Abstract
Synergistic effects of organo-modified Mg-Al layered double hydroxide (LDH) and triethyl citrate (TEC) on the properties of poly(lactic acid) (PLA) were demonstrated. PLA/LDH nanocomposites in the absence and presence of TEC were fabricated via solution casting technique. Morphological analysis revealed that as the LDH concentration increases, the number of aggregations is also increased; however, introduction of TEC considerably enhanced the dispersion quality of LDHs. Differential scanning calorimetry results showed that the addition of LDH and TEC had no significant influence on the crystallinity of nanocomposites obtained from solution casting. In contrast, once the samples were cooled from melt, the concurrent use of LDH and TEC led to a dramatic enhancement in the crystallinity of PLA (
Introduction
As a bio-based plastic, poly(lactic acid) (PLA) has gained a huge amount of attention since it possesses mechanical properties similar to those of commodity plastics such as polyethylene and polystyrene. 1 –6 PLA is a fully biodegradable polyester exhibiting high modulus, high strength, and good level of clarity. 7,8 Another advantage of PLA is the fact that its decomposition products lead to no significant harm to the environment. In general, PLA has shown a promising potential to replace the petro-derived polymers. However, due to the poor thermal stability and slow crystallization rate, PLA applications are being highly restricted. The crystallinity of PLA has a crucial influence on thermal stability, barrier, and mechanical properties. For instance, the barrier properties of PLA could be improved for packaging applications through an increased extent of crystallinity. 9 However, the enhanced crystallinity of PLA could also reduce its clarity and biodegradation rate. Hence, numerous studies have been devoted to control the crystallization process of PLA depending on the final application. One of the efficient solutions for this issue is the addition of nucleating agents including talc, 10 montmorillonites, 11 carbon nanotubes, 12 and layered double hydroxides (LDHs). 13 Among the diverse range of additives, the introduction of different LDHs into the PLA matrix has received attention in recent years because of the improvements in optical, mechanical, thermal, and barrier effects. 14 –16 LDH is a class of synthetic anionic clays with host–guest nanolayer material containing positively charged metal hydroxide sheets, intercalated anions as guest and water molecules. 17 In addition to many advantages of LDH nanoparticles, they have also been used as nucleating agents to enhance the crystallinity of polymers. Geng et al. 13 have used organically modified LDHs to enhance the overall properties of PLA. Their results showed that LDHs acted as excellent nucleating agents leading to greatly increased crystallization rate of PLA. In another study, amidated benzoic acid intercalated LDHs were incorporated into PLA matrix through reactive extrusion. 18 Based on the literature, one of the issues concerning the use of LDHs in polymers is their quality of dispersion, which is directly associated with the final performance of the resulting composite. Quispe-Dominguez et al. 16 have reported the utilization of magnesium-aluminum (Mg-Al) LDHs intercalated by dodecylbenzene sulfonate to improve the dispersion of LDHs in PLA matrix.
Imparting higher mobility of polymer chains, for example via using a plasticizer, has been regarded as an efficient method of enhancing crystallization rate of PLA. 19,20 Moreover, the improved toughness of PLA as a result of using a plasticizer could extend its application window especially in food packaging industry. 21 PLA plasticization with low molecular weight materials is associated with some limitations such as lowered strength, modulus, and thermal stability. 22 Therefore, a combination of nanoparticles and different types of plasticizers has been suggested to enhance the crystallinity, toughness, and also thermal stability of the PLA composites. 22,23 Li and Huneault 23 concluded that a combination of heterogeneous nucleation and chains mobility could significantly increase the crystallinity of PLA. Among a diverse range of plasticizers for PLA, citrate esters, which are derived from naturally occurring citric acid, have gained much attention due to being nontoxic and biocompatible. 24 Among them, triethyl citrate (TEC) has the lowest molecular weight and results in higher elongation at break as compared with the other citrate esters. Moreover, TEC was proved to considerably enhance the enzymatic degradation of PLA. 24
In this study, the effects of concurrent utilization of organo-modified LDHs (nucleating and reinforcing agent) and TEC (plasticizer) were evaluated on the properties of PLA with the aim of counterbalancing the adverse effects of plasticizer on mechanical and thermal properties. To this end, morphology, crystallization, mechanical properties, and thermal stability of PLA/LDH/TEC composites were investigated and the results were correlated.
Materials and methods
Materials
PLA (Ingeo 4043D grade) with density of 1.24 g mL−1, weight-average molecular weight of 100,000 g mol−1, and D-lactide content of 4.8% was supplied by NatureWorks LLC (Minneapolis, USA). TEC with density of 1.14 g mL−1 was purchased from Sigma-Aldrich, St. Louis, USA. Dichloromethane as a solvent for PLA was supplied by Merck (Darmstadt, Germany) and used as received. Magnesium nitrate, aluminum nitrate, and sodium dodecylbenzene sulfonate (SDBS), used for synthesis of organo-modified LDHs, were procured from Merck (Darmstadt, Germany).
Synthesis of organo-modified LDH
The complete procedure utilized for the synthesis of organo-modified LDHs was reported elsewhere. 17 Briefly, a NaOH solution (1 M) was added dropwise to an aqueous SDBS solution in a 250 mL three round-bottomed flask to keep the pH value at 10.5 ± 0.2 followed by dropwise addition of magnesium nitrate and aluminum nitrate solutions. Afterward, the solution was stirred for 30 min at 50°C, and then the solution was heated to 70°C and aged for 24 h. After cooling, the final white product was collected by filtration and washed thoroughly with distilled water to reach a neutral pH followed by drying in an oven at 75°C.
Samples preparation
A typical solution casting technique was utilized in this study for the preparation of samples. For the pure PLA film, a certain amount of PLA granules (1 g) was dissolved in 25 mL of solvent (dichloromethane) via magnet stirring for 30 min followed by casting in pre-cleaned glass plates. In the case of nanocomposite samples, certain amounts of LDH nanoparticles (2 and 4 wt%) were dispersed in the PLA solution via vigorous stirring for 6 h. Afterward, the suspensions were subjected to ultrasonic bath for 1 h to achieve a homogeneous suspension. In another series of samples, a certain amount of TEC (10 wt%) was also incorporated into the PLA-LDH suspensions. All the samples were dried at 30°C for 12 h. For simplicity, the nanocomposite samples were coded as LDHX in which X represents the concentration of LDH nanoparticles in the PLA matrix (2 and 4). Likewise, the LDHX-TEC samples represent those nanocomposites which were plasticized by TEC.
Characterization
Scanning electron microscopy (SEM) micrographs on the cross-sectional surfaces of samples fractured in liquid nitrogen were attained by a digital scanning electron microscope coupled with energy-dispersive X-ray (EDX) spectroscopy (VEGA//TESCAN instrument, Czech Republic) operated at 25 kV. To avoid electric charging, all the samples were gold coated (K450X; Emitech, Kent, UK). Differential scanning calorimetry (DSC) experiments were conducted in a Mettler TA 4000 thermal analysis system (Greifensee, Switzerland). Aluminum pans each containing 5–10 mg of samples were heated from 30°C to 200°C at a heating rate of 10°C min−1 followed by resting for 3 min to erase any thermal history. Afterward, the samples were cooled from 200°C to 30°C at a rate of 5°C min−1, and then heated again at a rate of 10°C min−1 to 200°C. Thermal stability of the samples was studied via Q 500 V6.5 apparatus from the ambient temperature to 600°C at the heating rate of 10°C min−1 under nitrogen atmosphere (flow rate of 50 mL min−1). The mechanical behavior was evaluated based on ASTM procedure D882-02 through a TA1 Texture Analyzer, AMETEK Lloyd Instruments, Hampshire, UK. At least five specimens were cut (10 × 10 mm2) from each formulation. The used conditions for tensile tests were a gauge length and crosshead speed of 30 mm and 1 mm min−1, respectively. The mechanical parameters measured were the tensile strength, tensile modulus, and elongation at break.
Results and discussion
SEM/EDX analysis
In order to study the effect of LDHs and TEC on the morphology of PLA films, the cross-sectional area of the samples was evaluated with SEM and the results are shown in Figure 1 at different magnifications. The lower magnification cross-sectional image for LDH2 (Figure 1(a)) indicates a brittle fracture and an overall uniform structure. However, there could be detected some agglomerations of LDH nanoparticles as large as 6 µm (as marked in Figure 1(b)). Based on Figure 1(c), the addition of TEC as a plasticizer changed the cross-sectional morphology and has resulted in a tougher fracture. Based on scanning a large area, large agglomerations, as found in LDH2, were not detected at the fracture surface of LDH2-TEC, indicating the positive influence of adding a plasticizer in dispersion quality of nanoparticles. Figure 1(d) shows the largest LDH agglomerations found on the fracture surface of LDH2-TEC which are in the range of 2 µm. Based on Figure 1(e), increasing the LDH concentration to 4 wt% notably changes the fracture surface of the film. As can be seen, the number of LDH aggregations was much higher due to the increased concentration. Despite the increased number of LDH aggregated structures, a good overall morphology could be detected for LDH4. Similar to LDH2, rather large LDH aggregations were also found in the case of LDH4 with sizes in the range of 6–8 µm (Figure 1(f)). The difference is that the number of such aggregations is higher in LDH4 with regard to LDH2. The significant influence of TEC as plasticizer could be seen in the case of LDH4-TEC sample (Figure 1(g)) for which the number of LDH aggregations was notably declined, which could be attributed to the enhanced mobility of PLA chains in the presence of TEC molecules leading to a better dispersion quality of nanoparticles in the course of mixing and drying processes. Figure 1(h) shows one of the few aggregations for this sample based on scanning a large area. In conclusion, one could infer that the introduction of TEC as plasticizer into PLA/LDH nanocomposites could lead to significantly better dispersion quality of nanoparticles resulting in an enhanced performance of the final product.

Cross-sectional morphology of (a, b) LDH2, (c, d) LDH2-TEC, (e, f) LDH4, and (g, h) LDH4-TEC at magnifications of ×2000 and ×5000.
Figure 2 shows the cross section at magnification of 500× along with the EDX mapping of Mg element, which is indicative of the LDHs’ distribution. Figure 2(a) shows the thickness of nanocomposite film which is estimated around 140 µm and Figure 2(b) implies that the LDH nanoparticles have been uniformly distributed throughout the whole cross-sectional area further confirming the observations from SEM results.

(a) Cross-sectional area of LDH2-TEC and (b) its corresponding EDX mapping for Mg.
DSC analysis
In order to investigate the effects of nanoparticle and plasticizer additions on the crystallization behavior of PLA, DSC analysis has been conducted. The first heating scans from room temperature to 200°C are illustrated in Figure 3. Three thermal transitions for PLA could be detected for all the samples, namely, glass transition, cold crystallization, and melting. Regarding the glass transition region, it is seen that the glass transition temperature (
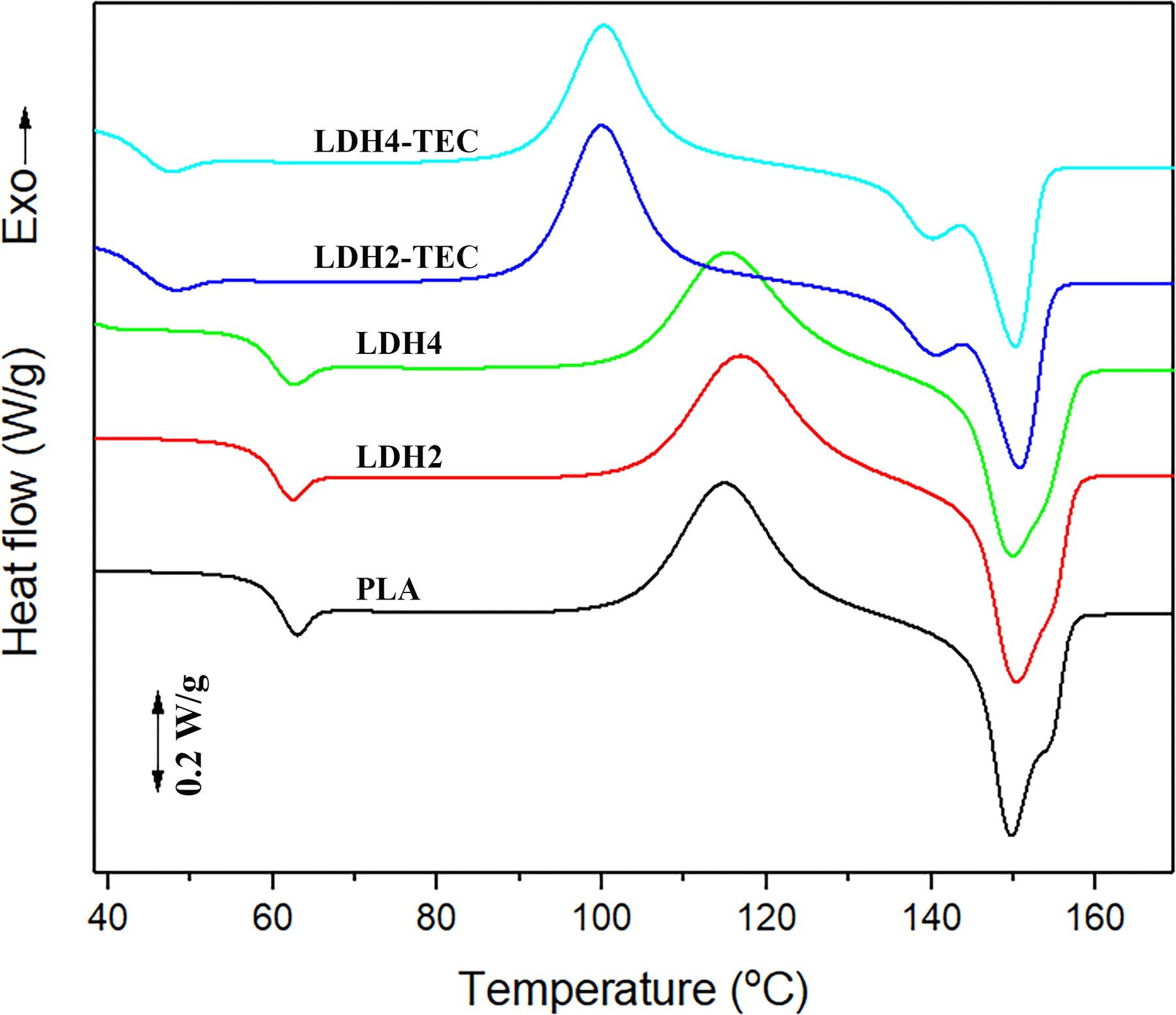
First heating DSC scans for the neat PLA, LDH-loaded and LDH/TEC-loaded nanocomposites.
The quantified parameters obtained from first DSC heating scan for all the samples.
LDH: layered double hydroxide; TEC: triethyl citrate; PLA: poly(lactic acid); FWHM: full width at half maximum; DSC: differential scanning calorimetry.
Based on Figure 3, the melting endotherm of neat PLA is bimodal, which could be ascribed to the formation of crystals with different degrees of perfection due to the slow rate of crystallization and the reorganization of crystalline PLA. 22 The first endothermic peak (149.7°C) is related to the melting of cold crystallized PLA chains, whereas the second peak (shoulder at 154.3°C) could be attributed to those PLA crystals, which have reorganized to more perfect crystals once they found sufficient thermal energy in the course of cold crystallization. Based on Table 1, the introduction of LDH imposed no significant changes in the endothermic behavior of PLA since the melting temperature range and also the sequence of peaks remained nearly unchanged. However, once LDH and TEC were added concurrently, a distinct endotherm appeared at around 140°C, which is most probably related to the melting of α′-type crystals that possess a lower degree of perfection. The main reason for the creation of these less perfect crystals is the fact that cold crystallization has occurred at 100°C in the case of LDH/TEC-loaded nanocomposites. Based on the literature, the α′-type crystals could be formed at crystallization temperatures lower than 120°C. 27 The second endotherm for LDH/TEC-loaded samples appeared at 150–150.5°C, which is similar to that of other samples. In conclusion, the PLA crystals were only of α-type in the cases of neat PLA and LDH-loaded nanocomposites; however, a small portion of PLA crystals was of the α′-type in the cases of LDH/TEC-loaded nanocomposites, and thus, the majority of crystals were of the α-type which are more ordered having a higher degree of perfection. 28 Apart from all the changes made upon addition of LDH/TEC, the melting enthalpy for the samples remained in a certain range (12–13 J g−1), indicating the fact that PLA crystallization from solution state is not majorly influenced by the addition of nanoparticles and plasticizer.
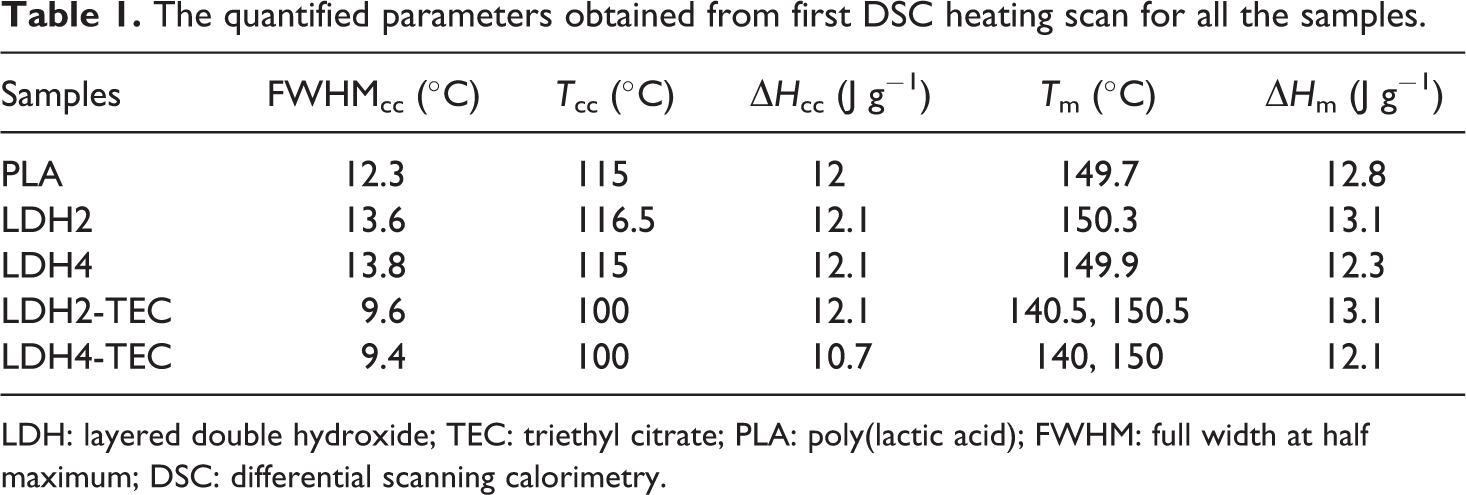
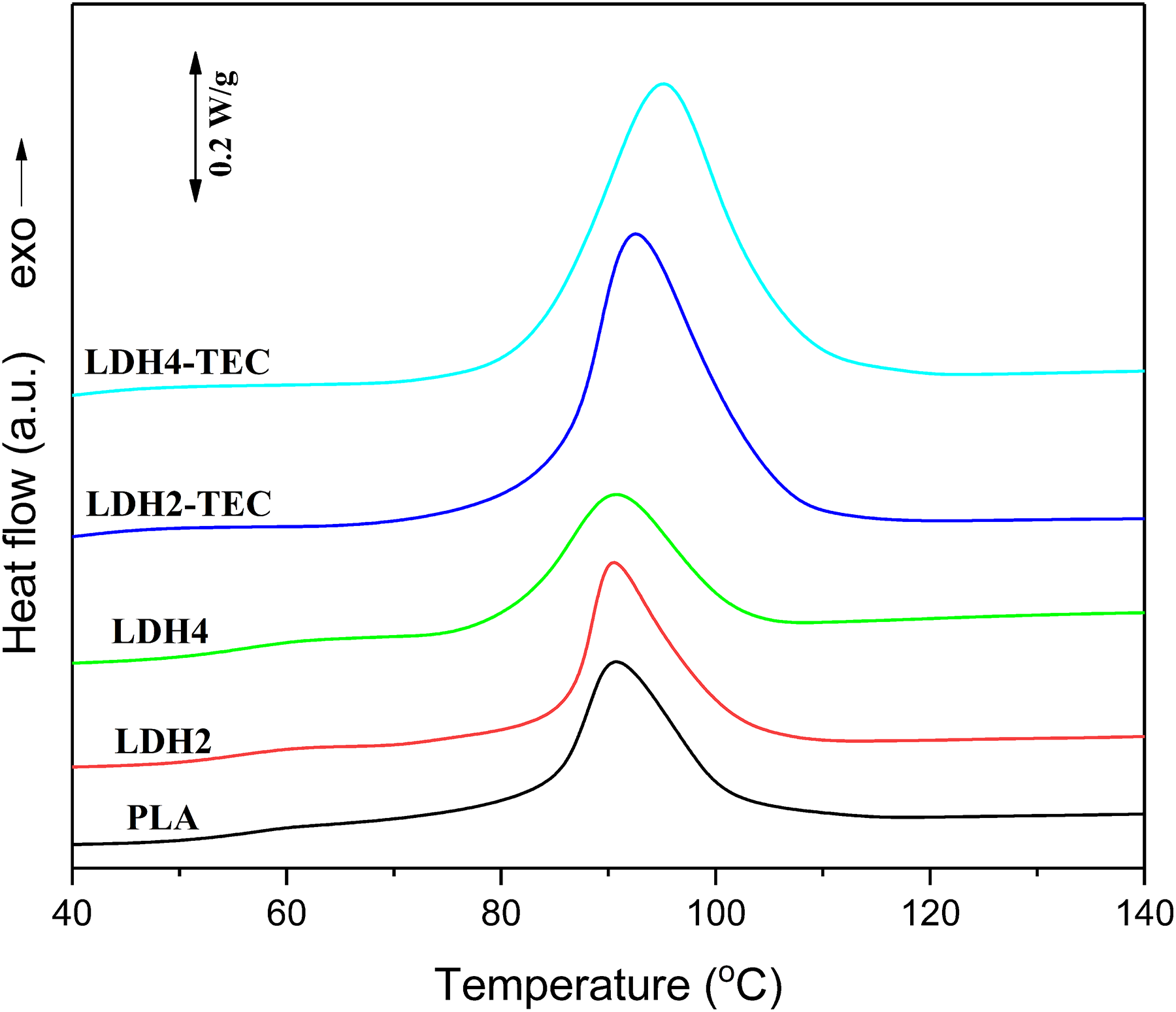
After removing the thermal history, the samples were cooled and the cooling scans could be observed in Figure 4. A distinct exothermic peak could be detected for all the samples with the peak temperature in the range of 90–95°C. One can infer that the crystallization process was carried out once cooled from the melt state. It could be observed that the crystallization peak for LDH2 and LDH4 samples is quite similar to that of the neat PLA. On the other hand, once the hybrids of LDH and TEC were utilized, the crystallization peak was essentially augmented. Finally, the second heating scans were also performed for all the samples and the results are shown in Figure 5. The quantified results are reported in Table 2. It can be seen that the enthalpy of cold crystallization is increased from 5.6 J g−1 for the neat PLA to 14.6 and 25.2 J g−1 for LDH2 and LDH4, respectively, indicating that LDH acts as an efficient nucleating agent for those PLA chains that have not found the opportunity to be crystallized during the cooling process from melt state. LDHs have previously shown their great potential as nucleating agents leading to an improvement in the crystallinity of polymers.
13
In contrast to the LDH-loaded samples, once the plasticizer was also added, the enthalpy for cold crystallization peaks was highly suppressed and the values of Δ
Cooling DSC scans for the neat PLA, LDH-loaded and LDH/TEC-loaded nanocomposites.
Second heating DSC scans for the neat PLA, LDH-loaded and LDH/TEC-loaded nanocomposites.
The quantified parameters obtained from second DSC heating scans for all the samples.
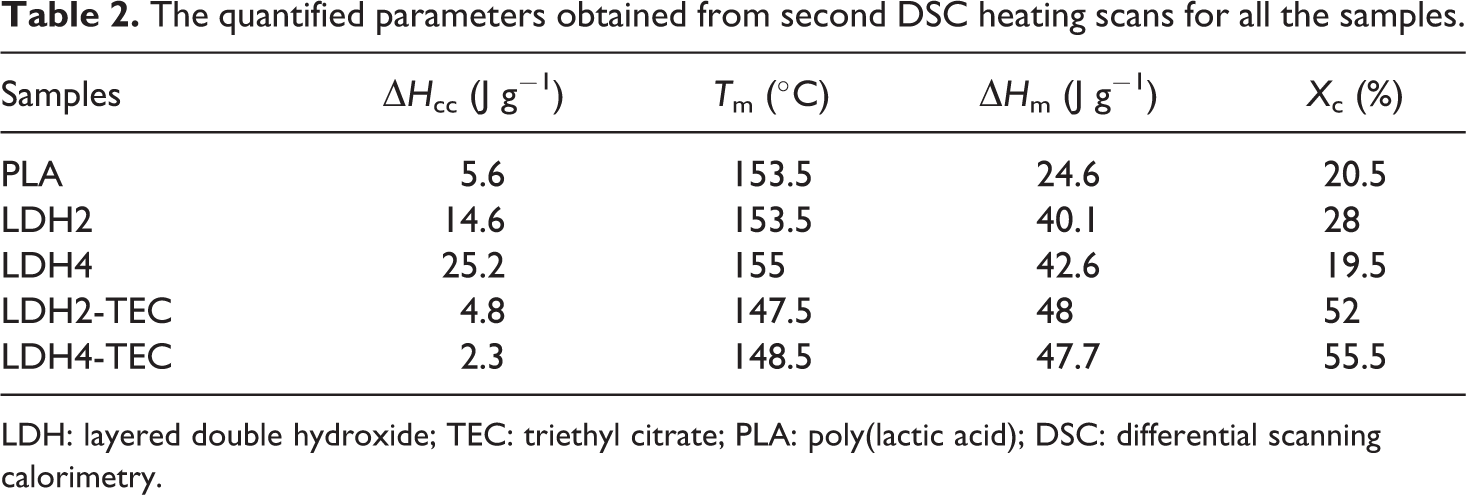
LDH: layered double hydroxide; TEC: triethyl citrate; PLA: poly(lactic acid); DSC: differential scanning calorimetry.
Based on Figure 5, another difference between the first and second heating scans is the appearance of unimodal endotherms for all the samples. This observation means that no recrystallization phenomena has occurred for the neat PLA film and LDH-loaded nanocomposites. Moreover, no α′-type PLA crystals have been formed for the LDH/TEC-loaded nanocomposites, which could be due to the highly suppressed cold crystallization in the case of LDH2-TEC and LDH4-TEC. Based on Table 2,
Quite oppositely, the
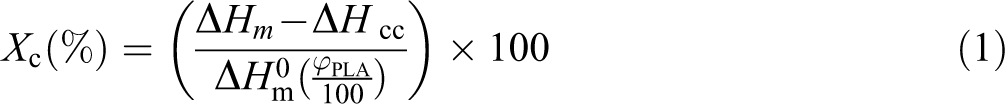
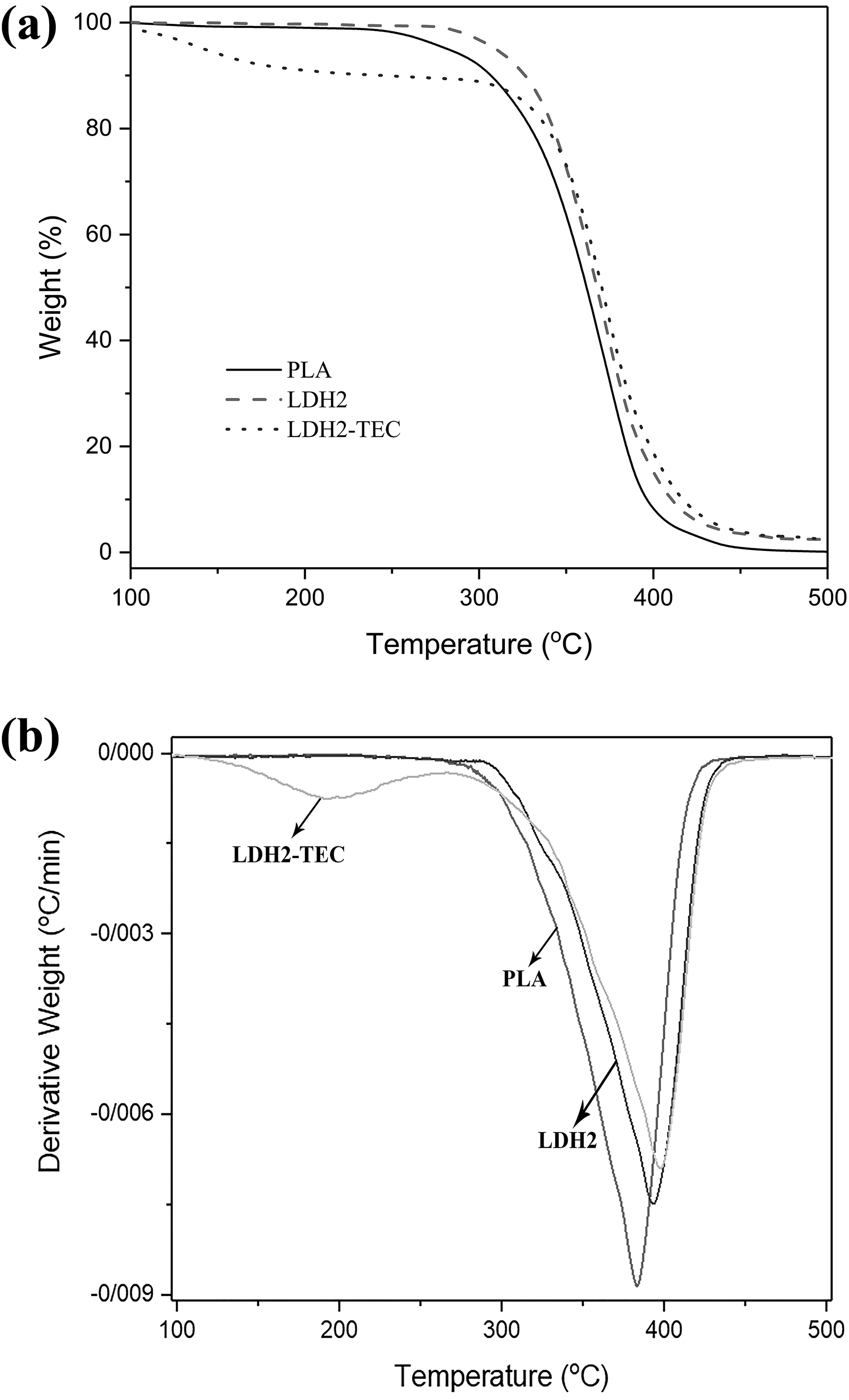
where Δ
A significant enhancement in the degree of crystallization was attained once a combination of LDH and TEC was utilized. The
Mechanical analysis
Addition of plasticizer into PLA formulations usually leads to a reduction in tensile modulus and strength; however, the concurrent utilization of nanoparticles could offset the adverse effects of plasticization. Based on Table 3, the pure PLA film exhibited high tensile modulus (2.3 GPa) and tensile strength (51 MPa) and a low elongation at break (
The mechanical properties of the neat PLA film and PLA-based nanocomposite films.
LDH: layered double hydroxide; TEC: triethyl citrate; PLA: poly(lactic acid).
Thermogravimetric analysis
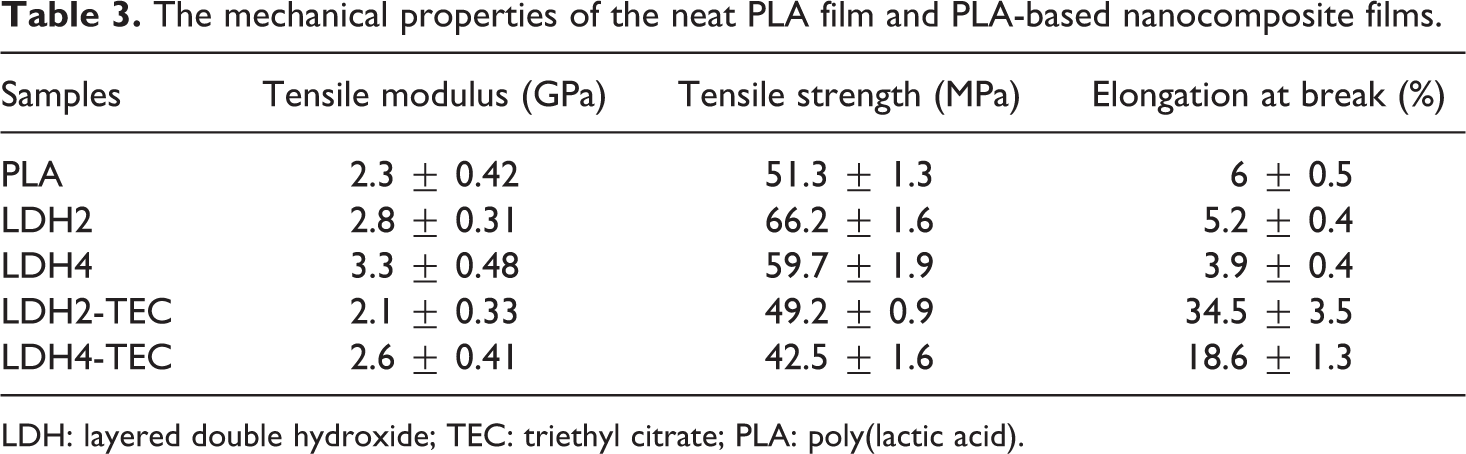
Thermal stability of the neat PLA film and nanocomposites loaded with 2 wt% of LDH was examined by thermogravimetric analysis (TGA). Both TG and derivative weight loss (DTG) curves are shown in Figure 6. As is clear in the TG curves, the onset of degradation is shifted to higher temperatures upon addition of LDH nanoparticles. The degradation is a single-step process for the neat PLA and LDH2; however, two separate stages could be detected for the degradation of LDH2-TEC. The first stage occurs at the range of 120–250°C in which TEC is completely degraded. Afterward, the degradation of PLA starts and the TG curve is slightly shifted to higher temperatures as compared with LDH2, which implies the better dispersion of LDH nanoparticles as a result of TEC incorporation.
(a) TG and (b) DTG curves for the neat PLA film and nanocomposites loaded with 2 wt% of LDH.
Based on the DTG curves shown in Figure 6(b), the onset of degradation is increased from around 275°C for the neat PLA to around 290°C for LDH2, indicating the efficient role of organo-modified LDHs in improving the thermal stability of PLA. Moreover, the peak on the DTG curve, which characterizes the maximal rate of thermal degradation, shifts from 383°C to 393°C upon addition of only 2 wt% of LDH nanoparticles. As stated earlier, Figure 6(b) also shows that concurrent incorporation of LDH and TEC results in the appearance of another degradation step within the temperature range of 120–250°C, which is attributed to decomposition of TEC molecules. As compared with LDH2, the addition of TEC resulted in a slight shift in the DTG peak to higher temperatures. Moreover, TEC slightly reduces the maximal rate of thermal degradation, which has been previously reported. 30 Generally, the addition of LDH resulted in lowering the maximal rate of thermal degradation and also enhanced the thermal stability of PLA majorly due to the barrier role of LDHs, the induced heat insulation and the minimized permeability of volatile degradation products. 17
Conclusions
In the current study, PLA/LDH nanocomposites were fabricated via solution casting technique using different concentrations of LDH and also in the absence/presence of TEC as plasticizer. The organo-modified LDHs were efficiently dispersed into the PLA matrix; however, the higher the content of LDH, the lower the dispersion quality. A large number of aggregations were found in the case of LDH4 due to the rather high concentration of nanoparticles (4 wt%). Addition of TEC was found to be effective in improving the dispersion quality of LDHs, and thus, the number of aggregations was highly reduced. The reason for this positive influence was ascribed to the enhanced chain mobility during the dispersion and drying processes. DSC analysis demonstrated that the crystallization from the solution has not been very effective, and thus, the role of LDH and TEC addition was found to be insignificant. In contrast, LDH and TEC acted synergistically in the crystallization from melt, and consequently, the degree of crystallization was significantly enhanced. Apart from the important role of enhanced PLA chains mobility in the course of crystallization, the positive effect of TEC on dispersion quality of LDHs could also be considered responsible in improved crystallization rate of PLA. In other words, the nucleating role of LDHs was reinforced by the addition of TEC leading to higher degree of crystallization. The synergistic effects of LDH and TEC were also observed in mechanical properties of nanocomposites. In fact, LDH counterbalanced the adverse effects of PLA plasticization by TEC leading to higher toughness of PLA/LDH/TEC systems. Such results could be employed in extending the applicability of PLA in food packaging industry.
