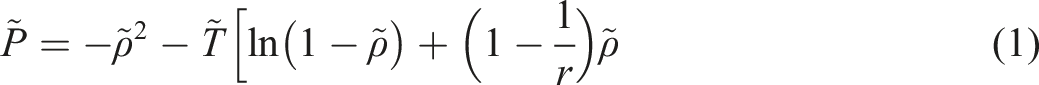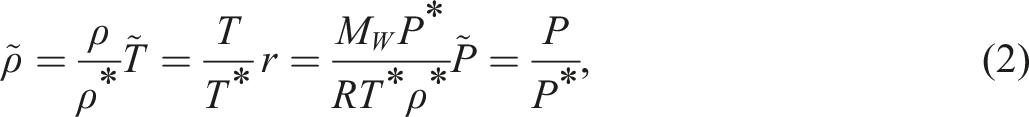Abstract
Bubble nucleation and growth were investigated in low density polyethylene (LDPE) using nitrogen (N2). The solubility and diffusivity of N2 in low density polyethylene were determined by a magnetic suspension balance (MSB) system. The bubble nucleation and growth during foaming was assessed using a visualization batch foaming system. It was observed that the increase in temperature and pressure decrement, decreased the bubble density, respectively. The bubble size during the foaming was also studied through numerical model and the effect of various simulation variables on the bubble growth was investigated. It was concluded that the numerical model could predict the foaming process. Moreover, according to the bubble pressure profile, the N2diffusion are formative factors in controlling the bubble growth.
Introduction
Low density polyethylene (LDPE) is extensively used for producing numerous containers, foam and plastic sections for some components. The polymer foams have been used recently due to their greater characteristics, compared to solid polymers.1–4 In foaming of polymers blown with blowing agents, numerous parameters such as saturation pressure and the thermodynamic instability could control the final bubble density, size, and growth rate.4,5 Thermodynamic instability, may be provided through a rapid pressure release.6–9 So, the nucleated bubbles could be stabilized when the bubble reaches the critical value.10–13 Furthermore, the bubble nucleation and growth and the final structure of foamed polymer is dependent on the materials characteristics.14–16 The simulation of bubble nucleation and growth during foaming have considered in sequence of studies.17–19 Han and Yoo advanced a molding process for foaming of polystyrene with different injection rates in foam extrusion and the experimental and numerical data were compared. 17 In another works the simulation for the heterogeneous nucleation of microcellular foams was studied and different parameters such the temperature and pressure, concentration of nucleating agents was investigated.18–22 The nucleation and growth of bubbles in non-Newtonian and Newtonian fluids were also are studied by Frank et al. 23 Ishikawa et al. 24 worked on the numerical studies of polypropylene foaming blow with CO2. Another simulation model for foaming process was projected by Sun et al. in which the concentration layers was used in the model and the effects of physical properties of the polymer on the process were investigated. 25
In this study, the bubble nucleation and growth in LDPE/N2 were studied using a batch foaming system. The bubble radius and density versus time were then plotted at various temperatures. The bubble nucleation and growth were also simulated using numerical models. The effect of various simulation variables on the bubble growth was investigated. Normally, considering the numerical model and our experimental data, we could predict the bubble growth behavior at different conditions.
Experimental
Material
The LDPE (grade of 022a) with density of 925 kg/m3 and average molecular weight of 350,000 gr/mol (MI = 21) was supplied form petrochemical complex, Iran. The melting temperature of used LDPE determined nearly 110°C by DSC (Netzsch-DSC200 F3 Maia) analysis. N2 was also used as the blowing agent. To generate the supercritical phase, N2 gas was pressurized using a plunger pump (Isco 260D, USA).
Characterization experiments
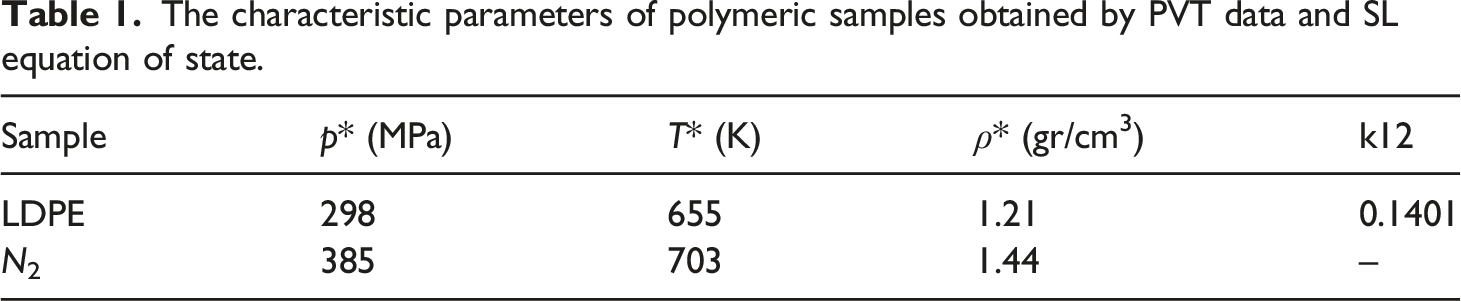
The characteristic parameters of polymeric samples obtained by PVT data and SL equation of state.
The foam visualization was also conducted using a lab-scale batch foaming set up attached with Bell view computer software. 30 The other works also used the similar systems. 31 Our batch system possesses a visual observation chamber made of high-pressure stainless steel. The N2 supplies to the chamber through a mass flow meter. The sapphire window on the chamber together with a high-speed digital camera is designed to visualize the bubble nucleation and growth. The foaming and foam visualization of PVA was conducted during the depressurization step. The recorded bubble nucleation and growth were then analyzed using an image processing software. 30
Bubble growth modeling
For modelling of a bubble growth in a polymer solution, some equations must be used; the force balance on the melt, mass balance of the gas in the polymer melt and the mass balance on the bubble. The shape of the bubble seems to be spherical. The ideal gas equation was used for N2. The Newtonian behaviour was supposed during the entire process. United equation of momentum and continuity of the melt close to the bubble in the radial component of spherical coordinate gives
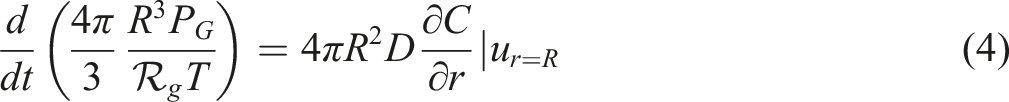

In equation (5), C is the N2concentration and D is the N2diffusion coefficient. Equation (5) describes the concentration profile in the polymer melt. The mass balance equation is expressed by integrating the polynomial profile. To know the concentration gradient at the bubble-melt interface with 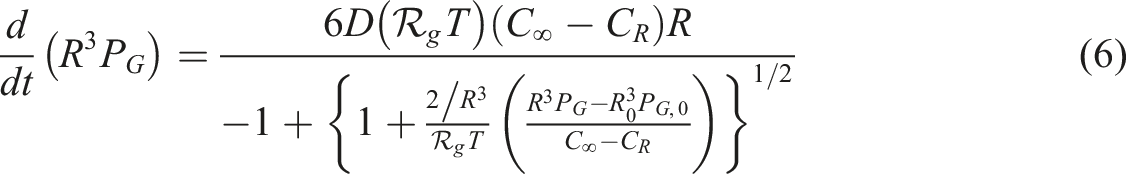

The initial conditions of bubble radius and bubble pressure are as follows

Results and discussion
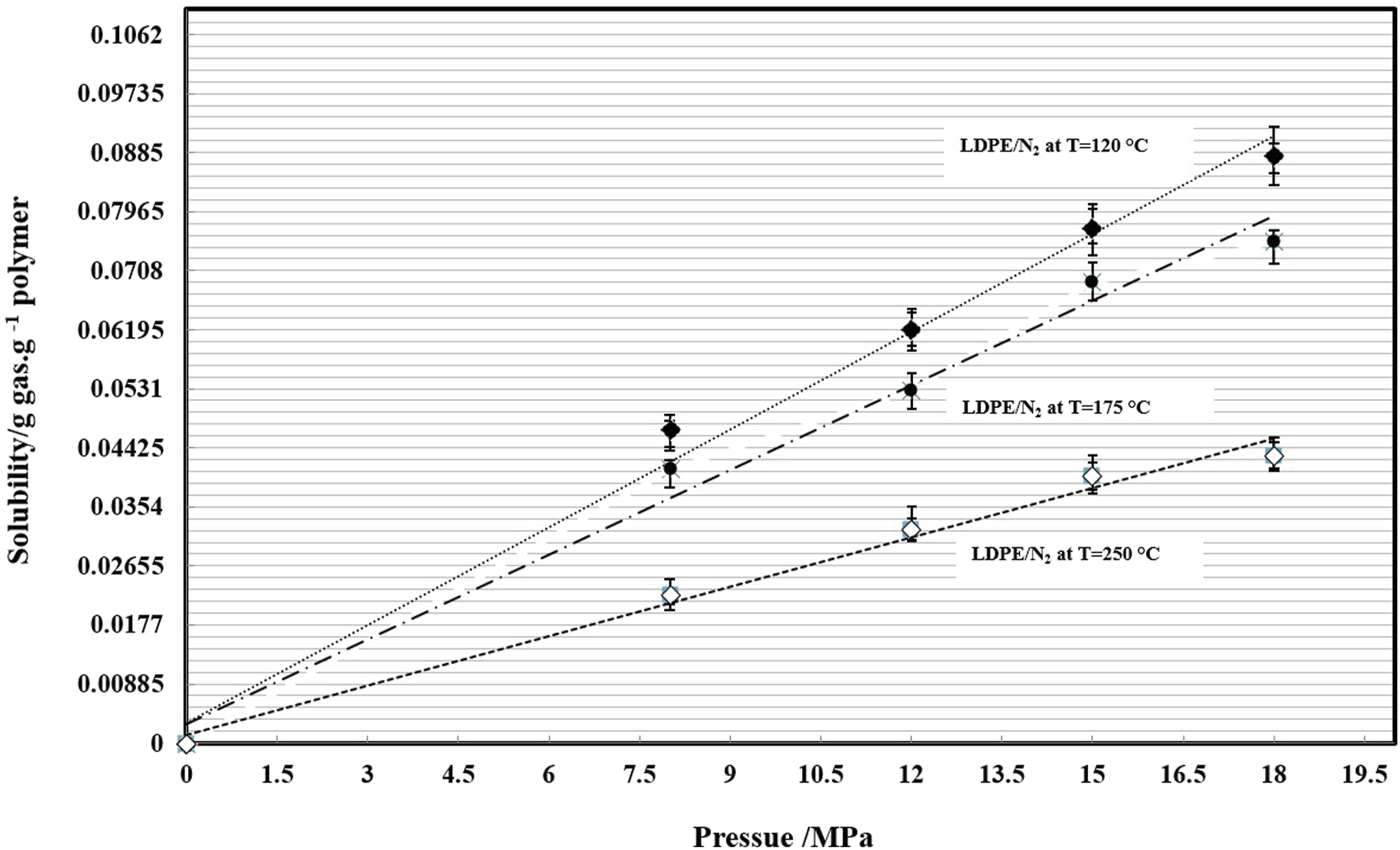
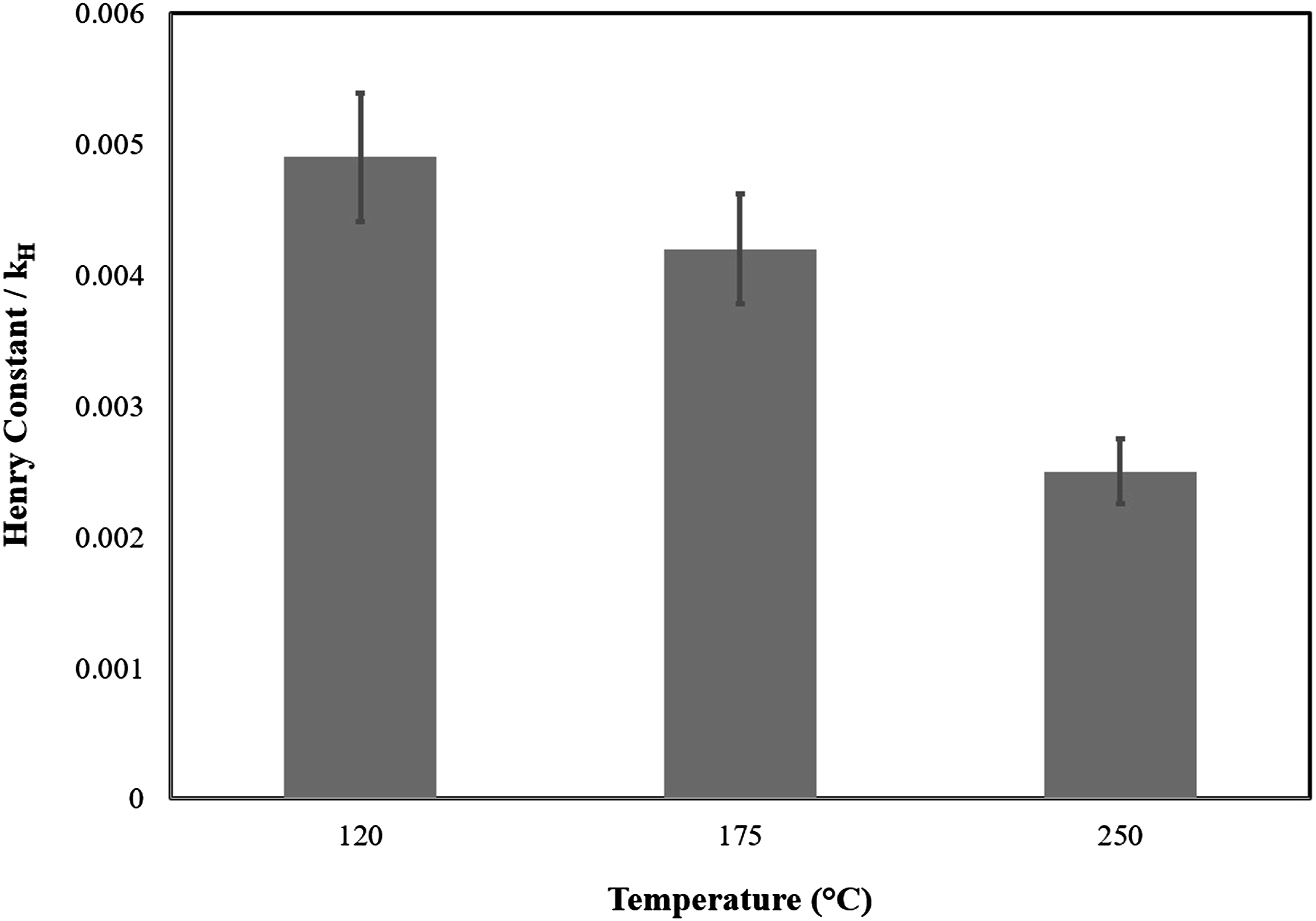
The pressure-volume-temperature data used to derive the Sanchez-Lacombe (SL) equation of state is shown in Figure 1. Figure 2 shows the solubility of N2 in LDPE at different pressures and temperatures, the N2 solubility in LDPE increases with pressure while with increasing of the temperature, the N2 track out of the LDPE and the solubility decreases. The Henry’s constant and its temperature dependency is shown in Figure 3. It is clear that with increasing of temperature the Henry constant decreased. The PVT data used to derive the Sanchez-Lacombe (SL) equation. Solubility of N2 in LDPE at different temperatures and pressures. The Henry’s constant temperature dependency.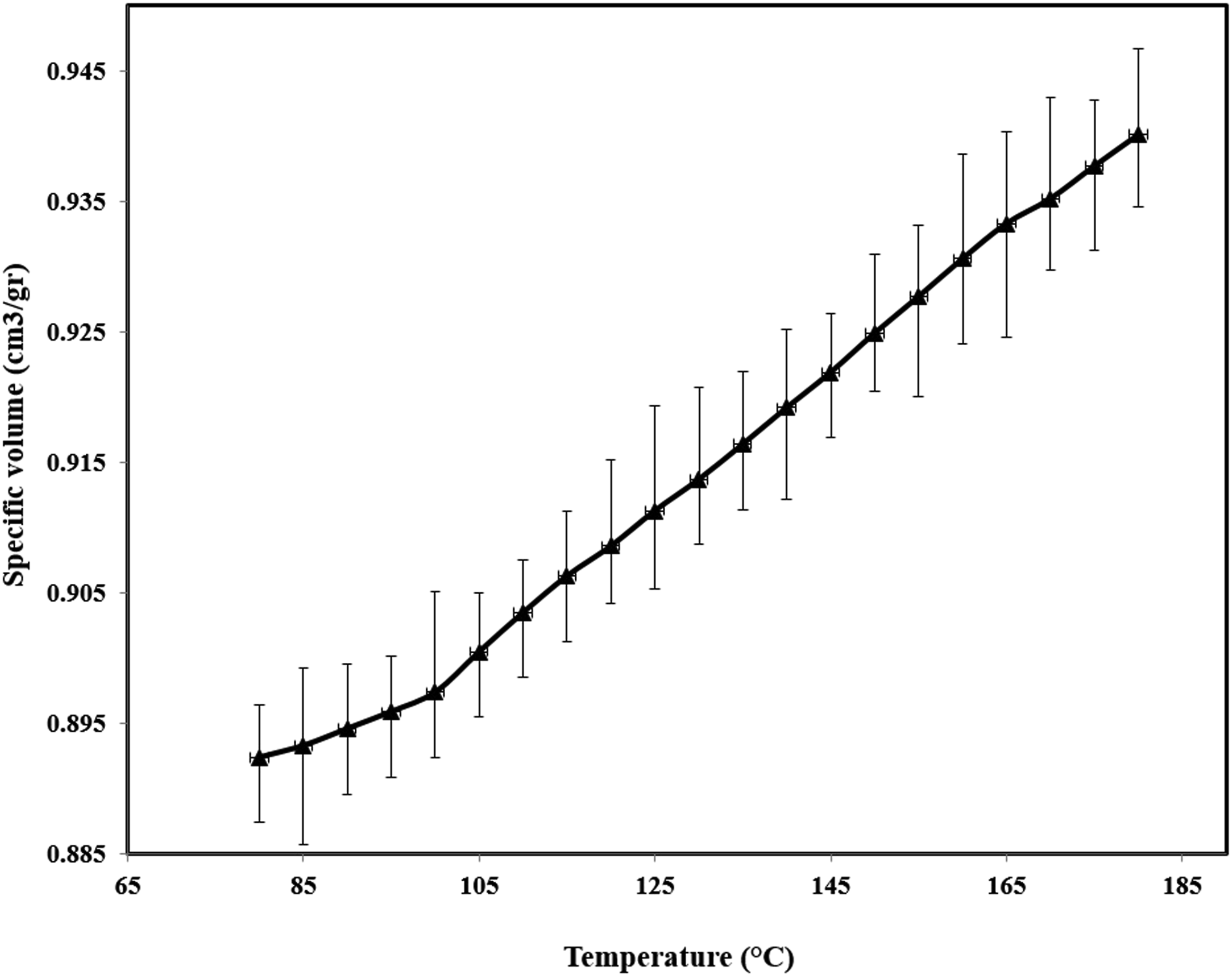
The N2 diffusivity data (Figure 4) are increased by increasing of the temperature and pressure. The temperature dependency of the Diffusion coefficient of N2 in LDPE is determined in Figure 5 which with temperature increment the diffusivity data increased. In some works the diffusivity of N2 or other gases in LDPE was determined but the grade of used polymer and temperature and pressure ranges cause the differences in diffusivity data.32,33 To run the experiments, the sample were saturated at 175°C and N2 pressure of 9 MPa. The foaming and the bubble visualization were then conducted during the depressurization step (1.44 MPa/s). The recorded micrographs during the process are shown in N2 Diffusivity in LDPE at different temperatures and pressures. N2 Diffusivity tempetture dependency of LDPE.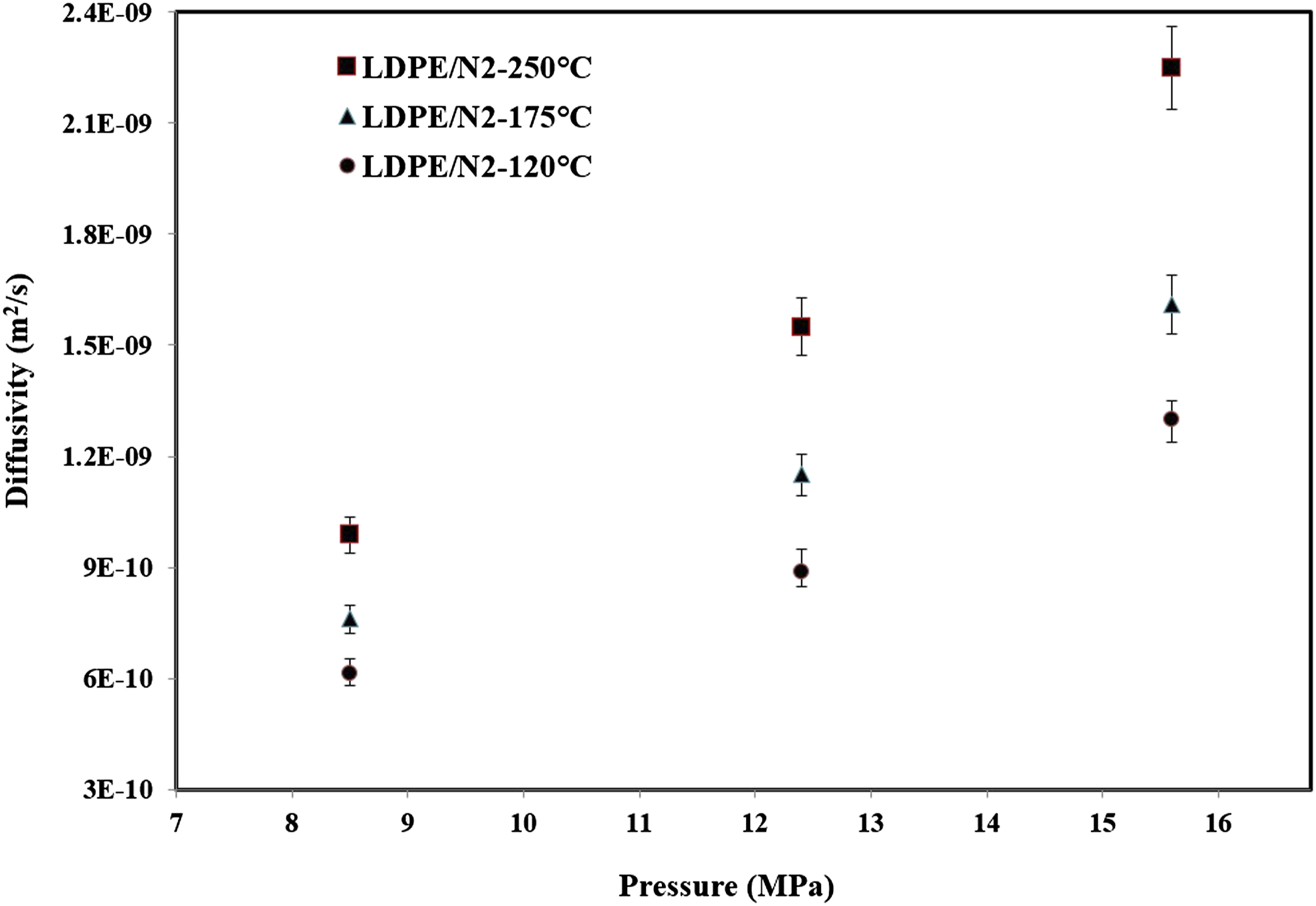
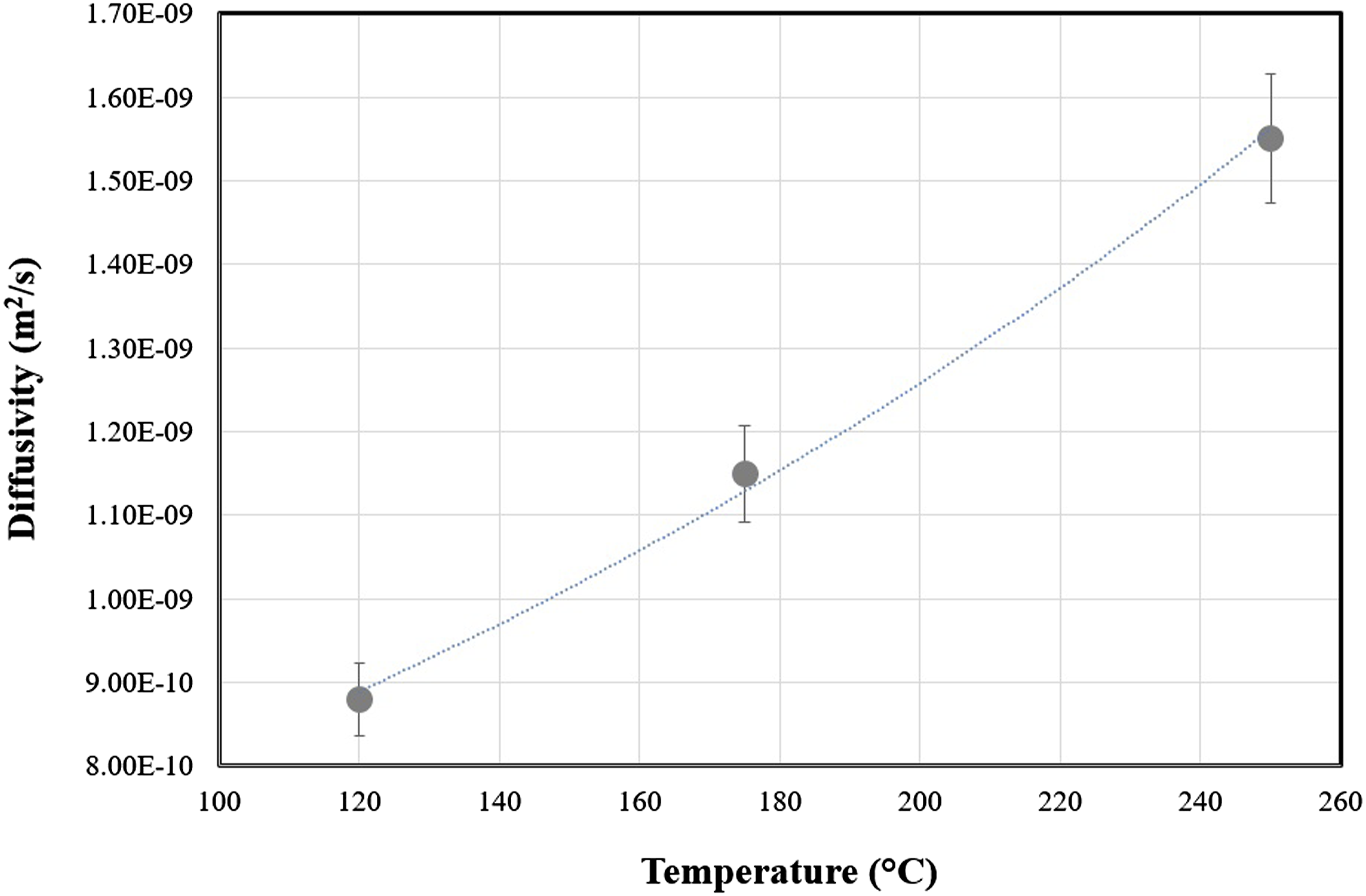
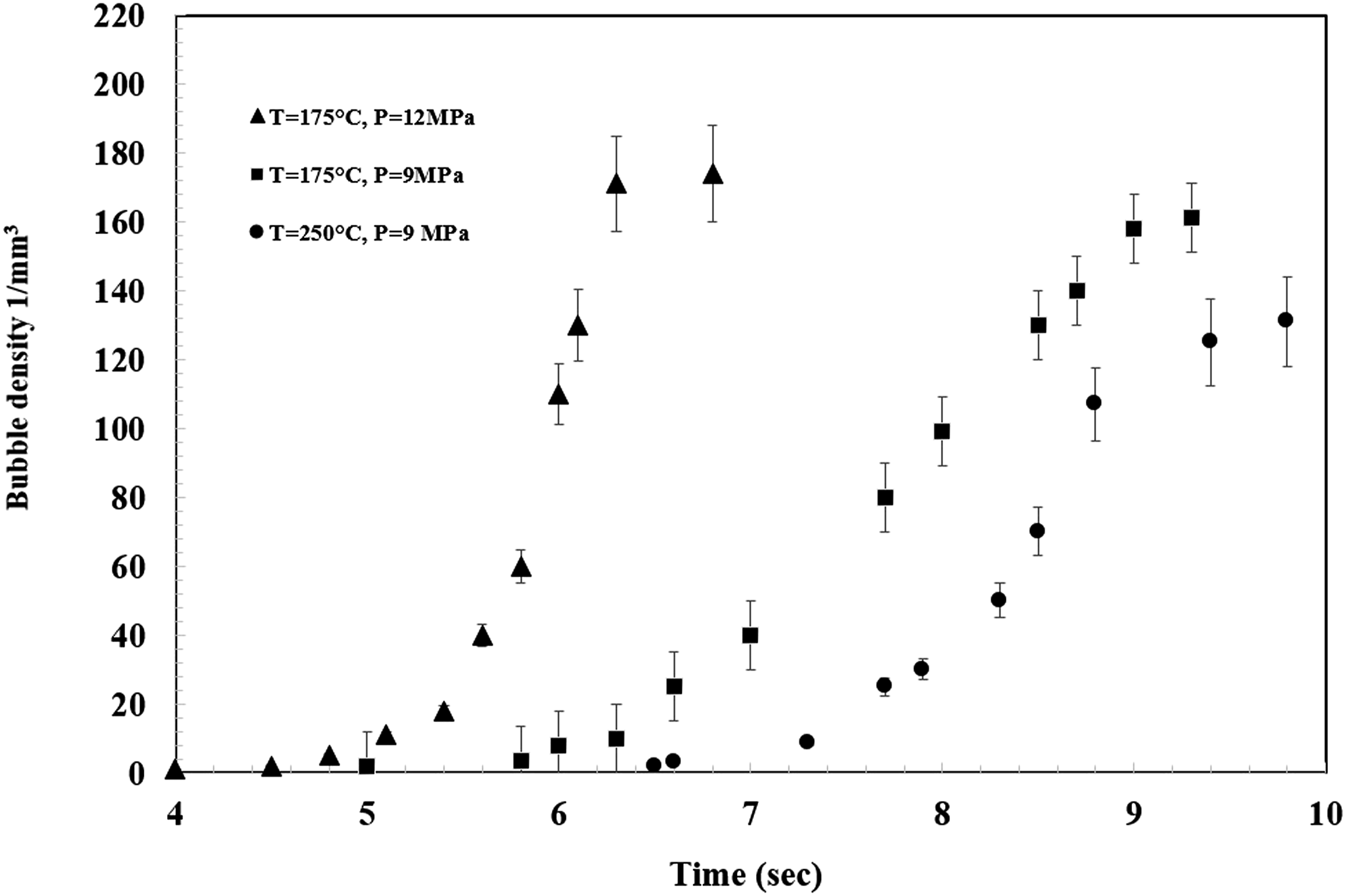
Figure 6. Furthermore, the bubble density could be determined at foaming times. The bubble density versus time at different temperatures and pressures is showed in Figure 7. It is clear that at constant temperature, as the pressure increased, the bubble density increases. In this state the available gas for dissolving in polymer increased and more amount of N2 diffusive into the polymer, so the bubble density increased. In constant pressure, with increasing of the temperature, the available N2 for diffusing into the nucleated bubbles in polymer decreased and the bubble growth could also happen slower. Therefore the bubble density decreased with temperature increment. Optical micrographs recorded during foam visualization at pressure release rate of 1.44 MPa/s. Effect of pressure and temperature on the average bubble density of PVA.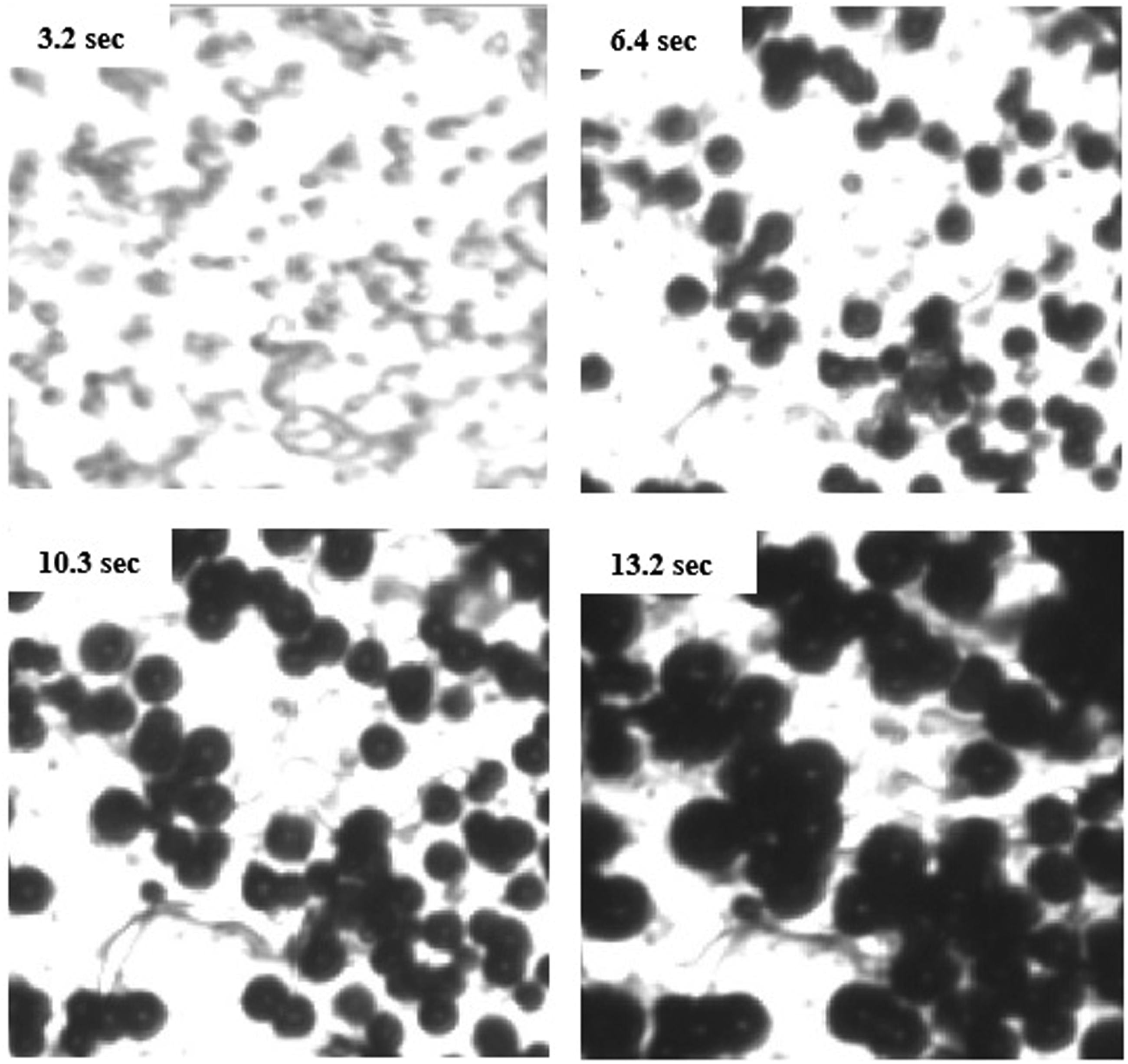
The parameters for the LDPE/N2 foaming simulation.
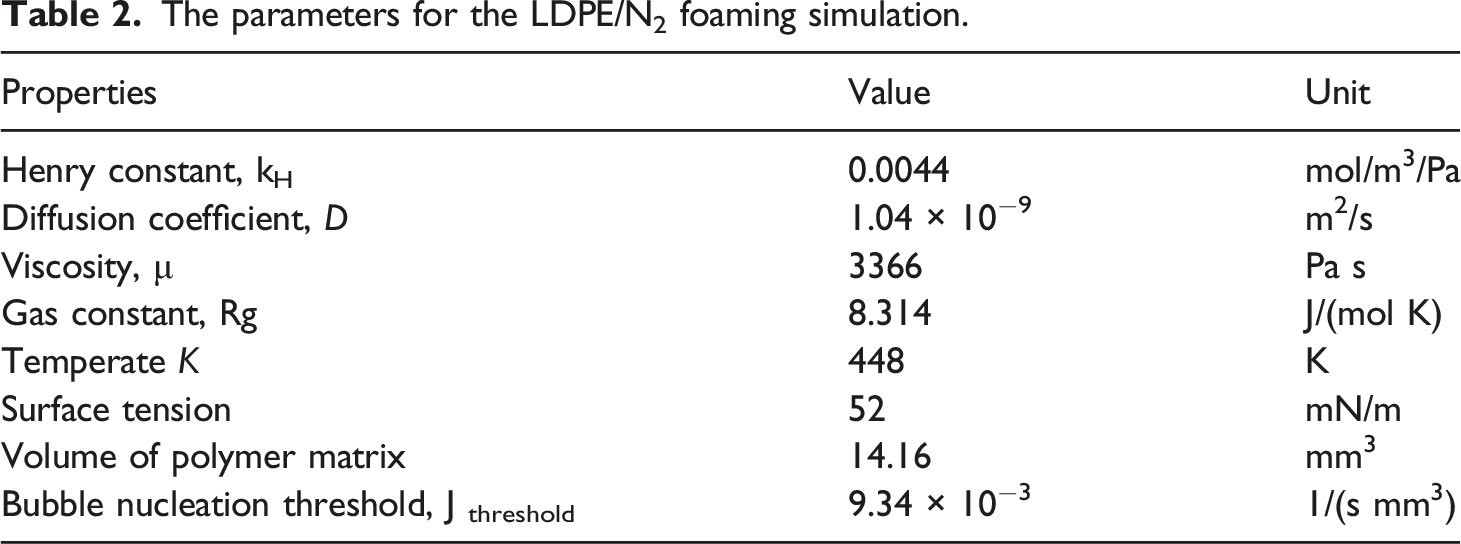
Figure 8 compares the bubble radius values versus time at different conditions. In these figure the model could predict the process, due to a overlapping with the experimental data. When the pressure increased, the final bubble size also increased. A larger pressure means that the LDPE can dissolve more N2 molecules and the N2 solubility increased. At the same time, the higher pressure causes the N2 molecules to diffuse to the bubbles faster and the bubble growth rate will increase. With increasing of the temperature at constant pressure, the vice versa behavior was seen, in which the gas solubility decreased and the final bubble size also decreased. Experimental data and simulated results.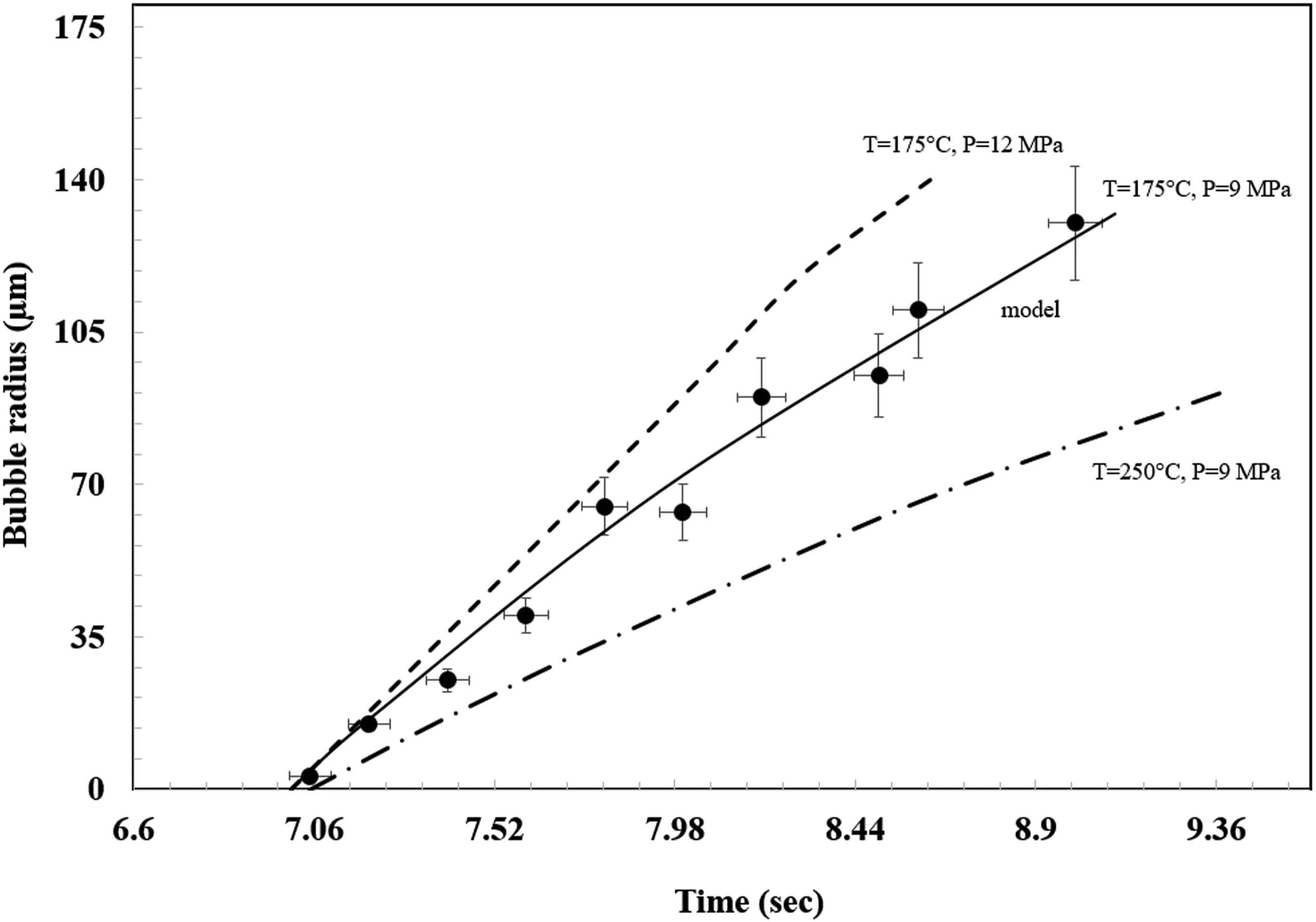
Conclusions
Bubble growth simulation for LDPE/N2 system was done using a visual batch set up. The solubility and diffusivity of N2 in LDPE were determined by a magnetic suspension balance system. It was concluded that the N2 solubility increased with temperature decrement and pressure increment, respectively. As well the diffusivity of N2 in LDPE increased with increasing of temperature and pressure. Considering the recorded images using a visual batch system, the bubble density versus time was plotted at different conditions. It was resulted that with increasing of temperature and decreasing of pressure, the bubble density was decreased. It was shown that the model seems to be reliable with the experimentally values. It was also decided that, we could extract the appropriate parameters and predict the foaming performance considering different factors via the numerical model.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.