Abstract
A solvent thermal method which depends on a thermal process under critical temperature and pressure was used to prepare cobalt iron oxide magnetic nanoparticles with a molar ratio 2. The prepared particles were in the form of nanoparticles with diameter ranging from 5 to 10 nm and with amorphous structure. Magnetic hysteresis behavior with saturation magnetization 36.31 emu/g and coercivity 4 Oe were observed for the nanoparticles. Polyvinyl alcohol was loaded with different concentrations of cobalt iron oxide nanoparticles using casting technique. Hysteresis loops for the polymer films were observed and both the saturation magnetization and coercivity were increased from 0.36 to 16.03 emu/g and 115 to 293 Oe for samples containing 5 and 20 wt% of nanoparticles, respectively. The elastic modulus of films was increased from 2.7 to 4.9 GPa for unloaded and loaded samples with 20 wt%, respectively. The storage modulus of the polymer films was found to obey the percolation behavior.
Keywords
Introduction
Magnetic nanocomposite materials are considered as one of the sources of the discovery of spectacular new phenomena, with potential applications in the different fields. Magnetic nanoparticles as one of a broad spectrum of nano-scale materials have gained remarkable importance due to their intrinsic properties. Magnetic nanoparticles are successful as anticancer materials, drug delivery agents, magnetically recoverable catalysts, magnetic resonance imaging devices, removal of oil spills from water, etc. 1 Magnetic nanoparticles are metallic, bimetallic nanoparticles, metal oxides, ferrites, or super-paramagnetic iron oxide nanoparticles.1–3 Spinel cobalt ferrite, CoFe2O4 is one of the most promising magnetic materials in terms of high coercivity, moderate saturation magnetization, excellent chemical stability, mechanical hardness and large magneto-crystalline-anisotropy.4,5
Magnetic nanoparticles have been widely used in the synthesis of polymeric nanocomposites, which used in various applications such as heterogeneous catalysts, sensors, magnetic resonance imaging, decontaminating agents for wastewater, environmental remediation, data storage and optical filters.6–13
Polyvinyl alcohol (PVA) with an excellent chemical resistance which extensively used for biomedical applications is semi-crystalline, non-toxic, biocompatible, biodegradable polymer.5,14–17 Consequently, the loading of PVA with magnetic nanoparticles, especially CoFe2O4, have been studied earlier and the influence of size and shape of the magnetic nanoparticles on the magnetic and mechanical properties of such nanocomposites have been investigated.5,18–21 Rashidi and Ataie 22 synthesized a spherical single-phase 20 ± 4 nm mean particle size of CoFe2O4 via the mechanical alloying method and then embedded it inside the PVA polymer matrix by intensive milling. The maximum saturation magnetization and coercivity of prepared CoFe2O4 were 52.2 emu/g and 832 Oe, respectively. These values were decreased to 37.8 emu/g and maximum coercivity 10.3 Oe for PVA loaded with 80.0 wt% CoFe2O4. A sonochemical method 23 was used to synthesize Fe3O4 and obtained spherical particles with a diameter of about 100 nm with a saturation magnetization of 66 emu/g and coercivity of 39 Oe. The addition of 25 wt% of this filler to the PVA polymer matrix reduced the saturation magnetization to 9.8 emu/g while increased the coercivity to 105 Oe. Jayakrishnan and Ramesan 24 incorporated Fe3O4 inside polyvinyl alcohol and polyacrylic acid (PAA) blend and found saturation magnetization 0.0996 emu/g and coercivity of 260.59 Oe for samples containing 20 wt% from the filler. Both the tensile strength and hardness (shore A) were increased from 20.24 MPa and 34 for neat PVA-PAA to 25.07 MPa and 43 for PVA-PAA loaded with 20 wt% Fe3O4, respectively. Preparation and characterization of CoFe2O4/PVA nanocomposites by various methods have been reported in recent years.20,25–29
In the present work, magnetic nanoparticles from cobalt iron oxide with a molar ratio 2 were prepared and characterized using XRD, TEM and VSM. Cobalt iron oxide magnetic nanoparticles were then embedded with different concentrations inside PVA host using casting technique. Magnetic hysteresis of PVA loaded magnetic nanoparticles films was studied. Static and dynamic mechanical properties of such magnetic polymeric films were also studied and correlated with the magnetic properties.
Experimental
Materials
Iron (II) acetate 99.99% trace metals basis, cobalt (II) acetate 99.995 trace metals basis, methanol, ethanol and Polyvinyl alcohol (PVA) with molecular weight 89,000 and 99+% hydrolyzed were supplied by Sigma-Aldrich, England.
Samples preparation
Preparation of cobalt iron oxide nanocomposite
A solvent thermal method which depends on the thermal process under critical temperature and pressure was used to prepare cobalt iron oxide with a molar ratio 2 of Co/Fe. Schematic presentation of the setup is shown in Figure 1. An amount equal 0.077 mol of iron acetate and cobalt acetate were reacted with an excess amount of methanol by vigorous stirring for a long time. After completing the reaction, 400 mL of ethanol was added to the mixture. Then, it was placed in a pressurized vessel as shown in Figure 1. The decomposition process for the contents of the mixture was carried out under supercritical conditions (temperature 250°C, 1°C/min and pressure 70 bar). The decomposition process was achieved directly with gel embedded with the solvent. The heating of the mixture inside the autoclave was slowly increased to a temperature to be higher than the critical temperature. The pressure follows the rise of temperature and reaches a value higher than the critical pressure. At this moment, the pressure releases slowly from autoclave with fluxing with nitrogen gas to prevent further oxidation for the product and to avoid liquid condensation within the pores of the powder. During the reduction of pressure, the autoclave was cooled down to room temperature. Finally, the powder was easily collected. For comparison, iron oxide without cobalt was prepared by the same technique.
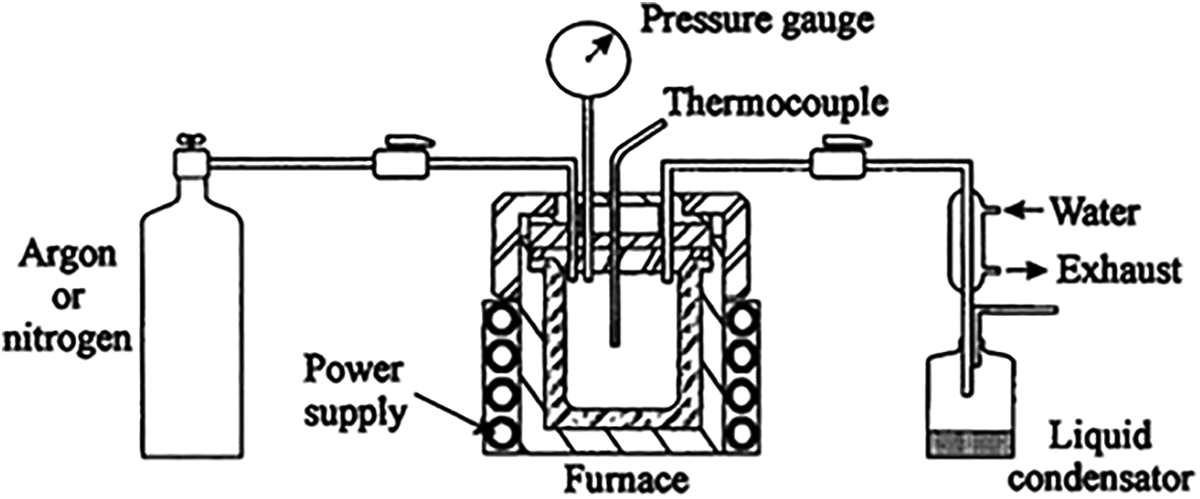
A schematic illustration of the setup used for the preparation process.
Preparation of PVA cobalt iron oxide nanocomposites
Initially, the desired amount of PVA was dissolved in hot distilled water at 90°C on a magnetic stirrer for 2 hours. The desired amount of previously prepared Co/Fe-2 nanoparticles was dispersed in distilled water and ultrasonicated with a sonication amplitude 60 μm 30 for 30 min followed by magnetic stirring for further 24 h to get Co/Fe-2 suspended solutions. The two solutions were then mixed and ultrasonicated with same sonication amplitude for 30 min and stirred for 24 hr to ensure good dispersion of the Co/Fe-2 nanoparticles. The solvent (distilled water) was evaporated at room temperature and film samples were prepared. Samples with different Co/Fe-2 concentrations (0.0, 1.0, 2.5, 5.0, 10 and 20 wt%) loaded PVA were prepared and kept inside vacuumed dissector. The film thickness (0.1 mm) was measured using a digital micrometer (MOTOTOYO, China) with 0.001 mm resolution.
Characterizations
XRD and TEM
X-ray diffraction (XRD) allowed us to determine the structural and crystal-study of the prepared materials through a Rigaku RINT 2200. Energy-dispersive X-ray spectroscopy (EDX) was exploring the composition of the prepared materials through electron probe micro analyzer JED 2300.
High resolution transmission electron microscope (HR-TEM, Tecnai G20, FEI, the Netherlands) was used for imaging purposes.
Magnetic properties
Magnetic properties were measured using a Vibrating Sample Magnetometer (VSM) 7400-S series, Lake Shore Cryotronics, Advicing Science, USA. Iron oxide samples and cobalt iron oxide samples were used as powders, while Co/Fe-2-PVA samples were in the form of films. The magnetic parameters (saturation magnetization, remanent magnetization, coercivity, and remanence) were determined from the shape of magnetization curves and the hysteresis loops.
Tensile tests
Samples used were in the form of strip films of length 3 cm, width 0.5 cm and thickness 0.1 mm. The tensile stress-strain tests were carried out on film specimens at room temperature and at a force rate of 1.5 N/min using the DMA Q800 instrument with film clamps (TA Instruments LLC, Delaware) at room temperature.
Dynamic mechanical tests
The mentioned DMA Q800 instrument was used through the dynamic mechanical studies. For these tests, a film clamp was used in dry mode. Tests were performed with the strip film samples of length 3 cm, width 0.5 cm and thickness 0.1 mm under tension, while the frequency is changed. A static preload force (0.01 N) was applied to the sample before the dynamic oscillating force to prevent film buckling.
During measurements, the instrument was programmed to maintain the static load at 125% of the force required to oscillate the sample. It is important that the film remained in its linear viscoelastic region during measurement (to ensure that the properties observed were independent of the deformation applied and truly reflected molecular motions), and so experiments were recorded maintaining constant strain. Generally, for thin polymer films, linear viscoelastic behavior can be assured with a strain less than 0.1%, and so this limit was used.
All measurements and tests were repeated many times on different portions (five samples) of the main film sample to ensure the reproducibility of results.
Results and discussions
Size and shape of iron oxide and cobalt iron oxide nanocomposites
Figure 2 illustrates the TEM images of the prepared iron oxide without cobalt. The particles, as detected from the images, appear like nanosheets with an average size in the range of 100 nm. This means that the structure of α-Fe2O3 can be described as the stacking of sheets of octahedral (six-fold) coordinated Fe3+ ions between two closed-packed layers of oxygen. Each oxygen is bonded to only two Fe ions because Fe is in the trivalent state. Therefore, only two out of three available oxygen octahedrons are occupied. The Fe–O sheets are held together by strong covalent bonds.

TEM images of iron oxide.
On the other hand, after the addition of cobalt, TEM images shown in Figure 3 revealed that the prepared cobalt iron oxides are nanoparticles and tend to aggregate in the form of clusters. This indicates a good adhesion among them and it may be due to their magnetic properties. Besides, irregularly shaped particles are observed in many locations because of the diffusion and aggregation of the nanoparticles. TEM images shown in Figure 3(b) and (c) show spherical shapes of nanoparticles with sizes ranging from 5 to 10 nm. Cobalt, iron, and oxygen were clearly identified as detected by the EDX analysis shown in Figure 3(d).
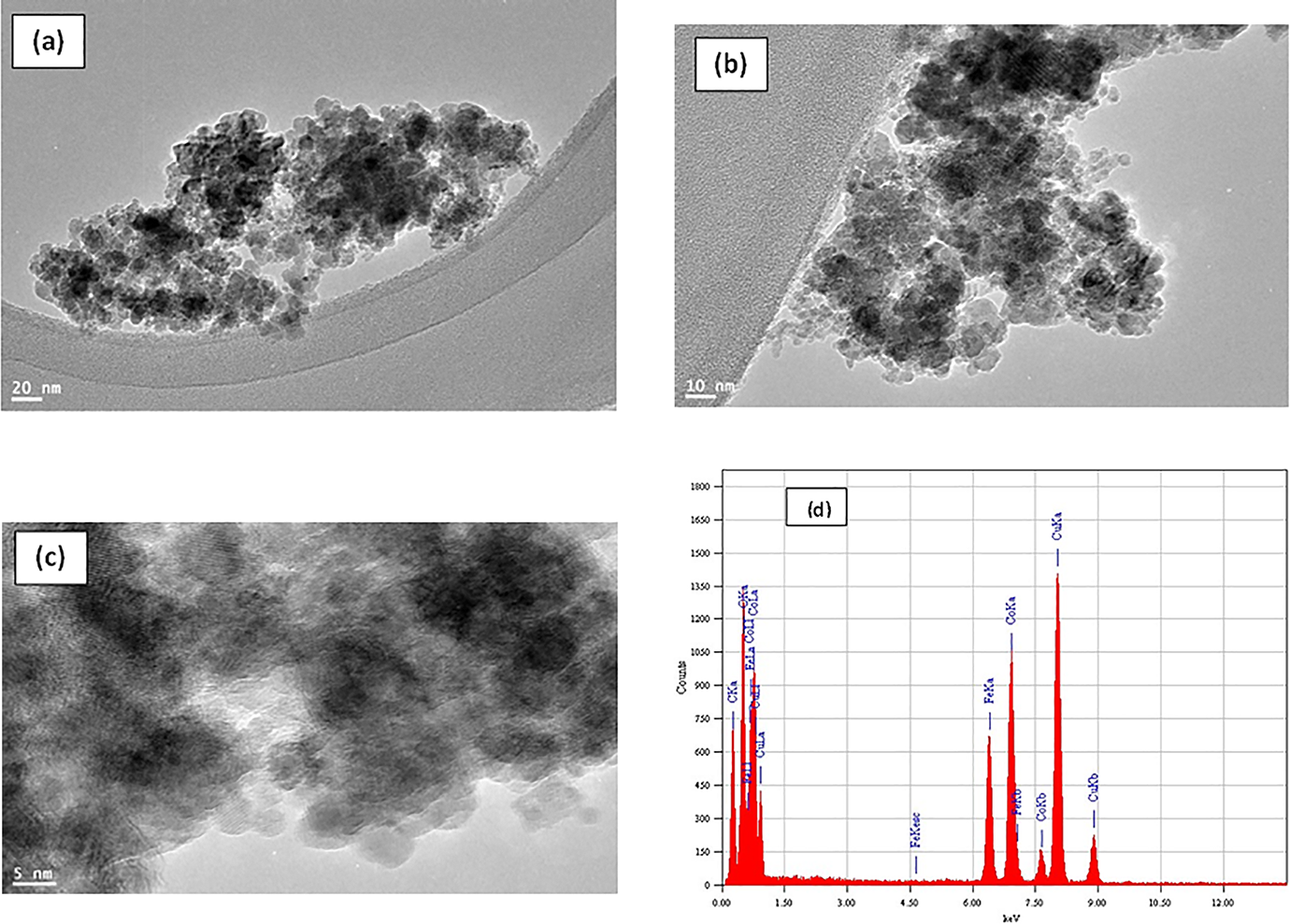
(a to c) TEM images at different locations of cobalt iron oxide (Co/Fe-2) with molar ratio 2 and (d) EDX pattern of cobalt iron oxide (Co/Fe-2) with molar ratio 2.
Structure of iron oxide and cobalt iron oxide nanocomposites
The X-ray diffraction pattern of the prepared iron oxide is shown in Figure 4(a). The pattern indicates that the iron oxide is similar to hematite Fe2O3. Sharp peaks at d-values 0.180 nm (024), 0.22 nm (113), 0.251 nm (110), 0.269 nm (104) and 0.368 nm (012) are observed. This indicates a single phase of α-Fe2O3 and resembling the JCPDS file 33-0664.
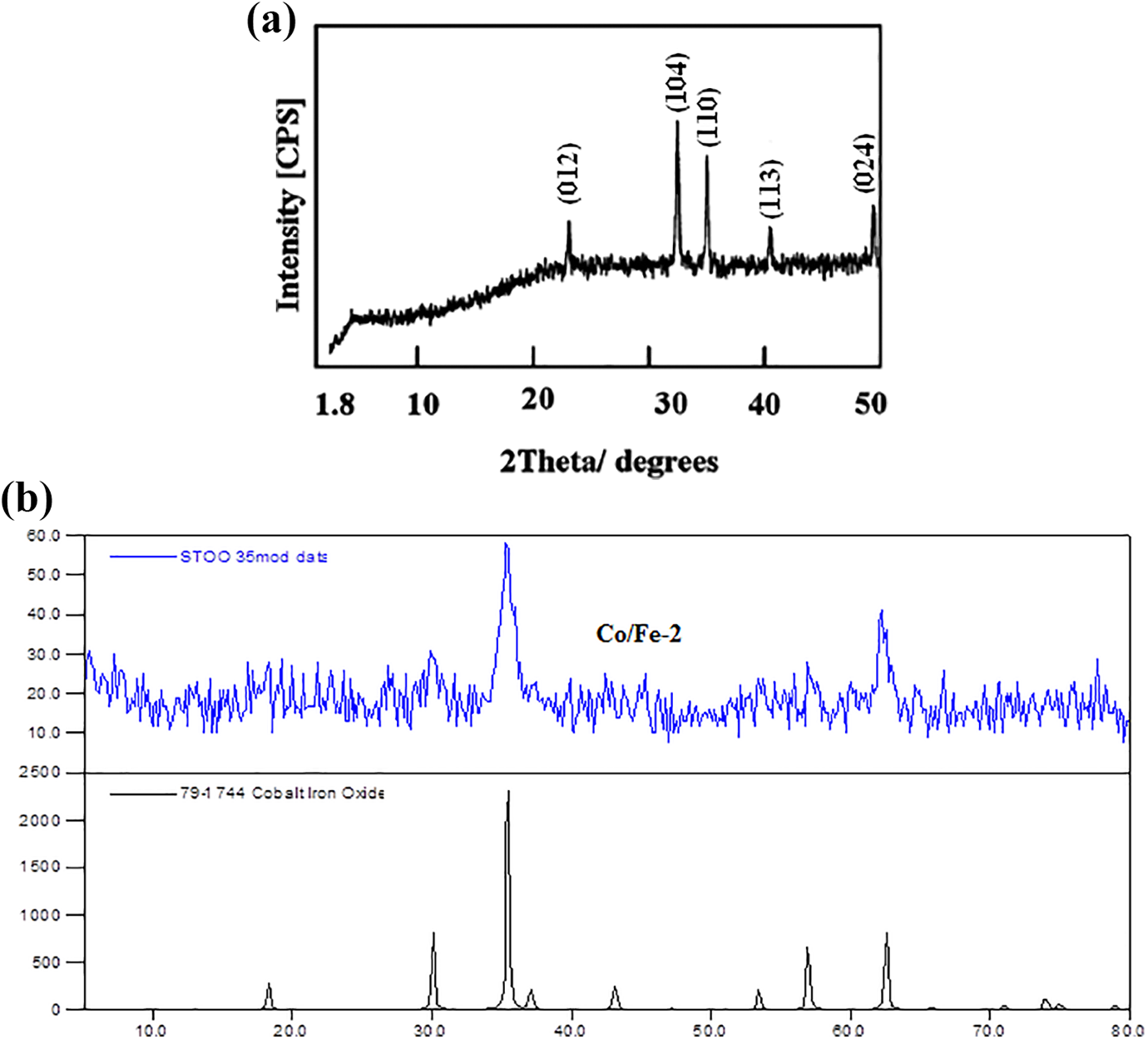
XRD of (a) iron oxide and (b) cobalt iron oxide (Co/Fe-2) with molar ratio 2.
On the other hand, Figure 4(b) shows the diffraction pattern of Co/Fe with molar ratio 2. The pattern revealed the presence of main peaks at planes (311) and (440) beside the appearance of week peaks at planes (220) and (511) that are related to the spinel structure of CoFe2O4 and matching with JCPDS file 79-1744. It is known that the preferred orientation of the planes in ferrites is (311). While, the peaks at planes (440) and (220) are sensitive to octahedral and tetrahedral structures; respectively.31,32 It is clear that these peaks are broad and are attributed to the nanosize of the sample. All peaks get broader with the addition of cobalt leading to an amorphous structure of cobalt iron oxide.
Magnetic properties of cobalt iron oxide and PVA loaded Co/Fe-2 nanocomposites
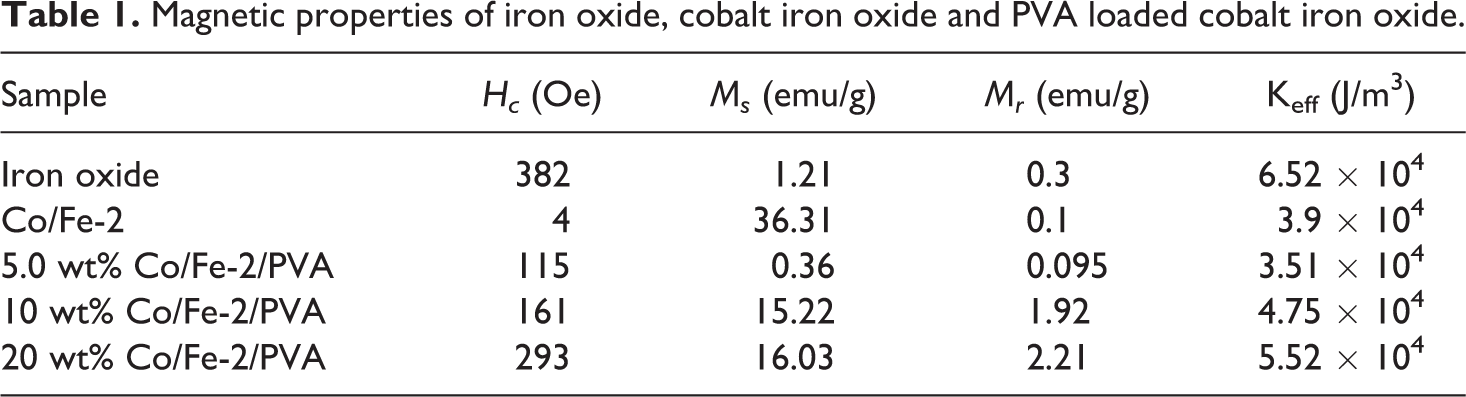
The room temperature of the magnetization versus the applied magnetic field (M-H) hysteresis loops of the prepared iron oxide and CoFe2O4 are shown in Figure 5. The inset shows an enlarged view of loops at the center. A typical soft ferromagnetic state of the CoFe2O4 sample can be seen from the magnetic hysteresis loops.33–36 The values of the coercivity (Hc), the remnant magnetization (Mr) and saturation magnetization (Ms) are listed in Table 1. Saturation magnetization is found to jump from 1.21 emu/g for iron oxide to 36.31 emu/g for Co/Fe-2 nanocomposites. This indicates that Co particles deeply enhance the magnetic properties of CoFe2O4 nanocomposites. Besides, the value of the coercive field Hc deduced from the hysteresis loops drops from 382 Oe for iron oxide to 4 Oe for Co/Fe-2 which an indication of magnetism which is observed in a single domain fine magnetic nanoparticle system called super-paramagnetism [2]. Super-paramagnetic atoms don’t contribute to the remnant magnetization Mr and this is revealed from the measurements in which the value of Mr decreased from 0.3 emu/g for iron oxide to 0.1 emu/g for Co/Fe-2 sample.
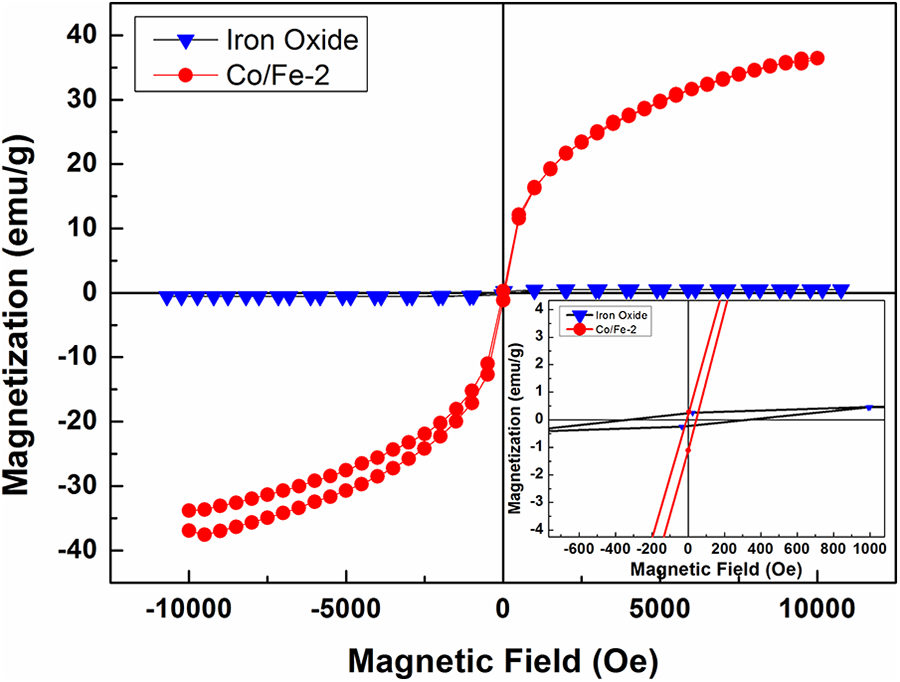
Magnetization-magnetic field (M-H) curves of iron oxide and cobalt iron oxide (Co/Fe-2) with molar ratio 2.
Magnetic properties of iron oxide, cobalt iron oxide and PVA loaded cobalt iron oxide.
From the above discussions, results indicate that the ferromagnetic state of α-Fe2O3 particles is shifted to the super-paramagnetic state as the Co added. The presence of Co in α-Fe2O3 nanocomposites has also been found to affect the coercivity Hc, remnant magnetization Mr and saturation magnetization Ms of the samples. This result allows the creation of numerous synthesis methods of CoFe2O4 nanocomposites for optimizing scale up production and use in technological applications.
Magnetic properties of PVA loaded Co/Fe-2 nanocomposites
Figure 6 illustrates the M-H hysteresis loops of PVA loaded with different concentrations of Co/Fe-2 nanofiller at room temperature and the magnetic data are also presented in Table 1. The magnetic properties of these polymeric nanocomposites arise mainly due to the addition of cobalt iron oxide nanofiller. It is expected that, the saturation magnetization of PVA loaded with Co/Fe-2 is less than that of Co/Fe-2 itself. This appears well from Table 1, saturation magnetization of Co/Fe-2 is 36.31 emu/g while for sample PVA loaded with 20 wt% Co/Fe-2, equals 16.03 emu/g. The presence of PVA layers on the surface of magnetic nanoparticles can cause surface anisotropy, crystalline disorder, reduction in the particle-particle interaction and exchange coupling energy which results in a significant decrease in Ms values.18,22,37 As the amount of magnetic nanofiller decreased, the saturation magnetization also decreased as a result of increasing the un-active polymeric layers surrounding the nanoparticles and may be increasing the separation distances between magnetic nanofiller which lead to a decrease in magnetic particle-particle interaction. Moreover, saturation magnetization depends mainly on the total mass of the magnetic nanofiller and varies as a function of the loaded weight. 38
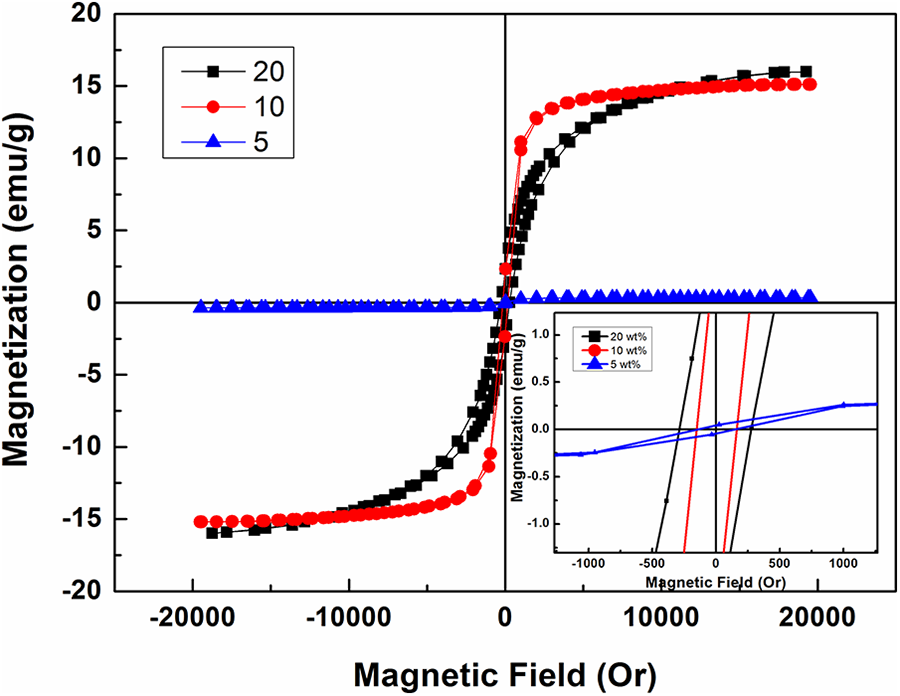
Magnetization-magnetic field (M-H) curves of PVA loaded with different weight ratios of cobalt iron oxide (Co/Fe-2) with molar ratio 2.
It is observed that the coercivity of PVA loaded with 20 wt% Co/Fe-2 nanocomposite is reduced as compared to Co/Fe-2 from 382 to 293 Oe. This reduction may be due to the decrease in the total mass of magnetic nanofiller and the residual strain which are the key factors in the coercivity. 39
From the above discussions and a theoretical fact, 40 it is noticed that the coercivity of a material decreases with increasing the content of the magnetic filler. This behavior is reversed for PVA loaded with different concentrations of Co/Fe-2 samples studied in this work. Table 1 revealed that as the content of magnetic nanofiller Co/Fe-2 increased, the coercivity of the nanocomposites increased too. On studying the hysteresis loop of the material, the given loop is probably due to the rotation of the magnetic nanofiller inside the polymer matrix when it is placed in an external magnetic field. The magnetic particles thus get magnetized and form chain-like structures lining up along the magnetic force lines. As the field reversed, magnetic particles tend to flip over to get their momenta parallel to the newly established influencing field. Magnetic particles inside the polymeric matrix are surrounded with polymeric layers that tend to render the flipping process and so need higher field value in the opposite direction. As the amount of contained magnetic nanofillers increased, more magnetic particles are bounded in places which need much higher fields to flip. In this respect, we can say that, as the material becomes hard, coercivity increased and this will be discussed in detail during the discussions of the elastic modulus in the next section.
The coercivity can be related to the strain-induced magnetic anisotropy, Keff. This magnetic anisotropy can be calculated by fitting the high field regions (H » Hc) to the law of approach to saturation (LAS) using the equation22,41:
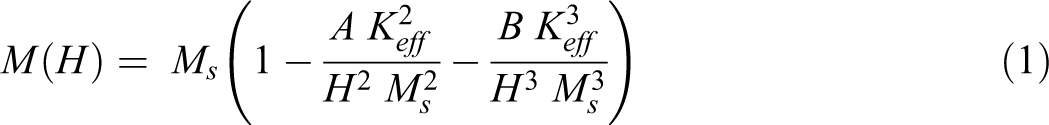
where M is the magnetization and H is the applied magnetic field, Ms is the saturation magnetization, Keff is the effective magnetic anisotropy, A and B are fitting parameters with values equal 0.4 and 0.0884 for magnetic nanoparticles and 0.55 and 0.0384 for polymer nanocomposite samples. Figure 7(a) and (b) represent the fitting of equation (1) for magnetic nanofiller and polymer nanocomposite, respectively. The values of saturation magnetization coincide with the experimental values. The calculated values of the effective magnetic anisotropy are listed in Table 1 which appears to be in direct relation with the coercivity.
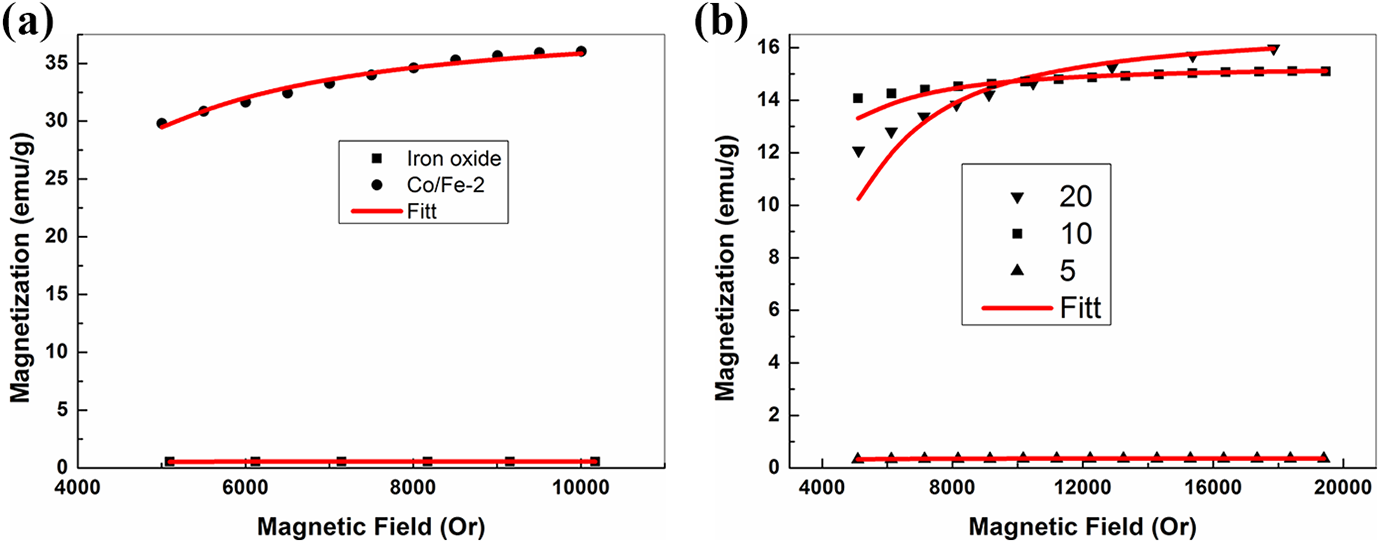
Magnetization-magnetic field (M-H) curves and fitting curves to LAS of (a) iron oxide and cobalt iron oxide (Co/Fe-2) with molar ratio 2 and (b) PVA loaded with different weight ratios of cobalt iron oxide (Co/Fe-2) with molar ratio 2.
Mechanical properties of PVA loaded Co/Fe-2 nanocomposites
Figure 8 presents the stress-strain curves of PVA loaded with different amounts of Co/Fe-2 magnetic nanofiller at 300 K. All curves show straight lines relations between stress and strain in the low stress-strain ranges. This reflects the Hookean behavior of these composites at low strain values. The slopes of these linear Hookean regions are mainly the elastic modulus which is presented in Figure 9 as a function of the magnetic nanofiller loadings. The addition of Co/Fe-2 nanofiller to PVA matrix increases the elastic modulus from 2.69 GPa at 1.0 wt% to 4.92 GPa at 20 wt% loadings. Figure 9, also, illustrates the dependence of the coercive field Hc on Co/Fe-2 loading. Both E and Hc are found to increase linearly with Co/Fe-2 weight percent ratio w according to the equation:

where Y denotes either E or Hc, a is the slope and equals 0.105 MPa for the elastic modulus and 12.06 Oe for the coercivity and b is a constant equals 2.77 MPa for E and 49 Oe for Hc; respectively. The increase in the elastic modulus with increasing Co/Fe-2 content may be attributed to the entanglement and coiled configuration of polymer chains around the filler particles which prevent or restrict chains elongations with the applied stress.42–44 When an external magnetic field is applied to these materials, magnetic moments are forced to align parallel to the field and may be stacked in place due to the entanglement of polymer chains. When the external magnetic field is removed, coiling and entanglement configuration around the magnetic particles restrict its motion and cannot return to the original state producing some remnant magnetization, and so an external field in opposite direction is needed to remove this magnetization which is the coercive field. As the content of magnetic nanoparticles increased, more magnetic moments are prevented from the re-orientation process leading to more remnant magnetization and consequently high coercive field values. From this discussion, one can conclude a relationship between elasticity and coercivity which is, high elastic modulus materials have high coercivity.
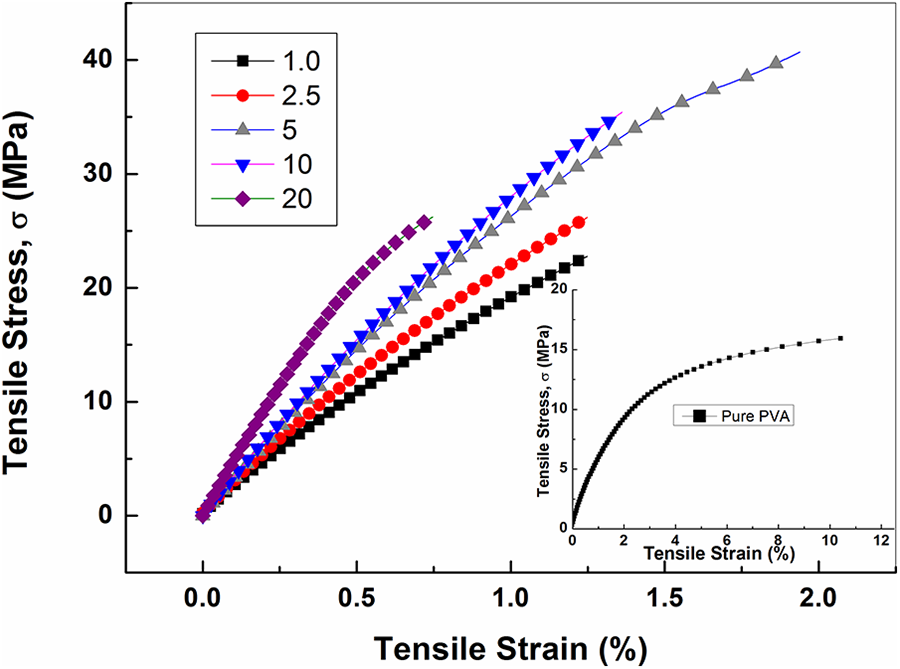
Tensile stress-strain curves of PVA loaded with different weight ratios of cobalt iron oxide (Co/Fe-2) with molar ratio 2 at a force rate of 1.5 N/min.
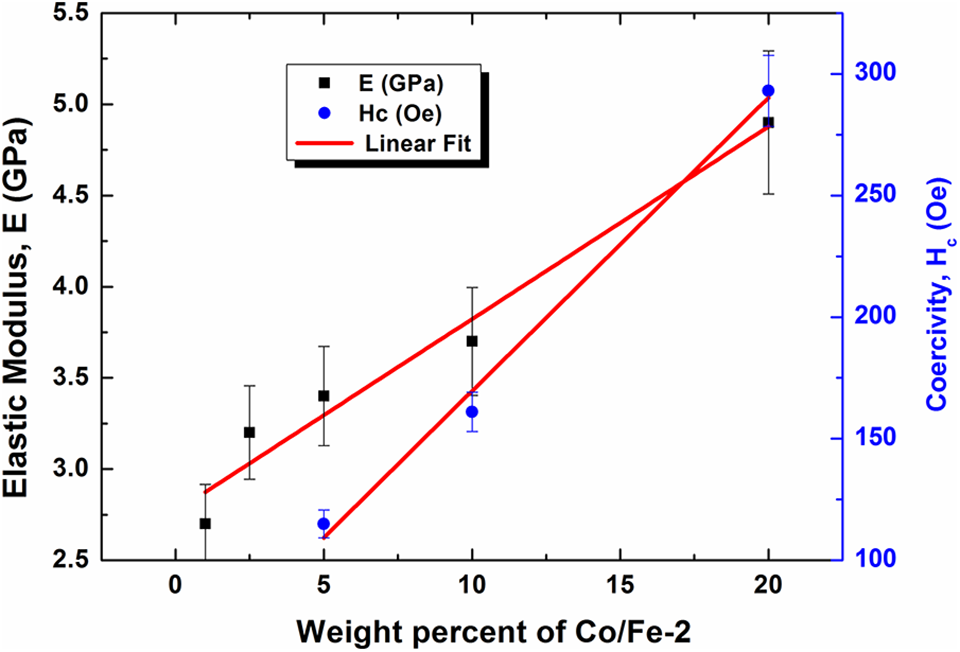
Dependence of the elastic modulus and coercivity on the weight percent of cobalt iron oxide (Co/Fe-2) with molar ratio 2.
The dependence of the tensile strength and the elongation at break on the weight ratio content of Co/Fe-2 of the studied samples are shown in Figure 10. Both have maximum values at 5.0 wt% of Co/Fe-2 loading which indicates that the material is converted into brittle at concentrations greater than 5.0 wt% of Co/Fe-2. At low concentrations of Co/Fe-2, polymer chains cover all the particles and stresses act on chains only. As the amount of Co/Fe-2 increased with good dispersion, interactions of chains endings and the filler particles surfaces as well as more entanglement and coiling of polymer chain take place. This appears as an increase in the tensile strength and strain at break. At a certain concentration (5 wt%) the interaction and entanglements with coiling of chains reach maximum and thus, both tensile strength and strain at break reach maximum values. Above loading concentration of 5 wt%, agglomerations of filler nanoparticles are formed (appears as a composite with less polymer chains) which resulted in a decrease in the chains-fillers interactions as well as less chains entanglements and coiling. This cause a decrease in both tensile strength and strain at break and the material becomes like brittle one.
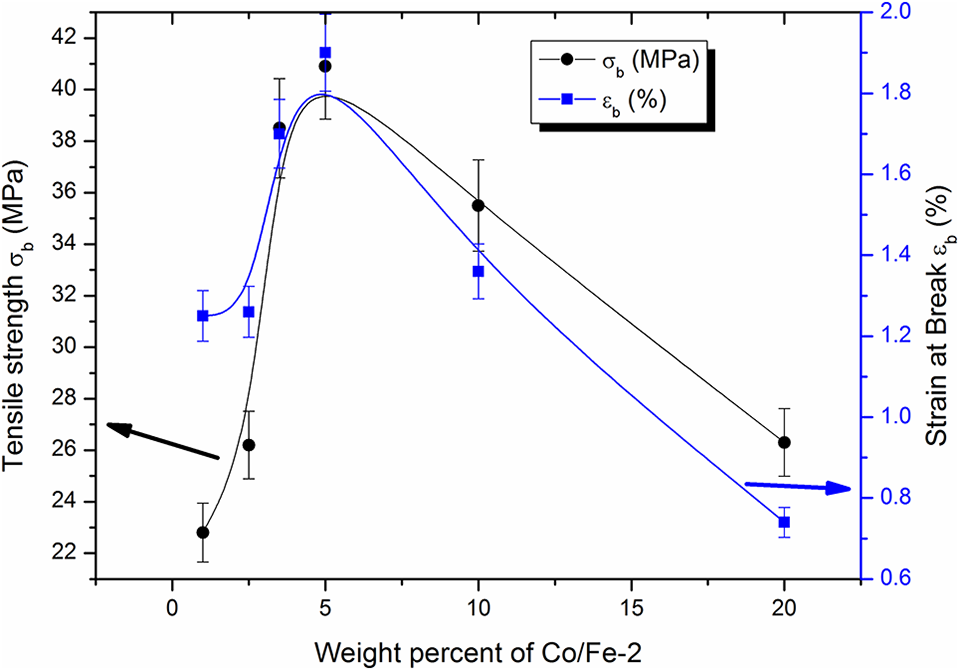
Dependence of the tensile strength and strain at break on the weight percent of cobalt iron oxide (Co/Fe-2) with molar ratio 2.
Figure 11 illustrates the dependence of the storage modulus, E/, on the driving frequency, f, for PVA loaded with different concentrations of Co/Fe-2 at room temperature. The storage modulus shows a linear dependence on the logarithm of frequency with slopes 0.139, 0.175, 0.200, 0.335 and 0.381 GPa for 1.0, 2.5, 5.0, 10 and 20 wt% of Co/Fe-2 loadings, respectively. The figure shows an increase in E/ with increasing both frequency and Co/Fe-2 content. This rheological response is similar to the relaxation behavior of the typical polymer composite systems. 45 It is known that the polymer chains are fully relaxed and exhibit characteristic homopolymer-like terminal flow behavior, resulting in that the flow curves of polymers being expressed by the power-law E/α f2.46–49 Krishnamoorti and Giannelis, 48 reported that the slopes of E/(f) for polymer/layered silicate nanocomposite were much smaller than 2 which are the values expected for linear homodispersed polymer melts. They suggested that large deviations in the presence of a small quantity of layered silicate might be due to the formation of a network structure in the molten state. The increase of the values of the slope of E/ versus frequency with increasing Co/Fe-2 content is an indication of the interaction that occurred between polymer chains and Co/Fe-2.
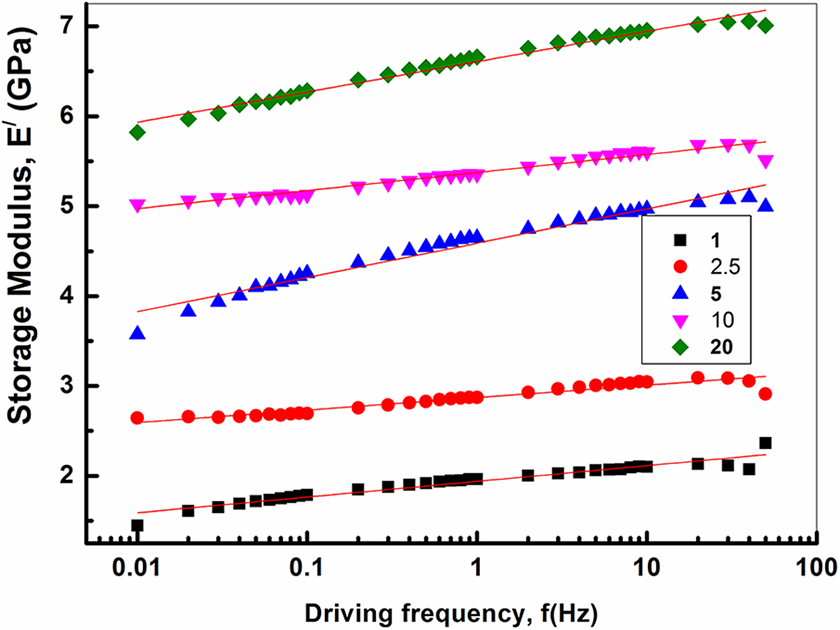
Dependence of the storage modulus on the driving frequency of PVA loaded with different weight ratios of cobalt iron oxide (Co/Fe-2) with molar ratio 2.
The change in the storage modulus of the Co/Fe-2/PVA nanocomposites as a function of Co/Fe-2 content at room temperature and different frequencies of 0.1, 1.0 and 10 Hz is shown in Figure 12. The storage modulus increases markedly with increasing Co/Fe-2 content. This increases perhaps because the Co/Fe-2 can be more easily dispersed effectively in the PVA matrix, causing strong interactions.
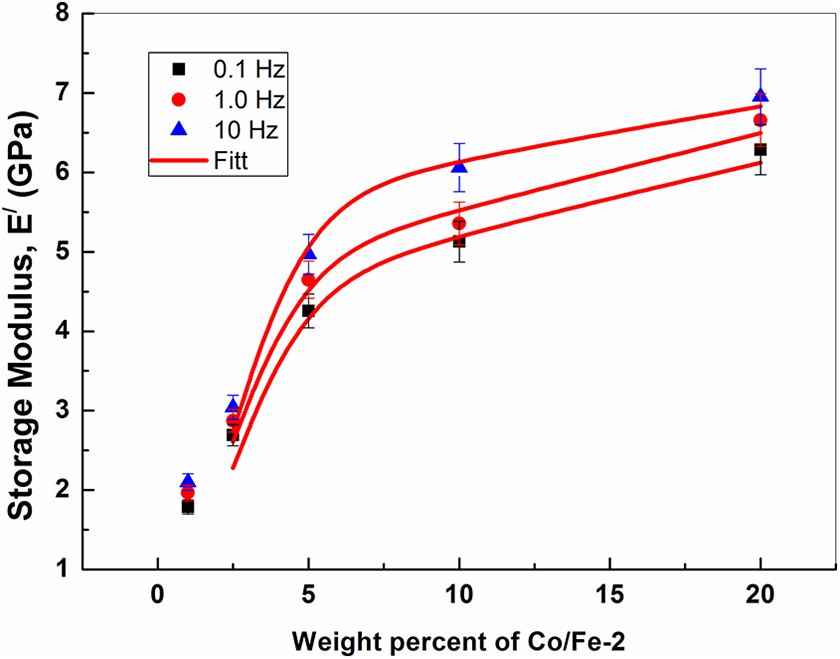
Dependence of the storage modulus on the weight percent of cobalt iron oxide (Co/Fe-2) with molar ratio 2 at different frequencies.
The dependence of the storage modulus on the concentration of Co/Fe-2 (Figure 12) appears to have what is known as rheological percolation behavior. To determine the rheological percolation threshold of Co/Fe-2-polymer composites, the relation between the storage modulus and the concentration of the filler in a medium is drawn into a modified power-law equation:

where, w is the Co/Fe-2 loading,
In percolated systems, one can observe a drastic change of the viscosity at a fixed frequency for a given concentration of the filler. This indicates that the Co/Fe-2–polymer composite reaches a rheological percolation threshold at which the filler particles block the motion of the polymer chains. The power-law equation (3) was used to determine the value of the rheological percolation threshold. The function was fitted to the experimental data points of E/ at 0.10, 1.0, and 10 Hz for
Conclusions
Iron oxide and cobalt iron oxide magnetic nanoparticles with a molar ratio equal 2 are prepared using a thermal solvent method under a critical temperature and pressure. The diameters of the prepared nanoparticles range from 5 to 10 nm. X-ray diffraction pattern of the prepared iron oxide indicates that the iron oxide is similar to hematite Fe2O3, while the addition of cobalt leads to an amorphous structure of cobalt iron oxide. VSM measurements give a saturation magnetization 36.31 emu/g and coercivity 4 Oe. The prepared Co/Fe-2 has a magnetism which is observed in a single domain fine magnetic nanoparticle system called super-paramagnetism. Magnetization-magnetic field hysteresis loops are observed for PVA loaded cobalt iron oxide nanocomposites. The area of the loop, saturation magnetization, coercivity and the elastic modulus are all increased with increasing the content of magnetic nanoparticles. Mechanical and magnetic measurements reveal that, high elastic modulus materials have high coercivity. A percolation behavior is found for the dependence of the storage modulus on the magnetic nanoparticles contents with a percolation threshold value 2.32 wt% at a driving frequency of 1.0 Hz.
Footnotes
Acknowledgement
The authors acknowledge the Deanship of Scientific Research at King Faisal University for the financial support under the annual research project (Grant NO. 170053).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The authors acknowledge the Deanship of Scientific Research at King Faisal University for the financial support under the annual research project (Grant NO. 170053).
