Abstract
The structural, optical, morphological and thermal properties of poly (methyl methacrylate) (PMMA)/boehmite nanocomposite films were studied by Fourier transform infrared spectroscopy (FT-IR), UV-Vis spectroscopy, X-ray diffraction (XRD), scanning electron microscopy (SEM), thermogravimetric analysis (TGA) and differential scanning calorimetry (DSC) studies. The FT-IR spectra of composites showed the characteristic band of boehmite nanoparticles at 518 cm−1 indicating an effective interaction between PMMA and boehmite. The UV-visible measurements showed that the optical energy gaps for the direct permitted transitions decreased as boehmite content increased. The orientation of nanoparticles observed from XRD revealed that the addition of boehmite increases the crystallinity of PMMA. The SEM analysis showed the uniform distribution of boehmite in the PMMA matrix. The shift in glass transition and melting temperatures to higher temperature domains were revealed by DSC analysis. From the TGA studies, the reinforcement of PMMA with boehmite nanoparticles increases thermal stability. The electrical conductivity was measured using the impedance technique, and it was studied as a function of nanoparticles loading, applied frequency ranging from 100 to 106 Hz and at different temperatures. The AC conductivity of the composite increases with frequency and temperature. The activation energy was also measured for all samples at various applied frequencies, and it was observed that it decreased with increasing nanoparticle loading. The dielectric constant of PMMA increases with nanoparticle concentration up to 7 wt%. The modulus, tensile strength, hardness, and impact properties of the PMMA/boehmite nanocomposite films were significantly enhanced by the reinforcement of nanoparticles into the polymer matrix. The excellent optical properties, mechanical strength and electrical properties obtained for 7 wt% loaded nanocomposite films enable them to be used in the construction of electromagnetic induction shielding materials, conducting adhesives, and flame-resistant flexible optoelectronic devices.
Keywords
Introduction
Combining two or more polymers has become an effective strategy to produce advanced polymeric materials with improved physical and chemical properties compared to the original polymers.1,2 The final product properties depend on factors such as the method of mixing, the mixed polymer proportions, the temperature at which mixing is done, and the degree of mixing of individual polymeric components.3–5 The interfacial interactions between polymer functional groups in the mixture govern the final physical and chemical properties of the material. In addition, the incorporation of nanoparticles into the polymer mixture enhances their properties due to the interaction between them and the functional groups of the mixture. This significantly enhances the electrical, dielectric, optical, and thermal properties of the resulting polymer nanocomposite.6–10
In electrochemical energy-storing devices, they contain electrochemically active material-coated current collectors (electrodes that are usually fabricated from metal oxides and carbon-based nanomaterials) and an electrolytic media. The polarization characteristics of virgin materials are usually enhanced with the incorporation of nanomaterials into electrolytes and electrodes, resulting in enhanced electrochemical characteristics of energy-storing devices.11–13
Acrylic-based polymers are one of the major thermoplastic polymers due to their excellent thermal, mechanical and optical properties.14,15 Among acrylic polymers, poly (methyl methacrylate) (PMMA) and poly (butyl methacrylate) are extensively investigated due to their high surface clearance, good transparency and mechanical properties. PMMA, also known as acrylic glass, contains a methyl ester group, which makes it bond with other polar polymers or polar fillers.16,17 Because of its insulating and brittle nature, PMMA cannot be used in applications that need conductivity or ductility. Several experiments have been performed in order to improve the electrical and mechanical properties of PMMA.18–20
Several inorganic nanoparticles have been widely studied to improve the properties of polymers. Among these nanoparticles, boehmite nanoparticles occupy a unique position due to their water solubility, high surface area, multiple hydroxyl groups that can be modified, and good dispersion capacity in various polymer matrixes. 21 Boehmite is a typical aluminium oxyhydroxide (AlO2H) with several peripheral hydroxyl groups that allow for strong interactions with polar polymers.22,23 The addition of boehmite nanoparticles to epoxy enhances the fracture toughness, shear modulus, shear stress and modulus at room temperature. 24 According to Jux and co-workers, the improvement in such properties depends on the concentration of nanoparticles. For instance, the tensile modulus and fracture toughness were increased by 26% and 62% respectively with the addition of 15 wt% boehmite to the epoxy matrix. 25
The aim of the present work is to investigate the effect of boehmite nanoparticles on optical, thermal, mechanical, electrical and dielectric properties of PMMA systems. The optical properties were investigated by UV-Vis spectroscopic studies. The structural and morphological properties were obtained by FT-IR and scanning electron microscopy (SEM) images. The thermal properties were accessed by TGA and DSC analysis. The mechanical properties such as impact strength, tensile properties and hardness. Ultimately, the temperature-frequency dependent electrical properties were investigated as a function of boehmite loadings. The obtained results indicated that developing optoelectronic devices with sufficient charge storing capacity in the near future would be a powerful application.
Experimental
Materials
The PMMA, with a molecular weight of 125000 g/mol was obtained from Hi-media. The boehmite nanoparticles (< 20 nm), chloroform and cetyltrimethylammonium bromide (CTAB) were purchased from Merck India. All chemicals were used directly without further purification.
Preparation of PMMA/boehmite nanocomposite films
In the present work, PMMA/boehmite nanocomposite films were prepared by a simple solution mixing process. In this method, first we prepare a homogeneous solution of PMMA by mixing it with the appropriate amount of chloroform. The clear solution was then poured into a petri dish and allowed to dry at room temperature for 48 h. The pure PMMA was peeled from the petri dish for further analysis. PMMA nanocomposite films was prepared by mixing the homogeneous solution of PMMA with different contents of boehmite nanoparticles. First, the nanoparticles were dispersed in a suitable amount of chloroform, followed by the addition of CTAB to avoid the agglomeration of the nanoparticles. Various percentages of boehmite nanoparticles (3, 5, 7 and 10 wt %) were dispersed into the homogeneous solution of PMMA with vigorous stirring for 30 min. The solution was then transferred into a petri dish and dried at room temperature for 2 days. The films were peeled from the petri dishes and kept in vacuum desiccators.
Characterization
The UV-Vis spectra of polymer nanocomposite films dissolved in chloroform were measured in the range of 200–800 cm−1 using a JASCO UV-Visible spectrophotometer. Fourier transform infrared spectroscopy (FT-IR) spectra measurements of polymer nanocomposite films were studied using a model JASCO-4100 FT-IR spectrometer in the range of 4000–400 cm−1 region and KBr pellets were used for casting the sample solution. The X-ray diffraction (XRD) pattern from 5° to 90° was recorded with a Bruker D8, Advanced Diffractometer by exposing the samples under analysis to Cu-Kα radiation at 40 kV. The morphology of the prepared nanocomposite films was analysed using a JSM-7610F plus Schottky field emission scanning electron microscope. Differential scanning calorimetry (DSC) measurements were achieved by Shimadzu DSC-50 (10 C/min) between room temperature and 275°C under the nitrogen atmosphere. The hybrid materials produced were analysed using a TGA-50 Shimadzu thermogravimetric analyser at a heating rate of 10°C/min. LCR meter (Model-IM 3570) was used to obtain the dielectric constant and AC conductivity of PMMA and PMMA/boehmite nanocomposite films.
Results and discussions
UV-Visible spectroscopy
Figure 1 shows the UV-Vis spectrum of PMMA and its nanocomposite films with different contents of boehmite nanoparticles. Pure PMMA has a noticeable peak at 245 nm, which corresponds to the n - π٭ electronic transition. The incorporation of nanoparticles shifts the absorption peak to a higher wavelength due to the enhanced surface area of the nanoparticles. For instance, the absorption peak shifts to 246, 247, 270, 250 nm for 3, 5, 7 and 10 wt % boehmite loaded nanocomposite films. Furthermore, it can be noted that all samples do not show any characteristic absorption bands in the visible region, indicating the transparent nature of the film.
26
UV-Visible spectrum of PMMA and PMMA/boehmite nanocomposite films.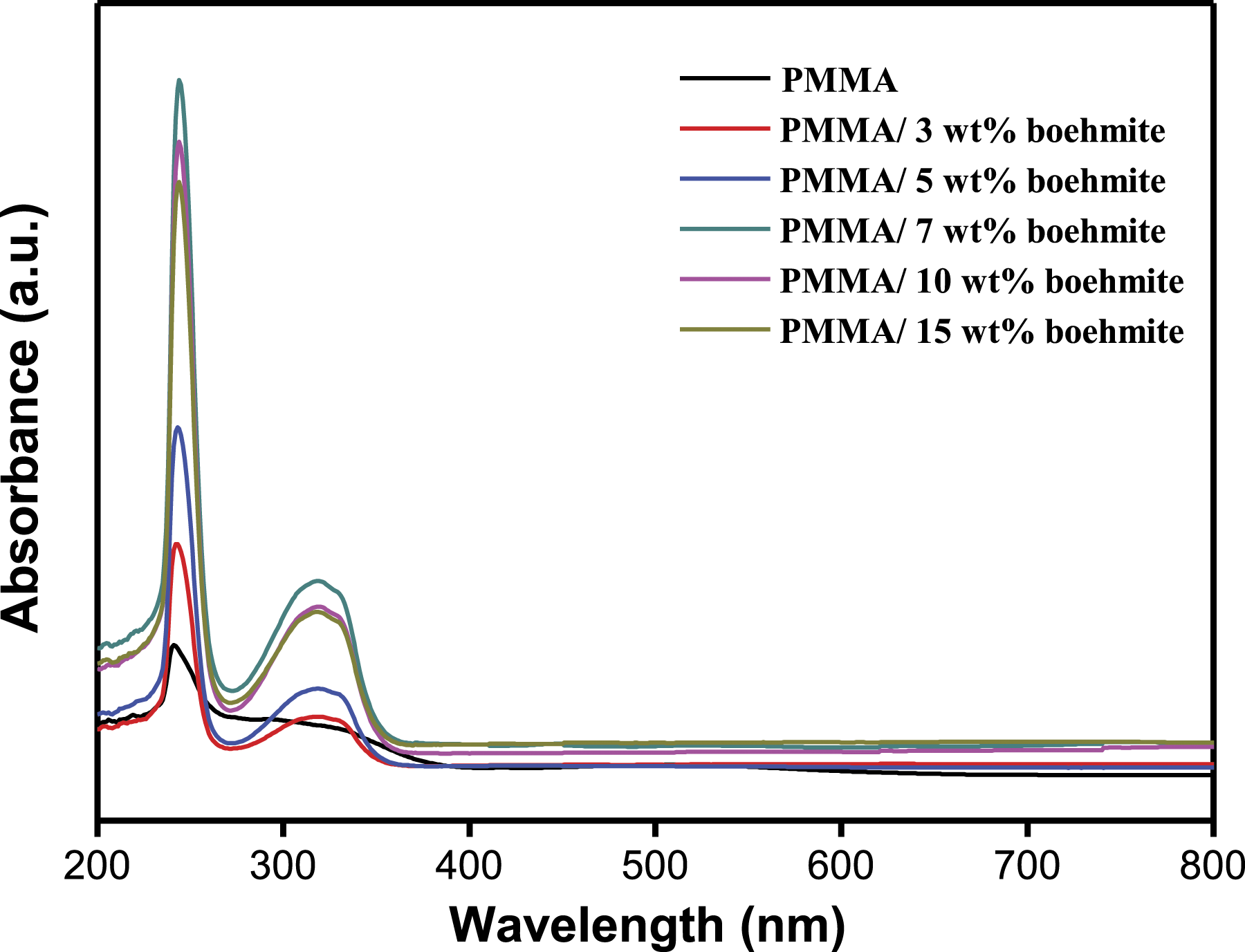
The optical bandgap is a crucial constraint on the performance of photocatalytic materials because the electronic excitation between the valance and conduction band occurs when the incident photon has an energy that is equal to or greater than the bandgap energy. Tauc and Menth proposed the following expression for obtaining the optical bandgap Tauc plots of PMMA and PMMA/boehmite nanocomposite films.
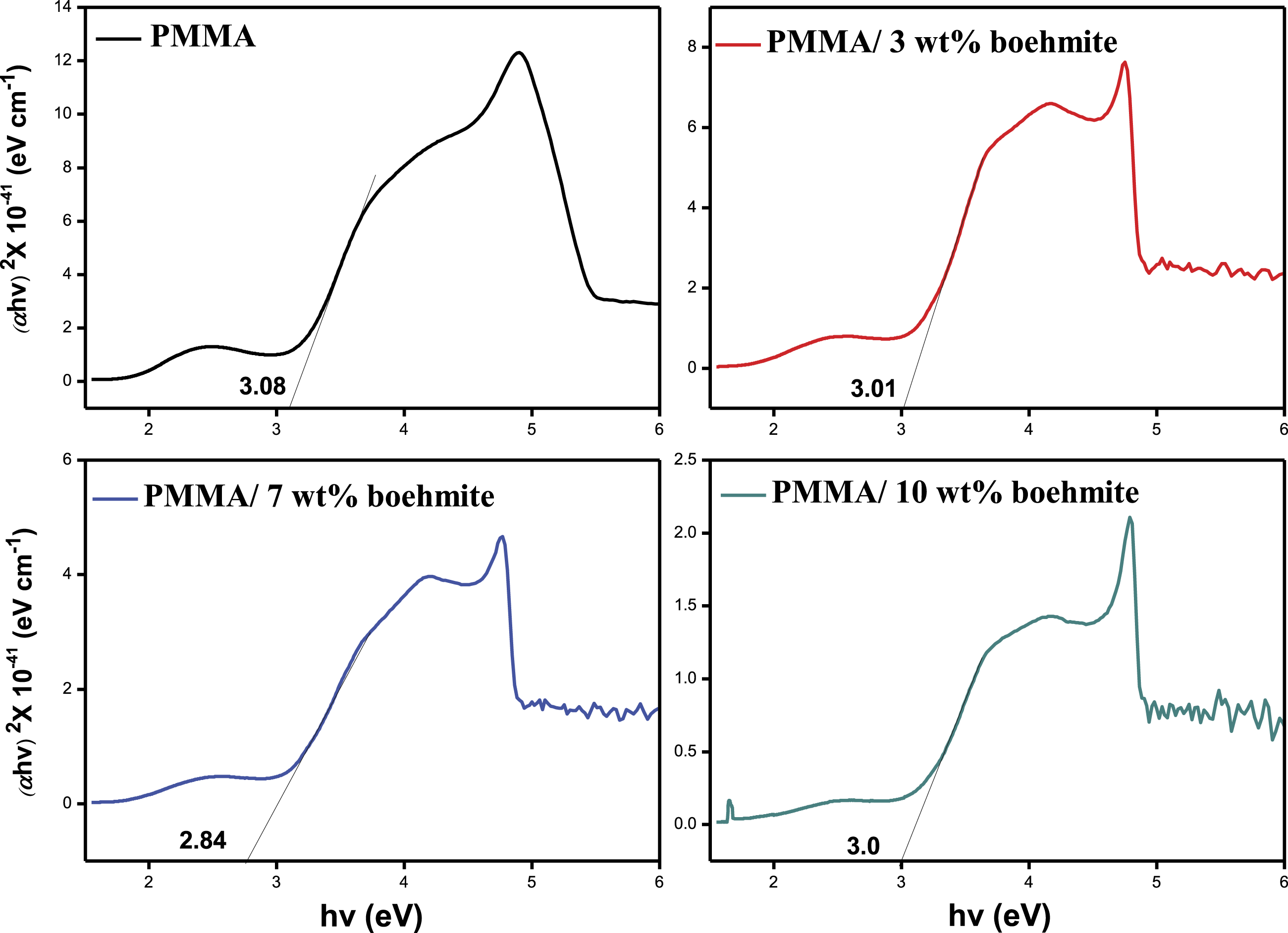
FT-IR spectra
Figure 3 shows the FT–IR spectra of boehmite nanoparticles, PMMA and PMMA/boehmite nanocomposite films. Boehmite shows a strong absorption band at 3305 cm −1 due to the presence of hydroxy groups in the AlOOH layers in the samples. The sharp peak at 1074 cm−1, as well as the three peaks at 740, 602, and 486 cm−1, are linked to Al-O bond vibration. The shoulder at 1653 cm−1 is the bending mode of the adsorbed water.
23
The FT-IR spectra of pure PMMA show the intense absorption peaks at 3000 and 2950 cm −1 correspond to CH stretching vibrations of the CH3 group and CH bending vibrations of CH2 group. The absorptions in the region of 1262 and 1060 cm −1 are attributed to C – O stretching vibration and wagging vibration of CH, respectively.
28
The band at 1712 cm−1 is the C = O stretching of the ester group from methacrylate.
29
From the different contents of boehmite coupled PMMA composite, it is found that the hydroxyl band of boehmite appears in the composite mainly due to the interaction of the carbonyl oxygen atom of the ester with the hydroxyl group available on the solid surface of the metal oxide particles. Moreover, the characteristic absorption of the C = O group in the composite is shifted to a higher wavenumber region (from 1712 to 1727 cm−1). The spectra of 5 and 10 wt % boehmite/PMMA composite show an additional peak at 518 cm−1 is the stretching vibration of boehmite nanoparticles in the PMMA chain. The appearance of new peaks or the shift in spectral band arises from hydrogen bonding between hydrogen and oxygen atoms in C = O or from the strong adhesion of AlOOH to the C = O group, or from both. Thus, from the FT-IR spectra, it is confirmed that the nanoparticles are successfully embedded within the macromolecular chain of PMMA. FT-IR spectrum of boehmite, PMMA and PMMA/boehmite nanocomposite films.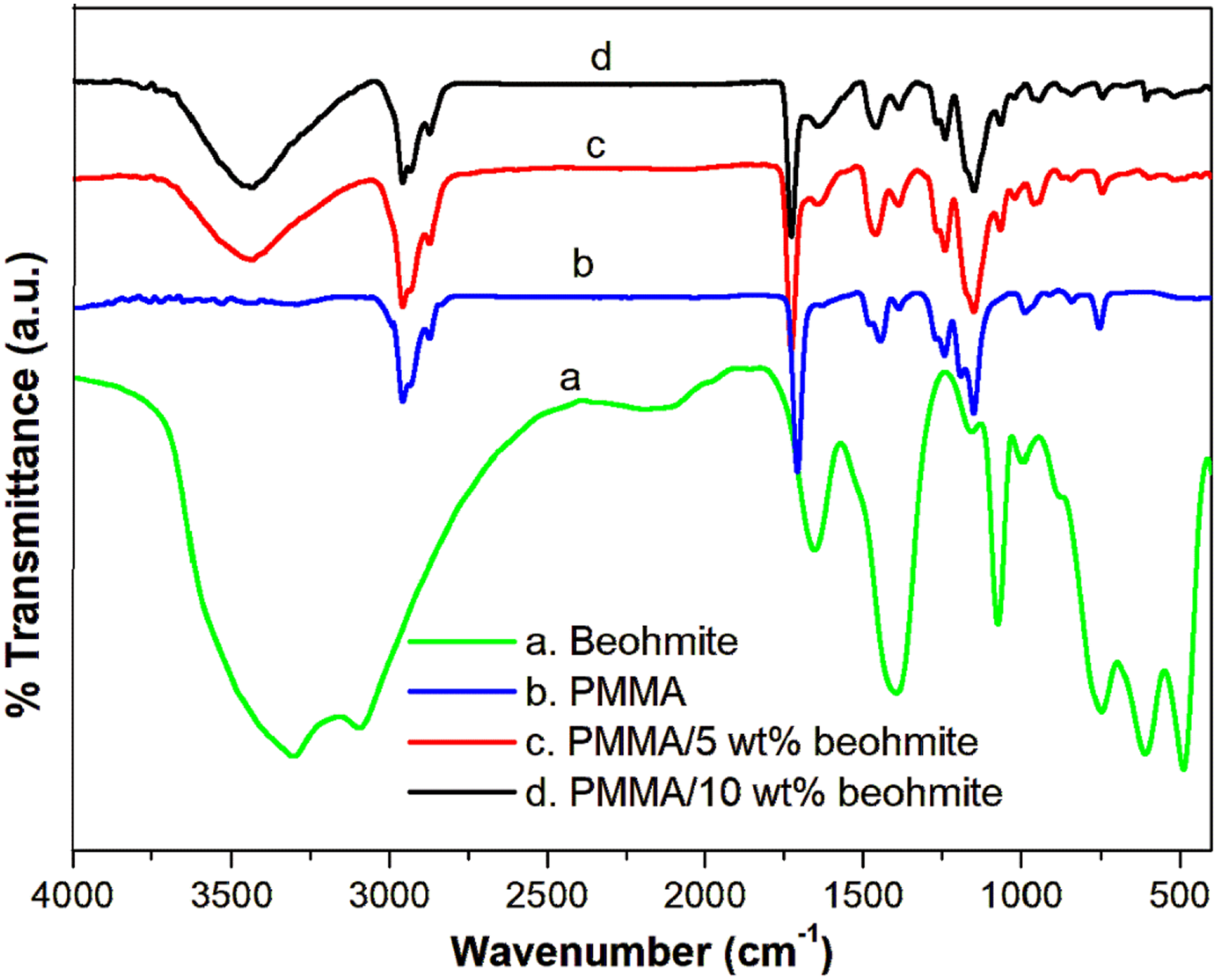
X –ray diffraction analysis
XRD patterns of PMMA, boehmite and PMMA with different concentrations of boehmite nanoparticles are exhibited in Figure 4. The diffraction pattern of boehmite nanoparticles shows several crystalline peaks at 2θ = 13.84, 27.86, 38.32, 49.34, 64.62 and 71.81° which corresponds to (020), (021), (130), (150), (151) and (200) lattice planes of boehmite and ascribes to JCPDS file no. 21-1275.
30
XRD patterns of PMMA, boehmite and PMMA/boehmite nanocomposite films.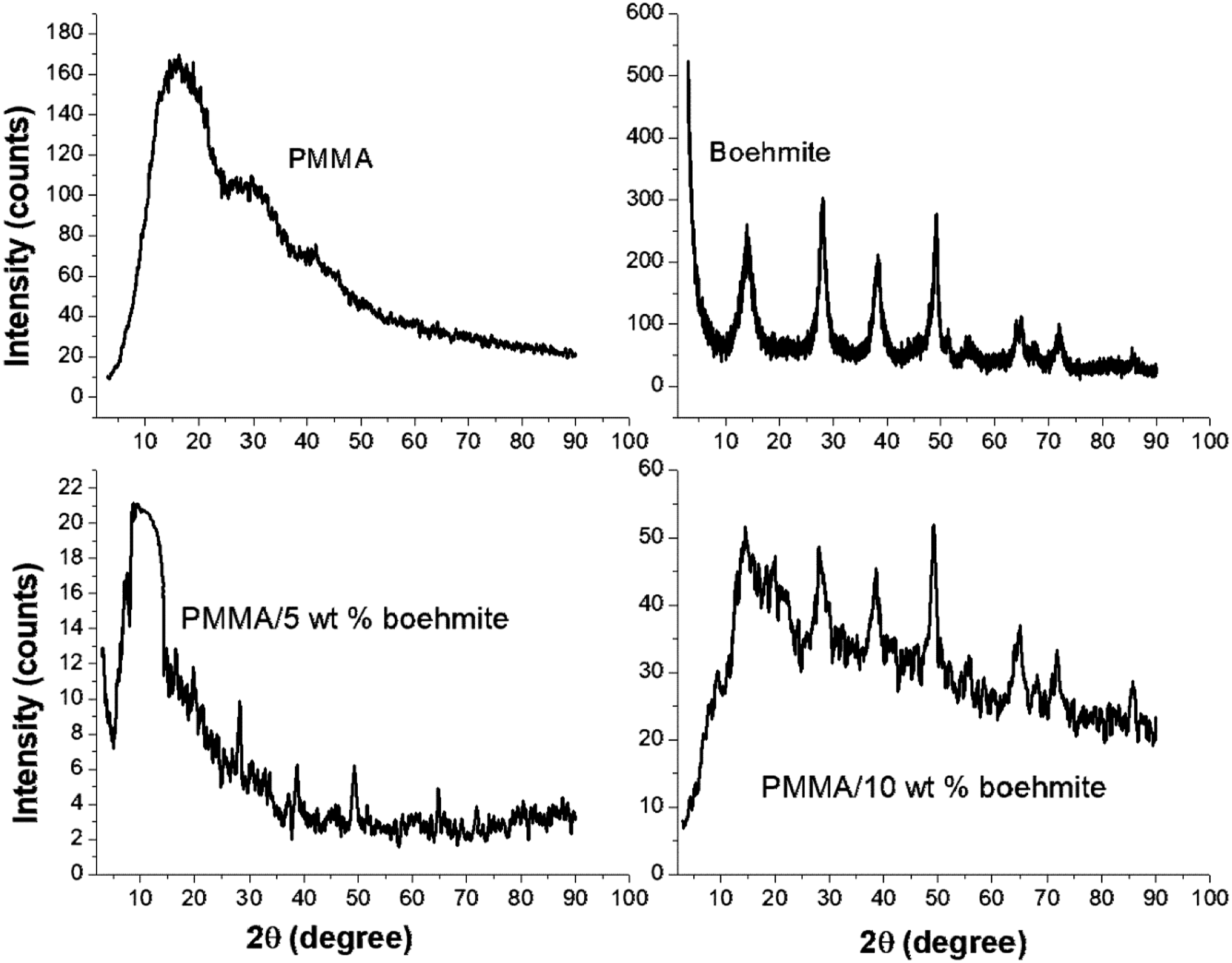
The PMMA exhibits a broad peak at 2θ = 15.97° and two weak peaks at 2θ = 29.64° and 41.04° indicating the amorphous portion of the PMMA. 31 However, the addition of boehmite nanoparticles causes the major XRD peaks of the PMMA to be slightly displaced to 2θ = 14.29°. It can be seen that the intensity of the amorphous peak of PMMA decreases with an increase in concentration of nanoparticles, whereas the crystalline region of samples increases. The decrease in broadness shape of the major XRD peak in PMMA after the addition of nanoparticles reflects the ordering inside the main chains. In addition, more peaks are observed in the polymer nanocomposite films which indicates the crystalline nature of polymer nanocomposite films. This decrease in broadness and shift in XRD peak with the addition of boehmite suggest a strong interaction between the ester group of PMMA and the hydroxyl group of nanoparticles, which improves the mechanical, electrical, and dielectric properties of PMMA, as discussed later. The increase in the degree of crystallinity with the addition of boehmite nanoparticles indicated that the structure of PMMA is strongly influenced by the concentration of nanoparticles. The XRD gives another piece of evidence for the formation and interaction of nanoparticles with the polymer chain. The percentage crystallinity of PMMA and its nanocomposites is estimated from the area of the XRD peak. PMMA has a crystallinity of 22.6%, while boehmite has a crystallinity of 82.3%. The incorporation of 5 and 10 wt% boehmite containing PMMA enhances the percentage crystallinity to 26.8 and 32.4% respectively.
Scanning electron microscopy
Figure 5 shows the SEM images of the dispersion of boehmite nanoparticles in PMMA. It can be seen from the figure that the pure PMMA shows a homogenous structure of morphology with a smooth surface. The SEM image of polymer composite with 5 wt % of boehmite shows morphology with some irregularity caused by the insertion of nanoparticles in the polymer matrix. The 7 wt % composite shows a uniform structure with the presence of hemispherical boehmite particles. This uniform structure morphology is due to the strong interfacial interaction between boehmite and the ester segment of the PMMA chain. As the concentration of nanoparticles increases to 10 wt %, the surface of the composite becomes coarser, indicating that the nanoparticles are irregularly arranged with few formations of voids in the composite. The dispersed nanoparticles are elongated in the PMMA matrix which is due to the poor interaction between the nanofillers and the polymer chain. Further, the particle-to-particle distance in the polymer is too short at higher loadings, which leads to the agglomeration of nanoparticles in the polymer nanocomposite. SEM images of PMMA with (a) 0 (b) 5 (c) 7 and (d) 10 wt % of boehmite.
Differential scanning calorimetry
The glass transition (Tg) and melting temperatures (Tm) of PMMA nanocomposite films with various loadings of boehmite nanoparticles is shown in Figure 6. PMMA is commonly used as a soft segment in a variety of applications due to its low glass transition temperature and durability.
32
The glass transition and melting temperatures of pure PMMA are appears at 68.5°C and 144°C, respectively. However, the Tg of PMMA/boehmite nanocomposite films increased to 75.4 and 79.5°C, respectively. Similarly, the Tm of PMMA is increased to 148.7 and 152.5°C, respectively. The addition of nanoparticles into the polymer matrix leads to strong interfacial bonding between the polymer chain and the nanoparticle surfaces and therefore the interfacial layer affects the glass transition temperature of the polymer matrix. In other words, the strong interaction of boehmite with PMMA leads to close contact with the polymer region, resulting in an increase in thermal transition values.
33
Therefore, it can be concluded that the thermal properties such as glass transition and melting temperatures are affected by the concentration of nanoparticles. DSC plots of PMMA with different contents of boehmite nanoparticles.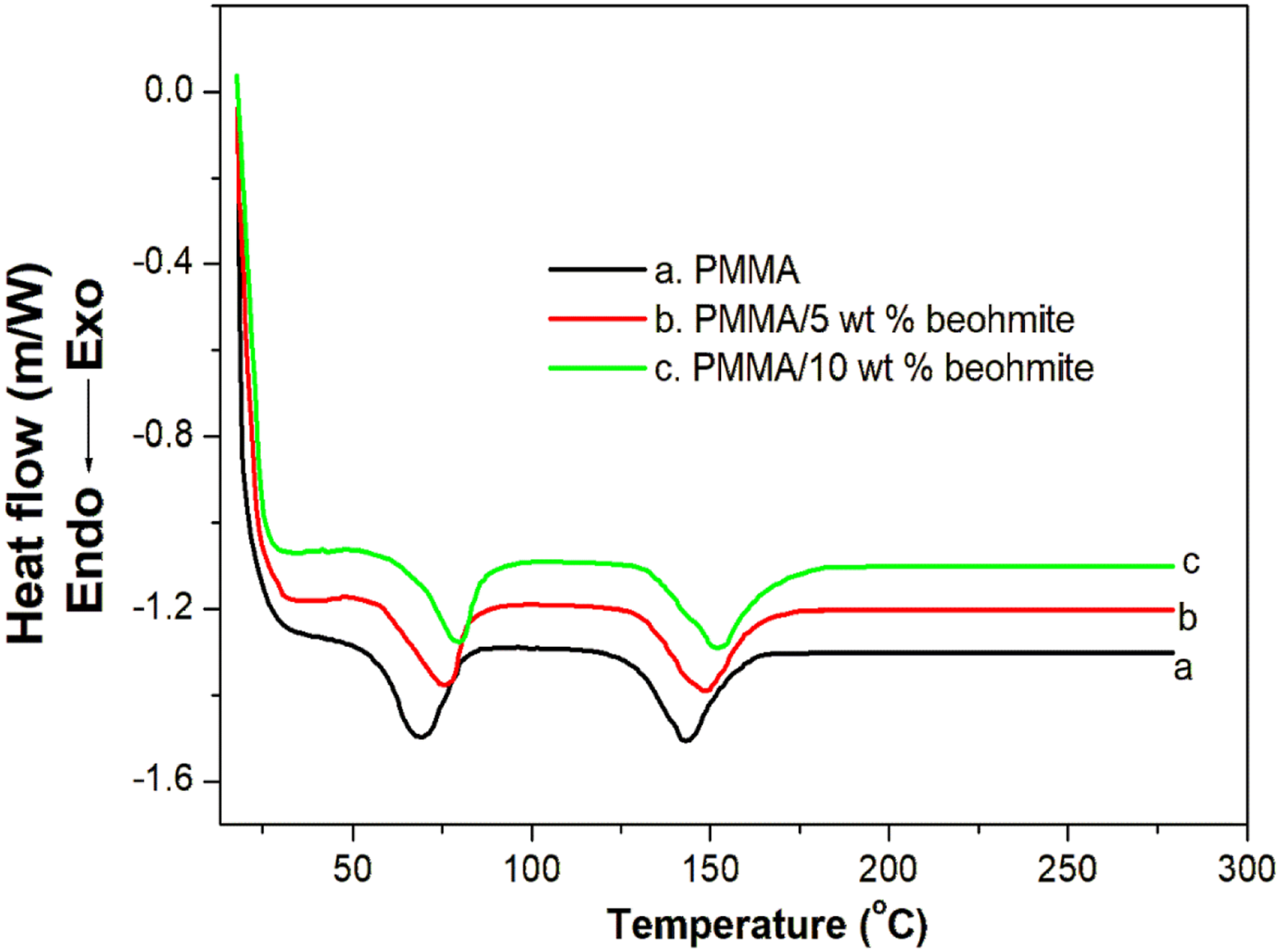
TGA analysis
The thermal degradation behavior of PMMA and PMMA containing 3, 7 and 10 wt% of boehmite nanoparticles is given in Figure 7. It is clear that all the samples show a single-stage decomposition pattern. Boehmite nanoparticle-reinforced PMMA nanocomposite films showed higher thermal stability than the parent polymer. The thermal degradation of PMMA occurs at 312°C, while the nanocomposite film with loadings of 3, 7 and 10 wt% shows major degradation at 324, 330 and 338 °C respectively. The increased thermal stability of nanocomposite films is mainly due to the uniform dispersion of boehmite nanoparticles and the strong intermolecular interaction between nanoparticles and the PMMA chain.
34
The final char residue that remained at 500°C for PMMA is 1.8% while the composite with 3, 7 and 10 wt% boehmite showed the char residue 6, 10 and 14%, suggesting the improved flame retardancy of the PMMA nanocomposite films. The PMMA/boehmite has greater thermal stability than NiO nanoparticles reinforced PMMA.
35
This is because PMMA/boehmite nanocomposites have stronger polar-polar interactions than PMMA/NiO nanocomposites. TGA plots of PMMA with different contents of boehmite nanoparticles.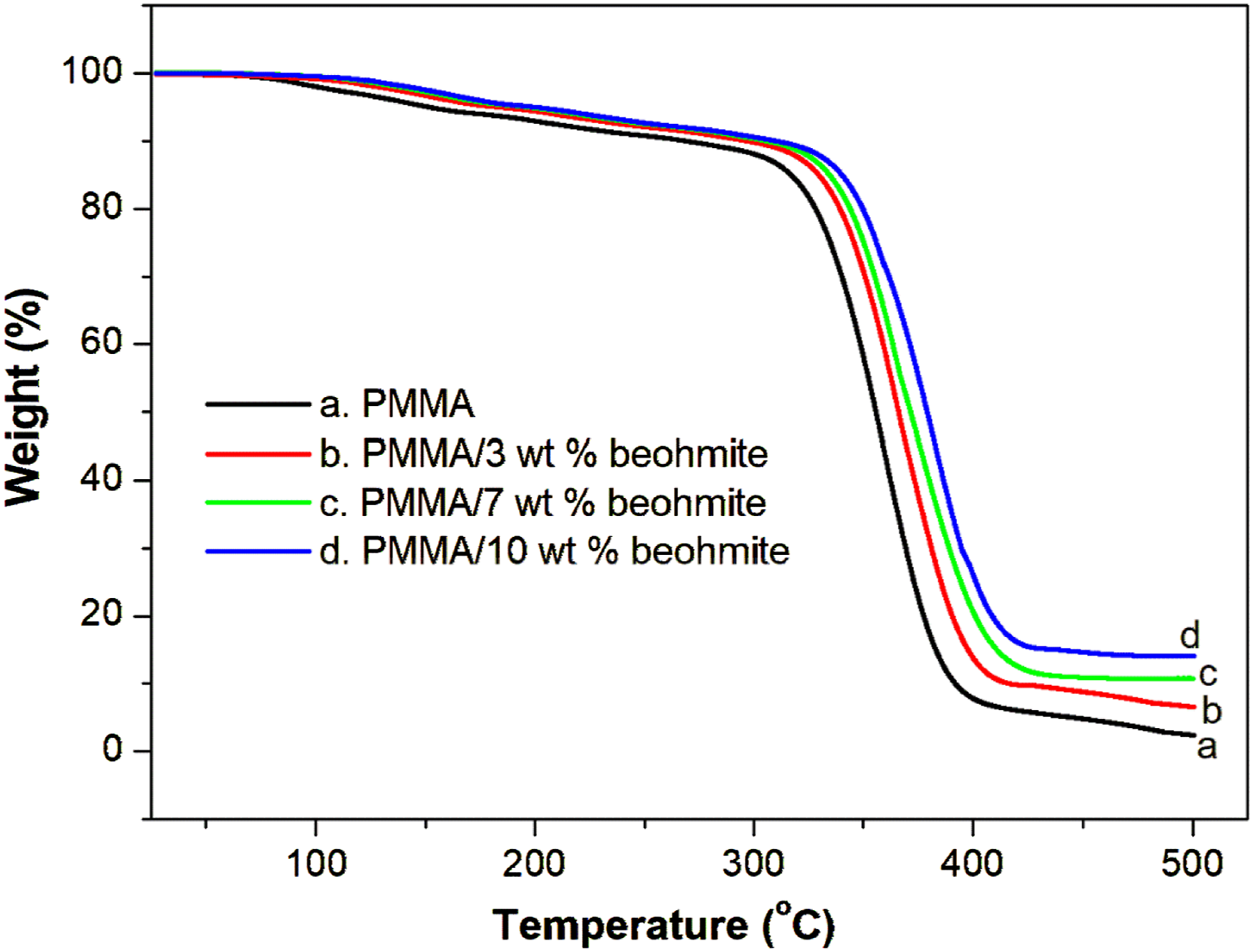
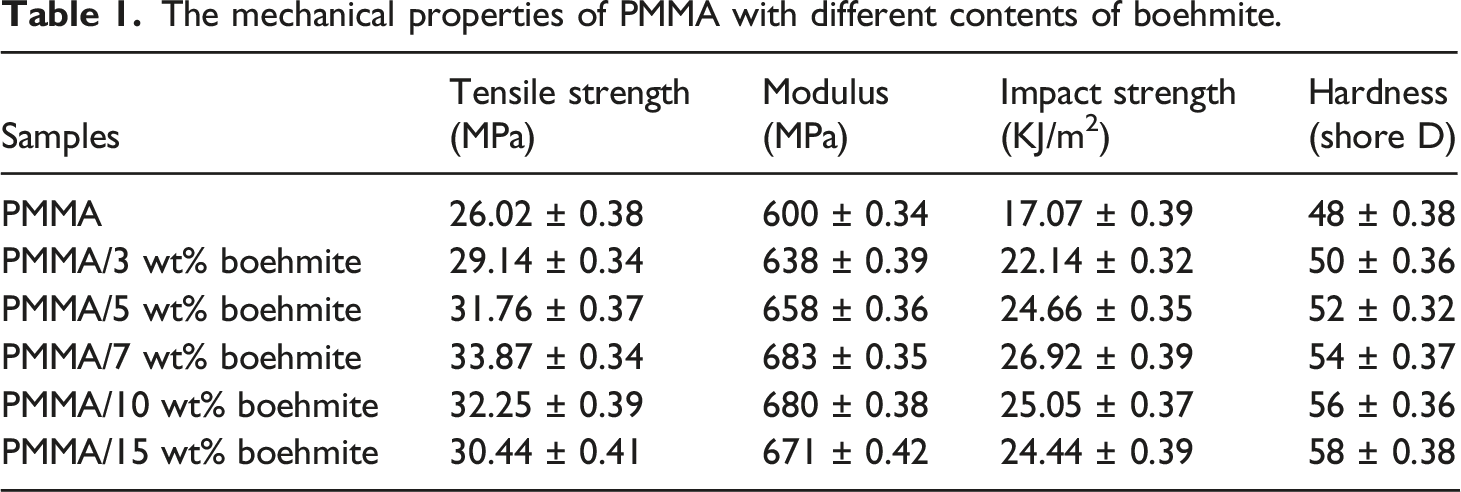
Mechanical properties
The mechanical properties of PMMA with different contents of boehmite.
AC conductivity
Figure 8 shows the experimental results for the AC electrical conductivity of the prepared films at different temperatures in the frequency range (0.01–1.0 MHz). This figure shows three different regions: the low frequency, high-frequency, and in-between regions (plateau region). We note that this behaviour obeys the relationship:
38
Variation of AC conductivity of PMMA and its nanocomposite films at different temperatures.
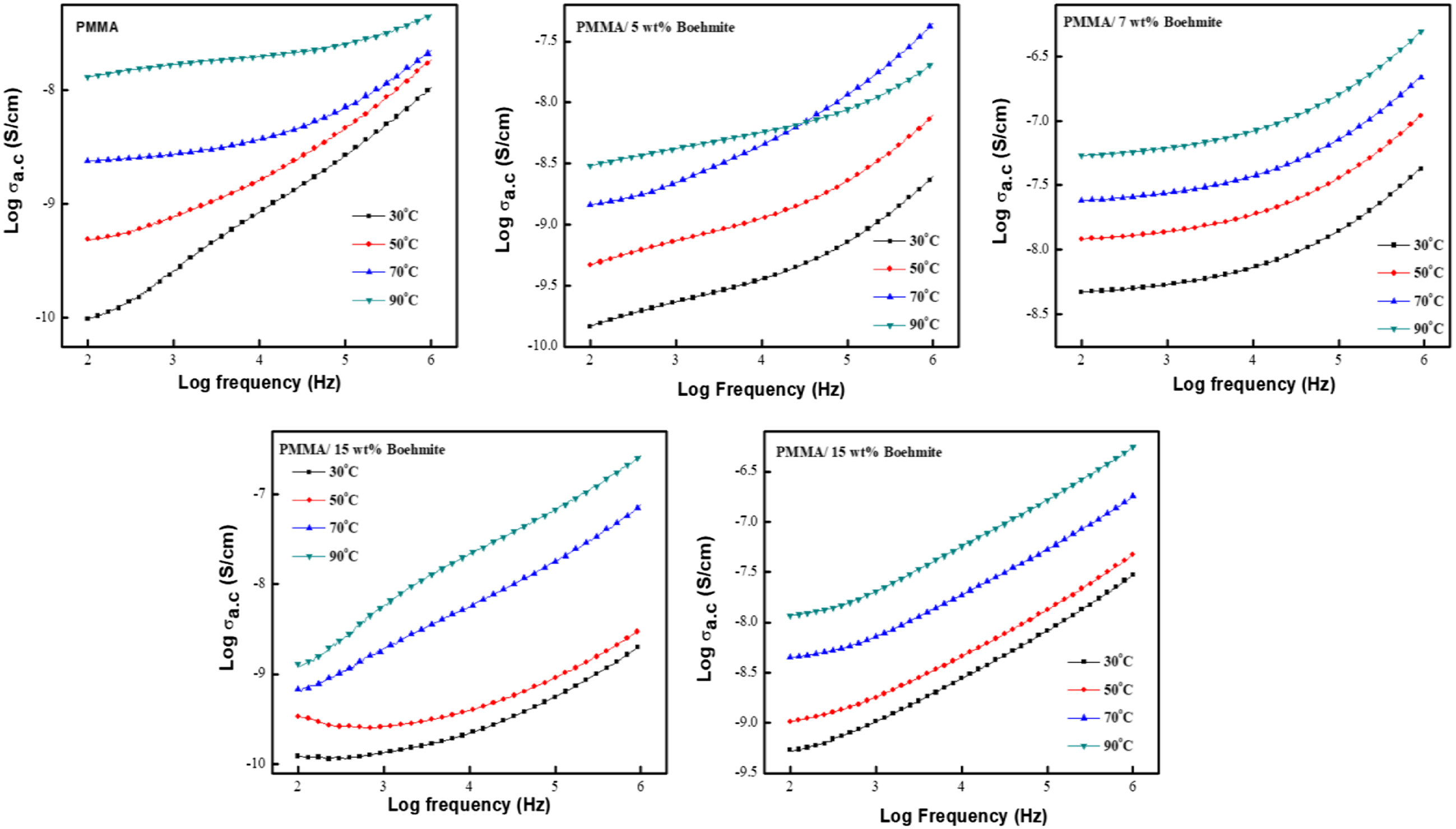
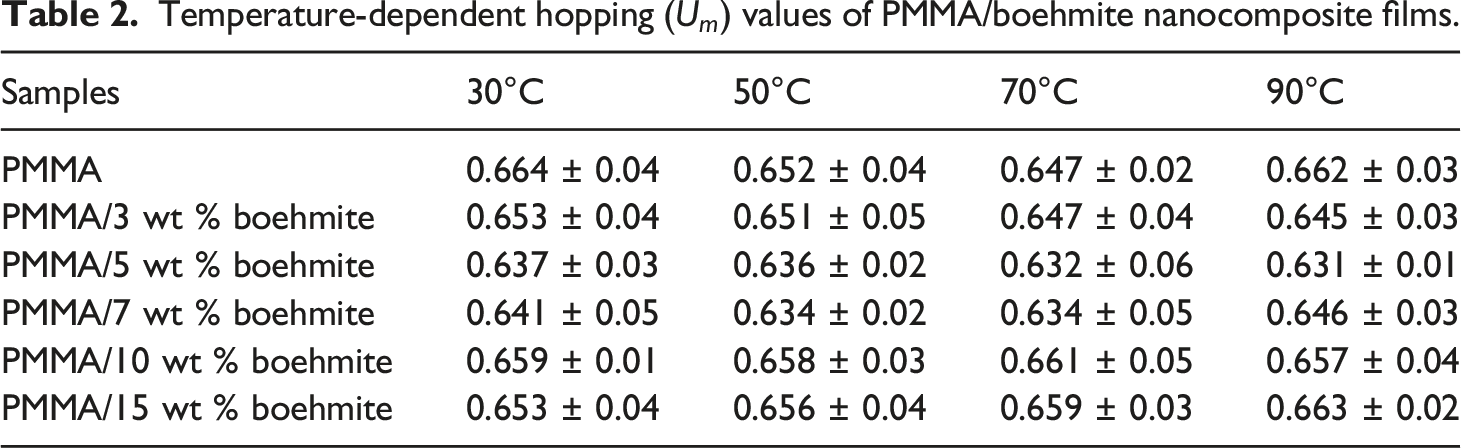
From Figure 8, it can be seen that the temperature change does not affect the electrical conductivity behaviour of the prepared samples, but it enhances the electrical conductivity of the prepared samples. This is because the mobility of chain segments becomes flexible, and the rotation of side groups becomes soft. In addition, the hopping of free charges from one site to another is enhanced through the heating process. More dipoles are also oriented at high temperatures, leading to a higher equivalent surface charge density and conductivity value.38,39 The transition frequency from frequency-independent to frequency-dependent conductivity called critical frequency, increased as the nanofiller content increased, indicating that the transition region shifts to a higher frequency as the boehmite nanoparticle content increases. 40

Temperature-dependent hopping (

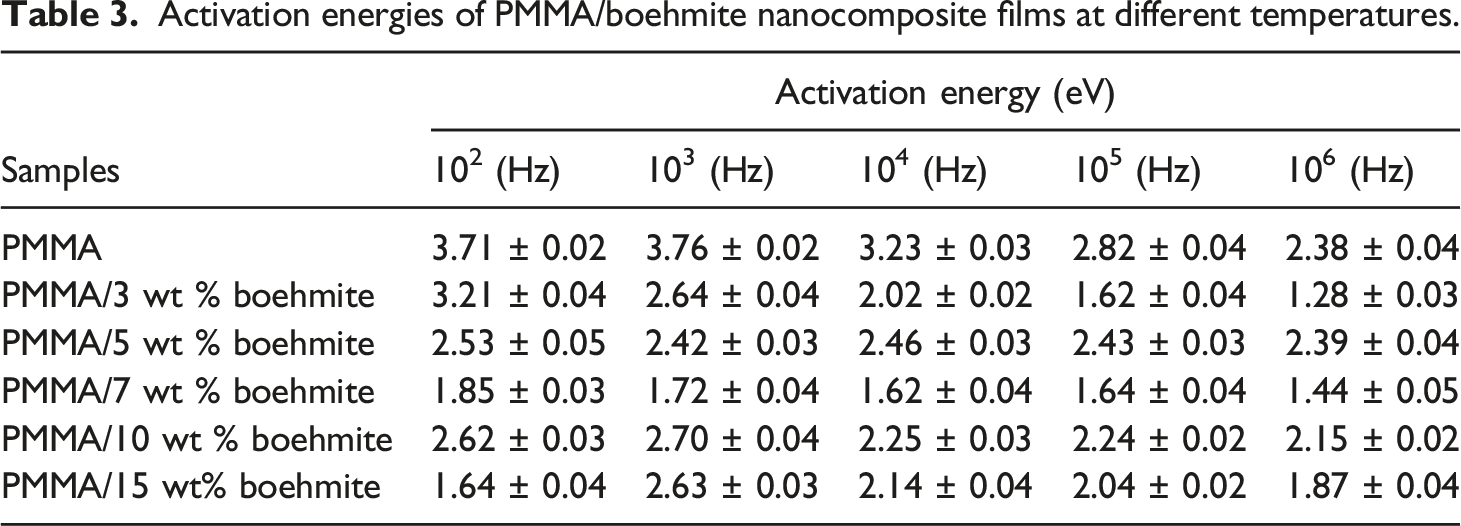
Activation energies of PMMA/boehmite nanocomposite films at different temperatures.
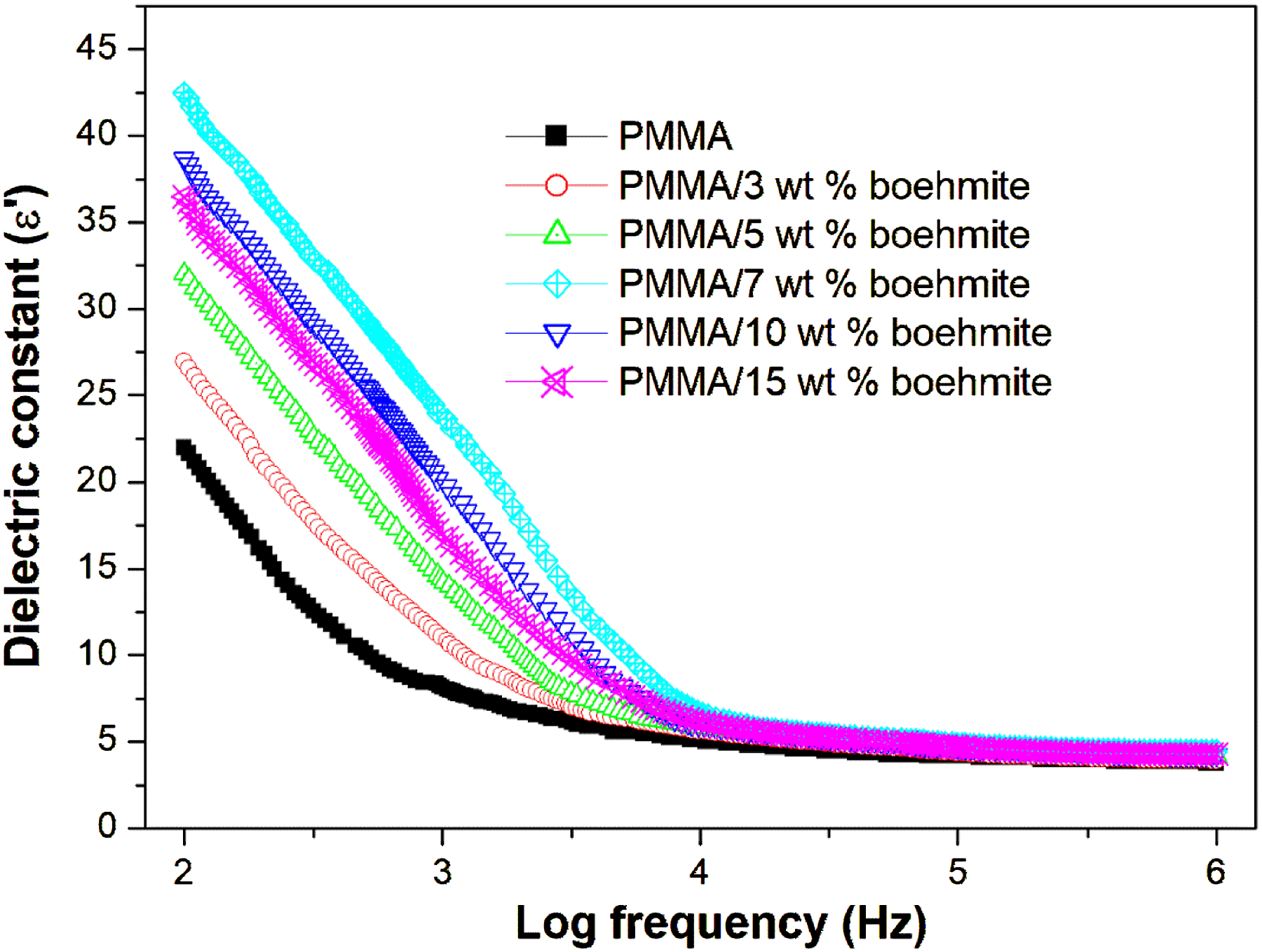
Dielectric constant
The importance of studying the dielectric properties of polymers is that they provide important information about the nature of their capacity and the nature of their conductivity. We note that the value of the dielectric constant at high frequencies is a constant for all the prepared films, as depicted in Figure 10. This is because dipoles, despite their abundance, are unable to orient themselves or follow the applied field changes. Furthermore, in the lower frequencies, the value of the dielectric constant increases with decreasing frequency due to the tendency of dipoles in large molecules to orient with the direction of the field.
43
In addition, the value of the dielectric constant in this region increases with the increase in the concentration of boehmite, reaching the maximum value at 7 wt% and then decreasing with the addition of more nanofiller. This result depicts the electrostatic interactions between the functional groups of the mixture and boehmite, which may increase the mini capacitors within the samples. The increased polarization of the molecules of these films due to the addition of boehmite means an increase in their ability to store electrical energy, which makes them a suitable candidate for the applications of flexible film capacitors that contain polymeric nanocomposite films. PMMA/boehmite has higher optical, thermal, electric, and dielectric properties than the ZnO nanoparticles incorporated PMMA,
44
indicating potential applications in the fabrication of optoelectronic devices with high charge storage capacity. Activation energy plot for PMMA and PMMA/boehmite nanocomposite films. Variation of dielectric constant of PMMA with different contents of boehmite.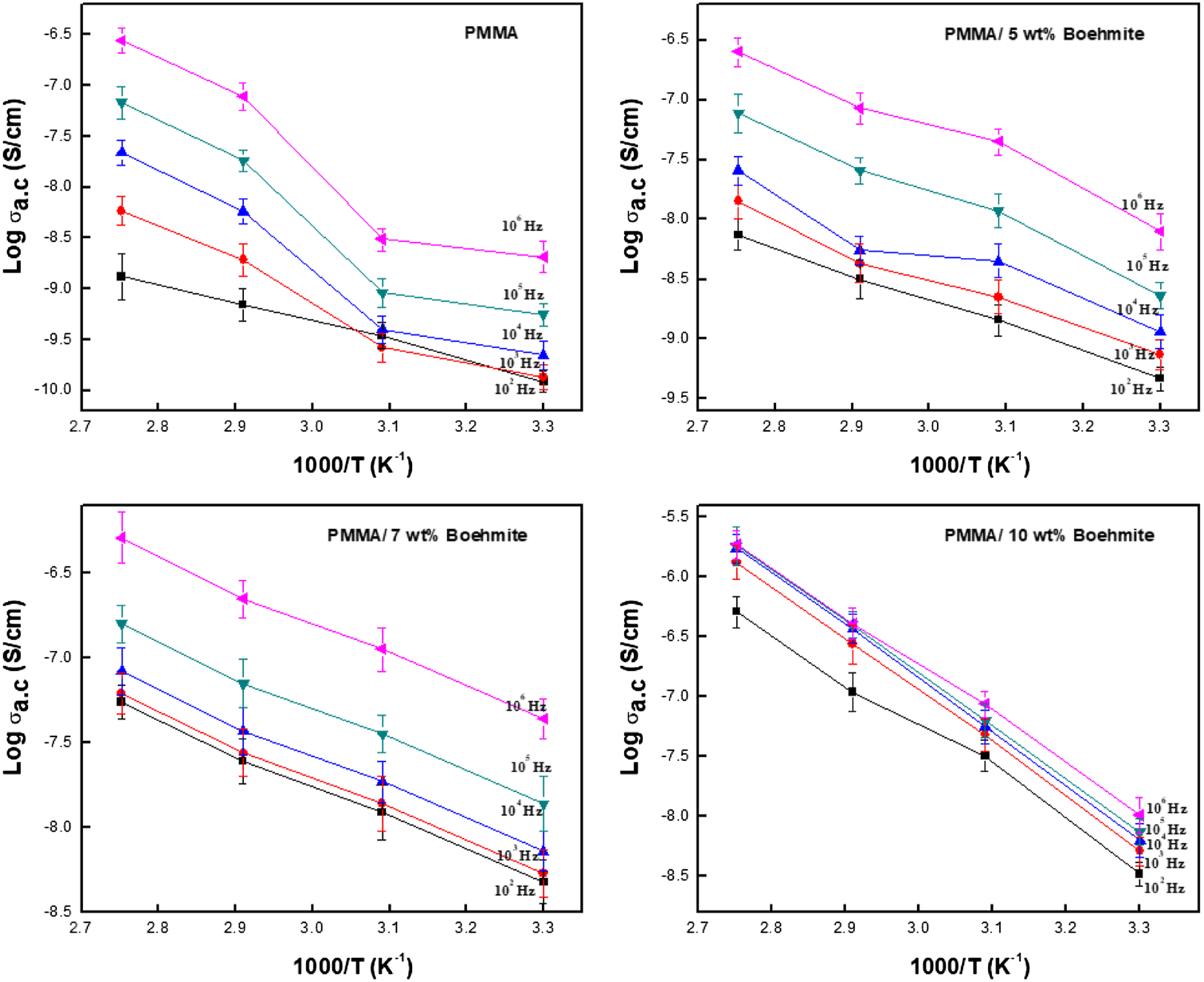
Conclusions
Boehmite-reinforced PMMA was successfully prepared by an unpretentious, non-toxic, and cost-effective dissolution casting method. The UV- Vis spectra emphasize a shift in absorbance maxima, which signifies strong electrostatic interaction of boehmite in the PMMA matrix. The optical bandgap energy decreases with filler loadings up to 7 wt%. FTIR spectra of PMMA/boehmite nanocomposite films indicated a characteristic peak around 500–700 cm−1, which indicates the interaction between boehmite and the PMMA matrix. The XRD results revealed a systematic and well-ordered arrangement of boehmite in the PMMA matrix. The SEM images reveal the uniform dispersion of boehmite in PMMA matrix. DSC analysis emphasis the enhanced glass transition temperature and thermogravimetric analysis (TGA) analysis reveal the superior thermal stability of PMMA/boehmite nanocomposite films relative to the bare PMMA matrix. Mechanical tests revealed that PMMA and 7 wt% boehmite had the highest tensile and impact strengths, while bare PMMA had the greatest elongation at break. Hardness was observed to increase with the concentration of boehmite filler in PMMA matrix. The AC conductivity increases as a function of filler loading, frequency and temperature. The enhanced AC conductivity was further supported by the calculated activation energy. The dielectric constant decreases with frequency and increases with filler loadings. Overall, the results demonstrated that the PMMA nanocomposite films with high tensile strength, optical properties, conductivity and dielectric constant can be employed to manufacture EMI shielding and flame-resistant flexible optoelectronic devices.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
