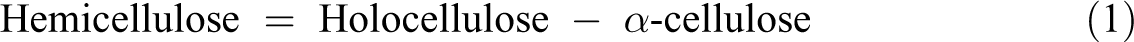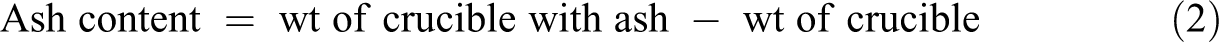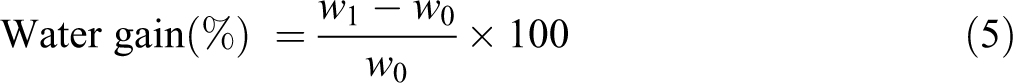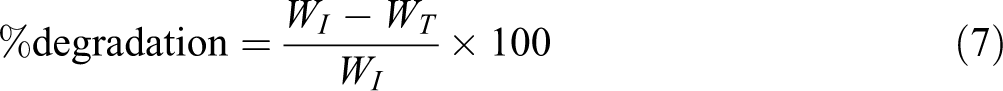Abstract
The present work focuses on surface functionalization, characterization, biodegradability and barrier properties of MOSF as a practicable reinforcement in PVA matrix. Film-forming dispersions at different concentrations of alkali and acid treatments were casted at room temperature. The effect of surface modifications on the developed film’s compositional, physical, mechanical, biodegradability and barrier properties were analyzed. Fourier transform infrared spectroscopy (FTIR), X-ray diffraction (XRD), Scanning electron microscopy (SEM) and Atomic force microscopy (AFM) were used to describe the functional composition, formation and surface morphology of the film. The 5% acid treated film significantly increases the tensile strength (33.69 MPa) and flexural strength (56.612 MPa), which was close to the frequently used LDPE and HDPE package films. Composite films were moisture absorptive but simultaneously capable to maintain uniformity and composition upon modifications. Lower water vapor permeability (1.42 × 10−10gs−1 m−1 Pa−1), eminent biodegradability proved the suitability of composite film for various packaging applications.
Introduction
In the last two decades, the use of non-biodegradable plastics derived from synthetic polymers such as PET (polyethylene terephthalate), HDPE (high density polyethylene), PVC (polyvinyl chloride), LDPE (low density polyethylene), PP (polypropylene), PS (polystyrene) etc.—which are generally petroleum based has increased remarkably. 1 One of the present day research trends is to replace the non-biodegradable plastics with plastics made up of renewable raw materials, particularly plants. This is closely associated with the fact of increasing consumer interest for high quality with serviceable life products and hence maximizing the awareness of environmental issues linked with the non-degradable packaging 2 The thirst of using the renewable material has given upliftment in the field of natural fiber polymer films. Natural fiber polymer films have engaged much attention because of their exceptional biodegradable and biocompatible nature, and the scope of its potential applications such as in automobile 3 and other manufacturing industries. 4 Presently, various natural fibers have been explored with polymers including, wheat straw, hemp, kenaf, bagasse, sisal, banana, flax, alginate etc.5–7 A notable disadvantage of these lignocellulosic, hydrophilic natural fibers is their poor compatibility and adhesion with hydrophobic matrix. This issue can be remediated by different chemical and surface treatment methodologies like alkylation, acidification, benzoylation etc. which have been experimented and suggested by researchers to improve the compatibility and adhesion,8–10 In recent times, researchers from different part of the world are considering to use locally available novel natural fiber which has various uses with outstanding properties. It will not only reduce the burden of traditional lignocellulosic fiber but will also strengthen the economy of rural sector by providing diversity in application.
Moringa Oleifera seeds, derived from Moringa Oleifera Lam (MOL) commonly known as “drumstick tree” is representative of Moringaceae ancestry and is locally available to the sub-tropical and tropical regions of Himalayan trail in the north-western India, Pakistan, Afghanistan and Bangladesh. 11 In recent times, MOL has accepted a greater recognition worldwide because it is very economical, easy and fast growing in changeable scale of location, space and geographical state. Its elevated production yield, versatile application of all its vegetative compositions such as flowers, pods, leaves etc. has a conventional use in medicinal field, in agronomy and water purification purposes. 12 Extensive work reviews are available on various parts of Moringa Oleifera tree to provide its adequacy in respective applications. Sampath and Kumar 13 extracted Moringa Oleifera (MO) fruit fiber from Moringa pods and reinforced with Ly556 epoxy by using hand-layup technique. Alkali treated fiber exhibited better mechanical, water absorption properties as compared to untreated fiber. Ayrilmis et al. 14 fabricated cellulose epoxy nanocomposite and found that the mechanical and thermal properties are improved when compared with neat epoxy. Amina et al. 15 fabricated MO seed oil based PVC films improved with silver nanoparticles and found an enhancement in thermal stability of film. Mimusops elengi seed filler were reinforced with polypropylene polymer matrix. Morphological properties showed that the filler had a good adhesion on the polymer matrix which ultimately increased the mechanical properties. 16 Delonixregia seed filler reinforced with recycled LDPE through compression molding process and increment in tensile and flexural properties of the composite in contrast with the pure LDPE polymer was observed. 17 Guo et al. 18 prepared packaging film with bagasse fiber reinforced with PVA matrix through solution casting method and found that 3 times more tensile strength than pure PVA film. Lamaming et al. 19 fabricated film with oil palm biomass waste and PVA matrix and showed improved thermal and mechanical properties than neat PVA films. Shukla et al. 20 extracted microcrystalline cellulose from rice husk and blend with PVA matrix through solution casting method and found that the mechanical and barrier properties of film increased significantly.
The literature survey supports the scrutiny of using thermosetting polymer in Mo filler/fiber or in other novel fibers with the aim to explore different properties while building the composites. The advantages of thermoplastic polymers over thermoset polymers are high processability, good cost-performance ratio, good resistance to fatigue ratio, required low handling temperature requirement. Furthermore, they have a capability to withstand chemical changes and provide considerable depletion in toxicity, fumes and flammability performances. On account of natural fillers as a reinforcing material and thermoplastic matrix composites discover a broad field of application including packaging, textiles but not restricted to plastic segments, stationary articles, recyclable holder vessels of different types, laboratory apparatus and automobile parts. Considering the above reality, an effort has been made to build composite film through Moringa Oleifera seed filler (MOSF) and PVA (polyvinyl alcohol) thermoplastic polymer matrix. PVA, a synthetic polymer, is non-toxic, water dispersible with outstanding film-forming, chemical resistance, thermal stability and biodegradable properties. Moreover, its usability in material industry such as coating and packing material has been explored widely. 21 Several papers are accessible on biodegradable composite packaging film fabricated from PVA and sustainable biopolymers such as starch, 22 chitosan, 23 halloysite nanotubes, 24 zinc sulfide, 25 sodium alginate, 26 polyvinyl acetate, 27 and carboxymethyl cellulose. 28 However, to our understanding till now no work is reported on various characterization of MOSF-PVA blended film.
Depending on these inspection and requirement for better research toward potential industrial implementation of biodegradable films, the goal of this work was to build novel biocomposite, environmental friendly films by blending PVA with MOSF using a solution casting method with glycerol (plasticizer), citric acid, glutaraldehyde (cross-linkers). Also, to analyze some attributes of these films such as physical, mechanical, thermal, water absorption, biodegradability, crystallinity and morphological properties to inspect their capability as packaging material.
Experimental
Material used
MO seeds were obtained from Matras exporters, Madhavaram, Chennai, India. To remove surface impurities, seeds were rigorously washed with distilled water. These were sun dried for 24 hrs, following which they were kept in an electric oven at 70°C for 48 h. Seeds were cut and assorted into pieces after which they were grounded in an electric mixer. Afterward, sieving of the filler to acquire significant fraction of size 300–500 μm. Ultimately, it was air dried at 105°C for 4 h and desiccated in dry atmosphere after which the process started. PVA (Polyvinyl alcohol) with an average molecular weight of ∼1,60,000, viscosity of 27–33 cP, alcoholysis degree of 86.50–89.00 mol% purchased from M/S Himedia laboratory Pvt. Limited (Mumbai, India).29–31 The chemicals for compositional (lignocellulosic) analysis mainly, benzene, ethanol, NaOH and sulfuric acid were supplied from SD fine chemical, India and M/S Himedia laboratory Pvt. Limited (Mumbai, India) respectively. Distilled water was produced in lab by distilled water unit.
Methods
Chemical modification of MOSF
Alkali treatment
MOSF were soaked in NaOH solution having varying concentrations of 2.5 wt%, 5 wt%, 10 wt% for a period of 60 mins. The liquor to fiber ratio was kept at 20:1.32,33 To remove the impurities and maintain a neutral pH level, 3–4 times washing of these fillers was carried out with distilled water. These washed fillers were then placed in a hot air oven for 2 days at a constant temperature of 70°C.
Acid treatment
MOSF were soaked in H2SO4 solution having varying concentrations of 2.5 wt%, 5 wt%, 10 wt% for a period of 60 mins. The liquor to fiber ratio was kept at 20:1.32,33 To remove the impurities and maintain a neutral pH level, 3–4 times washing of these fillers was carried out with distilled water. These washed fillers were then placed in a hot air oven for 2 days at a constant temperature of 70°C.
Preparation of PVA-MOSF composite films
MOSF-PVA composite films were fabricated by solution casting method as follows. A PVA solution was prepared by liquefying 8 g of PVA in 100 ml of distilled water and further 2 g MOSF, 1 g glycerol, 0.50 g citric acid, 0.025 glutaraldehyde and 0.03 g clay were suspended in the solution. The blend was steadily heated to 90°C with continuous stirring until a complete dispersion was established. The composite film developed without cross-linkers and plasticizers were rigid and ruptured during drying on the casting plates. Hence, cross-linkers and plasticizers were added into the solution to attain more flexible and workable composite films. During the heating process of the dispersion, evaporative loss may occur which can be compensated by addition of water. The solution was rested for some minutes and then ultra-sonicated for 20 mins to permit elimination of air bubbles added at the time of stirring. The dispersion was then spread on casting plates of dimension 140 mm × 140 mm × 5 mm. The plates were based on uniform surface and incubated for 48 h at room temperature. Later, the dried sample of film was peeled off from the casting surface. For different characterizations, different portions from each film at various positions (edges, center) were evaluated. Figure 1 demonstrates the preparation of MOSF-PVA composite films.
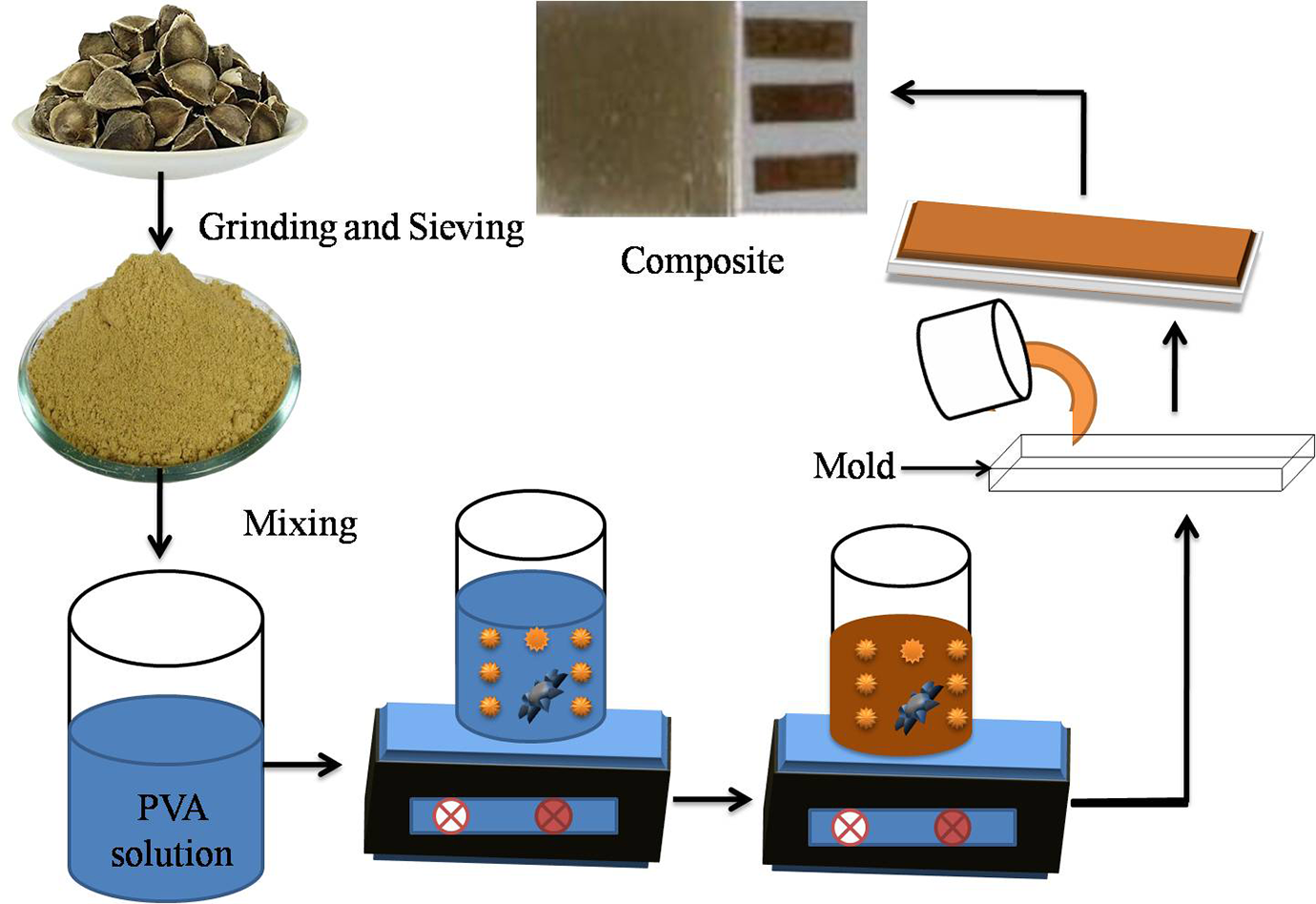
Process diagram illustrating the fabrication of MOSF-PVA composite film.
Characterization of MOSF and MOSF-PVA films
The compositional analysis of MOSF of the untreated and treated stages was detected through a standard protocol provided by the Technical Associated of Pulp and Paper Industry, considering the refinement reported by Silvério et al. 2013. 34 This method is built on consecutive removal and separation of different fraction of lignocellulosic material.
Lignin content
Lignin content was evaluated as described in ASTM E1721. 35 2 g of sample was taken in flask with addition of 15 ml of 72% H2SO4. The mixer was stirred at appropriate rpm for 2.5 h at 25°C thereafter further addition of 200 ml of distilled water into the mixer. Boiling of the mixture was done for 2 h and followed by cooling. In the next 24 h the lignin was separated and taken into the crucible with continuous hot water washing to make it acid free. Subsequently drying was carried out at 105°C and then the contents were put in a desiccator. It was weighed until it achieved constant weight
Holocellulose content
Holocellulose content was calculated as described in ASTM D1104. 36 3 g of oven dried samples were weighed and taken into a round neck flask with 160 ml of distilled water. This was mixed with 0.5 ml of glacial acetic acid and 1.5 g of NaCl. The water bath was used at 75°C for 1 h followed by the addition of 0.5 ml glacial acetic acid and 1.5 g NaCl in the flask. Further addition of acetic acid and NaCl were carried out two times in an hour. Ice bath was used to cool the contents of the flask below 10°C. The contents were filtered and washed with distinct liquids such as acetone, ethanol and H2O. At last weighing and drying was carried out.
α-Cellulose content
The final cellulose plus hemicellulose thus obtained was placed in a beaker with an addition of 10 ml of 17.5% NaOH. Stirring of filler was done through a glass rod. Once after every 5 mins, NaOH solution was added to the blend continuously for half an hour at 20°C. 33 ml distilled H2O was added in the beaker and stirred for 1 h. Filtration of the holocellulose residue was carried out and kept into the crucible. Successive washing was carried out with variable liquids such as 100 ml of NaOH (8.3%), 200 ml of H2O, 15 ml acetic acid (10%). Contents were dried and weighed.
Calculation of hemicellulose content was carried out through
Ash content
Preheating the silica crucible at 575°C for 4 h was carried out in a muffle furnace followed by desiccating it and cooling down for 2 hours. Weight of the crucible was noted and pre-dried 1 g of Moringa Oleifera seed filler sample was placed in a crucible and kept in for one day. Afterward, the final content in the crucible was weighed and ash content calculated through:
Fourier transform infrared spectroscopy of MOSF
In order to analyze the functional group, FTIR spectroscopy was used. FTIR analyses of untreated and treated fillers were performed with Nicolet 6700 series model. The powdered filler sample was mixed with potassium bromide in the ratio of 1:9 to form pellets. These pellets were then analyzed in the wavelength starting from 600 cm−1 to 400 cm−1.
X-Ray diffraction of MOSF
X-Ray Diffraction (XRD) was known to examine the crystal structure and crystallinity of the filler. Instrument used for the analysis was Bruker AXS D8 Diffractometer working on Cu-Kα radiation (λ = 1.5406 Å) with 40 kV and 30 mA. Analysis was carried out in the 2θ range of 0–50°C with goniometer speed of 0.02°/s. The crystallinity index (CrI) was calculated using the following formula:
CrI can be intended using intensity at primary peak (∼21.9° for cellulose) and because of amorphous peak intensity (evaluated at the minimum ∼18° between the primary peak and the minor peak at ∼16°).
Film thickness
Film thickness was evaluated using portable digital micrometer (TMI model no. 49-87, Delhi, India) with an accuracy of 0.0001 m. Measurements were performed on five random positions and the mean value of thickness was used to evaluate the different properties of the composite films such as water vapor permeability and mechanical properties.
Film density
To evaluate the film density, specimens of 1 × 1 cm2 were weighed after 24 days of keeping in a desiccator holding calcium sulfate desiccant. Densities of dry matter were evaluated through equation:
M is the dry mass of the film (g), A is the area of the film (1 cm2), δ is the thickness of the film (cm), ρs is the density of dry matter (g/cm2). 36 The density of film was determined as the average of three precisions.
Moisture absorption test
For water uptake test (ASTM D570), 37 specimen size was 10 mm × 10 mm. The square size film to be used for the test were kept in desiccator for 24 h. Thereafter, specimens were weighed initially then placed in petri dish filled with distilled water (pH = 6.5) and saline water (pH = 8) at 30°C. The weights of film were recorded periodically after gently wiping them with filter paper. Repetition of the process was carried out until a constant weight was achieved.
Calculation for % water uptake was carried out through 38
where w1 and w0 are weights after time t and initially respectively.
Mechanical properties
The mechanical properties of the MOSF-PVA films were evaluated with ASTM D882 39 and ASTM D790 40 in Instron model 5565 UTM (Universal Testing Machine). The films were cut into rectangular strips of 60 × 6.35 mm for tensile test and 100 × 25 mm for flexural test in dimension with head speed of 0.8 mm/min and initial grip separation of 20 mm. Properties like tensile strength, tensile modulus, flexural strength, flexural modulus were determined. The samples were prepared in triplets and runs were taken to attain dependable facts.
Morphological properties
The morphological properties of filler and MOSF-PVA films were determined by SEM (Scanning Electron Microscopy) and AFM (Atomic Force Microscopy). SEM (model: LEO 435 VP) having magnification of 10x–300kx was used to discover the surface features of composite film. AFM was used to have a quantitative approach to know the surface morphology and roughness of the MOSF. Instrument used for the analysis was NT-MDT (model: NTEGRA Prima) working on the tapping mode which consisted of silicon nitride cantilever with scan rate and area of 0.55 Hz and 20 × 20 µm respectively.
Water vapor permeability (WVP) test
Gravimetric determination is used to measure the water vapor transmittance rate of composite films. According to standard test method (ASTM E96-95) WVP was tested. 41 Briefly, films without bubbles or deformity were cut into sizes a bit larger to the glass cup diameter. All the cups were fully filled with CuSO4 desiccant. Underside of the film covered the opening of the glass cup, so that the support portion of the film experienced low RH (relative humidity). The overall system was positioned in a desiccator carrying NaCl. During first 9 h of testing, the cups were weighed periodically after every 1 h. Later, the weights of the cups were recorded in 24 h interval for 3 days. Differences in the weights of the cups were noted and time plots of each sample were measured through the slope of the plots. All samples were measured three times and mean value was taken.
where, ΔW/ΔT is weight gain per unit time (g/s), A is exposed area of the film (m2), X is average thickness of the film (m), ΔP is the difference in the water vapor pressure of 75% RH(relative humidity) of desiccator (NaCl) and 0% RH(relative humidity) of CuSO4
Biodegradability test
Laboratory scale soil burial test was performed to investigate biodegradability of composite film, using the methodology of Laxmeshwar et al. 42 and Behjat et al. 43 Initially, weight of the vacuum dry sample was 0.55 g (WI). Randomly, the vacuum dried samples were buried in the tray containing moisturized soil (taken from the ground of IIT Roorkee campus). The tray carrying the sample and soil was incubated at constant temperature of 23°C for 3 months. Moisture of the tray was maintained through constant spraying of water at regular intervals. This helped in dynamic growth of microorganism to decompose the specimens. Plastic films were used to cover the tray as it cooperated in reducing the evaporation from the soil. The degradation of specimen was noted by removing the specimen from soil after every 15 days of interval for 3 months. After that, continuous and careful washing of the sample with water was carried out to separate the film from debris or soil to confirm that the degradation stopped. The specimens were dried under vacuum till constant weight is achieved (WT). therefore, the % degradation was calculated using equation:44–46
Results and discussion
Physical and compositional characteristics of MOSF
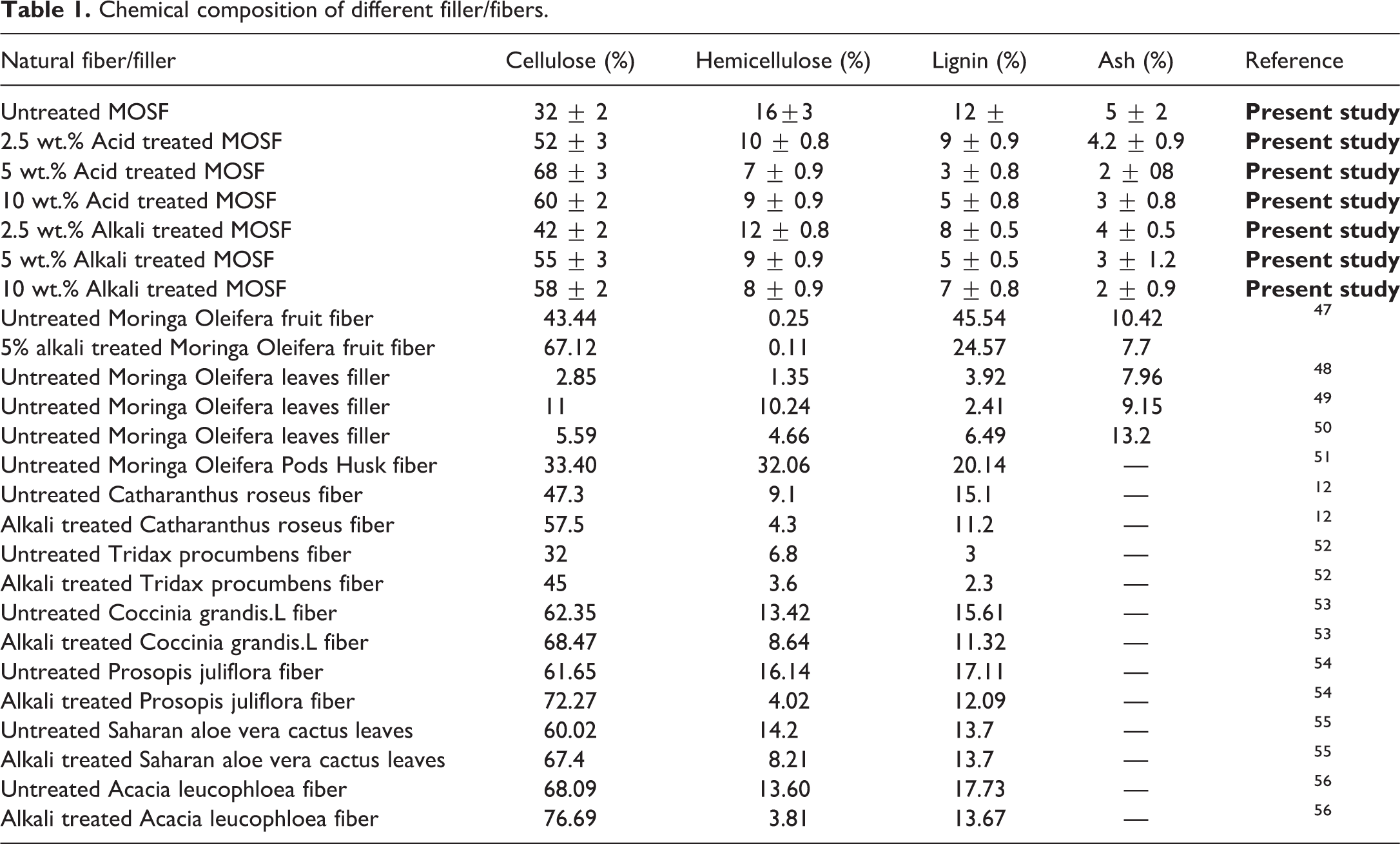
To understand the chemical examination of the raw and treated filler, confirmation of lignin, ash and holocellulose (cellulose + hemicellulose) content is important. The comparative results of the present study are shown in Table 1. In the present study, the raw filler consists of 32% ± 2 cellulose, 16% ± 3 hemicellulose, 12% ± 2 lignin and 5% ± 2 ash content. The ash content is comparable with results attained by Seifi and Alimardani. 57 The lignin and holocellulose content of MOSF till now has not been reported in literature. Chemical modification of the filler was carried out to enhance the adhesion of filler-matrix. This results in composites with better mechanical strength due to effective stress transfer between the filler and the polymer matrix.58–60 Table 1 shows that after the chemical modification, the MOSF gained a large amount of mass as impurities, lignin, waxy and hemicellulose contents are removed. This brings the information of overall increment of cellulose content in fillers. Therefore, this suggests that higher the cellulose content, more is the crystalline part in the filler which ultimately helps in forming a better film material. 61 Chemical modification helps in smoothening the filler surface by removing the impurities, waxy components, lignin and hemicelluloses. All concentrations of acid and alkali treatments remove the non-lignocellulosic content with lignin and hemicellulose, but 5% w/v acid and alkali concentrations removed the maximum amount of these. At 10% w/v chemical treatment, cellulose content is decreased possibly because of destruction created by higher concentration of acid and alkali.
Chemical composition of different filler/fibers.
The average thickness of the MOSF-PVA film was from 0.03 mm to 0.04 mm. The neat PVA film thickness was found to be 0.12 mm, which is 3–5 times thicker than the composite film. The reason behind the more thickness of the PVA film could be its hydrophilic nature, which binds water molecules and absorb more water. Contrarily, MOSF-PVA film is thinner because the addition of filler (MOSF) in the matrix reduced the water absorption tendency of the PVA. Moreover, increasing the filler content did not affect the thickness of the film significantly. This observation is similar with the research of Müller et al., 62 which says that increment in the content will not affect the thickness considerably.
Another major physical property is the density of film. MOSF-PVA film density was 2.50 g/cm3 whereas, neat PVA film is 3–4 times less dense i.e. 0.69 g/cm3. This result was due to the addition of filler, plasticizer, cross-linkers in the neat PVA which could increase the water resistive nature of the film.
Surface morphology of MOSF and MOSF-PVA composite film
SEM analysis of MOSF: SEM analysis of the raw and treated MOSF was achieved to determine the surface morphology and characteristics. Figure 2, displays the SEM micrographs of raw filler and chemically treated filler. Figure 2(a) indicates that the raw filler surface appears to be uniform and smooth. The reason behind this could be the excessive waxy, lignin contents present in the filler. The acid treated filler surface (Figure 2(b)) appears to be rough due to partial or uneven removal of waxy, lignin and hemicellulose content. This strengthens the properties of filler and thus benefits in better adhesion of filler-polymer matrix as it exchanges the stress load between them thereby enhance the bonding property.63–65 The alkali treated filler surface (Figure 2(c)) brings out a cylindrical formation of cellulose nanocrystals. The reason behind this could be the over elimination of lignin and holocellulose content i.e. deterioration of exposed celluloses caused by the reaction of high temperature and concentration of alkali. Similar results are observed by the literature66–68 but in acid hydrolysis.

SEM images (a) untreated MOSF, (b) acid treated MOSF, (c) alkali treated MOSF.
SEM analysis of MOSF-PVA film: Figure 3 indicates the SEM images of MOSF-PVA composite films. The neat PVA film (Figure 3(a)) appears to be smooth and regular with no pores or splits, proving its good structural unification, analogous with the result of Wahyuningsih et al. 69 The addition of untreated filler with PVA matrix is shown in Figure 3(b). The fillers are detectably evident on the surface of the PVA showing a poor bonding properties. The cause trailing the poor bonding is the hydrophobic attribute of filler which causes the pull-outs and cracks. 51 Comparatively, the 5% alkali treated composite film (Figure 3(c)) shows continuous assessment of filler which demonstrate excellent interfacial adhesion between MOSF and PVA matrix. Similarly, the 5% acid treated composite film (Figure 3(c)) is showing a homogeneous distribution with better filler-matrix bonding. The compact structure with good evident structural unification leads to allowable mechanical strength.

SEM images (a) neat PVA sheet, (b) untreated MOSF with PVA matrix, (c) 5% Alkali treated MOSF with PVA matrix, (d) 5% Acid treated MOSF with PVA matrix.
AFM of PVA and MOSF-PVA film: Figure 4((a), (b) and (c), (d)) indicates the 2D and 3D images of neat PVA, MOSF-PVA, chemically treated MOSF-PVA films. The neat PVA film shows a uniform and smooth surface with the average roughness of 23.4233 nm. When the MOSF is added to the PVA matrix, the surface of the composite film grows rougher with the average roughness of 40.3499 nm. Comparatively, the 5% alkali and acid treated composite films shows comparably rougher surface with the average roughness of 172.6233 nm and 183.4653 nm, respectively. This shows that MOSF is thoroughly mixed at the molecular extent and ultimately enhances the interaction between the MOSF-PVA matrix. Roughness of the surface improves the adhesion between the matrix and filler as it interchanges the stress load which leads to better interlocking and thus enhancing the bonding properties.

2D and 3D AFM images (a) neat PVA sheet, (b) untreated MOSF with PVA matrix, (c) 5% alkali treated MOSF with PVA matrix, (d) 5% acid treated MOSF with PVA matrix.
Mechanical properties of MOSF-PVA composite film
Figure 5((a)–(d)) represents the average value of mechanical strength and modulus of the untreated and chemically treated (acid and alkali) MOSF-PVA films respectively. Certainly, Figure 5((a) and (b)), gives a clear picture showing that values of tensile strength and modulus of 5% acid and alkali treated MOSF-PVA film is maximum i.e. ∼12% increment with comparison to neat PVA film. This enhancement is due to the improved contact angle of filler with the matrix. 70 Truly, by removing the impurities, waxy contents from the filler helps in creating rougher surface, improved interlocking and hence interface character improves. 71 On the other hand, removal of lignin and hemicellulose content also helps in increasing efficiency of cellulose content. Acidification helps in improving the dispersion of filler in matrix, which increases the aspect ratio. This enhances the effectiveness of filler as reinforcement material and thus increases the strength of the film. 72
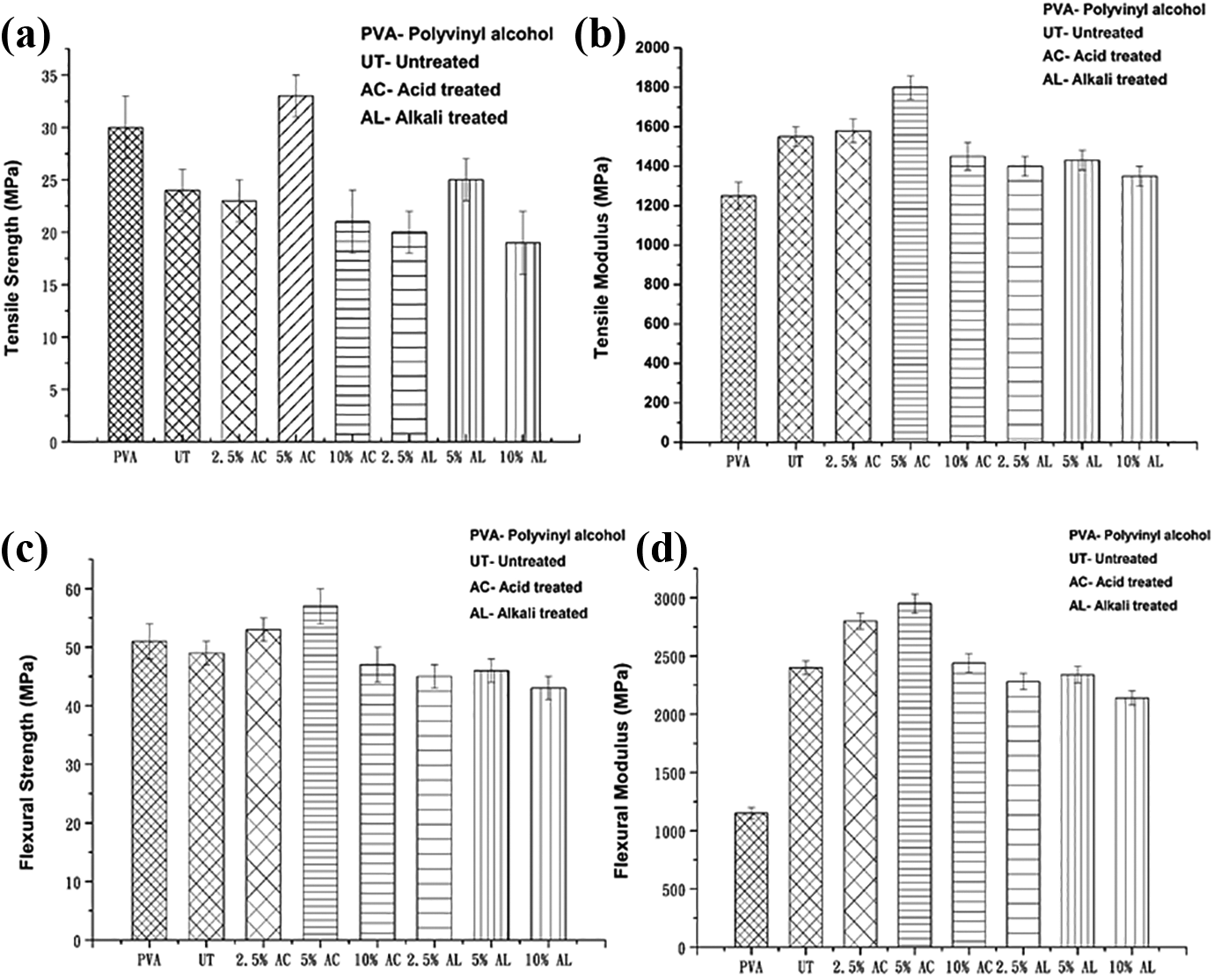
((a) and (b)) Tensile strength and modulus of acid and alkali treated PVA films. ((c) and (d)) Flexural strength and modulus of acid and alkali treated PVA films.
Increasing the acid concentration is supposed to have an increment in the tensile strength and tensile modulus of the film, but here it shows that 10% acid treated composite film doesn’t exhibit any excessive increment in the value as compared to neat PVA film and untreated film. This behavior indicates that high concentration of acid can damage the structural integrity of the filler which leads to decrease in the strength of the composite. As described in the report of Mwaikambo and Ansell, 73 higher concentrations of chemical treatment would eventually damage the filler and accordingly decrease the mechanical properties of the composite. Similar trends are observed in the tensile modulus graph. Alkali treated film has no significant increment in tensile strength and tensile modulus when compared to untreated films. This may be because of higher concentration of alkali demolishes the crystalline affiliation of cellulose and reduces the cellulose chain length. This refers to reduction in mechanical properties of filler because of its increased amorphous nature. 74
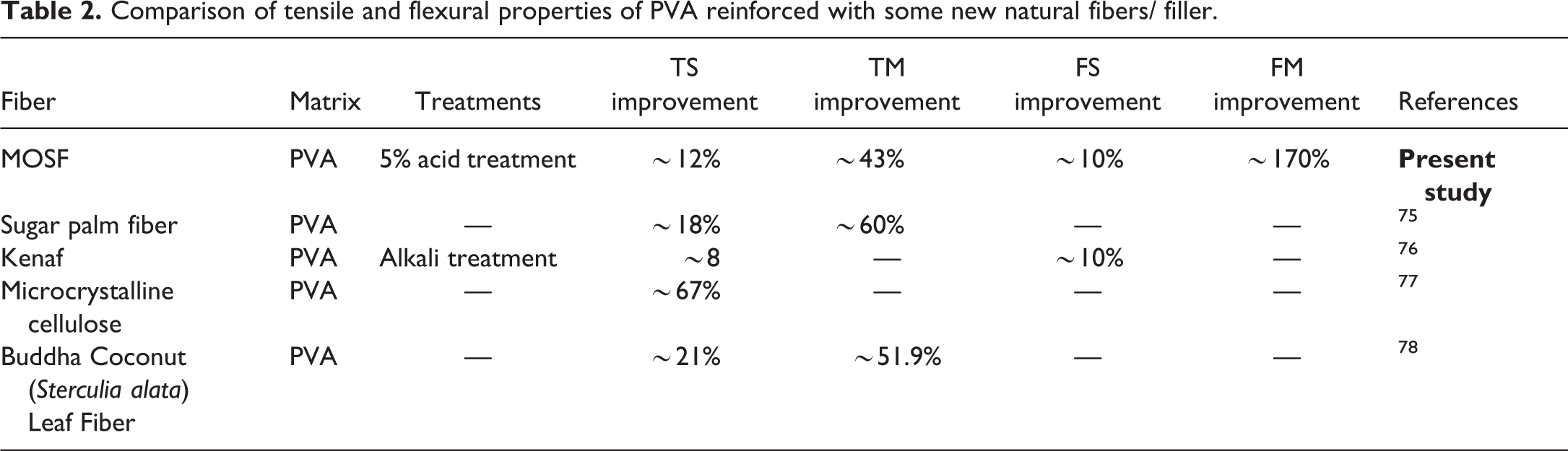
Similar effects are detected in flexural strength and modulus of untreated and chemically treated MOSF-PVA film. From Figure 5((c) and (d)), it is certain that improvement in both flexural strength and modulus up to 5% chemical treatment occurs, beyond that degradation in mechanical properties takes place. More than 10% increment in flexural strength and 170% increment in flexural modulus occurs as compared to neat PVA film. Flexural moduli of all the films are higher contrary to neat PVA film. This reveals that filler increases the stiffness of the film. A comparative result of this study with different studies reported in literature of PVA matrix reinforced with natural fiber is shown in Table 2.
Comparison of tensile and flexural properties of PVA reinforced with some new natural fibers/ filler.
Moisture absorption test of MOSF-PVA film
One of the substantial disadvantages of using a PVA matrix material is its tendency to absorb water and any rectification in the property is extremely important. So, moisture absorption rate is an important factor to check the films as for packaging material. The test is carried out by immersing the films into two distinct types of water states, which are saline water (pH = 8) and distilled water (pH = 6.5). To check applicability of these composites, two distinct types of water state are provided which indicates real-life circumstances. Moisture absorption curve with varying filler treatments immersed in distilled water and saline water during 192 his shown in Figure 6((a) and (b)). Figure 6(a), displays the % weight gain of film build using alkali and acid treated filler with PVA in distilled water and Figure 6(b) in saline water environment. The graph for both alkali and acid treated composite film shows 50% to more than 300% weight gain. 10% alkali and acid treated film in both the water states resist the maximum moisture absorption and exhibit almost constant gain in weight after the first 24 h. 2.5% alkali and acid treated film in distilled water as well as in saline water shows the highest weight gain and continue to rise for next 6 days prior to achieve constant weight. The reason behind this could be the network arrangement established by incorporating plasticizers, cross-linkers and clay with PVA, which block the H2O (water) molecules from dissolving and enhancing the resistive nature for the moisture absorption. Blend films with MOSF, cross-linkers, plasticizers acquire less moisture than neat PVA film. Strong hydrogen bonding reduce the free OH groups of cellulose, thus generate a decrement in moisture absorption of the film. Moreover, increase RH (relative humidity), increases the moisture absorption. PVA and other contents are hydrophilic with sufficient amount of OH groups. Therefore, in high humidity, water sensitivity will increase leading to higher moisture absorption. 79
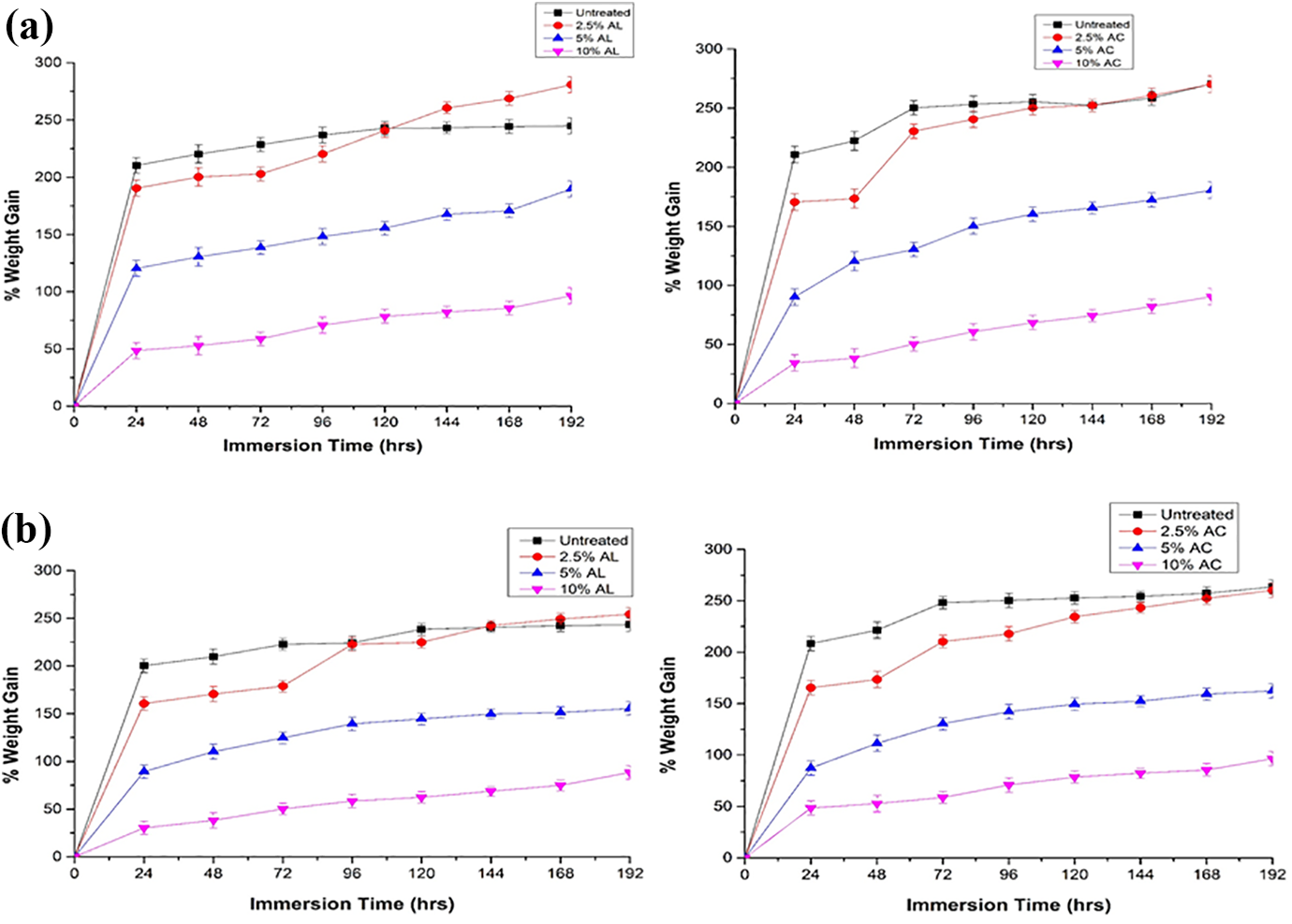
(a) % weight gain of alkali and acid treated MOSF with PVA in distilled water, (b) % weight gain of alkali and acid treated MOSF with PVA in saline water.
Figure 6((a) and (b)) shows that moisture absorption of MOSF-PVA composite immersed in distilled water is higher than in saline water. This is because of salt molecules which lower the action of water molecules and results in accession of salts particles on the surface of sheet which could further obstruct the water uptake. 80
X-Ray diffraction of MOSF
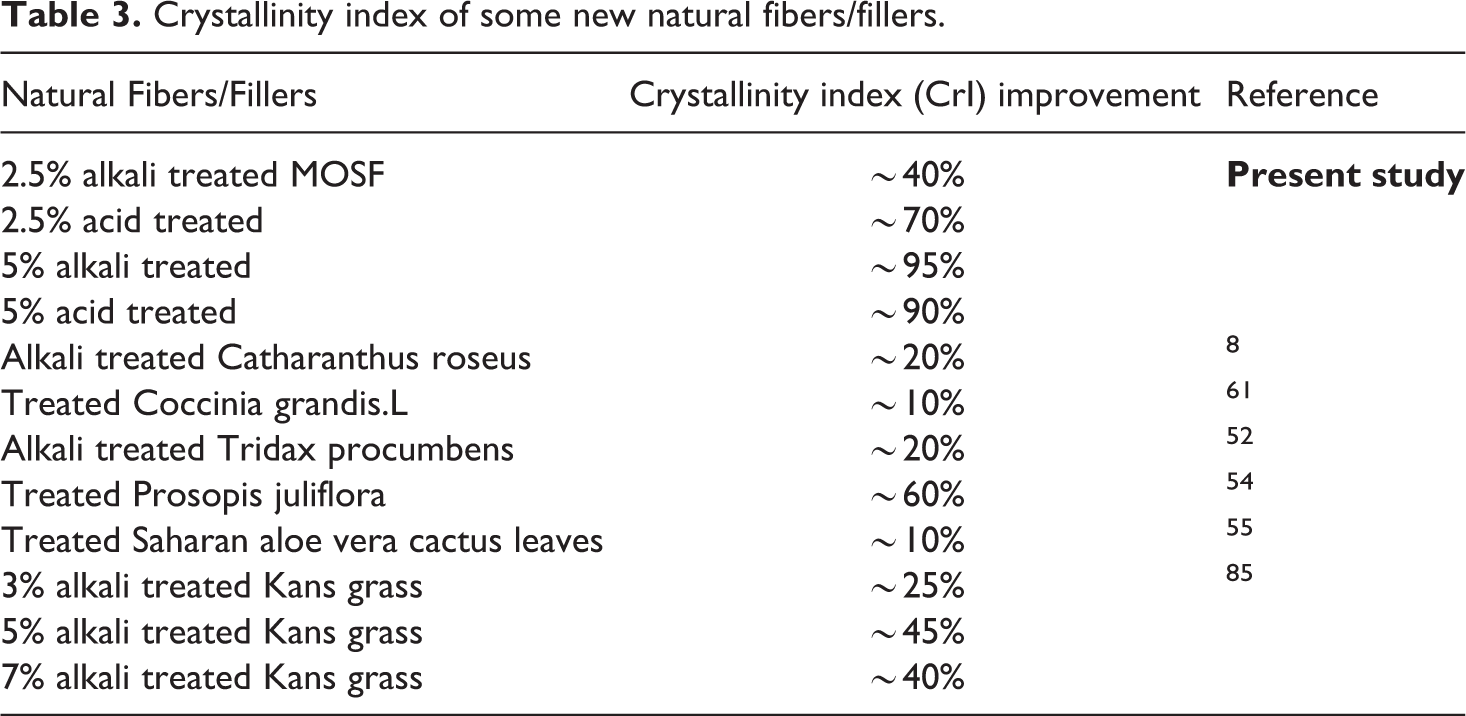
Figure 7(a) exhibits the pattern of alkali treated filler with untreated filler. Figure 7(b) shows x-ray diffraction (XRD) pattern of acid treated filler with untreated filler. The peaks of untreated, acid and alkali treated are visible at 2Ɵ 21.9° indicating the presence of cellulose. The crystallinity index (CrI) of untreated filler has been evaluated using equation 3 that comes out to be 36.85% which is lower than the untreated Coccinia grandis.L (52.17%), 57 untreated Prosopis juliflora (46%) 58 but higher than that of untreated Tridax procumbens (34.46%), 56 untreated Catharanthus roseus (25.09%). 12 The CrI of 2.5% alkali and acid treated are 53% and 66% respectively. This increment is due to starting removal of amorphous lignin and hemicellulose content from the filler. 81 5% alkali and acid treated filler is exhibiting highest CrI, i.e. 76% and 72% respectively, possibly as a consequence of complete reaction of the filler with the acid which results in diminishing all the impurities, waxy, amorphous content. Also, redistribution of the crystalline portion occurs. No more enhancements in CrI of 10% alkali and acid treated filler is observed due to extreme delignification of filler which causes deterioration of the filler surface. Analogous improvement in CrI of other natural fibers like Acacia leucophloea, 60 Juncus effuses L., 82 Saharan Aloevera, 59 etc. has also been detected in literature.83,84 A comparative result of this study with different studies reported in literature of various natural fibers is shown in Table 3.
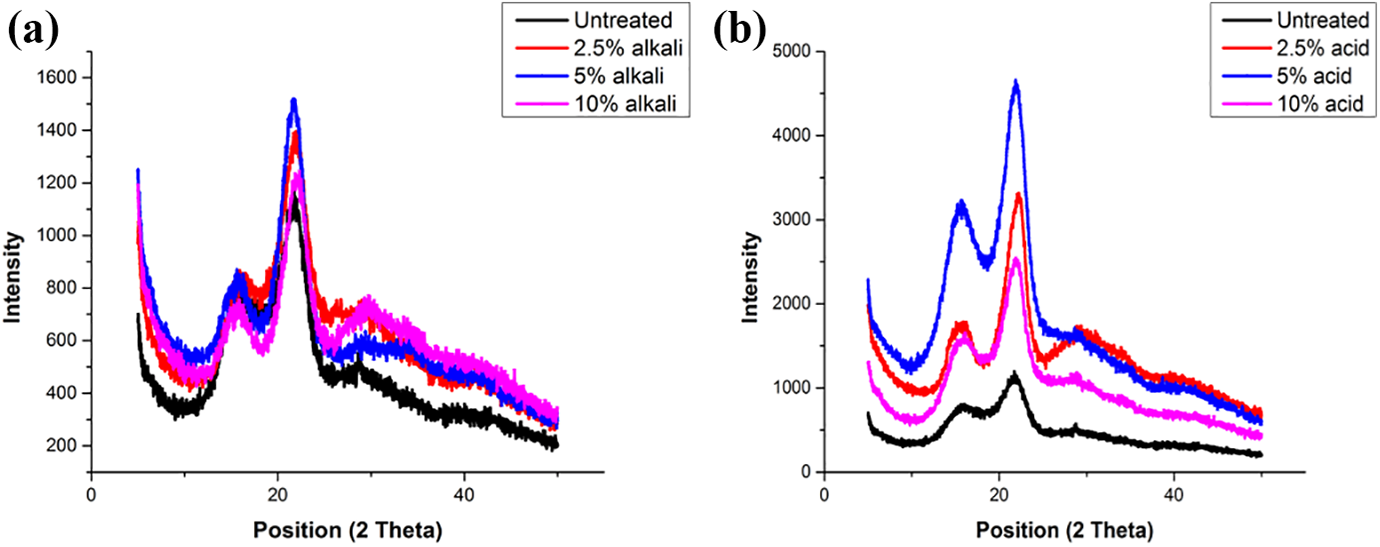
XRD spectra of (a) untreated and alkali treated MOSF, (b) untreated and acid treated MOSF.
Crystallinity index of some new natural fibers/fillers.
Fourier transform infrared spectroscopy of MOSF
Figure 8((a) and (b)) exhibits the FTIR spectra of untreated and treated MOSF that indicates the existence of multiple functional groups showing the complex behavior of the material. For untreated MOSF, the absorption bandwidth unifies at 3377.59 cm−1 possibly assigning a hydroxyl group and/or N-H stretching of the secondary amide bond formed due to the existence of proteins, fatty acids, cellulose or lignin content in the filler. 86 The absorption peak at 2926.50 cm−1 has a close similarity with the symmetric and asymmetric stretch of C-H and C-H2 bonds present due to the fatty acid content in seeds. It also tells about the presence of hemicellulose and cellulose content in the seeds. 87 Peaks at 1770.43 cm−1, 1426.07 cm−1 are observed because of C-N stretch of amide and N-H stretching present due to the existence of protein in the seed. 86
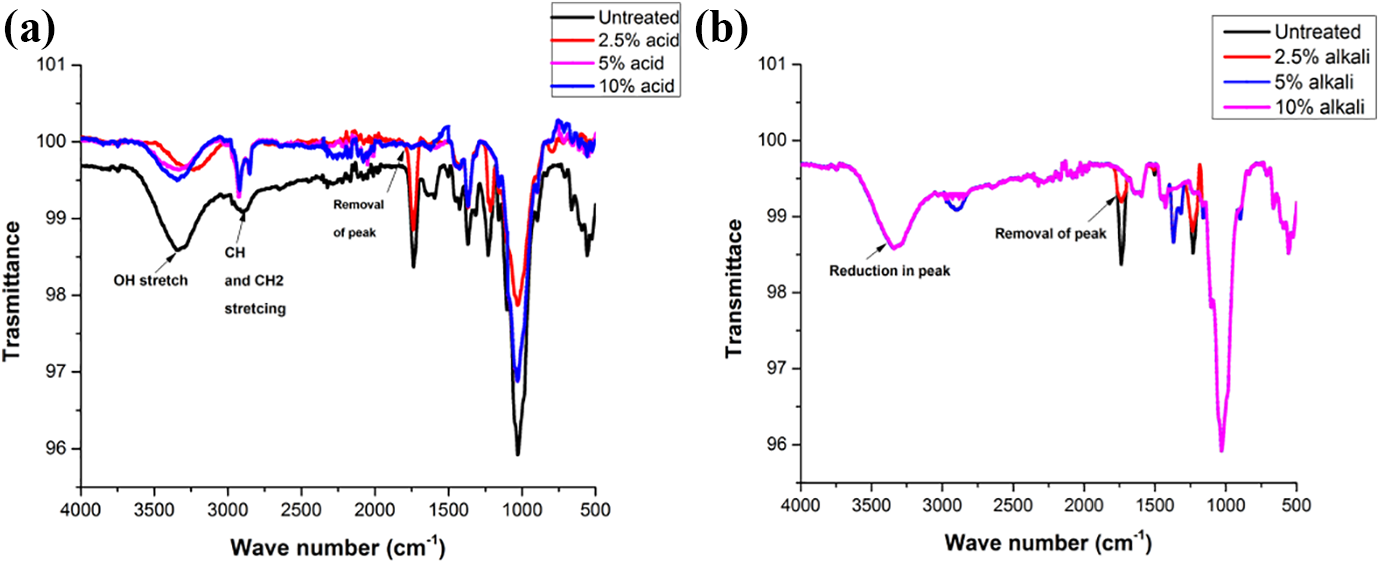
FTIR spectra of (a) untreated and acid treated MOSF, (b) untreated and alkali treated MOSF.
The spectrum for 5% acid treated filler is close to the untreated filler except there is removal of peak at 3377.59 cm−1. Similar peak is observed at 2926.42 cm−1 but with a higher intensity due to the predominant effect of lipid constituents present in the seeds, which is in similar proportion as of proteins. 87 In case of alkali treated filler there is a complete removal of peak at 1770.43 cm−1, 1426.45 cm−1 which reveals the complete removal of the impurities, lignin and hemicelluloses content from the filler. Rest of the peaks attain a similar pattern.
Water vapor permeability test of MOSF-PVA film
One of the crucial purposes of food packaging material is to reduce transfer of moisture between the enclosing environment and the food. Therefore, low WVP is preferable to optimize the environment of food packaging and possibly enhance the serviceable life of the food product.
Figure 9 exhibit the WVP of various composite films formed through untreated, treated MOSF and PVA. WVP of neat PVA film is 1.82 × 10−10 gs−1m−1Pa−1. 2.5% acid treated film doesn’t significantly affect then WVP whereas 5% acid treated film shows the least WVP of 1.42 × 10−10gs−1m−1Pa−1. This could be due to the fact that the molecular interaction of MOSF-PVA helps in preventing the diffusion of the free water molecules diffusivity through the composite film. 10% acid treated film increased the permeability value. This is due to the free hydroxyl group present on the surface of the film and its enhanced interaction with the water molecules present in the environment, thereby increasing the water vapor transmission. Moreover, the other factors could be the presence of no of polar groups in the matrix, rate of diffusivity and solubility of water molecules in the film.88,89
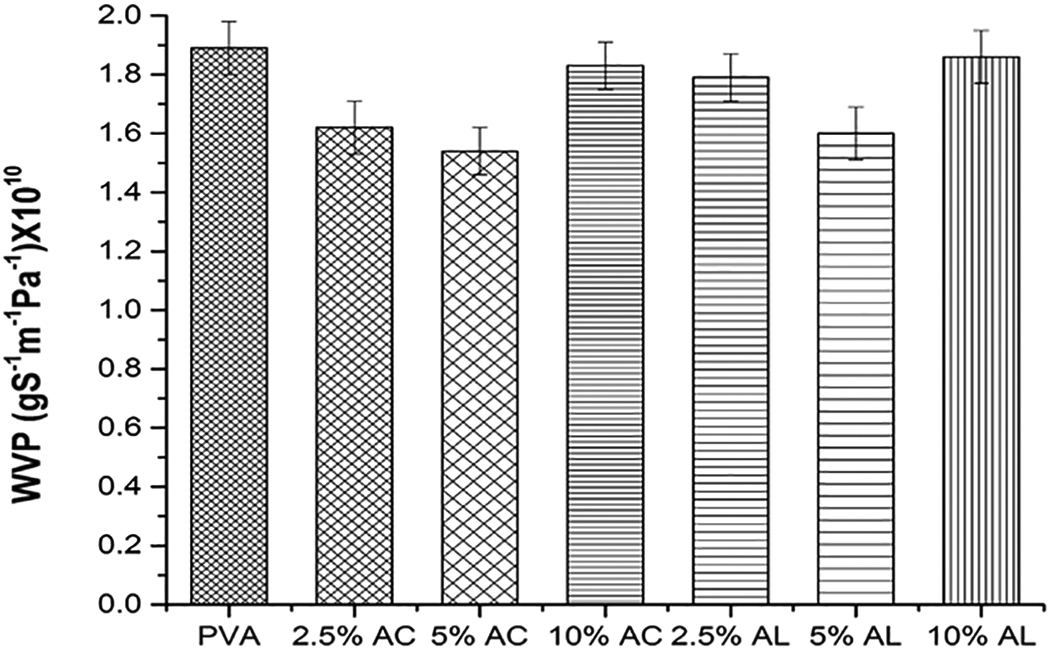
Water vapor permeability of composite films.
Similar results are reported by Limpan et al, 90 who found that increasing the concentration of chemical treatment, decrease the WVP. Bonilla et al. 91 reported the WVP value of chitosan/PVA composite film between 6.11 and 19 × 10−10gs−1m−1Pa−1which is a wide range and relatively higher than the present study. The WVP value of PVA reported in present study is higher than many synthetic polymers i.e. 0.0126 × 10−10gs−1m−1Pa−1for PVC, 0.0091 × 10−10gs−1m−1Pa−1for LDPE and 0.002 × 10−10gs−1m−1Pa−1for HDPE. 92 After all, the WVP value for MOSF-PVA film is somewhat higher than the work reported by Tajik et al. 85 Therefore, the MOSF-PVA film is suitable for some food packaging applications. Alkali treated film shows the similar trend with less significant results as compared to the acid treated film.
Biodegradability test
Figure 10((a) and (b)) shows the effect of chemical treatment on the degradation of MOSF-PVA films. The neat PVA film doesn’t lose weight in starting 60 days of soil burial test. Contrarily, 5% alkali and acid treated film immediately loses weight in starting 15–30 days of burial, recording the highest weight loss percentage to be more than 26%, after which there is a constant increment throughout the burial period. Therefore, biodegradation of MOSF-PVA film is much faster than that of neat PVA film. For both the 2.5% and 10% acid treated film, the degradation activity is significant throughout the 3 month. Initially, 30 days of burial, 2.5% acid treated film loses 5–7% weight where 10% acid treated film loses 2–5% weight. They maintain losing weight to get ∼13% and 7% weight loss respectively, and then they continued to be constant for 90 days. Surprisingly, 2.5% and 10% alkali treated film doesn’t lose any weight for 30 days. This could be due to the concentration of alkali deteriorates crystallinity of cellulose content in the filler resulting in forming a shorter chain length. This may have contributes to the lesser degradation of film in the soil. 93
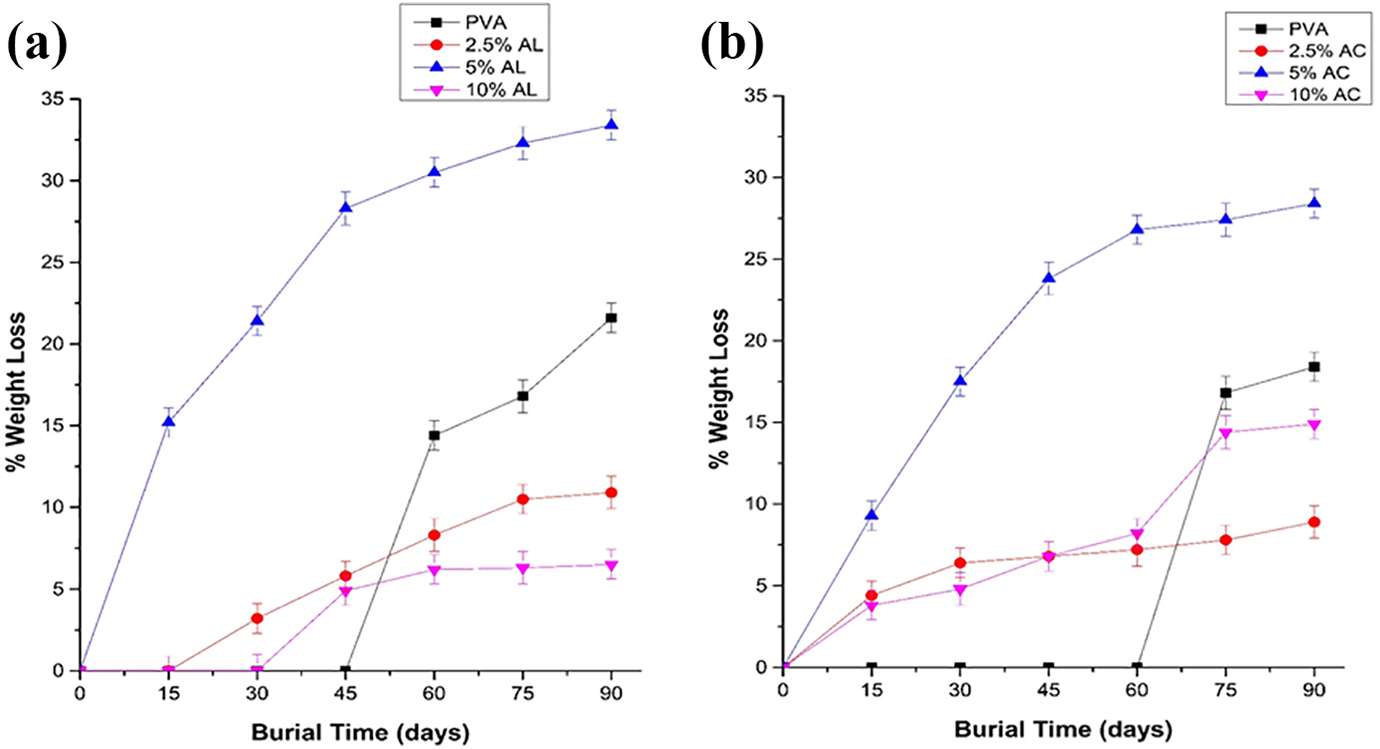
Biodegradability test (a) acid treated films, (b) alkali treated films.
The factors mainly affecting the weight loss are composition of mixture, various physical, chemical and biological criteria consisting of different microorganism and then species present, physical parameters such as pH, temp, oxygen, nutrients for degradation action, soil characteristics (i.e. particle size distribution), contaminants quality, quantity and its bioavailability.94-96 Therefore, the results advocate the suitability of MOSF-PVA film as it has improved the biodegradability in comparison to neat PVA.
Conclusion
This is the first study that reveals the possibility of forming biodegradable composite film through Moringa Oleifera seed filler (MOSF) and polyvinyl alcohol (PVA) via solution casting method. MOSF could be an encouraging raw substance for the development of biodegradable composite film and coatings. As confirmed by FTIR, SEM and AFM data, MOSF has superior or similar functional and morphological properties when compared to the other natural fillers used these days. The mechanical and moisture absorption property of 5% acid treated film was much better than those untreated MOSF/PVA and neat PVA films. Increasing the concentration of chemical treatments discloses the abatement in mechanical and water permeable properties. XRD analysis reveals that MOSF and PVA are suitable and chemical treatment increases the crystallinity of MOSF/PVA blends. The results initiated in this study evidently specify that there is an utmost necessity of surface modification to achieve biodegradable composite film. The chemically treated film also showed a superior biodegradability in comparison to neat PVA film. It can be concluded that acid treatment in the ideal surface methodology to upgrade the overall characteristics of the film that can have an appropriate use in several parts of science and environment-friendly packaging applications.