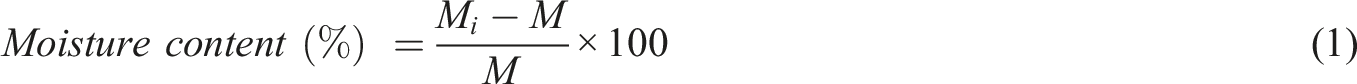Abstract
Multi-component nanocomposites based on chitosan, cellulose/chitosan nanofibers, and essential oils were developed in this study through solution casting technique. Both surface and structural properties of the nanocomposites were characterized. Surface morphology results revealed that the chitosan nanofibers were more localized at the films’ surface than the cellulose nanofibers, leading to enhanced hydrophobicity (contact angle = 95°). Moreover, the essential oils were distributed as micrometer-scale oil domains at the films’ surfaces, making them heterogeneous, leading to a much more hydrophilic character (contact angle = 30–36°). X-ray diffraction results revealed the disrupting role of nanofibers on the crystalline structure of chitosan; however, upon the addition of essential oils, the crystalline network was less affected than the pure chitosan film. The enhanced stiffness of the films in the presence of nanofibers was demonstrated by dynamic mechanical analysis. In contrast, the addition of essential oils highly reduced the stiffness, making the films much more flexible, which could benefit food packaging applications. Antibacterial properties were highly increased in the presence of essential oils, and the barrier properties were also improved, especially by the use of chitosan nanofibers. In conclusion, the developed nanofiber-loaded chitosan films could be considered innovative packaging to promote food conservation.
Introduction
Macro-plastic pollution resulting from food packaging has been a crucial threat to the environment during the last few decades, especially since the macro-plastic is degraded to micro-particles and nano-particles. 1 One of the most effective solutions for this problem is biopolymers.2,3 Despite being biodegradable, biopolymers could also improve the food quality, which leads to extending their shelf life through impeding the microbial growth in the food product.4–7 As one of the most well-known members of the biopolymer family, chitosan presents outstanding properties due to its biodegradability, antimicrobial activity, biocompatibility, non-toxicity, and film-forming ability.8–10 One of the major drawbacks of chitosan is its relatively high water vapor permeability (WVP) due to its intrinsically hydrophilic character. To solve this problem, hydrophobic materials such as lipids were suggested to be introduced to chitosan. 11 One of the well-known lipids, widely used in chitosan-based formulations, is the essential oils that could impart functional properties to the chitosan-based films and coatings.12–15 One of the properties that essential oils add to the chitosan films is the increased antibacterial activity and improved barrier properties. 16 Thyme (Thymus vulgaris) is a species from the Lamiaceae family with potent antimicrobial and antioxidant properties. Its essential oil has been extensively utilized in edible films and coatings. 17 Also, from the Lamiaceae family, Satureja (Satureja mutica) essential oil with strong antibacterial properties has been less studied in polymeric films designed for food packaging applications. 18
Cellulose nanofibers possess remarkable optical, mechanical, and barrier properties, and thus, their introduction to the polymer matrix could lead to highly improved mechanical, thermal, and permeability properties.19,20 Cellulose nanofibers consist of alternating crystalline and amorphous domains with a diameter of around 20 nm and several micrometers in length. Cellulose nanofibers are usually produced via disintegration of wood pulp through mechanical pressure processes. 21 Due to their good barrier and mechanical properties, cellulose nanofibers have recently been added to edible films and coatings. 22 For instance, Zhao et al. 23 have used cellulose nanofibers as reinforcing additives for chitosan/alginate films. The addition of cellulose nanofibers improved the mechanical properties and WVP of the films. In another research, cellulose nanofibers were used to reinforce papaya puree films and led to enhanced mechanical and thermal properties. 24 Chitosan nanofibers have also been widely used in the design of polymeric nanocomposites. In the current study, both cellulose and chitosan nanofibers were individually and concurrently added to the chitosan films, and the physicochemical and surface properties were characterized through scanning electron microscopy, X-ray diffraction, UV-vis analysis, ATR-FTIR, differential scanning calorimetry, dynamic mechanical analysis, wettability, and finally, antibacterial properties. Moreover, a hybrid of Thyme/Satureja essential oils was also added to the formulation of nanocomposite films. The effect of concurrent use of nanofibers and essential oils was investigated. The developed multi-component films exhibited promising potential to be used in food packaging applications due to their biodegradability, antibacterial activity, low water vapor permeability, and being edible.
Material and methods
Materials
Chitosan with a molecular weight of 250,000 g/mol, a degree of deacetylation of 80%, and a viscosity of 500 cPs was supplied by Sigma Aldrich (USA). Glacial acetic acid, Tween® 80, and glycerol were provided by Sigma Aldrich (USA) and used as received. Thyme and Satureja mutica essential oils were purchased from Pars Afra Darou Co. (Tehran, Iran) and Barij Essence (Kashan, Iran), respectively. Chitosan nanofiber aqueous gel (2.5% w/v, 85% degree of deacetylation) with nanofiber diameter of 40 nm and cellulose nanofiber aqueous gel (2.5% w/v) with nanofiber diameter of 35 nm were kindly supplied by Nano Novin Polymer Co. (Gorgan, Iran) (Supplement Figures S1 and S2).
Preparation of films
Chitosan acidic solution with a concentration of 1% (w/w) was prepared by adding 1 vol% acetic acid into the solution, followed by the addition of glycerol (30 wt% with respect to chitosan). Finally, the solution was cast in plastic plates and left to dry at room temperature for 48 h. In the case of nanofiber-loaded films, after achieving the chitosan solution, different concentrations of cellulose and chitosan nanofibers (2 and 4 wt%) were individually added to the solution and magnetically stirred for 6 h to attain a homogeneous suspension. This series of samples were coded as CeX and ChX in which Ce and Ch account for cellulose and chitosan nanofibers, respectively, and X denotes their concentration (2 and 4 wt%). In the case of essential oil-loaded films, the nanocomposite suspensions were prepared upon the addition of nanofibers, and then, the mixture of essential oils (1% w/w) with Thyme: Satureja ratio of 1:1 was added to the suspension. To better dispersion of the oily phase into the chitosan matrix, a proper surfactant (Tween 80) was added to the suspension (20% with respect to the essential oil content). Afterward, the whole mixture was homogenized by Ultra-Turrax (IKA T25-Digital Ultra-Turrax, Staufen, Germany) at 7000 rpm for 3 min, then degassed under vacuum for 10 min to remove all the bubbles. In one sample, the hybrid of nanofibers (2 wt%) was also utilized to formulate the nanocomposite. This series of samples were coded as Ce-ST, Ch-ST, CeCh-ST, in which S and T represent Satureja and Thyme essential oils.
Characterization
Scanning electron microscopy (SEM) was conducted on a digital SEM (VEGA//TESCAN instrument, Czech Republic). X-ray diffraction (Siemens D5000 system equipped with a copper target, Kα radiation source: λ = 1.5406 Å) was employed to monitor the crystalline structure of chitosan. UV and visible light absorption were evaluated using a PerkinElmer Lambda 365 Spectrophotometer (Waltham, MA, USA) with 1 cm cell quartz. ATR-FTIR was carried out on a Bruker IFS 66 spectrometer (Bruker, Germany) equipped with a SPECAC GOLDEN GATE diamond ATR accessory. Dynamic mechanical thermal analysis (DMTA) was performed by a STARE SYSTEM DMA instrument (METTLER-TOLEDO, Switzerland) in a bending mode with the frequency of 1 Hz, the temperature range of 10–150°C, and the heating rate of 10°C.min−1. To determine the glass transition temperature of the films, differential scanning calorimetry (DSC) experiments were conducted on a Mettler TA 4000 thermal analysis system (Greifensee, Switzerland). Aluminum pans containing 5–10 mg of samples, were heated from room temperature to 350°C at a heating rate of 10°C.min−1. A video-based contact angle measurement system (OCA 15, DataPhysics Instruments GmbH, Filderstadt, Germany) was employed to evaluate the wettability of the films. The moisture content of the films was measured by determining the weight loss of the samples (20×20 mm2) upon drying in an oven at 110°C to reach the constant weight based on the following equation:
Antibacterial analysis
Escherichia coli (E. coli) (PTCC 1330) cells were provided in the freeze-dried form from Persian Type Culture Collection (PTCC, Tehran, Iran). The bacteria were first cultured in a flask with 9 mL of Lauria Bertani broth followed by incubation at 37°C and oscillation at the frequency of 200 rpm for 18 h. After that, 100 µL of the culture suspension (10
5
CFU/mL) was spread on the surface of Mueller Hinton agar plates. Films were cut with the area of 100 mm2 and then placed on the agar plate, which was incubated at 37°C for 24 h. Finally, the plates were examined for a zone of inhibition (ZOI) by measuring the total diameter of the inhibition zone, including the film. To perform the colony counting experiment, the samples were removed, washed with PBS, and then placed in 0.5 mL TSB, followed by ultrasonication for 10 min, and finally, serially diluted. The number of survival microorganisms was attained by counting the colonies. The bacteriostatic reduction rate (BR) was calculated using the following relation, where B and A are the bacterial colonies of the chitosan film and the composite film, respectively.
Results and discussion
SEM analysis
The surface morphology of the prepared edible films was investigated by SEM analysis, and the results are shown in Figures 1 and 2. The effect of cellulose and chitosan nanofiber inclusions is studied in Figure 1. The low magnification images shown in the top row of Figure 1 indicate the difference between the samples loaded with cellulose and chitosan nanofibers. The incorporation of chitosan nanofibers led to the formation of much smoother and more uniform surface morphology compared with cellulose nanofiber-loaded samples. This difference could be ascribed to the higher compatibility of the chitosan matrix with the chitosan nanofibers. Moreover, no discernible changes could be detected between 2 and 4 wt% of both the added nanofibers indicating that the surface structure is not very sensitive to the concentration of nanofibers. Surface morphology of (a,b) Ce2, (c,d) Ce4, (e,f) Ch2, and (g,h) Ch4 at magnifications of 2000 and 60,000×. Surface morphology of (a,b,c) Ce-ST, (d,e,f) Ch-ST, and (g,h,i) CeCh-ST at magnifications of 2000, 10,000 and 30,000×.

In contrast to the low magnification images, the high magnification SEM images in the bottom row of Figure 1 imply that the surface layer of chitosan nanofiber-loaded samples is more influenced by the localization of nanofibers at the top layer of the films. It can be speculated that the added cellulose nanofibers were more localized at the depth of the films. In contrast, the added chitosan nanofibers were also localized at the surface of the films. It could be again inferred that the surface structure of the films is not highly dependent on the concentration of the nanofibers. The strong presence of chitosan nanofibers at the surface layer of the films is evident based on Figures 1(f) and (h).
The surface morphology of the essential oil-added samples is shown in Figure 2. In this series of samples, a hybrid of Thyme and Satureja essential oils was concurrently added to the chitosan matrix in the presence of nanofibers. Figures 1(a) to 1(c) shows the surface morphology of Ce-ST. The formation of irregularly shaped domains at the surface layer of the films could be attributed to the essential oils’ phase. The diameter of the oil domains is estimated to be in the range of 2–5 µm, which is comparable to the size of essential oil domains in other research. 26 The high magnification SEM image shown in Figure 2(c) clearly shows a magnified view from one of those oil domains. It is rational to conclude that the essential oils distributed at the surface layer have been evaporated due to the high volatility of essential oils. As a result, those domains could be detected, which are a trace of the oil phase at the surface layer of the films. In the case of Ch-ST, it is seen that the essential oil domains were more evenly distributed at the surface of the films, which could be attributed to the higher compatibility of chitosan nanofibers with the chitosan matrix. As opposed to the surface morphology of Ch2 (See Figure 1(f)), the chitosan nanofibers were not detected at the surface layer of the films, which could be attributed to the role of essential oils in the surface morphology of the films. It also can be observed that the oil domains were somewhat circular as opposed to those irregularly-shaped oil domains in the case of Ce-ST. A more evident appearance of the oil domains could be observed in Figure 2(f), further showing the interface of oil domains with the chitosan matrix. In the case of CeCh-ST, the surface morphology is more similar to that of Ce-ST, implying the significant influence of cellulose nanofibers on the surface morphology of the films, and the oil domains became irregularly shaped.
XRD analysis
To evaluate the crystal structure of the films, XRD analysis was performed on all the samples, and the results are depicted in Figure 3. As expected, several sharp peaks could be detected in the XRD pattern of pure chitosan film. The peaks that appeared at 2θ of 8.6 and 11.6 can be attributed to the anhydrous crystalline chitosan (020 planes), and the distinct peak that appeared at 2θ of 18.4° is the hydrated crystal character (110 planes).
27
According to Figure 3(a), the addition of 2 wt% cellulose nanofibers highly diminished the crystallinity of chitosan since no sharp peaks could be observed in the diffraction pattern of the Ce2 sample. There could only be detected a broad hump implying the amorphous structure of chitosan in the Ce2 sample. It could be postulated that the addition of 2 wt% of cellulose nanofibers has disrupted the crystalline structure of chitosan. Quite unexpectedly, further increment of cellulose nanofiber concentration to 4 wt% led to a crystalline structure since the sharp peaks at the same angles could be observed. It could be inferred that a higher concentration of cellulose nanofibers resulted in the formation of aggregations within the bulk of the film leading to a weaker effect of nanofibers on the crystalline structure of chitosan. Chitosan nanofibers exhibited a different influence than cellulose nanofibers on chitosan’s crystallinity. Based on Figure 3(a), it is evident that, regardless of the chitosan nanofiber content, the crystallinity of chitosan was highly hampered. The reason for this behavior could be ascribed to the high compatibility of chitosan nanofibers with the chitosan matrix leading to the disruption of the crystalline network of chitosan in the presence of chitosan nanofibers. (a,b) XRD diffraction patterns, and (c) the UV-vis spectra for the samples.
Figure 3(b) illustrates the XRD diffraction of essential oil-loaded samples. In the case of cellulose nanofibers, it is clear that the presence of essential oils had a very positive effect on the crystallinity of chitosan since the crystal peaks have appeared as opposed to their absence in the case of Ce2. This behavior could be attributed to the plasticizing role of essential oils which leads to enhanced chain mobility of chitosan macromolecules, and thus, a greater extent of crystallinity. The same behavior could be observed in the case of Ch-ST, for which the presence of essential oils also led to a higher crystallinity compared with the Ch2 sample. It could also be observed that CeCh-ST exhibits a high degree of crystallization. However, it should be pointed out that the peak intensity of the crystal peaks was lower for all the nanofiber and essential oil-containing samples compared with the pure chitosan.
UV-vis analysis
Absorption values at different wavelengths.

To better compare the absorption behavior of the samples, the absorption values at different wavelengths are listed in Table 1. Interestingly, the essential oil-loaded samples exhibited higher absorption values in all three UV regions (UV-C, UV-B, and UV-A). The absorption values exceeded 0.9 in essential oil-loaded samples, much higher than chitosan film (0.6–0.7). These results indicate that the addition of essential oils to the films increases UV light absorption. The enhanced absorption of UV light could be beneficial for food packaging applications due to the increased antimicrobial behavior of the films. 28 According to the absorption data at the visible range, it can be seen that the absorption value was increased from 0.69 for the pure chitosan film to 0.80 and 0.74 for Ce2 and Ch2, respectively, implying the reduced transparency of the films. It is seen that the chitosan nanofiber-loaded film is more transparent than cellulose nanofiber-loaded film. In the case of essential oil-loaded films, the absorption values were measured as 0.83, 0.63, and 0.67 for Ce-ST, Ch-ST, and CeCh-ST, respectively. These results reveal that the transparency of chitosan film was notably reduced in the case of Ce-ST, but the transparency of Ch-ST is higher than the pure chitosan film. The hybrid sample also shows an absorption value close to the pure chitosan film.
ATR analysis
To monitor the structural changes resulting from essential oil and nanofiber additions, ATR-FTIR was employed, and the results are shown in Figure 4(a). All the characteristic peaks of chitosan have appeared in the ATR spectrum of the chitosan film and its corresponding composites. For all the samples, a broad peak appeared within the wavenumber region of 3000–3600 cm−1, which belongs to N-H and O-H stretching. Two absorption peaks at the wavenumbers of 2925 and 2868 cm−1 could be ascribed to C-H symmetric and asymmetric stretching, respectively, which are the characteristic of polysaccharides. The distinct peak at 1550 cm−1 corresponds to the N-H bending of the primary amine. The peak at the wavenumber of 1404 cm−1 belongs to the CH2 bending. The high-intensity peak at a wavenumber of 1029 cm−1 corresponds to the C-O stretching.
29
The peak at the wavenumber of 809 cm−1 could only be detected for the essential oil-loaded films and is attributed to the C-H aromatic out-of-plane deformation. This peak is absent in the case of pure chitosan film and nanofiber-loaded film. In this study, the main constituents of the used essential oils are thymol and carvacrol, which have aromatic structures. (a) ATR-FTIR spectra and (b) Storage modulus (E′) values for the samples.
DMTA analysis
Dynamic mechanical thermal analysis was employed to investigate the effect of nanofibers and essential oils on chain mobility and stiffness of the chitosan matrix. The storage modulus (E′) results are shown in Figure 4(b). The E′ values for all the samples decrease with increasing temperature, and at temperatures higher than 60°C, which is the glass transition range for chitosan, the E′ values decrease more rapidly. Quite expectedly, the E′ values were reduced for all the essential oil-loaded films compared to that of chitosan film. On the other hand, the E′ values were increased for the nanofiber-loaded films (Ce2 and Ch2) as compared with the pure chitosan film. It is seen that Ce2 exhibits higher E′ values within the glassy region than Ch2. It can be concluded that cellulose nanofibers have imparted higher stiffness into the chitosan films. The E′ value at the temperature of 30°C is 1160 Pa for the pure chitosan film, which increased to 1548 and 1360 Pa for Ce2 and Ch2, respectively, indicating a 33 and 17% enhancement than the chitosan film. In the case of essential oil-loaded samples, the film loaded with cellulose nanofibers (Ce-ST) showed higher E′ values than Ch-ST and CeCh-ST. The E′ value decreased from 1160 Pa for the pure chitosan film to 754, 441, and 478 Pa for Ce-ST, Ch-ST, and CeCh-ST, respectively. This behavior could also be attributed to the intrinsically higher stiffness of cellulose nanofibers rather than chitosan nanofibers.
DSC analysis
Glass transition temperature (Tg), zone of inhibition (ZOI), moisture content, and water vapor permeability (WVP) for the samples.

Wetting analysis
To determine the wetting behavior of the films, the wettability of the samples was obtained by measuring the water contact angles (WCAs). According to Figure 5, the WCA value for the pure chitosan film was obtained as 70°, which was reduced to 64° upon introducing 2 wt% of cellulose nanofibers. On the other hand, the WCA value was notably increased to 95° upon adding 2 wt% of chitosan nanofibers. Such increased hydrophobicity could be attributed to the induced surface roughness in the case of Ch2, as shown in SEM images (Figure 1(f)). The induced nano-scale roughness is the reason for the observed boost in hydrophobicity of the film.32,33 In contrast to nanofiber-loaded samples, the hydrophilicity of the films was highly increased upon the concurrent addition of essential oils and nanofibers. As shown in SEM images (Figure 2), the surface layer of the films became heterogeneous upon the addition of essential oils. In fact, the oil phase was distributed as several micrometer-sized domains within the top layer of the films making the surface morphology heterogeneous, which leads to increased hydrophilicity, mainly because the oil phase has been evaporated. No significant differences were observed between the essential oil-loaded samples, and the WCA was obtained in the range of 30–36°. Water droplet profiles for the chitosan film and its corresponding composites. The WCA values are also reported as insets.
Antibacterial properties
The nanofiber-loaded films were not successful in imparting antibacterial properties, as shown in Supplement Figure S3. Based on the quantified results shown in Table 2, ZOI was attained as 16 mm for Ce2 compared with 10 mm for Ch2, indicating the higher antibacterial activity of cellulose nanofibers compared to chitosan nanofibers. In contrast to the nanofiber-loaded films, the samples containing essential oils exhibited a profound antibacterial behavior. A clear ZOI has appeared around the film samples with the size of 20, 21, and 22 mm for Ce-ST, Ch-ST, and CeCh-ST, respectively. The solid antibacterial character of essential oils has been well established in the literature. 34 The observed profound antibacterial activity could be attributed to the participation of essential oils’ constituents (thymol and carvacrol) in the lipid layer of the cell membrane leading to cell membrane deterioration. 35
In addition, the potential of the films to prevent bacterial colonization was evaluated via plate counting technique. Figure 6 shows the typical colonization by E. coli bacteria on agar plates from the liquid medium cultured with the pure chitosan film and the hybrid nanocomposite film (CeCh-ST). Considering the number of surviving cells (CFU/mL), the bacterial adhesion reduction (%) of E. coli was obtained as ∼92% for CeCh-ST. It is seen that the hybrid nanocomposite film demonstrated a profound antibacterial activity against E. coli, which is originated from the fact that growth of microorganisms in medium was inhibited by the presence of bioactive components of the loaded essential oils in CeCh-ST. In fact, the antimicrobial ingredients of the loaded essential oils gradually released from the chitosan film matrix, thus preventing microbial growth Images of colony forming units of bacteria after 24 h incubation for (a) pure chitosan film and (b) CeCh-ST nanocomposite film.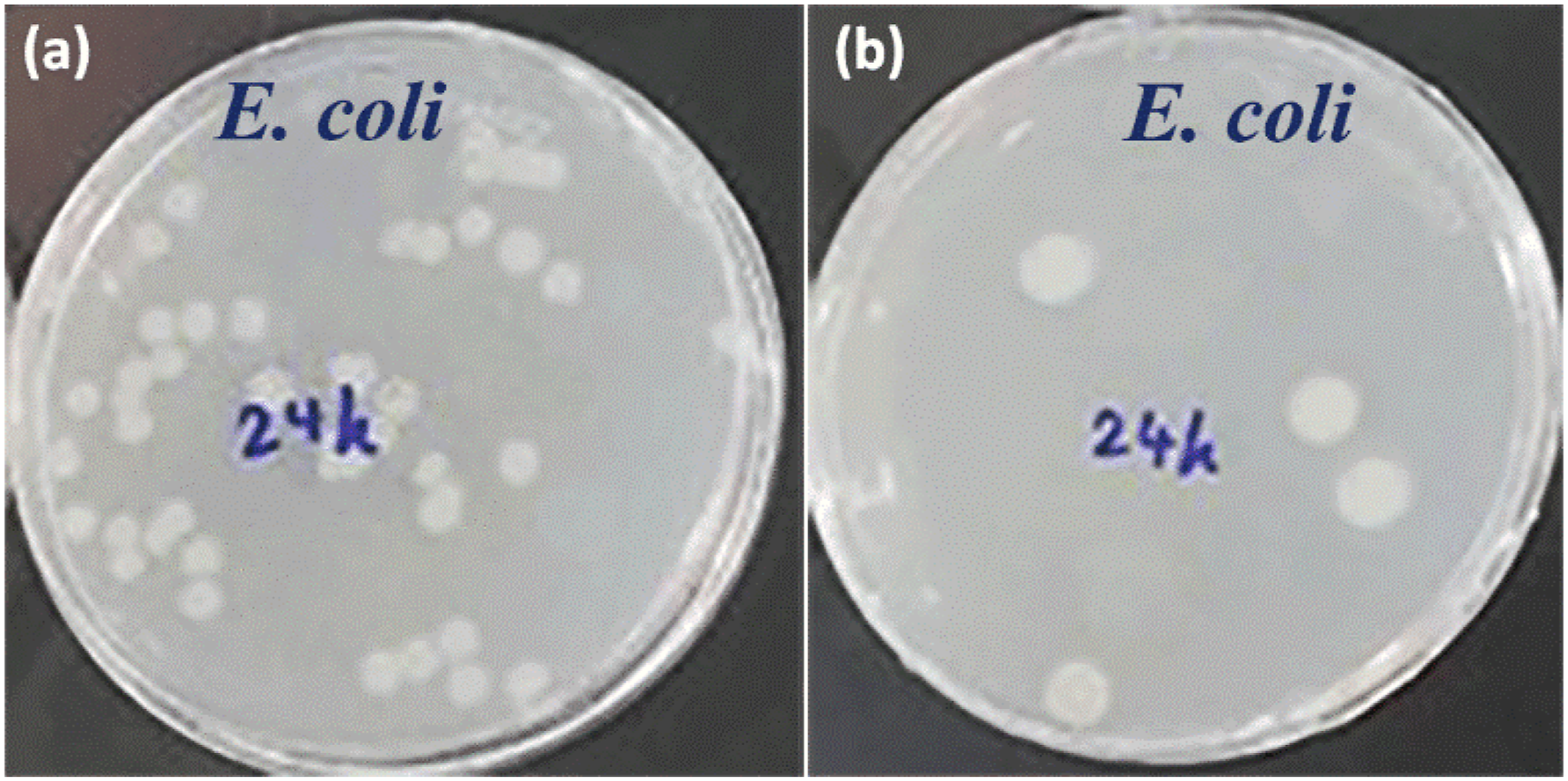
Barrier properties
The moisture content of the films was measured and reported in Table 2. As is seen, the moisture content was increased from 17% for the pure chitosan film to 20.4% for cellulose nanofiber-loaded film (Ce2), which could be due to the hydrophilic character of cellulose nanofibers and also the reduced crystallinity of chitosan in the presence of cellulose nanofibers. On the other hand, the moisture content was reduced to 15.1% in the case of chitosan nanofiber-loaded film (Ch2). The reason for the lower moisture absorption could be the enhanced hydrophobicity of Ch2. In the case of essential oil-loaded films, the moisture content was increased compared to their corresponding nanofiber-loaded films and the pure chitosan film. This result could be explained by the highly enhanced hydrophilic behavior of the films as a result of evaporation of the essential oils from the surface layer of the films, which led to the formation of heterogeneous surface morphology.
WVP values are also listed in Table 2, indicating an increase from 3.96 g/m.s.Pa for the pure chitosan film to 4.57 g/m.s.Pa for the cellulose nanofiber-loaded film (Ce2). On the one hand, cellulose nanofibers provide a tortuous path against the diffusion of water vapor molecules. On the other hand, they have disrupted the crystalline network of chitosan and thus offer a more expansive space for the diffusion of water vapor molecules. The latter mechanism dominated, and thus the WVP was increased in the case of cellulose nanofiber-loaded film. In contrast to Ce2, the WVP value was reduced to 3.12 g/m.s.Pa for the chitosan nanofiber-loaded film (Ch2). This result could be explained by the higher hydrophobicity of Ch2 (WC = 95°). In the case of the essential oil-loaded films, the WVP of Ce-SH (4.23 g/m.s.Pa) was higher than that of pure chitosan film. The other two samples exhibited lower WVP values. Based on Table 2, the WVP of Ch-ST and CeCh-ST were reduced to 2.81 and 3.34 g/m.s.Pa, respectively. The lowest amount of WVP was observed for Ch-ST, which can be attributed to the tortuous path imparted by the presence of chitosan nanofibers and also the presence of oil phase within the bulk of the films.
Conclusions
We developed edible nanocomposite films based on chitosan, cellulose/chitosan nanofibers, and Thyme/Satureja essential oils. Morphological analysis revealed that the chitosan nanofibers were more localized at the films’ surface compared with the cellulose nanofibers. The surface aggregation of chitosan nanofibers caused a nano-scale roughness to the films, which has resulted in an enhanced hydrophobic character (WCA = 95°). The UV-vis analysis demonstrated that the UV absorption was highly increased in the case of essential oil-loaded films. We also found that the combination of chitosan nanofibers and essential oils results in a more transparent film with respect to the pure chitosan film. DMTA analysis showed that the cellulose nanofibers caused higher storage modulus values within the glassy region than chitosan nanofibers. The addition of nanofibers was also found to have an increasing effect on the glass transition temperature of the chitosan films indicating the decreased chain mobility of chitosan in the presence of nanofibers. The antibacterial assay revealed that the nanofiber-loaded films exhibited no pronounced antibacterial activity against E. coli. In contrast, the essential oil-loaded films were highly successful in imparting antibacterial properties into the films. The sample loaded with the hybrid of nanofibers exhibited a slightly higher antibacterial behavior. In conclusion, the developed multi-component edible nanocomposite films based on chitosan, cellulose/chitosan nanofibers, and Thyme/Satureja essential oils exhibited a promising potential in the barrier and antibacterial properties, making them highly suitable for food packaging applications.
Supplemental Material
Supplemental Material - Preparation and characterization of antibacterial chitosan nanocomposites loaded with cellulose/chitosan nanofibers and essential oils
Supplemental Material for Preparation and characterization of antibacterial chitosan nanocomposites loaded with cellulose/chitosan nanofibers and essential oils by Neda Sadat Aghayan, Javad Seyfi, Mohammad Javad Asadollahzadeh, Seyed Mohammad Davachi and Maryam Hasani in Polymers and Polymer Composites
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.