Abstract
Mesoporous silica nanoparticles were synthesized by base-catalyzed hydrolysis of tetraethyl orthosilicate in the water-acetone medium. For surface modification of mesoporous silica, commercially available silane, 3-(trimethoxysilyl)propyl methacrylate was reacted with mono-thioglycerol via 2,2,6,6-tetramethylpiperidin-1-yl)oxidanyl (TEMPO) aided thiol-ene reaction, and the obtained silane was incorporated on the surface of neat mesoporous silica to graft extended alkyl chains terminating in hydroxyl groups. The prepared MSNs were characterized by thermal gravimetric analysis (TGA), Fourier transform infrared spectroscopy (FT-IR), nitrogen adsorption-desorption analysis, scanning electron microscopy, and dynamic light scattering. These MSNs were compared to conventionally used commercial silica as an additive in tubular quenched polypropylene films (TQPP) for their anti-blocking, thermal, mechanical, optical, and barrier properties. Organic modification of MSNs decreased the blocking force in TQPP films. The mechanical properties including % elongation and tensile strength increased for TQPP films with mesoporous silica, while it decreased for films containing commercial silica. Both modified and unmodified MSNs decreased haze and enhanced gloss in TQPP films, showing improved optical properties as compared to commercial silica particles. In TQPP film with modified MSNs, a reduced value of the water contact angle was obtained, indicating better wettability of the surface of the TQPP film.
Keywords
Introduction
Plastic films provide several benefits over rigid forms of packaging materials available, as they are lightweight with appreciable barrier properties, and can be reused or recycled. According to the Research and Markets global survey for 2021–2026, the plastic film market is expected to rise from USD 184 billion in 2020 to around USD 220 billion by 2026. Polypropylene (PP) is known for its excellent properties, such as high chemical and corrosion resistant nature, low cost, and easy processability. PP films have good heat resistance and therefore can be sterilized at high temperatures, making it an attractive candidate for packaging films. Apart from this, PP is translucent, possesses good flexibility, tensile strength, and high gloss.1,2
Nanoparticles have been utilized as additives in preparing packaging films to improve mechanical, barrier, and thermal properties.3–9 In the last few decades, researchers have worked towards developing high performance coatings or films, by combining the properties or applications of organic polymers with versatility offered by inorganic materials. However, nanoparticles can often lead to aggregation owing to their small size, that can lead to deterioration of inherent properties of the resin. A common concern encountered by manufacturers is the blocking phenomenon, which can be described as sticking of polymer films together due to Van der Waal’s forces of attraction. The adhesive force increases when the distance between two adjacent films is decreased, or the films are pressed together. To counter this issue, anti-blocking additives, such as silica, magnesium silicate, calcium carbonate, aluminum silicate etc. are added to the polymer composite formulation. Silica is known for its abundance in terms of availability as well as for its existence in amorphous, crystalline, and colloidal forms. Silica particles are used commercially as efficient anti-blocking agents as they create roughness on the polymer surface and minimize the contact or adhesion between two adjacent films. It is also popular for its reinforcing properties in elastomers, use in adhesives, cosmetics, as an adsorbent or dehumidifying agent, and in the food industry as an anti-caking agent to control the rheological properties of the product.10,11 They also display other interesting properties, including good chemical and thermal stability, permeability, low refractive index (similar range as polyolefins), and high surface area. 12 The incorporation of silica particles has proven to improve a wide range of properties of the base polymer, such as, electrical and mechanical properties, weathering and thermal resistance, optical properties, flame retardancy, impact properties, and abrasion resistance.13–15 The chemical purity conferred by the synthetic silica particles finds applications in food and medicine packaging as it is devoid of any toxins and heavy metals. Literature reports have suggested that the incorporation of silica nanoparticles in PP resins can potentially improve the lifetime of the product under temperature and high-field stresses, as well as insulating and dielectric properties.16–18 Crosby et al. evaluated the performance of combination of different types of commercially available silica and other anti-blocking agents and analyzed their performance in terms of openability of tubular quenched water polypropylene (TQPP) films, and as clarifying agents. They observed that formulations with anti-blocking agents with higher particle size of silica resulted in lowest coefficient of friction or best anti-blocking property but resulted in poor optical properties, as overall haze in the film increased. On the other hand, formulations with silica of relatively smaller particle size resulted in lower haze or better clarity but were not efficient enough as anti-blocking agents. 19 Bellel et al. deposited thin silica films on polypropylene surface through plasma induced chemical vapor deposition method and observed that the enhanced polarity resulted in its improved wettability. 20 Jankong and co-workers observed an increase in the tensile strength of PP films from 23 MPa to 26 MPa at 5% loading of silica nanoparticles but observed phase separation and agglomeration at higher loading of silica. They also observed an increase in oxygen permeation because of the smaller spherulite size of PP after incorporation of silica. 21 Mesoporous silica has been known to provide an edge in improving the properties of polymer composites as its inherent pore volume provides easy access to the polymer chains. It is used in several wide-ranging applications, including polymerization, drug-release, catalysis, membrane separation etc. The extensively high surface area of mesoporous substrates allows effective surface modification to alter the bulk properties as desired.22–24
Though silica nanoparticles have been extensively explored as additives to improve various properties of PP films, mesoporous silica hasn’t been studied for PP film formulations. In this paper, we have synthesized mesoporous silica particles and analyzed its performance as an additive on thermal, mechanical, and optical properties of tubular quenched polypropylene (TQPP) films as compared to the commercial silica grade used commonly in industries for preparing TQPP films. To improve the dispersion of mesoporous silica in PP matrix and decrease agglomeration, its surface was modified to graft an extended alkyl chain terminating in hydroxyl groups. The incorporation of non-modified (control sample) and modified mesoporous silica particles improved the tensile strength as well as % elongation of TQPP films, while commercial silica deteriorated its mechanical properties. Enhancement in gloss and clarity (low haze) was also observed with both kinds of mesoporous additives, while commercial silica increased haze of TQPP films owing to agglomeration. Surface modification of mesoporous silica also decreased the blocking force in TQPP films as compared to control mesoporous silica. To the best of our knowledge, an optimum balance of optical, mechanical, barrier properties as well as anti-blocking ability hasn’t been reported with mesoporous silica particles in PP films.
Experimental section
Materials
PP Homopolymer grade PP1100FS, was received from Indian Oil Corporation Limited, Panipat Refinery, India in powder form without containing any additives. Commercial grade silica grade Sylobloc 45H was received from W.R. Grace. Tetraethyl orthosilicate (TEOS, 98%) and 3-(trimethoxysilyl)propyl methacrylate were supplied by TCI, India. Methanol (HPLC grade) and liquor ammonia (30%) were obtained from Fischer Scientific. Cetyltrimethylammonium bromide (CTAB, 98%) was procured from Spectrochem, India. Mono-thioglycerol was procured from Sigma-Aldrich. Solvents used included ethanol (99.9%; Analytical CS reagents), anhydrous toluene (99.5%; from SRL), and acetone (from Merck). All the chemicals were used as received.
Synthesis of control and organically modified mesoporous silica
Synthesis of control mesoporous silica
Control mesoporous silica (CMS) was synthesized via base-catalyzed hydrolysis of silane precursor TEOS in a water-acetone solution mixture using CTAB as a template. A typical procedure involved the dissolution of 35 mmol of CTAB in 640 mL of distilled water, followed by the addition of 370 mL of acetone and 200 mL of liquor ammonia solution. The reaction mixture was stirred at 30 °C for a duration of 20 min. When CTAB was completely dissolved, 250 mmol of TEOS was added to the reaction which resulted in immediate precipitation of silica particles. The reaction mixture was stirred for another 2 h at the same temperature. The silica particles obtained were precipitated by centrifugation at 7000 rpm, followed by 3–4 times washing with distilled water and twice with ethanol. The precipitated silica particles were collected and dried in a hot air oven at 80 °C. After drying, silica powder was collected and dried at a temperature of 160 °C for 2 h, followed by calcination at 550 °C for 8 h for complete removal of the surfactant CTAB. The product yield post-calcination was around 13 g.
Synthesis of modified mesoporous silica with alkyl chain terminating with hydroxyl groups
The first step of surface modification of mesoporous silica involved a reaction of 17 mmol of 3-(trimethoxysilyl)propyl methacrylate with 18 mmol of mono-thioglycerol in 15 mL of methanol. The reaction was stirred for 16 h at 35 °C. The formation of modified silane was confirmed using 1H NMR spectroscopy. Further, 30 g of CMS was dispersed in 300 mL of anhydrous toluene, followed by addition of modified silane solution in methanol (approximately 21 mL). The reaction mixture was then refluxed at 120 °C for 48 h. The modified mesoporous silica with grafted alkyl chain terminating in hydroxyl groups (abbreviated as MS-OH) was collected post-precipitation via centrifugation and washed sufficiently with toluene and ethanol. The obtained MS-OH was dried for around 16 h in a hot air oven at 90 °C. The synthetic scheme for the synthesis of CMS and MS-OH has been given in Scheme 1. Synthetic route for (A) control mesoporous silica (CMS) and (B) modified mesoporous silica with grafted alkyl chain with terminal hydroxyl groups (MS-OH).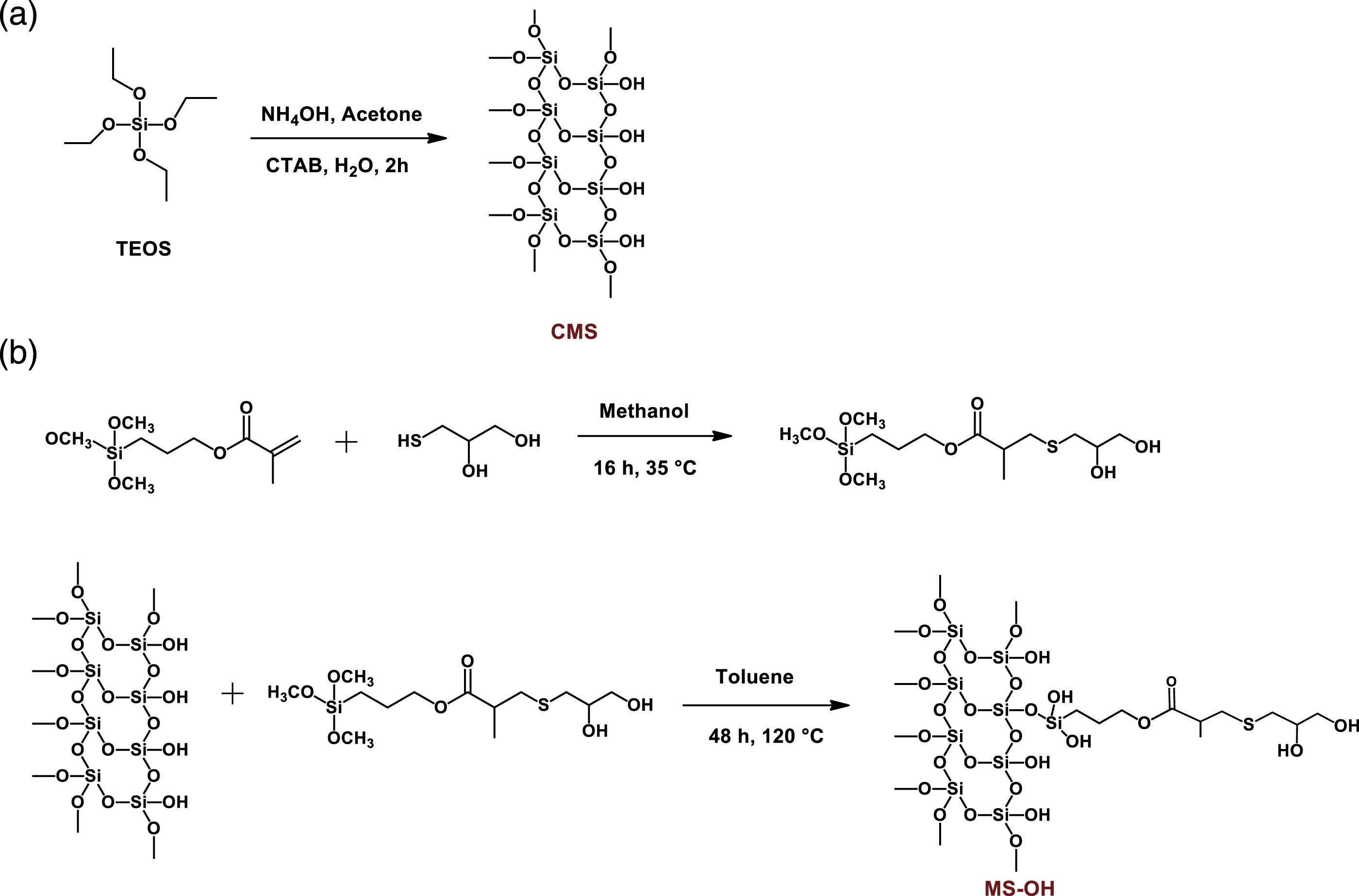
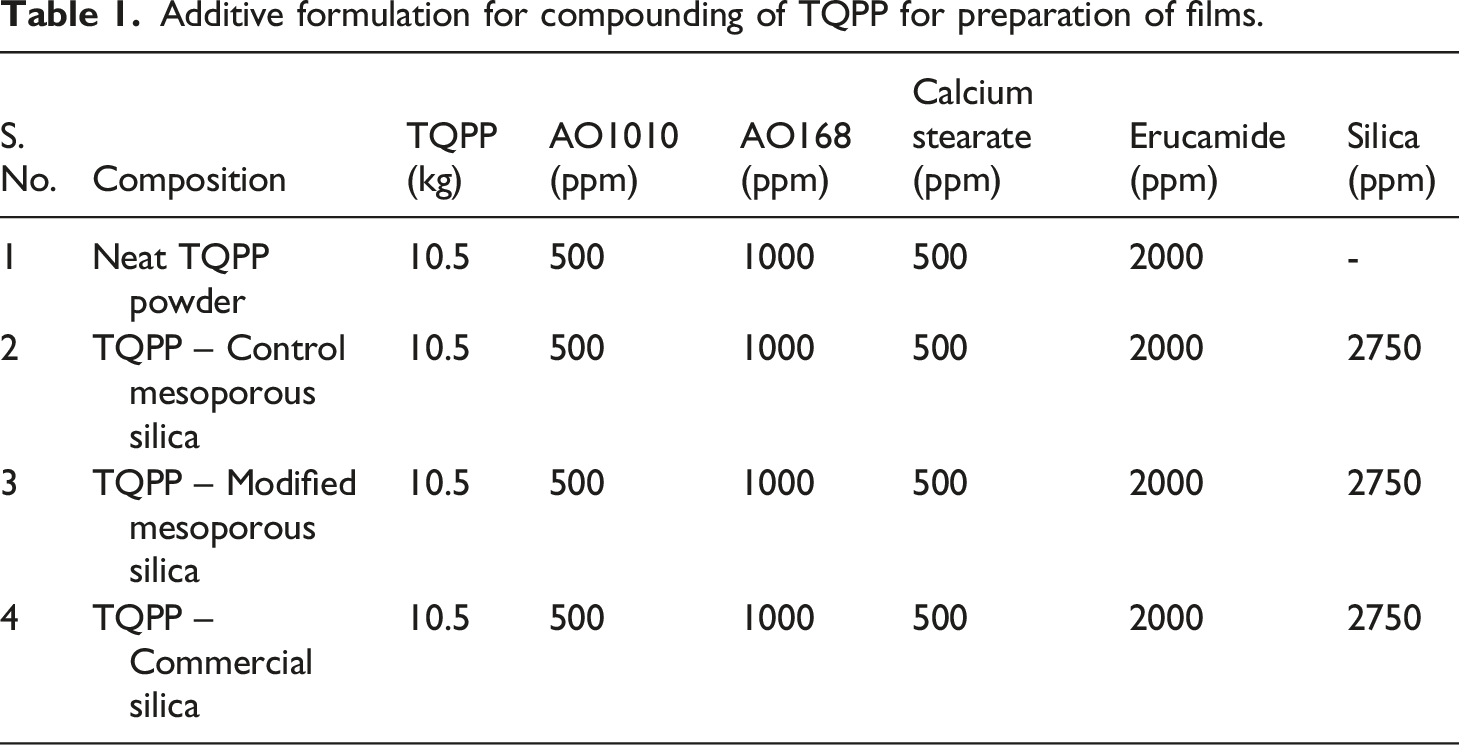
Compounding of TQPP with silica and preparation of films
Additive formulation for compounding of TQPP for preparation of films.
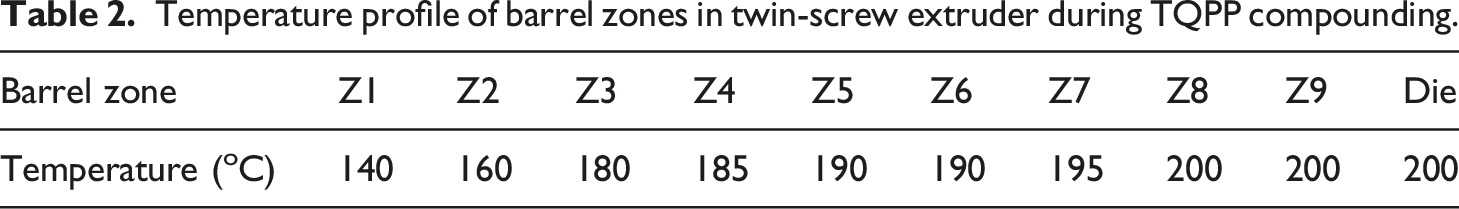
Temperature profile of barrel zones in twin-screw extruder during TQPP compounding.
The TQPP films listed above were prepared using blown-film extrusion process. The compounded homopolymer granules were fed in an extruder, and the extrudate coming out of the die was blown by air pressure and consequently quenched using water maintained at 15 °C.
Characterization techniques
Thermogravimetric analysis
Thermogravimetric analysis (TGA) for neat TQPP and TQPP-silica films was performed on Q-500 TA instrument. A sample weighing approximately 5–6 mg was analyzed at a heating rate of 10 °C/min in a nitrogen environment. All samples were dried at 90 °C overnight in a hot air oven prior to analysis.
Differential scanning calorimetry
Thermal characterization of prepared TQPP films by Differential scanning calorimetry (DSC) was carried out on Temperature Modulated Mettler Toledo DSC one star system instrument under a nitrogen atmosphere. A known sample weight of 5–6 mg was heated from 30 to 200 °C, held at 200 °C for 5 min to remove prior thermal history, and then cooled down to 30 °C. Cooling as well as a heating rate of 10 °C/min was used to record DSC thermograms.
Fourier transform infrared spectroscopy
Fourier transform infrared spectroscopy (FT-IR) spectra for the prepared mesoporous silica samples were recorded using a Thermo Nicolet IR 200 spectrometer within the spectral range of 4000-400 cm−1. KBr pellets for testing were prepared as circular disc of 1 mm diameter using a hydraulic press.
Dynamic light scattering
Dynamic light scattering (DLS) measurements of mesoporous silica, as well as commercial silica, were conducted on Malvern Zetasizer NANO ZS 90. Nearly 2 mg of the sample was dispersed in 40 mL of ethanol by probe-sonication for 5 min. The reported hydrodynamic diameter is an average of at least three measurements.
Field emission scanning electron microscopy
The morphology of all the silica samples and their particle size were analyzed on JEOL JSM-6610LV at 10 kV.
Nitrogen adsorption-desorption analysis
The surface area of mesoporous silica samples and commercial silica was determined using AutosorbiQ-XR-AG-AG Viton Quantachrome BET surface analyzer. The samples were dried for approximately 12 h in a hot air oven at 90 °C. Samples were degassed in vacuum and by heating at 100 °C prior to the experiment.
Melt flow index
The melt flow rate for neat TQPP and TQPP compounded with all three types of silica in consideration was determined using Gӧttfert MI 4. The reported Melt flow index (MFI) values were recorded against a load of 2.16 kg at 230 °C.
Contact angle measurements
Specimens of dimensions 2 × 2 cm2 were used to estimate the wettability of all the prepared TQPP films via sessile drop method using Drop Shape Analyser (DSA100 E). For the experiment, 2 μL drop of ultra-pure water was carefully deposited on the film surface and the angle between water and film surface was determined using Kruss Advance software. The contact angle values are reported as the mean of 20 measurements for each sample.
Optical properties
Surface Gloss was determined with BYK Gardner photometric unit of model Micro TRI/Gloss. The measurements were recorded at 60° angle as per the ASTM D-523 procedure. The % Haze in the TQPP films was estimated using Konica Minolta CM36DG Haze meter and the experiment was done as per ASTM D1003 standard.
Blocking force
Blocking Force between the adjacent film surfaces was quantified in Gram force units and the measurements were recorded according to ASTM D3354 standard using Dynisco D9047/230V Instrument.
Mechanical properties
The tensile strength and % elongation of all the prepared TQPP films was estimated using LLyods LR10 K Plus UTM instrument via procedure described in ASTM D882. Five specimens were tested for each sample and average values have been reported.
Barrier properties – water vapor transmission rate (WVTR) and oxygen transmission rate (OTR)
WVTR of all the prepared TQPP films was determined using Mocon-Permatran-W-3/33 MG Plus by procedure described in ASTM F1249 at 38 °C at 90% relative humidity. Oxygen Transmission Rate (OTR) of the films was examined using OTR Mocon 2/22H as per ASTM D3985 at 23 °C at 0% relative humidity.
Results and discussion
Characterization of mesoporous silica nanoparticles
As described above, base-catalyzed hydrolysis of TEOS in a water-acetone solution yielded CMS. In the first step of the post-modification procedure, 3-(trimethoxysilyl)propyl methacrylate was treated with mono-thioglycerol via thiol-ene chemistry using TEMPO as an initiator.24–28 The reaction mixture was continuously stirred until the disappearance of NMR signals corresponding to vinyl protons. The synthesis of modified silane was confirmed using 1H NMR, as shown in Figure 1. 1H NMR (400 MHz, CDCl3), δ ppm 4.08 (t, 2H), 3.7 (m, 1H), 3.62 (d, 2H), 3.51 (s, 9H), 2.81 (m, 1H), 2.72 (m, 2H), 2.53 (m, 2H), 1.7 (m, 2H), 1.2 (d, 3H), 0.65 (m, 2H). 1H NMR spectrum of modified silane formed after reaction of 3-(trimethoxysilyl)propyl methacrylate with mono-thioglycerol.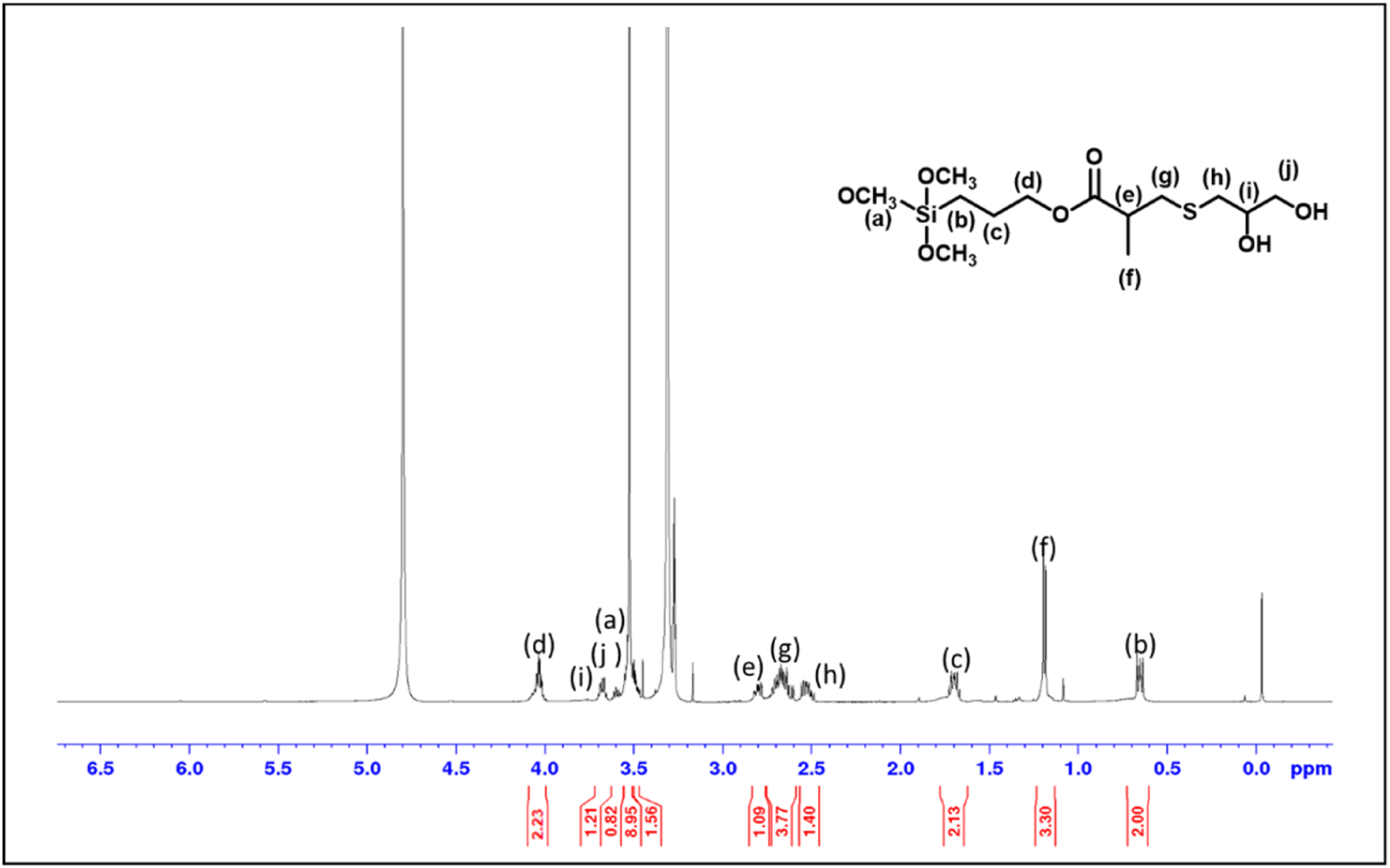
Thermal stability of all the silica samples was analyzed by TGA, as shown in Figure 2. The weight loss of around 17.7% for the modified mesoporous silica sample was due to the organic group grafted on the surface of mesoporous silica. The onset degradation temperature for MS-OH was around 343 °C which makes it suitable to process with TQPP. The initial dip in the weight loss observed in MS-OH is due to the water trapped in the pores of silica. The weight loss observed in the case of commercial silica can be attributed to the loss of water molecules adsorbed on the surface followed by condensation of surface hydroxyl groups. TGA curves of control mesoporous silica, modified mesoporous silica, and commercial silica.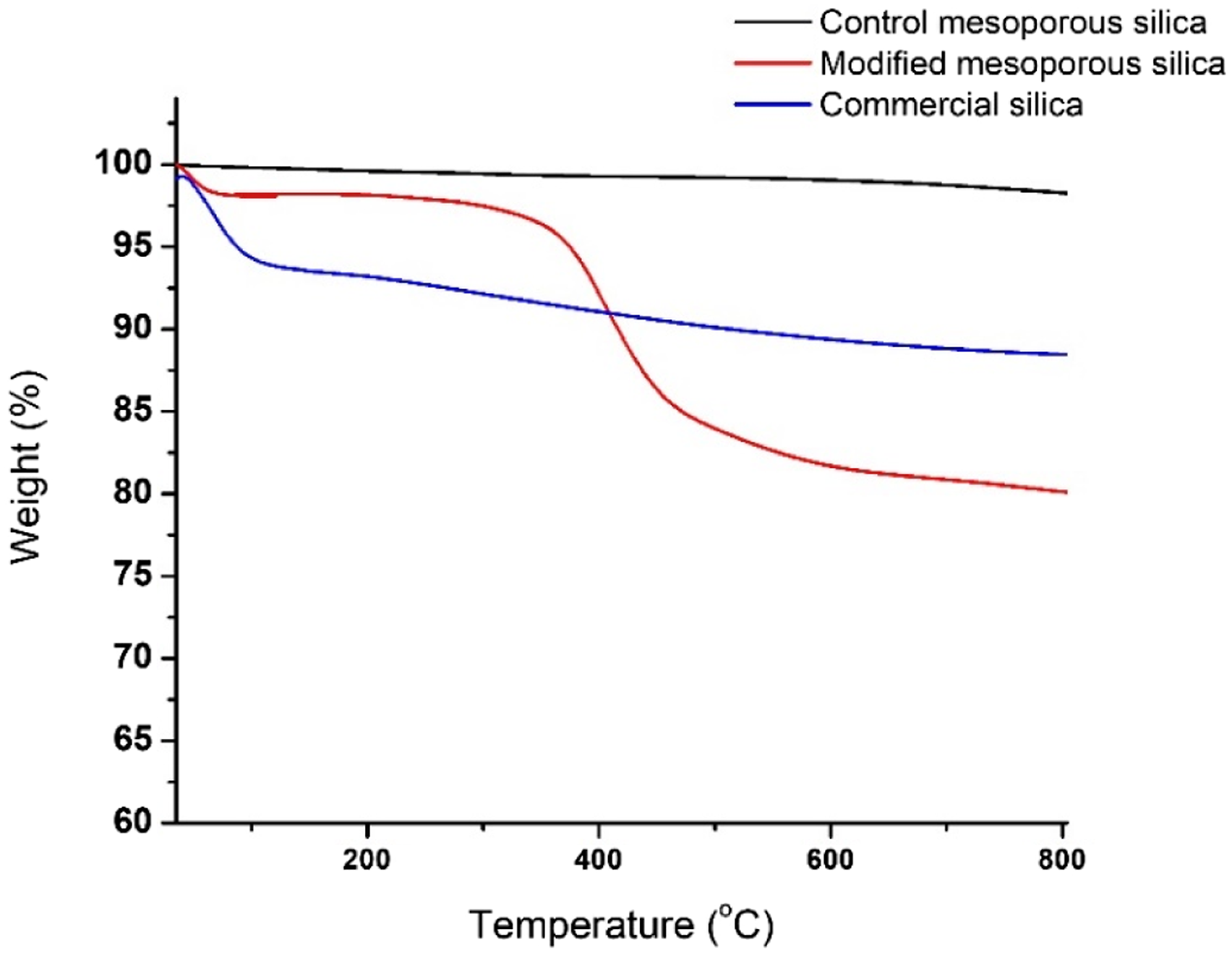
FT-IR analysis was carried out to identify the functional groups incorporated on the surface of MSNs post organic functionalization (as shown in Figure 3). The characteristic absorption bands of silica present in both neat and modified silica particles include a broad band at 1090 cm−1 due to Si-O-Si asymmetric stretching, 954 cm−1 corresponding to silanol Si-OH stretching, 810 cm−1 due to Si-O-Si symmetric stretching and 455 cm−1 because of Si-O-Si asymmetric bending. Additionally, a broad band at 3400 cm−1 due to OH stretching and around 1630 cm−1 corresponding to OH bending was observed. The organic functionalization on neat mesoporous silica particles was confirmed by a distinct band around 1720 cm−1 due to carbonyl stretching, 1460 cm−1 due to CH2 bending vibrations, and CH asymmetric and symmetric stretching at 2958 cm−1 and 2861 cm−1, which were absent in CMS. FT-IR spectra of control mesoporous silica and modified mesoporous silica.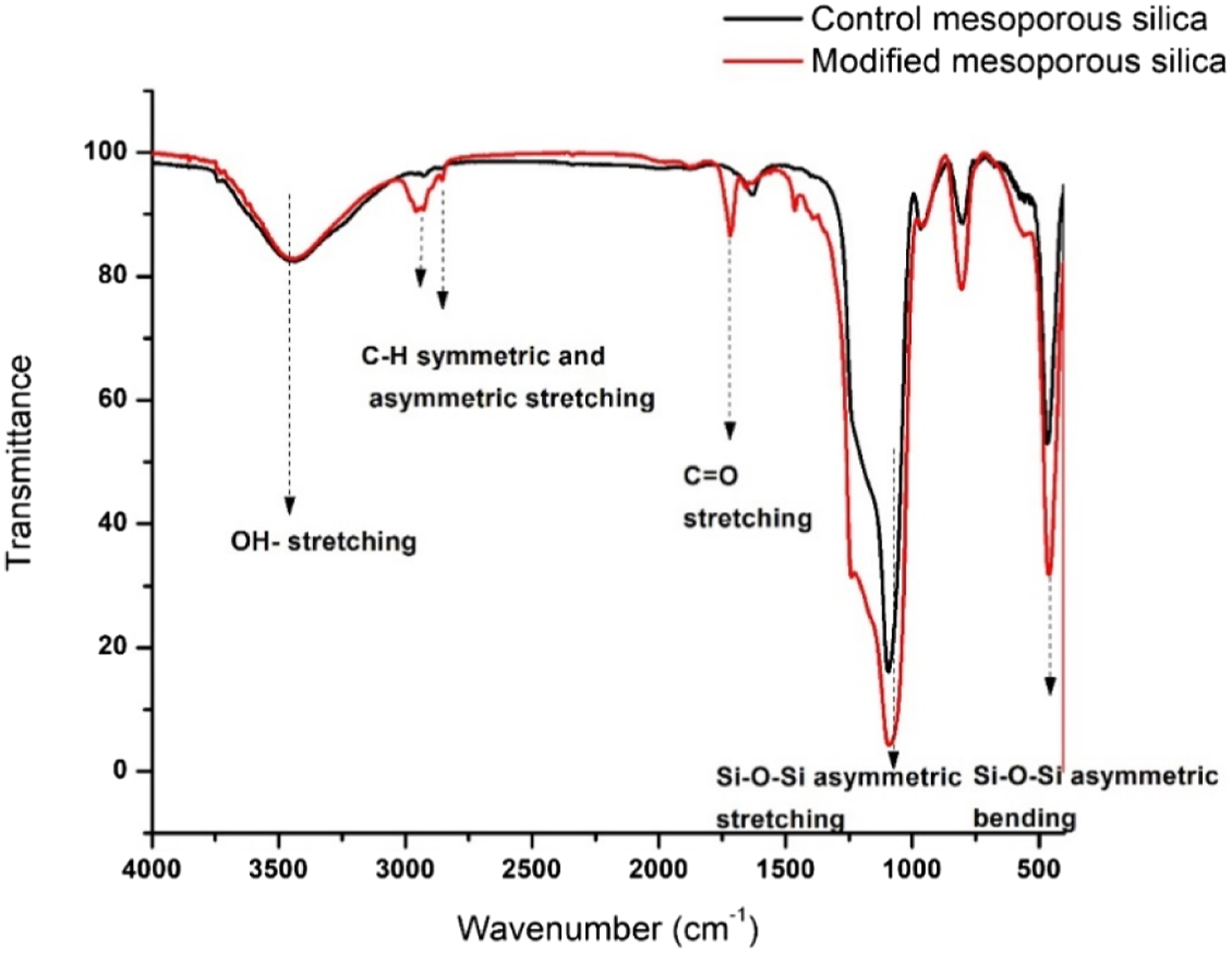
Field emission scanning electron microscopy (FE-SEM) of the mesoporous silica samples and commercial silica was carried out to study the shape and morphology of particles. The silica particles of CMS and MS-OH have spherical morphology, as shown in Figure 4(a) and (b), and lie in the range 400–700 nm. Commercial silica can be seen as large clusters of particles having irregular shape and size, as seen in Figure 4(c). Figure 4(d) shows a highly magnified FE-SEM image of commercial silica, where it can be seen clearly that the silica particles of extremely small size are aggregated together to form large clusters. FE-SEM images of (A) Control mesoporous silica (X 25,000) (B) Modified mesoporous silica (X 25,000) (C) Commercial silica (X 15,000) (D) Commercial silica (X 100,000).
Nitrogen adsorption-desorption analysis was done to determine the surface area and average pore size of all the three kinds of silica in consideration. As seen in Figure 5, both CMS and MS-OH exhibited Type IV adsorption isotherms, which is typically shown by all mesoporous substrates. It is also evident that the mesoporous structure was not altered post-surface modification of mesoporous silica. The BET surface area and pore diameter of all three kinds of silica have been listed in Table 3. The surface area for CMS and MS-OH was 1446 m2/g and 912 m2/g respectively. The surface modification of CMS to yield MS-OH resulted in decrease in the pore diameter as well, i. e. from 2.31 nm to 1.95 nm. Reduction in the surface area and pore diameter indicated incorporation of organic moiety on the surface of mesoporous silica. These observations are consistent with the literature reports.29,30 Commercial silica exhibited Type V adsorption isotherm, that is shown by porous materials displaying weak adsorbent-adsorbate interactions at low pressures, hence convex to the relative pressure axis. The surface area of commercial silica was found to be 416 m2/g. BET adsorption isotherms of control mesoporous silica, modified mesoporous silica, and commercial silica. Parameters obtained from BET (surface area and pore diameter) and DLS (particle size and Z-average size).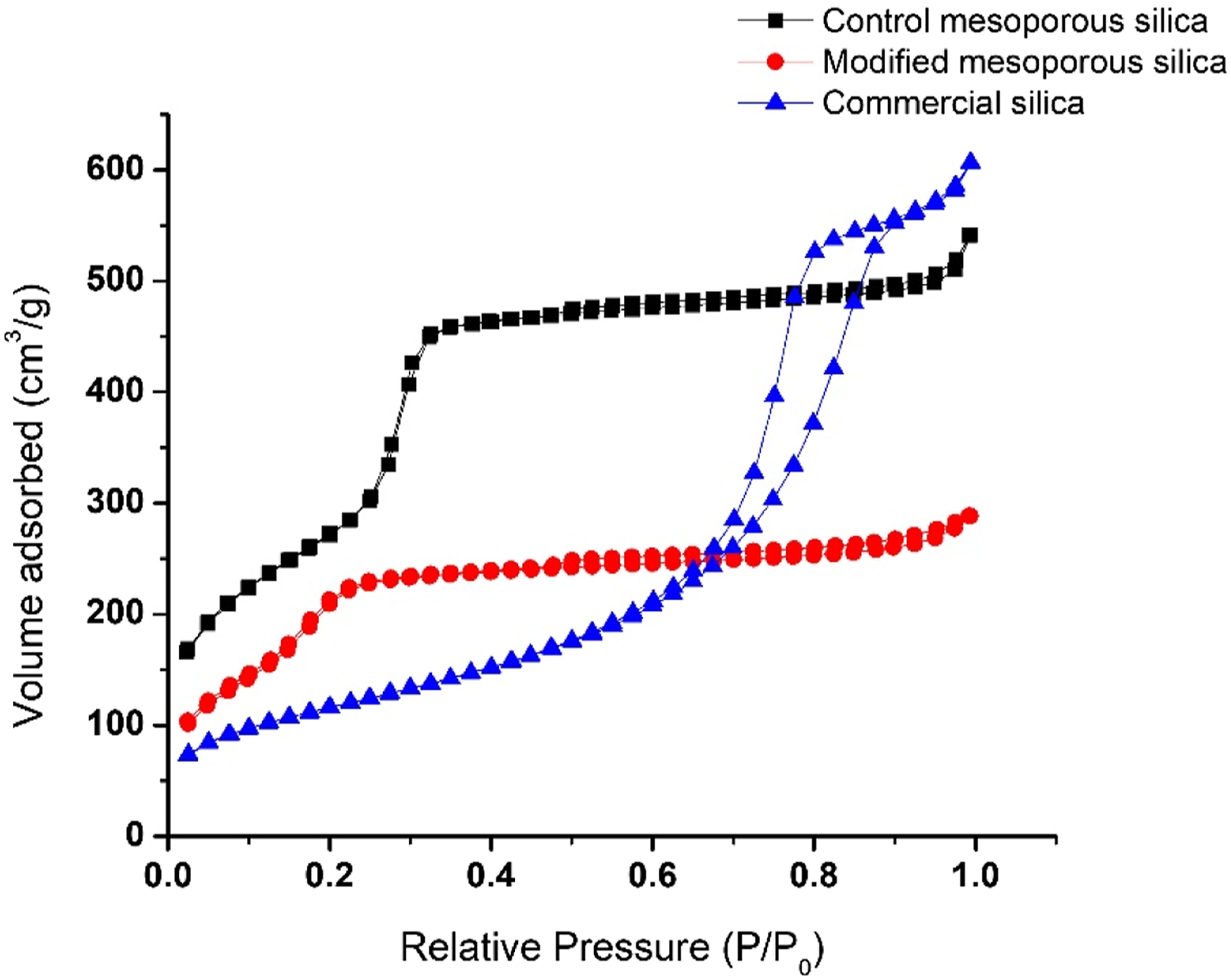
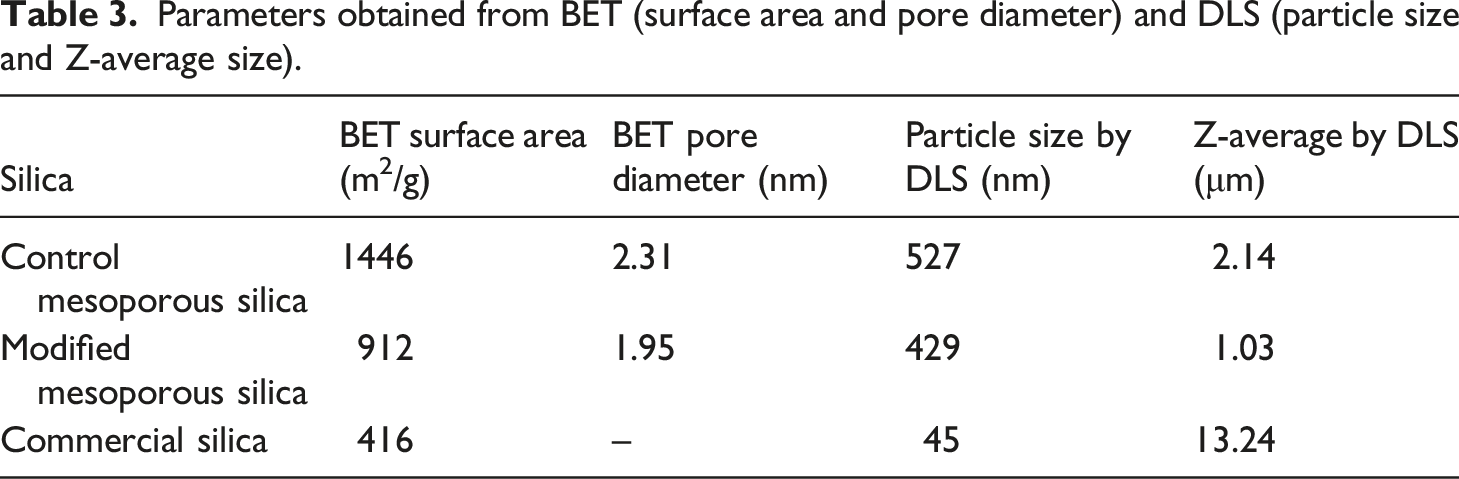
The particle size distribution in the silica samples was studied by dynamic light scattering (DLS). The average particle size and Z-average size (refers to the average size of the aggregate present in the solution) of CMS, MS-OH, and commercial silica have been listed in Table 3. The size of aggregated molecule decreased after surface modification in mesoporous silica, which indicated better dispersion of silica particles in the test solution. The commercial silica particles had the smallest size of individual particle that could be the reason for the largest size of aggregates or clusters present. The results obtained from DLS were found to be consistent with the FE-SEM images shown above (Figure 4).
The process of creating tubular water quenched PP film (TQPP) includes extrusion of film from an annular die that yields a tube through which air is passed into its interiors. Contact is created between the tube and the water bath underneath the die. The tube is then flattened into a film and subjected to drying and winding onto a roll. The conventional formulation for preparing a TQPP film includes PP resin with antiblock/slip additives. All the TQPP films prepared were analyzed for their thermal, optical, barrier, and mechanical properties. The blocking force as well as wettability of each film was also examined.
The thermal stability of the films was evaluated by TGA analysis (Figure 6). As seen in Table 4, the addition of ppm level of all three types of silica particles improved the thermal stability of PP. The onset degradation temperature (T
onset
) was increased from 379 °C to upto 409 °C for modified mesoporous silica as well as commercial silica. The organic functional group grafted on the inorganic framework of silica in modified mesoporous silica led to an increase in T
onset
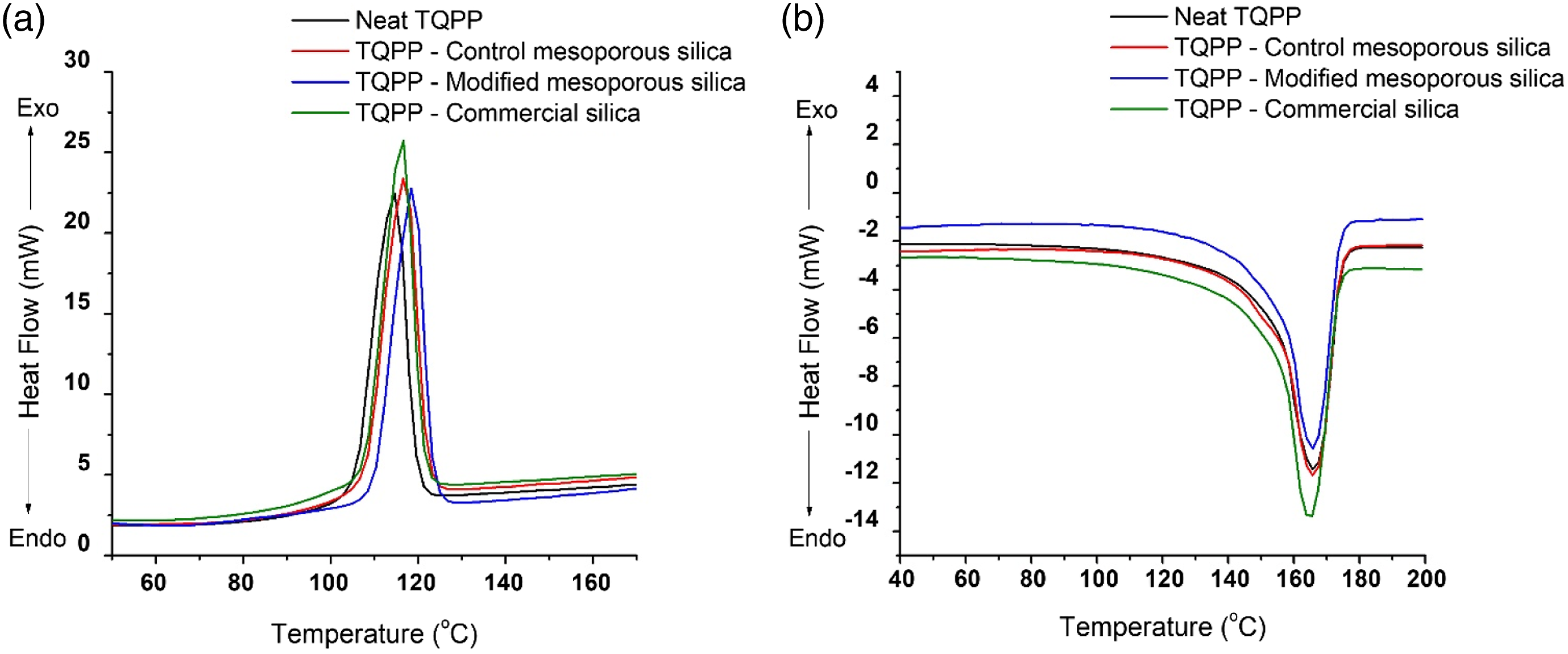
for modified mesoporous silica as compared to the control mesoporous silica. The crystallization behavior of TQPP films was examined via DSC analysis (as shown in Figure 7) and the corresponding parameters have been listed in Table 4. The melting point of all the films remained constant, while the peak crystallization temperature (T
c
) increased for all the TQPP-silica films as compared to the neat polymer. Maximum effect of heterogenous nucleation was observed for TQPP film with modified mesoporous silica. The improved dispersion of silica particles and increased interaction of organic group on silica surface with the PP chains increased the nucleating ability of silica particles in the matrix. As observed from FE-SEM and DLS results, particles in commercial silica sample are present in agglomerated form having size ranging in 5–13 microns. Increased particle size and lower surface area of commercial silica particles in comparison to mesoporous silica sample can be the possible reason for not enough rise in T
c
for commercial silica sample.
21
Similar conclusions were drawn by Borysiak et al., where improved dispersion, higher porosity and reduced particle size resulted in higher nucleating ability of silica particles in PP composites.
31
TGA curves of prepared neat TQPP and TQPP-silica films. Thermal parameters obtained from DSC - melting point (T
m
), peak crystallization temperature (T
c
), heat enthalpy of fusion (ΔH), as well as onset degradation temperature (T
onset
) determined by TGA. DSC thermograms depicting (a) cooling curves and (b) melting curves of neat TQPP and TQPP-silica films.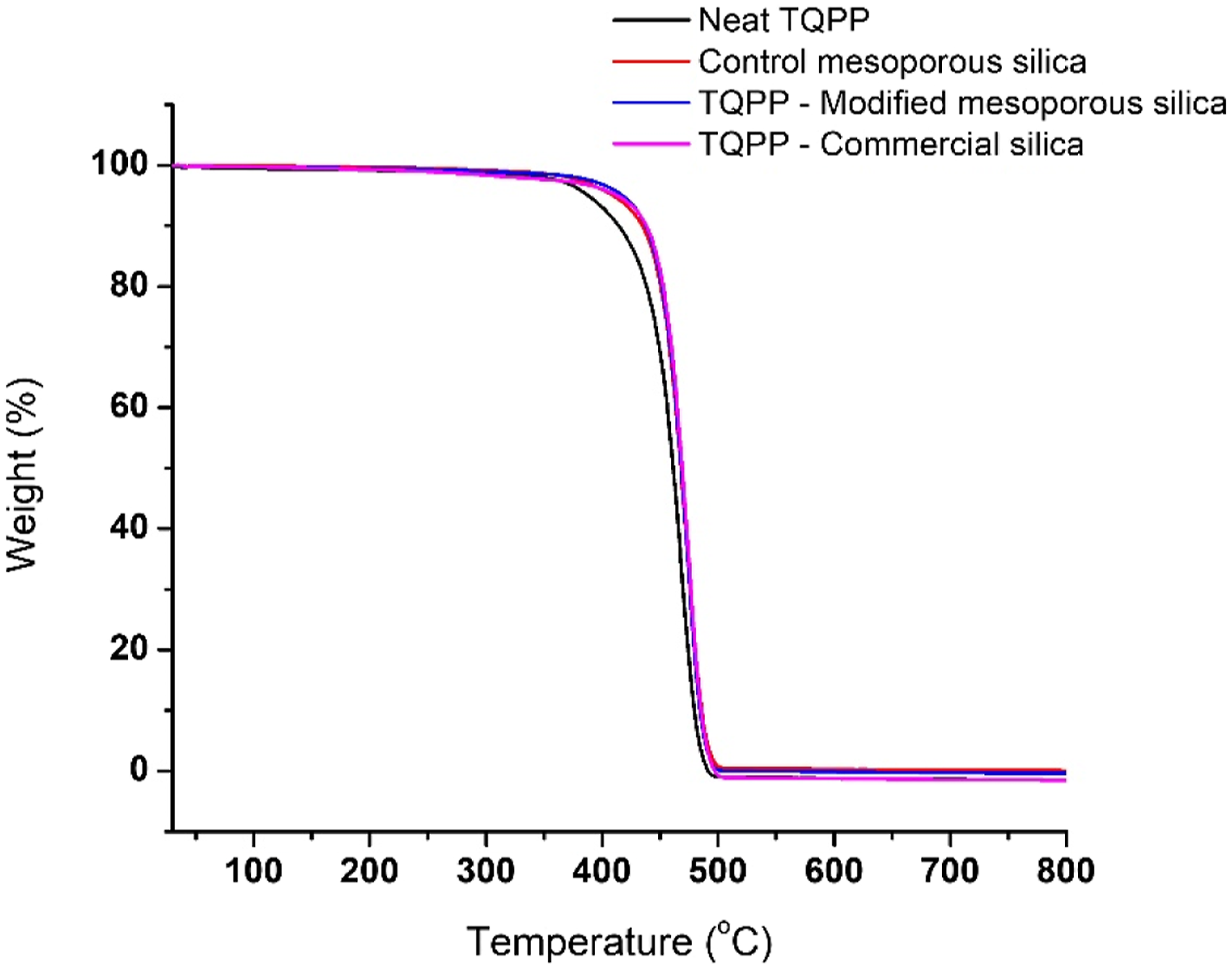

Blocking Force values and optical parameters including gloss and haze of neat TQPP and TQPP-silica films.
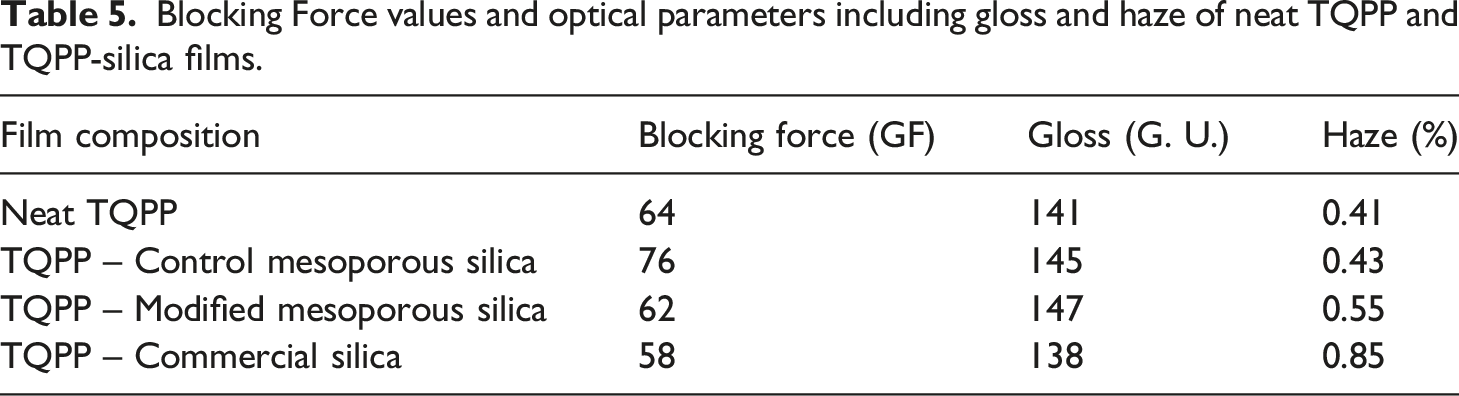
For packaging applications, the visual appeal of the films enclosing the articles, i. e. high clarity, low haze, and high gloss, is another crucial factor for both manufacturers and customers. The refractive index of synthetic silica (1.46) is quite similar to that of PP (1.49), which makes it a suitable additive for maintaining or improving the optical characteristics of polymer films. These optical properties are mainly a function of antiblocking aid, slip additive and internal haze arising in the crystalline structure. It has been observed that the gloss and clarity in films is compromised with higher loading of anti-blocking agents or slip additives. 33 Therefore, it is extremely important to obtain an appreciable balance of openability and optical properties of films. Haze is referred to the % of incident light which is scattered by more than 2.5 degrees by the surface of the film, while gloss is the ability of the film surface to reflect the incident light in any direction. Stehling et al. observed that the primary cause of haze in blown films was found to be the roughness of the film surface. 34 Bheda and co-workers also studied haze as a function of surface roughness or surface scattering, which correlates to the kind of anti-blocking additive used. 35 As seen from the values of haze and gloss in Table 5, commercial silica particles displayed poor optical properties in comparison to neat TQPP film. The large particle size of commercial silica particles (presence of agglomerates) lead to relatively more surface roughness and decreased the gloss of TQPP films and increased the internal haze development to more than double that observed for neat TQPP.19,36 The addition of control mesoporous silica showed no significant change in haze values as well as increased the gloss of the film. The purity of synthetic mesoporous silica particles reflected better optical properties as compared to commercial silica particles. Modified mesoporous silica particles increased the surface gloss due to improved dispersion in the matrix, while slightly increasing haze value as compared to neat TQPP because of increased anti-blocking property. This value (0.55%) was still much lower than that observed with commercial silica (0.85%).
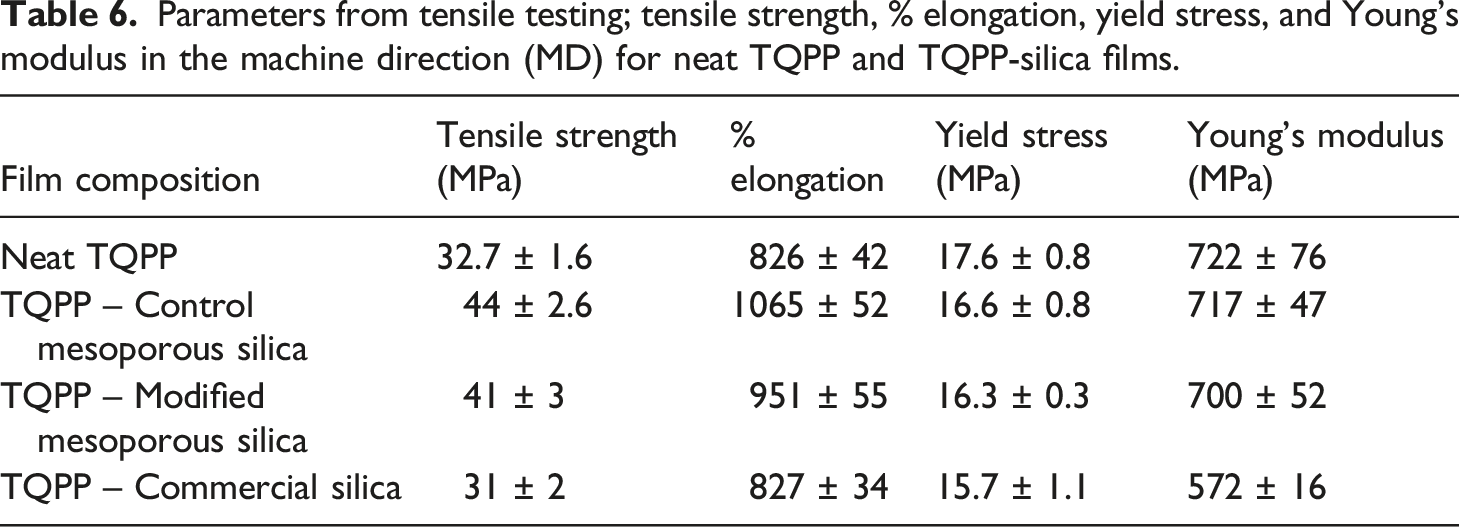
Another important parameter of packaging films is their resistance against stretchability and the extent of elongation. Literature reports have suggested that the well-ordered structure and extensively high surface area of mesoporous substrates improved dispersion and mechanical properties in PP as compared to the non-porous additives.37,38 The stress versus strain plot for neat TQPP and TQPP-silica films has been given in Figure 8 and the tensile parameters have been listed in Table 6. Even with the addition of such a small quantity of silica, the tensile strength for TQPP film increased up to 37.5% and 28% with CMS and MS-OH respectively, in comparison to neat TQPP. The extent of elongation is an important parameter to consider as it is a direct indicator of ductility, which is in turn an essential requirement in the case of packaging films.
39
As seen in the values listed in Table 6, the % elongation of the film was also enhanced up to 28% with CMS and 15% with MS-OH, indicating an increase in the ductile behavior of prepared TQPP-mesoporous silica films, which can also be clearly seen in Figure 8. There was absolutely no change in tensile properties with the addition of commercial silica particles in TQPP film. However, the Young’s modulus and yield stress of TQPP film decreased in the case of commercial silica as additive, while it nearly remained the same for neat TQPP and TQPP-mesoporous silica films. It is possible that the surface roughness due to agglomerates of particles in commercial silica samples is resulting in the crack generation and therefore counteracting the reinforcing properties of silica.
40
Stress versus strain curves of neat TQPP and TQPP-silica films. Parameters from tensile testing; tensile strength, % elongation, yield stress, and Young’s modulus in the machine direction (MD) for neat TQPP and TQPP-silica films.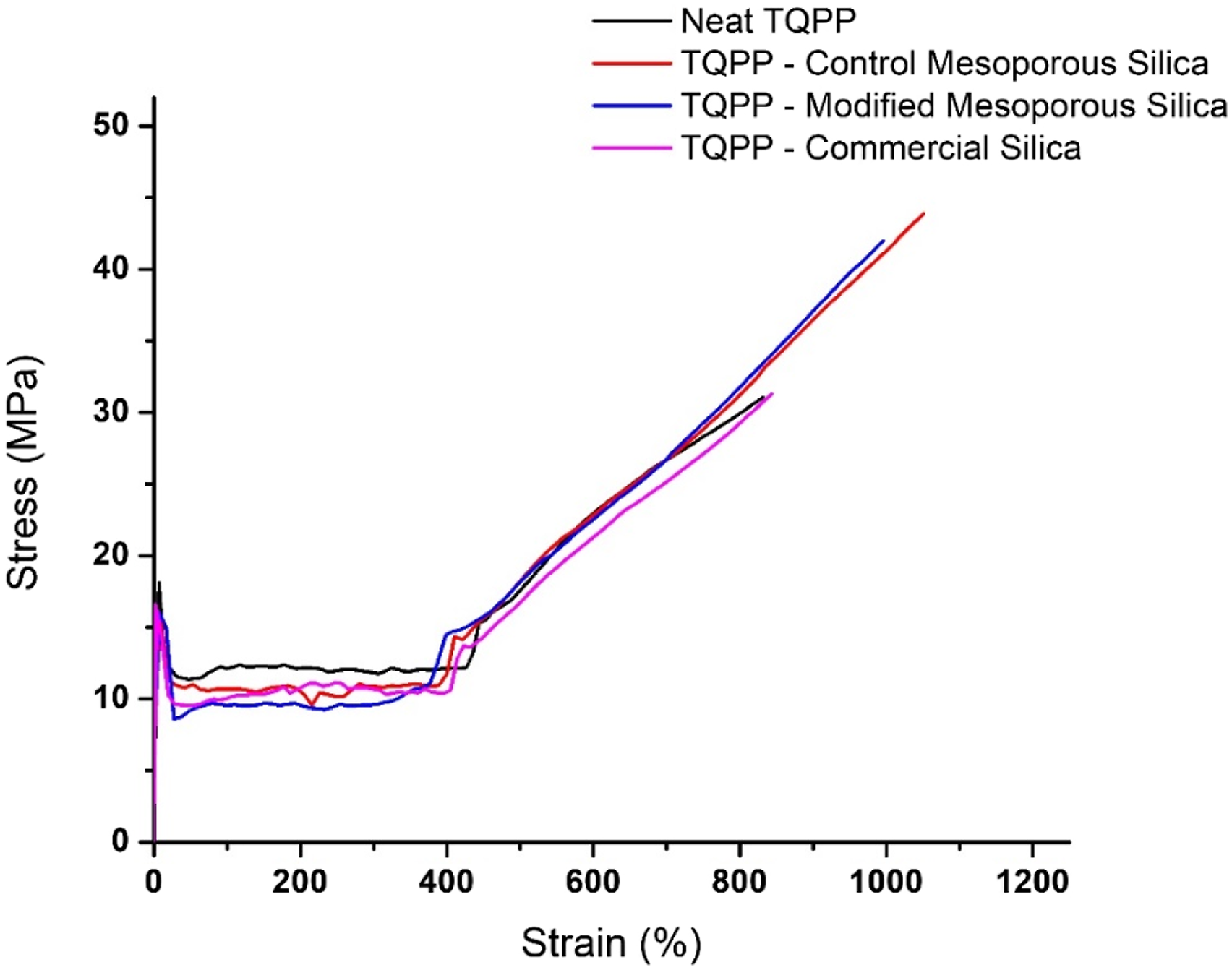
Contact angle, values of WVTR permeation, and OTR for neat TQPP and TQPP-silica films.
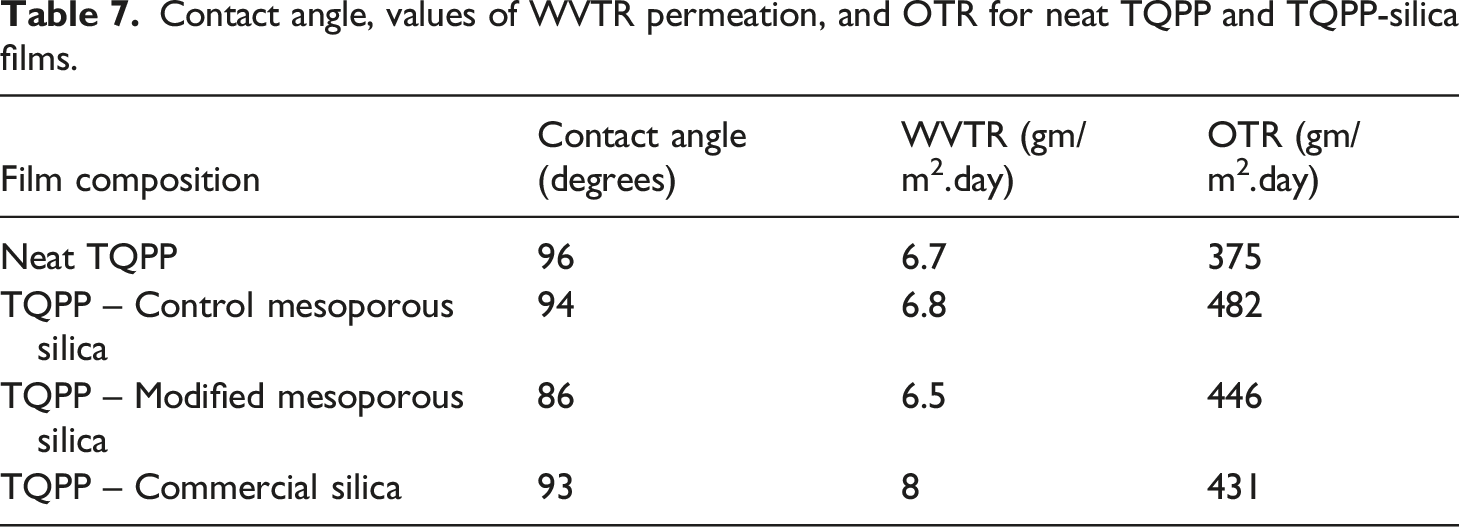
The evaluation of oxygen and moisture barrier properties are other important factors required for preparing packaging films for different purposes. For example, during the packaging of food items, it is necessary to maintain aroma, and flavor inside the packaged film and prevent the migration of undesirable elements from outside into the film. High barrier properties are required for enclosing coffee or roasted items, medicines, or nutrition-based products while low barrier properties are required for frozen fruits and vegetables.2,43,44 For the last many decades, plastics and polymer films have replaced glass and metal packaging due to the low cost and easy availability of the former. To evaluate the barrier properties of prepared TQPP films, water and oxygen transmission rate was determined and the corresponding values of WVTR and OTR have been reported in Table 7. PP has good water barrier properties due to its hydrophobic nature but is susceptible to the permeability of gases. 45 Due to the non-polar backbone of PP and the lower loading of silica, the values of water vapor permeability remained mostly constant for the prepared films. There was a slight increase in the WVTR value for TQPP film with commercial silica. This could be because of increased interfacial voids due to the agglomeration of silica nanoparticles. The results were more pronounced in oxygen transmission rate as compared to water vapor transmission. There have been reports published in the literature about an increase in oxygen permeability through PP on the addition of silica, possibly because of disruption in the polymer chain packaging.21,46–49 As seen from the values of OTR in Table 7, the value increased from 375 to 482 on the incorporation of control mesoporous silica. The possible reason could be the large surface area and easy permeation of gas through the mesopores. For TQPP-MS-OH film, the value of OTR decreased as compared to film with CMS, as the path length for the diffusion of oxygen slightly increased after surface modification of mesoporous silica. OTR values for TQPP-modified mesoporous silica sample were close to that of TQPP film with commercial silica. An increase in the OTR value for the latter in comparison to neat TQPP could be attributed to the voids created by agglomerates of nano silica, thereby increasing the free volume in the polymer matrix for easy gas permeation. The slightly lower OTR value in commercial silica film samples as compared to CMS could be because of the higher aspect ratio of silica particles that help in reducing gas diffusion as compared to silica particles of spherical morphology of aspect ratio nearly as unity. 50
Conclusion
Mesoporous silica nanoparticles (MSNs) were organically functionalized using silane that was in turn chemically modified by thiol-ene reaction. TQPP films were prepared with control mesoporous silica (CMS), modified mesoporous silica (MS-OH), and commercial silica as additives, and their effect on the anti-blocking force, mechanical, optical, and barrier properties were analyzed. Surface functionalization on mesoporous silica reduced the blocking force in TQPP films. Mesoporous silica particles show superior mechanical properties as compared to commercial silica particles in terms of higher tensile strength and % elongation in the machine direction. Higher clarity (lower haze) and improved gloss values were observed in TQPP-mesoporous silica films in comparison to that of commercial silica. Modified mesoporous silica decreased the water contact angle from neat TQPP film, indicating improved wettability. Control and modified MSNs showed slightly better WVTR values than commercial silica particles. The oxygen transmission rate was found to decrease with MS-OH as compared to CMS.
Footnotes
Acknowledgements
AJ acknowledges the Ministry of Education and Indian Oil Corporation Ltd, India for fellowship. LN acknowledges the Science and Engineering Research Board.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by. Department of Science and Technology, India (SB/S3/CE/061/2015) for financial support.
