oven with the same conditions adopted in our previous works.40,41 Thus, samples were fully immersed, periodically wiped off using absorbent paper and weighted using a digital balance of 0.001 g accuracy. Ageing conditions were chosen in order to (i) point out the impact of temperature on the diffusion as well as the evolution of the bulk properties of investigated materials (ii) infer the similarity and/or the discrepancy of trends with distilled water bath previously studied.40,41
Both materials (PA6 and PA6GF30) were taken off twice for testing, at equilibrium state (moisture uptake is steady) and at the end of ageing (80 days).
The moisture uptake M (t) was measured through gravimetric method as following in equation (1)
Afterwards characterizations were then performed.
Characterization
Mechanical tests were carried out at room temperature using a Universal Testing Machine model WDW-E with a capacity of 10 kN. Tests were performed in accordance with ISO 527 procedure at a constant crosshead speed of 5 mm/min using a longitudinal extensometer with a gauge length of 50 mm. For each test, the average value of at least three specimens were used.
Afterwards, failure features of tested materials were coated with a thin layer of gold using a metallization device. Then, they were examined using a scanning electron microscopy (SEM VOVA NANO SE 450/650) operating under an accelerating voltage of 10 and 20 kV.
In order to assess the plasticization effect of GW ageing, Dynamic Mechanical Analysis (DMA) measurements were performed on 8000 Perkin Elmer analyzer at a heating rate of 3°C/min on samples with dimensions 51 × 10 × 4 mm3 between −50°C and 180°C (frequency scan: 1 Hz, ΔL = 5 μm). The glass transition temperature (Tg) was determined from the max of damping factor (tanδ) of tested.
The change of chemical structure was evaluated through infrared spectroscopy analysis using Agilent Cary 630 FTIR spectrophotometer. The scan of samples was conducted in the range from 4000 cm−1 to 650 cm−1 at room temperature
Each spectrum is the co-addition of 74 scans at 4 cm−1 resolution with a total measurement time of 30 s.
Results and Discussion
Mass Uptake
Change in Mass and Diffusion Coefficient
The change in mass of PA6 and PA6GF30 samples after immersion in GW, are displayed in Figure 1. Results show that the moisture uptake for both materials increases linearly with ageing time (first stage) until reaching a plateau indicating saturation (Figure 1). This saturation is reached after a fairly short time for both materials immersed at high temperature (Figure 1). For instance, the equilibrium state is attained approximately after 87 h of ageing at 90°C (Table 2). Nevertheless, these durations are commonly slowed down with the presence of glass fibers especially for samples aged at 50°C and 70°C. This confirms that the absorption mechanism even for glycol water ageing is strongly dependent on the ageing temperature as demonstrated by Thomason.
53
Furthermore, after saturation a slight decrease in the mass uptake after saturation is noticeable (Figure 1). The reduction reaches 6.4% and 7.3% for PA6 samples aged at 70°C and 90°C, respectively. Nonetheless, for PA6GF30 aged under the same temperatures, the decrease did not exceed 3%. This is attributed to the hydrolysis of the macromolecular chains in the polymeric matrix known as leaching phenomenon (Figure 1). This phenomenon remains barely visible for PA6GF30 samples than for PA6 ones aged at the same conditions (Figure 1). Change in mass versus square of time of (a) PA6 and (b) PA6GF30 aged in glycol water at different temperatures. Immersion duration (hours) of PA6 and PA6GF30 at saturation states for different ageing temperatures.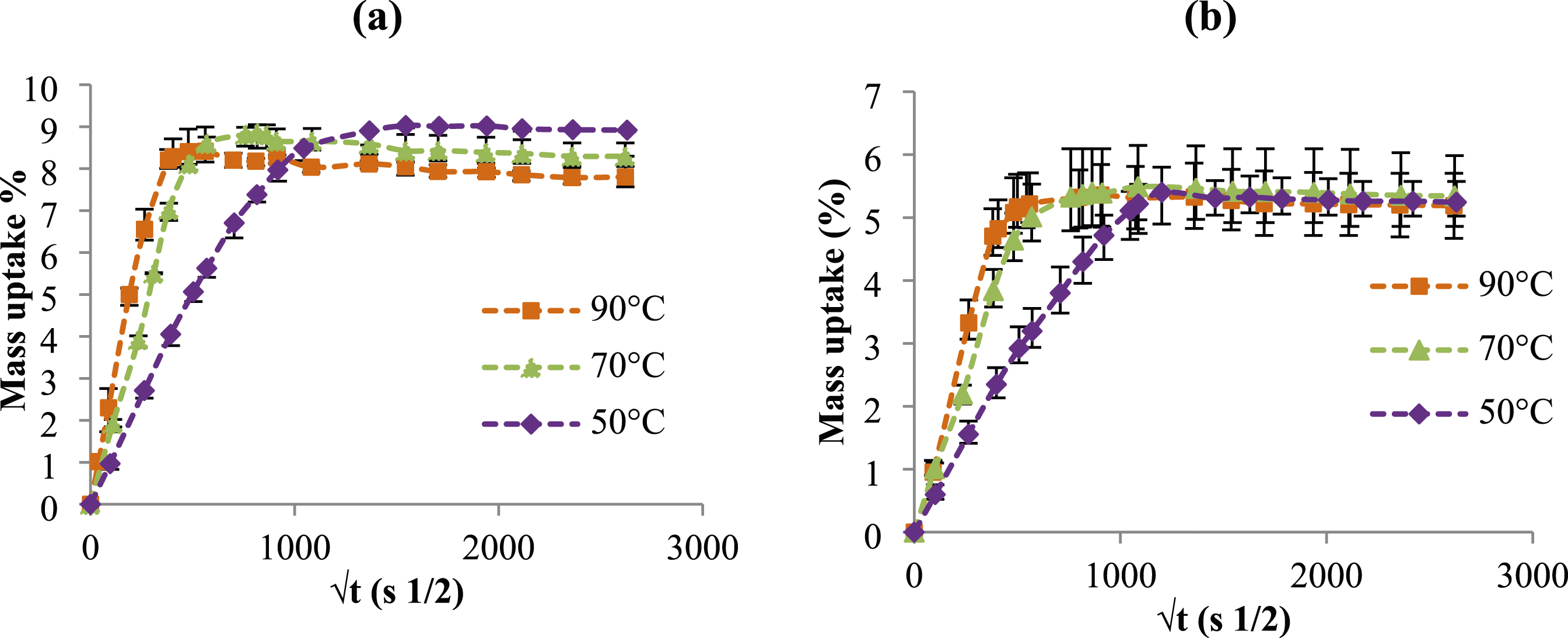

Regarding the ageing fluid, a comparison was established between results gathered in distilled water reported elsewhere40,41 and those in glycol water (Figure 1; Table 2). According to change in mass plots, it seems that the absorption mechanism in GW is somewhat similar to the absorption mechanism in distilled water. Nevertheless, for leaching phenomenon it was found that it is more noticeable for samples aged in distilled water than for those aged in glycol water. Thus, it reaches 20% in distilled water for PA6 samples aged at 90°C until it is only 7.3% in GW. Similarly, for PA6GF30 samples aged in GW, the mass loss varies according to the ageing temperature where the highest reduction values were measured for samples aged at 90°C. This decrease is limited to only 8% for samples aged at 50°C in distilled water although in glycol water it is almost absent. These results confirm that distilled water is the most aggressive ageing environment that may cause polymer degradation.
Mass uptake at saturation Ms (%) of PA6 and PA6GF30 in glycol water GW and distilled water DW 33 at different temperatures.
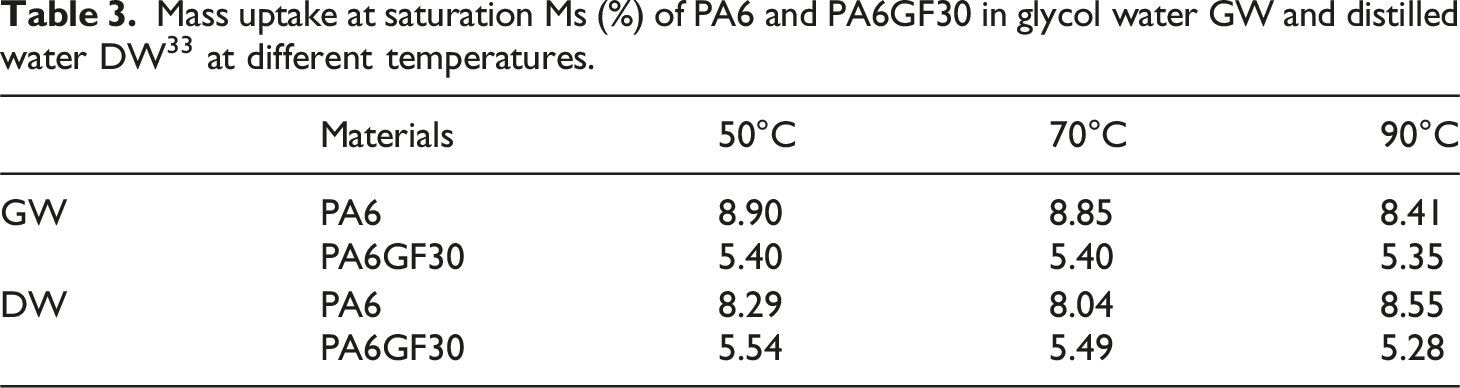
This finding assumes that the presence of glass fibers reduces the moisture uptake at saturation. For this reason, we try to measure the water uptake only in the composite matrix without taking into account the weight of the fiber using the equation suggested by Carrascal et al.
59
and Ledieu.
32
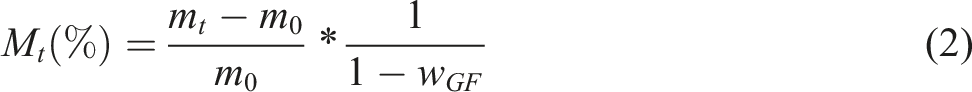
Results obtained show that the absorption curve relative to the matrix is always located below the polymer (PA6) independently of the aging temperature (Figure 2). Thus, the absorption mechanism is always hampered by the presence of fibers and is dependent on the contribution of the hygroscopic phase.52,53 Hence, the reduction of the volume fraction of the polymer (PA6) induces a reduction of the mass uptake. This result confirms that only the polymer is responsible for the absorption mechanism. Moreover, it can be obviously noted that the gap between both curves (i.e. polymer and matrix) decreases when ageing temperature increases. This finding is related to the leaching phenomenon that occurs at long term ageing which is attributed to the severe damage of studied polymer. Therefore, the GW diffuses in the interface region which may constitute a preferential location for GW accumulation. This result let us postulate that the long term ageing at elevated temperature in GW mixture affect the interfacial adhesion between fiber and matrix. Water absorption in PA6 (polymer), matrix and PA6GF30 (composite) after ageing in glycol water mixture at (a) 50°C, (b) 70°C and (c) 90°C for 80 days.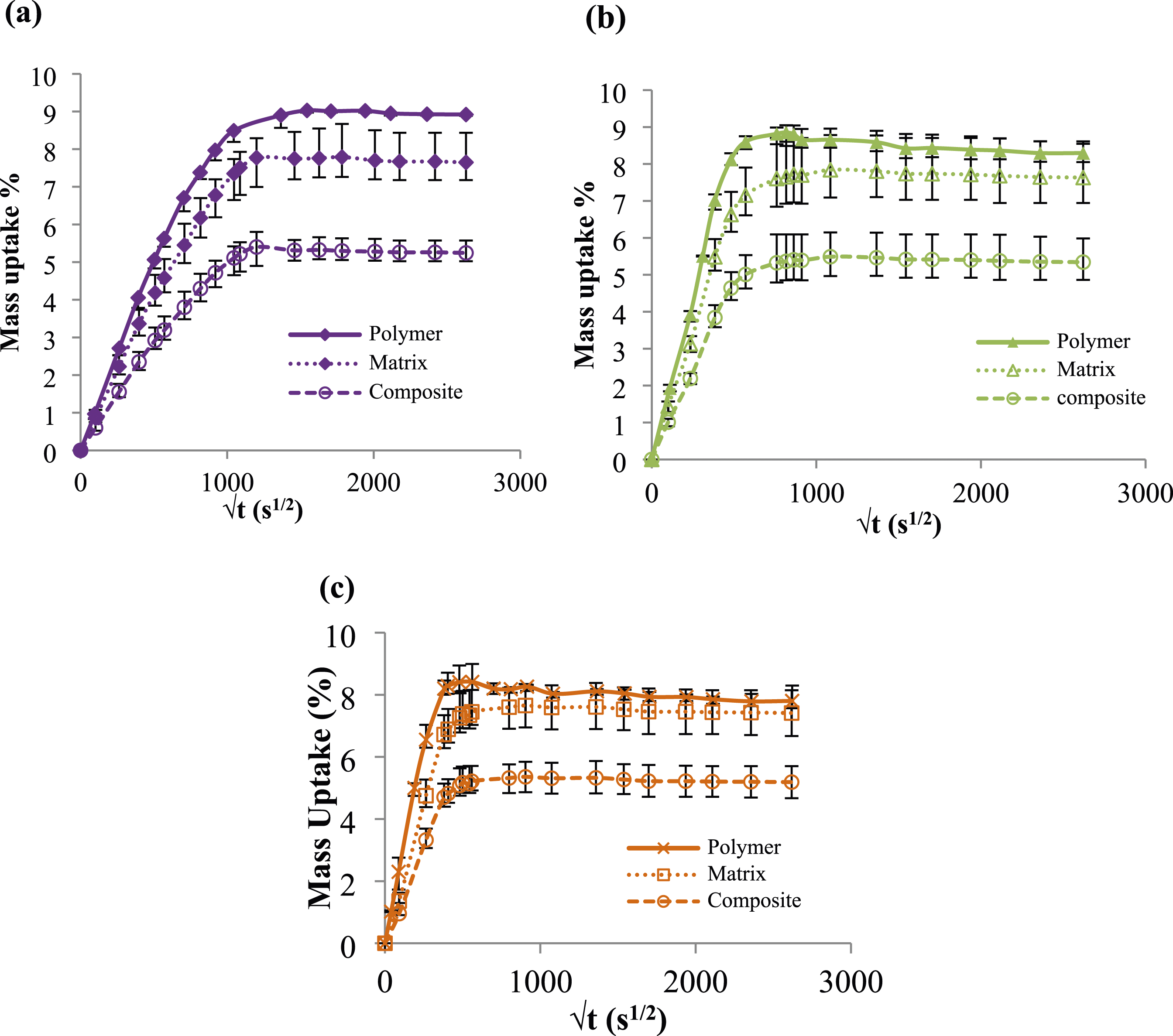
This tendency confirms that the polymer is the only contributor on the ageing process which constitutes the hydrophilic portion as reported by.51,52 Therefore, the presence of fibers restricts absorption 51 and induces a reduction in the moisture uptake.60,61
According to Figure 1, the glycol water diffusion initially follows a Fickian process. Therefore, the diffusion coefficient ‘D’ can be computed using the slope of the change in mass Mt/M∞ as a function of √t,
Results obtained show that for the same material the diffusion coefficient increases with to the ageing temperature (Figure 3) to reach the highest values for samples aged at 90°C (24.33 10−12 m2s−1 and 12.65 10−12 m2s−1 for PA6 and PA6GF30, respectively). However, for PA6 samples aged at 50°C, it is sixfold lower than those aged at 90°C (≈410−12 m2s−1). This finding confirms again that temperature plays the role of an activator for the absorption mechanism even in water-glycol as it was shown in the case of distilled water.
62
Moreover, this coefficient is greatly influenced by the presence of glass fibers in the polymer. Thus, for the same ageing temperature PA6 have the highest values in comparison with PA6GF30. This result confirms that glass fibers slow down the absorption process as found for distilled water.
51
Evolution of diffusion coefficient D with ageing temperature and immersion environment (DW: distilled water and GW: glycol water)
33
.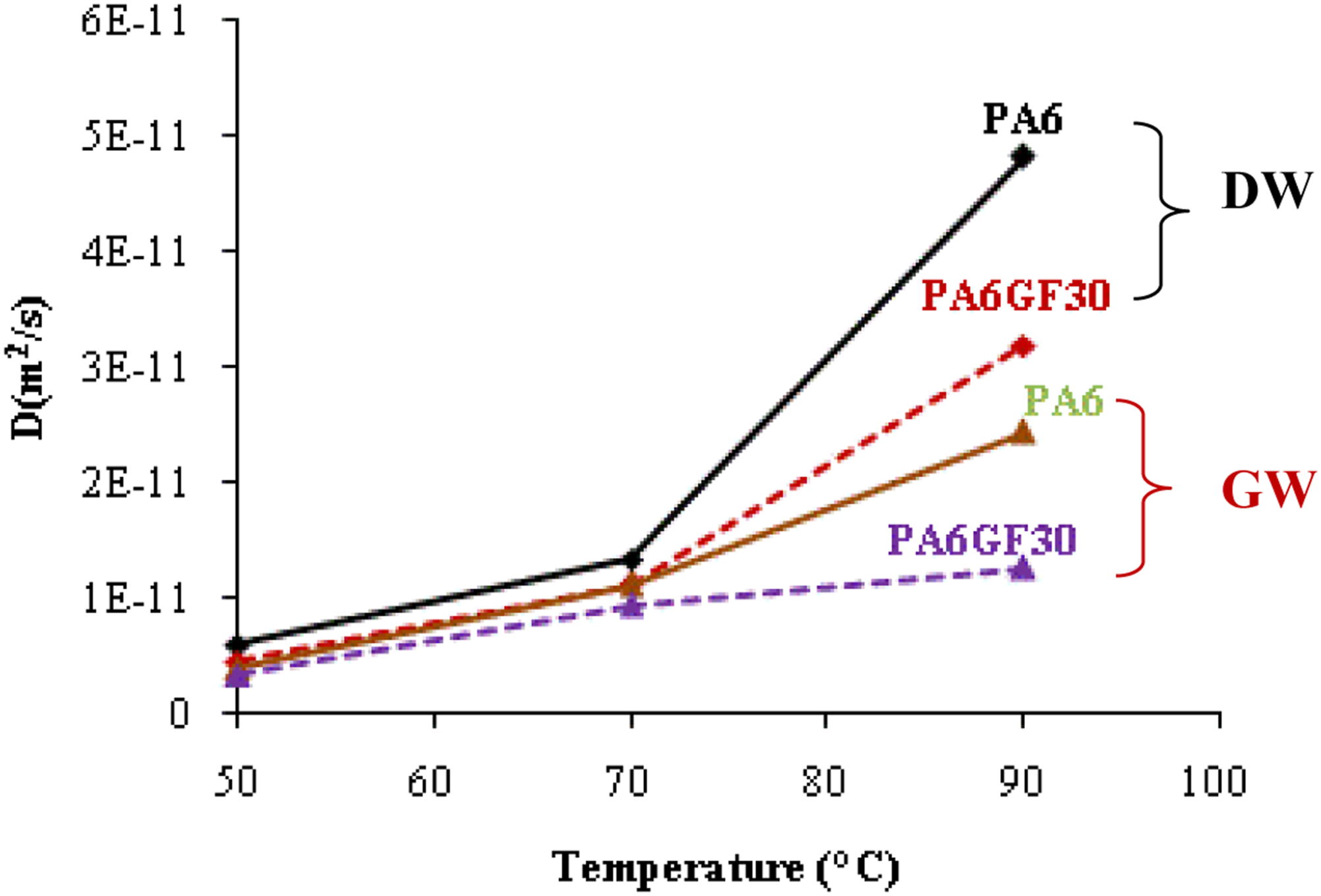
Moreover, it is noted that whether for PA6 or PA6GF30, the diffusion is slower in glycol water than during ageing in distilled water (Figure 3). Accordingly, this difference in diffusion behavior may be related to the larger size of the ethylene glycol molecules as reported by. 51 Nevertheless, contrarily to glycol, water molecules are polar and small in size, which may also contribute to diffusion kinetics. 32
Activation Energy
The diffusion mechanism in polymers and/or in composites is strongly dependent on the conditioning temperature. It is a process kinetically activated by temperature as reported by.62–64 As the absorption mechanism follows a Fick’s law and depends on the ageing temperature, the diffusivity is related to temperature by an Arrhenius law given by:
The activation energy can be calculated using the curve Ln(D) = f (1/T) plotted in Figure 4. We found Ea (PA6) = 44,24 kJ mol−1. This result is in good agreement with that found by Ledieu 2010 who found Ea = 48 kJ mol−1 for PA66 aged in an equal volume mixture of water and glycol (50:50) at 130°C. Estimation of the activation energy of the diffusion process in glycol water for PA6 (full line) and PA6GF30 (dashed line).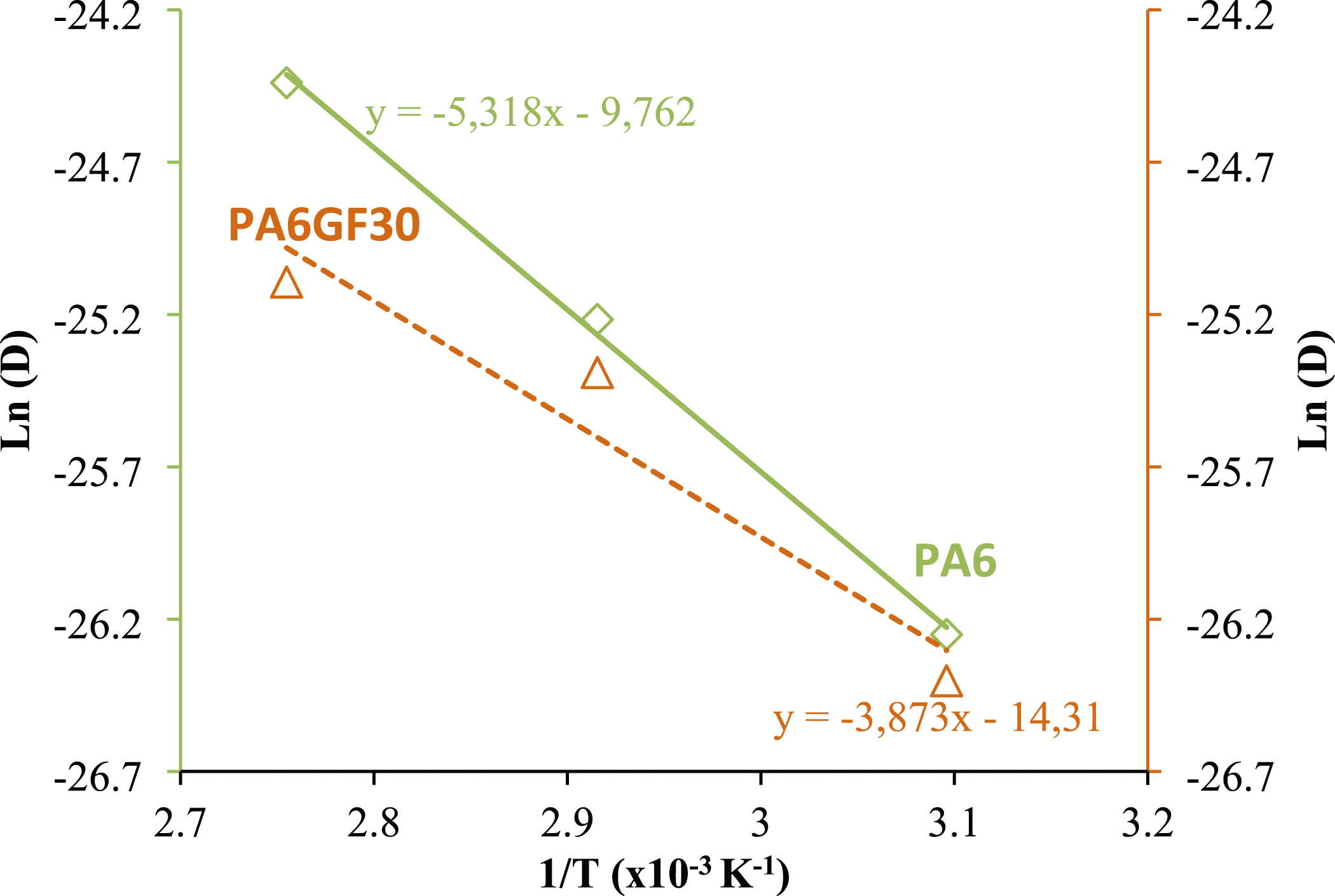
Nevertheless, for PA6GF30 samples, it was noted that the diffusivities show a non linear Arrhenius plot over the entire temperature range studied which make difficult to calculate the activation energy of composite material in this environment.
These measured values still lower than those found for PA6 and in PA6GF30 aged in distilled water 41 indicating that the glycol water has sufficient kinetic energy to break the chain bonds of investigated material and consequently form a new product.
Surface Damages: Color Change
As shown in Figure 5, contrarily to hygrothermal ageing
41
and glycol water ageing recently studied by,
56
the only visible damage caused by immersion in glycol water for 80 days only was the color change “no crack formation”. This change is barely visible after ageing at 50°C but it becomes more pronounced for higher ageing temperatures for both materials. This appearance may be the result of thermo-oxidation as observed in the case of distilled water. In order to explain this aspect, Grigg
65
has speculated that this yellowing is the result of the formation of either conjugated double bonds due to the consecutive reaction of azomethines, which gives rise to the chromophore that accounts for the observed yellowing of PA’s during oxidation as reported by Grigg.
65
Color change of (a) PA6 and (b) PA6GF30 after immersion in glycol water at different temperatures.
The comparison of both fluids shows that the ageing process in distilled water is beginning with yellowing at early stage of ageing40,41 and continuing up a slight blackening especially at 90°C after 80 days of ageing (Figure 6). Nevertheless, for those aged in glycol water, yellowing is detectable at the same ageing temperature for the same ageing duration (Figure 6). According to Dong and Gijsman
66
and Rudzinski and al,
24
this color change was detected for PA samples aged between 120 and 180°C due to the occurrence of thermal oxidation phenomenon. Nevertheless, in this work ageing was carried out at temperatures below 120°C which let us wonder if this color changes is an indication of thermo oxidation or not. Comparison of color change after long term ageing of (a) PA6 and (b) PA6GF30 in distilled water (DW) and glycol water (GW) at 90°C.
Infrared Spectroscopy Analysis
The infrared spectra were used to point out the chemical changes of the investigated materials that may occur after 80 days of immersion in glycol water at different temperatures (Figures 7 and 8). The superposition of all spectra of PA6 and PA6GF30 shows that most of the characteristic bands of the functional groups of PA6 and PA6GF30 are slightly shifted, indicating a possible interaction between the materials and the ageing environment (Figure 7(a) and (b)). Moreover, based on the previous studies and in order to monitor the spectral changes promoted by glycol water ageing, we focused our analysis on two intervals: from 2700 cm−1 to 3800 cm−1 and from 1640 cm−1 to 1840 cm−1 (Figure 8). According to these spectra, an increase of the absorption band located at ≈ 3400 cm−1 after 80 days in glycol water ageing for both materials was noticed. This band is assigned to the stretching vibration of free N-H groups (Figure 8(a) and (b)) as reported by.41,67,68 The appearance of this broad band corresponds mostly to the fluid (mixture of glycol and water) absorbed by the studied material as reported by32,54 when they studied a separated ageing of polyamide in glycol and water. Meanwhile, a noticeable wide band was detected on carbonyl species absorption area (1733 cm−1) for PA6 aged at 90°C for 80 days. This band is barely detectable for PA6GF30 aged at the same conditions with a slight increase of another peak located at 1716 cm−1 (Figure 8(c) and (d)). Infrared spectra of (a) PA6 and (b) PA6GF30 aged in glycol water at different temperatures for 80 days. Infrared spectra of (a,c) PA6 and (b,d) PA6GF30 aged in glycol water at different temperatures, from 2800 to 3600 cm−1 and (c) in the carbonyl area 80 days.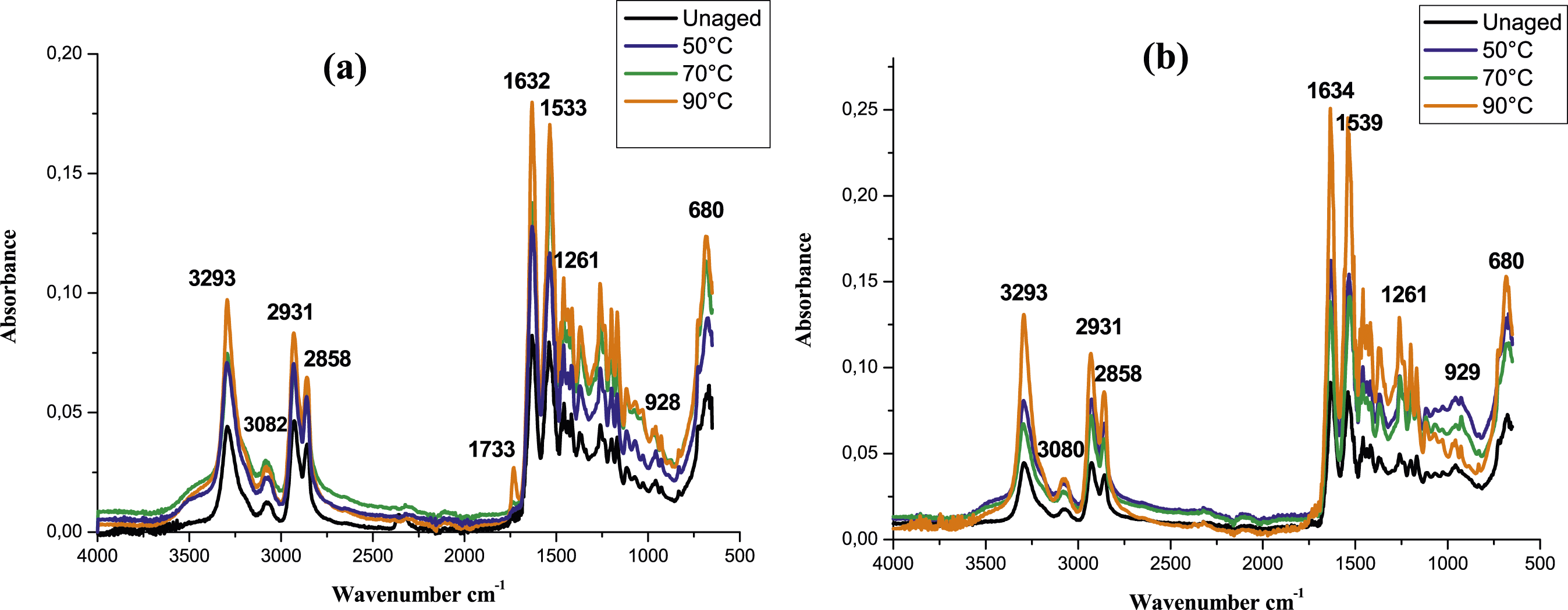
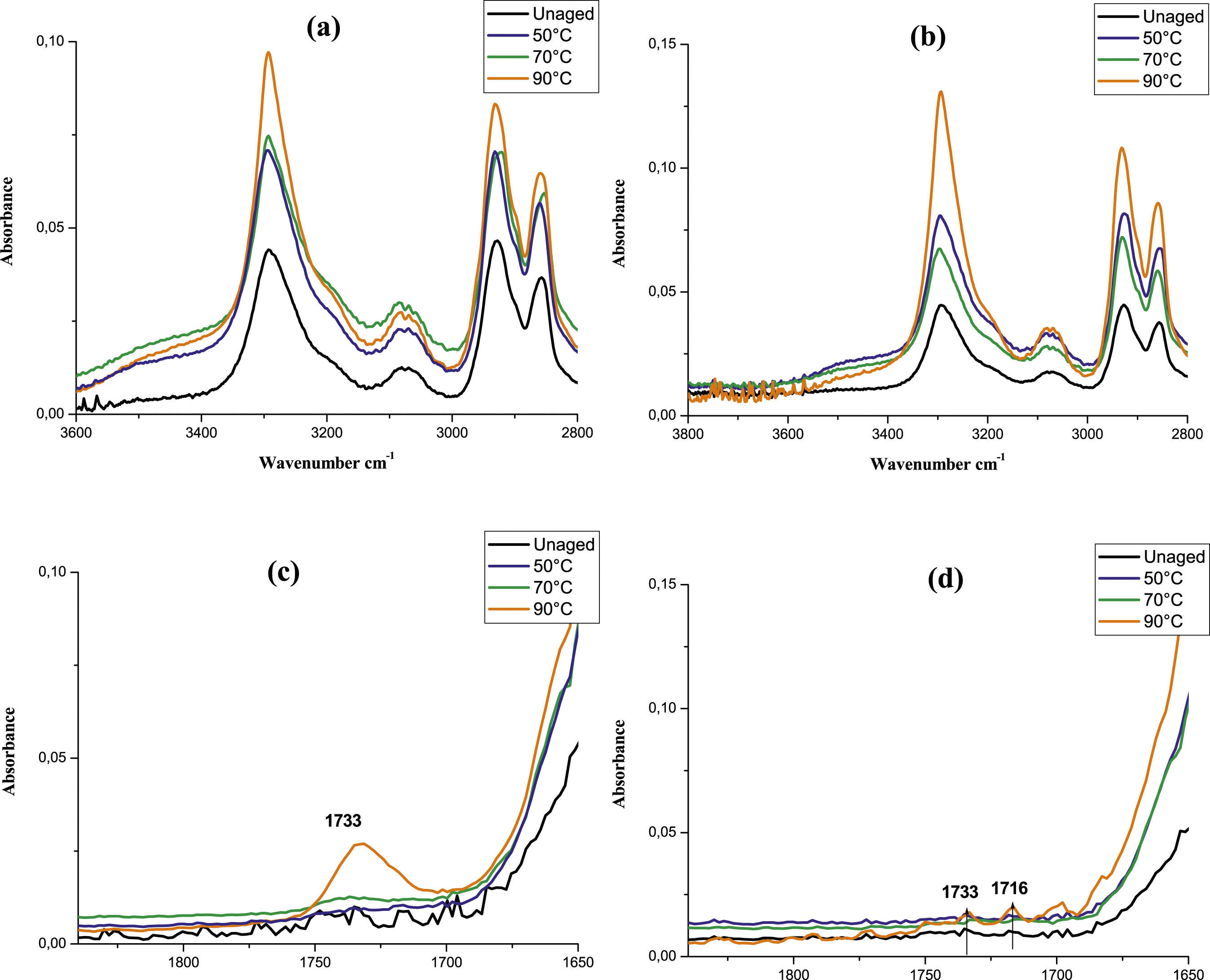
Does these species are related to a glycolysis reaction that PA6 undergoes during glycol water ageing?
In order to respond to this question, a comparison with previous investigations conducted in similar conditions or close to ours is crucial. Typically, according to literature leading with glycol ageing, the major byproduct resulting from glycolysis process is ester species (1734 cm−1).2,32 For instance, Kim et al. 54 confirmed the glycolis process of polyamide after few hours of glycol ageing at 275°C. Thus, they noted an ester bond newly formed which absorbance increases with glycolysis time. They ascribe this change to the occurrence of glycolysis reaction resulting from the interactions of glycol with PA6. In fact, the disruption of the bond between carbon and nitrogen of the amide portion of nylon 6,6 leading to the degradation of the polymer into oligomers having ester groups. 54 This finding is in good accordance with data. 2 Nevertheless, Hong et al. 50 and despite the long duration immersion, showed that Nylon 66 and Nylon 66 composite didn’t underwent a glycolysis process after 1000h of conditioning in aqueous Ethylene Glycol solution (50/50) at 108°C. The same result was found by Ledieu 32 when they studied the ageing of the same materials conditioned in the same bath at higher temperature (130°C) for 700h. For both experimental investigations, glycolysis assumptions were denied by the absence of ester band in the Infrared spectra after glycol water ageing at temperature beyond 100°C for more than 700 h. Recently, Lee and Kim, 57 proved that hydroxyl groups of monoethylene glycol (MEG) react with PA66 chains via hydrolysis process when they are aged at 130°C for long duration (1008 h). All these papers confirm that glycolysis process occurs at very high temperature and does not take place at 130°C.
The adopted temperature in our study remains below 100°C which discard the occurrence of glycolysis process in our conditions. Moreover, the appearance of such products was found in papers dealing with oxidation in dry47,48,65,66,69 and wet environment.41,68,70 Accordingly, the noticeable new peaks formed after glycol water ageing are certainly the result of oxidation process. Specifically, the former (1733 cm−1) is related to an ester where its absorbance remains more detectable for PA6 samples aged at 90°C than other samples (Figure 8(c) and (d)). However, the latter is attributed to the formation of diametric acids or ketones as reported by.
66
These species, known as oxidation products, suggest that studied material undergoes thermo-degradation inducing consequently yellowing with its extent depends on both parameters: temperature and time. This oxidation, which is barely detected for PA6 samples thermally aged at the same temperature (Figure 9), is a consequence of prevailing temperature and either water or glycol water mixture in the samples. Thus, wet conditioning induces thermal degradation as reported by Arash et al.
70
Infrared spectra of (a) PA6 and (b) PA6GF30 before and after thermal ageing for 80 days in the carbonyl area.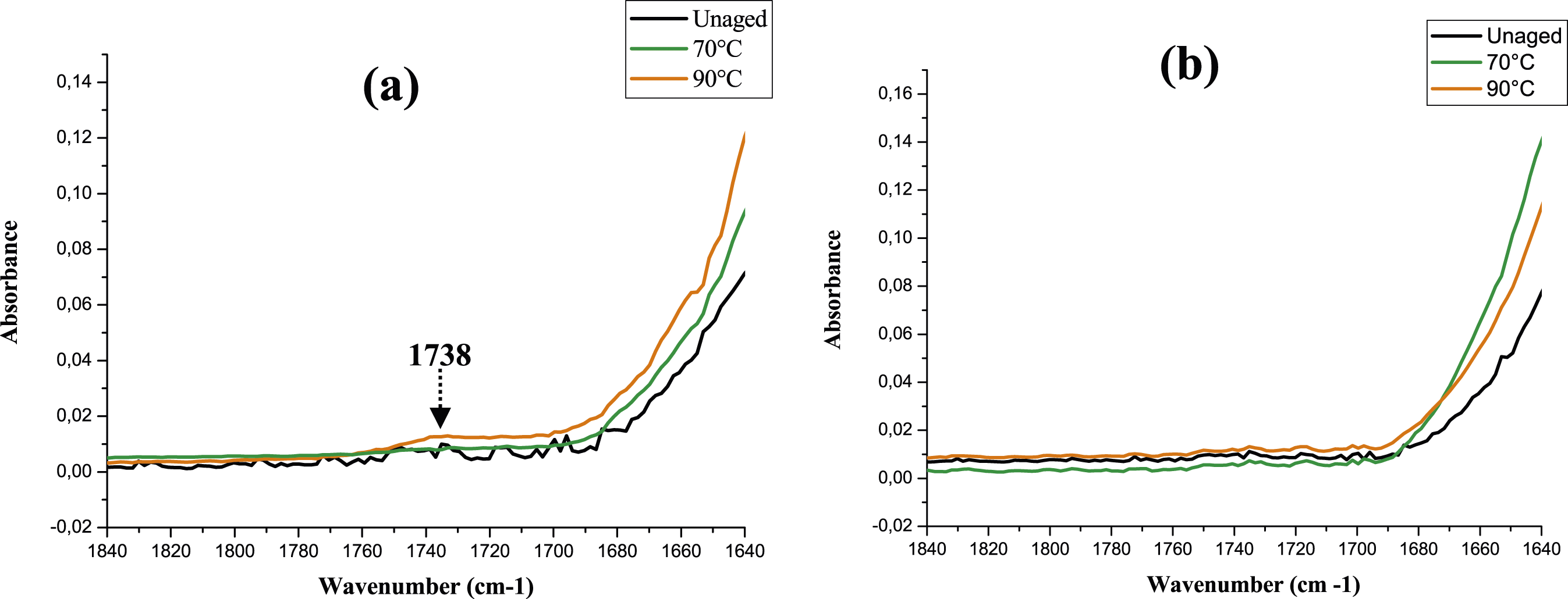
Regardless the ageing bath (glycol/water or water), PA6 remains more damaged40,41 especially at high temperature than composite material (PA6GF30). In fact, the polymer is the only contributor on the ageing process. Meanwhile, it can be assumed that the composite materials is highly resistant to external impact, due to the presence of glass fibers that may improve heat resistance as reported by Lee and Kim. 57
Accordingly, it is reasonable to assume that all these changes were chemical in nature and proved the degradation of investigated materials in glycol water bath.
Dynamic Mechanical Analysis (DMA)
DMA tests were carried out in order to highlight the impact of ageing in glycol water on the glass transition temperature Tg. The evolution of the damping factor tan δ for both materials is presented in Figure 10. As expected, the position of α transition, characteristic of Tg is affected after immersion in glycol water due to the plasticizing effect of water and/or glycol. Thus, it shifts towards lower temperatures as shown in Figure 10. Furthermore, it can be noticed the appearance of a new peak corresponding to the relaxation α′, the amplitude of which increases with the ageing temperature for both materials (Figure 10). This evolution was previously observed in previous studies40,41,71 for samples aged in distilled water. The decomposition of the α relaxation into two transitions which are attributed to water diffusion respectively within intra-spherulitic (strong linked water molecules) and inter-spherulitic (weak linked water molecules) phases.71,72 Damping peak (tanδ) an storage modulus of (a,c) PA6 and (b,d) PA6GF30 before and after ageing at 50°C, 70°C and 90°C in glycol water for 80 days.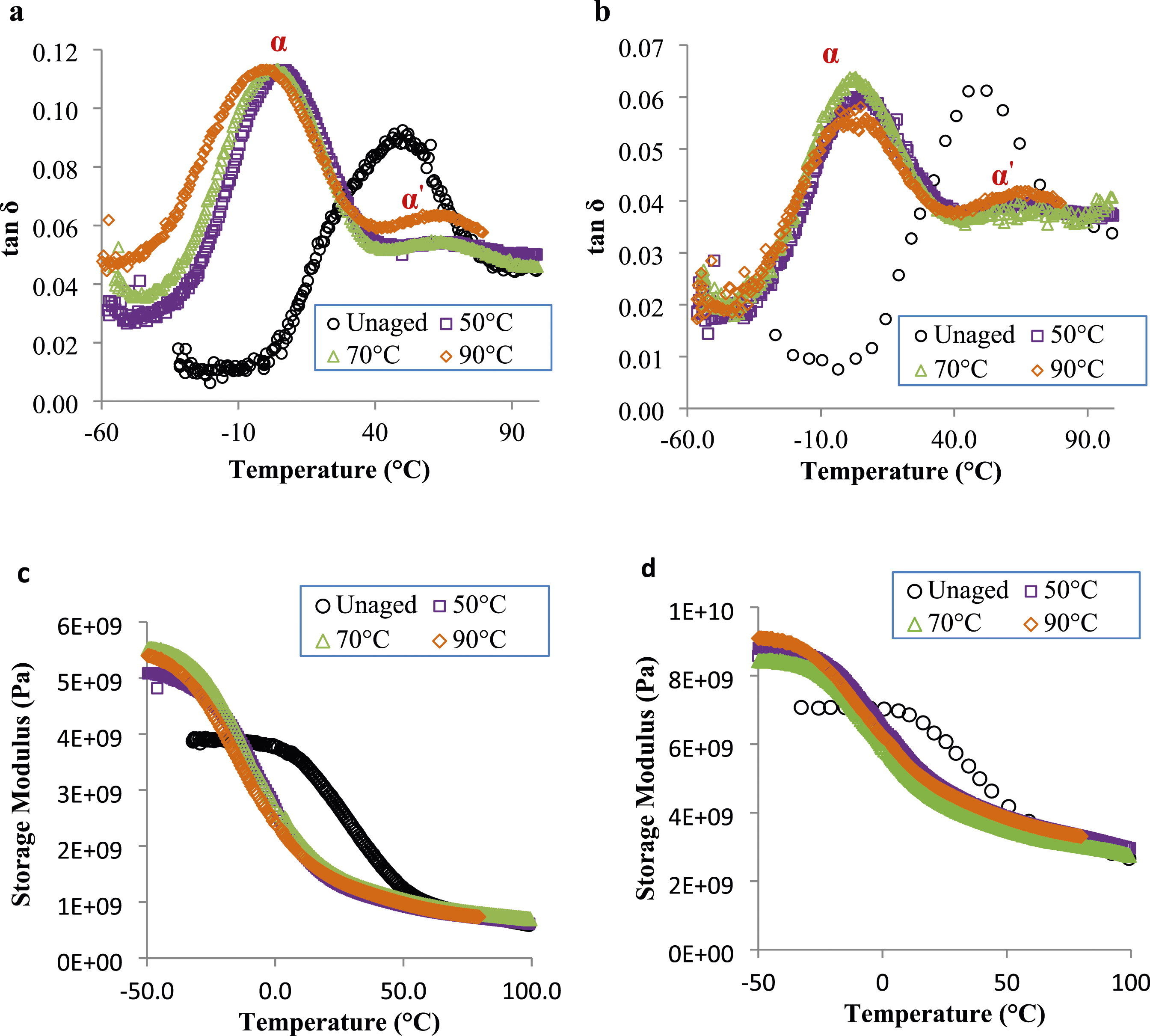
Evolution of glass transition temperature of PA6 and PA6GF30 before and after ageing in GW for up to 80 days.

Furthermore, it is noticeable that the peak related to PA6 samples aged at 90°C is relatively very large and begun at a different position. This is an indication of the existence of different chain lengths due to chain scission that was previously proved by the leaching phenomenon in the mass uptake curves (Figure 1). This hydrolysis process induces an increase of the local motions of macromolecular chains (Figure 10(a)). However, for PA6GF30, all curves are superimposed indicating that the chain mobility is similar for all temperatures (Figure 10(b)). Accordingly, PA6 samples especially those aged at 90°C undergo a significant chain scission.
Regarding the storage modulus (Figure 10(c) and (d)) a clear reduction of modulus in the 0–35°C temperature range with increasing ageing temperature is observed. This evolution is ascribed to the plasticization effect of polyamides due to the ingress of fluid into the polymer as reported by. 3 Simultaneously, an increase in the modulus in the sub-ambient temperature range can be noticed (Figure 10(c) and (d)). According to Thomason and Porteus, 3 this increase has been attributed to the ability of water to form bonds between chain segments at low temperatures which are sufficiently stable to produce an increase in modulus.
Mechanical Characterization
Mechanical characterization remains crucial to assess the impact of glycol water ageing on PA6 and PA6GF30 after 80 days of ageing.
Mechanical tests were conducted on samples before and after ageing (Figure 11). Samples were tested twice; after saturation and at the end of ageing (80 days). Evolution of (a) elastic modulus, (b) tensile strength and (c) elongation at break of PA6 at saturation state (full color) and after 80 days of ageing (Patterns fill) in glycol water at different temperatures.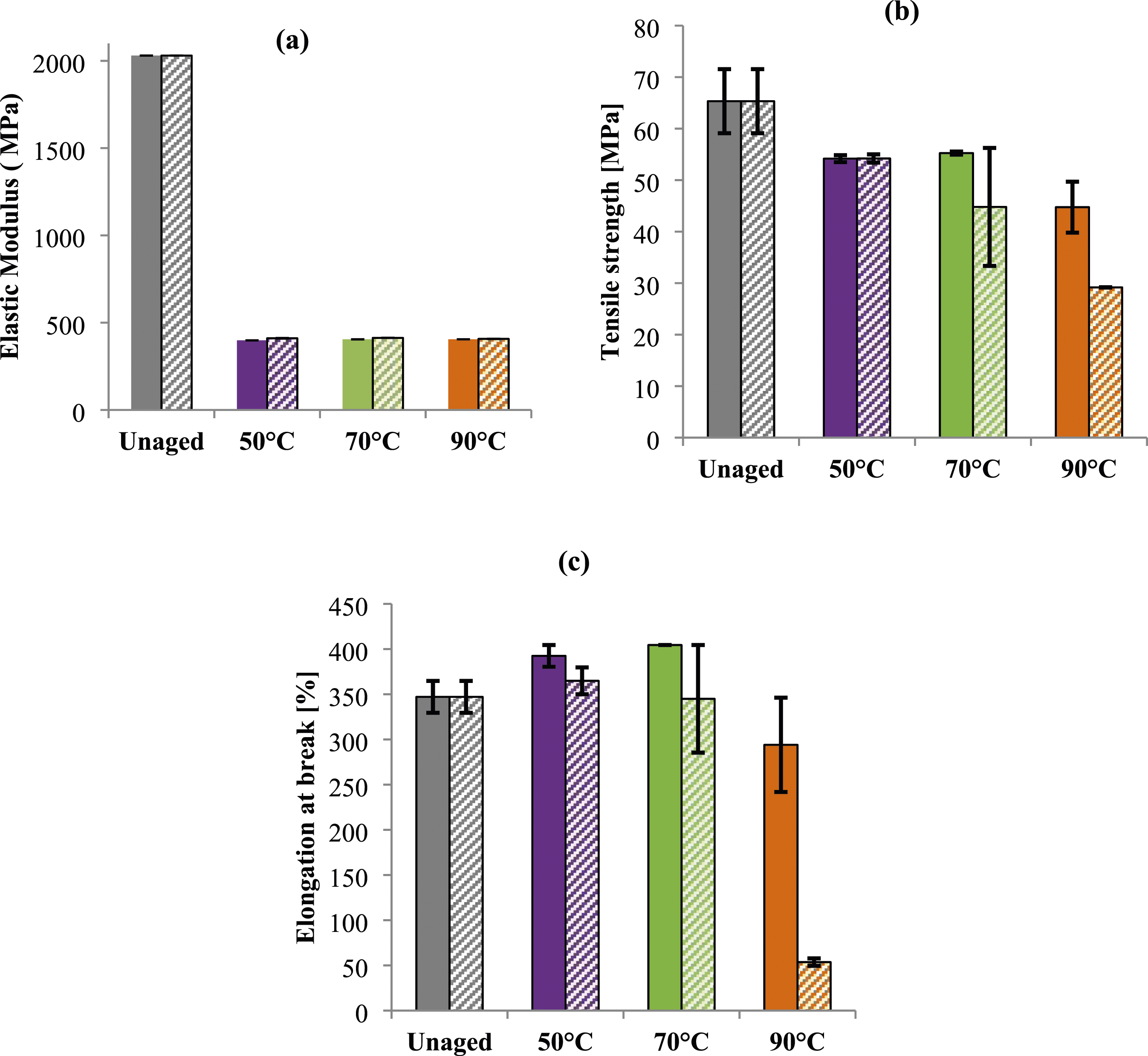
PA6 Samples
At early stage of ageing, regardless of the gap between getting saturation as shown in Table 2, the elastic modulus of PA6 samples falls catastrophically for all samples. This drop is estimated of roughly 80 % for all ageing temperatures (Figure 11(a)). However, for tensile strength the decrease depends on the conditioning temperature (Figure 11(b)). Thus, the reduction measured attains the lowest values (28%) for samples aged at 90°C (Figure 11(b)). With regard to the loss measured for PA6 samples aged at 90°C, it may be reasonable to assume that high temperature may act like long duration of conditioning. Thus, it triggers its degradation even for short duration (87 h), leading as a consequence to a significant loss of stiffness and strength. Nevertheless, the elongation at break was less affected (Figure 11(c)). In fact, PA6 behavior remains ductile and no significant embrittelment was noticed (Figure 11(c)). This result can be explained by the interaction between the water and the amide groups of the polymer proved by infrared spectroscopy. Solvent molecules can fit into the molecular network by replacing or breaking existing hydrogen bonds. According to the analogy of such interaction described by Chekkour et al., 56 Obeid, 73 Vega et al. 74 and Lim et al. 31 water can interact with polymer by forming either a single hydrogen bond “weakly linked water molecule” or two hydrogen bonds “firmly bound water”. The former is obtained when two molecules of water form bridges between the already hydrogen-bonded of two polyamide molecular chains. 22 Nevertheless, the latter is obtained when a water molecule forms double hydrogen bonds with two carbonyl groups. 31 In the same context, Hong et al. 50 showed that the glycol water ageing induces a destruction of the intra and intermolecular hydrogen bonding. This interaction induces a loss of the structural strength of the polymer and as a consequence a decline of both stiffness and strength due to the plasticizing effect of the conditioning bath.
After 80 days of ageing, the decrease of elastic modulus remains almost the same for all temperatures (Figure 11(a)). Thus, acting as a plasticizer GW can disrupt the chain polymer interactions which was proved by the decrease of the glass transition temperature previously observed. Therefore, regardless the ageing temperature, the overall stiffness of the material is reduced. Meanwhile, the tensile strength decreases progressively with ageing temperature and reaches 28% and 53%, at 70°C and 90°C, respectively (no change was observed for samples aged at 50°C) (Figure 11(b)). This reduction remains less pronounced in comparison with Elastic modulus especially for temperature close to the glass transition temperature. This finding can be explained by the fact that the polymer chain’s ability to resist deformation and breakage under stress are slightly reduced after ageing at low temperature due to glycol molecules. This ability disappears progressively at high temperature (90°C) confirming the obtained results.
Regarding the elongation at break, a loss of ductility was detected confirming the occurrence of irreversible damage. Thus, a catastrophic drop was measured for sample aged at 90°C estimated by 85% in comparison with unaged ones (Figure 11 (c)). This finding confirms the assumption of high ageing temperature. Thus, for the same ageing duration (80 days), the noteworthy reduction was measured for samples aged at 90°C. However, for other ageing temperatures (50°C and 70°C) the elongation at break remains almost steady and samples keep a ductile behavior (Figure 11(c)). This can be explained by the fact that, despite the hardly detectable interactions between the polymer and the hydroxyl roots of the alcohol function in the glycol (absence of the alcohol function of glycol in the infrared spectra previously presented), the presence of 20% of glycol in the bath reduces the damages of tested materials that exhibit a rubbery behavior as reported by Chekkour et al.
56
According to them, the interaction between the glycol and the polymer leads to an increase of the deformability due to the increase of the distance between macromolecular chains and the subsequent free volume.
56
Therefore, and contrarily to water ageing results recently published in our previous work,40,41 the effect of glycol water remains physical in nature for samples aged at relatively low temperatures (50°C and 70°C) confirming results previously discussed. This result was confirmed by SEM observations of failure features of PA6 after ageing. Thus, after immersion in glycol water at 50°C for 80 days, (Figure 12) the presence of fibrils with a rough surface are discernible highlighting the plastic deformation after glycol absorption. Nevertheless, when the ageing temperature increases (70°C), the surface remains rough, and the fibrils disappear progressively at 90°C and the surface becomes quasi-smooth (Figure 12) testifying that at high ageing temperature for long duration the glycol water effect is no longer physical in nature and failure occurs without necking. Thus, the entanglement network responsible for the plastic deformation disappears after ageing and, the Van der Waals interactions are the only intermolecular forces (for cohesion) as explained by.23,47 Indeed as shown by Laügt et al.
55
that the embrittling of the polymer was related to the reduction of the molar mass. SEM observations of PA6 after glycol water ageing at different temperatures for 80 days.
Accordingly, time and temperature are crucial parameters in the environmental degradation of polymers. 74 Thus, an increase in these parameters leads to a faster degradation rate.45,56 Therefore, similarly to water ageing, conditioning at high temperature for relatively long durations induces a chemical degradation leading to a catastrophic loss of mechanical properties.
PA6GF30 Samples
For PA6GF30 samples, the loss of both stiffness and strength occurs at early stage of ageing and remains till the end of ageing for all ageing temperature (Figure 13(a) and (b)). This loss resulted from the destruction of the interfacial zone between glass fibers and the matrix as reported by.5,7,41,50,56 The stability of this region is crucial for the effort transmission from the matrix to the fibers. After ageing and after the first hours of immersion as previously reported by Hong et al,
50
this adhesion is weakened due to glycol water absorption. Qualitatively the degree of this bad adhesion was assessed by the change noticed after ageing (Figure 14). Thus, the surfaces of the glass fibers initially covered by the polymer become smooth as shown in Figure 14 with the presence of stretched fibrils for all the fracture surfaces. These features testify the significant reduction in interfacial bonds between fibers and matrix after glycol water ageing. This reduction results in a progressive loss of adhesion which is extremely affected whether after immersion in distilled water or in water-glycol as reported by.
32
Consequently, once this adhesion is affected and regardless of the ageing temperature, the load transfer efficiency will be reduced and a drop in mechanical properties will be assessed.7,50,57 Evolution of (a) elastic modulus, (b) tensile strength and (c) elongation at break of PA6GF30 at saturation state (full color) and after 80 days of ageing (Patterns fill) in glycol water at different temperatures. SEM observations of PA6GF30 after glycol water ageing at different temperatures for 80 days.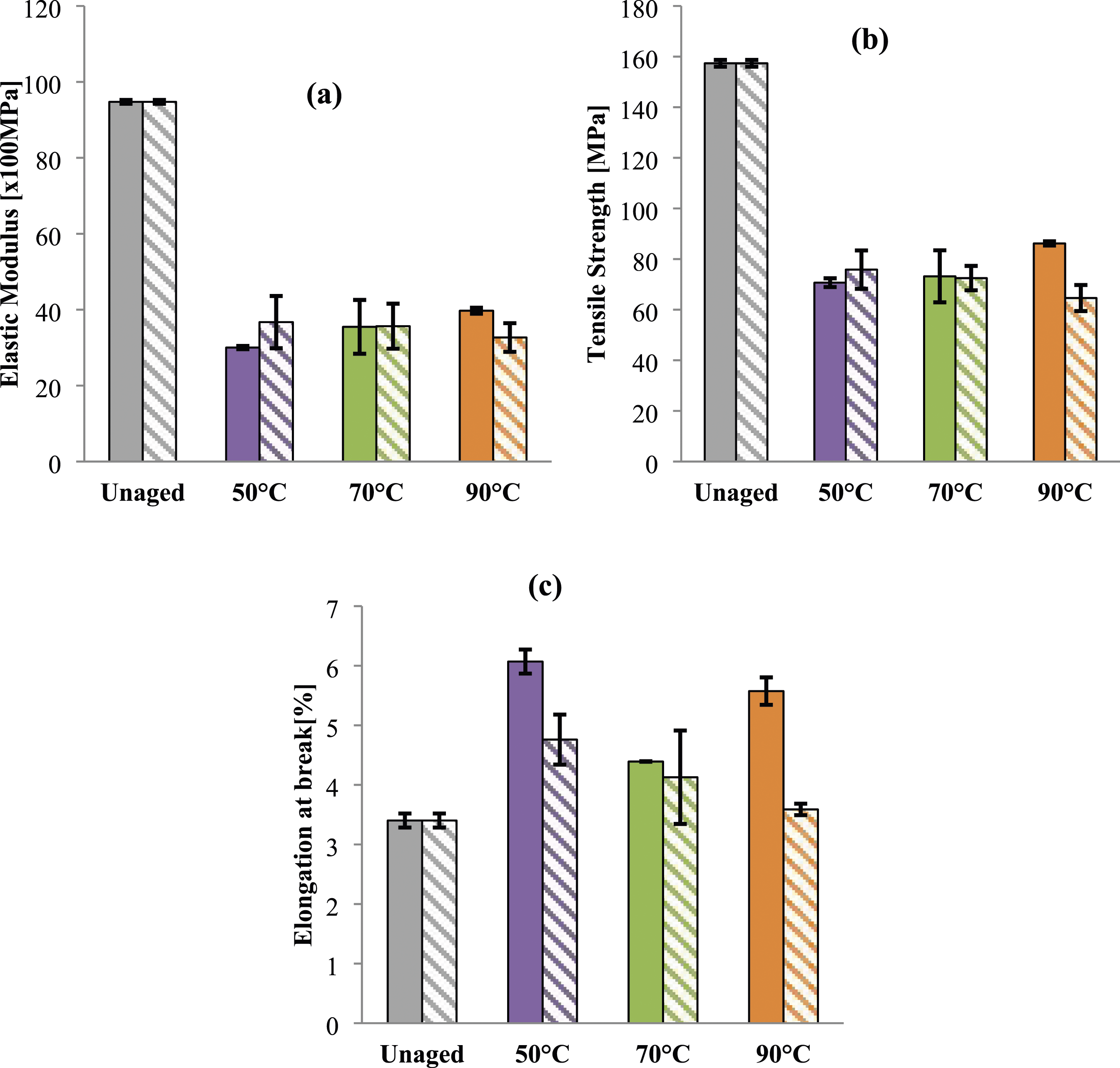

Meanwhile, at 50°C, a slight increase of elastic modulus and tensile strength was observed (Figure 13(a) and (b)). This let us assume the occurrence of changes in the crystalline structure as reported by Laügt et al. 55 According to them, these changes concerned especially the lamellae thickness that induces an increase in the stiffness of the tested material. On the other hand, an increase of elongation at break was noticed at early stage of ageing (Figure 13(c)) and it decreases progressively with ageing temperature after 80 days due to the plasticizing effect. This result is in accordance with previous data.46,50,56,75 Moreover, the absorption of the fluid swelling effect of PA matrix can induce a mismatched fiber–matrix volume expansion creating consequently a residual stress, which in turn can reduce the fiber–matrix interfacial properties as reported by Autay et al. 5
Conclusions
This experimental investigation deals with the impact of glycol water ageing on the bulk properties of PA6 and PA6GF30 under different temperatures for 80 days. After long term ageing, hydrolysis of the macromolecular chains in the polymeric matrix known as leaching phenomenon was noticed. This phenomenon remains pronounced for PA6 samples than for PA6GF30 ones aged at the same conditions. Meanwhile, the plasticizer effect of glycol water ageing was proved by the significant drop in Tg. The harmful effect of such fluid on the investigated materials was proved by the overall changes observed for both tested materials especially at high ageing temperature. This effect was detected from the first hours until the end of the immersion period. Thus, a catastrophic drop of mechanical properties of PA6 was measured with a significant embrittlement. For composite material the reduction of mechanical properties results in a progressive loss of adhesion which is extremely affected after ageing. Thus, once this adhesion is affected the load transfer efficiency will be reduced and a drop in mechanical properties will be assessed. All these changes confirm the occurrence of irreversible damage testifiying that at high ageing temperature for long duration, the glycol water effect is no longer physical in nature and a change in chemical structure takes place. This damage was ascertained by Infrared Spectroscopy analysis that pointed out the appearance on ester and ketone species. These species, known as oxidation products, suggest that studied material undergoes thermo-degradation.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.