Abstract
In this work, the thermal stability of wood flour (WF) was modified using a boric acid treatment. To further understand the impacts of incorporating various proportions of titanium dioxide (TiO2) nanoparticles loaded on polycarbonate (PC)/wood flour. The rheological, mechanical, thermal, electrical, and interfacial properties were studied. Various properties of the resultant PC/WF/TiO2 nanocomposites were then characterized. The viscosity of the nanocomposite was shown to decrease with shear rate and temperature. XRD investigation indicated that, by introducing TiO2 nanoparticles into PC/WF, the amorphous phase decreased, and the degree of crystallinity increased, as compared to bare PC/WF, from 65.78 to 42.29. In addition, the TGA profile showed that the nanocomposites have a higher resistance to thermal degradation than PC/WF alone. It was observed that the char residue resulting from the TGA of PC/WF/10%wt TiO2 increased to 5.69% compared to pure PC/WF (1.32%). It was found that an appropriate amount of TiO2 in PC/WF caused a significant increase in tensile strength, elongation at break, and on impact strength. These were attributed to an increase in the interface of the dispersed phase. The incorporation of nanoparticles enhanced the thermal conductivity of the nanocomposites, which was found to be higher when a TiO2 content of ∼3 wt% was used. The electrical conductivity of the PC/WF/TiO2 nanocomposite (<3 wt%) hindered the movement of free-moving ions, therefore reducing the nanocomposite’s electrical conductivity. However, at > 3 wt% of TiO2, due to the aggregation of nanoparticles, TiO2 nanoclusters formed in the PC/WF composite. Therefore, the additional space so created for the movement of conduction ions at the interface may have resulted in increased ion mobility. This could be the one of explanations for the increased electrical conductivity associated with higher wt% of TiO2. Results showed that treating the PC/WF/TiO2 nanocomposites with boric acid decreased both the rate of heat release and the total heat release.
Keywords
Introduction
As an engineering plastic, polycarbonate is a durable, versatile building material used in a lot of construction projects. It can be used both indoors and outdoors in residential, commercial, and industrial applications such as in skylights, windows, partitions, and domes. There are many reasons engineers, architects, and designers like building with polycarbonate panels. Some of these reasons are that they are lightweight and sturdy, come with high-optical clarity, high-impact and high-heat resistant, and extremely flame resistant.
1
PC is also insensitive to moisture and durable to various weather conditions.
2
Consumer electronics, automobiles, medical apparatus and instruments, structures, packaging, and other areas have all made extensive use of this material.
3
Many additives have been combined into PC-based materials to expand its application domains and overcome its limitations, such as has poor abrasion properties, scratch resistance, high cost, and hard coating. Manufacturers and researchers are interested in wood-polymer composites (WPCs) made of biomass fibers and polymers because of their advanced, environmentally friendly attributes, biodegradability, and antiseptic properties.
4
They have been widely employed in the automotive, transportation, construction, and decorative industries.
5
Wood flour (WF), is used to prepare the organic fillers to form wood-plastic composites. Furthermore, wood flour a kind of biomass material that is environment-friendly, and low density with low cost, making it an excellent choice as a filler, improving toughness and energy recovery, causing lower pollutant emissions, and lowering non-renewable sources usage.
6
Ahlem et al.
7
investigated the addition of TiO2 nanoparticle to polypropylene and its effect on the material’s thermal, rheological, and crystallinity levels. It was discovered that TiO2’s influence is strongest at low frequencies, and because of shear thinning, the relative effect decreases as frequency increases. The polymer chains' entangled state precludes shear flow at lower frequencies, which results in high viscosity. The mean interparticulate distance is high at 2.5wt% TiO2, which causes a slightly increase in viscosity due to a minimal hydrodynamic disruption. The complex viscosity significantly reduces with the nanoparticle concentration when 5wt% TiO2 is added to polypropylene. The complex viscosity decreases at greater TiO2 contents (>5Wt%). Since TiO2 nanoparticles agglomerate at larger concentrations, this decline is probably caused by it. The surface that the nanocharge has with the polymer matrix is reduced as a result of this agglomeration. Zhang et al.
3
showed that the wood flour dispersed in the polycarbonate matrix as micro or nanoparticles. The introduce of treated wood flour with boric acid increased the thermal stability of the resulting composites. The flexural and tensile modulus linearly increased with the increase of wood flour content by 147.2% from 3.6 GPa for pure PC to 8.9 GPa for the WPCC40 and 27.7% while the tensile modulus increased by 129.6%, respectively. This increase can be ascribed to the stiffness of the wood flour. Meanwhile, the addition of wood flour would reduce the cost of PC composites. However, both biomass fibers and polymers are inherently ignitable, which means that WPCs carry a considerable fire risk. Hence, it is important to decrease the flammability of WPCs to expand their commercial utilization. Generally, flame-retardants and non-combustible particles are added to WPCs to improve their flame retardancy.
8
Titanium dioxide is a critical inorganic filler/reinforcement agent material used in polymer composite materials. It sees widespread use in a variety of applications, such as optoelectronic and photocatalytic activities, electrochromic applications, hydrogen storage, and gas sensors.9,10 Inorganic reinforcing agents such as titanium dioxide are added to thermoplastics to improve their mechanical characteristics such as yield stress and modulus of elasticity. On the other hand, increasing the proportion of reinforcing agent content might result in a substantial loss of impact strength and elongation at break. According to Zhenhua,
11
composites made of poly(methyl)methacrylate (PMMA) and titanium dioxide were created in a variety of concentrations. On the mechanical characteristics and thermal behavior of PMMA composite film, the impacts of fiber content and modification were examined. When used as a binary modifying agent for TiO2 filler, organosolv lignin and acrylic acid enhanced the strength and ultimate strain of treated PMMA composites relative to untreated ones. Chaisaenrith and Pavasupree conducted a thorough investigation of the impact of titanium dioxide (TiO2) on the flame retardant char-forming effect of ammonium polyphosphate in polypropylene. An intumescent nitrogen-containing resin was also used. Titanium dioxide improved the fire resistance by making the material stronger, forming more char with a higher expansion and an increased porosity.
12
The results of using nano-titanium dioxide to manufacture paint and flame-retardant coatings revealed the product’s excellent antipollution properties and flame resistance.
13
The impact of fillers like mica and fly ash, at various concentrations, on the mechanical, thermal, electrical, rheological, and morphological characteristics of polyester thermoplastic elastomer were investigated by Sreekanth et al.
14
The goal of this work is to find an optimal reinforcement agent content (TiO2) with good dispersion that can be added to the PC/WF composite while considering various issues such as rheological, mechanical, thermal, and electrical properties. To modify TiO2 nanoparticles, a silane coupling agent (3-aminopropyltriethoxysiane, APTES) was to improve the TiO2 dispersion and interfacial adhesion between the TiO2 and PC/WF composite. As a result, the viscosity of the nanocomposites increased. This finding an agreement with reported literature.
15
Figure 1 displays several factors that may affect the mechanical, rheological, electrical, and thermal properties of nanocomposites and organic/inorganic fillers to be valuable material in electronics, automobiles, medical apparatus and packaging for hydrocarbon liquid such as oil which used in lubricating. The factors that impact the mechanical, rheological, electrical, and thermal properties of nanocomposites materials.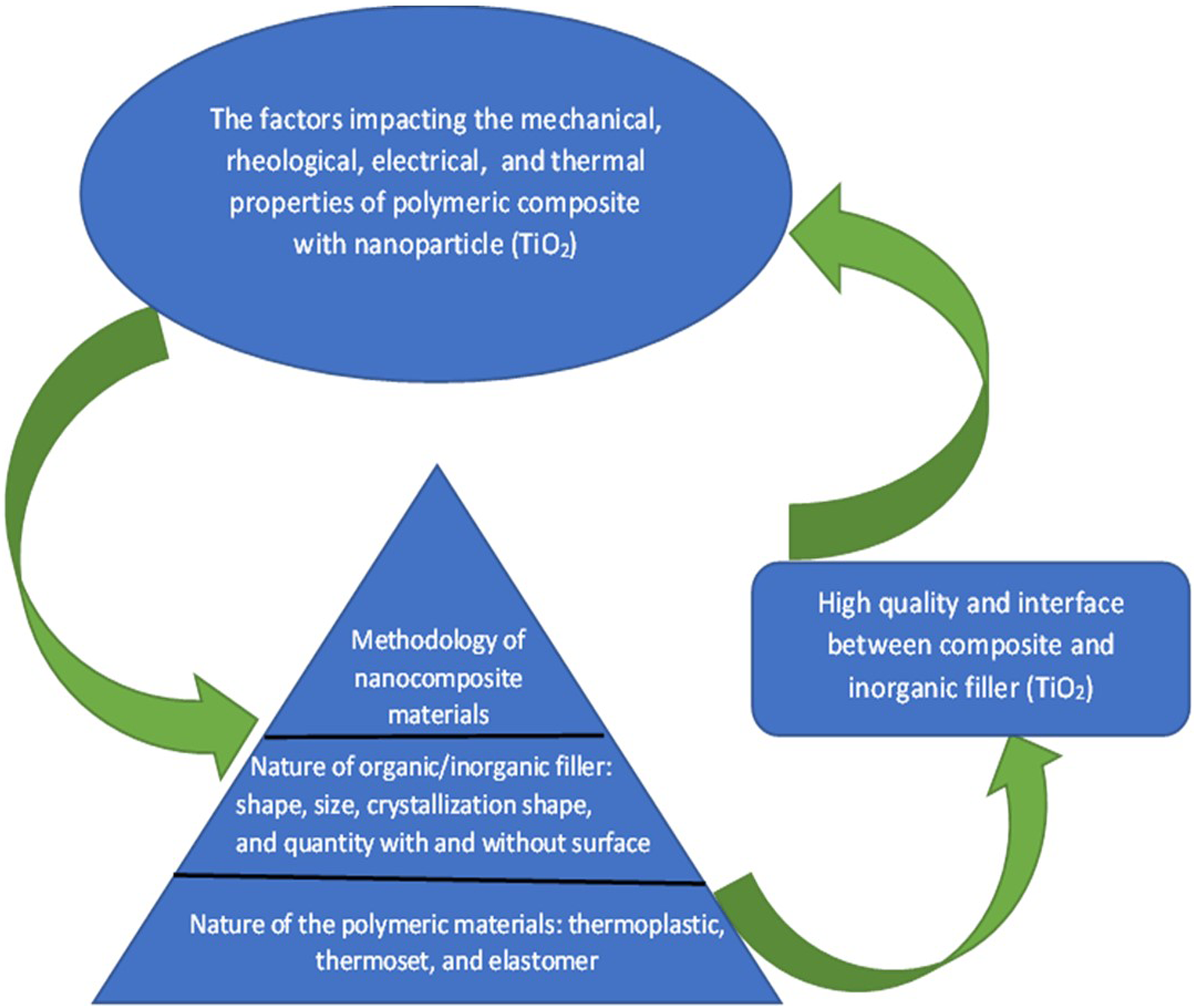
Experimental
Materials
Commercial polycarbonate with a melt index at 300°C of 19.6 g/10 min, a polydispersity of 2.16 weight, a glass transition temperature of about 144.5°C, a density of 1.20 gm/cm3, and with an average molecular weight, Mw, of about 57,404, was supplied by BASF, Germany. Commercial wood flour, 120 mesh sieves (125 μm) purchased from Ontario Sawdust Supplies was used as a filler. The moisture content of the WF was estimated by ASTM D4442 to be 5–6 wt. Commercial titanium dioxide, as supplied by Sigma Aldrich and with a particle size of 60.41 μm, was used as nanoparticle. 3-aminopropyltriethoxysiane (APTES) was purchased from Sigma Aldrich.
Wood flour modification
Wood flour was immersed in a 5 wt% boric acid solution for 4 h. The solid-to-liquid ratio was 1:4. The treated WF was then filtered and oven-dried for 24 h at 110°C to release the last of the moisture content. The control group was treated with deionized water before being oven-dried for 24 h at 110°C.
Surface modification nanoparticles of TiO2
PC, WF, and TiO2 particles were dried in a vacuum oven at 80°C for 12 h to decrease the amount of absorbed moisture on their surfaces. To ensure thorough dispersion, 4 g of TiO2 nanoparticles were ultrasonically dispersed for 25 min in a mixed solution of 100 mL anhydrous ethanol and 40 mL deionized water. 0.4 g of hydrolysis-treated APTES was added to each sample. The pH of the solutions was gradually elevated to 5 and 10 using ultrasonic treatment at 50°C for 2 h. The dispersed particles were then separated from the solvent using a centrifuge (10 min at 10,000 rpm), followed by washing with ethanol and water alternately for at least two cycles to remove excess silanes. To re-disperse the centrifuged particles in fresh solvent, they were put in ultrasonic bath for more than 15 min to make sure a visually well dispersed suspension was regained before centrifuging again. The modified TiO2 nanoparticles were dried in a vacuum oven at 80°C for 12 h. 2 g of modified TiO2 nanoparticles were added to the PC/WF particles and dispersed with a stirring at speed of 300 rpm for 3 h in mild anhydrous ethanol. Modified PC/WF/TiO2 particles were generated after drying the mixes for 12 h at 80°C. The modification of APTES on the TiO2 nanoparticle surface is depicted in Figure 2. Chemical grafting of coupling agent (APTES) onto TiO2 nanoparticles surface.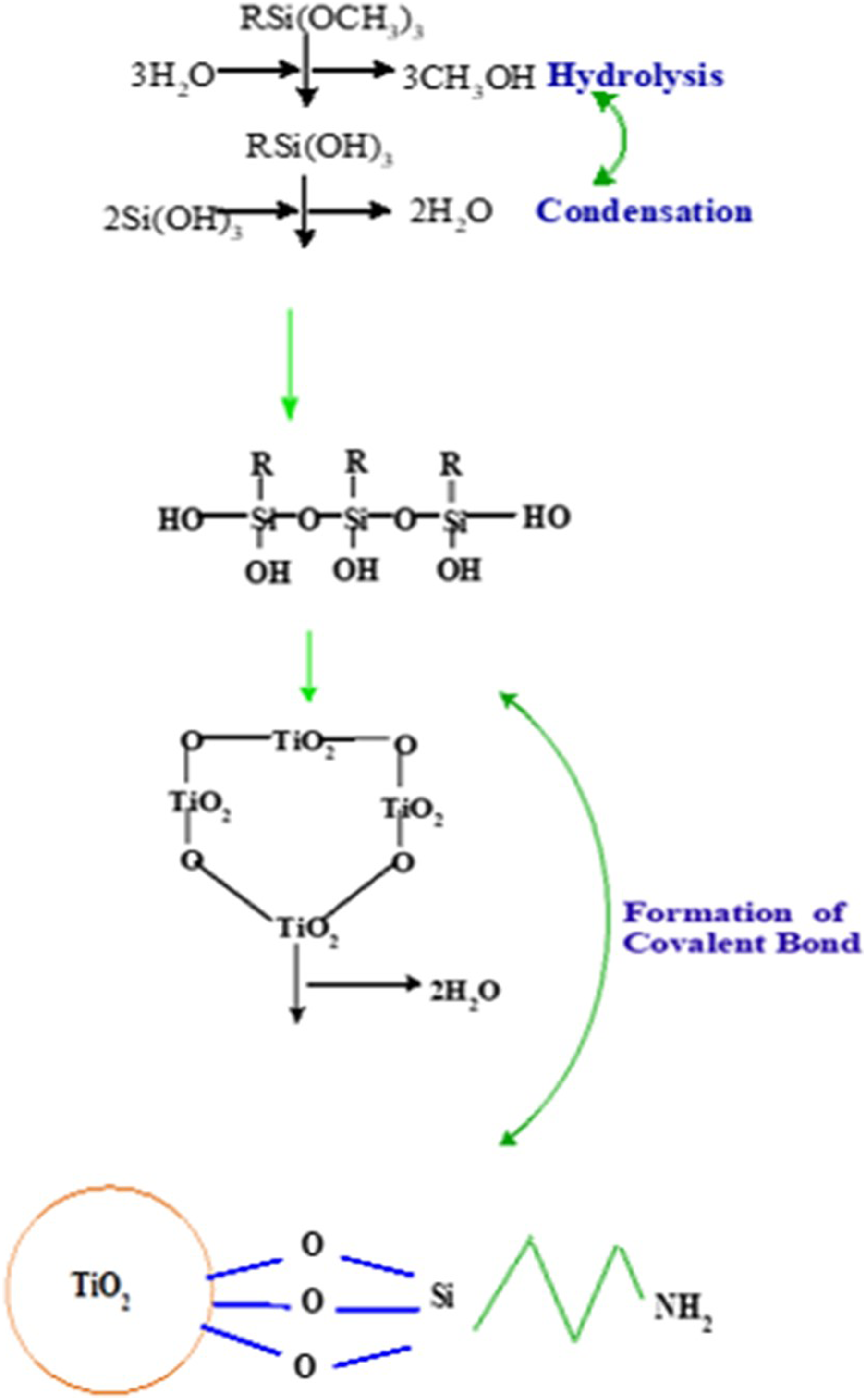
Preparation of specimens
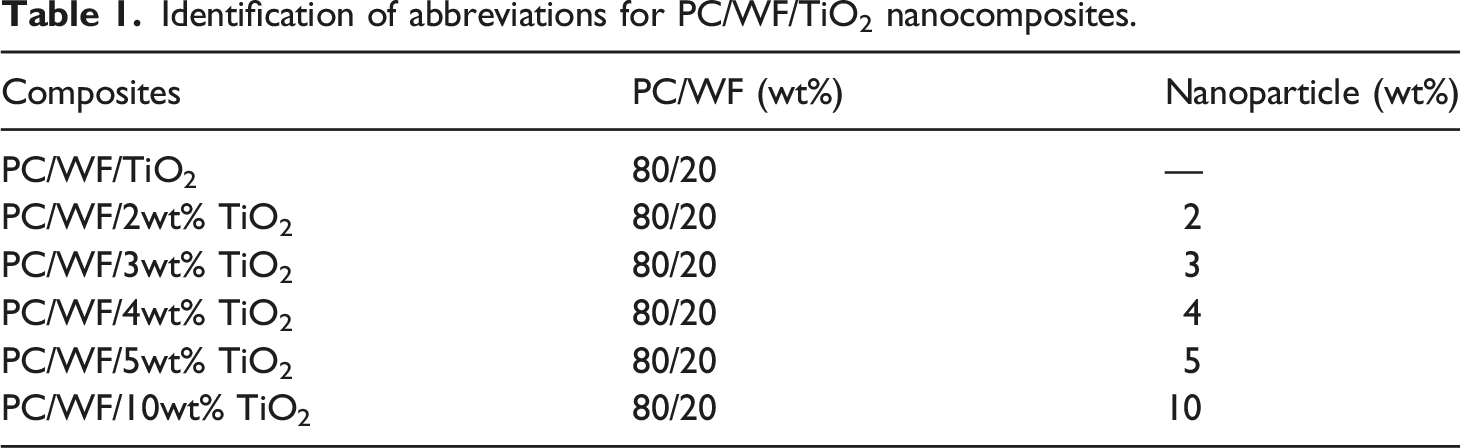
Identification of abbreviations for PC/WF/TiO2 nanocomposites.
Rheological measurement
Torque measurement
A Brookfield DV-III Ultra Programmable Rheometer was used for the rheological experiment, which measures the fluid parameters of torque, shear stress, and viscosity at the given shear rates required for polymer melts at three temperatures (230, 250, and 270°C), and five rotational speeds (100, 120, 140, and 180 rpm). PC/WF/TiO2 and six compositions of their nanocompsites in the ratios of 80/20, 80/20/2, 80/20/3, 80/20/4, 80/20/5, and 80/20/10 were measured. Polymer granules of various compositions were weighed to fill the chamber to 70% capacity. The weighted granules were premixed by tumbling and dried in an oven for 2 days at 80°C. The temperature and rotating speed of the rheometer were programmed using the rheometer’s microcontroller.
Data interpretation for torque and viscosity
The equations from (1)–(9) were obtained from the study by Laguna et al.
16
which was used to investigate torque data in terms of fundamental units. The torque 

where 



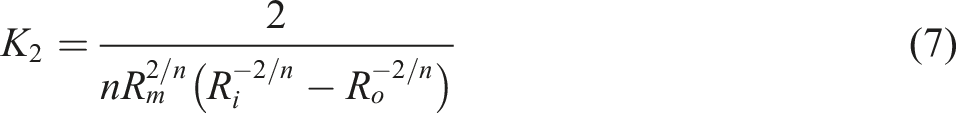
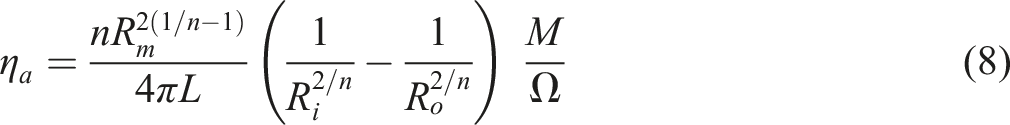

Characterization of nanocomposites
X-ray diffraction (XRD)
A Bruker D8 Advance with Cu-Kα radiation (λ = 0.15418 nm) was used to record the XRD of synthesized nanoparticles and their PC/WF/TiO2 nanocomposites. The applied current was 30 mA, and the accelerating voltage was 40 kV. In the step scanning mode, the XRD patterns were scan size of 0.02°/step at a rate of 147.4 s/step.
Thermal property analysis
Thermogravimetric analysis (TGA) was performed to demonstrate effect of TiO2 nanoparticles on thermal stability of a pure PC/WF. TGA provides measurable information about the weight-loss process. TGA was performed on a TGA Q-50, TA instruments. Approximately 10 mg of the samples were heated from ambient to 800°C at a heating rate of 20 °C/min in a nitrogen atmosphere introduced at a flow rate of 80 mL/min.
Mechanical properties measurement
Impact strength tests
The impact strength is measured according to GB1043-79 standard test. The machine used is CHARPY XCJ-40-. The following relationship was used to calculate the impact strength:
17

Hardness tests
Shore A hardness was determined using a ZWICK hardness machine, which is available at the National Company for Chemical and Plastic Industries. Tester type 7901 was used, and the test was performed in accordance with DIN 53505, ASTM (D1706-61), and ISO DR 988.
Tensile strength tests
Tensile properties were determined on a dumbbell-shaped die-cut sample using an Instron tensile tester machine (model 1445) in accordance with ASTM standards (D638). 18 Specimens were cut and their thickness measured using a micrometer with a resolution of 0.025 mm (0.001 inch).
Thermal conductivity measurement
The thermal conductivity of the PC/WF/TiO2 nanocomposite was measured using Lee’s disk technique, with the nanocomposites placed between two brass plates (plate a, plate b). The experiment’s heat source was a constant voltage and current of (6 V and 0.25 A), respectively. The following are the thermal conductivity equations (11)–(13)
19
:
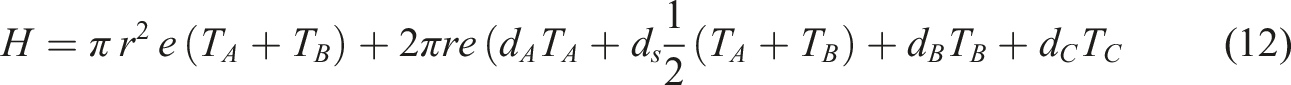
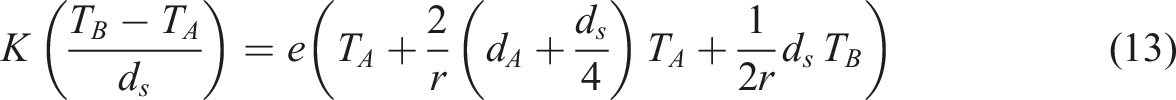
Electrical Measurement
Discharging a high voltage through a PC/WF composite linked with TiO2 was used to evaluate its electrical characteristics. At ambient conditions, DC electrical conductivity was measured using the usual four-probe technique. Four similarly spaced probes are placed on the material in this procedure. A current source produces an increasing current, 
Cone calorimeter
Cone calorimeter (CONE) tests were conducted in accordance with ASTME 1354-2009 standard. The specimen was covered with aluminum foil and positioned horizontally with an external heat flux of 50 kW.m-2. The specimen’s measurements were 100 × 100 × 3 mm-3.
Results and discussion
Shear stress versus shear rate plots
A log-log plot of Log-Log plots of shear stress versus shear rate for PC/WF/TiO2 nanocomposites at (a) 230°C, (b) 250°C, and (c) 270°C. The power law index versus percentage weight of TiO2 (2, 3, 4, and 10 wt%) at various temperatures (230, 250, and 270°C).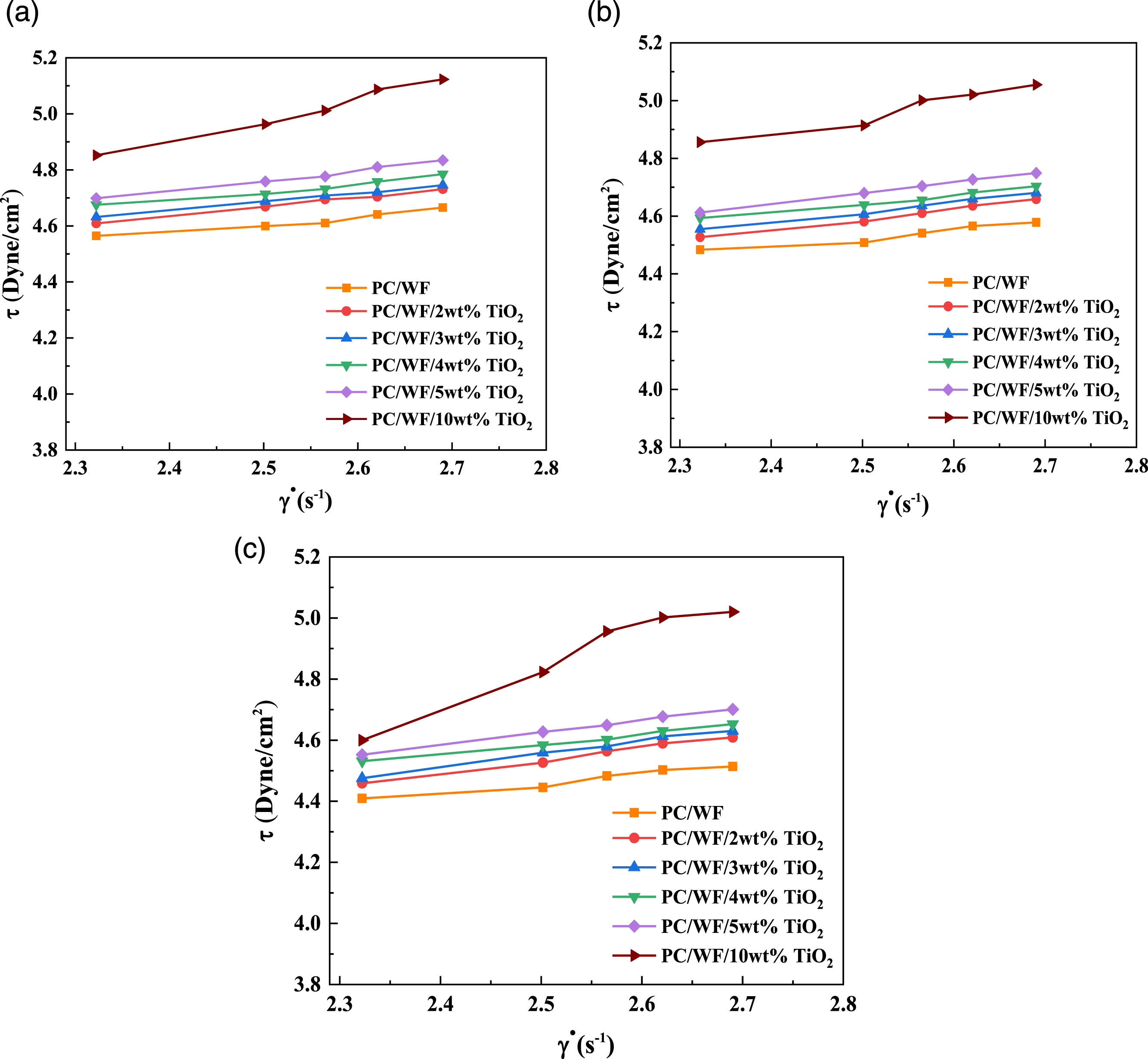
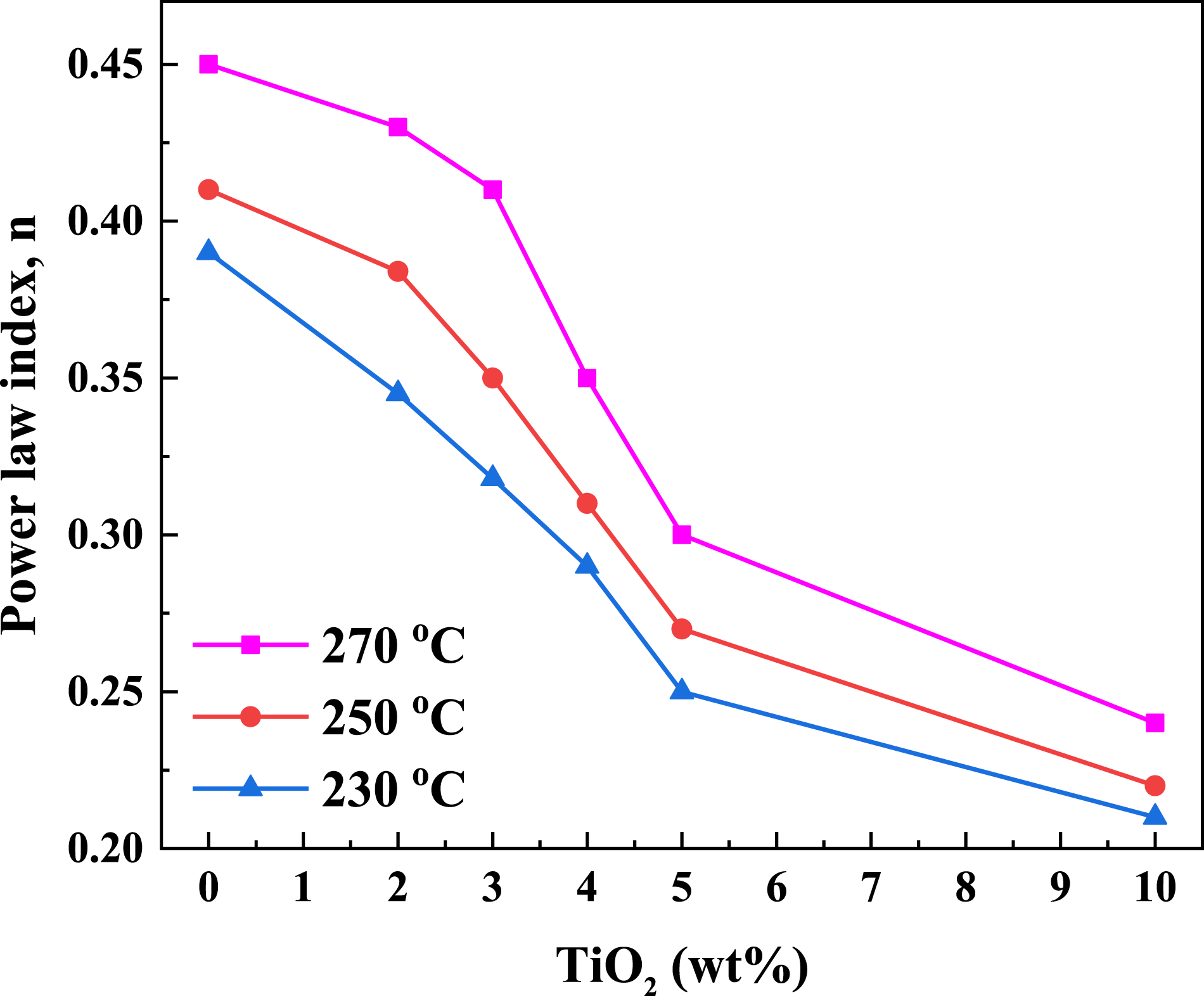
Apparent viscosity
Equation (7) was used to calculate the apparent viscosities of the mixes. The apparent viscosity rose quickly with increasing amounts of reinforcing agent, especially at low shear rate. This phenomenon can be described in terms of TiO2 nanoparticle interactions, which tend to attract the TiO2 nanoparticle into unfavorable to flow orientations, resulting in increased viscosity. At low shear rates, the composite viscosity appears to be driven primarily by the strength of the bonding in the network structure, which appears to be temperature independent. These findings are consistent with those reported by Parvaiz.
26
All synthetic nanocomposites reveal a shear thinning nature that increases proportionally with polymer concentration, a characteristic reflected by the decayed increase in measured shear stress forces with increasing shear rates,
27
as shown in Figure 5(a)–(c). The apparent viscosity of the PC/WF/TiO2 nanocomposites was found to be higher than that of PC/WF with increasing the content of TiO2 in the mix. Thus, when shear rate increases, molten state may be gradually sheared away, resulting in a decrease in intermolecular interactions and, as a result, a decrease in apparent viscosity.
28
The influence of temperature on nanocomposite viscosity at high shear rates may be shown as follows: when particle contacts are larger, alignment is greater, and viscosity is lower. These results are in good agreement with those reported by Abbas et al.29,30 As the temperature rises, the viscosity drops because viscous stresses prevail. Log-Log plots of apparent viscosity (ηa) vs. shear rate for PC/WF/TiO2 nanocomposites at (a) 230°C, (b) 250°C, and (c) 270°C.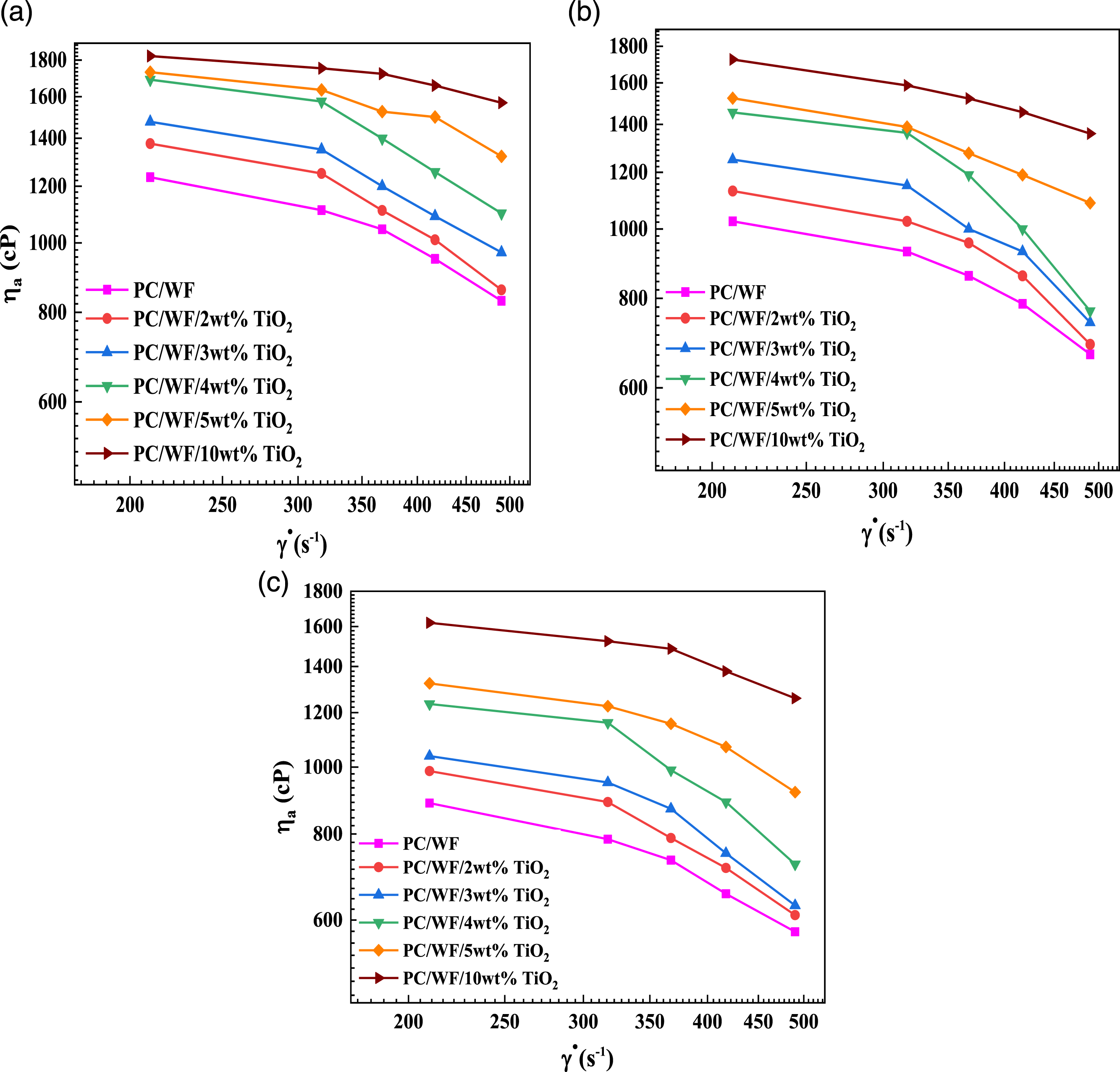
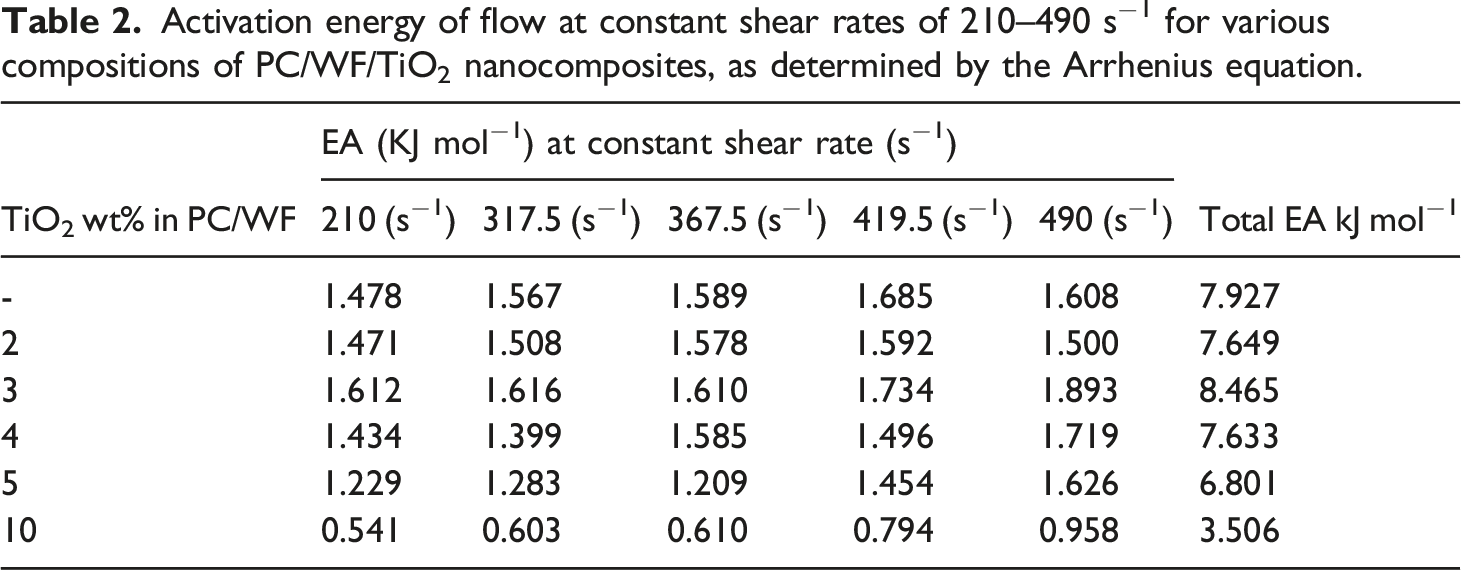
Activation energy
Activation energy of flow at constant shear rates of 210–490 s−1 for various compositions of PC/WF/TiO2 nanocomposites, as determined by the Arrhenius equation.
XRD characterization
As shown in Figure 6, bare PC exhibits a broad diffused diffraction peak at around 2 XRD patterns for PC, WF, TiO2, and PC/WF/TiO2 nanocomposites with various TiO2 content.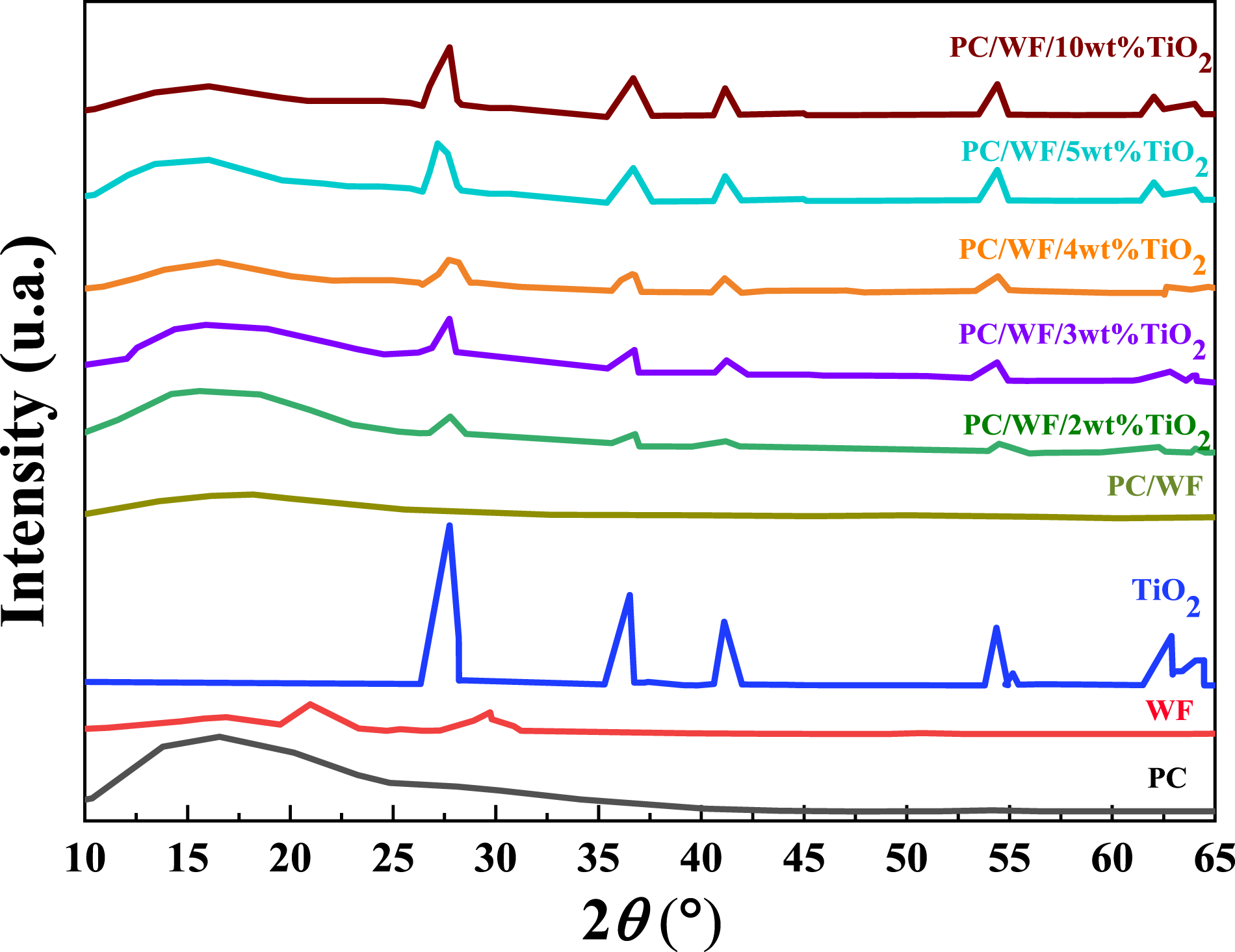
PC/WF/2%wt TiO2 > PC/WF/3%wt TiO2 > PC/WF/4%wt TiO2 > PC/WF/5%wt TiO2 > PC/WF/10%wt TiO2 > PC/WF (65.78, 63.21, 59.39, 57.56, 50.68, and 42.29). A shift to lower angles of the characteristic diffraction peak in the TiO2 doped PC/WF nanocomposites and suggests an increase in interlayer spacing of the TiO2, which is resulting in as intercalation. 38
Thermal property analysis
Thermogravimetric analysis (TGA) study was carried out to examine TiO2’s impact on flame retardancy. By triggering the changes in mass of a sample of material over time as a function of temperature and time, thermogravimetric analysis was used to study the thermal degradation process of polymers. Interfacial interaction plays a crucial role in the degradation of polymeric nanocomposites, i.e., an excellent interfacial interaction allows particles to act as restriction sites for the mobility of a polymer chain, makes the scission of a polymer chain harder at lower temperature, and therefore shifts the degradation temperature of the material to higher temperature. Nevertheless, better interfacial interaction between additives and polymer chain introduced by the deeper penetration of smaller particles in the polymer matrix will also restrict the mobility of the polymer chain. ‘‘Deeper penetration’’ means that a further interaction between nano-scale particles and polymer chain segments occurs, beyond the simple interaction between nano-particles and polymer chains. Figure 7(a) depicts the TGA curves for PC/WF and PC/WF/TiO2 nanocomposites (2%, 3%, 4%, 5%, and 10 wt%). As can be observed, the samples degrade in two stages. In addition to the evolution of H2O and CO2 over the entire mass loss range, alcohols, ethers, and carbonates are predominantly found at the first stage of PC thermal degradation, and the carbonates produced undergo rearrangements and eventually char during the second stage.
39
Some easily degradable wood elements, on the other hand, such as hemicellulose and extractives, deteriorated throughout the melting process.
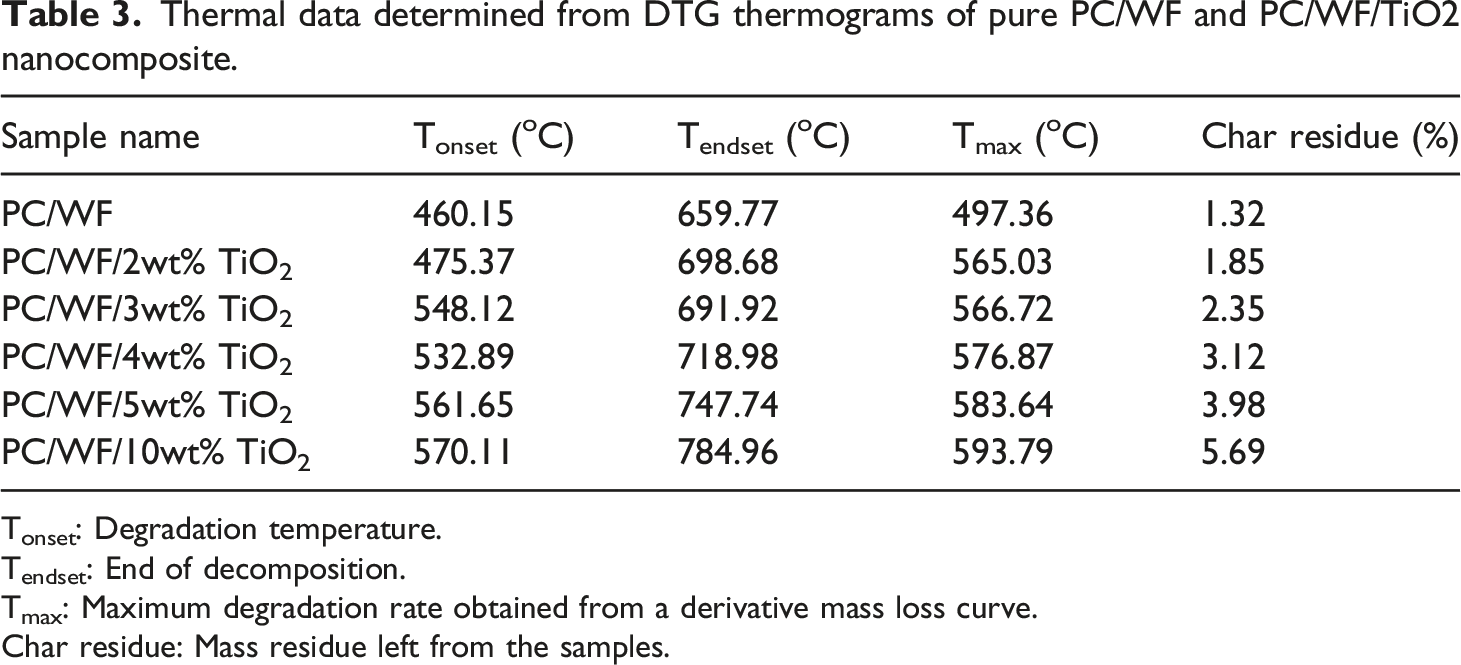
3
Table 3, thermal data obtained from TGA thermograms of the PC/WF and PC/WF/TiO2. TGA curves showed that nanocomposite have higher resistance to thermal degradation compared to PC/WF and it reveals to increase as the TiO2 loading increases. Moreover, variations in the DTG temperature as a function of TiO2 nanoparticle content are represented in Figure 7(b). The PC/WF composite starts to decompose at 460.15°C, with the process complete at 659.77°C, which is lower than the PC/WF/TiO2 nanocomposites (Figure 7(b)). Tonset and Tendset for the nanocomposites were both shifted to higher temperatures as compared to the PC/WF, which had a smaller char residue (1.32%). The higher adhesion force at the PP/WF/TiO2 interface may be responsible for the rise in onset and endset temperatures. Particles with high interfacial interactions can impede the mobility of polymer chains, making polymer chain scission harder at lower temperatures. As a result, the nanocomposite’s degradation temperature shifts to a higher temperature.
40
With the addition of TiO2 nanoparticles, the temperature at which maximum degradation rate occurs is raised as compared to PC/WF. For reinforced nanocomposites, residue amounts to 1.85%, 2.35%, 3.12%, 3.98%, and 5.69% for TiO2 loadings of 2, 3, 4, 5, and 10 wt%, respectively. This demonstrates that the samples treated with a flame retardant (TiO2) created noticeably more solid residues, the char’s surface was more compact. Heat is successfully kept from penetrating the substrates. This observation is consistent with the literature.
41
Thermal analysis of pure PC/WF with and without TiO2 (a) TGA and (b) DTG. Thermal data determined from DTG thermograms of pure PC/WF and PC/WF/TiO2 nanocomposite. Tonset: Degradation temperature. Tendset: End of decomposition. Tmax: Maximum degradation rate obtained from a derivative mass loss curve. Char residue: Mass residue left from the samples.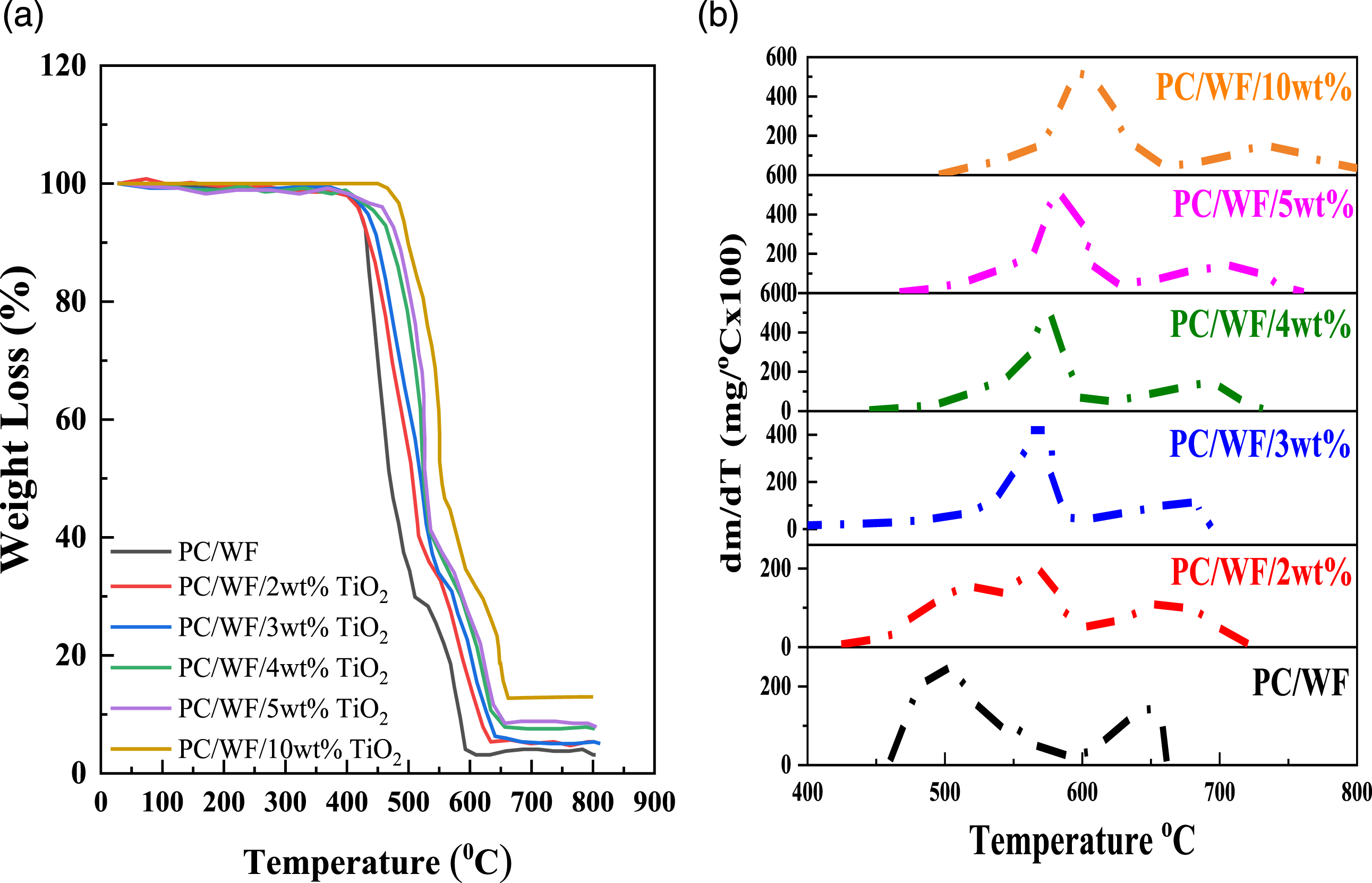
Mechanical properties
A functional composite must have a certain mechanical strength across a wide range of applications. The impact strength, tensile strength, and elongation at break of the PC/WF/TiO2 nanocomposites initially increased and then decreased with increasing TiO2 fraction. The nanocomposites generally performed well when the TiO2 content was set at 3 wt%, as this optimal amount of TiO2 had a toughening effect on the PC/WF and optimal interfacial compatibility between the TiO2 nanoparticle and the matrix, as shown in Figure 8. The TiO2 content was found to affect the change in impact strengths of the PC/WF composites. It can also be seen that the TiO2 fraction and its state of dispersion had a significant impact on the toughness of the materials. A smaller amount of TiO2 filling improved the hybrid materials' impact strength, but as its fractional weight increases the impact strength decreases significantly, as shown in Figure 8(a). This phenomenon can be explained by changes in TiO2 dispersion; finer dispersion reduces impact strength to a lesser extent.
42
Figure 8(b) also shows the utilization of PC/WF doped with TiO2 nanoparticles. With increasing TiO2 content rather than 3wt%, the harnesses of PC/WF/TiO2 nanocomposites decreased as shown in Figure 8(b). This decrease indicates that as TiO2 loading increases, the nanocomposite become brittle due to the stress concentration effect of TiO2. Hardness decreases with increasing TiO2 content due to the formation of cracks around the TiO2 nanoparticles and the potential formation of particle agglomerates and voids, resulting in local matrix detachment from the particles.
43
As shown in Figure 8(c), the tensile strength of PC/WF composites increase with increasing TiO2 content. This is due to mechanical anchorage between the TiO2 and the PC/WF. As a result, interfacial adhesion is critical in increasing the tensile strength of composites. The greater the composite’s interfacial adhesion, the greater the stress that can be transferred to inorganic particles from the matrix, resulting in higher tensile strength.
44
Tensile strength is reduced when the nanoparticle level exceeds 4% by weight, at which point larger agglomerates of nanoparticles can be formed as displayed in Figure 8(c). The decreased tensile characteristics of nanoparticle-reinforced PC/WF could be attributed to the decreasing surface area of the higher particle loading (larger agglomerates) through a less efficient stress transfer mechanism.45,46 Figure 8(d) also shows that the elongation required to break the nanocomposites has a strong relationship with the interfacial properties of the PC/WF. The elongation to break the coupled PC/WF-treated, particle-reinforced nanocomposites increased dramatically when compared to the neat matrix. A reduction in elongation to break nanocomposites with more than 3wt% TiO2 is to be expected due to the development of cracks around the TiO2 nanoparticles and the possible formation of particle agglomerates and voids, resulting in local matrix detachment from the particles.
43
Mechanical variations of: (a) impact strength, (b) hardness (Shore A), (c) tensile strength, and (d) elongation at the break of the pure PC/WF as a function of the TiO2 content in wt%.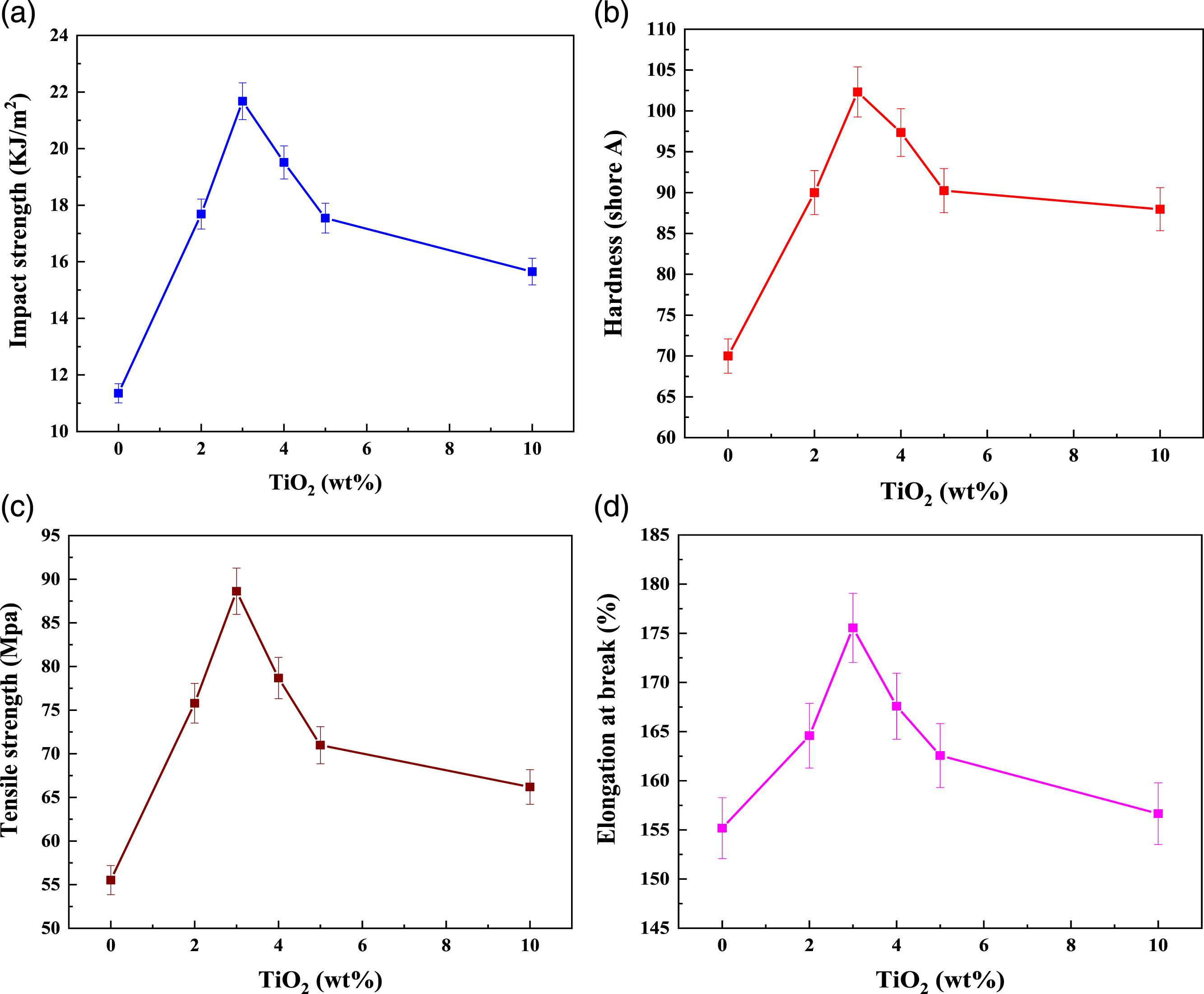
Based on the associated mechanical properties PC/WF/TiO2 nanocomposites, a TiO2 concentration of 3wt% was found to be optimal in PC/WF composites resulting in limited the polymer chains thereby strengthen the interface between TiO2 and PC/WF. This is confirmed by activation energy results (refer Table 2).
Regression analysis of impact strength and hardness.
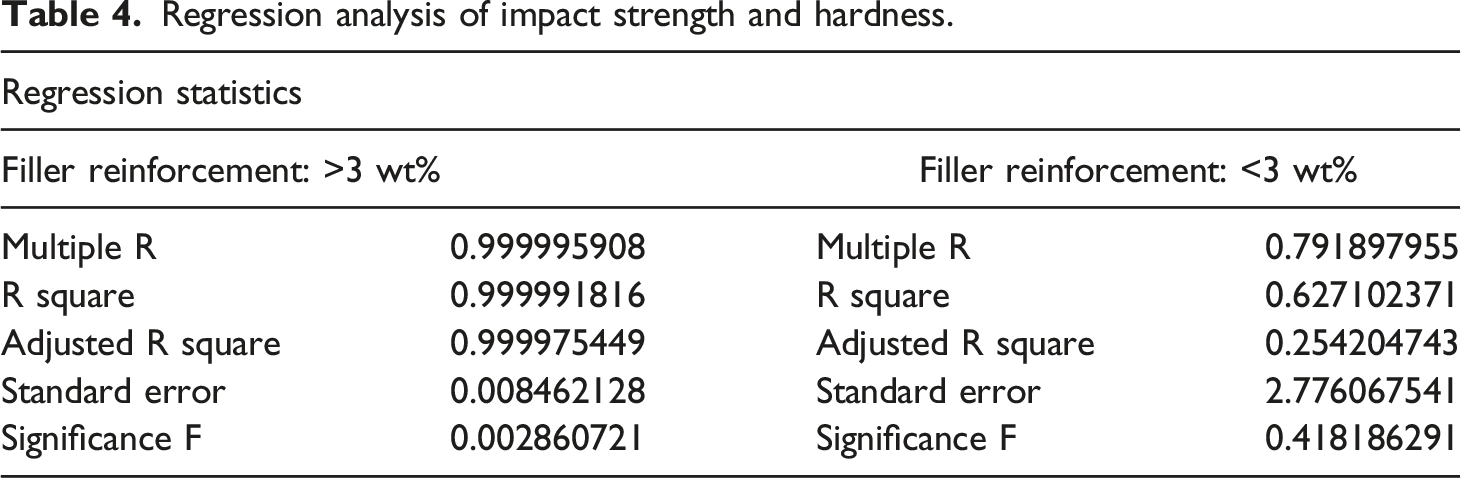
Regression analysis of impact strength and tensile strength.
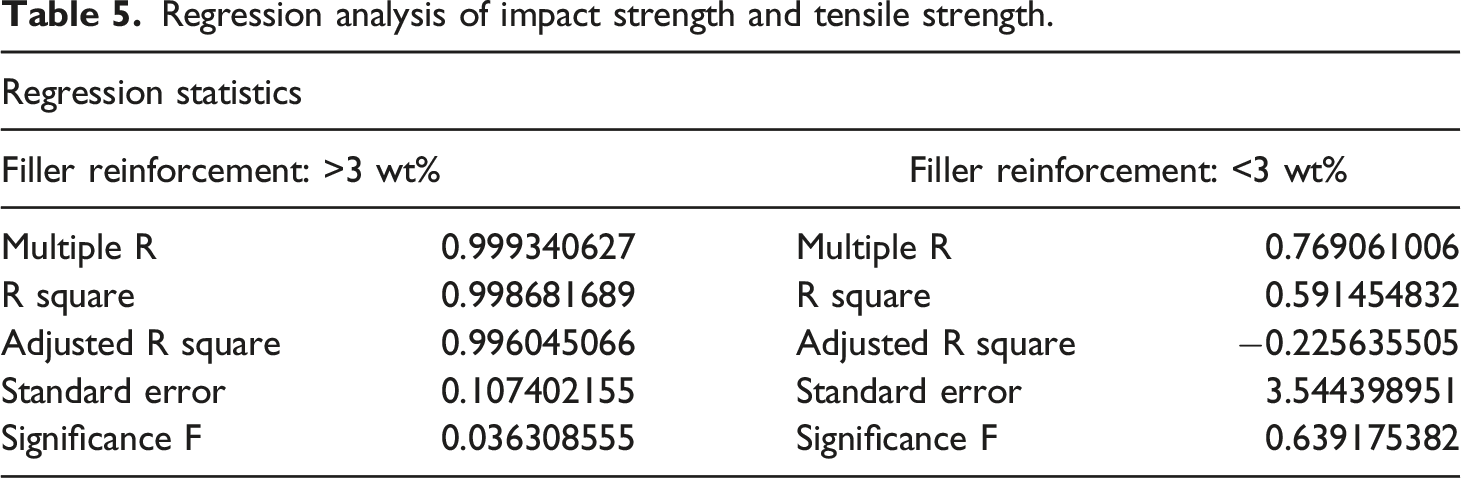
Regression analysis of impact strength and elongation at break.
The quantification of the interfacial interaction of PC/WF/TiO2 nanocomposites
The interfacial adhesion between the TiO2 nanoparticles and the composite has a significant impact on the mechanical properties of the composites, particularly the tensile strength. Pukanszky et al.
50
created a model to predict the variation in the yield stress of a composite with the particulate TiO2 nanoparticle fraction using equations (15)–(17).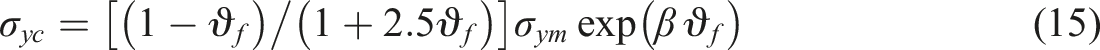

parameter Ln(φ) versus weight fraction curves of PC/WF as a function of TiO2 content in wt%.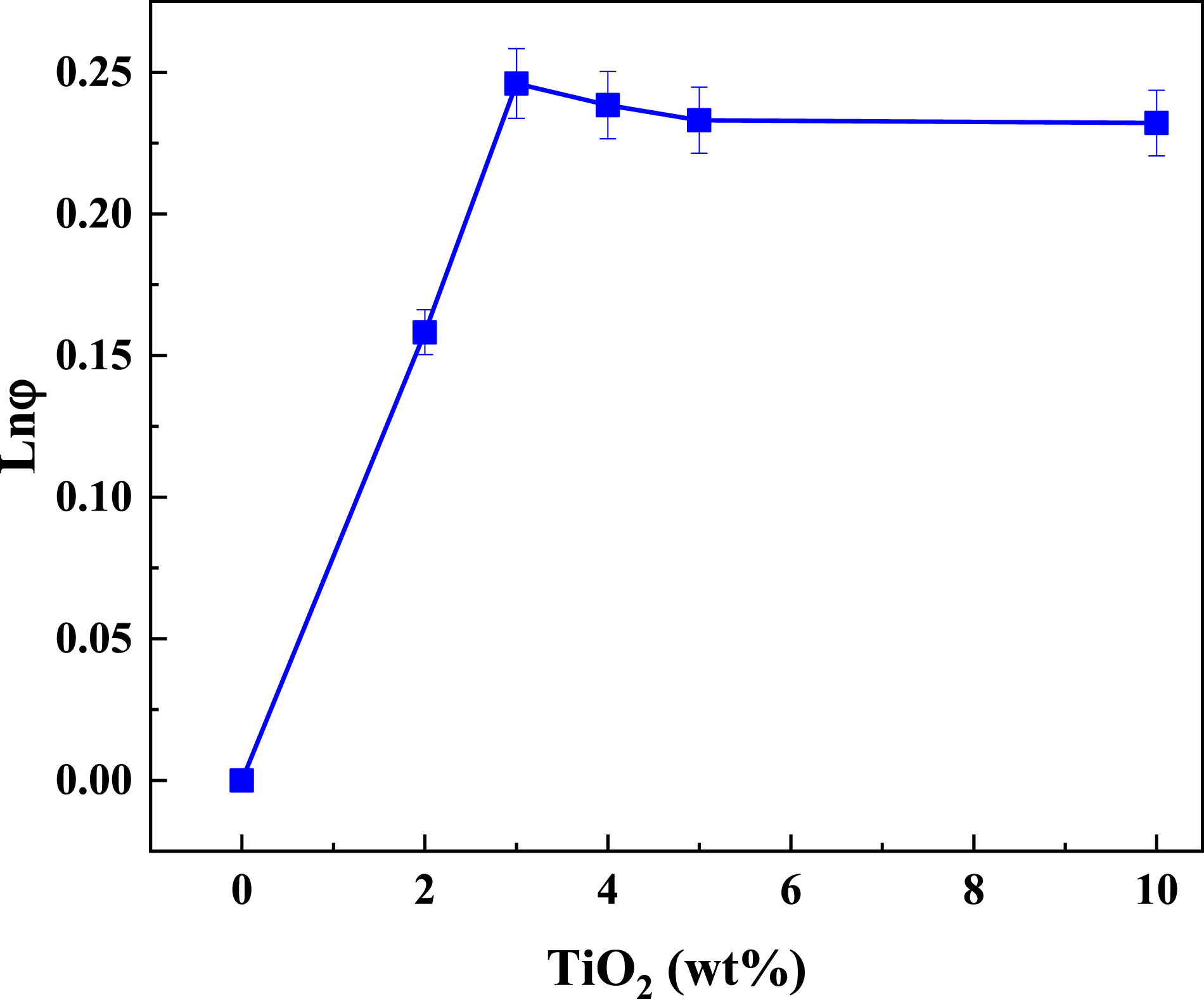
Regression analysis of quantity and impact Strength.

Regression analysis of quantity and hardness (Shore).

Relationship quantity and tensile strength.

Relationship quantity and elongation at break.

Thermal conductivity
Figure 10 depicts thermal conductivities of PC/WF/TiO2 have been determined at various weight concentrations of TiO2. The value of thermal conductivity increases monotonically with increasing weight fraction of TiO2 because the addition of the molecular TiO2 nanoparticle can remarkably improve the adhesion of two-phase interfaces When a thermally insulating polymer like polycarbonate and wood flour are reinforced with any conductive reinforcing agent (having conductivity higher than that of PC/WF) like TiO2, the thermal conductivity of the nanocomposite starts enhancing and at higher TiO2 concentrations, the nanocomposite becomes conductive in nature. Initially, at low TiO2 concentration, since the number of the conductive particles is insufficient to form a continuous conducting path at less than 3wt%, the conductive domains are insulated from each other by the PC/WF medium and the heat conducting behavior cannot be observed. The TiO2 nanoparticles are isolated by the PC/WF in the case of low TiO2 content. As the TiO2 concentration increases, in reinforced PC/WF composites, TiO2 nanoparticles gradually get connected and conduction network are formed at more than 3wt% of TiO2. The precise location of the percolation threshold (at 3wt% of TiO2) is impacted by various factors including the size, aspect ratio and size/spatial distributions of the conductive particles. Immediately after the percolation threshold, a slight increase in the concentration of conductive TiO2 nanoparticles is found to greatly increase the bridges in the conducting network leading to substantial improvement in thermal conductivity of the composites Further increase (5–10 wt%) in the concentration of the TiO2 nanoparticles, however, only causes the volume of the conducting domains to increase without any significant increase in the pathways for heat, leading to a monotonic increase in conductivity. These results are in good agreement with previous reported literatures.54,55 Thermal conductivity of PC/WF as a function of TiO2 content in wt%.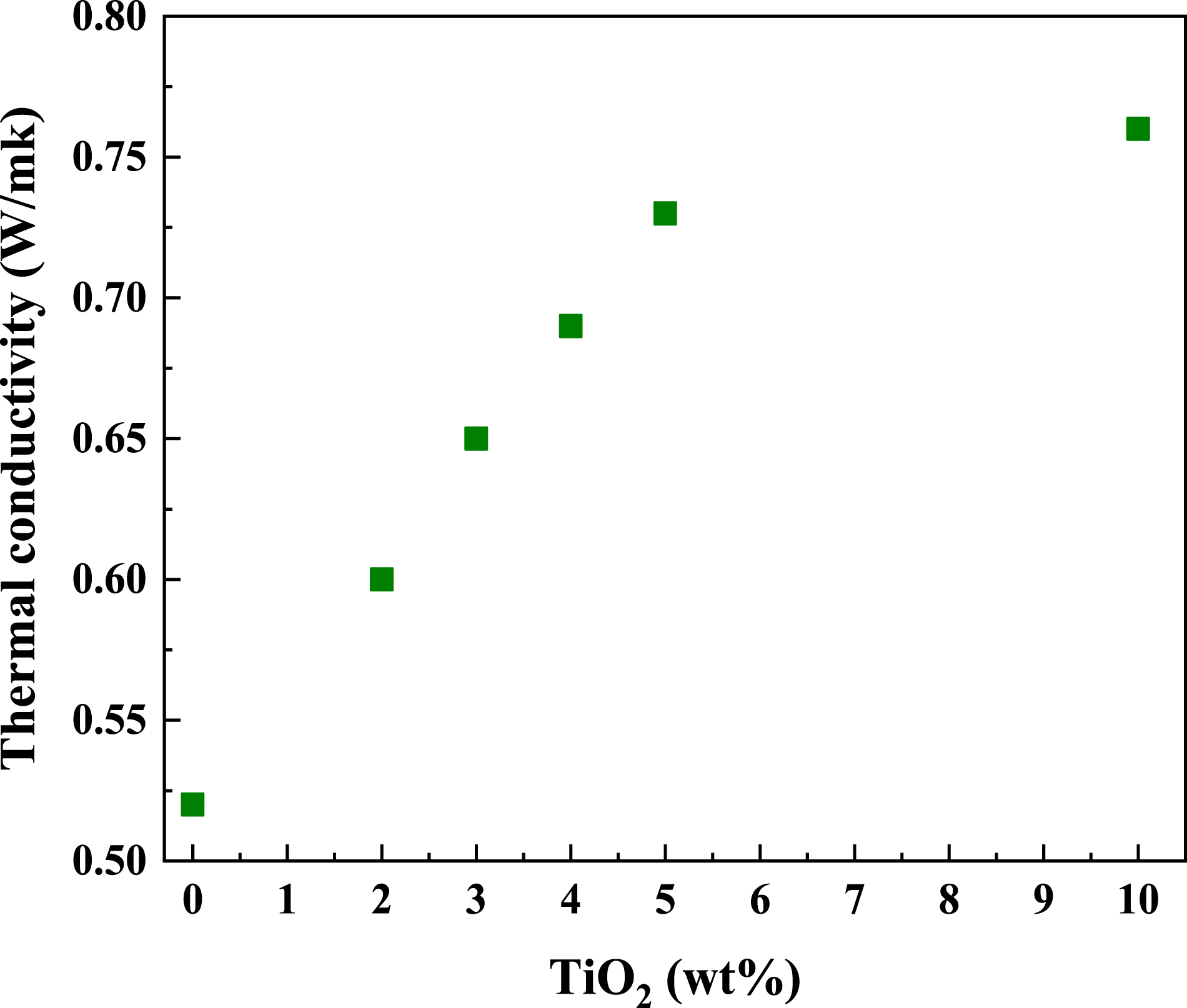
Electrical resistivity
Figure 11 illustrates the electrical resistivity of PC/WF with varying TiO2 concentrations. The electrical percolation threshold is defined as the weight fraction of the TiO2 nanoparticle at which the measurements begin to show percolation behavior. The amount of filler in the polymer alters the current flow mechanism. A current flow is not achievable with a very low filler concentration (I) because the gap between two TiO2 particles is too large to allow for quantum tunneling. The gap narrows as the filler content (II) rises, allowing quantum tunneling to occur. When the filler amount is increased even further (III), the filler particles make direct electrical contact.
56
The TiO2 aggregates in the PC/WF with a low TiO2 weight fraction (e.g., 3 wt%) are relatively more separated as the conductive TiO2 nanoparticle weight fraction approaches the percolation threshold (3 wt% TiO2 content). Because the distances between the TiO2 nanoparticles in the PC/WF are relatively large, little, or only slight changes in electrical resistivity can be observed at lower TiO2 loadings. Increasing the TiO2 content causes TiO2 to become more crowded, resulting in a gradual decrease in electrical resistivity. The electrical resistivity decreases dramatically near the percolation threshold, and the material transitions from being insulating to conductive. This indicates that TiO2 nanoparticles collide or are close enough to allow electron hopping via tunneling, resulting in continuous conductive paths or networks. Because the conductive network has already been established, additional TiO2 loading does not significantly reduce volume resistivity once the percolation threshold is reached. A further increase in the volume fraction of TiO2 powder (>4 wt%) narrows the gap slightly, resulting in only a few more conductive channels. As a result, the decrease in resistivity occurs much more gradually. These findings are consistent with those obtained in Lan.
57
Electrical resistivity of PC/WF as functions of TiO2 content in wt%.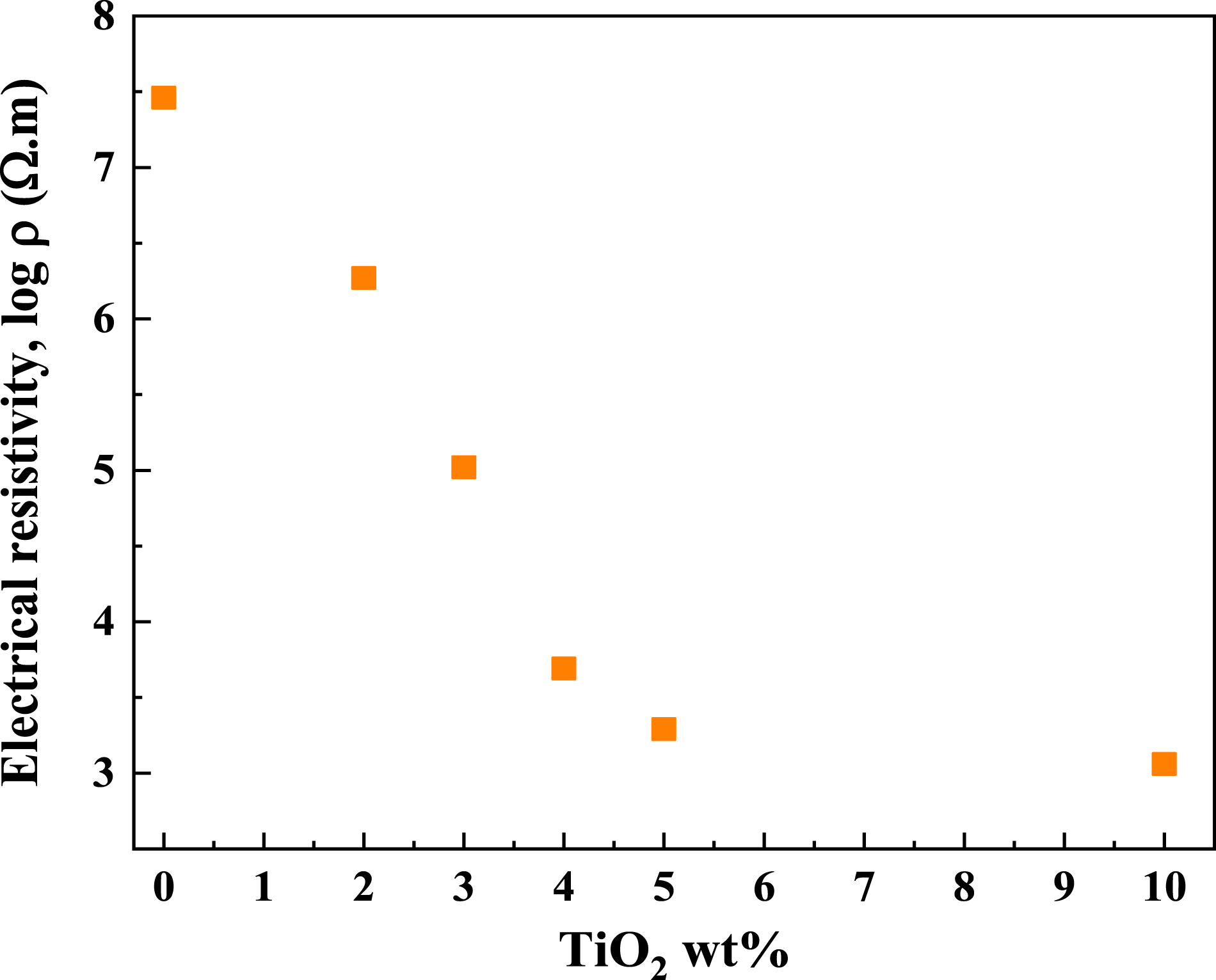
Cone calorimeter analysis
To assess the qualities of materials for combustion, the cone calorimeter, based on the oxygen consumption concept, was frequently utilized. Cone calorimeter experiments were used to examine the flammability properties of PC/WF as functions of TiO2 content (3 and 10 wt%) nanocomposites with and without boric acid (BA). Figure 12 displays the heat release rate (HRR) and the total heat release rate (THR). Boric acid and wood mix to form a novel chemical structure that modifies the way that wood deteriorates under heat.
58
Boric acid makes it easier for cellulose and hemicellulose to be thermally decomposed into biochar rather than flammable gas. In other words, a boric acid treatment caused more char to remain after burning wood. The layer of carbon that has developed isolates oxygen and heat, slowed the combustion of the composite and reduced its HRR. However, the char layer resulting from wood flour suppressed the HRR of the polymer resin in composites (Figure 12(a)). Similar trends were observed in THR (Figure 12(b)). In comparison to PC, wood flour releases less heat since it has more oxygen and aromatic chemical groups. These results demonstrate that boric acid may reduce the HRR and THR of wood while burning while increasing the flame retardancy of wood flour, along with those observed in the literature.
59
Flame–retardant properties of PC/WF as functions of TiO2 content (3% and 10% wt%) nanocomposites with and without boric acid (BA). (a) Heat release rate (HRR), (b) total heat release THR.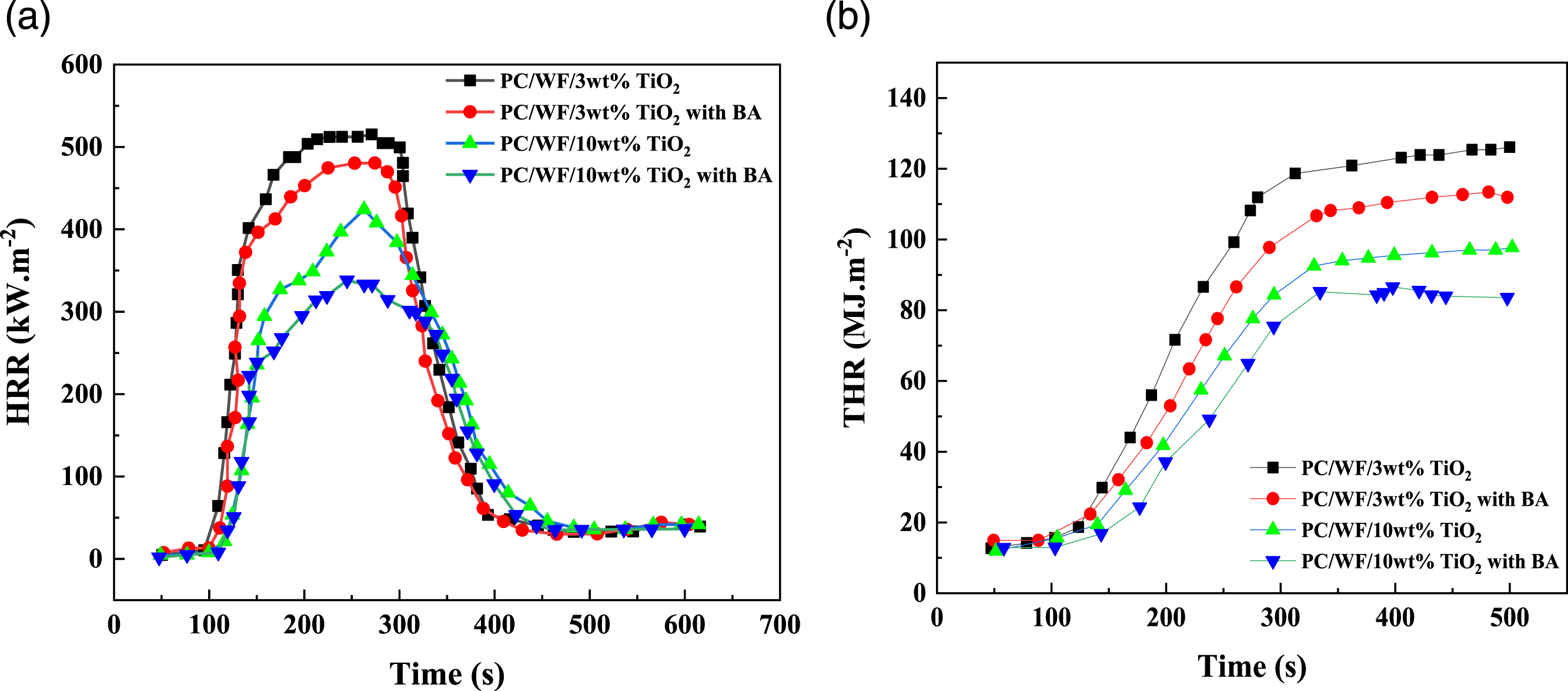
Conclusion
In this study, the thermal stability of wood flour was first enhanced using boric acid treatment and the treated wood flour was then filled into PC and then reinforcement with TiO2 to prepare PC/WF/TiO2 nanocomposites with various rutile TiO2 contents. Meanwhile, their various properties characterized and discussed. The main conclusions obtained are as follows: 1. The addition of TiO2 nanoparticle to PC/WF improves the rheology processability. As TiO2 concentration rises, it is observed that perceived viscosity also rises. As a result of the addition of TiO2 nanoparticles to the PC/WF composite, the viscosity of composites has increased. Besides, the viscosity of nanocomposites decreases as temperature rises. 2. The energy barrier inhibiting the migration of the polymer chain from one place to another is related to the activation energy. As a result, the observed greater activation energy of the composites at 3wt% of TiO2 indicates that the interfacial adhesion was also enhanced in addition to the polymer and reinforcement agent’s increased dispersion. 3. XRD investigation showed that by embedding TiO2 nanoparticles into PC/WF, the amorphous phase decreased, and the degree of crystallinity increased compared to bare PC/WF from 65.78 to 42.29 (PC/WF/TiO2) nanocomposites. Whereas incredibly uniform TiO2 dispersion can provide stronger interactions in the PC/WF matrix, causing incredibly rapid nucleation and development. 4. The DTG and TGA curves showed that nanocomposites have a higher resistance to thermal degradation than bare PC/WF. The onset, endset, and Tmax temperatures of the PC/WF composite thermal degradation were all enhanced by the TiO2 concentration, however. Consequently, the TiO2 concentration in metal hydroxide hybridized WPC could be more efficient in enhancing thermal stability against high temperature degradation. It was found the char residue resulted from TGA of PC/WF/10%wt of TiO2 increased to 5.69% than bare PC/WF (1.32%). 5. TiO2 nanoparticle addition to PC/WF was studied to see how its effects on mechanical characteristics. Greater impact properties, elongation at break, and tensile strengths are seen in the TiO2-doped PC/WF composites. The incorporation of 3wt% of TiO2 nanoparticles into PC/WF composite exhibited to excellent performance in term mechanical characteristics of this mixture. 6. The thermal conductivity of composites reinforced with TiO2 is greater than that of PC/WF because of the high interfacial thermal resistance between the reinforcement agent and the composite. 7. The electrical conductivity decreases at lower concentration of TiO2 (<3 wt%) due to the hindrance of ion mobility. The increased conductivity associated with higher concentrations of TiO2 indicates the formation of more void space at the interface due to the formation of TiO2 nanoclusters. 8. The thermal stability of nanocomposite materials is increased by boric acid’s promotion of char formation from wood flour. Results obtained show that boric acid may decrease the HRR and THR of burning wood while enhancing the flame retardancy of wood flour.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
All data generated or analyzed during this study are included in this published article’ in the main manuscript
