Abstract
The study investigated the effect of paraffin wax, expanded graphite, and their dispersion on the properties of the blend of polypropylene (PP) and ethylene vinyl acetate (EVA). The idea is to reduce interfacial tension in blends of PP and EVA while enhancing the properties of blends in the process. The chosen optimum contents of the paraffin wax and EG in the 80/20 PP/EVA blend were 10 and 6 wt.%, respectively. The morphological features show that the expanded graphite (EG) particles had high affinity for each other. Moreover, EG filler particles are localized inside the minor EVA phase and some at the interface. Rheological properties revealed that the flow of the binary composites increased when 10 wt.% wax is added suggesting that wax enhanced the processability of the composites. The Cole-Cole plots deviated from the semi-circular arc suggesting heterogeneity in the blend and PP/EVA/Wax/EG composite. The addition of paraffin wax and EVA delayed crystallization of PP, while earlier crystallization of PP was promoted by the presence of EG. Generally, all samples caught fire after 10 s, started dripping and are thus given a V-2 rating according to the Underwriters Laboratories test standard 94 results (UL-94).
Introduction
Polypropylene (PP) is amongst important thermoplastics utilized in various applications, namely automobile and home appliances, due to its ease of processability. 1 However, the most noticeable drawback of PP is its low toughness and impact resistance at either temperature below or around the glass transition temperature (Tg). 2 The polymer industry developed high impact PP based materials through copolymerization of PP with small fractions of ethylene to overcome the drawbacks associated with native PP. Another approach is to blend PP with softer or elastomeric polymers, namely ethylene vinyl acetate (EVA) to overcome its inherent drawbacks. 3 However, blends of PP and EVA are thermodynamically immiscible due to poor interfacial compatibility between the constituents of the blend.4,5 Therefore, the anticipated properties for the blends of PP and EVA may be achieved at the expense of the good stiffness of PP. Usually, nanofillers are incorporated into the blends of PP and EVA to achieve a balance in the properties. Carbonaceous nanomaterials such glassy carbon, 6 carbon nanotubes,7,8 and graphene or graphite9–11 have been used. Dos Anjos et al. 6 produced glassy carbon containing PP/EVA nanocomposites through melt extrusion while keeping about 60/40 PP/EVA ratio in the samples. The authors reported an immiscible dispersed morphology in the non-compatibilized 60/40 PP/EVA blend and high interfacial adhesion in a maleated PP containing blend. The glassy carbon preferentially localized inside the EVA phase and interface of the blended polymers. A thermal stability of maleated PP containing blend increased when 0.5 to 1 wt.% glassy carbon was incorporated. The authors attributed these results to higher thermal stability of glassy carbon than the blended components, and its ability to form a barrier that prevents the diffusion of gases arising from degraded material. However, Dos Anjos et al. 6 did not account for the relatively lower thermal stability PP/EVA/glassy carbon nanocomposite at 3 wt.% glassy carbon loading in comparison to 0.5 and 1 wt.% loadings. Generally, it is known in studies of polymer composites that dispersion levels, distribution of fillers, and interfacial interactions between the components play an important part for enhancing the properties. Therefore, the relatively lower thermal stability at 3 wt.% glassy carbon loading can be due to poor dispersion or agglomerates obtained at higher filler loading. Liu et al. 7 prepared composites comprised of either 60/40 or 80/20 PP/EVA and functionalized multiwalled carbon nanotubes using a twin-screw extruder. Their results revealed development of a more perfect co-continuous network structure of functionalized multiwalled carbon nanotubes in 60/40 PP/EVA relative to the 80/20 PP/EVA. Moreover, functionalized multiwalled carbon nanotubes exhibited poor effect on crystalline structures of PP/EVA, whereas EVA dominated the crystallization of PP during non-isothermal crystallization. In addition, rheological characteristics of nanocomposites showed that single- and dual-network structure of functionalized multiwalled carbon nanotubes exists in 80/20 and 60/40 PP/EVA, respectively. 8 Varghese et al. 9 investigated the influence of thermally reduced graphene oxide (TRG) on morphological, thermal, and rheological properties of polypropylene/maleic anhydride-graft-ethylene-vinyl acetate blends. Maleated EVA plays a compatibilizing role for promoting better dispersion and interfacial interaction TRG in the major PP phase. It was reported that TRG sheets localized inside maleated EVA for lower TRG composition (<3 wt.%), whereas at 5 wt.% TRG, the dispersed maleated EVA phase appeared smaller and a co-continuos morphology was attained. Incorporation of either TRG or maleated EVA and the differences in the morphological features of the nanocomposites had no marked influence on crystallization and melting temperatures of PP. The TRG containing nanocomposites had better thermal stability than neat polymers and the blend. The improvement in thermal stability was ascribed to the role played by TRG in forming a thermal shield that hindered the thermal decomposition during heating. Moreover, the rheological behaviours of the nanocomposites enhanced upon introduction of TRG, and this was ascribed to interactions between TRG and EVA-g-MA. Recently, Zebiri et al. 10 investigated the combined effect of the graphene oxide surface silanization and the addition of compatibilizers on the morphological, thermal, and mechanical properties of PP/EVA nanocomposites. The size of EVA reduced when compatibilizers were added. The ‘thermal stability and some tensile properties were improved upon addition of a combination functionalized graphene oxide and compatibilizers to PP/EVA. The authors linked the better characteristics of composites to better dispersion and interactions of graphene oxide nanoparticles with matrix in the presence of compatibilizers. Expanded graphite (EG) has generated a lot of interest from researchers in developing polymer nanocomposites.11–14 EG is relatively cheaper than other carbonaceous nanomaterials such as carbon nanotubes. However, better EG dispersion in polyolefins during melt processing is a challenge because EG forms non-covalent attractions. Lately, Fotoohi et al. 15 investigated the tensile and fracture behavior of PP/EVA/EG nanocomposites using nonmodified EG and EG that was surface modified with ammonium polyphosphate (EG-g-APP). At a fixed 10 wt.% EVA for equal compositions of PP/EVA/EG-g-MA and PP/EVA/EG, PP/EVA/EG-APP had better stiffness than the PP/EVA/EG composites. The authors attributed their results to less filler aggregation and better interfacial adhesion in PP/EVA/EG-APP composites. Mochane and Luyt 16 reported on morphology, thermal and flammability characteristics of EVA/wax/EG. The authors showed that the particles of EG agglomerated in EVA/EG composites, whereas better EG dispersion in EVA/wax/EG is promoted by wax. In turn, better thermal stability and lower peak value of the heat release rate were attained in EVA/wax/EG composites. The lower molecular weight, wax is able to penetrate between the EG layers and separate them resulting in better dispersion, hence improvement in the properties. To the best of our knowledge on PP/EVA/filler nanocomposites, there are no reports available on PP/EVA/wax/EG nanocomposites. Herein, the objective is to investigate how the morphology, thermal, and flow properties of the 80/20 PP/EVA blend vary in the presence of EG and wax, and establish a relationship between the properties of PP/EVA/wax/EG composite system to broaden its application. In literature, the dispersion of graphite prior incorporation into various polymers is enhanced by sonication in the presence of organic solvents or surfactants such as sodium dodecyl sulfate. The drawback of this method is that most organic solvents are toxic and prolonged sonication utilizes more energy along the process. Therefore, in the current study, EG and wax were incorporated without prior mixing to PP and EVA to investigate whether wax can improve the dispersion of EG in PP/EVA/wax/EG composites. An insight into how localization of EG particles and morphology of the blend composites affect the thermal properties, rheology and flammability of the composites is provided.
Materials and methodology
Materials
Paraffin wax was bought at Sasol (Sasolburg, South Africa). The wax is medium-soft-Fischer (M3 grade) with density of 0.90 g/cm3, melting at of 60°C and comprised of 99% straight chain hydrocarbons. Ethylene vinyl acetate 7350M grade was supplied by Formosa Plastics Corporation (Taipei, Taiwan). It is comprised of 18% vinyl acetate. Its melt flow index (MFI) at (190°C/2.16 kg) and density are 2.5 g/10 min and 0.941 g/cm3respectively. Polypropylene (HHR 102 grade) melting in the range of 160–165°C was purchased from Sasol (Sasolburg, South Africa). Its MFI at (190°C/2.16 kg) and density are 2 g/10 min and 0.901 g/cm3, respectively. Expandable graphite was purchased from Qingdao Kropfmuehl Graphite (Hauzenberg, Germany).
Fabrication of EG and melt processing of the samples
Codes and amounts of ingredients in blend composite samples.
Characterization of the blends and blend composites
Morphology
The microstructure of samples fractured in liquid nitrogen were analyzed with scanning electron microscope (SEM), model (JSM-7500F, JEOL, Japan) set at a voltage of 3 kV. The samples were made conductive by carbon sputtering prior SEM analysis. The state of localization and dispersion of EG in composites was determined with transmission electron microscope (TEM), model (JEM-2100, JEOL) from Japan. The blend composites were microtomed cryogenically to 80 nm sections with a diamond knife in a LEICA EM FC6 microtome. The EVA phase of the blend was stained with osmium tetroxide and analysis was done at a voltage of 200 kV.
Rheological properties
Rheology was carried at 190°C under nitrogen using Physica MCR501 (Anton Paar, Austria) rheometer with a 25 mm diameter parallel-plates configuration. A strain amplitude of 0.5% and frequency scans from 0.1 to 100 rad/s were applied.
Thermal properties
Differential scanning calorimetry (DSC) measurements were done to determine the crystallization temperature (Tc) and melting temperature (Tm) and enthalpy of melting (ΔHm during second heating. Samples were heated from −65 to 200°C at 10°C/min under nitrogen atmosphere with DSC-Q2000 instrument (TA Instruments, USA). The samples were cooled to −65 and reheated to 200°C at the previous rate. The DSC analysis for each sample was done on three independent runs.
Polarized optical microscopy
The growth of spherulites growth and morphology in molten state were studied with polarized optical microscope (POM). To be specific, thin films were placed between two glass cover slides and heated to 200°C at a rate of 10°C/min on hot stage Linkam THMS (Linkam Scientific Instruments, Ltd., Surrey, KT, UK), Samples were kept at 200°C for 5 min, and cooled to 140°C at 10°C/min, and held at 140°C to allow isothermal crystallization of the PP matrix for 30 min during which images were taken.
Thermogravimetric analysis (TGA)
A thermogravimetric analyzer, model (Perkin Elmer Pyris-1) was used for TGA analysis. Samples weighing 10 mg were heated at a heating rate of 10°C/min under nitrogen atmosphere from 25 to 800°C.
UL-94 vertical burning test
Flammability properties of the specimens with dimensions of 80 × 13 × 3 mm were investigated by UL-94 vertical burning tests. Test specimens were exposed to flame from the Bunsen burner for 10 s and then the Bunsen burner was removed to allow the specimen to continue burning.
Results and discussion
The morphological features of blend composites
Presented in Figure 1 are the micrographs of the blends and ternary composites. The SEM image for 80/20 PP/EVA (Figure 1(a)) shows dispersion of the minor EVA phase as droplets in the PP matrix. The two-phase morphology of the 80/20 PP/EVA blend suggests that the blended polymers are immiscible. In Figure 1(b), the dispersed EVA droplets are easily visible indicating that the interfacial tension between PP and EVA is high when compared to Figure 1(a).
17
This suggests that the presence of 10 wt.% wax in PP/EVA/Wax blend promoted heterogeneity. Despite their non-polar structures, PP and wax are generally immiscible because PP crystallizes in a helical form in which there are three monomer units per turn of a helix,
18
and wax likely crystallizes in a planar zig zag form.
19
The droplet size is reduced when EG is incorporated to the blend of PP and EVA (Figure 1(c)) and PP/EVA/Wax/EG (Figure 1(d)). Expanded graphite consists of the stacks of the nanosheets and it has a layered structure that is similar to layered silicates. Because of its porous structure, polymer chains may enter in some of the EG layers. Later, it will be shown that the EG particles are localized inside the minor EVA phase, while some are found at the interface. Therefore, EG particles at the interface of PP and EVA for the blend composites enhanced compatibility, hence a reduction of EVA droplet size. SEM micrographs of surface morphology for (a), (b) blends, and (c), (d) blend composites.
The TEM micrographs in Figure 2 show the EVA minor phase appears dark and forms a droplet morphology within the lighter PP matrix. The darker colour of EVA is due to its relatively higher electron density than PP. The TEM micrograph of the PP/EVA/EG composite (Figure 2(a)) reveals localization of EG particles inside the darker minor EVA phase as agglomerates, while some EG particles also localize at the interface of blended polymers. The preferred localization of EG particles in minor EVA phase as opposed to the PP phase can be attributed to two effects. Firstly, EVA has low melting temperature than PP. Therefore, during processing the EG particles would favour localizing in the phase which has lower melting temperature. Secondly, EVA has lower viscosity than PP and it is much easier for EG particles to localize into a less viscous phase than into a more viscous PP phase during processing. In Figure 2(b), wax is represented by the lighter phase whereas the darker phase represents EVA. The EG particles still favour localization inside the EVA phase (Figure 2(b)) irrespective of the existence of wax, but some EG particles are found at the interface and in the wax phase. The EG particles show a tendency of aggregating suggesting that they have a higher affinity for each other than EVA, and the PP matrix. The melt processing method used for preparing the composites was not very effective in breaking up the aggregates. TEM micrographs of the blend composites. (a) PP/EVA/EG. (b) PP/EVA/Wax/EG.
Rheological properties analysis
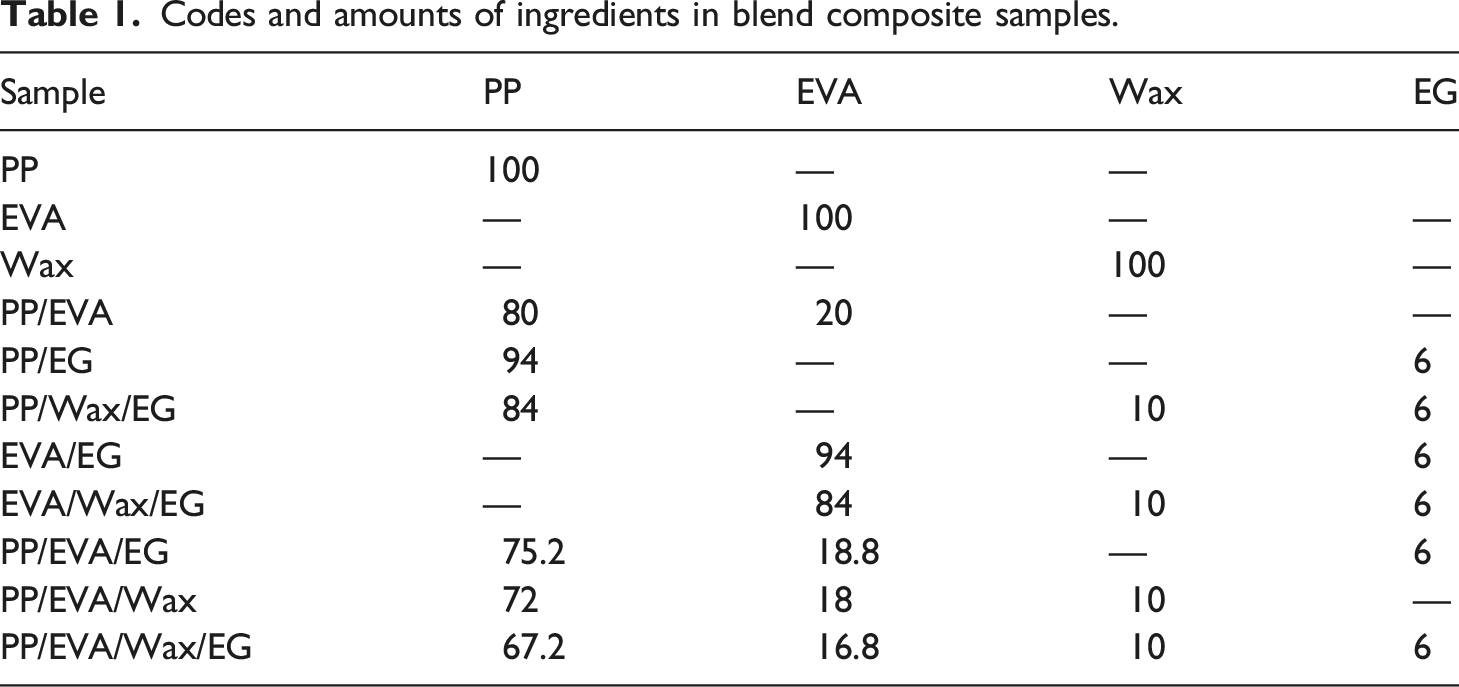
Depicted in Figure 3 are the plots of complex viscosity and storage modulus ( (a), (b) Complex viscosity versus ω, and (c), (d) 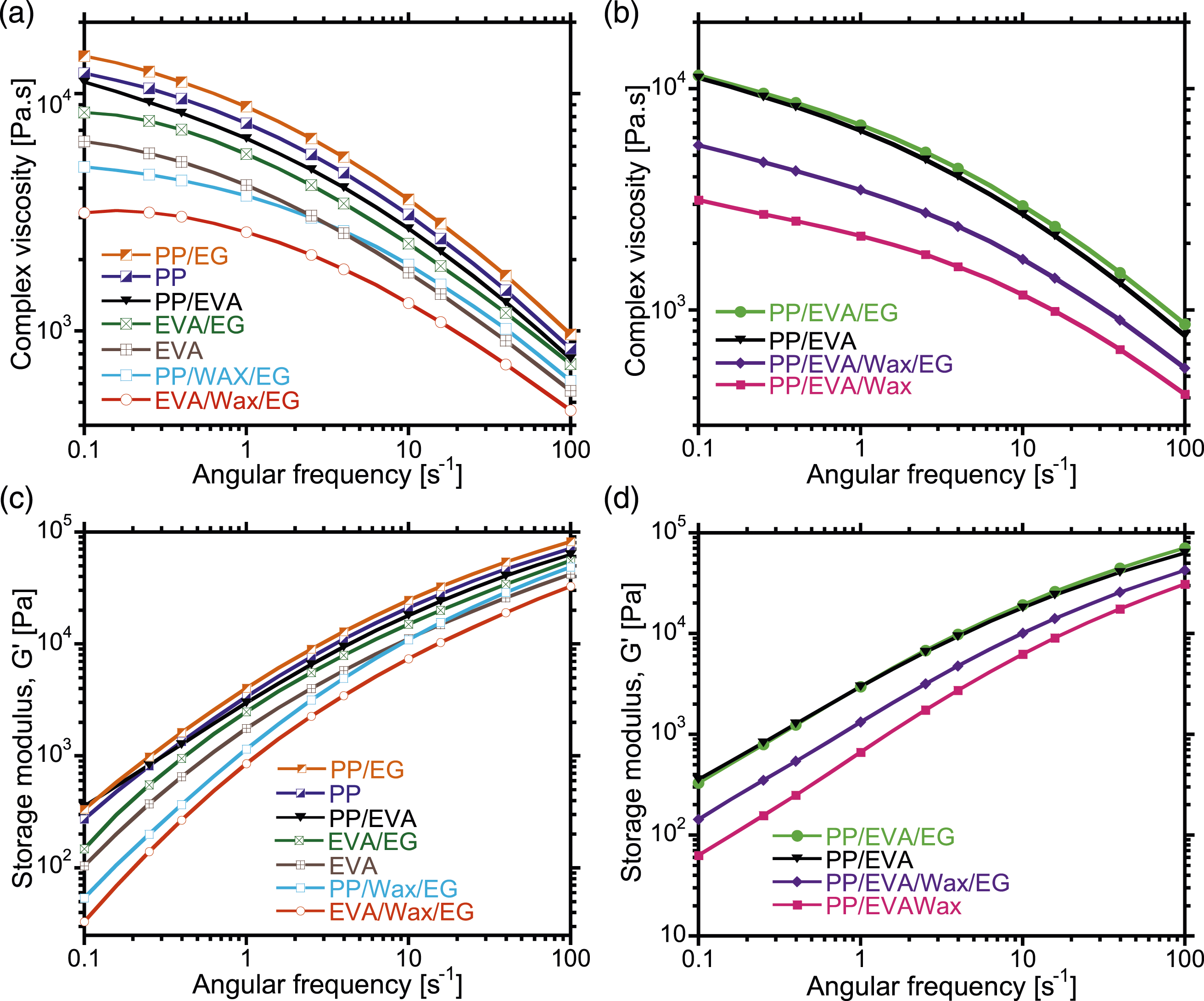
In Figure 4, the rheological behavior of neat PP was viscoelastic-liquid (i.e., Crossover frequencies of neat polymers, blends and EG-filled composites. The 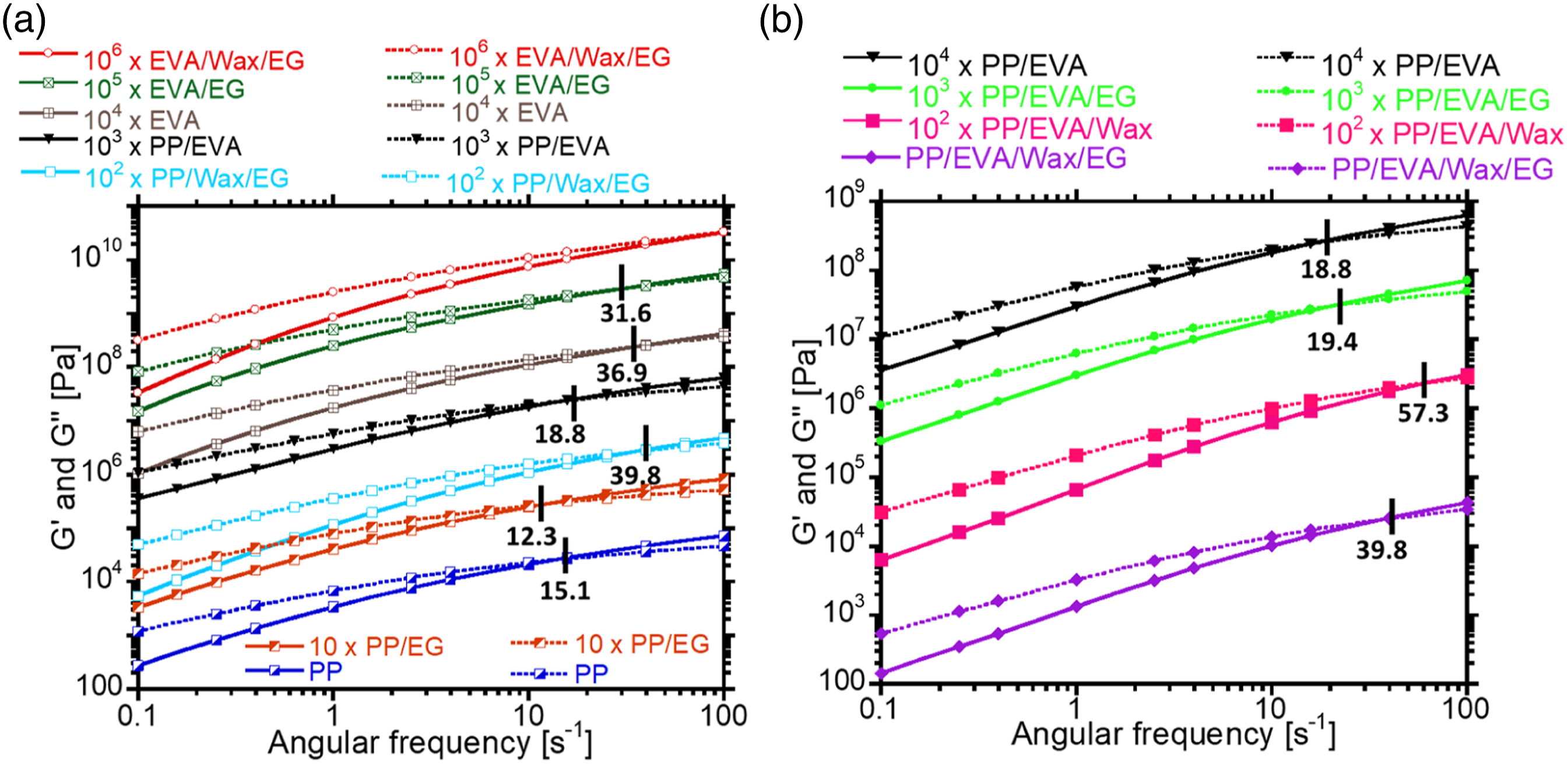
In rheology, the Cole-Cole plots (where Eta″ = G′/ω and Eta′ = G″/ω) are useful for investigating homogeneity or miscibility between the blended components. It is seen in Figure 5(a) that the Cole-Cole plots for both pristine EVA and PP have a semi-circular configuration, a characteristic of homogenous single-phase system.
22
The PP/EVA blend deviates from semi-circular configuration of pristine EVA and PP in such a way that a tiny tail occurs at higher The cole-cole plots of (a) non-filled samples, (b) blends and ternary composites.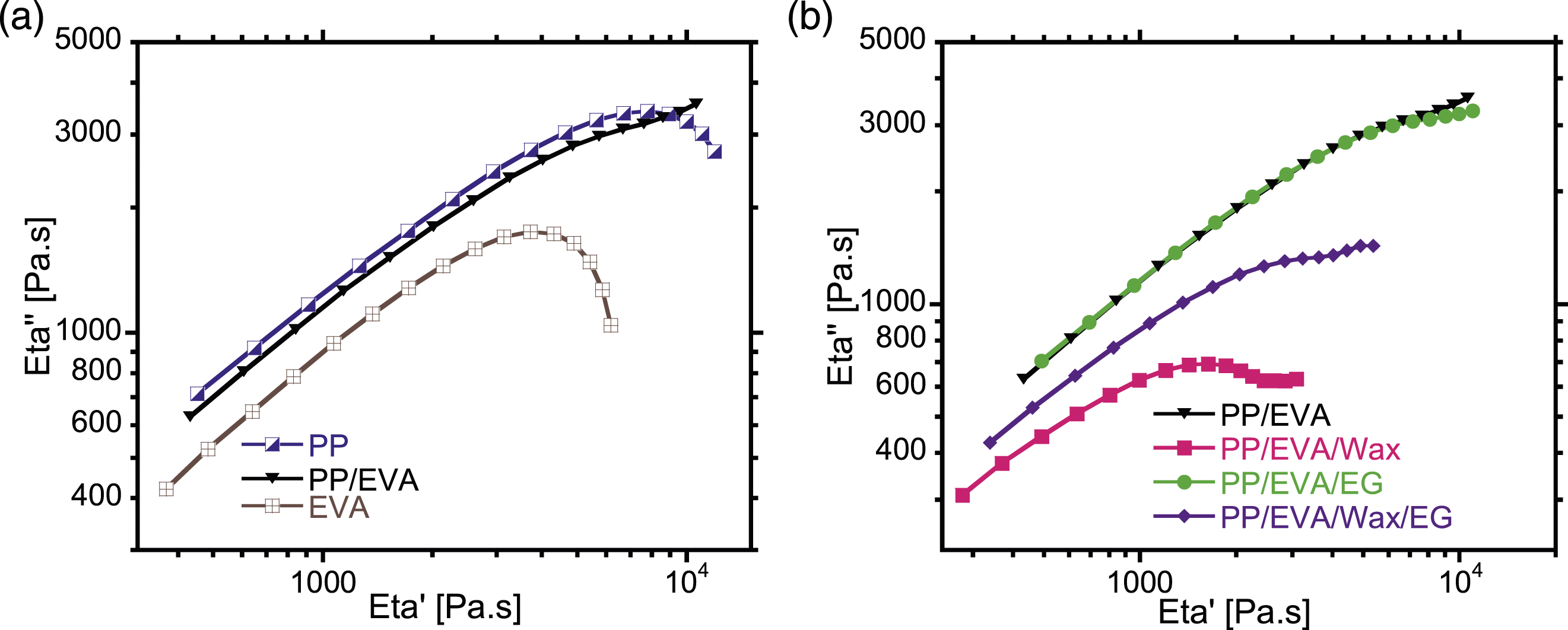
Thermal analysis
The second heating and cooling profiles of the samples are shown in Figure 6 and Table 2. In Figure 6(a), PP and EVA have melting temperatures ( DSC thermograms for (a)-(b) heating, and (c)-(d) cooling of the samples. Melting and crystallization parameters obtained with DSC.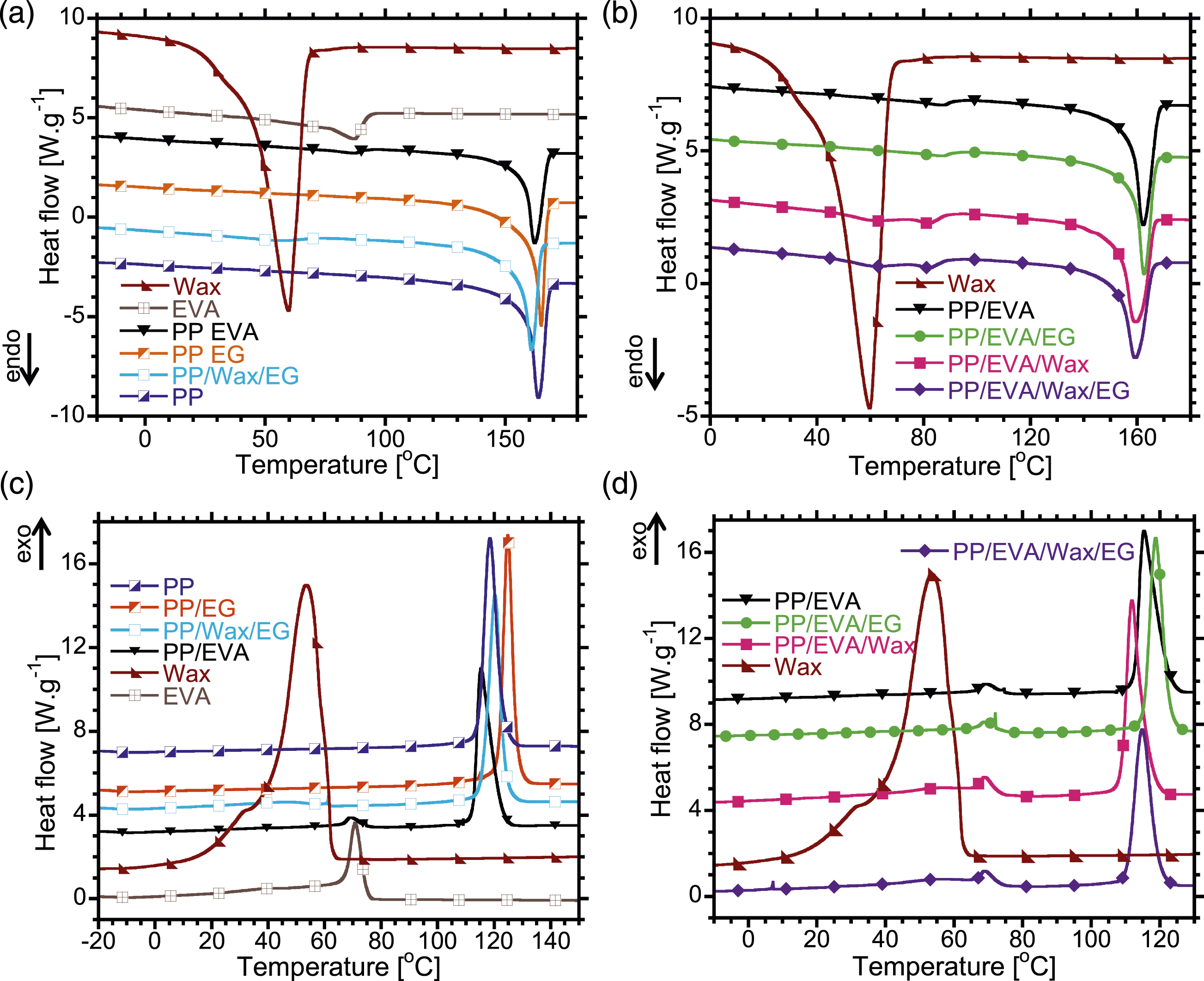
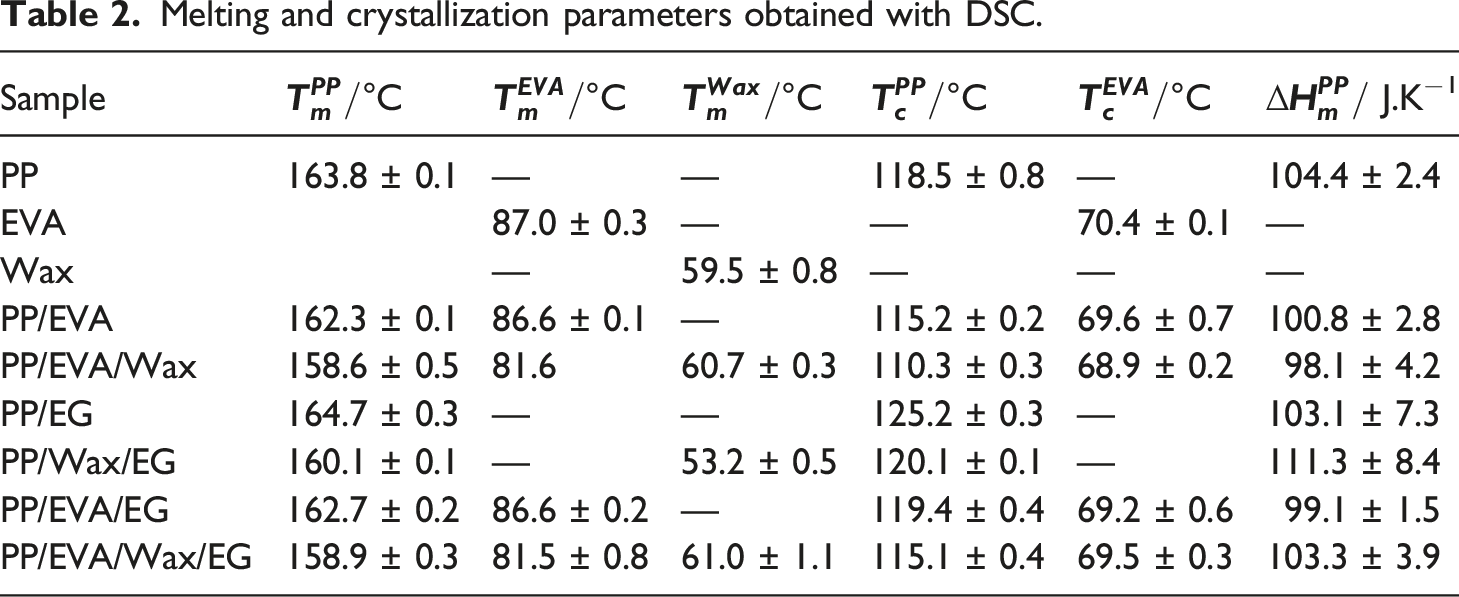
In Figure 6(b), the PP/EVA blend has a small peak at 86.6°C associated with melting temperature of EVA (
In Figure 6(c), the crystallization temperature of EVA
In Table 2, the
In Figure 7(a), the larger spherulites of PP may suggest faster spherulitic growth rate. However, this may also be associated with the nucleation ability of the material. There is a pronounced increase of the number of spherulites when 6 wt.% EG is added to PP (Figure 7(b)), whereas their size is reduced indicating nucleation by EG. The number of spherulites reduces per unit area when wax is added to PP/EG (Figure 7(c)) supporting the previous observations, which highlighted that wax delays crystallization in PP. Fewer spherulites are seen for the PP/EVA blend (Figure 7(d)) in comparison to neat PP. The decline in the number of spherulites is due to existence of melted EVA, which acts as a diluent, and disturb crystallization of PP during isothermal crystallization at 140°C. However, wax present in the blend (Figure 7(e)) also disturbs the crystallization of PP further, resulting in small and imperfect crystals. Many and small spherulites form when EG is added to the PP/EVA blend (Figure 7(f)) suggesting nucleation of PP by EG. The number of spherulites per unit volume is slightly lower in PP/EVA/EG in comparison to PP/EG composite and due to EVA, which disturbs crystallization of PP. There is lesser number of spherulites per unit volume for the PP/EVA/wax/EG when compared with PP/EVA/EG composite. Existence of wax in PP/EVA/wax/EG composite delays crystallization of PP. Therefore, it is deduced that although wax existence improves the processability of investigated samples, it also reduces nucleation of PP which in turn increases the cycle time during injection molding process. Therefore, EG is a promising candidate for mitigating against the effect of wax in reducing nucleation of PP. POM images of samples crystallized isothermally at 140°C for 30 min. (a) PP. (b) PP/EG. (c) PP/Wax/EG. (d) PP/EVA. (e) PP/EVA/Wax. (f) PP/EVA/EG. (g) PP/EVA/Wax/EG.
Thermal stability of the blend and blend composites
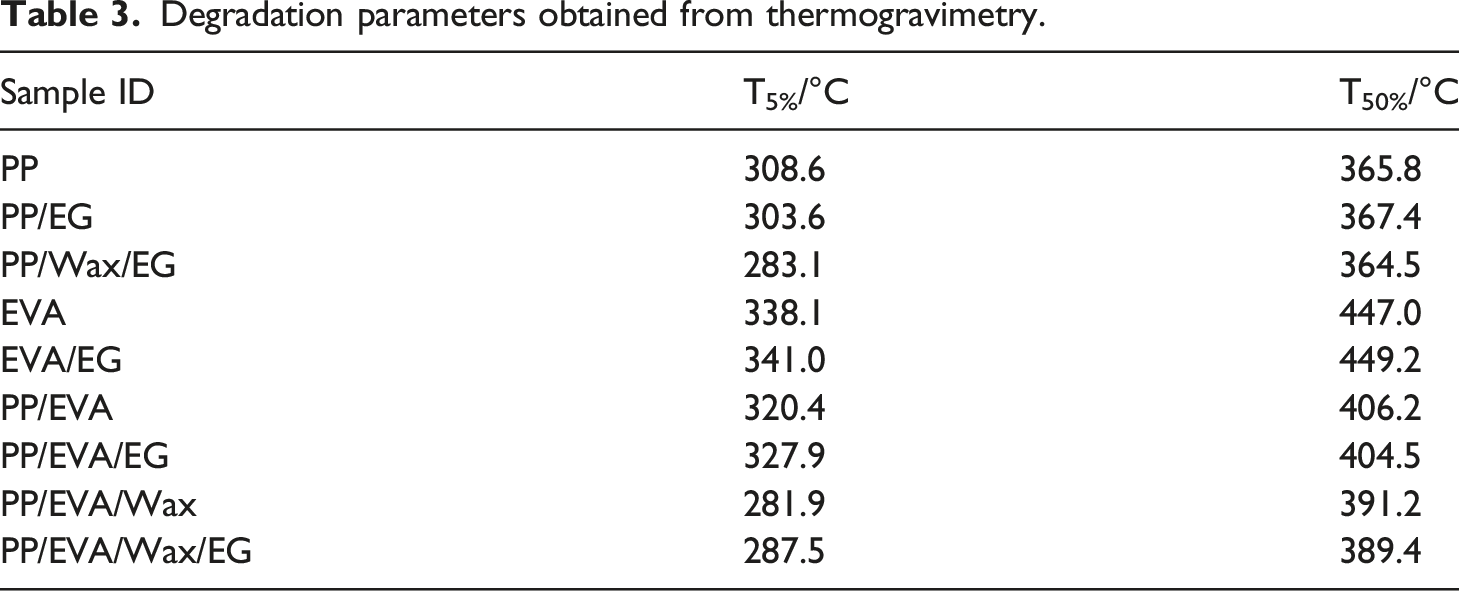
The thermal degradation curves of PP, PP/EG, PP/Wax/EG, EVA, and EVA/EG composite are shown in Figure 8(a). A summary of the decomposition temperatures of the samples are tabulated in Table 3. The thermal degradation curves of the neat PP, PP/EG, and PP/Wax/EG composite show a single degradation step, which is mainly due to thermal degradation of PP main chain. PP/EG composite has a slightly lower T5% than pristine PP suggesting a relatively lower thermal stability for the PP/EG composite. Kaczor et al.
24
reported a decline in thermal stability of PLA when graphite fillers were incorporated. They ascribed the findings to the high thermal conductivity of graphite which facilitated heat transfer in the sample volume, thus reducing the thermal decomposition temperature. The PP/Wax/EG composite has lower thermal stability compared to pristine PP and PP/EG composite. Wax has a low molecular weight. Therefore, the short chains of wax generally evaporate at a lower temperature than the degradation temperature of PP. Introduction of short wax chains in the PP/EG composite promotes degradation to occur earlier, hence lower thermal stability for the PP/Wax/EG composite. It is observed in Figure 8(a) that pristine EVA and the EVA/EG composite show a two-step degradation profile. The two-step degradation profiles are related to deacetylation and the decomposition of the main chain.
25
The EG fillers in EVA/EG moderately enhanced thermal stability of EVA. This is attributed to thermal energy being initially absorbed by the EG filler such that sufficient energy to initiate the degradation of EVA only becomes available at slightly higher temperatures.
16
Pristine PP has less thermally stable than pristine EVA and this is clarified by the T5% and T50% values for these two polymers in Table 3. T5% and T50% of PP increase when EVA is added signifying improvement of the thermal stability. Introduction of EG into PP/EVA slightly enhanced the thermal stability further. Earlier, the TEM micrograph (Figure 2(b)) showed that EG fillers localized inside the minor EVA, and at the interphase of PP and EVA. Therefore, such localization of EG enabled confinement of heat which resulted in less heat becoming available to initiate the degradation of the blend at low temperatures, and sufficient energy became available at higher temperatures to initiate degradation of PP/EVA blend. In addition, the surfaces of EG were likely to adsorb the volatile degradation products, thus retarding their diffusion out of the PP/EVA/EG composite which resulted in the T5% being observed at a slightly higher temperature. Thermal stability of PP/EVA is lowered when wax is introduced to form PP/EVA/wax blend, and this is attributed to a lesser thermally stable wax which evaporates at low temperatures than the degradation temperature of PP/EVA. However, PP/EVA/Wax/EG composite has a relatively better thermal stability compared with PP/EVA/Wax due to EG particles which retarded diffusion of volatile decomposition products. It is noted that despite containing equal amounts of EG, the PP/EVA/Wax/EG has lower thermal stability compared with PP/EVA/EG composite. The PP/EVA/Wax/EG composite contains wax, whereas wax is absent in the PP/EVA/EG composite. Therefore, short chains of wax evaporate at lower temperature than the onset degradation temperature of PP/EVA/Wax/EG composite influence degradation to occur earlier. Hence a lower thermal stability of PP/EVA/Wax/EG composite. Thermogravimetry curves of the samples. Degradation parameters obtained from thermogravimetry.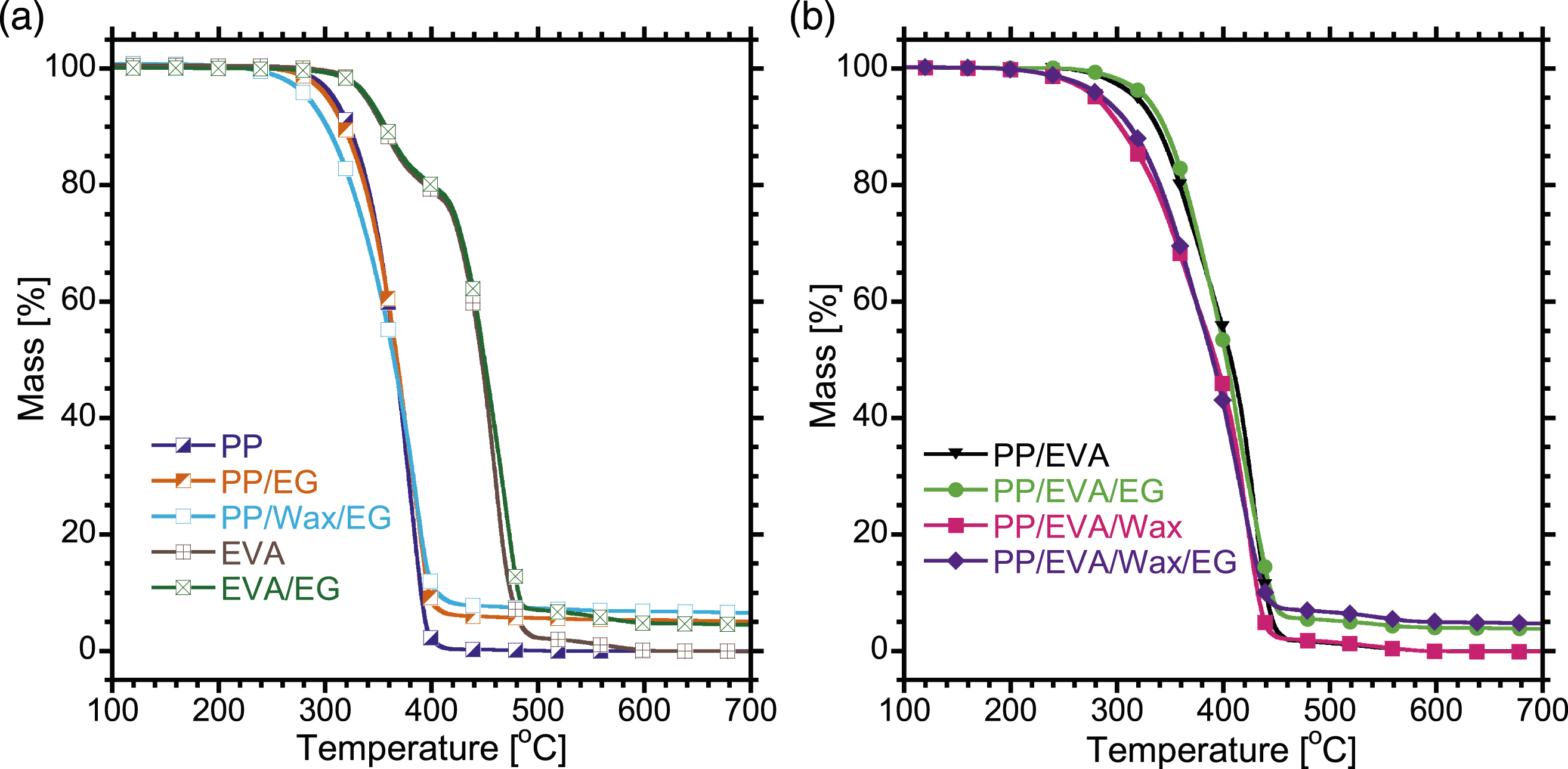
Fire behaviour of samples
UL-94 was done to investigate the dripping behavior of the samples. After ignition for the first 10 s in all samples shown in Figure 9, the fire propagated, and the samples were burnt out. Upon catching fire, the PP/EVA and PP/EVA/Wax blend dripped continuously, whereas dripping was significantly reduced in PP/EVA/EG and PP/EVA/Wax/EG composites. The total flame time (TFT) and drip flame time (DFT) for neat PP/EVA blend were recorded at 98.5 s and 11.5 s, respectively. Upon incorporation of wax for the PP/EVA/Wax blend, TFT was lowered to 73.5 s, whereas DFT is similar at 12 s. A lower TFT for PP/EVA/Wax blend is attributed to its lower viscosity caused by the presence of wax as previously observed in the rheological section. A high melt viscosity is desirable because it reduces the probability of dripping.
26
The DFT of the blend is increased to 56 s when EG is incorporated to the neat blend to form the PP/EVA/EG composite. A longer DFT for the PP/EVA/EG composite is due to EG which formed a char layer and in turn suffocated the fire by disrupting the heat and oxygen from penetrating the PP/EVA blend, hence an improved DFT. DFT of the PP/EVA/EG composites is lowered to 48 s when wax is incorporated to form the PP/EVA/Wax/EG composite. Wax lowered viscosity and promoted dripping. Since all the samples in Figure 9 caught fire after 10 s of exposure to fire and continued burning, they are therefore given a V-2 rating. The use of polymer composites in electronic applications, for example electrical cables, requires a material to have a V-0 rating. In V-0 rating, the burning stops within 10 s after applying a flame for 10 s to a test specimen. Therefore, our future study will be based on investigating the synergistic effects of EG and diammonium phosphate on flammability of PP/EVA and PP/EVA/Wax phase change blends. Flammability photos of (a) PP/EVA, (b) PP/EVA/Wax, (c) PP/EVA/EG, (d) PP/EVA/Wax/EG composites.
Conclusions
Herein, binary, and ternary PP/EVA/Wax/EG composites were prepared through melt processing. The results demonstrated that the presence of paraffin wax in the PP/EVA blend and PP/EVA/EG composite improved the processability. It was further realized that paraffin wax also induced plasticization of the major PP phase and delayed its crystallization. Overall, the EG particles acted as nucleating agents and promoted earlier crystallization of PP in PP/EG, and PP/EVA/Wax/EG composite when compared to pristine PP, and PP/EVA/Wax, respectively. In addition, it is deduced that paraffin wax had little influence on the dispersion state of EG as the majority of EG particles had higher affinity for each other. Moreover, paraffin wax was noted to promote the mobility of the polymeric chains in PP/EVA/wax blend and PP/EVA/wax/EG composite as observed from the Cole-Cole plots. In the subsequent study, more attention shall be directed to enhancing dispersion of EG in the blend composites and the flammability. To achieve better dispersion, we intend to simultaneously mix both the EG, and paraffin wax prior to incorporation into the blend. Furthermore, an intumescent flame retardant (IFR) will be incorporated, and synergy effect of EG and IFR on flammability of PP/EVA and PP/EVA/Wax blend will be studied.
Footnotes
Acknowledgements
We acknowledge the national research foundation, South Africa (NRF) and Council for Scientific and Industrial Research (CSIR), South Africa (086ADMIN).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
