Abstract
Current research intended the fabrication of highly durable conducting rubber products of low cost and wide applicability in the field of electronics. Chlorinated natural rubber (Cl-NR) reinforced with copper alumina (Cu-Al2O3) nanocomposites were fabricated by a solvent-free and economically viable industrial compounding technique with special attention to structural, morphological, rheometric curing behaviour, flame retardancy, electrical conductivity, dielectric, thermal and mechanical properties. FT-IR spectra confirmed the presence of Cu-Al2O3 in Cl-NR. The XRD investigation showed the crystalline peaks of Cu-Al2O3 in Cl-NR nanocomposites. The SEM and TEM results indicated that the nanoparticles were uniformly dispersed into the chlorinated rubber in the nano regime. The DSC results indicated that the addition of nanoparticles into the rubber matrix increased the glass transition temperature of the composites. The conductive metal oxide particles in the rubber accelerate the vulcanization process, which could be more beneficial in the industrial sector. The AC conductivity, dielectric properties, tensile strength, modulus, tear strength, heat build-up and hardness of nanocomposites were greatly increased, whereas the elongation at break, abrasion loss and resilience were decreased with the addition of nanoparticles to the chlorinated natural rubber. These increased mechanical properties of rubber nanocomposites with improved processability, controlled morphology and electrical properties are important parameters in the designing of flexible flame retardant electronic devices, electromagnetic induction shielding and conducting adhesive materials.
Keywords
Introduction
Demand for flexible electronic gadgets along with environmental protection leads to a continuous search for bioresorbable polymer composites. Over the last several decades, nanocomposites have opened a hot area in the field of scientific research all over the globe to manufacture high quality polymer products from naturally available resources.1,2 Many research works on natural rubber (NR) have been reported earlier as they are available in plenty and low in cost.3-6 Commercial natural rubber (cis 1,4–polyisoprene) has many outstanding properties such as low hysteresis, high resilience as well as superior dynamic and mechanical properties.7,8 Though the high molecular weight, as well as its long allylic carbon backbone, causes poor resistance to ageing, weathering, abrasion, oil and flame.9-11 Thus, the reactive double bonds present in the backbone could be arrested by various chemical methods to improve its properties. Among the various chemical modifications reported, chlorinated derivatives of NR have shown excellent solvent and oil resistance in addition to good thermal properties.12,13 Optimum chemical and physical properties are obtained by controlled chlorination of NR with special attention to the time and extent of chlorination. Halogenated polymers are widely used in engineering applications due to their high mechanical and dielectric properties, resistance to chemicals, oils and increased flame retardancy and adhesive nature.14,15 The reinforcement of NR with polar fillers or polymers limits its application due to the absence of reactive functional groups in the rubber or the incompatibility between the rubber and reinforcing materials. So, the chlorinated natural rubber not only increases the mechanical and dielectric properties but also enhances the interaction between reactive polymers or fillers with the elastomeric matrix.
Compared to conventional fillers, nano dimensional fillers are effective owing to their large surface area, high surface-to-volume ratio and uniform dispersion in the polymer matrix, which opens ample scope for enhancement in properties of rubber composites.16-18 The key factors that change the processability and performance of rubber nanocomposites are the extent of interfacial interactions and the density of crosslinks developed in the matrix. It is very tedious to adjust the crosslinks between the matrix and the filler particles as it determines the mechanical property, thermal and swelling resistance properties. Nano clay, silica and carbon nanotubes are some of the commonly used nanofillers in rubber chemistry.19-21 Among the broad variety of nanofillers used for the development of rubber nanocomposites, metal nanoparticles having specific functional groups are of great interest due to their inexpensive and easy preparation as compared to expensive nanofillers such as carbon nanotubes and graphene. In addition to this, metal or metal oxide nanoparticles possess high electrical, magnetic, dielectric and thermal properties. 22 Copper is an industrially important metal because of its excellent thermal and electrical properties. 23 However, the structural integrity of copper is very poor due to its shuttling oxidation states. 24 This shuttling of oxidation states can be arrested by doping fine particles of aluminium oxides. Alumina possesses unique behaviour such as high strength, structural integrity and excellent resistance even to very high temperature annealing. 25 Similar to copper particles, alumina also exhibits high thermal and electrical properties. 26 Thus, the alumina impregnated copper has gained much attention due to high temperature applications. 27 The synthesized copper alumina nanoparticles are used as potential filler in NR for the enhancement of thermal, electrical and dielectric properties . 28 However, the mechanical properties of such nanocomposites are poor due to the incompatibility between the polar filler particles and non-polar NR. The incompatibility between NR and Cu-Al2O3 can be overcome by the introduction of a reactive functional group in NR through chemical modifications. The polar functional group present in the chemically modified NR can effectively interact with the polar filler particles leading to accelerated curing with higher dynamic stability. Hence, the present research approach centred on reinforcement of chlorinated NR using Cu-Al2O3 opens a new era in the field of flexible conductive elastomeric nanocomposites.
This article focused on the fabrication of chlorinated NR/copper alumina nanocomposite by a simple and cost-effective two-roll mill mixing technique. The composites are characterized structurally and morphologically by various techniques such as FT-IR, XRD, FE-SEM and HR-TEM. Thermal characteristics are analysed by DSC analysis. The cure characteristics, mechanical properties, flame resistance, electrical conductivity and dielectric properties are also evaluated with respect to different loading of nanoparticles in chlorinated NR matrix.
Experimental
Materials and methods
Natural rubber–ISNR five was purchased from the Rubber Research Institute (RRII), Kottayam. Chloroform (CHCl3), sodium hydroxide flakes (NaOH), cetyltrimethylammonium bromide (CTAB), isopropyl alcohol were purchased from Himedia Chemicals. Dicumyl peroxide (DCP) was obtained from regional chemical suppliers. Cu-Al2O3 with a particle size of 15 nm was synthesized as reported earlier. 28
Synthesis of chlorinated natural rubber
Chlorination of NR was performed by the in-situ reaction of chloroform with aqueous NaOH (70%) in presence of phase transfer catalyst CTAB. For this, 100 g of NR was dissolved in toluene in a reaction vessel. The phase transfer catalyst dispersed in toluene was added to NR solution followed by chloroform and dropwise addition of aqueous alkali. The mixture was stirred for 4 h at room temperature. The product was separated from the solution and washed with water until it was free from chlorine. Finally, the chlorinated NR was coagulated with alcohol and dried to get a constant weight. The chlorine molecules attached to the main chain of NR were determined by chemical analysis and it was found to be 13% chlorine.
Fabrication of chlorinated natural rubber/copper alumina nanocomposites
The reinforcement of Cu-Al2O3 into chlorinated NR was done by a simple and worthwhile mechanical mixing method using lab size two-roll mill mixer. First, the Cl-NR was masticated for 5 min by passing through the two rolls at a frictional ratio of 1:1.25. The Cu-Al2O3 loading in the Cl-NR composite systems varied from 1 to 10 parts per hundred (phr). Different loadings of nanoparticles mixed with CTAB were added to 100g chlorinated NR in an open two-roll mill and the mixing was carried out at room temperature. Finally, the vulcanizing agent dicumyl peroxide (DCP) (6phr) was added to the running compounds in two-roll mill and the mixing was continued for 8 min as per the ASTM D-15-627 method. A systematic abbreviation is used for the prepared composites indicating first the type of rubber (CN) and then the proportion in phr of Cu-Al2O3. For instance, a sample denoted as CN-5 Cu-Al2O3 contains chlorinated natural rubber and 5phr of Cu-Al2O3.
Characterization techniques
A Fourier transform infrared spectrometer (FT-IR series model JASCO 4100) was used for structural analysis of Cl-NR/metal oxide nanocomposites at a scanning rate of 20 and 4 cm−1 resolution from 400 to 4000 cm−1. X-Ray diffraction analysis of copper alumina powder and the thin sheet of Cl-NR with different contents of nanoparticles were recorded on Bruker AXS diffractometer using CuKα radiation (λ = 1.5406 A˚) with an accelerating voltage of 30 KV. The diffractogram was recorded in terms of 2 θ in the range 100–700 at a scanning rate of 10/minute. FE-SEM (Carl Zeiss, VP500, field emission scanning electron microscopy) was used to study the morphology of fabricated composites. A high-resolution transmission electron microscopy (JEM-2100F, HR-TEM, JEOL Co., Japan) was employed to analyse the size, shape and structure of rubber nanocomposites. For this, the composite samples were cut by Cryostat Microtome at a temperature of −40 oC. Shimadzu DSC-50 instrument was employed to find out the glass transition temperature of the samples at an optimum heating rate of 100C per minute. Electrical conductivity and dielectric properties of the Cl-NR filled compounds were determined using a circular-shaped thin film (radius 1 cm and a thickness of 1 mm) by the Hioki impedance analyzer (HIOKI 3570 model) over a frequency range window of 102–106 Hz at room temperature. The cure characteristics of rubber nanocomposites were studied using a Monsanto Rheometer R-100 at a rotational frequency of 100 cycles per minute as per ASTM standard D-2084 (2001) at 1500C. The moulding of each rubber sample was carried out in a hydraulic press at 40 MPa pressure as per the corresponding optimum cure time obtained from the Rheometer. Dumb-bell shaped tensile and angle shaped tear samples were punched out from the vulcanized sheets and the mechanical properties were measured using a Zwick Universal Testing Machine with a load cell of 10 kN capacity as per ASTM D 412–16 and ASTM D 624–00, respectively. The hardness of composites was carried out by Zwick H04–3150 as per ASTM D 2240–95 (2004). The rubber composites having a thickness of 6 mm were used for the hardness measurement. Abrasion loss of the samples was monitored with a DIN abrader (53516). Goodrich flexometer was used to measure the heat generated in the cured rubber composites (heat build-up) during compression as per ASTMD 623–2007. The cylindrically shaped rubber sample with a height of 25.4 mm and diameter of about 12.7 mm was subjected to compressive stresses (108 N) at a frequency of 30 Hz under controlled conditions and the heat generated during the compression was measured. The flame retardancy of the rubber composites was carried out by the limiting oxygen index (LOI) test using a Stanton Redcroft FTA flammability tester, under nitrogen–oxygen atmosphere as per ASTMD-2863-08. The thin sheets of the vulcanized samples with an overall length of 80 mm and width of 30 mm were used for the LOI analysis.
Results and discussion
Fourier transforms infrared spectroscopy (FT-IR)
The FT-IR spectra of CuAl2O3, NR, chlorinated NR and composites of chlorinated NR with different loadings of Cu-Al2O3 are shown in Figure 1. The FT-IR spectrum of Cu-Al2O3 shows the absorption peak at 544 cm−1 is due to the Cu–O stretching.
27
The bands observed at 3399 and 1626 cm−1 are the absorbed water molecules on the surface of the metal-oxide nanomaterial. The spectra of NR show the major characteristic peaks at 2944 cm−1 (stretching of C-H) 1664 cm−1, (stretching of CH=CH), 1452 cm−1 (bending vibration of C-H), 1373 cm−1 (CH3) and 838 cm−1 (vibration of CH2=CH2).
24
The strong absorption bands at 2963 cm−1, 2928 cm−1 and 2862 cm−1 are the stretching vibrations of CH3, CH2 and CH groups in chlorinated NR macromolecules, respectively.
29
The methyl group shows an absorption band at 1381 cm−1 and the band at 981 cm−1 is the cis CH=CH of rubber.
30
The strong band at 833 cm−1 is assigned to =CH wagging and this absorption is commonly used as the fingerprint region of NR. The characteristic chlorinated segment present in the chemically modified NR is observed at 775 cm−1.
31
Cyclopropyl ring formed across the double bond showed an intense band at 1091 cm−1. It can be seen from the IR spectrum of nanocomposites that the attachment of nanoparticles in the rubber is visible as a peak at 521 cm−1 together with all the characteristic absorption bands observed in Cl-NR. Here the metal oxide particles caused a small shift in the wavenumber in Cu-Al2O3 filled Cl-NR as compared to pure Cl-NR. The appearance of a new absorption band with the shift in absolute frequencies of the nanocomposites may be due to the change in geometry by the interaction between Cu-Al2O3 nanoparticles and Cl-NR. FT-IR spectra of NR, Cl-NR, copper alumina and Cl-NR/copper alumina composites.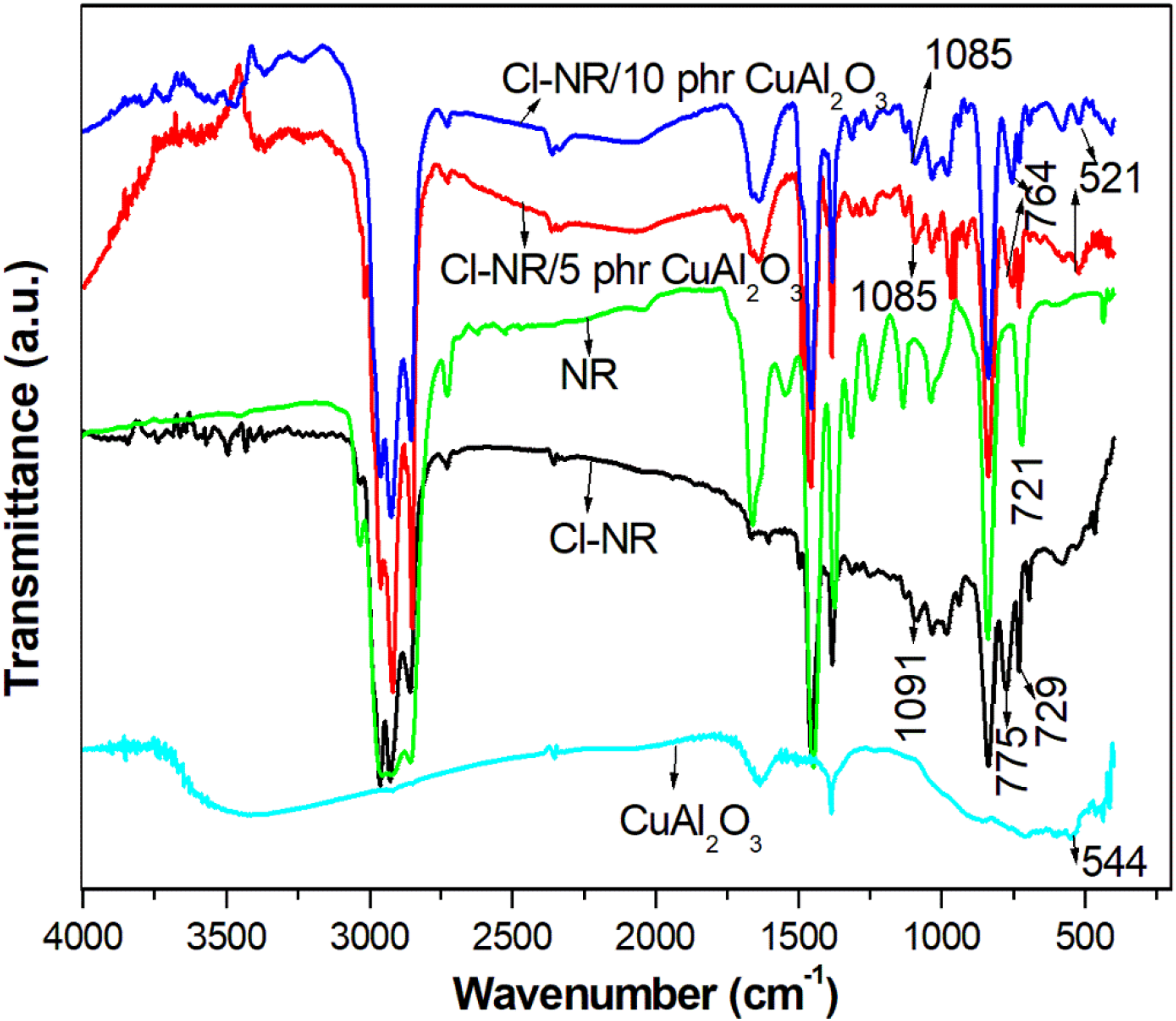
X-Ray Diffraction Pattern (XRD)
XRD is one of the powerful techniques to characterize the structural changes of chlorinated NR chain upon the addition of nano Cu-Al2O3 is presented in Figure 2. The narrow crystalline bands at 2θ = 25.350, 32.520, 35.350, 38.670, 43.350, 48.680, 52.500, 57.640, 60.730, 66.090 and 68.390 are the crystalline characteristics of synthesized Cu-Al2O3 nanoparticles.32,33 The broad peak corresponding to the diffraction of chlorinated NR found at 2θ = 21.810 indicates the amorphous nature of chlorinated counterpart. The additional peaks at 2θ = 29.680, 48.080 specify the crystalline nature of chlorinated NR by the stereospecific addition of dichlorocarbene to the double bonds present in NR. The reinforcement of nanoparticles into Cl-NR is observed in nanocomposites with several crystalline peaks of Cu-Al2O3 with the narrowed amorphous peak of chlorinated NR. For the composite with 3, 5 phr filler content, the amorphous peak at 2θ = 21.500 in Cl-NR is shifted to 19.850. Upon the increase in the content of nanofiller in polymer, the crystalline peak intensity increases and the broad amorphous region of rubber are narrowed with a considerable shift of diffraction peak to the lower region. This improvement in crystallinity can be explained as a result of the interaction of nano Cu-Al2O3 with the chlorinated segments of the NR matrix. Similar results are obtained for chlorinated styrene butadiene rubber with metal oxide nanoparticles.
34
The better filler-polymer adhesion provides a high degree of crystallinity in the chlorinated NR. XRD pattern of Cu-Al2O3, Cl-NR and Cu-Al2O3-loaded nanocomposites.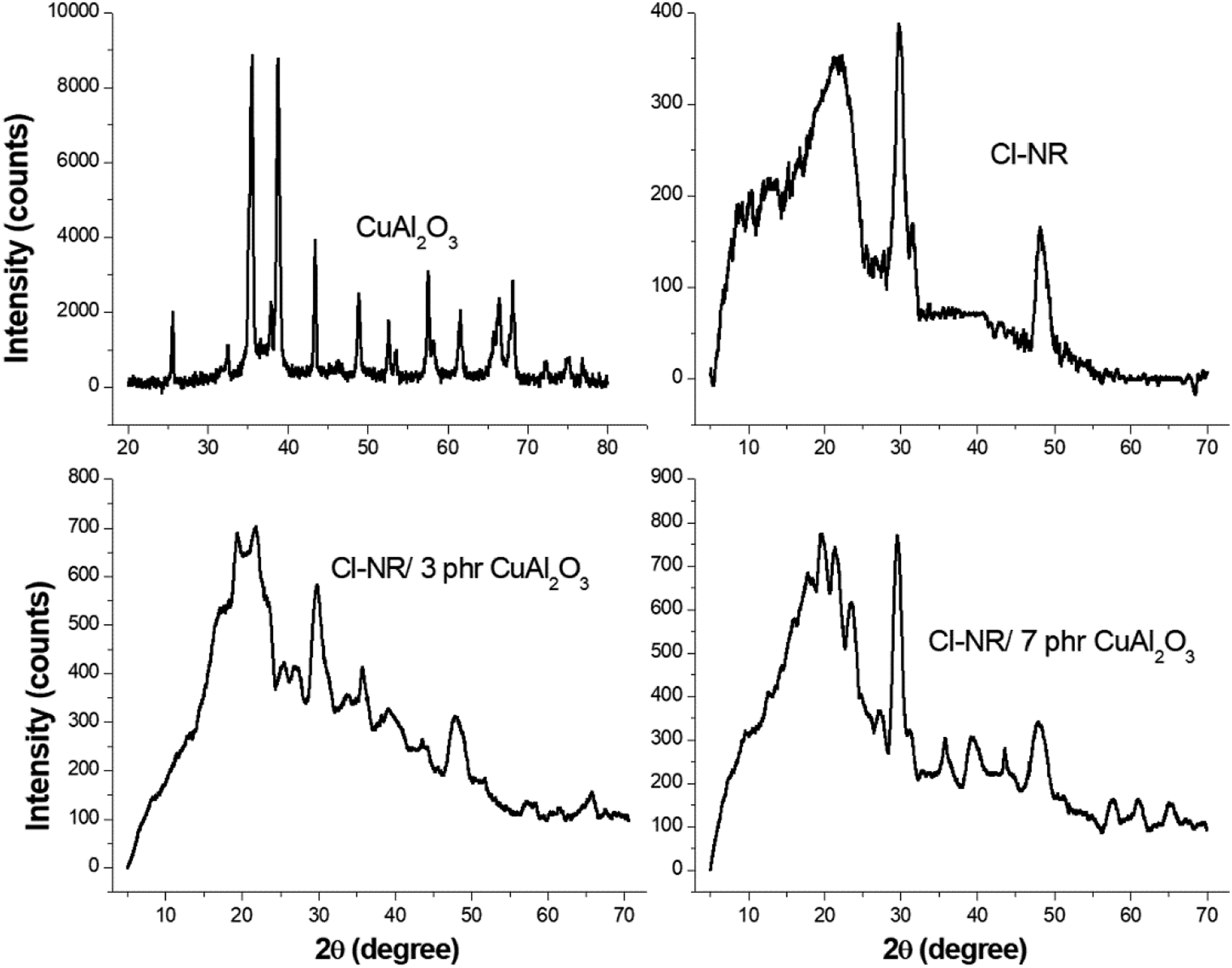
Scanning electron microscopy (SEM)
The surface structure of chlorinated NR with Cu-Al2O3 particles is exhibited in Figure 3. The morphological image of chlorinated NR (Figure 3a) indicates that the chlorination of polymer matrix forms a distinctive layered type structure due to the attachment of chlorine entity on the main chain of polyisoprene. It is evident that the irregular surface of the Cl-NR is changed by the introduction of nanoparticles in the rubber matrix. Composite with 3 phr filler loading shows the uneven distribution of nano Cu-Al2O3 in a localized manner (Figure 3b). In the case of 5 phr nanocomposite, regular and uniform distribution of nano Cu-Al2O3 in the Cl-NR matrix is visible with the absence of layered rough surface on rubber matrix as represented in Figure 3c. This uniform arrangement explains the high interfacial adhesion between the filler and the chlorinated matrix.
35
Figure 3d shows some agglomerations as the filler-filler interaction outweighs the polymer-filler interaction. These agglomerations are caused by the coupling of filler particles as the loading exceeds the limit of uniform occupancy (7phr). SEM micrographs of (a) Cl-NR, (b) Cl-NR/3 phr (c) Cl-NR/5 phr and (d) Cl-NR/7 phr Cu-Al2O3. TEM images of (a) Cl-NR/5 Phr Cu-Al2O3 and (b) Cl-NR/10 Phr Cu-Al2O3.

Transmission electron microscopy (TEM)
TEM micrographs of chlorinated NR filled Cu-Al2O3 are exhibited in Figure 4 Uniform dispersion of spherical shaped particles of nanofiller is well observed in 5 phr loading. The strong interaction of polar chlorinated NR with the filler particles causes the uniform dispersion and this result is in good agreement with the SEM image given in Figure 3c. However, the TEM image of 10 phr filler loaded composite shows an agglomerated structure. Here, greater stress developed at higher loading (10phr) is due to the high filler-filler interaction than polymer-filler interaction which leads to unequal distribution of fillers in the polymer.
Differential scanning calorimetry (DSC)
Figure 5 shows the DSC thermograms of chlorinated NR and its nanocomposites with Cu-Al2O3. The effect of loading of nano Cu-Al2O3 on the amorphous nature of chlorinated NR is identified by the physical changes taking place in the material, in particular, the glass transition temperature (Tg) of composites. This change in physical characteristics (Tg) is determined from the tangents of the DSC profile. Chlorinated NR shows a Tg value at −26.310 C and for the composites with 5 and10 phr loadings, the Tg value is shifted to −23.84 0C, −21.76 0C, respectively. This change in transition is the variation in degree of freedom of the macromolecular chains of chlorinated NR, especially the amorphous zone of rubber by the influence of temperature.
36
The increase in Tg with the loading of filler in the rubber is due to the strong interfacial interaction between Cu-Al2O3 nanoparticles and the chlorinated segments of Cl-NR. The increase in Tg composite indicates the increased rigidity of the material with the effective incorporation of nanofiller. The filler particles present in the rubber composite reduce the segmental mobility of chlorinated NR and thereby stiffness of the material increases and this result is in good agreement with the lignocellulose filled carboxylated nitrile rubber system.
37
DSC thermogramof chlorinated NR and its nanocomposites.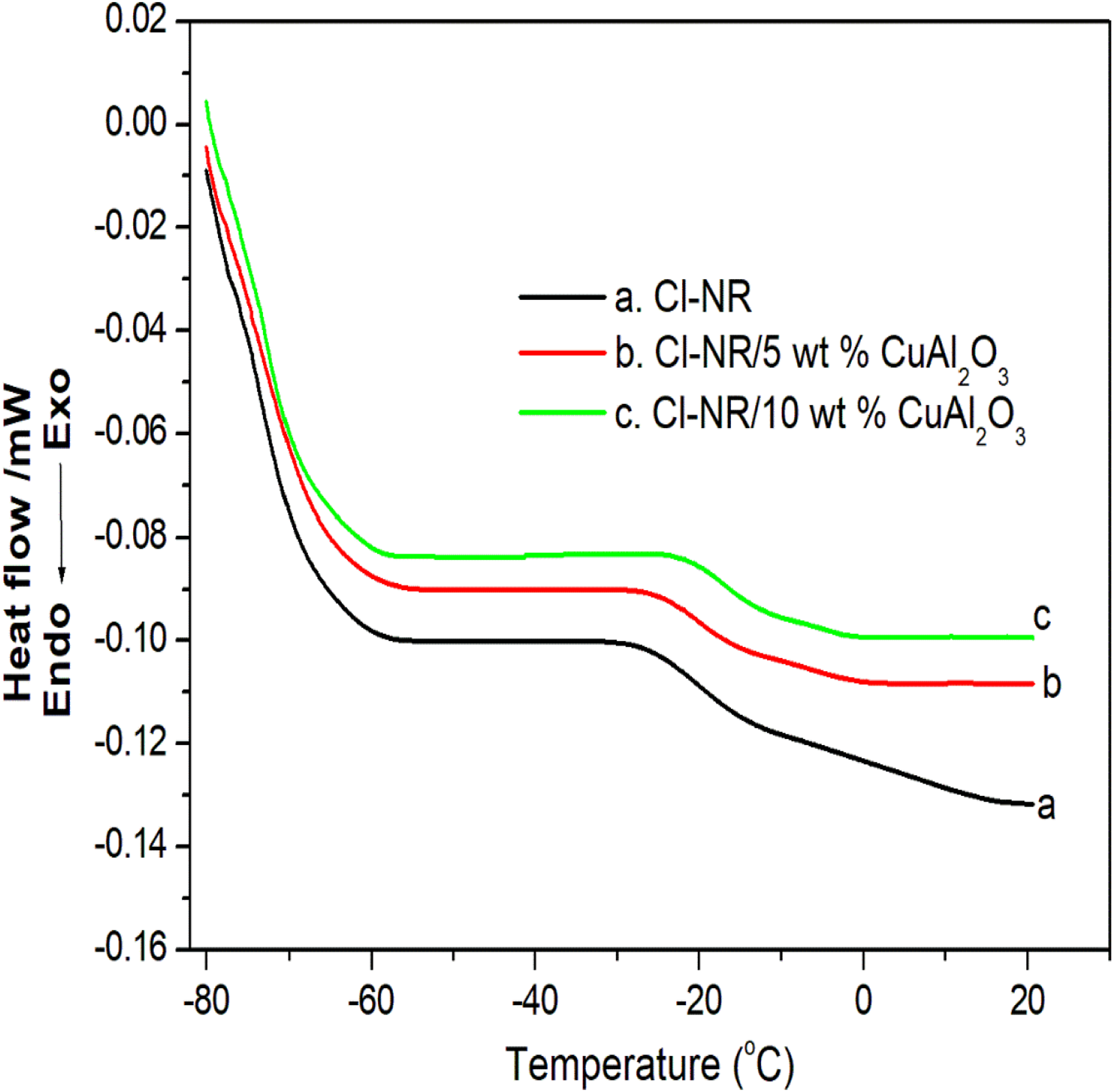
AC Conductivity
AC conductivity of chlorinated NR and composites with different loadings of copper alumina is presented in Figure 6. It can be seen that the addition of 1–5 phr volume fraction of Cu-Al2O3 abruptly increases the conductivity of Cl-NR and the conductivity is also increasing with the frequency as a result of electronic conduction in the polymer matrices. The reinforcement of conductive metal oxide nanoparticles creates continuous dispersion of fillers with regularly packed macromolecular chains in the rubber matrix until a critical concentration is reached (5phr loading). Above this loading, the conductivity slightly decreases with the addition of Cu-Al2O3 because of the establishment of stress developed in the conductive pathways. The interfacial zone between the copper alumina and chlorinated NR counterpart confirms the presence of oriented dipoles, which causes higher conductivity than the bare polymer matrix. The AC conductivity also depends on the crystallinity, polarity and orientation of dipoles in the chlorinated matrix.38,39 It is clear from the XRD data that the crystallinity is found to increase as filler loading increases, thus as a result of increased crystallinity and improved interfacial interaction, the AC conductivity of composites increases. AC conductivity of chlorinated NR and its nanocomposites.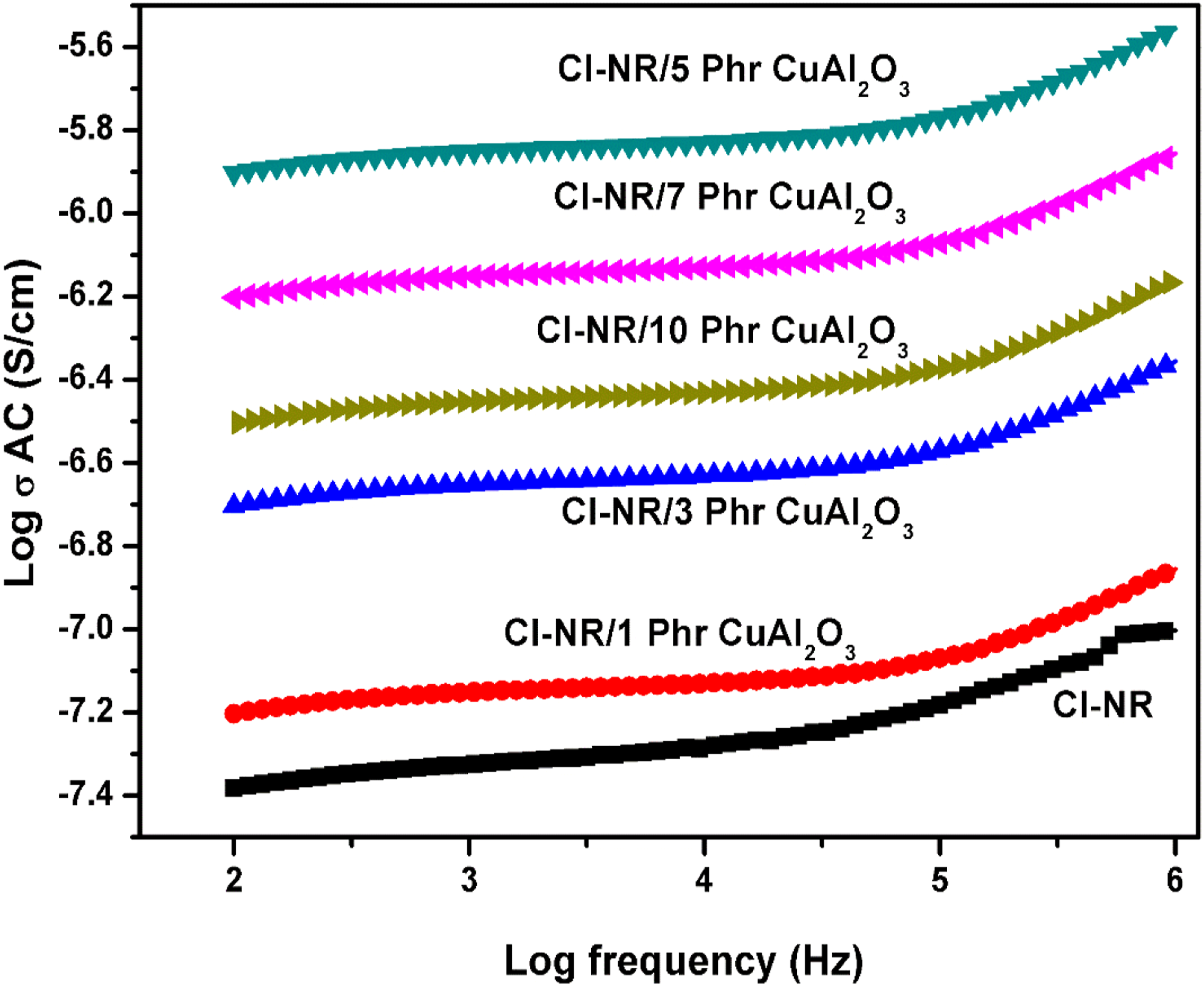
Dielectric constant
The dielectric constant is used to understand the alignment of charge carriers developed in the composites as a result of the polarization mechanism in the polymer matrix upon the application of high frequency.
40
Figure 7 explains the variation of dielectric constant with frequency for chlorinated NR and chlorinated NR with different concentrations of Cu-Al2O3. It is clear from the figure that the dielectric constant decreases as the frequency changes from lower to higher and reaches near a constant value at a higher frequency. At lower frequencies, the polar moieties in the chlorinated NR matrix align uniformly, which increases the dielectric permittivity. Upon the increase of frequency, the dipolar moieties find difficulty to orienting uniformly with the electric field, as a result, the dielectric constant decreases. Copper alumina nanoparticles show ionic polarization and hence the composites show a higher dielectric constant than that for bare chlorinated NR. The maximum dielectric constant is observed for 5 phr loading and the nanoparticles are unevenly arranged in the polymer composites at higher loading of nanofillers (in concordance with the SEM results discussed earlier). The significant increase in dielectric constant for 5 phr loading arises from the localization of charge carriers along with the mobile phases or by the formation of a network inside the chlorinated matrix. Moreover, the interaction between the polar nanofiller and the chlorinated segments of the rubber is responsible for the higher dielectric constant of Cl-NR/Cu-Al2O3 composite. Dielectric constant of Cu-Al2O3 filled Cl-NR at different frequencies.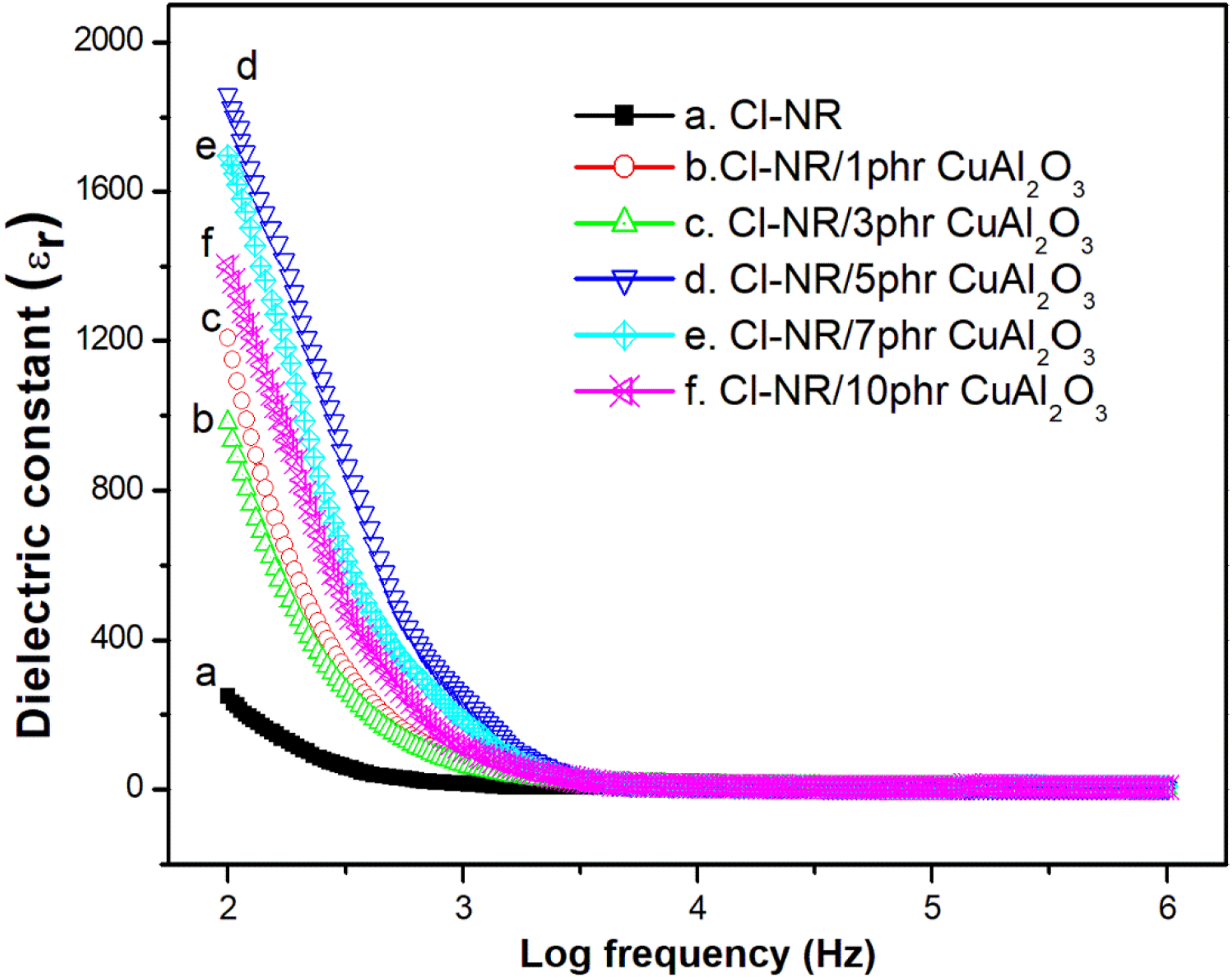
Dielectric Loss Tangent (Tan δ)
Figure 8 shows the variation of dielectric loss tangent with frequency ranging from 100 to 106 Hz for chlorinated NR and composites with different loadings of Cu-Al2O3. All the composites show an exponential decrease with frequency sweep. An initial higher value of tan δ decreases as the frequency increases indicates the rotational motion of polymer chains.
41
The Cu-Al2O3 filled composites show higher tan δ than that for parent chlorinated NR. Dielectric loss tangent is a function of dielectric conductivity and it increases upon the increase in filler loading. Maximum tan δ is thus observed for 5 phr loaded composite and beyond this loading, a slight decrease is found, respectively, for 7 and 10 phr loading. The regular arrangement of macromolecular chains resulting from the adhesion between the polymer chain and nanofiller is observed only up to 5 phr and beyond this loading; the nanoparticle starts agglomeration causing irregularity in the arrangement of filler in the rubber matrix. At high frequencies, the tan δ value of chlorinated NR is observed to be lower than the Cu-Al2O3 filled Cl-NR indicating their application in high frequency electronic devices. Variation of dissipation factor of Cl-NR and its composites.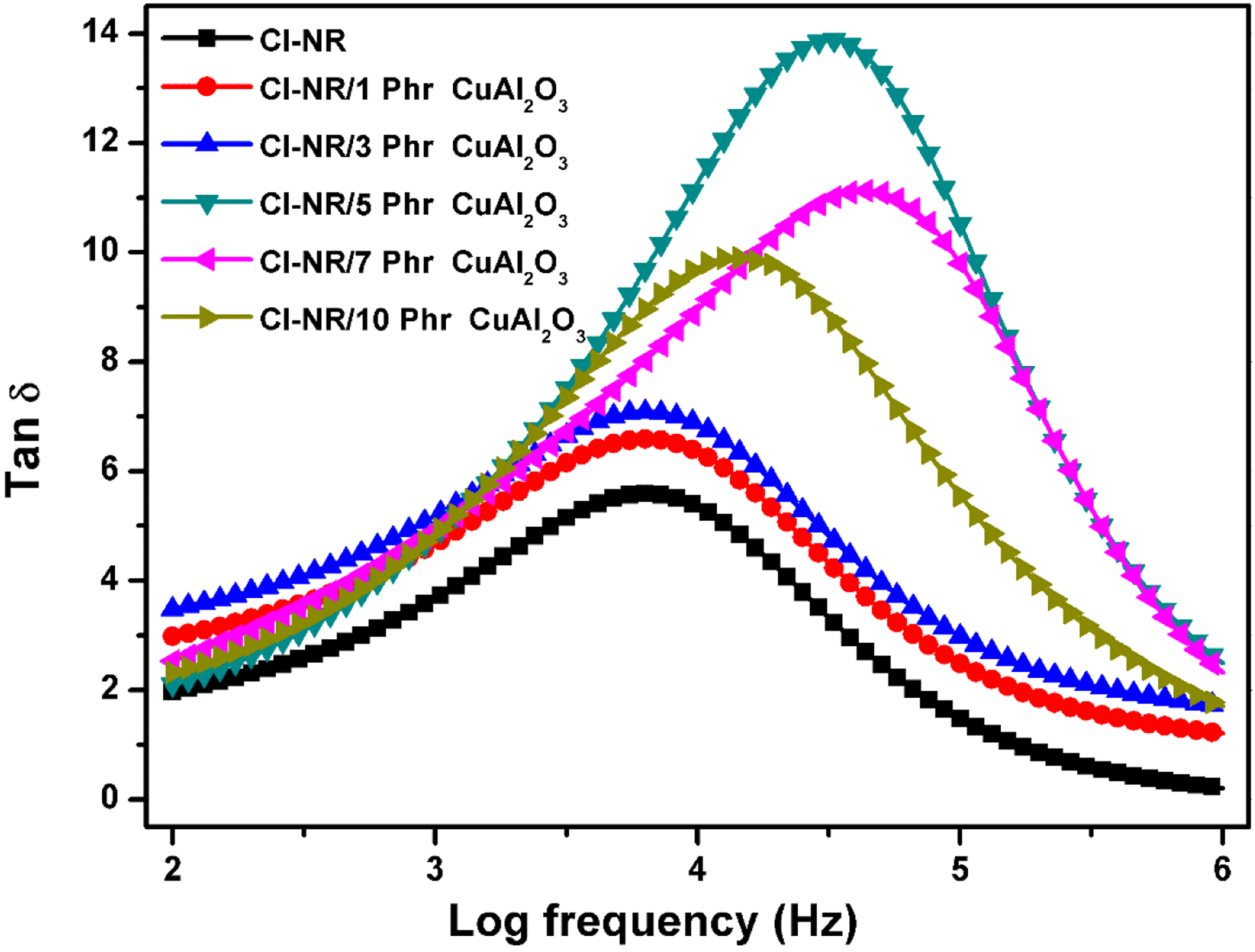
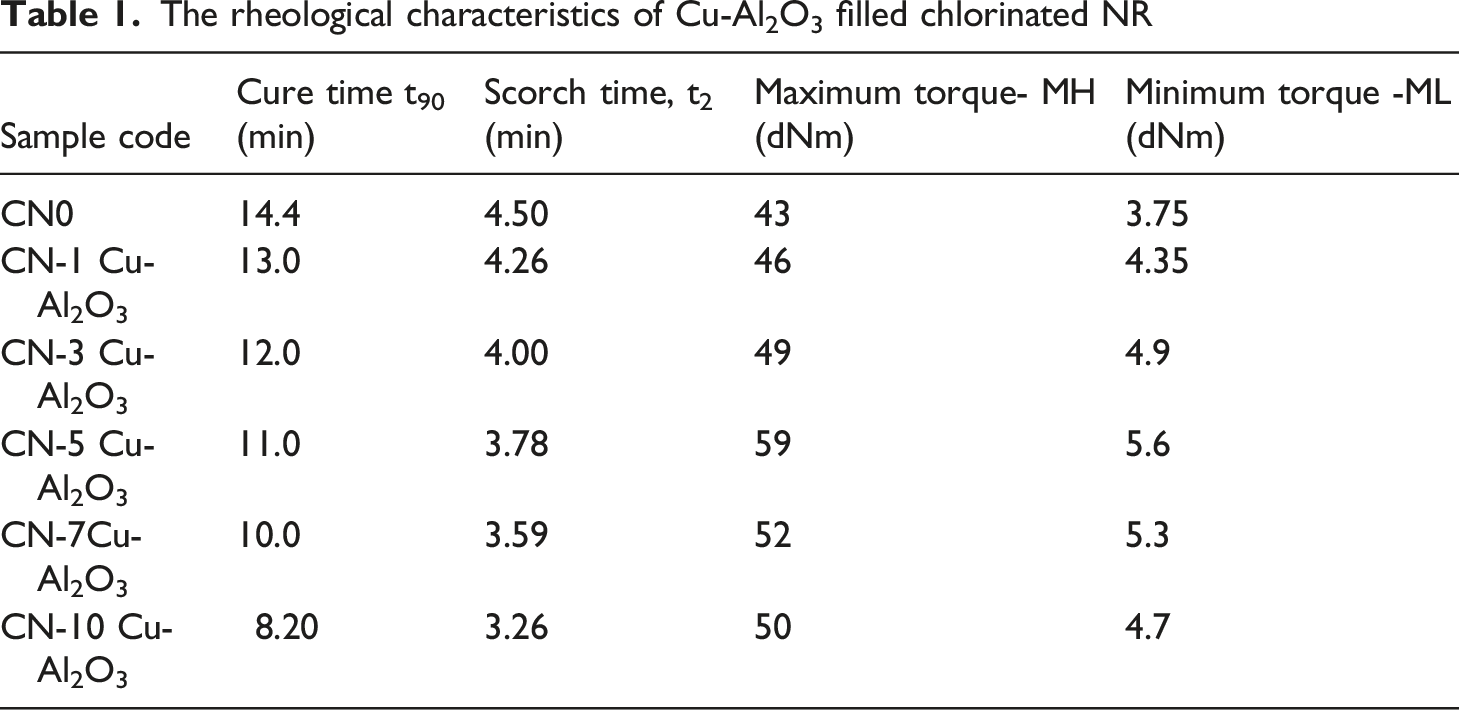
Rheological characteristics of the prepared vulcanizates
The rheological characteristics of Cu-Al2O3 filled chlorinated NR
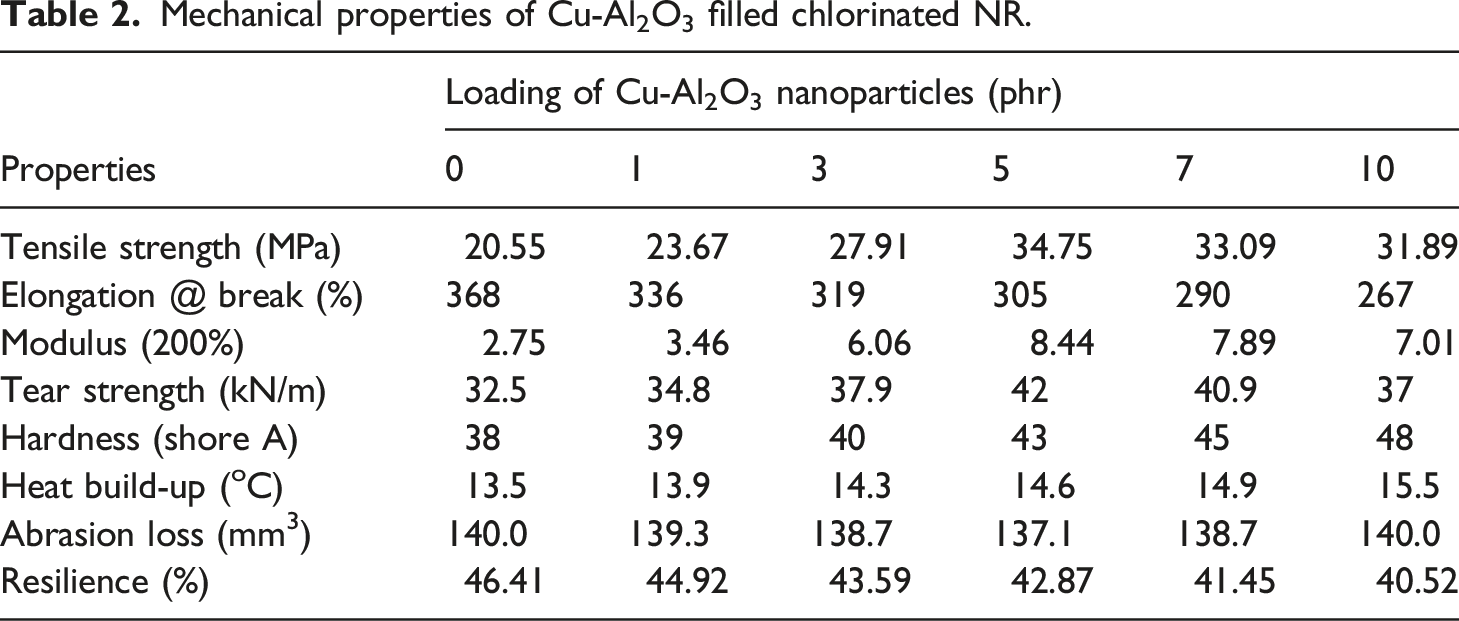
Mechanical properties
Mechanical properties of Cu-Al2O3 filled chlorinated NR.
Flammability behaviour
The flame retardancy of Cu-Al2O3 incorporated rubber matrices is determined as limiting oxygen and the results are given in Figure 9. It is well known that the halogenated segments present in polymer exhibit the intumescence effect. These properties have a great significance in restricting the fire by forming a swollen char or a protective covering on the surface of polymeric materials and thereby resisting further combustion.
46
Chlorinated NR shows the limiting oxygen index (LOI) value of 23.6%, which is higher than the oxygen content present in atmospheric air. It is found that the LOI value increases with filler loading in chlorinated NR, indicating the increased flame resistance of composites compared to chlorinated NR. It is reported in the literature that substances with LOI values above 20–21% are categorized as relatively safe.47,48 Thus, the fabricated composites of chlorinated NR are exhibiting high flame resistance, which are safe products for building materials and indoor applications. The chemical interactions between the metal oxide nanoparticles and the macromolecular chain of rubber affect the heat reduction because of the char layer promotion from the chlorinated segments of rubber resist the burning of rubber composites. Similar results are obtained for the hydroxyapatite nanoparticles reinforced chlorinated nitrile rubber composites.
49
Variation of limiting oxygen index of Cu-Al2O3 filled chlorinated NR.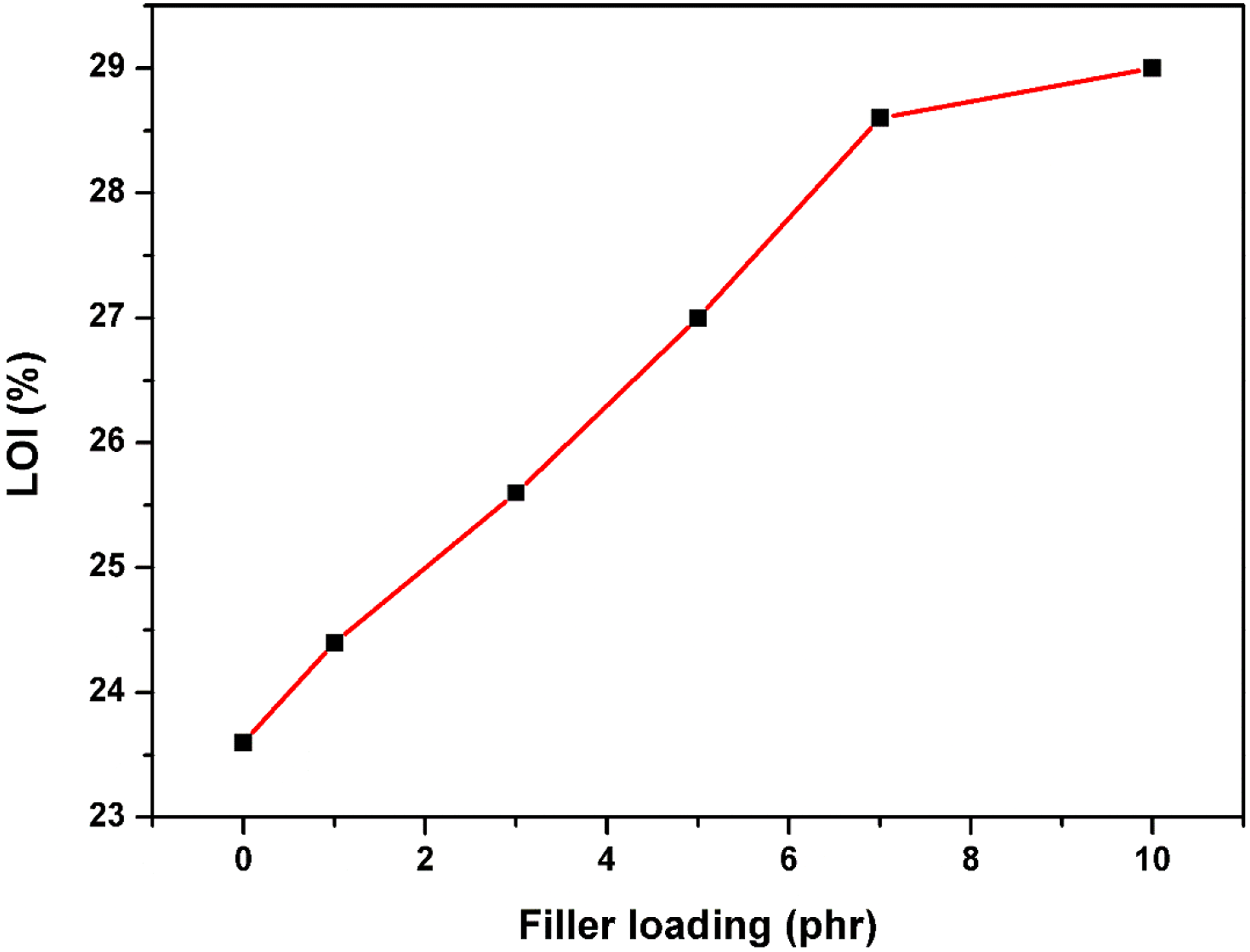
Conclusions
In summary, present innovation demonstrates the reinforcing of copper alumina into chlorinated natural rubber by a simple industrial compounding technique. The FT-IR spectra of Cl-NR/Cu-Al2O3 nanocomposite showed the attachment of nanoparticles at 506 cm−1 along with the characteristic peaks of chlorinated NR. The XRD of chlorinated NR was amorphous while the filled composites showed the crystalline peaks of Cu-Al2O3 indicated the ordered arrangement of nanoparticles. The uniform dispersion of spherical or hemispherical shaped nanoparticles in the chlorinated rubber matrix was evident from the FE-SEM and HR-TEM images. The glass transition temperature of the rubber composites increased with filler loading. The decrease in vulcanization time with the addition of Cu-Al2O3 indicates the easy processability of flexible elastomeric composites. Evaluation of mechanical properties confirmed the increased mechanical strength of fabricated products up to 70% compared to bare polymer. The AC conductivity and dielectric properties showed a major dependence on the filler content. The intumescence effect caused by the chlorine moiety of rubber with the metal oxide particles leads to superior flame retardancy. The composite with 5 phr samples showed the maximum tensile strength, modulus, abrasion resistance, tear resistance, AC conductivity and dielectric constant indicate the strong chemical interactions between the filler and the polymer. From this research, it can be concluded that the Cu-Al2O3 is potential filler in chlorinated natural rubber and it may find applications in electromagnetic interference shielding (EMI), conductive adhesives and flame retardant electronic devices.
Footnotes
Acknowledgements
The authors greatly acknowledge the financial assistance from KSCSTE, Government of Kerala, India (Order No.566/2017/KSCSTE).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Kerala State Council for Science, Technology and Environment (566/2017 kSCSTE).
