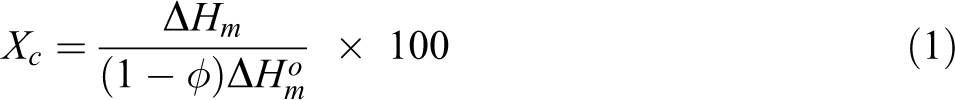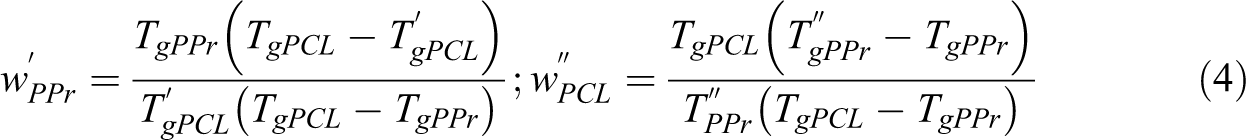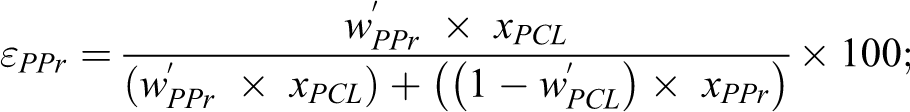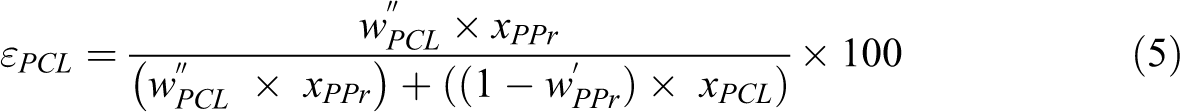Abstract
The influence of polycaprolactone (PCL) and nano titanium phosphate, pristine (TiP) and Jeffamine™ modified (JETiP), was investigated in composites derived from recycled polypropylene/polycaprolactone/titanium phosphate (PPr/PCL/TiP and PPr/PCL/JETiP). The TiP was synthesized and chemically modified with amine and the composite processed in a Haake rheometer. Improvement of PPr thermal stability was revealed. Reduction of PPr cooling crystallization temperature (9°C) and crystallinity degree (∼5–10%) were registered. Hydrogen nuclear magnetic resonance time domain revealed enhance of molecular mobility which was associated to the diminution of PPr crystallinity. The X-ray diffraction pattern of the composite PPr/PCL/JETiP did not show phosphate diffraction angle. It was understood as polymer intercalation into modified titanium phosphate galleries. At low temperature, the storage modulus increased by the presence of PCL and phosphates evidencing reinforcement. Both PPr/PCL/TiP and PPr/PCL/JETiP composites yielded phase separation systems revealed by two peaks in the loss modulus curves. Also, the high PPr participation effectiveness degree on the PCL-rich phase was understood as owing to the interaction between PCL and phosphates.
Introduction
Since its development and insertion in our daily life, the growth of polypropylene (PP) as commodity polymer is huge. It presents low density, excellent mechanical properties, chemical stability, low reactivity, atoxicity and high processability to productivity ratio. Owing to that, it is widely found as household items and electrodomestic appliances. There is ongoing increase of the studies on its properties improvement and applications.
Flores-Rojas and collaborators provided a study on antimicrobial activity to polypropylene by covalent immobilization of lysozyme. 1 Blends of PP and modified PP with the insertion of silver nanoparticles (AgNPs) produced to generate a bactericidal effect were studied by Oliani and co-authors. 2 Zhang and Kraus investigated the influence of anisotropic titania nanoparticles on the structure formation in a semicrystalline IPP matrix. The authors stated that the IPP/TiO2 nanocomposites exhibited clearly anisotropic molecular orientations. 3 Viewing additional safety of lithium ion batteries, He et al. described the building of separators constituted by coating of poly-p-phenylene terephthalamide (PPTA) onto commercial polypropylene. 4
Successful grafting of polypropylene (PP) onto graphene oxide (GO) surfaces was reported by Woo and Park. The authors asserted the increase of modulus, tensile strength, and elongation at break. 5 Physical anchoring of maleic anhydride grafted polypropylene or polypropylene onto acid functionalized multiwall carbon nanotube (f-MWNT) was accomplished by Wang and collaborators. At 0.1–1 wt% concentration, these coated matters were added to pristine PP to achieve nanocomposites. It was stated that the stronger interaction in coated PP or MA-g-PP/f-MWNT interphase justified the increase of PP crystallization rate and melt elasticity. 6 Complex specific heat of the crystallization and melting transitions of nanocomposites of isotactic polypropylene (iPP) and carbon nanotubes (CNT) as function of CNT weight percent and temperature scan rate were investigated by Kalakonda and co-authors. 7 As compatibiling agent, PP grafted with amine-alcohol (PPgDMAE) was added to polypropylene (PP)/graphene oxide nanocomposites. A significant improvement of mechanical thermal stability and electrical properties were registered. 8 The effect of the loading of carbon nanofibers on the electrical properties of PP/ vapor grown carbon nanofibers nanocomposites was reported. The percolation threshold was estimated at about 5.5% wt of carbon nanofibers. 9 A review approaching the effect of several nanofillers on thermal, mechanical, breakdown strength and energy storage properties on PP nanocomposites was published by Cheng and co-authors. 10 Yuan et al. developed PP melting composites filled with melamine-modified graphene oxide improving flame retardancy. 11 The combination of reduced graphene oxide (rGO)–carbon nanotube with iron (CNT-Fe) was used as filler for achieving melting PP nanocomposites. The authors reported magnetic and conducting properties at room temperature. 12 Compression and microinjection molding specimens of polypropylene melting nanocomposites filled with carbon black, carbon nanotubes, and graphite nanoplatelets were evaluated. The authors identified higher percolation threshold of microinjection specimen when compared to the compression molded one. 13 Magnetite (Fe3O4) was incorporated to polypropylene matrix and structure and dielectric properties were assessed. The increase of dielectric permittivity was attributed to the nanocomposites polarization ability. 14 Rheological and electrical properties of nanocomposites based on polypropylene filled with multiwall carbon nanotubes (MWCNTs) were investigated by Chafidz et al. 15
Volume resistivity of PP matrix showed dependence on MWCNTs loading. Pimelic acid was chemically attached onto TiO2 nanosphere surface. It was composed with polypropylene acting as nucleating agent promoting β-crystal crystalline arrangement. 16 In situ melt blending method was used by Cao et al. to synthesize polypropylene/dodecyl mercaptan-functionalized silver nanoparticle nanocomposites. According to the authors, a promising antibacterial and food simulant materials for biomedical and industrial applications were achieved. 17 Idumah published a review with recent developments of flame retardants in PP composites, nanocomposites and nano-biocomposites. 18 According to Ong and Chow, the halloysite nanotube can accelerate the crystallization rate of PP/polyethylene nanocomposites. 19 Improvement of mechanical and thermal properties in PP nanocomposites filled with nanocrystalline cellulose was registered by Al-Haik and co-authors. 20 Although it is synthesized by fossil matter polycaprolactone (PCL) is biodegradable polyester. With many attributes such as slow degradation rate, mechanical properties, ease of processing and molding it find applications in the biomedical, agricultural and food packaging sectors. 21 There are many studies devoted on mixing of polycaprolactone with other polymers and composites filled with inorganic and organic additives. Khandanlou and co-authors investigated PCL solution-casting composites filled with rice straw. The presence of organic matter improved the tensile modulus but thermal stability, tensile strength and elongation at break decreased. 22 An interesting control drug release film based on 5-fluorouracil-polycaprolactone was studied by Bi and co-workers in order to inhibit postoperative bleb scarring in rabbit eyes. 23 In order to provide an additive for fabrication of fused filament, Pires et al. investigated structural and thermal properties of bioactive glass-reinforcing PCL. It was registered that the glass particles induced the reduction of PCL crystallinity degree and enhanced its crystallization rate. 24 Melt-drawn microfibrillar composites derived from polycaprolactone/poly(lactic acid) (PCL/PLA) and halloysite nanotubes were ascertained. The researches pointed out that biocompatible and biodegradable fibrils were attained owing to the halloysite reinforcement and the rising of PLA crystallinity. 25 Plencner and collaborators developed a scaffold for ventral hernia regeneration build from surgical polypropylene functionalized with polycaprolactone nanofibers and thrombocytes. In all tests, the scaffold imparted viability of mouse fibroblasts and additional properties improvements. 26 A kind of biodegradable and biocompostable thermoplastic based upon polycaprolactone/thermoplastic starch/additives (MB) and chemically treated hemp fibers were added to polypropylene thinking on packaging application. The authors revealed that MB and hemp fibers acted as PP nucleating and retarding agents, respectively. 27 In order to simulate gecko toe pads structures, polypropylene, polyethylene and polycaprolactone nanofibers were fabricated using a porous membrane as template. Wetting and adhesive properties were assessed. 28 Binary system based on polycaprolactone and carbon nanotubes was added to polypropylene, polyethylene terephthalate and polyamide-6 in order to produce conductive polymer composites. The blend PP/PCL-nanotube (50/50 wt/wt%) showed co-continuous morphology and selective localization of nanotubes into PCL phase. 29 Since the end of 19th century, the chemical routes for attaining tetravalent metal phosphates (Zr, Ti and Sn) came to light. The importance and huge development of these materials must be specially dedicated to Clearfield due to its fruitful contribution in this area. These phosphates are inorganic matters capable of acting as ionic exchanger, catalyst, ionic conductor and so on.30–34 Rajini et al. reported the synthesis of organomodified titanium phosphate with antimicrobial, nematicidal and anticancer biological activities. 35 Our research group devotes attention to synthesize and modify zirconium and titanium phosphates with long chain amines. Additionally, they are used as fillers to achieve nanocomposites with improved properties.36–40
The effect of polycaprolactone and nano titanium phosphate, pristine (TiP) and Jeffamine™ modified (JETiP) on the polypropylene matrix was performed. Thermal, crystallographic, dynamic-mechanical and molecular mobility characteristics were assessed. The material was designed for application in miscellaneous utensils.
Experimental
Materials
Recycled polypropylene (PPr) was supplied by COMBRARE Ltda; polycaprolactone (10,000 g/mol) (PCL), titanium isopropoxide –Ti[OCH(CH3)2]4, 97%, P.A., and ether-amine oligomer (Jeffamine™ M600) were purchased of Sigma-Aldrich; phosphoric acid (H3PO4), 85%, P.A., was provided by Vetec Química Fina Ltda.
Titanium phosphate synthesis and long chain amine intercalation
Synthesis of titanium phosphate was carried out by mixing titanium isopropoxide (20 mL) and orthophosphoric acid (35 mL), at molar ratio of 1:8, maintaining under stirring and refluxing, for 24 hours. After that, the reaction medium was inserted into dialysis membrane and submitted to successive washes in water bath for removing the residual acid. By filtration and drying, a white powder was attained labeled as TiP. To intercalate Jeffamine inside the TiP galleries, ethanollic dispersion of TiP (1 g in 15 mL absolute ethanol) was mixed with ethanollic solution of Jeffamine (1.19 g), with aid of one drop of ethylamine, at room temperature, under stirring, for 24 h. After separation, it was dried at 90°C until constant weight being labeled as JETiP.38,40 The schematic representation of synthesis and intercalation is depicted in Figure 1.
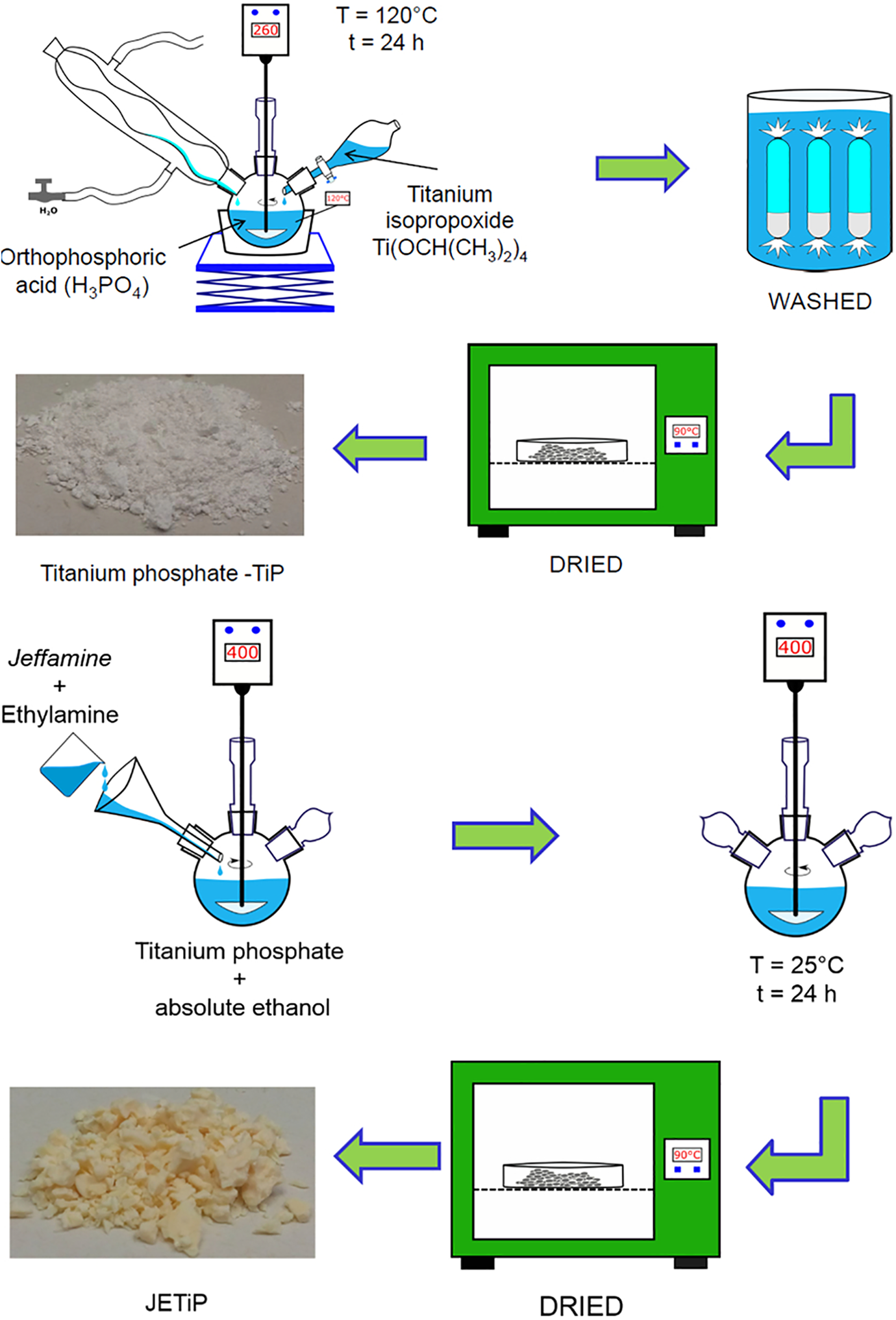
Schematic representation of TiP synthesis and intercalation.
Composite preparation
In order to aid the incorporation of the TiP and JETiP into PPr matrix, a masterbatch was yielded. PCL solution (5 g in 20 mL of chloroform) was dripped to a TiP suspension (4 g in 30 chloroform), at 25°C, under mechanical stirring, for 24 hours. Following, the reaction medium was sonicated during 30 minutes, dried at 50°C, until constant weight. The mass of PCL and phosphates was thought to attain the PCL/Phosphate proportion 26/4 wt/wt%. The masterbatches were labeled as PCL/TiP and PCL/JETiP. Prior to processing, PPr and masterbatches were dried. In a Haake torque rheometer coupled with a mixing chamber, adequate amount of the precursors were mixed in order to keep PPr/PCL/Phosphate proportion of 70/26/4 wt/wt/wt%, processed at 190°C, 60 rpm, during 15 minutes. For comparative purpose, PPr sample was processed. The samples received the following abbreviations: PPr, PPr/PCL/TiP and PPr/PCL/JETiP. By compression molding, specimens of 30x10x1 mm were processed at 190°C and submitted to the characterization. Briefly, Figure 2 shows the steps of composite preparation.
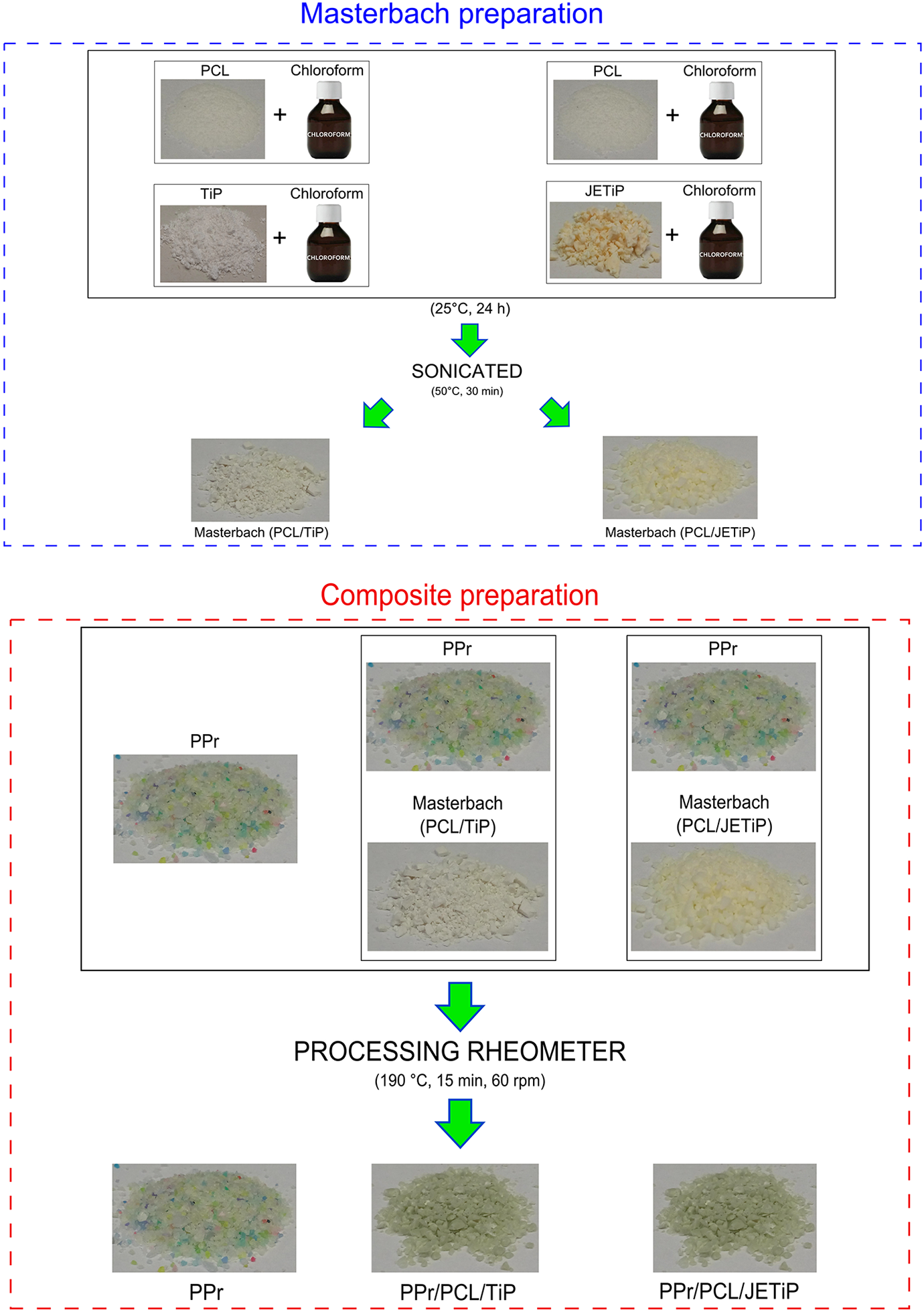
Summarized scheme of composite preparation.
Characterization
Fourier transform infrared spectroscopy (FTIR)
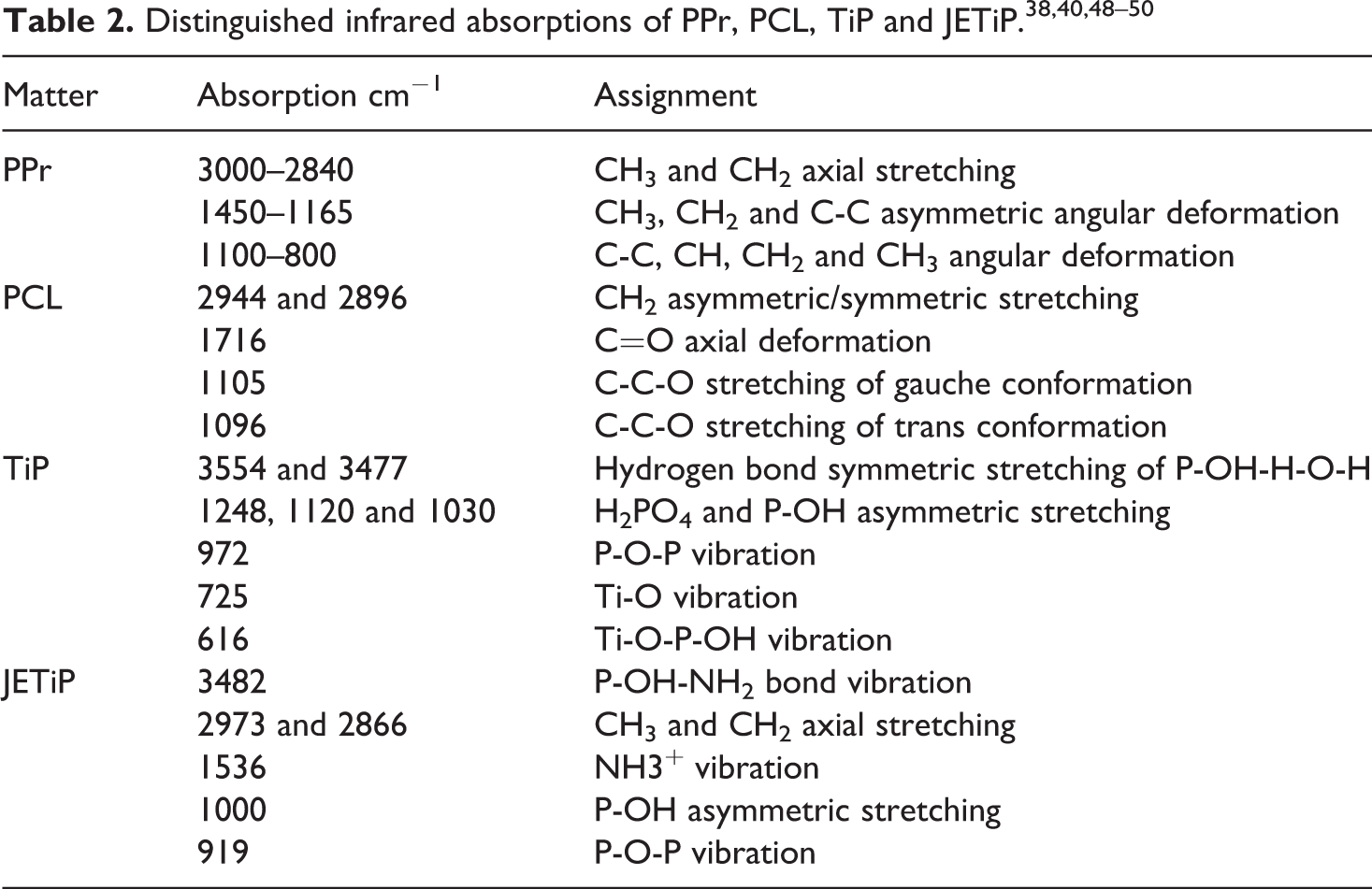
Infrared spectroscopy was used to characterize the chemical structure. The analysis was performed using a Perkin Elmer equipment model Frontier in sample film from 4000–600 cm−1 with attenuated total reflectance accessory (ATR), considering 20 scans and 4 cm−1 resolution.
Differential scanning calorimetry (DSC)
Samples exo and endothermic processes were monitored by differential scanning calorimetry following ASTM D3418. 41 The test was conducted at TA equipment model Q1000 employing three thermal heating cycles and two cooling ones. Initially, the sample was heating 0–200°C, at 10°C.min−1, keeping 2 min for erasing the thermal history. Immediately after, a cooling cycle was applied at equipment maximum rate. A second heating cycle was performed applying the first heating conditions. After that, a second cooling cycle at 10°C.min−1 was performed. Finally, a third heating cycle similar to first one was carried out. Cooling crystallization temperature (Tcc) and melting crystalline temperature (Tm) were collected from the second cooling and third heating cycles, respectively. Melting enthalpy (ΔHm) was taken for calculating crystallinity degree (Xc) according to the ratio described below where ΔHm, J.g−1 is the experimental melting enthalpy, ΔHom is the melting enthalpy of 100% crystalline PP, ΔHom = 165 J.g−1. 42 Xc calculation took in consideration the amount of PCL and filler weight fraction (Φ) in each composite. Also, the 100% crystalline PCL, ΔHom = 139 J.g−1, was used for determining of polyester crystalline percentage (Equation 1). 43
Hydrogen nuclear magnetic resonance time domain (1HNMRTD)
Hydrogen nuclear magnetic resonance time domain (1HNMRTD) was performed to characterize molecular mobility by determining the longitudinal relaxation time (T1H). The test was conducted in a Maran Ultra 23 low-field NMR device. The relaxation time (T1 H) was measured in time intervals of 2 s, taking 40 points, at 30°C. The result was expressed in terms of domain curve.
Wide angle X-rays diffractometry (WAXD)
Sample crystallographic organization was evaluated by wide angle X-ray diffractometry in a Rigaku Miniflex equipment, model DMAX 2200, using CuKα radiation (wavelength, λ = 0.155418 nm), with a voltage of 30 kV, 15 mA current, with a 2θ range of 0–60° and resolution of 0.05°. When possible, the phosphate interplanar distance was determined by Bragg equation (Equation 2) where n = diffraction order (n = 1), λ = wavelength (Å), dhkl = interplanar distance (Å), and θ = diffraction angle (°).
Thermogravimetry analysis (TGA)
TA Instruments equipment model Q500 was used for evaluating the thermal properties from 30 to 700°C, at 10°C.min−1, with nitrogen as carrying gas. Mass loss and derivatives curves were registered. The initial degradation temperature (Tonset) and temperature of maximum degradation rate (Tmax) were considered.
Dynamic-mechanical analysis (DMA)
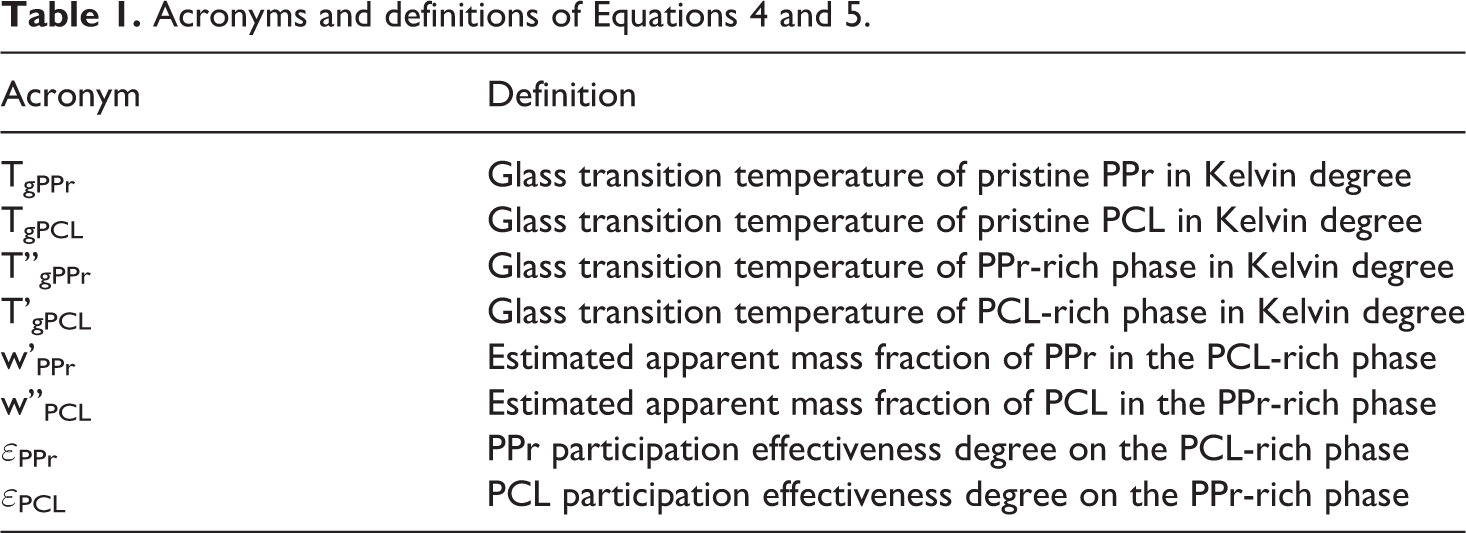
Dynamic-mechanical analysis (DMA) evaluates the dependence of the storage modulus (E’), the loss modulus (E”) and the delta tan tangent δ (E”/E’) with the temperature variation under mechanical and dynamic action. The analysis was performed in TA equipment model Q 800 equipment, using rectangular specimens with dimensions of 8.0 x 1.0 cm with 0.1 cm thickness, temperature range from −120 to 60°C, 2°C.min−1, frequency 1 Hz. E’ and E” of pristine PPr, PPr/PCL/TiP and PPr/PCL/JETiP were registered. Also, the glass transition temperature (Tg) was taken from E” curves. In order to assessing the interaction between polymers, the Fox’s equation (Equation 3) was calculated where w1, w2, Tg1 and Tg2 are PPr and PCL polymer mass fractions and their glass transition temperatures, respectively. 44 Additionally, the polymers miscibility degree was determined through the Equation 4 and 5 taken from elsewhere.37,45–47 The acronyms and definitions of Equations 4 and 5 are summarized in Table 1.
Results and discussion
Fourier transform infrared spectroscopy (FTIR)
Figure 3 depicts absorptions in the spectral region at around 3600–3000 and 1800–600 cm−1 in order to understand on the existence of interactions between polymers and phosphates. The overview of the precursors dominant infrared absorptions are disclosed in the Table 2.38,40,48–50 Between 3600–3000 cm−1, TiP absorptions at 3554 and 3477 cm−1 (hydrogen bond symmetric stretching of P-O-H and H-O-H) appeared. For JETiP, the band at 3554 cm−1 reduced intensity while the TiP band at 3477 cm−1 was shifted to 3482 cm−1 due to the reaction between phosphate -P-O-H group and Jeffamine –NH2 one. Sharply, polycaprolactone C=O axial deformation at 1716 cm−1 was not shifted. In the interval of 1300–600 cm−1, PPr, PCL, TiP and JETiP showed a series of absorptions. If PPr/PCL/TiP e PPr/PCL/JETiP spectra are confronted to the pristine PPr it is possible to infer that bands overlapped of polycaprolactone, TiP and JETiP with PPr ones led to change the profile of polyolefin infrared fingerprint region. In our vision, the result does not yet allow to infer on the interaction between PPr and the others constituents.
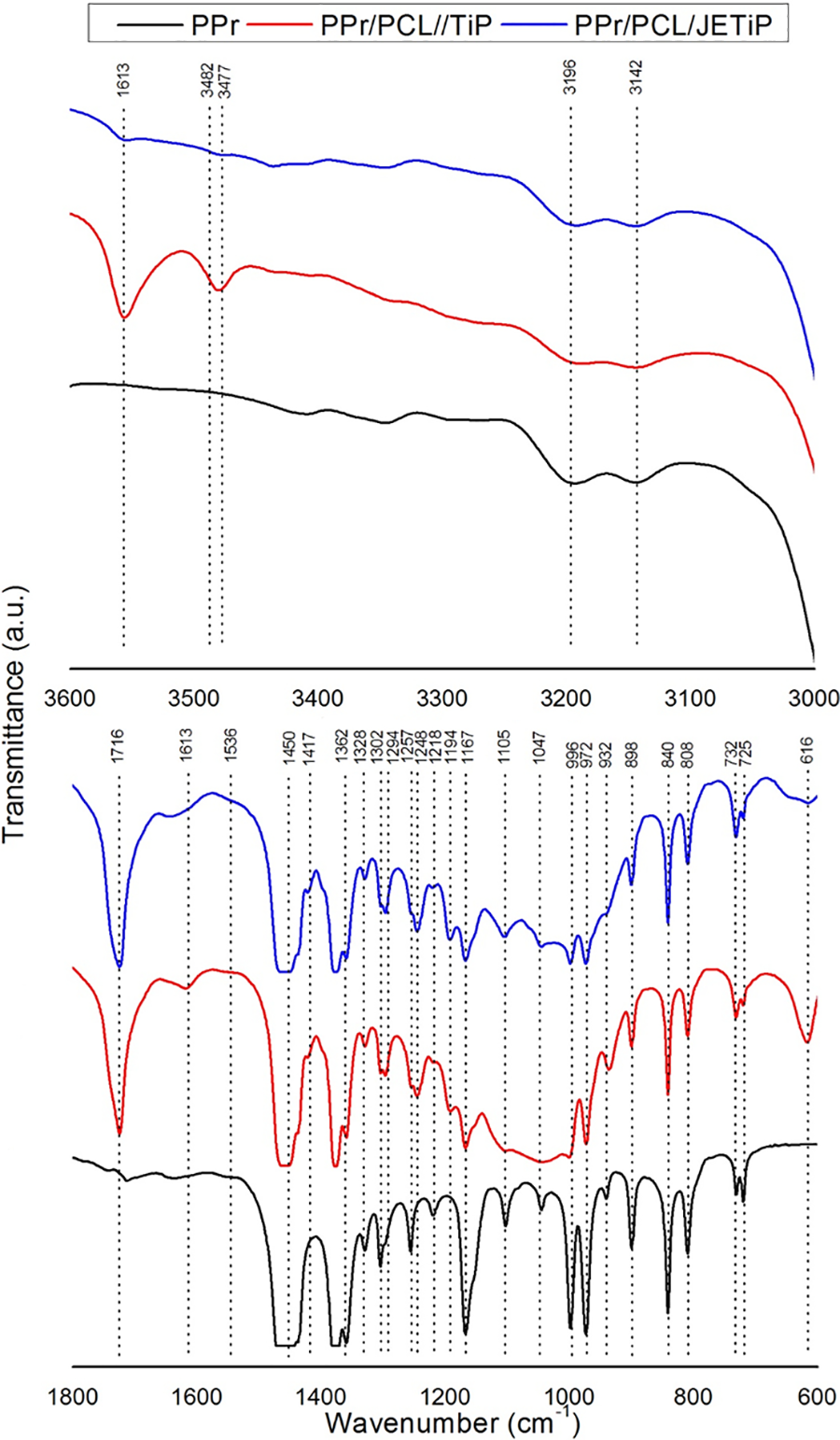
PPr, PPr/PCL/TiP and PPr/PCL/JETiP in the spectral region at vicinity of 3600–3000 and 1800–600 cm−1.
Differential scanning calorimetry (DSC)
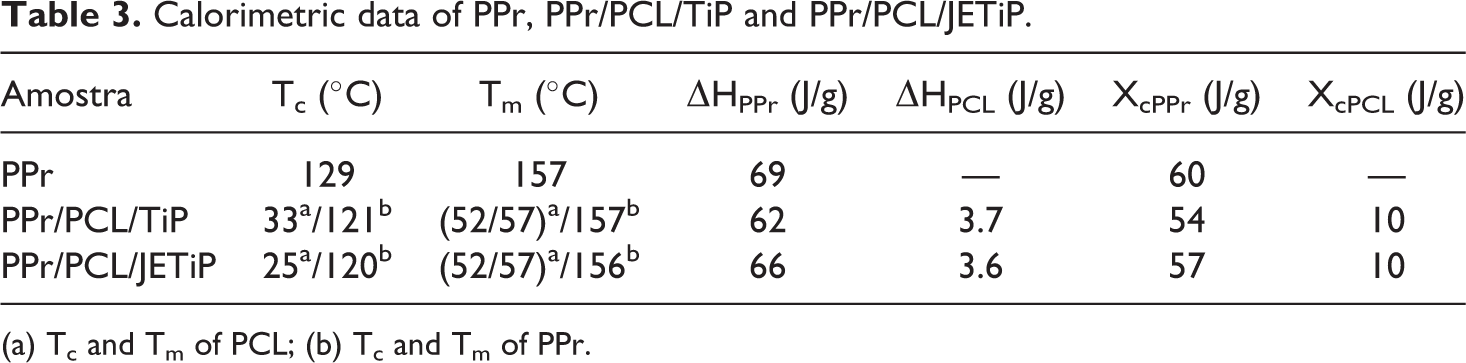
Figure 4 introduces the PPr, PPr/PCL/TiP and PPr/PCL/JETiP first and second heating, second cooling and third heating cycles. Table 3 illustrates the calorimetric data. With respect to the glass transition temperature (Tg), its approach is in the dynamic-mechanical analysis section. The cooling crystallization curve of PPr appeared as an intense peak coupled with a shoulder. PPr heating cycle curve indicated two melting peaks being low intense one at lower temperature (130°C) and main peak at 157°C. As the polypropylene was recycled, we considered that the lowest peaks at cooling and heating cycles were related to the polyethylene as low waste matter. Herein, its calorimetric evaluation was not considered. Then, the PPr cooling and melting temperatures were 129 and 157°C, respectively. The PPr crystallinity degree (Xc) attained 60%.
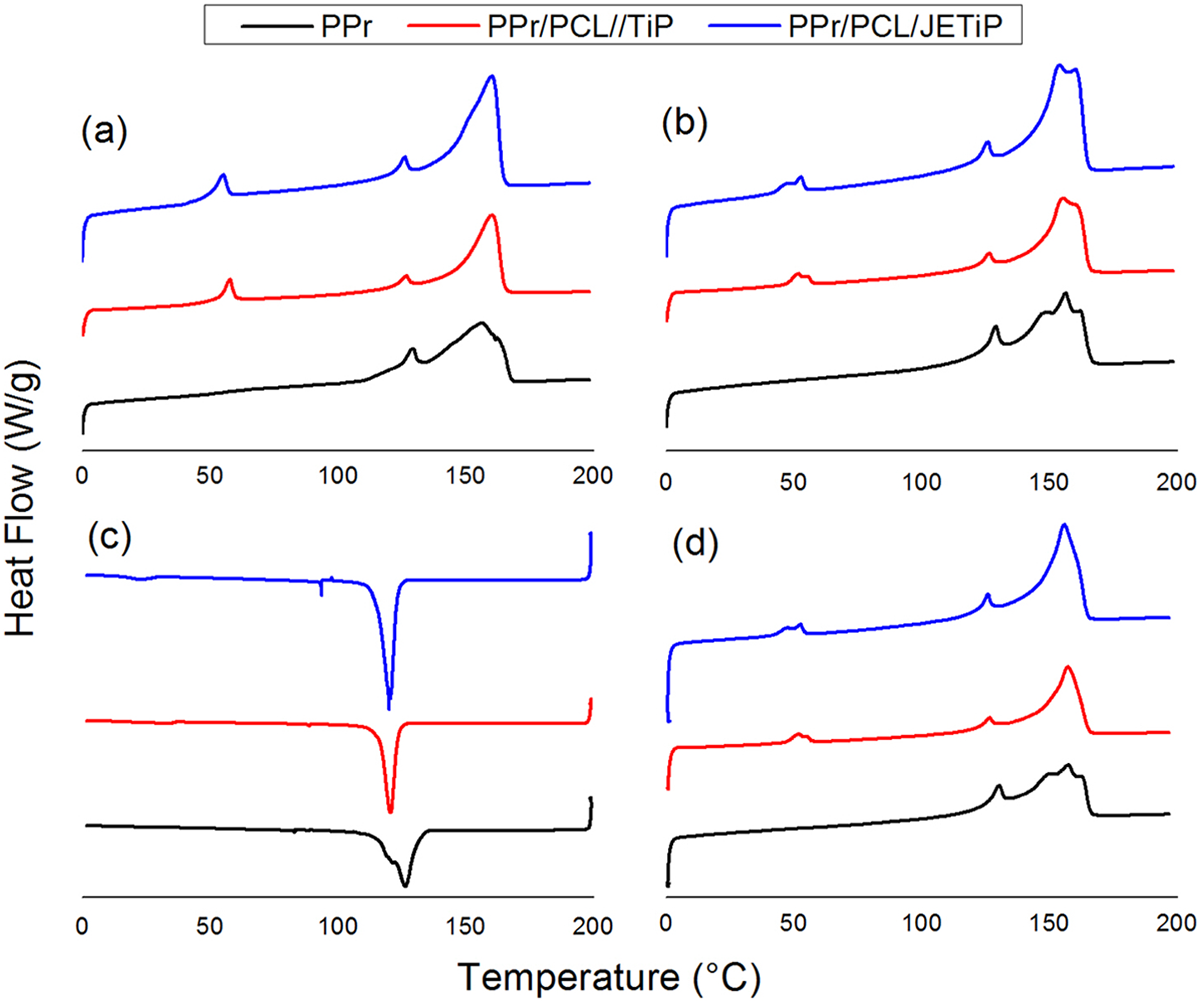
Calorimetric curves of PPr, PPr/PCL/TiP and PPr/PCL/JETiP: (a) first heating cycle, (b) second heating cycle, (c) second cooling cycle, and (d) third heating cycle.
Calorimetric data of PPr, PPr/PCL/TiP and PPr/PCL/JETiP.
(a) Tc and Tm of PCL; (b) Tc and Tm of PPr.
PPr/PCL/TiP and PPr/PCL/JETiP cooling curves presented the crystallization peaks of PCL and PPr. In both composites, the Tcc of PCL is depicted as low intense. For PPr/PCL/JETiP composite, its value was lower than that observed for the PPr/PCL/TiP one. The difference could be attributed to the interaction between polycaprolactone and Jeffamine™ during the PCL crystallization/solidification processes. On the contrary, the Tcc of PPr appeared as an intense peak at temperature much lower (120°C) than that observed for PPr. Sharply, it indicated that in some extent PCL, pristine and modified TiP slowed down the PPr crystallization process. In the melting curves of PPr/PCL/TiP and PPr/PCL/JETiP were viewed the melting peaks of PCL, polyethylene and PPr. As mentioned above, the polyethylene was considered as low waste matter in PPr and its calorimetric evaluation was not taken into account. For both composites, the Tm of PCL was manifested as too low intensity peak with two maximum probably due to the different crystal sizes. The Tm of PPr remained invariable. With respect to the crystallization degree in the nanocomposites, the content of crystalline portion in PCL was considered low. The Xc of PPr decreased—10 and 5% for PPr/PCL/TiP and PPr/PCL/JETiP, respectively. This was ascribed to the hindrance of PCL chains and phosphates to the PPr chains diffusion to the crystallization centers. If added to PP, polymers and inorganic matters may influence its calorimetric properties. Ternary composites composed by polycaprolactone biodegradable thermoplastic matrix, polypropylene and hemp fiber revealed increase of PP cold crystallization temperature and reduction of crystallinity degree. None effect of hemp fiber and PP on PCL crystallization was observed. 27 In their study on PP nanocomposites containing graphene oxide grafted with 3-aminopropyl-triethoxysilane and maleic-anhydride-grafted PP, Woo and Park detected an increase of PP crystallization temperature attributed to the nucleation effect. Melting temperature did not show significant variation. 5 Herein, the composites did not manifest variation on PP melting temperature (crystal thickness) but the presence PCL and phosphates induced the delay of PP crystallization process.
Hydrogen nuclear magnetic resonance time domain (1HNMRTD)
Hydrogen nuclear magnetic resonance time domain has been used to evaluate organization, heterogeneity and particle dispersion, interaction between polymer and filler, molecular dynamic and relaxation of polymeric blends and composites/nanocomposites.51–53 Figure 5 and Table 4 show the domain curves and relaxation times (T1H) of PPr, PPr/PCL/TiP and PPr/PCL/JETiP, respectively. Sharply, for all materials, the domain curves revealed two domains—flexible (lower relaxation time) and rigid (higher relaxation time). Around 5000–15,000 ms, the domain was associated to the hydrogen with high degree of freedom and that easily coupled to the hydrogen nucleus. In that interval, the composites domain curves were slightly shifted to lower time. This was ascribed to the higher abundance of hydrogen atom induced by PCL and Jeffamine™. Between 100,000 and 1,000,000 ms, the peaks were sharper and associated to the domains where the hydrogen relaxation was hampered due to the stiffer and ordination of the chain arrangements. Also, the composites domain curves were displaced to lower time motivated by the presence of PCL and Jeffamine™. The relaxation time followed this sequence: PPr > PPr/PCL/Ti > PPr/PCL/JETiP. The progressive reduction was imputed to the lowering of PPr crystallinity induced by PCL and phosphates as described in DSC section.
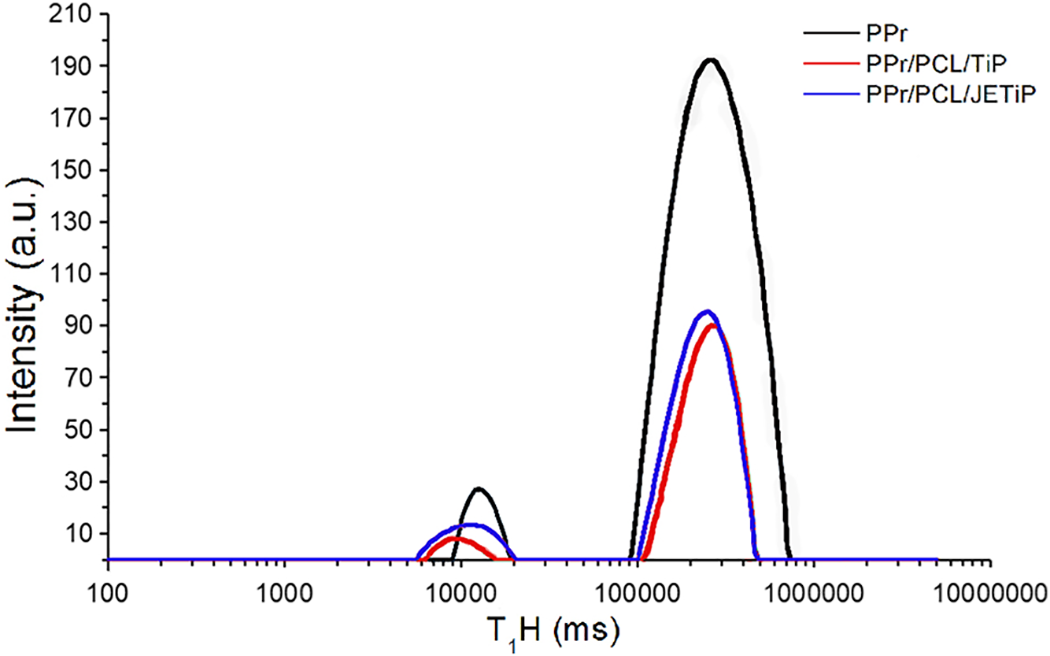
Domain curves of PPr, PPr/PCL/TiP and PPr/PCL/JETiP.
Relaxation time of PPr, PPr/PCL/TiP and PPr/PCL/JETiP.

Wide angle X-ray diffraction (WAXD)
X-ray diffractograms are seen in Figure 6. The diffraction pattern of PPr showed four diffraction angles 2θ at around 14.65, 17.50, 19.10 and 22.05° attributed to the crystallographic planes (110), (040), (130) and (111) in agreement with reported by Bafana et al. and Mariano et al.54–55 These angles and planes are related to PP α-monoclinic crystalline arrangement. Mixing of α and β crystalline forms was attained by Gonzalez-Calderon and co-authors in their investigation on PP nanocomposites filled with titanium oxide, pristine and modified by pimelic acid. 16 The diffraction patterns of PPr/PCL/TiP and PPr/PCL/JETiP composites showed the diffraction angles of PPr. PCL diffraction angles at around 2θ = 21.65 and 23.95° 56 seem likely to be overlapped by the PPr ones. Probably, it was due to its low crystallinity degree as mentioned in DSC section. For the PPr/PCL/TiP composite, the TiP diffraction angle (hkl 002, 2θ ∼ 12°, black arrow) was maintained indicating that there was not polymers intercalation into the pristine phosphate galleries. In the PPr/PCL/JETiP composite, the absence of TiP diffraction angle was understood as the polymers intercalation into phosphate lamellar spacing. Thus, it was supposed that each kind of phosphate led to formation of standard and exfoliated composite structures.
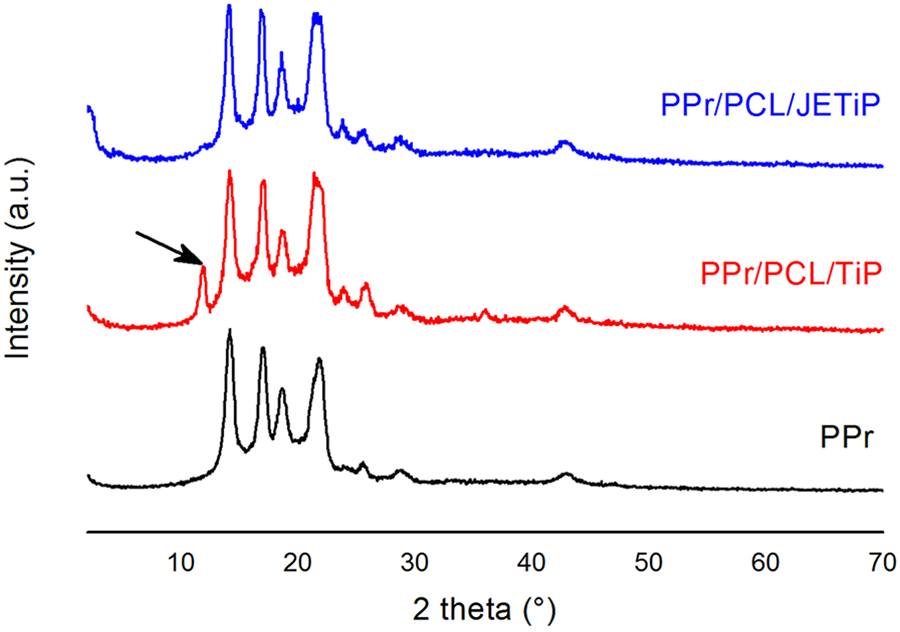
X-ray diffractograms of PPr, PPr/PCL/TiP and PPr/PCL/JETiP.
Thermogravimetry analysis (TGA)
Figure 7 exhibits the mass loss and derivative curves of PPr, PPr/PCL/TiP and PPr/ PCL/JETiP. Table 5 introduces the degradation temperatures. Only a single step of degradation was observed for PPr. Tonset and Tmax occurred at 413 and 435°C, respectively. Tonset of the composites showed drastic reduction at around 70°C and 100°C for PPr/PCL/TiP and PPr/PCL/JETiP, respectively. With respect to the temperature of maximum degradation rate, the composites revealed two distinct Tmax. Those at the lowest temperatures were interpreted as being concerned the PCL-rich phase. The Tmax of PPr/PCL/JETiP was lower than that observed for PPr/PCL/TiP. It was understood that Jeffamine™ could be operated more drastically on the reduction of the PCL-rich phase thermal stability. The highest Tmax were attributed to the PPr-rich phase. The values were higher than that detected to pristine PPr. One degradation step and improvement of PP thermal stability were found by Cao et al. in their work on nanocomposites derived from PP filled with pristine and dodecyl mercaptan-functionalized silver nanoparticles. 17 With respect to the PPr thermal stability in the composites, it was deduced that its increase was due to the phosphate matters which imparted heat insulation effect. Summarizing the TGA results, it must be emphasized that systems with phase separation were achieved, Jeffamine™ diminished PCL-rich phase thermal stability and the two kinds of phosphate enhanced the PP rich phase thermal stability.
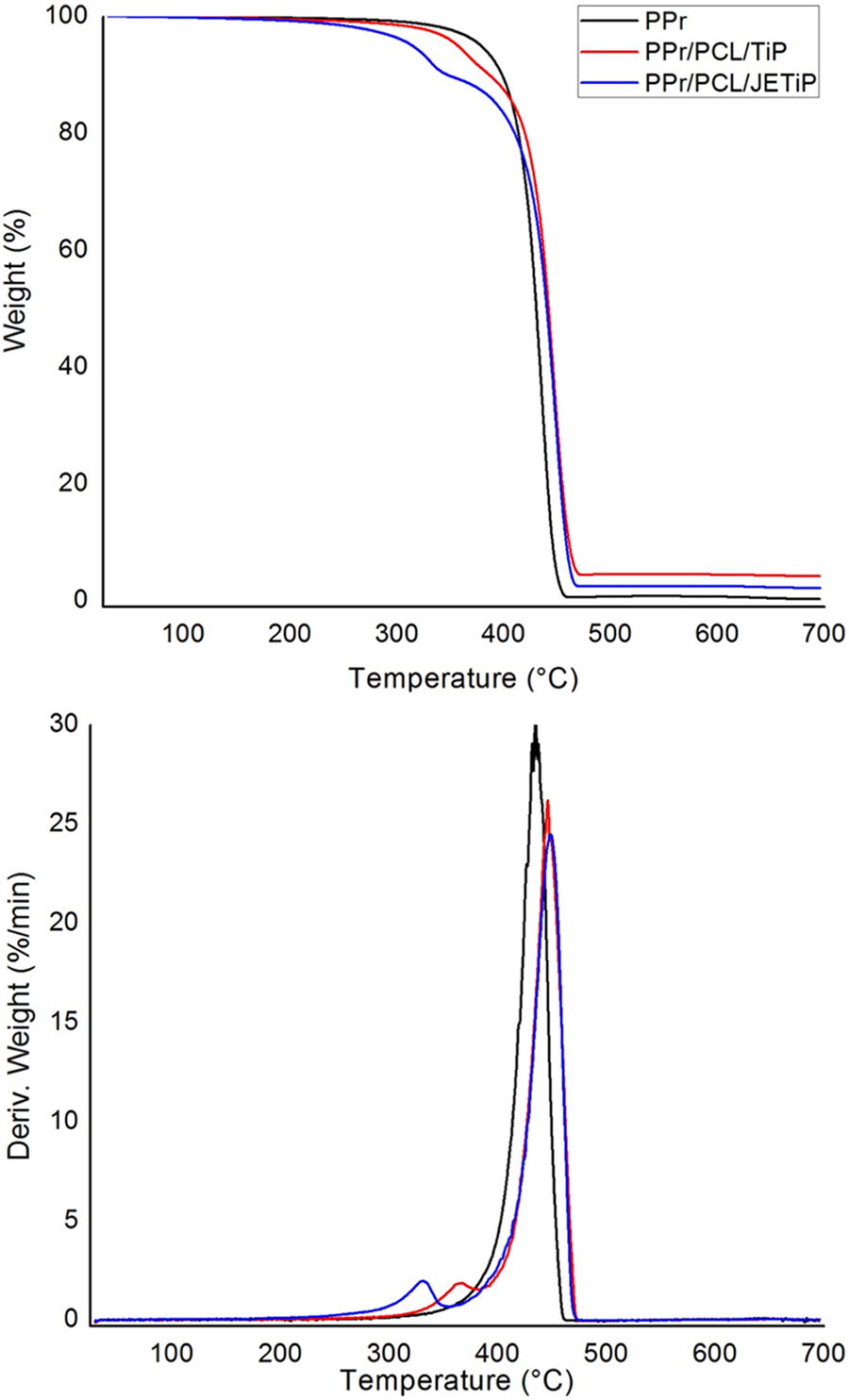
Mass loss and derivative curves of PPr, PPr/PCL/TiP and PPr/PCL/JETiP.
Thermogravimetric data of PPr, PPr/PCL/TiP and PPr/PCL/JETiP.
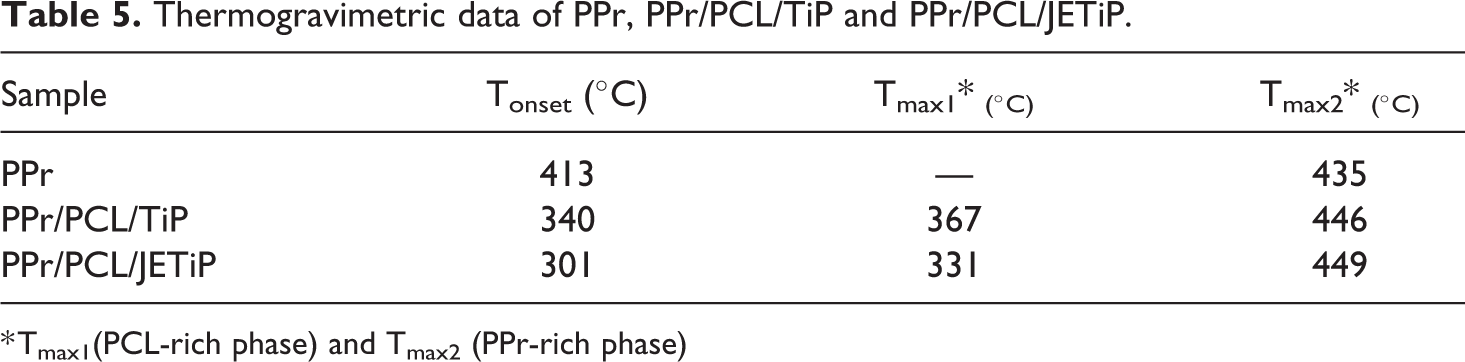
* Tmax1(PCL-rich phase) and Tmax2 (PPr-rich phase)
Dynamic-mechanical analysis (DMA)
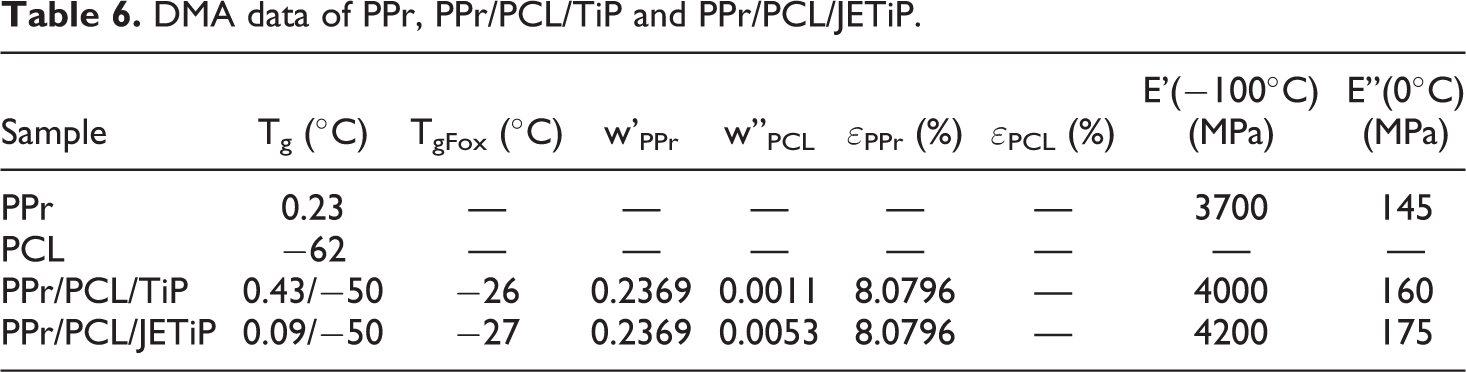
Storage (E’) and loss (E”) moduli curves of the samples are illustrated in Figure 8. Table 6 presented the DMA data. The curve of E’ showed temperature dependency. Below 0°C, it showed the following tendency PPr/PCL/JETiP > PPr/PCL/TiP > PPr which underline some reinforcement action of PCL and phosphates. Above this temperature, it was noticed the overlapping of the E’ curves. Concerning to the E”, the PPr curve showed a very intense peak with maximum around 0°C interpreted as representative of the PPr glass transition temperature (Tg). For both composites, E” curves depicted two relaxations. One located at around −50°C and a second one in the vicinity of 0°C. The first one was associated to the Tg of the PCL-rich phase. At around 0°C, it was detected a steady relaxation attributed to the PPr-rich phase. Brito et al. mentioned decrease of glass transition temperature in their investigation on PP composite reinforced with green coffee cake. 57 Studies on miscibility of binary systems of PCL and synthetic polymers are controversial. Although are biodegradable synthetic polyester, polycaprolactone and poly(lactic acid) (PCL/PLA) formed immiscible system even in the presence of halloysite as registered by Kelnar and co-authors. 25 In their research on poly(vinyl chloride) (PVC) and PCL blends, Rusu et al. reported to achieve a homogeneous system in blend with 40% in mass of PCL. 58 If the evaluation of the PPr participation effectiveness degree on the PCL-rich phase and PCL participation effectiveness degree on the PPr-rich phase is considered it is possible to assess on the polymers and phosphates interactions. The εPPr value was significant while its counterpart εPCL was null. In summary, immiscible systems were obtained. There is evidence that the interaction of PCL and phosphates is significant. Also, both PCL and phosphates aided the mixing of some PPr chains into PCL-rich phase.
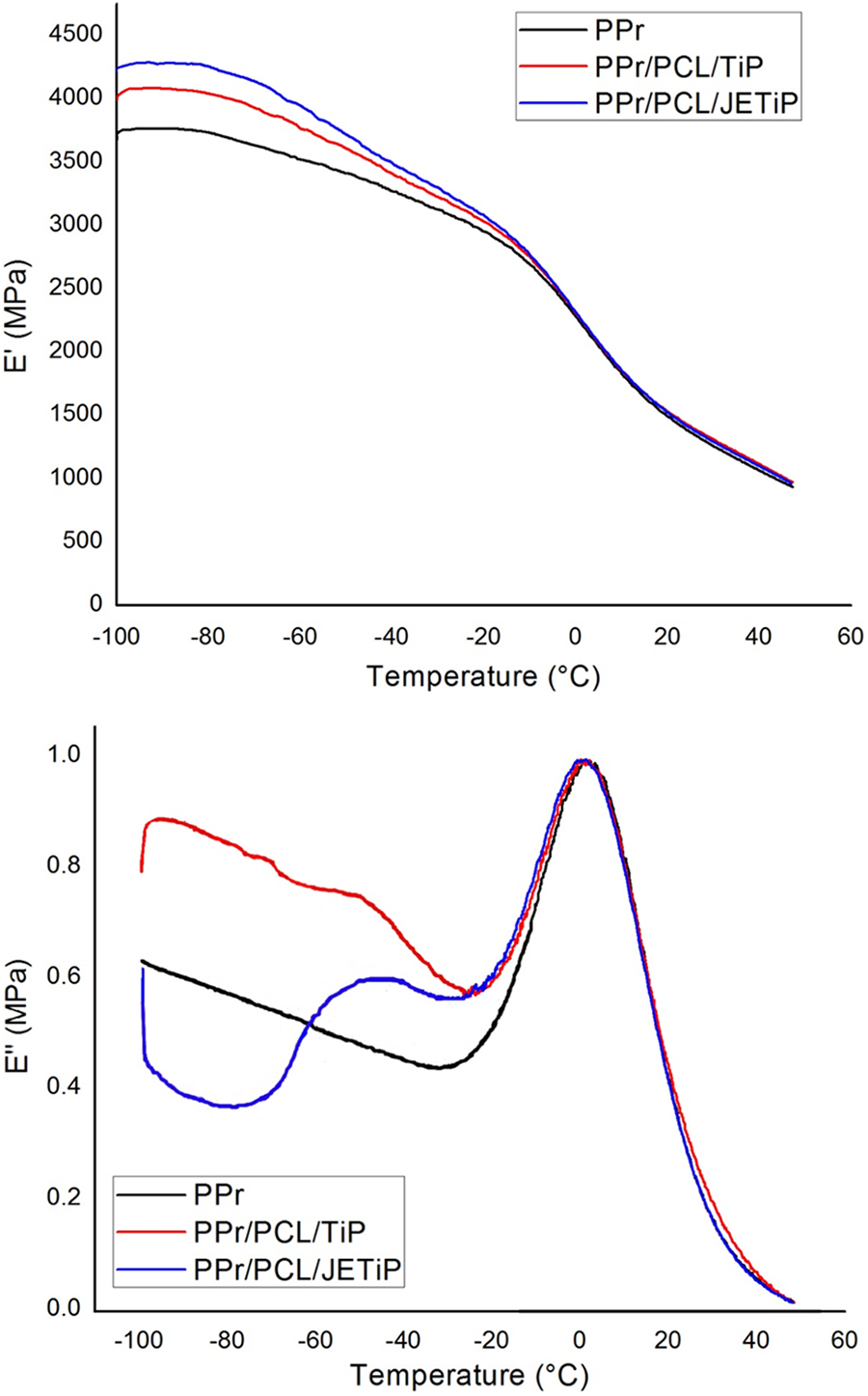
E’ and E” curves of PPr, PPr/PCL/TiP and PPr/PCL/JETiP.
DMA data of PPr, PPr/PCL/TiP and PPr/PCL/JETiP.
Conclusions
Composites based on recycled polypropylene (PPr) filled with polycaprolactone (PCL) and pristine/Jeffamine-containing titanium phosphates were investigated. Thermogravimetry indicated two-phase system and the increase of PPr thermal stability. According to the kind of phosphate, X-ray diffraction revealed that composites with standard and exfoliated structures were achieved. Also, polymer intercalation was evidenced. PPr cooling crystallization temperature and crystallinity degree were reduced meaning the influence of PCL and phosphates on the polyolefin crystallization process. Both PCL and phosphates induced the diminution of PPr molecular relaxation. DMA results corroborated the formation of two-phase system. Also, the εPPr endorsed the evidence of higher interaction between PCL and phosphates.
Footnotes
Acknowledgements
The authors thank Conselho Nacional Desenvolvimento Científico e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001 and Universidade Federal do Rio de Janeiro (UFRJ) for supporting this work.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.