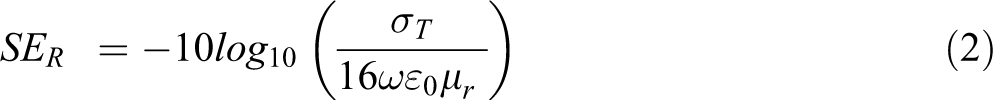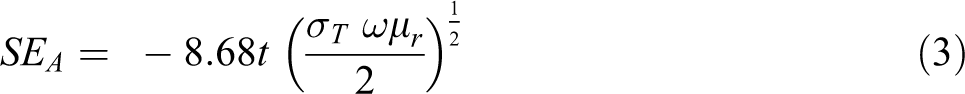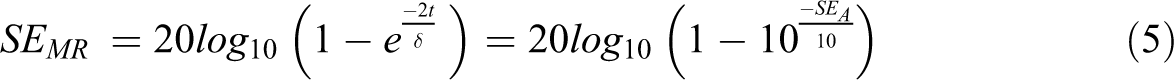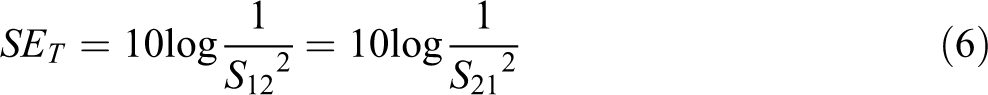Abstract
Based on recent advancements, the emission of electromagnetic radiation has become a serious issue of electromagnetic interferences. These electromagnetic interferences comprise of various undesirable emission of radiation that can create unwanted deprivation of equipment or structure performance. If these complications remain unresolved can create extreme damage to the security operation or communication system of several electronic devices. Various studies have been made to resolve these problems. In past years, electrically conductive polyaniline has attained a unique position due to the prosperity of its features. The absorption of microwave and EMI shielding characteristics of the electrically conductive polyaniline can be described in the relation of great electrical conductivity with strong relaxation and, polarization effects due to the existence of stronger bonds or localized charges. In the present article, the advancement in electromagnetic interference shielding with the use of nanosized polyaniline particles and their derivative nanocomposites is discussed by various parameters such as the size of the particle, absorption properties, magnetic properties, reflective properties, electrical conductivities, and dielectric properties. The electromagnetic absorption performance of conducting polyaniline nanocomposites associated with dielectric nanomaterials (graphene) and magnetic materials (γ-Fe2O3) are widely discussed. The most appropriate polyaniline-based nanocomposites can be identified by this context. Due to increasing demand of electrically conductive polyaniline for shielding materials, it is considered to be review for better understanding in advancement. This article presents recent electromagnetic interference shielding materials knowledge of nanosized polyaniline and their nanocomposites as well as their possibilities and challenges.
Keywords
Introduction
The advancement in electronic and telecommunication industries has been increasing in modern years. The radiation of these electromagnetic signals has become a dangerous situation due to the increased usage of electromagnetic systems. The radiation from electromagnetic interferences not only creating problems to the electronic system but they have also deep effects on the health of human, 1 such as dangerous diseases including brain tumor, 2 as well as leukemia3,4 Due to the extreme level of growth of broadcasting, electrical system, and telecommunication. it becomes impossible to prevent the emission of all kinds of electromagnetic fields in the maximum part of the earth. 5 In recent times, electromagnetic pollution results from different electrical instruments surrounded by an all-around human being. Based on recent studies, the research on cancer by the international agency has categorized the magnetic fields of very low frequency that can be carcinogenic to humans. 6 But there is no categorization made for EM radiations of radiofrequency, however, few research studies revealed it as harmful nature. 7 Electromagnetic interference (EMI) also creating serious problems for several nearby electrical types of equipment as the efficiency of these systems enables it to be retained in assemblage.8,9
It is essential to decrease electromagnetic interference by reducing errors and noise in electronic systems. 10 The electromagnetic compatibility regulations and specifications have been established by several countries to reduce the challenges created by electromagnetic interference. The blocking of conducting or radiating electromagnetic radiations into shielding material is known as electromagnetic interference (EMI) shielding. The shielding material isolates the system either a circuit, an apparatus, or a room from the external source of EM radiation. 11
Due to high density, corrosiveness, weak mechanical flexibility, tedious and costly processing restrict the use of metals and metallic composites for EMI shielding nanomaterials. However, the advanced EMI shielding nanomaterials are currently focused primarily on polymer matrix, carbon matrix, and ceramic matrix nanocomposites.12,13 The requirements for a typical electromagnetic interference shielding nanomaterial are outstanding thermal stability, high electrical conductivities, and low density. 14
In past years, electrically conductive polymers have attained a unique position due to the prosperity of applications.15–23 The electrically conductive polymers as well as their nanocomposites having different nanoparticles are utilized for the synthesis of semiconductor, lightweight batteries, sensors, and shielding materials.24–27 The absorption of microwaves and EMI shielding characteristics of the electrically conducting polymers28,29 can be described in a relation to high electrical conductivity as well as strong relaxation with polarization effects due to the existence of bound or localized charges.30,31 Polyaniline has a unique position among all conductive polymers due to its high electrical conductivity excellent stability in the environment, non-redox doping, and cost-effectiveness.32–34 These characteristics could be additionally modified with regulating polymerization parameters, use of dopants, fillers, doped aniline, and precise comonomers.35–38 Figure 1 shows the properties of polyaniline. PANI demands dispersion for binding in the matrix, due to minimal inherent particular strength, to produce nanocomposites for the beneficial product at the commercial level. While, percolation threshold moves to be elevated because of phase-separated morphology, low compatibilities, and a low ratio of the conductive polymer nanoparticles. Therefore, the mechanical properties which frequently affected by the electrical properties required a high concentration of electrically conductive polymers in the matrix for nanocomposites.
Properties of polyaniline.
It is reported from previous studies that the incorporation of electrically conductive polymers39,40 to the carbon nanomaterials can improve the electromagnetic absorption applications by overcoming these restrictions, 41 such as the electromagnetic absorption improved by the incorporation of polyaniline nanoarray on the interface of graphene. The features of polymer-nanofillers nanocomposites depend on the dispersion and attraction of the matrix of polymer. 42 The incorporation of ferrites/reduced graphene oxide/single-walled carbon nanohorn (Fe3O4 layered RGO with SWCNH) into the matrix of polydimethylsiloxane increased electromagnetic radiation absorption. The dipole as well as interfacial polarization with the decline in surface reflectiveness because of skin depth subsidized to improved electromagnetic absorption. 43 While, some intrinsically conductive polymer along with graphene has an extensive range of permeability (µ), permittivity (ε), and electrical conductivity for microwave absorption. 44 Hence the performance of shielding material can be enhanced by the arrangement of polymer along with carbon nanomaterials. 45
Generally, magnetic materials as ferrites 46 are also abundantly used for electromagnetic absorption 47 due to their great permeability, stability, and magnetic loss. 48 The blend of dielectric and magnetic loss provides outstanding shielding material by magnetic materials. 49 The properties of conductive polyaniline can also be improved with the incorporation of inorganic filler.50–52 Nanostructured ferromagnetic fillers are attractive because of the absorbing properties result from the enormous surface area with small size. As conducting polyaniline combine with magnetic particles to produce ferromagnetic composites that have an excellent structure to examine the probability of showing unique functionality. The nanocomposites containing nanostructured ferromagnetic nanoparticles have distinct magnetic properties and can be useful for the absorption of microwaves. The electromagnetic absorption capacity of many conductive polymers nanocomposites associated with dielectric nanomaterials (graphene) and magnetic nanomaterials (γ-Fe2O3) have been extensively studied. In a large range of hetero nanosize, electrically conductive polyaniline complemented by certain dielectric and magnetic nanofillers has modified electromagnetic absorption characteristics by reducing electromagnetic waves.53,54 Due to increasing demand of electrically conductive polyaniline for shielding materials, it is considered to be review for better understanding in advancement. This article presents recent electromagnetic interference shielding materials knowledge of nanosized polyaniline and their nanocomposites as well as their possibilities and challenges.
Electromagnetic interference (EMI) theory
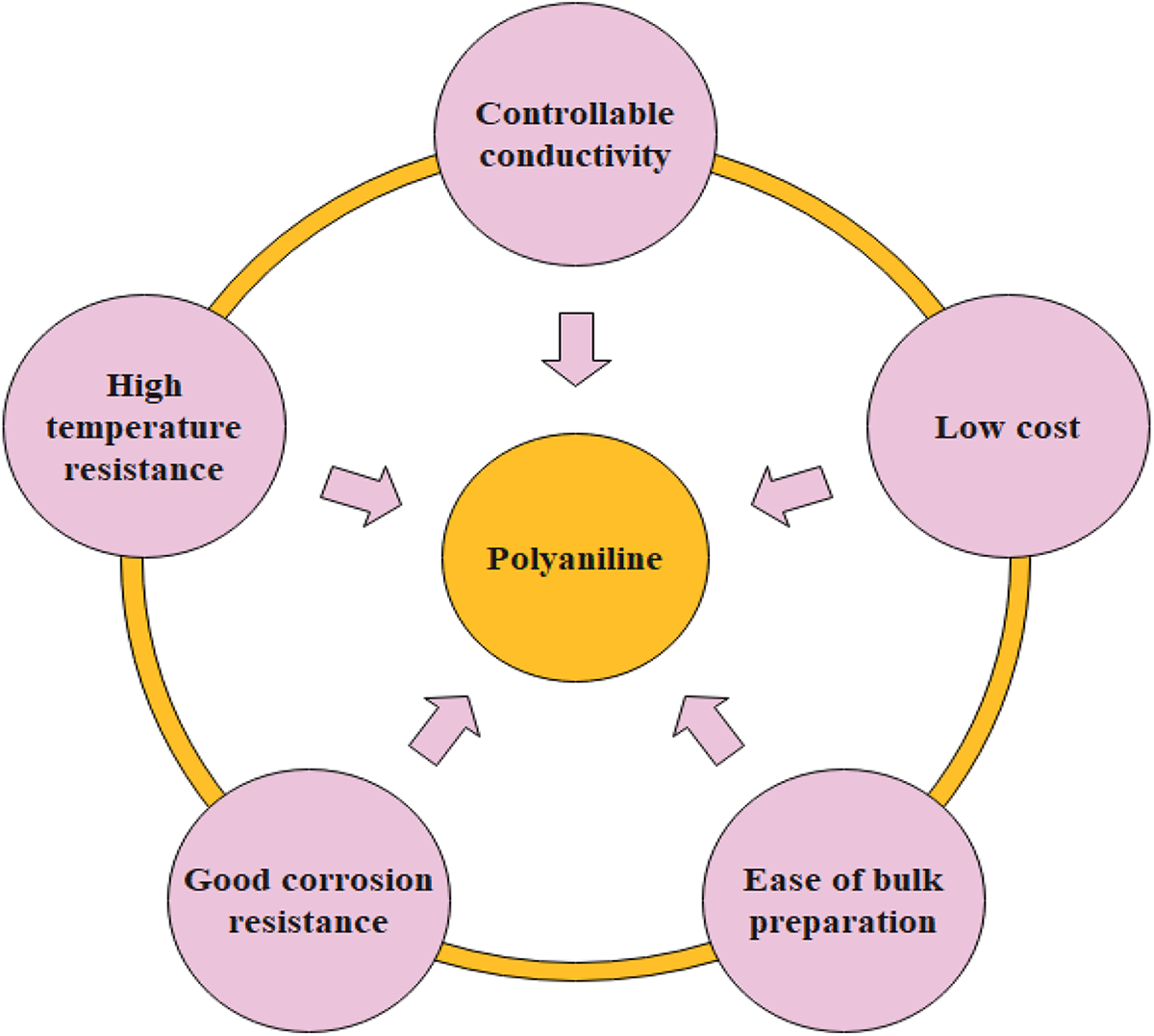
According to electromagnetic interference (EMI) theory, permittivity, permeability, and electrical conductivity are the leading factors for microwave attenuation. 55 By the use of polymer nanocomposites, connecting networks or electrical conductivity of conducting nanofillers as well as the dipoles of electrical and magnetic fields creates the improved EMI shielding effectiveness. Generally, the shield comprises conductive nanomaterials that control the transmittance of electromagnetic radiation within it. A variety of shielding nanomaterials are scientifically considered for attenuation of the electromagnetic signals to a substantial level. The effectiveness of shielding nanomaterials for attenuation is calculated by the vector network analyzer. Meanwhile, operational frequencies of radiations are extensively dispersed in frequency ranges of GHz, the parameter for the investigation of electromagnetic attenuation should be in wideband frequency ranges. When incoming electromagnetic (EM) waves interact with the shielding material, it attenuates several procedures such as absorption, reflection, as well as multiple reflections and depends on the shielding features. Figure 2 Represents the EMI shielding mechanism. The total shielding efficiency is obtained by sum up the shielding of absorptions (SEA), reflections (SER), and multiple reflections (SEMR) and is stated as 56 :
Mechanism of EMI shielding.
Shielding by reflection (SER)
The reflection in the microwave region, the waves reflected due to the mismatch impedance among the coming electromagnetic signals with the shielding nanomaterial. The reflection occurs on the surface of the shielding when EM waves strike. The high electrical conductive materials containing free charge carriers which make them efficient wave reflectors. The extent of reflected rays could be calculated by the use of the subsequent equation. 57
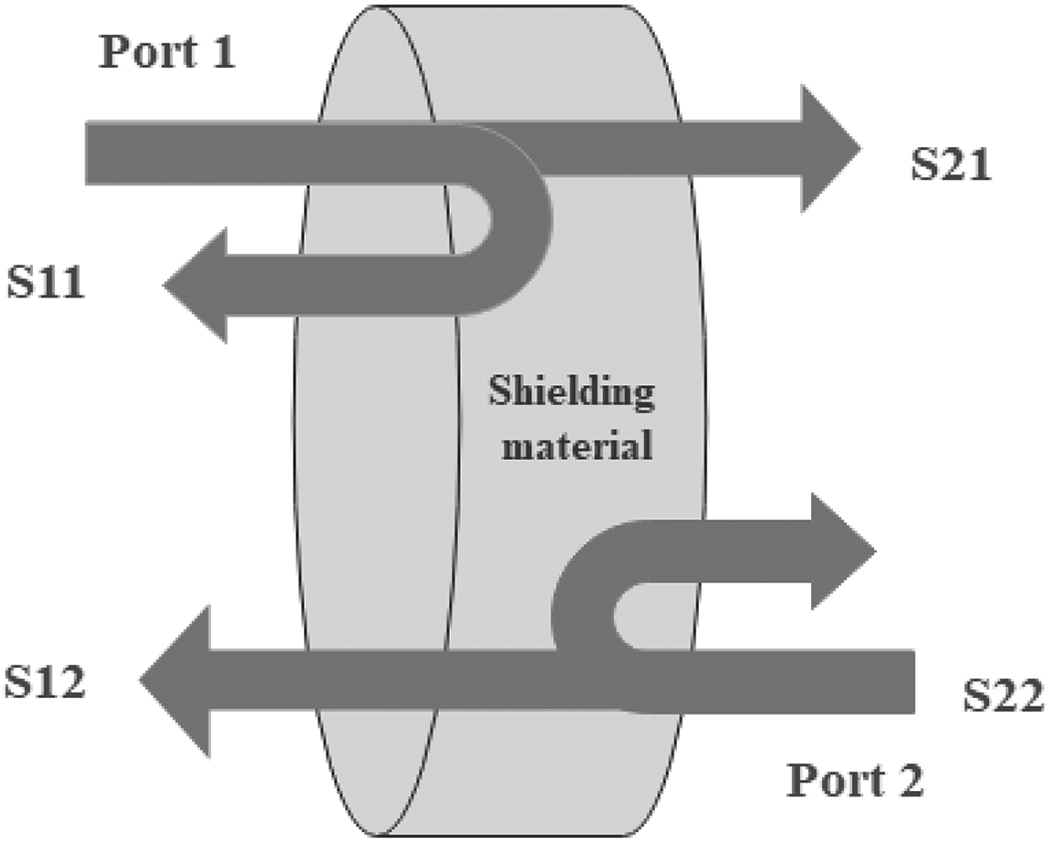
where, σT are total conductivity of shielding nanomaterials and µr relates to the relative permeability. It is reported from the equation that the reflection in the microwave region is primarily regulated by the concentration of electrical conduction in the shielding as well as shielding permeability.
Shielding by absorption (SEA)
The rays in the microwave region comprise electric as well as a magnetic field. The Ohmic losses occur because of the interaction of these fields and the dipole of the shielding material. The dipoles of magnetic and electric fields are necessary for shielding materials for the absorption of EM waves. The extent of EM wave propagation declines exponentially within the thickness of the shielding materials. The absorption is primarily because of magnetic hysteresis losses and current generated in the shield i.e. Ohmic losses. The extent of shielding through absorption can be calculated from the subsequent relation 58 :
where t denotes the thickness of the shielding nanomaterial. The extent of shielding effectiveness due to absorption is based on the results of the electrical conductivity value and permeability of the shielding nanomaterials. The thickness of the shield may improve the efficiency of shielding by the electromagnetic radiation absorption. These absorptions mounts by increasing frequency as compared to the reflection approach. While with increasing frequency the reflection reduces; the reliance on logarithmic functions. Thus, the maximum attenuation of electromagnetic waves is enhanced by increasing frequency.
Shielding effectiveness by multiple reflections (SEMR)
Generally, the extreme frequency radiation is only incorporated adjacent on an electrically conductive shielding surface with the extent of decreasing the field exponentially by increasing thickness. The shielding nanomaterials having low thickness needed to minimize the level by 1/e at the incident field is characterized as skin effect (δ) and may be calculated by use of the subsequent equation 59 :
The electromagnetic (EM) radiation reflects at multiple borders within the shielding nanomaterials when the shielding thickness is low compared to the skin effect. The extent of shielding by multiple reflections can be measured by the use of the following equation 60 :
The shielding efficiency by multiple reflections is thoroughly associated with the capability of absorption by shielding nanomaterial. The nanocomposites material having multiple boundaries and dispersed filler in the structure are most important for SEMR. It is clear from the equation that the thickness and high absorption ability (≥SEA) of the shield can easily neglect the value of SEMR. The main reason is that on maximum frequency, although moving from one surface edge to another edge the extent of electromagnetic radiation converts into insignificant because of the absorptive phenomenon.
Vector network analyzer (VNA)
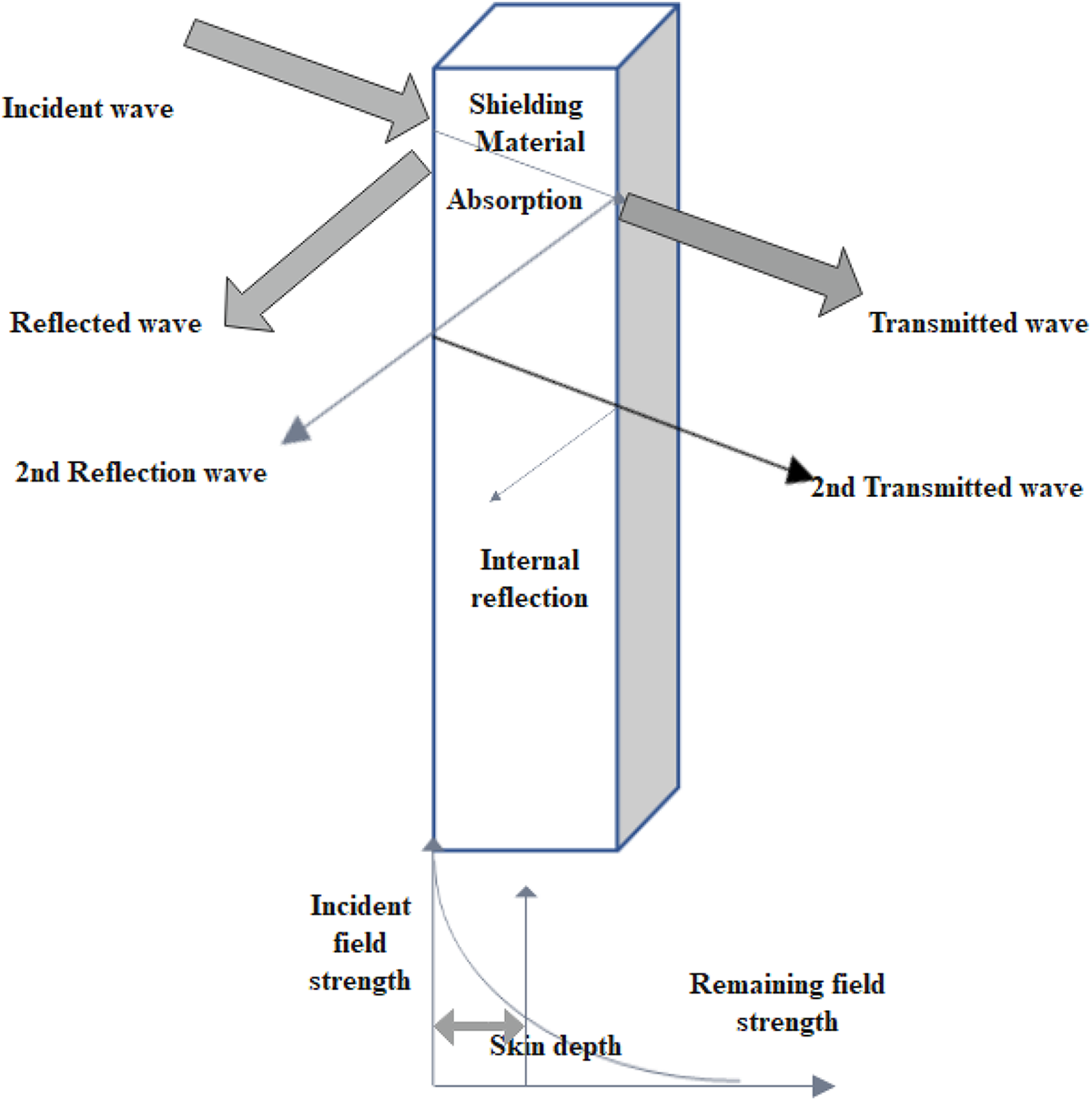
A vector network analyzer shows in Figure 3 is used to calculate the scattering parameters. It is extensively used for understanding the mechanism of attenuation in the microwave frequency range by using different factors like permeability and permittivity. The efficiency through absorption, reflection, and total effectiveness can be calculated by the use of scattering factors which are measured from vector network analyzer and are stated as 61 :
In which, S11 represents the onward reflection coefficient, while S12 represents the inverse transmission coefficient then S21 represents the onward transmission coefficient. The reflection, as well as transmission power through the sample, are represented by the scattering parameter.
Vector network analyzer.
Properties of polymer-based nanocomposites
The polymer-based nanocomposites have a variety of features such as dielectric properties and magnetic properties of nanocomposites which are as following:
Dielectric properties
Several parameters such as nanoparticle dispersion, features of the matrix, intrinsic conductivity, the affinity of the matrix within nanoparticles, and the characteristic ratio of nanoparticles governed the electrical conductivity properties of the polymeric nanocomposite. At percolation, exhibits an extreme level of increment in the conductivity is exhibited by the DC conductivity of the nanomaterials. 62 Based on the classical percolation theory, 63 the electrical conductivity of polymer nanomaterials obeys a power-law relation which is known as scaling law due to the presence of conducting nanoparticles. This relationship is expressed as:
where p and pc represent the concentration ratio of nanofiller and percolation threshold respectively. While t represents the perilous exponent that varies with structural dimension. If t for two-dimensional systems is 1.3 but in a three-dimensional network it will vary between 1.6 and 2. It is studied that the achievement of improved electrical conductivity is because of the production of a conducting nanofiller arrangement within the polymeric matrix. By using a highly significant ratio of conducting particles, the percolation threshold may be decreased.
64
From previous studies, it is cleared that the EM attenuation is optimized by conducting a network of the nanoparticle. A variety of different strategies such as covalent as well as non-covalent grafting with functionalization of polymer chains were proved to improve nanoparticle dispersion in the past. Dielectric properties such as real, imaginary, and complex permittivity
Magnetic properties
As the polymers have no magnetic properties in behavior but magnetic features conceivable with a combination of conductive polymer with magnetic particles. The radiation in the microwave region has both magnetic and electrical fields and their magnetic and electrical dipoles in the shield govern the absorption of waves.
The absorption of EM waves in magnetic fields can be emitted due to the interaction of magnetic dipoles that primarily depend upon permeability (
Polyaniline
Polyaniline, a conducting polymer, exhibits various forms, and each form of polyaniline finds a variety of technological applications. Figure 4 shows different arrangements of polyaniline. PANI is considered to be the essential conductive polymer due to its easy preparation, biocompatibility, proton conductivity, 66 and high conductivity value. 67 The polyaniline in leucoemeraldine form, which is a reduced form has vast properties in Lithium-PANI battery with different electrochromic systems. The polyaniline in emeraldine base form is insulating which is half of reduced and half of the oxidized form. The emeraldine base form discovers several features like corrosion resistant layer for mild steel, iron, and HCl gas in sensors. The conducting polyaniline derives from the doping of emeraldine form, which finds applications in industries such as electrode material 68,69 like batteries, electrochromic glasses, sensors,33,70,71 solar cells, anticorrosion materials, supercapacitors, and EMI shielding. The pernigraniline (fully oxidized arrangement), which discovers vast features in the field of inline optics. The polyaniline conductivity varies with the level of protonic acid and the structure of the nanomaterial. In the molecular structure of polyaniline, the polymer chain flanked due to the presence of reactive NH group which creates high chemical flexibility of the system in contrast to other phenylene-based polymers.
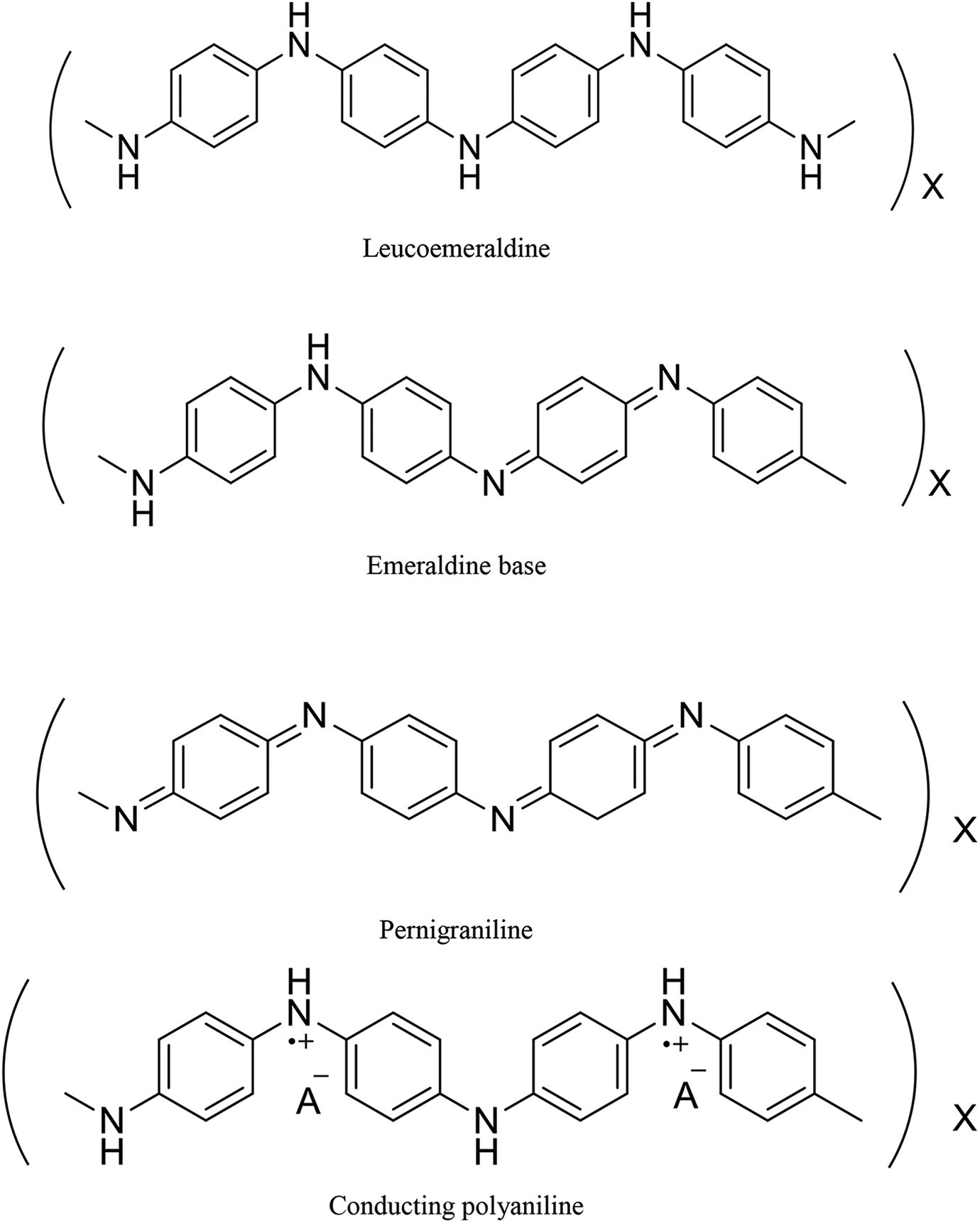
Different arrangements of polyaniline.
In the presence of an acidic medium, the polymerized aniline convert into an emeraldine base structure or protonated formation of polyaniline. 72 This process is somehow recognized as a unique type for polymerization of cations. 73 The propagation of the polyaniline chain ends as the highest conductive arrangement of polyaniline, the emeraldine oxidation state, is formed during polymerization. This arrangement, with the treatment of an alkali solution or, maybe with an excessive amount of water, can be changed to the emeraldine base. 74 The transitional form of polyaniline forms the imine sites that can be protonated simply, during a prominent transition of insulator and conductor, and brought because of the positive charge appearance within the matrix, although the amount of p-electrons remnants same. Figure 5 shows the formation of emeraldine salt. As a result, emeraldine salt has unique properties such as optical, paramagnetic, and conductive properties. 75
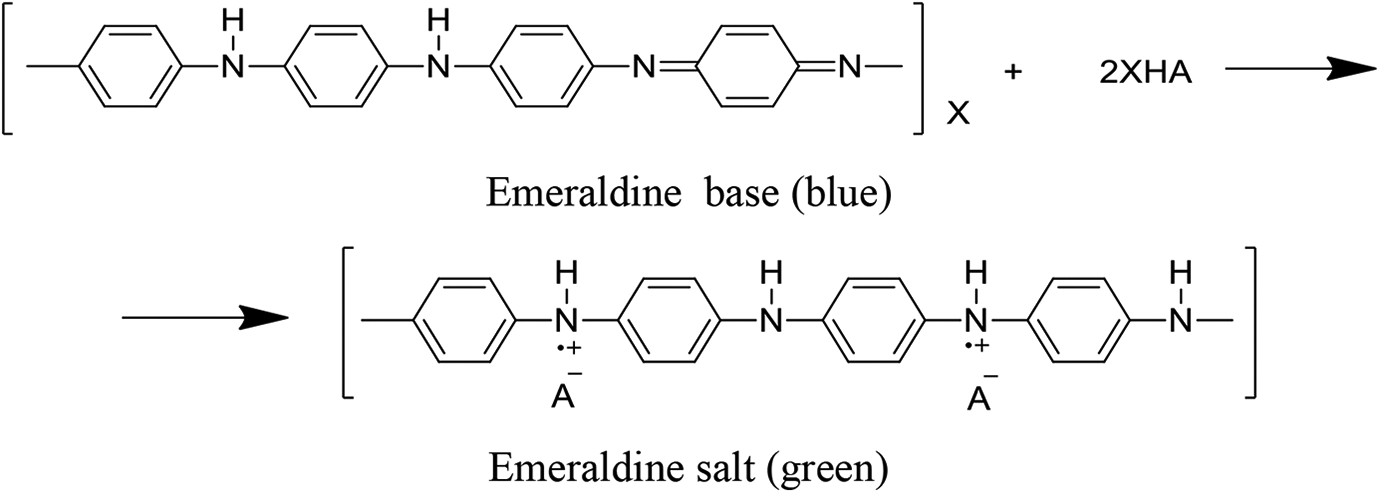
Formation of emeraldine salt.
The properties of conductive polyaniline are not just because of the extent of oxidation and protonation as well as the conformational and structural parameters, that have been pretentious during polymerization conditions of aniline. 76 So, this is one of the main tasks in polyaniline synthetic chemistry, that the progress of technical approaches toward the formation of conducting nanocomposites having doped polyaniline by the greatest mixture of the factor. These characteristics should also in the contest by other necessities for the most appropriate nanocomposite material.
The conductivity procedure of PANI is diverse as compared to a further electrically conductive polymer such as polythiophene, polypyrrole, and polyfuran, due to the significant contribution of the heteroatom in the formation of p-band. In the hasty production of electrical systems, EMI is a type of additional unnecessary product. The plastics are replacing the metallic cupboards of the electrical casings due to their visual applications. Though, plastics could not be entered for providing are electrostatic charge due to their transparency for EM signals. Recently, electrostatic charge dissipation and electromagnetic interference are controlled by the most effective means, which is the use of various kinds of conducting nanocomposites having conducting fillers. 77 The optimization of conductive nanocomposites of polyaniline can be attained by assembly with conventional polymers. 78
Synthesis of polyaniline
The polyaniline (PANI) can be synthesized by aniline monomer either by electropolymerization 79 or with oxidative polymerization. 80 The ECS is supported in the aqueous medium of appropriate solvent by using one section or two sections of the electrochemical cell. 81 The synthesis technique of polyaniline (PANI) by both methods (chemical 82 and electrochemical methods) is evaluated by the conductivity, XRD, solubility, and SEM.
Electrochemical synthesis (ECS-PANI)
A single compartment cell is used for the development of electrical polymerization of aniline. In the cell 3.1 milliliters of aniline (0.15 M), 0.2 liters of distilled water, and 20 ml of 1 M conc. HCl was used. The amount of product is directly proportional to the amount of monomer used per batch. But, the use of extraordinary monomer quantity leads to a reduction in productivity because an extreme quantity of products, on the electrode surface, cannot be accommodated which results in material depletion. Therefore, 3.1 milliliters of an aniline monomer is preferred. The 0.7 V potential difference was used on the electrode boundaries against SCE. The time is taken by the reaction 2 h. At the graphite electrode, the PANI produced which was first collected, washed by deionized water, afterward filter as well as dry at room temperature in the muffle furnace. The obtained polyaniline was then mixed with a large quantity of NH3 until the removal of HCl and afterward again doped with PTSA (50 mol). At last, the obtained paste was the first filter, then use distilled water of washing, and at last drying at room temperature at the muffle furnace. 83
Chemical synthesis (CS-PANI)
In CS-PANI, the same configuration can be taken as in the ECS-PANI but an equal amount of oxidizing agent with respect to aniline is used such as ammonium persulfate (APS) rather than continuous voltage. 190 ml of deionized water, 0.15 M monomer of 3.1 milliliters aniline, and twenty milliliters of conc. HCl (1 M) was placed in the flask and stirring it. 7.6 grams of ammonium persulphate (APS) was taken in 10 ml of deionized water. Afterward, it is mixed with the other aniline solution gradually, stir for 2 h at room temperature through a magnetic stirrer. The particles with dark green color were gotten and filtered with filter paper. After it, use deionized water for washing and then drying at room temperature in a muffle furnace. The obtained polyaniline was then mixed with a large quantity of NH3 until the removal of HCl and afterward again doped with PTSA (50 mol). At last, the obtained paste was the first filter, then use distilled water of washing, and at last drying at room temperature at the muffle furnace. 83
Comparison between CS-PANI and ECS-PANI
Owing to the protonation level and the oxidation state, polyaniline (PANI) can either be insulating or conducting in nature. The leucoemeraldine form and pernigraniline form show insulating behavior, while the doped and protonated emeraldine base system shows significant conduction, but the rest of the other forms are in between. 84 The mechanism of conduction is described by various means such as the polaron/bipolaron formation30,31 and hole theory conduction or the hopping mechanism.85,86 The crystallinity is increased by increasing the conductivity of the mobile hole or electron interchain and the mobility of the equivalent interchain. The reduction in the spacing of the d-band increases the possibility of hopping of interchain which in turn increases the conductivity. Generally, it is reported that the doping value increasing (up to 50%) for polyaniline produce modification in the value of conduction by the polaron formation, however extensive increase in doping value (>50%) the electrical conductivity is again initiate decreasing because the formation of bipolaron. 85 Electrical conductivity of the composite does not significantly increase unless and until composite achieve a certain definite size. This is due to delocalization of electron cloud in long-range, as the conjugated double bond cannot form unless the composite attains definite size. However, the increase in weight of the molecule, also has some types of limitation such as alteration in symmetry of chain can be produced, that have adverse effects on the delocalization structure. 87 So, the conduction in polyaniline is primarily changed with the molecular structure and may be oxidation state, degree of crystallinity, degree of doping, the spacing in d-band, and chain length of molecular weight.
The DC conductivity of polyaniline prepared by the electrochemical synthesis (1.7 × 10−4 S cm−1) is somewhat low compared to the polyaniline synthesized by the chemical synthesis method (6.3 × 10−4 S cm−1). The electrical conduction in chemically synthesized polyaniline is nearly 3.5 times greater as compared to the polyaniline that is electrochemically synthesized. The level of crystallinity for the electrochemically synthesized polyaniline was approximately half of the CS polyaniline. That is because of the alteration in preparation techniques.
In the electrochemical synthesis, the release of the electron is due to the attraction of aniline, as a monomer, to the anode (graphite) to form a cation radical. The polymer is synthesized by cation radical successively. 88 The probability of orientation can be low because of the anode which is attracted and therefore the level of crystallinity as well as the chance of orientation could be less due to the attraction of aniline monomer to the anode (graphite) in the electrochemical polyaniline synthesis. During the chemical synthesis, the cation radical of aniline is produced by ammonium persulphate even the polymerization mechanism is similar for both reactions. 89 So, the cation radicals can easily orient and are free throughout the improvement in the polymerization. It can be the main cause of the better level of crystallinity in the chemical polyaniline synthesis. The degree of crystallinity and an amorphous zone have an inverse relationship with each other. Therefore, the solubility is increased because the solvent can enter between chains easily. In the case of ECS-PANI, it could be one of the main factors for increasing solubility. The maximum degree of crystallinity in the chemical synthesis of polyaniline evaluating the formation of greatly ordered chains, that created more possibilities for the intrachain hopping as well as interchain hopping, which means that higher conduction in the chemical polyaniline synthesis is due to the carrier charge mobility. The chemical synthesis has higher chemical oxidation effectiveness by APS than that of electrochemical synthesis in the oxidation process. The constant removal of deposited aniline with polyaniline deposition at the surface of the electrode can cause a decrease in the electrode efficiency for more deposition and removal. The synthesis method has an outstanding effect on the structural pattern and morphologies of PANI. 90 It is reported that both CS-PANI and ECS-PANI have spherical particles in structure. The particle magnitude of the electrochemical synthesis of polyaniline is 20 to 40 mm, while the particle diameter of the chemical synthesis of polyaniline is 0.1 to 1.0 mm. The polyaniline particle magnitude by the electrochemical synthesis is almost 20 to 400 times greater as compared to the particle size of polyaniline by the chemical synthesis. The production of bigger size may be as a result of the higher capacity of smaller particles to coalescence into bigger one units throughout electrochemical synthesis rather than the chemical synthesis. The capacity may be due to polyaniline coating deposition after dispersion on the similar boundaries of the electrode until the multiple dispersion have a screening effect (the discharging effectiveness of cathode reduces by each coating) significantly decrease the effective discharge of cation radicals on the surface of the screened electrode. 91
Preparation techniques for polyaniline blends and nanocomposites
A synthesis method of PANI-based nanocomposites are represented in Figure 6 which were prepared by dispersion of fillers in the polymer matrix through various methods which are as following:
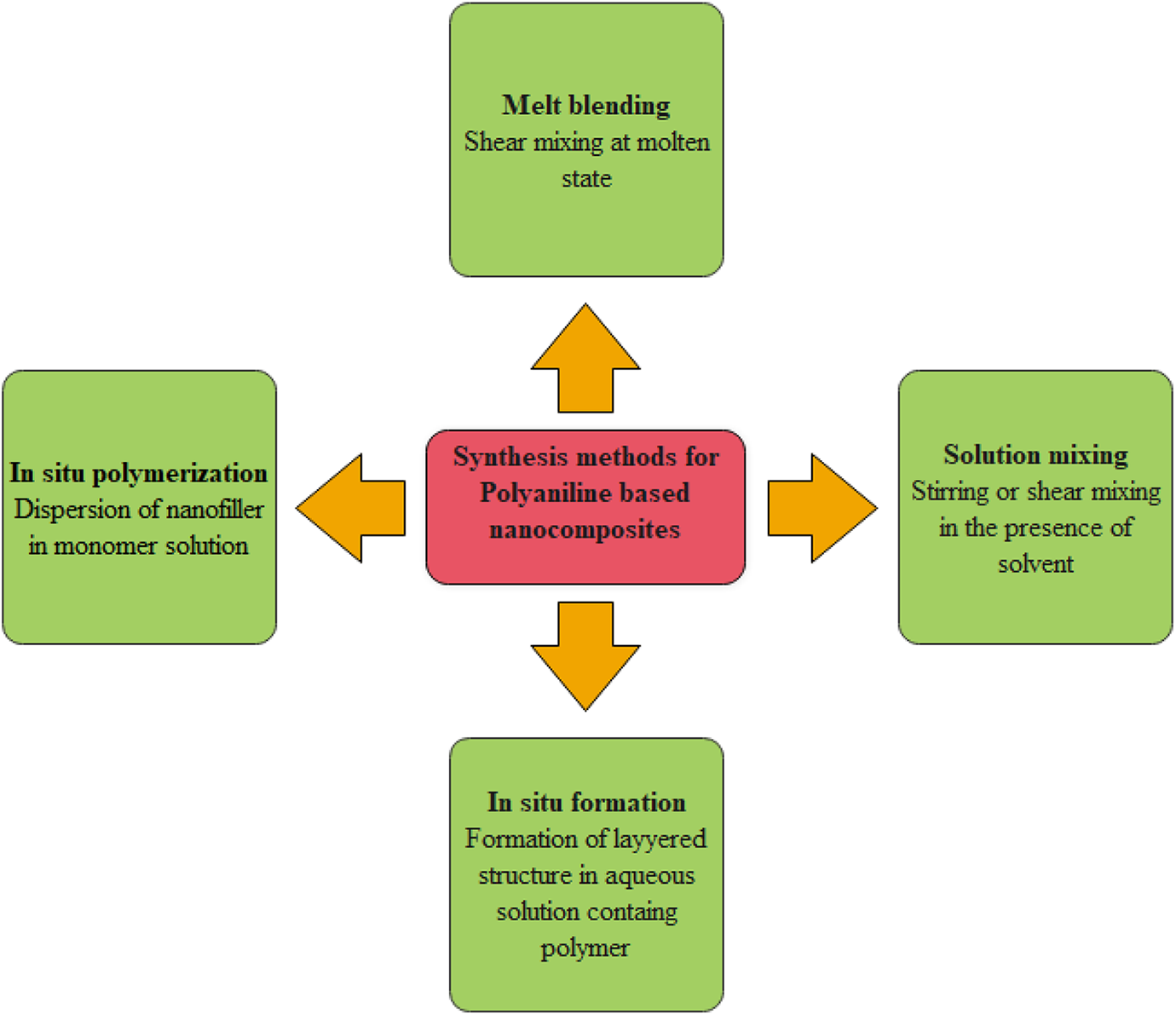
Synthesis method for PANI-based nanocomposites.
In situ intercalative polymerization
One of the most efficient methods for the production of nanocomposites is in situ polymerization. The nanofiller is added to the monomer/oligomer during the process of polymerization. The result shows a stronger attraction among the conducting nanofiller and polymeric matrix. The nanocomposites synthesized by the in situ polymerization method contain superior mechanical features rather than other techniques such as the melt blending technique and the solution mixing method. One of the primary disadvantages of in situ polymerization is the use of a large amount of electrical energy for dispersing the nanofiller in the polymeric matrix, which is not suitable for mass production. 92
Sol-Gel method
Sol-Gel method for synthesis of nanocomposite is a technique in which the aqueous solution having inorganic materials and the polymer form a layered structure. In the development of layered structures, the water-soluble polymer behaves as a template. 93 The technique includes the incorporation of organic and monomers molecules at the sol-gel matrix. The organic or inorganic networks form between the sol-gel matrix and the polymer. During the development, the polymers create nucleation and when they grow, the inorganic host filler is surrounded within layers when they grow. 94
Solution mixing
The polymers are soluble in the solvent, while nanofillers are dispersed in the subsequent solution in the solution mixing technique which is based on the solvent system. 95 This technique allows the nanofillers to be uniformly diffused in the polymeric matrix,96–103 while the layered nanofillers can be swellable. At first, the fillers are grown in a variety of solvents such as water, toluene, or chloroform. As the nanofillers and polymers are mixed in the solution, the chains of polymer displace and intercalates the solvents within the interlayer structure. When the solvent removes, the exfoliated or intercalated structure remnants within polymer composites. Solution mixing technique for different polymer nanocomposites includes the dispersal of nanofillers in the polymer solution by precise evaporation of the solvent, high-speed mixing, and nanocomposite film casting. High-speed mixing can be attained by reflux, shear mixing, magnetic stirring, or the primarily used technique of sonication. 104
Melt blending
Melt blending is an ideal technique for making polymer with clay nanocomposite having a polymer matrix composed of elastomeric and thermoplastics materials. 105 This method for polymer blending is generally acceptable due to economically effective as well as enhancing the quality of materials. 106 It is a direct, environmentally stable as well as cost-effective technique. The technique is based on mixing various mixing instruments including extruder, two-roll mill, and internal mixer instead of the use of solvent.92,107–109 Generally, the polymer is heated then mixed with the estimated amount of the intercalated clay by using a mixing device. Melt blending uses an inert gas like neon or argon to proceed with the reaction. At first, the polymer and intercalant are dry mixed, then it is melted in a mixer to shear sufficiently to make the desired polymer/clay composites. This technique has many advantages compared to solution intercalation or in situ polymerization. Melt blending is environment friendly because of the nonexistence of solvents having organic nature. The technique is suitable for recent industrial developments, such as injection molding and extrusion. The melt blending technique has become widespread because of its advantages in industrial development.
Polyaniline (PANI)-based EMI shielding materials
Among the different electrically conductive polymers, PANI has attained significant attention worldwide because of its thermal, chemical as well as excellent environmental stability. PANI is present in several oxidation states ranging from completely reduced form to partially oxidized form to completely oxidized form that is leucoemeraldine, emeraldine, and pernigraniline respectively. The widely studied form of polyaniline is the emeraldine salt derives from the emeraldine base. The semiconductive emeraldine base having the conductivity of 10−5 S cm−1 converts into a conductive form of emeraldine salt with a conductivity of 102 S cm−1 by using a simple doping technique. 110 Polyaniline-based conductive nanocomposites were an innovator of electromagnetic interference shielding materials. Now we come to venture from the wide references for conducting PANI-based nanocomposites.
Polyaniline-based carbon nanocomposites
The different kinds of nanomaterials such as carbon-based composites have been broadly used as EMI shielding nanomaterials. A wide range of absorption frequency, strong absorption features, excellent thermal stability, antioxidant ability, and low density are perfect microwave absorbing properties. 111 As the conductive polymer doesn’t attain all these properties, these nanomaterials could be considered as the good applicant of polymer to achieve good microwave absorption properties. As carbon in the form of carbon nanotubes are an excellent choice to combine with the polymer due to its high conductivity value and low density.112–116 The nanofiller like multiwalled carbon nanotubes are outstanding fillers having excellent characteristics of physical features, hence they are generally used for polymer-based nanocomposites for obtaining required properties.117–119
Saini et al. synthesized polyaniline coated multiwalled carbon nanotube through in situ oxidative polymerization. The polyaniline/multiwalled carbon nanotube nanocomposites were contained a better conductivity value of (19.7 S /cm) than pure polyaniline (2.0 S cm−1) and multiwalled carbon nanotube (19.1 S cm−1). With the increase of multiwalled carbon nanotube, the shielding efficiency by reflection improved from −8.0dB to −12.0dB. While the SE by absorption enhanced from −18.5dB to −28.0 dB by increasing coating of CNT. The shielding efficiency (SE) of −27.5dB to −39.2 dB by dominated absorption of nanocomposites was measured in 12.4–18.0 GHz frequency range of Ku-band with a minimum 2 mm thickness of pellets. 120
Wang et al. prepared polyaniline and graphene aerogel (GA) to study the microwave absorption properties. The covalent bond was formed between polyaniline nanorods and graphene aerogels covalent bonds rather than non-reactive groups for connecting. The transfer of accelerated electrons improved the irregular dispersion of electron clouds at graphene aerogels’ surface. The development of polyaniline nanorods on graphene aerogels efficiently increased the multiple reflections of electromagnetic radiations and interfacial polarization, which increased electromagnetic radiation absorption. The measured polyaniline (PANI)/graphene aerogel (GA) exhibited high electromagnetic interference shielding −42.3 dB in 11.2 GHz frequency range with 3 mm thickness of nanomaterials. 121 The incorporation of natural graphite flakes improved polyaniline for the advancement of MWCNTs-based composites was prepared by Gupta et al. The multilayer graphene was prepared by ball milling for polyaniline natural graphite flakes/multiwalled carbon nanotubes composites. Figure 7 shows the processing of PANINGF–MWCNTs nanocomposites. The measured electromagnetic interference shielding efficiency was enhanced by increasing the concentration of MWCNTs in the nanocomposite from 1% to 10% wt. The highest value obtained for electromagnetic interference shielding efficiency of −98 dB for composite contained 10 wt% multi-walled carbon nanotubes content. The extreme value of electromagnetic interference shielding was dominated by the phenomenon of absorption that was owing to the collective effect of decreasing carrier mobility and increasing space charge polarization. The reduction of carrier mobility created a constructive outcome of the shore hardness cost because of the compact bonding among the nanocomposites. 122
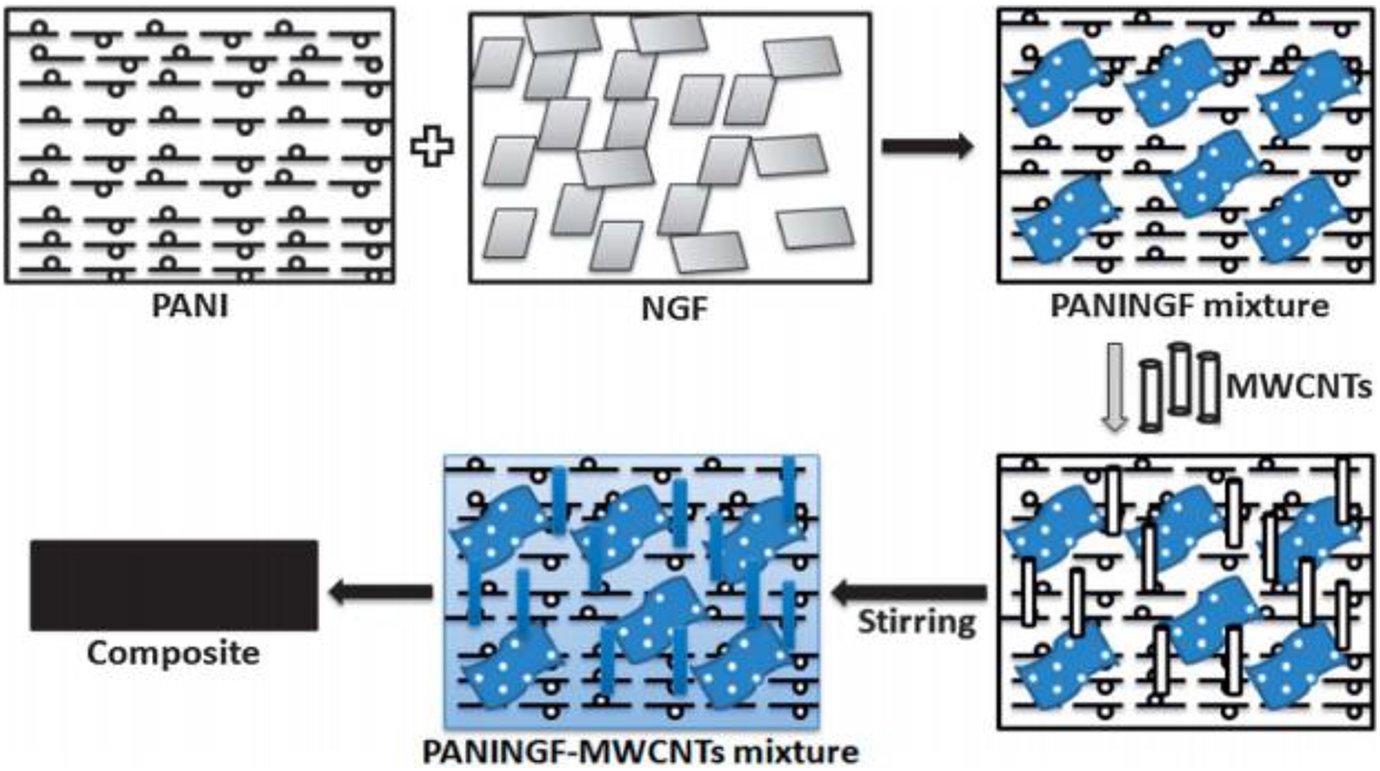
Schematic representation of PANINGF/MWCNTS composites. 122
In another work, Zou et al. analyzed the incorporation of polyaniline to carbon nanotubes coating deposited on fabrics to obtained extraordinary EMI shielding characteristics. After the CNT and PANI combination by the in situ polymerization, a compressed coating was attained of polyaniline on the surface of the carbon nanotube, thus formed a highly efficient conducting network. Figure 8 depicts the morphology of cotton fiber having the clear surface of the raw fiber with nanosize stripes. The compact coating of PANI/CNT nanocomposites by deposition covered the fabric surface. The creation of a conductive network of polyaniline/ carbon nanotube-coated fabric has outstanding shielding efficiencies of 23.0 dB for electromagnetic interference in the frequency range of 4.0–6.0 GHz. 123

SEM images of (a) Pure fabric (b, c) PANI/CNT coated fabric with various magnification. 123
Khasim prepared polyaniline (PANI) / graphene nanoplatelet (GRNP’s) nanocomposite films with a thickness of ∼1.5 mm by in situ polymerization. With the addition of graphene nanoplatelet, a hefty impact of the high electrical conductivity value for polyaniline/graphene nanoplatelet nanocomposites was created. The electrical conductivity was improved by enhancing the percentage of graphene nanoplatelet within the matrix made up of polyaniline. The incorporation of graphene nanoplatelet (GRNP’s) in the PANI matrix led to the dispersal of further aniline within the graphene nanosheets through polymerization technique. The effect of polarization was improved with modified dispersion of further aniline monomers at the surface of nanoparticles which precedes to improve the charge density as well as electrical conductivity values. The electromagnetic interference shielding efficiency for pure polyaniline was observed as −0.5 dB, while for polyaniline/graphene nanoplatelet nanocomposite having 10 wt% concentration of Graphene nanoplatelet, the electromagnetic interference shielding efficiency value was about −14.5 dB. The values of skin depth reduced as a purpose of applied frequency for pure polyaniline and Polyaniline/Graphene nanoplatelet nanocomposite nanocomposites, which evaluated their reliance on the difference of conductivity of the nanocomposites. The values of skin depth showed a strong reliance on the concentration of graphene nanoplatelet in the matrix of polyaniline. As the concentration improves in the matrix, the value of skin depth (δ) for the nanocomposites reduces. The rise in the concentration of graphene nanoplatelet led to the improved assembly of nanosheets that preventing the penetration of electromagnetic radiation too much deep within the shielding material. 124 Figure 9 shows conductivity values for various nanocomposites.
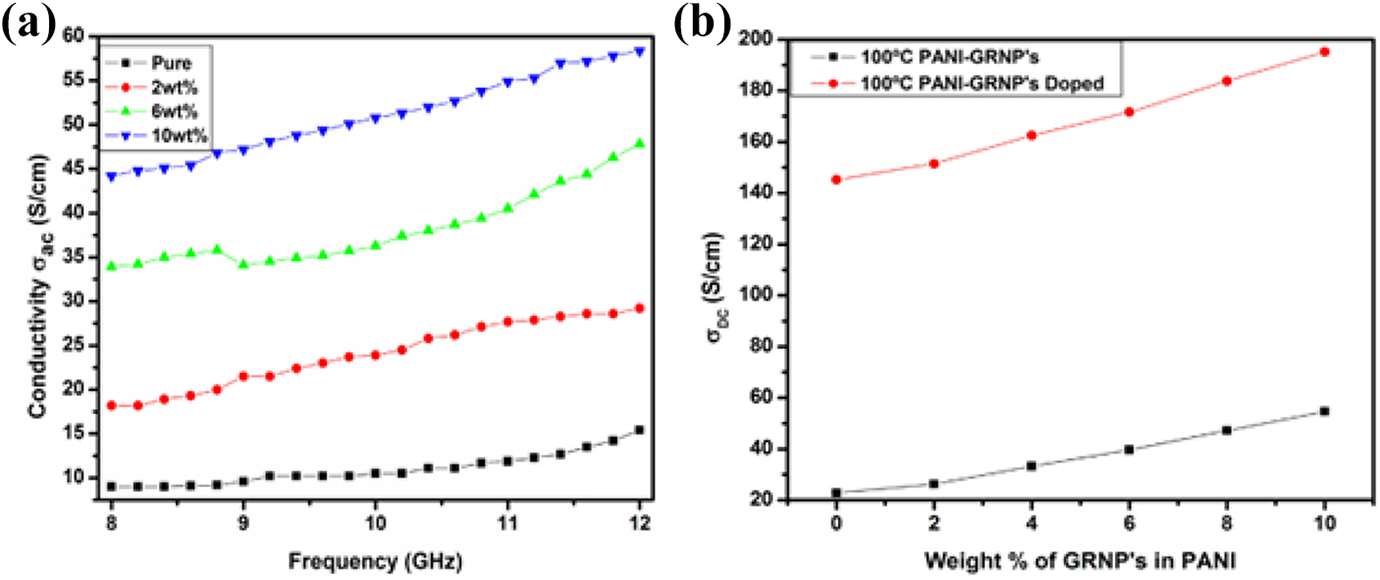
AC conductivity value for pure PANI and PANI/GRNP’s nanocomposites (b) DC conductivity as a function of graphene nanoplatelets content in polyaniline. 124
F. Ma et al. prepared PANI/PVC blends having a concentration of (7/89%) with the incorporation of an additional nanofiller of reduced graphene oxide to produce a conductive network having an electrical conductivity of 7.2 × 10−2 S cm−1. Wang et al. prepared stable as well as thin films of polyvinyl chloride/graphene nanocomposite having 20% of polyaniline content. The obtained electrical conductivity was 18 S cm−1 by a minimum of 30–60 µm thickness. 125 The coating of electrically conducting pallets of surfactant to polyaniline with graphene oxide was synthesized according to Cheng et al. having 10 S cm−1 value of conductivity and improved flexibility features. 126 In recent work, polyaniline/polyvinyl chloride/graphene nanopallets nanocomposites were synthesized with improved conductivity value. The EMI shielding value of PANI/PVC/GNP nanocomposites was obtained in the 10MHz–20 GHz frequency range. The shielding efficiency of PANI/PVC with 15 wt% was about 27dB that was enhanced to ∼ 51 dB with the addition of 5wt% graphene nanoplatelets, because of the absorption phenomenon. The improved shielding efficiency was attained primarily in the 11–20 GHz frequency range. 127 Kumar analyzed highly conductive polyaniline/vapor grown carbon fiber hybrid nanocomposite for flexibility features, electrical and EMI shielding characteristics. Polyaniline-based hybrid nanocomposites were produced by incorporation of vapor-grown carbon fiber as conducting nanofiller. The electromagnetic interference shielding efficiency of 51 dB in the X-band and electrical conductivity of 1.89 S cm−1 were measured. 128 The rise in the electromagnetic interference shielding efficiency was recognized to the increased electrical conductivities of the nanocomposites by incorporating vapor-grown carbon fiber. The electrical conductivities of the nanocomposites having 5 wt% concentration of vapor-grown carbon fiber were almost four times greater than the nanocomposites without vapor grown carbon fiber. The performance of vapor grown carbon fiber for the absorption of electromagnetic radiations as well as the formation of numerous phases inside the nanocomposites were further essential parameters for having an outstanding modification of electromagnetic interference shielding efficiency in the material.129,130
Modak synthesizes electrically conducting nanocomposites for EMI shielding. Polyaniline/graphene nanocomposite was synthesized through in-situ chemical oxidative polymerization of aniline in addition to graphene. 131 A sequence of nanocomposites by changing the percentage of functionalized graphene was prepared. The electrical conductivities of composites were reduced for 1%w/w graphene concentration as compared to pure polyaniline. While the conductivities of nanocomposites were attained to be improved with the increase in %weight of graphene. The electromagnetic interference shielding efficiencies of composites were evaluated as modified by increasing graphene concentrations. The value of electromagnetic interference shielding efficiency found to be highest at 5%weight concentration of graphene that was 51–52 dB in the 2 to 12 GHz frequency range which was above the value of efficiency at the commercial level than the needed value of electromagnetic interference shielding efficiency (20 dB) and could be utilized as lightweight electromagnetic interference shielding nanomaterials to shield electrical equipment and machines from electromagnetic radiations. 132 The improvement in electromagnetic interference (EMI) shielding efficiency by improving frequency was due to a reduction in skin depth (depth at which the field drops to 1/e of its original strength) of the materials. 133 Shakir et al. synthesized the PANI/PVC/TRGO nanocomposites. Figure 10 shows FTIR pattern of PANI. The polyaniline has characteristic peaks at 1100 cm−1 as well as 3500 cm−1 parallel to -C-N and -N-H stretching. Figure 11 shows the DSC results of polyaniline-based composites. The crystalline polyaniline has no effect on other nanoparticles present in nanocomposites and has sharp melting point of 138°C. 134 The total shielding efficiency of 56 dB was obtained by these nanocomposites.
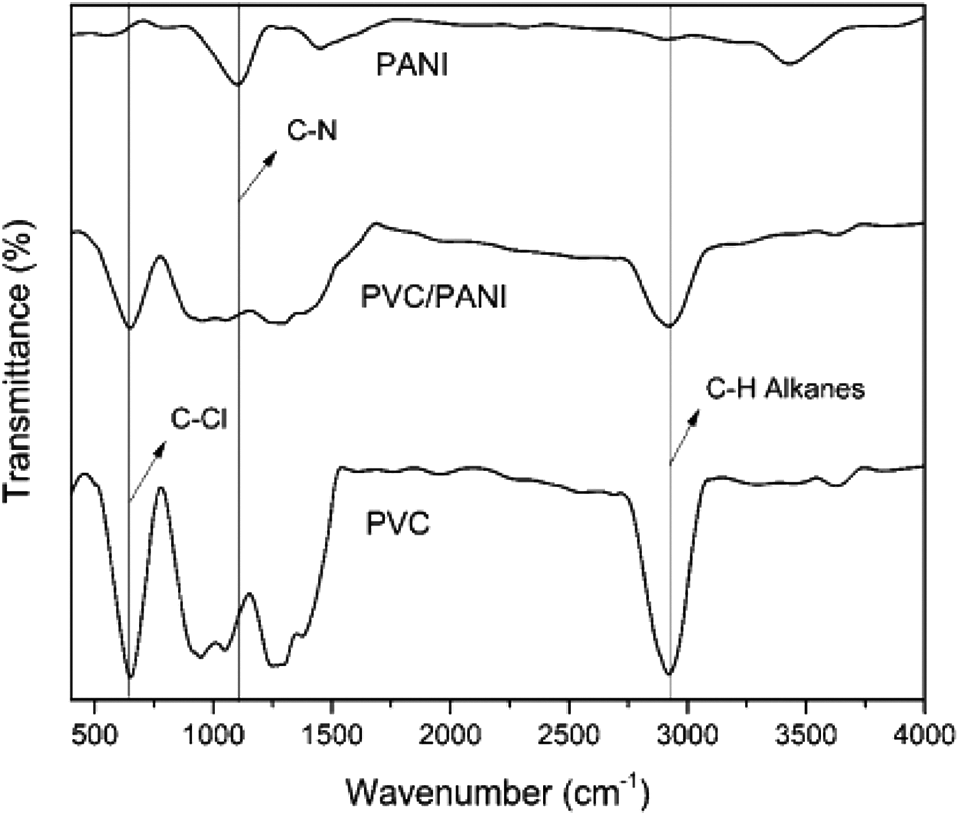
Graphical representation of FTIR of PANI composites. 134
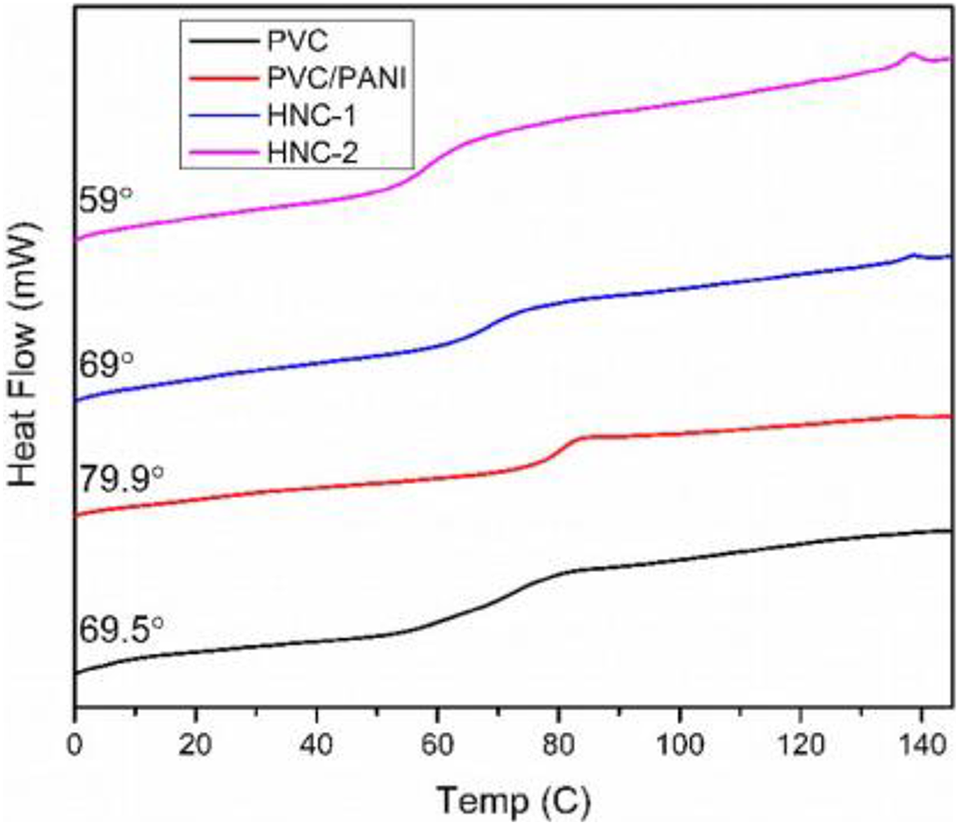
Graphical representation of DSC of PANI composites. 134
Polyaniline ferrites-based nanocomposites
Electrically conductive polyaniline-ferrite nanocomposites 135 having mutually electrical as well as ferromagnetic properties got huge attention, and developed as the high function and encouraging research zone.136,137 Conductive polyaniline-based ferrite nanocomposites having a well-planned system providing an innovative efficient blend containing organic as well as inorganic nanomaterials.138,139 As it is already depicted that electrically conductive polyaniline can significantly shield EM radiations produced by an electrical source, while magnetic sources produce extreme frequency EM radiations that could be efficiently shielded by nanomaterials containing magnetic particles.137,140 Therefore, the introduction of magnetic nanoparticles and conductive polyaniline nanomaterials inside multifunctional nanocomposites create new opportunities for achieving effective shielding for many EM resources.
Anum et al. synthesized polyaniline/polyvinyl chloride/barium hexaferrite-based nanocomposites by solution casting techniques. Figure 12 shows the XDR pattern of polyaniline. Two characteristics peak were observed at 19° and 25°. The shielding efficiency of 78dB was obtained by using these nanocomposites in a 0.1–20 GHz frequency range. 141
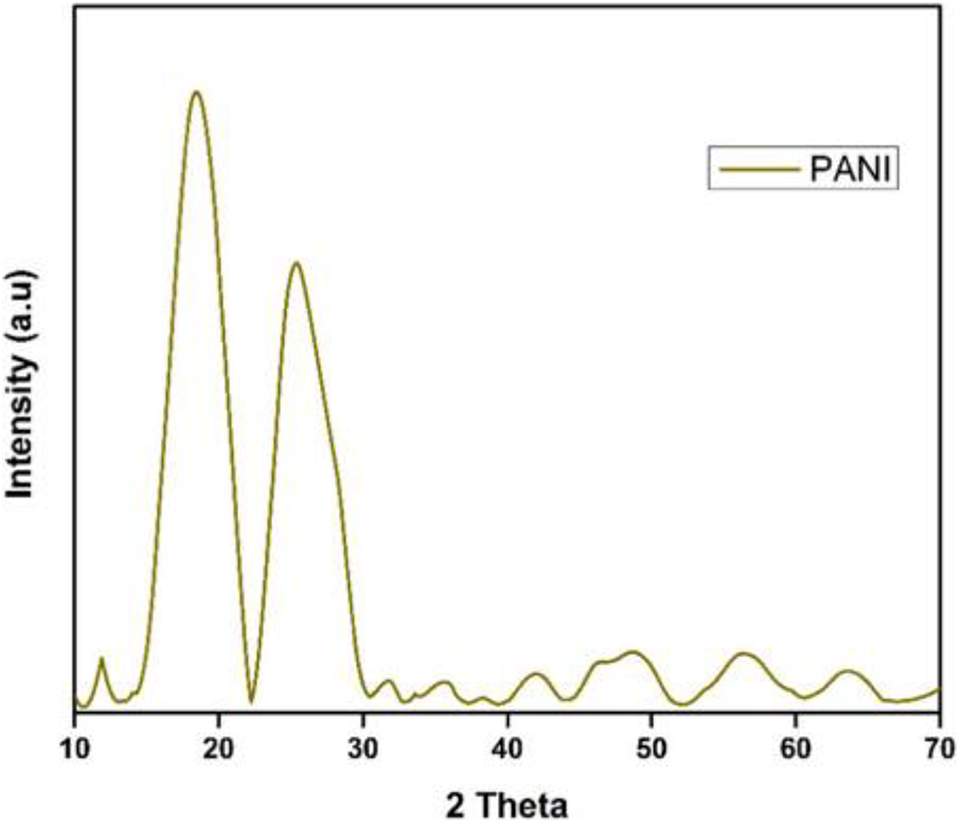
Graphical representation of PANI nanoparticles. 141
In recent work, Saini et al. synthesize copper doped cobalt ferrite nanocomposites with polyaniline by in situ polymerization at different ratios of ferrite. The nanocomposites in the applied magnetic field showed ferromagnetism at room temperature. The measured absorption loss was 26.4dB and 19.7dB for 10 wt% and 20 wt% of ferrite respectively. While reflection loss was 8.7dB and 6.3dB respectively. The highest shielding effectiveness value showed up to 35.1dB for the composites with a 20 wt% concentration of CoCuFe2O4 in X-band frequency of 8–12 GHz range with a minimum thickness of 2.2 mm. 142
In another work, Hosseini et al. prepared conductive polyaniline (PANI) and with core-shell structure of manganese ferrite (MnFe2O4) nanocomposites through in situ polymerization in the presence of an oxidant such as ammonium persulfate (APS)and a surfactant or dopant such as dodecylbenzene sulfonic acid (DBSA). Negatively charged core nanoparticles were synthesized through the emulsion polymerization technique. 143 The least reflection loss vale of −15.3 dB was measured in the 10.4 GHz frequency range with 1.4 mm thickness of coating. 144
Ezzati et al. synthesize conductive polyaniline and Ni/Mn substituted barium strontium ferrite composites by in situ polymerization and sol-gel technique. Figure 13 shows the processing mechanism of the PANI/Ferrite composite. Electric transition produced among polymer and ferrite was because of interaction between conductive and magnetic phase. The coating of PANI on ferrites reduced the saturation magnetization, slightly change the coercivity, and increase conductivity. The coercivity of polyaniline/ferrite composites was less than natural ferrite nanoparticles.145–147 The shielding efficiency was −21.68 dB in the 10.6 GHz frequency range with a 4 mm minimum thickness. The microwave absorption as well as reflection loss improved after the incorporation of polyaniline content from 11.2 GHz to 10.6 GHz. 139
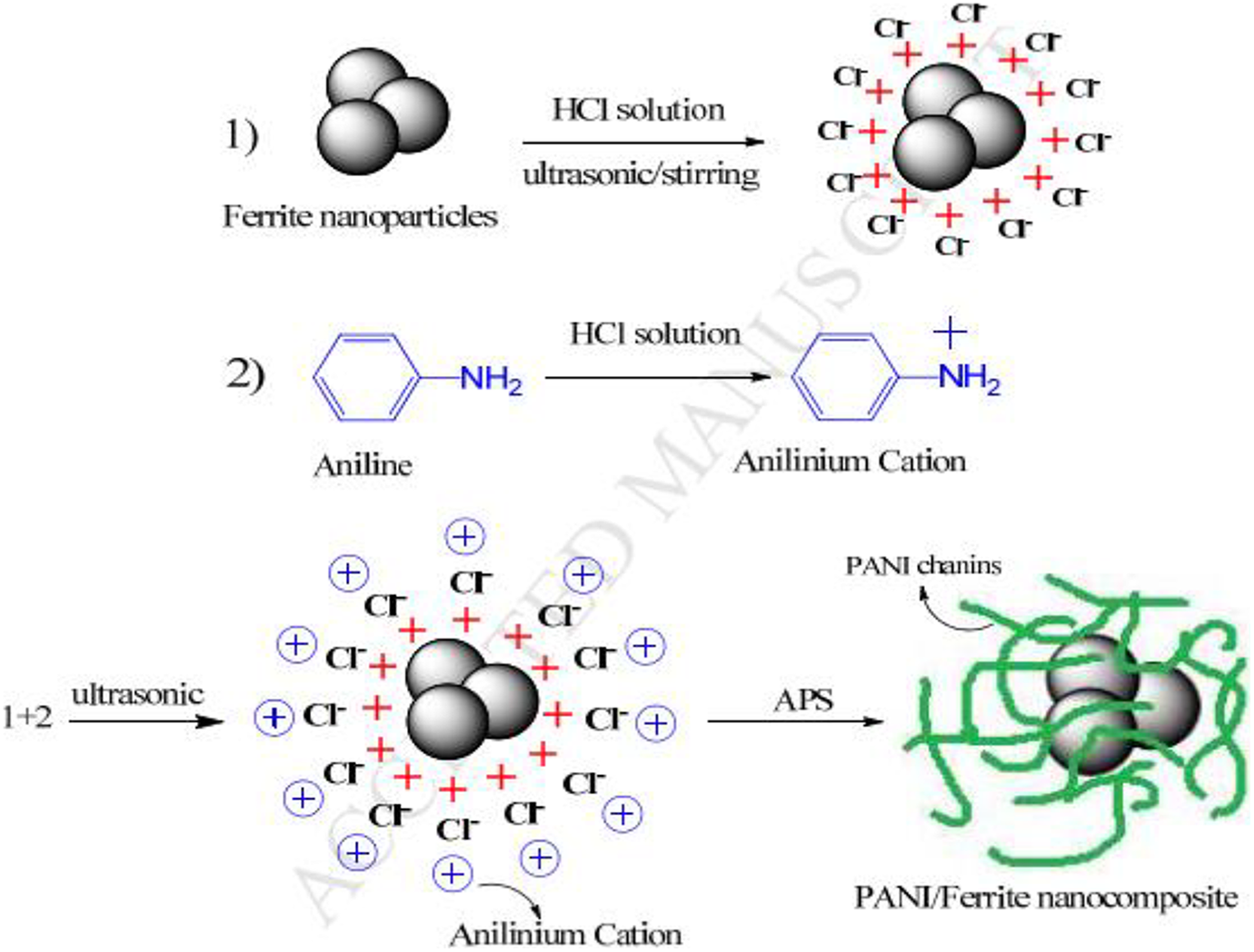
Synthesis mechanism of Polyaniline/Ferrite nanocomposite. 139
Wang et al. used in situ emulsion polymerization method to synthesize electrically conductive 148 and high crystalline polyaniline having magnetic nanoparticles of Mn0.5Zn0.5Fe2O4 having greater permittivity with better magnetic property. 149 The electrostatic force, hydrogen bonding, and paramagnetic force strongly bonded The ferrite nanoparticles were incorporated in the polyaniline surface by various strong forces such as electrostatic force, hydrogen bonding, and paramagnetic force to enhanced the thermal stability of the PANI nanostructures. Figure 14 indicated the uniform distribution of ferrite nanoparticles on the polyaniline surface. Polyaniline-based ferrite composites have acceptable shielding efficiencies (∼6 to 20 dB) in the 30 MHz to 200 MHz low-frequency range and improved with increasing polyaniline content. As Guan et al. and Li et al., the nanomaterial could be recognized for EMI shielding if the shielding efficiency is greater than 5 dB and 15 dB, in range of low frequency respectively.150,151 The incorporated ferrite in PANI nanostructures was thought to be an outstanding nanomaterial for the absorption of EM waves as it has a great number of dipole polarizations at the ferrite particles surface as well as on the polymer surface. 152

SEM images of (a) nanosize ferrite particles (b) nanosize PANI particles (c, d) nanocomposites of PANI/Ferrite. 152
Xu et al. synthesized nanoparticles of BaFe12O19 through reverse microemulsion method 153 to synthesize PANI/ferrite nanocomposite by in situ polymerization. Coralloid nanostructures were prepared irrespective of the size of inorganic particles, crumbling or virtual-spherical; while the inorganic particles influenced the development of coralloid nanoparticles with different weight percentages. There were only nucleation sites for polyaniline nanofibers instead of chemical interaction between polyaniline and ferrites. The absorption properties in the microwave region by polyaniline/barium hexaferrite nanocomposite were improved significantly than pure barium ferrite nanoparticles, which rarely exhibited waves absorption in the microwave region of 2–18 GHz due to its resonance frequency was 47.6 GHz. 154 The shielding efficiency was 19.7 dB in the 14.6 GHz frequency range with a 2 mm thickness. 155
Gandhi et al. synthesize Polyaniline and CoFe2O4 nanocomposite by in situ emulsion polymerization procedure by which ferrite particles were incorporated in polyaniline matrix. Magnetic and conductive nanoparticles possess a conductivity of 0.2 S/cm and saturation magnetization of the order of 22.4 emu/g. Nanocomposites have advanced activation energy which showed a degree of bonding and thermal stability between polyaniline and CoFe2O4. The incorporation of magnetic nanoparticles in polyaniline creates scattering with more interfacial dipolar polarization because of nano-size that improved shielding effectiveness value. The shielding efficiencies through absorption were measured as 21.5 dB whereas the shielding efficiencies by reflection were insignificant ∼3.5 dB in the Ku-band range of 12.4–18 GHz. 156 Figure 15 demonstrated shielding efficiency as a function of frequency.
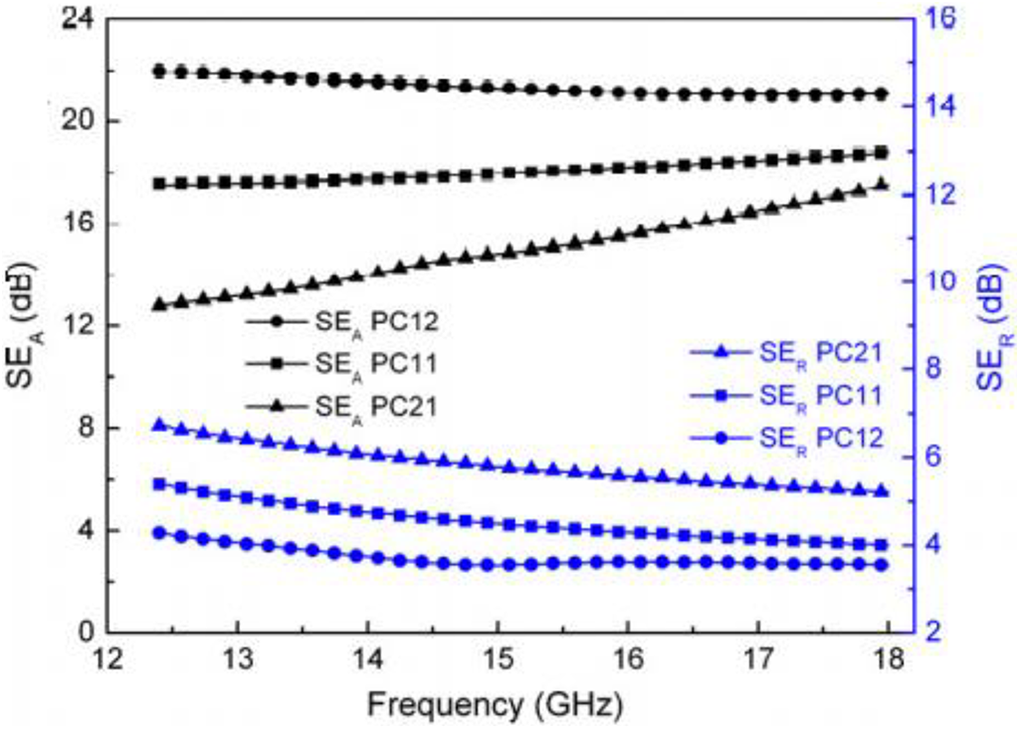
Shielding efficiencies of nanocomposites as a function of frequency. 156
EMI shielding efficiency of lead hexaferrite (PFO) and electrically conductive polyaniline nanocomposite in Ku and X band frequencies was observed by Choudhary et al. Pure polyaniline have shielding efficiency ∼−15 dB at 18 GHz frequency range while lead hexaferrite/polyaniline nanocomposite has SET of 24 dB at a frequency range of 18 GHz with a 3 mm shielding thickness that could shield ∼ 99% of the incoming electromagnetic magnetic radiation. 157
Polyaniline-based conducting polymers nanocomposites
Conductive polymer nanocomposite materials have outstanding features for electromagnetic interference shielding mechanism due to corrosion resistance, low weight, suitable electrical properties as well as dielectric features for preventing interferences, disturbances, and electrostatic discharge among the electrical systems.158,159 Most of the researchers have been used polyaniline-based polymeric nanocomposites because of their excellent properties and easy processing. 160 It was stated as the distinctive structure of conductive polymer composites, such as separated structures, 161 layer-by-layer assemblage, 162 double percolation system, and foam networks, and 163 multilayer structures, assist to modify the absorption of electromagnetic waves
Dhawan et al. prepared electrically conducting nanocomposite of PANI having polystyrene and poly methyl methacrylate by using melt blending technique in the180°C to 220 8°C temperature range. These electrically conducting nanocomposites were efficiently used for the degeneracy of static charge. The electrically conducting nanocomposites of polyaniline/polystyrene exhibited 58 dB shielding efficiency for EMI at the range of 101 GHz. 19 Sudha et al. synthesized EMI shielding nanomaterials from the conducting polymer mixtures of nanosize PANI with polyaniline-clay composites having a host matrix of ethylene-vinyl acetate. Electrically conductive nanosize polyaniline and polyaniline-clay composites were produced by using amphiphilic dopants, 3-pentadactyl phenol 4- sulphonic acid achieved by low-cost renewable product nutshell of cashew liquid and dodecylbenzene sulfonic acid by the in situ intercalative emulsion polymerization technique. 164 Nanosize polyaniline-clay composites (PANICN) were self-assembled protonated polyaniline in the composites. 165 The shielding efficiency with 2 mm thickness was observed of 40–80 dB at 8 GHz frequency range with 15% loading of conductive filler which makes these conducting nanocomposites potential applicant as EMI shielding materials for electronic devices. 166
The incorporation of powdered polyacrylate on polyaniline was synthesized by Niu through chemically oxidative polymerizations by the dispersion of polyacrylate with aniline monomer in HCl. After the dedoping of powders with water of ammonia, again doped by camphor sulfonic acid for making it conducting. The conducting polyaniline/PA nanocomposite coating was synthesized with bead milling camphor sulfonic acid and dope polyaniline incorporated polyacrylate powders. The level of polyaniline/camphor sulphonic acid increased the conducting values of the polyaniline/polyacrylate coating and polyaniline- camphor sulfonic acid/polyacrylate powders while observing the percolation threshold in the range of 0.3 and 0.4, respectively. The main purpose deceits that polyaniline/ camphor sulfonic acid was placed among the polyacrylate powder within the pressed pellets with excellent conduction routes was designed in fact a single layer of polyaniline/ camphor sulfonic was found at the interface of the polyacrylate powder, which is parallel to the effect of double percolation. 167 The EMI shielding observation evaluated that the efficiency of shielding for polyaniline/polyacrylate coating and powder can be amplified with and polyaniline- camphor sulfonic acid amount, with an achieved value of 60 dB having a minimum 0.1 mm thickness. 168
The EMI shielding and dielectric properties of polyaniline (PANI) and styrene-acrylonitrile (SAN)-based polymer blends were analyzed by Saboor et al. through an impedance analyzer. Polyaniline (PANI) was used as a conductive filler which was dispersed in an insulative SAN matrix by varying concentrations range from 5 to 50 wt%. The phase-separated morphology was formed by polyaniline in the SAN matrix and ∼40 wt%, was the percolation threshold concentration which forms an interconnected system of polyaniline in the styrene-acrylonitrile matrix. The PANI/SAN polymer blends have enhanced dielectric properties significantly at the percolation concentration. Permeability, permittivity, and AC conductivity of styrene-acrylonitrile were improved by different magnitude orders. The maximum rate of shielding efficiency through reflection was 164 dB at a frequency range of 1 kHz at ∼40 wt% percolation threshold concentration having maximum
To consent the electronic system to exist deprived of dangerous electromagnetic waves, the development of modern absorbing as well shielding nanomaterials having better functioning in high effective frequency band was essential. Lakshmi et al. developed a conducting polymer nanocomposite suitable for EMI shielding along with electrostatic charge dissipation. Conducting nanomaterials of polyaniline with polyurethane nanocomposite thought for shielding features. The EMI shielding properties by microwave absorption and microwave reflection for polyaniline/polyurethane nanocomposites were analyzed in the microwave region as well as the S-band region. The width of the shielding nanomaterial has a huge effect on the shielding efficiency of nanocomposite and these nanosize materials were excellent for the effectiveness of shielding in the 2.23–8.82 GHz range. The number of dips, status, and concentration was reliant on the thickness of samples.170,171 The shielding effectiveness of 18.2 dB and 26.7 dB obtained with a minimum 0.62 mm and 1.9 mm thickness respectively in the X-band frequency range of 8.82 GHz frequency range the X-band. Whereas the shielding efficiency of 10.3 dB and 15.5 dB obtained at 10.18 GHz range of frequency which proved ideal material for shielding is at 8.82 GHz. 172 Wojkiewicz developed the polyaniline/polyurethane (PANI/PU) nanomaterials for efficient shielding properties in the microwave frequency range. These conducting materials have a low percolation threshold because their electrical features can be simply changed by the magnitude proportion of PANI blend or maybe through chemical technique. By the use of two set-off waveguides, the shielding efficiency is estimated to be found of 30–90 dB at 8.22–18 GHz frequency range for three different samples having lightweight and excellent mechanical properties. 173
Zhang synthesizes a highly flexible polyaniline/polyethylene terephthalate nanocomposites paper having effective electromagnetic shielding capability by facile and cost-effective in situ polymerization technique. The synthesized polyaniline/polyethylene terephthalate nanocomposite significantly provides the electrical conductivity of 0.78 S·cm−1. The smaller skin depth revealed a stronger absorption capacity. 174 The smallest skin depth attained the highest absorption. Multiple reflections also improve total shielding effectiveness.175,176 Figure 16 shows the shielding mechanism of nanocomposites. The nanocomposites were demonstrating an effective shielding proficiency of 23.95 dB under the excellent features of mechanical bending deformation. The durability for bending was mainly ascribed to the appropriate coating thickness, the strong interfacial interaction, and an exceptional nanoporous heterostructure. The interfacial polarization, electric dipole, multiple reflection behavior, and capacitive structure effect gave a shielding efficiency with a minimum 0.39 mm thickness. 177
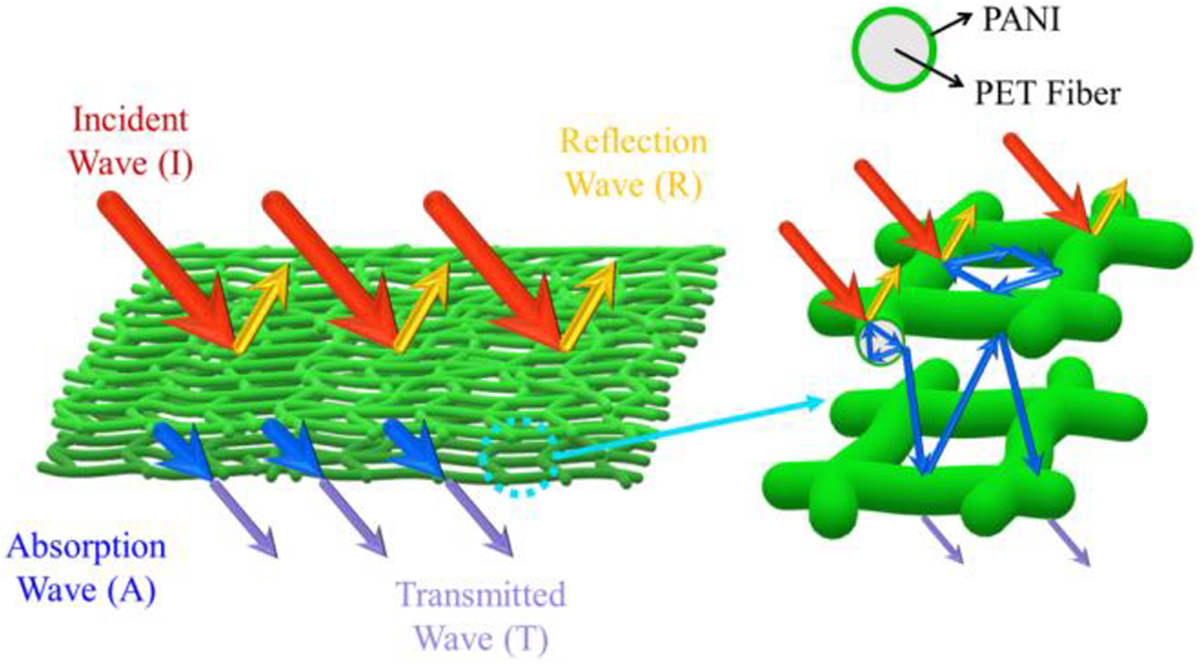
Schematic representation of the shielding mechanism of PANI/PET nanocomposite film. 177
Bora et al. used a solution mixing technique to synthesize polyaniline/polyvinyl butyral nanocomposites. The improvement of shield conductivity and dielectric loss, as well as electromagnetic reduction with a frequency, resulting in an outstanding electromagnetic interference shielding efficacy in the Ku-band and X-band region. In the frequency region of X-band, the obtained SE was about 26 dB for electromagnetic interference shielding efficiency (shielding because of absorption phenomenon about 21 dB while shielding because of reflection about 5 dB) that thought to enhanced about 30 dB (shielding by absorption is about 26 dB while shielding by reflection is about 4 dB) in Ku-band. An improvement of shielding, the dielectric loss, conductivity, and electromagnetic attenuation continuous with frequencies have resulted in outstanding features of polyvinyl butyral/polyaniline films for electromagnetic interference shielding. 178 Figure 17 demonstrated difference of total EMI shielding efficiency in the (a) X-band region and (b) Ku-band region.
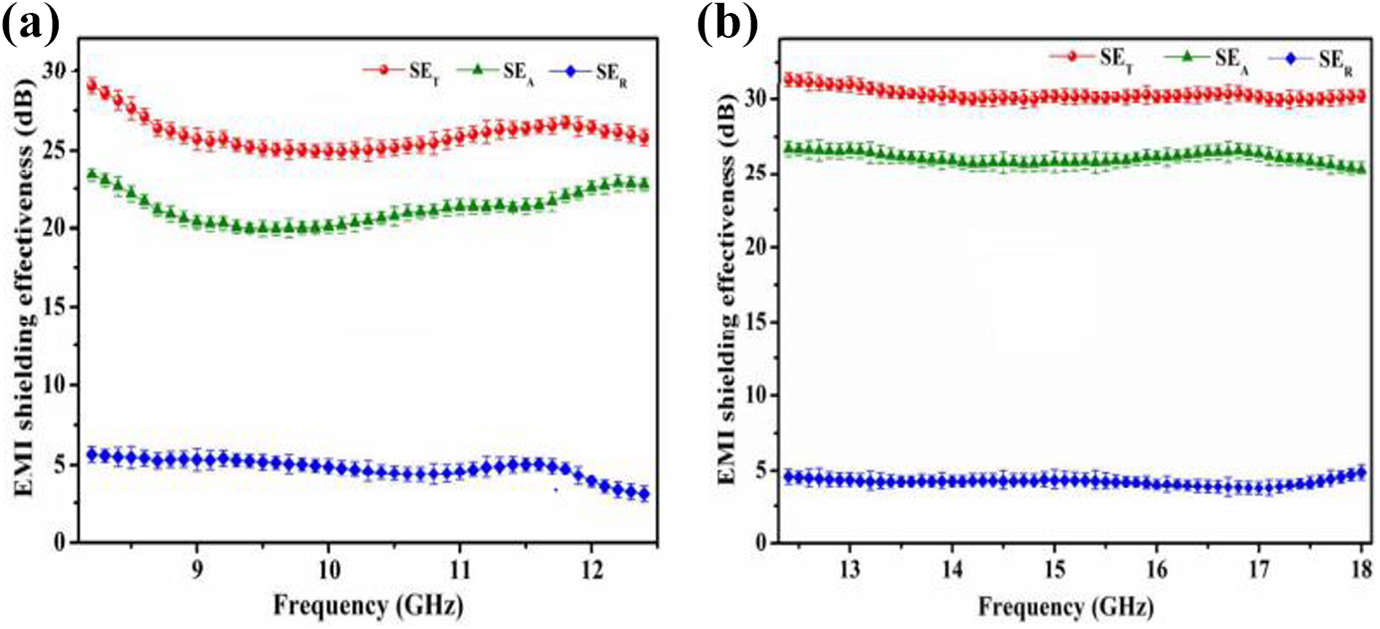
EMI shielding efficiencies in (a) X-band (b) Ku-band. 178
The electrically conductive blends of nanofiller such as polyaniline-based clay composites with polystyrene were prepared by Sudha et al. using the matrix as host through one step by emulsion polymerizations to create a low-cost renewable product. These interactions were produced among the basic nanoparticles with a matrix. The electrical conductivity of 7.6 × 101 S/m was obtained from a low-cost PANICNPS blend. The effectiveness of excellent thermally stable and shielding material was analyzed 10–20 dB at frequency ranges of 8 GHz which converts into promising applicants having properties for EMI shielding as well as for the protection of the electrical device, it acts as an antistatic ejection matrix. 179 The consequence of the amount and behavior of conducting nanofiller incorporation at the concentration of the percolation threshold has an important effect on the efficiencies of shielding. 180
Polyaniline-based multiple nanocomposites
Polyaniline-based multiple nanocomposites are used for improved electromagnetic interference shielding properties. The electromagnetic absorption characteristics of many electro-conductive polyaniline nanocomposites associated with graphene ass dielectric and ferrite as magnetic nanomaterials have been widely studied. In this work, Kauser prepared polyaniline/polystyrene microspheres/multiwalled carbon nanotube (PSMS/PANI/ MWCNT) nanocomposite for EMI shielding. The dispersion polymerization system to synthesize polystyrene microspheres from styrene monomer was used. In situ oxidative polymerization method to synthesize polystyrene microspheres/polyaniline/multiwalled carbon nanotube (PSMS/PANI/ MWCNT) was used. Figure 18 shows the structural formation scheme. Fine morphology of core-shell particles was obtained by combination technique which was also compared with previous research.181–183 The perfect morphology of nanocomposites modified the rise in shielding efficiencies. The incorporation of conductive polyaniline and nanofillers improved the organized network structure which absorbed and reflected the incident EM waves and enhanced efficiencies of shielding nanomaterials. The efficiencies of shielding material also improved with the modification in conductivity value from −14 to 0.87 mho m−1. The polyaniline/polystyrene microspheres/multiwalled carbon nanotube composites showed EMI shielding efficiency of 23.2 dB in the 8.2 to 12.4 GHz frequency ranges. 184
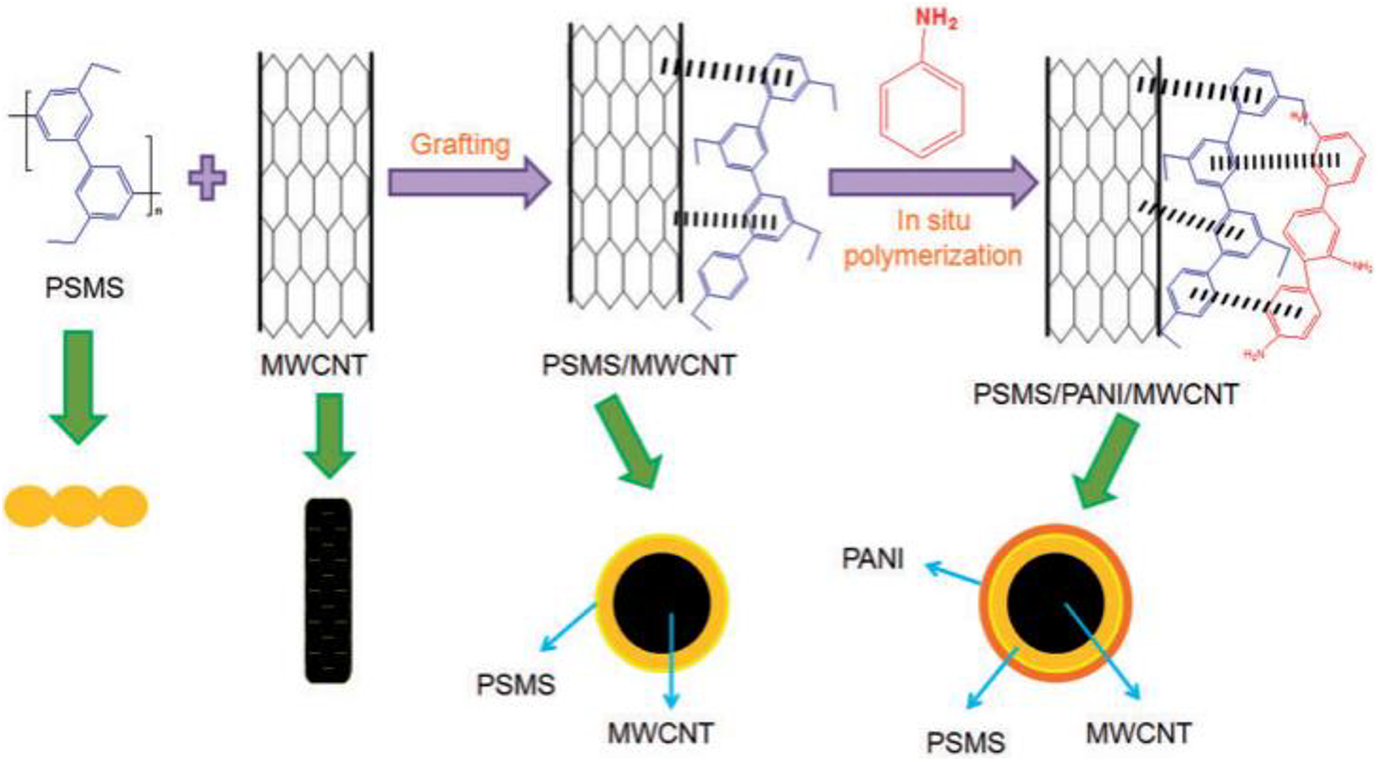
Schematic formation of PSMS/MWCNT and PANI/PSMS/MWCNT composites. 184
It was also studied that the impedance matching of electric and magnetic as well as microwave absorption property can be enhanced with the combination of the carbonyl iron and various nanomaterials having dielectric loss properties such as ZnO 185 MnO2 186 as well as polypyrrole. 187 Jafarian et al. prepared microwave absorber composites created on a mixture of polyaniline coated with multiwalled carbon nanotubes and microspheres iron carbonyl (CI) by an easy blending method. The hollow iron carbonyl microsphere was synthesized through pitting corrosion based on Yin et al. 188 The complex features like microwave absorption, permeability, and permittivity of the nanocomposites were obtained at 8.5–12.5 GHz ranges. The maximal reflection loss was obtained –25.5 dB at 11 GHz having a thickness of 2.0 mm with a bandwidth of 3.6 GHz. 189 Jaiswal et al. prepared polyaniline incorporated ferrite and reduced graphene oxide nanofillers by solution mixing technique for properties of microwave absorption in defense applications. The complex permeability and permittivity were evaluated in the 2 to 18 GHz frequency ranges. The −10.26 dB reflection loss was measured in the X band having a bandwidth and thickness value of 8.47 GHz and 3 mm respectively. These composites were considered outstanding for applications of EMI shielding in microwave absorption region. 190
Manna prepared polyaniline/carbon/ferrites nanocomposite as an effective lightweight EM radiation absorption. C/Fe3O4 was made with the hydrothermal technique on which polyaniline was coated with in situ polymerization technique. The reflection loss of PANI/C/Fe3O4 nanocomposites was measured ∼33 dB at 2–8 GHz, while the shielding effectiveness owing to absorption was ∼47 dB and reflection was ∼15 dB. The maximum rate of shielding efficiency was because of the presence of magnetic and dielectric integration as well as dual interfaces in PANI/C/Fe3O4. The high dielectric loss value by relaxation and interface polarization effects in PANI/C/Fe3O4 subsidized toward greater absorption in microwave region capacity of PANI/C/Fe3O4 as compared to PANI/Fe3O4 and C/Fe3O4 nanocomposites. It was commanded to modified the natural resonance, interfacial polarization, effective anisotropy energy, dielectric polarization, and trapping of electromagnetic waves by internal reflection in PANI/Fe3O4/C. 191
Zhang et al. prepared reduced graphene oxide/polyaniline/silicone dioxide/ferrites nanocomposite through low-cost approaches. The EM restrictions which confined the interfacial polarization, multiple reflections, the charge transfer, scatterings, the magnetic loss, Debye dipolar relaxation methods, the matching wavelength with distinctive impedance improved the function of electromagnetic radiation absorption. Figure 19 shows the morphology of and RGO/PANI/SiO2/α-Fe2O nanocomposite. The shielding efficiency because of high reflection loss for nanocomposite of RGO/PANI/α-Fe2O3/SiO2 was found to be as −50.06 dB at 14.4 GHz frequency with 2.3 mm of thickness. 192

SEM morphologies of (a) RGO/PANI/Fe2O3/SiO2 composites (c, d) EDS pattern. 192
Lakshmi et al. prepared manganese-based ferrite chemically improved with polyaniline (PANI) and polymethylmethacrylate (PMMA) EMI shielding nanomaterials that provided ∼44 dB shielding efficiency in the 8−12 GHz X band frequency range. 193 Bera et al. prepared nanoporous polyvinylidene fluoride/ (ferrosoferric oxide coated PANI/SWCNH nanocomposites by an easy solution blending technique. Conductive nanofiller developed the porous morphology and conducting network in nanocomposite during solution blending process by discharging out from the NaCl salt. The nanocomposite evaluated excellent electromagnetic interference shielding efficiency was because of its nanoporous arrangement and existence of an electrically conducting system (PANI-SWCNH) with ferromagnetic (Fe3O4) particles. The nanocomposite occupied a high conductivity value of ∼2.5 × 10−2 S/cm with a thickness of 2 mm, permeability property, and maximum dielectric loss. The shielding effectiveness of −29.7 dB was measured in the region of Ku-band. 194
Jelmy synthesizes electro-conductive Au/polyaniline/multiwalled carbon nanotube composites through in situ polymerization with Au particles and multiwalled carbon nanotube (MWCNTs). The nanocomposites have improved electrical conductivity of 36.06 S cm−1 which creates improvement in absorption and reflection losses, which improved the efficiency of shielding the nanomaterials. 129 The least reflection loss exhibited extreme absorption of the electromagnetic signals by the shielding nanomaterial. The Au-MWCNT/PANI nanocomposite has a reflection loss of −56.6 dB. The total shielding efficiency of nanocomposite was obtained at −16 dB in the 8–12 GHz range of X-band. Due to the presence of conducting nanofillers, the advanced dielectric properties and maximum electrical conductivities create the development in electromagnetic interference shielding and absorption in the microwave region. 195 Figures 20 and 21. shows reflection loss and EMI SE respectively
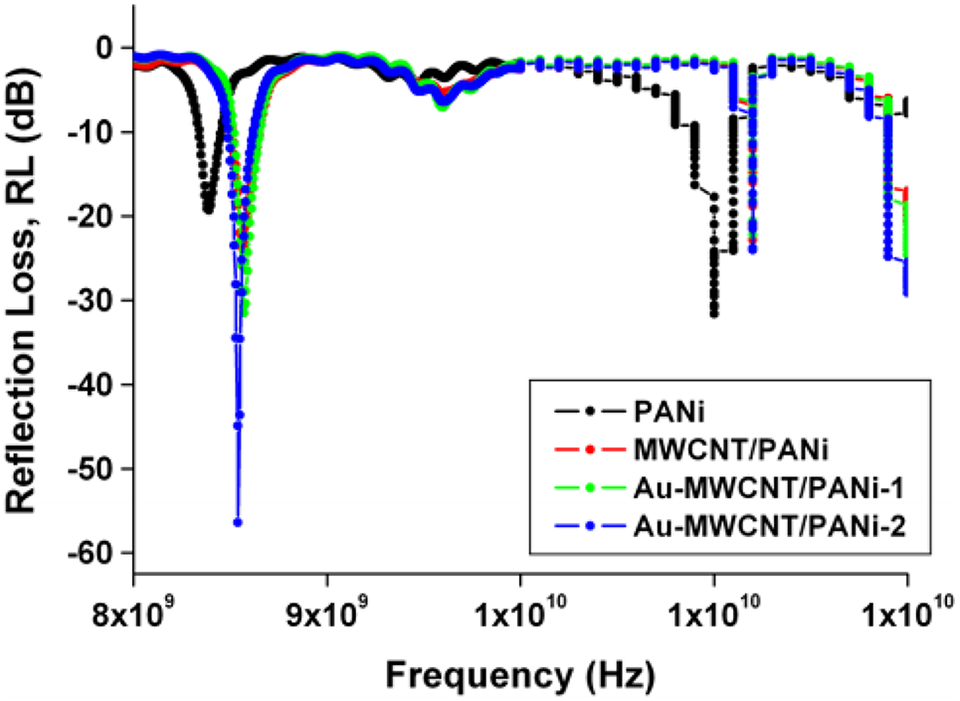
Reflection loss in X- band as a function of frequency. 195
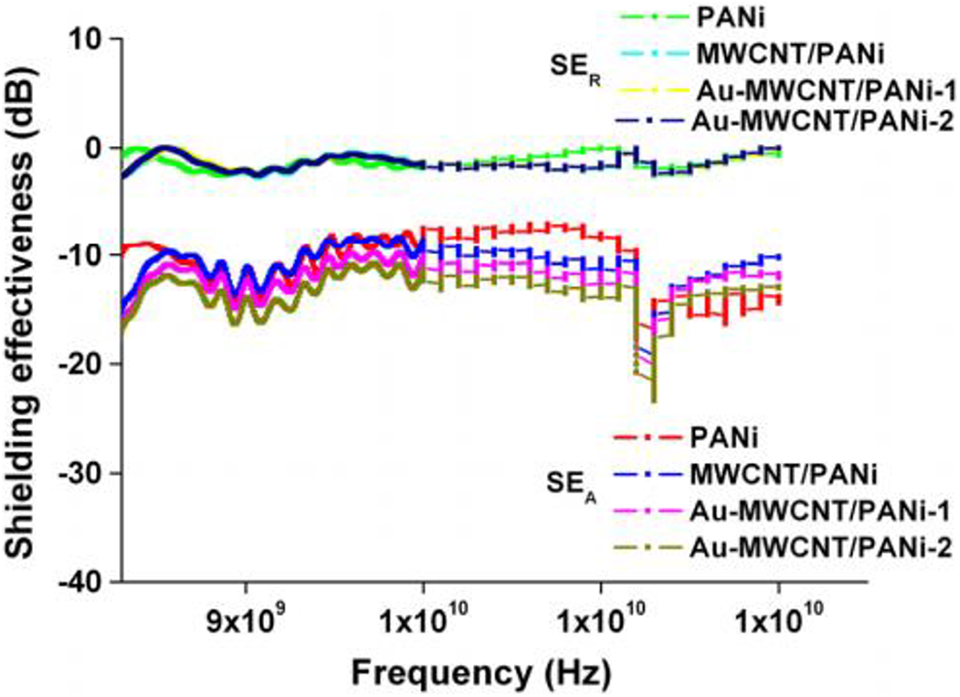
EMI shielding efficiencies of various nanocomposites in X-band. 195
In this work, Sabet et al. used many needle-like magnetic nanomaterials to modified the microwave absorption property of PANI. He prepared PANI/CNT/CoFe2O4 and PANI/CNT/NiFe2O4 to study microwave absorption behavior. The electrical resistance of polyaniline was 40 Ω cm–2. When carbon nanotubes (CNT) were added to the polymeric matrix by 5% weight percentage, the electrical conductivities were improved deeply while electrical resistance was reduced to 10.5 Ω cm–2. The improvement in electrical conductivities by incorporating carbon nanotubes was also allocated to the effect of dopant or the process of charge transfer from polyaniline to carbon nanotube. 196 The addition of nanosize ferrite to PANI/CNT created a further increment in the conductivity was observed while the electrical resistance of PANI/CNT/NiFe2O4 and PANI/CNT/CoFe2O4 were calculated about 9.7Ω/cm2 and 10Ω cm–2 respectively. These outcomes were also related to the results of Khairy and Gouda. 197 The shielding efficiency due to maximum reflection for the nanocomposite of PANI/CNT/CoFe2O4 was 32 dB in the 7.8 GHz frequency range while shielding effectiveness for PANI/ CNT/NiFe2O4 was 33 dB in the 6.8 GHz frequency range with a 0.5 mm thickness. 198
Liu prepared nanocomposites of PANI/graphene/CoFe2O4 through in situ polymerization. The nanocomposite showed ferromagnetic behavior. The enhancement in dipole polarization was because of the production of dielectric loss by nanosize of CoFe2O4 particles. The presence of multiple interfaces in the nanocomposite functioned as a polarized center, contributing to electromagnetic radiation absorption property because of the presence of interfacial polarization. Polyaniline and sheets of graphene are absorbers for dielectric loss, while CoFe2O4 particles are absorbers for magnetic loss. The electromagnetic radiation absorption was improved by different properties of nanocomposites such as dielectric loss, magnetic loss, matching impedance. The highest reflection loss by the PANI/graphene/CoFe2O4 nanocomposites was 47.7 dB in the 14.9 GHz frequency range with 1.6 mm thickness. 199
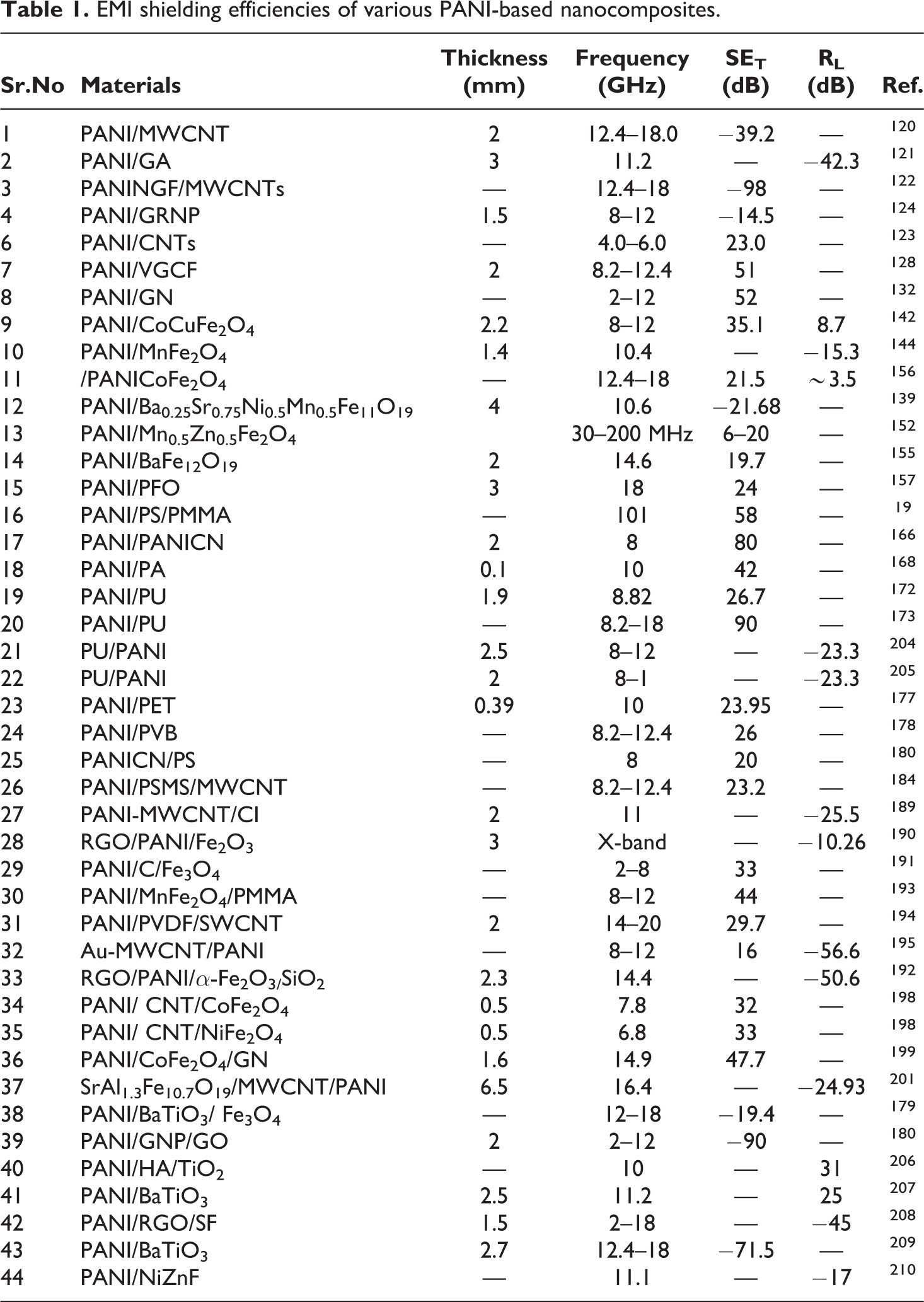
Peymanfar used a sol-gel technique to prepare aluminum substituted strontium hexaferrite nanoparticles. The polyaniline-based magnetic nanocomposites (PANI/MMWCNT/SrAl1.3Fe10.7O19) were prepared by a sonochemical technique through in situ polymerization method. The EMI shielding effectiveness was obtained at a frequency range of 12.4–18 GHz Ku-band. The value obtained by reflection loss evaluated the nanocomposites as an outstanding absorber for radiation for Ku-band, which was 224.93 dB at the range of 16.40 GHz. Based on dielectric properties and free-electron theory, 200 the absorption characteristics of the magnetic nanoparticles (SrAl1.3Fe10.7O19) were modified with the addition of conductive polyaniline and multiwalled carbon nanotubes. The dispersing agent present in the functional group of the MWCNTs improved the value of the nanocomposites. 201 The shielding efficiency was measured at 10 dB with a thickness of the thickness of 6.5 mm. The interfacial polarization was the main factor that attenuates the EM radiations. 202 The coating of polyaniline on the MWCNT/SrAl1.3Fe10.7O19 surface by a sonochemical technique through in situ polymerization created the interfacial polarization among SrAl1.3Fe10.7O19, MWCNT as well as polyaniline. 203 A comparison is shown of various composites with their shielding effectivness in various regions in Table 1.
EMI shielding efficiencies of various PANI-based nanocomposites.
Conclusion
In this article, the advanced polyaniline-based nanocomposites are discussed as an application of EMI shielding nanomaterials. The main shielding concept for electromagnetic emission of radiation by the effect of particle size, absorption properties, magnetic properties, dielectric properties as ell ass concentration of conductive nanofillers are described. The attractive properties of polyaniline nanoparticles and their corresponding nanocomposites like easy processability, broad bandwidth, low matching thickness, broad bandwidth, and maximum absorption proved as a potential candidate in future electromagnetic interference shielding applications. It demonstrated as the addition of electrically conducting polymers to the carbon nanomaterials can improve the electromagnetic absorption applications by overcoming these restrictions, such as the electromagnetic absorption improved through polyaniline nanoarray coating on the graphene surface. They frequently use magnetic materials as ferrites (γ-Fe2O3) for electromagnetic absorption is due to its elevated permeability, stability as well as magnetic loss. The characteristics of conductive PANI can also be improved by the combination of inorganic filler. Nanostructured ferromagnetic fillers are attractive because of the absorbing properties result from the enormous surface area with small size. The electromagnetic absorption performance of many conducting polyaniline-based multiple nanocomposites associated with dielectric materials such as graphene and magnetic materials such as ferrites have been also discussed studied. This review article highlight recent progress in the area of research for achieving innovative ideas about effective shielding nanomaterials at a different level.