Abstract
Agricultural waste shells are another source of calcium carbonate (CaCO3). Waste shells contain natural polymers such as polysaccharides and protein which can react with active groups in a polymer by melt mixing to form a compatible layer in a composite material. The formation of a compatible layer helps to improve the compatibility and material toughness of a composite. At the same time, the presence of CaCO3 in waste shells can also help to improve the hardness of a material. Therefore, this work mainly focused on the preparation of composite materials using different kinds of shell powder, and also studied the effects of the interface structure between shell powder and polycaprolactone (PCL) and properties of the composite materials. In particular, this work, studied the effects of the crystal forms of various shell powder as well as the content of polysaccharide and protein on the tensile performance and thermal properties of composite materials. The results implied that the tensile performance of the composites prepared from different kinds of shell powder were slightly different. The composite prepared from shell powder containing aragonite-structured CaCO3 had a better elastic modulus than the composite prepared from shell powder containing calcite-structured CaCO3. Likewise, the composite material prepared from scallop shell powder had the best toughness and thermal properties. This work has laid a foundation for exploring the effects of all sorts of shell powder on the structure and properties of composite material.
Keywords
Introduction
Shells are not only diverse in nature, but also complex and changeable in shape and structure. 1 They are widespread in mollusks and are important biomineralized materials. Among the common shells available are oysters, scallops, conch, scapharca subcrenata, razor clam, clam shell, corbicula shell, etc. 2 The main compositions in shells are calcium carbonate (CaCO3) and organic material, however, the content of CaCO3 in shells is about 95% while the organic matter component just about 5%. The CaCO3 mainly divided into calcite calcium carbonate and aragonite calcium carbonate crystal forms, of which the latter is less stable to temperature and pressure. 3 The organic matter component in shells includes soluble and insoluble proteins, carbohydrates (such as chitin and glycoproteins) and lipid (such as fatty acids and cholesterol). The function of the organic matter component is to regulate the formation, transformation and growth of calcium carbonate crystal nuclei in the process of shell bio mineralization, and ultimately determine the microstructure of the shell. 4 Varying types of shells have different crystal forms of CaCO3 as well as the contents of organic matter which can have a great influence on the thermodynamic and tensile performance of composite materials prepared from shells. There are also differences in the structure and properties of varying types of shells.5–11
In the 1950s and 1960s, researchers conducted in-depth research on the bio minerals of most shells of fishes and had a relatively complete understanding of the structural characteristics of most shells of fishes. 12 In the 21st century, scientists from many disciplines conducted research into shells of fishes. For instance, Haidao Chen 1 observed the structure of thirteen (13) species of bivalves from six (6) families under the scanning electron microscope. The results illustrated that there were certain differences in the surface and cross-section of the microstructures of different types of shells mainly in the composition and arrangement of the crystals. According to the results on surface morphological characteristics, the cuticle of the shell is divided into five (5) types, namely smooth and flat, irregular polygon, granular, honeycomb and gully. 12 In addition, the multi-level structure in various shells was mentioned in the research of Hou Xue. 13 Different crystal structure and its arrangement will cause the difference of shell hardness while the organic components in the shells are the most important factors in the toughening mechanism. 14 Furthermore, Zuopeng Lin 15 found that shell powder can still maintain its porous structure after calcining at 500–900°C. The results showed that they had a porous inner structure with many small pores constituting a total pore volume of 82%. It was also found that the pores and specific surface area of oyster shells increased after calcination at 900°C for 2 h. The characteristics of the microstructure of shells from different families are various and are likely to affect the compatibility and hardness of composite materials.
In our previous work, shell powder was melt-blended with polycaprolactone (PCL) and utilized PCL-based polyurethane prepolymer (PCLPU) as a compatibilizer.16,17 Because PCL is an excellent renewable plastic raw material with great mechanical properties, and shells are biomass resources with high strength. Therefore, the initial combination of the two has achieved complementary advantages, and the addition of compatibilizer also achieved good interface compatibility of the composite material obtained. However, varying kinds of shell powder have different crystal forms as well as the content of the organic matter component, what’s more, the tensile performance of the composite materials prepared from different kinds of shell powder may also be distinct. In this work, different kinds of shell powder were used to prepare a series of shell powder-polycaprolactone-based (SP-PCL) composites to study the effects of various kinds of shell powder on the structure and properties of PCL-based composites.
Experimental
Materials
Diphenylmethane-4,4′-diisocyanate (MDI, CAS no,101–68-8) was acquired from Sigma-Aldrich Fine Chemicals (St. Louis, MO). Polycaprolactone (PCL) and PCL diols were received from Perstorp UK Ltd with M w of 50,000 g mol−1 and 2000 g mol−1 respectively. Six different shells (200-mesh) including oyster scallop, clam shells, razor clam, scapharca, conch were collected from the waste discarded by the seafood stores in Shandong province, China.
Synthesis of PCLPU
In order to prepare PCL diols-based polyurethane prepolymer with -NCO groups at both ends (named PCLPU), the molar ratio of -NCO to -OH was set to 2.0. At first, 80.0 g of PCL diols was added to a three-necked flask (250 mL), and after it was melted to a liquid state (around 60°C), stirring at 300 r/min was set. The moisture content in the PCL diols was removed by drying the system in a vacuum at 110°C for 30 min. Subsequently, the temperature of the system was decreased and then 20.0 g of MDI was charged when it reached to 60°C. The mixture in the flask gradually turned from translucent to transparent. After 15 min of adding the MDI, vigorous stirring of the mixture was continued. The mixture was then kept at 80°C for 1 h, and finally white PCLPU was obtained. Figure 1(a) exhibits the preparation process of PCLPU. (a) The preparation process of PCLPU; (b) Clam shell; (c) PCL particles.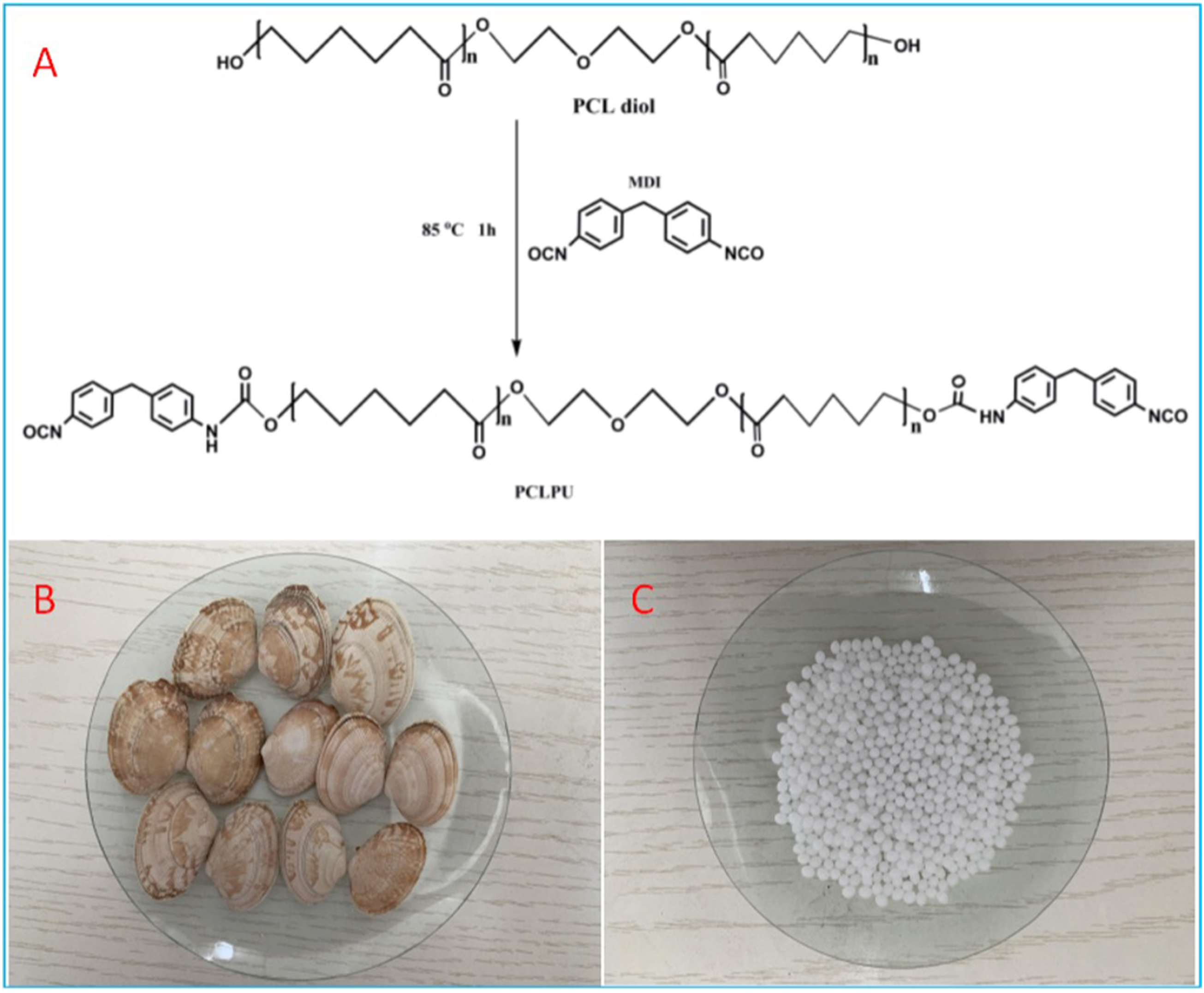
The preparation of shell power and composites
Related names and photos of the six types of shells.

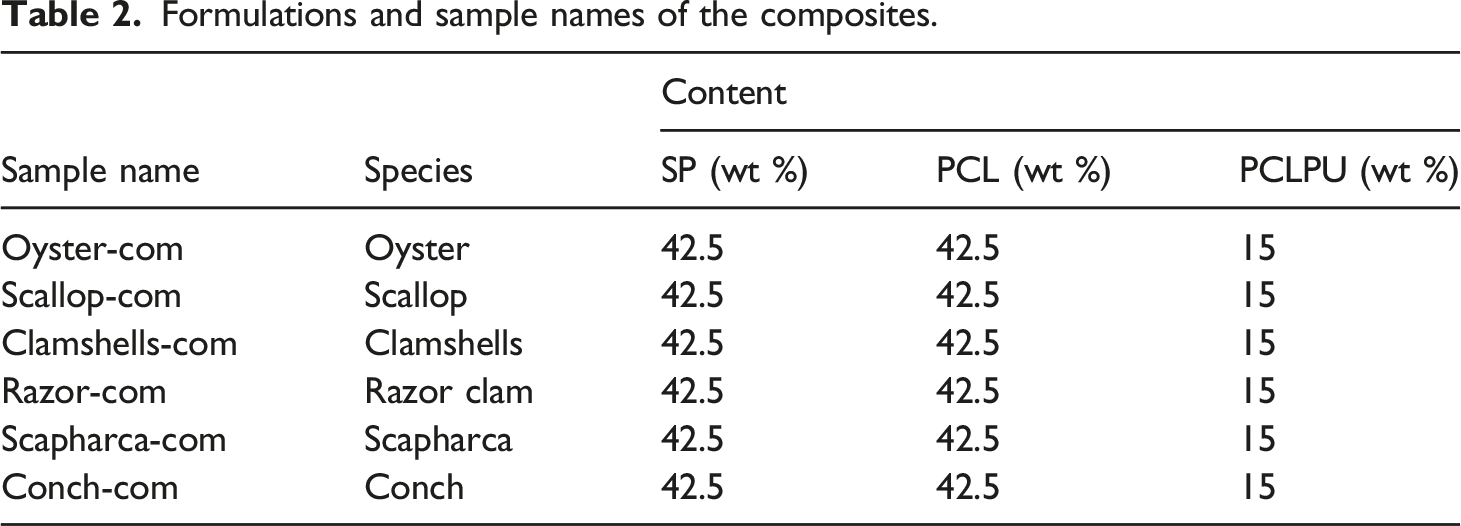
Formulations and sample names of the composites.
The SP-PCL composites were compression-molded at 100°C for 10 min with the hot pressing pressure of 20 MPa by R3202 hot press (Wuhan Qien Science and Technology Co, Ltd, Wuhan, China) to obtain composite sheets with average thicknesses of 1.0 mm. And then the GB/T1040-2006 standard was implemented to cut the resulting sheets into 5A type dumbbell shapes. (Figure 2 shows the dumbbell-shaped samples of the prepared composite). The dumbbell-shaped sample of shell-based composites.
Characterization
The materials were identified by a Nicolet 6700 fourier transform infrared (FT-IR) spectrometer instrument (Thermo Nicolet, Madison, WI, USA).16,18
Frozen-fractured cross-sections of the samples were observed using scanning electron microscopy (SEM. JSM-5610LV, Japan).16,18
X-Ray diffraction (XRD) was performed by a XRD instrument (D8 Advance, Bruker Instrument, GER.) using Cu-Kα radiation (1.542 nm) at 2θ values ranging from 10° to 80°, and cathode at 40 kV and 40 mA.16,18
The differential scanning calorimetry (DSC) was conducted in a nitrogenous atmosphere by a DSC analyzer (TA-DSC2500, USA). The samples of 10 mg were cooling from room temperature to −50°C at a rate of 10°C·min−1, and kept at −50°C for 2 min, then heated to 280°C. The melting point (T
m
) and melting enthalpy value (∆H
m
) were then determined. The following equation was used to calculate the degree of crystallinity (X
c
):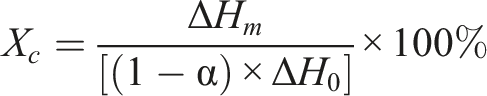
The melting enthalpy value of PCL for complete crystallization is 139.5 J/g expressed as ΔH 0 . α is the mass fraction of filler in the composite material.16,18
Thermogravimetric analysis (TGA) was measured under a nitrogen atmosphere by STA 449 C analyzer (NETZSCH Instruments Inc. MA, USA). The specific conditions were as follows: the heat rate was 10°C min−1 and the temperature range was 40–900°C.16,18
The ASTM D882-18 standard test method for tensile characterization was carried out to evaluate the elongation at break (єb) and tensile strength (σb) of these composite samples by CMT6503 test machine (Shenzhen SANS Test Machine Co Ltd).16,18 The dumbbell-shaped sheets for tensile performance were placed at room temperature to equilibrate for 1 week before was tested.
Results and discussion
Chemical analysis
Figure 3 shows the FT-IR results for the six kinds of SP and the SP-PCL composites prepared. It is obvious from Figure 3(a) that the different kinds of SP have similar characteristic absorption peaks, indicating that the main components of the various shells are roughly the same. The characteristic peaks of SP raw materials are 706 cm−1 (CO32-, symmetric), 860 cm−1 (CO32-, asymmetric), 1078 cm−1 (CO32-, symmetric), 1446 cm−1 (CO32-, asymmetric), 2522 cm−1 (CO32-, asymmetric), 2918 cm−1 and 3305 cm−1 (-OH).16,19-21 The prominent absorption peak at about 3450 cm−1 overlapps with the N-H stretching of polysaccharides or proteins (NH-absorption band at 3260 cm−1) and assign to the O-H stretching vibration. In addition, the peak at 2918 cm−1 corresponds to the C-H stretching vibration,
16
while the peak at 1790 cm−1 represents the C = O stretching vibration.
21
Similarly, 1240 cm−1 and 1500 cm−1 represent the stretching vibrations of N-H.20,22 Literature report suggests that the band at 1789 cm−1 corresponds to C = O stretching vibration and N-H in-plane bending
21
of the organic matter component of the shell powder. It can be seen from the Figure 3 that although the types of shell powder are various, the IR peak positions of the organic matter groups are roughly the same, it can be presumed that the shell powder samples contains natural polymers with hydroxyl groups or -NH- groups. It is reported in the literature that the band at 1789 cm−1 corresponds to the C=O stretching vibration and the N-H in-plane bending vibration. (a) IR images of six kinds of SP; (b) IR images of SP-PCL composites prepared by SP.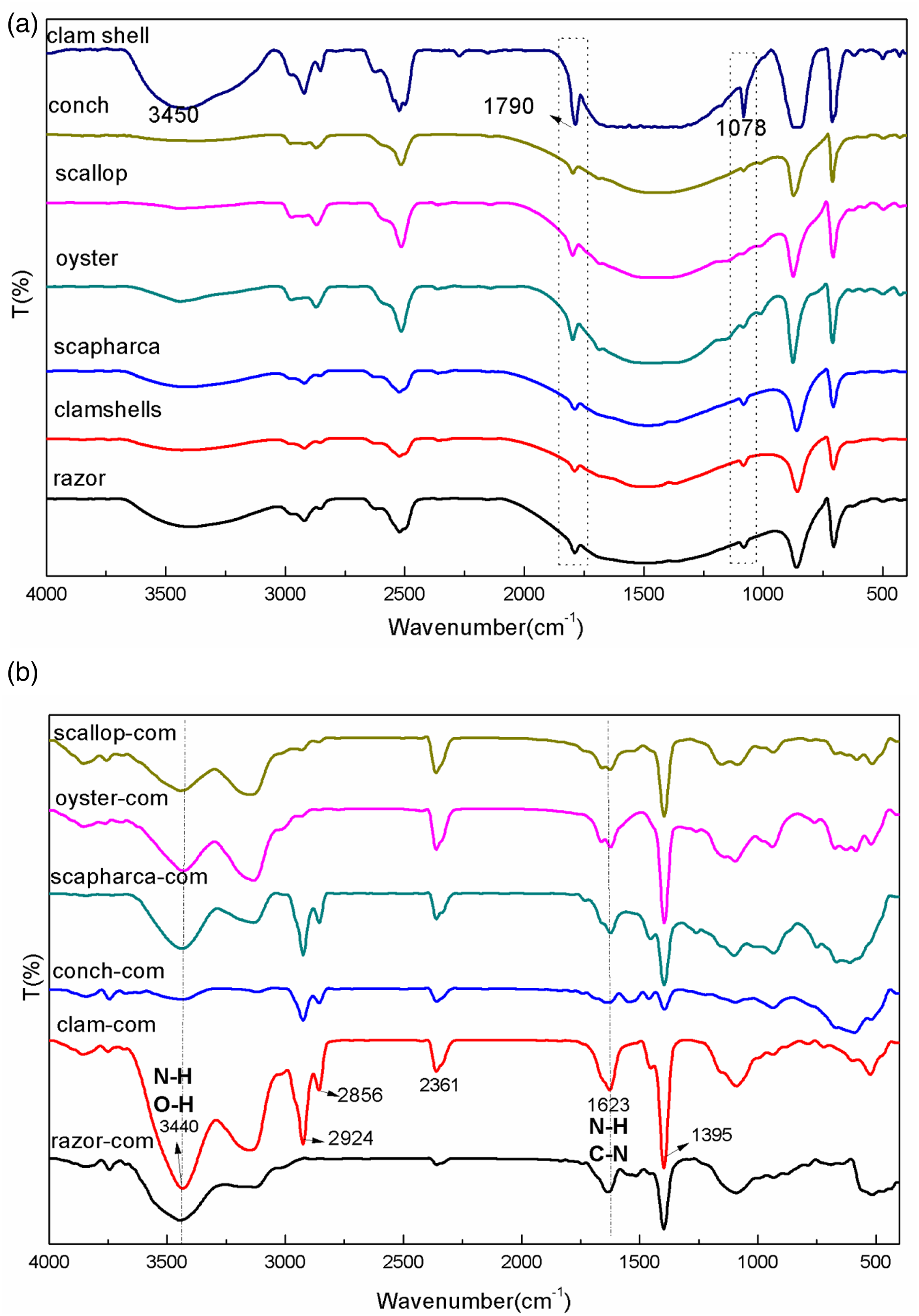
Figure 3(b) shows the infrared absorption spectra of six kinds of SP-PCL composites. Apparently, the peaks at about 3440 cm−1 and 1530 cm−1 belong to the stretching and bending vibrations of N-H and C-N bonds in urethane, 22 respectively. In addition, the characteristic peak at 2361 cm−1 represents the organic matter and HCO3- as well as urethane bond around 2360 cm−1 as supported by Xia’s work 23 and our previous work. 16 The absorption peak at 1735 cm−1, for the urethane stretching vibration, 19 can be seen in the FT-IR spectra of the different composites, but not in the SP. The above observations indicate the formation of urethane bonds between the -NCO groups in PCLPU and the -OH groups in SPs to form a well-compatible interface structure.
The layers in the shell of many mollusks have been widely studied. Although the microstructure of a shell is relatively simple, it is composed of aragonite pearl flakes and organic flakes connected by a “brick and tile” structure.
5
Consequently, the composition of the structure makes it easy to form a cross-linked structure due to the enhanced interaction between the macromolecules which leads to a rough cross-sectional morphology of the material. From the SEM images of the cryogenic fractured surface of SP-PCL composites prepared by the different kinds of shells (Figure 4), it can be found that the structure of oyster shell powder and scallop shell powder in the composites are like “prism”, while the conch shell has a “flaky-bridged” structure, with closely arranged layers connected by organic matter.5–11,13,24,25 Unlike the clamshell fillers, they do not contain oblique prism layers in the microstructure, but have a rough surface structure similar to “honeycomb”, which is an aragonite structure. On the fractured surface of the scapharca-com composite, the smooth interface of the scapharca can be seen, which is similar to a sheet-like structure but with larger thickness than that of the conch shell. As for the fractured surface of the razor-com composite, the razor powder shows unevenness with no obvious special structure.
1
Although these six kinds of shells have distinct microstructures when used as fillers, the prepared composites have a common feature. Specifically, the interface between the filler and the PCL matrix in all the composites is well bonded and there are no obvious ravines and voids (seen form the red ovals).
16
This phenomenon indicates that the filler and the matrix have superior interfacial compatibility. The principle is that the soft segment of the PCLPU compatibilizer can interact with PCL polymer chains via physical crystallization, while the terminal isocyanate group of PCLPU can react with the hydroxylamino group on the surface of shell powder to form a covalent urethane bond.16,23 SEM images of cryogenic fractured surface of SP-PCL composites. (oyster-com A1: ×1000, A2: ×3000; scallop-com B1: ×2000, B2: ×3000; conch-com C1: ×1000 C2: ×3000; clamshells-com D1: ×1000 D2: ×3000; scapharca-com E1: ×1000 × 102: ×3000; razor-com F1: ×1000 F2: ×3000).
Analysis of crystallographic and thermal stability properties
The XRD of six kinds of shell powder shows two distinct crystal forms, as shown in Figure 5(a). Among them, oyster, scallop and conch shells exhibit the same calcite (CaCO3, JCPDS card number 05–0586) crystal form.
26
Meanwhile clam shell, scapharca and razor shell show the aragonite (CaCO3, JCPDS Card No. 05–0453) crystal form. According to literature reports, the aragonite crystal form can be converted into the calcite crystal form by grinding.
27
The XRD patterns of the SP-PCL composites (Figure 5(b)) are from SP or PCL,
16
but the peaks of the shells superimpose the peaks of PCL. Among them, oyster-com, scallop-com, and conch-com have similar XRD peaks, while scapharca-com, clamshells-com, and razor-com are also similar. In particular, the crystal forms of the composites prepared by different kinds of shell powder are not consistent.3,19,27,28 XRD patterns of (a) six types of shells; (b) SP-PCL composites.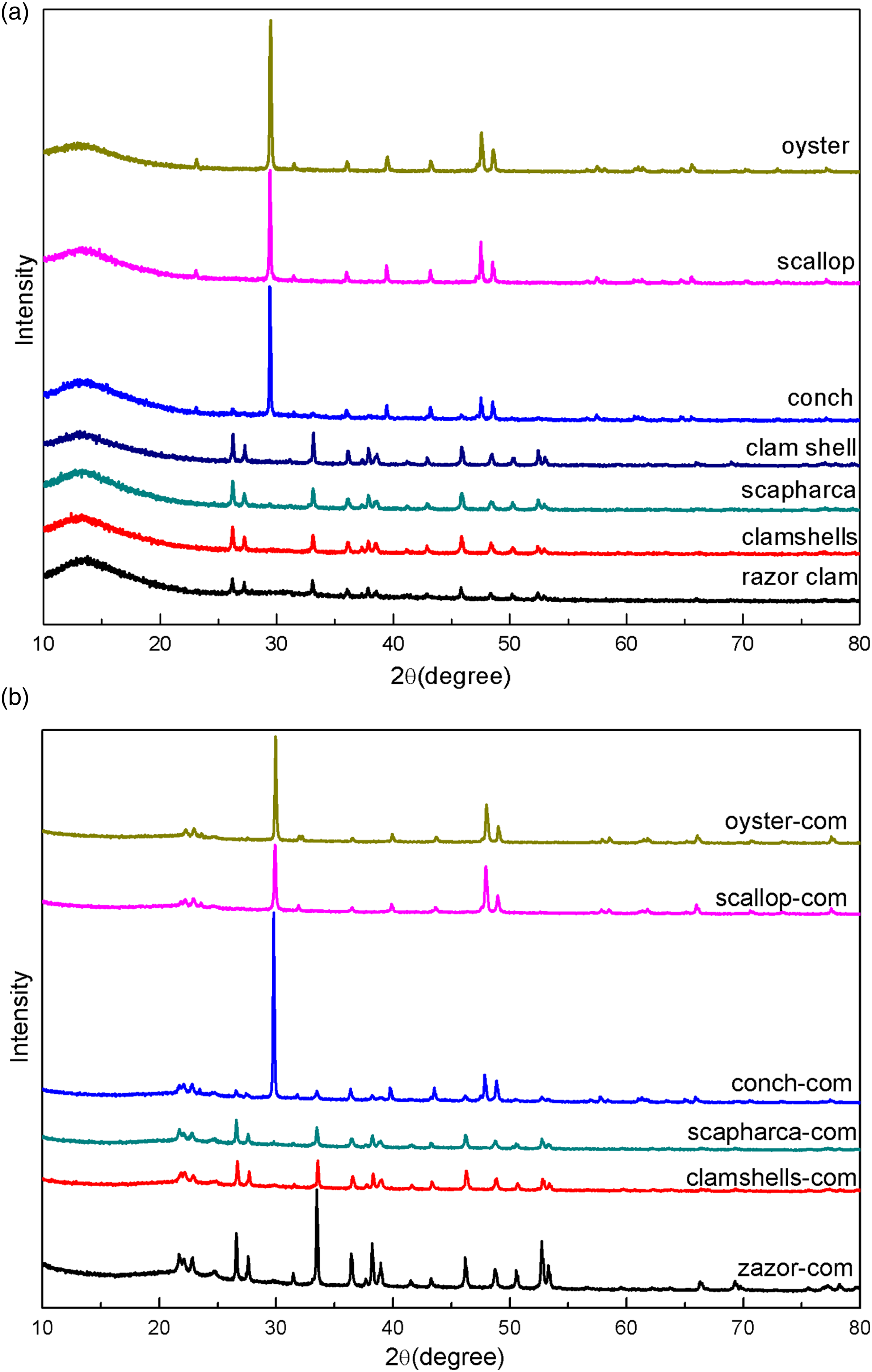
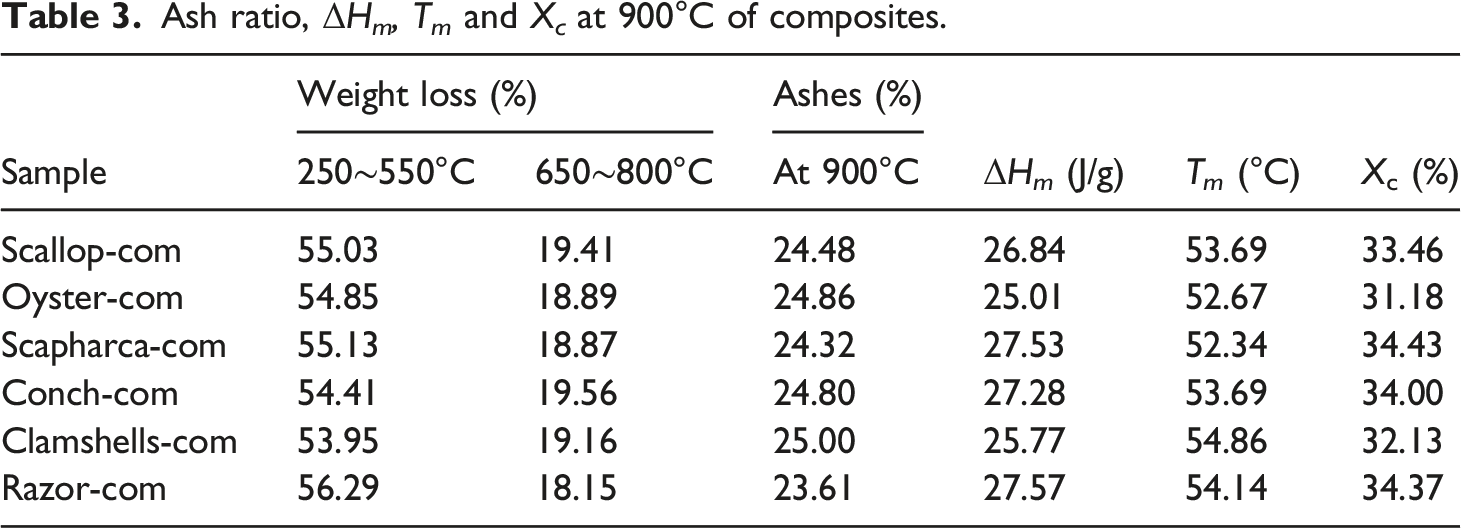
Figure 6 shows the melting temperature and crystallization properties of PCL in SP-PCL composites. The results show that the material prepared by clamshell has the highest thermal stability with a corresponding melting temperature of PCL as 54.86°C. Meanwhile the composite prepared by using oyster shell has the lowest thermal stability with a melting temperature of 52.67°C. In terms of crystallinity, the crystallinity of oyster-com is the lowest (only 31.18%), while the crystallinity of the clamshells-com is 34.43%. The work of Ge et al.
4
shows that shell powder like other calcium carbonate particles, has a nucleation phenomenon, which is beneficial to the crystallization of PCL. A comprehensive comparison shows that the difference in melting temperature and crystallinity of SP-PCL materials prepared from different shells are not significant. The composites prepared from razor shell have relatively good T
m
and X
c
of 54.14°C and 34.37%, respectively (see Table 3). DSC images of composites prepared from six shells. Ash ratio, ΔH
m
, T
m
and X
c
at 900°C of composites.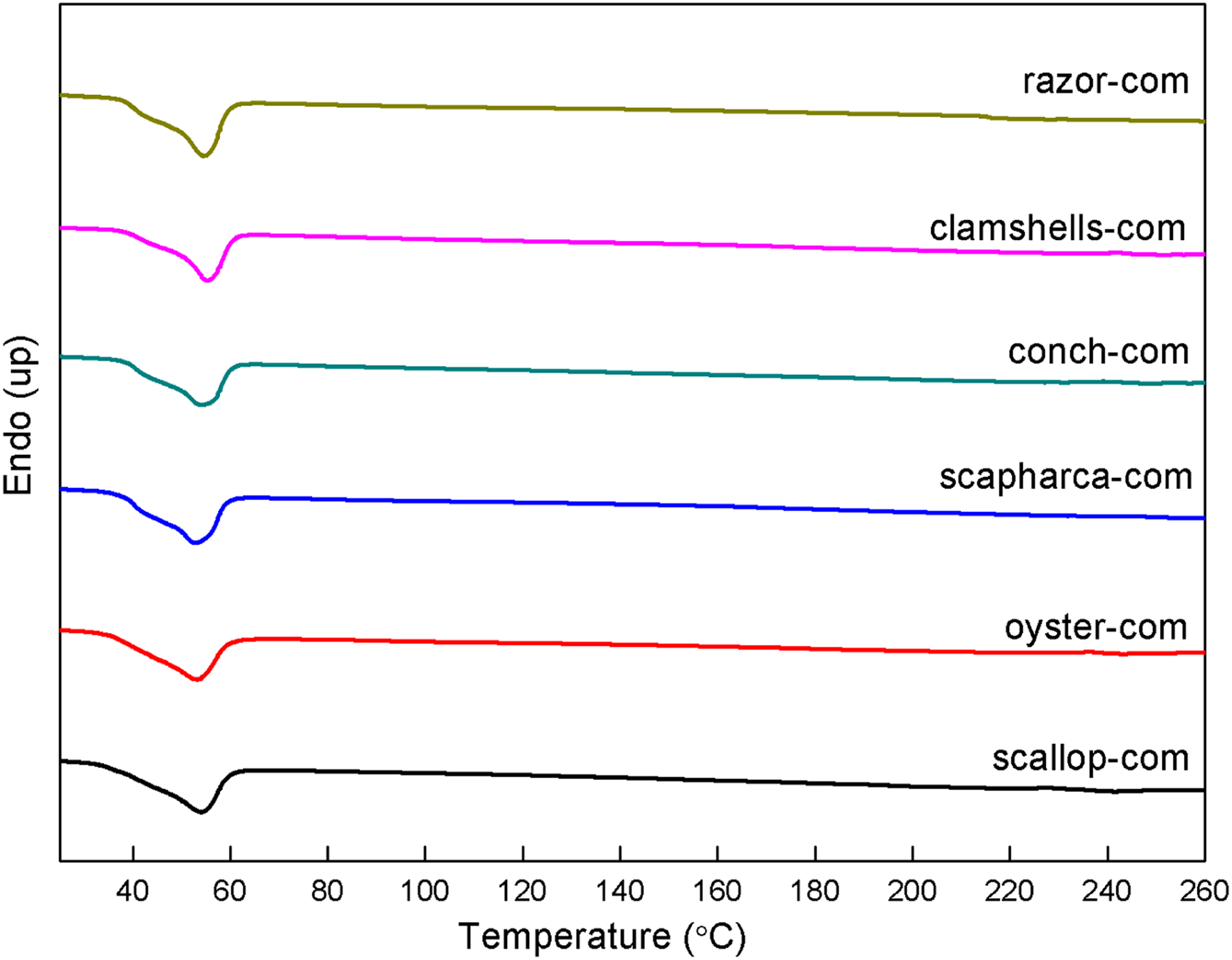
From Figure 7 it can be found that the different composites have similar trends with increasing temperature and a relatively favorable thermal stability. Except for a small amount of water loss at the initial stage, the first significant decomposition stage occurs at 250°C–550°C. The thermogravimetric loss at this stage is attributed to the decomposition of PCL, PCLPU and organic matter in shell powder .16,24 Careful observation reveals a weak curve fluctuation at 430°C, which is as a result of the decomposition of organic substance while the thermal transformation of aragonite to calcite in the shell.3,21,23,27 The decomposition temperature of organic substance in calcite is generally lower than that of aragonite .
14
Another major weight loss range occurs at 580–750°C, which mainly corresponds to the decomposition of calcium carbonate into calcium oxide and carbon dioxide. As a natural organic-inorganic filler, shell powder exhibits excellent thermal stability due to its strong binding force between organic components and calcium carbonate.
14
Therefore, the thermal stability of the composites prepared from the different shells is also superior. TG curves of six kinds of shell-prepared composites.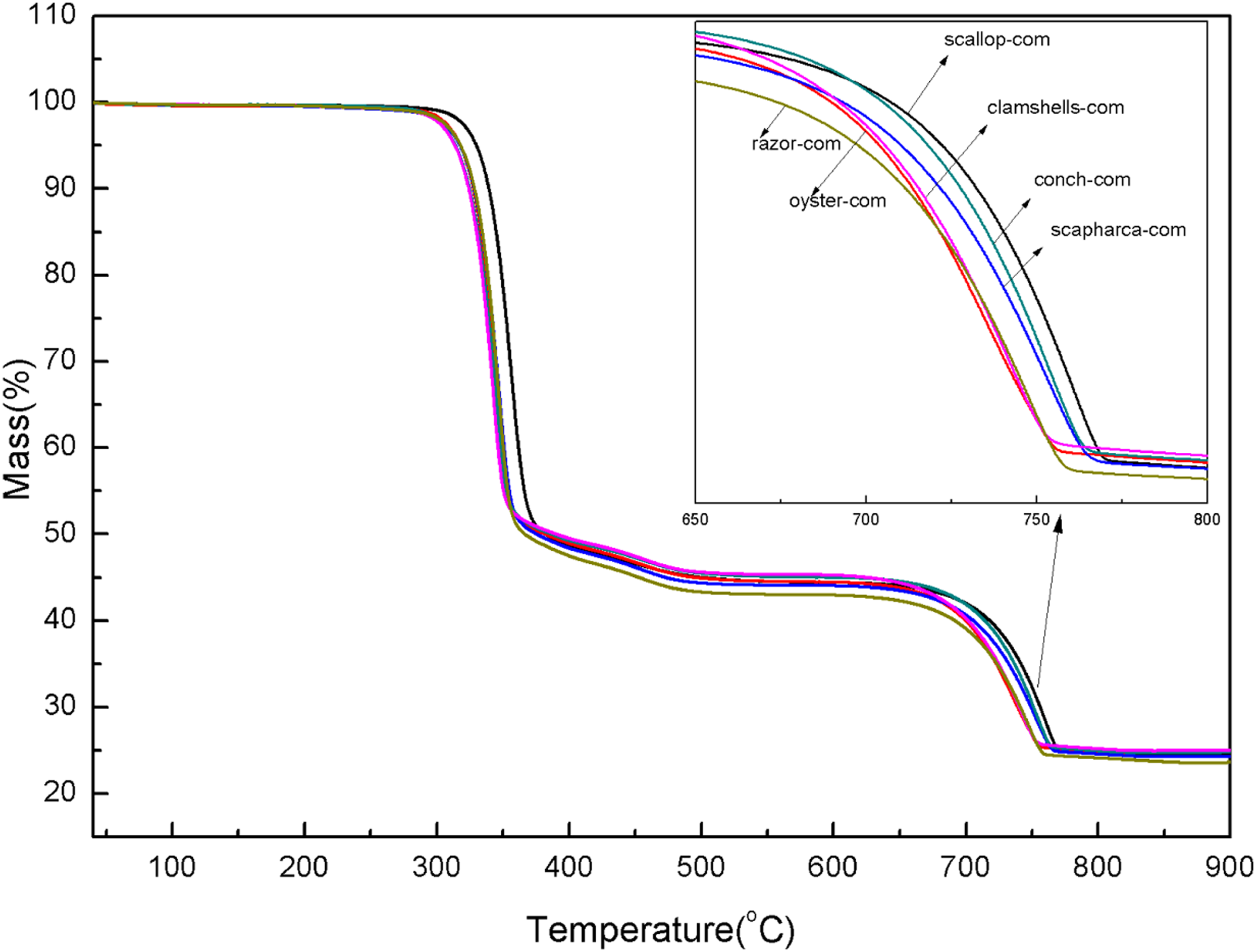
Analysis of tensile properties
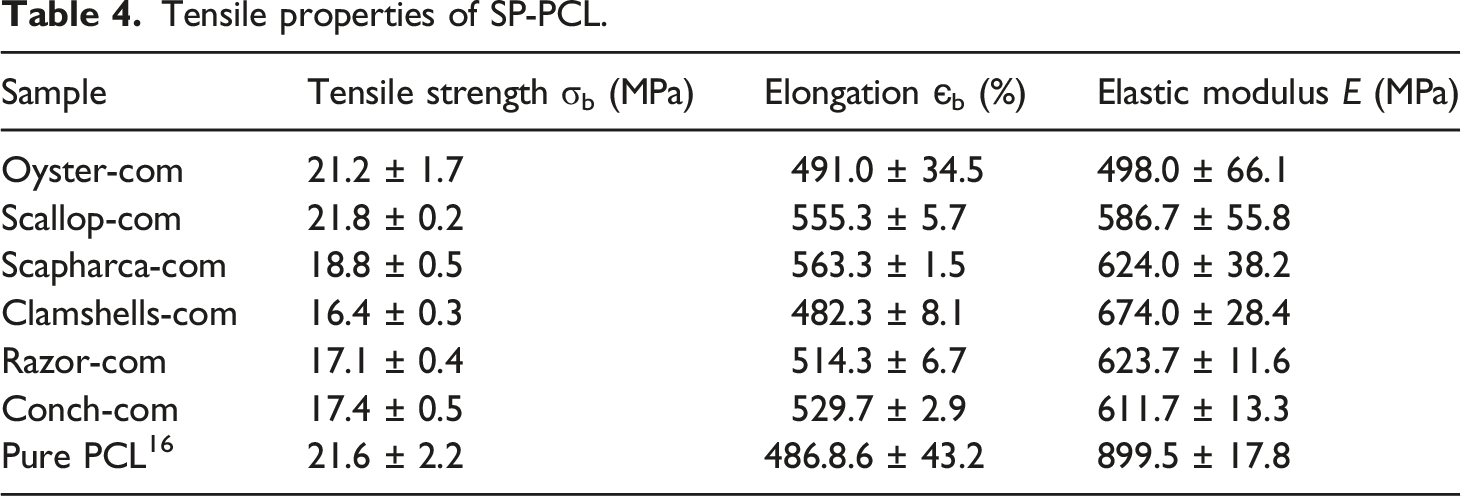
Tensile properties of SP-PCL.
From the analysis of the results, the structure of the SP-PCL composite material can be depicted diagrammatically as shown in Figure 8. Obviously, no matter the kind of shell powder utilized in preparing the composite, the bonding principle for SP-PCL composite material is that PCL and PCLPU interact via physical cross-linking, while, the -NCO groups in PCLPU form urethane bonds with the hydroxylamino present in the organic matter of the shell powder to create a compatible interfacial system. However, due to the variations in the amount of organic matter component in the various shell powder used, the number of urethane bonds formed in the various composites were also diverse. Consequently, the various composite materials exhibit some differences in the tensile performance. For example, the interface structure of Razor-com (with less urethane bonding interactions) may look like the diagram in Figure 8(a), while interface structure of Oyster-com (more urethane bonding interactions) may resemble the diagram in Figure 8(b) with better tensile performance. Diagram image of shell-prepared composites.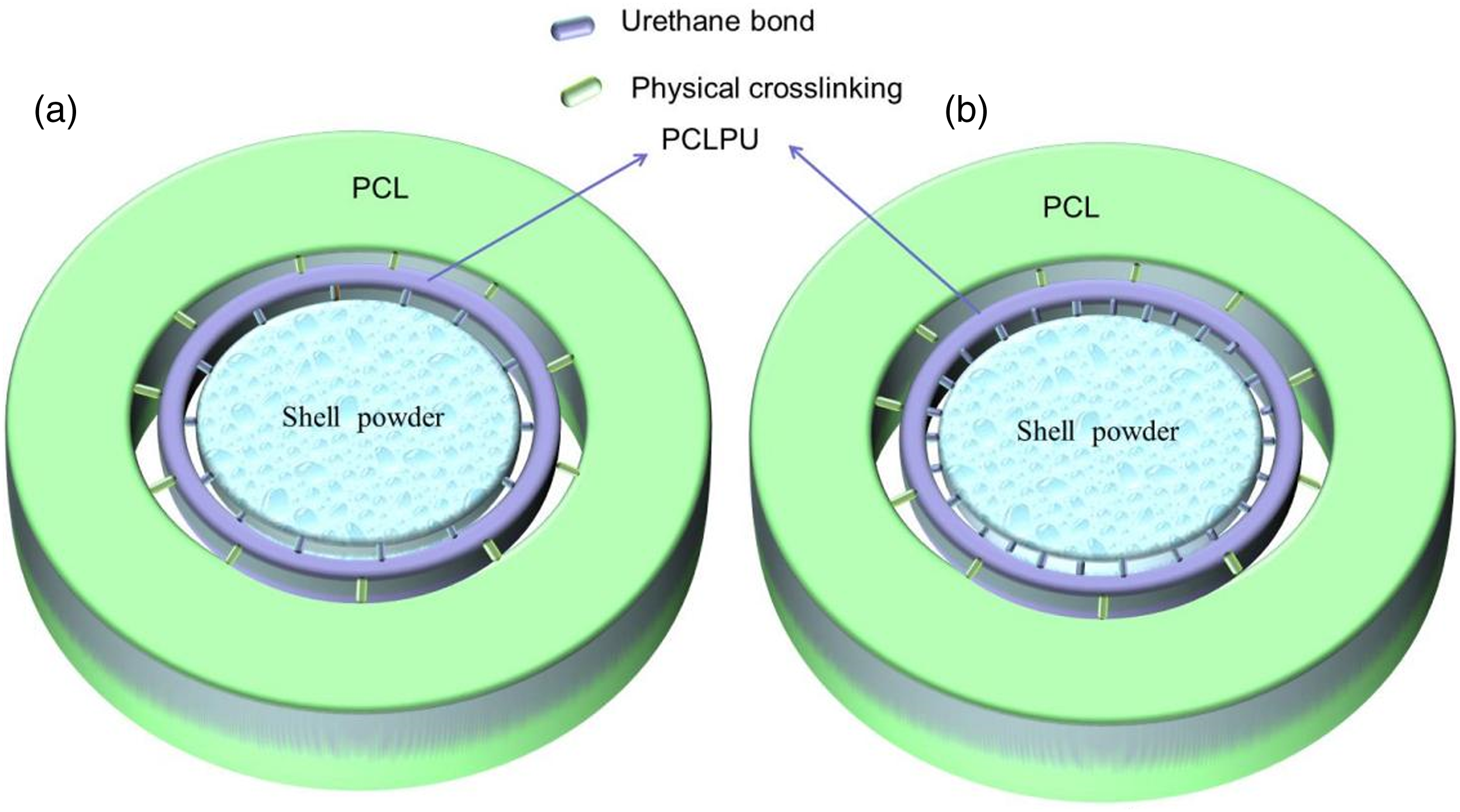
Conclusion
In this work, SP-PCL composites by using six (6) kinds of shell powder were prepared. The SP-PCL composites were prepared by using 15 wt % of the PCLPU compatibilizer to explore the effects of different kinds of shell powder on structure and properties of the composites. The FT-IR results showed that the organic matter composition and structure of the various shell powders were similar. Furthermore, XRD revealed that the six kinds of shell powder explored in this work could be divided into two crystal forms. The composite prepared from shell powder with aragonite crystal form had a higher elastic modulus of 27.4% higher than that of the composite prepared from shell powder with calcite crystal form. Among, the various composites prepared, the scallop-com composite had the highest thermal decomposition temperature, while the clamshell-com composite exhibited the maximum improved melting temperature. Analysis of SEM and tensile test results showed that although the microstructures of the different kinds of shell powder were slightly distinct, all the composites showed excellent interfacial compatibility and tensile performance. The differences in crystal structure and content of the organic matter composition ultimately affected the reactivity between the shell powder and the polyurethane prepolymer. Application of these discarded shells as filler in the preparation of various composite materials could ensure judicious use of natural resources and reduce cost as well as environmental burden in the form of pollution. This work has also laid a foundation for the full utilization of shell as a natural resource in the degradation of packaging composite materials, plates and films, etc.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to thank the Program of Introducing Talents of Discipline to Universities of China (111 program, B17019) and the foundation of Scientific Research Program of Hubei Provincial Department of Education (Grant No. Q20213001) for the material supply.
