Abstract
Numerous studies have reported the use of 3D-printed biocompatible intramedullary (IM) pins of various metals, alloys, and composite materials for a variety of orthopedic requirements in canines. One of the limitations of these IM pins is that they are non-resorbable, and required surgical procedures to be followed for their removal after performing the required function in the subject. Some studies have been reported on polyvinylidene fluoride (PVDF) composite-based smart IM pins that are partially absorbable. But hitherto less has been reported on the compressive properties of such 3D printed PVDF composite matrix. This study reports the compressive properties of 3D printed (by using fused filament fabrication (FFF) process) PVDF composite (90% PVDF-8% Hydroxyapatite (HAp)-2% Chitosan (CS) by wt.) for a partially absorbable implant for canines. The result suggested that the best setting for the FFF process are nozzle temperature (NT) 235°C, printing speed (PS) 60 mm/s, and raster angle (RA) 45° for the compressive properties (peak load (PL) 1321 N, break load (BL) 1188.9 N, strength at peak (SP) 10.83 MPa, and strength at break (SB) 9.75 MPa). Furthermore, analysis of the PVDF composite (at 10 N load, 300 s, and 60 r/min) outlined wear of 314 µm, friction force of 1.2 N, and a coefficient of friction of 0.14 was observed. The results are supported by the morphological analysis (based on scanning electron microscopy (SEM), % of porosity, grain size No.) of the fracture site.
Keywords
Introduction
The removal of an intramedullary (IM) pin is typically considered when the fracture has healed completely and the canine can sustain self-weight during running or galloping. In such cases, the IM pin is no longer needed for stabilization.
1
Several times the IM pin comes out through the distal end of the femur bone during running or galloping then the IM pin needs to be removed, so there is a need for extra surgery for the removal of the IM pin.
2
To remove the IM pin, a post-operative surgery which is usually time-consuming needs to be performed by the veterinary surgeon. This surgery is an extra burden on the subject, and some complications may occur after post-operative surgery like, infection, nerve injury, a reaction to anesthesia, etc.1,2 The metallic implant put inside the body provides initial stability, allowing for proper alignment and fixation of the implant/scaffolds. The metallic implants are non-biodegradable and have stress shielding issues. For addressing the stress shielding problem, some studies have reported the use of hollow implants.
3
The bioabsorbable thermoplastic components can play a crucial role in the implant’s degradation process.
4
As the thermoplastic material degrades over time, it gradually transfers the load-bearing capacity to the surrounding bone or tissue, allowing for natural healing and remodeling. The degradation kinetics can be tailored by adjusting the composition, molecular weight, or blending of different thermoplastic materials.3,4 This controlled degradation facilitates the transfer of mechanical forces and supports the natural healing process. The bioabsorbable thermoplastics composite gradually degrades, providing a favorable environment for tissue ingrowth and remodeling. This combination of stability and controlled degradation promotes bone healing and integration, reducing the risk of implant-related complications.5,6 In the recent past, an attempt was made to 3D print a partially bioabsorbable implant of PVDF composite for orthopedic application.
7
This work is the extension of the previously reported study in which the β-phase of PVDF composite was ensured for acceptable mechanical properties. The selection of the composition (90%PVDF-8%HAp-2%CS) was based on the formation of more β-phase required for better mechanical properties in PVDF composite. It has been reported that when the load is applied to the PVDF (CH2CF2) materials, the crystal changes the phase (i.e., from α to β). In β-phase, the mechanical properties are enhanced due to the arrangement of the crystals as shown in Figure 1. α-to-β phase transformation during applied load in PVDF materials.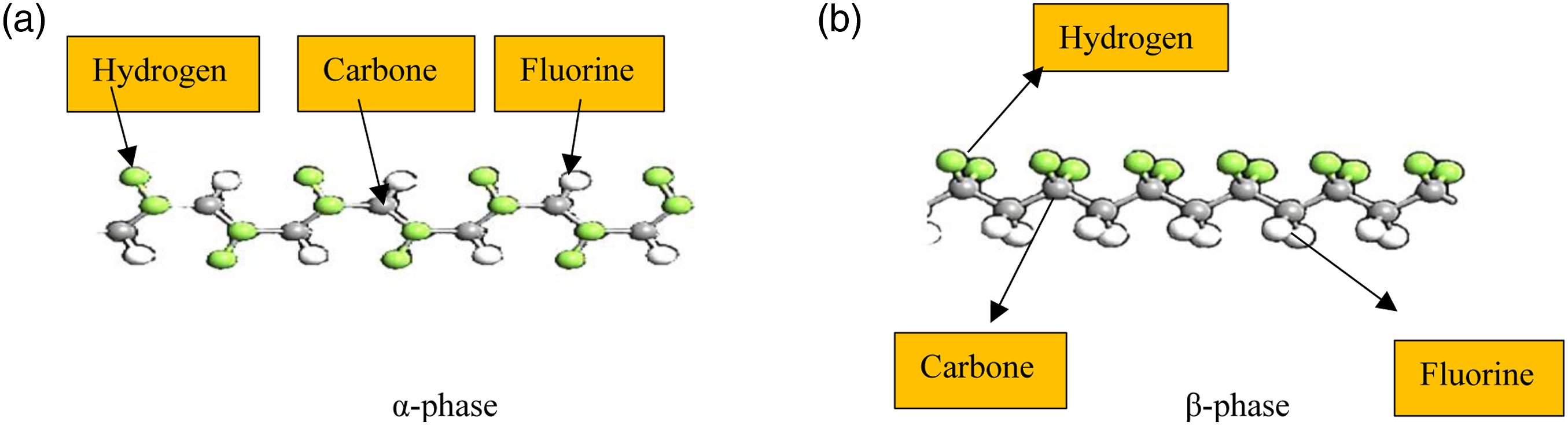
Because PVDF contains 45%–55% crystallinity, a previous study of the crystal arrangement of several PVDF composites (with varying wt. percentages of PVDF-HAp-CS) was performed by using XRD analysis. The formation of β-phase was also supported by the crystallinity of 90%PVDF-8%HAp-2%CS observed as 37.8% through differential scanning calorimetry (DSC) analysis. 7 The compressive strength of materials is a critical parameter during the material selection and design optimization process for implants/scaffolds, 8 which may be tailored as per the subject requirements.
Numerous researchers consider various materials, such as metals, ceramics, polymers, and composite materials, with different compressive strength profiles for implant/scaffold applications. 9 By evaluating the mechanical properties, including compressive strength, researchers can identify materials that meet the specific requirements of the implant/scaffold, ensuring optimal performance and patient outcomes.9,10 The compressive strength requirements for implants/scaffolds can vary depending on individual patient characteristics, such as age, weight, and specific medical conditions. 11 The 3D printing process can customize and personalize implants/scaffolds to match the patient’s needs. 12 Compressive strength data can aid in the development of patient-specific implants/scaffolds by tailoring the material properties to meet the unique mechanical demands of each patient, enhancing treatment outcomes and patient satisfaction. Various article report on biomimetic and tissue-mimicking materials that closely replicate the mechanical properties of natural bone. Compressive strength plays a crucial role in achieving biomimetic behavior, where the implant/scaffold mimics the mechanical properties of the surrounding bone. 13 By matching the compressive strength of the natural bone, the implant or scaffold can better distribute loads and prevent stress shielding, promoting better integration and reducing complications. 14 3D printing enables the production of patient-specific implants by using patient imaging data to create personalized designs. 15 This customization capability is particularly advantageous for partially absorbable implants, as it allows for precise tailoring of the implant’s shape, size, and internal architecture to match the patient’s anatomy. 16 This can lead to improved fit, functionality, and patient outcomes. 3D printing techniques, such as selective laser melting (SLM) or fused deposition modeling (FDM)/FFF can fabricate implants with complex geometries and intricate internal structures. This capability is beneficial for partially absorbable implants, as it allows for the creation of porous and meta structures within the implant. 17 These structures can enhance osseointegration, facilitate nutrient and oxygen exchange, and promote tissue ingrowth, ultimately improving the implant’s long-term stability and performance. The mechanical properties of partially absorbable implants are essential for their stability and load-bearing capacity during the absorption period. 3D printing techniques offer the ability to optimize the implant's mechanical performance by adjusting parameters such as printing speed, nozzle temperature, raster angle, infill density, and material composition. 18 This allows for tailoring the implant’s compressive strength, elastic modulus, and fatigue resistance to withstand physiological loads while maintaining structural integrity during the absorption process. 19 3D printing techniques provide flexibility in terms of manufacturing complex shapes, porous structures, and patient-specific designs. They offer opportunities for incorporating bioactive agents, controlling the distribution of HAp-CS particles, and optimizing the composite’s mechanical properties and degradation behavior. 20
These reinforcements are made up of biocompatible materials such as ceramic phosphocalcic, which is made up of 3-hydroxybutyrate-co-3-hydroxyvalerate (PHBV) polymer with a weight fraction of 40%/60% and tricalcium phosphate (β-TCP) and dehydrated 3-hydroxybutyrate-co-3-hydroxyphosphate (DCPD). 21 Biomaterials called “bone substitutes” are used in surgical therapy to replace missing or broken bones in numerous circumstances. 22 Based on the previously reported studies for thermal stability, and mechanical qualities many thermoplastic composites are commercially manufactured at a laboratory scale. 23 In one of the reported studies, N, N-dimethylacetamide, PVDF, and polyethylene glycol (PEG) were dissolved with different PEG concentrations in the homogenous mixture. To create films, the mixture was painted on glass plates that were primed with acrylic resin 24 . By using a solution mixing and hot-pressing technique, composites of three distinct carbon nanotubes (CNTs) made of PVDF were created. 25 The composites of PVDF, polyurethane (PU), and polyaniline (PANI) as electromagnetic infrared (EMI) shielding materials were examined by some researchers. 26 In another study, a twin-screw extruder (TSE) was used to prepare the filament for FFF. The TSE process parameters for producing smart material strands with a uniform dispersion of barium titanate (BTO) and graphene (Gr) in the PVDF matrix were investigated. 27
The PVDF composite prepared by reinforcement of HAp, and CS has been reported as one of the acceptable biocompatible and bioabsorbable solutions. 7 PVDF is a biocompatible and mechanically robust polymer known for its excellent wear resistance and stability. HAp, a natural component of bone, provides bioactivity and promotes osseointegration. 28 The presence of HAp within the composite promotes bioactivity, allowing for improved tissue integration with the implant. HAp provides a favorable surface for cell attachment and proliferation, facilitating the formation of new bone tissue and enhancing the implant's stability. The composite’s bioactivity can be further enhanced by incorporating bioactive molecules or growth factors, which can be released from the implant over time to promote tissue regeneration. CS, derived from chitin, possesses antimicrobial properties and facilitates cell adhesion.29,30 The combination of these materials in the composite offers a unique set of properties suitable for partially bioabsorbable implants. PVDF contributes to the composite’s mechanical strength, allowing it to withstand physiological loads and maintain structural integrity during the implantation period. The composite’s mechanical strength can be tailored by adjusting the composition and processing parameters during fabrication. PVDF-HAp-CS composite is partially bioabsorbable, 31 while PVDF alone is non-biodegradable, CS is biodegradable and can be gradually absorbed by the body over time. This allows for controlled degradation of the composite, reducing the long-term presence of foreign materials in the body. The degradation rate can be influenced by adjusting the CS content in the composite, allowing for customization based on the specific requirements of the implantation site. 32
The literature review reveals that numerous studies have reported the use of 3D-printed biocompatible IM pins of various metals, alloys, and composite materials for a variety of orthopedic requirements in canines. One of the limitations of these IM pins is that they are non-resorbable, and required surgical procedures to be followed for their removal after performing the required function in the subject. Some studies have been reported on PVDF composite-based smart IM pins that are partially absorbable. But hitherto less has been reported on the compressive properties of such 3D printed PVDF composite matrix. This study reports the compressive properties of PVDF composite (90% PVDF-8% HAp-2%CS by wt.) fabricated by FFF for a partially absorbable implant for canines.
Materials and methods
The PVDF granules of extrusion grade were procured from Solvay, Gujarat (India). The pellet has a density of 1.75 g/cm3, a melting point between 170 and 180°C, and a glass transition temperature of −40°C. The Marine Hydrocolloids, Kochi, Kerala, India, provided the CS (pH 7.0–9.0) and HAp (colorless and brittle). The adopted methodology for this study is shown in Figure 2. Methodology for the optimization of FFF parameters.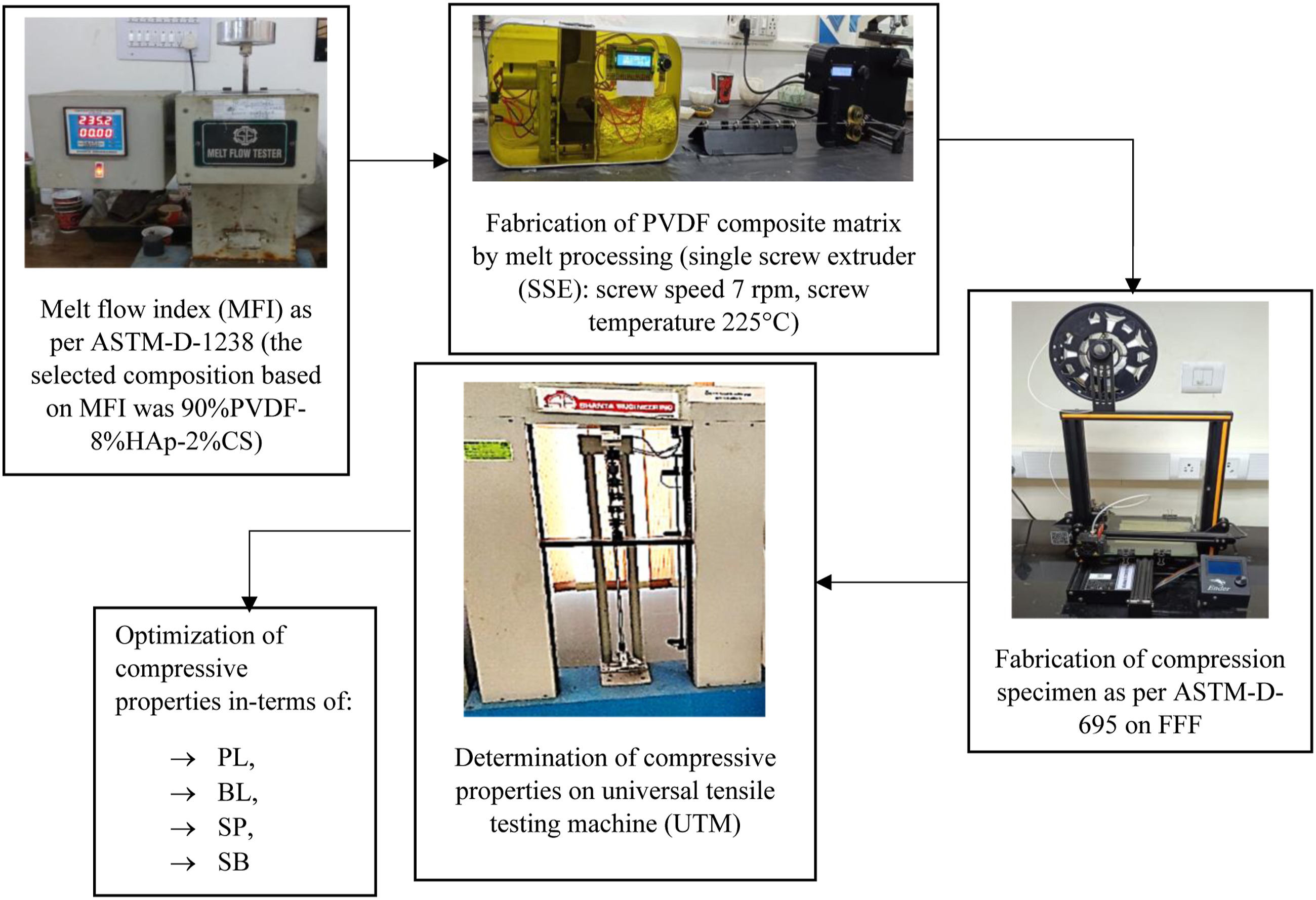
Experimentation
Factor and level for optimization of FFF.

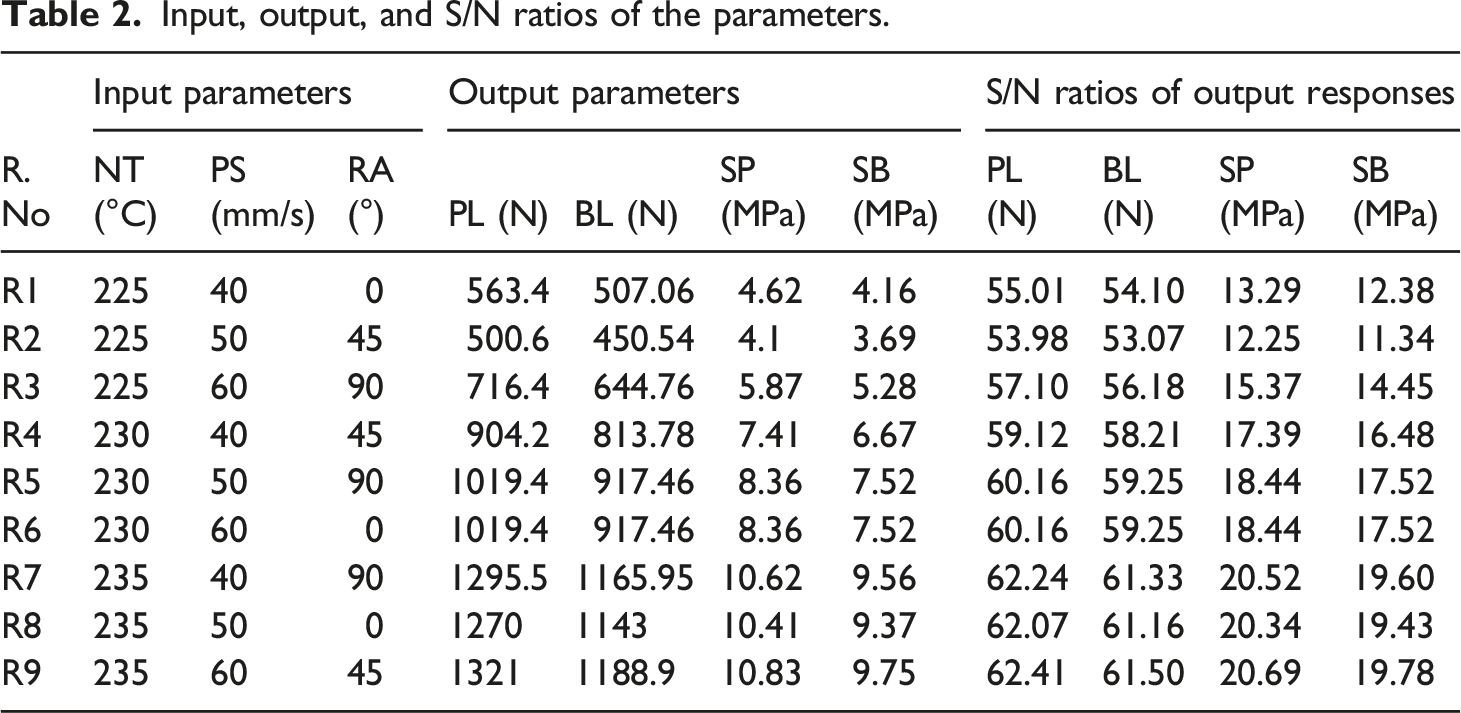
Input, output, and S/N ratios of the parameters.
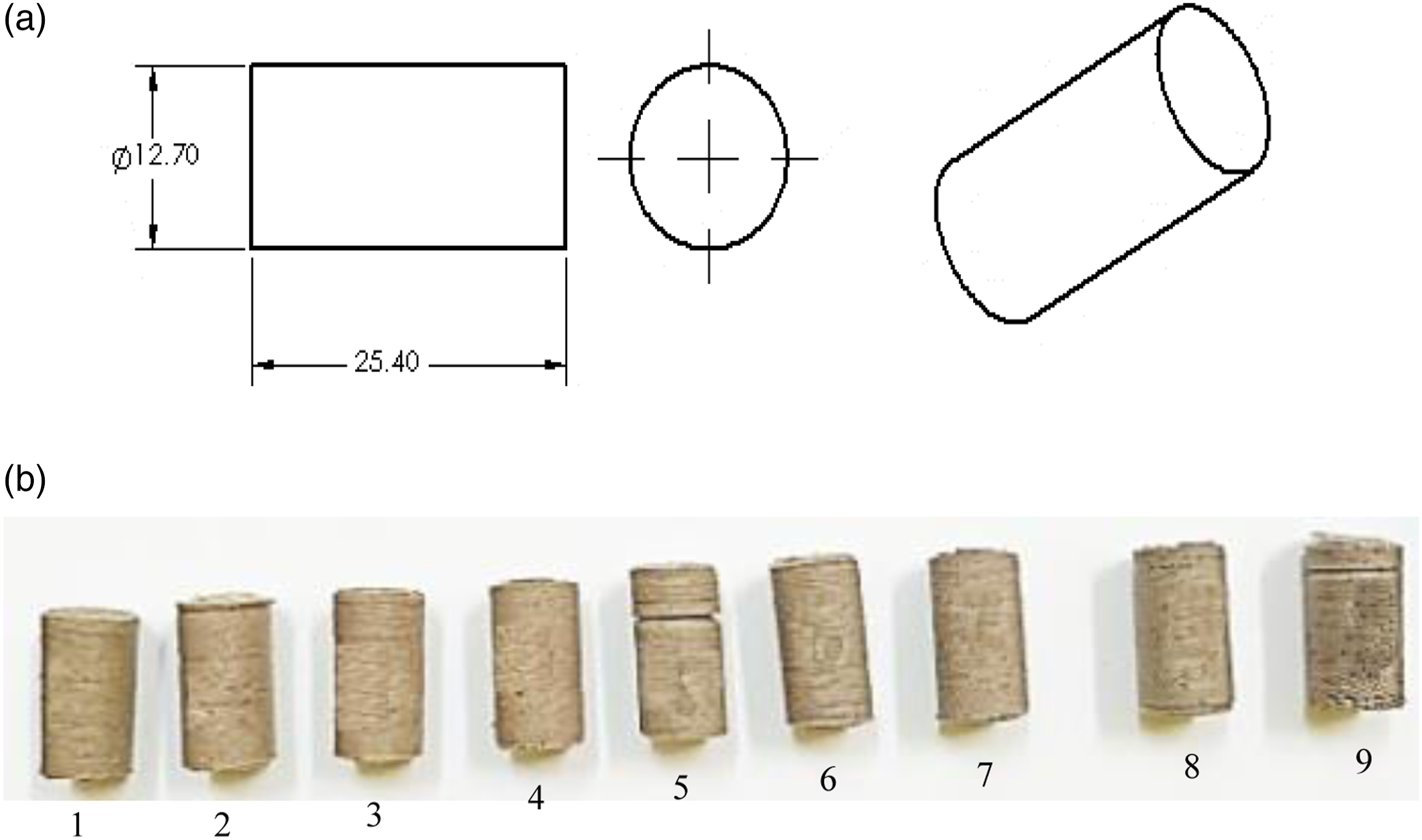
(a) Compression specimen, (b) 3D printed compression specimens.
To comprehend the mechanism causing the material's deformation, stress-strain curves have been plotted (Figure 4). The compressive test was performed at UTM setup (Make Shanta Engineering, Pune, India) for the strain rate 20 mm/s, span distance 25 mm, and area of cross-section of the specimen 126.67 mm2. Stress versus strain graphs.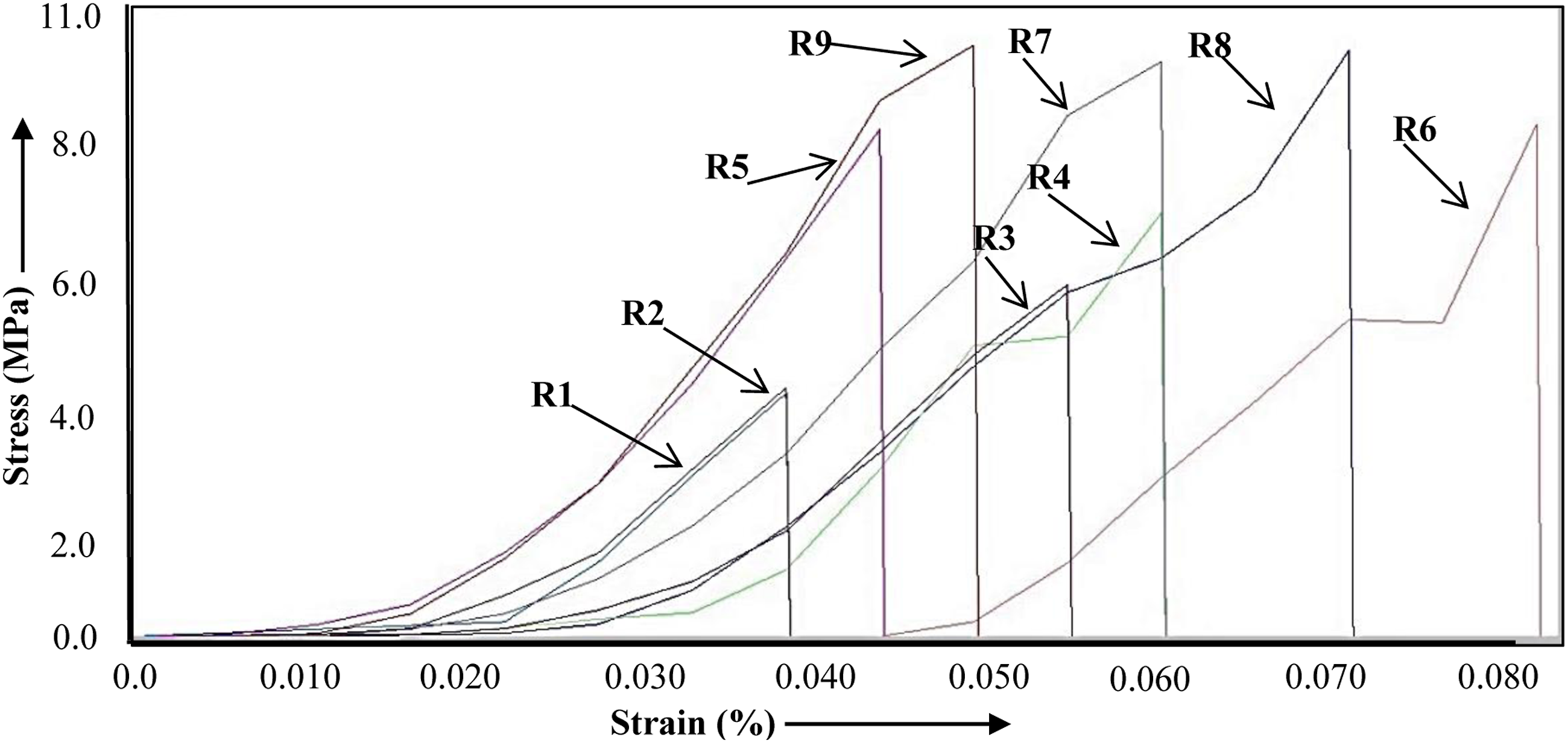
The gathered data was statistically analyzed using the Minitab-19 software. The signal-to-noise (S/N) ratio was used to examine the observed responses. S/N ratios for PL, BL, SP, and SB were calculated for the “larger is better” case.
Results and discussion
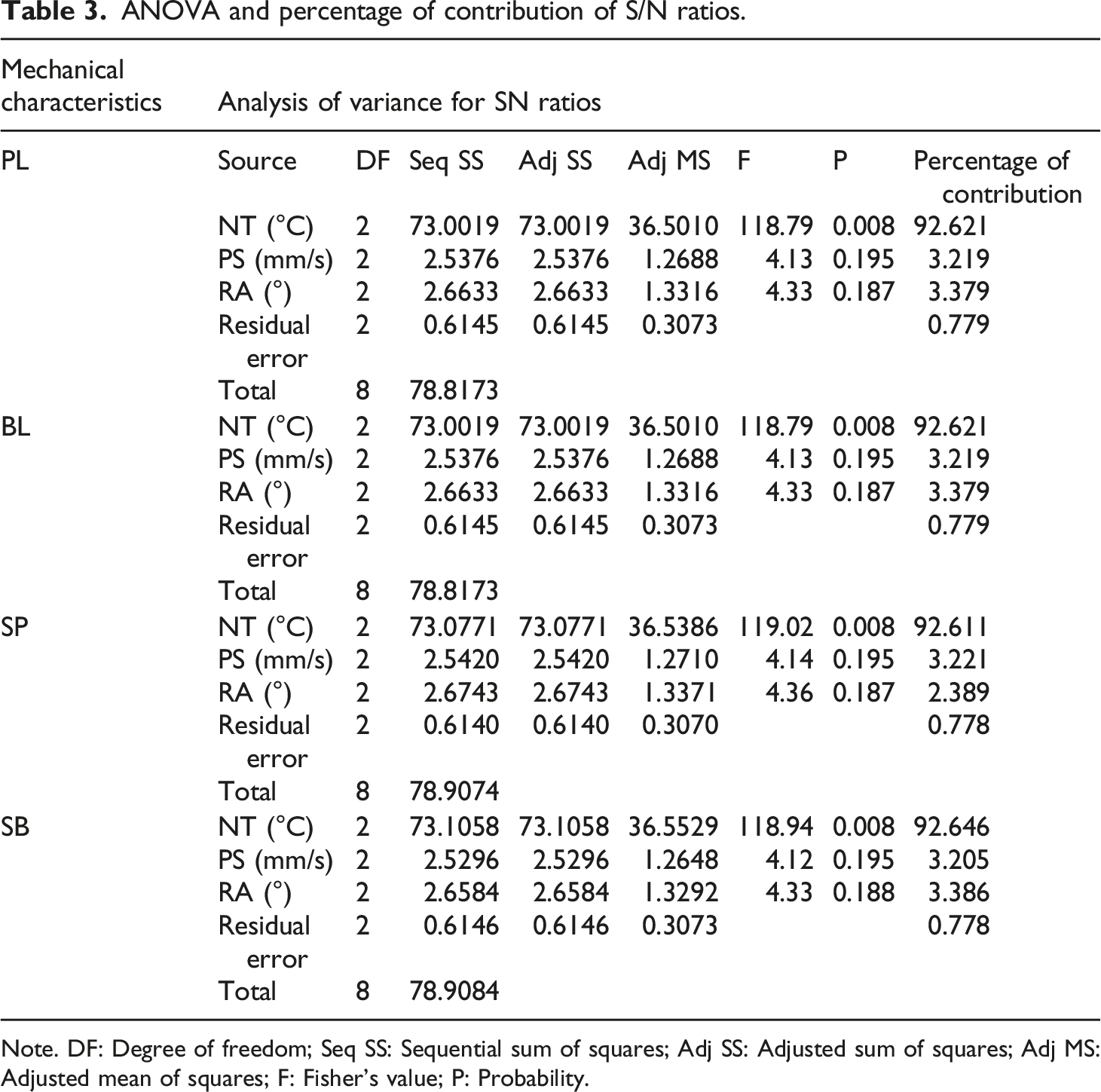
ANOVA and percentage of contribution of S/N ratios.
Note. DF: Degree of freedom; Seq SS: Sequential sum of squares; Adj SS: Adjusted sum of squares; Adj MS: Adjusted mean of squares; F: Fisher’s value; P: Probability.
Response and rank table for S/N ratios.
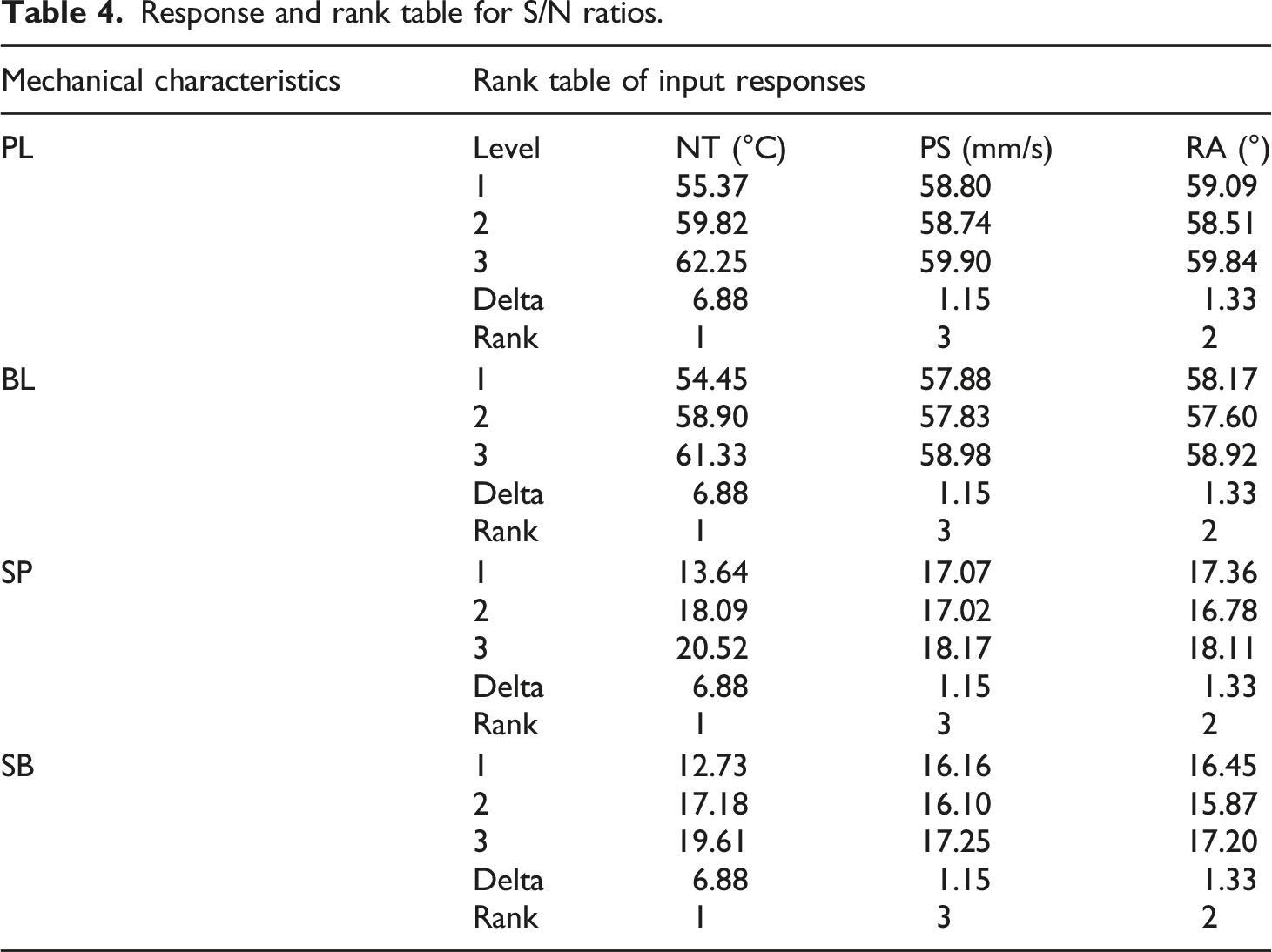
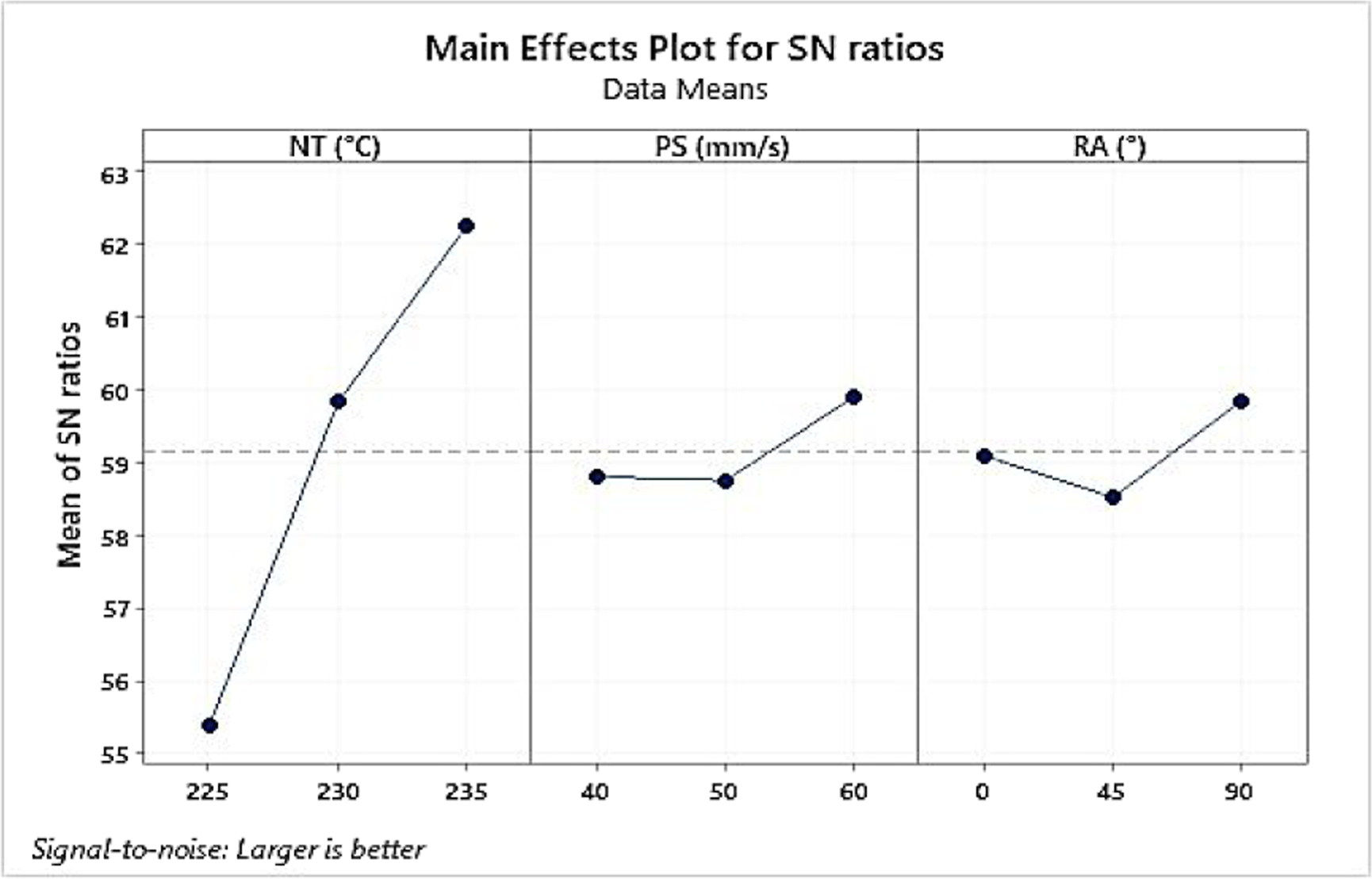
Main effect and residual plots for S/N ratios.
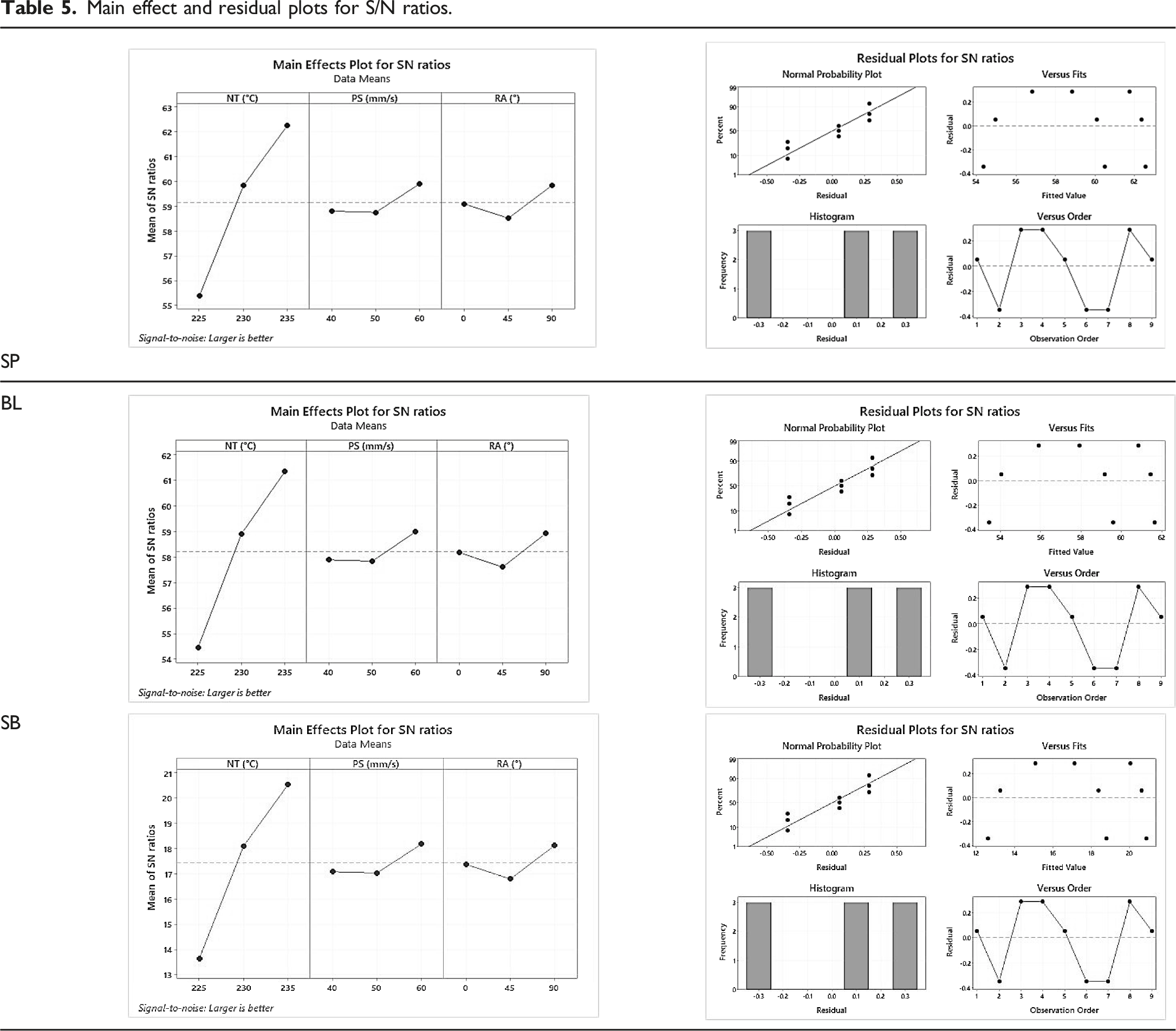
The p-value for the PL, BL, SP, and SB was found 0.008 for NT similar in all cases, which is below 0.05 and confirms the substantial control of process variables.
As observed from Table 5, by enhancing the NT in the selected range, the PL, BL, SP, and SB were improved.
This may be because raising the NT resulted in an appropriate flow of materials while laying layers, resulting in higher layer adhesion. As a result, raising the NT enhanced the PL, BL, SP, and SB. At the same time, PL, BL, SP, and SB were also increased by increasing the PS and higher PS may improve the interlayer fusion. However, PS was observed as an insignificant parameter (Table 3). Furthermore, increasing the RA from 0 to 90°, the first decrease then increases in PL, BL, SP, and SB was noticed. Here again, this parameter was observed as insignificant for the output properties. The residual error contributed 0.779% for PL and BL, and 0.778% for SP and SB, hence the process may be considered under statistical control.
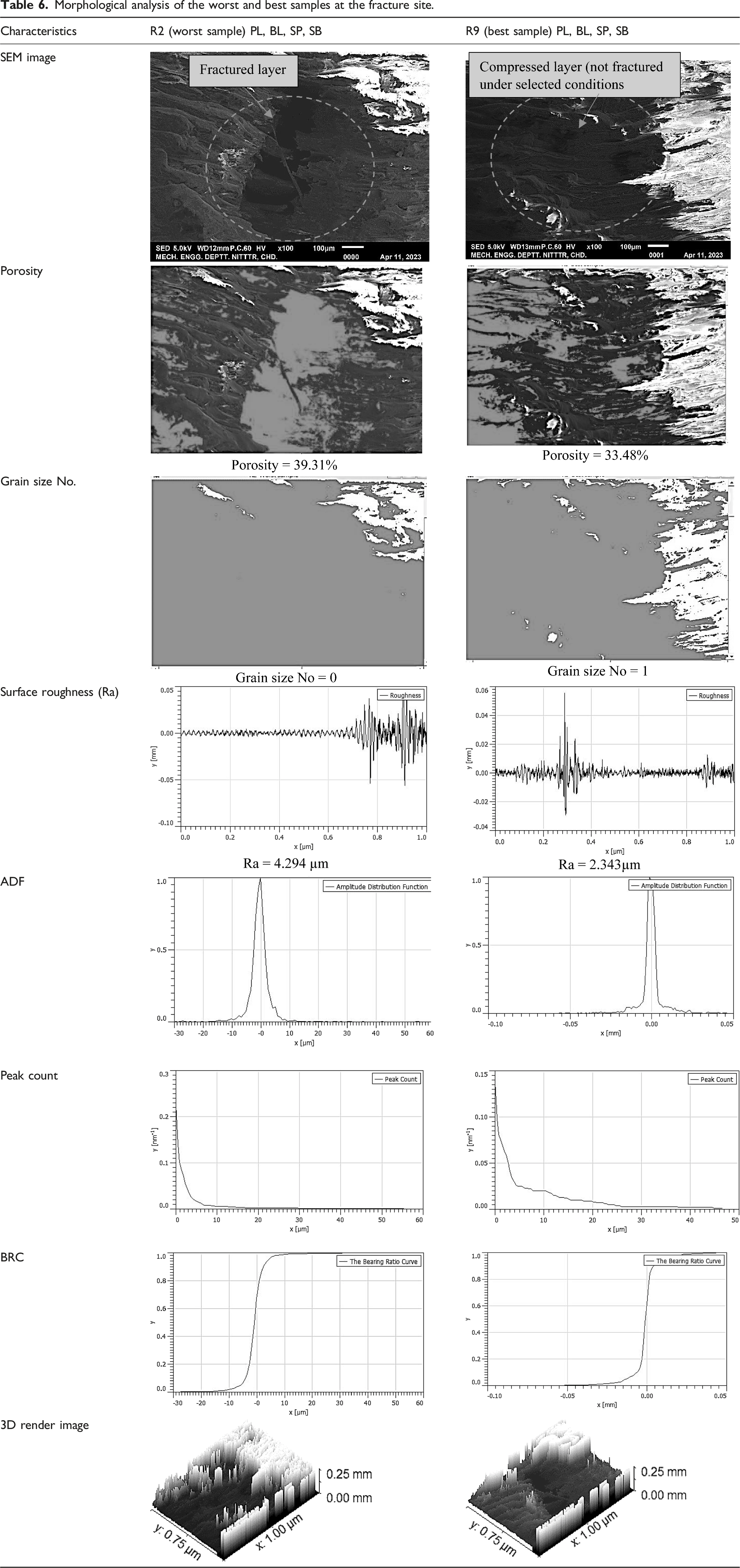
Morphological analysis of the worst and best samples at the fracture site.
A cylindrical pin of PVDF composite having a diameter of 12.70 mm and length of 25.40 mm (as per Figure 3(a)) was used on a pin-on-disc setup (as per ASTM G99-05). The sandpaper of fine grit size number 2000 was placed over the circular disk with the help of tape as shown in Figure 5(a). A 10 N load was applied for 300 s at a speed of 60 r/min. The sample clamped in the sample holder is shown in Figure 5(b). The wear of the worst (R2) and best (R9) samples was observed at 353 µm, and 314 µm respectively as shown in Figure 5(c). The frictional force of R2 and R9 was observed as 4.6, 1.2 N as shown in Figure 5(d), similarly for the coefficient of friction of R2 and R9 was 0.47, 0.14 was recorded as shown in Figure 5(e). The weight loss of R2 and R9 was measured as (initial weight – final weight) 0.493 g, 0.348 g respectively. (a) Track path after wear, (b) PVDF composite during wear, (c) wear curve, (d) frictional force, (e) coefficient of friction of the best and worst sample.
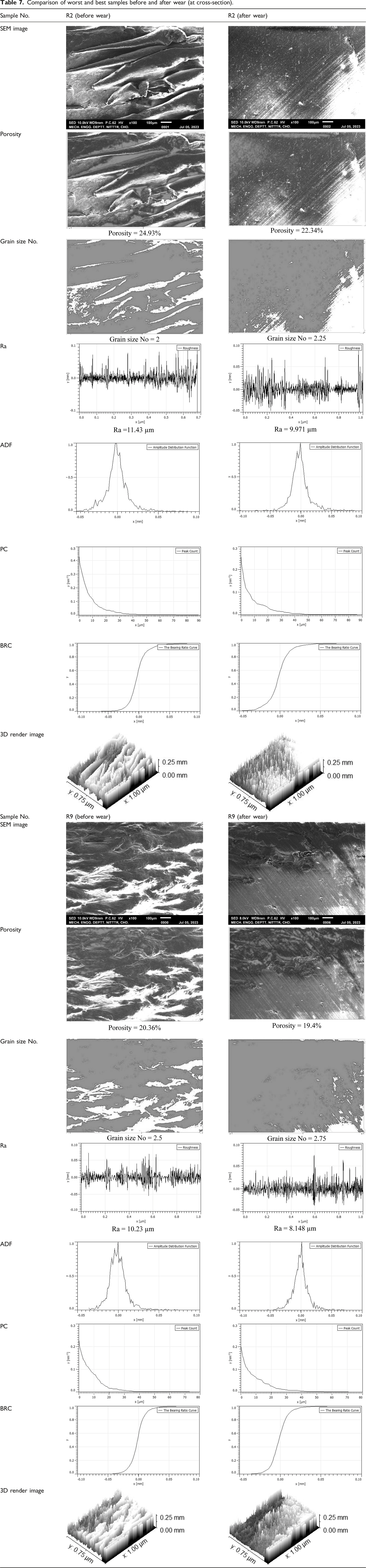
Comparison of worst and best samples before and after wear (at cross-section).
This study was performed to explore the compressive properties of PVDF composite (90% PVDF-8% HAp-2%CS by wt.) fabricated by FFF for a partially absorbable implant for canines, it has been ascertained that the selected sample R9 may be commercially explored for fabrication of partially bioabsorbable implants under compressive loading in line with the work reported by previous researchers.4,5 The results are harmonized with previous studies34,35 and may be useful to select orthopedic biomaterials. The selected composition and processing route will affect the useful life of orthopedic implants and eventually improves the standard of living for those receiving orthopedic care. One of the major advantages of the proposed solution is that the need for post-surgery for the removal of IM pin of metallic alloy as reported previously 5 after healing the complete bone may be eliminated. This will help to prevent post-operative infections, which is one of the biggest problems in orthopedics.
Conclusions
This study highlights the improvements in orthopedic implant materials, especially for reducing post-operative surgical procedures such as the removal of IM metallic pins in canines. The result suggested that the best set of parameters for samples prepared by FFF are NT 235°C, PS 60 mm/s, and RA 45° (for the compressive properties). The best observed mechanical properties were PL 1321 N, BL 1188.9 N, SP 10.83 MPa, and SB 9.75 MPa. Furthermore, the statistical analysis reveals that NT is the only significant parameter for PL, BL (with percentage contributions of 92.62%), SP, and SB (with percentage contributions of 92.62%92.61, 92.64 respectively). The results are supported by the morphological analysis (SEM, porosity%, grain size No., etc.) of the fracture site, which clearly shows significant improvement at the best-suggested settings. The fractured layer was noticed in the case of the R2 sample, whereas compressed layers (without fracture) were noticed for R9 samples (with porosity% as 39.31%, grain size No. 0, Ra: 4.294 µm, for R2 and porosity% as 33.48%, grain size No. 1, and Ra 2.343 µm for R9). The wear for best sample R9 was observed as 314 µm, friction force 1.2 N, and coefficient of friction 0.14. For the worst sample R2, wear was 343 µm, friction force 4.6 N, and coefficient of friction 0.47. Furthermore, like fracture site morphological analysis, better results were noticed for the wear surface at cross-section for sample R9. For R2 (worst sample) porosity% (before and after wear was observed as 24.93% and 22.34%, grain size No. 2, and 2.25, Ra 11.43 µm and9.971 m respectively), and for R9 (best sample) porosity% (before and after was noticed as 20.36% and 19.4%, grain size No. 2.5 and 2.75, Ra 10.23 µm and 8.148 µm respectively). Further studies may be conducted to prepare the digital twin of partially bioabsorbable PVDF composite implant under compressive loading for online health monitoring of the subject.
Footnotes
Acknowledgements
The author(s) are thankful to DST (GoI) for providing research facilities under the FIST project (File No. SR/FST/COLLEGE/2020/997).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Department of Science and Technology, Ministry of Science and Technology, India with grant number SR/FST/COLLEGE/2020/997.