Abstract
Alkaline pretreated hemp fibers were modified by steam explosion and/or silanization before being compounded with polylactic acid (PLA). The fungal biodegradation of the PLA/hemp fibers composite was investigated using
Introduction
The growth of the worldwide green economy has driven demand for biodegradable plastics to replace petroleum-based plastics.1,2 Biodegradable aliphatic polyesters are considered to be the most promising biodegradable and environmentally friendly plastics due to their hydro-degradability, thermoplasticity, and favorable physico-mechanical properties. 3 Polylactic acid (PLA) is a biodegradable aliphatic polyester produced from corn, beet, wheat, and other renewable starch sources. 4 Since the mechanical properties of PLA are similar or superior to those of conventional polymers, it is one of the most widely used biodegradable plastics.5–7 Although PLA presents some drawbacks such as brittleness, low impact strength and poor resistance to thermal deformation, 8 its properties can be improved if the polymer is used in a composite. PLA composites that included natural fibers such as kenaf,9,10 hemp,11,12 flax 13 and sisal fibers14,15 have been used in automotive interiors, 16 construction materials, 17 and packaging materials. 18
Some of these applications require durable products, particularly outdoor applications in humid environments. For these products, the microbial degradation of the composite must be carefully considered to ensure satisfactory long-term performances.
19
Over the past two decades, the biodegradability of PLA has been investigated using aerobic microorganisms such as actinomycetes, bacteria, and fungi in laboratory settings.
20
The present work aims to investigate the properties of a PLA/hemp fiber composite after fungal degradation by
Materials and method
Materials and chemicals
PLA 3052D was supplied by NatureWorks Co. Ltd. (Plymouth, MN, USA). Hemp (
Alkaline pretreatment of hemp fibers
Hemp fibers were pretreated with a 6% (w/v) aqueous solution of sodium hydroxide at room temperature for 48 h. After treatment, the fibers were washed with water several times until the pH of the water was neutral (pH = 7). The fibers were then dried at room temperature and cut into 0.2–0.5 cm lengths.
physicochemical modifications of alkaline pretreated fibers
After alkaline pretreatment, the hemp fibers were modified by using three different physicochemical treatments: steam explosion, silanization, and steam explosion followed by silanization (Figure 1). Steam explosion was performed at ∼210°C at a pressure of 18 kg/cm2 for 5 min. The codes of samples are shown in Table 1. Flow chart of the PLA/Hemp fibers composites. Abbreviation and composition of the PLA/hemp composites.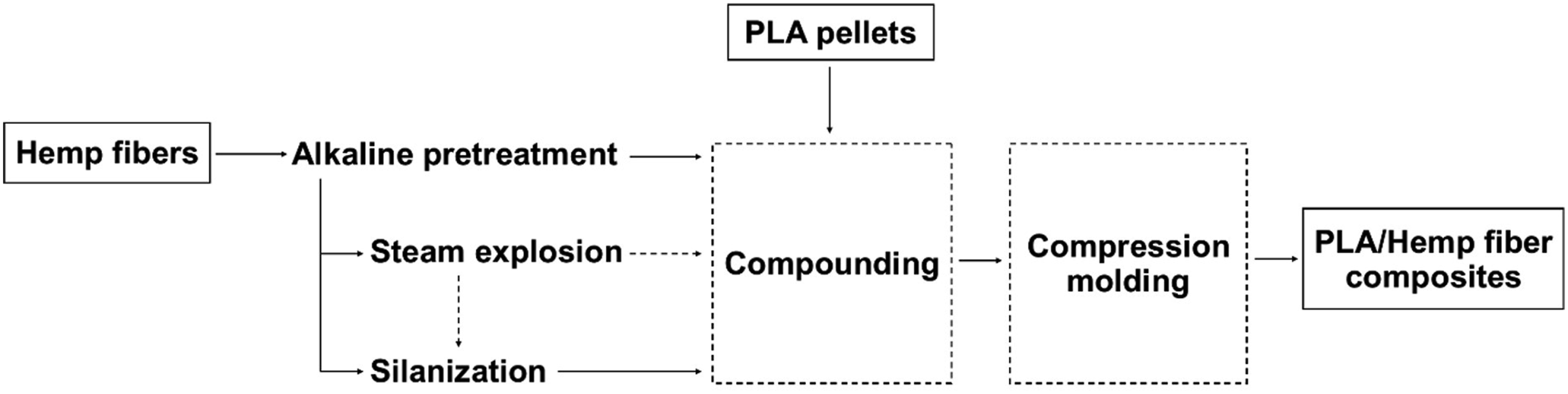

Before silanization, the alkaline-pretreated fibers were soaked in 5% (w/v) sodium hydroxide at a ratio of 1:10 (w/v) at room temperature for 1 h and washed with distilled water until neutral pH was obtained. The silanization of fibers was then performed according to the following reported method. 19 A silane coupling agent was prepared as follows: 3% (w/v) MPS was dissolved in 60% (v/v) ethanol, and the solution was adjusted to pH ∼ 4 by adding 5% (v/v) of acetic acid. Hemp fibers (2.5% (w/v)) were immersed in the silane coupling agent at room temperature for 6 h. The silane-treated fibers were collected and dried in an oven (Memmert UN110, Schwabach, Germany) at 120°C for 2 h.
Preparation of PLA/Hemp fiber composites
PLA pellets were dried at 80°C to remove excess moisture. The PLA pellets were compounded for 5 min with modified hemp fibers (30% by weight of PLA) using an internal mixer (Barbender, Germany) at 180°C and 60 rpm. The obtained composites were cut into small pieces and test specimens 2 mm thick were prepared in a compression molding machine at 180°C, applying pressure in three stages: 0 psi for 3 min; 1,000 psi for 2 min and then, after releasing the pressure, 1,500 psi for 1 min.
Fungal biodegradation testing
The fungal biodegradation by
Characterization of PLA/Hemp fibers composites before and after fungal biodegradation
Mechanical properties
The fiber composite samples for the flexural test were investigated according to ISO 178 (three-pointed bending mode) using a Universal Test Machine (Instron Model 3365) at room temperature. The maximum load capacity of this equipment is 1 kN, and the crosshead speed was set at 2 mm/min. In terms of the impact strength, the fiber composite samples were tested according to ISO 179 using a Zwick/Roell 5102 Pendulum Impact Tester operating with a pendulum energy of 1 J. At least five specimens were tested to determine the average values including their standard deviations of the results.
Scanning electron microscope (SEM)
The morphology of fractured surfaces of samples after impact testing was observed by a scanning electron microscope (JSM-5800 LV, JEOL, Japan) with an accelerating voltage of 20 kV. The fractured surfaces were sputtered and coated with a thin layer of gold prior to analysis.
Molecular weight by gel permeation chromatography (GPC)
To determine the degree of fungal biodegradation, specimens were analyzed before and after incubation by gel permeation chromatography (GPC) with refractive index detection (Shimadzu Prominence, Tokyo, Japan). Samples were dissolved in tetrahydrofuran (THF) to a final concentration of 0.1% (w/v). The obtained solution was filtered through a 0.45 µm nylon membrane. The filtered solution (20 µL) was injected into the mobile phase of THF at 40°C at a flow rate of 1 mL/min. Polystyrene of known molecular weight was used as the calibration standard.
Chemical structure by fourier transform infrared spectroscopy
Changes in the chemical structures of samples caused by fungal biodegradation were investigated by using Fourier transform infrared spectroscopy (FTIR) in attenuated total reflectance mode (Bruker Tensor 27 FTIR Spectrometer, Billerica, MA, United States). Spectra were recorded from 32 scans from 4000 to 600 cm−1 at a resolution of 4 cm−1. The obtained spectra were pre-processed by baseline correction and vector normalization using the OPUS 7.5 software (Bruker Optics Ltd., Ettlingen, Germany). The semi-quantitative degree of biodegradation was evaluated from the carbonyl index, which was calculated from the ratio of the band intensities of carbonyl ester groups (C = O) (I1750) and CH3 bending (I1452),
12
using equation (1):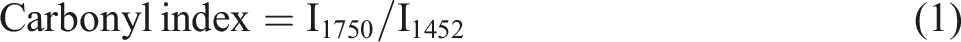
Thermal properties by differential scanning calorimetry
The melting behavior and crystallization of samples before and after fungal biodegradation were examined under a nitrogen atmosphere by differential scanning calorimetry (DSC) (Perkin-Elmer DSC7, Waltham, MA, USA). The temperature was raised from 30 to 190°C at a rate of 10°C/min and then held constant at 190°C for 1 min. Samples were then cooled to 30°C at 10°C/min before being heated again in the same condition as the first cycle. The enthalpy values derived from DSC profiles were used to calculate the degree of crystallinity (χc) using the following equation (2):
Thermal degradation by thermogravimetric analysis
The thermal and degradation stability of samples were investigated by thermogravimetric analysis (TGA) (Perkin-Elmer TGA 8000, Waltham, MA, USA). Samples were heated from 25°C to 600°C at a rate of 10°C/min under a nitrogen gas flow.
Surface free energy (SFE) analysis
The hygroscopic properties of samples before and after fungal biodegradation were evaluated by contact angle measurement (Data Physics OCA 15EC, Filderstadt, Germany). A single 5 μL drop of distilled water, formamide or ethylene glycol was deposited from a syringe onto the sample surface. Images were captured by video camera, and analysed to calculate the contact angle (θ). The surface energy of samples was calculated using the Owens and Wendt approach, 26 dividing the surface energy of samples into dispersive and polar components. The resulting contact angle measurements in the case of each of the nanocomposite films were used to calculate their SFE.
Statistical analysis
All experimental data were expressed as means
Results and discussion
Mechanical properties
Mechanical properties of PLA/hemp fiber composites.

*neat PLA The different superscript letters (a, b, c, d and f) for mechanical properties indicated a statistically significant difference at
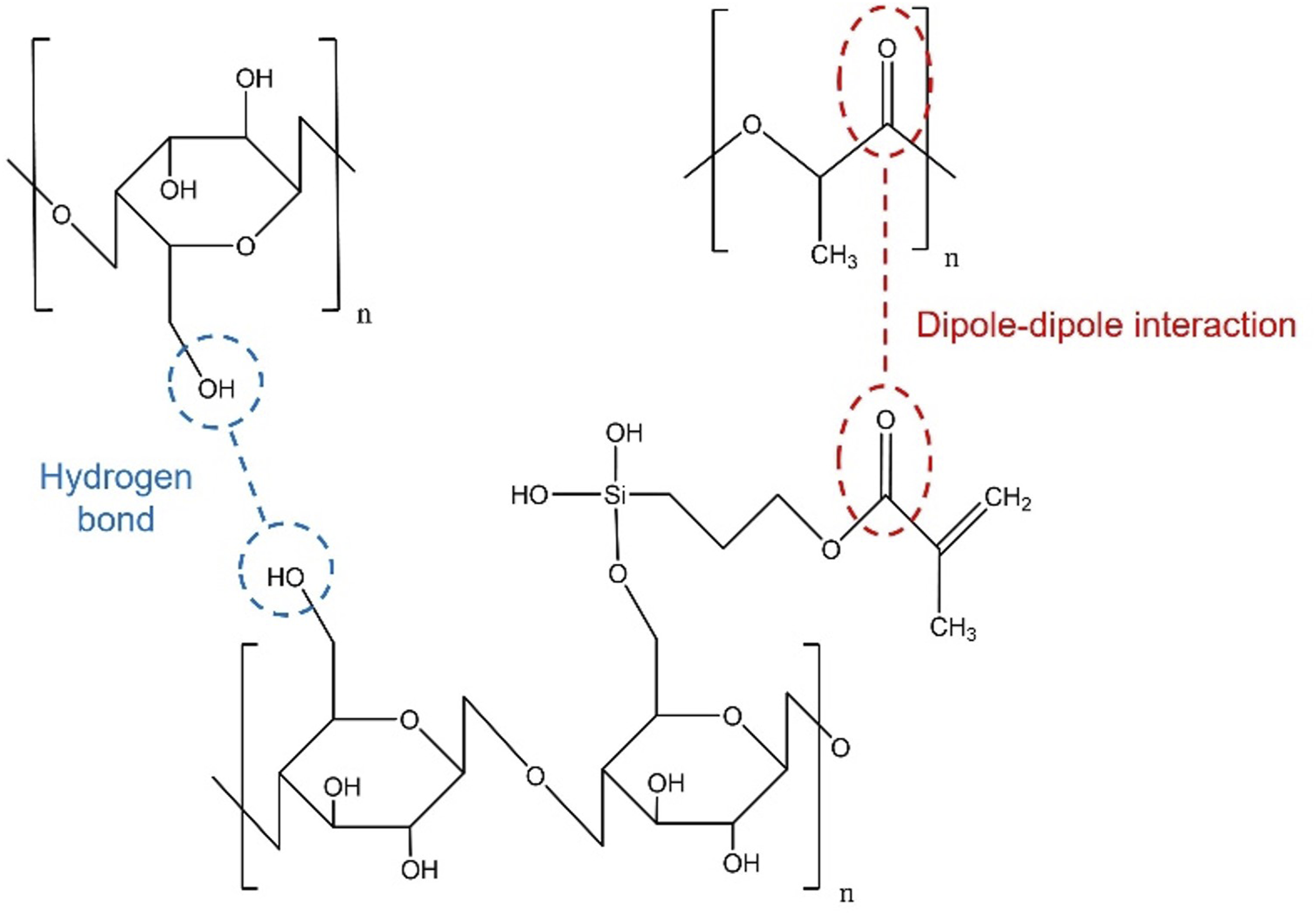
The interaction between the silanized fiber and PLA matrix.
Furthermore, enhanced hydrophobicity after salinization could prevent water absorption in composites. Consequentially, the service lifetime of composites could be prolonged as the mechanisms of hydrolytic and enzymatic degradation were obstructed.15,28 Nevertheless, SEM images in Figure 3(b) (PLA/A-S) and 3(d) (PLA/A-SE-S) revealed that the silanized and steam explosion-silanized fibers were more damaged than unsilanized fiber when subjected to an impact test. As described previously, during silanization, the fibers might be partially decomposed to weaken. Therefore, these caused it to lose the capability of instant load receiving. SEM images at cross section after impact testing; (a) PLA/A, (b) PLA/A-S, (c) PLA/A-SE and (b) PLA/A-SE-S.
The silanization of hemp fiber in PLA/A-S and PLA/A-SE-S composites as seen in Figures 3(b) and 3(d), revealed a rougher surface after impact testing. Jaw fish shape (rougher surface) in cross section was crucial evidence to confirm that the composites require more energy to destroy. This confirmed the improved interfacial adhesion between the silanized fiber and matrix of both composites. In addition, embedded fiber, as seen in PLA/A-SE-S (Figure 3(d)), could strongly confirmed the better compatibility of hemp fiber with the PLA matrix over PLA/A-SE (Figure 3(c)). However, based on the results of mechanical properties in Table 2, it was found that the composites with silanization of fiber (PLA/A-S and PLA/A-SE-S) had a lower impact resistance than those without silanization (PLA/A and PLA/A-SE). The results indicated that there might be other factors involved. Principally, the strength of composites is mainly dependent Even though the compatibility between fiber and matrix has improved, leading to better load transfer from matrix to fiber, the insufficient strength of the matrix and fiber may lead to the lower capability of composites to withhold the external load.
The silanization of fiber under acidic conditions might damage the fiber structure, as described previously. As seen in Figures 3(b) and 3(d), the “breakage fiber” in both figures indicates defects due to their weakening structure. Subsequently, the composites were easily cracked when receiving loads.
Measurement of durability of PLA/hemp fiber composites via fungal growth test
Molecular weight change
The weight and number -average molecular weight (
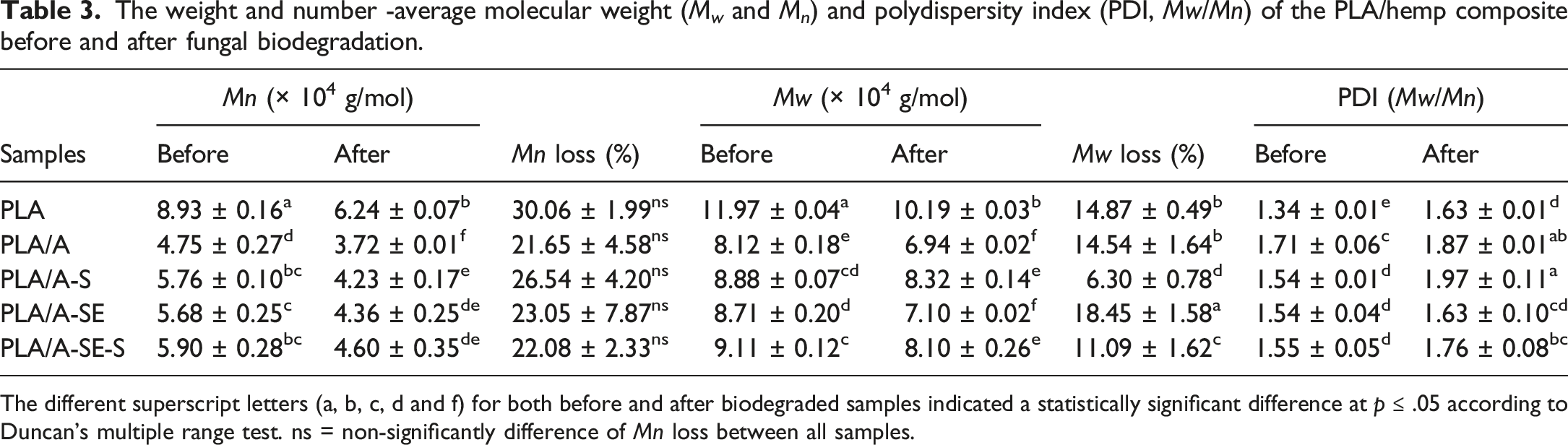
The different superscript letters (a, b, c, d and f) for both before and after biodegraded samples indicated a statistically significant difference at
Chemical structure changes
The carbonyl index of the PLA/hemp composite before and after fungal biodegradation.

The different superscript letters (a, b, c and d) for both before and after biodegraded samples indicated a statistically significant difference at
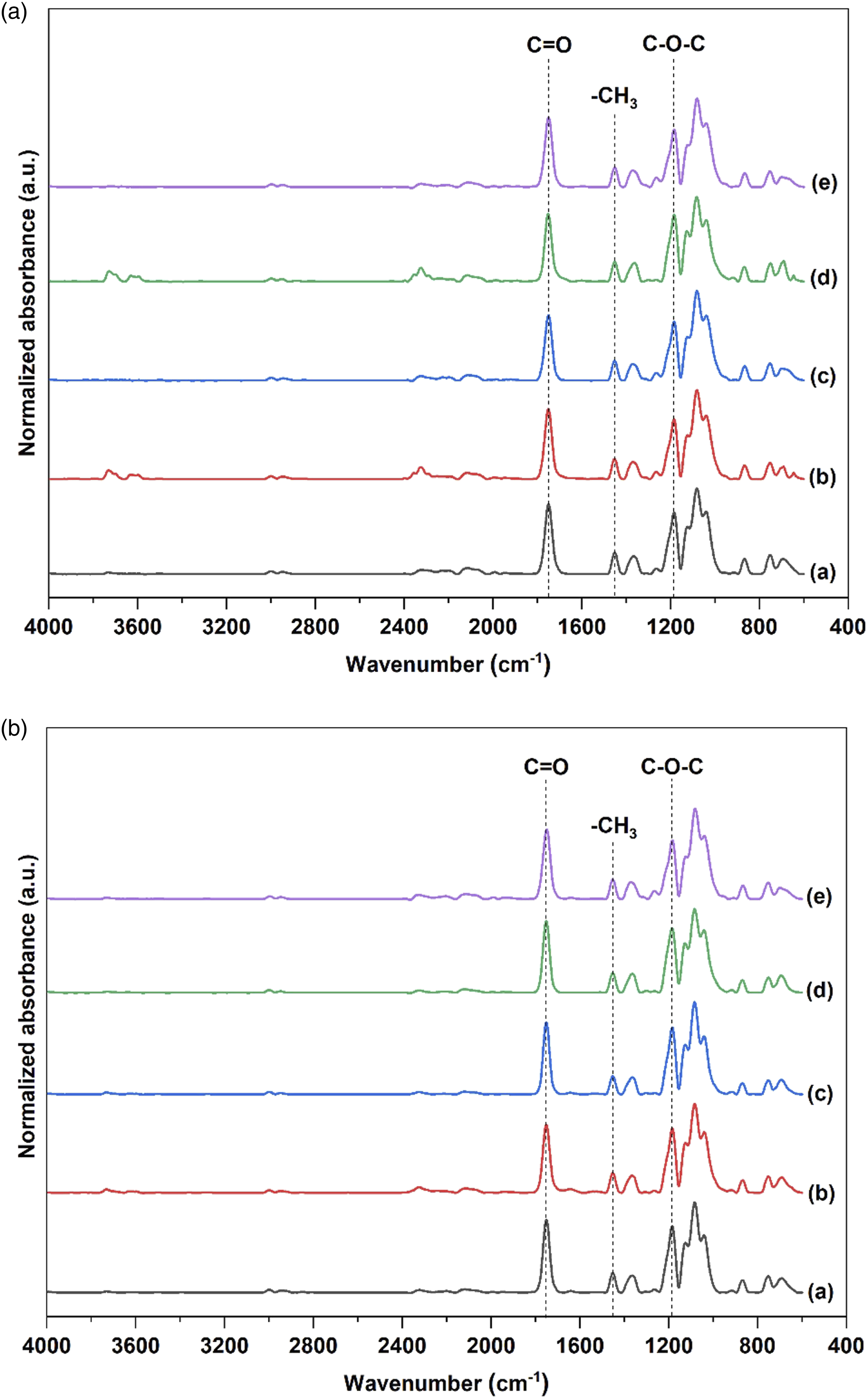
FTIR spectra of PLA/Hemp biocomposites (a) before and (b) after fungal biodegradation: (a) PLA, (b) PLA/A, (c) PLA/A-S, (d) PLA/A-SE and (e) PLA/A-SE-S.
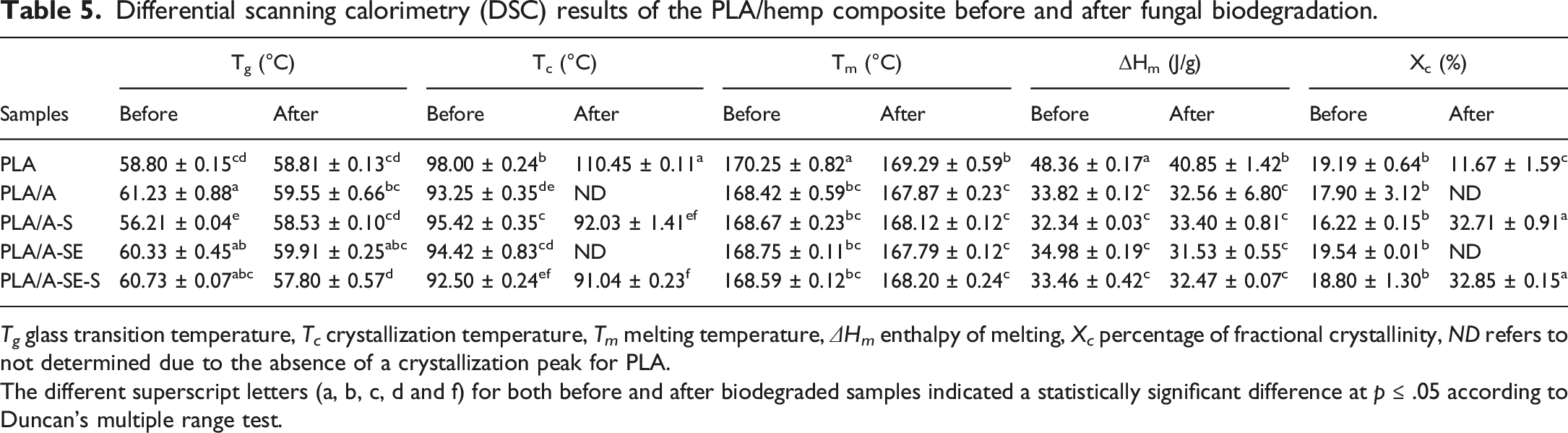
Differential scanning calorimetry
The influence of fungal biodegradation on the thermal behavior of the studied PLA/hemp composites was investigated by DSC. The DSC thermograms of all samples were presented in Figure 5, and the data from DSC analysis were summarized in Table 5. The glass transition temperature (Tg) of neat PLA did not change, while the Tg values of the PLA/hemp composites were significantly different after exposure to Differential scanning calorimetry (DSC) thermograms of PLA/Hemp biocomposites (a) before and (b) after fungal biodegradation: (a) PLA, (b) PLA/A, (c) PLA/A-S, (d) PLA/A-SE and (e) PLA/A-SE-S. Differential scanning calorimetry (DSC) results of the PLA/hemp composite before and after fungal biodegradation. The different superscript letters (a, b, c, d and f) for both before and after biodegraded samples indicated a statistically significant difference at 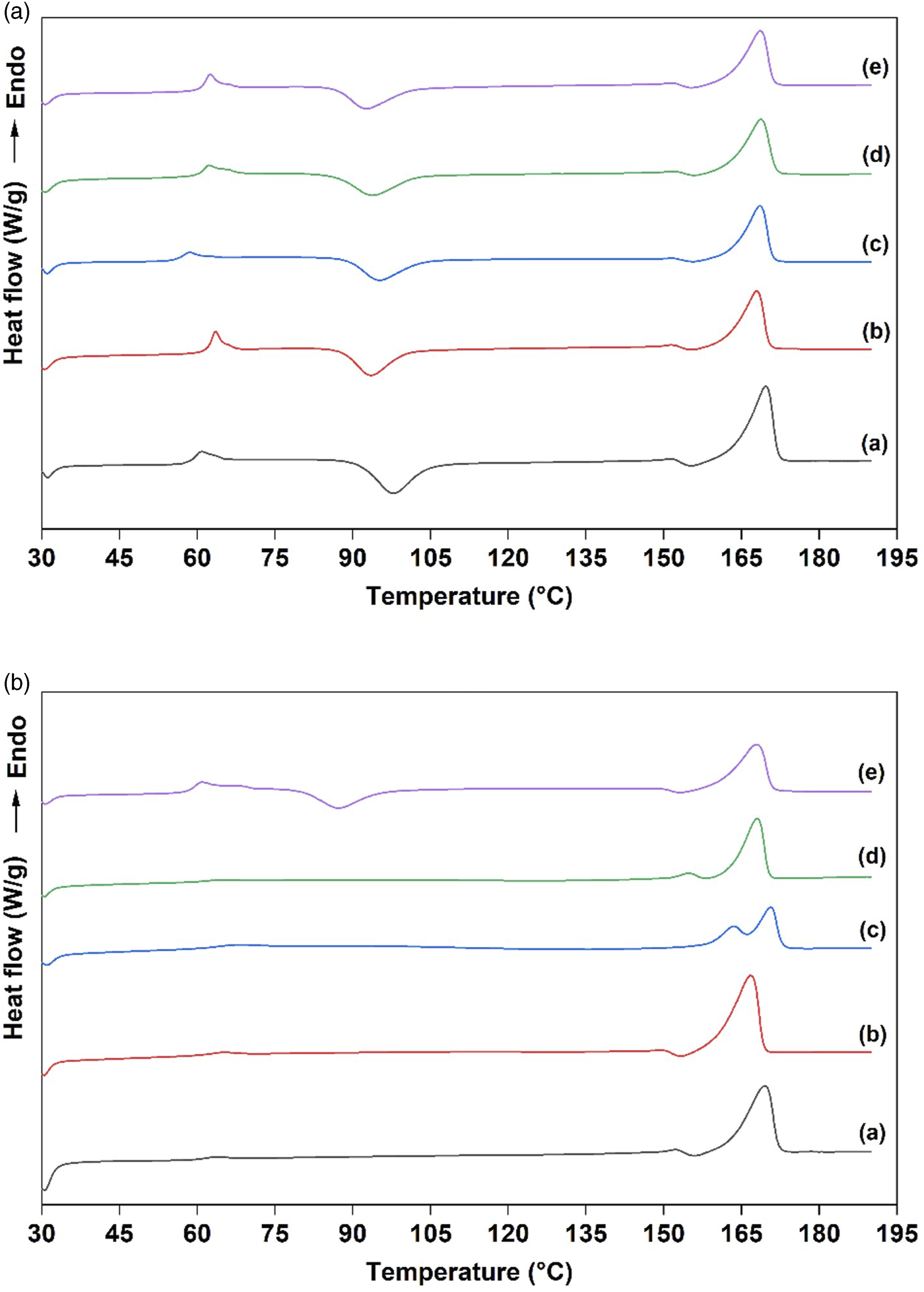
Thermogravimetric analysis
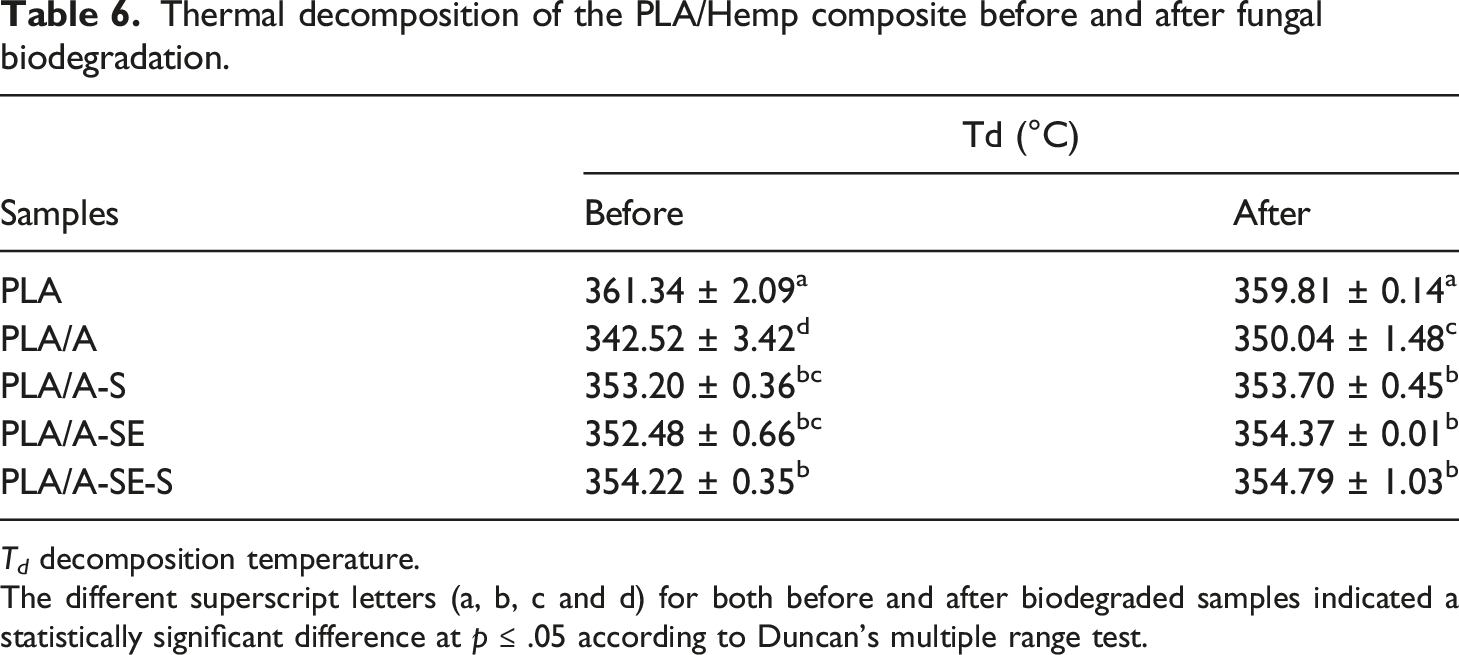
The thermal stability and decomposition temperature (Td) of samples were analyzed by TGA before and after fungal biodegradation (Figure 6). The Td values of biodegraded samples were significantly reduced (5 °C–19°C) ( The thermograms of thermogravimetric analysis (TGA) and derivative thermal gravimetric analysis (DTG) of PLA/Hemp biocomposites (a) before and (b) after fungal biodegradation: (a) PLA, (b) PLA/A, (c) PLA/A-S, (d) PLA/A-SE and (e) PLA/A-SE-S. Thermal decomposition of the PLA/Hemp composite before and after fungal biodegradation. The different superscript letters (a, b, c and d) for both before and after biodegraded samples indicated a statistically significant difference at 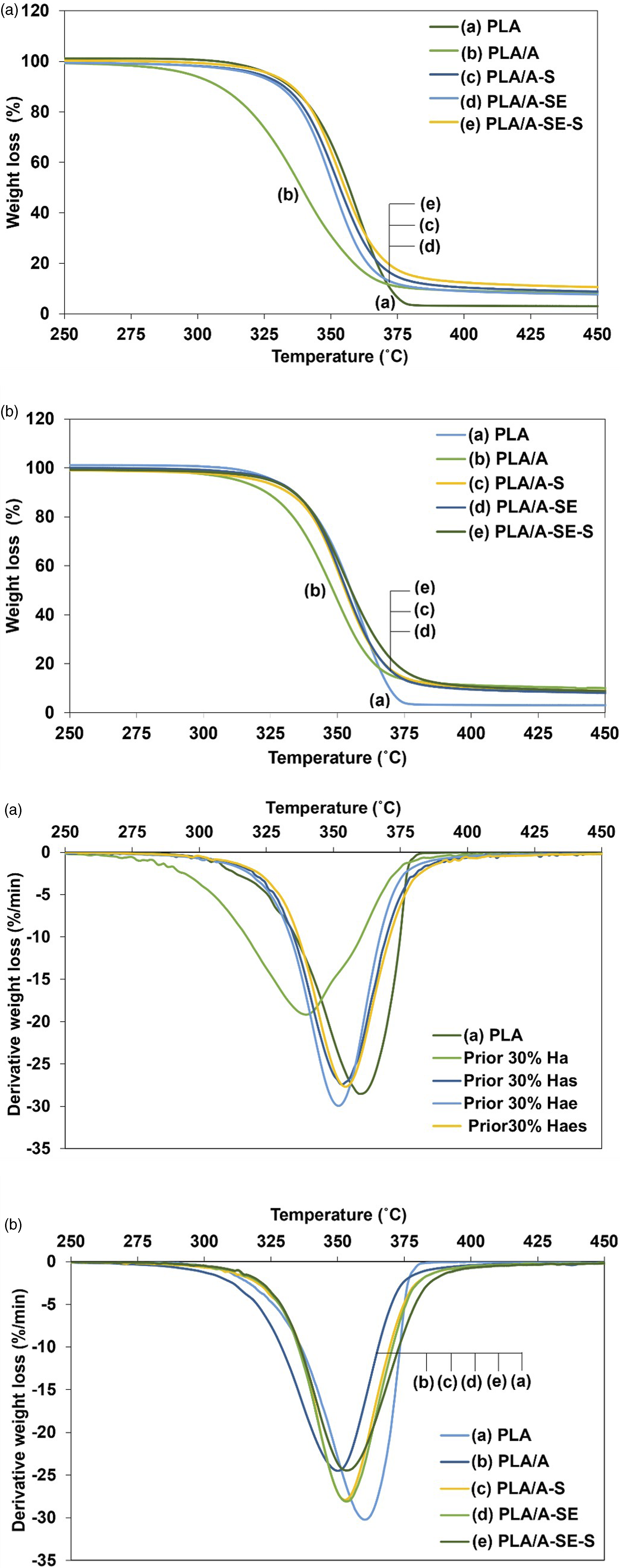
Surface tension
The total, polar and dispersive surface tensions of PLA/hemp biocomposite before and after fungal biodegradation.
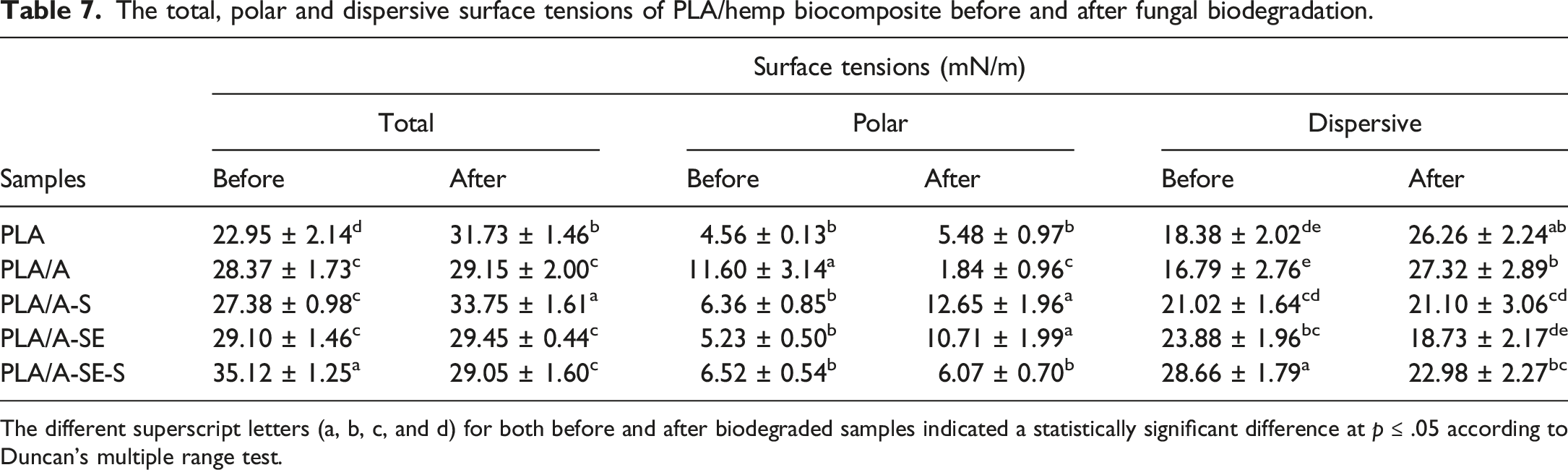
The different superscript letters (a, b, c, and d) for both before and after biodegraded samples indicated a statistically significant difference at
It was noticeable that steam explosion and/or silanization significantly increased the dispersive non-polar surface tension of PLA/A-S, PLA/A-SE and PLA/A-SE-S samples (
Many previous studies reported that the improved hydrophobicity of lignocellulosic fiber composites, influenced the delay of biodegradation due to inappropriate conditions for microbial growth. 42 The moisture or water absorbed in composites could facilitate inducing fungal or bacterial invasion. 43 The enhancement of hydrophobicity on PLA/hemp fiber composites led to their decreased moisture or water absorption. This was subsequently followed by less hydrolytic and enzymatic degradation, respectively. Therefore, it is considered a sustainable approach for expanding the service lifetime without any antimicrobial agent incorporated.
Conclusion
The durability of PLA and PLA/hemp fibers composites in outdoor applications was investigated by simulated fungal biodegradation for 28 days, following the ISO 846: 1997 standard method involving an aqueous medium containing
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was financially supported by the National Research Council of Thailand (NRCT), under grant no. SCI600609S.
