Abstract
The goal of this work is to investigate the effect of incorporating organoclays on the cross-link structure, morphology, and thermomechanical properties of cross-linked organoclay/polyvinyl chloride grafted with styrene and maleic anhydride (PVC-g-(St-MA)) copolymer nanocomposites (CPN). Cloisite30B (C30B) and Cloisite15A (C15A) organoclays were used for the preparation of cross-linked PVC-g-(St-MA) nanocomposites by the solution mixing route. The nanoclay content in nanocomposites varied from 0.2 wt% to 1 wt%. The chemical structure and interaction between PVC-g-(St-MA) cross-linked segments and nanoclays were studied by Fourier transform infrared (FTIR) peaks deconvolution method. FTIR spectroscopy suggested the lowest extent of hydrogen bonding interaction for C30B containing sample, which decreased with clay content increment. The morphology of nanocomposites was studied by X-ray diffraction and transmission electron microscopy methods. Morphological observation revealed a near to the exfoliation state for organoclays in PVC-g-(St-MA) nanocomposite containing 1 wt% C30B. However, C15A/PVC-g-(St-MA) nanocomposite (C15A-CPN) exhibited partially intercalated and agglomerated morphology. Differential scanning calorimetry examination was conducted to measure the glass transition temperature (Tg) of the segments. At the same clay content, the Tg of the C30B containing nanocomposites were higher than that of C15A samples. The mechanical properties of these nanocomposites were also investigated. As a consequence, C30B-CPN showed improved mechanical properties compared to C15A-CPN and cross-linked PVC-g-(St-MA) samples.
Introduction
Polyvinyl chloride (PVC) is one of the major thermoplastics, which is extensively used in different fields such as automotive, packaging, and pipe production. This is due to its high chemical resistance, low cost, and self-extinguishability. 1 Its main drawbacks are brittleness and low thermal stability that lead to discoloration due to the formation of conjugated polyene sequences. However, PVC formulations usually comprise of various additives to improve toughness, mechanical properties, and thermal stability of the matrix. 2
Within recent years, various nanoparticles have been used not only to toughen PVC but also to improve its plasticizer migration, 3 rheological properties,4,5 thermal stability,6–8 and enhanced gas barrier properties.9,10 These improvements are obtained at lower nanoclay contents than those of traditional fillers.
One prominent nanoparticle is montmorillonite (MMT), which has proved to be an effective filler for the development of new polymeric nanocomposites.10–13 MMT clay layers carry negative charges, which are neutralized by hydrophilic, exchangeable metal ions located on the surface of the layers. An organoclay (nanoclay) is often produced through a cation exchange of its surface sodium ions by hydrophobic quaternary ammonium ions. The resulting materials consist of inorganic platelets separated by the organic modifier which are grouped to form aggregates. These materials are hydrophobic and more compatible with polymers compared to unmodified clay. These organophilic clays are then incorporated into the polymer matrix via in situ polymerization of monomer with nanoclays, solution blending, or melt intercalation under stress and shear. Each method has its own advantages and limitations.14–16
Intercalation occurs while the polymer chains penetrate among clay layers, leading to an increase in d-value. A proper exfoliated nonocomposite is obtained as all the silicate layers are delaminated and fully dispersed to the individual nanoclay platelets in the polymer matrix. 17 While a homogeneous dispersion of clay layers in a matrix is reached, it is expected that the mechanical and thermal properties of the nanocomposite to be improved compared to neat polymer and even to the conventional composite with similar clay content. 18
However, the dispersion of the nanoclays in a polymer matrix is a big challenge due to their high surface energy and large specific surface area that leads to a strong tendency for agglomeration.11,19–21 Considerable efforts have been made to identify the variables that may lead to intercalated or exfoliated structures. The most relevant variables are the types of organic modifiers, mixing methods, the structure of polymers, and cure temperature (specially for polyurethanes).22–25 As far as the mixing method is concerned, several procedures such as ultrasonication, high-shear mixing, calendering, tip sonication, and conventional stirring have been widely studied via both melt-processing and solution-based techniques.23,26,27 Since all of these parameters are related to each other, a general conclusion has not been reached yet.
According to studies reported until now, most investigations on clay/PVC nanocomposites (copolymer nanocomposites (CPN)) have been focused on plasticized formulations28–32 and little depth examination has been performed in rigid ones. Although the clay addition showed the enhanced mechanical properties, the presence of alkyl ammonium salt has been pointed out as a responsible factor for the catalytic effects on PVC degradation. Unlike hectorite, bentonite clay usually contains lattice iron and may also contain other catalytically active sites that could promote the degradation of organic compounds. The decomposition of alkyl quaternary ammonium leaves strongly acidic sites and active micromolecular matter which may catalyze the dehydrochlorination or discoloration of PVC.33,34
One of the PVC applications is as the foam materials which are often used as low-density core materials in sandwich structure composites. Among the different approaches to improve the mechanical performance of these foams, cross-linking is an effective way to strengthen up the PVC foams. One of the methods for preparing cross-linked PVC (c-PVC) foams is based on the grafting reaction of an unsaturated acidic anhydride and a monomer (such as styrene (St)) on the PVC chains, following the cross-linking reaction between acidic anhydride and isocyanate compounds. 35 On the other hand, nanoclays can play a key role in PVC foams. A small amount of well-dispersed nanoclays in PVC may serve as nucleation sites to facilitate the bubble nucleation process. Furthermore, clay nanoplatelets can reduce gas diffusivity in the polymer matrix, and the presence of nanoclays may enhance the mechanical and physical properties of PVC foams. Novel nanocomposite foams based on the incorporating nanoclays in c-PVC foam may lead to a new class of materials that are lightweight, high strength, and multifunctional. To the best of our knowledge, there is no acknowledged report about how the addition of nanoclays can affect the formation of cross-linking structure and thermomechanical properties of c-PVC foams. Since the cross-linking and foaming reactions are taking place at the same time, this work is only dedicated to the investigation of PVC with styrene and maleic anhydride (PVC-g-(St-MA)) cross-linking in the presence of nanoclays.
In this study, we examine the influence of two organically modified MMTs on cross-linking reactions of PVC-g-(St-MA) as well as on morphology and thermomechanical properties of cross-linked nanoclay/PVC-g-(St-MA) nanocomposites. At the beginning, PVC-g-(St-MA) was synthesized by grafting PVC St and maleic anhydride (MA) monomers. Two nanoclays (Cloisite15A (C15A) and Cloisite30B (C30B)), in different contents, were dispersed in PVC-g-(St-MA) matrix by solution blending. Eventually, cross-linked PVC-g-(St-MA) nanocomposites were prepared by mixing PVC-g-(St-MA) nanocomposite and diisocyanate and then immersing the casted samples in boiling water. The reactions involved in CPN preparation were investigated by Fourier transform infrared (FTIR) spectroscopy. The effect of these reactions on morphology and thermal properties of CPN was studied by X-ray diffraction (XRD), transmission electron microscopy (TEM), and differential scanning calorimetry (DSC) methods. Tensile tests were performed to characterize the mechanical properties of CPN in terms of tensile strength, Young’s modulus, and elongation at break.
Experimental
Materials
PVC (emulsion grade, E6834) with a K-value of 67–69 was provided by Arvand Petrochemical Company (Iran). St monomer, a chemically pure reagent, was purchased from Merck (Germany) and washed with 10% of NaOH solution to remove the inhibitor, then washed with distilled water, and dried by molecular sieve before use. MA monomer and toluene diisocyanate (TDI) were used as received without purification (Merck). Benzoyl peroxide (BPO) as initiator was chemically pure and recrystallized with methanol before use. Cyclohexanone, tetrahydrofuran (THF), and methanol solvents were of pro-analysis grade (Merck).
Two organophilic clays with trademarks C30B and C15A were obtained from Southern Clay Products (Gonzales, Texas, USA). Both clays were spaced out with a quaternary ammonium salt. C30B has been modified with methyl bis-2-hydroxyethyl tallow quaternary ammonium ions. The modifier concentration or cation exchange capacity (CEC) was 90 meq/100 (g clay) in C30B. C15A has been modified with dimethyl dihydrogenated tallow quaternary ammonium ions, where the tallow was similar to the one in C30B, the modifier concentration or CEC was 125 meq/100(g clay). The interlayer space (d001 basal spacing) for nanoclays obtained from our XRD experiments were 3.15 nm for C15A and 1.85 nm for C30B. In both nanoclays, the moisture content was less than 2 wt%.
Synthesis of PVC-g-(St-MA)
The graft copolymerization process was performed in a round-bottomed flask equipped with a reflux condenser under nitrogen atmosphere. At first, PVC powder was dissolved in cyclohexanone and then the solution was stirred and heated. While the desired temperature was reached, St and MA monomers and BPO initiator were added in predetermined proportions and were reacted under nitrogen purge at 95°C for 2 h. The reaction products were precipitated by methanol and washed several times with methanol and then dried in a vacuum oven at 60°C up to a constant weight. The precipitate would be a mixture of grafted copolymer of PVC-g-(St-MA), un-grafted St-MA copolymer, and polystyrene homopolymer. PVC-g-(St-MA) was extracted in a Soxhlet extractor with benzene for 24 h.
Preparation of cross-linked PVC-g-(St-MA) and nanocomposites
To prepare cross-linked PVC-g-(St-MA) sample, 0.3 g of PVC-g-(St-MA) was dissolved in 25 mL anhydrous THF under a steady flow of nitrogen in a two-necked flask. Then, 1 phr of TDI was added dropwise to the solution and stirred for 15 min. Afterward, the whole solution was poured into a glass plate and allowed to create a thin film.
As the solvent gets evaporated, a thin film of PVC-g-(St-MA) and TDI mixture was formed. Cross-linking was carried out by immersing the samples in boiling water for 5 h.
For preparing cross-linked CPN, the following procedure was conducted. Anhydrous THF was used to facilitate the dispersion of the nanoclays. Different contents of nanoclays by wt% (see Table 1) were added to THF under rapid stirring for 15 min, followed by ultrasonication for 1 h to completely disperse the nanoclays. Afterward, 0.3 g PVC-g-(St-MA) was added to the mixture under nitrogen atmosphere, and the mixture was stirred at 1000 r min−1 for 30 min at room temperature. Cross-linking was carried out at the same manner as described in the preparation of neat cross-linked PVC-g-(St-MA) sample. The samples’ acronyms are presented in Table 1. A schematic illustration of the preparation procedure for cross-linked CPN is shown in Figure 1.
Acronyms of typical neat cross-linked PVC-g-(St-MA) and cross-linked nanoclay/PVC-g-(St-MA) nanocomposite samples.

PVC: polyvinyl chloride; St: styrene; MA: maleic anhydride; TDI: toluene diisocyanate; C15A: Cloisite15A; C30B: Cloisite30B.
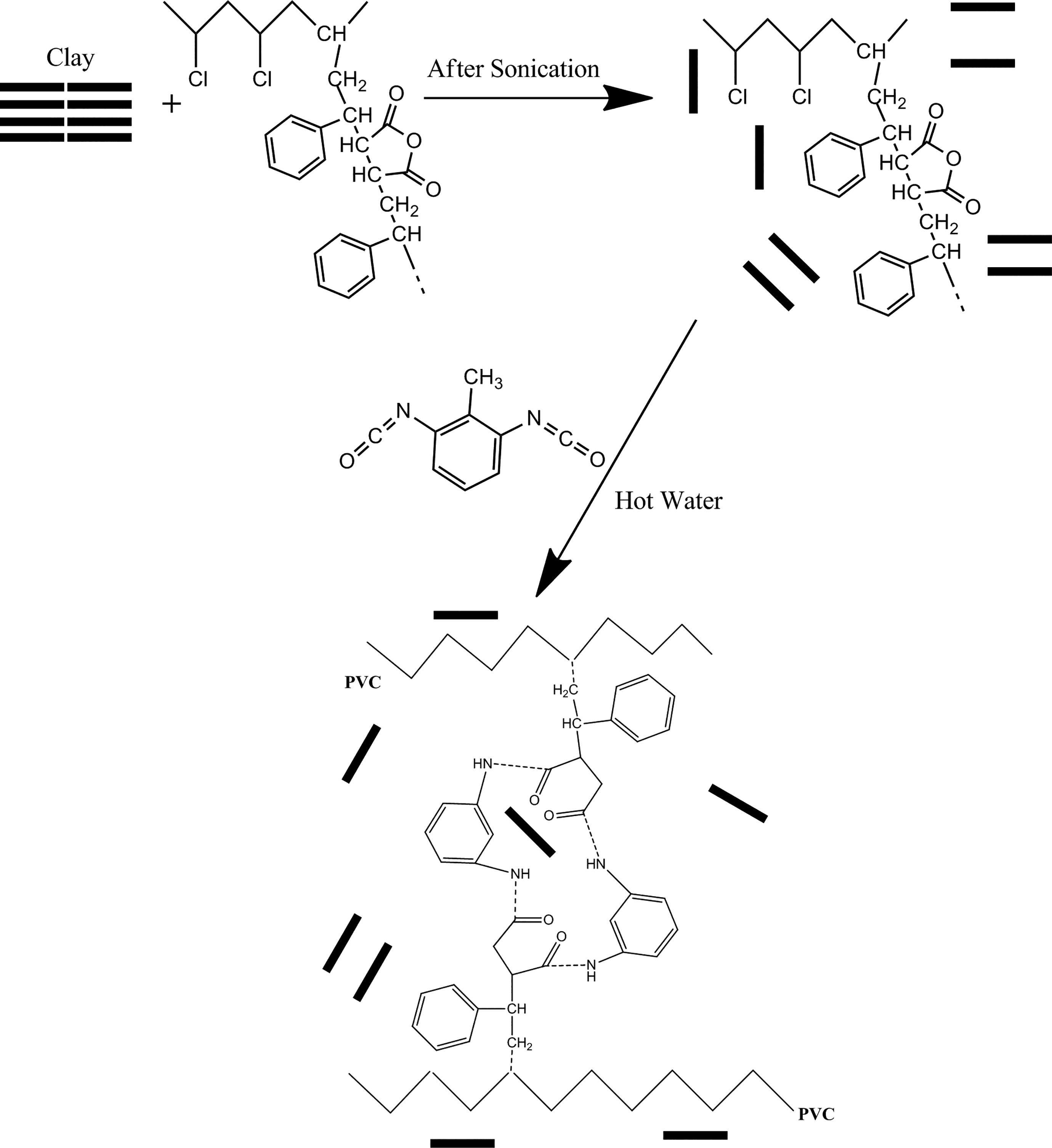
A schematic illustration of cross-linked nanoclay/PVC-g-(St-MA) nanocomposite formation.
Characterization methods
Gel content measurement was used to determine the samples’ degree of cross-linking by means of Soxhlet extraction for 24 h with THF. The final residue was dried in a vacuum oven at 60°C for at least 24 h, and the gel content (the percentage of insoluble material in THF) was defined as the percentage of the residue weight to the initial cross-linked PVC-g-(St-MA) sample weight.
FTIR spectra were recorded with a Bruker FTIR spectrometer (Tensor 27, Germany). The samples were prepared by solution casting method. The measurement range was 400–4000 cm−1 with 24 scans per measurement and a resolution of 4 cm−1.
Thermal properties of the samples were determined using a DSC (Netzsch DSC-200 F3, Germany). Around 6 mg of the sample was encapsulated in an aluminum pan. Then the sample was heated from 25°C to 130°C at a heating rate of 10°C min−1 under nitrogen purge to measure its glass transition temperature (Tg) according to the ASTM 3418-15 standard.
X-Ray diffractometer (type D5000, Siemens, Germany) equipped by a scintillation counter, copper Kα radiation (λ = 0.1540 nm) with an accelerating voltage of 35 kV, and current of 20 mA was used to record XRD patterns of various samples. The measurement range was from 1.5° to 10°, and step size was 0.0167.
TEM (Philips, the Netherlands) with an accelerating voltage of 200 kV was used to study the morphology of the casted films.
The tensile mechanical properties examination was carried out in a Zwick/Roell tensile machine (Z010, Germany). The measurements were conducted using a 10-kN load cell, with an elongation rate of 20 mm min−1, at room temperature on a dumbbell-shaped specimen prepared from cross-linked PVC-g-(St-MA) and its related nanocomposite films according to the ISO-527-2 type 5B standard. The tensile tests were repeated at least three times, and the average of the results was reported.
Results and discussion
Chemical characterization of cross-linked samples
Figure 2 shows FTIR spectra of BC-Pure-, BC-15A-1-, and BC-30B-1-casted films before immersing in hot water. Due to the low content of nanoclays in the samples, with incorporation of nanoclays into the pure cross-linked PVC-g-(St-MA) sample, no significant nanoclays absorption peaks were observed in the FTIR spectra. Along with the absorption bonds of pure PVC,36,37 there are two new absorption bonds appearing at 1780 cm−1 and 2280 cm−1 region in all samples. The bonds at 1780 cm−1 and 1830 cm−1 are attributed to the stretching vibrations of the carbonyl functional groups of the MA, while the bond at about 2280 cm−1 is related to the stretching vibration of (–NCO) groups of TDI. Furthermore, there is a weak bond in 3300 cm−1 region that is related to N–H stretching vibration.
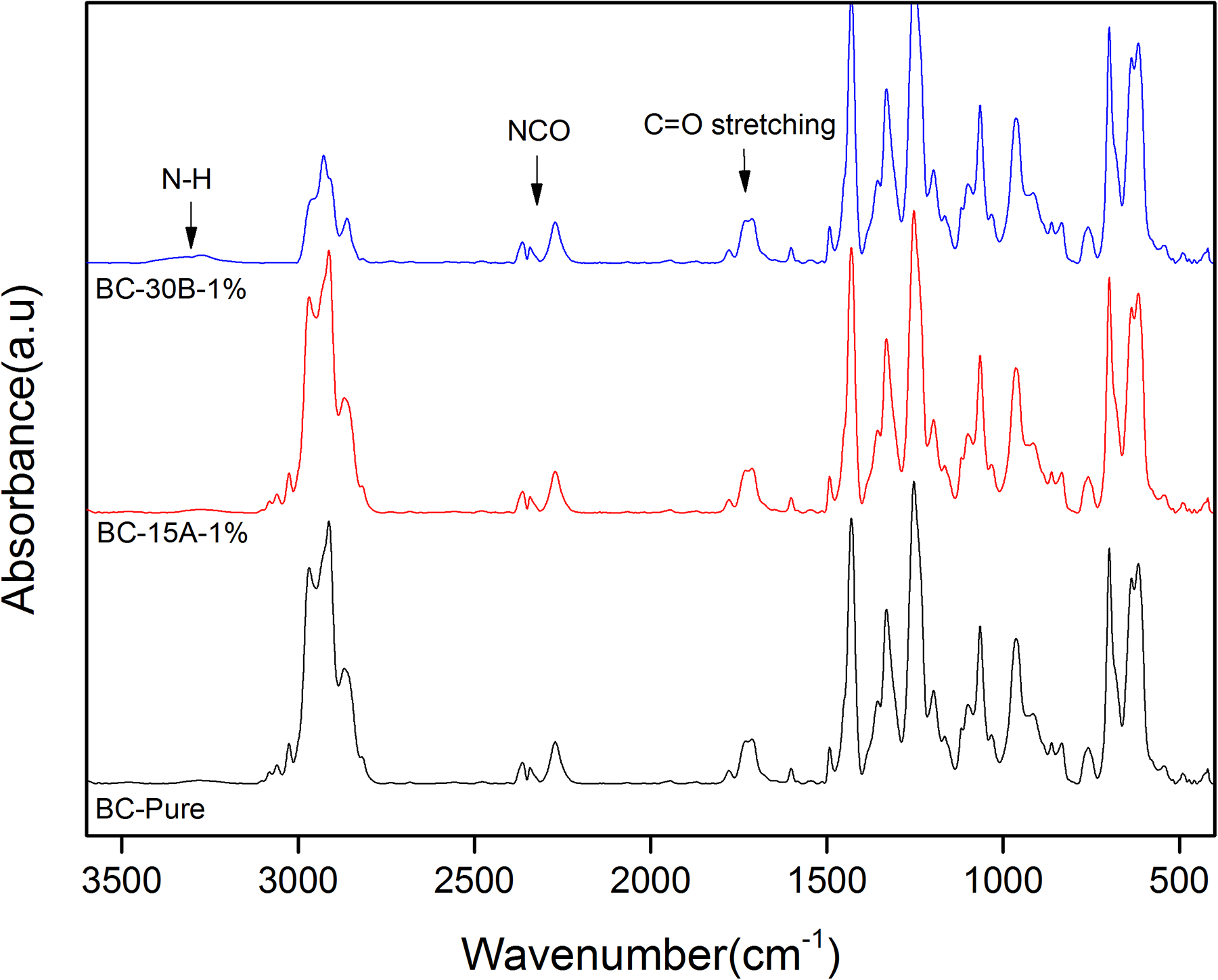
The FTIR spectra of BC-Pure, BC-15A-1, and BC-30B-1 samples.
By careful examination of–NH and –NCO peaks in FTIR spectra, it is revealed that the ratio of area under the –NH (ANH) and –NCO (ANCO) peaks relative to the area under –CH (2850–3100 cm−1) (ACH), as an internal standard, increases for BC-30B-1 sample compared to BC-Pure and BC-15A-1 samples. These observations can be attributed to the reaction of –NCO groups of TDI with structural –OH and –CH2CH2OH groups in C30B nanoclay, which confirm the covalently bonding of some –NCO groups to the C30B layers before immersion in hot water.38,39 A schematic illustration of the probable reactions between –NCO and the organic ammonium groups is shown in Figure 3.
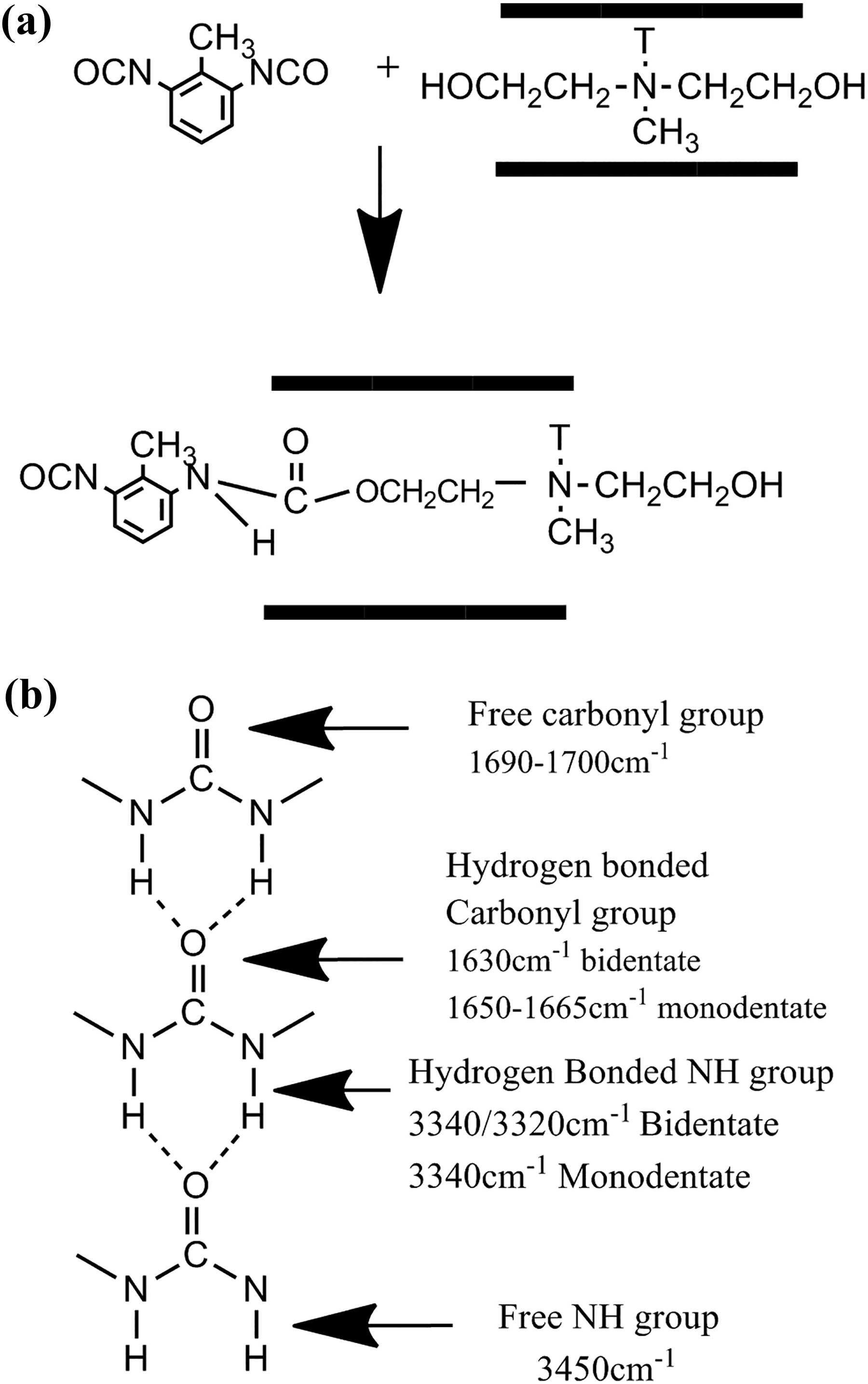
Schematic illustration of (a) the reaction between C30B and –NCO groups of TDI and (b) the band assignments for the amide carbonyl and N–H stretching modes.
The FTIR spectra of samples after immersing in hot water are shown in Figure 4. The disappearance of isocyanate and anhydride groups demonstrates that the reaction of MA groups in water leads to the maleic acid groups formation, and consequently the maleic acid groups reactions with amine groups, which resulted from the reaction of –NCO groups of TDI with water, are conducted during the cross-linking process. In turn, the appearance of new peaks at 3300 cm−1 and 1640 cm−1 are attributed to N–H and C=O stretching vibrations of amide and urea groups between PVC chains, respectively.40–42
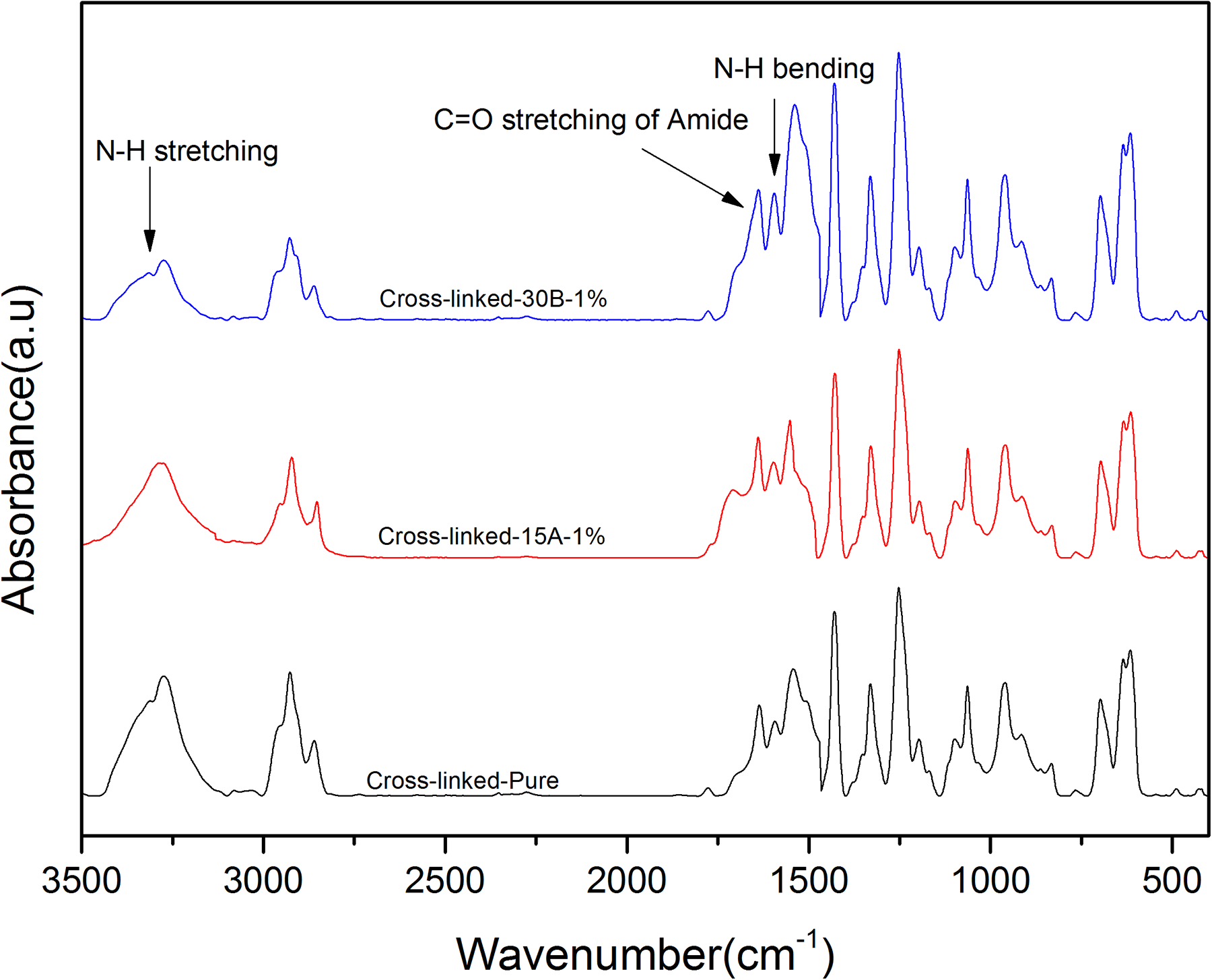
FTIR spectra of C-Pure, C-15A-1, and C-30B-1 cross-linked samples as the measure of hydrogen bonding intensity.
In the FTIR spectra of amide and urea, the peak at around 3300 cm−1 is corresponded to hydrogen-bonded –NH stretching and the peak at around 3450 cm−1 is attributed to free –NH groups (Figure 3(b)). The area under hydrogen-bonded NH stretching (AH–NH, in the range of 3150–3400 cm−1) and free NH stretching (AF–NH, 3450 cm−1) were evaluated by FTIR peak deconvolution method.43,44 The area under CH stretching (ACH, in the range of 2800–3100 cm−1) was used as an internal standard. In all samples, AH–NH/ACH ratio is much higher than AF–NH/ACH, which reveals that most of the –NH groups of the cross-links participate in hydrogen bonding with carbonyl groups of the grafted segments. However, AH–NH/AF–NH ratio varies for different samples. A comparative analysis of –NH zone deconvolution study suggests more hydrogen bonding for C-Pure sample (observed from hydrogen-bonded peak areas) than the nanocomposite samples and the C-30B-1 has the lowest amount of hydrogen bonding. Moreover, the area under free C=O stretching (AFCO, 1700 cm−1), hydrogen-bonded C=O stretching (AHCO, 1640 cm−1), and total C=O stretching (ACO, in the range of 1640–1760 cm−1) were calculated. The area under hydrogen-bonded carbonyl group stretching peak is much higher than free carbonyl group one. AHCO/ACO ratio decreases in cross-linked PVC-g-(St-MA) nanocomposites compared to pure cross-linked PVC-g-(St-MA) sample. Additionally, in case of C-30B-1 nanocomposite, the ratio of AHCO/ACO is slightly lower than C-15A-1 nanocomposite (see Table 2). Nanoclays dispersion may be a good reason for this behavior, such that the well-dispersed nanoclay layers probably interfere with hydrogen bond formation between cross-linked PVC-g-(St-MA) chains. Due to the polar nature of C30B, nanoclay layers may have an appropriate dispersion in C-30B-1 nanocomposite compared to C-15A-1 one, leading to an increase in the specific surface area such that nanoclay layers can act as barrier between cross-links against their interactions.
The ratio of areas for various functional groups in FTIR spectra of different samples.
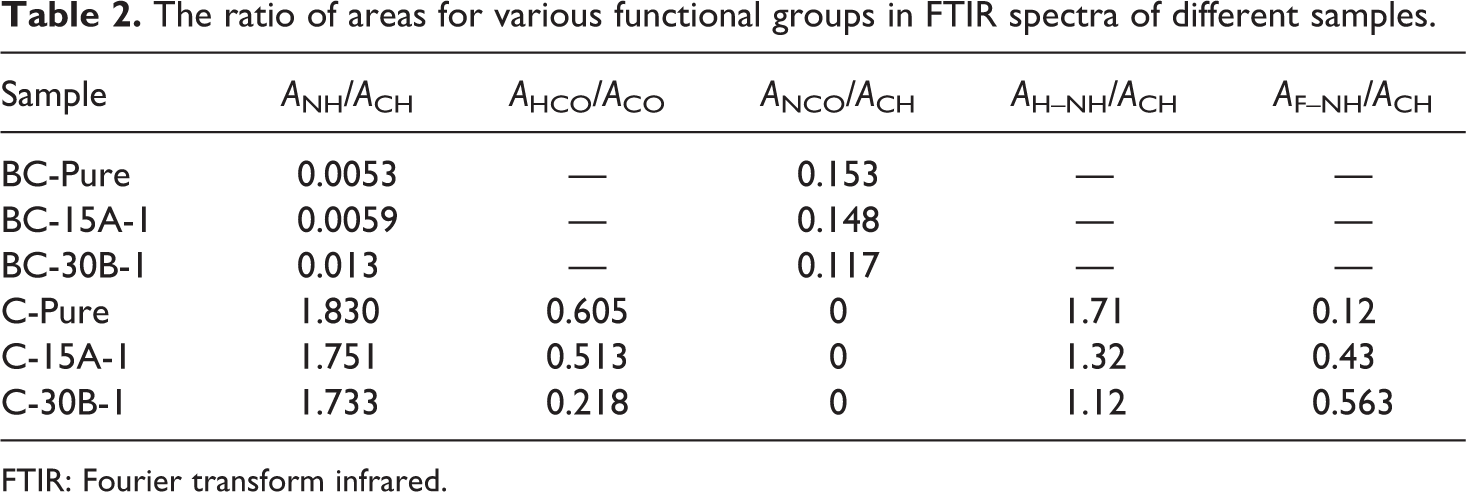
FTIR: Fourier transform infrared.
Gel content measurements
In Figure 5, the gel contents of cross-linked PVC-g-(St-MA) and related nanocomposites are shown. C-Pure sample has a maximum gel content of 56.5%. This figure shows that the gel contents of nanocomposites are almost unaffected with adding up of nanoclay content to 0.5 wt%, but it decreases with further increasing of nanoclays content. A limited range of clay could be incorporated into these cross-linked systems due to network structure of the PVC that prevented the migration of macromolecules within the galleries and thus made difficult the proper dispersion of higher amount of silicate layers. In other words, the PVC-g-(St-MA) molecules mixed with nanoclay in mixing step need to diffuse within the galleries to increase the d-spacing between the layers and consequently to attain a proper dispersion of clay throughout the matrix. This process should occur parallel to cross-linking phenomena in hot water. Under cross-linking condition, some PVC-g-(St-MA) chains would be confined by clay layers which prevent the cross-linking reactions. This behavior indicates that the presence of nanoclay in PVC-g-(St-MA) matrix inhibits to some extents from the formation of a cross-linking network. However, the gel content is still higher for C15A nanocomposites than C30B ones at the same nanoclay content. This can be attributed to the proper dispersion of C30B in the polymer matrix as well as to the structural –OH and –CH2CH2OH groups existing in C30B which may react with –NCO groups of TDI before their participation in cross-linking reactions. The C15A is more hydrophobic than C30B, so it is expected that PVC chains and TDI molecules to enter more easily into the C30B interlayer space than C15A. In case of the C15A, there are no functional groups that can react with TDI molecules. Therefore, TDI can only participate in main cross-linking reaction.
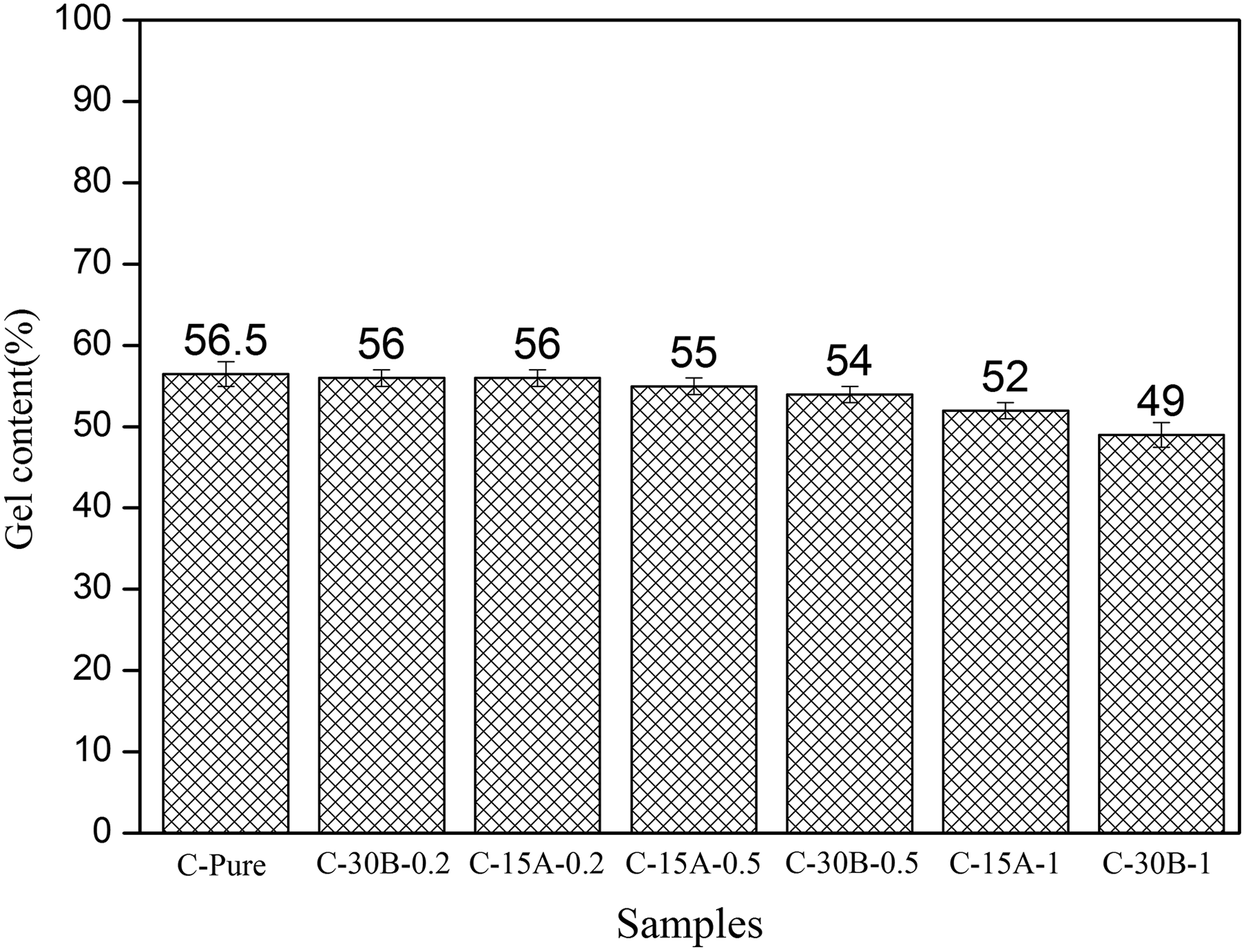
Gel content of various cross-linked PVC-g-(St-MA) and related nanocomposite samples. PVC: polyvinyl chloride; St: styrene; MA: maleic anhydride.
Thermal properties
The DSC thermograms of PVC, PVC-g-(St-MA), cross-linked PVC-g-(St-MA), and related nanocomposites are shown in Figure 6. Summary of the
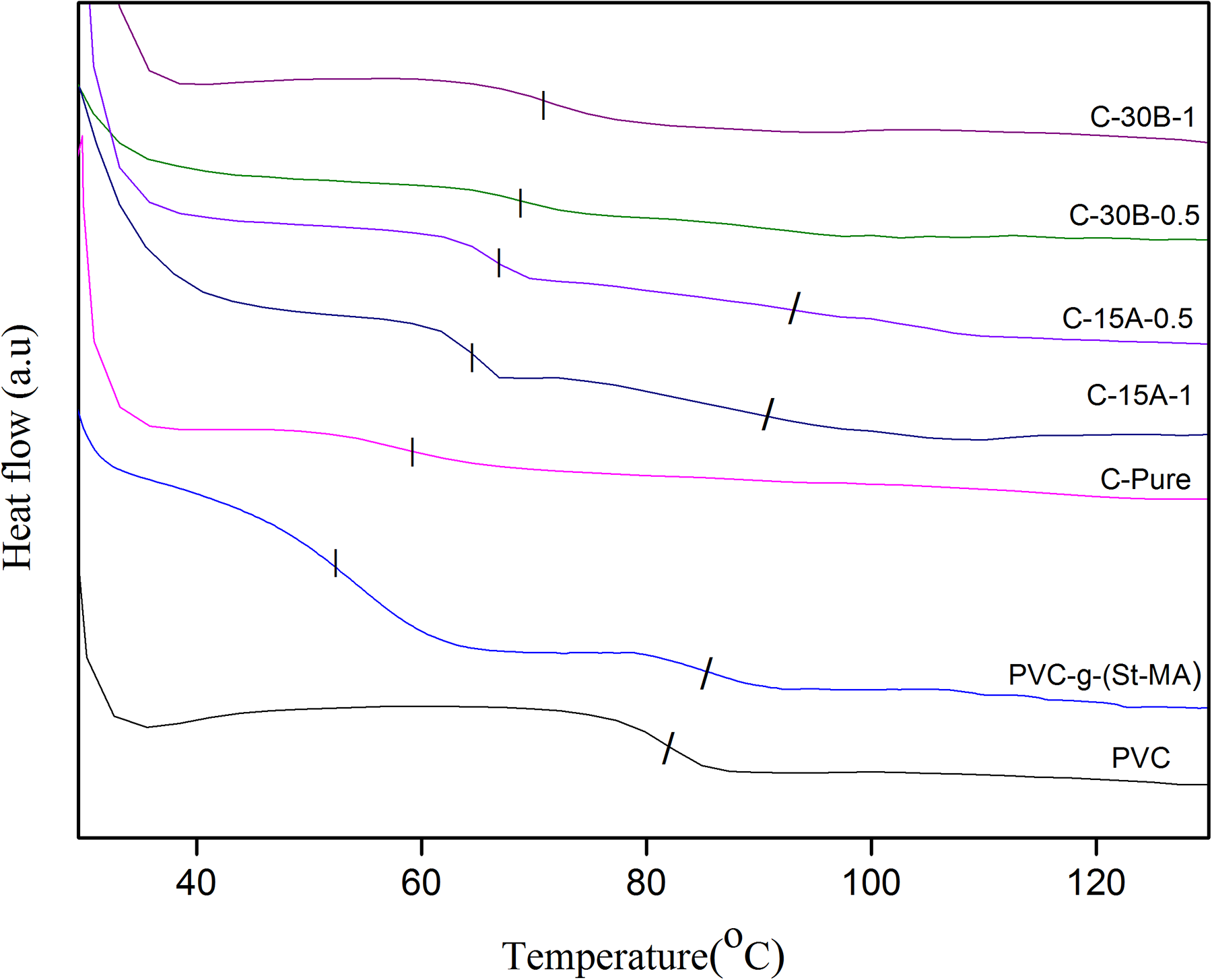
DSC thermograms of PVC and related nanocomposites with different nanoclay contents. DSC: differential scanning calorimetry; PVC: polyvinyl chloride.
Summary of the Tg of various samples.

Tg: glass transition temperature; PVC: polyvinyl chloride; St: styrene; MA: maleic anhydride.
In C-30B nanocomposites, although the gel contents are lower than pure cross-linked PVC-g-(St-MA) sample, a single Tg is observed at higher temperature compared to C-Pure, which corresponds to the cross-linked network. An increase of 8°C in Tg is observed by the addition of 0.5 wt% C30B. At nanoclay content of 1 wt% C30B, Tg is 70.9°C, which is higher than Tg of C-30B-0.5 sample. The DSC thermograms at the higher nanoclay content do not show any glass transition. The increase in Tg at lower C30B nanoclay contents could be attributed to decrease in chain mobility of PVC-g-(St-MA) matrix by proper dispersion of nanoclay layers between cross-linked PVC-g-(St-MA) chains. Since C30B nanoclay surface has the appropriate interaction with PVC-g-(St-MA) chains, it may assist the immobilization of polymer chains on the nanoclays surface, which leads to an increase in Tg. As it can be observed, at the same clay content, the Tg of C30B nanocomposites is higher than that of C15A ones.
In C15A-CPN, the addition of C15A increases Tg of the cross-linked network compared to pure one. In C-15A-0.5 sample, a glass transition for cross-linked nanocomposite is appeared at 66.8°C, which is reduced to 64.4°C in C-15A-1 nanocomposite. Surprisingly, the Tg of PVC chains, which has been disappeared in all cross-linked samples, is appeared again at 90.7°C in C-15A-1 and at 91.6°C in C-15A-0.5 samples. This behavior can be attributed to the reduction in the effective surface area due to C15A agglomeration at high nanoclay content as well as to the lower gel content of C-15A-1 sample compared to C-Pure PVC-g-(St-MA) sample.
Morphology of cross-linked PVC-g-(St-MA) nanocomposites
The XRD method can be used for evaluation of CPN, since PVC with an amorphous structure does not have any reflection in the region of 2θ from 1° to 10°; therefore, the reflection appearing on the XRD patterns are originated from nanoclays. The changes in 2θ are related to the alteration in interlayer spaces of nanoclays.
Figure 7 shows the XRD patterns of cross-linked CPN as well as the patterns of C30B and C15A nanoclays. C30B shows an intense reflection at 2θ = 4.8° with corresponding interlayer space (d001) of 1.85 nm, which is obtained via Bragg equation: nλ = 2dsinθ. For both C30B-CPN, C30B characteristic reflection is disappeared, suggesting that the basal spacing may be expanded over 6 nm (2θ < 1.5°). The nonappearance of an XRD reflection implies that during the cross-linking process, the layered structure of C30B is destroyed by the strong interaction between polar PVC chains and the silicate layers of C30B. PVC chains and TDI molecules can diffuse into the nanoclays interlayer space pushing the silicate layers to space out, and the cross-linking of PVC-g-(St-MA) is conducted through intra and extra-interlayer spaces. As a result, the exfoliated C30B-CPNs are formed.
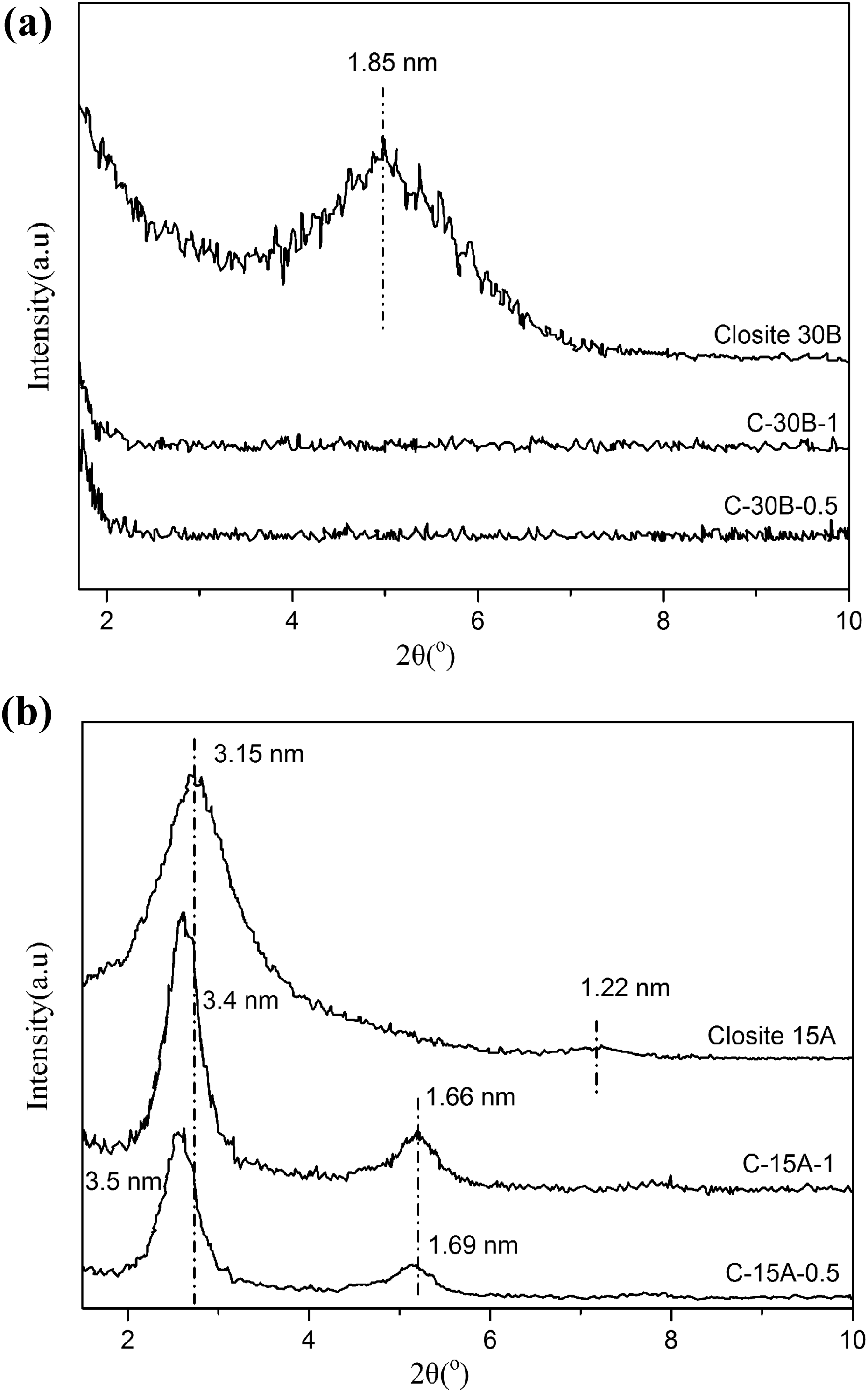
XRD patterns of cross-linked PVC-g-(St-MA) nanocomposites: (a) C30B nanocomposites and (b) C15A nanocomposites.
C15A nanoclay shows an XRD pattern which has a main reflection at 2θ = 2.8° that corresponds to an interlayer space (d001) of 3.15 nm and reflection at 2θ = 7.2° assigned to d002 basal spacing. Two examined C-15A-0.5 and C-15A-1 nanocomposite samples show basal reflections at slightly lower 2θ values than neat C15A nanoclay. These patterns correspond to d001-value of 3.5 nm and 3.4 nm for C-15A-0.5 and C-15A-1 nanocomposites, respectively, which are slightly higher than d001-value in C15A nanoclay. The small increment of d001-value suggests that PVC-g-(St-MA) chains intercalated in C15A nanoclays slightly. The increase in d002 spacing of C-15A-0.5 and C-15A-1 nanocomposites (1.69 nm and 1.66 nm, respectively) also indicates that these nanocomposites have partially intercalated structures. C15A is more hydrophobic than C30B, thus it is expected that PVC chains and TDI molecules to enter more easily into the C30B interlayer space than C15A. However, the alkyl ammonium cations in C15A have two alkyl chains (hydrogenated tallow), whereas C30B cations have only one alkyl chain. So it can be concluded that in C15A, more space is occupied by the alkyl ammonium ions, and there is less available interlayer space for PVC chains compared to C30B. Thus, the PVC chains are intercalated in a smaller proportion in C15A nanoclays than in C30B. 41
Following the observations from XRD analyses, TEM micrographs were taken to visualize the nanoclay dispersivity in nanocomposites. The typical TEM images of the C-15A-1 and C-30B-1 samples are shown in Figure 8. In TEM images for C-15A-1 nanocomposite (Figure 8(a)), more agglomerated nanoclays are observed, indicating poor dispersion of C15A in CPN matrix. Due to the excess amount of ion exchange capacity of C15A nanoclays (125 meq/100 g clay), their surfaces contain high modifier content, which the majority of their surfaces can be covered by the hydrophobic modifier and the affinity of PVC chains to C15A are inhibited. This may be another manifestation of the reports that interaction between nanoclay and polymer can be more imperative in the clay dispersion than the interaction between modifier and polymer. 48
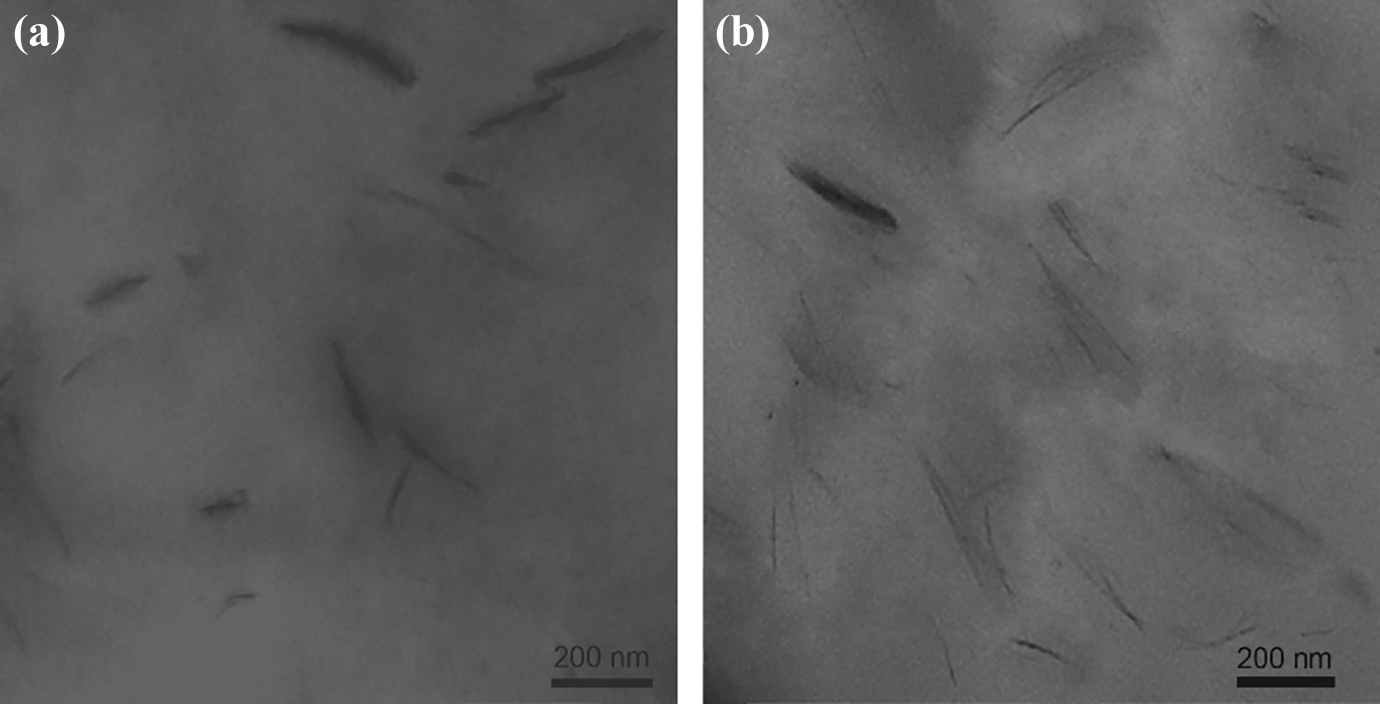
TEM images of cross-linked PVC-g-(St-MA) nanocomposites: (a) C-15A-1 and (b) C-30B-1.
Figure8(b) shows TEM micrograph of C-30B-1 nanocomposite, demonstrating the exfoliated/intercalated structures for C30B layers. These structures may reflect the compatibility and interaction between PVC-g-(St-MA) chains and the hydroxyl groups of C30B nanoclays, which facilitate the separation of their silicate layers. This observation is in good agreement with our XRD results.
Mechanical Properties
Typical stress–strain diagrams of neat PVC, cross-linked PVC-g-(St-MA) (C-Pure), and cross-linked CPN are shown in Figure 9. The mechanical properties for cross-linked PVC-g-(St-MA) and its nanocomposites are listed in Table 4. As it is observed, Young modulus and yield stress of C-Pure sample are increased about 78% and 43%, respectively, compared to neat PVC, which can attributed to cross-linked network formation. Also, it is apparent that the incorporation of nanoclays leads to an increase in Young’s modulus, which confirms the reinforcing potential of these nanoclays in examined CPN.
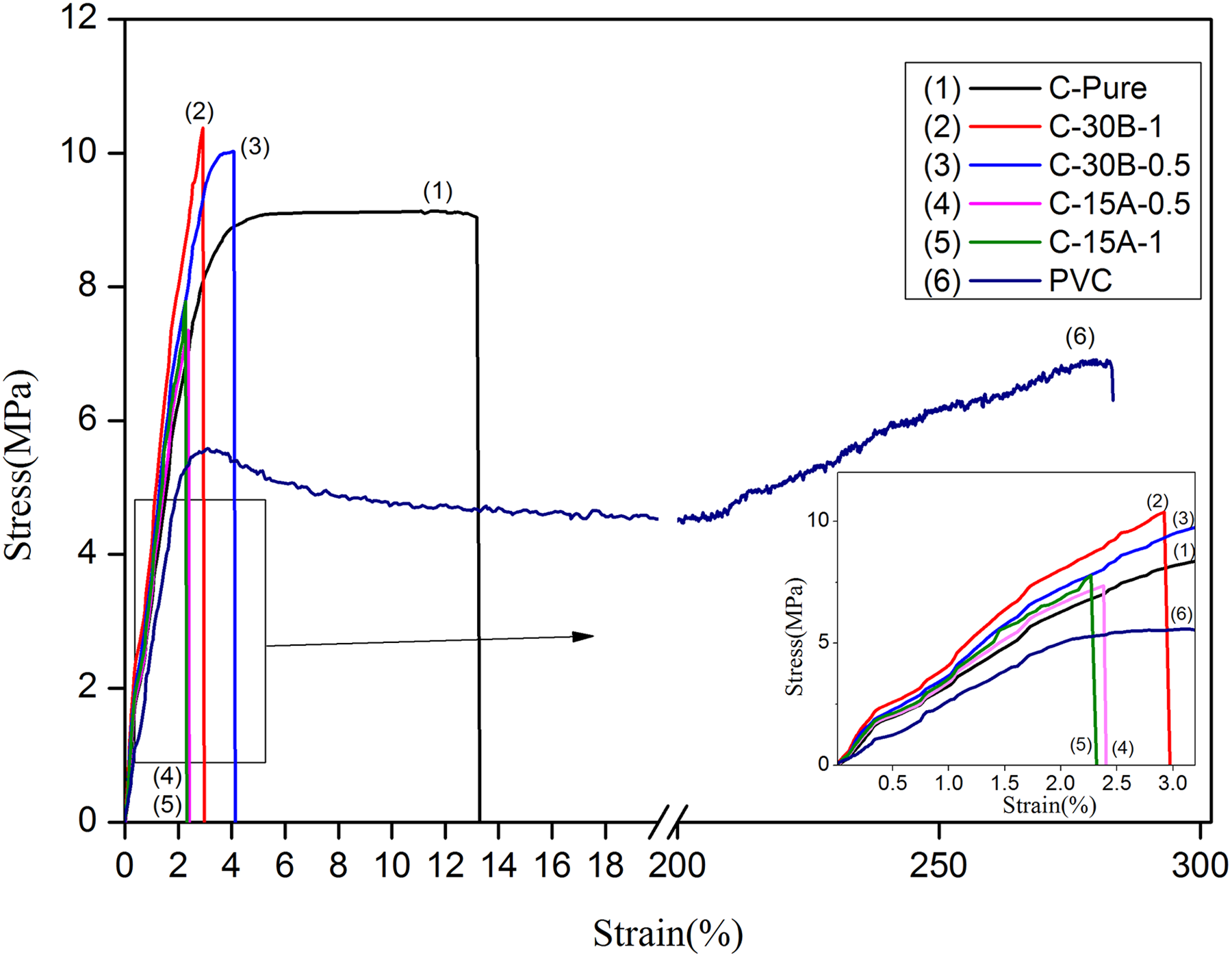
Stress–strain diagrams of PVC, C-Pure, and related nanocomposites.
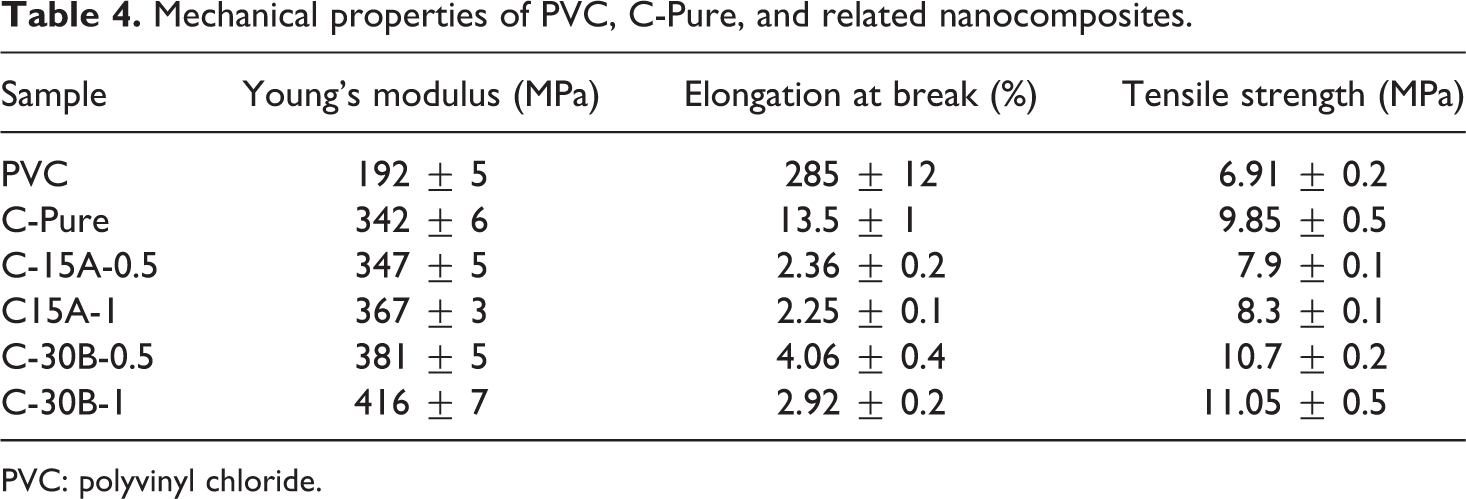
Mechanical properties of PVC, C-Pure, and related nanocomposites.
PVC: polyvinyl chloride.
In C30B-CPN, the improvement in tensile strength is conducted up to 1 wt% of nanoclay. While the amounts of the C30B are 0.5 wt% and 1 wt%, the tensile strengths of the C30B-CPN are 10.7 MPa and 11.05 MPa, respectively. Similarly, Young’s modulus values are increased from 342 MPa for C-Pure to 381 MPa in C-30B-0.5 and to 416 MPa in C-30B-1 nanocomposites; however, the elongation at break is decreased from 14% for C-Pure to 3–4% in theses nanocomposites. Such improvement in tensile strength and Young’s modulus can be attributed to the nanoclay–polymer interaction, as well as the cross-links between PVC-g-(St-MA) chains, which provide reinforcement. Consequently, the movements of the polymer chains are restricted to reduce the sliding through straining.
In the nanocomposites of C15A, the aggregation of nanoclay layers inside of the polymer matrix will decrease the effective interfacial interactions. As a result, the loading stress cannot be effectively transferred from PVC-g-(St-MA) matrix to nanoclays and the tensile strength of the nanocomposites will be dropped.
Conclusion
Nanoclay/PVC-g-(St-MA) nanocomposites containing C30B and C15A nanoclays were prepared by solution mixing method and cross-linked by immersing in hot water. The FTIR spectra of the samples showed that the incorporation of nanoclays has significant effect on hydrogen bonding between cross-linked segments, especially for C-30B-1 sample. Furthermore, the gel content of C-30B-1 nanocomposite was the lowest among all samples due to the good dispersion of C30B and the reaction capability of –CH2CH2OH modifier groups with –NCO groups of TDI. Morphological examinations by XRD and TEM revealed more appropriate clay dispersion in C30B nanocomposites than the C15A ones. As a result, much better exfoliated structure was observed in C-30B-1 nanocomposite, while C-15A-1 nanocomposite showed partially intercalated and agglomerated morphology. DSC measurements revealed that at the same nanoclay content, the Tgs of the nanocomposites containing C30B were higher than those C15A samples. Tensile stress–strain behavior of CPN indicated that nanocomposites had higher Young’s modulus compared to neat PVC and C-Pure samples. However, in C15A nanocomposites, the aggregation of nanoclay layers in PVC-g-(St-MA) matrix decreased the tensile strength of the nanocomposite compared to C-Pure sample.
