Abstract
Silica derived from a variety of sources and its functionalized form has been studied as an antiblock additive in polypropylene (PP). Commonly, inorganic antiblock additives are added to PP films to reduce the blocking and facilitate the separation of polymeric films. However, such types of additives can cause a reduction of clarity in transparent films. In the present work, a comparative analysis of silica obtained from various sources specifically from rice husk ash and its further functionalization/modifications using n-octyltriethoxysilane has been performed. Since silica synthesized via rice husk ash was obtained from waste (rice husk ash), this further solves the problem of ash disposal. The functionalized forms of various silica have been characterized using Fourier transform infrared (FTIR) spectroscopy, differential scanning calorimetry (DSC), and thermogravimetric analysis (TGA). The morphological analysis was performed using scanning electron microscopy (SEM). The melt flow index (MFI), yellowness index, and other mechanical characterizations including tensile and impact strength were performed for 30–40 μm thick tubular quenched polypropylene (TQPP) films. These films were evaluated to have high transmittance (above 93%), high clarity (above 98%), and very low haze (less than 2%) indicating high transparency and improved optical properties. The blocking force and optical properties are quite similar for TQPP film containing silica synthesized from rice husk ash and commercially grade silica and hence proving silica synthesized from rice husk ash to be an effective substitute for commercial silica in TQPP films.
Keywords
Introduction
The phenomenon of blocking is considered as an adhesion between two layers of polymer films occurring due to the van der Waal forces of interaction. This force increases with the decreasing distance between the adjacent layers of films which in turn results in adhesion between films when pressed together. To avoid this kind of adhesion between films, commonly, antiblocking additives are utilized. The antiblocking agents microscopically protrude from the surface and create roughness which reduces the contact area between the surfaces as well as the coefficient of friction which minimizes blocking. Silica, calcium carbonate, magnesium silicate, aluminum silicate, and calcium phosphate are some of the common antiblocking agents 1 used with polypropylene. Silica can exist in crystalline, amorphous, and colloidal form and is a widely used antiblocking additive and is available abundantly on Earth. Silica particles microscopically protrude from the surface and create roughness which reduces the area of contact between the two surfaces and thus minimizing the adhesion between the two films. The micro rough surfaces reduce adhesion between the films, thus acting as an antiblock agent.
The other uses of silica are, as reinforcing filler in elastomers, as a component in adhesive, as an adsorbent, dehumidifying agent, anticaking agent in the food industry to control the rheological properties, and as a cleansing agent in toothpaste. Polypropylene (PP), a commodity plastic, has been widely used due to its low cost. 2 Excessive and wide applications of PP have called for various alterations required for an assortment of usage. Numerous modifications have been performed with PP, and the new properties achieved have been studied in detail and are well reported.3–5 To improve the properties and to enhance the performance of PP in packaging applications, various fillers 6 and additives are utilized with PP. Resch et al. 7 investigated the morphology of different types of PP cast films prepared using different formulations and processing conditions. To such films, various types and amounts of slip, antiblocking, and other acid scavengers were added, and their properties were studied. Particularly, the optical properties of films were investigated along with their correspondence to surface morphology. 8 A linear correlation was observed between the optical property and surface roughness of the films. There are diverse types of antiblocking aids that have been widely used, such as, natural silica, talc, organic zeolites, and synthetic silica. Synthetic silica has been widely used and reported for good optical properties as well as an antiblocking agent. Essche et al. 9 reported a highly efficient antiblocking agent using silica. Slip agents were further used to decrease the frictional coefficient making the surface smooth. However, sometimes slip agents can be adsorbed on the surface of antiblocking agent, for example, synthetic silica, and thus results in a higher coefficient of friction. The inorganic antiblocking agents provide excellent antiblocking property but find challenges in optical properties such as clarity. Also, not only clarity but there is other concern about the interactions with other additives which needs to be addressed. 10 The size of silica particles used as an additive in PP films also influences properties and performance of PP/silica composites. Hwang et al. 11 investigated the role of silica particles size in PP/silica composites with their foamed samples produced using microcellular injection molding process. An improvement in performance and processing benefits were reported. An increase in tensile strength and viscosity was observed when nanosilica was incorporated into PP. Also, the cell size of resulting foams decreases due to the incorporation of these micro/nanosilica particles. Wu et al. 12 investigated in detail mechanical properties of nanosilica/PP composites. The introduction of the pre-treatment method for the particles by surface modification of silica was performed, in this treatment, grafting of the polymer was performed on the precipitated nanosilica surface. This pre-treatment of silica reportedly showed improvement in strength and toughness of prepared composites. This pre-treatment was performed to facilitate dispersion of silica nanoparticles.
Rice husk which is discarded as an agricultural waste is rich in silica and can be an economically viable raw material for the extraction of silica.10–12 Among the various conventional technologies for energy conversion, the pyrolysis method has been widely adopted technology.13–17 Furthermore, rice husk can be also used as a renewable fuel in cogenerating plants, considering its high calorific value (4012 Kcal/kg). During its combustion, about 20–25 wt.% of rice husk ash (RHA), containing more than 90% silica, with traces of other metal oxides was produced.15–17 Tsai et al. 18 reported fast pyrolysis studies for rice husk in a fixed-bed heating furnace. The influence of heating rate, temperature, holding time, particle size, flow rate, sweep gas (N2) etc., were studied. In addition to silica derived from RHA, RHA itself has been used as a reinforcing filler. One of the prime properties of RHA is the low thermal conductivity and high thermal shock resistance, thus making it an appropriate material for the fabrication of composite19–21 and its applications in constructions industries.
The applications of RHA as a filler in PP have been reported by Faud et al. According to Ismail et al. burning RHA in an open-air leads to the formation of two types of fillers. The upper layer of RHA upon burning yields carbonized layer termed as black rice husk ash (BRHA), whereas the inner layer which burns at higher temperature is termed as white rice husk ash (WRHA). RHA silica and RHA has been reported as a filler for a variety of polymers, for example, natural rubber, polypropylene, etc.25–29
Although various uses for rice husk and RHA have been suggested before, but its disposal or utilization remains a major concern. RHA gives an economical pathway for silica extraction from agricultural waste rice husk, which also manages ash disposal.30–32 Duarte et al. 33 studied composite based on the RHA silica/PP which was processed by injection and extrusion molding. They assessed the effect of processing variables on the mechanical and thermal properties of these composites. Guswara and co-workers 34 have studied the enhancement in properties of PP composites with a detailed study on the effect of morphology, mechanical, and physical properties.
Chowdhury et al. 35 reported an improvement in mechanical and physical properties of PP composites based on RHA. They introduced RHA, low density polyethylene, and jute fibers to PP. Here, the effect of RHA as a filler was analyzed based on mechanical properties. Thus, reported RHA is a low-cost alternative to CaCO3 filler in PP composites. Sharma and co-workers 36 also demonstrated rice husk ash as an alternative filler to CaCO3 for PP composites. Bimodal porosity exhibited by RHA provided enhanced mechanical and thermal properties via capillary effect. Aprilia and co-workers37,38 reported use of RHA in PP leading to the composites with improved thermal stability.
In this work, the objective is to synthesize different types of silica as well as to prepare functionalized silica. The introduction of hydrophobic group in functionalized silica was performed, and assessment with non-functionalized silica was made in PP composites. As prepared non-functionalized and functionalized silica have been characterized and studied as an antiblock additive in polypropylene films which can find application in textile overwraps and garment bags. Three different sources of silica were studied, commercially available silica, silica synthesized via Stöber process, and silica from biomass (rice husk ash). All the different types of non-functionalized and functionalized silica were incorporated in PP, and mechanical, thermal, and optical properties were studied in detail.
Experimental
Materials
Commercial grade of silica Sylobac 45H from W.R.Grace was used. n-octyltriethoxysilane and tetraethylorthosilicate (TEOS) were procured from TCI India. Toluene, ammonium hydroxide, ethanol, and sodium hydroxide were procured from Fisher Scientific. Polypropylene (grade PP1110MG) in powder form was supplied by Indian Oil Corporation Limited (IOCL), Panipat refinery, India. A-1010 (sterically hindered phenolic primary antioxidant), A-168 (phosphite-based secondary antioxidant), calcium stearate, and erucamide (amide of C22 mono-unsaturated erucic acid) were of commercial grade. Rice husk silica was generated from rice husk obtained locally at a temperature of 800°C. The silica was further treated using in-house developed procedure to obtain 99% pure silica as determined using gravimetric procedure.
Fourier Transform Infrared Spectroscopy (FTIR): Uniform-sized (1 mm thick) pellets were prepared from the samples of non-functionalized and functionalized silica with KBr powder (1/10) using a hydraulic press. FTIR spectra were recorded on a ThermoNicolet IR 200 spectrometer in the spectral range of 400–4000 cm−1.
Thermogravimetric Analysis (TGA): Thermogravimetric analysis was performed on TA Instruments TGA Q 500. TGA was carried out at a heating rate of 10°C/min from room temperature to 850°C to study the thermal stability of different types of silica synthesized and to study the degradation profile of compounded polypropylene.
Differential Scanning Calorimeter (DSC): Differential scanning calorimetry was performed on TA Instruments DSC Q 2000 using heating and cooling rate of 10°C/min in temperature range of −20–200°C. The effect of silica on the crystallization temperature of polypropylene was analyzed using DSC.
Scanning Electron Microscopy (SEM): SEM analysis was carried out on Zeiss EVO-50 machine. SEM analysis for all the types of non-functionalized and functionalized silica was carried out. It was followed by particle size determination of silica particles using ImageJ software.
Tensile and Impact Testing: Tensile specimens of compounded PP were prepared in Haake Minijet II microinjection molding machine. The cylinder temperature was maintained at 200°C and mold temperature was kept at 90°C. The air pressure was kept at 750 bar. Tensile testing was performed on Zwick Z010 Universal Testing Machine (UTM) at a strain rate of 50 mm/min. Samples for tensile testing were prepared according to ASTMD638 type V. Five microinjection molded samples were tested for each composition. Tensile testing of TQPP films was carried out at a strain rate of 200 mm/min. Impact strength was determined using Tinius Olsen impact testing machine.
Film thickness: Thickness of the films was measured at 20 points, and then the average thickness of the film was calculated.
Melt Flow Index (MFI): MFI of compounded PP was determined using Gottfert MFI tester. MFI was calculated at 230°C using load of 2.16 kg.
Yellowness Index: Yellowness index of compounded PP was determined using Hunterlab yellowness index testing machine using ASTM E313.
Transmittance: It is the ratio of transmitted light to incident light. It was determined using ASTM D1003. Transmittance values of all the types of TQPP films prepared were determined.
Haze: It is the % of incident light scattered by more than 2.5° by the film. It was determined using ASTM D1003. Haze value of all the types of TQPP films prepared was determined.
Clarity: It is the light flux deviating less than 2.5° from the incident light. % clarity of all the types of TQPP films prepared was determined.
Oxygen Transmission Rate (OTR): OTR of all the TQPP films was determined using ASTM D3985 at 23°C at 0% relative humidity.
Water Vapor Transmission Rate (WVTR): WVTR of all the TQPP films was determined using ASTM F1249 at 38°C at 90% relative humidity.
Blocking Force: Blocking is the phenomenon of adhesion between adjacent layers of film. Blocking force was determined using ASTM D3354.
Commercial silica and its functionalization
Sylobloc 45H from Grace was used as the commercial silica. 10 g of commercial silica, 10 mL of n-octyltriethoxysilane, and 100 mL of toluene were taken in a 250 mL round-bottomed flask, and the mixture was refluxed at 120°C for 48 h. The precipitate was washed with toluene, ethanol, and water. The precipitated silica was dried in an oven for 24 h followed by characterization of functionalized silica obtained.
Silica synthesized by Stöber method and its functionalization
For the synthesis of silica, ammonium hydroxide was taken in a 2 L reactor flask. A mixture of ethanol and tetraethyl orthosilicate (268 mmol) was added to the reactor. This mixture was stirred for 24 h at 35°C. The precipitate was filtered and washed with ethanol and water. The precipitated silica was then dried in an oven for 24 h followed by characterization of silica obtained. For the functionalization of the prepared silica, silica along with n-octyltriethoxysilane and toluene were taken in a 250 mL round-bottomed flask and refluxed at a temperature of 120°C for 48 h. The precipitate was washed with toluene, ethanol, and water. The functionalized silica was then dried in an oven for 24 h. It was followed by characterization of functionalized silica obtained and its comparison with non-functionalized silica as well as with other types of silica synthesized.
Silica from biomass and its functionalization
Rice husk ash contains 90% silica as determined by the treatment of ash with hydrofluoric acid. Rice husk ash was initially ground to form powdered rice husk ash. A required solution of sodium hydroxide was prepared. A required amount of powdered rice husk ash and sodium hydroxide solution was taken in a round bottom flask. This mixture was stirred under reflux conditions for 2 h at a temperature of 140°C. The hot mixture was filtered using a G3 flask with the help of a vacuum pump. The clear filtrate of sodium silicate solution was obtained. Sodium silicate solution was allowed to cool to room temperature. The solution was then neutralized with a sulfuric acid solution (1:4 of sulfuric acid to water) till a pH of 6 was obtained and silica gets precipitated. Drop-wise addition of sulfuric acid was done with continuous stirring, and demineralized water was also added to dilute the highly viscous solution. During neutralization by sulfuric acid solution, pH was checked and maintained at regular intervals. Hot water washing at a temperature of 90°C with continuous stirring followed by decantation was carried out with demineralized water multiple times for sodium sulfate removal. The remaining product was dried at a temperature of 140°C to obtain white amorphous silica powder. Silica powder thus obtained was then analyzed using various characterization techniques like FTIR, TGA, SEM, ICP-MS etc. The calcination of silica powder was also performed in the furnace at a temperature of 600°C, and further weight loss was recorded. Rice husk ash silica was functionalized using a similar procedure used for the functionalization of commercial silica and Stöber silica. Briefly, RHA silica along with n-octyltriethoxysilane and toluene were taken in a 500 mL round-bottomed flask, and the mixture was refluxed at 120°C for 48 h. The solution formed was washed with toluene, ethanol, and water, and the functionalized RHA silica was then dried in an oven.
Processing
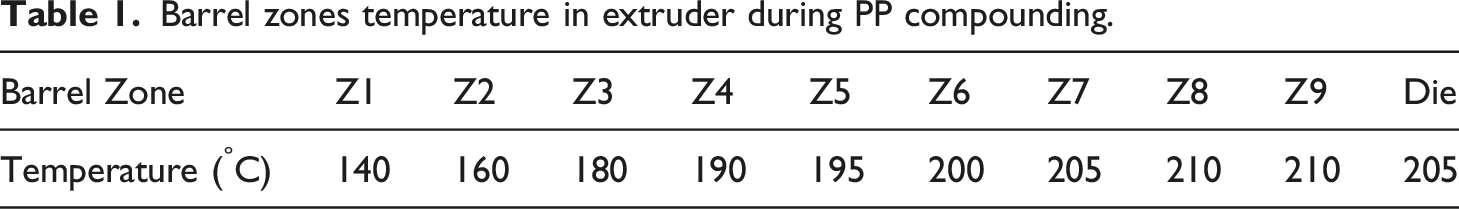
Barrel zones temperature in extruder during PP compounding.
Additives used in compounding with its function and dosage.

Designation for different samples of PP compounded with silica.

Tubular quenched polypropylene film formation
Thickness of TQPP films.

Results and discussion
As discussed above, a variety of silica were synthesized and functionalized with n-octyltriethoxysilane. All the functionalized and non-functionalized silica were characterized using a variety of techniques. The FTIR spectra for all functionalized and non-functionalized silica are shown in Figure 1. For commercial silica (CS) and functionalized commercial silica (FCS), Figure 1(a), the bands at 1086 cm−1 and 803 cm−1 correspond to Si-O-Si asymmetric stretching and bending vibrations, respectively. The bands at 3452 cm−1 and 1635 cm−1 correspond to OH stretching and bending vibrations, respectively. The bands at 2927 cm−1 and 2859 cm−1 correspond to C-H asymmetric and symmetric stretching, respectively. These bands confirm the functionalization of commercial silica (FCS). FTIR of various functionalized and non-functionalized silica.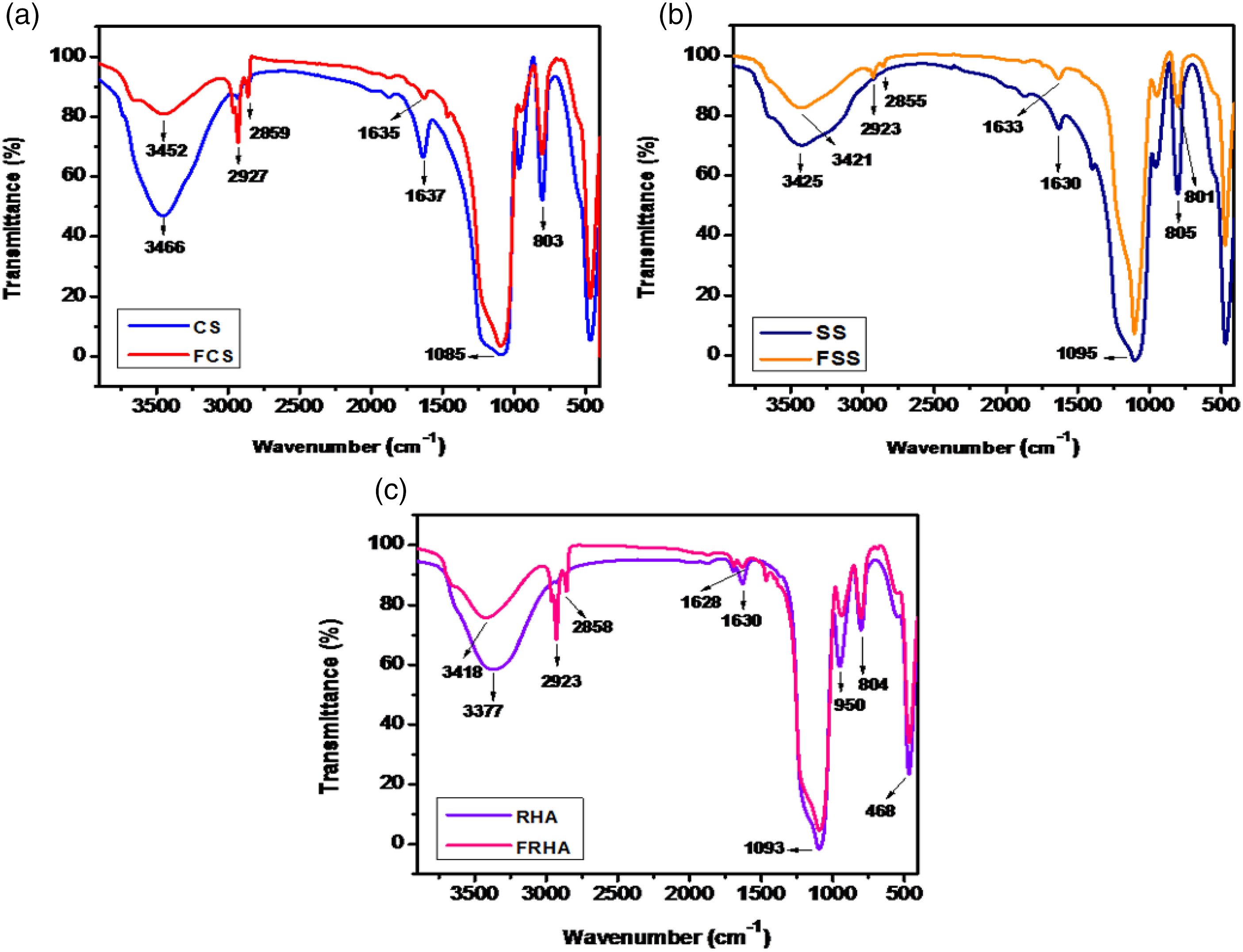
Figure 1(b) corresponds to bands for Stöber silica (SS) and functionalized Stöber silica (FSS), and the bands at 1100 cm−1 and 805 cm−1 correspond to Si-O-Si stretching and bending vibrations, respectively. The bands at 2923 cm−1 and 2855 cm−1 correspond to C-H asymmetric and symmetric stretching, respectively. Similarly, Figure 1(c) shows bands corresponding to RHA silica (RHA) and functionalized RHA silica (FRHA). For RHA silica, the bands at 1093 cm−1, 950 cm−1, and 804 cm−1 correspond to Si-O-Si asymmetric stretching, symmetric stretching, and bending vibrations, respectively. These bands confirmed the formation of silica. The bands at 3377 cm−1and 1628 cm−1 correspond to OH stretching and bending vibrations, respectively. The bands at 2923 cm−1 and 2858 cm−1 correspond to C-H asymmetric and symmetric stretching, respectively. These bands confirmed the functionalization of RHA silica.
Thermogravimetric analysis was performed to determine the weight loss in different types of functionalized silica. Figure 2 represents % of weight loss for various silica samples. TGA plot shows that non-functionalized silica (CS, SS, and RHA) has excellent thermal stability and there was no significant weight loss up to a temperature of 850°C. Whereas for functionalized commercial silica (FCS), there was a 9% weight loss in the temperature range of 400–600°C which was due to the decomposition of organic component in the functionalized silica. A significant weight loss was observed for functionalized Stöber silica (FSS). The 6% weight loss at 100°C was attributed to water loss while 5% weight loss in the range of 400–600°C was attributed to the loss of organic component. In RHA silica, 10% water loss was observed initially at a temperature of 100°C. After that, no significant weight loss can be observed which shows that the silica synthesized has excellent thermal stability. Whereas in functionalized RHA silica, 12% weight loss in the temperature range of 400–600°C was observed due to the decomposition of organic component in the functionalized RHA silica. TGA plots for a variety of functionalized and non-functionalized silica.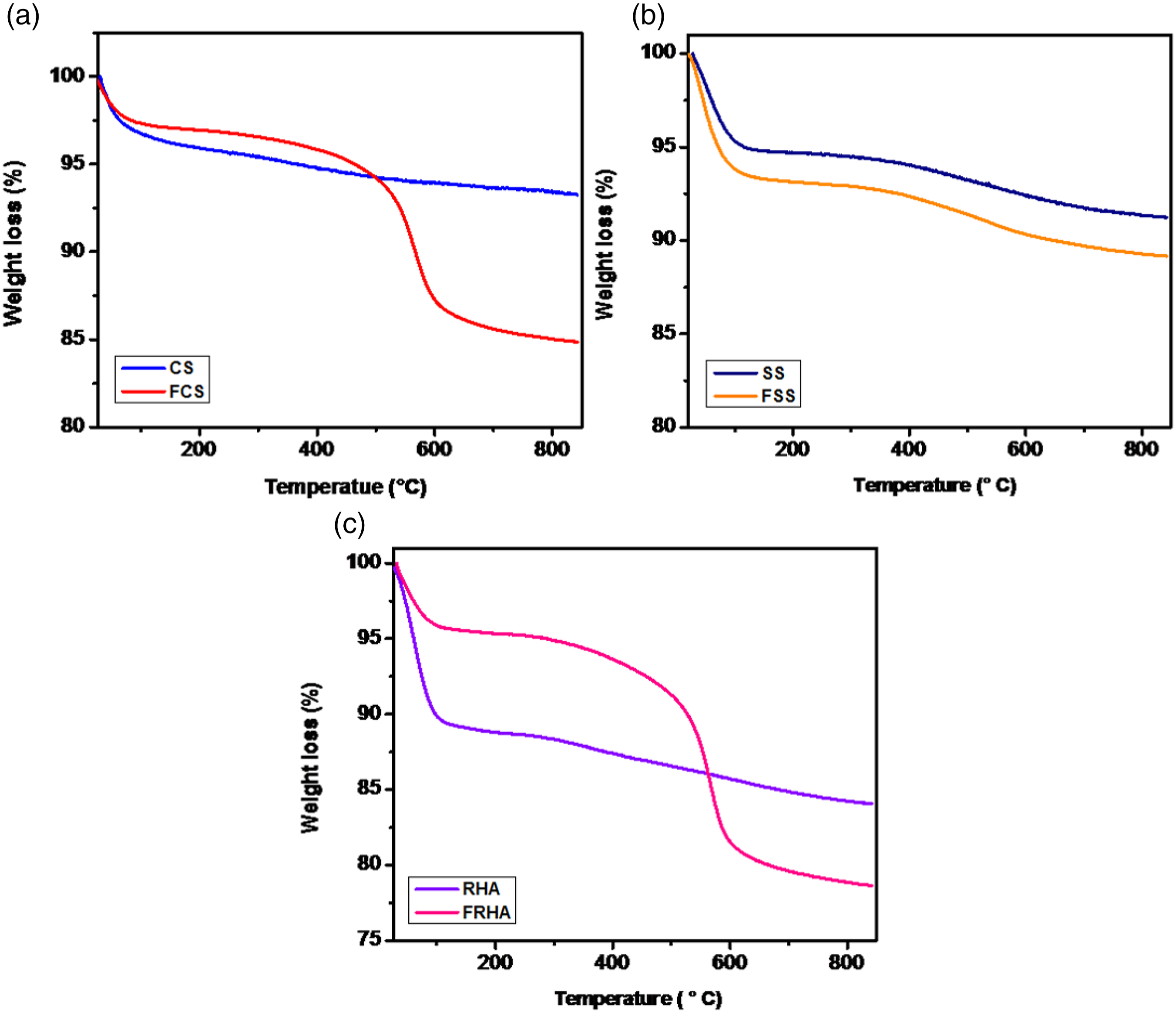
The morphology of all functionalized and non-functionalized silica samples was studied using scanning electron microscopy. Figure 3(a1, a2) depicts non-functionalized commercial silica (CS) having silica particles of non-uniform shape and size. Figure 3(b1, b2) shows functionalized commercial silica (FCS) which also shows non-uniform shape and size. Scanning electron micrographs of (a1, a2) commercial silica (CS), (b1, b2) functionalized commercial silica (FCS), (c1, c2) Stöber Silica (SS), (d1, d2) functionalized Stöber Silica (FSS), (e1, e2) RHA silica (RHA), and (f1, f2) functionalized RHA silica (FRHA).
SEM analysis for Stöber silica in Figure 3(c1, c2) shows well-formed spherical particles of silica. The particles seem to be of almost uniform shape and size. Each individual particle is clearly visible, and the boundary of particles is uniform and free from any defects. On analyzing particle size of Stöber silica, it was found that the particles were in the size range of 350–500 nm with the majority of them having size of 400–450 nm. Upon functionalization of Stöber Silica (FSS) as shown in Figure 3(d1, d2), particles obtained are quite uniform and regular in shape with most of the particle size in the range of 250–300 nm. Silica particles synthesized from rice husk ash (RHA) shown in Figure 3(e1, e2) is in agglomerate form and it tend to form large clusters. Distinct particles cannot be identified even at high magnification which implies that size of the particles is extremely small. Upon functionalization of RHA silica (FRHA) depicted in Figure 3(f1, f2), large agglomerates are seen and confirm the extremely small size of these particles.
Compounding and processing of polypropylene with a variety of silica
MFI and yellowness index of PP compounded with a variety of functionalized and non-functionalized silica.

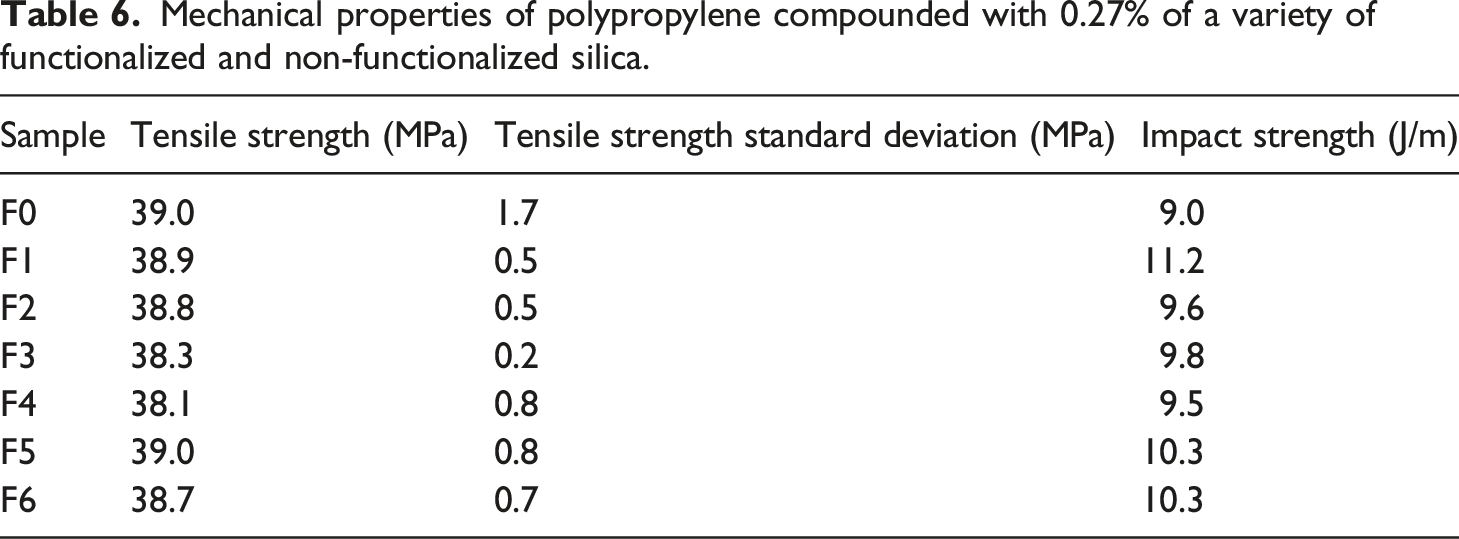
Mechanical properties of polypropylene compounded with 0.27% of a variety of functionalized and non-functionalized silica.
Thermal analysis of PP compounded with a variety of silica was performed using DSC and TGA. Figure 4 shows DSC plots, and Table 7 depicts the melting temperature, crystallization temperature, and % crystallinity of the samples analyzed. DSC plot for PP compounded with a variety of functionalized and non-functionalized silica. DSC analysis of PP compounded with a variety of functionalized and non-functionalized silica.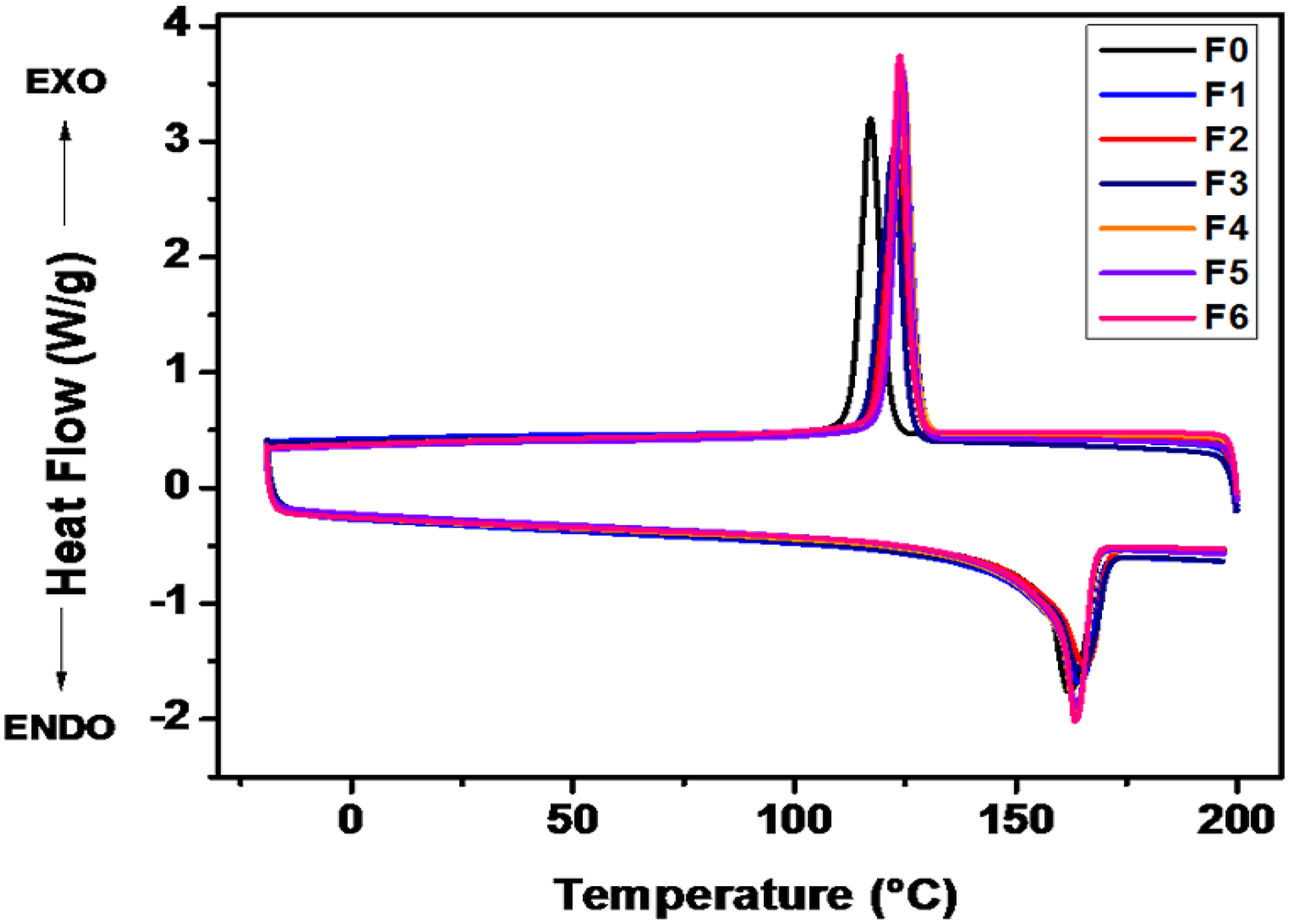
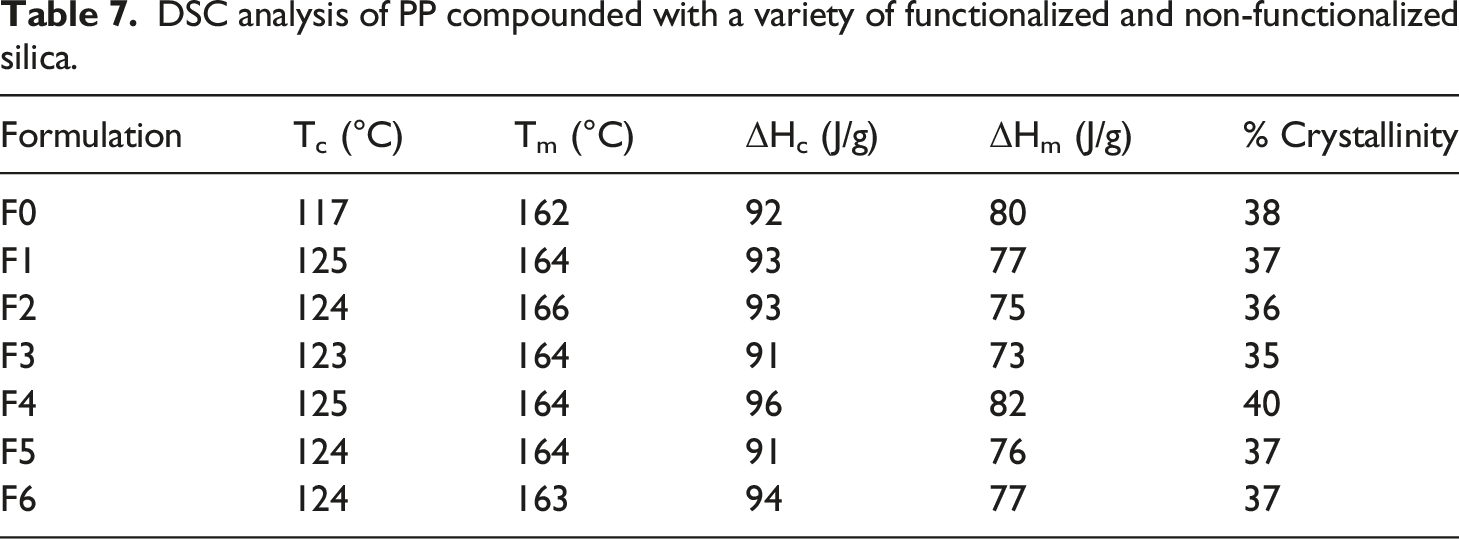
From the DSC analysis, it is evident that all the formulations show roughly the same melting point. However, formulations F1 to F6 have higher crystallization temperatures than formulation F0. Also, the cooling cycle curves convey that silica present in formulations F1 to F6 is leading to faster crystal formulation.
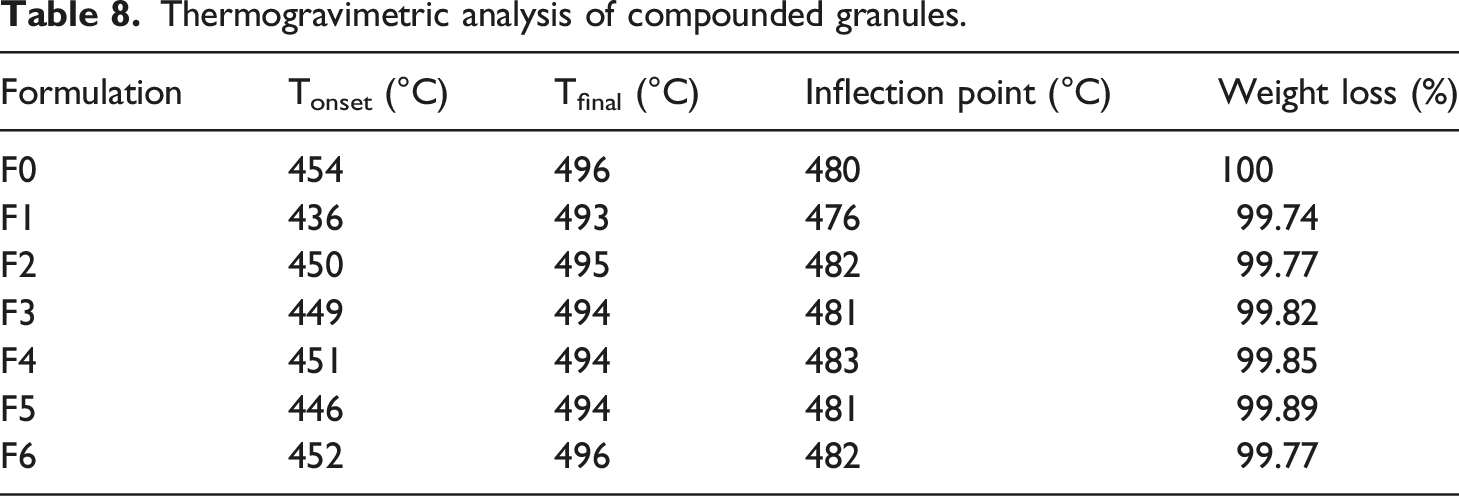
Figure 5 shows TGA analysis of the PP compounded with a variety of functionalized and non-functionalized silica. TGA plots show that the weight residue is there in samples F1 to F6 which is due to the presence of silica. The degradation temperature and % weight loss has been dicussed in Table 8. TGA plot of PP compounded with a variety of functionalized and non-functionalized silica. Thermogravimetric analysis of compounded granules.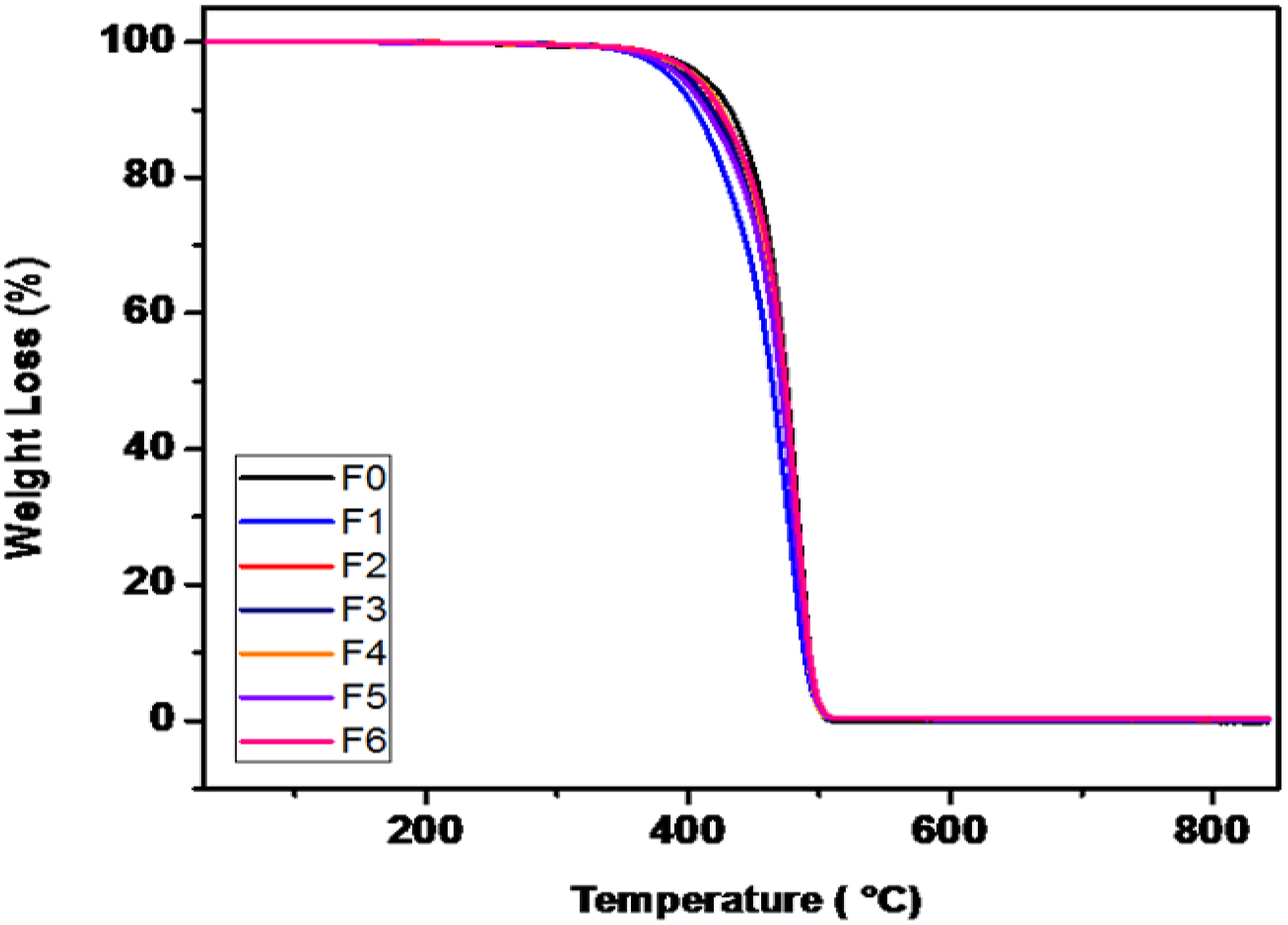
Tensile strength of TQPP films in transverse and longitudinal direction.
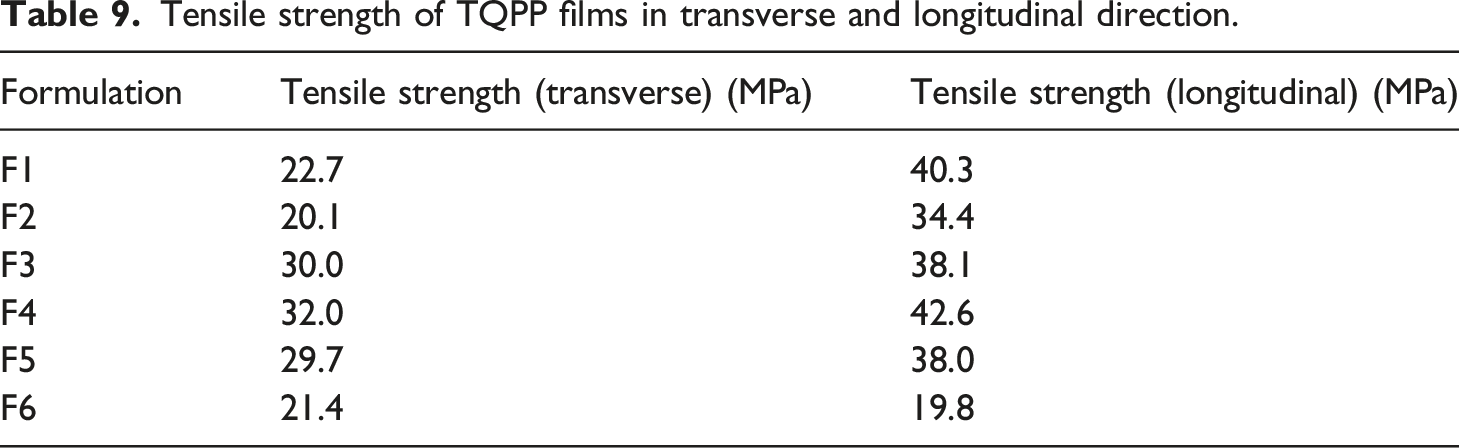
On analyzing the tensile results, formulations containing commercial silica, Stöber silica, and silica derived from rice husk ash show similar tensile strength values. Due to the orientation of the polymer chains in the longitudinal direction, it was observed that the tensile strength of all the films is higher in the longitudinal direction than transverse direction (Table 9).
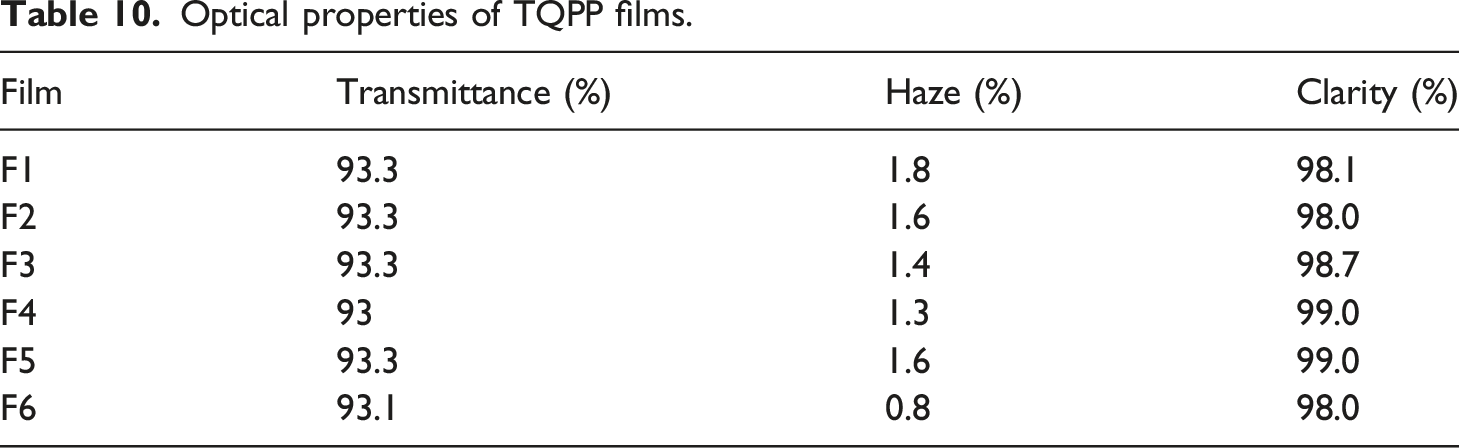
Optical properties of TQPP films.
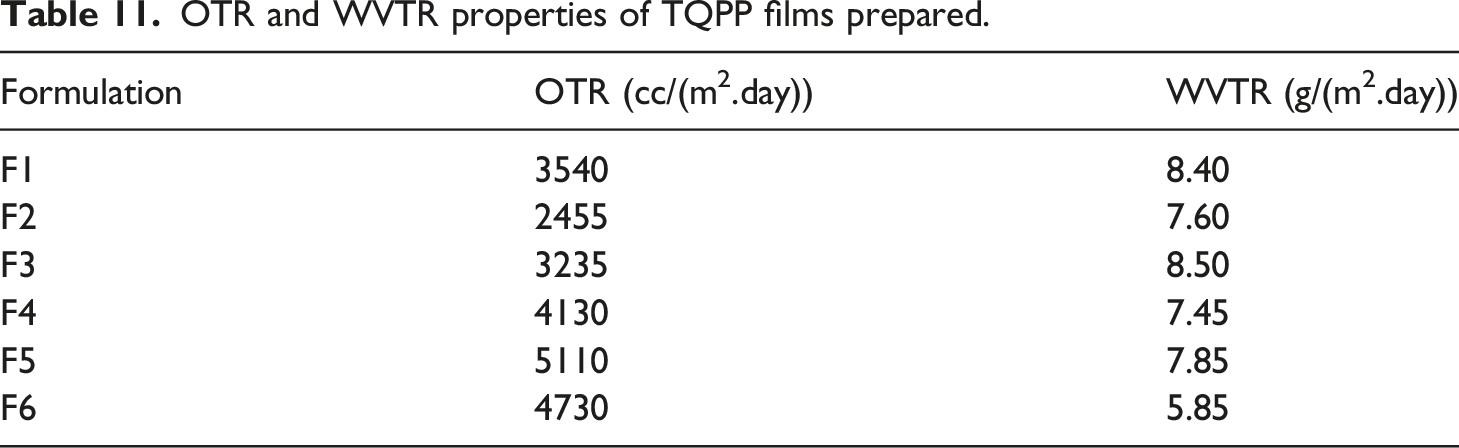
OTR and WVTR properties of TQPP films prepared.
Blocking force values of TQPP films.

The higher blocking force of F5 film over F1 can be attributed to the fact that silica particles synthesized from rice husk ash are in agglomerate form while commercial silica particles are stabilized by stabilizers and thus devoid of any agglomerations possible. Also, the shape of commercial silica particles also plays an important role in lowering of blocking force due to its irregular shape. Furthermore, it was found that films containing functionalized silica have higher blocking force as the hydrophobic part of the functionalized silica can interact with polypropylene chains.
Conclusion
In this study, silica from various sources has been functionalized with n-octyltriethoxysilane. Since silica synthesized from rice husk ash is obtained from waste (rice husk ash), it solves the problem of ash disposal and by-products obtained during synthesis are recoverable. It can be used as an excellent substitute for commercial silica. FTIR curves confirm the synthesis and functionalization of all the types of silica. TGA curves confirm that different types of silica synthesized have excellent thermal stability. SEM analysis depicts that silica particles of commercial silica and its functionalized version are irregular in shape and are not agglomerated. Stöber silica and its functionalized version particles are spherical in shape with distinct particles clearly visible while silica particles synthesized from rice husk ash and its functionalized version are in agglomerated form. Mechanical properties like tensile strength and impact strength show that since the concentration of silica is very less, it is not behaving as a reinforcing filler. MFI values were found suitable for the preparation of TQPP films. Yellowness index values are low which indicates that the film which will be formed will be highly transparent. TGA of compounded granules estimates that the degradation temperature of all samples is similar and % weight which is remaining is because of silica. DSC analysis clearly indicates that silica is affecting the crystallization temperature. It is clear from the tensile strength testing of TQPP films thattensile strength was found be higher in the longitudinal direction than transverse direction. Assignable to the 30–40 μm thickness of the TQPP films, they were evaluated to have high transmittance (above 93%), high clarity (above 98%), and very low haze (below 2%) which indicate that the films are highly transparent and thus with improved optical properties. On comparing, blocking force value of film containing silica synthesized from rice husk ash is analogous with blocking force value of film containing antiblock grade commercial silica. The films formed using functionalized silica have lower WVTR values than films made using non-functionalized silica which is due to the presence of hydrophobic part in the functionalized silica. Moreover, blocking force and optical properties are quite similar so silica obtained from rice husk ash can be effectively used as antiblock grade silica in TQPP films which can further find applications in food packaging, textile overwraps, and garment bags.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
