Abstract
Biological elastomer i.e., medium-chain-length poly-3-hydroxyalkanoate (mcl-PHA) and its graft copolymer were investigated for reactive coating application. They were composited with silver-doped zinc oxide (Ag-ZnO) to produce films with photocatalytic activities. The activities were assessed from the photodegradation efficiencies of methylene blue, Rhodamine B and Orange II dyes. The PHA composite films demonstrated sustained photocatalytic activities over repeated usage compared to neat PHA. The presence of Ag also inhibits microbial growth on the film surface. The composite film exhibited negligible toxicity effect when tested against Ceriodaphnia dubia. Potential and advantages of Ag-ZnO−PHA composite film in reactive coating applications are discussed.
Introduction
A surface can be coated with polymeric material to fulfil functional or aesthetic requirements. Delicate adherence, protection, and imparting desirable qualities to the underlying surface may be realized via polymeric coatings. 1 Coating a metal surface with polymer materials often improve its resistance to heat and corrosion. For example, acrylics found widespread applications as low-cost corrosion protection coatings for aircraft, road-building machinery, automotive components etc.2,3 Bio-based polyurethane exhibits potential as an ecofriendly protective coating against abrasion, fire etc.4,5 Designing and creating organic coating with intrinsic conductivity, such as polyaniline, to prevent corrosion has been demonstrated.6,7
Coating technology embedded with photocatalyst is being adopted in many applications to provide a self-cleaning property e.g., building construction, textile industry, solar panel and many more. 8 The attraction of photocatalytic self-clean technology is that it greatly reduces the fouling layer adhesion, extends the cleaning cycle intervals, and minimizes the consumption of polluting detergents.8–10 Organic dirt and grimes can be easily rinsed off the material surface after decomposition by the photocatalysts, customarily semiconductor materials such as titanium dioxide (TiO2), zinc oxide (ZnO), zirconium dioxide (ZrO2) among others.11–13 Along with the semiconductors, the presence of oxygen and water molecules, UV component in light (<390 nm) and its intensity are essential for the photocatalysis-mediated decomposition. 14 Studies have reported that doping of semiconductors with noble metal such as silver (Ag) boosts the photocatalytic activity by augmenting the receptiveness of the semiconductors towards visible light.15,16
Thermoplastic composites such as ZnO−polyamide-imide, 17 TiO2−polystyrene, 18 Ag-BaTiO3−polyvinylidene fluoride, 19 TiO2−polyvinylchloride 20 showed good piezoelectricity and photocatalytic properties as well as excellence in thermal, chemical and mechanical stability, attributes that are useful in functional coating application. Biopolymers such as galactomannan, chitosan and polylactic acid, enhanced with photocatalysts (e.g., graphene oxide, TiO2 and ZnO), are a few examples of green composites that have been investigated for their potential in the coating technology.21–23 On the other hand, the use of microbial-produced medium-chain-length poly-3-hydroxyalkanoate (mcl-PHA) film decorated with silver-doped zinc oxide (Ag-ZnO) for coating application is yet to be reported, thus the motivation of the current investigation. The amorphous thermoplastic mcl-PHA has two glass transition at −50 and 15°C, affected by the polymer’s main chain and side-chain, respectively. 24 The photocatalytic activities of Ag-ZnO−PHA film composites were studied by determining the extent and rate of organic dyes degradation in aqueous solution. Furthermore, its performance in extended use and ecofriendly potential were also studied.
Methodology
Synthesis of mcl-PHA and its graft-copolymer
Mcl-PHA was produced through microbial cultivation of Pseudomonas putida Bet001 with lauric acid as the sole carbon source. The details of its biosynthesis, extraction and purification were reported in previous studies. 25 The neat mcl-PHA was copolymerized with poly-methyl acrylate (PMA) via grafting through technique using ultrasonication. 26 To this end, solution mixture of mcl-PHA, methyl acrylate (MA) and hydrogen peroxide (H2O2) in acetone was subjected to sonication (30% amplitude, 20 kHz) for 2 min. The ultrasonicator (VCX500, 500 W, 20 kHz, Sonics, USA) is equipped with metal tapered microtip (3 mm diameter) immersed into the mixture solution at about 1-cm depth from the liquid surface. The amount of mcl-PHA, H2O2 and acetone were fixed at 0.15 g, 20 μL and 5 mL, respectively, while MA amount was varied between 0.06 and 0.12 M. The synthesized graft copolymer (PHA-g-PMA) was precipitated with methanol followed by centrifugation (3500 r/min) in the recovery process. The supernatant containing residual MA and H2O2 was decanted to obtain purified samples designated as PHA-g-PMA[0.06] and PHA-g-PMA[0.12].
Synthesis of silver-doped zinc oxide (Ag-ZnO)
Similar method was applied to synthesis Ag-ZnO microparticles. 27 ZnO (1 g), AgNO3 (1.698 and 3.397 g) and hexamethylenetetramine (HMTA) (1.402 and 2.804 g, respectively) were continuously stirred together in 200 mL of deionized water at 100°C for 6 h. They were purchased from Sigma-Aldrich. Then, the mixture solution was centrifuged at 3000 r/min for 10 min to separate the solid particles. Light grey precipitate obtained was washed thoroughly in distilled water to remove residual unreacted chemicals before it underwent calcination at 300°C for 2 h. The purified Ag[0.05]-ZnO and Ag[0.10]-ZnO microparticles were obtained from 1.698 and 3.397 g of AgNO3, respectively.
Preparation of Ag-ZnO−PHA composite film
Polymer solutions (0.5 g L−1) were casted onto glass surface, followed by oven-drying at 50°C, and air-drying at room temperature for 1 h and overnight, respectively. Transparent polymer films with area and thickness of 4 cm2 and 0.2 mm, respectively, were acquired. Then, 0.3 mL of photocatalyst in methanol, each containing 0.05 mg of ZnO, Ag[0.05]-ZnO and Ag[0.10]-ZnO were applied dropwise onto separate polymer films. The solutions were allowed to air-dried at room temperature overnight prior to experiments. All preparations of polymer composite films exhibited transparent appearance (shown in Figure S1 in the Supporting Information).
Photocatalytic activities of Ag-ZnO−PHA composite films
The composite film was placed on a horizontal platform immersed within a beaker containing 100 mL of 2.5 mg L−1 methylene blue (MB) solution to represent an organic pollutant. The solution was continuously stirred while allowed to equilibrate for 15 min in the dark. The photodegradation of MB was commenced under UV light at 254 nm using UV lamp (model UVGL-58) at a power rating of 6 W, and intensity of ∼850 μW/cm−2 for 100 min. At ten minutes interval, 4 mL of the dye solution was sampled for UV-Vis measurement at 664 nm, and subsequently returned into the beaker. UV light was switched off during sampling and measurement. Experiments were conducted in triplicate. The dye photodegradation efficiency (%), and time-evolution of the degradation process (Ct) were determined based on the formula used in a previous study. 28 The investigation of photocatalytic activity was repeated using two other dyes, i.e., Rhodamine B (RB) and Orange II (OII). Similar to MB, the initial concentration of the RB and OII solutions were fixed at 2.5 mg L−1. They were measured using UV-Vis spectrophotometer 551 and 486 nm, respectively.
Anti-bacterial and toxicity studies
Agar plate cultures of Escherichia coli, Bacillus cereus and Bacillus subtilis were used in testing anti-bacterial property of Ag-ZnO−PHA composite films. To this end, sterile cotton buds were dip into the bacterial suspensions (100 ppm dilution of 0.01 g L−1), and smeared onto pristine agar plates. Then, Ag-ZnO−PHA composite film (1 cm2) was placed onto the agar surface, with the decorated surface facing downwards. The culture plates were then incubated for 7 days at 35°C.
Ceriodaphnia dubia was employed to assess possible toxicity of Ag-ZnO−PHA composite films. The toxicity kit (Ceriodaphtoxkit F) was purchased from Microbiotest, Belgium. Standard freshwater (1 L) was prepared by mixing the provided CaSO4, MgSO4 and KCl solutions. The freshwater was used as a medium for hatching the water flea, and as toxicant diluent on the tested materials. To this end, Ag-ZnO−PHA film composites were immersed in 5 mL of standard freshwater in a parafilm-covered small beaker, and stored for 5 days at room temperature in the dark. The beakers were agitated slowly on a rotary flask shaker. Then, the freshwater samples were collected for the toxicity testing. Experiments were conducted in triplicate. Fresh water preparation, egg hatching and toxicity testing were carried out according to instruction provided in the kit standard operating procedure.
Results and Discussion
Photocatalytic activities of Ag-ZnO−PHA composite films
Figure 1 shows the spectra of MB absorbance measured regularly during the photodegradation experiment using different polymer composites i.e., neat PHA, PHA-g-PMA [0.06] and PHA-g-PMA [0.12] decorated with different photocatalysts (1a, 1b and 1c, respectively). The reduction in MB absorbance spectra is attributed to the photocatalytic activities of the polymer composites. All PHA-based composites showed consistent MB reduction indicating definite photocatalytic activities. The degradation of the dye compound was primarily due to the presence of photocatalysts on the surface of the polymer films. The neat PHA and its graft copolymers (PHA-g-PMA) are not part of the photocatalytic activities, which had been confirmed in a previous study.
28
The time evolution of MB absorbance spectra for different polymer composite films irradiated with UV light. (a) neat PHA, (b) PHA-g-PMA[0.06] and (c) PHA-g-PMA [0.12] were decorated with 0.05 mg of different photocatalysts viz. ZnO, Ag[0.05]-ZnO and Ag[0.10]-ZnO.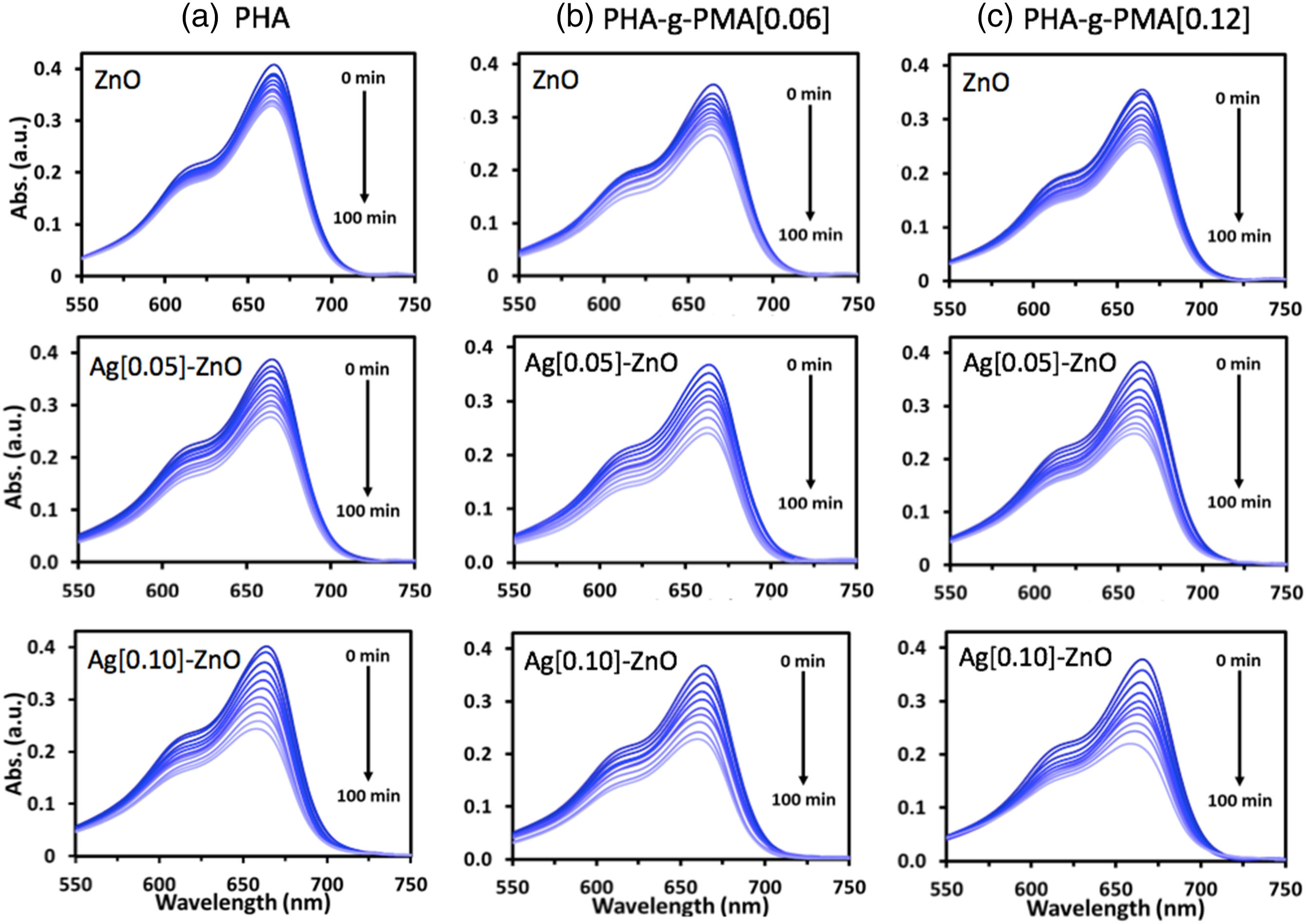
The profiles of MB removal (%) for all polymer composites are shown in Figure 2(a). The composites showed a linear increase in the photodegradation efficiency of the dye over time. Nevertheless, higher photodegradation efficiencies were observed when PHA graft copolymers i.e., PHA-g-PMA[0.06 and 0.12] were used as the supporting matrices relative to the neat PHA. In a previous study, superior wettability of PHA-g-PMA towards methanol ameliorate the dispersal of methanol-suspended ZnO microparticles during the decoration step.
28
The microparticles are well-distributed and adsorbed to the film surface with minimal aggregation, thus attributing to the observed increase in photodegradation efficiency of decorated PHA-g-PMA samples as shown in Figure 2(a). For neat ZnO and Ag-doped ZnO, the latter showed enhanced photodegradation efficiency. Higher percentage of MB removal was obtained with higher Ag amount doped to the ZnO, affirming the expediency of Ag in augmenting the responsiveness of ZnO microparticles towards light energy in photocatalysis. (a) MB removal/photodegradation profiles and (b) ln (Ct/C0) versus irradiation time (t) plots for the MB photodegradation using different polymer composite films. All films were decorated with the same amount of photocatalysts (0.05 mg).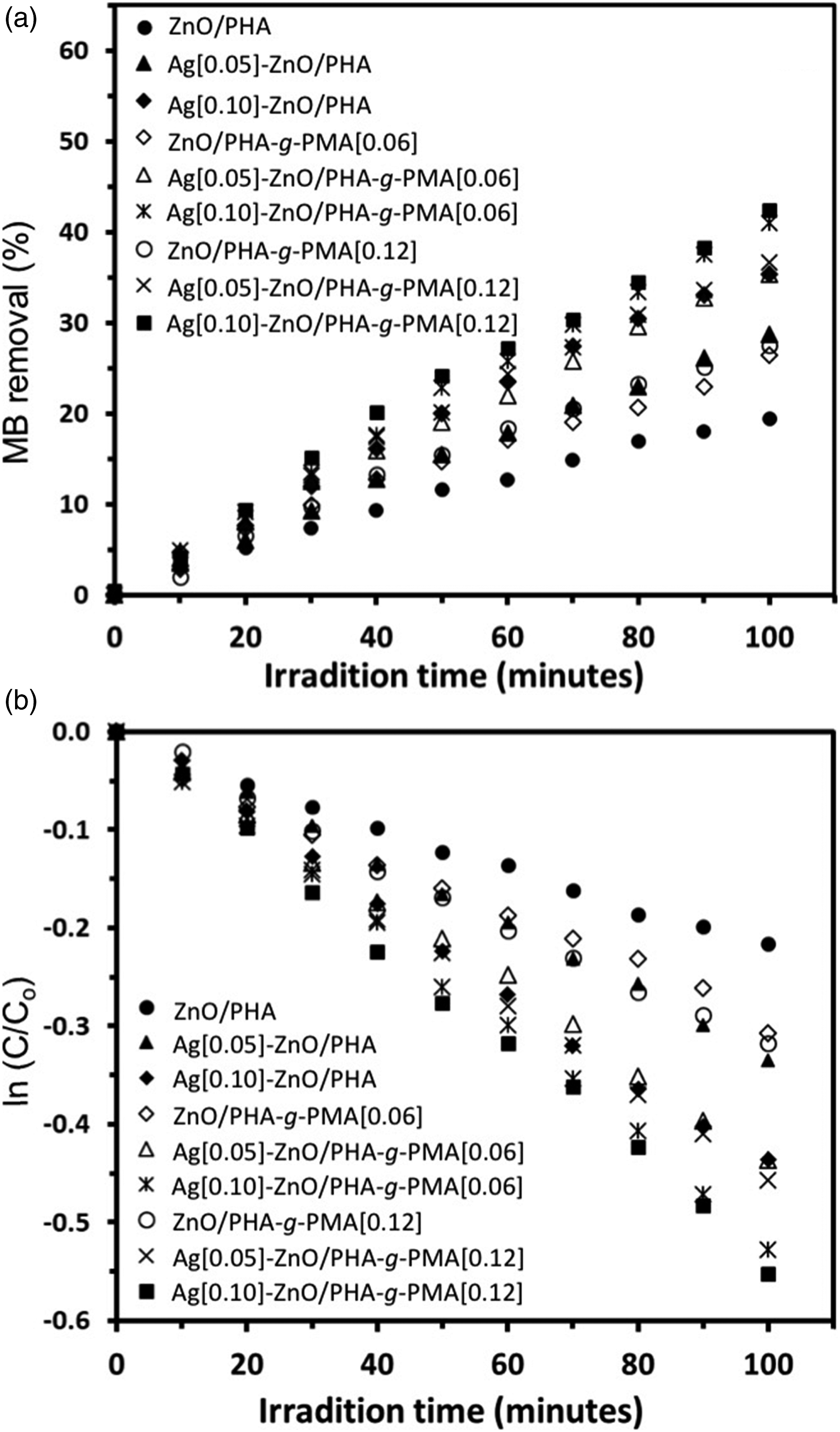
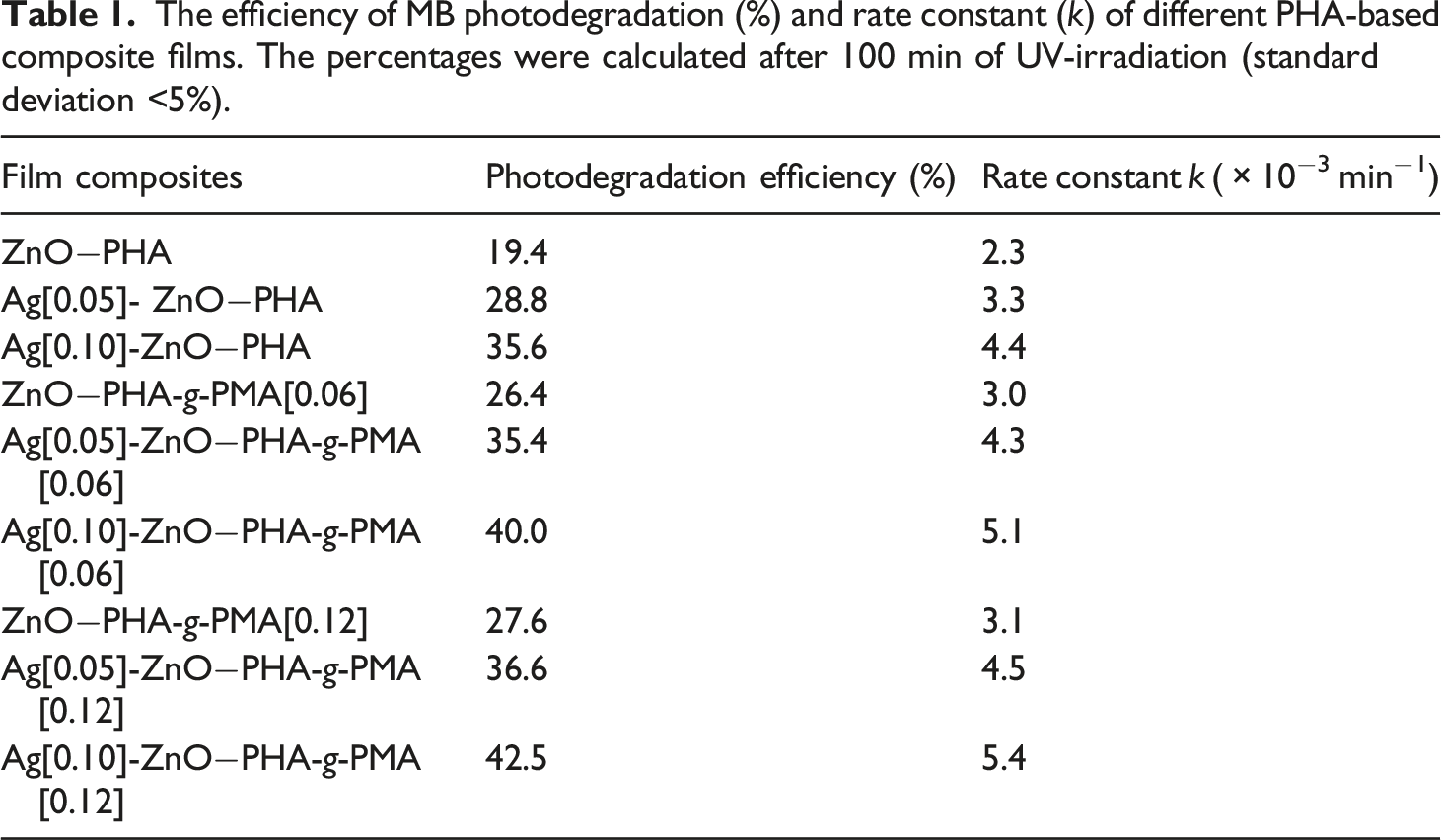
The efficiency of MB photodegradation (%) and rate constant (k) of different PHA-based composite films. The percentages were calculated after 100 min of UV-irradiation (standard deviation <5%).
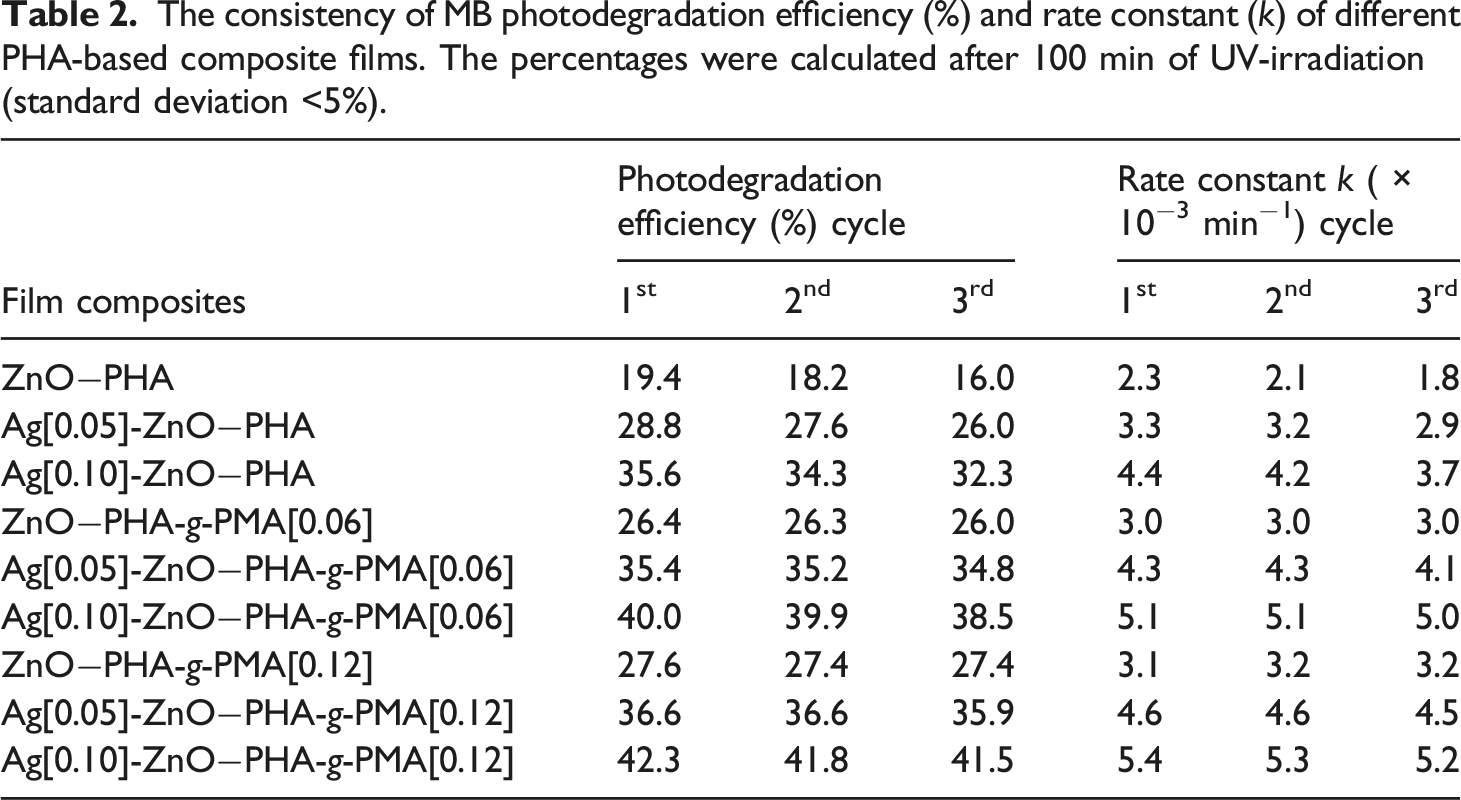
The consistency of MB photodegradation efficiency (%) and rate constant (k) of different PHA-based composite films. The percentages were calculated after 100 min of UV-irradiation (standard deviation <5%).
Photocatalytic degradation of complex dye compounds
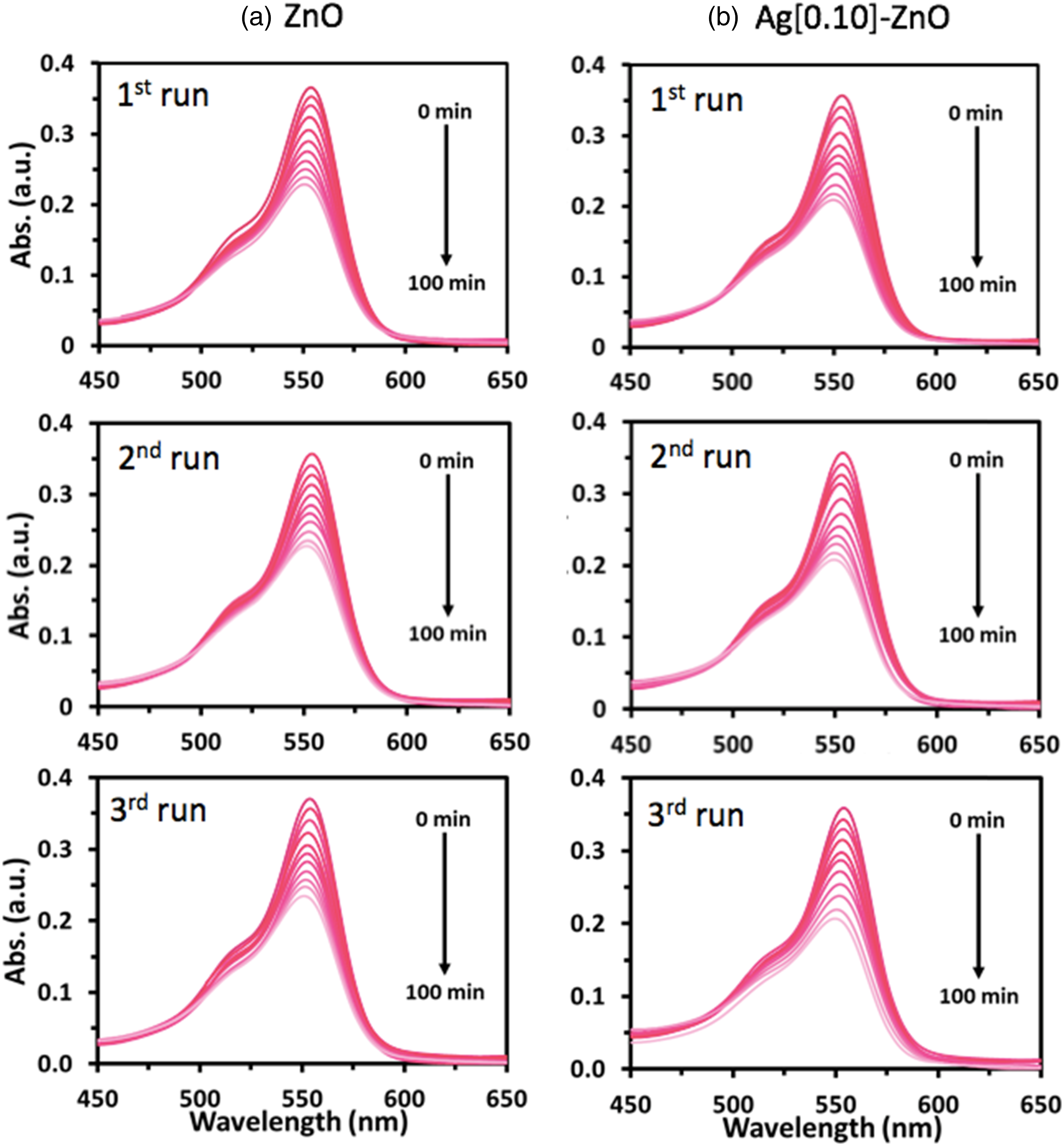
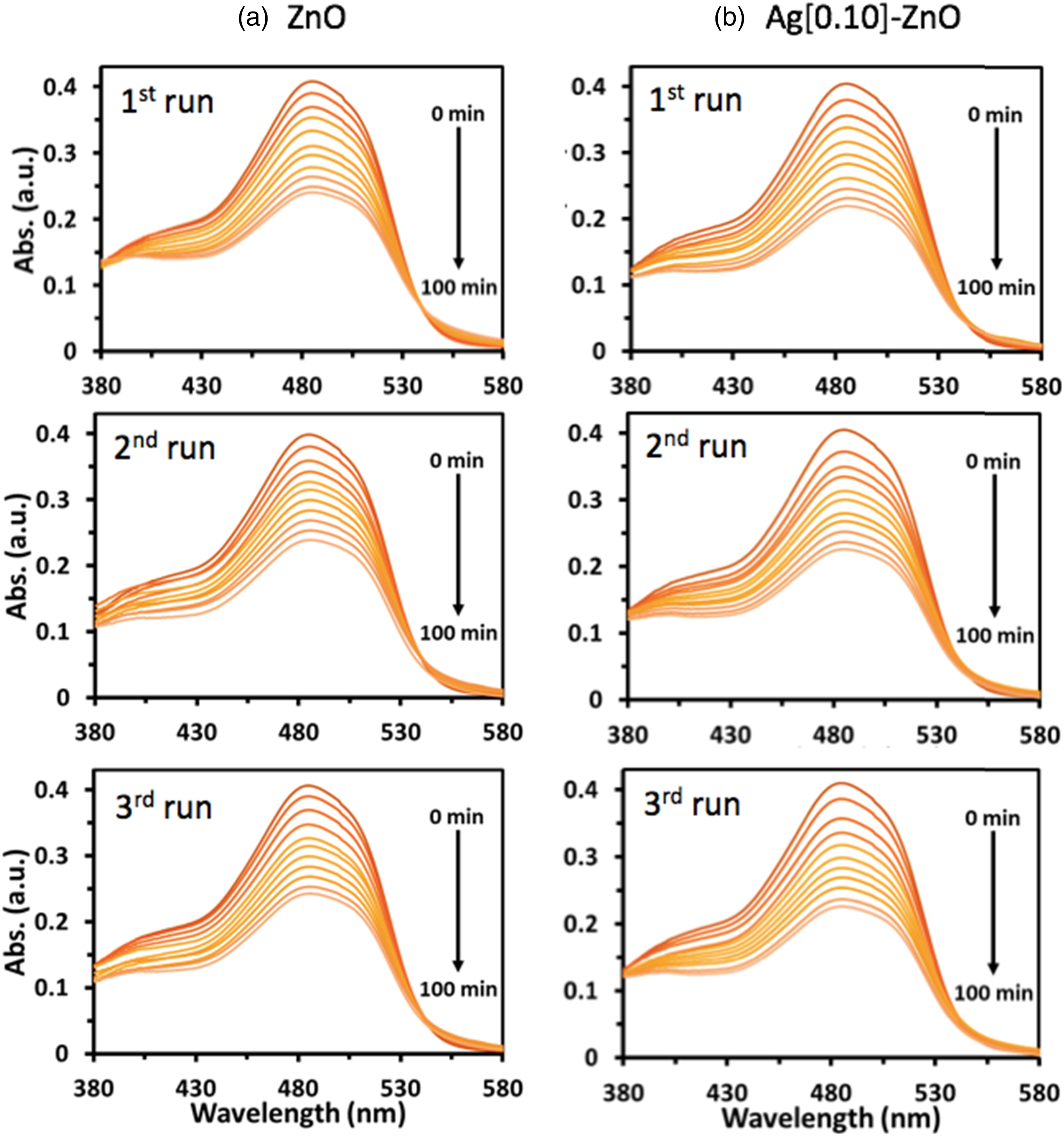
Figures 3, 4 and 5 compared the time evolution of methylene blue (MB), Rhodamine B (RB) and Orange II (OII) absorbance spectra, respectively, when irradiated with UV light. It is evident that both photocatalysts i.e., ZnO and Ag[0.10]-ZnO (Figure 4) decorated on the PHA-g-PMA[0.12] supporting matrix, are able to degrade the three different organic dyes through photocatalysis. Interestingly, the later showed higher degree of dye photodegradation, further substantiating the earlier proposition that silver doping of the semiconductor enhances its photocatalytic activity. Overall, the results showed the potential of investigated photocatalysts in degrading different complex pollutants when they are in reaction proximity. The time evolution of MB absorbance spectra for polymer composite films when irradiated with UV light. Graft polymers, i.e., PHA-g-PMA[0.12] was used as the supporting matrix to decorate (a) ZnO and (b) Ag[0.10]-ZnO. The time evolution of RB absorbance spectra for polymer composite films when irradiated with UV light. Graft polymers, i.e., PHA-g-PMA[0.12] was used as the supporting matrix to decorate (a) ZnO and (b) Ag[0.10]-ZnO. The time evolution of OII absorbance spectra for polymer composite films when irradiated with UV light. Graft polymers, i.e., PHA-g-PMA[0.12] was used as the supporting matrix to decorate (a) ZnO and (b) Ag[0.10]-ZnO.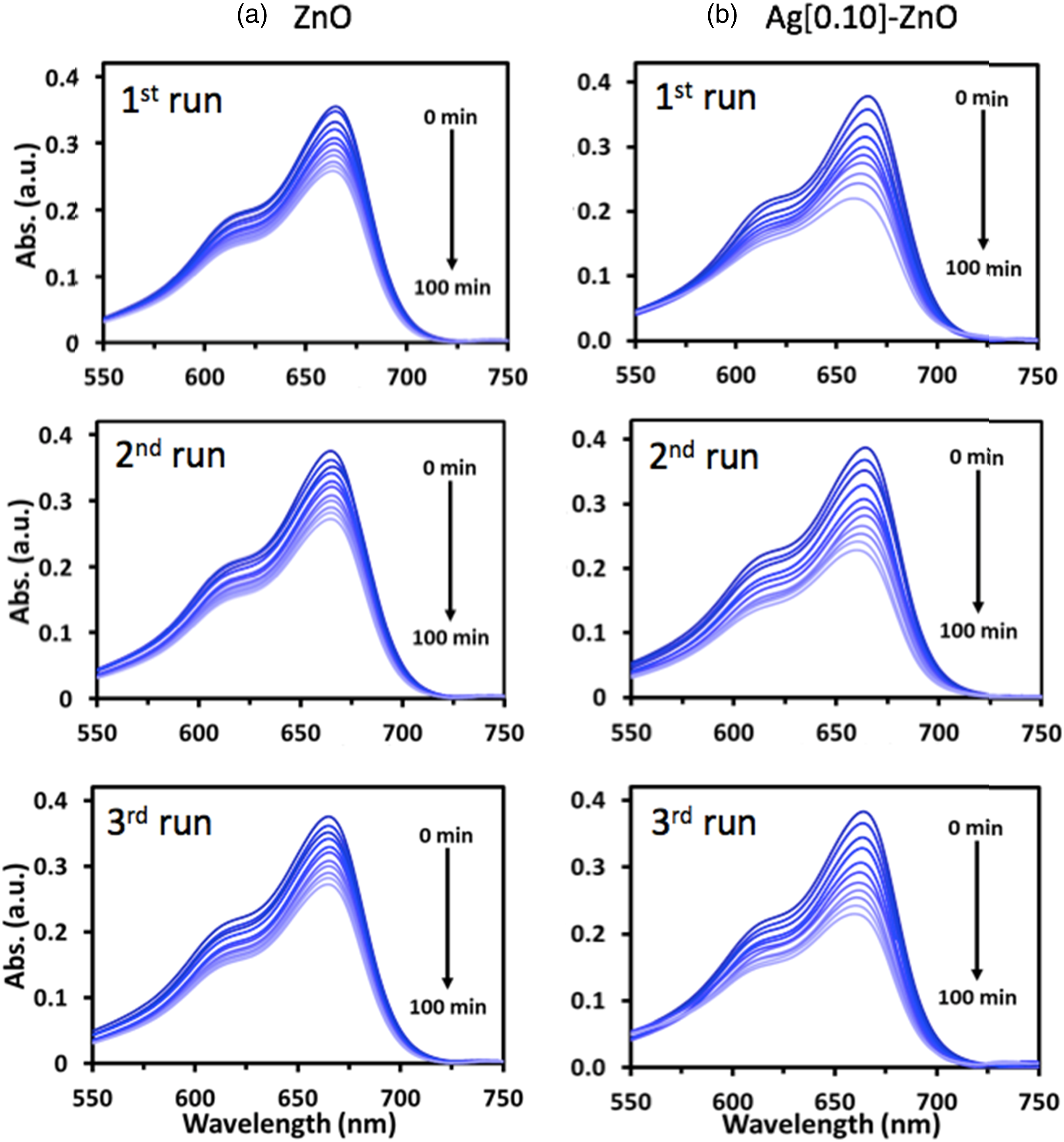
The consistency of dye photodegradation efficiency (%) and rate constant (k) of different PHA-based composite films. The percentages were calculated for 100 min of UV-irradiation (standard deviation <5%).
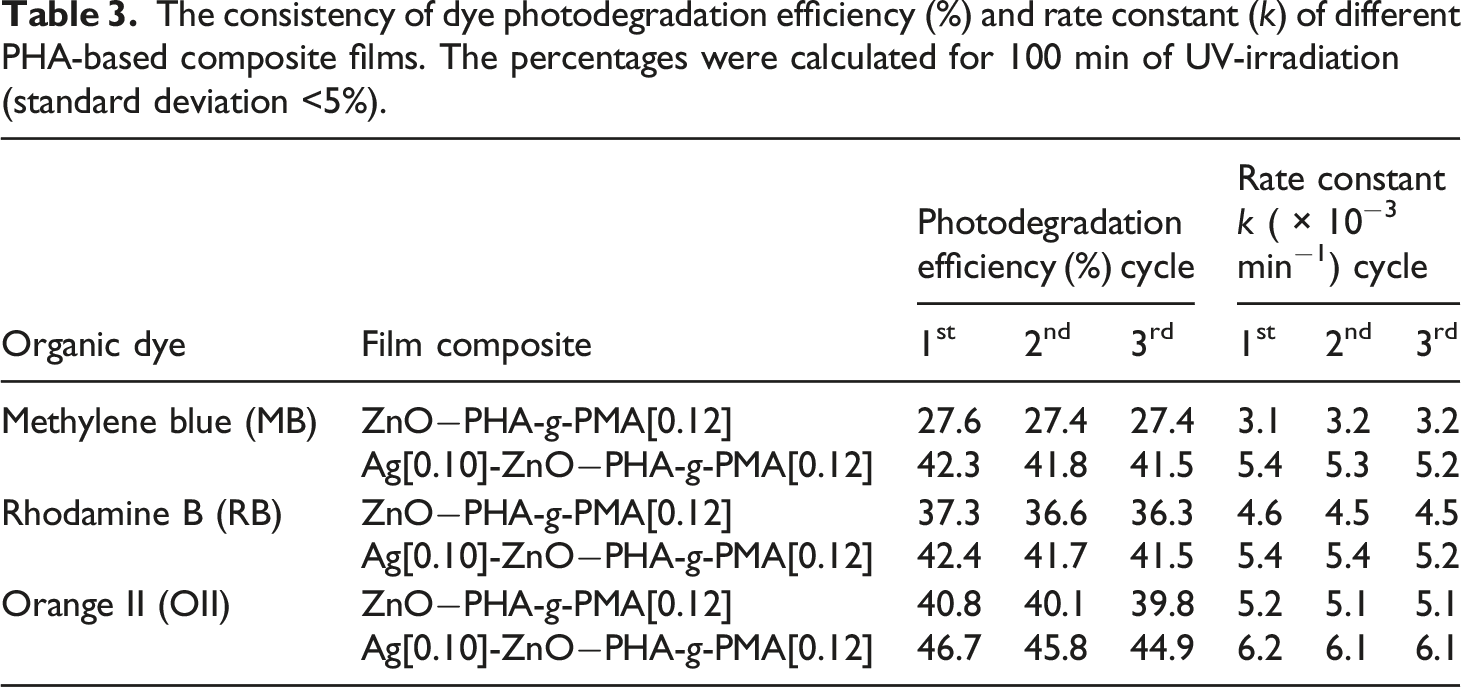
The supporting matrix i.e., PHA-g-PMA[0.12] provided consistent and reliable structural support to the overall photocatalytic performance (Table 3). The PHA graft copolymer assisted in preserving the photocatalytic activity over repeated applications, primarily through its shielding effect towards photocatalyst exuviating off the surface under hydrodynamic forces, concomitantly contributing to the preparation durability. Figure 6 shows the elemental diffraction X-ray (EDX) analysis of neat PHA and PHA-g-PMA[0.12] decorated with Ag[0.10]-ZnO (6a and 6b, respectively). Both composite films experienced hydrodynamic shear force when immersed within a beaker containing 100 mL of stirred distilled water overnight. Neat PHA-based composite film showed some aggregations of Zn and Ag particles on its surface (Figure 6(a)) afterwards, as compared to the graft copolymer film (Figure 6(b)). From the elemental analysis (Table 4), the apparent concentration and atomic percentage of Zn and Ag on neat PHA preparation were notably diminished, in contrast to PHA-g-PMA[0.12] preparation. The results support the hypothesis that PHA graft copolymer film provides shielding effect towards photocatalyst particles from sloughing off the surface under hydrodynamic stress, thus preserving the composite films photocatalytic performance. Elemental mapping of (a) neat PHA film composite and (b) PHA-g-PMA[0.12] film composites decorated with Ag[0.10]-ZnO before- and after immersion in a continuously stirred distilled water overnight. Elemental analysis on the surface of Ag-ZnO−PHA composite films (standard deviation <5%).
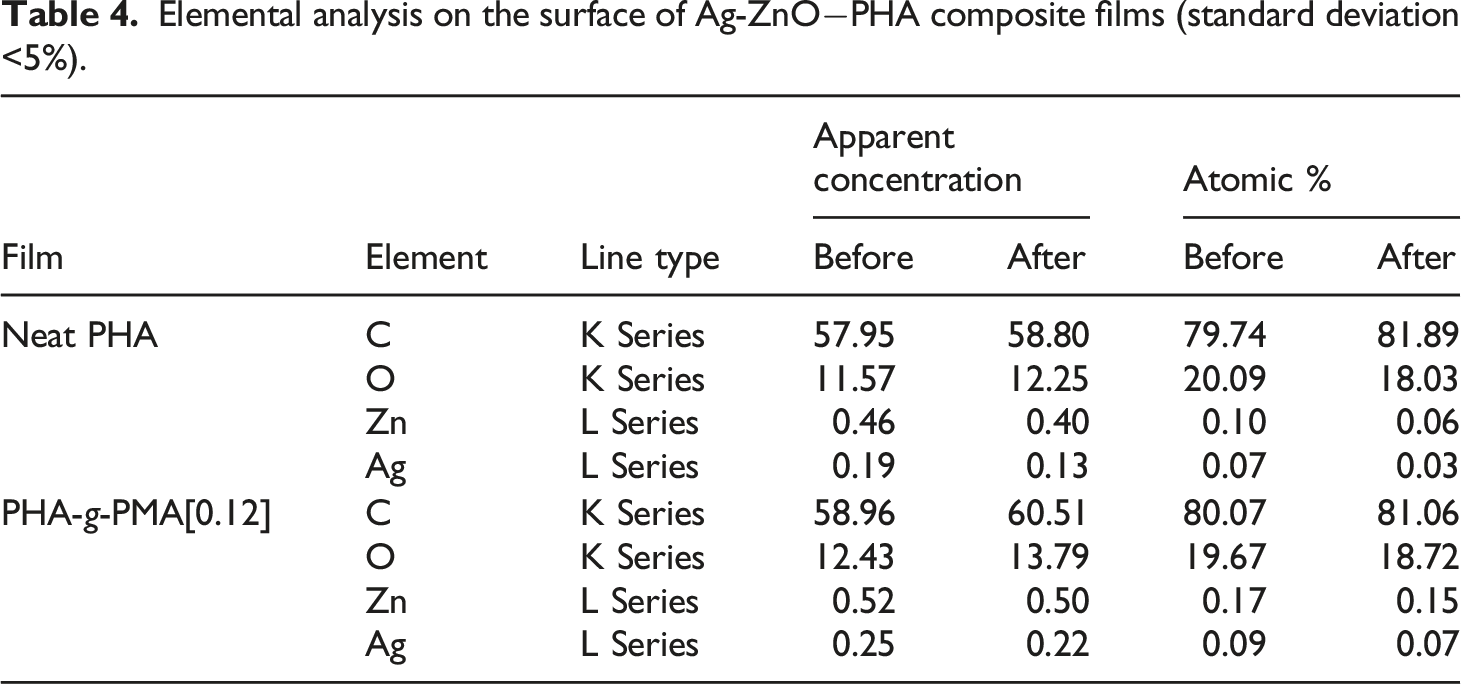
Antibacterial and toxicity studies of Ag-ZnO−PHA composite film
Figure 7 shows the PHA-g-PMA[0.12] composite films after being incubated for 7 days on different bacterial plate cultures. Bacterial growth of tested species were observed on the PHA-g-PMA[0.12] films that were decorated with neat ZnO based on the presence of white-colored biofilm on the polymer film as shown in Figure 7(ai to ci). PHA is a biodegradable polyester originally produced by microorganisms as a storage material during metabolizable carbon supply crisis. Hence it is likely to be used as growth substrate by the three bacterial species studied. However, for PHA-g-PMA[0.12] films decorated with Ag-doped ZnO, biofilm was entirely absent or showed almost negligible growth (Figure 7(aii to cii)). Moreover, growth inhibition area was observed on the Bacillus subtilis plate culture, as indicated by the arrows in Figure 7cii. These observations provide evidence of antibacterial property of Ag-ZnO particles. Similarly, antibacterial property of Ag-ZnO against Staphylococcus aureus and E. coli have been reported.
31
In general, Ag particles are known to inhibit the growth of many pathogenic bacteria through multiple mechanisms namely promoting protein dysfunction, damaging membrane cell etc.32,33 Therefore, in addition to enhancing photocatalytic performance, decorating the PHA graft copolymer film with Ag also provides deterrence to biofilm growth that may result in PHA biodegradation, and reducing its film transparency. Aforementioned advantages highlight the potential of Ag-ZnO−PHA composite film for the application reactive coating technology, which will be discussed further in the last section. Antibacterial testing of PHA graft copolymer composites against (a) Escherichia coli, (b) Bacillus cereus and (c) Bacillus subtilis. PHA-g-PMA[0.12] films were decorated with neat ZnO and Ag[0.10]-ZnO (i and ii, respectively).
Toxicity testing of samples on Ceriodaphnia dubia.
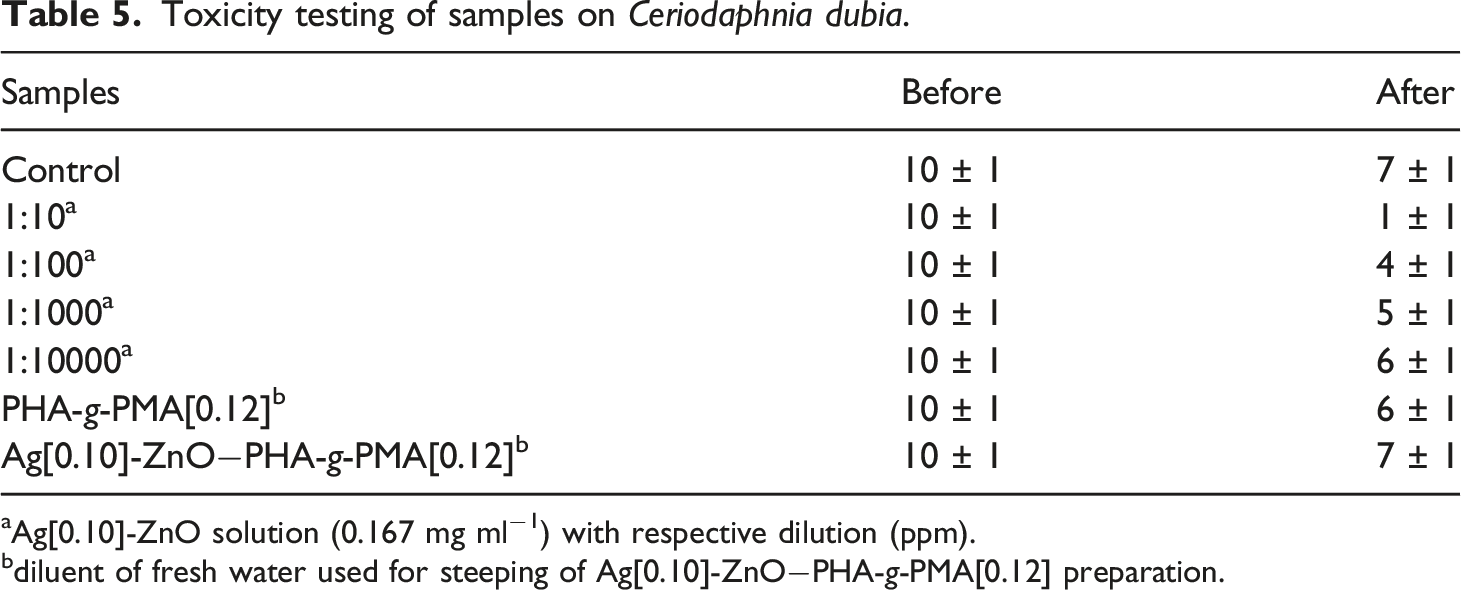
aAg[0.10]-ZnO solution (0.167 mg ml−1) with respective dilution (ppm).
bdiluent of fresh water used for steeping of Ag[0.10]-ZnO−PHA-g-PMA[0.12] preparation.
Potential and advantages of Ag-ZnO−PHA composite film in reactive coating technology
According to the X-ray fluorescence (XRF) analysis (Figure S10 in the Supporting Information), two additional peaks at ≈ 3 and 3.5 keV observed in the Ag-ZnO sample, similar to those reported in other studies, 36 indicate the presence of metallic Ag element. Furthermore, two additional peaks in the X-ray diffraction (XRD) spectra at 2θ values of 38° and 44° indicate elemental growth of silver in the ZnO lattice (Figure S11 in the Supporting Information). This result corresponds to a previously reported study, 37 which concluded that Ag atoms were grown on the surface of ZnO rather than loaded into the ZnO lattice. According to the study, a spin-orbit energy difference of 7 eV (373.5 and 366.5 eV) in X-ray photoelectron spectroscopy (XPS) analysis was due to a metallic silver cluster. 37
The photocatalysis occurs primarily on the surface of the PHA film where the Ag-ZnO particles are positioned (Figure 8(a)). Upon UV radiation, the electrons (e−) from the valence band (VB) of ZnO are photoexcited to the conduction band (CB) with the generation of the same number of holes (h+). Meanwhile, due to the lower Fermi level of Ag-ZnO compared with the CB energy of ZnO, the photoexcited e− will be transferred from ZnO to Ag due to potential energy difference. In this way, the accumulation of the photogenerated e− occurs in Ag particles along with the transfer of photogenerated h+ to the surface of ZnO, inducing the separation of the photogenerated e− and h+, as shown in Figure 8(a). Afterwards, the e− will be taken up by O2 molecules thus generating strong oxidizing ⋅O2− species that are able to degrade organic materials. Meanwhile, the generated h+ can react with hydroxyl (OH−) or H2O to generate hydroxyl radicals (·OH). They could effectively degrade organic materials into products like CO2 and H2O, which are expected to further improve the photocatalytic efficiency.
30
It is reported that the addition of Ag enhances the photocatalytic activity of ZnO as much as 34% because of excited electrons sequestration by Ag nanoparticles, which prevents electron-hole recombination.
38
The enhanced photocatalytic activity is attributed to the synergistic actions between Ag nanoparticles and clusters in zinc oxide, where charge separation plays a dominant role.
39
Some studies have also shown that Ag plays an important role in making some semiconductor composites more catalytic. For example, Ag aids in the photocatalytic activity of KNbO3 in the synthesis of NH3 under visible light energy
40
and the piezocatalytic activity of PbBiO2I in the degradation of Rhodamine B under ultrasonic vibration energy.
41
(a) Photocatalytic mechanism, and (b) potential applications of transparent Ag-ZnO−PHA based biopolymer composite film as self-clean coating technology.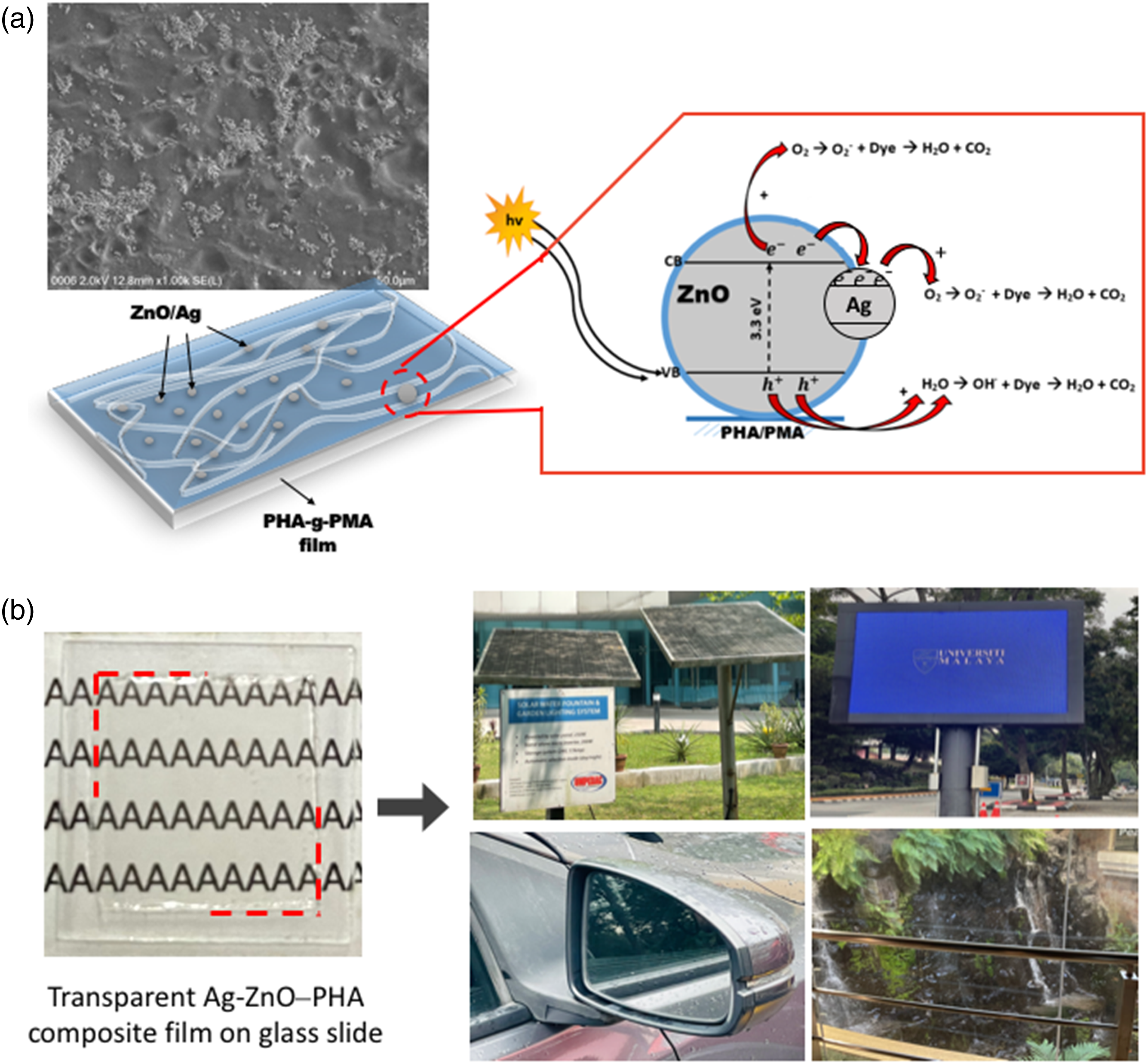
From the results, it can be deduced that Ag-ZnO–decorated PHA film exhibits good potential in self-clean coating technology. The transparency of Ag-ZnO−PHA composite film will not obscure the surface underneath (Figure 8(b)). Nowadays, transparent self-clean materials are much sought after particularly in photovoltaic applications. The Ag-ZnO−PHA film coating may allow sunlight to pass through, and at the same time preventing organic dirt from forming a covering layer on the solar panel surface thus preserving the performance of the solar-electrical energy conversion. Additionally, screen monitor, display window, mirrors are some examples that could benefit from Ag-ZnO−PHA film coating (as illustrated in Figure 8(b)).
In addition to transparency, high hydrophobicity characteristic of PHA makes them insoluble in water, and provides some resistances to hydrolytic degradation while prolonging and sustaining photocatalytic performance. Decoration of Ag-ZnO particles may also extend the life of PHA film composite by increasing the film hydrophobicity, and prevent biofouling. Incorporation of ZnO particles has been shown to enhance the hydrophobicity of coating film resulting in a marked reduction in the surface maintenance of metal panel. 42 In another study, the contact angle measurement of the polymer nanocomposites showed an increase in its hydrophobicity when incorporated with silver-doped zinc oxide nanoparticles. 39
Both PHA graft copolymer and Ag-ZnO can be synthesized through green approaches using renewable resources thus making them sustainable and ecofriendly. There are numerous wastes (food, agricultural etc.) that have been shown as viable carbon sources to produce PHA. 43 ZnO nanoparticles can be synthesized from various biological materials e.g., plant extracts, fruit extracts, fungi, algae and bacteria. Plant extracts offer a simple and effective approach to synthesize nanoparticles at large scale, 44 and could help to reduce the particle size by increasing the surface area of the material. 45 ZnO is a Federal Drug Administration (FDA)-approved inorganic compound currently used in a wide range of commercial applications namely food packaging, textile and biomedicine.38, 46–48 Recent studies have proposed that association of Ag to nanoparticles such as ZnO may reduce the reactivity of silver ion, potentially making it feasible for clinical- and therapeutic applications. 49
Conclusion
Ag-ZnO−PHA based composite film is demonstrated for its applicability in photocatalysis. Sought after attributes such as high transparency and hydrophobicity are amendable for self-clean coating technology. Both the PHA and photocatalyst compounds can be sustainably synthesized using renewable raw materials. Ag-ZnO−PHA based composite film is deemed ecofriendly since polluting synthetic microplastic component is absent as well as favorably reduces the consumption of synthetic detergents for cleaning and maintenance.
Supplemental Material
Supplemental Material - Photocatalytic Ag-ZnO − poly-3-hydroxyalkanoate composite film for translucent coating application
Supplemental Material for Photocatalytic Ag-ZnO − poly-3-Hydroxyalkanoate composite film for translucent coating application by Khairul Anwar Ishak, Chong-Boon Ong, Nur Azmina Mohamed Safian and Mohamad Suffian Mohamad Annuar in Journal of Thermoplastic Composite Materials
Footnotes
Acknowledgements
The authors acknowledge Centre for Fundamental and Frontier Sciences in Nanostructure Self-Assembly (FSSA) and Low Dimensional Materials Research Centre (LDMRC), Universiti Malaya for providing the lab and instrument facilities.
Author contributions
All authors equitably contributed towards the manuscript preparation, i.e., design of study, data acquisition/analysis/interpretation, and drafting/revising of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported financially by the Ministry of Higher Education Malaysia via the Fundamental Research Grant Scheme (FRGS/1/2020/STG01/UM/01/1). The Universiti Malaya also partially supported this research through High Impact Research Grant [UM.C/625/1/HIR/MOHE/05], Impact-Oriented Interdisciplinary Research Grant [IIRG017A-2019] and UM International Collaboration grant (ST021-2022). Internal Research Grant (2019-2021) by International University of Malaya-Wales also partially funded the research.
Availability of data and material
All data generated or analyzed during this study are included in this article and its Supporting Information.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
