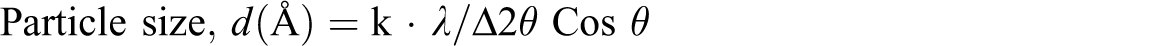Abstract
Synergistic stabilizing effect of layered double hydroxides (LDHs) with micro- as well as nano-sized calcium carbonate (CaCO3) on thermal stability of polyvinyl chloride (PVC) was studied and evaluated. Nano-sized CaCO3 was prepared in the laboratory by matrix-mediated growth and controlled (in situ deposition) technique. Crystal size of nano-CaCO3 was estimated by studying x-ray diffraction pattern. The PVC composites were prepared using Brabender Plastograph instrument. Various formulations were prepared by taking fixed amount of LDH and varying the concentration of micro-/nano-sized CaCO3. PVC sheets of 1 mm thickness were prepared on compression molding machine. Thermal stability in terms of thermal degradation was studied by thermogravimetric analyzer. Surface morphology and mechanical property were studied using scanning electron microscopy and universal tensile machine. Increase in the onset temperature of the PVC sheets was considered as imparting a better thermal stability to PVC. Better synergistic effect on the stabilization of PVC was observed in case of LDH with nano-sized CaCO3 and compared with micro-sized CaCO3 .
Introduction
Concept of nanocomposites of polymer deals with the effect of nano-sized fillers on the properties of composites. In contrast to the conventional systems where reinforcement is of the order of microns, discrete constituents of the order of a few nanometers represent polymer nanocomposites. 1 Uniform dispersion of these nano-sized filler particles produces ultra-large interfacial area per unit volume between nanofiller and a polymer matrix. Immense internal interfacial area and the nanoscopic dimensions fundamentally differentiate polymer nanocomposites from traditional composites and filled thermoplastics. These new materials improve the performance of classic materials by acquiring new modified properties, resulting in favorable cases of unique synergism between the polymer materials. 2,3
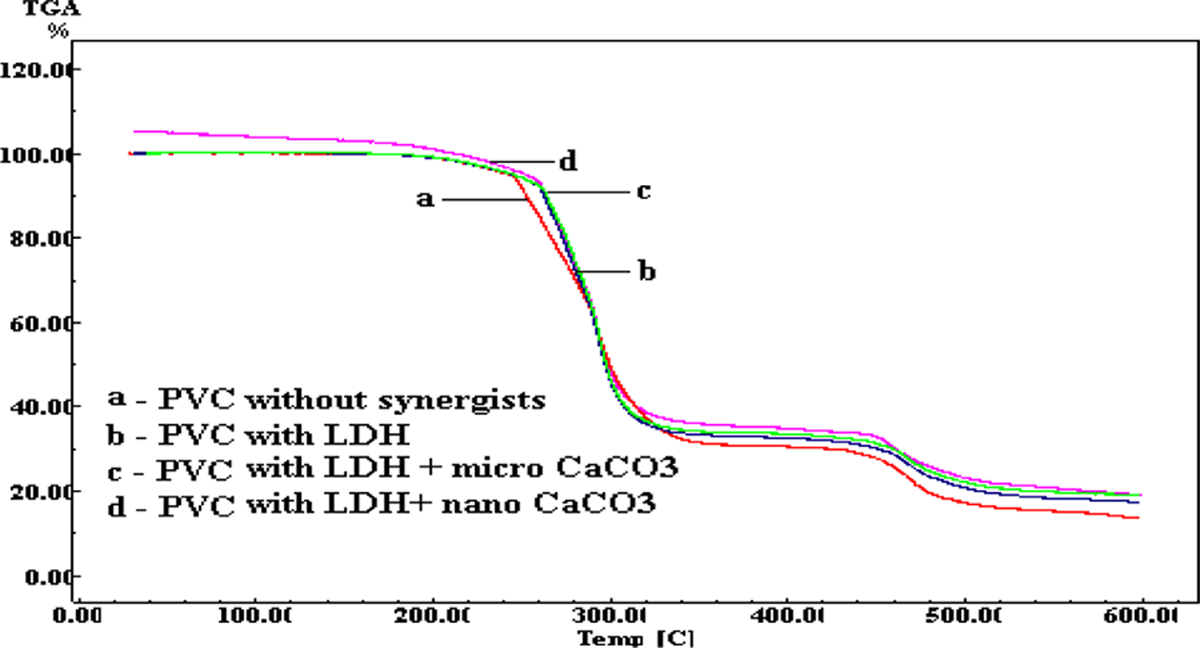
Polyvinyl chloride (PVC) is one of the most versatile polymers used in many field of applications. However, low thermal stability during the processing of PVC is a major concern and leads to PVC degradation. During PVC processing, thermal degradation occurs due to dehydrochlorination which develops conjugated double bonds in the PVC backbone. 4,5 This degradation is responsible for the sudden change in the PVC color and deterioration of mechanical properties. To overcome the problem of thermal stability of PVC, various heat stabilizers are being used. Among the stabilizers, lead based, organotin compounds and mixed metal soaps constitute the majority.
Extensive research has been carried out to use the nontoxic heat stabilizers for PVC. One of its kinds is layered double hydroxides (LDHs). LDH is an inorganic compound having HCl-scavenging ability to improve the thermal stability of PVC. 6 Lin and coworkers reported that optimum level of LDH is 1–2 phr at which substantial improvement in thermal stability of PVC can be observed. 7 Zhu and coworkers studied the effect of calcium carbonate (CaCO3)/lithium chloride (Li2CO3) on the degradation behavior of PVC and their effect on HCl-scavenging ability. 8 Literatures also have reports that various fillers like silica, clay, CaCO3 and metal hydroxides or oxides may enhance the properties of polymers including PVC. 9 Some of these fillers, like CaCO3 and metal hydroxides or oxides, act as HCl scavenger. Mechanism for scavenging HCl involves reactions of fillers with HCl liberated from PVC, which results in the unavailability of HCl for autocatalytic dehydrochlorination. Therefore, there is decrease in the rate of further thermal degradation of PVC. HCl-scavenging effect of particulate fillers such as red mud, calcium carbonate (CaCO3) and dolomite on the thermal degradation of PVC was investigated by thermogravimetric/mass spectrometry. 10 Also Stephanie and coworkers investigated the synergistic effect of CaCO3 and poly(vinyl butyral) on the thermal stability of PVC. 11 Gupta reported the synergistic effect of hydrotalcite and diketones with metal stearates on the thermal stabilization of PVC with improved thermal stability of the PVC. 12 However, the synergistic effect of CaCO3 and LDH on the thermal stability of PVC has not been studied so far.
Hence in the present investigation we studied the synergistic effect of LDHs with micro- as well as nano-sized CaCO3 on the thermal stabilization of PVC. In this work, we compounded the samples with the help of Brabender Plastograph machine, using LDHs and CaCO3 in various proportions. Finally, the PVC sheets were prepared on compression molding machine, and the thermal stability of PVC composites was studied by thermogravimetric analysis (TGA). Morphology and mechanical property were studied to investigate the dispersion of fillers in the polymer matrix and reinforcement of matrix by the filler, respectively.
Experiment
Materials
A suspension-grade PVC (Reon grade–67) was obtained from the Reliance Industries Ltd., India. Calcium stearate, processing aid and stearic acid were kindly given by the Aryavarat Chemicals Ltd (Jalgaon, Maharashtra, India). All analytical grade chemicals and solvents such as glutaric acid, zinc oxide and toluene were used as obtained from S D Fine Chemicals (Mumbai, India). Other chemicals such as calcium chloride, potassium carbonate and polyethylene glycol (PEG; molecular weight 6000) were procured from the Qualigens India Ltd (Mumbai, India).
Synthesis of zinc glutarate
Zinc glutarate was synthesized from zinc oxide and glutaric acid. 13 The powdered zinc glutarate was dried in a vacuum at 100°C for 24 h. Yield of zinc glutarate prepared in the laboratory was 99.8%.
Synthesis of nano-sized CaCO3
Solution of calcium chloride (111 g, 1 M) was prepared by dissolving it in distilled water (100 mL). The PEG solution (372 g, 0.062 M) was prepared by dissolving it in hot distilled water (100 mL). These two solutions were mixed properly and digested gently for 12 h. Third solution, that is, potassium carbonate (106 g, 1 M) dissolved in distilled water (100 mL) was then added slowly to the above mixture under constant stirring. The newly formed particles of CaCO3 were allowed to digest overnight. Nanoparticles were filtered, washed thoroughly with distilled water until the particles were freed of PEG traces and then dried at 110°C for 2 h. CaCO3 filler was heated at about 250°C for removing traces of moisture before compounding. 14 –18
Preparation of the PVC/CaCO3 nanocomposites
Different PVC formulations were prepared by varying concentrations of nano-sized CaCO3 (1, 3 and 5 phr), keeping the LDH amount constant (2 phr). At the same time, commercial CaCO3 PVC composites were also prepared for comparison of their performance with nano composites of PVC. All PVC formulations contain Ca stearate/Zn glutarate as primary stabilizer, plasticizer and lubricant. These formulations were dry blended in a mixer for 6–7 min. Then the dry blended PVC formulations were processed in a Brabender Plastograph internal mixer (GmbH & Co. KG, Duisburg, Germany) at 160°C for 5 min and at a mixing speed of 45 r/min. Successively, the PVC composites obtained were compression molded on heated compression molding machine at 170°C for 10 min. PVC sheets of 1 mm thickness were obtained, which were used for further analysis.
Characterization
X-Ray diffraction and transmission electron microscopy (TEM)
The particle size of the synthesized nano-sized CaCO3 particles was determined using x-ray diffractometer (model: Xpert MPD; Philips, Holland) in the range of 0–1000 cps and diffraction angle of 0°–40° in sovitzkey Golay’s mode. Later, the size and morphology of the prepared nano fillers were confirmed using TEM (Philips Tecnai-20) at an accelerating voltage of 200 kV. The sample of synthesized fillers was dispersed in acetone in an ultrasonic bath for sufficient time before the analysis of particle size using the particles disperser on a C-coated grid
Thermogravimetric analysis
Thermal degradation behavior of the PVC samples was studied using Shimadzu thermogravimetric analyzer (TGA 50, Japan). Temperature programming was done from room temperature to 600°C at a heating rate of 20°C/min.
Scanning electron microscopy
Extent of dispersion of fillers and surface morphology of the PVC composites were inspected with scanning electron microscope (SEM JEOL JSM-6360A, Japan). Prior to analysis, the PVC samples were coated with platinum under vacuum.
Mechanical testing
Tensile strength of PVC sheets was measured using a universal tensile testing machine (Polyplast, Mumbai, India), according to ASTM D 638. The crosshead speed for testing was 50 mm/min, and all measurements were performed five times to obtain the average.
Results and discussion
Particle size determination
Particle size of synthesized nano-sized CaCO3 was determined using x-ray diffraction pattern as shown in Figure 1, using the Scherer’s formula. XRD of nano-CaCO3. CaCO3: calcium carbonate; XRD: x-ray diffraction.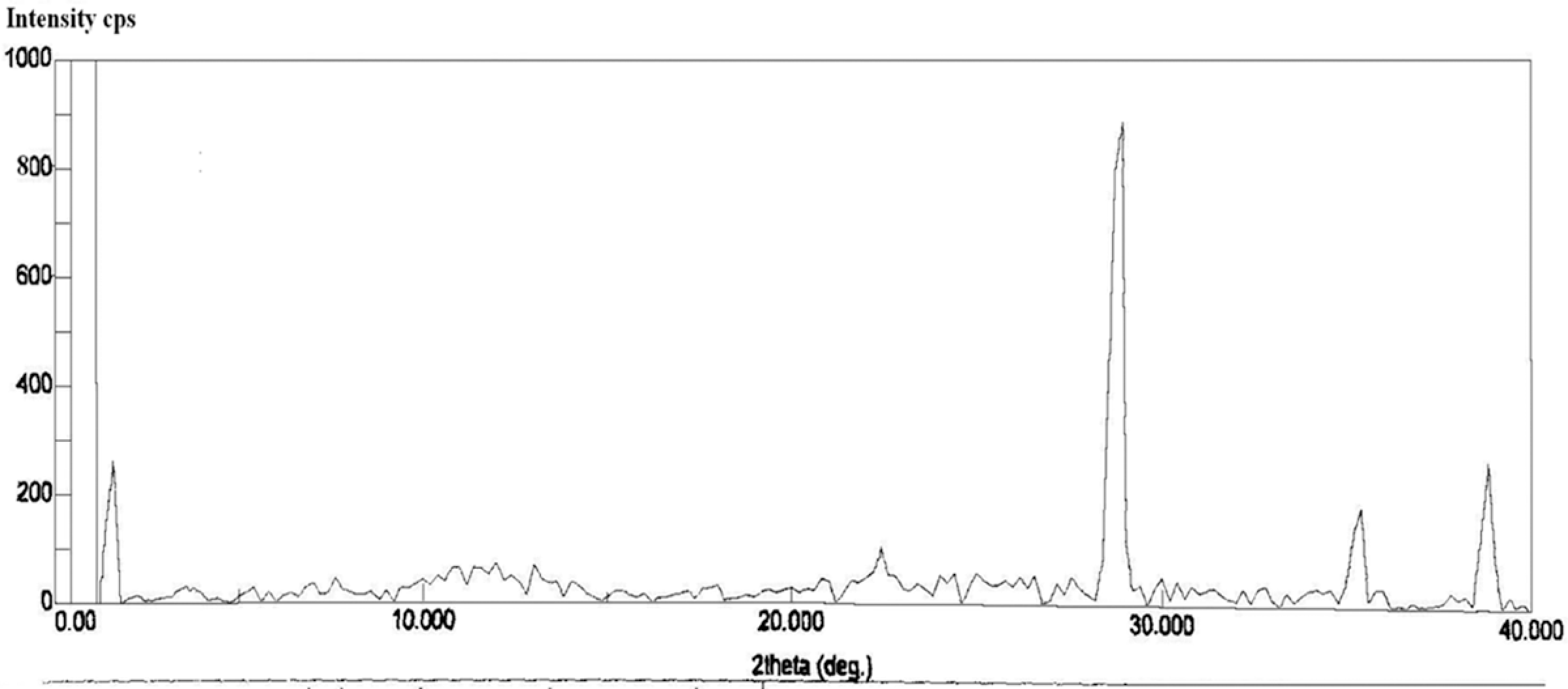
In the current study, the size obtained for synthesized CaCO3 work was found to be in 44–66 nm range, which was later confirmed by TEM.
Particle size of synthesized CaCO3 nanoparticles confirmed from TEM
In the present work, the particle size of the synthesized CaCO3 filler was found to be in the range of 40–70 nm as verified using TEM micrograph (Figure 2).

Transmission electron microscopy micrograph of calcium carbonate nanoparticles prepared by in situ deposition technique.
Thermal stability of PVC
TGA was used to investigate the thermal stability of PVC. The typical TGA curves of representative PVC samples are shown in Figure 3. It is reported in the literature that PVC degradation undergoes a two-step process. 19 The first step involves dehydrochlorination of PVC around 200°C–350°C. The second step (350°C–550°C) mainly involves the breaking of the backbone chain of PVC due to the loss or pyrolysis to low-molecular-weight fragments produced by the decomposition of PVC.
TGA thermograms of PVC samples (a) PVC without synergists (b) with 2 phr LDH (c) with 2 phr LDH + 3 phr micro-CaCO3 (d) 2 phr LDH + 3 phr nano-CaCO3. TGA: thermogravimetric analysis; PVC: polyvinyl chloride; LDH: layered double hydroxide; CaCO3: calcium carbonate.
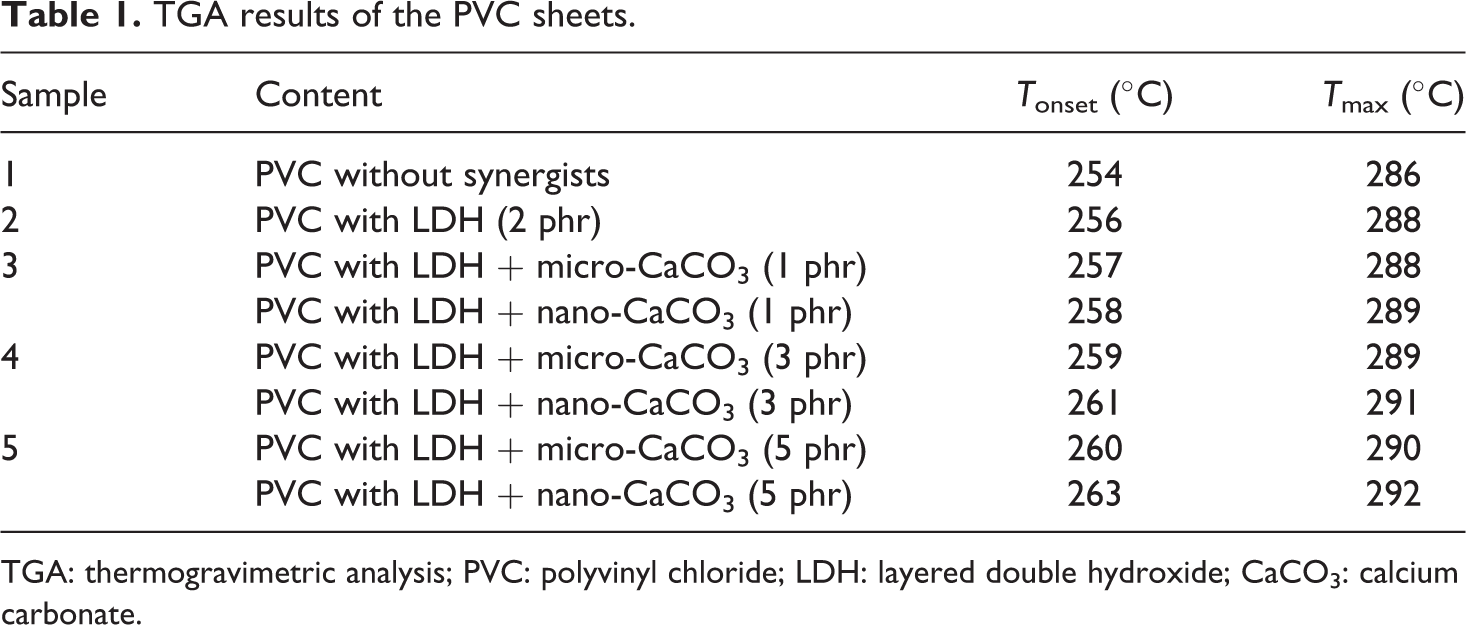
The data obtained from the TGA are shown in Table 1 and represented in Figure 4. The results show that the thermal stability of PVC is improved in all the case, that is, with the addition of LDH only in combination with micro-sized and nano-sized CaCO3 in the PVC stabilizer system. Addition of only LDH along with the conventional Ca/Zn stabilizer system increases the onset temperature, while use of micro- as well as nano-sized CaCO3 along with LDH further improves the thermal stability of PVC in terms of increased onset temperature. However, slight increase in the inflection temperature (T
max) was observed. Better thermal stability is thus observed when the PVC was added with 2 phr LDH in association with 3 and 5 phr nano-sized CaCO3. As compared to the micro-sized CaCO3 in the stabilizer system, nano-sized CaCO3 show better results for the thermal stability of PVC. This may be attributed to more surface area available for nano-size CaCO3 to interact with liberated HCl during PVC degradation as compared to interactions with micron size CaCO3.
20
This indicates that the thermal stability of PVC improves by LDH which further enhanced synergistically by the addition of CaCO3. These results could be explained by noting that calcium ions present in the nanoparticles are properly dispersed in a PVC matrix, scavenged the liberated HCl during the PVC degradation and subsequently formed calcium chloride.
21
Along with this, the ion exchange mechanism of LDH with the release of HCl from PVC prevents further autocatalytic dehydrochlorination process, which in turn results in better thermal stability of PVC achieved by scavenging the liberated HCl.
5,6
This clearly points out that there is a synergism between LDH and nano-sized CaCO3. The mechanism of reaction may be written as follow
11,22
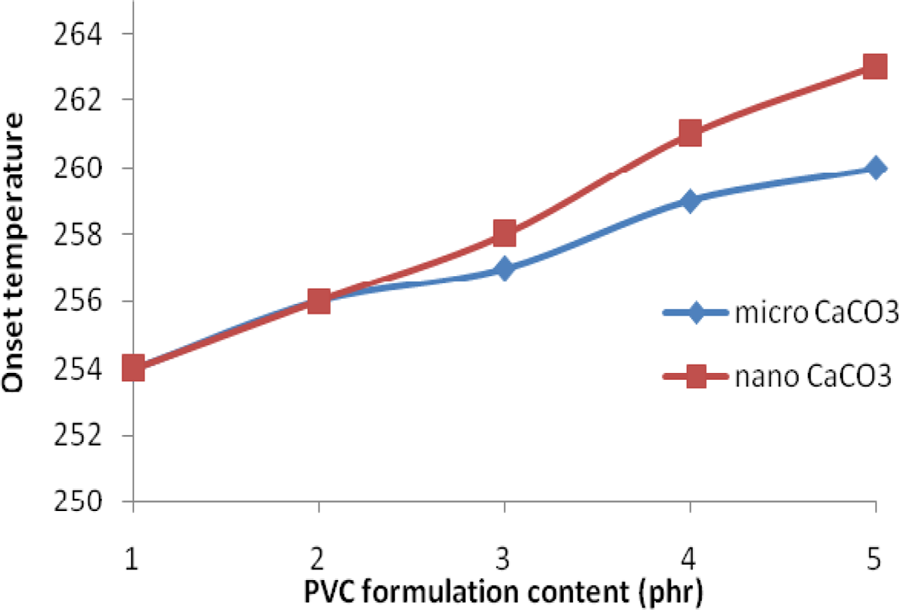
Onset temperatue of PVC samples without and with synergists: (1) PVC without synergists, (2) PVC with 2 phr LDH, (3) PVC with 2 phr LDH + 1 phr micro- and nano-CaCO3, (4) PVC with 2 phr LDH + 3 phr micro- and nano-CaCO3 and (5) PVC with 2 phr LDH + 5 phr micro- and nano-CaCO3. PVC: polyvinyl chloride; LDH: layered double hydroxide; CaCO3: calcium carbonate.
TGA results of the PVC sheets.
TGA: thermogravimetric analysis; PVC: polyvinyl chloride; LDH: layered double hydroxide; CaCO3: calcium carbonate.
Morphological study
Figure 5 depicts the representative SEM micrographs of the surface morphology of the PVC sheets containing 3 phr of micro- and nano-sized CaCO3. The SEM micrographs show that the micron size CaCO3 filler (Figure 5) are not dispersed properly in a PVC matrix and formed bigger agglomerates than that of its nano-size CaCO3. Incorporation of filler in Brabender Plastograph EC imparts sufficient dispersion in both micro- and nano-sized CaCO3. Proper dispersion gives maximum interfacial area for nano-sized CaCO3 to scavenge the liberated HCl during the high-temperature processing of PVC. Therefore, it prevents the autocatalytic action of HCl, which stops and prevents further dehydrochlorination of PVC. Thus, we conclude that the proper dispersion of nano-sized CaCO3 provides better HCl-scavenging properties than the micron-sized one CaCO3.

SEM of PVC sheets having 3 phr of (a) micro-CaCO3 and (b) nano-sized CaCO3. PVC: polyvinyl chloride; CaCO3: calcium carbonate; SEM: scanning electron microscopy.
Mechanical testing
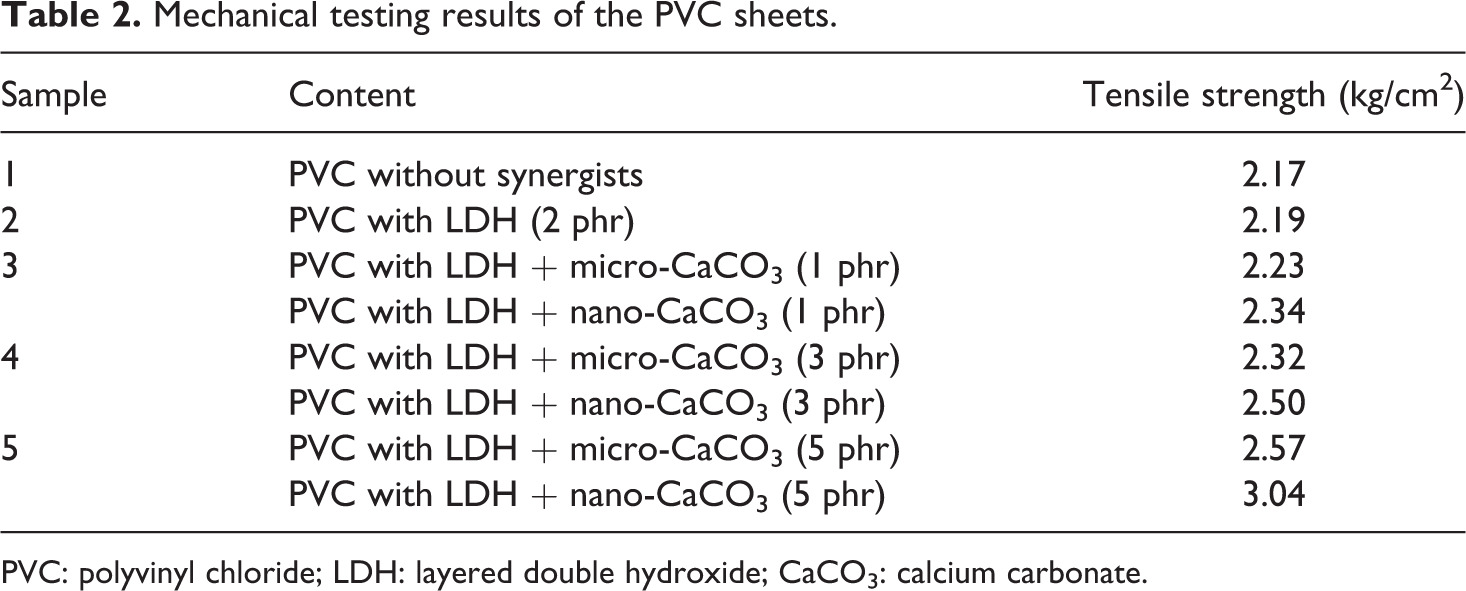
The effects of commercial and nano-CaCO3 with LDH on the tensile strength of PVC sheets are shown in Figure 6, and the corresponding data are given in Table 2. From the figure it was observed that the initial tensile strength of the PVC composites does not changed substantially but increased further with the addition of micro as well as nano-CaCO3 along with LDH into the PVC stabilizer system. By the addition of 1, 3 and 5 phr CaCO3 with LDH in PVC, the tensile strengths of the nano-CaCO3–filled PVC composites were found to be much better than those of the micro-CaCO3–filled PVC composites. The most substantial increase in the tensile strength value was observed at 3 and 5 phr of nano-CaCO3. This is because the size of nano-CaCO3 is much smaller and through their higher surface area can have better interactions with PVC matrix than those of micro-CaCO3. This provides better interfacial interaction between matrix-filler surface, which results in the improvement of the mechanical strength of the polymer matrix. 23
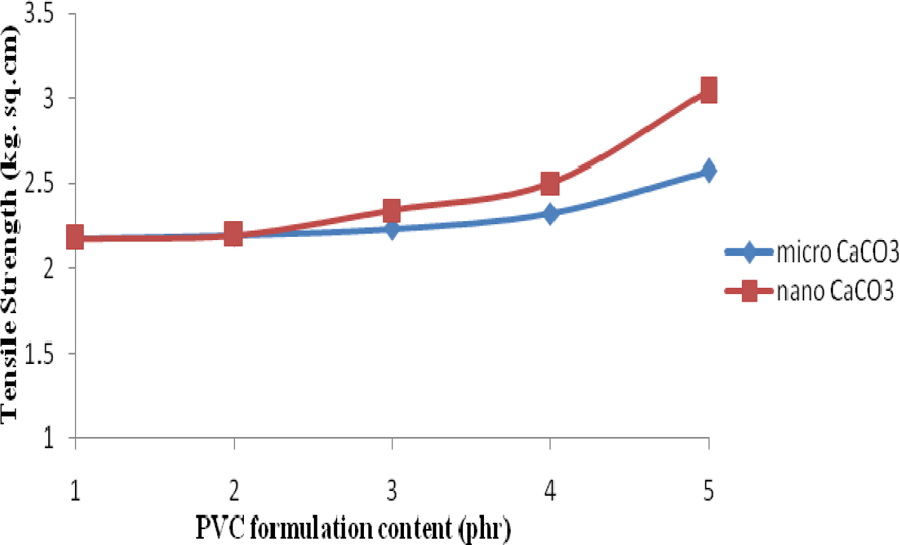
Tensile Strength of PVC samples without and with synergists: (1) PVC without synergists, (2) PVC with 2 phr LDH, (3) PVC with 2 phr LDH + 1 phr micro- and nano-CaCO3, (4) PVC with 2 phr LDH + 3 phr micro- and nano-CaCO3 and (5) PVC with 2 phr LDH + 5 phr micro- and nano-CaCO3. PVC: polyvinyl chloride; LDH: layered double hydroxide; CaCO3: calcium carbonate.
Mechanical testing results of the PVC sheets.
PVC: polyvinyl chloride; LDH: layered double hydroxide; CaCO3: calcium carbonate.
Conclusion
Nano-sized CaCO3 was prepared in the laboratory and its particle size was found to be in the range of 40–70 nm. We prepared different PVC composites with varying content of micro- and nano-sized CaCO3, by keeping the LDH amount constant. Thermal stability of PVC sheets were studied by TGA. Addition of nano-sized CaCO3 increases the onset temperature of PVC more substantially than that of micro-CaCO3. Larger surface area provided by nano-sized CaCO3 and better scavenging reaction of HCl resulted in enhanced thermal stability of PVC and better tensile strength than the micro-sized CaCO3-filled PVC. Better synergism was observed in LDH with nano-sized CaCO3 stabilizing system on the thermal stability of PVC compared to the synergistic effect of LDHs with micro-sized CaCO3.
Footnotes
Acknowledgement
The authors thank the Aryavarat Chemical Limited, Jalgaon, India, for providing additives.
Funding
This work was supported by the University Grant Commission (UGC), New Delhi, India.