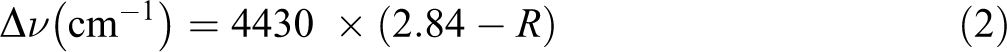Abstract
Biodegradable poly(butylene succinate) (PBS)/Alfa fiber biocomposites were prepared through the compression molding method. Scanning electron microscopy images were acquired to assess the effects of reinforcement and homogenization of mixtures and to determine the characteristics of the microstructure. The rheological properties, melting, and crystallization behavior of neat PBS and its biocomposites were investigated. Regarding the thermal properties, it was observed that the presence of Alfa fibers facilitates the crystallization of the PBS matrix, which suggests that Alfa cellulose fiber acts as a nucleating agent. The rheological analysis suggests that the biocomposites show a better dynamic behavior with the addition of Alfa fibers. Indeed, the incorporation of fibers increased the complex modulus and complex viscosity of the composites. Also, increasing the percentage of fibers in the matrix induces percolation, the shift and change in the slope of Cole–Cole curve of the PBS/Alfa fiber composite compared to that of neat PBS indicate that the PBS microstructure has changed with the addition of fibers. Moreover, the improvement of biocomposites properties is believed to be largely attributable to the homogeneous dispersion of the Alfa fibers within the polymer matrix and also to the strong interfacial interactions between the two constituents.
Introduction
Over the last few decades, the use of natural resources in the development of composite materials has attracted increasing interest.1–5 In fact, both the industrial and the academic communities have been largely interested in the use of natural fiber-reinforced biopolymer composites due to low cost,6,7 low density, 8 and eco-friendliness of natural fibers which possess the potential to reinforce the matrix9–11 and are available in large quantities in the environment.
Biomass fibers offer many advantages and have a low impact on the environment; they possess good specific mechanical properties. 12 In addition, they are renewable resources and are naturally biodegradable. With regard to their production, these fibers can compete with synthetic fibers in many areas and can replace them to conquer new markets. 13
Alfa grass (Stipa tenacissima), also identified as esparto grass, is widely distributed in Northwest Africa and Southern Spain. 14 The use of Alfa fibers as reinforcements in composite materials has recently been reported and recommended. Several researchers15–20 have used Alfa fibers to reinforce biodegradable polymer matrices for a variety of applications such as packaging and automotive industries.
Alfa fibers are generally extracted from the stems of Alfa plant by means of a chemical extraction process using an alkaline solution.21–23 The Alfa grass consists mainly of lignin, pectin, cellulose, and hemicellulose. Alfa fibers are composed of cellulose chains which play an essential role in the reinforcement of materials.
Currently, the study of interfacial adhesion between fibers and the polymer matrix is highly recommended because plant, polar, and hydrophilic fibers have a poor compatibility with apolar and hydrophobic matrices.
Furthermore, better knowledge of the condition of fibers, during the mixing and shaping operations, is necessary for the development of these materials. The present work was carried out in that context.
The poly(butylene succinate) (PBS), a biodegradable polymer material, is an aliphatic thermoplastic polymer resin that belongs to the polyester family. It presents excellent biodegradability and has mechanical properties that are comparable to those of thermoplastics such as polyethylene, polypropylene, and polystyrene.24–26 This polymer can be prepared using injection, extrusion, compression, and lamination molding techniques.27,28 It has better thermal processing properties than other biodegradable polymer materials, 25 but its production costs are quite high. For this reason, the mixture of the polymer and plant fibers can be interesting, not only for reducing the material cost but also for the matrix reinforcement.9,29–34
The present work focuses on the development and characterization of the biodegradable composite reinforced by Alfa fibers (PBS/Alfa) through a preliminary study of the interfacial strength, between the fibers and the polymer matrix, as well as the thermal and rheological properties. Note that the study of the rheological properties of that plant fiber-reinforced composite is required in order to optimize the experimental parameters and verify the interaction and adhesion between the fiber and the matrix.
The biodegradable PBS/Alfa composites were fabricated by the compression molding method, and the effect of fiber addition on the thermal and rheological properties of the composites was investigated as well.
Experimental
Materials
Commercial-grade PBS was purchased from Anqing Hexing Chemical Co., Ltd (Anhui, China). Its average molecular weight (Mw) is 1.29 × 105 Da and relative density 1.26. Alfa fibers were extracted from the Algerian esparto grass. The Alfa plant was harvested in the month of October and was subjected to several pretreatments in the laboratory; the stems were dried in an oven for 24 h at 60°C, soaked in salted water for 24 h, then washed with distilled water, and finally dried in the oven for 24 h at 60°C. Alfa fibers were extracted using a 2N sodium hydroxide (NaOH) aqueous solution; this process consisted of cooking the plant stems in the 2N NaOH solution at 100°C for 2 h under atmospheric pressure; afterward, they were bleached in a 40% aqueous sodium hypochlorite (NaOCl) solution for 1 h. The alkali treatment removed a certain amount of lignin and pectin which would limit the adhesion of fibers to the polymeric matrix. 35 The resulting fibers were abundantly washed with distilled water and dried at 60°C, for 12 h.
Afterward, during the biocomposite fabrication, the fibers were ground in order to avoid their winding and possible cutting which would lead to heterogeneity in the dimensions of the reinforcements of the material and consequently to the modification of its properties.
After 10–20 s of grinding, differences in diameters and lengths could be noticed. Then after another 30-s grinding period, the fiber dimensions were nearly identical. It seemed that the grinding (10–20 s) could reduce the length of the fibers while the second one (30 s) helped to decrease both the length and the diameter.
Preparation of the PBS/Alfa composite
The Alfa fibers and PBS were dried at 80°C in a conventional oven for 6 h before use. Prior to composite fabrication, the two ingredients were mixed with a fiber content of 2, 5, and 10 wt% at the temperature of 160°C and the mixture obtained was pulverized mechanically. The obtained powder was placed in a steel mold and then molded in a compression manner using a hot press (Carver, Inc., USA) at 160°C for 15 min.
The thickness of the resulting biocomposites was accommodated according to the specimen requirements for each analytical method.
Characterization
The optical microscopy analysis was performed on the diameter and morphology of treated and untreated Alfa fiber by means of optical microscope (Olympus Optiphot POL Polarizing Transmitted Light Microscope BX41, Tokyo, Japan). Sample picture at 10× magnitude was collected with the help of the microscopes’ camera.
Scanning electron microscopy (SEM) structure images were acquired of specimens using the scanning electron microscope Hitachi TM1000, Corporation Ibaraki, Japan. The accelerating voltage was 15 kV.
Fourier transform infrared (FTIR) spectra of all composites and neat samples were determined by FTIR measurements in the attenuated total reflectance (ATR) mode using the FTIR spectrophotometer from Agilent Technologies Cary 640 series, Ltd., Australia, with a total of 16 scans with a 4 cm−1 resolution. The samples were then scanned within the range from 4000 cm−1 to 500 cm−1.
The crystallization behavior and physical properties of neat PBS and PBS/Alfa fiber composites were determined using the differential scanning calorimeter (DSC) instrument TA Q2000, New Castle, DE, USA. The equipment was calibrated for heat flow, temperature scale, and baseline (capacitance sensor for Tzero technology) using the standard calibration procedure. Afterward, the biocomposite samples were heated above glass transition temperature (Tg) at 10°C min−1 up to the temperature of 130°C. Then, the melted samples were cooled down to −70°C at the rate of 10°C min−1; this was the first cycle. In the second cycle, the samples were heated again at 10°C min−1 up to 130°C.
The melt rheological measurements were carried out using a rheometer (Discovery Hybrid DHR2—TA Instrument). The frequency sweeps were from 600 rad s−1 to 0.1 rad s−1. The measurements were carried out at 160°C, under small-amplitude oscillatory shear mode (2%) using a rheometer equipped with a 25-mm diameter parallel plate geometry. Note that the 2-mm-thick sample disks were used for all tests.
Results and discussion
Structure and morphology of Alfa fiber and PBS/Alfa fiber composites
Vegetable fibers consist mainly of cellulose, hemicellulose, lignin, pectin, and waxes. Their properties depend on the proportion of each constituent. 36
The chemical extraction of the fibers was carried out using NaOH to dissolve the noncellulosic substances such as lignin, pectin, hemicellulose, as well as the different constituents forming the reserve and the outer layer of the stem of the plant.
Surface morphology of Alfa fibers before and after alkali treatment is shown in Figure 1(a) and (b). In the figure it was observed that the untreated fiber surface (stem of Alfa) is rough, the impurities material such as hemicellulose, pectin, lignin, and waxes is removed after different steps of alkali treatment and the surface becomes smoother. The fibrillation is also found to arise as the binding materials removed as shown in Figure 1(b).

Micrograph of Alfa fiber surface morphology (a) before and (b) after alkali treatment.
After chemical treatment and the grinding of the fiber, most of the components of the plant were dissolved, leaving mainly cellulose filaments and a small amount of lignin, as illustrated in Figure 2.

SEM graphs of the fibrils of treated Alfa fiber.
The chemical composition of the fibers’ surface was analyzed by FTIR using ATR. The FTIR-ATR spectrum obtained allows analyzing the chemical structure of the stem of Alfa plant then after the alkali treatment, as shown in Figure 3.
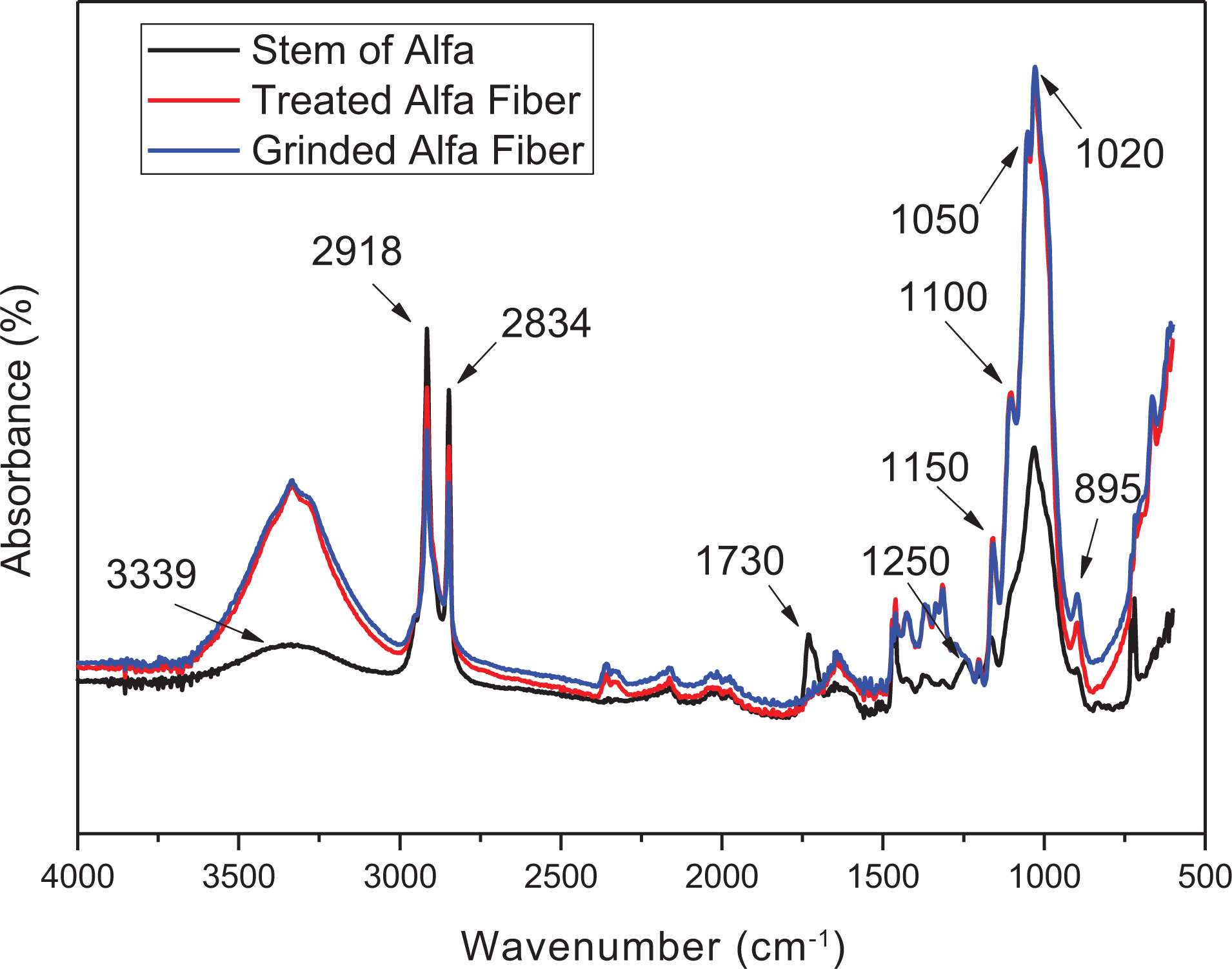
FTIR spectra of the stem of Alfa and treated Alfa fibers.
Figure 3 shows the main peaks detected. Indeed, the band at 897 cm−1 assigned as C–O–C stretching at the β-glycosidic linkage, an intense band with shoulders at 1030 cm−1 corresponding to the C–O stretching modes of the hydroxyl, the C–OH of the cellulose backbone (C–O secondary and C–O primary alcohols) corresponded to the 1056 cm−1 and the C–O–C β-glycosidic linkage bond of cellulose was detected by the stretching vibration band at 1150 cm−1. Peaks due to alcohol groups of cellulose (OH deformation) were located at 1360 cm−1 and at 1315 cm−1 corresponded to CH2 wagging of cellulose. The 1426 cm−1 band was attributed to the symmetric CH2 bending of cellulose. In addition, some saturated C–H stretching vibrations from CH and CH2 are remarked at 2850 and 2910 cm−1, beside the hydroxyl stretching broad band that is centered at 3300 cm−1. These absorption peaks are characteristic of cellulose. 37
Thus, by comparing the three spectra of the raw and treated fibers, we observe the decrease in the intensity of the band at 1460 cm−1 attributed to the lignin and the disappearance of the vibration peak at 1254 cm−1 assigned to the C–O stretching vibration of the acetyl group in lignin component. This is due to the delignification mainly by the chemical treatment and also by the bleaching step with the NaOCl solution. Indeed, NaOCl is an oxidant that reacts with the constituents of lignin, inducing its extraction of natural fibers. 38
In addition, there is a disappearance of the band at 1734 cm−1 (C=O stretching vibrations) in the treated fibers spectrum; these bands are associated to the hemicellulose. 39 This result is confirmed by the image obtained by the SEM in Figure 2, showing the fibrils of Alfa fiber obtained after the chemical treatment, the interfibrillar material should be the mixture of lignin and hemicellulose which have removed.
Furthermore, Figure 4 exhibits the ATR-FTIR spectra of Alfa stems, extracted cellulose, and commercial cellulose. One can clearly observe that the spectrum of extracted cellulose is identical to that of commercial cellulose, which suggests that the extraction operations of lignin, pectin, and hemicellulose were successfully achieved.
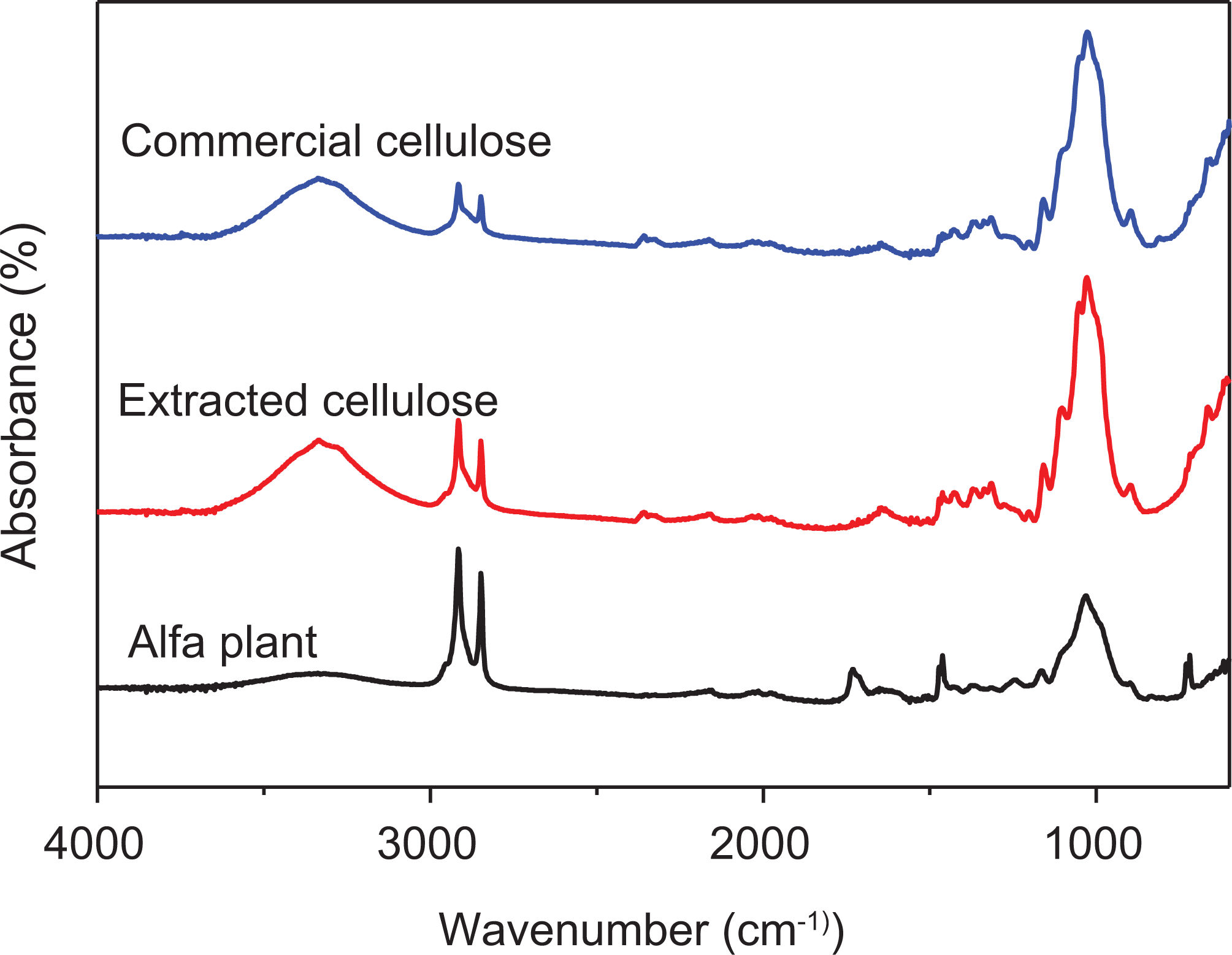
FTIR-ATR spectra of Alfa stem, extracted cellulose, and commercial cellulose.
The broad peak representing the –OH group in the region extending from 3000 cm−1 to 3400 cm−1 is intense, indicating that the number of hydroxyl groups of the cellulose increased after the fibers were subjected to alkaline treatment. 40 This is certainly due to the involvement of the hydroxyl groups in the bonds following the alkaline treatment, as shown in Figure 4. The rigidity of the fiber is generally attributed to the packing of cellulose chains and the interaction of their adjacent chains. The stronger and more numerous the hydrogen bonds are, the greater the rigidity is. 41 One may, therefore, assert that the hydrogen bonds are responsible for different characteristics of natural fibers,37,42 such as their mechanical and thermal properties.43,44
On the other hand, it is worth noting that the intramolecular hydrogen bond vibration in cellulose appears at around 3432 cm−1.37,45
The energy of the hydrogen bonds EH for several OH stretching bands was calculated using the following equation 46 :
where ν0 is the standard frequency corresponding to free OH groups (3650 cm−1), ν is the frequency of the bonded OH groups, and k is a constant (1/k = 2.625 × 102 kJ).
The hydrogen bond distances R may be obtained using equation (2), originally proposed by Pimentel and Sederholm. It is expressed as follows: 46
where Δν = ν0 − ν.
Note that ν0 is the monomeric OH stretching frequency, which is equal to 3600 cm−1.
Moreover, ν is the stretching frequency observed in the infrared spectrum of the sample.
The hydrogen bonds for the bands around 3342 cm−1 showed higher energy values (23.085 kJ) as compared to other natural fibers like curaua, jute, and kenaf, 47 which may suggest a higher number of intramolecular hydrogen bonds in cellulose. These higher energy values are certainly related to the shorter hydrogen bond distances (2.768 Å) as shown in Table 1, which may engender higher interactions between intramolecular cellulose chains. These findings are in favor of the higher hydrogen bond energy values of Alfa fibers in comparison with other vegetal fibers.
Energy EH and distance R of hydrogen bonds of Alfa stems and fibers.

It is well-known that the diameter size of natural fibers varies depending on the fiber’s length, after 30 s of grinding, differences in diameters and lengths could be noticed and this is the reason why we made a statistical measurement of the fiber diameters. Hundred fibers were chosen at random from the collection of Alfa fibers. Figure 5 shows the values of Alfa fibers diameter, which was distributed in the ranges 2–34 µm with an average value of 5 μm, this value is attributed to the diameter of the cellulose filaments which have diameters ranging from 5 µm to 10 µm and lengths ranging from 50 µm to 2000 µm with an average value of 300 μm. This large distribution of the Alfa fiber diameter is inherent in natural fibers of various origins.
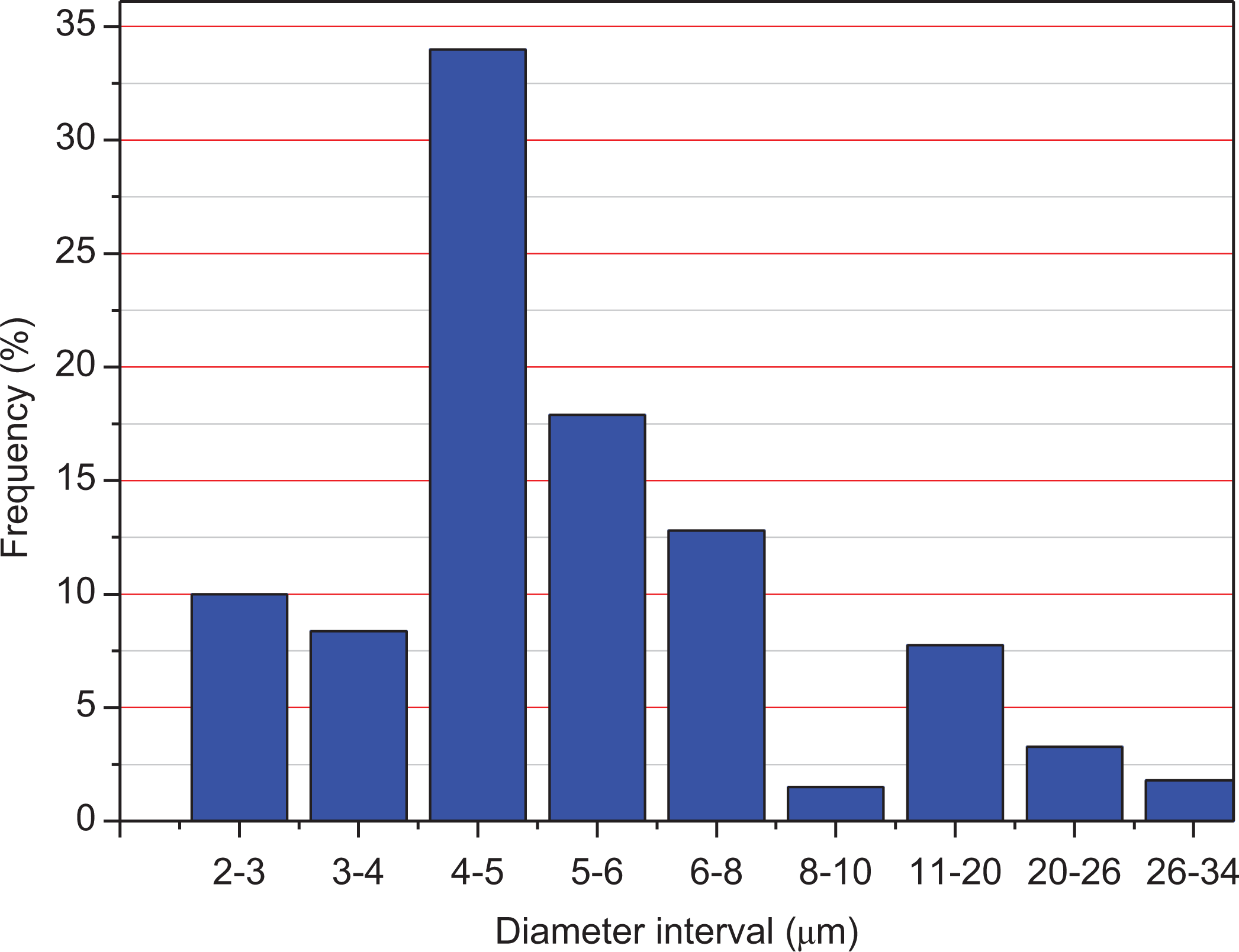
Diameter distribution of modified Alfa fiber.
The morphology of PBS/Alfa composites was analyzed by SEM as shown in Figure 6. It is observed that the PBS/Alfa 10 wt% exhibits a filamentary structure in comparison to PBS/Alfa 2 wt% which shows a smooth surface and fibers are coated by the matrix.

SEM images of the Alfa/PBS composites: (a) Alfa/PBS 2 wt% and (b) Alfa/PBS 10 wt%.
It is observed that the interface interaction between the Alfa fiber and the PBS matrix is good; Alfa fibers mainly consisted of cellulose fibrils which are uniformly embedded in the polymer matrix without the interfacial gap formation. This result was attributed to the enhanced fiber/matrix adhesion with the help of an alkaline treatment of Alfa fiber.
Physical properties
The differential scanning calorimetry (DSC) was used to investigate the effects of the addition of fibers into PBS on the physical properties of composites. The thermal characteristics including the crystallization temperature (Tc), melting peak temperature (Tm), melting enthalpy (ΔHm), and crystallization enthalpy (ΔHc) are summarized in Table 2.
Melting and crystallization behavior of neat PBS and PBS/Alfa fiber composites from DSC analysis.
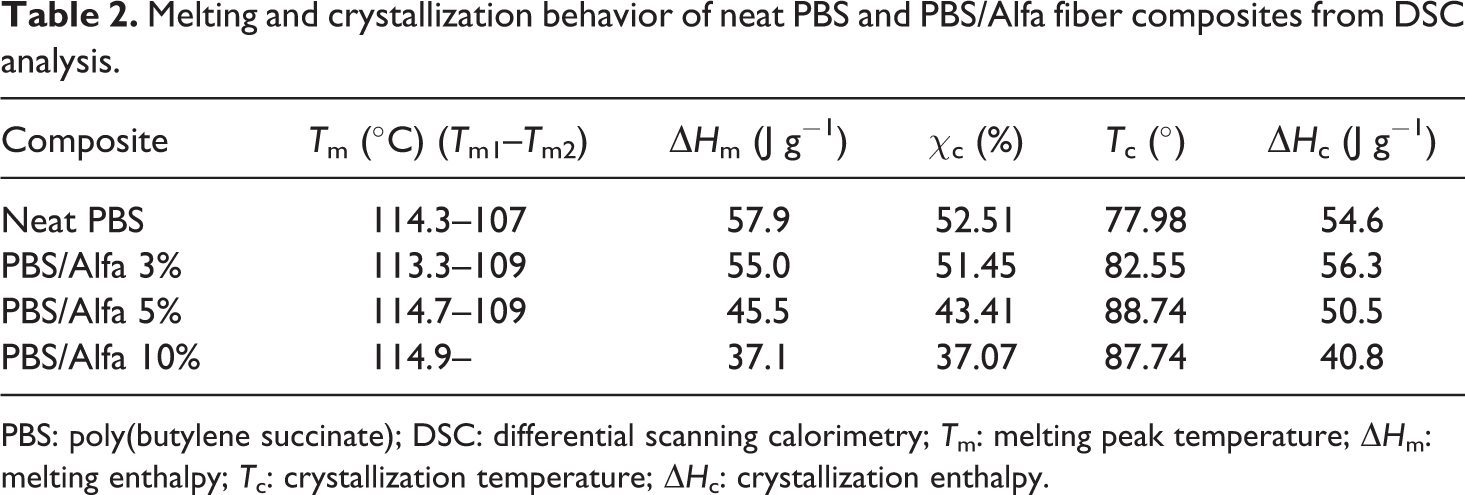
PBS: poly(butylene succinate); DSC: differential scanning calorimetry; Tm: melting peak temperature; ΔHm: melting enthalpy; Tc: crystallization temperature; ΔHc: crystallization enthalpy.
Figure 7 shows the DSC thermograms obtained during the second scan. An increase in the Tc is clearly noted depending on the percentage of Alfa cellulose embedded in the matrix.
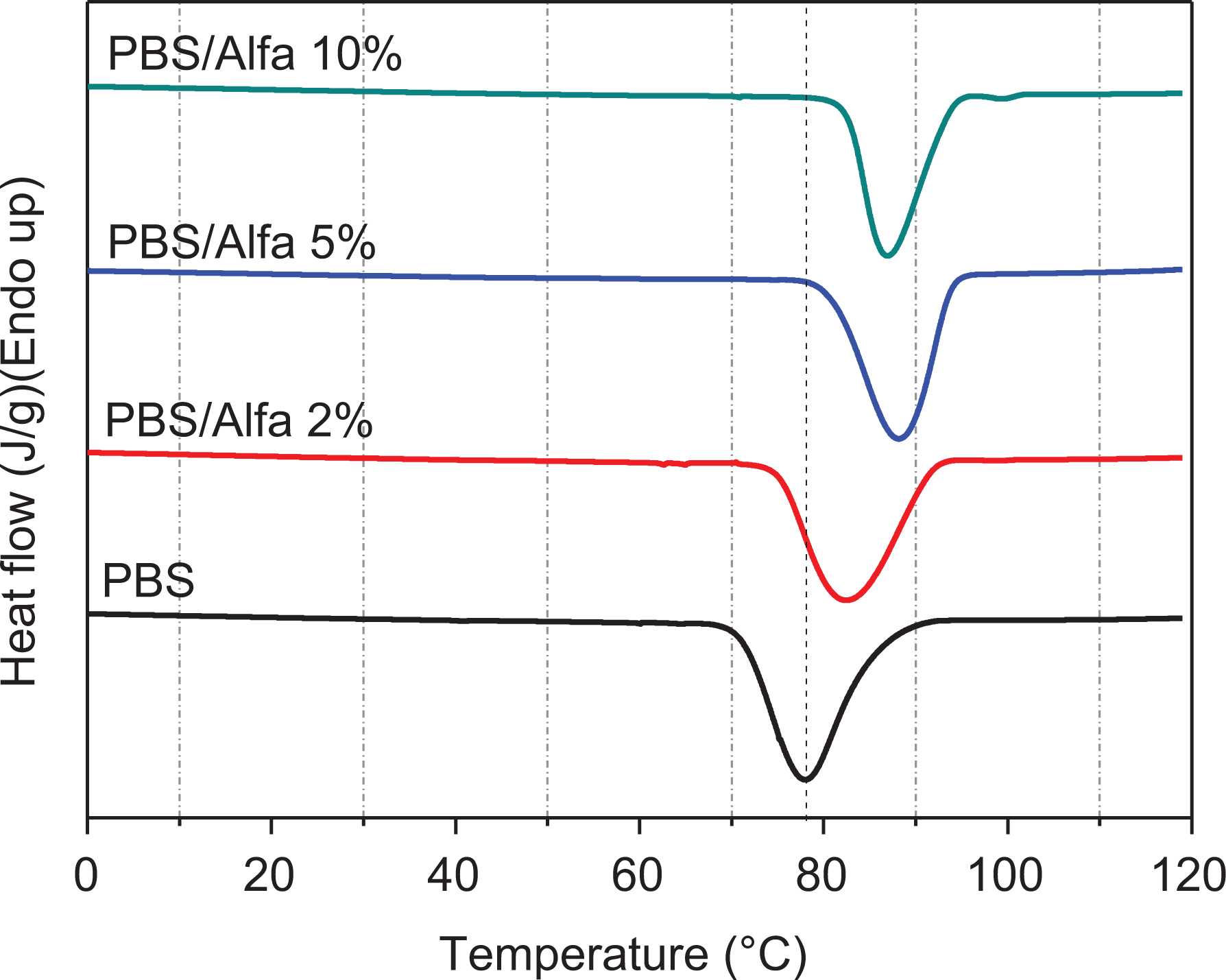
DSC cooling thermograms of neat PBS and its biocomposites.
Figure 7 indicates that the Tc value of neat PBS is about 78.10±1°C which shifts to a higher temperature of 88±1°C with the addition of Alfa cellulose, suggesting that the presence of Alfa fibers facilitates the crystallization of the PBS matrix and enhances its crystallinity. This indicates that Alfa cellulose served as a nucleating agent and enhanced the crystallization of PBS within the composites, as is most often indicated for PBS reinforced with organic fillers like fibers.26,30,48
The decline in the crystallinity rate, which can be observed in Table 1, can be explained by the fact that the crystalline integrity of the PBS matrix decreased with increasing Alfa fiber loading. This means that the incorporation of fibers increased the nucleation density but reduced the spherulite size, which led to poor crystalline integrity as a result of crystal growth confinement.
Figure 8 indicates that neat PBS exhibits two melting endothermic peaks of Tm1 and Tm2 during the melting process (low to high temperatures, respectively). Generally, the complex behavior in this melting phase may be interpreted as a melting–recrystallization–remelting process. Duplicates of melting peaks are mentioned for various semicrystalline polymers.49,50
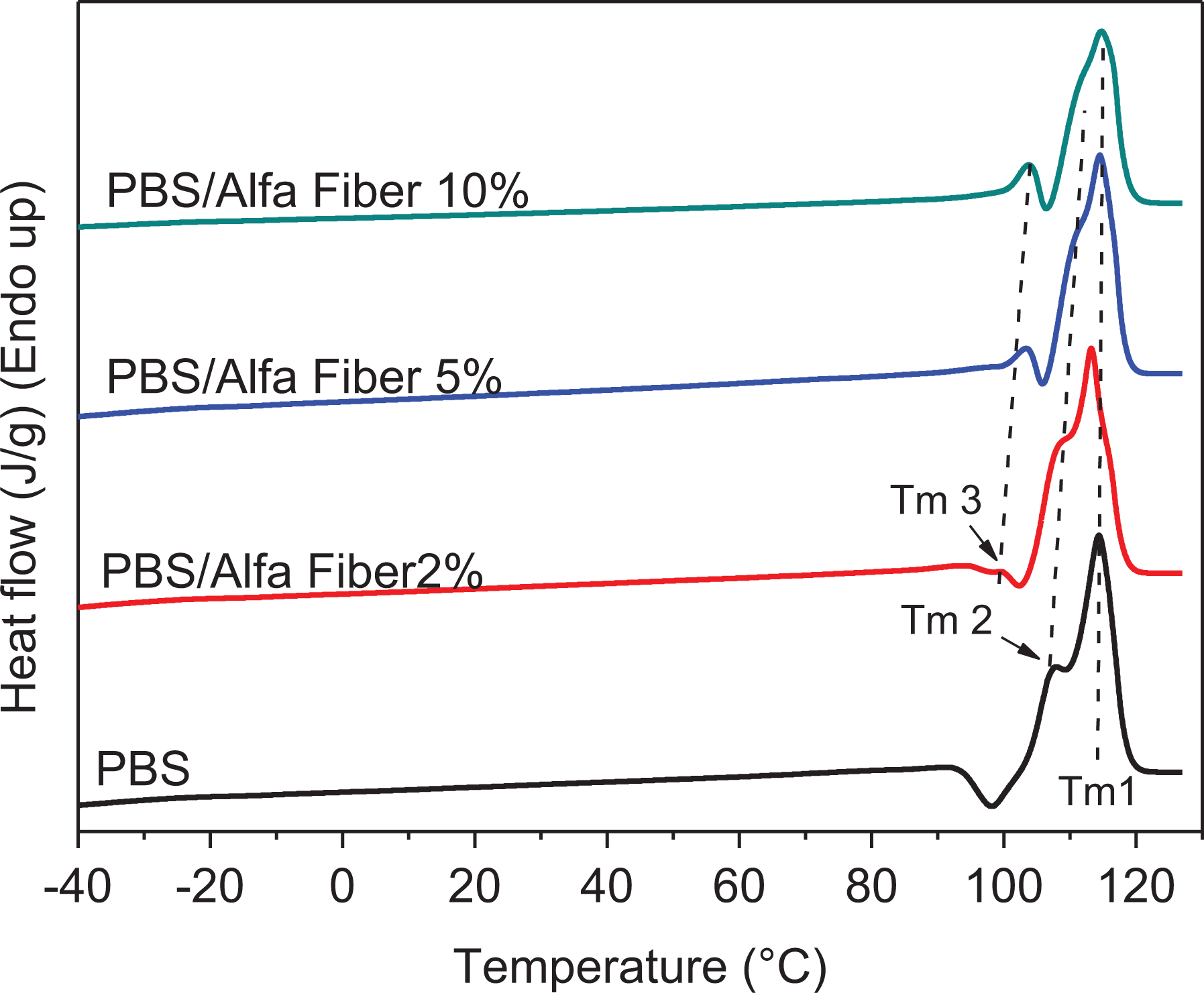
Subsequent melting with a heating rate of 10°C min−1 of PBS and its biocomposites based on Alfa fibers.
The graphs depicted in Figure 8 highlight the presence of endothermic peaks along with an exothermic peak. The temperatures Tm1, Tm2, and Tm3, ranging from the highest to the lowest, correspond to the melting peaks. The melting peak (Tm3) appears when the fiber is added to the polymer; the temperature and intensity corresponding to this peak increase as the fiber content in the composite rises. Similarly to Tm3, the melting peak corresponding to Tm2 increases with the percentage of fibers in the composite. Note that the melting temperature Tm1 remains unchanged.
Wang et al. 51 used modulated DSC and were able to highlight and confirm the above behavior which presents three fusion endotherms that are attributed to the coexistence of two phenomena, namely the presence of two different lamella distributions with different thermal stabilities (Tm2 and Tm3) and a melting-recrystallization phenomenon which appears during the heating of the polymer (Tm1). As the fiber content in the matrix increases, the melting peak (Tm1) and the recrystallization peak (Tm2) merge progressively, reflecting the fact that the crystals formed are sufficiently stable, and the melting–recrystallization phenomenon hardly takes place during heating.
Figure 8 suggests that the incorporation of Alfa fibers into PBS leads to composites that exhibit Tm2 and Tm3 values higher than those of neat PBS; in addition, the peaks corresponding to Tm1 with Tm2 are overlapping which indicates that the resulting crystals are sufficiently stable.
In general, Tm3 corresponds to the melting temperature of original crystals, 52 though they represent only a portion of the melting endotherms due to overlapping with the exotherm, as shown in Figure 8. The melting peak (Tm3) appears for PBS/Alfa fiber composites; in addition, the magnitude of this peak and its corresponding temperature increase as the fiber content in the composite goes up. The formation of original crystals may be attributed to the evolution of the crystals formed at the interfacial region between the fibers and the PBS matrix.
Rheological properties
The rheological measurements are used for controlling the quality of raw materials, for assessing the manufacturing process/final product and also for predicting the material performance. 53 Dynamic rheological analysis can provide information about the internal structure of materials, processing of polymeric composites, dispersion state of fillers, and the interaction between fillers and the matrix.54,55
The dynamic rheological properties of neat PBS, PBS/Alfa 2%, 5%, and 10% composites were determined for the purpose of characterizing the dispersion state of the Alfa fiber within the polymer matrix, the level of interaction between the fibers and the polymer matrix, as well as the melt viscosity of the composite.
Figure 9 displays the evolution of viscosity and dynamic modulus (G′ and G″) as a function of the frequency ω for neat PBS and PBS/Alfa fiber composites.
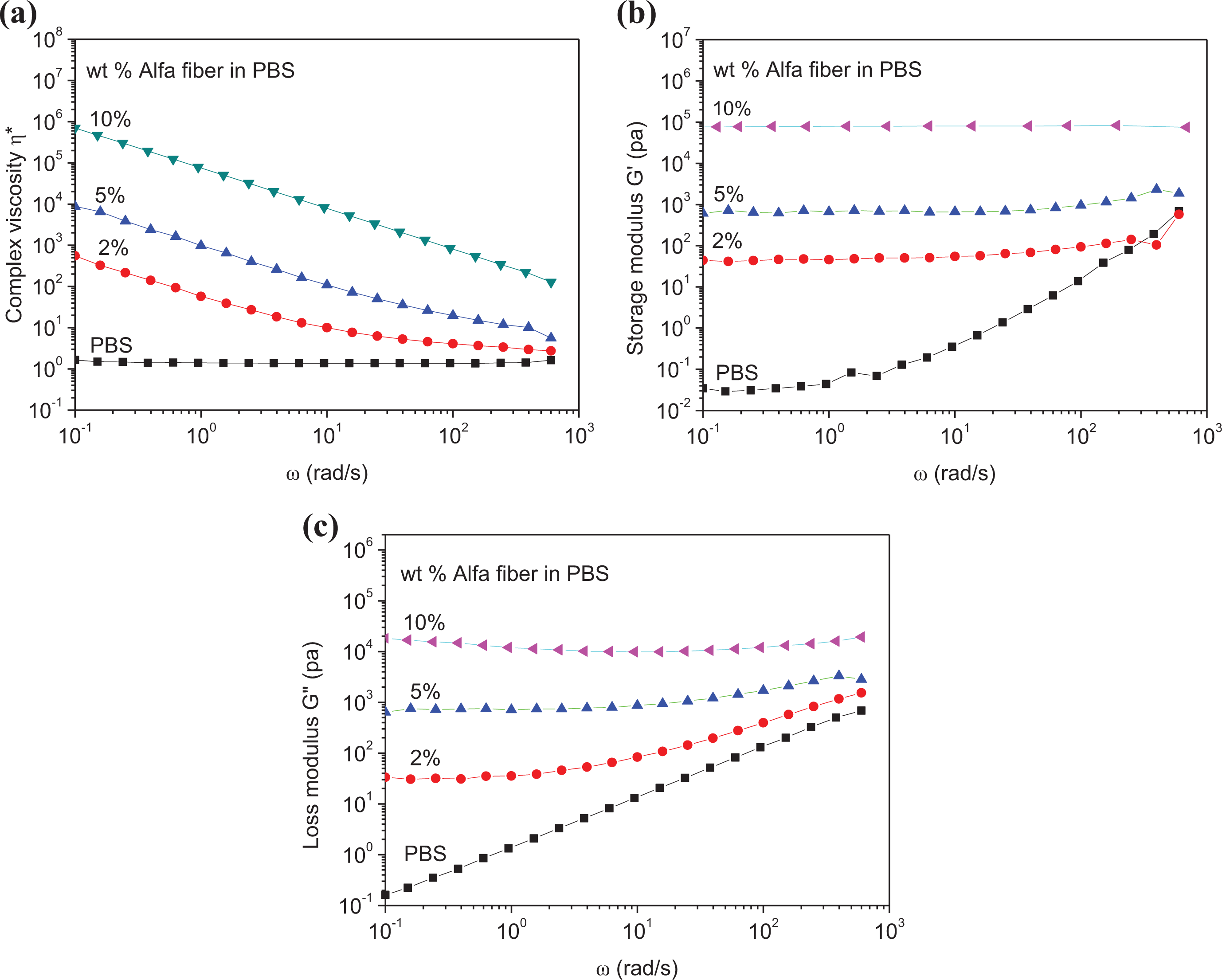
Melt rheological properties as a function of fiber content and frequency at a temperature of 160°C: (a) complex viscosity η, (b) storage modulus, and (c) loss modulus.
In view of the low viscosity of PBS, it is expected that the greater the amount of cellulosic fibers in a mixture is, the higher the viscosity of this mixture will be. One can easily check that the complex viscosity of neat PBS is much lower than that of the biocomposite materials; this observation is also true for G′ and G″ as well.
Figure 9 shows that the biocomposite containing 10% of fibers is more viscous than neat PBS even at high frequencies. Neat PBS exhibits small frequency dependence only, whereas the biocomposites present a very strong shear-thinning effect indicating that the biocomposites exhibit a good dispersion/distribution of the Alfa fiber in the PBS matrix. 56 Note that the effect of fibers is more pronounced at low frequencies. This effect diminishes with increasing frequencies due to the shear-thinning phenomenon.
Furthermore, Figure 9(b) illustrates the dependence of the storage modulus (G′) on frequency, for neat PBS and PBS/Alfa fiber biocomposites. One can see that neat PBS showed no frequency dependence, which is characteristic of a Newtonian behavior. However, the PBS/Alfa fiber biocomposites exhibited a very strong shear-thinning effect and displayed a non-Newtonian behavior in the low-frequency region. The neat PBS sample exhibits a typical terminal behavior at low frequencies, indicating a liquid-like response. However, this terminal behavior disappears gradually as the fiber loadings go up. For an Alfa fiber loading of 2 wt%, the PBS/Alfa fiber biocomposites display an evident solid-like response in the low-frequency region, indicating the occurrence of an elastic deformation-dominated flow.
Moreover, Figure 9 indicates that modulus G′ of the composites (2, 5, and 10 wt% fibers) is higher than that of neat PBS, indicating the transfer of stresses from the matrix to the fibers. In addition, one can also observe that when the frequency rises, the modulus G′ and G″ increase as well; this is certainly attributed to the response of the relaxation time of polymer chains.57,58
The variation of G′ and G″ as a function of frequency shows a plateau for low frequencies; this can be explained by the interconnected structure of fibers. Also, increasing the percentage of fibers in the matrix induces percolation. 59 The percolation network in a dynamic fluid is generally defined as a hydrodynamic structure, which is a transient structure caused by particle–particle interactions in the oscillatory process. 60 The interconnected structures lead to an apparent yield stress that is noticeable through the dynamic measurements of G′ and G″ as a function of the frequency; this is confirmed by the appearance of a plateau in the low-frequency region.
Also, the complex modulus (G′ and G″) increase was attributed to the addition of rigid fibers; this would lead to a change in the polymer chain displacement and to the modification of the molecular dynamics. 61 This finding allows confirming the good interfacial adhesion between the fibers and the polymer matrix. In addition, the removal of the noncellulosic portion of the fiber during the alkaline treatment in addition to the presence of the hydroxyl (–OH) groups in the cellulose on the surface of the fiber promoted the interfacial bonding between the fiber surface and the polymer matrix. 62 The enhancement of the interfacial adhesion had positive effects on the final properties of the composites produced.
Figure 10 presents the evolution of the storage modulus G′ as a function of the loss modulus G″, with the frequency as a parameter analogous to Cole–Cole plots used in dielectric spectroscopy.
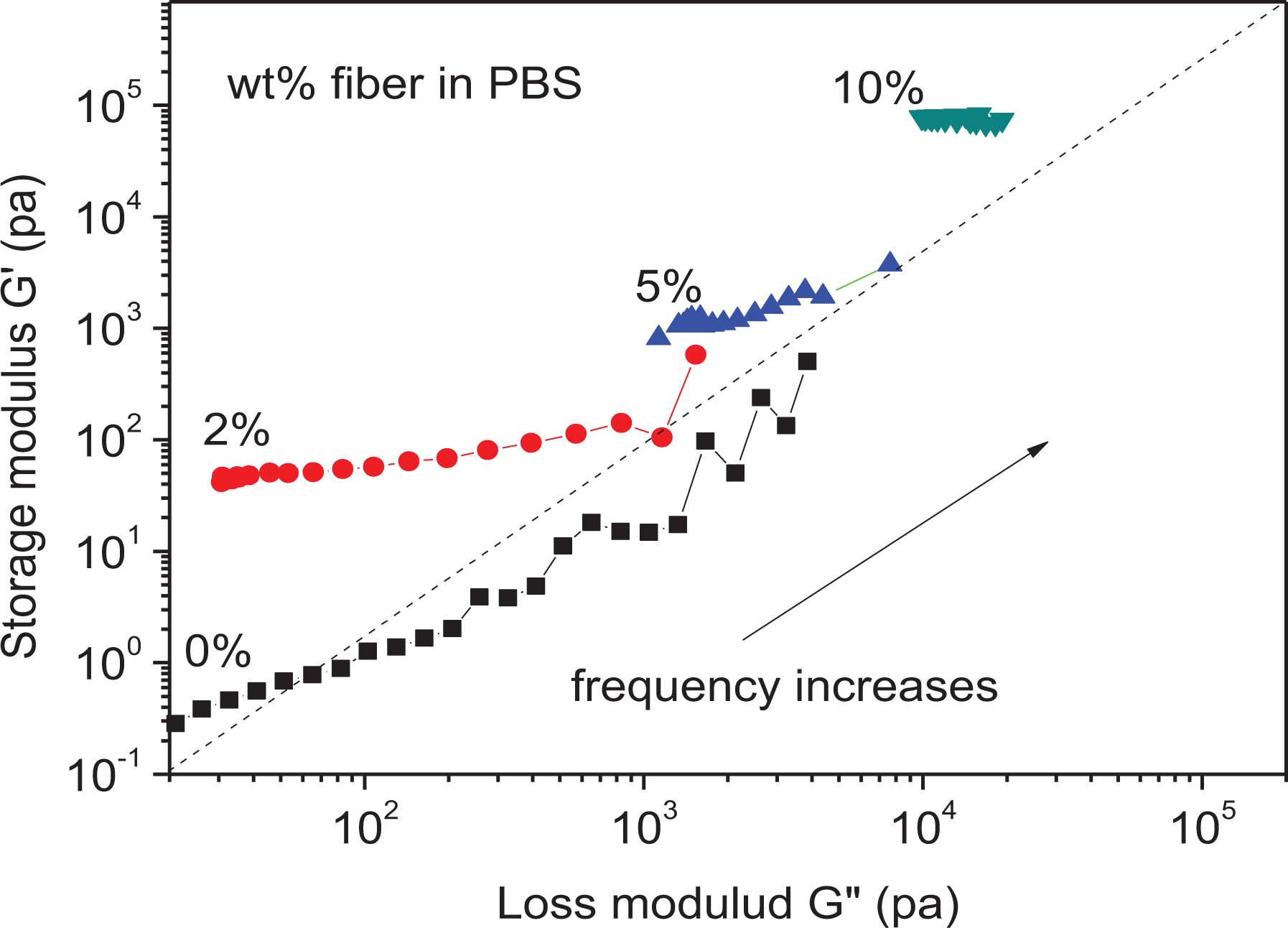
Storage modulus G′ versus loss modulus G″ of the biocomposite, at different fiber contents (wt %).
Several authors63–65 used the log G′–log G″ curve to investigate the effects of various parameters such as temperature, branching and broadening of the Mw distribution, and incorporation of fillers, on the microstructure of a polymer. It was proposed that the log G′–log G″ curves for a polymer should be similar under different conditions if the microstructure of that polymer does not change.
In this work, the log G′–log G″ curve was used to find out the structural differences between neat PBS and PBS/Alfa fiber composites at 160°C. One may easily observe that the slope of the log G′–log G″ curve decreases with increasing fiber content. The shift and change in the slope of curve of the PBS/Alfa fiber composite compared to that of neat PBS indicate that the PBS microstructure has changed with the addition of fibers; this is due to the formation of a percolation network.
Conclusion
The present work aims to elaborate and characterize a biodegradable polymer/Alfa fiber biocomposite. The mixture consists of a biopolymer, that is, PBS and cellulose fibers (Alfa fibers). The Alfa fibers were extracted from Alfa stems and were next characterized by the optical microscopy, SEM, and FTIR spectroscopy. Rheology and DSC were used for the study of the rheological and physical properties of the PBS/Alfa fiber composites.
The analysis of the thermal behavior of the composites allowed asserting that the presence of Alfa fibers facilitates the crystallization of the PBS matrix and enhances its crystallinity. This suggests that Alfa cellulose acted as a nucleating agent, which promoted the crystallization and crystallinity of PBS within the biocomposites. The decrease in the crystallinity rate can be explained by the fact that the incorporation of fibers increased the nucleation density but reduced the spherulite size as a result of crystal growth under confinement.
The Tm3 melting peak appeared with the incorporation of Alfa fibers. In addition, the intensity of this peak and the corresponding temperature increased as the fiber content in the composite went up. The Tm3 melting peak was attributed to the melting of original crystals. The formation of these original crystals could probably be attributed to the evolution of crystals formed at the interface between the PBS matrix and the fibers.
Furthermore, the rheological behavior of the composites was investigated using oscillatory rheometry, at a constant temperature (160°C). It was found that viscosity increased significantly for higher Alfa fiber concentrations. It is worth noting that the incorporation of fibers increased the dynamic properties of the composites. Also, the fibers dispersed in the molten polymer remained solid, which promoted the flow stream and consequently viscosity.
It was noted that the viscosity increased as the elastic melt complex modulus (G′ and G″) became larger.
The variation of G′ and G″ as a function of frequency shows a plateau at low frequencies, which can be explained by the interconnected structure of fibers (percolation effect).
The improvement of the composite behavior can be mainly attributed to the homogeneous dispersion of Alfa fibers within the polymer matrix and to the strong interfacial interaction between the two constituents.