Abstract
This article looks at liquid molding of polyamide 6 (PA6) via vacuum assisted resin transfer molding (VARTM) of discontinuous recycled carbon fiber composites. Its mechanical, thermal, and optical characterization is compared to hydroentanglement/compression molding. Liquid-molded composites show consistent improvement in their tensile and impact properties at three different weight fractions in comparison to hydroentanglement/compression molding. There was roughly a 10 and 13% increase in its tensile strength, modulus, and impact strength properties at 30 and 40% weight fractions and almost a 120% increase at 50% weight fraction. Fourier-transform infrared spectroscopy and differential scanning calorimetry data show that the caprolactam was synthesized to PA6 and was comparable to commercial grade PA6 used in this research. Scanning electron microscopy studies show poor wet out in the case of hydroentanglement/compression molding as compared to VARTM. The combination of better mechanical performance and lower processing temperature (165°C) shows promise in being a viable method to process PA6-based recycled fiber composites.
Introduction
In today’s competitive markets, thermoplastic composites are being used more frequently owing to their superior specific strength, better toughness, and recyclability. 1 The automotive industry alone makes up more than half of the market demand and in the United States alone, the market for composite materials is over a billion pounds. 2,3 Common industrial processing methods of thermoplastic composites include compression and injection molding which yields continuous and discontinuous fiber composites. In recent years, there has been a surge in the use of long fiber–reinforced thermoplastics (LFT) in various industries. 4 These are a class of materials that are comprised of reinforcing fibers that are 5–25 mm in length, compared to 0.5–1.0 mm typically found in short fiber–reinforced thermoplastics (SFT). 5 These longer fibers provide several property enhancements such as high impact strength, improved modulus at elevated temperatures, and better dimensional stability for LFT compared to SFT composites. 6 The most common type used is in the form of pellets which can be injection molded or extrusion-compression molded to form a part. The technology to manufacture LFTs has improved during the last 10 years which has resulted in drastic improvements in the quality of these materials. Currently, compression and injection molding of pellets has been the preferred method of producing LFT composites.
A new subsector being developed in many industries is the use of recycled materials for processing. The main materials being reclaimed in the composites sector are expensive synthetic fibers and high-end engineering thermoplastics from offcuts and end-of-life parts. 7,8 Carbon fiber, due to its high price, seems to be the choice for recycling or reclaiming as much as possible. 9,10 Currently, recycled fibers are either used to make pellets or are used as fillers when combined with thermoplastics. A recent process being implemented is the hydroentanglement or wet-laid process to make preforms using recycled discontinuous fibers. 11 –13
Hydroentanglement is a method to make preforms or mats using discontinuous fibers which can subsequently be used to make thermoset- and thermoplastic-based composites. A common method to make thermoplastic composites is to use chopped thermoplastic fibers dispersed in the preform or mat. It is relatively easy to make thermoset composites using vacuum assisted resin transfer molding (VARTM) via hydroentanglement due to its relatively lower viscosity. In the case of thermoplastics, owing to their high viscosities, is quite difficult to process via VARTM. The logical options to use VARTM for thermoplastics seem to be either high pressure, small flow distances, or lower viscosity thermoplastics. One way to achieve the latter is through anionic ring-opening polymerization (AROP) of lactams. 14,15 AROP is a form of chain-growth polymerization in which the terminal end group of a monomer behave as a reactive center for more cyclic monomers by either having their bonds broken or by the opening of the ring in the monomer. One of the more common studied forms of AROP is the ring opening of cyclic monomer ε-caprolactam to form polyamide 6 (PA6).
As mentioned above, the key element of this manufacturing process is the ring-opening polymerization (ROP) of a monomer called ε-caprolactam, 16 which polymerizes in situ on reinforcements to produce a PA6-based composite. While this process has not been commercially used to a great degree, its genesis is traced back to 1938 by Carothers. 17 The process involved a hydrolytic condensation of dicarboxylic acid and diamine at temperatures greater than 250°C to produce a high molecular weight polymer and water. This was then drawn to thin nylon wires and used in various products. 18 The first use of a catalyst to reduce the polymerization time was performed by Joyce and Ritter in 1941. 19 An alkali earth metal was used, namely sodium caprolactam, to assist in the ring opening of ε-caprolactam in an inert atmosphere, which brought the cycle time down to 90 min. 19 The current, co-catalytic method for anionic polyamide production was discovered by Šebenda and Králiček in 1958 as reviewed by Wichterle. 20 The polymerization reaction was carried out with ε-caprolactam as the monomer, sodium caprolactam as the catalyst, and various imides such as N-acetyl caprolactam acting as initiators. The main advantage of this method was polymerization could be done at a much lower temperature. 20 This meant that the polymerization proceeded to near unity as the thermodynamic nature of the reaction propagated the polymer chains while maintaining a high concentration of the monomer throughout.
This research aims to mechanically, thermally, and optically characterize, quantify, and compare recycled carbon fiber–based PA6 composites via hydroentanglement and ROP of caprolactam via VARTM. This research aims to show the viability of VARTM using ROP as a feasible method to use recycled carbon fibers of various weight fractions.
Materials and methodology
The carbon fibers used for this research are quarter-inch chopped IM7 carbon fibers obtained from Material Innovation Technologies (MIT, Fletcher, NC). Since these fibers were chopped prior to pyrolysis, it was difficult to recoat the discontinuous fibers with sizing in a uniform fashion. As a result, the fibers have no sizing on it. While there is a drop in properties of the fibers post pyrolysis, studies have shown that this drop of its tensile properties is generally around 3–10% 21 –23 The average fiber length used is 0.25 inch (6.35 mm) with an average diameter of 5.783 ± 0.122. This diameter was attained by studying the diameter of 120 fibers taken in a scanning electron microscope (SEM) at 3000× magnification. The diameters were calculated using an imaging software. For the hydroentanglement process, the resin used were nylon fibers with an average length of 0.125 inch (3.175 mm) and a linear density of 6 dpf obtained from Minifibers Inc (Johnson City, TN). For the ring-opening synthesis of nylon via VARTM, the materials used are a combination of ε-caprolactam as the monomer, Bruggolen C1 (caprolactam magnesium bromide) as an activator, and Bruggolen C20 (hexamethylene-1,6-dicarbamoyl-caprolactam) as the initiator.
The processing of hydroentanglement process is shown in Figure 1. The setup was constructed in-house. Mats or preforms made via hydroentanglement are obtained by mixing fibers and resin in water and filtering the mix on a fine mesh.
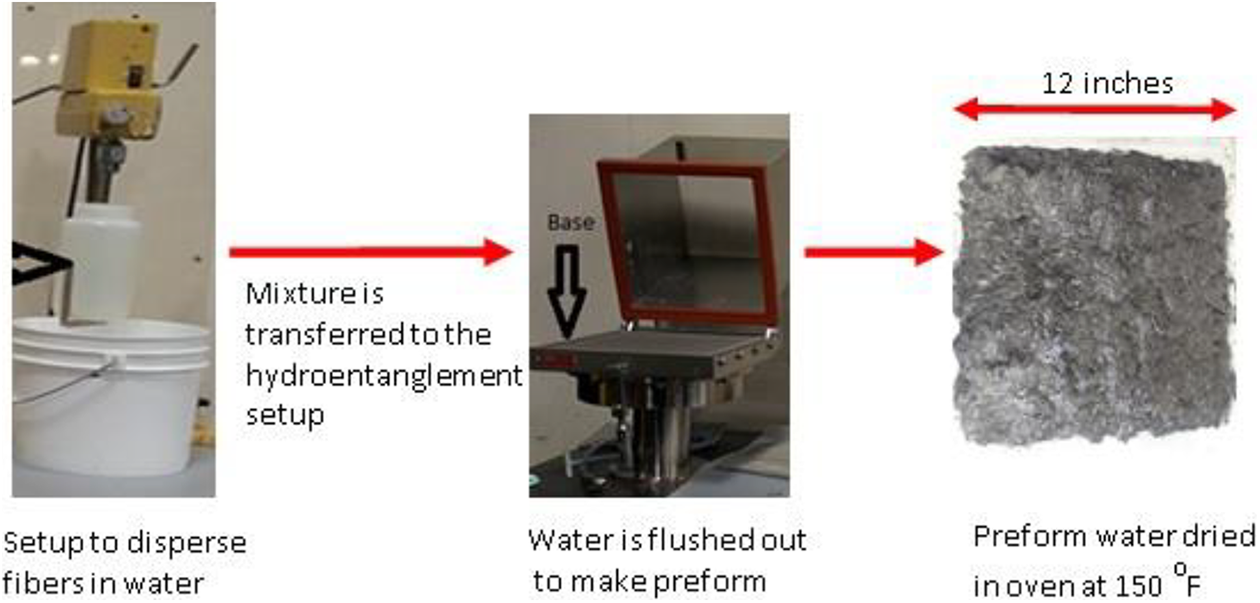
Hydroentanglement process.
The recycled fibers are first agitated for around 20 min in water through a mechanical stirrer. The amount of fiber used is based on the final weight fraction desired. The similar process is done for the resin as well. The individual mixtures are added together and this is agitated via a mechanical stirrer for about 15 min. This mixture is then poured in the container with the mesh placed on the base. The water is filtered out leaving behind the mat. The remaining water in the mat is then removed by placing it in an oven at a temperature of 150°F for about 12 h. The final mat is shown in Figure 2. The final panels are made using compression molding. The layers of preforms were stacked in a 12″ × 12″ tool. Frekote 700-NC release agent, obtained from Northern Composites, was used on the surface of the tool. The layers were then compression molded at 8 ton pressure and at 525°F with a dwell time of 20 min and subsequently air cooled to room temperature.

Final preform via hydroentanglement process.
The method of ring-opening synthesis of caprolactam via VARTM process is as follows. A solution of 7% caprolactam magnesium bromide (Bruggolen C1) by weight of ε-caprolactam and ε-caprolactam monomer is prepared by melting at 100°C under dry nitrogen. Frekote 700-NC release agent is applied to the VARTM platens and air dried. Discontinuous carbon fiber preforms or mats made via hydroentanglement are placed in the VARTM which is then closed and sealed under vacuum. The tool is heated to 165°C to dry the fibers for approximately 1 h. The initiator by weight of ε-caprolactam, 2.5% hexamethylene-1,6-dicarbamoyl-caprolactam (Bruggolen C20), is added to the monomer solution. The panel is infused from the center fill port through vacuum. The tool is held at 165°C for 15 min and cooled in air. A schematic of the setup is shown in Figure 3, respectively.
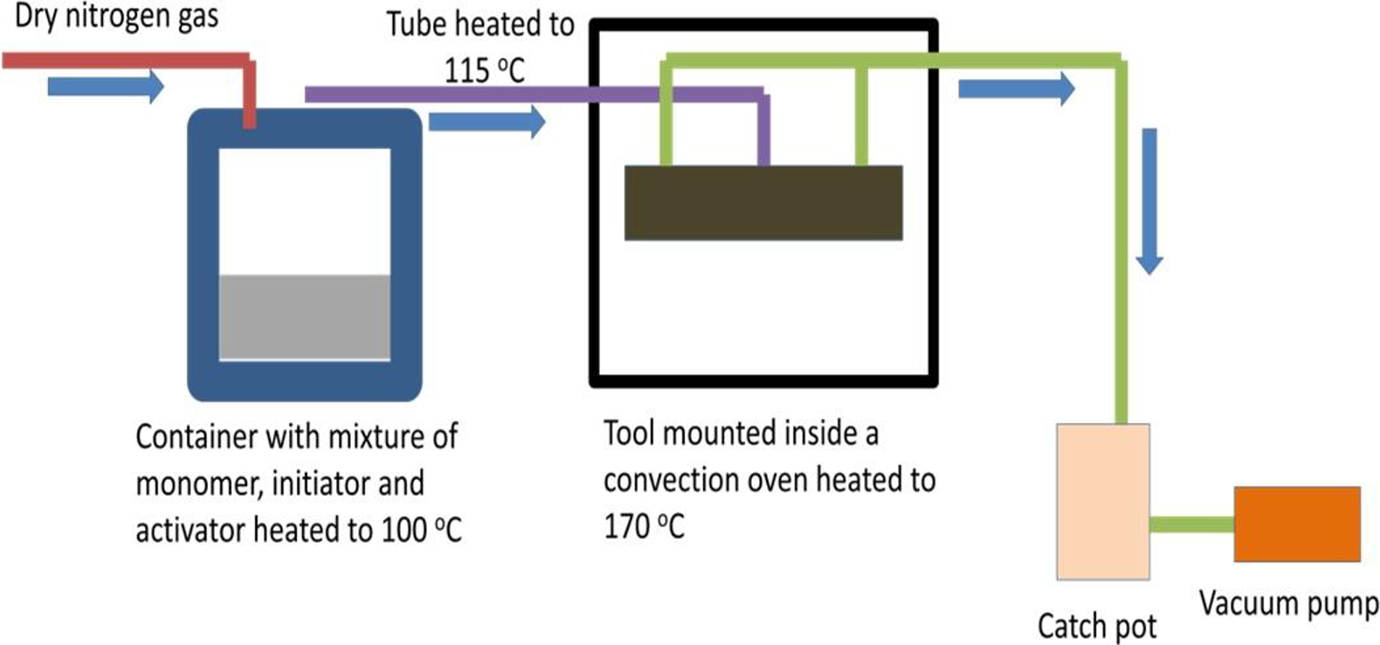
Setup of the VARTM process.
Tensile and impact tests were conducted to evaluate the mechanical performance of the samples. All the samples tested for tensile and impact properties were prepared as per ASTM D3039 and ASTM D256, respectively. For the tensile samples, the ends were tabbed with emery cloth. The samples tested for Izod were notched as per the standard. Fourier-transform infrared spectroscopy (FTIR) studies were conducted to study the spectra of the resin used in order to validate the formation of PA6 via ring-opening synthesis. Differential scanning calorimetry (DSC) analysis was carried out to study the thermal nature of the resin. A heat–cool cycle was conducted between the temperature range of 25–250°C.
The key aspect of these processes is that there is negligible to no fiber attrition. For the hydroentanglement process, the fibers are agitated in water and the shear component during mixing is far too low to break fibers. Also since the fibers are already discontinuous and only 0.25 inch, there is no fiber attrition. As for the compression molding step, it is an application of heat and pressure on the preforms and at this step the fiber attrition is negligible. In the case of VARTM, the monomer mixture used for infusion has a very low viscosity of 42–45 cps. At this low viscosity, there is not enough shear to break fibers.
Results and discussion
The burn off results for the panels made via hydroentanglement and VARTM are presented in Table 1, respectively. The temperature profile of the burn off is shown in Figure 4.
Burn off results for hydroentanglement and VARTM process.
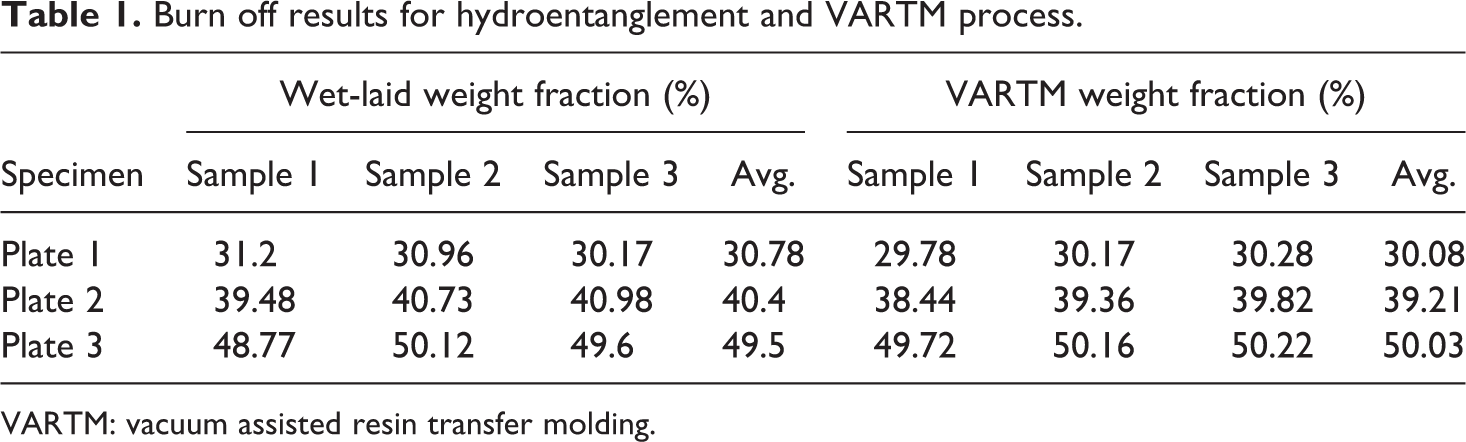
VARTM: vacuum assisted resin transfer molding.
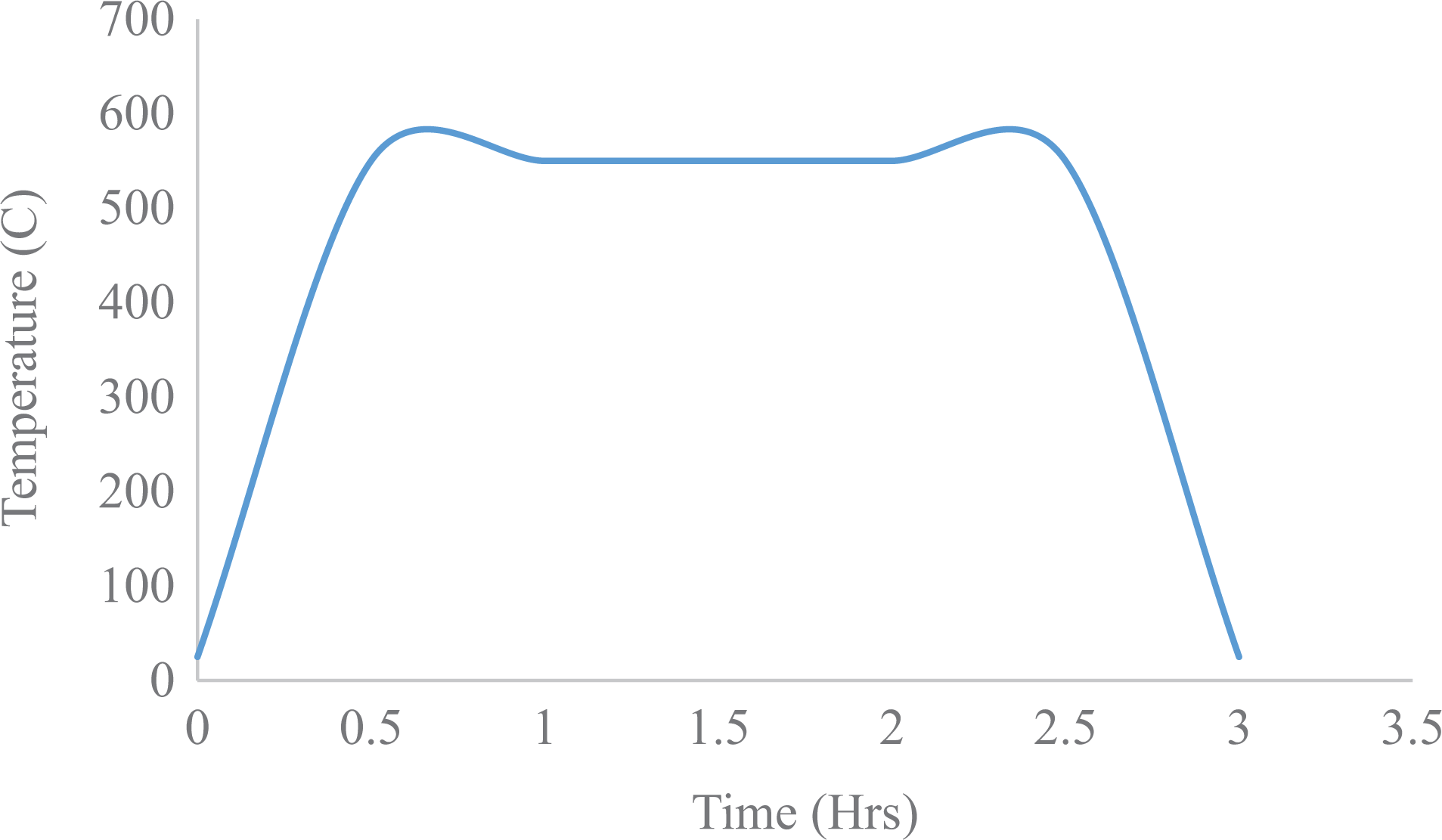
Burn off temperature profile for the samples.
Mechanical characterization
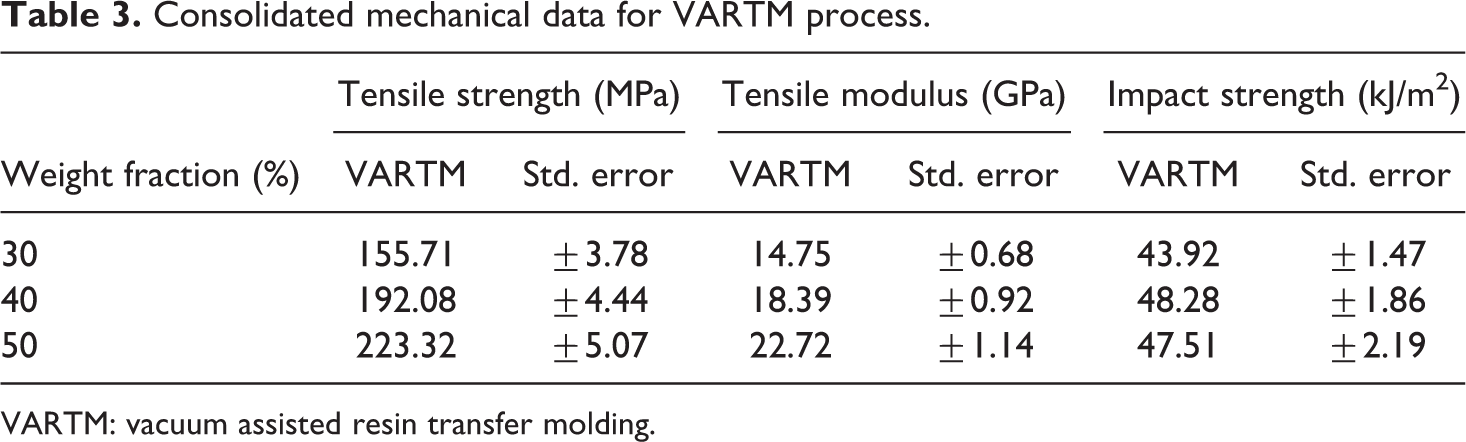
A comparison of the tensile data for the hydroentanglement process and VARTM process is shown in Figure 5. The average tensile strength for the panels of weight fractions 30%, 40%, and 50% via hydroentanglement were 141.43, 172.27, and 92.86 MPa, respectively. In comparison to VARTM process, the panels of similar weight fractions yielded 155.71, 192.08, and 223.32 MPa, respectively. This results in an increase of 10.1%, 11.5%, and 140.5%, respectively, which shows that there is an overall superior performance of panels made via VARTM as compared to hydroentanglement process.
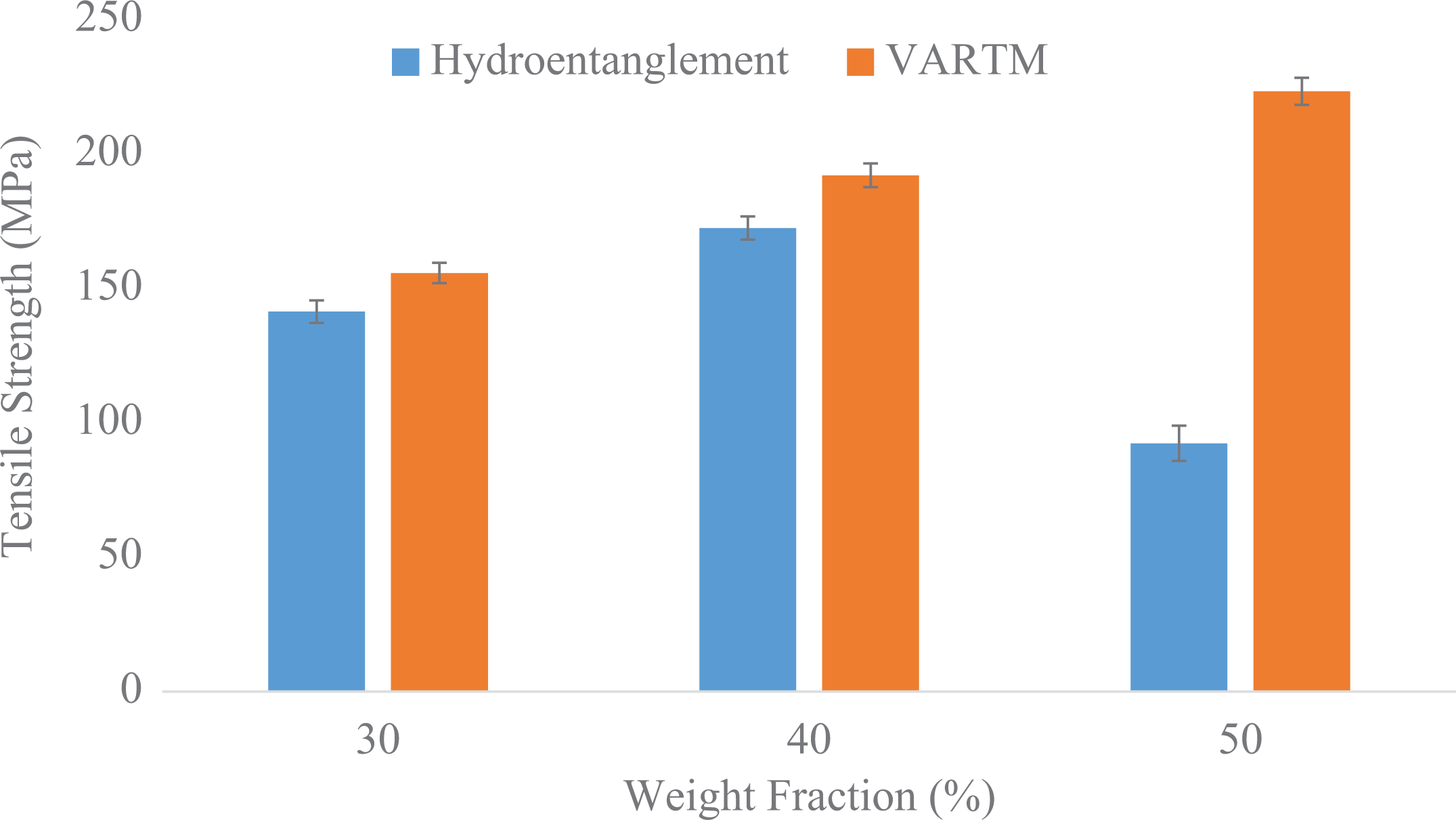
Comparison of tensile strength between hydroentanglement and VARTM process.
It is important to note that there is a significant drop off in the data for 50% weight fraction samples made via hydroentanglement. In fact it can be seen that the properties for hydroentanglement panels fall off after 40%. The main reason for this is the poor wettability of the fibers as the weight fraction increases and is explained in more detail in the “Morphology characterization” section.
Similarly, the tensile modulus for the hydroentanglement panels for the abovementioned weight fractions are 13.49, 17.27, and 10.38 GPa, respectively. As compared to the panels made via VARTM, the modulus was found to be 14.75, 19.39, and 23.72 GPa, respectively. The data are shown in Figure 6. There is an over-improvement in the VARTM panels for the abovementioned weight fractions by 9.34%, 12.27%, and 128.51%, respectively.
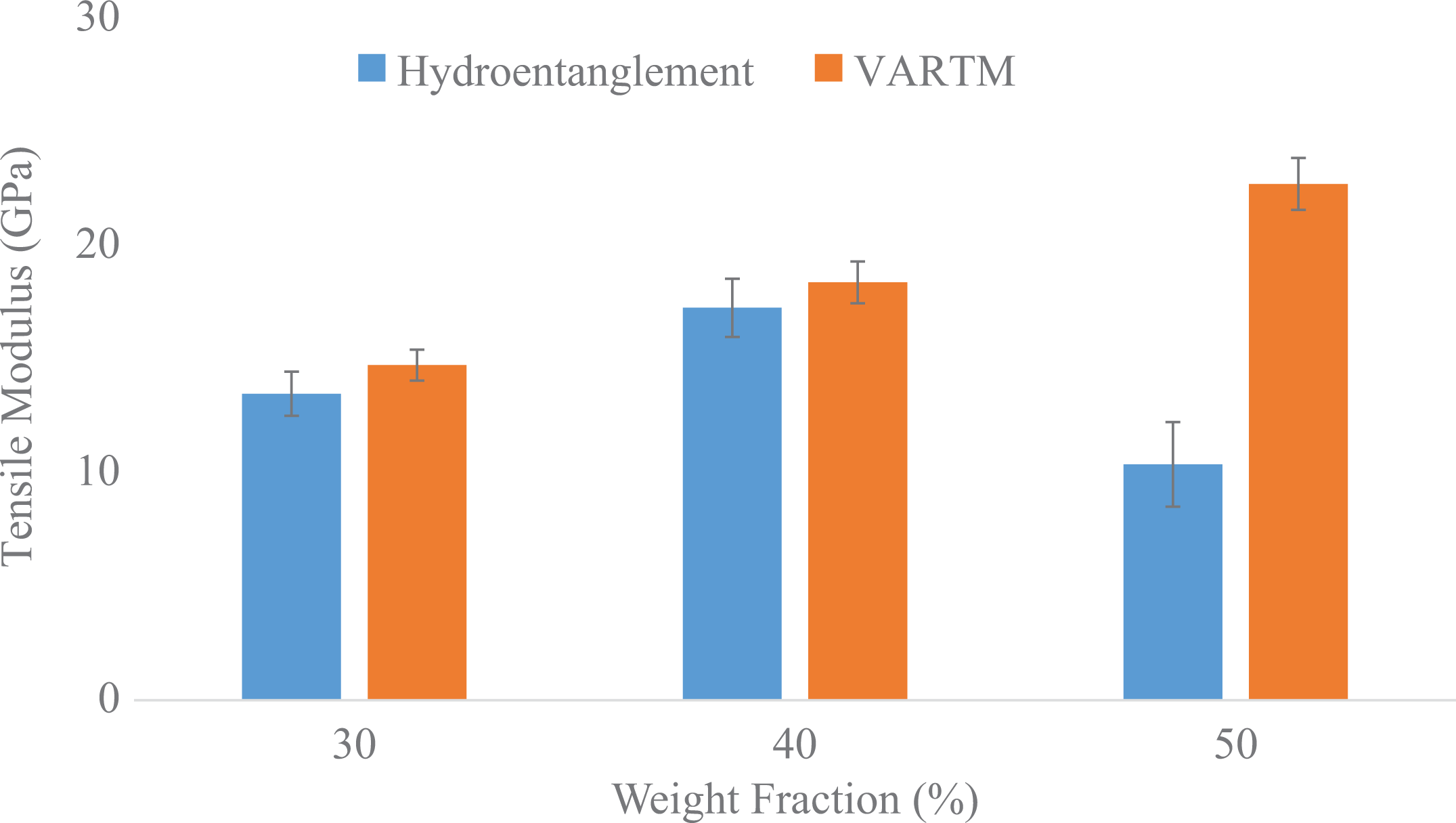
Comparison of tensile modulus between hydroentanglement and VARTM process.
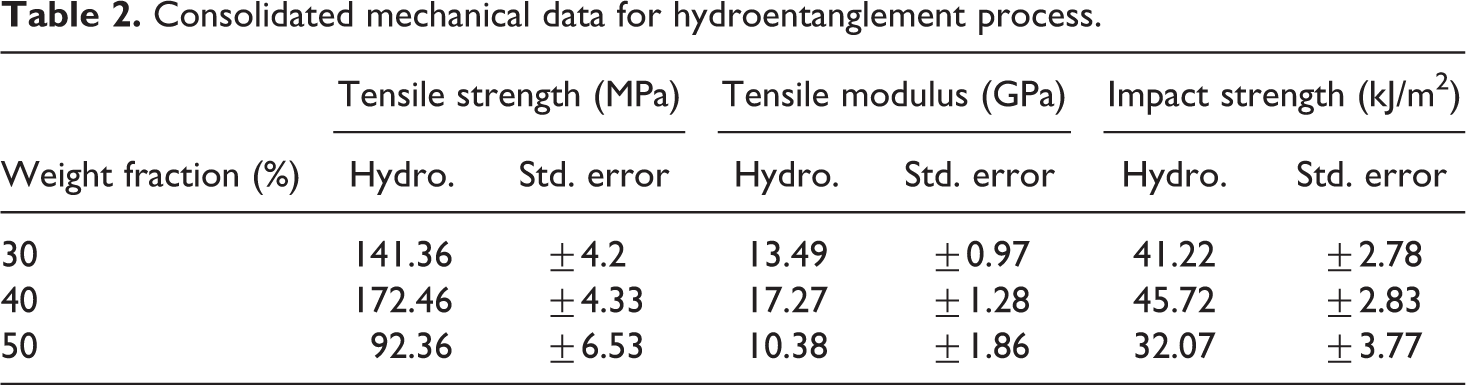
A comparison of the impact data for the hydroentanglement process and VARTM process is shown in Figure 7. The data for the panels of weight fractions 30%, 40%, and 50% via hydroentanglement were 41.22, 45. 72, and 29.07 kJ/m2, respectively. In comparison to VARTM process, the impact strengths were 43.92, 48.28, and 47.51 kJ/m2, respectively. The data follow a trend which seems to be in accordance with the tensile data obtained. The consolidated data for the mechanical tests are shown in Tables 2 and 3, respectively.
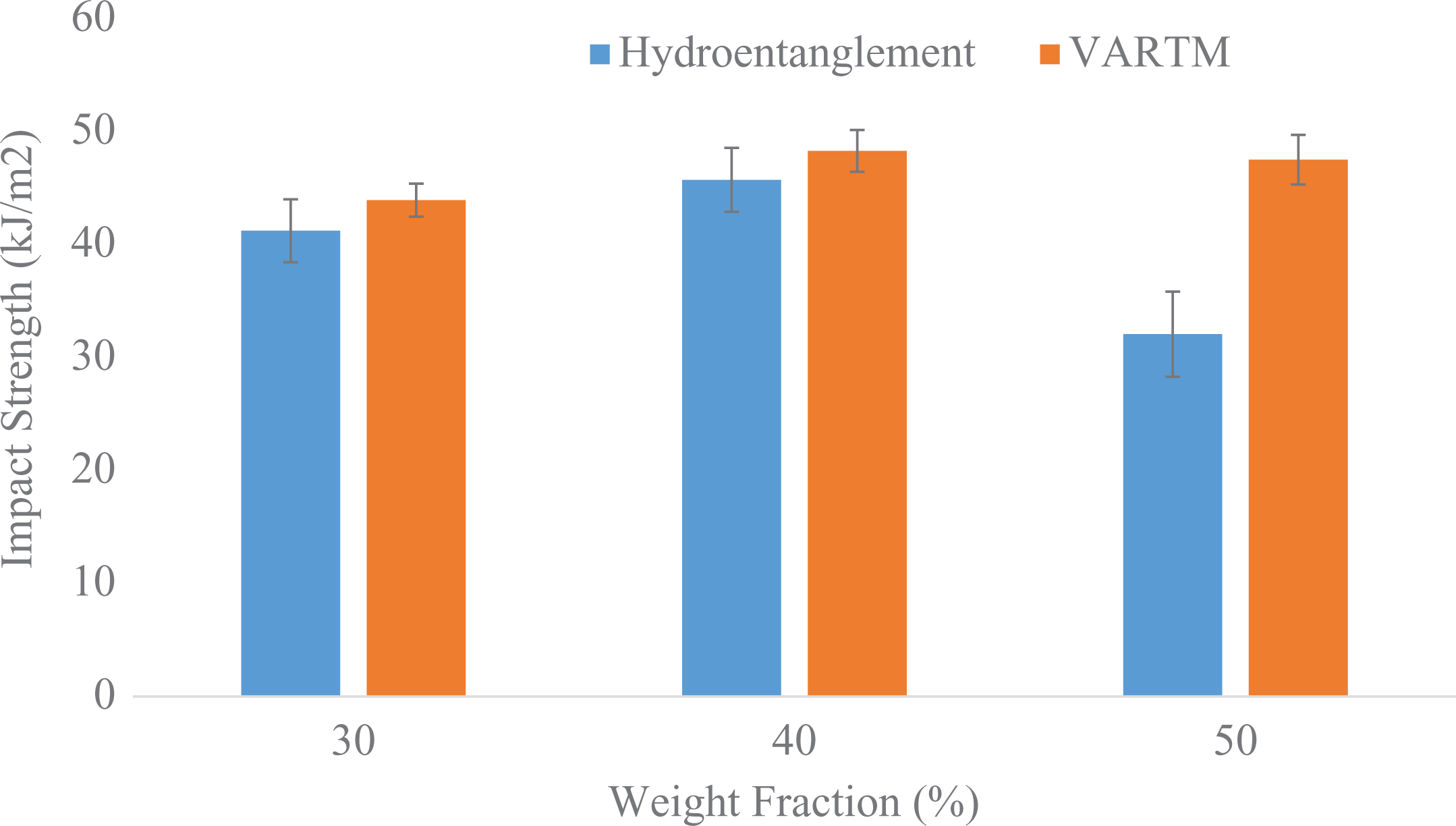
Comparison of impact data between hydroentanglement and VARTM process.
Consolidated mechanical data for hydroentanglement process.
Consolidated mechanical data for VARTM process.
VARTM: vacuum assisted resin transfer molding.
Thermal characterization
The FTIR data for the nylon fibers and nylon obtained via ring-opening synthesis are shown in Figures 8 and 9, respectively. In the case of nylon fibers and the nylon synthesized from caprolactam, the spectra around 3300 cm−1 are assigned to the N±H stretching in the vibrational mode, while those around 1635 and 1540 cm−1 correspond to the C=O stretching in the amide I band region and NH deformations in the amide II band region, respectively. 24,25 These characteristic bands show that it is PA6 spectra.
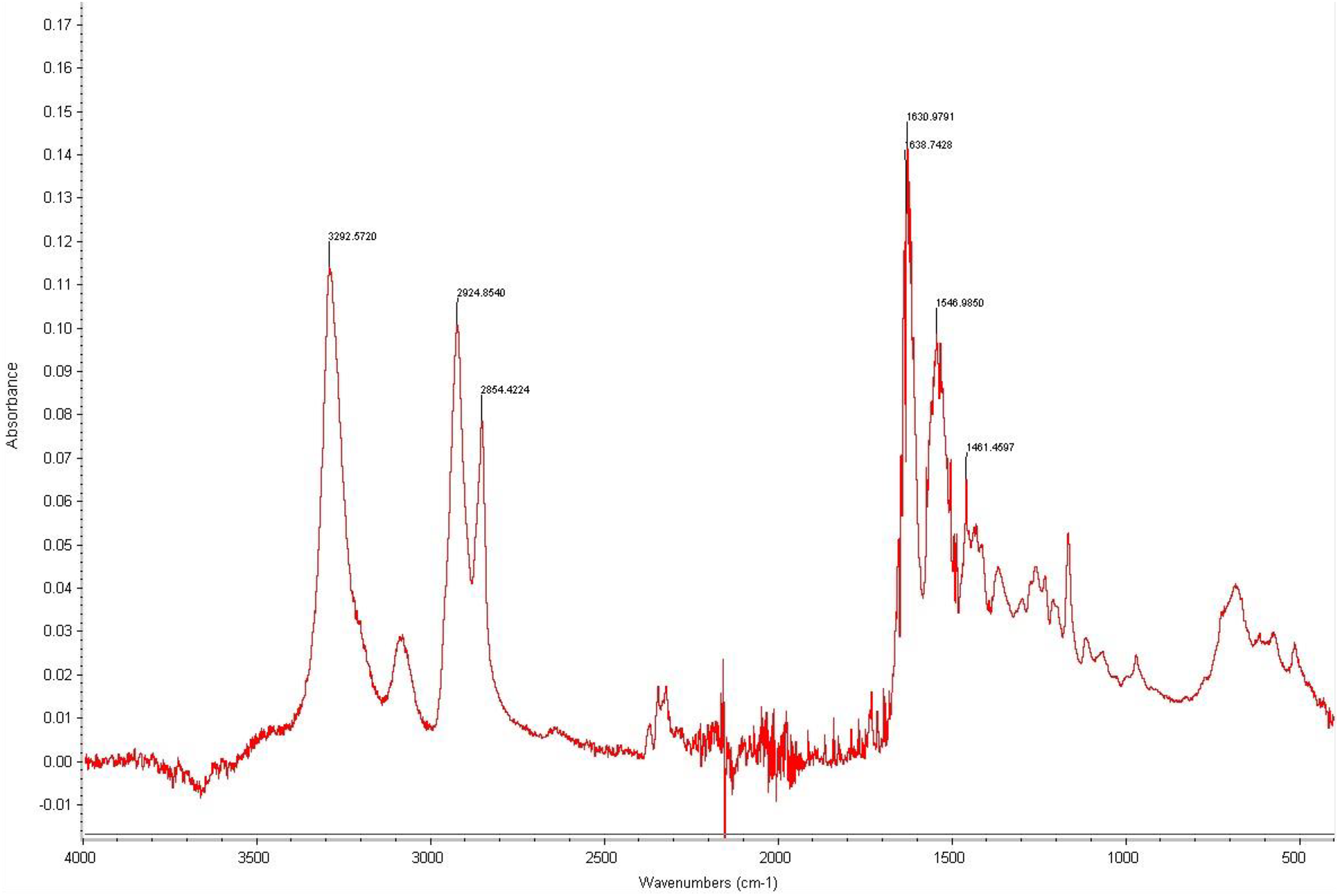
FTIR peak for nylon fibers used in hydroentanglement.
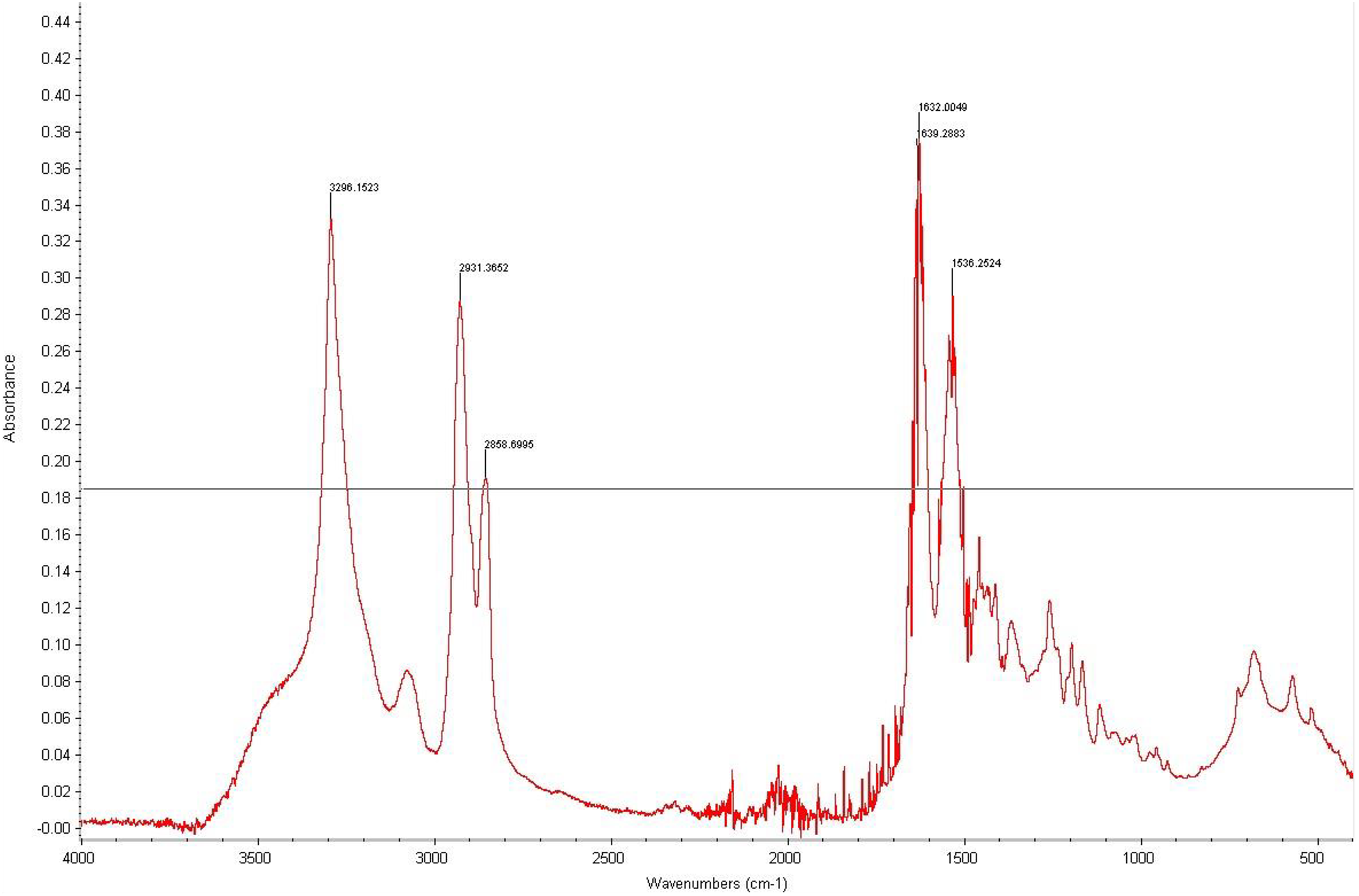
FTIR peak for nylon synthesized via ring-opening synthesis of caprolactam.
The DSC data for nylon fibers and nylon synthesized via VARTM process are shown in Figures 10 and 11, respectively. The glass transition temperature (T g) for fibers used in hydroentanglement was at 48.13°C and for nylon synthesized via ROP was 43.44°C, respectively. The T g for the polymer is dependent on the processing temperatures. Since the VARTM process can synthesize nylon at 165°C as compared to commercially available nylon fibers at 225°C, this allows the synthesized nylon to have a lower T g. This falls between the range of T g seen in the literature. 26 The melt temperatures for the two were 218.51°C and 213.95°C, respectively, which again falls within the range for nylon. This clearly shows the morphology of the two nylons used are very close and in terms of thermal performance.
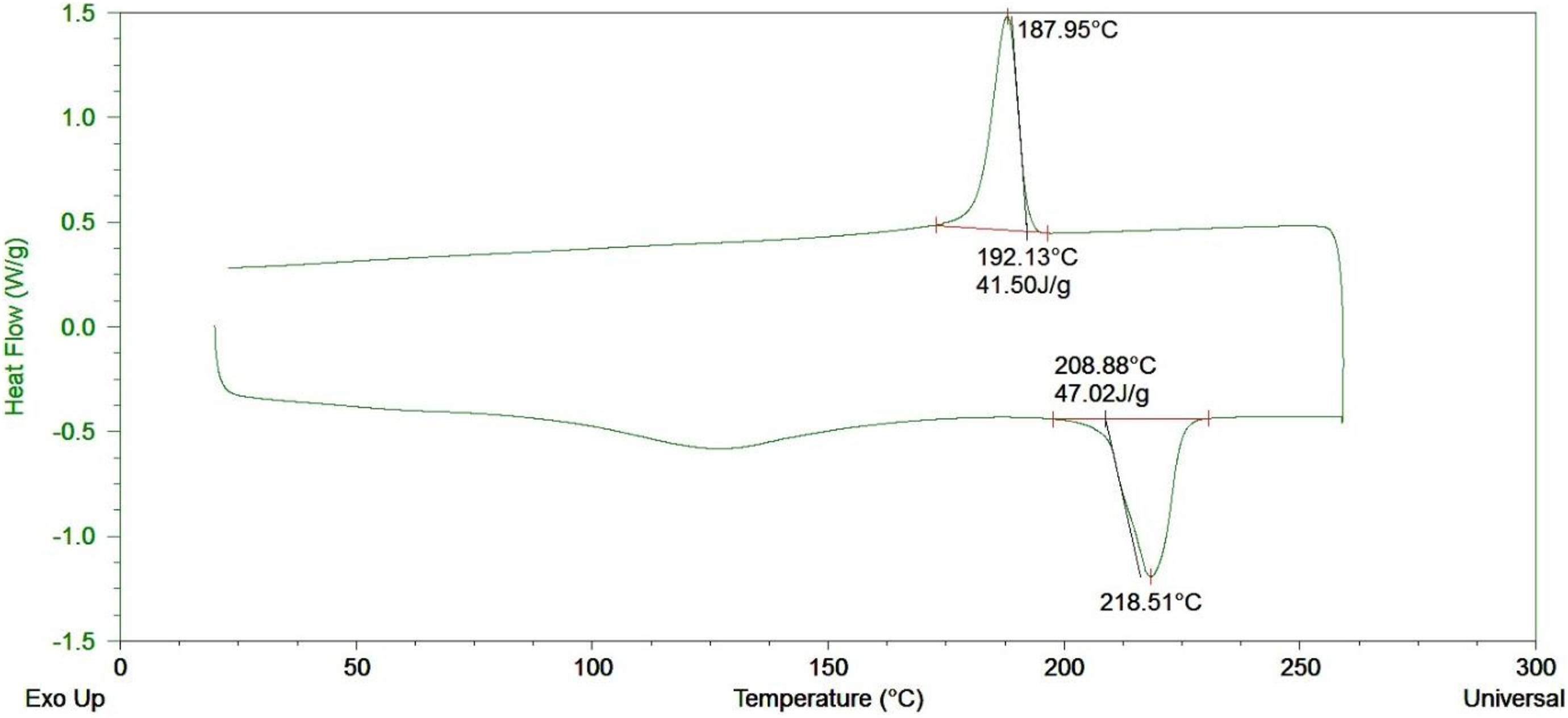
DSC curve for nylon fibers used in hydroentanglement.
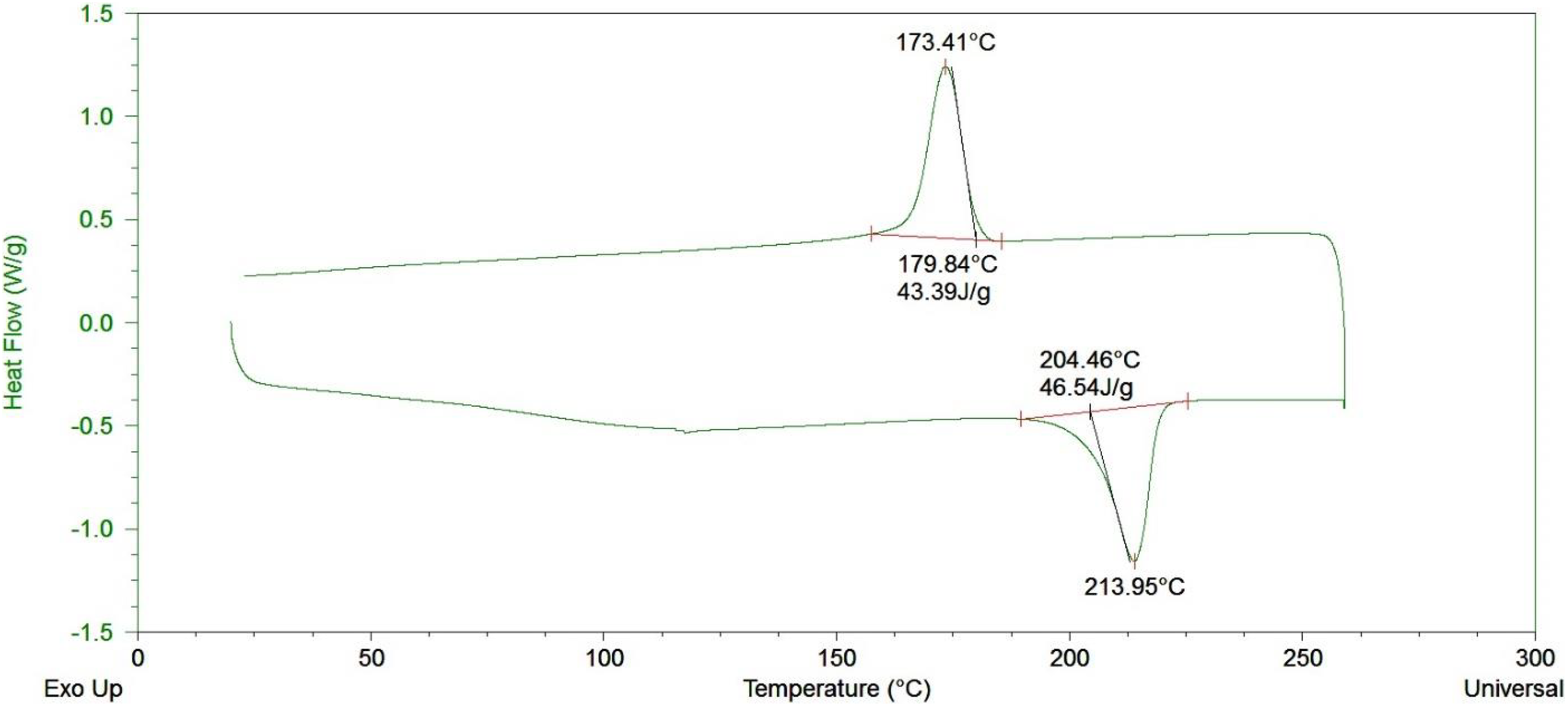
DSC curve for nylon synthesized via ring-opening synthesis of caprolactam.
Morphology characterization
A close-up of the fractured surface was taken using SEM. A depth of field of 5 kV was used to negate the possibility of localized melting of resin. Magnifications of 32× and 500× were used for both the cases, that is, hydroentanglement/compression molding and VARTM, as shown in Figures 12 and 13, respectively.

SEM data for the fractured surface of hydroentanglement specimens: (a) 30% weight fraction at 32× magnification; (b) 30% weight fraction at 500× magnification; (c) 40% weight fraction at 32× magnification; (d) 40% weight fraction at 500× magnification; (e) 50% weight fraction at 32× magnification; and (f) 50% weight fraction at 500× magnification. SEM: scanning electron microscopy.

SEM data for the fractured surface of VARTM specimens: (a) 30% weight fraction at 32× magnification; (b) 30% weight fraction at 500× magnification; (c) 40% weight fraction at 32× magnification; (d) 40% weight fraction at 500× magnification; (e) 50% weight fraction at 32× magnification; and (f) 50% weight fraction at 500× magnification. SEM: scanning electron microscopy; VARTM: vacuum assisted resin transfer molding.
In the case of the samples prepared via hydroentanglement, there is fiber pullout at 30%, fiber and some fiber bundle pullout at 40% weight fraction, and in the case of 50% weight fraction, there is a lot of fiber bundle pullout. This mode of failure is indicative of a weak interface between the fibers and resin which is a result of poor wet out of the fibers. 5 Also there seems to be a correlation between an increase in the amount of fiber pullout and increasing weight fraction. This suggests that at higher weight fraction, owing to the relatively high viscosity of nylon, makes it difficult to wet out the fibers leading to poorer mechanical performance.
On the other hand, samples prepared via VARTM process have a better distribution of resin among the fibers owing to a different failure mode. There is a lot more of fiber breakage at the point of impact which is indicative of a stronger interface leading to better load transfer and ultimately better mechanical performance. 5
The main reason for this in the case of VARTM is that the initial monomer mixture is heated to 100°C and at this temperature the viscosity is very low as shown in Figure 14. 27 As the mixture enters the tool, which is at 165°C, there is a delay of a few seconds or “induction time” before polymerization begins and then there is a drastic rise in its viscosity as polymerization occurs. 28 This induction time allows the low viscosity mixture to wet out the carbon fibers better thus leading to a stronger interface as the mixture polymerizes.
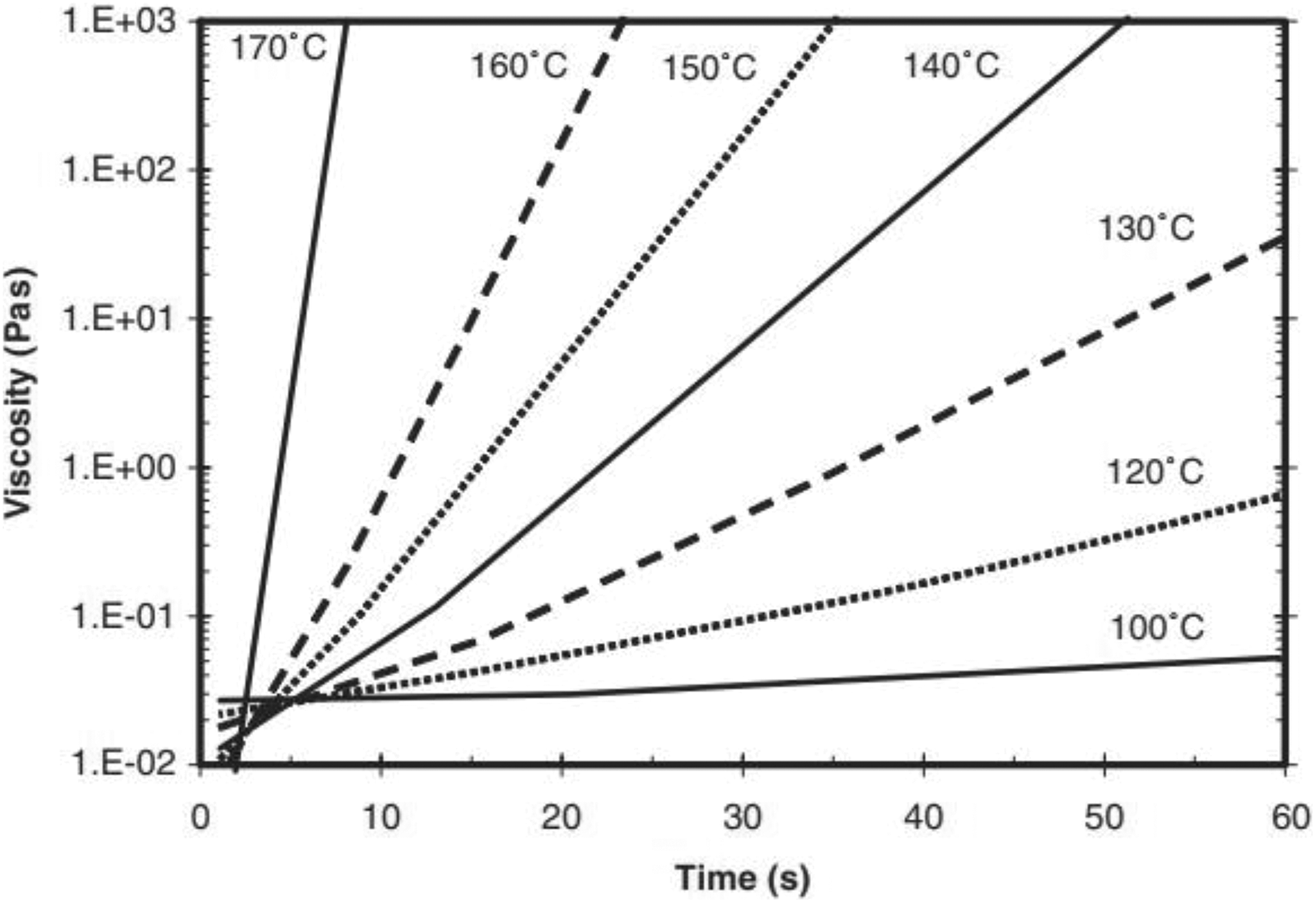
Time viscosity curves at different temperatures as adapted from Sibal et al. 27
Based on the mode of failure seen in the SEM images, hydroentanglement process has an issue of wet out at higher weight fractions due to the inherent high viscosity of the PA6 used. This results in a weak interface through the panel and thus results in a poor translation of its macro mechanical properties. On the other hand, VARTM process shows significantly higher wet out based on the failure mode leading to a stronger interface. Even at higher weight fractions, there is significantly better wet out of fibers leading to superior macro mechanical properties.
Conclusions
VARTM, a process predominantly used for thermoset systems, has been successfully used to infuse discontinuous recycled carbon preforms with PA6 matrix via liquid molding. When compared to hydroentanglement/compression molding, a process currently used for recycled fiber–based thermoplastic composites, VARTM showed superior mechanical performance especially at higher weight fractions. While there was an increase of roughly 10% and 13% in its tensile and impact properties at 30% and 40% weight fractions, there was roughly a 120% increase in its properties at 50% weight fraction. FTIR studies showed that the commercial nylon fibers and the synthesized nylon showed characteristic peaks around 3300, 1635, and 1540 cm−1 corresponding to N±H stretching vibrational mode, C=O stretching in the amide I band region, and NH deformations in the amide II band, respectively. DSC data showed that the thermal behavior of the synthesized PA6 was comparable to commercially obtained PA6 fibers in terms of its T g and melt temperature. SEM studies revealed that there was a poor wet out of fibers in the case of hydroentanglement leading to fiber pullout at lower weight fractions, and at higher weight fractions of 50%, considerable fiber bundle pullout due to a weak interface bond between the fibers and PA6. This poor wet out was more pronounced at higher weight fractions thus leading to a significant drop off in its mechanical properties. VARTM specimens however showed a more consistent wet out of the fibers and as a result had more fiber fractures indicating a stronger interfacial bond. This in turn translated to better and more consistent mechanical properties. The main reason for better wet out seems to be the initial low viscosity of the caprolactam mixture when infused.
