Abstract
A detailed study of the dispersion of magnetite (Fe3O4) nanoparticles in the polypropylene (PP) matrix, the degree of coagulation, and the dependence of magnetic properties of PP + Fe3O4 nanocomposites on the size of nanoparticles is reported. The size distribution of nanoparticles in polymer matrix and morphology of the nanocomposites were studied by the means of scanning electron microscope (JEOL JSM-7600 F) and atomic force microscopy (NT-MDT). It was found that when the Fe3O4 nanoparticles are introduced into the PP matrix, their coagulation takes place. The increase in the size of the Fe3O4 nanoparticles depends on their volume content in the polymer matrix, the viscosity of polymer, mixing time, and so on. The magnetic properties of PP + Fe3O4 nanocomposites were experimentally and theoretically studied. It was found that the magnetic hysteresis parameters of the nanocomposites directly depend on the size and concentration of the Fe3O4 nanoparticles in the matrix. Theoretical calculations were compared with experimental results obtained from M(H) measurements. Discrepancy between theoretical and experimental magnetic values have been explained.
Introductıon
Hybrid nanomaterials, particularly magnetic polymer nanocomposites, have gained significant interest due to the combination of unique properties such as excellent magnetic properties, stability, and good biocompatibility of inorganic and organic component in one material. 1 Magnetic nanoparticles (NPs) in polymer solutions present typical colloidal system consisting of continuous phase which is a dispersed medium and dispersed phase (NPs). The stability of the colloidal system is one of the key problems and all properties of such a system directly depend on the size and the dispersion of the NPs in polymer medium. 2 NPs are very active due to their high surface energy. The high surface energy of many types of particles is lowered by their coagulation. It is well-known that it is very important to ensure the narrow size distribution and good dispersion of NPs in tissue fluid for applications. Consequently, determining the nanoparticle size after introduction into the polymer is important from both theoretical and experimental point of view. The change in the size of NPs in a polymer medium affects some properties, including magnetic properties of this system, which in turn are largely dependent upon the size of filler. 3
At present, there is widespread use of composite materials based on thermoplastic polymers. Polypropylene (PP) is a thermoplastic polymer used in a wide variety of applications. Its properties are similar to polyethylene, but it is slightly harder and more heat resistant. In 2013, the global market for PP was about 55 million tons. 4
Magnetic NPs including iron, nickel, cobalt, and their hybrids and oxides are among the most attractive mineral NPs due to their numerous potential applications in various electronic, military, and medical industries. 5 Recently, iron oxide NPs have attracted much consideration due to their unique properties, such as superparamagnetism, surface-to-volume ratio, greater surface area, low toxicity, and easy separation methodology. Magnetite (Fe3O4) NPs are one of the commonly used compounds due to its considerable performance. 6,7
Nanocomposites based on thermoplastic polymers and Fe3O4 NPs promise new applications in many fields such as absorbent of electromagnetic radiations, environmental remediation, sensors, and other systems. 8 –12 The study of the effect of the dispersion of magnetic NPs on the magnetic properties of nanocomposites and the relationship between structure and properties is still the relevant question.
In the present work, the effect of Fe3O4 NPs concentration on the final size (distribution) and magnetic properties of nanocomposites based on PP matrix and Fe3O4 NPs was theoretically and experimentally investigated.
Experimental details
Materials
Isotactic PP (PP grade Moplen HF500 N, homopolymer): density—0.92 g/cm3 at 25°C; M w = 250,000; M n = 67,000; melt mass flow rate (MFR) = 11.5 g/10 min (230°C, 2.16 kg); melting temperature = 162°C. Fe3O4 NPs were synthesized via the chemical coprecipitation method in an alkaline medium. 11 The average NPs size were 7–15 nm. 12
Methods
X-ray diffraction
X-ray diffraction (XRD) analysis of samples was performed on Rigaku Mini Flex 600 XRD diffractometer at ambient temperature. The used radiation was monochromatic copper (Cu) K α from a Cu X-ray tube operating at 15 mA and 30 kV. The samples were scanned in the range of 2θ angle of 10–80°.
Scanning electron microscopy
The distribution of Fe3O4 NPs in the polymer matrix has been studied by scanning electron microscopy (SEM, JEOL JSM-7600 F). Scanning was carried out in SEI mode with an accelerating voltage of 15 kV and a working distance of 4.5 mm.
Atomic force microscopy analysis
The morphology of the nanocomposites was studied using atomic force microscopy (AFM) Integra Prima (NT-MDT, Zelenograd, Russia). For the scan used special silicon cantilevers fabricated by plasma etching method with the needle radius of curvature of 20 nm and the resonance frequency of 1–5 Hz. Scan size was 2 × 2 mm. The measurements were performed in the semicontact microscopy mode in air; needle change of the cantilever oscillation amplitude was fixed, determining the surface topography. The scanning speed and the number of scanned lines of the image are, respectively, 256 and 1969 Hz
Method for studying magnetic properties
Measurements: magnetization curves acquired at 300 K (room temperature) by a Quantum Design SQUID magnetometer in the field range ±80 kOe.
Synthesis of nanocomposites
The polymer nanocomposite materials were prepared as follows: isotactic PP was solved in toluene solvent, at a temperature 120°C. Fe3O4 NPs were added to the polymer solution at different volume contents and stirred for 2 h to prepare a homogeneous mixture. The mixture was transferred to a Petri dish and dried in a vacuum oven during the day. From these samples, by hot-pressing at the melting temperature of PP and a pressure of 10 MPa, 100 μm thin film was obtained. Cooling the films after hot pressing was carried out in water at the cooling rate 20 grad/min.
Results and discussion
Figure 1 shows X-ray diffractograms of pristine PP, PP nanocomposites with 0.6 vol%, 2 vol%, and pure Fe3O4 NPs in the 2θ range between 10–80°. The characteristic line observed at 30.36° (220), 35.68° (311), 43.3° (400), 57.36° (511), and 62.95° (440) fully correspond to the inverse cubic spinel phase of Fe3O4, according to ICDD (PDF-2/Release 2011 RDB) DB card number 01-073-9877. The diffraction peaks corresponding to the crystalline planes (311) and (220), which are characteristic of Fe3O4, become more and more prominent with the increase of Fe3O4 inclusion.
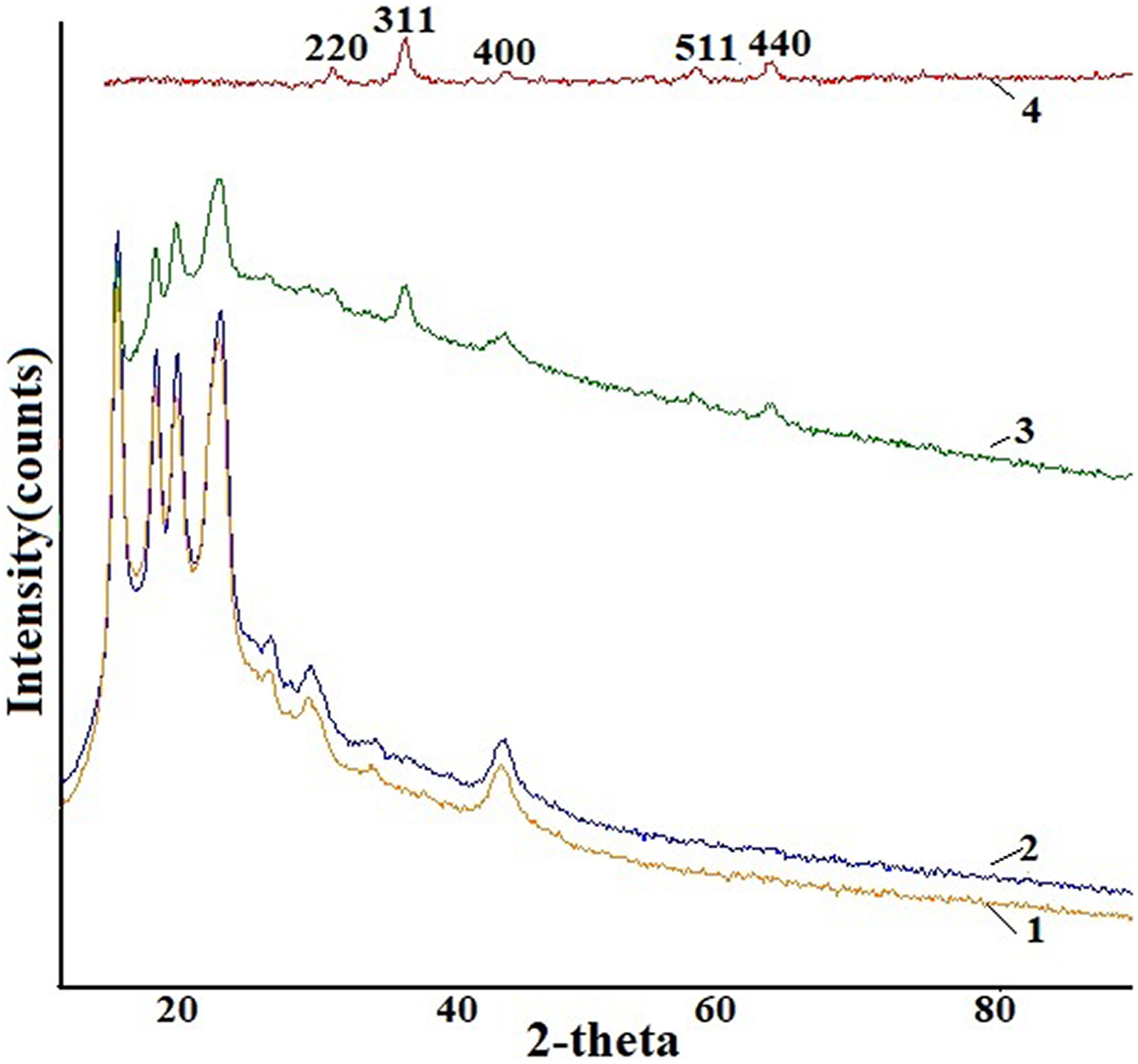
X-ray diffractograms of pristine PP, PP-based nanocomposites. (1) PP, (2) PP + 0.6 vol%, (3) PP + 2 vol%, and (4) pure Fe3O4 nanoparticles. PP: polypropylene; Fe3O4: magnetite.
The Fe3O4 particle size in polymer matrix and morphology of PP + Fe3O4 nanocomposites were investigated by scanning electron microscope JSM-7600F. The SEM images of the PP + Fe3O4 nanocomposites with different volume content of Fe3O4 NPs are shown in Figure 2. As it can be seen from the figure, for 0.6 vol%, 1 vol%, and 2 vol% contents of the filler, the sizes of Fe3O4 NPs in the matrix were 7–15 nm, 16–18 nm, and 20–29 nm, respectively.

SEM images of nanocomposites based on PP + Fe3O4. (a) PP + 0.6 vol% Fe3O4; (b) PP + 1 vol% Fe3O4; (c) PP + 2 vol% Fe3O4. SEM: scanning electron microscopy; PP: polypropylene; Fe3O4: magnetite.
The particle sizes observed from SEM images were used to determine statistical parameters of the particle size distribution.
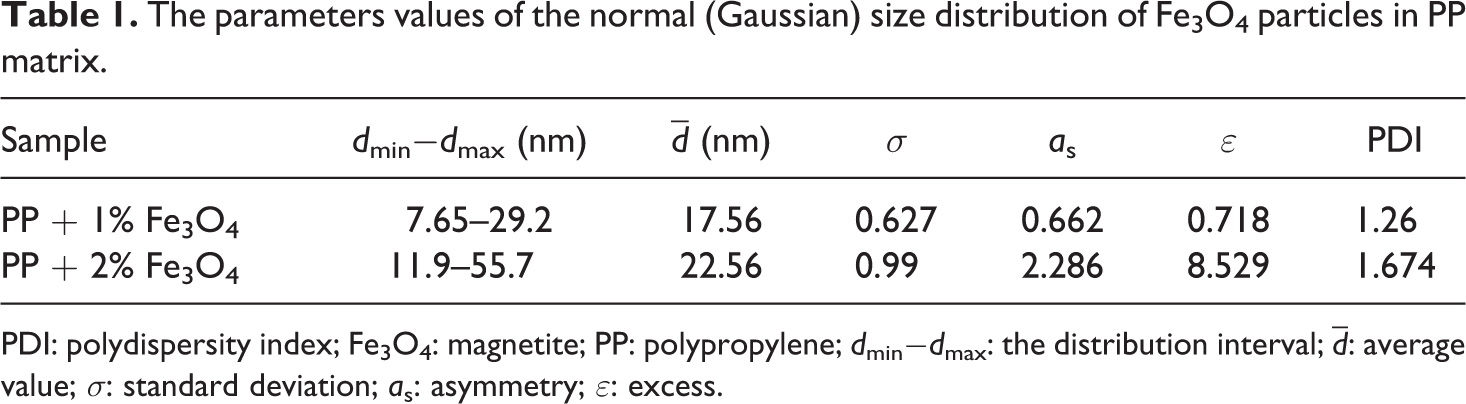
The parameter’s value of the normal and lognormal size-distribution of the Fe3O4 NPs in the PP matrix are reported in Tables 1 and 2, respectively.
The parameters values of the normal (Gaussian) size distribution of Fe3O4 particles in PP matrix.
PDI: polydispersity index; Fe3O4: magnetite; PP: polypropylene; d
min
−d
max: the distribution interval;
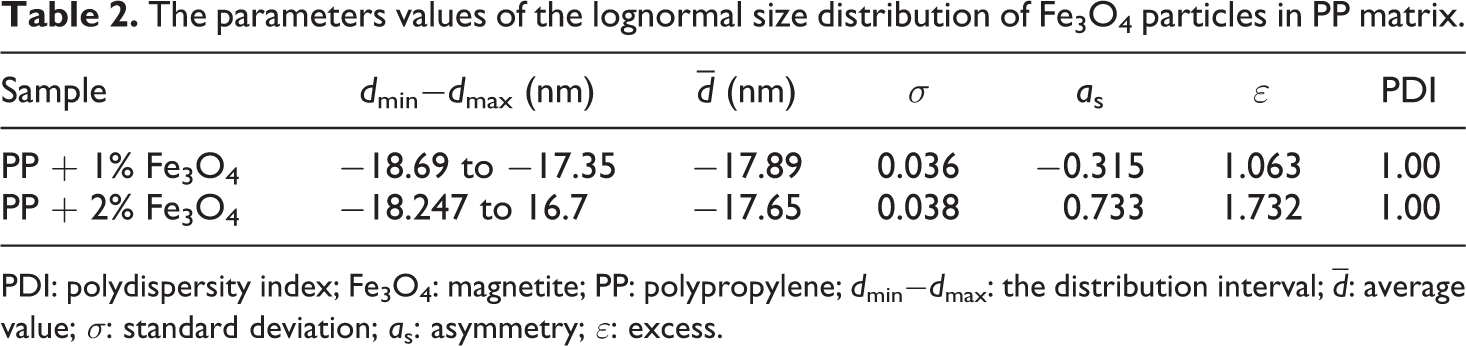
The parameters values of the lognormal size distribution of Fe3O4 particles in PP matrix.
PDI: polydispersity index; Fe3O4: magnetite; PP: polypropylene; d
min
−d
max: the distribution interval;
As it can be seen from the Table 2, the values of the standard deviation indicate a narrow lognormal particle size distribution. The values of the poly dispersity index confirm the monodispersity PP + Fe3O4 nanocomposites system. It means that Fe3O4 NPs homogeneously dispersed in the polymer matrix and can be described by a lognormal distribution function 13,14
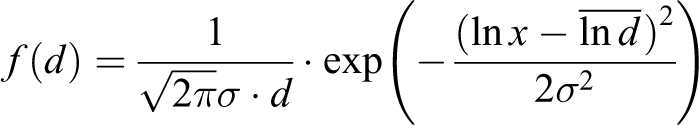
It is also clear from the table that the nature of the distribution of the Fe3O4 particles in the polymer matrix, as a function of their concentration and time, does not change. Only the distribution parameters:
Figure 3 shows the lognormal distribution of the Fe3O4 NPs at the different volume contents in the PP matrix:
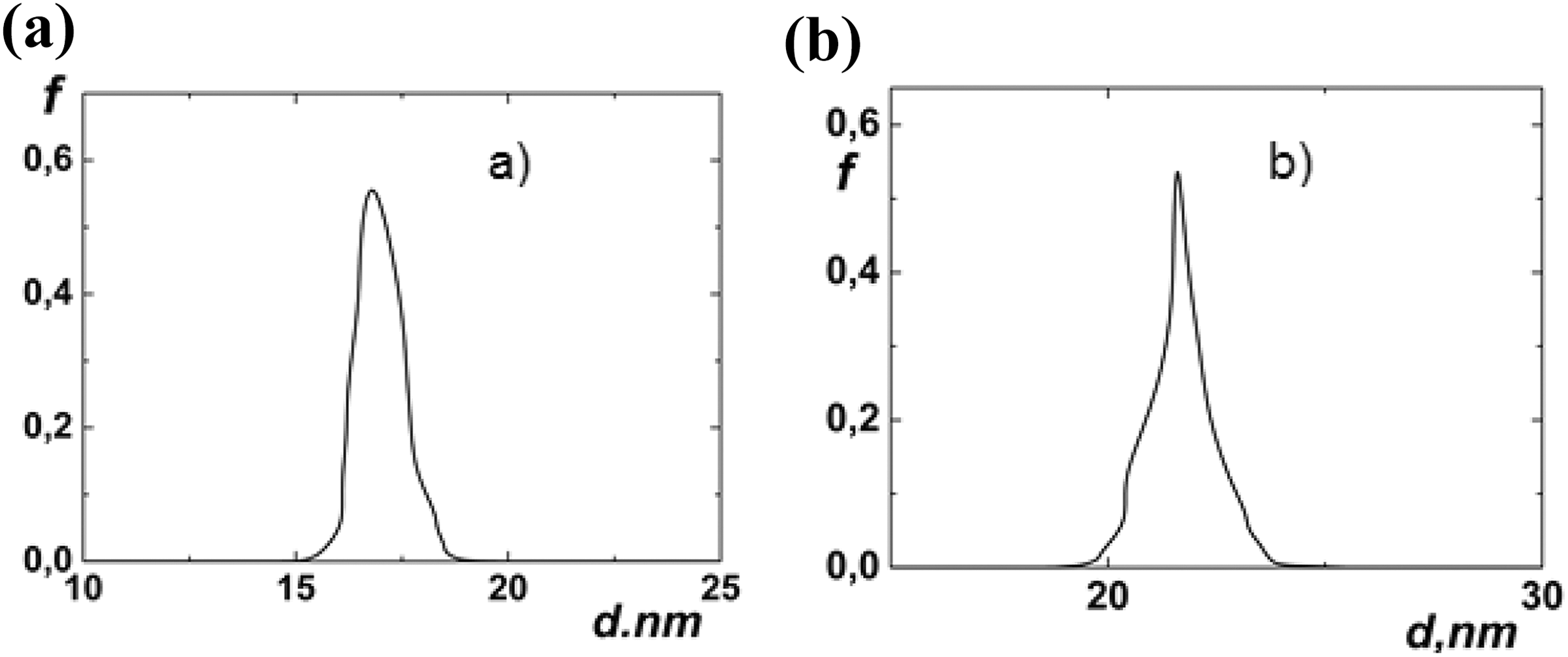
The size distribution function of Fe3O4 particles in the PP polymer matrix: (a) PP + 1% Fe3O4; (b) PP + 2% Fe3O4. PP: polypropylene; Fe3O4: magnetite.
The dependence of the average size of Fe3O4 NPs in the PP matrix on their concentrations was studied. Figure 4(a) and (b) represents the dependence of the average particle size of Fe3O4 NPs on its volume content—d(φ).
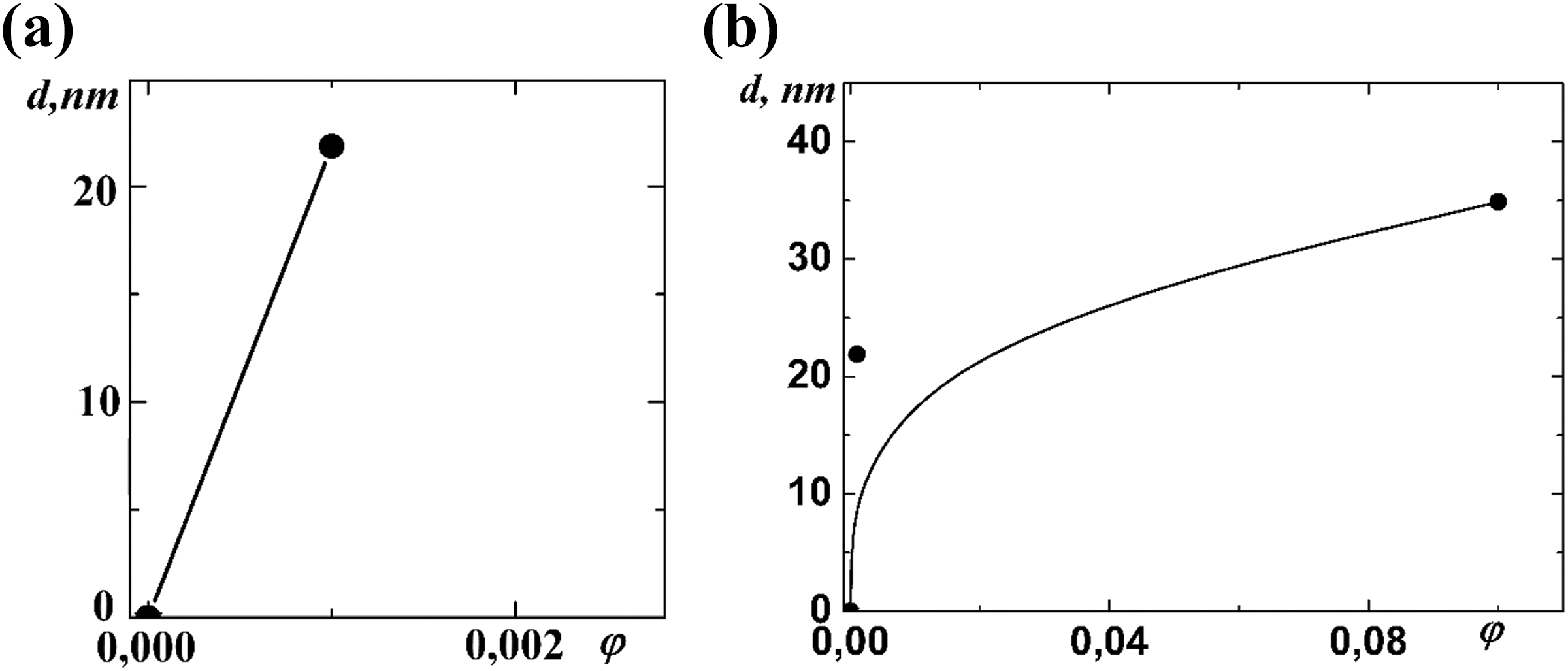
Dependence of the diameter of Fe3O4 particles on their concentration in polymer PP matrix. (a) Low content of the filler; (b) high content of the filler. PP: polypropylene; Fe3O4: magnetite.
As it can be seen from Figure 4(a) and (b) at low concentrations of the Fe3O4 NPs d(φ) dependence is the linear function, for the high content of the filler is square root function respectively. Coagulation of particles also depends on the viscosity of the PP that it has during the preparation of the composite. In a medium with a lower viscosity, the coagulation process will happen faster. As the concentration of the filler in the matrix increases, the viscosity of the medium changes and the coagulation condition changes as well.
The linear part of the d(φ) dependence (low concentration) can be described using the following equations

At high concentrations of the NPs, the saturation occurs, reaching the maximum value of the coagulated particle diameter, which can be described with an exponential law (Figure 4(a)).
The theory of coagulation of NPs in solutions at their low concentrations is known. According to this theory, the number of particles during coagulation decreases according to the law. 16,17
The value of k 0 is velocity constant and defined by Smolukhovsky as below 2 :

where η – viscosity of the dispersive medium.
By the definition
The solution is
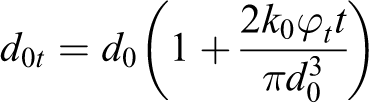
The expression obtained is valid for low volume content of particle. As it can be seen, the diameter of NPs aggregates is a linear function of concentration, which is consistent with the previously obtained result (Figure 4(b)). At high concentrations of the filler, the last expression should be rewritten with the help of such a function that the value of the function should asymptotically approach the fixed value “d,” when the value of the variable is high.
The values of “k” found from equations (1) and calculated with the help of equation (2) were compared. To determine the viscosity of the mixture, the Walter formula was used 17
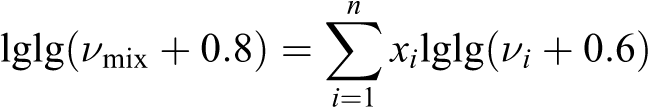
where v mix and v i are the kinematic viscosity of the mixture, and the ith component, and x i is the fraction of the component in the medium. From the latter, it is possible to determine the kinematic (further dynamic) viscosity of the mixture
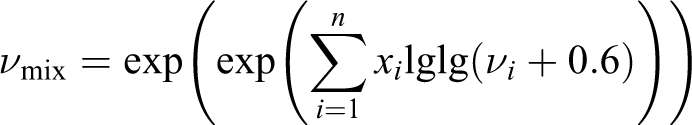
The obtained result is consistent with AFM illustrations.
AFM observation showed that the size of the dispersed phase increases with increasing filler content in the nanocomposites (Figure 5). The sizes of Fe3O4 NPs are 25–30 nm, 50–80 nm, and 65–90 nm for 0.6 vol%, 1 vol%, and 3 vol% contents, respectively.These results were found to be consistent with the results obtained from the theoretical calculations. There are differences between the magnet sizes and the actual geometrical dimensions of NPs. Table 3 reports the magnetic sizes of Fe3O4 NPs. The obtained experimental results correlate with theoretical data very well.
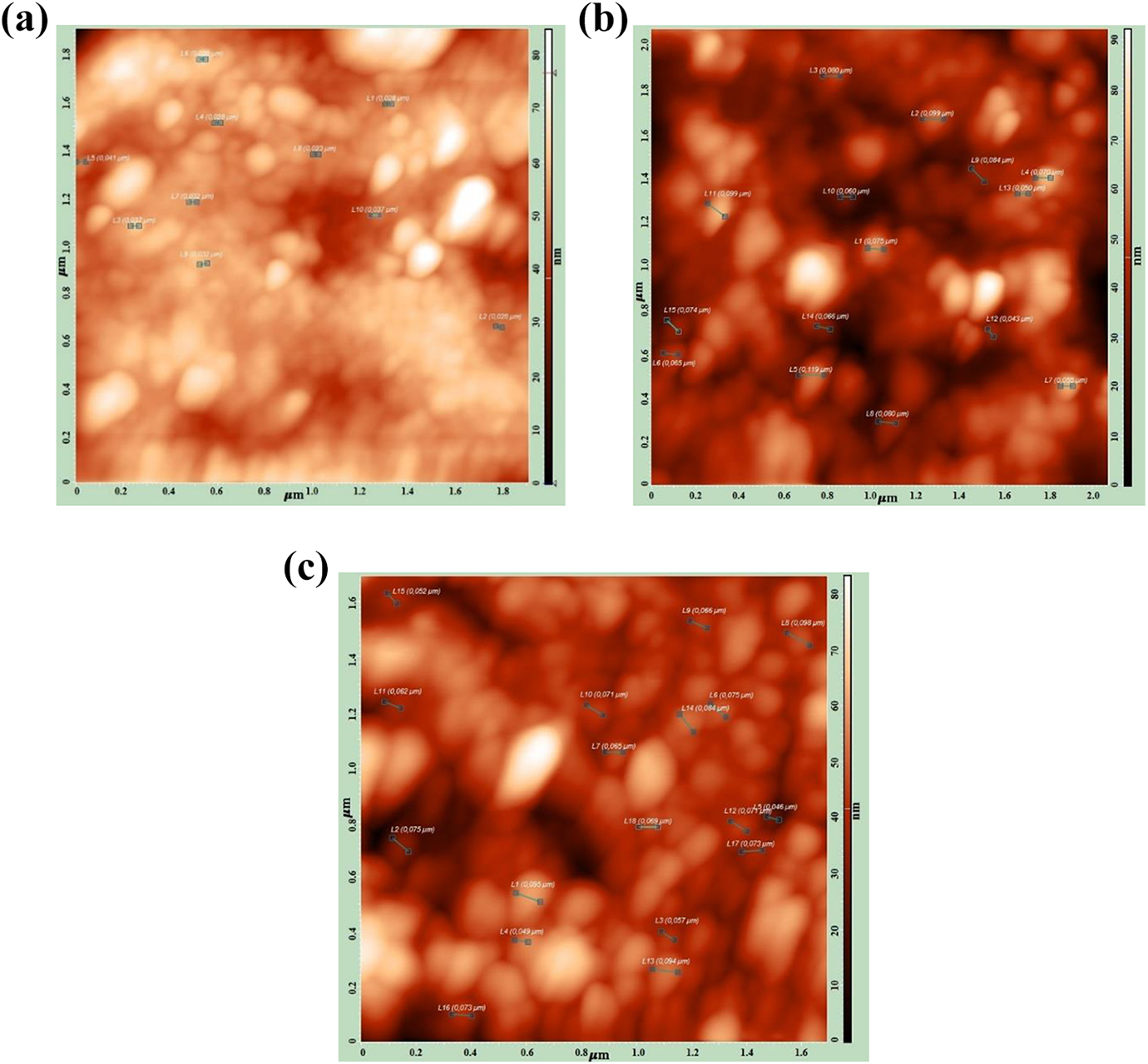
AFM image of PP + Fe3O4 nanocomposites. (a) PP + 0.6 vol% Fe3O4; (b) PP + 1 vol% Fe3O4; (c) PP + 2 vol% Fe3O4. PP: polypropylene; Fe3O4: magnetite; AFM: atomic force microscopy.
Magnetic sizes obtained from MFM study.
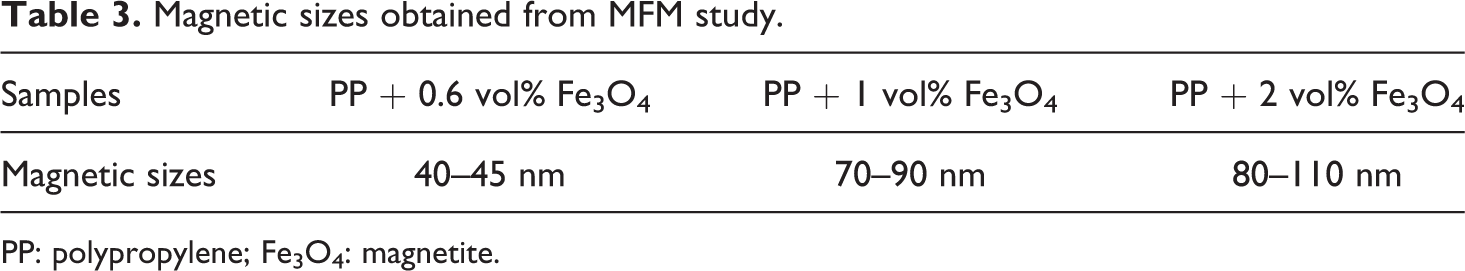
PP: polypropylene; Fe3O4: magnetite.
The magnetic properties of PP + Fe3O4-based nanocomposites were studied by the experimental and theoretical investigation. Figure 6 illustrates the experimental hysteresis loops of magnetic polymer composite materials with different volume content of the Fe3O4.
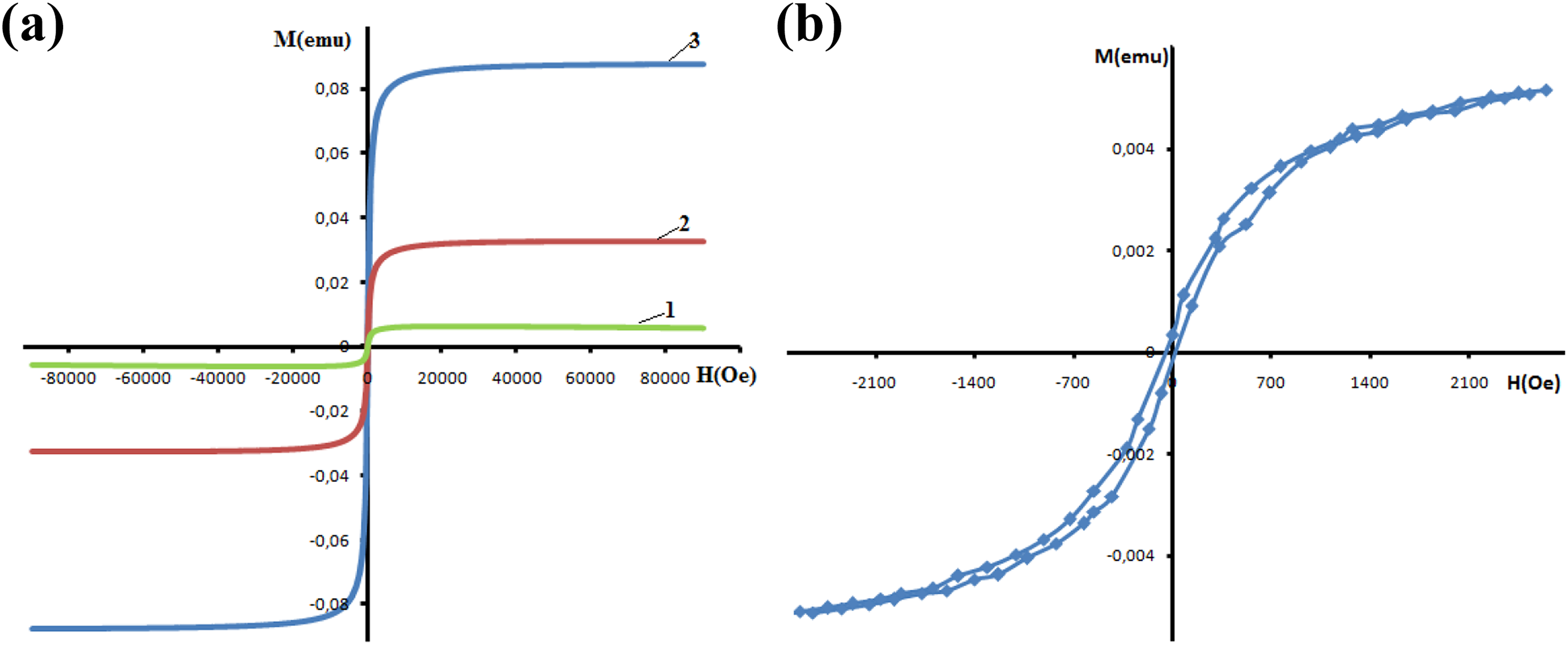
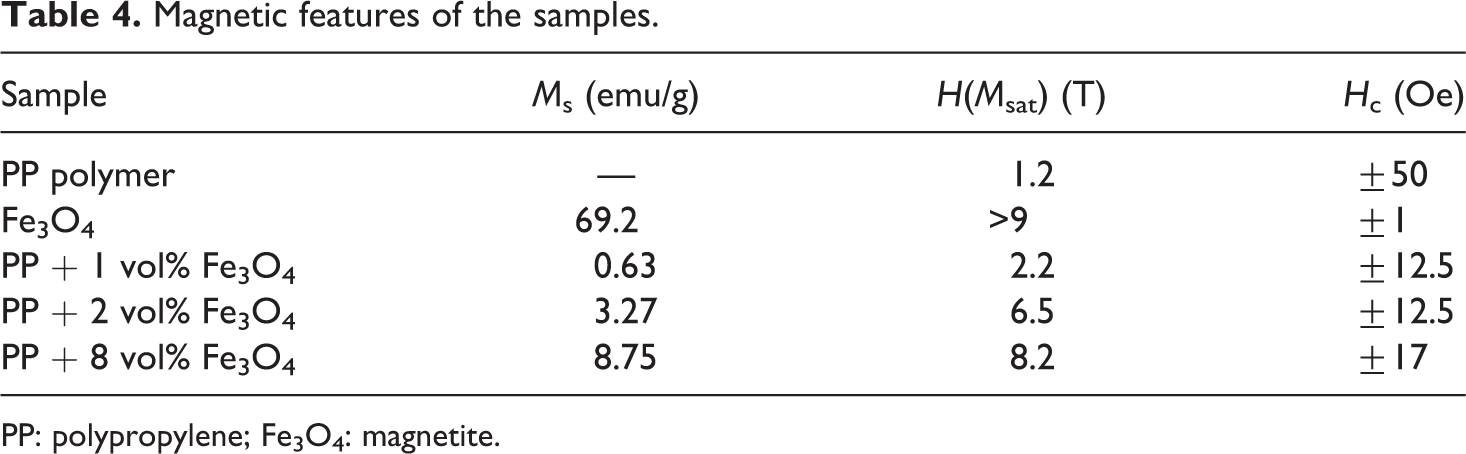
(a) M(H) magnetization versus magnetic field curves of PP + Fe3O4 based nanocomposites. (1) PP + 1 vol% Fe3O4; (2) PP + 2 vol% Fe3O4; (3) PP + 8 vol% Fe3O4. (b) Experimental magnezitation versus magnetic field M(H) curves of PP + Fe3O4-based nanocomposites under low magnetic field for PP + 1 vol% Fe3O4. PP: polypropylene; Fe3O4: magnetite.
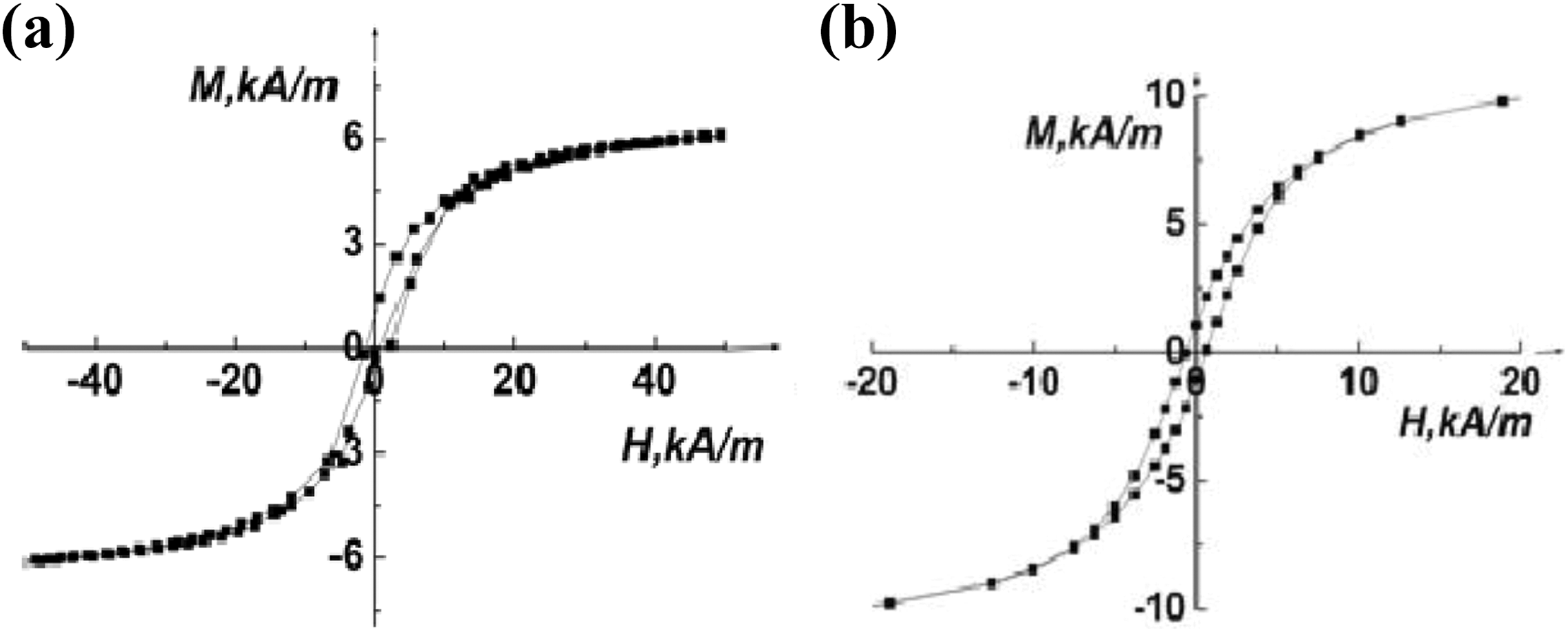
All the curves have a similar shape (superimposable). The curves near the origin demonstrate a small hysteresis loop(Figure 6). The coercive field (H c) of about ±12.5 Oe, ±12.5 Oe, and ±17 Oe is observed for PP + 1 vol% Fe3O4, PP + 2 vol% Fe3O4, and PP + 8 vol% Fe3O4 samples, respectively (Table 4). Thus, the NPs and the nanocomposites exhibit weak ferromagnetic behavior with very low coercive field.
Magnetic features of the samples.
PP: polypropylene; Fe3O4: magnetite.
Figure 7(a) and (b) shows the theoretical magnetization versus magnetic field curves of the nanocomposites based on PP matrix and Fe3O4 with the size of 20 nm. Numerical differentiation was carried out by the method. 18,19
Theoretical magnetization versus magnetic field M(H) of PP + Fe3O4 nanocomposites (a) PP + 6 vol% Fe3O4; (b) PP + 8 vol% Fe3O4. PP: polypropylene; Fe3O4: magnetite.
As it can be seen from Figure 6(a) and (b), the magnetization curves have the hysteresis loop. This is typical for particles which have many magnetic domains. The hysteresis parameters of the nanocomposites (saturation magnetization, residual magnetization, coercive force, etc.) depend on the size and concentration of Fe3O4 particles. As the size and concentration of Fe3O4 particles increase, the number of magnetic domains increases in both particle and medium. As it can be seen from Figures 6 and 7(a), the magnetic field dependence of the magnetization obtained from the experiments and calculated theoretically is close to each other for low magnetic field.
It is known that for dispersed systems, during the magnetization, the value of the saturation magnetization can be given by the following equation

where M
s and
To explain this, suppose that the geometric sizes of the NPs (d t) are not equal to the magnetic sizes of the NPs (d m).
For comparison of magnetic sizes with the geometric sizes of NPs, we use the following equations
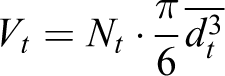
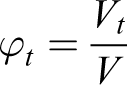
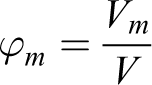
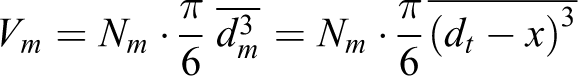
where V, Vt , and Vm are the volumes of medium, solid, and magnetic phase, respectively. Then, using the expression for the concentration of the magnetic phase, we obtain the relation between geometrical and magnetic phase
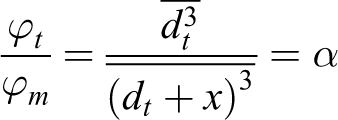
After the manipulation of the last expression, we obtain the cubic equation.
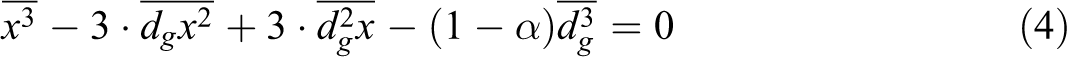
The last equation was solved by the Cardan method. 20 Table 5 reports the result obtained from the cubic equation. It should be noted that the magnetic properties of nanocomposites are independent on the type of the polymer.
Geometric and magnetic sizes of nanoparticles for different filler concentrations.
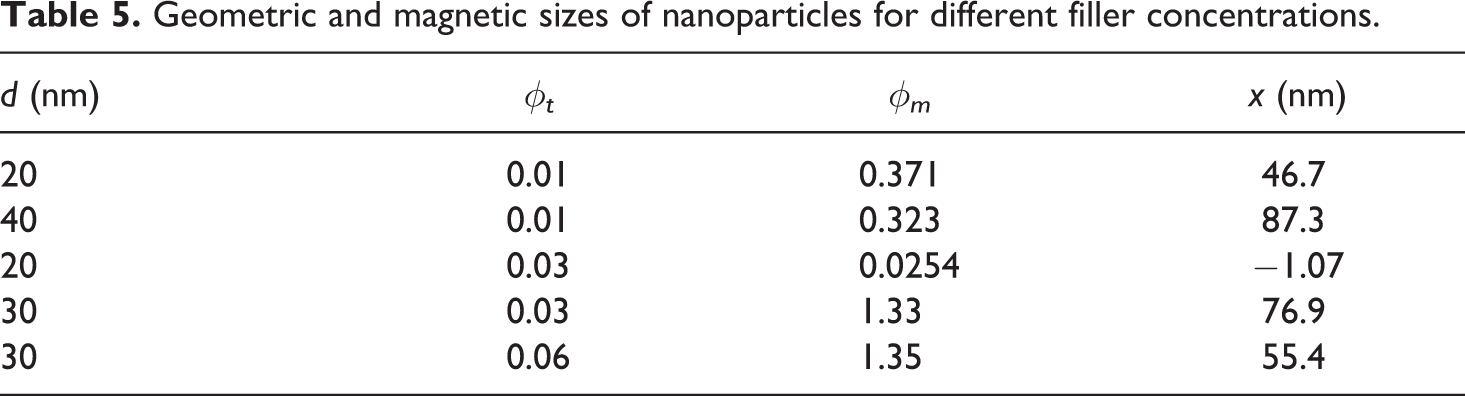
It can be concluded that the thickness of the polymer layer around NP rises with the increasing size of the NPs, and it also depends on the properties of the medium (molecular weight, polarity, viscosity, etc.).
It also can be seen from the table that in all cases the particle size increases, except for d = 20 nm, φ = 0.03 where the size decreases (i.e. the oxidization of the surface layer is close to the lattice parameter of the Fe3O4 NPs). The edge length of the unit cell is 0.839 nm. 21
Conclusion
It was found that the Fe3O4 particles are introduced into the polymer matrix, their coagulation takes place. The increase in the particle size depends on their concentration in the polymer matrix, the type of polymer (polar, non-polar, viscosity, etc.), reaction temperatures, and so on. When Fe3O4 particles are introduced into the polymer matrix, at the same time with the coagulation, the oxidation of their surface layer also occurs and the magnetic size decreases and hence the magnetic characteristics are reduced. It becomes clear that the magnetic hysteresis parameters depend on the size and concentration of Fe3O4 particles in the polymer matrix. Theoretical calculations were compared with experimental results obtained from M(H) measurements. At the same time, it has been revealed that, at low applied fields, the theoretical and experimental M(H) curves are of the similar shape. With increasing size of the NPs, the difference between theoretical and practical values increase and this is explained by the fact that, in theoretical approach, it is assumed that nanocluster consists of the single-domain NPs with one-dimension and the interactions that occur between the NPs and the matrix material are not taken into account.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
