Abstract
The water transport kinetics and moisture-induced thickness swelling behavior of wood-plastic composites (WPCs) in the presence of two imidazolium-based ionic liquids (ILs) were studied. The wood flour chemically treated with ILs were compounded through melt mixing of high-density polyethylene and coupling agent, and finally, the test specimens were produced by compression molding. Hygroscopic rates of the composites were evaluated by immersing them in the water at room temperature and monitoring moisture sorption and thickness changes for several weeks. Besides, the water diffusion coefficients and swelling rate parameters were measured for all composites. It was found that the water absorption and thickness swelling of the WPC specimens reduced as a result of ILs treatment. The SEM analysis revealed that the presence of ILs can improve the quality of adhesion between the polymer matrix and the cellulosic materials, to decrease the gaps in the interfacial region and block the hydrophilic groups. Studying the water sorption process in the specimens showed that the process follows kinetics and mechanisms described by Fick’s law. The highest water diffusion coefficients and swelling rate parameters were found in composites with untreated wood.
Keywords
Introduction
Wood-plastic composites (WPCs) are often subjected to the environmental conditions where the moisture changes rapidly. 1 Due to the uncertainties regarding WPCs stability in exterior conditions, their use is mostly limited to non-structural outdoor applications. 2 Thus, their exposure to the atmosphere or contact with aqueous media, have made it necessary to evaluate the water uptake characteristics of these materials.3–5 Water absorption and the consequent thickness swelling are the most important characteristics of WPCs exposed to environmental conditions and thus affecting their end-use applications.6–8 Therefore, as a limiting parameter, hygroscopic characteristics have to be taken into account in the design of WPCs for final applications.3–8
Chemical treatment is one of the most common and best-known methods to improve the dimensional stability of WPCs. It is well known that the interfacial adhesion between the polymer matrix and the lignocellulosic filler promoted during the chemical modification process. As functional groups such as hydroxyls are decreased, the equilibrium moisture content of wood decreases. This result is in improved dimensional stability, bio-durability.9–14 Although, the chemical treatment is known to be effective, that this method has some disadvantages in terms of degradation of lignocelluloses components, demand cooking at extreme conditions of temperatures, and pressures or strong chemical reagent bases so that special processing equipment is necessary.15–17 Also, many chemical modification methods are not “green” since hazardous and toxic chemicals are used. Therefore, the development of a new, cost-effective, and environmentally benign pretreatment technology for efficient utilization of lignocellulosic biomass in composite materials remains challenging. One promising alternative could be the use of ionic liquids (ILs).15–17 Ionic liquids are materials consisting only of cations and anions, and exist in the liquid state at room temperature. Possessing unique material properties such as low very pressure and chemical stability, they are expected to replace conventional organic materials in many applications. Moreover, ionic liquids are called “designer green solvents” as their physio-chemical properties can be tuned by selecting proper cation and anion.18–21
Considering the role of chemical treatment in WPCs manufacturing and its application in aqueous media, the objectives of this study were to investigate the effect of chemical modification of wood with ionic liquids on the moisture absorption and thickness swelling behavior of wood flour/polyethylene composites. Besides, the water diffusion coefficient and swelling rate parameter of long-term immersed WPC specimens depending on the ionic liquid treatment were determined.
Experimental
Materials
Industrial wood flour (WF) provided by Cellulose Aria Co. (Iran) and was used as the lignocellulosic filler. The particle size of WF was selected 40-mesh (425 μm) using a laboratory test sieve. Injection grade high-density polyethylene (HDPE, HD-52518) purchased from Jam Petrochemical Co. (Iran) and was used as the polymer matrix. It has a melt flow index of 18 g/10 min, a density of 0.952 g/cm3, and a molecular weight of 250000 g/mol. Some important characteristics of neat HDPE were as follows: Vicat softening point 122°C, tensile strength 24 MPa, tensile modulus 1760 MPa, elongation at break yield 11%, flexural strength 18 MPa, flexural modulus 1350 MPa, and impact strength 25 J/m. Polyethylene-grafted maleic anhydride (PE-g-MA, Epolene G-2608) as a coupling agent was obtained from Eastman Chemical Co. (Kingsport, TN, USA). It has a melt flow index of 6–10 g/10 min, an acid number of 6.5–11 mg KOH/g, and weight average of MW ≈ 65000 g/mol, as reported by the supplier. The reagents and solvents were purchased from Merck (Germany) and used without further purification.
Methods
Synthesis of ionic liquids
Two imidazole-based ILs, namely1-(3-trimethoxysilylpropyl)-3-methylimidazolium chloride (IL-Cl) and 1-(3-trimethoxysilylpropyl)-3-methylimidazolium thiocyanate (IL-SCN) were synthesized by an ion exchange reaction according to the previously described procedure. 22 The success of ILs synthesis has been confirmed by FT-IR and 13CNMR. Synthetic routes for ILs preparation are shown in Figure 1.
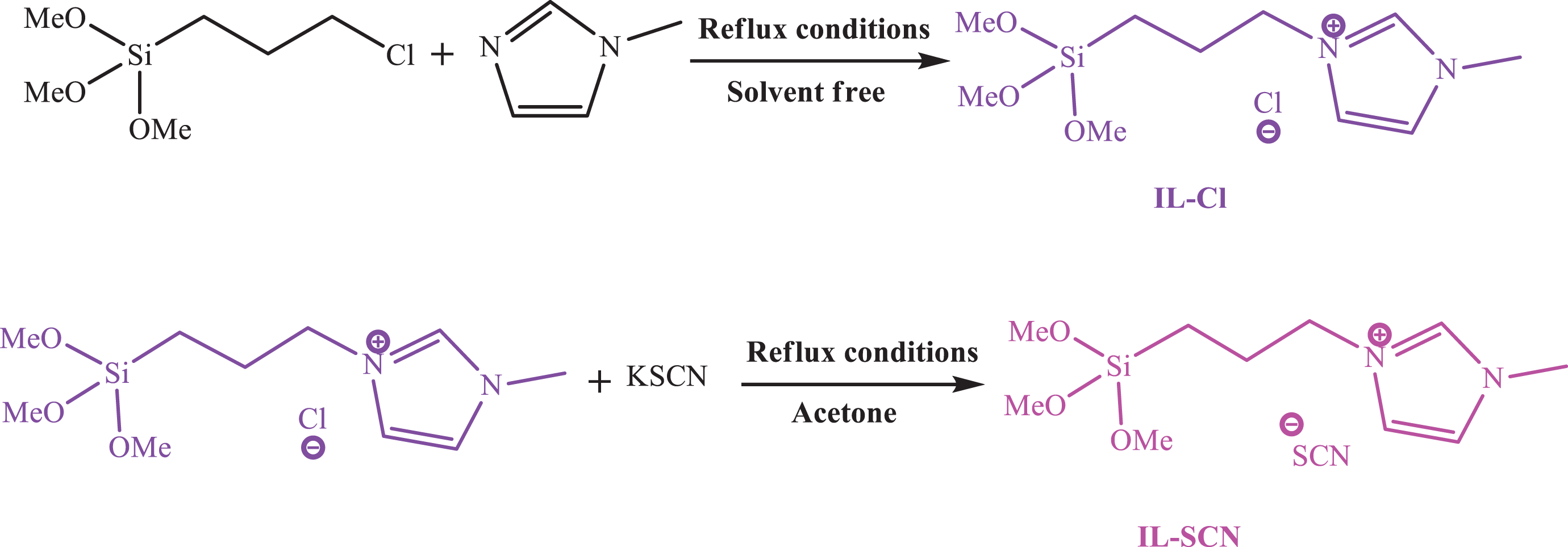
Synthesis reaction of IL-Cl and IL-SCN.
Pretreatment of wood with ionic liquids
50 g pre-heated dried wood flour and 11 g ionic liquid (molar ratio of 1:0.2) in toluene (300 mL) as solvent was added into a 1000 mL round-bottomed flask. The suspension was stirred vigorously in reflux conditions under the argon atmosphere for 4 h. Thereafter, the slurry was filtered and then ILs-treated wood was extracted with ethanol in the Soxhlet apparatus, to remove unreacted and unbounded IL chains. Eventually, ILs-treated wood was dried in the oven at an elevated temperature (80°C) until constant weight.
Compounding and composite board fabrication
The process of compounding various materials was performed in a Brabender compounder (model WHT 55, Duisburg, Germany) with an internal mixer. The mixing was carried out at 170°C with a rotation speed of 60 rpm and a blending time of 5 min. For each batch, the total starting mass of materials was 45 g. The weight ratio of WF (unmodified or modified) to HDPE was kept constant at 50% for all samples. The amount of coupling agent was fixed at 1 phr (parts per hundred of resin) for untreated samples. Following compounding, all the blends were cooled to room temperature and then granulated by using a pilot-scale grinder (Weisser, model WGLS 200, Germany). The granules were dried at 105°C for 4 h. The WPC boards were prepared by the compression molding process using a hot press (Toyo Seike Mini-test Press, model WCH, Japan) with a square steel mold and dimensions of 100 mm × 100 mm. The blends were first added into the cold mold. The platens were preheated to 170°C and minimum pressure (close to zero) was applied during the preheating step to maintain the contact between the platens and mold. The pressure was then increased slowly to 25 MPa in 2 min and held at this pressure for an additional 10 min. The mold was then removed from the hot press and cooled to room temperature in a separate cold press under the same pressure (2 MPa). The target thickness of the plaques was 2 mm. Finally, the WPC boards were conditioned at a temperature of 23 ± 2°C and relative humidity of 50 ± 5% for at least 40 h, according to ASTM D618 before testing.
Characterization
Fourier transform infrared–attenuated total reflection (FTIR-ATR) spectroscopy measurements were determined in the range of 400–4000 cm−1 with a resolution of 4 cm−1 using a Bruker Vertex 70 spectrometer. Elemental analysis was performed by a Vario EL III CHNS/O analyzer (Heraeus, Germany).
Specimens with a dimension of 20 × 20 × 20 mm were cut for the water uptake test under ASTM D7031. Five specimens of each sample were dried in an oven for 24 h at 102 ± 3°C. The weight and thickness of dried specimens were measured to a precision of 0.001 g and 0.001 mm, respectively. Then they were placed in distilled water and kept at room temperature for 2800 h. At predetermined time intervals, the specimens were removed from the water and, the surface water was wiped off using blotting paper and their wet mass values and thickness were measured. The values of water absorption and thickness swelling were calculated using equations (1) and (2), respectively.

where

where
Scanning electron micrographs of untreated and ILs-treated samples were taken with a Tescan Mira 3 XMU (Czech Republic) scanning electron microscope with field emission gun (FEG–SEM) with an acceleration voltage of 15 kV. All samples were sputter-coated with gold to a thickness of 25 nm before microscopic examination.
Results and discussion
Spectroscopic analysis
Successful modification of wood surfaces with ILs was evidenced by FTIR spectra. For untreated wood (Figure 2(a)), the peak at 1043 cm−1 is mainly from the stretching vibration of C–O groups. Peaks at 1626 cm−1 and 3434 cm−1 are attributable to O–H stretching and O–H bending vibrations, respectively. The peak appeared at 1741 cm−1 is due to the stretching vibration of the C=O group, originated from the acetyl groups of galactoglucomannan and aromatic/conjugated aldehydes and esters.23–25 Peaks at 2856 cm−1 (symmetrical stretching), and 2924 cm−1 (asymmetrical stretching) are corresponded to the C–H group, respectively. Both FTIR patterns of wood treated with IL-SCN (Figure 2(b)) and with IL-Cl (Figure 2(c)), exhibited all of the peaks corresponding to the parent untreated wood. Peaks at 940–1200 cm−1 are ascribed to Si–O and Si–O–C vibration, respectively. The peak at 1628 cm−1 represents mainly the stretching mode of C=N groups. Besides, the appearance of this peak may contain C–H vibration (symmetrical and asymmetrical), which is due to the saturated alkyl chains of the ILs. The higher intensity of peak at 1628 cm−1 for ILs-treated wood in contrast with the peak of the untreated wood confirms the anchoring of the 1-(3-trimethoxysilylpropyl)-3-methylimidazolium moieties on the wood surface. The position and intensity of peak at 1740 cm−1 sustain unchanged with the incorporation of ILs (Figure 2(b) and (c)). The sharp and single peak at 2060 cm−1 in Figure 2(b), corresponds to the C–N stretching vibration of [SCN] anions. However, minimizing the intensities of the hydroxyl signals around 3434 cm−1 because of the hydroxyls chemical modifications with ILs would be expected in treated woods, create new hydroxyl groups through the silanization process in these samples, leads to broadening of the assigned peaks at this area.
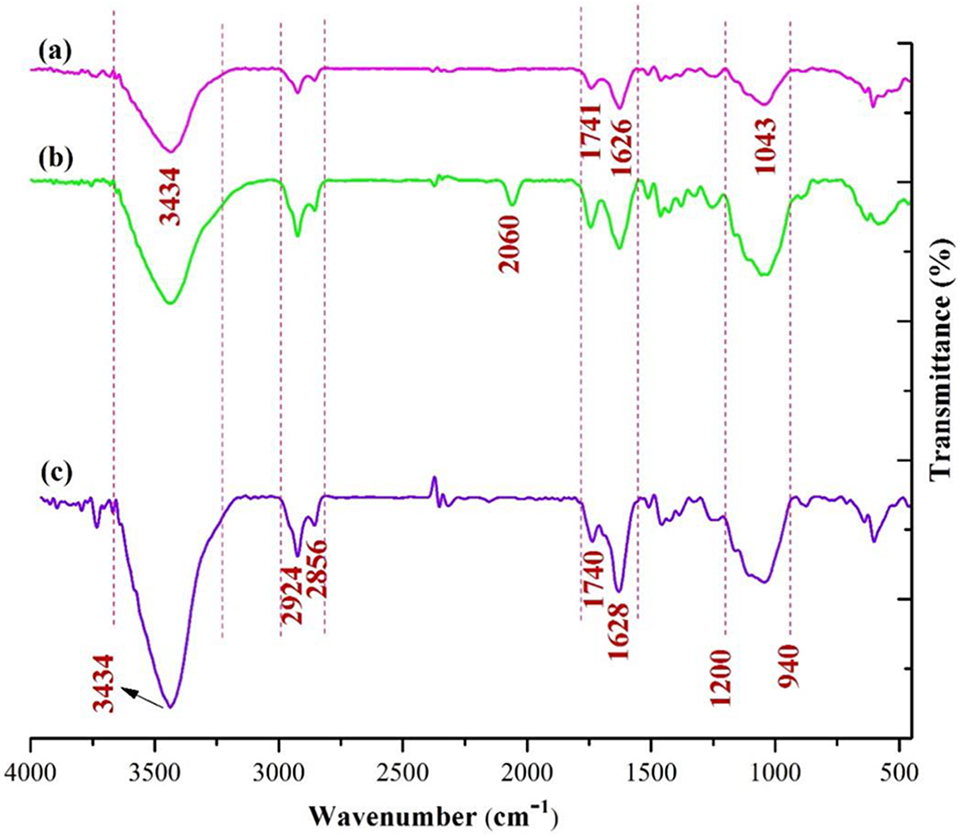
FTIR spectra of (a) untreated wood, (b) wood treated with IL-SCN, and (c) wood treated with IL-Cl.
Elemental analysis
The amount of ILs anchored on the wood surface was evaluated from elemental analysis. The results demonstrated that the loading of 0.53 mmol/g IL-SCN on the wood surface, corresponding to the S and N contents of 1.68% and 2.21%, respectively. The loading of IL-Cl substituted on the wood surface was found to be 0.44 mmol/g, with the N content of 1.22%.
Water absorption
The long-term water absorption curves of the WPCs depending on the ILs-treatment of the wood are illustrated in Figure 3, where the percentage of water absorption is plotted against time for all the specimens. It is seen that generally water absorption increases with immersion time. This trend continued up to where no more swelling was attained. The time to reach the saturation point was not the same for all the WPC formulations. As can be seen in Figure 3, those composites which were treated with ILs, absorbed less water compared to the untreated samples. This could be possible due to the better adhesion between matrix and cellulosic materials, the velocity of the diffusion processes decreases since there are fewer gaps in the interfacial region and also more hydrophilic groups as hydroxyls are blocked by applying ILs-treatment.16–17 Also, the hydrophobic nature of ILs could act as a supplementary barrier for water penetration into the specimens. 22
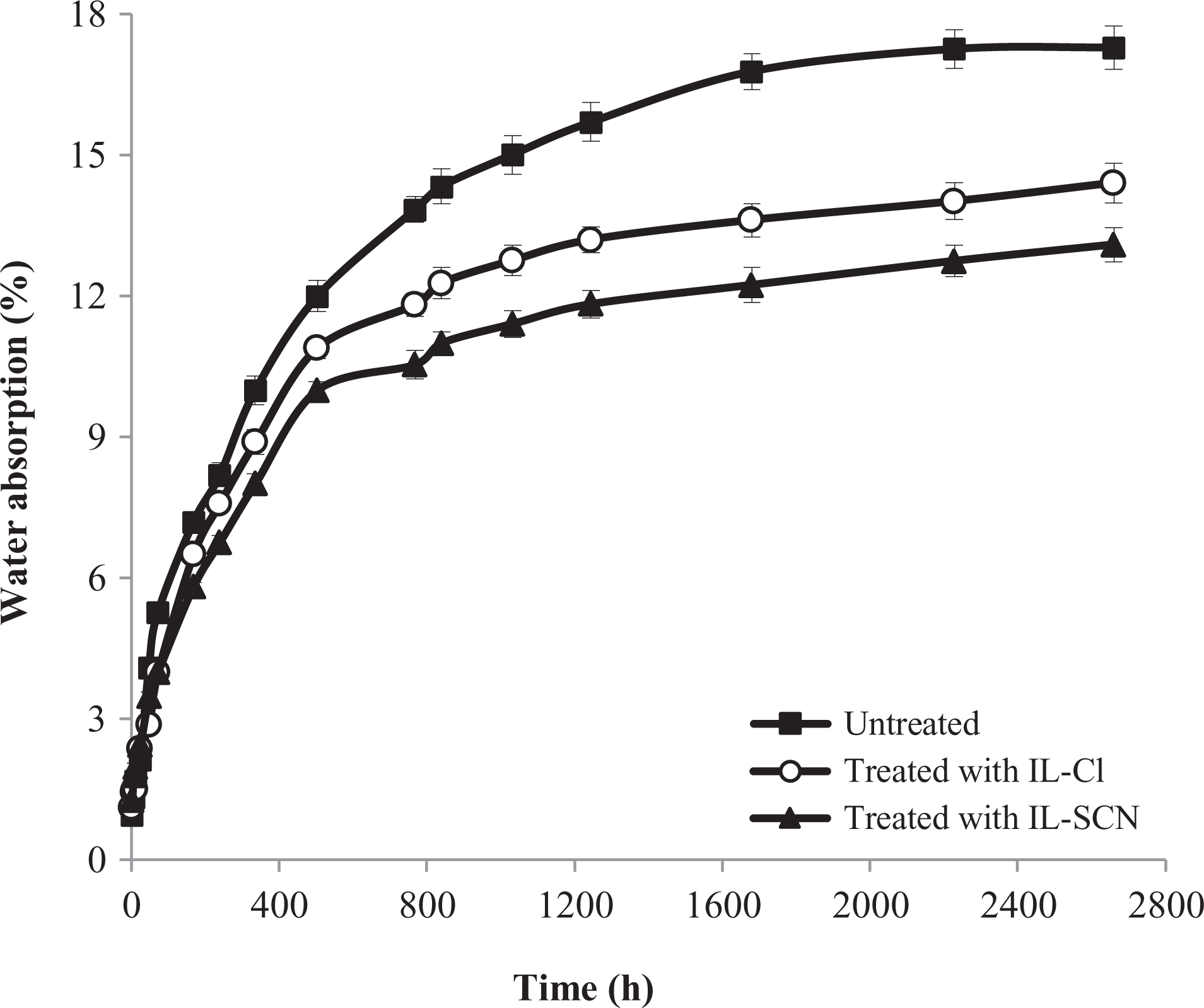
Effect of ILs-treatment on the long-term water absorption of WPC formulations.
A comparison between IL-SCN and IL-Cl with the same silanoxy alkyl imidazolium cationic centers, the difference in the size and the nature of the anions, induces alteration in the water uptake of the specimens. In the case of chloride anion [Cl]−, a higher hydrogen bond affinity toward the proton of the hydroxyl groups on the wood surface comes from the small size and greater negative charge density.26–28 This behavior keeps anions from good miscibility with the polymer matrix and as a result, hinders from the proper interfacial interactions. However, in the case of thiocyanate anion [SCN]−, the delocalization of negative charge on the greater surface of the linear molecule decreases the negative charge density 28 and could induce the attached ILs on the wood surface to have more miscibility with the polymer matrix. Moreover, a difference in the anchoring degree between both IL could be also related to the better interface compatibility in IL-SCN.28–30 The maximum water absorption value of untreated samples could be decreased from 17.34% to 14.45% and 13.18%, respectively, by applying the treatment of wood with IL-Cl and IL-SCN.
The SEM analysis revealed that the void content in the composite decreased after ILs-treatment (Figure 4(a) to (c)). Consequently, the fewer voids resulted in slow penetration of water into the depth of the composites. Furthermore, the presence of fewer voids helps to makes the WPC much less accessible for the moisture uptake due to decreasing of hydrogen bonds between hydroxyl groups of wood flour and water molecules. The ILs-treatment changes in the cell morphology and chemical structure decreased the water absorption in the WPCs with the filling of the porous structure by encapsulation the fillers in the matrix (strong bonding at the interface). Besides, it seems that the void volume in the composite material system which was affected by ILs-treatment could decrease the capacity for accommodating fewer water molecules and thereby decreased the hygroscopic behavior.
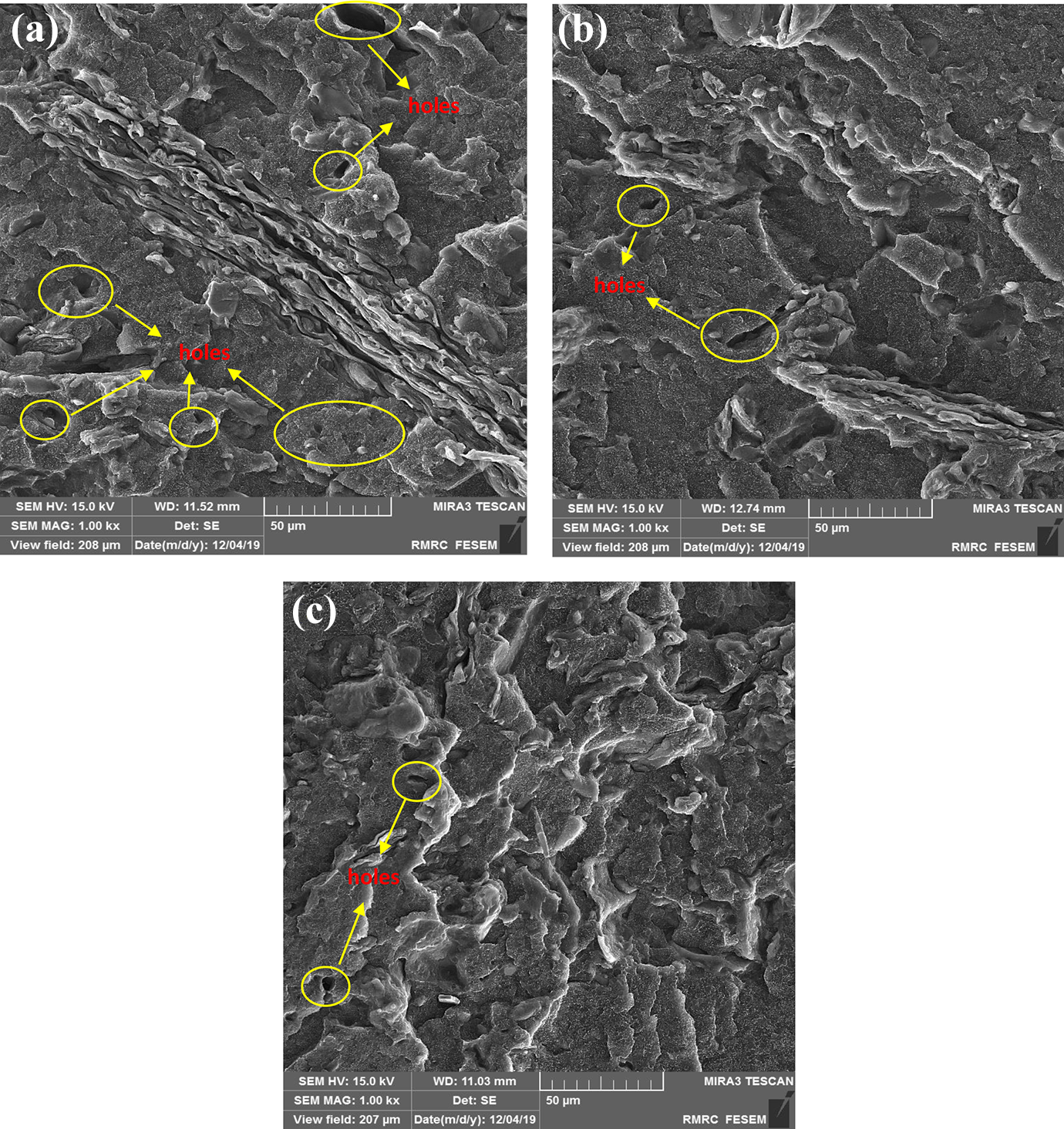
FE-SEM micrographs of WPCs with (a) untreated wood, (b) wood treated with IL-Cl, and (c) wood treated with IL-SCN.
In general, the water transport in natural fiber-reinforced polymer composites is governed by three significant mechanisms including: diffusion, capillary, and transport of water molecules, respectively. 31 The diffusion mechanism occurs inside the micro-gaps between the chains of polymers. The capillary transport mechanism occurs in the voids, micro-cracks, and gaps at the interfaces between fibers and the polymer matrix. The transport of water molecules occurs through the profusion of hydroxyl groups in natural fibers.31–33 According to these mechanisms, there are three cases of water transport behavior in the polymer composite systems that can be classified as: Fickian diffusion, relaxation controlled, and non-Fickian or anomalous. 31 These three cases can be distinguished theoretically by the shape of the sorption curve represented by equation (3).

where
To investigate the water diffusion behavior, an analysis of the kinetics of water absorption was performed via an empirical fit of the exponential model to the experimental sorption curves. A rearrangement of equation (4) yields:
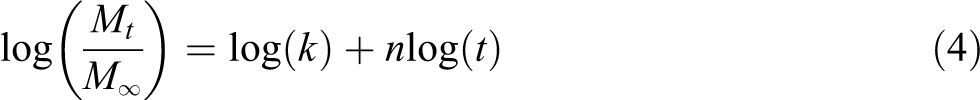
The coefficients (
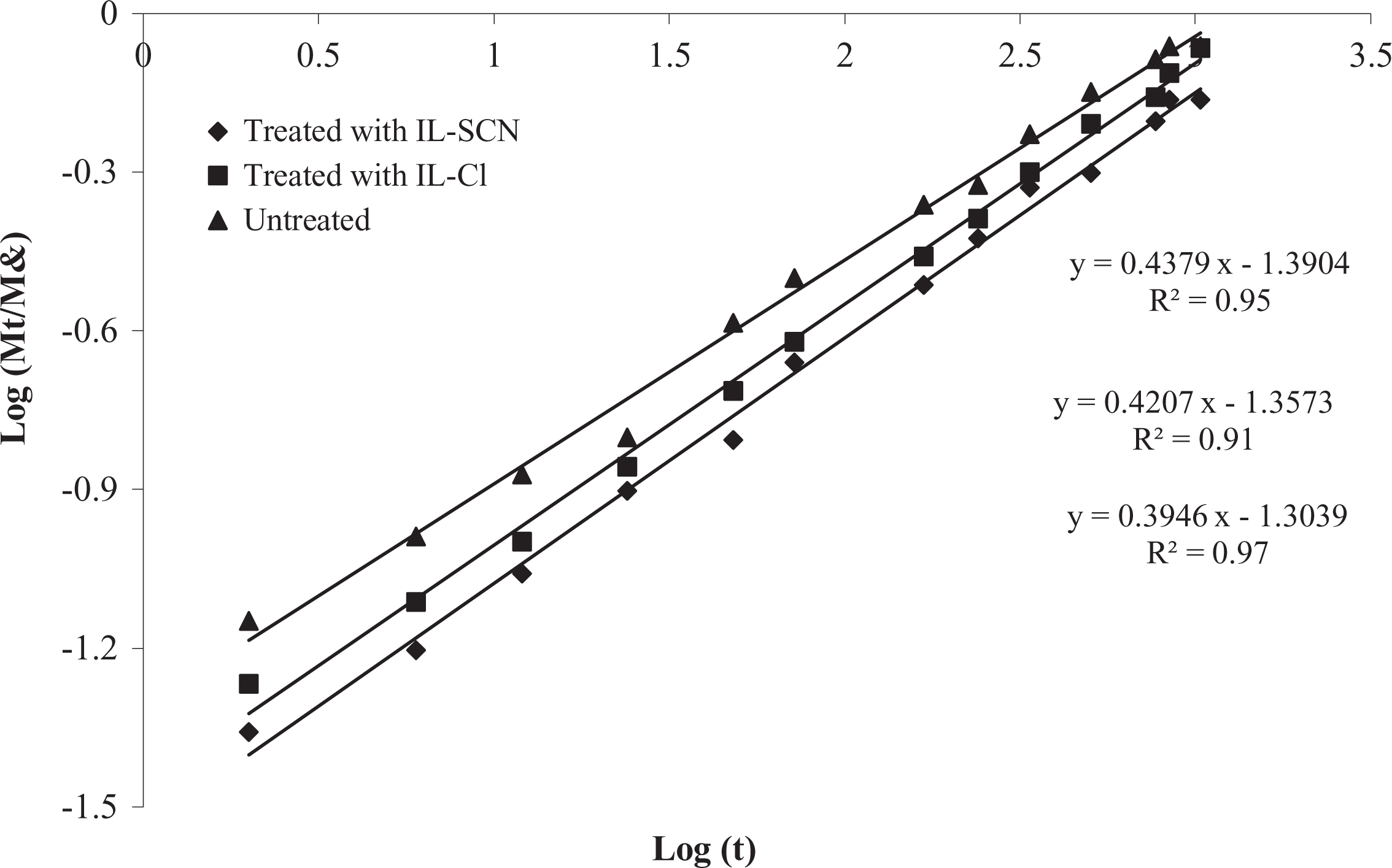
Effect of ILs-treatment on the diffusion case fitting for WPC formulations.
The values of
Water diffusion,
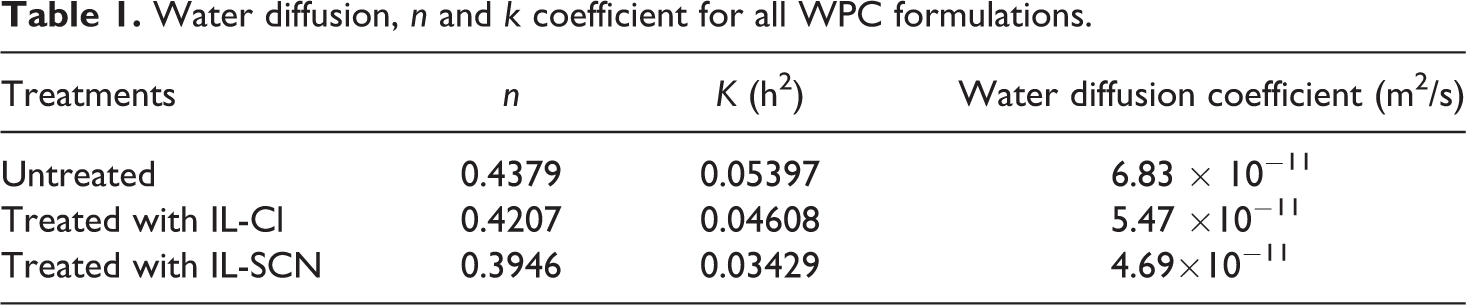
The water diffusion coefficient is the most important parameter of Fick’s model and shows the ability of water molecules to penetrate inside the composite structures. At early stages and small times (typically

where
The water diffusion coefficient for the WPCs was plotted as
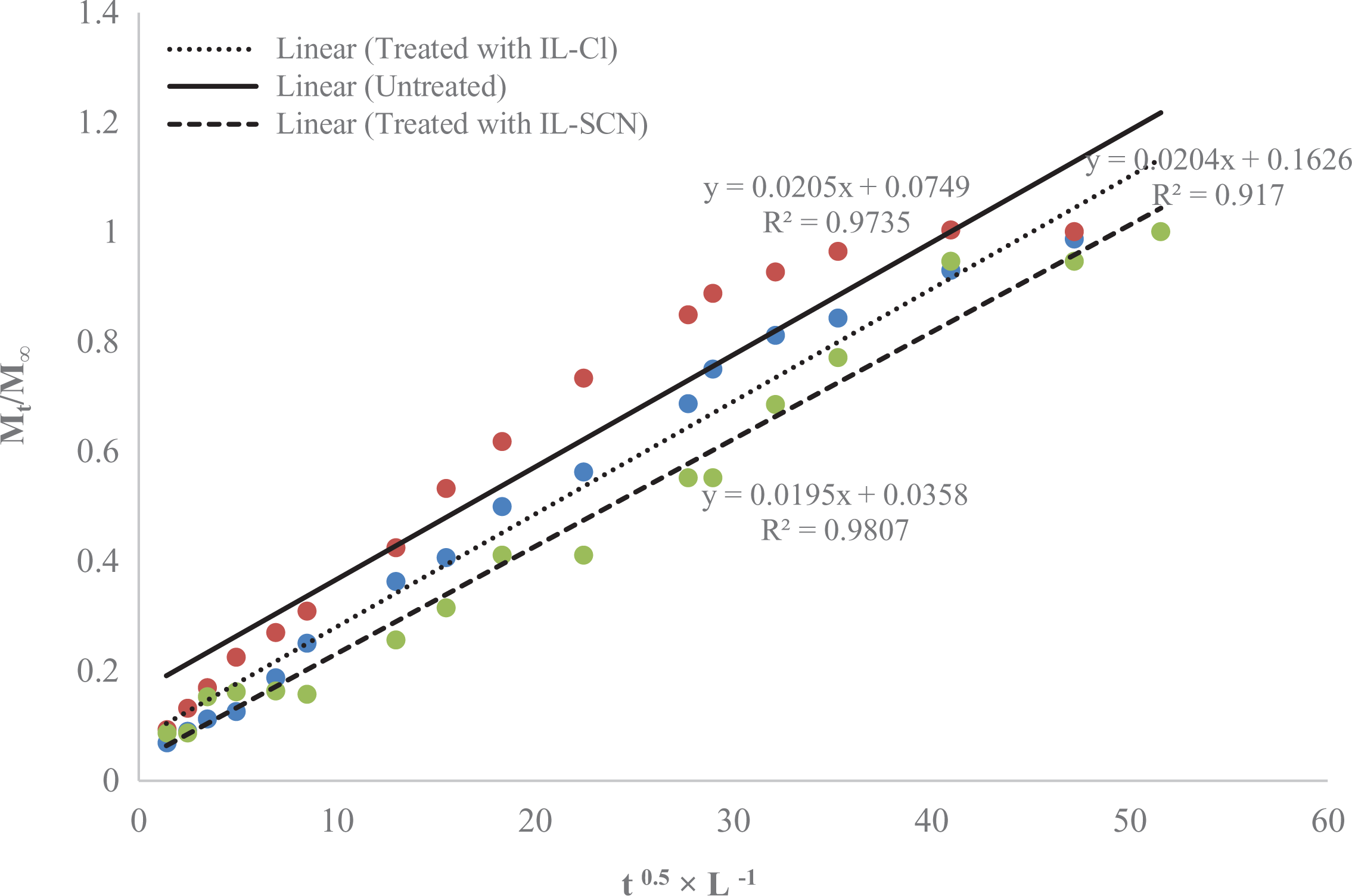
Effect of ILs-treatment on the water diffusion coefficient for WPCs.
Thickness swelling
The effect of chemical modification wood with ionic liquids on the long-term thickness swelling behavior of WPCs is presented in Figure 7. As can be seen, the thickness swelling of the specimens was reduced by applying ILs-treatment. Variations in thickness swelling of the composites are similar to variations in water absorption. The maximum thickness swelling of untreated samples was 3.91%, which was declined to 2.78% and 2.16% for treated samples with IL-Cl and IL-SCN, respectively.
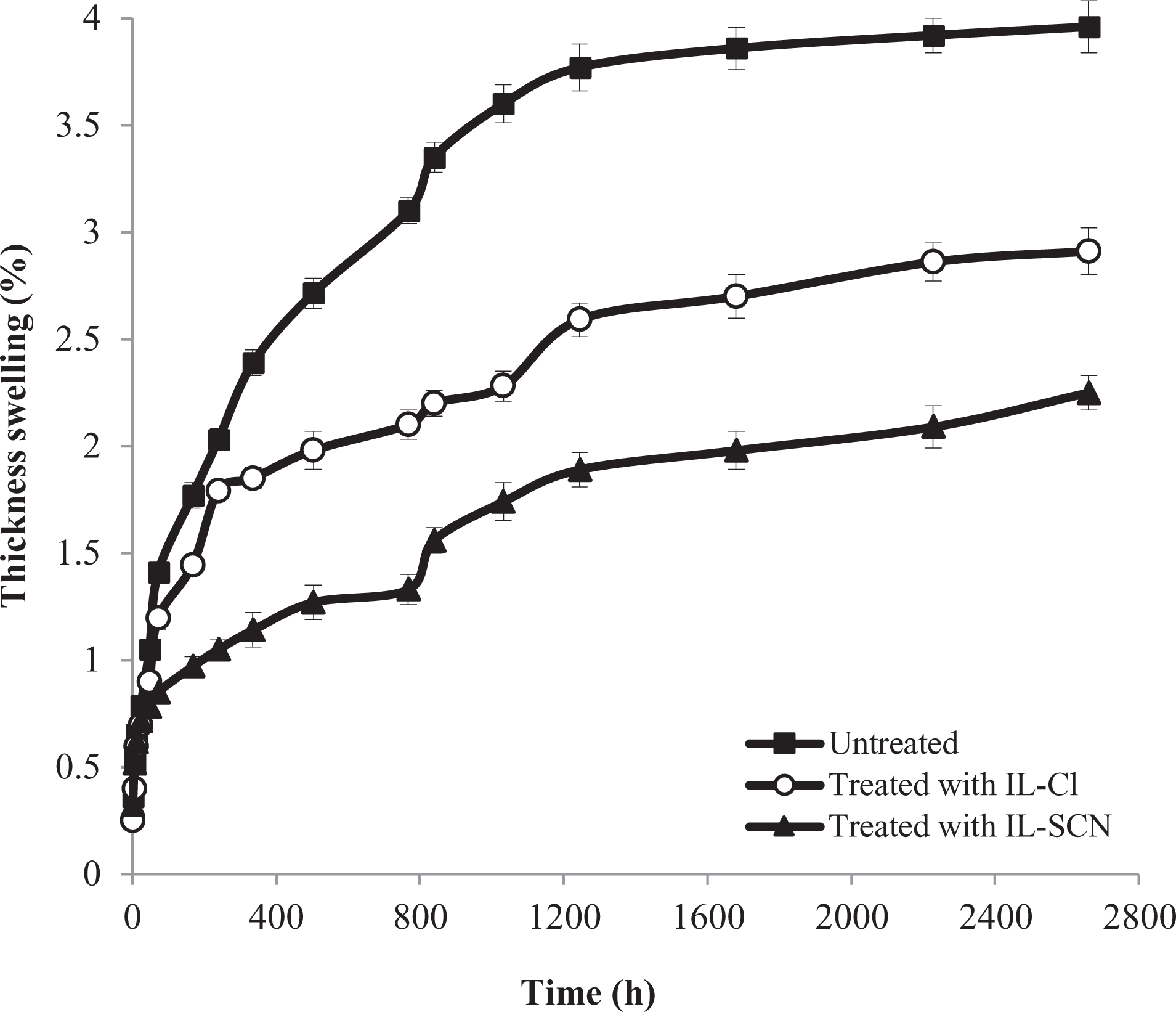
Effect of ILs-treatment on the long-term thickness swelling of WPC formulations.
For more convenient comparisons, the thickness swelling rates of composites were quantified by the model described and developed by Shi and Gardner.
34
In this model, a swelling rate parameter
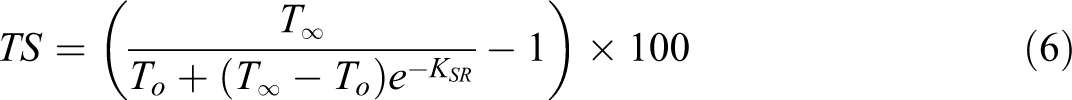
where
Rearranging equation (6) and taking the natural logarithm of both sides gives:
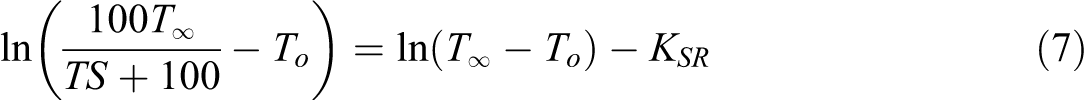
This would give a straight line with the slope of
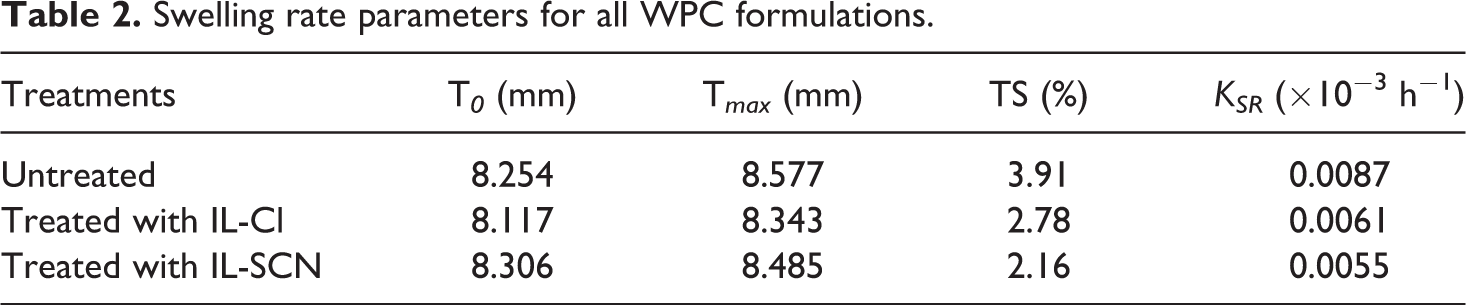
The swelling rate parameter (
Swelling rate parameters for all WPC formulations.
Coefficients of moisture expansion (

where
The
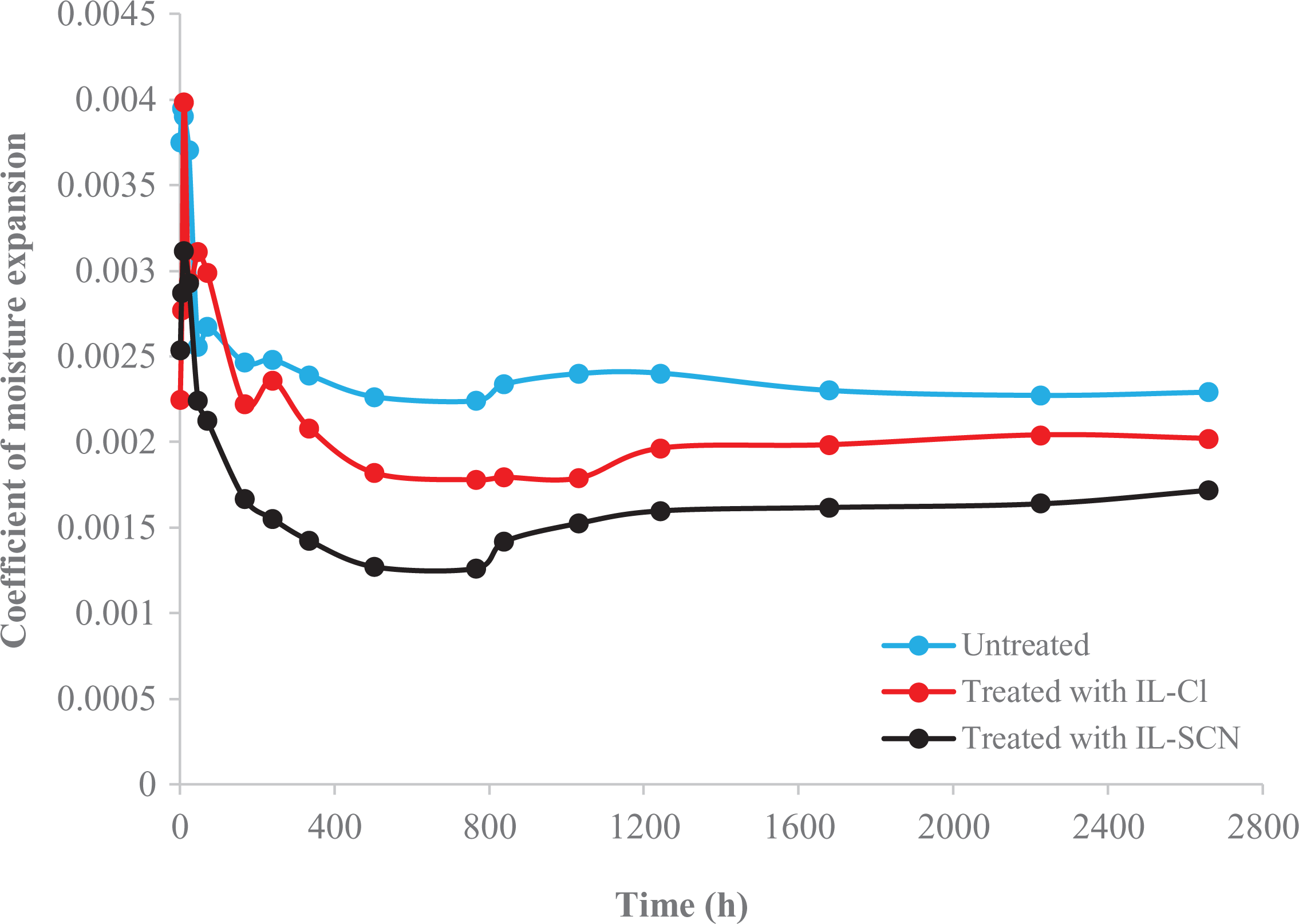
Effect of ILs-treatment on the coefficients of moisture expansion (
Conclusions
The main conclusions drawn from this study are as follows: The chemical treatment with ILs enhanced water resistance and dimensional stability in the studied formulations. The moisture absorption processes for all composite specimens were found to exhibit Fickian diffusion behavior. The highest equilibrium thickness swelling and swelling rate parameter ( SEM micrographs showed that the void content in the composites reduced as a result of ILs treatment.
