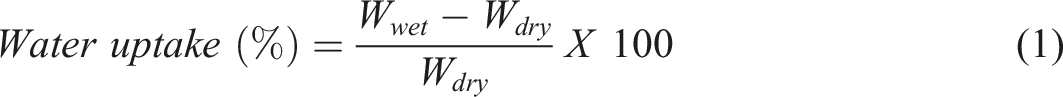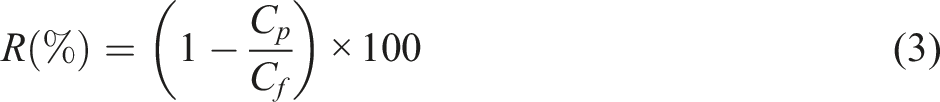Abstract
A novel mixed matrix membrane was prepared by blending polysulfone with two-dimensional boron nitride followed by its hydroxyl functionalization. Prepared nanocomposite membranes were characterized for the surface morphology, surface roughness, zeta potential and hydrophilicity. Uniform dispersion of boron nitride nanoparticles in polysulfone matrix and consequent changes in membrane properties were confirmed by scanning electron microscope images, atomic force microscope images, zeta potential, contact angle analysis, and X-Ray photoelectron spectroscopy. Differential scanning calorimetry (DSC) technique was used to determine the glass transition temperature (Tg) of the modified membrane and the results revealed the presence of BN in the membrane matrix. The highest water flux of 329.52 Liters per sqm per hour (LMH) was obtained in case of PS/BN (1%) as compared to 151.87 LMH of virgin membrane and Albumin rejection also increased by nanocomposites membranes up to certain concentration of Boron nitride. A unique behavior of temperature responsiveness of nanocomposite membrane was observed, where a steep increment in permeate flux by 205% at higher temperature was noted as compared to the virgin Polysulfone membrane where only 103% increase was recorded for the same temperature range. Moreover, the water uptake capacity of nanocomposite membranes were measured to understand the changes with nanomaterial incorporation. The membrane showed a good promise for high-temperature separation applications.
Keywords
Introduction
Membrane-based separation has been emerging as unparalleled and effective technique for water/wastewater treatment owing to its simplicity and low power consumption.
1
. In particular, Ultrafiltration has been used widely to remove dissolved macromolecules and finer impurities from wastewater in industrial applications.2–8 Polymeric membranes are usually preferred for ultrafiltration applications owing to their simple fabrication techniques and ease of achieving pore size of a few nanometers in diameter. These membranes have found applications in various wastewater treatment, dairy industry, bio-separation, and drug purification, etc.9–11 Ultrafiltration membrane can dramatically change flux or rejection in response to change in the operating conditions such as pH, temperature or chemical charge, etc
The inability of the polymeric membrane to resist high temperature limits its application in many industrial wastewater as well as desalination treatments. Such a requirement may have saved the materials and operating cost when operating with a hot stream with a temperature above 50oC.15–20 Various ongoing researches are focused on preparing polymeric membrane having thermoresponsive behavior, which eventually prolongs the performance of membrane at elevated temperature. For instance, Zhao et al. 13 (2013) in their study used PPESK UF membrane for treating condensed water from the petroleum industry and found that flux was stabilized around 80% of the initial value with the rejection of 90% of Iron ion. In another study, Liu et al. 17 (2017) prepared and investigated the PAEK-COOH UF thermal stable membrane behavior on textile dye as feed and found a significant increase in permeate flux from 95 L m−2 h−1 to 138 L m−2 h−1 with a subsequent decrease of rejection from 99.6% to 95.7% over the temperature range from 20oC to 95oC.
Polysulfone (PSf) is a widely used polymeric material for ultrafiltration membrane preparation because of ease of fabrication, resistance to excellent mechanical properties, higher chemical resistance, and wide pH operational range. However, the use of PSf membrane is restricted due to its hydrophobicity as it leads to low water flux and caused more serious fouling. PSf membranes are also more prone to thermal degradation and much vulnerable to fouling when operated at the high-temperature feed for long-run operation.21,22 These demerits could be overcome by blending certain additives, which possessed excellent thermal capacity as well as mitigating fouling behavior in polysulfone matrix.23–26 Various nanomaterials such as bentonite, 27 SiO2,28,29 and TiO2,30–33 with certain modifications have been incorporated in the polymeric membrane for imparting the thermo-responsive behavior of the membrane. With the recent advancement in nanotechnology, a new family of 2-D nanomaterials named “boron nitride (BN)” has gained a lot of attention in recent years in the field of membrane-based separation applications. Boron Nitride has a hexagonal layered structure with unique properties such as low density, high thermal conductivity, stability, high specific surface area and high resistance to oxidation with excellent chemical inertness. More importantly, the BN has a high-temperature tolerance up to 900°C in air, which makes them potential candidates as fillers for the polymeric membrane for increasing thermal permeance at elevated temperature.34–41 Having a higher specific surface area, BN provides an improved pathway for water molecule transport across the membrane, which results in the high rate of mass transfer through the BN channels.42,43 Taking advantage of these intrinsic properties of BN, various researches have been carried out in modifying membrane properties using boron nitride as nanofiller. For instance, Kallem et al. 44 (2021) investigated a 4% Hydroxyapatite/Born nitride-based PES membrane, where the membrane had shown the rise of 97% in permeate flux along with BSA rejection of 90% and a high flux recovery ratio. However, few researches have been done considering the impact of the thermal effect on polymeric membrane incorporating boron nitride (BN).
Considering this research gap, we demonstrate synthesis, characterization and application of mixed matrix membranes by intercalated BN in polysulfone matrix via phase inversion techniques followed by its hydroxyl (-OH) functionalization. Modified Membranes were checked for the potential of higher thermal stability along with water permeability and hydrophilicity. Antifouling properties were also enhanced by the addition of negative hydroxyl (-OH) charge groups on BN modified polysulfone surface. The prepared membranes were subjected to required characterization techniques such as SEM, FTIR, AFM, EDX, XRD, XPS, DSC, and zeta potential measurement. Performance of the membranes was evaluated in terms of pure water flux (PWF), albumin rejection. The thermal behavior of the prepared membrane was also investigated in terms of permeate flux and rejection from a temperature range of 22oC–48oC.
Experimental
Materials
Polysulfone Pellets (from Solvay Specialty Polymers USA), Boron Nitride Nanoparticles (Nano power (<150 nm) from Sigma Aldrich), N-N’ Dimethylformamide (SQ grade of Qualigens from Fisher Scientific India Pvt Ltd. India), Sodium Chloride [LR grade of Qualigens from Fisher Scientific India Pvt Ltd. India), Albumin (Tokyo Chemical Industry Co., Limited Japan), Colloidal silica (LUDOX), Sodium Hydroxide Pallets (Fisher Scientific India), Non-woven polyester fabric (AWA Japan) used as membrane support.
Preparation of mixed matrix membrane (MMM)
All the membranes were prepared through non-solvent phase inversion as reported in the literature.45,46 The dope solution was prepared by blending 15 wt. % polysulfone with DMF solvent. Prepared dope solutions were blended with oven dried boron nitride (BN) nanofillers with different weight fraction of 0.1%, 0.3%, 0.5%, 0.7%, 1.0%, 1.5%, and 2.0%, respectively, by vigorous stirring. This was followed by sonication for 1 h for complete dispersion and additional complete stirring for 30 min. The blended dope solution was poured and casted on non-woven polyester fabric using a knife blade by setting thickness in a flat sheet membrane casting machine; thereafter dipped in deionized (DI) water for gelation and the prepared membranes were kept in water for at least 24 h for complete removal of solvent. Then, the membrane was dipped and preserved in DI water for further testing.
Hydroxyl functionalization of MMMs
All the prepared MMMs were cut into definite sizes and were thoroughly washed with deionized (DI) water. They were then tightly placed on a glass to make sure that only the top layer was treated. The membrane was treated with 0.5 M NaOH solution at 50°C temperature for 1 h. Further, the membrane was cured at 80°C in a vacuum oven for 10 min followed by washing with DI water and further tested.
Membrane characterization
Scanning electron microscope JSM-7100 F (JEOL, Tokyo, Japan) was employed for membrane morphology observation of prepared membranes. Before SEM imaging, Membrane was fixed on metal support using conductive carbon tape and coated with gold by a sputtering device. Energy Dispersive X-ray (EDX) was done to evaluate the elemental composition of BN as well as the BN modified polysulfone membranes.
Surface porosity, pore size distribution, and average pore size were measured by using an open-source image-processing tool ImageJ. 47 For this, raw images from the SEM sample were taken for image processing analysis. The raw images were converted into refined images by removing the background noise. Segmentation of image was done to measure the pore area where, the pore surface was denoted by the darker pixels and membrane surface was denoted by brighter pixels. As a result, the refined image was transformed into a binary image and the pixel count were noted. The surface porosity (Sp) was measured with the ratio of the area of pores to the overall membrane surface area of the SEM image.
Atomic force microscopy measurements were conducted to evaluate the surface roughness of the membrane, using Atomic force microscope instrument NT-MDT, Moscow, Russia. Surface roughness parameters such as average roughness (Sa), and root mean surface roughness (Sq) were evaluated.
BN and mixed matrix BN-PSf membranes were taken for an X-ray diffraction study to analyze the peaks using an X-ray Diffractometer. Fourier transform Infrared spectroscopy of BN, pristine PSf, and mixed matrix BN-PSf membranes were done to study the chemical structural changes on account of boron nitride incorporation in membrane matrix.
Water uptake of the prepared membrane was measured using the following procedure. Membranes were firstly dried and their dry weight (W
dry
) was noted. Afterward, the membrane was kept in DI water for 24 h at room temperature and then, its wet weight (W
wet
) was noted after decanting the adhering water on the membrane. The water uptake of the membranes was calculated using below equation (1)
The surface zeta potential of prepared membranes were determined by an electrokinetic analyzer (Zeta-CAD, France). The surface zeta potential of a tight ultrafiltration membrane was carried out using a 1 mM KCl electrolyte solution.
Membrane performance evaluations
The membrane performances of the membranes were conducted using a cross-flow filtration system with required accessories. The membranes coupons of the area of 14.5 cm2 were inserted in the cells and compacted for 1 h at 3 bars for better permeate flux stability. Afterward, the filtration experiments i.e. pure water flux (PWF), albumin rejection, were performed at the same operating pressure. The pure water flux (PWF) denoted by
Antifouling test
The antifouling test was measured by the change in flux before or after fouling. At first, the pure water flux (J
wb
) of the membrane was measured, and then the prepared membrane was fouled with the solution of albumin (3000 ppm) at the pressure of 10.34bar (150 psi). The volume of permeate was collected every 30 min till 360 min. After filtration for 360 min, the membrane was washed with deionized water for 10 min and then the pure water flux was again measured (J
wa
). The flux recovery ratio (FRR) of the membranes were calculated using the following equation (4)
Results and discussions
BN nanoparticles characterizations
Boron Nitride (BN) nanoparticles were characterized using a range of techniques to study the basic characteristics. Scanning electron micrograph (Figure 1(a)) shows the structure of BN that are arranged in a layered structure with round shapes on the micrometer scale. Energy dispersive X-Ray (EDX) results revealed the purity of BN in atomic % in Figure 1(b). The crystalline structure analysis of BN using X-Ray diffraction (XRD) is shown in Figure 1(c). The XRD spectra show that BN is a highly crystalline structure and its sharp peak observed at 27°. The weaker peaks were observed owing to planes of (100)—(101), (004) and (110) at 42°, 55°, and 77°, respectively. The zeta potential of BN as mentioned in (Figure 1(d)) revealed its negative charge, that is, −26.4 mV. (a) Scanning electron micrograph, (b) EDX Spectrum, (c) XRD spectrum, and (d) zeta potential of Boron Nitride nanoparticles.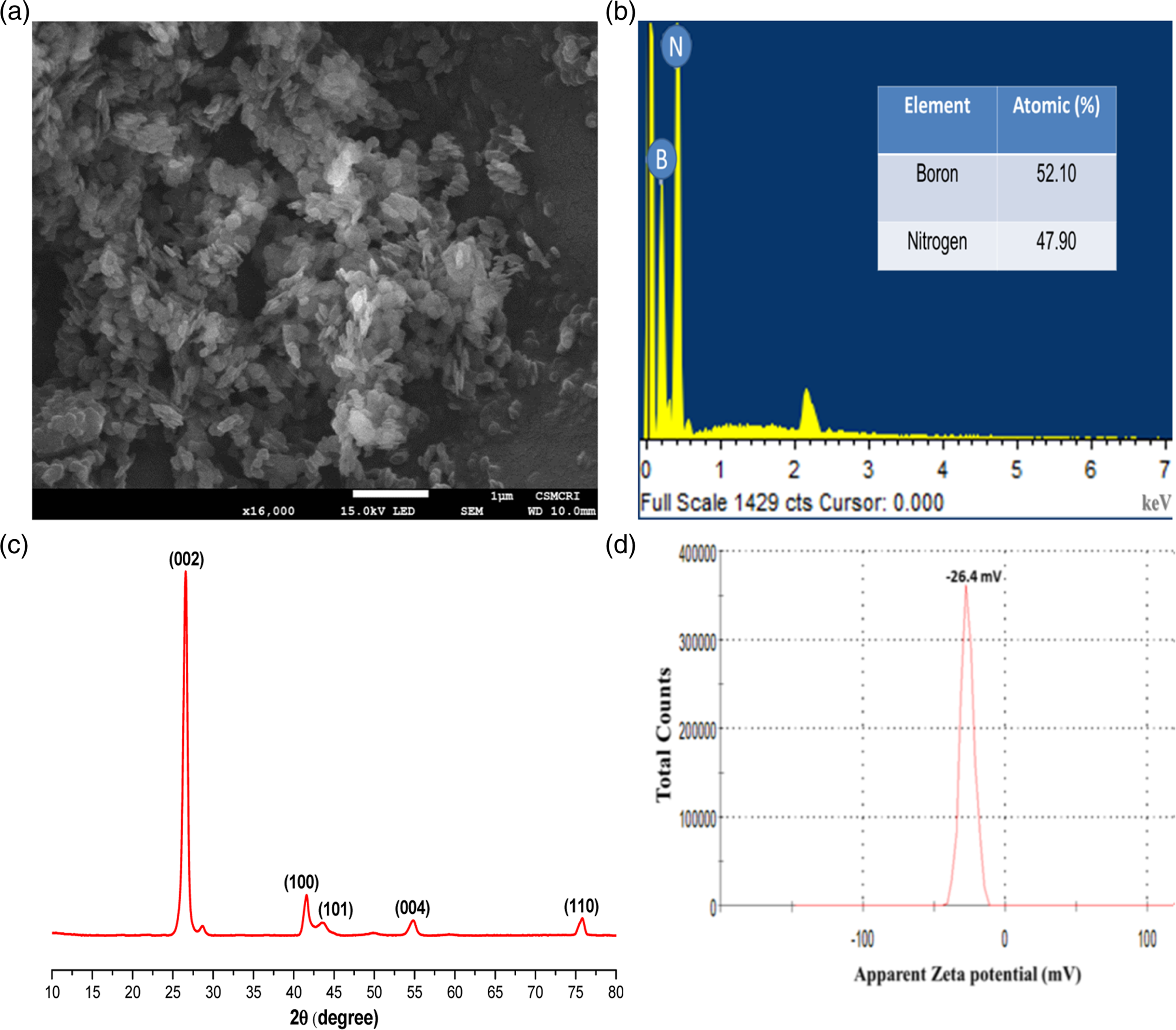
Characterization of membranes
Scanning electron microscopy
The surface and cross-section SEM images of the prepared membranes were obtained at different magnifications to study the effect of BN loadings on the PSf structure. Figure 2 represents the high-resolution top surface images of the prepared membrane. As seen in Figure 2(a) and (e), PSf membranes seemed to be exhibiting smoother surfaces with dense structures. However, with the incorporation of BN, there were substantial morphological changes in the membrane surface as well as in its cross-section. The morphologies of the membrane surface after BN addition showed an increase in the number of pores in the skin layer (Figure 2(b)–(d)). The modified membrane had a typical asymmetric structure which consisted of a finger-like porous sub-layer (2f, 2g, and 2h). The addition of BN increased the number of small pores with smaller pore diameters at the surface, causing a faster rate of mass transfer between solvent and non-solvent at the surface. After the formation of the top surface of the membrane, there was a slower rate of diffusion of solvent from the sub-layer of the membrane to the top surface, which causes the formation of a macro-void beneath the top surface as seen in the SEM cross-section. As shown in the figure, the energy dispersive X-ray (EDX) mapping of PS/BN (2%) membranes have confirmed the uniform dispersion of BN nanoparticles across the cross-section in the polysulfone matrix. The elemental composition (in atomic %) of PS/BN (2%) membrane were boron (17.48%), carbon (68.82%), nitrogen (3.53%), and oxygen (9.50%) (Figure 3). Surface Scanning electronic micrograph of (a) Pristine PSf, (b) PS/BN (0.5%), (c) PS/BN (1.0%), (d) PS/BN (2.0%) and cross section image of (e) Pristine PSf, (f) PS/BN(0.5%), (g) PS/BN(1.0%), and (h) PS/BN (2.0%). EDX mapping and spectra of cross-section image of PS/BN (2%) membrane. Surface porosity, average pore size, and Pore size distribution.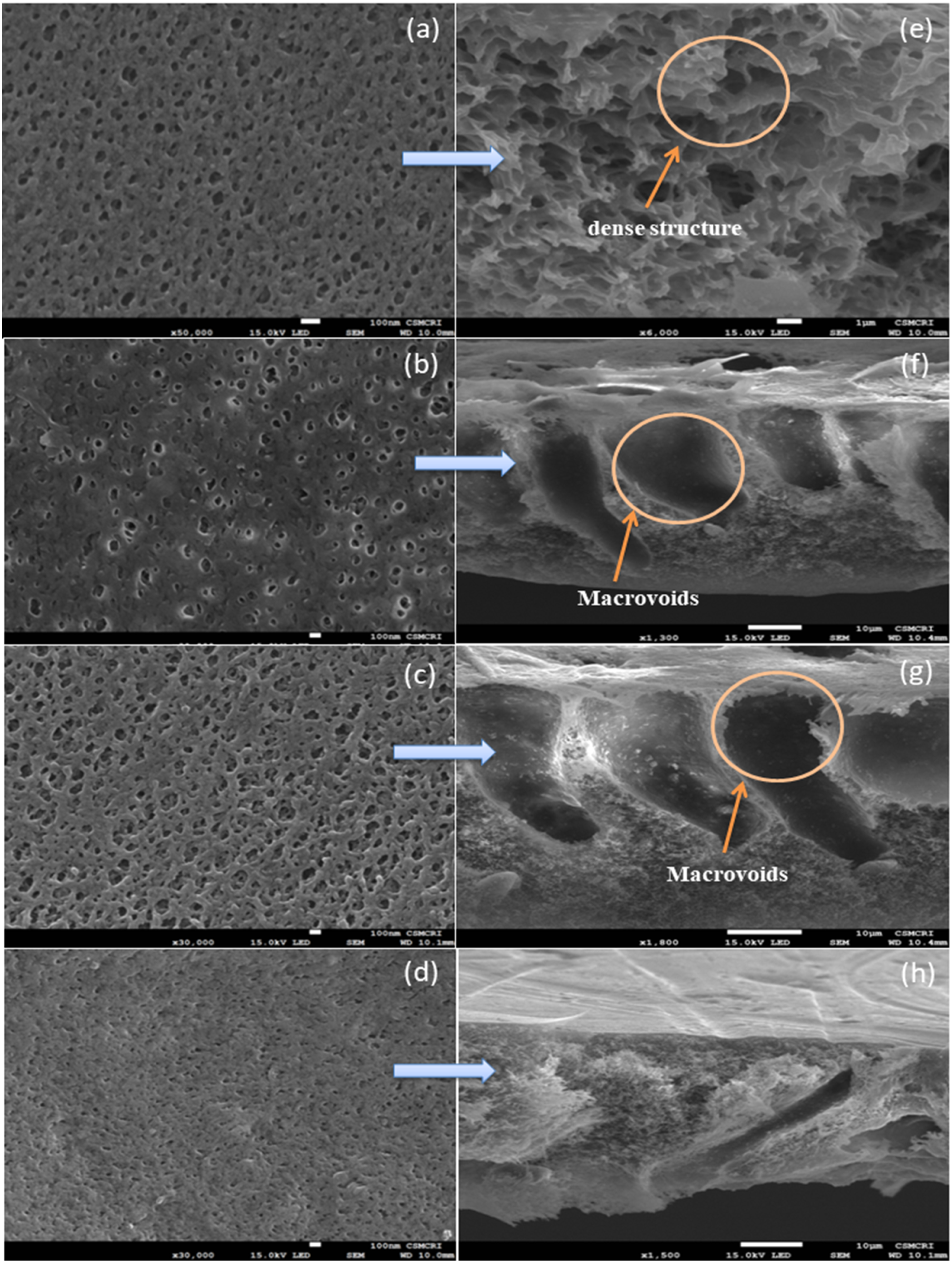

Pore size, pore size distribution, and surface porosity were measured using image processing analysis software. The pore size distribution of PSf, PS/BN (0.5%), PS/BN (1.0%), PS/BN (2.0%) are shown in Figure 4. PSf membrane has large number of pores with a pore diameter range of 60–80 nm and very few pores in the range of 20–60 nm. This trend is shifted to the lower range of pore diameter with the addition of BN nanoparticles. It was found that there was a higher number of small pore compared to the lower number of large pores in case of PS-BN nanocomposite membrane. The average pore size of PSf membrane was found to be 77 nm. Whereas the modified membrane PS/BN (0.5%) had an average surface pore size of 58 nm. It was found that there was a decrease in average surface pore size with an increase in BN nanoparticles loading. The average pore size of the modified membrane with 1% and 2% BN were found to be around 50 nm and 34 nm, respectively. Surface porosity was the measure of pore surface area of the membrane to the total membrane surface area. The surface porosity of pristine PSf was found to be 3.7%. It was seen that surface porosity increased with the addition of BN concentration. The surface porosity of the modified membrane PS/BN (0.5%) and PS/BN (1.0%) was found to be 4.8% and 9.8% respectively. However, there was a decrease in surface porosity with very high loading of BN nanoparticles (2%) and the value obtained was 4.4%. Pore size distribution of PSf and modified membranes.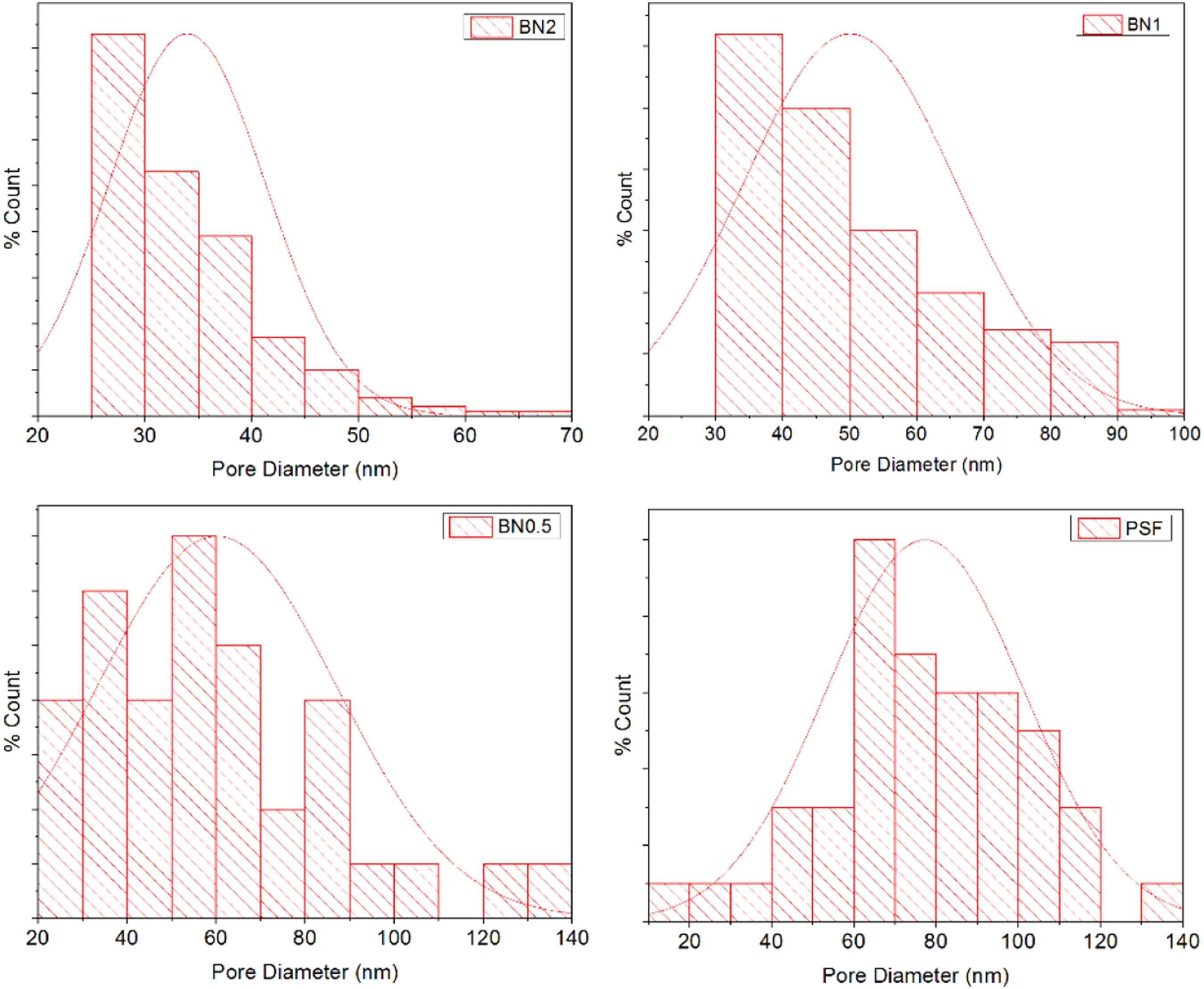
Atomic force microscopy
AFM analysis was conducted to study the effect of incorporation of BN on membrane surface roughness. The 3D AFM surface images of the different PSf, PS/BN (2%) with different loading concentrations scanned over the area of 2 µm x 2 µm are depicted in Figure 5. The roughness parameter represented by average roughness (Sa) and root mean square roughness (Sq) are also evaluated. The Sa and Sq value of pristine PSf membrane were found to be 161.8 nm and 194.4 nm, respectively, whereas for the PS/BN (0.5%), it changed to 239.4 nm and 155.4 nm. Further, the roughness of PS/BN membrane decreased to 155.5 nm and 133.6 nm due to the functionalization of boron nitride with sodium hydroxide. The highest roughness was observed for the membrane with 2% Boron nitride (BN) loading. There was a significant effect on membrane surface roughness with the addition of BN. Roughness parameters such as average roughness (Sa) and root mean surface roughness (Sq) increased with an increase in BN loading. It was due to non-uniform nanostructured BN which created ridges and valleys over the membrane surface. When polymer chains are precipitated during coagulation, they tend to be adsorbed onto the particle surface and form a smaller nodule, which causes the increase in surface roughness.
40
AFM 3D images of (a) PSf, (b) PS/BN (0.5%), (c) functionalized PS/BN (0.5%), and (d) PS/BN (2%).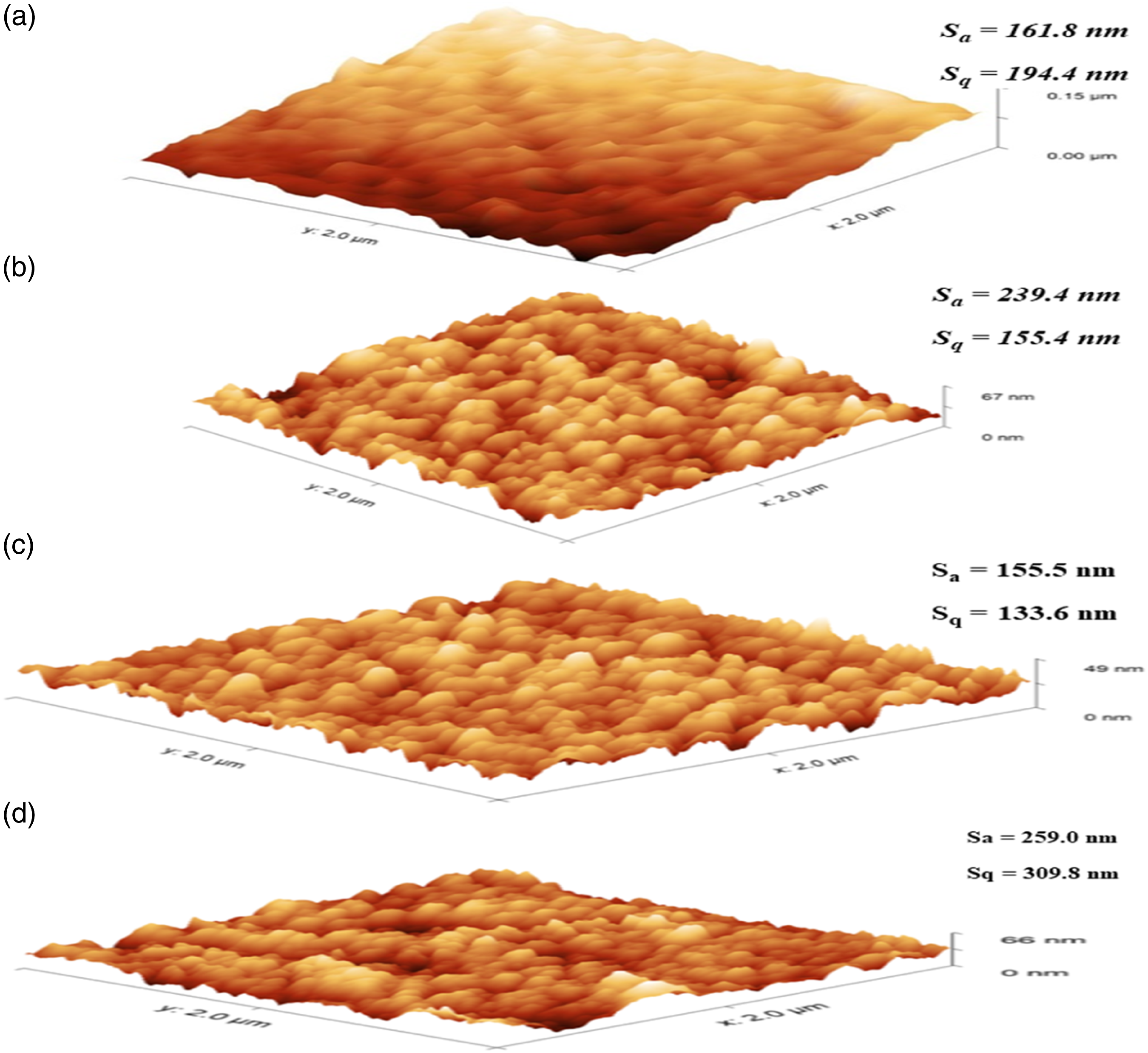
Fourier transform infrared (spectroscopy)
The FT-IR spectra of Boron nitride (BN), pristine polysulfone (PSf), and modified PS/BN (2%) nanocomposite membrane were recorded to evaluate the bonding among PS and BN. Figure 6 shows the FTIR spectra of BN, PSf, and PS/BN (2%). For pristine PS, the absorption bands around 1295 cm−1 are attributed to asymmetric stretching of sulfoxide, whereas the band near to 1150 cm−1 is attributed to symmetric sulfoxide stretching. The band near 1250 and 1040 cm−1 are assigned to C-O-C asymmetric and symmetric stretching, respectively. The band observed at 2874, 2970, and 3096 cm−1 shows the symmetric and asymmetric CH3 and aromatic –CH stretching, respectively. The band at 1585 cm−1 is attributed to the C=C conjugation of the aromatic ring. For modified membrane, PS/BN (2%), Out of plane B-N-B bending and in-plane B-N vibration are accountable for the absorption bands at 1181 and 1413 cm−1, respectively. A broad peak near 3208 cm−1 is attributed to -OH stretching vibration. FT-IR spectra of Boron nitride (BN), pristine polysulfone (PSf) and modified PS/BN (2%) membrane.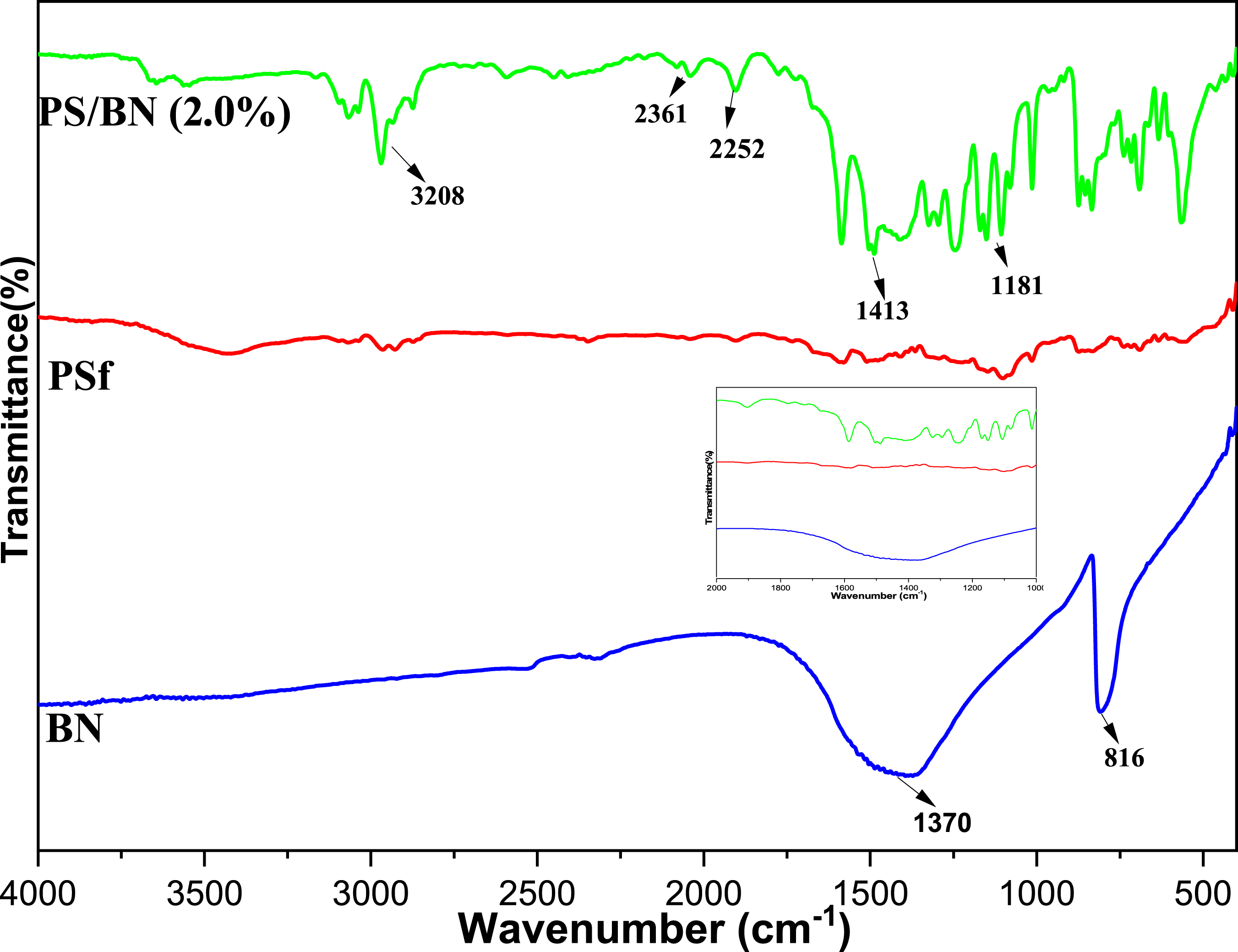
X-ray diffraction spectroscopy
Figure 7 showed the XRD spectra of Boron nitride (BN), pristine polysulfone (PSf), and modified PS/BN (2%) nanocomposite membrane, respectively. XRD spectra of BN are represented and the characteristic 2θ peaks were observed at 26.40o, 41.36o, 54.76o, and 75.92o, respectively.
41
Along with it, the XRD spectrum of pristine PSf shows peaks at around 17o, 23o and 27o indicating an amorphous structure for PSf. BN incorporated PSf membrane revealed the broadening of these peaks and a new peak formation at 32o. This confirms the dispersion of BN within PSf matrix. XRD spectra of Boron nitride and PSf, PS/BN (2.0%) membranes.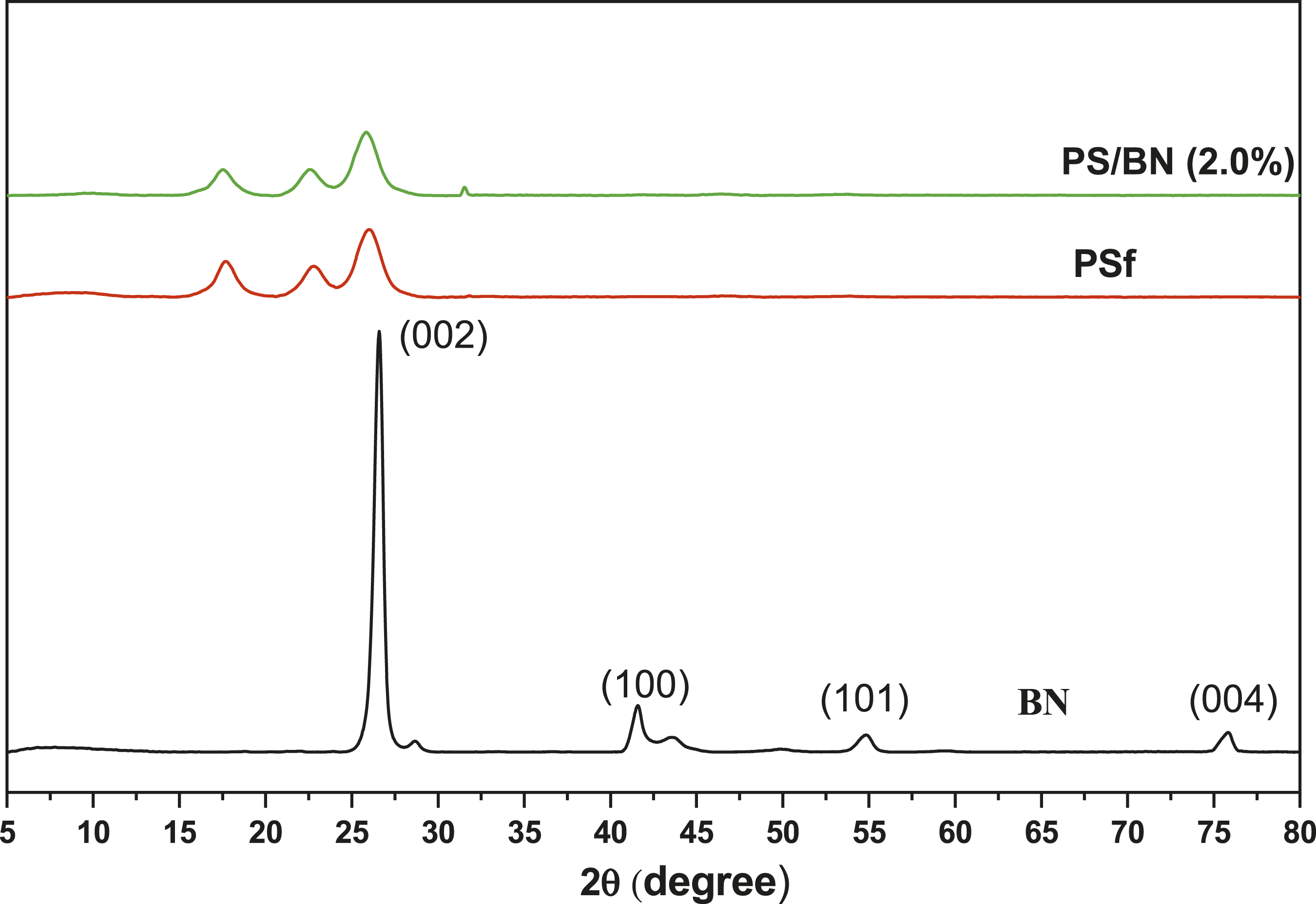
Differential scanning calorimetry techniques
The thermal stability of PSf membrane modified with BN was assessed by differential scanning calorimetry (DSC) techniques, where the data are recorded in heat-flow as a function of temperature. The DSC curve for the PSf and PS/BN membrane was represented in Figure 8. At the heating rate of 10 oC/min, two peaks were observed in PSf membrane at 40.46 oC and 181.26 oC, whereas three different peaks at 42.96oC, 120.02oC, and 180.71oC were observed for PS/BN nanocomposite membrane. For PSf membrane, there was a linear decrease in heat flow rate with increase in temperature throughout the process. Modified PS/BN membrane had shown that the heat flow rate increased because of higher conductivity and specific heat of boron nitride; thus shown a transition at 120.02oC.48,49 For the same temperature range, the thermal degradation of PS/BN required high enthalpy compared to PSf membrane. Similar findings were also seen in some other polymer/BN composites. It was due to the loading of boron nitride, which facilitated the faster rate of thermal transfer in the nanocomposites.50–52 DSC Curves of PSf and PS/BN(2%) membrane.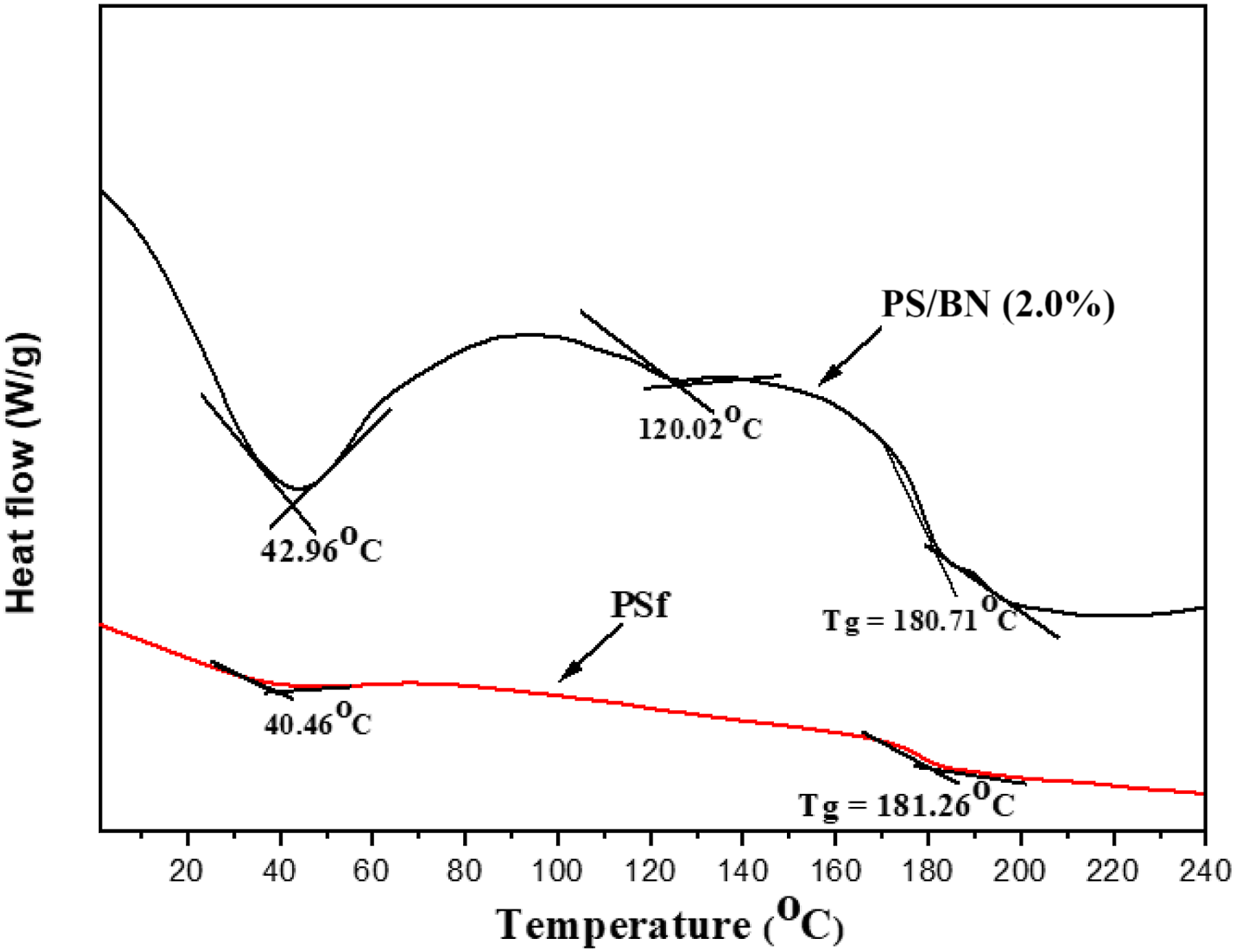
Water uptake capacity and hydrophilicity
The water uptake capacity of the membranes was measured to check the stability. Water uptake of the PSf and PS/BN composite samples is shown in Figure 9. Water uptake decreased with increasing BN content in the composites. Pristine PSf membrane had a water uptake value of 35.77%, whereas PS/BN (1.0%) has the least water uptake value 4.09%. This may be due to the change in pore structure and because of the incorporation of boron nitride in the membrane matrix. However, hydroxyl functionalization of MMMs increased the water uptake capacity. Variation of water uptake capacity with BN loading.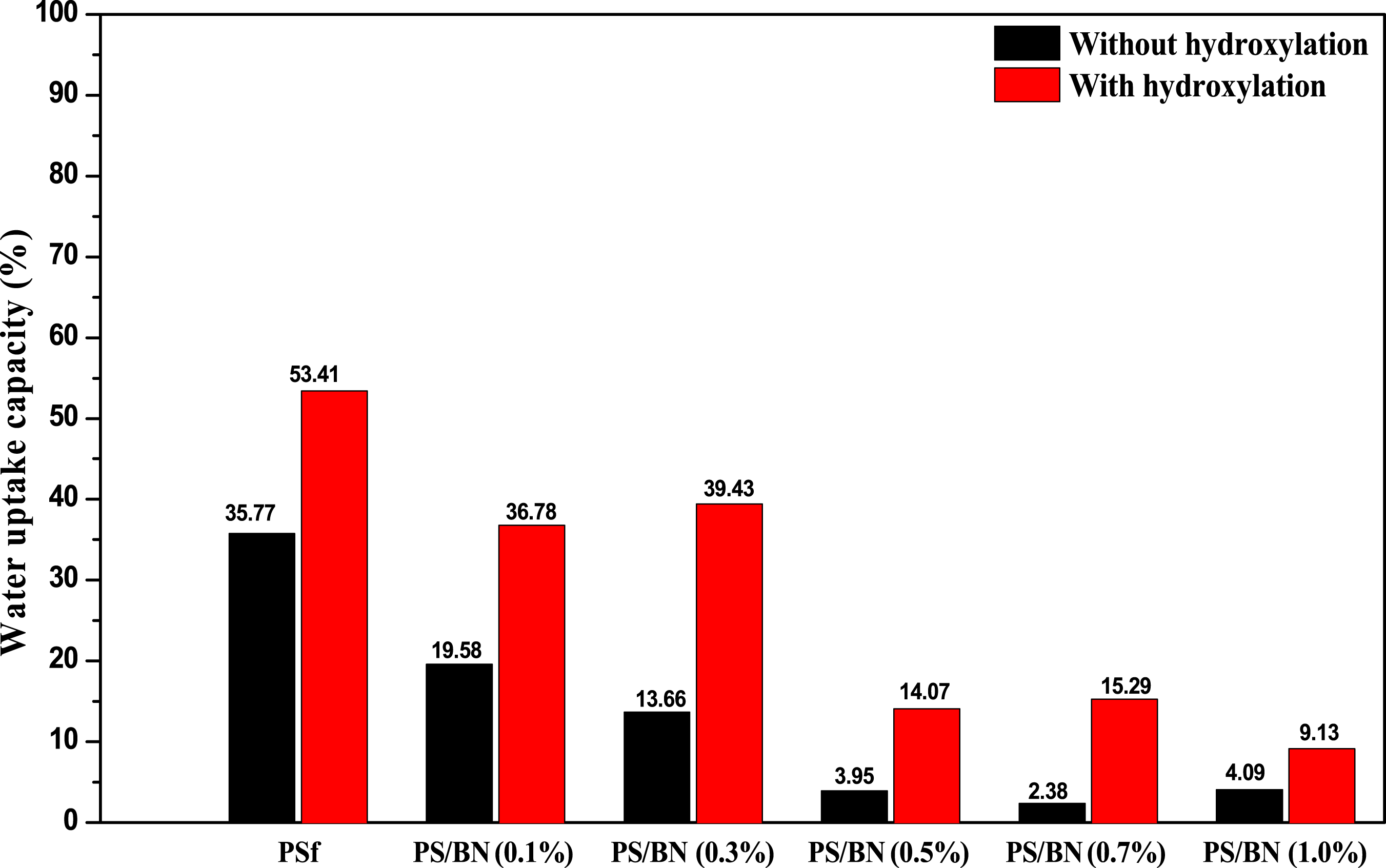
Hydrophilicity plays an important role, which could enhance the antifouling properties of the membrane. The hydrophilicity of the prepared membrane was measured by contact angle (CA) measurements. As seen in Figure 10, the CA of the pristine PSf membrane is 75.9o which is quite hydrophobic. The incorporation of BN in PSf leads to a slight decline in contact angle owing to an increase in surface porosity of the membrane. With the increase of BN loading from 0.1 to 2.0 wt. %, there was a decrease in contact angle up to 68.3o. It can be obsereved that the contact angle declines with hydroxyl functionalization. This results of water flux and Albumin rejection are shown in Figure 11. Contact angle (CA) of the prepared membrane. Pure water permeability (PWP) and Albumin Rejection of membranes.
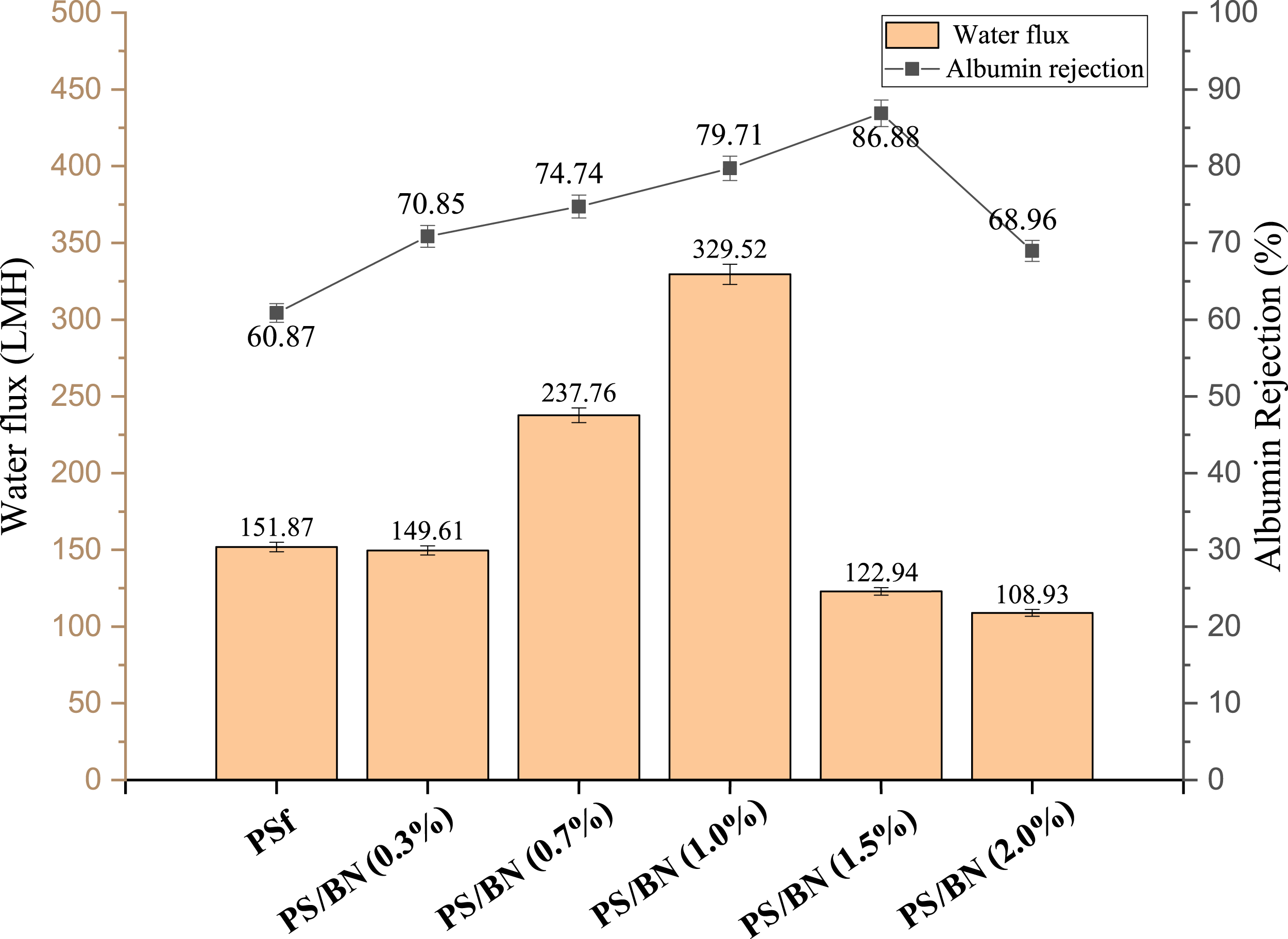
X-ray photoelectron spectroscopy
The chemical composition of PS/BN was evaluated by X-ray photoelectron spectroscopy (XPS). The XPS results survey of PS/BN nanocomposite is shown in Figure 12. In the survey scan, C, O, S, N, and B peaks are observed. The XPS survey peaks were observed at 502.5 eV, 398.6 eV, 278.2 eV, 188.8 eV, and 176.4 eV for O1s, N1s, C1s, B1s, and S2p, respectively. The chemical bonding in PS/BN has been inferred from the deconvoluted C1s, S2p, N1s, B1s peaks, and their corresponding spectra are displayed. The high-resolution deconvoluted spectra of C1s revealed that binding energy at 284.4 eV, 285.4 eV, 286.6 eV, and 288.1 eV are assigned to C-C, C-S, C-O, and O-C=O, respectively. In addition to that, the spectra of S2p reveal that 167.9 eV and 168.8 eV are assigned to C-SO3 and C-SO2, respectively. The N1 spectra reveal strong binding energy at 400.4 eV which is attributed to the B-N bond and a weak N-H bond also appeared at binding energy 398.2 eV. B–O and N–H groups that appeared over the BN surface may reveal the hydrophilicity or moisture content of BN. Thus, successful dispersion of BN inside the polysulfone matrix was confirmed by XPS analysis. (a) XPS survey spectra, the convoluted graph of (b) C1s, (c) S2p, (d) N1s and (e) B1s.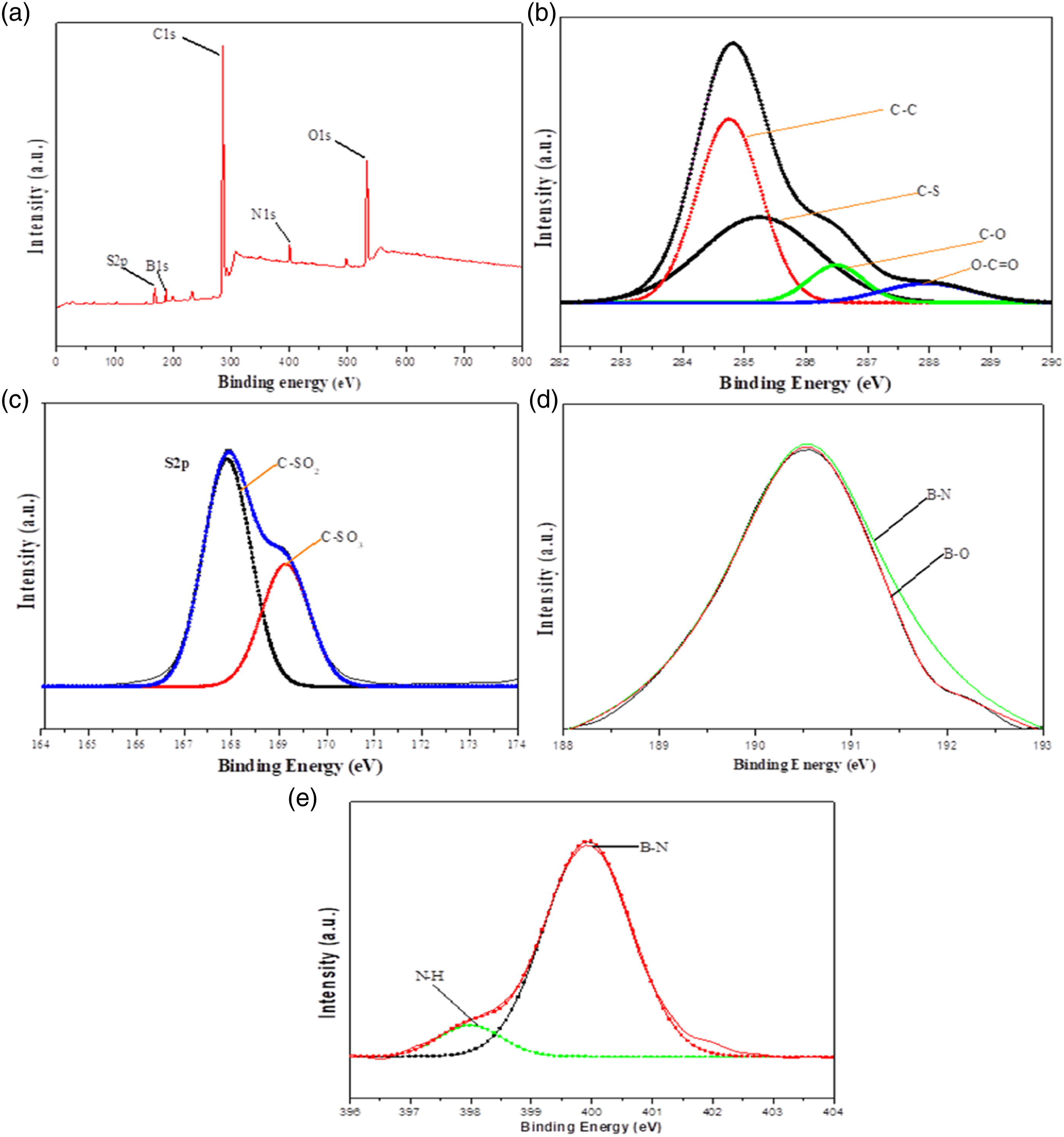
Water flux and albumin rejection
Figure 11 shows the water flux and BSA rejection for pristine PSf and modified membrane with BN loading (0.3, 0.7, 1.0, 1.5, and 2.0%). The pure water flux of the pristine PSf membrane is 151.87 LMH, whereas the highest flux of 329.52 LMH was achieved by the 1.0% BN nanocomposite membrane, increasing concentration of nanomaterial further, the decline in water flux was noted. The increased permeate flux should have resulted from the addition of Boron nitride which causes the increased in porosity of the membrane. At higher concentration of nanoparticles, the porosity declined as shown in Figure 13, which might have caused decline in water flux. Albumin rejection also increased with BN loading. While, the PSf membrane had the albumin rejection of 60.87%, the PS/BN (1%) membrane had increased albumin rejection upto 79.71%, and it increased further to 86.88% for PS/BN (1.5%). Boron nitride has negative charge (confirmed via zeta potential analysis) which could have become responsible for repelling negatively charged albumin, eventually increasing the albumin rejection. The water molecules have more favorable interactions with BN as it forms hydrogen bonds between the water molecules and boron-nitrogen atoms that have caused the faster rate of water molecules transfer through the BN pores.53,54 Thus, the water permeance data qualifies the membrane to be a better ultrafiltration membrane. Surface porosity (%) and Avg. pore diameter (nm) of pristine PSf and modified membrane.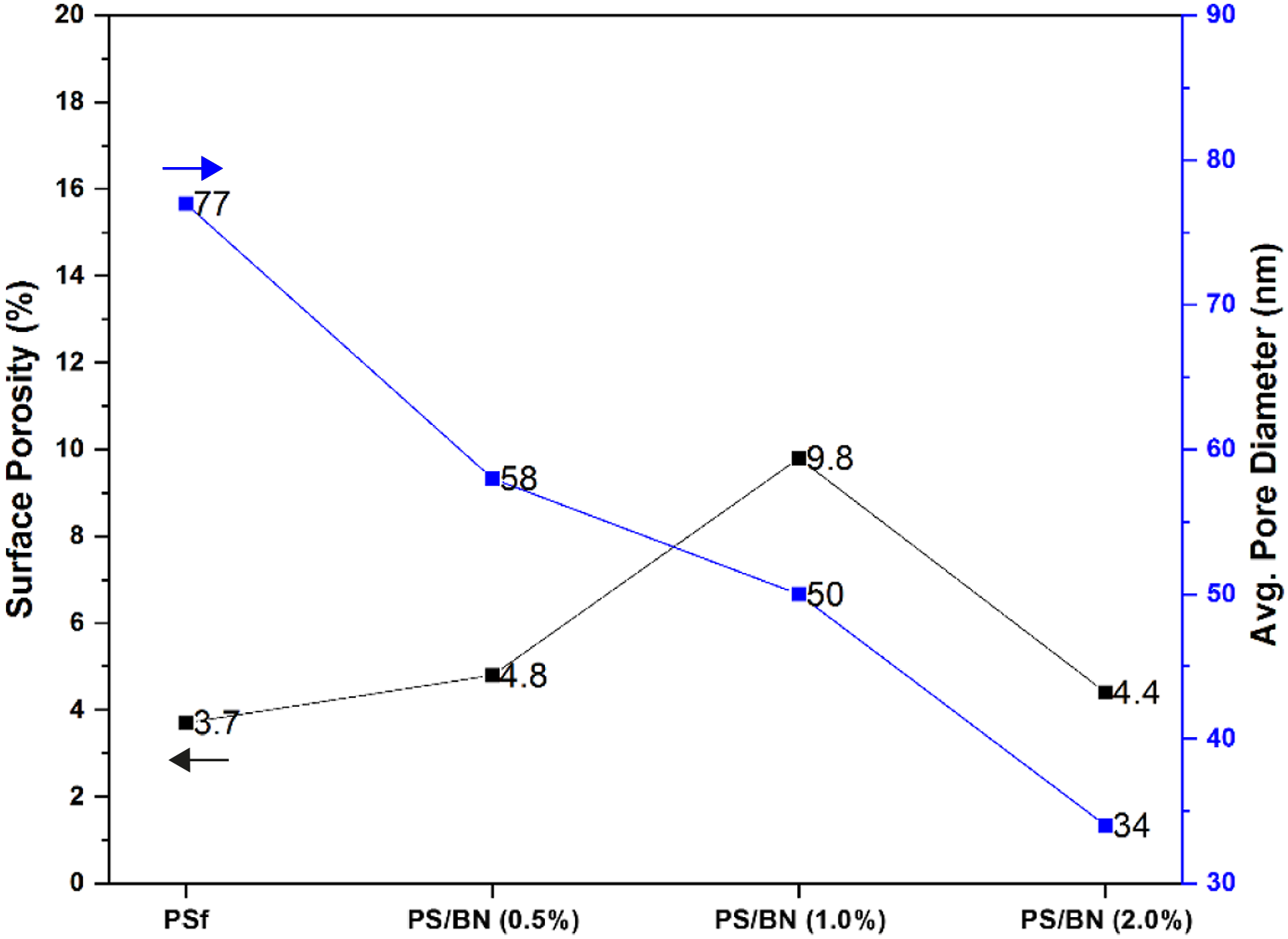
Effect of temperature on water flux
The thermoresponsive behavior of the membrane plays an important role in qualifying it for the treatment of high-temperature aqueous streams. The effect of temperature on the water flux through the thermos-responsive membrane (PS/BN (2%)) and pristine PSf membrane are shown in Figure 14. For pristine PSf membrane, there is a near linear increase in water flux with the increase in temperature. Whereas in the case of PS/BN (2%) membrane, this showed a significant spike in water flux at high temperature than pristine PSf membrane. It is owing to an increase in the faster diffusion of water molecules across the membrane surface. Diffusivity of water was increased steeply with an increase in temperature in the case of PS/BN (2%). As Boron nitride has high thermal conductivity and high specific heat, at elevated temperature, there is a faster rate of heat transfer which caused a faster rate of mass transfer across the membrane.48,53–55 The water-flux increment through the temperature-sensitive membrane (PS/BN (2%)) follows the similar pattern as PS membrane at lower temperatures. When the temperature was higher than 35oC, water flux was increased significantly with the rise of temperature. The pure water flux was increased by 205% when increasing the temperature from 22oC to 48oC in the case of PS/BN (2%) membrane, whereas the PSf membrane as shown merely 103% rise in water flux within the same temperature range. An increase in hydrophilicity
56
together with higher temperature responsiveness due to boron nitride could be the reason for enhancement in water flux. Temperature responsive behavior of the nanocomposite membrane.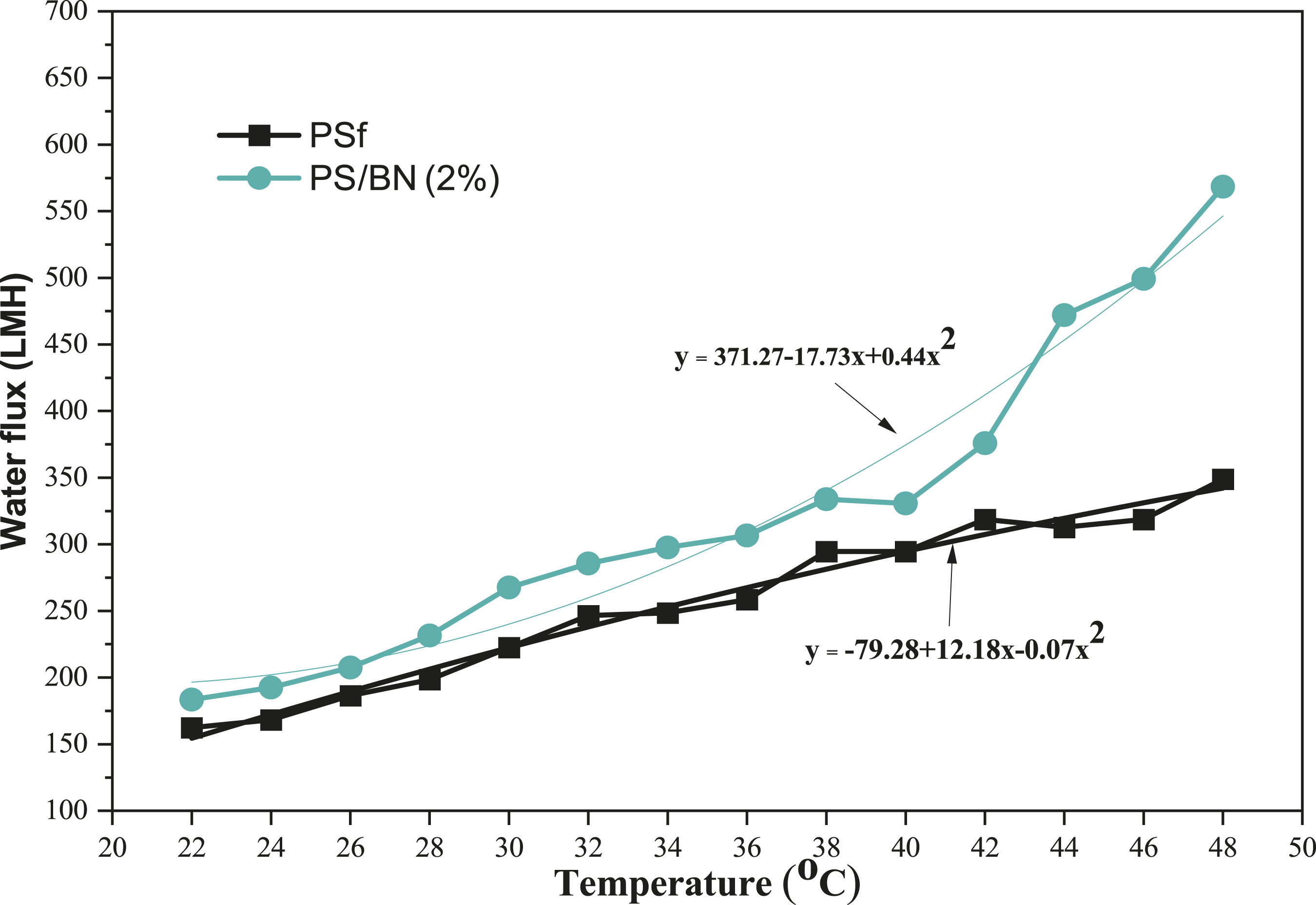
Antifouling studies
The antifouling properties of the prepared membrane were measured in terms of flux recovery ratio (%). Figure 15 showed the flux recovery ratio of PSf, PS/BN (0.5%), and PS/BN (1.0%). It can be seen that the FRR of pristine PSf membrane was 27.6%, whereas the FRR of PS/BN (0.5%) was 36.5% and FRR of PS/BN (1%) membrane was 25.9%. Flux recovery ratio (FRR%) of PSf and BN modified PSf membrane.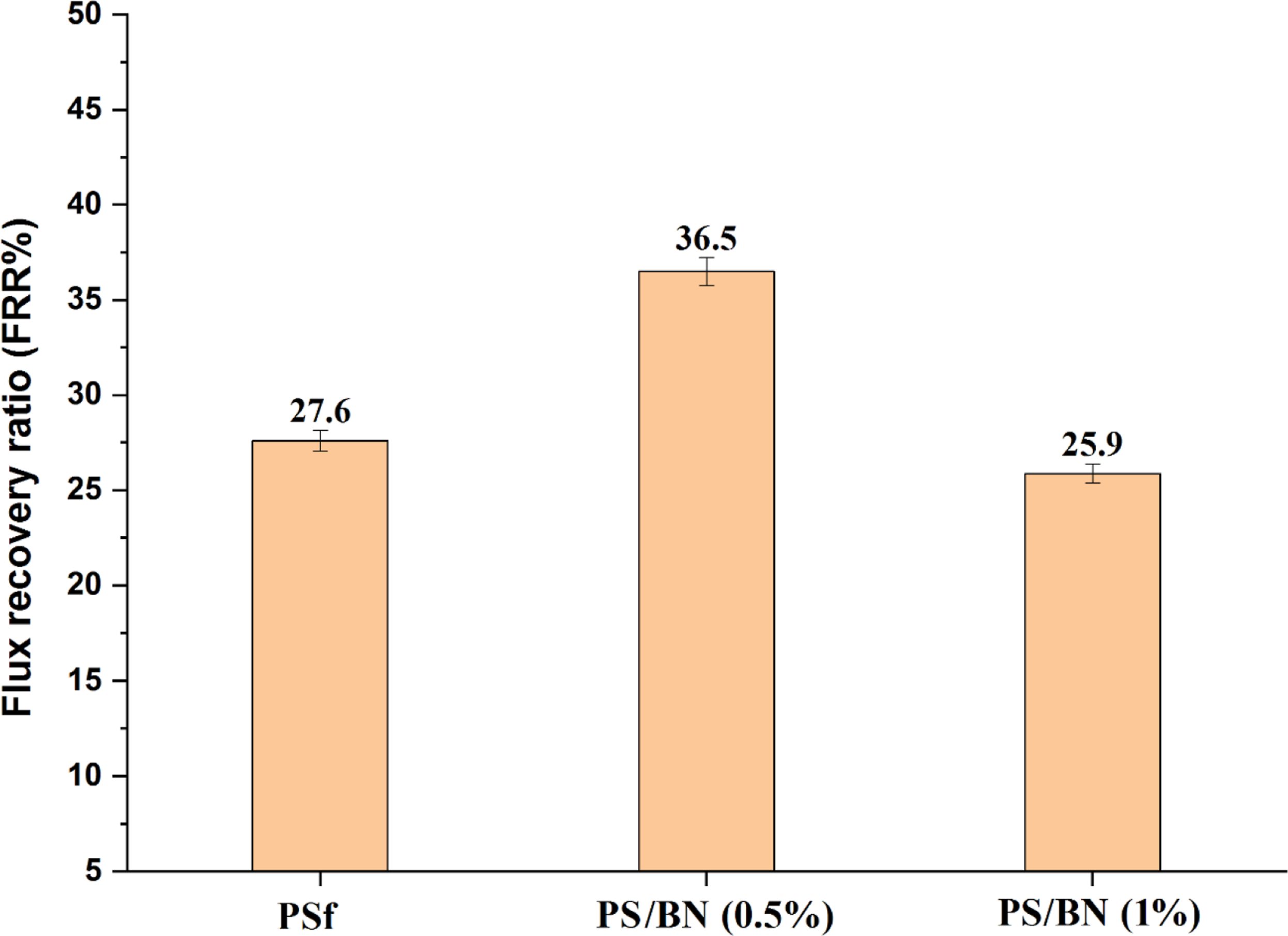
Conclusion
A novel mixed matrix membrane was prepared by blending polysulfone with two-dimensional boron nitride followed by hydroxyl functionalization of the membrane surface using 0.5 M NaOH. The membranes were characterized for surface and cross-sectional morphologies along with uniform BN impregnation by SEM and EDX, respectively. XRD and FT-IR confirmed the presence of boron nitride within the polysulfone membrane matrix. From the DSC analysis, a new peak was observed due to incorporation of Boron nitride, in which heat flow varied with change in temperature and an additional peak observed at 120.02oC. The boron nitride-modified PSf membrane had shown temperature-responsive behavior where water flux increases significantly with temperature rise, that is, 205% for nanocomposite as compared to 103% in virgin Polysulfone membrane for the same temperature range. It can be revealed that the membrane has shown a significant rise in permeate flux and albumin rejection. It opens the possibility for future work in applications involving high-temperature water/wastewater treatment or as the support membrane for thin-film composite Reverse Osmosis membrane.
Footnotes
Acknowledgments
Communication PRIS number CSIR-CSMCRI is 151/2021. The authors thank the funding support of the Department of Science and Technology project (Grant no. DST/TDT/WMT/fouled Membranes/2021/01). The authors thank the Analytical and Environmental Science Division and Centralized Instrument Facility, CSIR-CSMCRI for the help in different analyses. The authors thank Mr. Kaushik Dave for help in testing and Mr. Hardik Gajjar for image processing analysis for porosity measurements.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article was supported by Department of Science and Technology project (Grant no. DST/TDT/WMT/fouled Membranes/2021/01).