Abstract
Urea cyanurate, as a new nitrogen-rich organic-based heat stabilizer, for the enhancement to thermal properties of polyvinyl chloride (PVC) is prepared via the simple precipitation method, in this study. Characterization, thermal and mechanical properties of the stabilized PVC were examined by means of fourier transform infrared (FTIR) spectroscopy, thermogravimetric analysis (TGA/DTG), X-ray photoelectron spectroscopy (XPS) measurements, discoloration test, congo red test and the weight-loss method, thermal aging test, and tensile analysis. Results showed that the thermal stability of PVC is improved after the addition of urea cyanurate in PVC formulation and weight loss related to HCl releasing in the normal PVC processing temperature (180°C) is less than 1%. Enhancement of the thermal properties and stability time of the PVC lead to decreased degradation tendency. Oven-aging test result showed that the color change during degradation process of PVC samples improved by adding the urea cyanurate. The tensile strength of stabilized samples were about 42%, higher as compared to the pure PVC. The optimum and best results for improving the thermal and mechanical properties of PVC were obtained by incorporating the 3 wt % of the urea cyanurate.
Introduction
PVC (polyvinyl chloride) is a commonly used thermoplastics produced by the polymerization of vinyl chloride monomer used in a durable applications in the industrial field such as pipes, profile, films.1,2 Nevertheless, the main defect of PVC has corresponded to its weak stability when exposed to high temperatures. This limitation can be prevented and modified with thermal stabilizers, which commonly act in two ways: replacing the chlorine atoms in PVC matrix with other functional groups, for example, allylic, and tertiary chlorine atoms and absorption of hydrogen chloride (HCl) is released during the degradation of PVC.3–5
Over recent years, different compounds have been studied as PVC stabilizers, such as metal soaps,6–8 lead salts, 9 organic antimony, 10 and organotin.11,12 However organotin and lead salts stabilizers have good efficiency for stabilizing PVC. The toxicity of these stabilizers are the major reason for the limitation of their application as stabilizers.13,14 Recently, Ca/Zn thermal stabilizers, have attracted a lot of attention in this field via their nontoxicity.2,15 Although, an undesirable produced zinc chloride (ZnCl2) that formed from these stabilizers can catalyze the degradation of PVC products.16,17
Therefore, it is desirable to candidate single stabilizers with multiple functions to reduce cost and improve product quality.
In recent years, several studies have been conducted in the fields of nitrogen-containing compound synthesis, sustainable additives for PVC, and the investigation of the thermal and mechanical properties of this polymer as a result of modification with these additives.18–20
Nitrogen-containing compounds such as urea derivatives such as DAU, MAU, and MATU have shown good potential as an organic stabilizer.21–23
These nitrogen-containing organic-based thermal stabilizers indicated an excellent thermal stability for PVC products.24,25 The thermal stability mechanism of urea derivatives includes inhabitation of the autocatalytic effect of HCl by absorbing released HCl during the degradation of PVC and, after that reacting with PVC chains to avoid the formation of conjugated double bonds.26–28
Urea cyanurate (UCA) is a nitrogen-containing compound that is mostly used as a preservative in the storage of agricultural crops and in the production of cyanuric acid and also could be used as a thermal stabilizer in PVC.29,30 The important point in choosing of these nitrogen-containing organic compound is to consider the good compatibility of these thermal stabilizers with PVC along with no negatively effect on the transparency and other expected properties of the final product. 31
Thus, this paper optimized the synthesis method of UCA via the simple precipitation method. The compatibility of UCA with PVC was further studied by characterizing the thermal and mechanical analysis. Moreover, we report that UCA has a long-term thermal stability of PVC as an organic stabilizer.
Materials and methods
Materials
PVC (s.pvc grade) were obtained from Persian Gulf Petrochemical industries Co, Iran, suspension grade with k-value of 65, Viscosity number of 102 mL/g.
Urea (CO(NH2)2), with assey (ex N) ≥ 99.0%, density of 1.32 g/cm3 (20°C), melting point of 134°C, cyanuric acid C3N3(OH)3, with assey ≥98.0%, density of 2.5 g/cm3 (20°C), melting point of and other chemicals purchased from Merck Chemical Co. and were used without further purification.
Characterization
ATR-FTIR analysis
Attenuated total reflectance - Fourier transform infrared spectroscopy (ATR-FTIR) were recorded by a thermo nicolet nexus 470 spectrometer. The five scans were collected to enhance the signal-to-noise ratio, and these scans were recorded in the range 600–4000 cm−1 in the transmittance mode.
TGA/DTG analysis
The thermogravimetric analysis (TGA) and derivative thermogravimetry (DTG) of the stabilized PVC samples were recorded using STA Instruments (Bahr) in the range between 25°C and 800°C at a heating rate of 10°C/min in nitrogen atmospheres.
XPS analysis
X-ray photoelectron spectroscopy (XPS) measurements of the sample were performed by Thermo Scientific K-alpha spectrometer using monochromatic Al Ka radiation to analyze the surface electron states.
Thermal aging test
The thermal stability of PVC samples was evaluated by color change test and weight change test, according to the ISO 305:2014 method,
32
briefly: The PVC and stabilized PVC sheets were cut into about 20 mm × 15 mm strips, inserted in the temperature-controlled oven (Binder, Germany) at 180°C and checked at the specified time periods, and then subjected to visual examination. The efficiency of the thermal stabilizers was investigated by the visual comparison in color differences of the heated sample strips
Congo red test
The static thermal stability of PVC and PVC samples (3 g) with different amounts of UCA is measured via Congo red test. For this means, each PVC sample, was put into a test tube with congo red paper indicator, located approximately 2 cm above the sample. The assembled tube and sample were immersed in an oil bath at 180°C. The static thermal stability time was measured when the congo red paper began to turn blue. 33
Mechanical analysis
Mechanical properties were investigated via a tensile instrument (SANTAM CO., Iran). The tensile properties of the neat PVC and stabilized samples were measured based on ISO 527-2. 34 Stress-strain curves were obtained at a strain rate of 10 mm min−1 and 25°C. Each sample is measured three times and recorded for the average of tensile properties.
Sample preparation
Synthesis of urea cyanurate
UCA was prepared according to the following simple methods:
32
An aqueous solution (50%), of urea (12 g), was added to into a 500 mL round bottom flask and heated to 90°C, then cyanuric acid (1.29 g) and ammonium chloride (0.129 g) was added to the reaction mixture and stirred for 3 h. During the reaction, the temperature was kept at 90°C. After this reaction time, the wet precipitated UCA was recovered from the slurry mixture using a centrifuge. The wet UCA was dried at 60°C for 12 h to obtain UCA powder, and the yield was 76%. The synthesis route of UCA is shown in Scheme 1. Synthesis of UCA.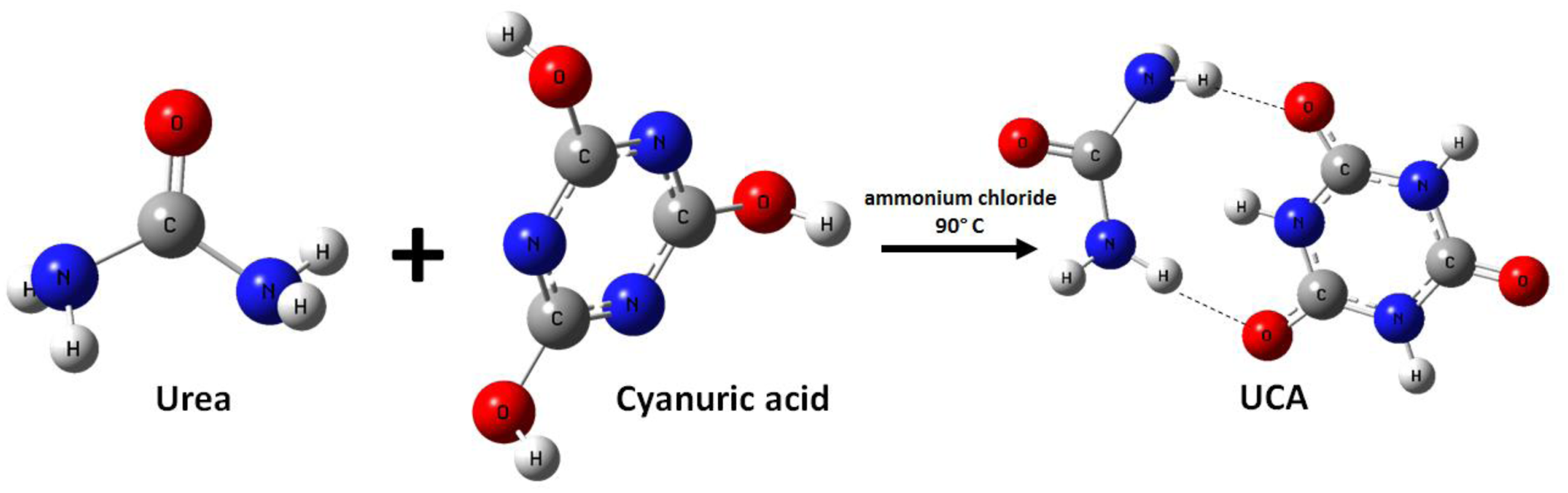
Preparation of PVC samples
A laboratory internal mixer (Plastograph70, Iran) was used to mix UCA with PVC in the 0% (PVC-UCA0, blank sample), 1% (PVC-UCA1), 3% (PVC-UCA3), and 5% (PVC-UCA5), for 5 min under the condition of 40 r/min and 180°C. The resulting compositions were compressed to sheets with a thickness of 3 mm at 120 bar and 190°C in a scientific laboratory hydraulic press (Netzsch, Germany), and cut to the specified diameter according to each test method.
Results and discussion
Characterization of UCA
The structure of synthesized UCA was confirmed by The Fourier transform infrared (FTIR) spectra, TGA and additional calculations for estimation of Gibbs free energy by DFT calculation and also XPS experiments.
FTIR analysis
The structure of cyanuric acid, urea and UCA were confirmed by The FTIR spectra are shown in Figure 1, and characteristic absorption bands data are listed in Table 1. FTIR spectra of urea, cyanuric acid, and UCA. Characteristic vibration bands in FTIR of urea, cyanuric acid, and UCA.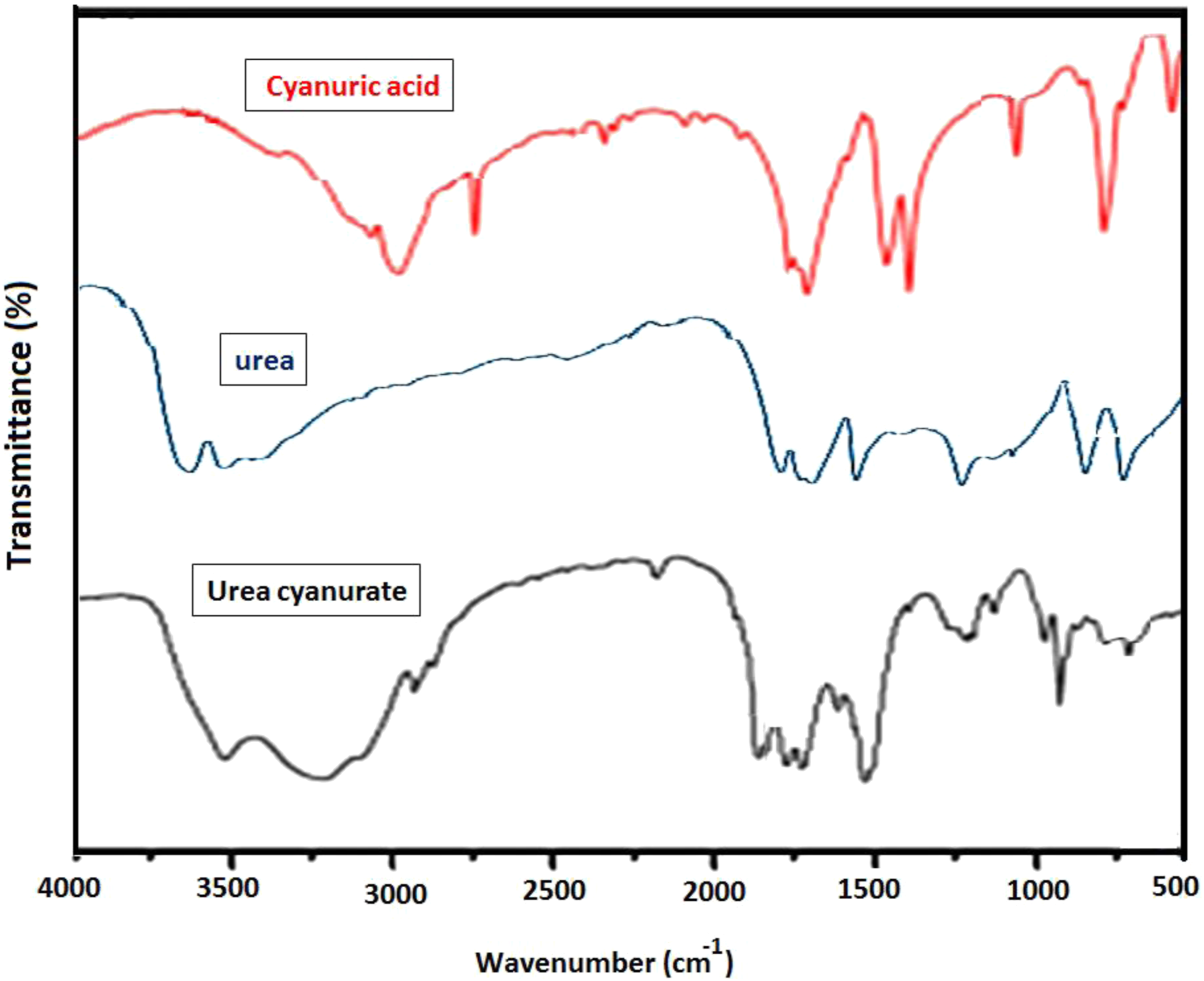

FT-IR measurements of urea showed absorption bands at 1381 cm −1 for the C-N stretch, 1750 cm −1 for the C=O stretch, and 1640 cm −1 and 3410–3550 cm −1 for the N-H stretch. 35
Cyanuric acid is in an aqueous media, containing weak acid as a mixture of keto-enol tautomer. 35 In acidic media the keto form is more stable than the enol form.35,36 As shown in Figure 1, The stretching vibration bands of 3200 cm−1 corresponding to the N-H in keto form, and another peak represented at 3080 cm −1 contributed to the stretching of the O-H band (enolic form). 37 Moreover, a sharp band was present at the 2860 cm −1 is resulted from the stretching vibration related from coupling of O-H and N-H during tautomerism. As shown in Figure 1, this peak disappeared in the UCA spectrum. It is suggested that the hydrogen bond formation between N-H and the oxygen of the carbonyl group in the keto form of the cyanuric acid part in the UCA sample, caused the stability of the keto form, and this fact in agreement with the estimated UCA structure (Scheme 1).
On the other hand, The strong absorption peaks at 3100 and 3550 cm−1 were assigned to the symmetric and asymmetric stretching vibrations from ─NH─ groups and hydrogen bonding, 1720 and 1770 cm−1 to the stretching vibration of ─C═O, and 1680 cm−1 to the stretching vibration of C= N, agreed with those of the UCA structure. Also, it could be observed that, the UCA spectrum of the series of characteristic peaks attributed to urea and cyanuric acid, shifted to higher wavenumbers relative to those because of the intermolecular hydrogen banding effect between urea, and cyanuric acid.
Thermal analysis
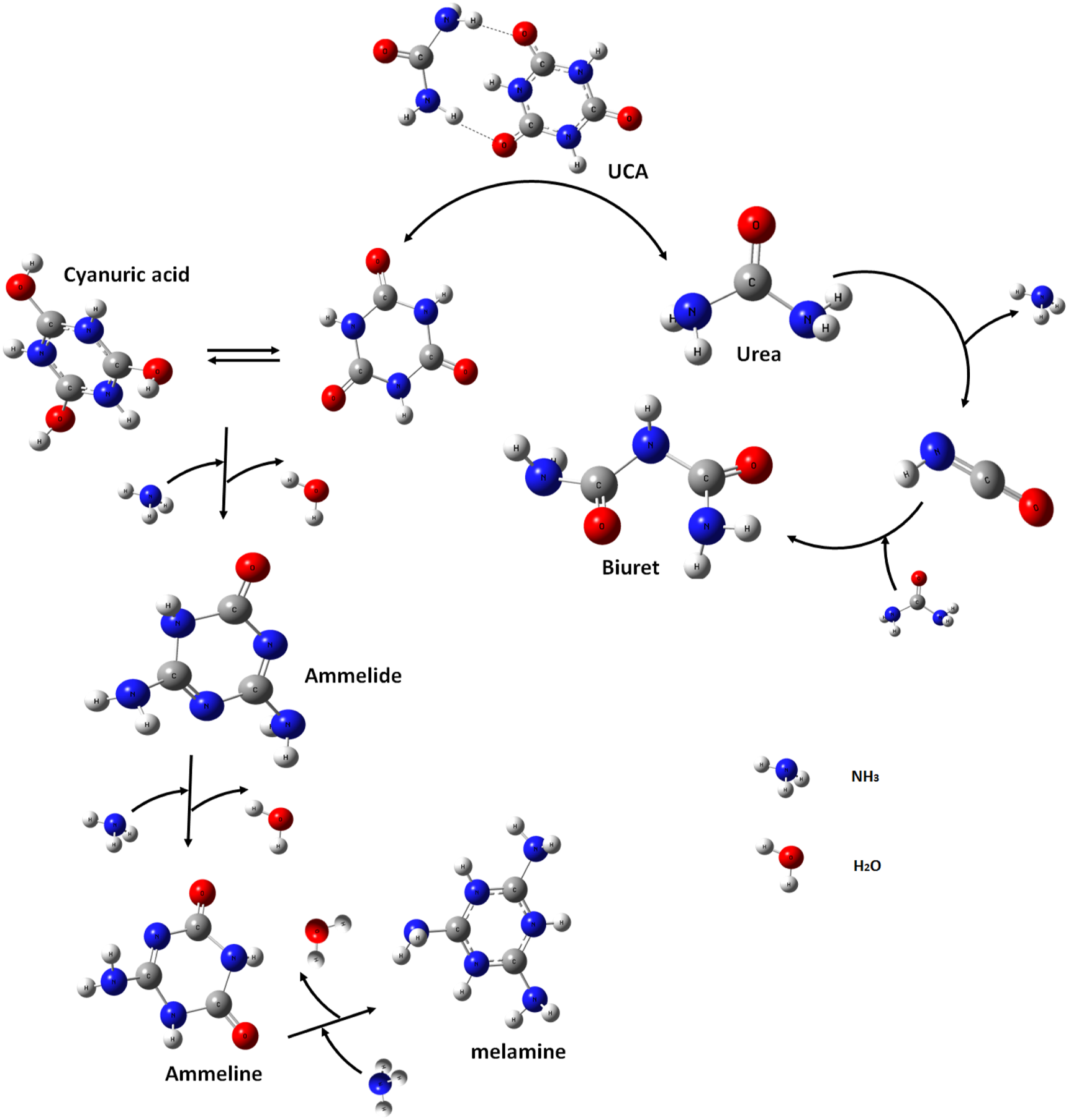
As is shown in Figure 2, the thermal properties of cyanuric acid, urea and UCA were characterized by TGA at a heating rate of 10 C/min. The thermal properties of urea indicated two decomposition stages. The first stage approximately occurs at a temperature of 280°C, associated with early evaporation of 70% of urea and biuret (Scheme 2). TGA thermograms of urea, cyanuric acid and UCA. Decomposition route and Possible produced compound of urea, cyanuric acid and UCA.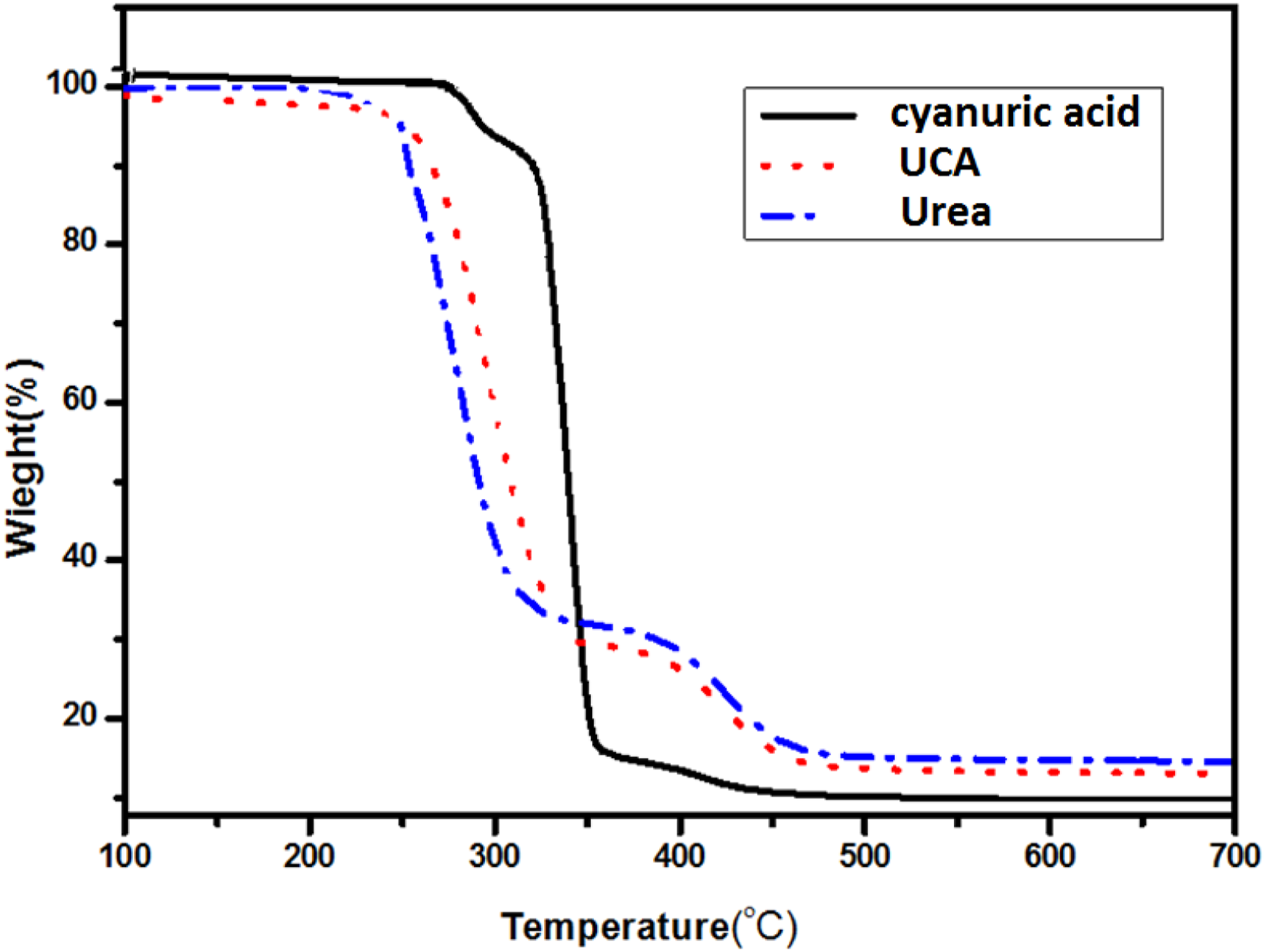
The second stage, corresponded to 30% urea/biuret decomposition, starting at the temperature of 338°C. 38
Cyanuric acid is more stable than urea due to its higher decomposition temperature and showed three decomposition stages 39 in this degradation process. The first stage at 310°C showed urea/biuret decomposition. The second decomposition stage at the temperature,corresponded to another fast decomposition process that verified cyanuric acid structure. 40 During cyanuric acid decomposition in the third stage above the 360°C, chemically more stable compounds, such as ammelide and melamine, are probably produced (Scheme 2).
According to the TGA curve of UCA, the weight loss of UCA in the normal PVC processing temperature (180°C) is less than 1%. The TGA curve of UCA shows a weight loss over 270°C in two steps. The initial decomposition stage within the temperature range of 270°C–345°C could be related to the release of carbon monoxide. The second mass loss step occurred in the range of 350–450°C, due to the decomposition of the cyanurate more stable produced compounds (ammelide, ammeline and melamine) 41 (Scheme 2).
Both of these stages are higher than the processing temperature of PVC. Therefore, UCA is stable at PVC processing temperature and could be used as PVC thermal stabilizer.
Thermodynamic parameters
With the availability of TGA data, thermodynamic aspects of the successfully synthesized UCA was also investigated in detail by calculation of thermodynamic parameters of urea, cyanuric acid and UCA.
For this purpose the enthalpy changes (ΔH°), the Gibbs free energy changes (ΔG°) of the process were performed with the Becke–Lee–Yang–Parr functional (B3LYP) DFT method 28 and the 6-311++G(d, p) basis set. 42
ΔH° value for UCA structure showed a lower energy about -438.78 Kcal/mol compared to the −235.10.40 Kcal/mol for the urea and the −564.15 Kcal/mol for cyanoric acid.
Additionally, the Gibbs free energy changes values (ΔG°) for all of the structures are also negative; by −438.20 Kcal/mol, −235.50 Kcal/mol and −564.30 Kcal/mol for UCA, urea, and cyanuric acid, respectively. These results refers to the spontaneously of the reaction at room temperature, which is in agreement with the experimental results.
According to the results, enthalpy changes values (ΔH°) of for the UCA sample is negative, so, means that the synthesis process goes through an exothermic path and indicating strong interactions between urea and cyanoric acid fragments in the UCA structure and suggest that the interaction of urea and cyanoric acid is a spontaneous and enthalpy-controlled process.
XPS measurement
The structure of the successfully synthesized UCA was also confirmed by XPS experiment. XPS provided information on the surface composition and chemical states of UCA and thus, clear information about its chemical structure and composition.
The XPS spectra of UCA are displayed in Figure 3. Figure 3(a) displayed the XPS spectra of UCA. The data showed that three signals appeared in the full spectrum of UCA at 286.45, 397.8 and 535.14 ev which corresponded to C 1s, N1s. and O 1s, respectively. This figure revealed that the UCA is composed of C, N, and O. XPS curve of UCA (a); high-resolution C1s (b), N1s (c) and O 1s (d) spectra of UCA.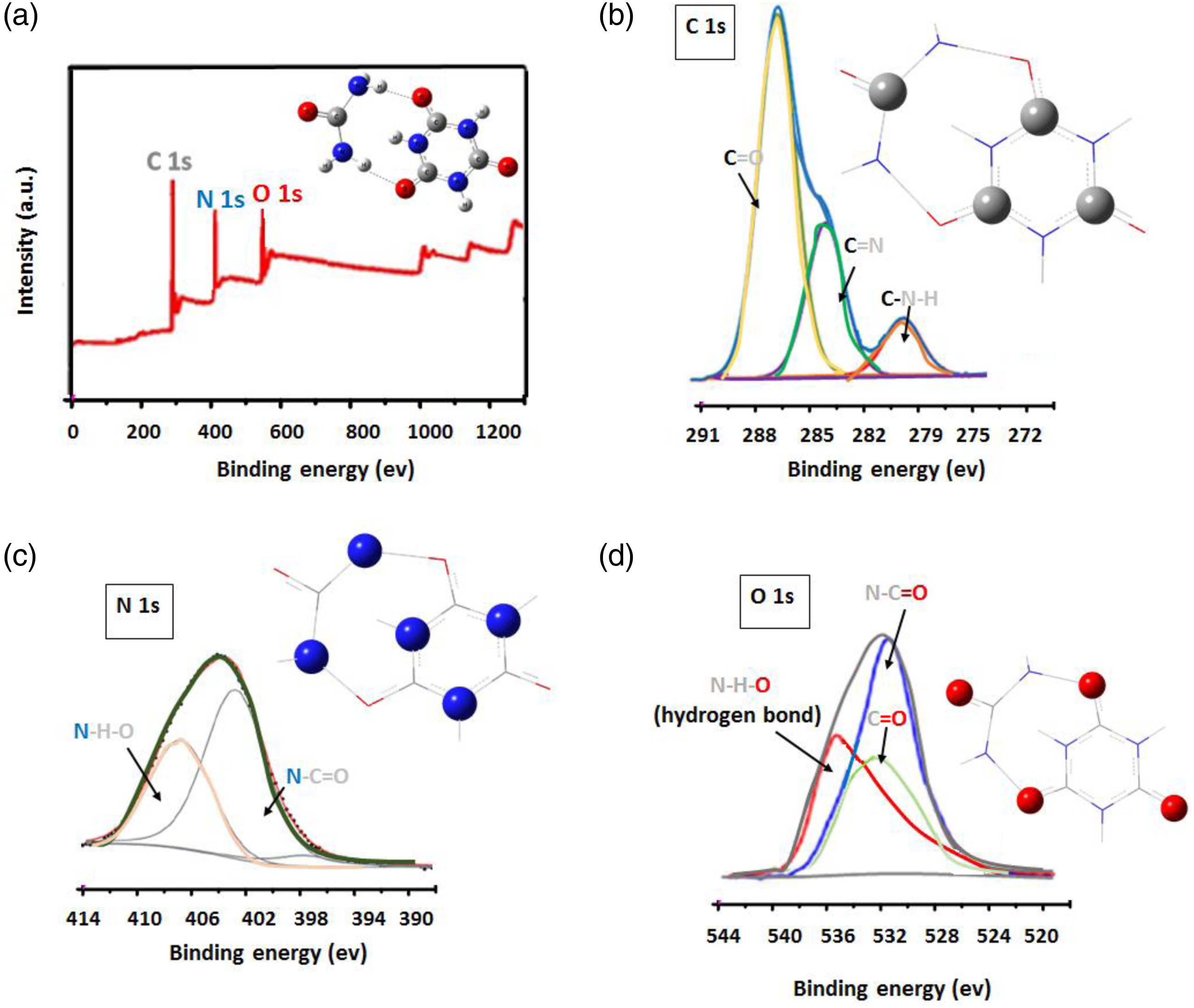
The high-resolution C 1s spectrum of UCA (Figure 3(b)) confirms the presence of C=O (288.3 eV), C=N (284.8 eV) and C-N-H (280.1 eV), groups in UCA structure. In the N 1s spectrum of UCA (Figure 3(c)) confirms the presence of N-H-O (408.5 eV) and N-C=O (402.4 eV) groups in UCA structure.
In the O 1s spectrum of UCA (Figure 3(d)) confirms the presence of O-H-N (536.1 eV), O=C (532.5 eV) and O=C-N (530.8 eV) groups in UCA structure, The O-H-N peak that represented in 536.1 eV clearly indicated that the Urea functionalized onto the surface of the CA structure via hydrogen bonding (Scheme 1).
According to the all results that obtained from FTIR, TGA and XPS experiments. The morphology of the successfully synthesized UCA was confirmed.
Thermal properties of stabilized PVC
The TGA and DTG curves of blank PVC and PVC stabilized samples with 1, 3 and 5 wt% of UCA stabilizer are shown in Figure 4. It these curves could be observed that a two stage thermal degradation of PVC, the first of which corresponded to dehydrochlorination of PVC and formation of conjugated polyenes, and the second stage involved polyene sequences to low molecular weight related compound.41,43 (a) TGA (b) DTG curves of PVC and stabilize PVC samples.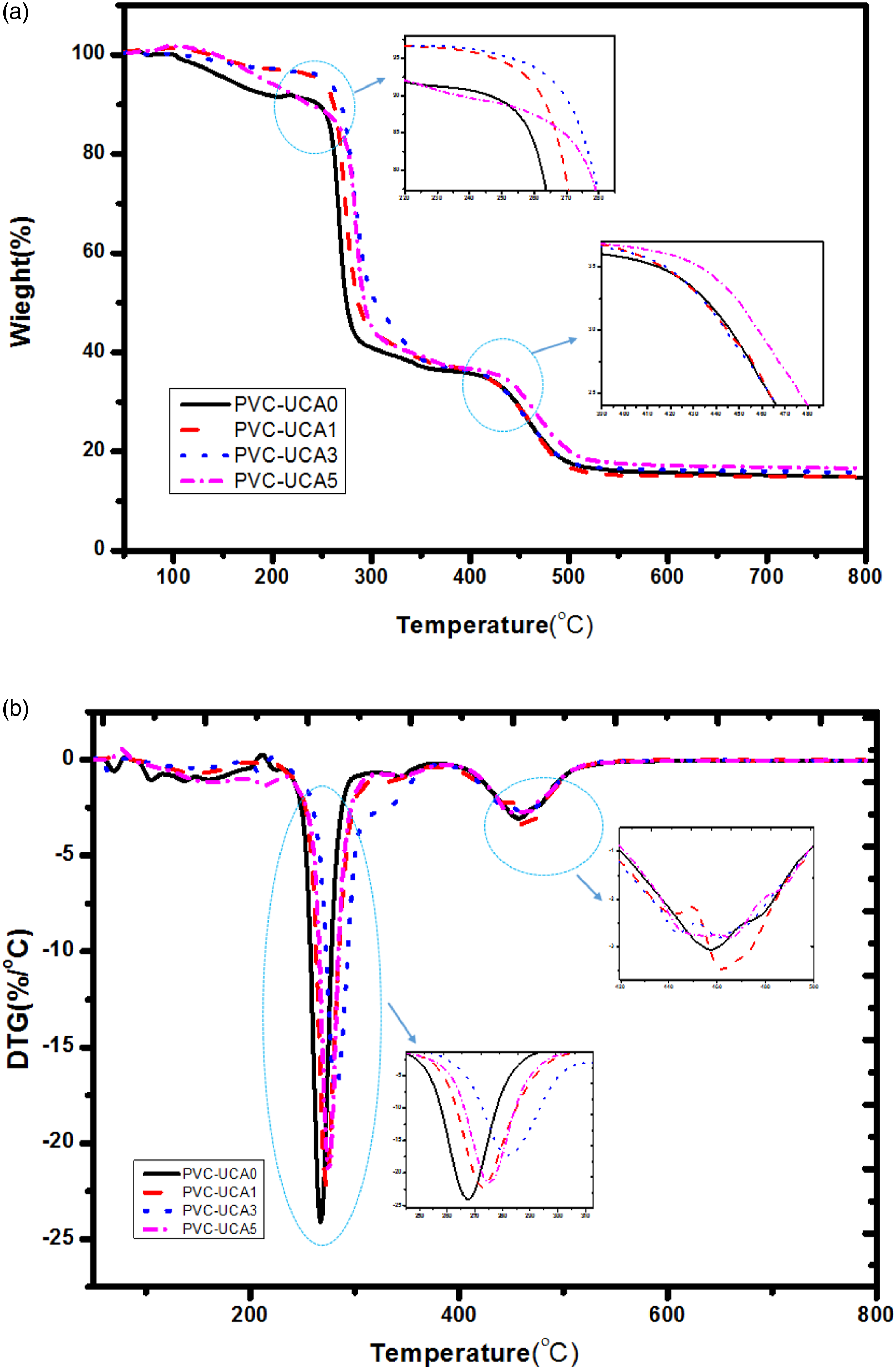
Thermal properties data of PVC and stabilize PVC samples.
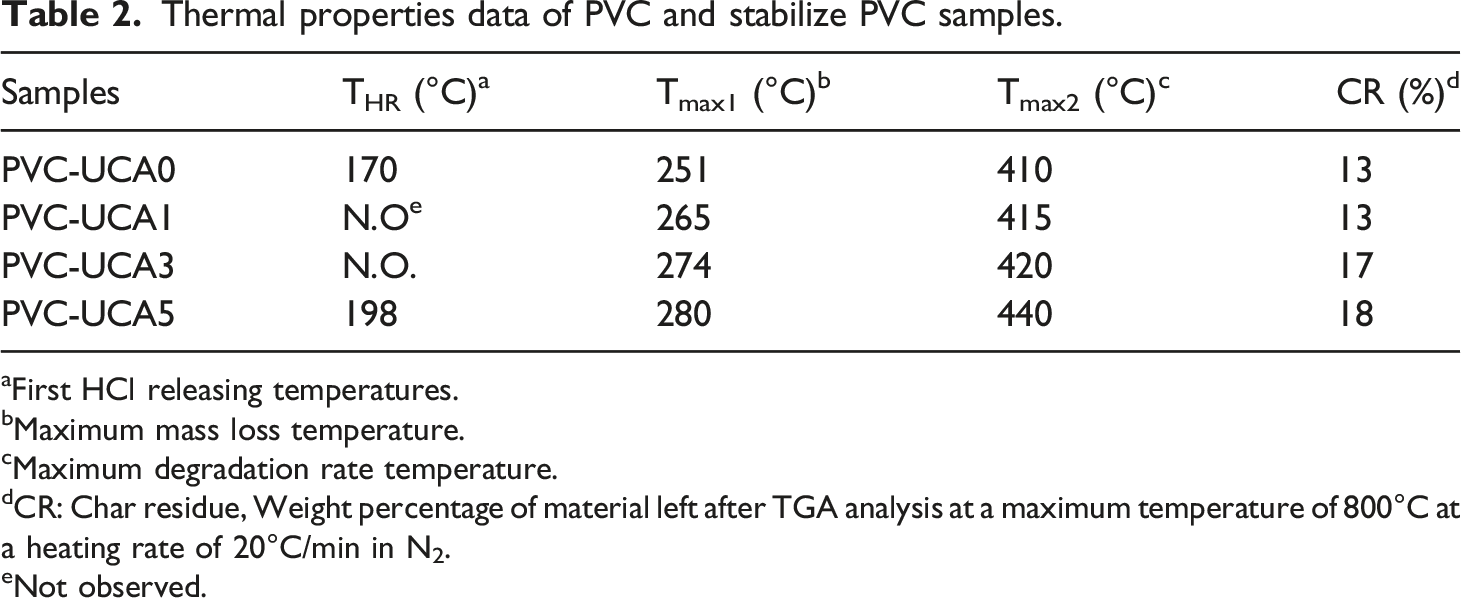
aFirst HCl releasing temperatures.
bMaximum mass loss temperature.
cMaximum degradation rate temperature.
dCR: Char residue, Weight percentage of material left after TGA analysis at a maximum temperature of 800°C at a heating rate of 20°C/min in N2.
eNot observed.
The first weight loss occurs below 180°C, corresponding to the HCl released from the chains of polymer (THR). It is noted that all the stabilized samples have no significant HCL releasing (in 1 and 3 wt% stabilized content) in the normal PVC processing temperature (180°C) or have a less amount of HCL releasing (in 5 wt% stabilized content) over this temperature, compared with blank PVC. This is attributed to the ability of these stabilizers to absorb the released HCl from stabilized PVC samples. Then, these data could be confirmed UCA has a good effect on the thermal stability of PVC.
As shown in Table 2 and Figure 4, by loading of UCA in the PVC matrix, the maximum mass loss temperature (Tmax1) and temperature at second decomposition (Tmax2) in all stabilized PVC samples enhanced as compared to the blank PVC.
Improvements in the thermal properties of PVC in the presence of UCA stabilizer were also seen in char residues of the stabilized samples as compared to the blank PVC at 800°C.
According to all the results in Table 2, the UCA in the 3% and 5% stabilizing levels has the best stabilization effect for PVC. Furthermore, the sample containing 3 wt% of UCA has no significant weight loss related to the HCL releasing and could be suggested on its excellent efficiency in the stabilization of PVC with the lower value of stabilizer.
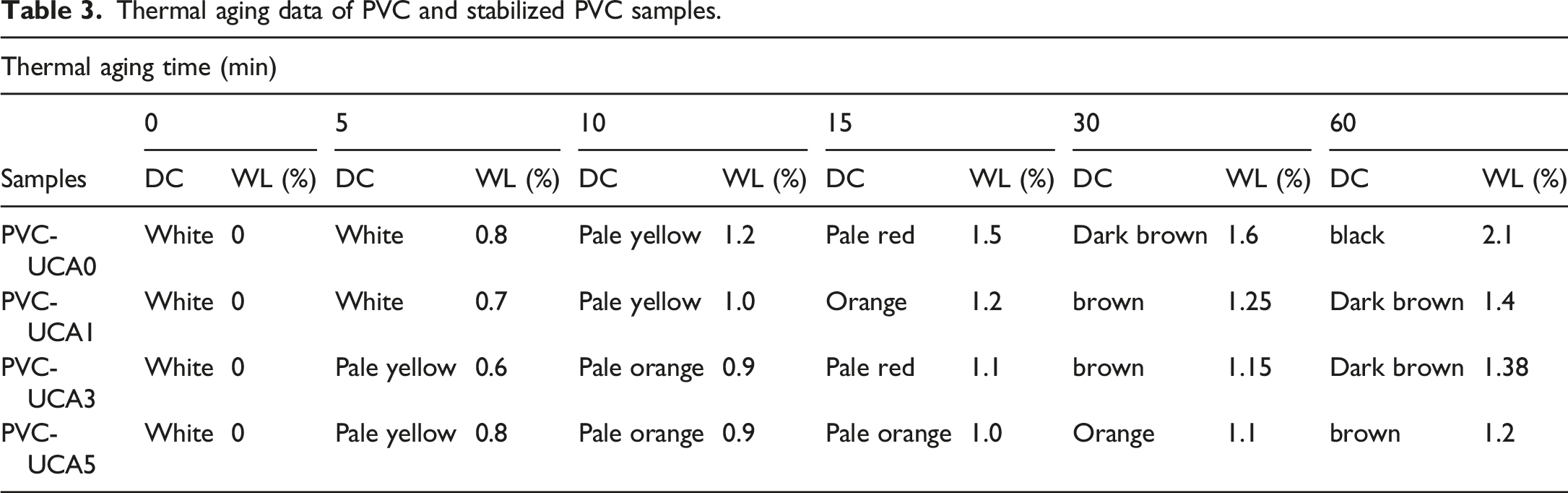
Thermal aging test
Thermal aging tests of the samples were investigated by discoloration test (DC) and Weight Loss evaluation (WL) methods. The corresponding curves of PVC and stabilized PVC samples are represented in Figures 5 and 6, and the data is listed in Table 3. Discoloration of PVC with different UCA stabilizers. Changes in mass loss (%) upon thermal degradation. Thermal aging data of PVC and stabilized PVC samples.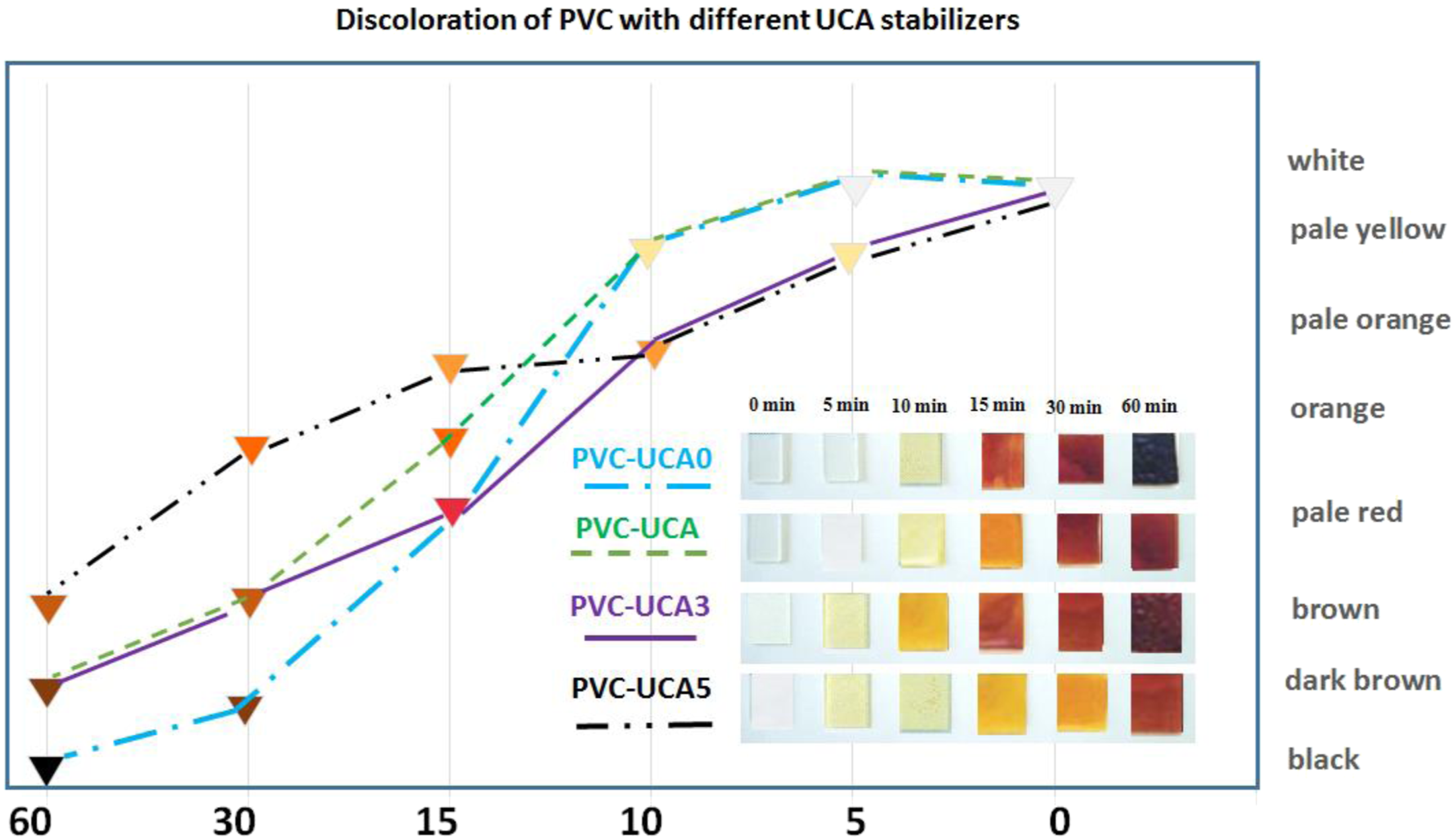
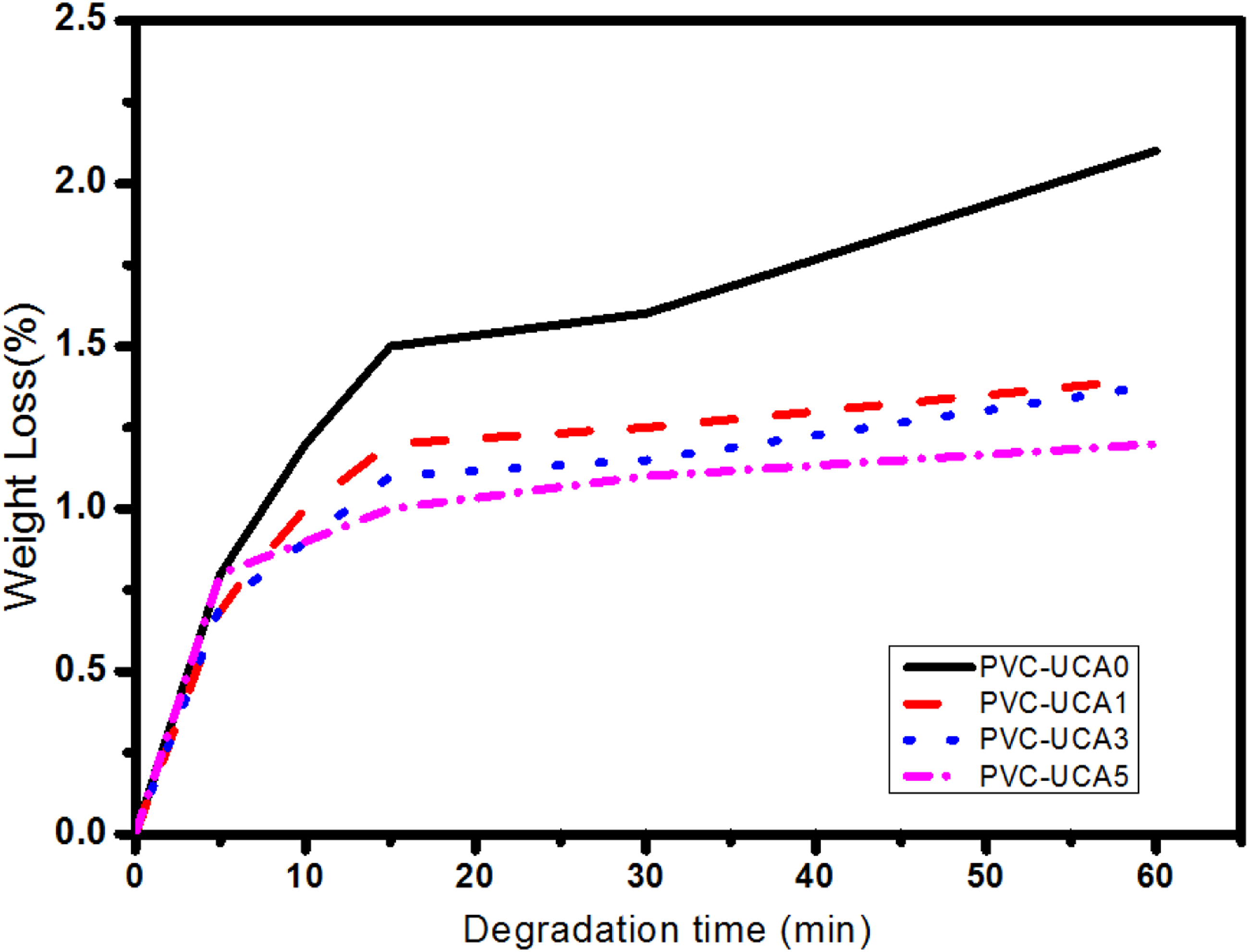
As shown in Figure 5 and Table 3, all the PVC samples containing UCA stabilizer exhibit better performance in the discoloration process against the blank PVC.
The better performance of the UCA stabilizer on PVC attributes to the nitrogen-rich network molecular structure of UCA that can absorb released HCl during the degradation of PVC which increasing the thermal stability of PVC.
The blank sample (PVC-UCA0) undergoes complete discoloration and turns black completely at the test time, in the range of 5–60 min. By the incorporation the UCA stabilizer in PVC matrix, obviously delays the complete the discoloration time of the stabilized PVC samples.
The PVC-UCA5 is slightly better in discoloration process than PVC-UCA1 and PVC-UCA3. It is also founded that the PVC-UCA3 and PVC-UCA5 exhibit the lowest initial change colors: this could be due to the network structure and appropriate amount of UCA for PVC that can significantly absorb HCl released during the degradation of PVC in initial period discoloration.
Dechlorination of PVC at high temperatures causes the chain break due to the removal of HCl. This removal process leads to the release of volatile toxic gas and discoloration of this polymer. Then, nitrogen-rich network stabilizers can stabilize PVC against degradation caused by oven aging. In this evaluation, PVC samples were heated for specified time period at 180°C in the temperature-controlled oven, and the weight loss (%) during these periods was calculated using equation (1),
8
and the results are shown in Table 3 and Figure 5.
As shown results in Table 3 and Figure 6, the weight loss for the blank PVC sample (PVC-UCA0) was higher than those obtained from UCA stabilized samples. In the periods lower than 15 min, the strip containing 3 wt% of UCA (PVC-UCA3) showed the lowest weight loss among the stabilized samples, but above 30 min, the (PVC-UCA5) sample showed more resistance against the aging weight loss; these results are well consistent with the results obtained from the TGA analysis.
Congo red test
Results of the static thermal stability time against dehydrochlorination of PVC containing UCA evaluated by the Congo red method at 180°C and 200°C are shown in Figure 7. The stability time of PVC samples.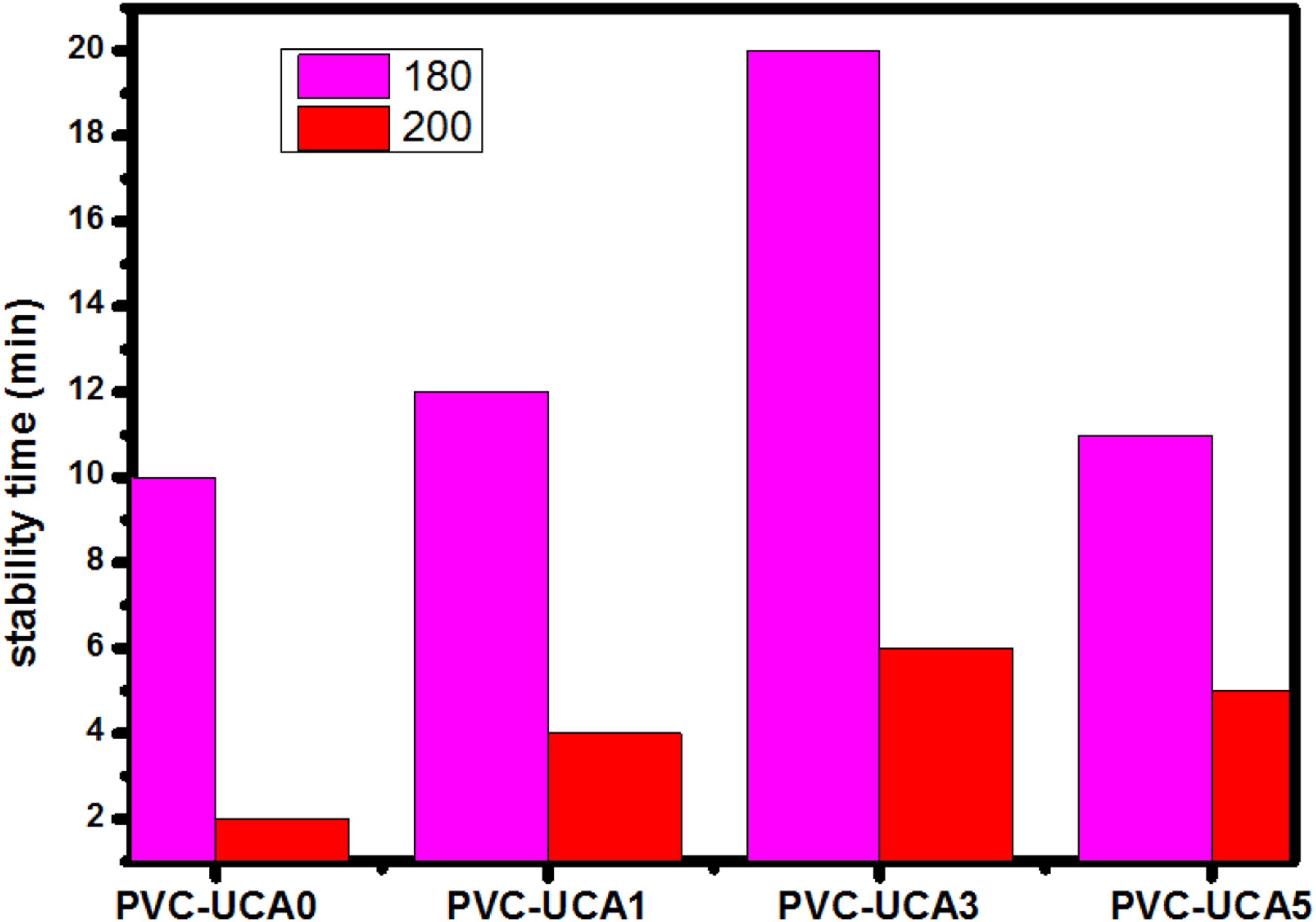
It is seen that the thermal stability time of the blank sample (PVC-UCA0) is 10 min at 180°C and 2 min in 200°C while those of PVC with incorporation of UCA at 180°C and 200°C are (12 min and 4 min) for PVC-UCA1, (20 min and 6 min) for PVC-UCA3 and (11 min and 5 min) for PVC-UCA5, respectively. In contrast, the thermal stability time of PVC containing 3 wt% is twice longer than pure PVC at the processing time of PVC (180°C) and three times longer than PVC in 200°C.
These results may be explained as the strong ability of UCA stabilizer, especially in PVC-UCA3 sample to absorb HCl released during the degradation of PVC.
Tensile strength
The mechanical properties of the PVC containing UCA stabilizer, including the tensile strength (Ts), and elongation at break (EB) are shown in Figure 8, and related data represented inTable 4. Tensile strain-stress curves for PVC-UCA samples with different UCA content. The tensile properties of the PVC-UCA samples with different UCA content.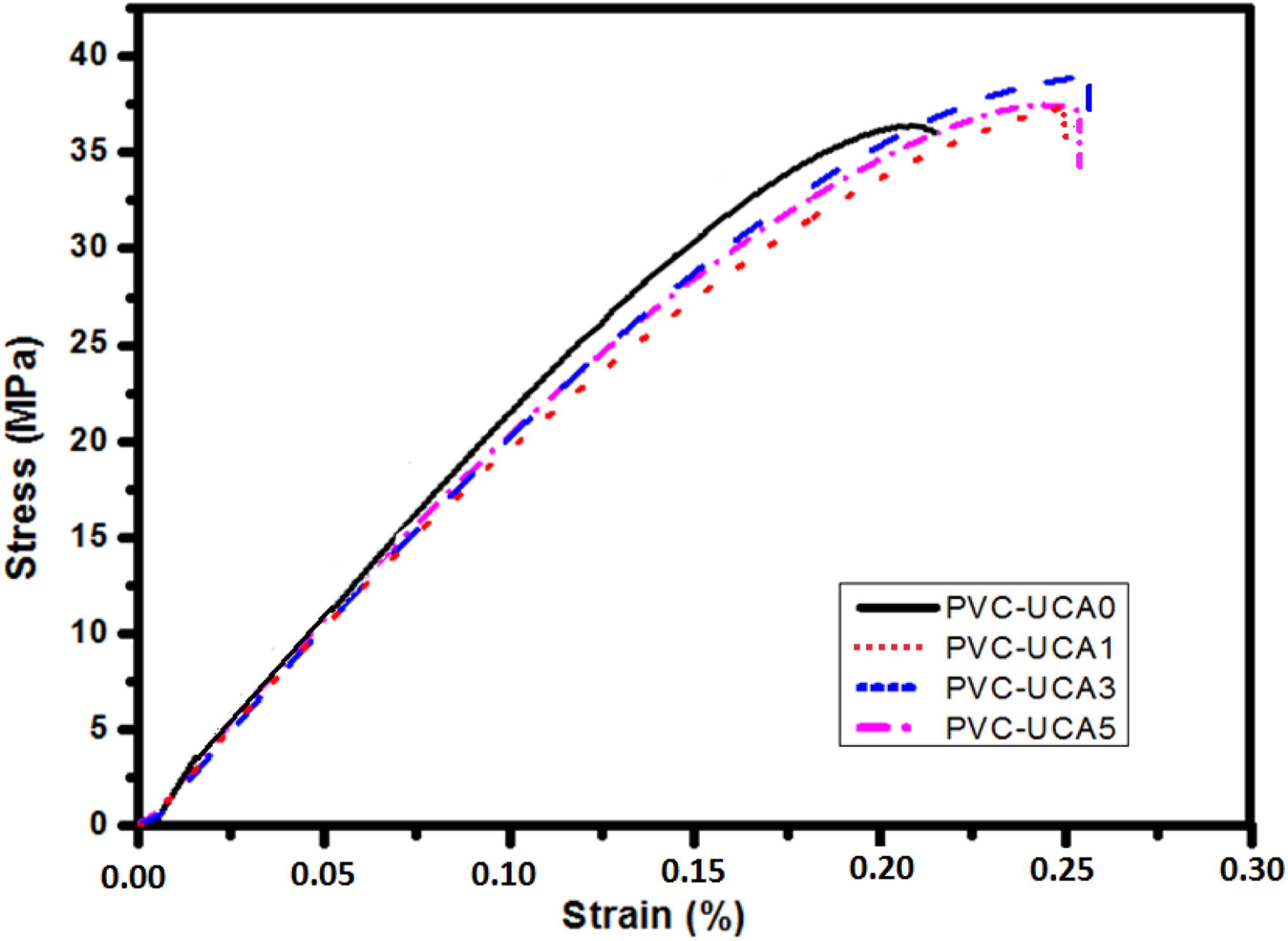
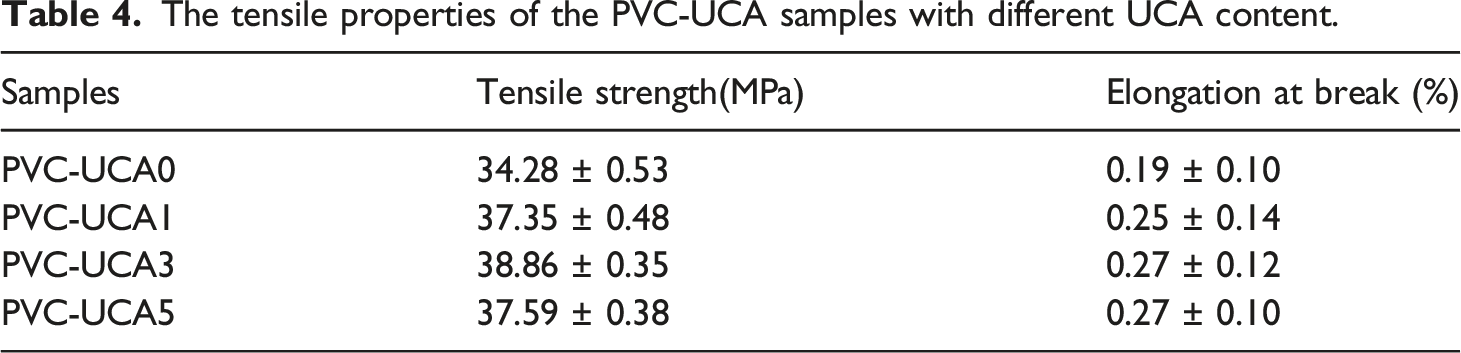
The tensile strength increased from 34.28 MPa for blank sample (PVC-UCA0) to 38.86 MPa for PVC-UCA3. The tensile strength (Ts) values of PVC-UCA1 and PVC-UCA3 and PVC-UCA5 were about 37.35, 38.86 and 37.59 MPa, respectively. The results show that tensile strength increased with increasing the amount of stabilizer and reached the maximum when incorporating 3% UCA in the PVC sample. Beyond 5% stabilizer addition, the tensile strength begins to decline.
According to the elongation at break values (EB), represented in Table 4 and Figure 8, all the PVC-UCA samples showed increased EB value increasing UCA contents, from 0.19% for the bank PVC-UCA0 to 0.27% for PVC-UCA3 and PVC-UCA5. The EB values of these two stabilized samples (3 and 5 wt%) which were about 42%, higher as compared to the bank PVC-UCA0.
These results reveal that UCA and PVC had the best interaction and compatibility in the 3% stabilizer content level, this information is useful in industrial production.
Conclusions
In this work, a simple reaction method was established for the UCA and this stabilizer wasapplied and evaluated as a thermal stabilizer for PVC.
The influence of UCA on PVC in the thermal stability effect was researched by thermal aging test, Congo red thermal stability, thermogravimetric method, and tensile test. The UCA stabilizer has a good effect in the discoloration process of PVC. The optimal percentage of UCA in PVC is 3% at this condition. The stability time of PVC increases from 10 min to 20 min (stabilized by 3 wt% UCA).
In UCA molecular structure contains several double bonds and nitrogen atoms in a network; they can absorb HCl during that produced from the PVC degradation to improve heat stability. Because of increasing stability in the production process, UCA shows a strong stabilizing effect on the tensile strength of PVC.
According to obtained results, this research proposed a new structural design of an effective thermal stabilizer for PVC.
Footnotes
Author contribution
Mahroo Khaleghi conceived of the presented idea, verified the analytical methods, performed the measurements, processed the experimental data, performed the analysis, drafted the manuscript and designed the figures, wrote the manuscript and discussed the results and contributed to the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
