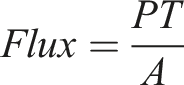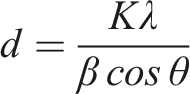Abstract
Flexile composite film of Poly-3-hydroxyalkanoate−Perovskite−Tween 80 (PHA-PV-T80) as a green membrane film was prepared. Pristine medium-chain-length poly-3-hydroxyalkanoate (mcl-PHA), produced through bacterial fermentation, was blended with different concentrations of perovskite (PV) and Tween 80 (T80) ranging from 0.01 to 0.0025 % (w/v) and 1.5 – 4.5 % (w/v), respectively, to fabricate the composite films. Ultrasound irradiation was applied to aid homogenous dispersion of blend solution prior to film casting. The composite film exhibited improved physical properties compared to the neat mcl-PHA film, which is more susceptible to ripping upon peeling. Thorough distribution of perovskite, facilitated by T80, were responsible for promoting increased side-chain crystallization, resulting in the formation of extensive rigid regions within the PHA matrix, which significantly improved proton permeability of the composite film. PHA composites showed higher proton flux (>3.5 × 10−4 mol.min−1.cm−2) compared to neat PHA (1.03 × 10−4 mol.min−1.cm−2) and commercial Nafion (1.22 × 10−4 mol.min−1.cm−2). The relationship between physicochemical attributes and proton flux characteristics of the composite films were discussed with regards to their potential application as proton exchange membrane.
Introduction
Recently, increasing efforts have been directed towards developing composite film from biological polymers,1–3 as they are attractive substitutes due to their low cost and biodegradable characteristics. 4 PHA, also known as poly (3-hydroxyalkanoates), is becoming a widely studied biopolymer.5–8 It is synthesized by microorganisms as a result of an imbalanced growth environment. 9 A few examples of PHA technological applications include the PHA-carbon nanotube composite for ion movement enhancement, 10 the PHA-PEGMA graft copolymer for electrode modification, 11 and graphene-grafted PHA for biosensors and nerve repair conduits. 12 The modified PHAs showed a significant increase in proton conductivity compared to their neat form.10–12
The compositing of inorganic nanostructured materials, such as perovskite oxides, into biopolymers started to gain prominence in recent years.13,14 They are commonly employed to enhance the physical characteristics and other properties when integrated as part of a composite film. 15 For example, strong homogenous composite was fabricated using strontium titanate (SrTiO3) and cellulose that exhibited efficient removal of chromium (VI). 16 The biocomposite film was able to remove approximately 99.5 % of Cr (VI) from the aqueous solution (8 µg/ml) at pH 2.0. 16 Delaminated perovskite CsCa2Nb3O10 was assembled with gelatin to produce a translucent film and its dielectric properties were studied, where significant gain in dielectric permittivity than dielectric loss was observed, indicating the film’s potential to be used in various applications such as improving artificial bone implants to stimulate tissue growth. 17 The non-toxic nature of perovskite oxides has also enabled it to be safely applied as a bioactive material in scaffold for tissue engineering. 18
Application of polymeric film in filtration technology is an attractive way to treat wastewater from cities and oil fields in second- or third-level processing.19,20 An ion-exchange membrane is often used in electrodialysis to remove salt from water and harmful chemicals from industrial wastewater.21,22 The polymeric film acts as a physical barrier or membrane that separates two different liquids and can control the direction of movement for different chemical species due to its selective permeability. One example is selective proton exchange membrane (SPEM), which only allows cations, like proton, to pass through due to its negatively charged polymer matrix.21,23,24 Perfluorosulfonic acid polymer (Nafion) is currently and widely applied as the SPEM due to its chemical stability, mechanical qualities, and high proton movement capacity.25,26 Nevertheless, high production cost, and environmental impacts surrounding its manufacturing and disposal have raised concerns. 27 A study found that incorporating TiO2 and SiO2 nanoparticles increased the proton conductivity of neat Nafion from ∼3 to 11 mS cm−1 at 40°C. 28 Similarly, the incorporation of 2D poly (triazine imide) increased Nafion’s proton conductivity from ∼0.08 to 0.12 S cm−1 at 30°C. 29 It is unequivocally demonstrated that even high-performance polymers such as Nafion require some degree of modifications with additives to improve its effectiveness as proton exchange membranes.
In this study, a biodegradable medium-chain-length PHA (mcl-PHA) was utilized as the base material to prepare a polymeric membrane film. Perovskite oxide (SrTiO3) and surfactant (Tween 80) were composited in specific proportion to reinforce the mcl-PHA film and promote the unidirectional flux of protons. In addition to improving physical attributes, SrTiO3 can also enhance proton conductivity of a membrane or composite film. 30 They are known to have a stable structure because of their balanced geometrical arrangement of basic atoms as well as valences. 31 Surfactant on the other hand, would help to improve the miscibility of two distinct phases in the polymer composite due to its unique amphiphilic structure.32–34 The use of mcl-PHA as a green host matrix for perovskite and surfactant in constructing a polymer nanocomposite for applications in membrane separation technology is yet to be investigated, hence clarifying the significance of the study.
Methodology
Materials
Strontium titanate (SrTiO3) nanoparticle with size <100 nm and Tween 80 were purchased from Sigma Aldrich (U.S.A), while Nafion 117 was supplied by Dupont (U.S.A). Nafion was activated based on an earlier study prior to its usage as a reference membrane. 10 Dichloromethane, ACS Reagent with ≥99.9% purity, was purchased from Merck (U.S.A). Tween 80 or polyethoxylated sorbitan, was purchased from Sigma Aldrich (U.S.A).
Biosynthesis of Mcl-PHA
P. putida BET001 was cultured using batch fermentation technique in shake-flasks to produce mcl-PHA with lauric acid as the main carbon and energy source. 35 The culture system was divided into two stages comprised of the cell growth stage and PHA accumulation phase. The cell growth stage was initialized with the cultivation of bacterial culture in rich medium for 24 h. Subsequently, cell biomass was harvested at 8000× g, 4°C (Thermo Scientific Sorval RC-5C Plus ultracentrifuge), washed with saline solution (0.9% w/v), then aseptically transferred to the E2 medium to undergo the PHA accumulation phase. 1.0% (w/v) of fatty acid and 0.35% (w/v) ammonium sodium phosphate dibasic were added into the medium as carbon- and nitrogen sources, respectively.
Following incubation of E2 medium culture for 24 h, cell biomass was harvested by centrifugation. Pellets were rinsed twice using 0.9% (w/v) saline solution and then dried in the oven at 50°C until they reached constant weight. Fatty acid residue was discarded by biomass dissolution in n-heptane.
Poly-3-hydroxyalkanoate extraction and purification
PHA extraction from dried biomass was carried out by refluxing 1.0 g dried cells in acetone (100 mL) for 4 h at 25°C ± 2.0. Then, the slurry was filtered using Whatman filter paper No.1. The filtered pellets were recollected and underwent two additional cycles of solvent reflux in 50 mL of acetone for another 4 h. Repetitive cycles were conducted to ensure maximum PHA extraction from the biomass. Each filtered solution from all three cycles was combined, and then concentrated by utilizing the rotary evaporator until approximately 10 mL remained.
The concentrated solution containing the polymer was then mixed with cold methanol (1:7) to induce PHA precipitation. After centrifugation at 1920× g for 10 min, supernatant was removed and the PHA sediment at the bottom was recovered to undergo another round of solvent precipitation process. This step was conducted several times to obtain highly purified PHA. Then, the polymer solution was cast onto a glass petri dish and left to air-dried until constant weight.
Fabrication of polyhydroxyalkanoate-perovskite-Tween 80 (PHA-PV-T80) composite film
Pre-determined mass percentages of SrTiO3-type perovskite (designated as PV) and Tween 80 (designated as T80), ranging from 0.01%–0.0025% (w/v) and 1.5%–4.5% (w/v), respectively, and mcl-PHA (0.4 g) were dispersed in dichloromethane (DCM) using ultrasound-assisted technique in water bath sonication (Elmasonic P, sonicator). The solution was sonicated at 80% power output for 40 min at frequency of 37 kHz. Water inside the bath was changed when necessary to ensure temperature remained constant (30°C) throughout the sonication period. After ultrasonication, the solution was vortexed to obtain a homogenous mixture prior to casting on a petri dish (11 cm2) to be air-dried to a constant weight. All steps above were repeated when fabricating membranes using different surfactant. Neat-PHA film was prepared using a similar technique to the PHA blends. The thickness of the membrane was measured using a digital micrometer (Mitutoyo, ±0.001 mm) based on the average of three random measurement points.
Membrane characterization
Physicochemical analysis
Functional groups present in the neat/composite membrane were determined using the Perkin Elmer FTIR-Spectrum 400, equipped with PIKE GladiATR hovering monolithic diamond ATR assessor. The spectra were recorded between 450 and 4500 cm−1 at room temperature.
Crystalline properties of neat mcl-PHA, perovskite and PHA-PV-T80 composites were studied using a high resolution XRD (Rigaku SmartLab) where scanning was performed at 25°C and between 5°≤ 2θ ≤ 80°.
Morphological studies of all membranes were carried out using the field emission scanning electron microscopy (SU8220 Hitachi). The observation was conducted under electron acceleration of 10 kV in a high vacuum atmosphere at room temperature.
Thermal analysis
Differential scanning calorimetry (DSC 8825/700) (Mettler Toledo) analysis was used to identify the glass transition temperature (T g ) and melting temperature (T m ) of all samples. All samples (about 7 mg) were loaded into sealed aluminium pans and heated from −50 to 100°C at a heating rate of 10°C min−1
Degradation temperature (T d ) of neat-PHA and PHA-PV-TW80 samples were measured using Thermogravimetric Analysis 4000 (TGA4000) (Perkin Elmer). The samples (about 7 mg) were subjected to a heating rate of 10°C min−1 from 30 to 500°C.
Measurement of proton flux across membrane
A double-chambered vessel comprising two H-shaped glass bottles was utilized as the setup for the proton transfer analysis (Figure S1 in Supporting Information). Each membrane was placed in-between both chambers for the experiment. Initially, left- and right-hand side chambers were filled with the same amount of ultrapure water, and allowed to equilibrate for a few minutes while being stirred at a constant rate. Then, 0.6 mL of HCL solution (0.1 M) was added to the left-hand side chamber. pH changes in the right-hand side chamber were recorded every 30 s until it reaches a plateau. The same experimental setup and procedure was applied for proton flux analysis in tap water. The experiment was conducted at 25 ± 0.1°C. Proton flux (×10−4 mol.min−1.cm−2) is calculated as follow:
Results and discussion
Preparation of PHA-PV-T80 composite films
Mcl-PHA possesses natural properties to create a ductile and transparent film. However, in our specific application, where the membrane thickness is 2.0 mm, the mcl-PHA is prone to ripping when peeled (Figure 1(a)). When composited with perovskite (PV) and Tween 80 (T80), the film showed less susceptibility to rip when peeled. Figure 1(b) shows the PHA composite film as a representative for all the composite films that were prepared in this study. PV and T80 are proposed to interfere with the “typical” arrangement of PHA chains in the following manner; perovskite promotes the formation of rigid regions within the PHA matrix, resulting in a more rigid film that is resistant to ripping when peeled. On the other hand, T80 facilitates uniform dispersion of PV throughout the hydrophobic matrix of mcl-PHA, enabling the formation of a widespread rigid framework. However, if T80 is incorporated at a concentration higher than 4.5% (w/v), the PHA film exhibited an uneven thickness, as shown in Figure 1(c). One possible reason for the observation is PHA chain ‘bundling’ promoted by disproportionate intercalation of T80 among PHA chains. Incorporating more than 0.01% (w/v) of PV resulted in observable whitish deposition due to PV crystallization (Figure 1(d)). Elucidation of the occurrence is presented in the final section. For the subsequent experiments, it is determined that the maximum percentages for composited PV and T80 to obtain desired films are 0.01 and 4.5% (w/v), respectively; while the lowest percentages for both compounds are 0.0025 and 1.5% (w/v), respectively. Film of (a) pristine mcl-PHA, (b) PHA-PV0.01%-T80 4.5% composite, (c) PHA-PV0.01%-T80 5.0% and (d) PHA-PV0.02%-T80 4.5%.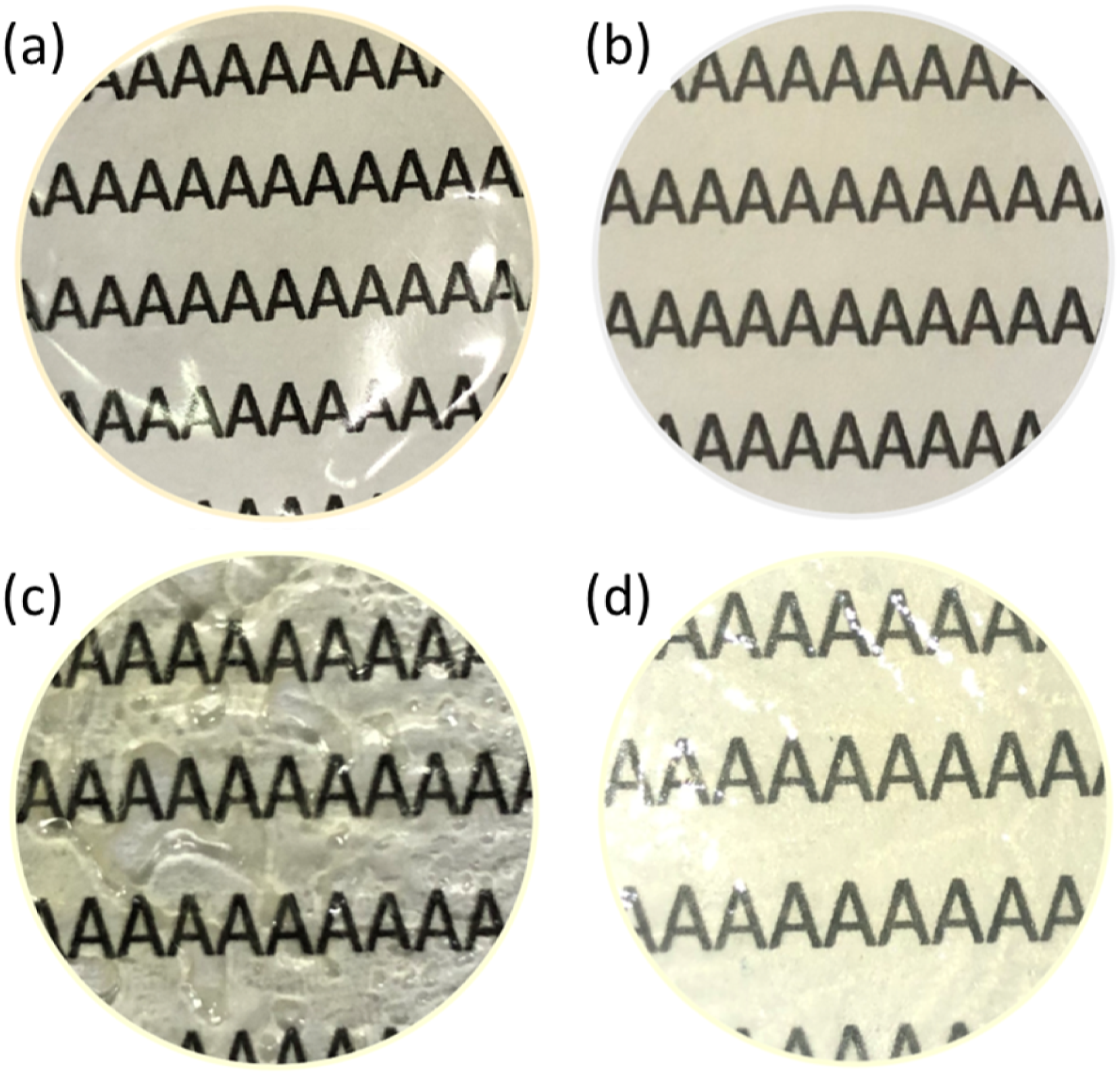
FTIR analysis of PHA-PV-T80 composites
As shown in Figure 2, both neat-mcl PHA and Polyhydroxyalkanoate-Perovskite-Tween 80 (PHA-PV-T80) composites yield signal peak of the representative moiety i.e., intense peak at ≈ 1730 cm−1 that signifies the stretching band of carbonyl group (C = O) of PHA and T80.36,37 Two absorption peaks at 2850 and 2925 cm−1 are both representing the stretching modes of anti-symmetric and symmetric CH3 and CH2 of alkyl chain, respectively. Meanwhile, peak signals from 700 to 1500 cm−1 stipulate the occupancy of CH2, CH3, -C-O-C and -C-C-. Bands 1050 – 1280 cm−1 specify the symmetric and antisymmetric stretching mode of C-O-C in PHA.
36
The absorption peak located near 720 cm−1 is assigned to the C−C skeletal vibrations of PHA hydrocarbon chain. FTIR spectra of neat mcl-PHA and (PHA-PV-T80) composites at room temperature; (a) high PV and (b) low PV concentration with increasing T80 amount. Solid and dashed arrows indicate the presence of T80 and perovskite in the PHA composite, respectively.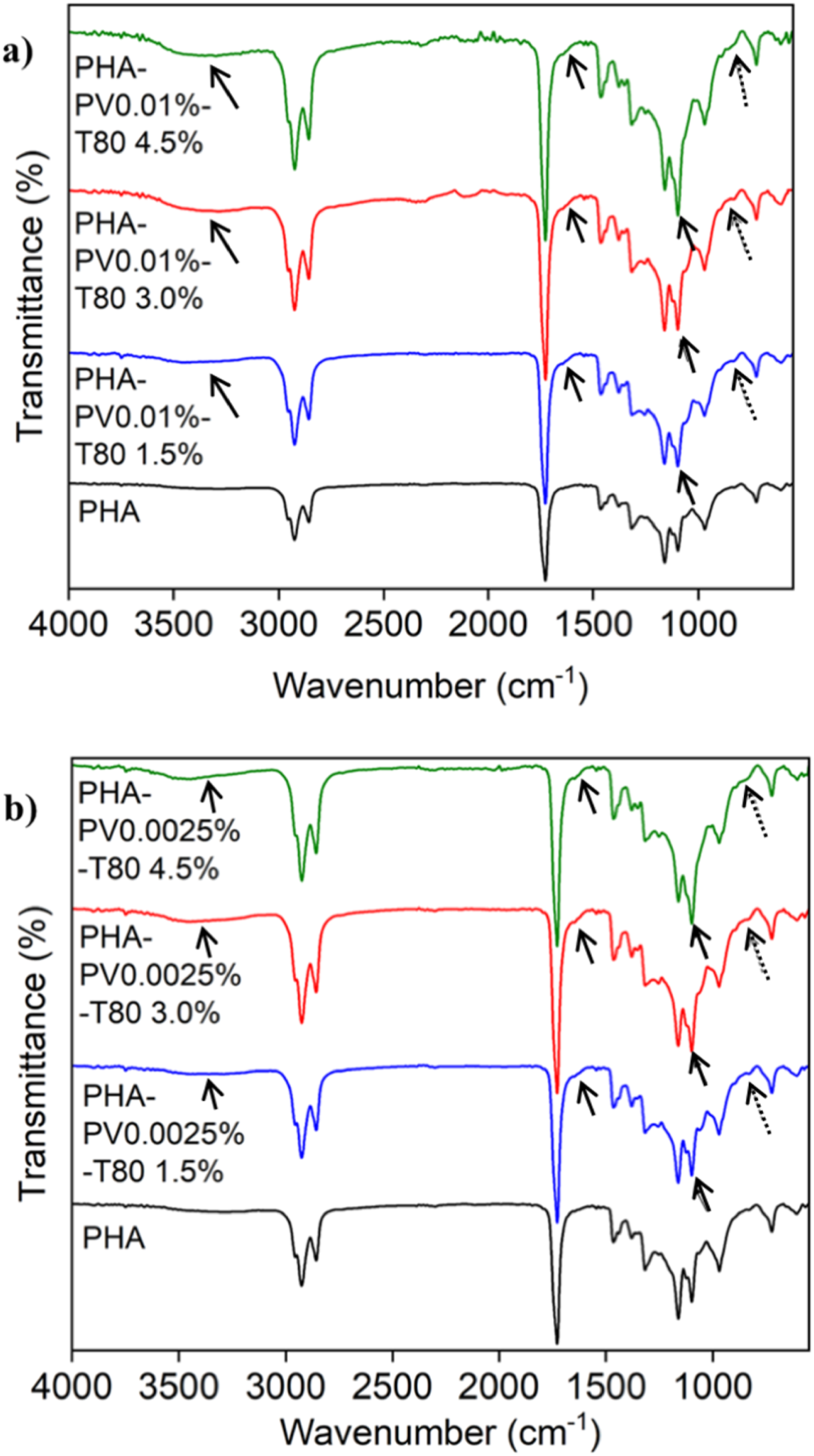
Increasing intensity of peak 1050 cm−1 and presence of big hump around 3700 cm−1, as shown by solid arrows in Figures 2(a) and (b), indicate the stronger presence of T80 in the PHA-PV-T80 composites. The former represents the ester bonds while the latter represents the hydroxyl groups of the T80 hydrophilic head. In addition, the unsaturation (C = C) of T80 alkyl tail can be seen from the tiny shoulder at 1650 cm−1 (shown by solid arrow). 37 Metal oxide bands usually appear within the range of 500 – 800 cm−1 depending upon the perovskite phase. 38 Broader peaks at ≈ 800 cm−1 were observed as shown by dashed arrow in Figures 2(a) and (b); attributed to the intercalation of perovskite particles within the PHA matrix. A more pronounced difference is evident in Figure S2 of Supporting Information.
XRD analysis of PHA-PV-T80 composites
Neat mcl-PHA and PHA-PV-T80 composites exhibit two diffraction peaks viz. 19.5° and 21°, as shown in Figure 3. These peaks represent the side-chain crystallization of PHA, which occurs in the rigid regions within the polymer matrices.5,39 It is suggested that higher concentration of perovskite within PHA-PV-T80 composites promote greater side-chain crystallization at room temperature. This is evident from significant difference in peak intensity at 19.5°−21° in PHA-PV0.01%-T80 (Figure 3(a)) compared to PHA-PV0.0025%-T80 (Figure 3(b)). The greater promotion of side-chain crystallisation would result in the creation of more rigid regions within the polymer matrix, thereby providing support for the hypothesis stated in the previous discussion of PHA-PV-T80 composite films preparation. The intensity of the peaks corresponding to the SrTiO3-type perovskite is reduced in the XRD spectra (Figure 3(a) and (b)) because of the small quantity incorporated in the polymer composite. SrTiO3-type perovskite yields 2θ = 22.5°, 32°, 40°, 46.6°, 57.6°, 67.7° and 77.3° as shown in Figure S3, Supporting Information. Assignments of these peaks are corroborated by previous studies.38,40 The interplanar of the perovskite is 3.91 Å, 2.76 Å, 2.25 Å, 1.95 Å, 1.59 Å, 1.38 Å and 1.24 Å, similar as reported in other publications using SrTiO3.40–42 Crystallite size SrTiO3 was calculated using the Debye Scherer formula: X-ray diffractograms of neat mcl-PHA and PHA-PV-T80 composites at room temperature; (a) high PV concentration and (b) low PV concentration with increasing T80 amount.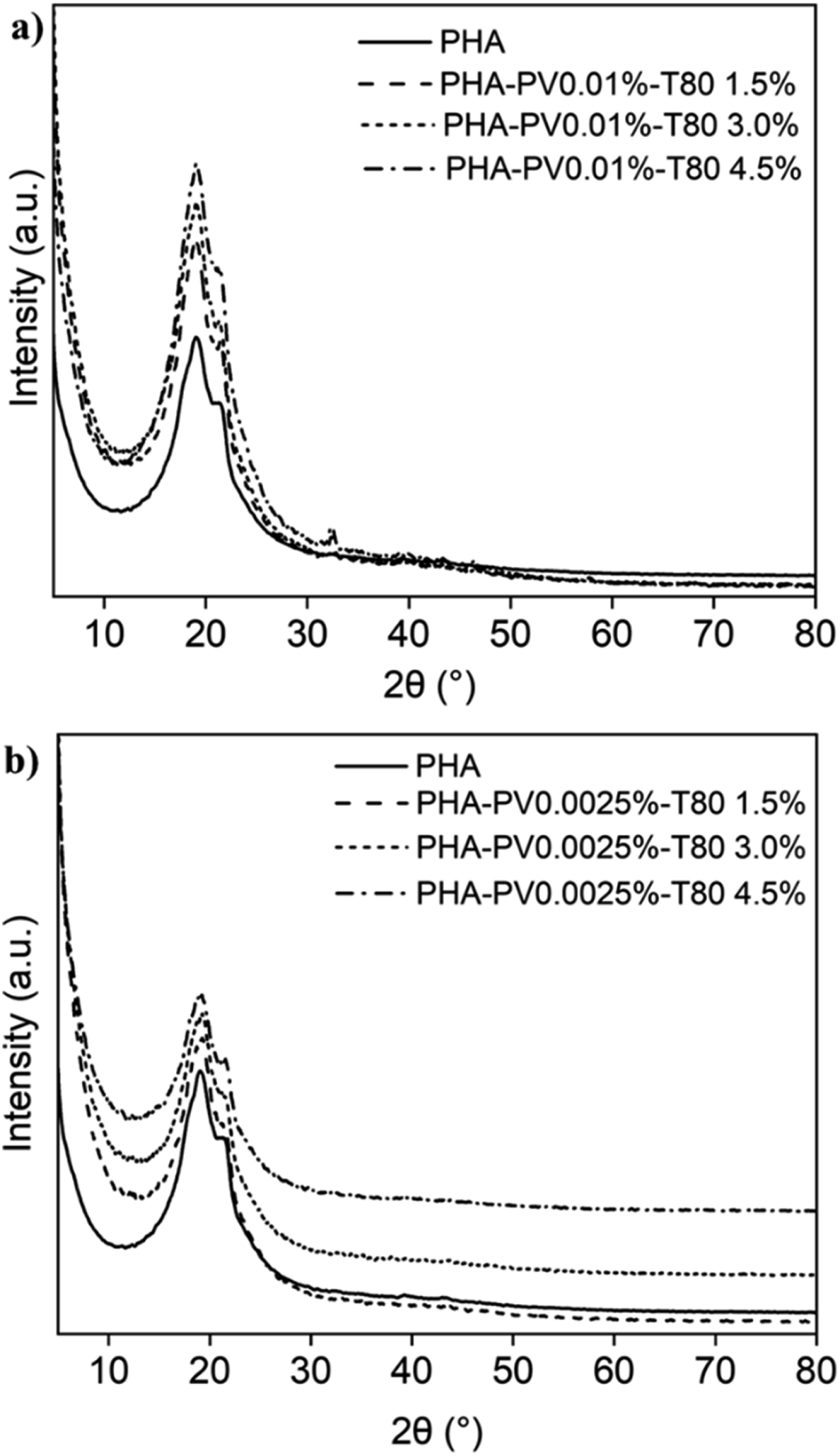
FESEM analysis of PHA-PV-T80 composites
Homogenous dispersion of perovskite (PV) and Tween 80 (T80) within film matrix can be observed from the FESEM images of PHA-PV-T80 composites in Figure 4; indicating the utility of ultrasonication dispersion technique applied in the fabrication process. Observable aggregation of powder or liquid phase is absent in the images. Generally, all composites exhibit non-uniform surface morphology compared to the smooth and uniform neat mcl-PHA
43
owing to broader presence of rigid regions within the composite matrix. It is worth noting that a more textured morphology was observed in the PHA-PV0.01%-T80 4.5% sample (Figure 4(e)); attributed to the extensive side-chain crystallization concomitantly the increase in rigid regions, as previously discussed. The maximum feasible amounts for incorporation into the composite without unwanted aggregation were determined at 4.5 % for T80 and 0.01 % for PV. These values provided the upper limits for subsequent experimental procedures. FESEM micrographs with higher magnifications are provided in the Supplementary Information (Figure S4– S6). FESEM micrographs of PHA-PV-T80 composites observed at 25°C; (a) PHA-PV0.01%-T80 1.5%, (b) PHA-PV0.0025%-T80 1.5%, (c) PHA-PV0.01%-T80 3.0%, (d) PHA-PV0.0025%-T80 3.0%, (e) PHA-PV0.01%-T80 4.5% and (f) PHA-PV 0.0025%-T80 4.5%.
Thermal analysis of PHA-PV-T80 composites
Two glass transitions evident in the DSC thermograms at ≈ −50 and 10°C (denoted as Tg1 and Tg3, respectively) were ascribed to the base polymer, i.e., mcl-PHA.
5
One additional glass transition (denoted as Tg2) at temperature range of −24 to −17°C was found in the PHA-PV-T80 composites only (Figure 5). It is hypothesized that incorporation of PV in the composites resulted in the increase of rigid regions across the polymer matrix, thus making the composite films sturdier while allowing the PHA as base material to retain some degree of flexibility. The melting temperature (Tm) of PHA-PV-T80 showed slight changes as compared to neat mcl-PHA (Figure 5(a) and (b)), which is suggested to arise from the effect of T80 intercalation in-between PHA chains, thus resulted in earlier melting process of the rigid regions. The presence of unsaturated alkyl chains in T80 may lead to a decrease in the PHA side chains interaction crucial for retaining the integrity among its main chains during temperature increase. DSC thermograms of neat mcl-PHA and PHA-PV-T80 composites; (a) high PV and (b) low PV concentration with increasing T80 amount.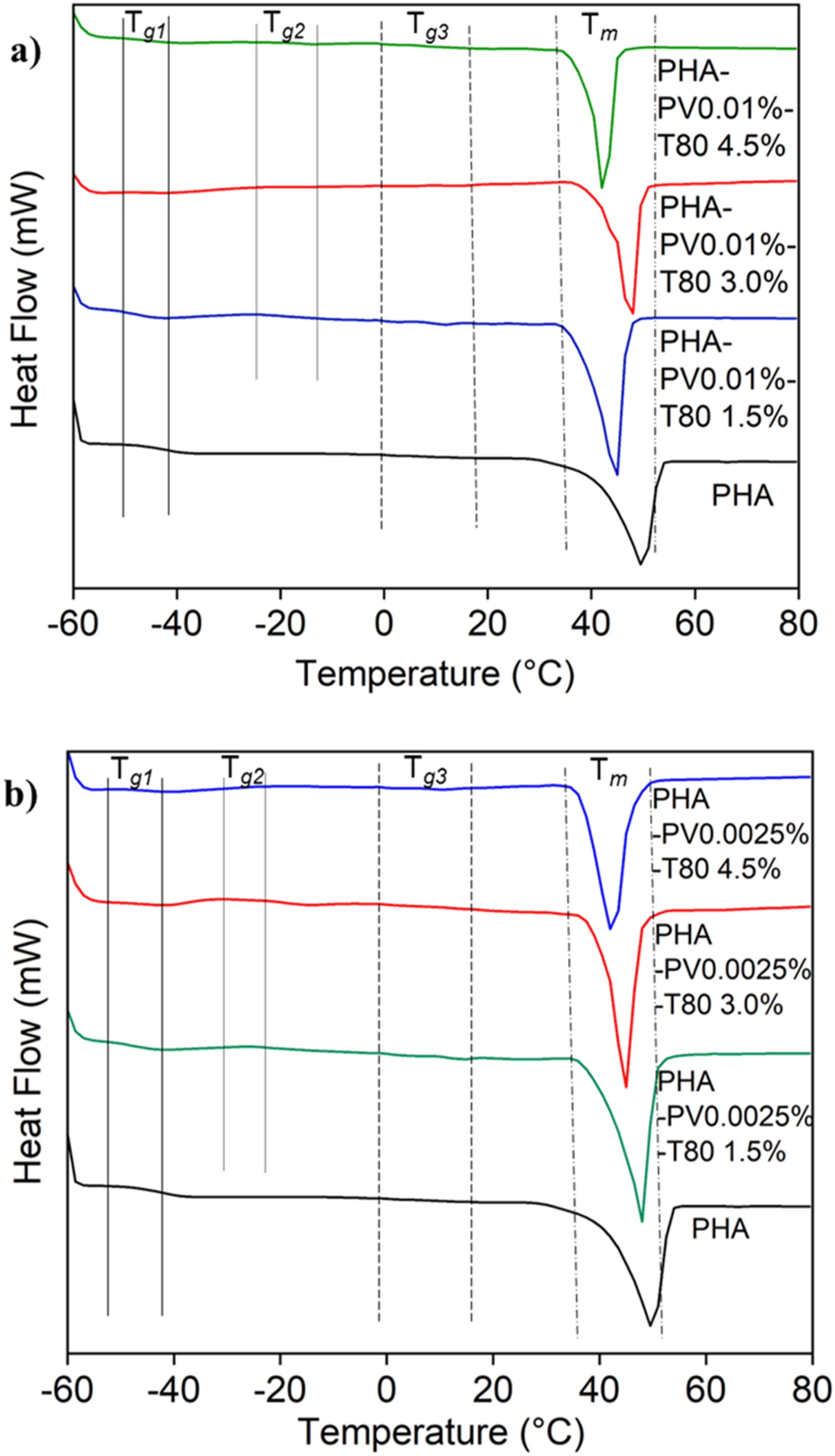
Figure 6(a) and (b) show the results of thermogravimetric analysis for neat mcl-PHA and PHA-PV-T80 composites. Neat mcl-PHA degrades at a temperature of 250°C, which agrees to a previous study.
39
Meanwhile PHA-PV-T80 composites were observed to have a slightly higher thermal degradation temperature (T
d
) from 260 to 275°C, indicating that the blending of PHA with surfactant and perovskite improved its thermal stability at higher temperature. A second-step TGA curve observed in all PHA composites is attribute to the degradation of T80, which degrade much later than PHA at around 370°C. Thermal weight loss profile for neat mcl-PHA and PHA-PV-T80 composites; (a) high PV concentration and (b) low PV concentration with increasing T80 amount.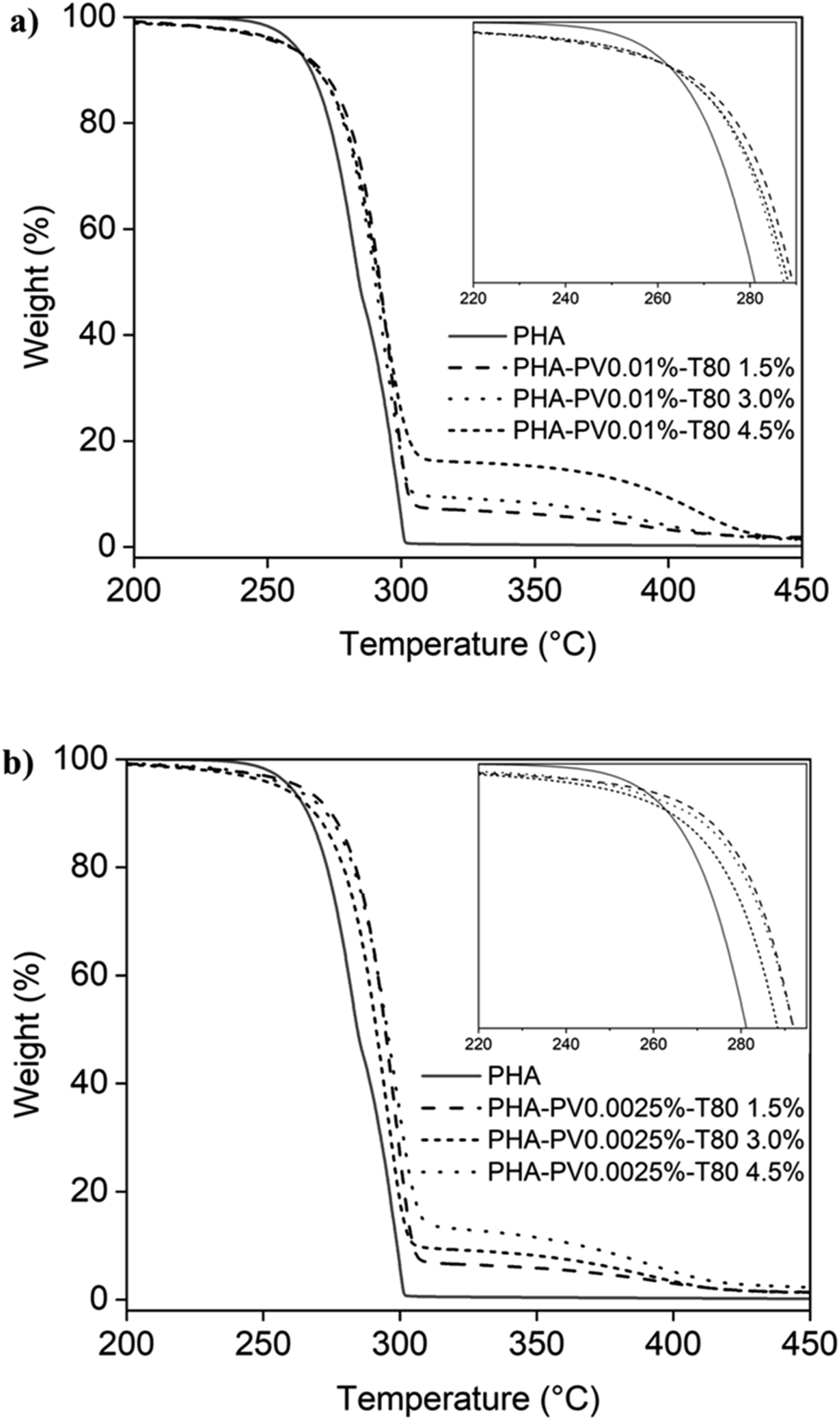
Based on spectroscopic-, X-ray diffraction, morphological- and thermal studies, it can be concluded that the fabricated composite films exhibit good physical characteristics for potential application as membrane material.
Proton Flux Analysis
Figure 7 shows the comparison of proton transfer in ultrapure water through PHA-PV-T80 composite films, and the proton flux (×10−4 mol.min−1.cm−2) for each film (Table 1). Neat PHA film, Nafion and PHA composites incorporated with Span 80 (SP80) was also tested for comparison. It can be seen that composite with T80 can transport protons at a higher efficiency as compared to other tested films, including neat mcl-PHA and Nafion (standard membrane used as control). It is suggested that extensive molecular interaction between the big hydrophilic head group (polyethoxyl) of T80 and perovskite, would form many proton-hopping sites. In contrast to T80, adding Span 80 (S80) as the surfactant in the composite films did not yield a significant result in proton flux as compared to neat mcl-PHA (Table 1 and Figure 7). This is probably due to the smaller head group of S80 that does not form many proton-hopping sites in the PHA-PV-S80 composite film, thus giving a significantly lower proton flux value than that of T80. pH changes profiles by using neat PHA, Nafion and different PHA composites as proton exchange membranes in ultrapure water medium. PHA composites with (a) high and (b) low perovskite amounts were tested. Proton transfer of PHA-PV-T80 and PHA-PV-S80 composites in ultrapure water (standard deviation <5 %).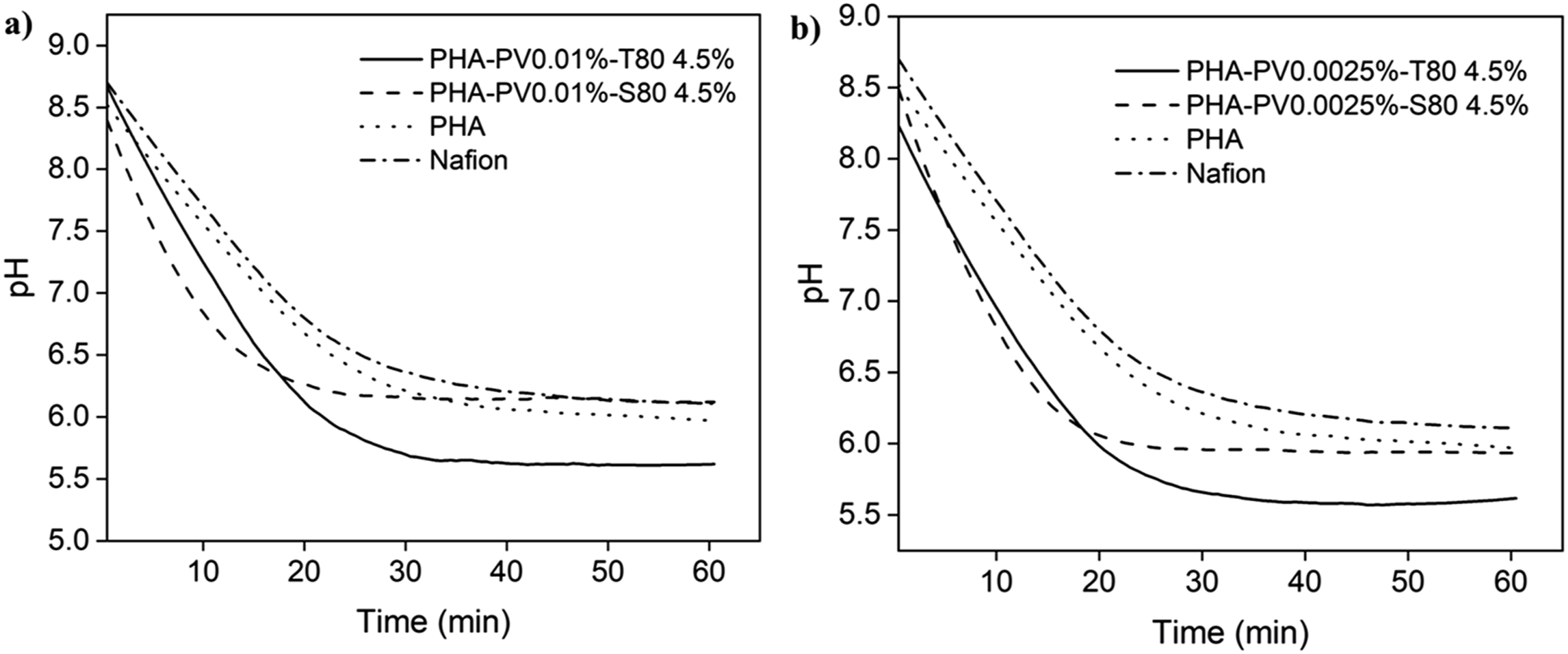
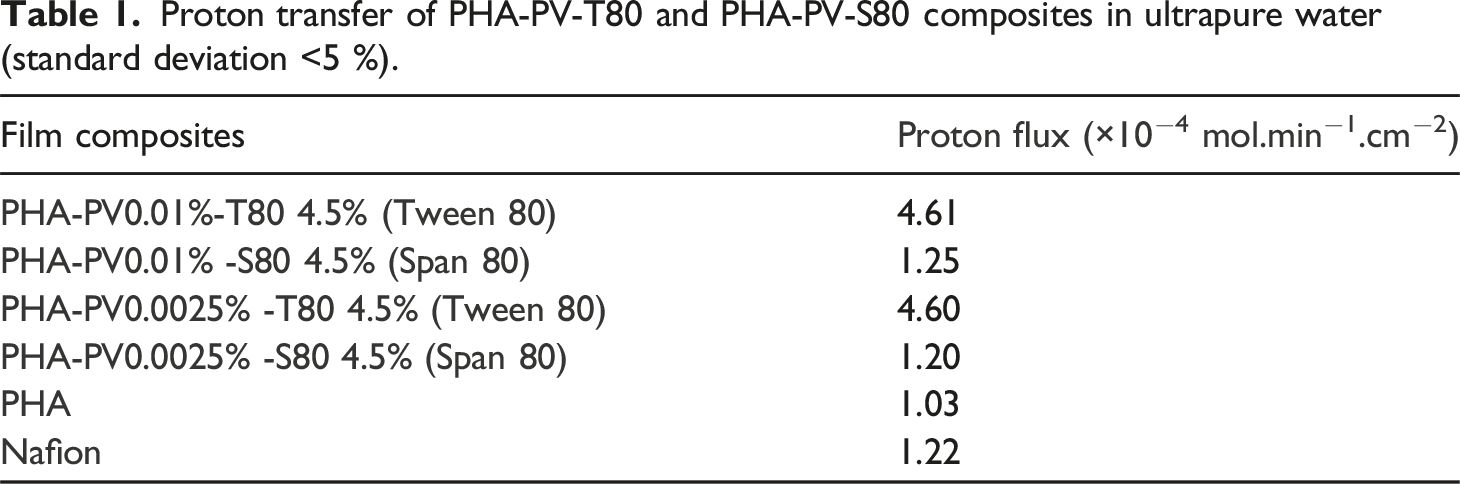
The ability of neat mcl-PHA to conduct limited proton transfers is attributed to hydrogen bonding between the hydronium ion (H3O+) with carbonyl (C = O) and ether (C-O-C) groups of ester bonds present in the mcl-PHA structure. However, the pH changes reached plateau at 6.3 with neat PHA compared to PHA composite with T80, pH 5.5; lesser number of proton hopping sites present in pristine mcl-PHA is hypothesized to be responsible for the observation made.
From Figure 8, it can be observed that different proton fluxes are obtained for each membrane when tap water was used as the conduit medium compared to ultrapure water. This is probably due to tap water containing other ions such as calcium, sodium, magnesium, potassium etc.,
44
that enhance proton transfer (Table 2). Nevertheless, it has been shown that PHA-PV-T80 composites are still able to transfer protons efficiently due to the selectivity of perovskite and surfactant present. pH changes profiles by using neat PHA, Nafion and different PHA composites as proton exchange membranes in tap water medium. PHA composites with (a) high and (b) low perovskite amounts were tested. Proton transfer of PHA-PV-T80 and PHA-PV-S80 composites in tap water (standard deviation <5 %).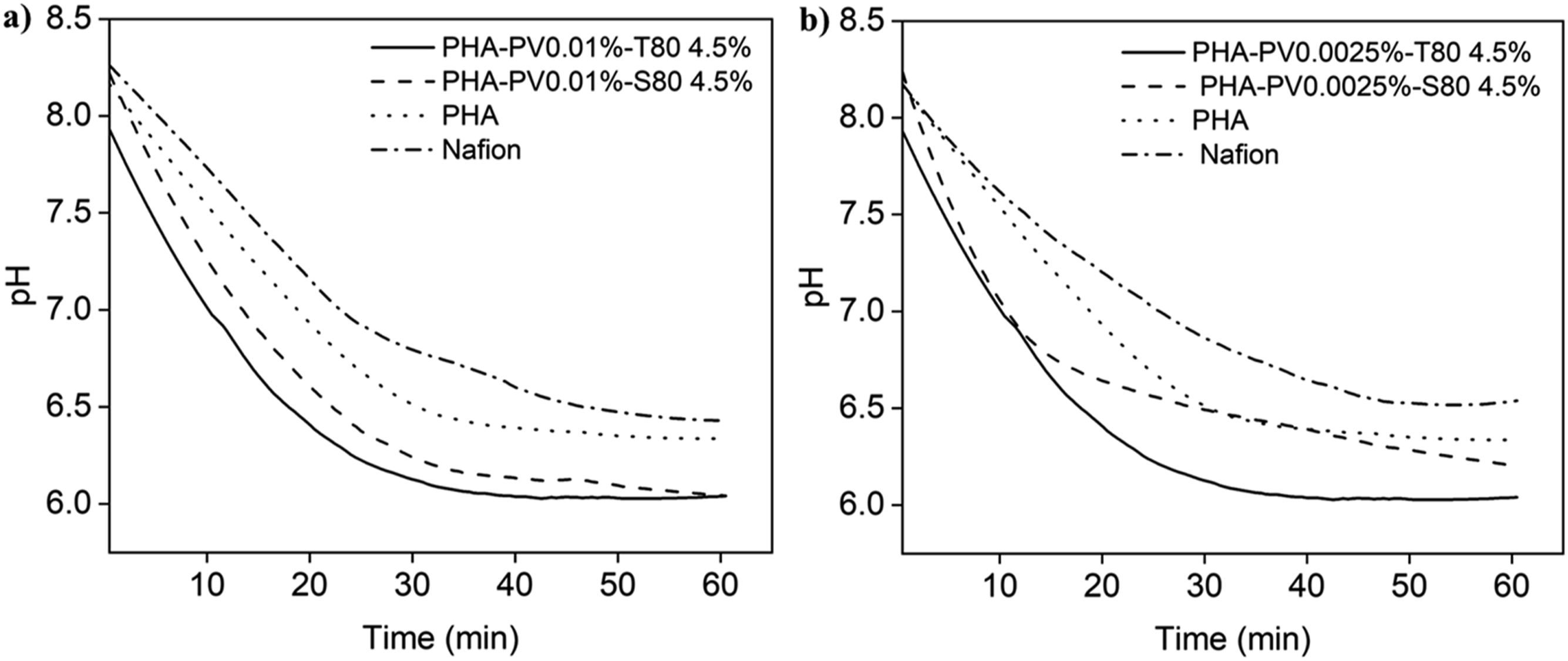
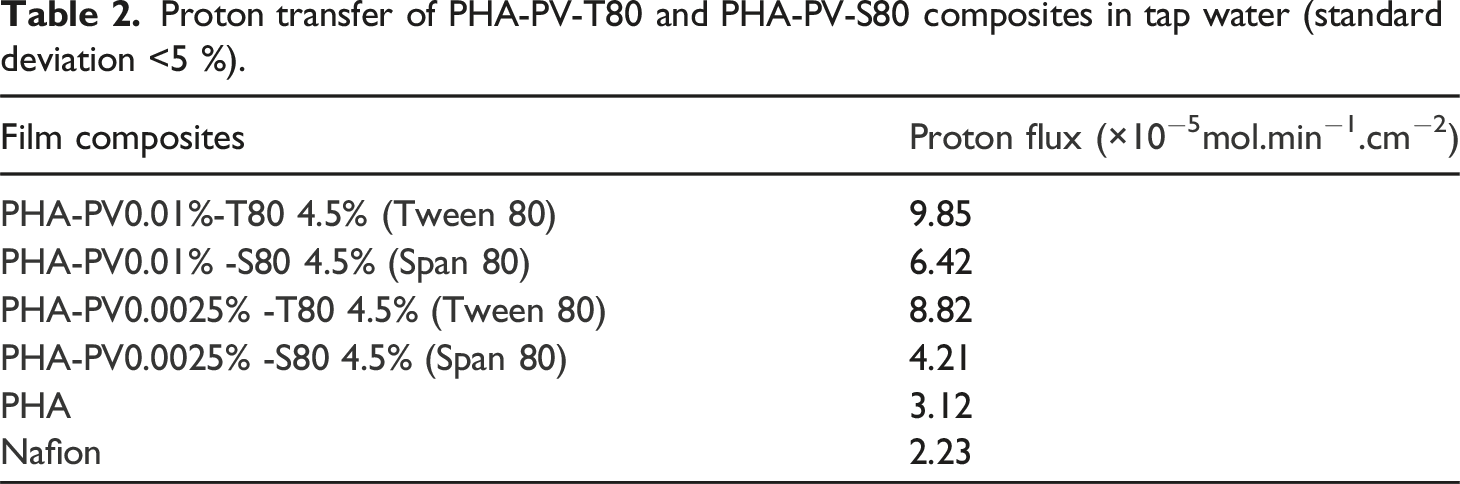
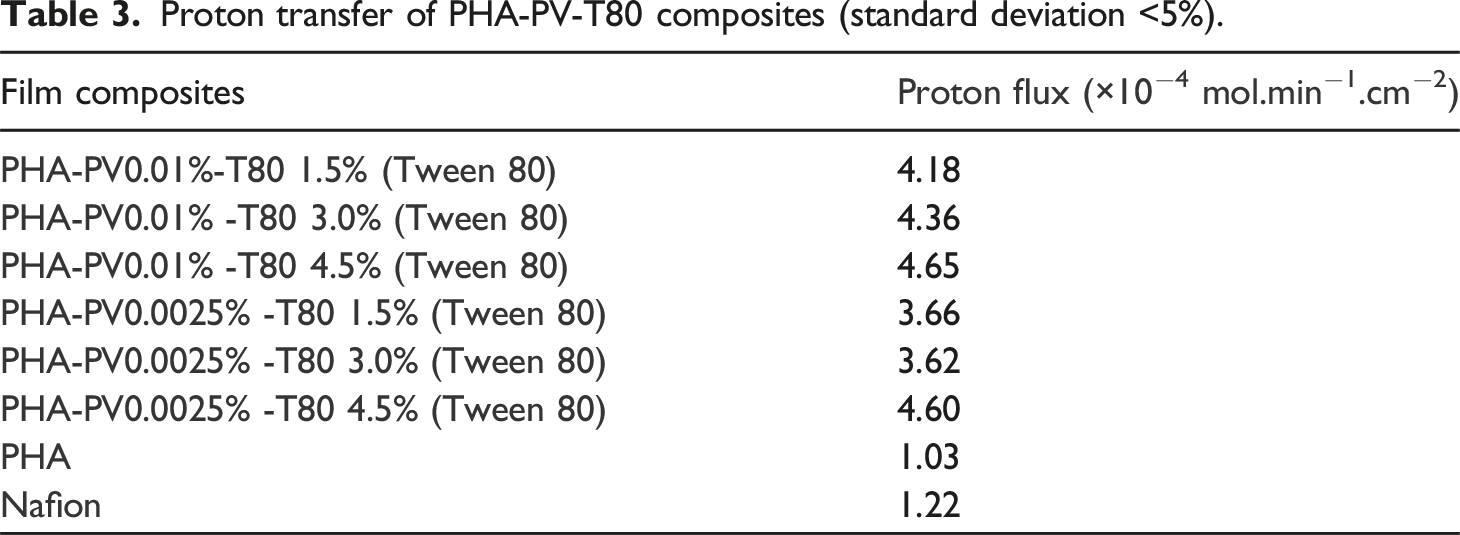
Figure 9 shows the efficiency of proton transfer through PHA-PV-T80 composite films in ultrapure water and the proton flux (×10−4 mol.min−1.cm−2) for each film (Table 3). It is observed that increasing the quantity of T80 in the composite preparation resulted in an elevation of the proton flux. However, the augmentation of the perovskite quantity yielded only slight variation in the proton flux. The observation highlights the significance of T80 within the context of the PHA proton exchange membrane. PHA composite with the highest concentration of surfactant (T80) exhibited the most efficient proton transfer attributed to greater number of interaction sites for hydrogen bonding on hydrophilic head group (polyethoxyl) of T80 and perovskite. pH changes profiles by using PHA-PV-T80 composites as proton exchange membrane in ultrapure water medium. PHA composites with (a) high and (b) low perovskite amounts were tested. Proton transfer of PHA-PV-T80 composites (standard deviation <5%).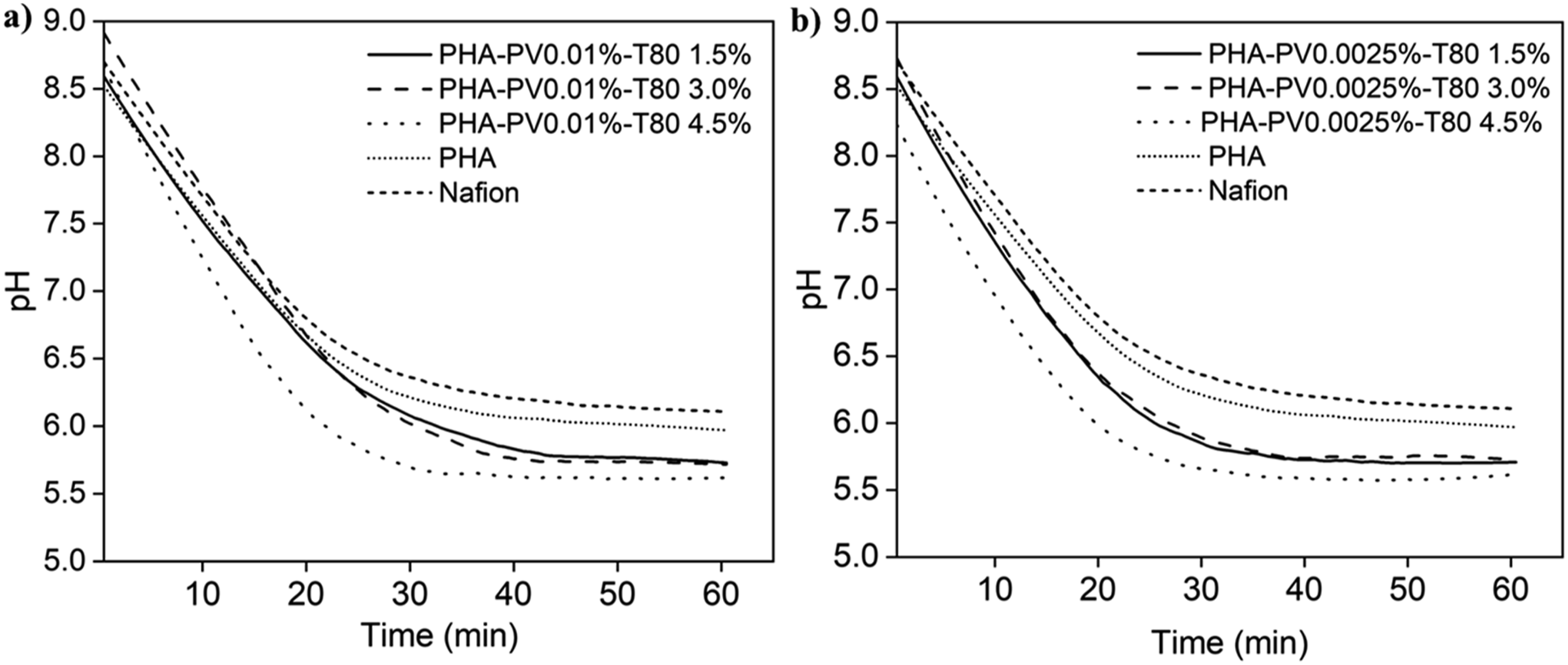
PHA-PV-T80 composite film as a proton permeable membrane
From the results, proton movement across the membrane thickness is suggested to be determined by the physicochemical properties of PHA-PV-T80 membrane. An optimal composited quantity of T80 and perovskite allow for a characteristic film well-suited for membrane application. As proposed earlier, T80 facilitates the uniform distribution of perovskite within the hydrophobic PHA matrix. Both compounds are hypothesized to intercalate among the main chains of PHA, composed of a sequence of ester bonds (Figure S7 of the Supporting Information). Consequently, the intercalation promotes formation of rigid regions within the PHA matrix, imparting some degree of stiffness to the composite film (Figure 10). Furthermore, the compositing of surfactant (Tween 80) and perovskite (SrTiO3) in the mcl-PHA film also imbues hydrophilic properties to the composite membrane from the surfactant head (polyethoxylated sorbitan) as well as oxygen vacancies found in the perovskite nanoparticles. Consequently, large number of water molecules are partitioned towards the membrane surface, and penetrate the composite film layer, creating an extensive network of hydrogen bonding with the composite matrix. Schematic hypothesis of proton transfer mechanisms across poly-3-hydroxyalkanoate−Perovskite−Tween 80 (PHA-PV-T80) composite film.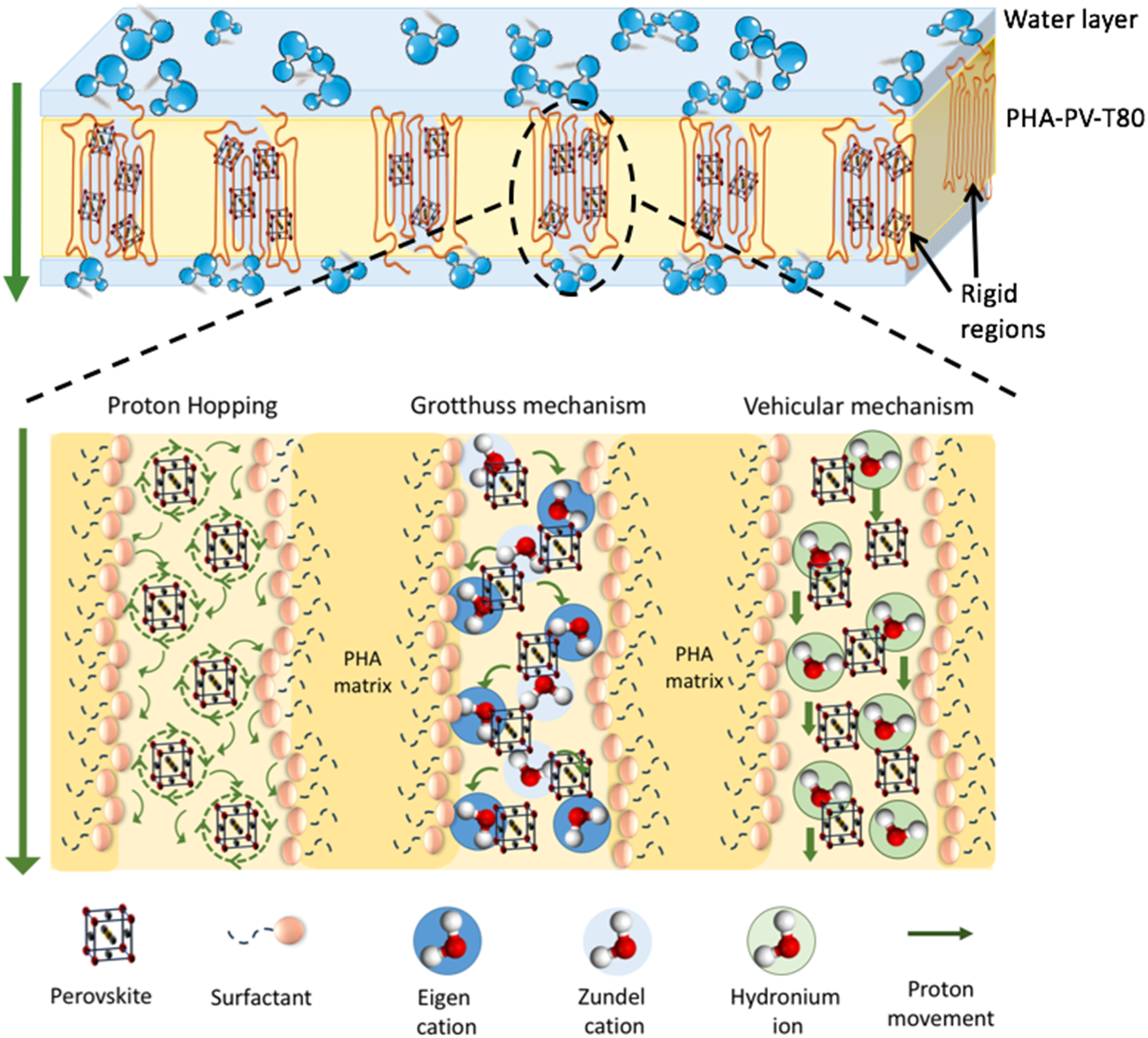
It is hypothesized that the movement of protons primarily occurs within the rigid regions, which are predominantly constructed from the presence of perovskite and surfactant. As illustrated in Figure 10, proton movement across the PHA-PV-T80 membrane (represented by green arrow) is suggested to occur via combination of three theorized mechanisms, i.e., Proton hopping, Grotthuss and Vehicular mechanisms
45
; (1) Proton hopping could potentially occur between adjacent oxygen of perovskite and ether (C-O-C) and carbonyl (C = O) groups of surfactants. Rotational diffusion and intra-octahedra proton hopping in trap region areas are possible migration paths used by the perovskite to transport protons. Rotating motion of the protons around the oxygen groups is fast, thus proton reorientation occurs at the next oxygen before transfer.
30
(2) The Grotthuss mechanism describes proton hopping/movement via a network of hydrogen bonding in hydronium ions as the intermediate state H5O2+ (Zundel cation) and H9O4+ (Eigen cation) in water molecules, playing the role as proton donor and proton acceptor, respectively, in the proton migration series.
46
(3) Vehicular mechanism describes direct proton movement in the state of hydronium ions (H3O+) within water molecules similar to diffusion.
47
Based on the results obtained from DSC and TGA, as shown in Figure S8 and S9 of the Supporting Information, it can be concluded that the thermal properties of the PHA composite films remained unaffected under the study conditions. It implies the absence of irreversible physical/chemical modification or impairments of the composite membranes structure subsequent to the proton transfer experiment. This demonstrates the potential of the PHA-PV-T80 membrane to be utilized as a proton exchange membrane in low-temperature fuel cell application, specifically in microbial fuel cell.
Conclusion
A facile preparation of medium-chain-length polyhydroxyalkanoate (mcl-PHA)-perovskite (PV)-Tween 80 (T80) film composites exhibited excellent proton permeability and proton flux characteristics when tested in an aqueous environment. Therefore, the PHA nanocomposite shows a good potential for applications such as proton exchange membrane. The integrity of the composite membranes remained unaffected following the proton transfer experiment, indicating a good physical attribute. The composite preparation may fulfill the need for an eco-friendly membrane material, for example, for water purification via filtration.
Supplemental Material
Supplemental Material - Fabrication of SrTiO
3
−Tween80 reinforced biological poly-3-hydroxyalkanoate composite film and its proton permeability
Supplemental Material for Fabrication of SrTiO3−Tween80 reinforced biological poly-3-hydroxyalkanoate composite film and its proton permeability by Nur Amirah Faizal, Khairul Anwar Ishak and Mohamad Suffian Mohamad Annuar in Journal of Thermoplastic Composite Materials.
Data availability statement
All data generated or analyzed during this study are included in this article.
Footnotes
Acknowledgements
The authors acknowledge Centre for Fundamental and Frontier Sciences in Nanostructure Self-Assembly (FSSA) and Low Dimensional Materials Research Centre (LDMRC), Universiti Malaya for providing the lab. and instrument facilities.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Higher Education under the Fundamental Research Grant Scheme (FRGS/1/2020/STG01/UM/01/1). The Universiti Malaya also partially supported this research through UM International Collaboration grant (ST021-2022).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.