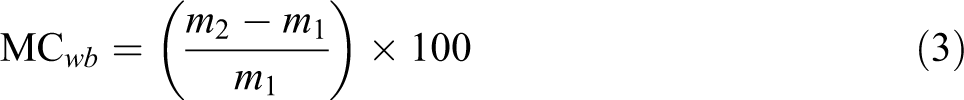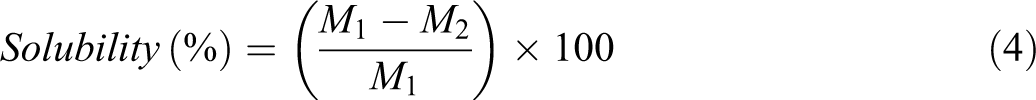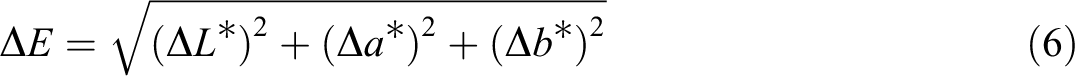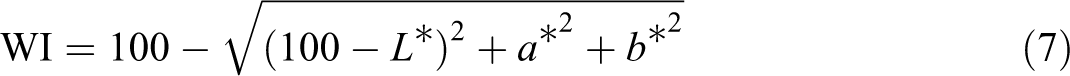Abstract
In this study, biodegradable/antioxidant active film based on potato starch was provided. Composite films of potato starch/Zedo gum/Salvia officinalis essential oil were provided in different percents of Zedo gum (0.5–1.5%) and S. officinalis essential oil (0–500 µl). The characters of composite film, including physical, mechanical, and antioxidant properties were studied. The results showed that adding different amounts of Zedo gum and essential oil to starch film causes a significant decrease (p < 0.05) in moisture content, solubility, and film water vapor permeability (WVP). The film containing 1.5% gum and 500 µl of essential oil had minimum moisture content, solubility, and WVP. Results showed that the addition of Zedo gum significantly increased the thickness and opacity of the films and reduced the transparency of the films. Increasing the essential oil is significant on the film opacity and leads to a decrease in the passage of light from the films. By adding different amounts of gum, the antioxidant content of the films was significantly increased compared to the control film (pure starch film). Also, increasing the concentration of essential oil in the film composition caused a significant increase in the antioxidant activity of the films (p < 0.05). Modified starch films containing 1.5% gum (without essential oil) with 62.64% antioxidant activity and film with 1.5% gum and 500 µl essential oils with 68.35% antioxidant activity had the highest antioxidant activity. The addition of salvia essential oil and Zedo gum significantly decreased the elongation and tensile strength of the potato starch film (p < 0.05). Finally, the potato starch composite film containing Zedo gum and salvia essential oil showed excellent physical and antioxidant properties and had an excellent ability to be used in the food packaging industry.
Keywords
Introduction
In the world, about 125 million tons of plastic are produced, of which about 30 million tons are consumed in packaging. Polymeric films are widely used in packaging industries due to their easy plasticity, cheap price, lightness, high chemical resistance, and a variety of physical properties, heat sealing capability, good printing, and easy manufacturing process. Although these polymers have different benefits, using these materials causes different problems like environmental pollution. These problems have attracted researchers in recent years to the use of natural biodegradable polymers in the production of packaging materials. 1 Biopolymers are considered as an environmentally friendly substitute for nonrenewable packaging materials. Biodegradable films are biomaterials that are made from natural and renewable resources and are easily decomposed after consumption under appropriate conditions of humidity, temperature, oxygen content, and naturally occurring metabolites of living organisms. These materials decompose and do not leave any toxic or harmful substance in the environment. 2
Starch is a polysaccharide that can be produced at a low cost. Due to the nature of starch, it has good filming ability, but because of some disadvantages it cannot produce a good film alone. The starch film has some disadvantages, including strong hydrophilic capacity and poor resistance to moisture as well as its poor mechanical properties compared to synthetic polymers, which limits the use of this biopolymer in various fields. 3 Different strategies have been proposed to overcome the limitations mentioned in relation to starch films, one of which is the use of hydrocolloids (gums) in combination with starch. The combination of hydrocolloids and starch changes the properties of starch films. For example, hydrocolloids can reduce retrograde rates. 4 Zedo gum (usually called Persian gum) is produced in three colors: white, yellow, and red from the trunk and branches of the almond tree. This tree grows often in the central regions of Iran. 5 Persian gum is found in white, light yellow, vegetable yellow, red, and brown, which is graded in this regard. Tannins are the natural polyphenols that are found in many herbs, including many foods, such as vegetables, fruits, and seeds. Tannin, or the tannic acid, is a mixture of glucose esters with glycine and 3-galvile gallic acid. Because gum contacts with the tree body, it can absorb the tannins from the tree, and with increasing life of gum on the tree, the amount of tannin increases and different degrees of yellow appear in the gum. 6 In recent years, attention has been focused on the active packaging in food production, which is due to the fact that this type of packaging is not only responsible for the protection of foodstuffs against agents, but it also has other benefits, such as the ability to determine the shelf life of the food using various sensors and reagents. 7 Biodegradable packaging based on edible films, which are mainly made of polysaccharides, proteins, fats, or a combination of them, due to the availability of natural ingredients, the ability to be renewed, and the lack of environmental pollution has special importance. On the other hand, the potential of these biodegradable films as carriers of antimicrobial and antioxidant agents for improving the quality, increasing the shelf life, controlling pathogens, and improving the organoleptic properties of the foodstuff causes increased application of these polymers in the food packing industry. Today, to reduce the use of chemical additives, the use of natural additives has been increased due to their antioxidant and antimicrobial properties as well as they have no negative effect on human health. 8 The use of antioxidant active packaging is one of the newest techniques for preventing the oxidation of oils and fats in foods. By releasing antioxidants from this type of packaging during storage of food, oxidation of fatty acids can be reduced. 9 Phenolic compounds and polyphenols are one of the most important phytochemical compounds due to the presence of hydroxyl groups in their chemical structure as a source of natural antioxidants. 10 Essential oils are extracted from herbs and spices that have antioxidant and antimicrobial properties. In addition, most of them are known as GRAS (generally safe). Although, their use as preservatives has been limited due to their strong flavor, but to solve this problem they can be used in edible films. Due to their (essential oil) fatty nature, these compounds are expected to reduce the vapor permeability in hydrophilic films. It has been shown that these materials have antioxidant and antimicrobial properties and can also change some film properties (stretching, appearance, and structure). 8 Salvia officinalis belongs to the Lamiaceae family. The leaves of this plant contain essential oil, tannin, and a bitter compound. 11 The essential oil of S. officinalis is usually produced from the wild type of the plant and is a yellowish–yellowish–greenish-colored liquid with a special smell. In addition, this essential oil has pinene, cineol, and borneol. The S. officinalis leaves contain organic acids, espresso oil, and saponin, which facilitates digestion, diuretic, augmentation, and bacterial growth retardation. The essence of the S. officinalis is very rich in the amount of camphor, and the amount of essential oil and essential oil of cinnamon is the most during flowering. The amount of cineol in the essential oil of freshly dried branches is about 25%. 12
Considering the many benefits associated with the essential oil of S. officinalis and Zedo gum, this study attempts to make a composite film of potato starch containing Zedo gum and essential oil of the S. officinalis. The composite film has an antioxidant active film characteristic and has the ability (physically and mechanically) to be used as an edible and biodegradable film in food packaging. 13
Material and methods
Materials and chemicals
Potato starch was provided by the German Merck company. Zedo gum from Freer Isfahan Company and essential oil of S. officinalis from Zardband Co (Isfahan, Iran). Yasouj were prepared. Glycerol and waterless calcium chloride were purchased from Sigma.
Preparation of Zedo gum solution
First, the gums with clear color were separated and powdered with a laboratory mill. After that, the powder of gum was passed through a sieve with 250 µm pores to obtain a uniform powder. The resulting powders were kept in non-permeable plastic bags until they were consumed. To prepare various concentrations of gum, gum powder (0.5, 1 and 1.5 g) dissolved in 100 ml of distilled water and stirred at magnetic stirrer for 1 h at 50°C. The obtained solutions were stored at 4°C for one night to complete the hydration process. 14
Film preparation
Starch (5 g) and 2 ml of glycerol were dissolved in 100 ml of distilled water and were heated and stirred at 90°C for 30 min. Different percentages of gum (0.5%, 1%, and 1.5%) were added to the starch solution for 10 min at 75°C with stirring. The solution was then cooled down to 40°C, then the essential oil of the S. officinalis was added at 250 and 500 µl to the solution for 10 min, then the final solution was stirred slowly for 60 min to remove the air bubbles. Then, 100 ml of the solution was dispersed into Teflon containers with a diameter of 18 cm and dried at 60°C for 18 h. 15
Test methods
Thickness measurement
To determine the thickness of films, micrometers were used with a precision of 0.01 mm. Measurements were performed in five different film regions and then their average was calculated. The average calculated thickness was used to determine the tensile strength and vapor permeability. 16
Measuring the water vapor permeability
The ASTME-96 standard was used to calculate the permeability to vapor. First, in the falcons (with a crateralarea of 0.0019625 m2), 2.5 mg of anhydride calcium chloride was discharged, and the flank of the falcon was covered with no wrinkles, bubbles, and holes with the help of molten paraffin. To maintain a relative humidity gradient of 75% of the films, a sodium chloride saturation solution was used inside the desiccator. The relative humidity difference in the two sides of the film at 25°C creates a pressure of 1753.55 Pa. In this way, the weight changes of the falcons were measured over time using a digital scale. The weight gain curve of the falcons was drawn over time and, after calculating linear regression, the slope of the resulting line was calculated. The water vapor transmittance (WVTR) and water vapor permeability (WVP) were calculated according to the following equations 17
where WVTR is in g/m2 s; L is the film thickness (m); ΔP is the partial difference in relative vapor pressure between the two sides of the film (Pascal); A is the film area (m2); WVP is in g/m s Pa.
Humidity measurement
To determine the film moisture content, the films were cut in 2 × 2 cm2, after weighing m 1 for 24 h at 100°C. Then dried specimens, weighing m 2 and moisture content of films were obtained using equation (3) 18
where MCwb is the moisture percentage based on relevant weight; m 1 is the sample weight before drying (g); m 2 is the sample weight after drying (g).
Measuring solubility
For the test, films that were cut in 2 × 2 cm2 and placed in an oven (100°C) for 24 h were weighed (M 1) and placed into a container (containing 50 ml distilled water). The container was placed in a shaker incubator (70 r/min and 25°C) for 24 h. The remaining pieces were then placed in the oven (100°C) for 24 h and then weighed (M 2). The solubility of the films was calculated using equation (4) 18
where M 1 is the dry matter of the film and M 2 is the insoluble matter of the film.
Opacity
The film opacity was determined by UV-Vis spectrophotometer (model UNICO 2100, UNICO-US) at defined wavelengths. For this purpose, 1 × 4 cm pieces were cut from the films and their thickness was measured at five points along the film, and then the film was placed in a transparent wall in the quartz tubes, and the sample absorption was read. The light transmittance was calculated from equation (5) 1
where A600 is an absorbance at wavelength 600 nm, and X is the average of the film thickness (mm).
Measure the surface color of the films
Colorimetric instrument (Minolta Model CR-410 Japan) was used to measure the surface color of the films. First, the device was calibrated with standard white pages, and then the films were placed on a white paper and their color was measured. This instrument measures four wavelengths in the area of the visible area waves. The results include the three color amounts with the quantitative indexes L*, a*, and b*, which respectively represent the brightness (from L = 0 for black to L = 100 for white), green to red (a = −60 for green to a = 60 for red) and blue to yellow (from b = −60 blue to b = 60 for yellow). Using the relationships, the values of the total color difference (ΔE), white index (WI) and chroma (C*), which show the gray color and the impurity of the film, were calculated according to the following equations
19
Determine the antioxidant content of the films
In this test, 2,2-diphenyl-1-picyrylhydrazine (DPPH) was used as a stable radical combination. Subsequently, pieces of the film (2 × 3 cm2) were immersed in ethanol for 10 min and vortexed until the digested film and antioxidant compounds were released. After centrifugation for 10 min at 3000 r/min, a topical ethanol solution (sample solution) was used to determine the DPPH inhibition. DPPH (2 ml) solution (0.06 mM, ethanol is solvent) was mixed with 1 ml of sample solution and 1 ml of ethanol. The control sample was prepared by mixing 2 ml of ethanol and 2 ml of DPPH solution and absence of sample solution of the film. After stirring for 1 min, the mixture was placed in a dark place at 25°C for 30 min, and then its absorbance was measured at 517 nm by a spectrophotometer (UV-2100, USA). The DPPH free radical activator inhibitory power (antioxidant activity) was calculated using equation (9) 20
where Abscontrol is the DPPH ethanolic sample absorption at 517 nm; Abssample is the ethanolic film sample absorption at 517 nm.
Determine mechanical properties
The stress–strain test is one of the most popular tests for determining the mechanical behavior of the films. Tensile tests were measured using the TAXT Tissue Tester (Plus, Stable Micro Systems UK) and according to Standard (ASTM D882-91). Before analyzing, samples were conditioned in a desiccator (saturated calcium nitrite) for a period of 24 h at a relative humidity of 55%. Then three samples from each of the films were cut with dimensions of 8 × 0.5 cm and placed between two jaws of the instrument. The initial distance between the jaws was 50 mm, and data were recorded by the computer. Measuring factors include tensile strength and elongation percentage to tear point. The tensile strength represents the maximum strength of a film against the applied tensile stress. 21
Fourier transform infrared spectroscopy
For Fourier transform infrared (FTIR) spectrometry, thin films with a thickness of less than 1 mm were provided. By mixing of a film sample with dry bromide potassium in a ratio of 1:20 and applying a pressure of about 60 kPa, a tablet was obtained, and the sample spectrum was obtained in the range of 500 to 4000 cm−1 with a resolution of 0.5 cm−1.
Scanning electron microscopy test
The structure of the films was investigated by TescanVegan-3 (Brno -Kohoutovice, Czech Republic) scanning electron microscope. To investigate the effect of the addition of Zedo gum extract and essential oil on the microstructures of the produced film, electron microscopic images were obtained from the film surface. Imaging of the samples was performed by scanning electron microscopy (SEM) at 15 kW.
Statistical analysis
In this study, to study Zedo gum variables (0.5–1.5%) and essential oil of S. officinalis (0–500µl), a factorial design with three replications was used. Minitab (version 17) software was used for data analysis and Tukey’s test was used to compare the means. Also, significant levels of data were considered at 5% probability level (p < 0.05). Meanwhile, the data in tables and forms were considered as a mean standard deviation (SD).
Result and discussion
Thickness, permeability, humidity, and solubility of films
Table 1 shows the comparison of average thickness, permeability, humidity, and solubility. Thickness is one of the important factors in films that directly affects the properties of vapor and oxygen permeation, as well as mechanical properties, and therefore this factor can affect the biological properties and shelf life of the packaged product. In all of these tests, the control sample had a significant difference compared to other samples (p < 0.05), so that the control sample had the least thickness between the samples. By adding different amounts of gum extract and essential oil to the control sample, the thickness of the films has increased significantly. The samples containing essential oil and gum-free have different thickness compared with samples that contain essential oil and gum. The samples containing essential oil and gum had a higher thickness than gum-free samples. Zedo gum can absorb water in the single-layer region and can increase dry matter of films, so removes water less than a control sample while drying. The total of these changes increases the thickness of the produced films. Various results have been reported on the effect of essential oil on the thickness of edible films depending on the nature of the essential oil composition and its non-polarity. Ghadermarzi et al. reported the effect of oregano essential oil on the characteristics of hydroxypropyl methylcellulose film. They reported that there is a significant difference between the thickness of films without essential oil and the film loaded with oregano essential oil. These researchers said that the reason for the lesser thickness of the essential oil film is due to the presence of essential oil in the empty spaces between the polymer chains. Due to the hydrophobic nature of essential oil, it can also be effective in preventing high moisture absorption by glycerol combined with the film, while glycerol can absorb the maximum moisture content in film without essential oil and increase the thickness of the film. 22 Also, Ghadermarzi et al. reported that carbohydrate compounds can absorb the moisture in their network structure and can change the thickness of the film. 23
Comparison of average (±SD) of thickness, WVP, humidity, and film solubility.a
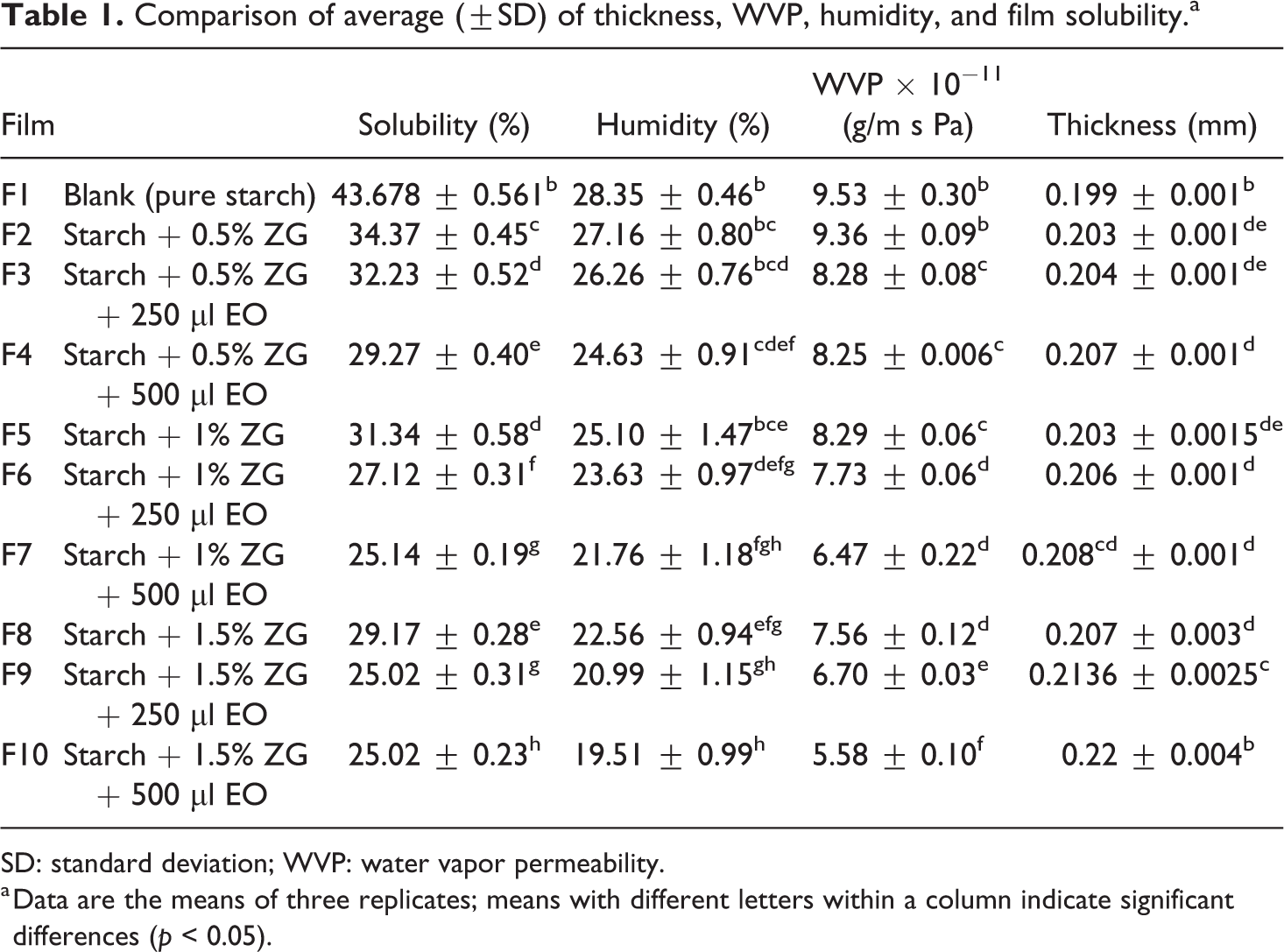
SD: standard deviation; WVP: water vapor permeability.
a Data are the means of three replicates; means with different letters within a column indicate significant differences (p < 0.05).
In terms of moisture content, by adding different amounts of gum to the control film combination, the moisture content of the films decreased. Among samples containing gum and not-containing essential oil (F2, F5, and F8 specimens), the F8 sample had a meaningful difference with two other films (p < 0.05). This means that by increasing the amount of gum from 0.5% to 1.5%, the moisture content of the films decreased significantly. Among the samples containing gum and essential oil (F3, F4, F6, F7, F9, and F10), adding different amounts of essential oil to the composition of films, the moisture content of the films decreased significantly compared to the films that did not contain essential oil. Increasing the amount of total solids is the reason for this phenomenon. In addition, fatty acids in the essential oil can cover the active group of starch and prevent hydrogen bonds between water molecules and active starch group. 24 Another reason in reducing the moisture content of films by adding gum is referring to this fact that the water molecules absorbed in the film can be linked to the gum group of alginates, so the moisture output is prevented during the drying of the films while water is present in the structure of the film. As mentioned earlier, this factor can be one of the reasons for increasing the film thickness. The essential oil combined with the film prevents high moisture absorption due to the hydrophobic nature of the essential oil, while the glycerol in the film without essential oil can absorb maximum moisture without any obstacles, which increases the thickness of the film. Films that contain essential oils create hydrophobic properties, and these films absorb less moisture. 22 Also, as the intermolecular hydrogen bond between the starch chains and the cross sections in the polymer structure are higher, the polymer matrix conjugation increases, as a result, the coherence and integrity of the film increase and its sensitivity to moisture penetration decreases. 25
Solubility in water can be one of the most important properties for edible films due to water resistance, especially in wet environments. In fact, the solubility of films in water determines the release of antioxidant and antimicrobial compounds from active films when used to cover food. Anti-water activity is an important feature of biodegradable and edible films, which indicates their effectiveness in protecting food with high water activity or fresh and frozen food. 26 Due to the hydrophilicity of starch, the starch films exhibit high water absorption and high solubility, which limit pure starch application as coatings and films. 27 Usually, gum and glycerol, due to water solubility, increase film solubility and lipids due to their insolubility-reduced film solubility. Of course, sometimes gum may reduce solubility due to interactions that can be done between the components of the film. 28 According to the results, the control film had high solubility content than other samples. By adding different amounts of gum to the composites of the film, the solubility of the films decreased significantly (p < 0.05). Also, the addition of different amounts of essential oil to the composition of films caused a significant decrease in the solubility of films compared to the control film and the films containing gum and without essential oil. It should be noted that this decrease in solubility was significantly increased with increasing essential oil content.
One of the important functions of food packaging is to prevent or minimize the transfer of moisture between the atmosphere and the food. For this reason, the lower WVP value is a desirable feature of the packaging film. 29 The WVP in films is related to the type of their application, so that any food can’t be packaged with the same polymer. Therefore, the use of many biodegradable films is limited due to high WVP. Particle size is one of the factors that affects the amount of WVP, so the smaller the size of the particles is, the lower the WVP is. 30 The results showed that the control film had the highest permeability to the vapor with a permeability level of 9.539 × 10−11 (g/m s Pa). Other films had a significant difference (p < 0.05) to each other, with the increase in the amount of gum in the film composition, the WVP of films decreased. WVP reduction has significantly increased by adding essential oil to the composition of the films. According to the results, the lowest WVP is related to F10 sample with 5.5829 × 10−11 (g/m s Pa). Glycerol loses intermolecular hydrogen bonds and increases the distance between starch molecules, and water vapor molecules pass through these molecules. Also, in the pure starch film sample, glycerol is placed between the amylose and amylopectin chains, so the distance between polymer chains increases and vapor penetrates from these pores of the film and this increases WVP. 31 In the films containing gum, the addition of gum adds a dense and loose pathway to penetrate the water molecules and reduce the film’s WVP. 32 Aguirre et al. added oregano essential oil to triticale protein and Atareset al. added ginger and cinnamon essential oils to soy protein isolates and concluded that the essential oil did not significantly change the permeability of the films. Other studies showed that the addition of essential oils to the film reduced the film WVP. 33,34 Researchers have mentioned the hydrophobic character of essential oils decreased the film’s WVP. 35,36 The results of this work for the film containing S. officinalis essential oil are consistent with the results of Pires et al. and Sanchez-Gomenez et al. 35,36 It should be mentioned that sometimes the addition of essential oil to the polymer reduces the polymerization crack, so can decrease the WVP. 33,37 Figure 1 shows the curve of average WVP, solubility in water and humidity.
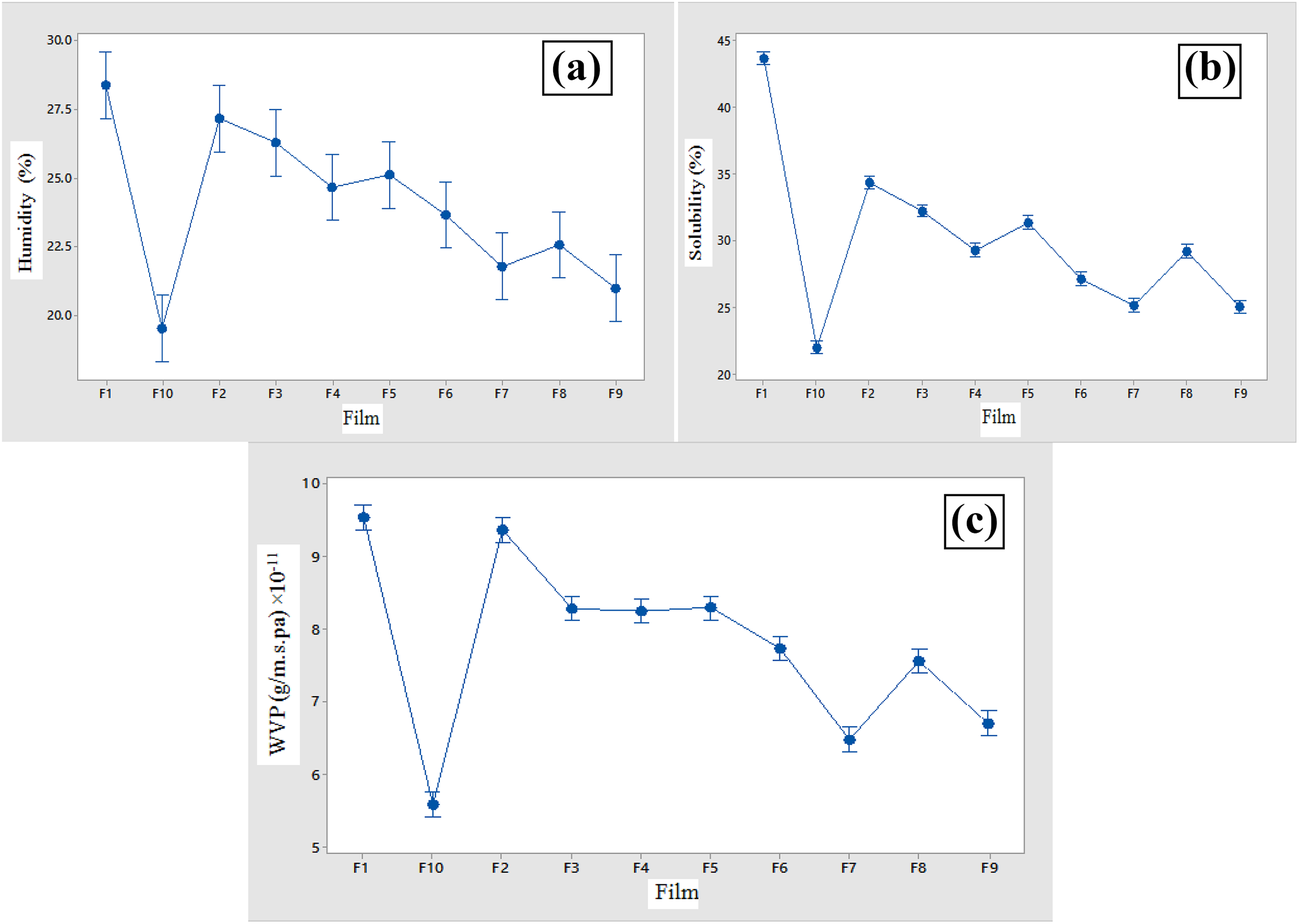
The humidity (a), solubility (b), and WVP (c) of different films containing essential oil and gum.
Film color study
The color characteristics of the packaging depend on the type of composition and the process used to make the film. Edible films are usually clear, milky, or colored. 23 The comparison of the average colorimetric parameters of the films is shown in Tables 2 and 3. The films obtained from pure starch (control sample) had a high transparency compared to other films (p < 0.05). The films containing gum and nonessential oil have high transparency in comparison to films containing gum and essential oil. This means that with the addition of different percentages of gum, the film’s degree of transparency has dropped. The transparency of the samples containing gum and essential oil was significantly different from that of the control film and nonessential oil films, which means that the film saturation rate (reduction of transparency) was increased by adding essential oil. Among the produced films, the F10 has the lowest L* value and is a film with a yellowish color. In general, by adding gum and essential oil in different amounts to films, the L* index has significantly decreased. Adding the essential oil of S. officinalis to the films usually reduces the film transparency and glassy state of the film, due to the formation of a rugged surface at the film’s surface during drying. Essential oil accumulates on the surface of the film and causes heterogeneity at the film surface. Of course, in low concentrations of the essential oil, these changes are not significant. 38 Also, the essential oil of S. officinalis is pale yellow, adding different concentrations of the essential oil to the composition of the films and the synergistic effect of the essential oil with gum significantly decrease the whiteness and transparency of the film. In terms of parameter a*, the control sample had a significant difference compared to other samples. The samples containing different amounts of essential oil (F3, F4, F9, and F10) did not differ significantly (p > 0.05) except for F6 and F7 samples that had a slight difference. In terms of parameter b*, the control film had a significant difference compared with the other samples. All the data from this parameter have positive values, which indicates that there is a yellow color in the films. The control film had the lowest yellowish (1.4833) and the F10 sample with the highest b* (4.5533) had the highest yellowish among the films. The overall color difference (ΔE) is a measure of the transparency of films. According to the results, there was no significant difference between the control film and the films containing gum extract and the essential oil.
Comparison of average (±SD) of a*, b*, and L*.a
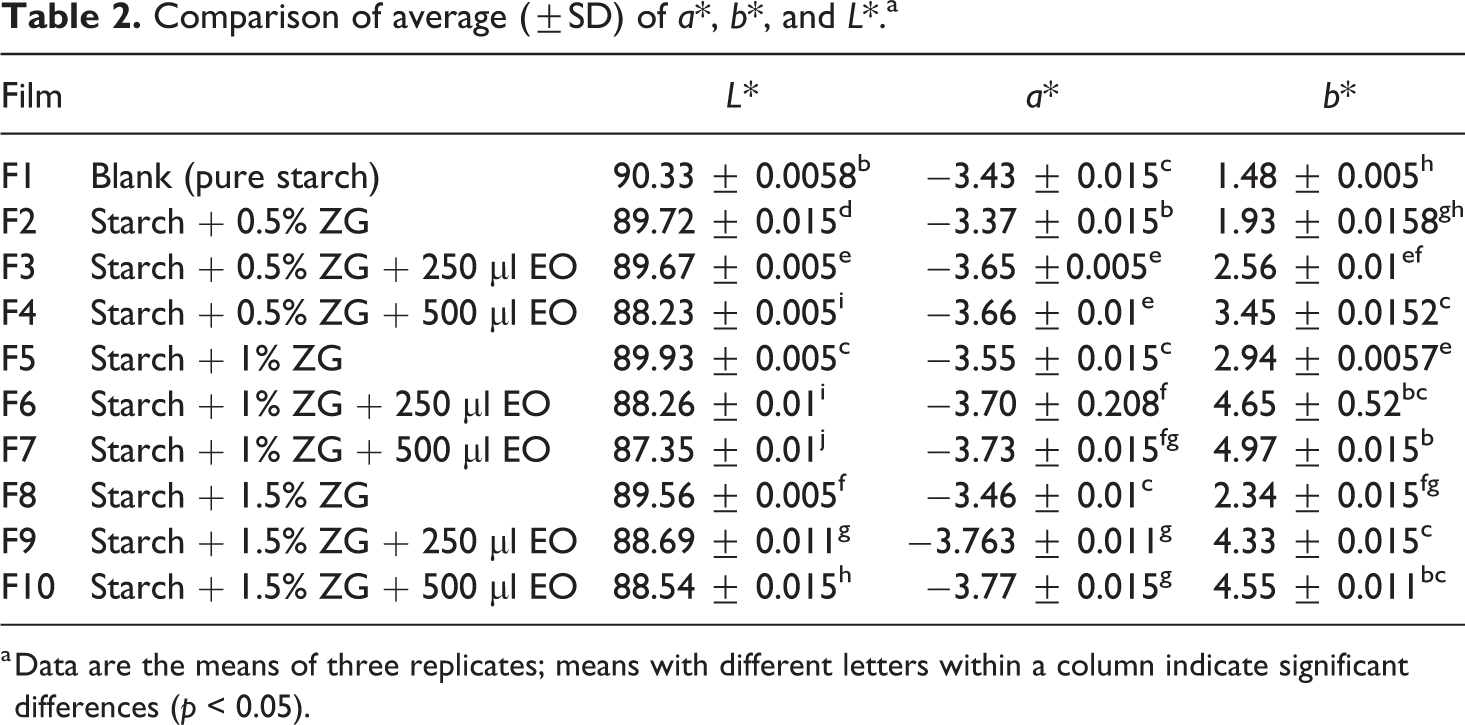
a Data are the means of three replicates; means with different letters within a column indicate significant differences (p < 0.05).
Comparison of average (±SD) of C*, ΔE, WI, and opacity.a
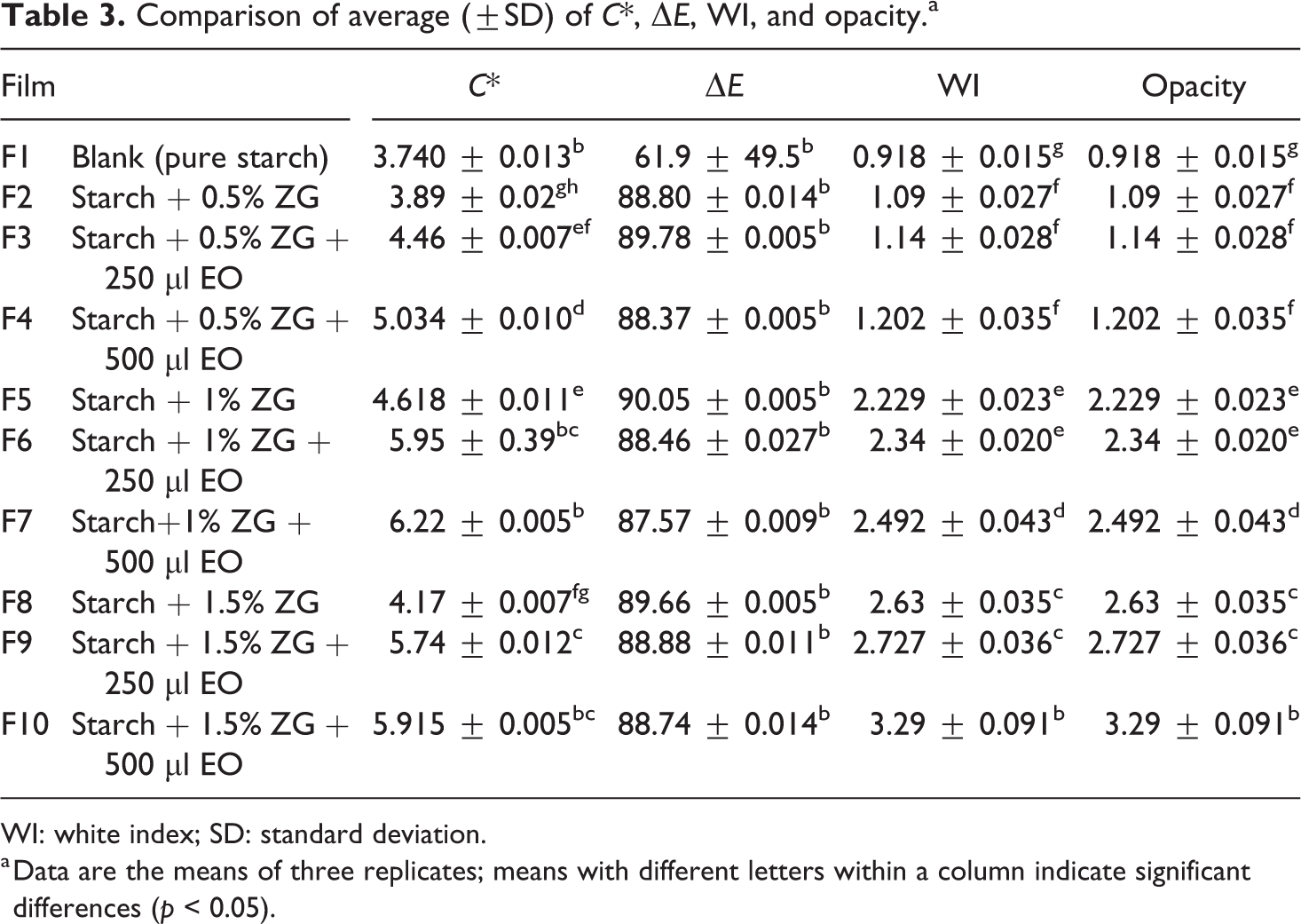
WI: white index; SD: standard deviation.
a Data are the means of three replicates; means with different letters within a column indicate significant differences (p < 0.05).
Among the samples, the F5 had the highest (90.05) and the control samples had the lowest (61.9) total color difference, compared to other samples. Chroma (C*) is a measure of the difference between a color of gray and a purity criterion. Calculating chroma in samples shows the highest degree of purity. The lowest level of purity was observed in the control sample. The levels of chroma (C*) F1 and F7 were similar and did not show significant differences. These two samples had a significant difference compared to other samples. The highest chroma was found for the F6 sample (956.5) and the lowest chroma value was for the control sample (3.4007).
In terms of whiteness index (WI), all samples were significantly different from each other. In this case, the control sample has the highest WI and the F10 sample has the lowest WI. By adding gum and essential oil to the film, WI values have decreased significantly. Atares and Chiralt also reported that the color of films generally depends on the type and concentration of essential oil. 39
Film opacity
Light irradiation is one of the most important factors in food corrosivity; some products are so sensitive to light that their color, odor, and taste are changed with the least oxidation from the effects of light. The major challenge for such products is to protect the packets from light. 40 Transparency is also an efficient and effective indicator for determining the size of the particle size, dispersed in the polymer matrix, so that particles and granules larger than the visible wavelengths block the light path and increase the turbidity of the films. 41 The average light transmission and film transparency are shown in Table 3. In examining the transparency of the films, the control sample had a significant difference compared to other samples (p < 0.05). This means that the control sample has a higher clarity than other samples (0.9186). F2 samples had no significant difference in opacity (p > 0.05). In general, with the addition of different amounts of gum to films, the turbidity of films has increased compared to the control sample. This increase in the amount of turbidity of films was significantly increased by adding the essential oil to the film composition (p < 0.05). By adding an essential oil to the film composition, the thickness of the produced films was higher than the control sample, which increased the turbidity of the films.
The effect of gum on the amount of opacity depends on the type of gum and its conditions of preparation. Some gums are dark and opaque, increasing the amount of turbidity. 42,43
Mechanical features
The mechanical properties, including the elongation and lengthening of the length to the breaking point (rupture) of the films, are presented in Table 4. The results showed that all samples had a significant difference in tensile strength except F4 and F10 compared to each other (p < 0.05). Thus, the control sample had the highest tensile strength (4.7272 MPa) and the F10 sample had the least resistance to stretching (1.6285 MPa).The plasticizers reduce the interactions between the amylose and the amylopectin chain by destroying the intra and intermediate molecular bonds of hydrogen. As a result, the gap between polymer molecules and molecular movements increases and the polymer’s flexibility increases. By reducing the interaction between the polymer chains, resistance to elasticity and elastic modulus decreases and the elasticity increases. 44 Hydrocolloids or gums are hydrophilic polysaccharides with high molecular weight and soluble in water, which, due to the nature of the polymer, high molecular weight, and interactions that occur between their chains, increase the viscosity of the film-forming solution. 45 On the other hand, hydrocolloids, due to high water absorption, increase the moisture content of starch films, and, moisture acts like plasticizers, it weakens mechanical properties, and increases the amount of stretchability. 46 Depending on the two opposing effects of hydrocolloids on the characteristics of edible films, the type and severity of the changes depend on the type of gum. Some gums such as xanthan reduce the amount of stretching and reduce stretching and elasticity. 47 Some gums such as carrageenan and pullulan also increase the stretch ability and reduce elasticity. 48 By adding various percentages of gum to the control film, the film tensile strength decreased significantly. Also, the samples containing gum without essential oil had a significant difference (p < 0.05). By increasing the amount of gum, the resistance to the stretch decreased, and the F2 sample had the highest (3.7274%) and F8 had the lowest (2.838%) resistance to stretching. According to the results, in terms of mechanical and tensile properties, the Zedo gum is very similar to xanthan gum, so by adding gum to the control films, elongation to fracture toughness and tensile strength were significantly reduced compared to the control sample. Also, films containing essential oil of S. officinalis had a significant difference compared to other films (p < 0.05). As a result, addition of essential oils to film, the film tensile strength has been significantly reduced. By increasing the amount of essential oil in the film, this decrease in the tensile strength has increased. The films containing essential oil showed a significant difference in the other film and have less resistance to stretching. In general, the presence of essential oils in the film structure causes the structural condensation and decreases the resistance to strength. According to the research carried out by various researchers, the addition of antimicrobial compounds to various polymers due to changes in the molecular level has the effect of altering the mechanical properties of films. This change is often due to film matrix degradation and reduces the film resistance to tension. 49 Ghadermarzi et al. reported that, due to the addition of S. officinalis to hydroxy propyl methyl cellulose (HPMC) film, the tensile strength and the young's modulus were decreased. The researchers argued that the essential oil concentration at high levels causes a weak reaction between the polymer and the oil and reduces the tensile strength. 23
Comparison of average (±SD) of STB and UTS.a
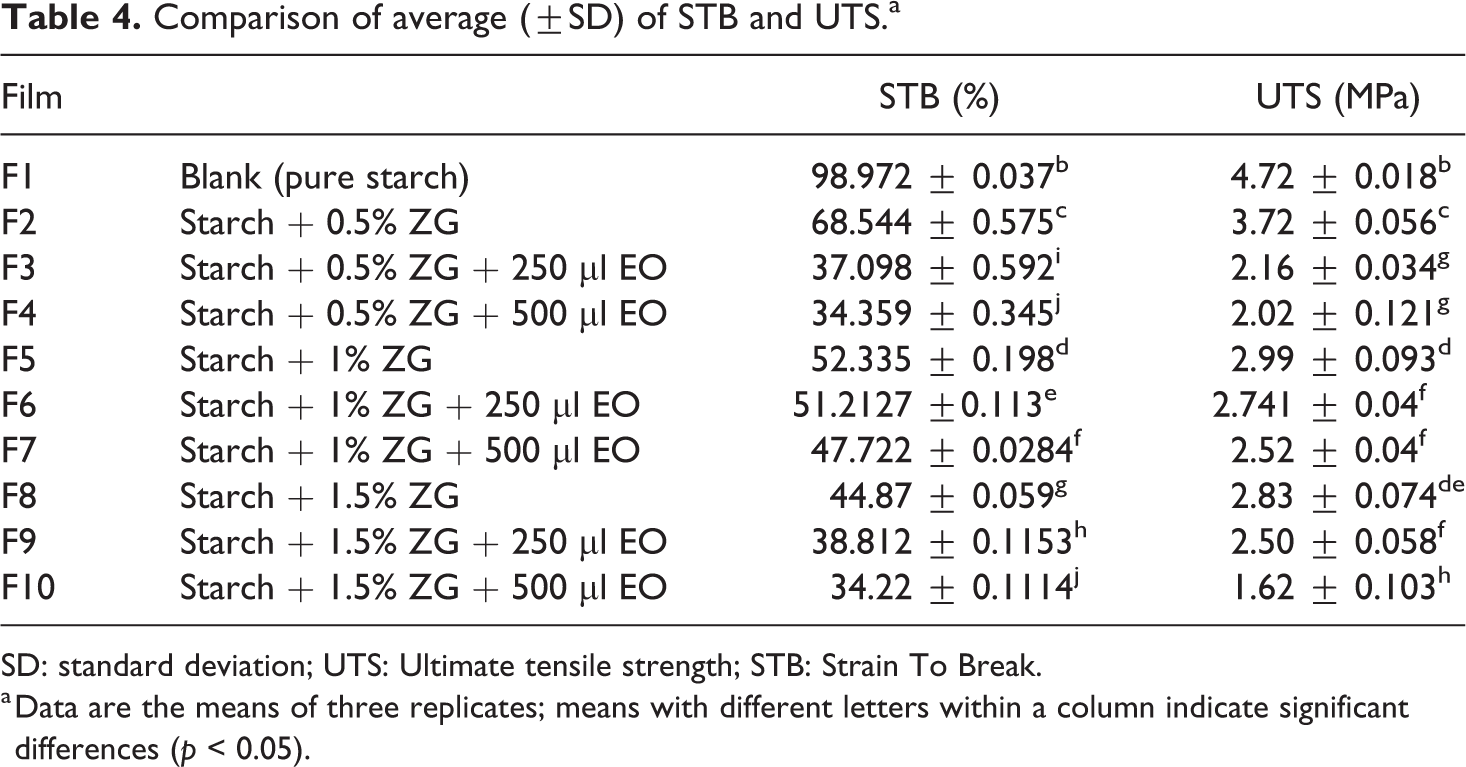
SD: standard deviation; UTS: Ultimate tensile strength; STB: Strain To Break.
a Data are the means of three replicates; means with different letters within a column indicate significant differences (p < 0.05).
Analysis of the strain to break data showed that the control sample had a significant difference (p < 0.05) with the maximum tensile strength (98.972%) compared to the other films. F2 had a significant difference (p < 0.05) in comparison with its similar samples (containing gum, no essential oil), which means that by increasing the amount of gum in the film, the STB was significantly reduced. The highest traction and elongation in this group is related to the F2 sample and the lowest is the F8 sample. Also, by adding different amounts of essential oil, the STB factor increased significantly. Figure 2 shows STB and UTS for provided films.
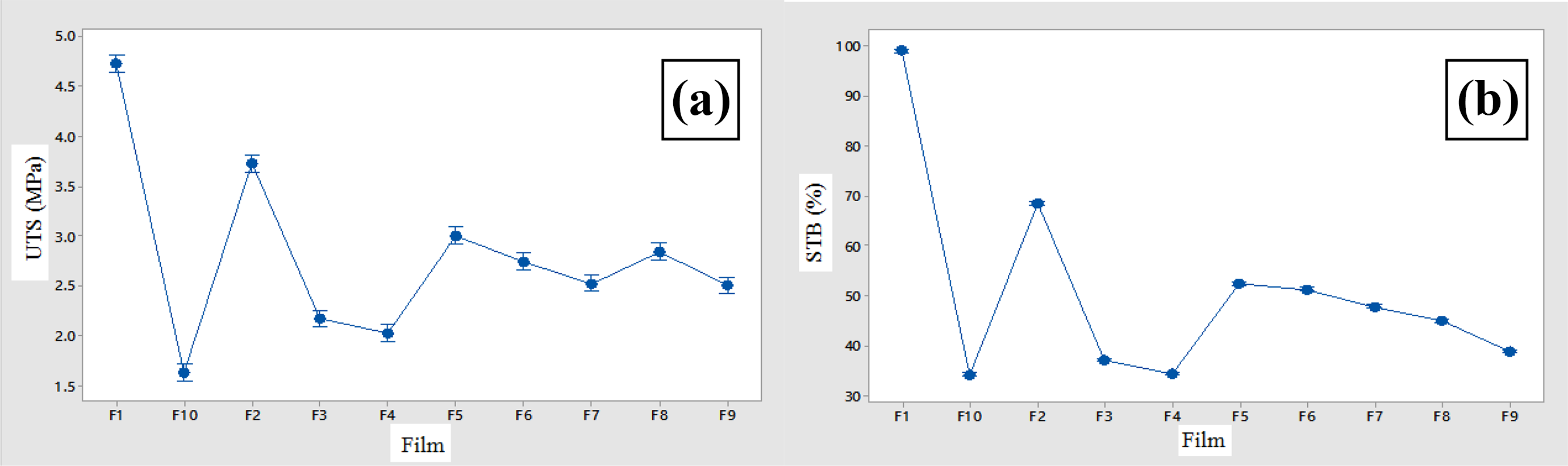
The UTS (a) and STB (b) of different films containing essential oil and gum.
Antioxidant activity
The comparison of the antioxidant capacity of the films is presented in Table 5. In essential oil of sage, there are oxygenated monoterpenes (about 45%) and oxygenated hydrocarbons (20%). The most important compounds of these groups are alpha-thujene, camphor, borneol, 1 and 8-cineole, beta-thujene, and bornyl acetate. These compounds have different antioxidant potencies in different conditions. 50 The antioxidant activity of the films is presented in Table 5 and Figure 3 shows the average antioxidant activity of the films. According to the study, the control sample antioxidant activity had a significant difference compared to other samples (p < 0.05). According to the results, by adding gum in different amounts to the control sample, the antioxidant activity of the films increased significantly compared to the control sample (p < 0.05). By adding different amounts of essential oil to film containing gum, the antioxidant activity of these films increased significantly (p < 0.05). The lowest antioxidant activity was for control film (7/31%) and the highest antioxidant value was related to the film containing 1.5% gum and 500 µl of essential oil (68.35%). In similar research, Ghadermarziet al., in studying the properties of edible film–based HPMC, containing essential oil of S. officinalis, reported that salvia essential oils showed a good ability to reduce the oxidation rate of soybean oil, so that the amount of unopened oil peroxide packed with film without essential oils The essential oil content was 92.7, 85.9, and 66.3 mEq of the oxygen was in kilograms of oil. 23
Comparison of average (±SD) of antioxidant activity.a
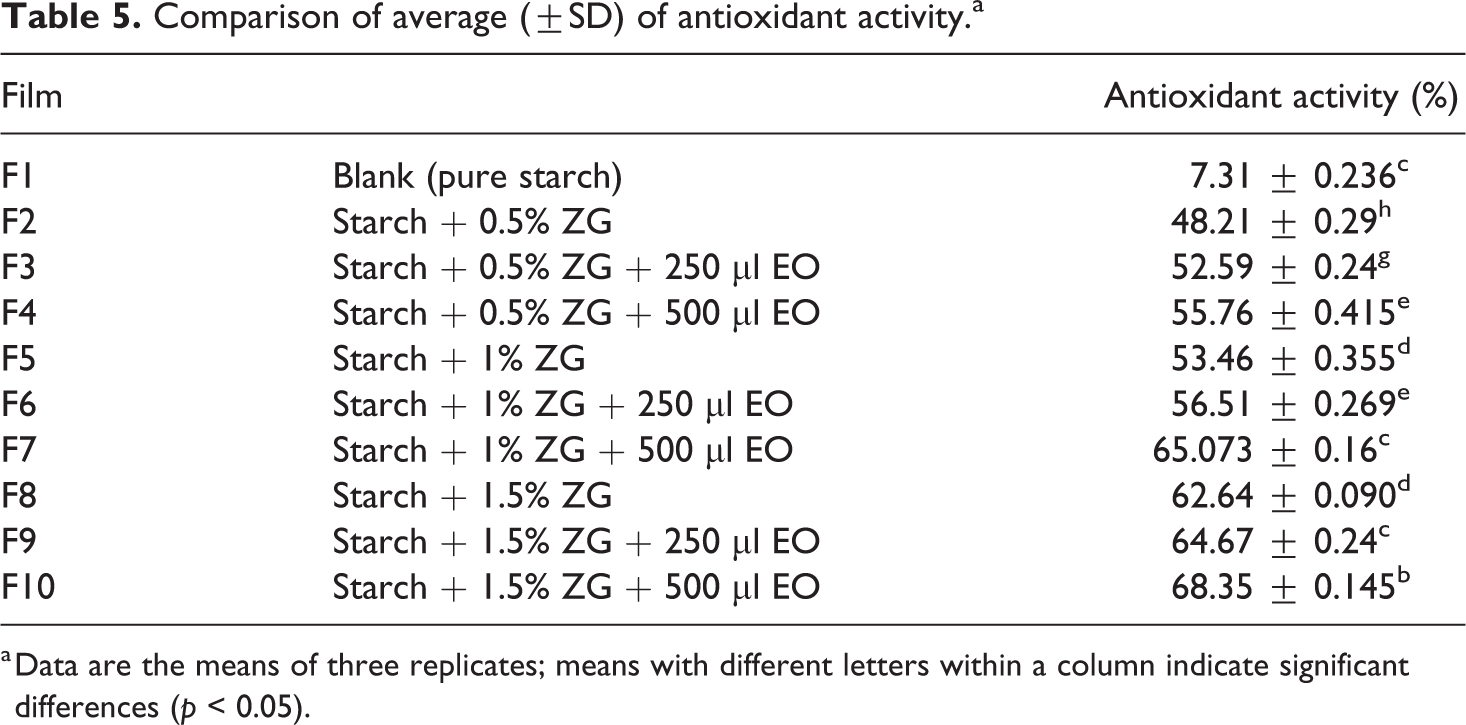
a Data are the means of three replicates; means with different letters within a column indicate significant differences (p < 0.05).
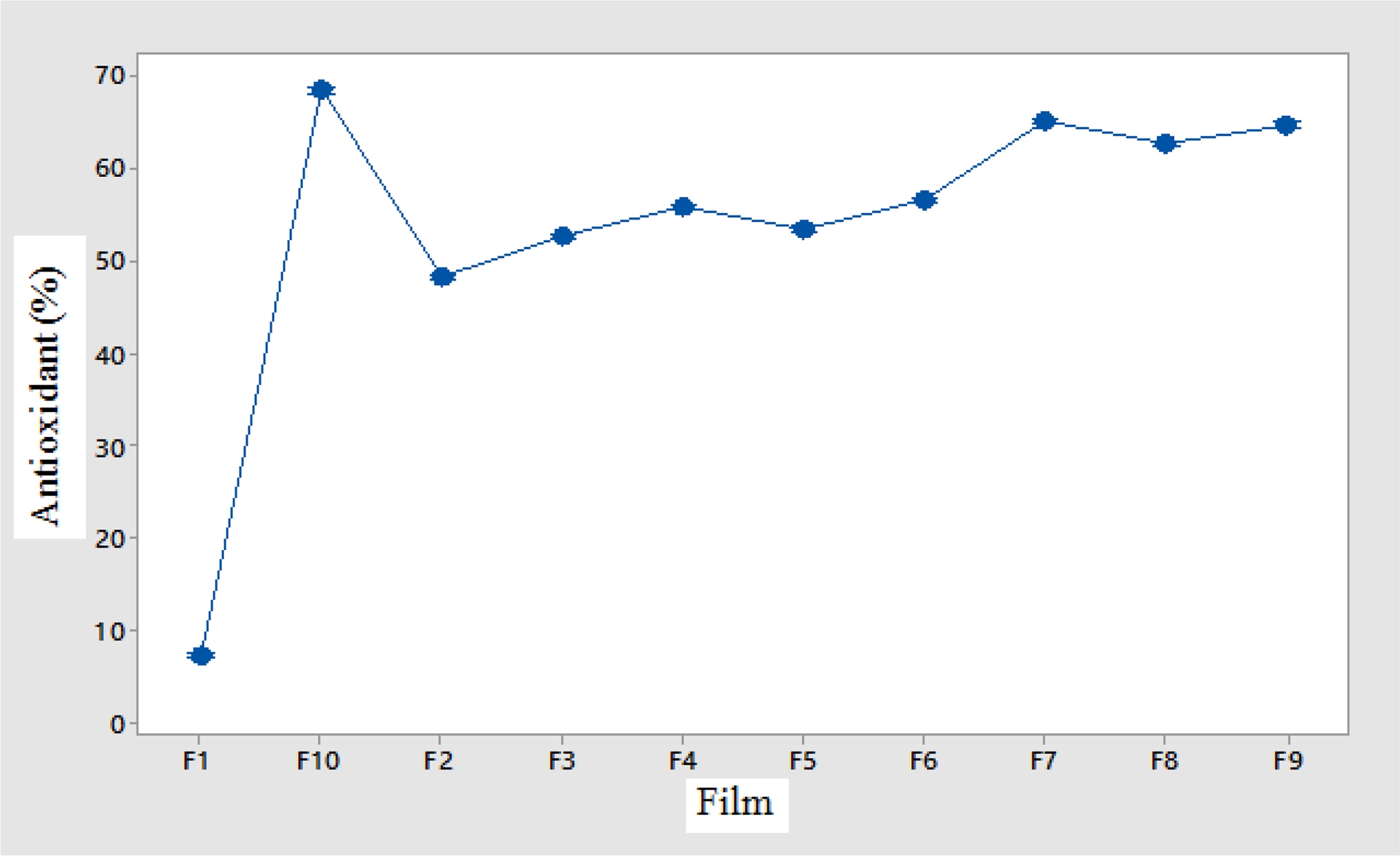
The antioxidant activity of different films containing essential oil and gum.
FTIR spectroscopy
FTIR spectra of pure starch films and starch films containing gum and essential oils are shown in Figure 4. According to the obtained spectra, the control sample or the pure film had a clear difference in the peak frequency with the other films, so that in the FTIR spectrum of the control film, the peak appearing in the wave number of 572.15 cm−1 indicates a strong bond between the C–C in the starch components. The frequency of 703 cm−1 and 742 cm−1 indicates a strong C–Br bond and represents the presence of a displaced benzene group (monobenzen) in the starch structure.
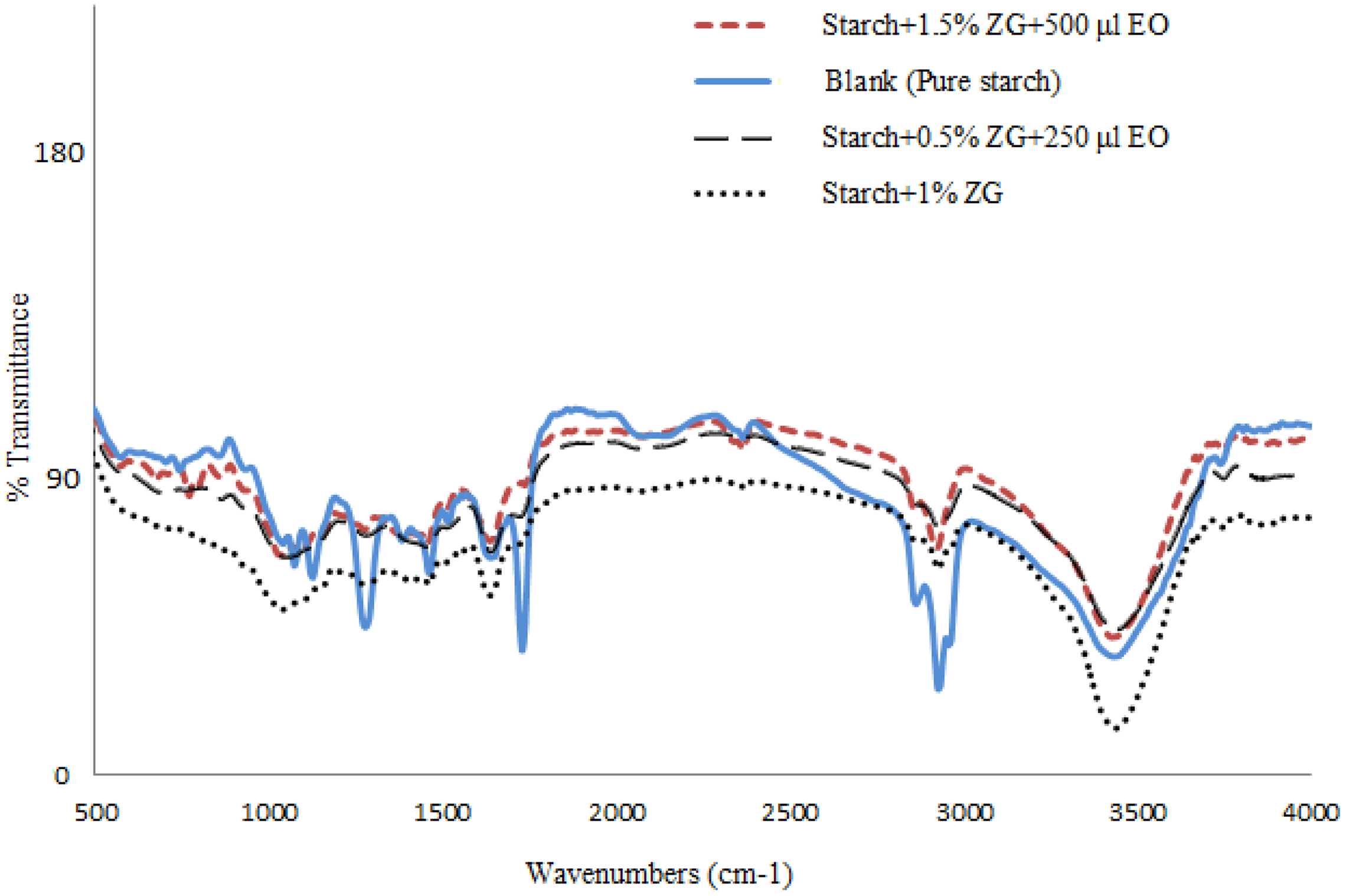
FTIR spectra of pure starch films and starch films containing gum and essential oils.
The peaks at 932 and 854 are also related to vibration of the C–O–C ring of starch. Also, the peaks of 1072 and 1124 represent the tensile vibration of C–O. The peaks at the 1382, 1557, and 1515 represent tensile vibrations of NO2. The peak at 1637 indicates a weak bond between C=C and a peak at 1728 cm−1 relative to the carbonyl group C=O. The peak at 2080 cm−1 indicates the presence of isothiocyanates, isocyanates, ketones, and allenes. The 292.94 cm−1 peak is related to the tensile vibration of the C–H bond and the presence of CH2 in the glucose ring of the starch chain. The peak at 2955 cm−1 represents a semi-strong C–C–H bond and 3431 cm−1 represents a broad and a variable band of carbonyl group. These bonds are usually strong, but the hydrogen bonding of the carbonyl group may be represented by a very poor O–H reactivity.
When two or more substances are combined, physical combinations against chemical interactions are characterized by variations in spectral peaks. 51 With the addition of gum and the essential oil to the films, the appeared peaks have some displacements, so that peaks with the following frequencies (cm−1), 572, 703, 742, and 854 in pure starch films were removed or shifted to other frequencies. The spectrum of the film containing 1% gum without essential oil was different from film containing essential oils in some wave numbers. The peaks at 500–100 were removed, the peak in the 1072 cm−1 shifted to the 1034, the peak at 1637 shifted to 1634, and the peak at 2926 shifted to 2925, which may indicate the interaction between starch and gum constituents. Also, by adding the essential oil to the composition of the films, new peaks appeared that present aromatic compounds in the essential oil in the film’s structure, so that the peaks created in the 673, 696, and 1508 cm−1 indicate the presence of aromatic compounds in the films containing essential oil. Spectrum of films containing essential oil had a clear difference in comparison to the films without essential oil, so that in film with essential oil, new peaks were created at 500–1500 cm−1, which could indicate the interaction between the essential oil compounds such as fatty acids. The main peak of pure starch is in the 3393 cm−1. This peak is related to the bond between the H and OH groups, which causes tensile vibrations in hydroxyl. 52 The peak at 1309 cm−1 is related to C–C–H, O–C–H and C–O–H stretching in the starch chains and the peaks at 1152 and 1027 cm−1 are related to the C–O–C bond in the C–O–C group. 53 The peaks of 851 cm−1 and 859 relate to the vibration of the C–O–C ring of starch. The peak at the 2927 cm−1 is related to the tensile vibration of the C–H in the glucose rings. The observed peaks in the region of 1500–1200 cm−1 are attributed to the bending vibration of the C–H groups and the O–H alcohol group bonds. 54 Han et al. studied the FTIR spectrum of starch and polyvinyl alcohol. They reported O–H stretching at 3426 cm−1 and a vibration of the C–O–C ring at 928 and 858 cm−1. 55 Noshirwaniet al. investigated active films based on carboxymethyl cellulose–chitosan–oleic acid containing ginger essential oils, which reported that by adding essential oil to the film, the peaks of the amine and hydroxyl groups are appearing in the frequencies compared to the blank film that shows the hydrogen bonding between the polymer bed and the ginger essence was established. 56 The researchers noted that the intensity of the symmetrical and asymmetrical methylene peak in CH2 and CH3 groups associated with lipids also increased after adding ginger essential oils, which indicates an increase in hydrophobicity of the films by adding ginger essential oils. 55 Also, Tongnuanchan et al. observed an increase in the intensity of these peaks in gelatin films containing ginger essential oils, which was attributed to the increase in film hydrophobicity due to the presence of hydrocarbon containing organic oils in the film structure. 57 Paluszkiewicz et al. studied the FTIR spectra of biodegradable film chitosan/nanoclay film and reported that the band appeared between 2750–3000 in the chitosan film spectrum related to the tensile band of the CH, CH2, and CH3 groups. 58 The peaks between the 3500 and 3000 belong to the stretching vibration of free hydroxyls and the symmetric and asymmetric stretching vibration of N–H in the amine group. 59
Scanning electron microscopy
The SEM test was used to observe and evaluate the morphology of films and polymers. The microscopic image (SEM) taken from the surface of the films is shown in Figure 5. As can be seen, the control sample or pure film has more bubble-like gaps and cracks than other films, which justifies high permeability characteristics of starch. By adding 1% gum to the composition of the film, the film surface is smoother and the amount of film gaps is significantly reduced, which indicates the appropriate combination of two polymers and the creation of appropriate links with each other, and the least ruggedness or roughness is observed in the film, which can be indicative of the ability to suitable combine of two starch and gum polymers. Of course, at the surface of the film, like the control sample, there are bundle bumps that can be attributed to the nature of starch and gum. Essential oils are complex and hydrophobic compounds, and it cannot be easily assumed that the addition of these hydrophobic materials to the film’s formula reduces its permeability to the vapor, but the effect that the essential oil has on the microstructure of the film determines its degree of permeability to vapor. 33 By adding different concentrations of the essential oil to the films, the film surface was smoother than the control sample and significantly reduced the amount of cracks and gaps in the film surface. One of the reasons for this is the oily structure of the essence, which, due to hydrophobicity, covers the surface of the film and closes the gaps. Obviously, by reducing the amount of cracks and slots in the film, the film’s permeability to water vapor and the degree of solubility of films are reduced due to cross-linking in their structure, and these films are preferred to pack foods that have a wet surface. This decrease in the amount of gaps is evident in the obtained images. The film containing 0.5% gum and 250 µl of essential oil is not significantly different from the control sample, but the film contains 1.5% gum and 500 µl of essential oil have a clear difference with the control film. Thus, it can be concluded that by increasing the percentage of gum, as well as high concentrations of essential oils, the film surface is smoother and the produced film is more favorable. Also, the presence of essential oil on the film surface, due to antioxidant and antimicrobial properties of essential oils, can prevent the contamination of the food in the packaged food and ensure the health of the food. Panahi et al. produced the starch film containing Pistaciaatlantica gum and reported that the film without essential oils has a uniform, coherent, and relatively smooth surface due to the continuity of the polymeric polymer network of starch film, while films containing 2% of Pistaciaatlantica essential oil have a lot of pores across the film than the control film. 60 Tabarikochaksaraye et al. examined the SEM images of the surface of the carboxymethylcellulose as control film and the composite film of carboxymethylcellulose–tragacanth and reported that the carboxymethylcellulose control film exhibited a smooth and compact surface and also composite film has a smooth surface, which shows that the two polymers are blended together uniformly. 61 Therefore, the coherent film surface may be due to the natural compatibility of two polymers. 62

SEM of the films: (a) blank; (b) 0.5% ZG, 250 µl EO; (c) 1% ZG, 0 µl EO; (d) 1.5% ZG, 500 µl EO.
Conclusion
Today, the pollution from synthetic polymers has attracted the attention of all researchers to the use of biodegradable materials, and over the past two decades, the study on the biodegradable materials derived from proteins and carbohydrates has become widespread. These macromolecules can potentially be a suitable replacement for synthetic polymers derived from petroleum derivatives. In the present study, antioxidant active films based on potato starch were studied. The effect of adding gum (0.5%, 1%, and 1.5%), and essential oil of S. officinalis (0, 250, and 500 µl) on various properties of starch film was examined. According to the results, by adding different percentages of gum and essential oil to the film, the thickness of the films increases, and this increase in thickness with the addition of the essential oil is more than with the addition of the gum. This increase in thickness affects the transparency of the films, which increases the film turbidity compared to the control film. By increasing the amount of gum and adding essential oil, the moisture content of the films decreased, which shows the advantage of using of gum in the pure starch, which makes this polymer desirable for food packaging. The high solubility and permeability was observed in the control film, where solubility and WVP were reduced by adding gum and essential oil. By adding different percentages of gum to film samples, the films antioxidant activity was increased and films exhibited antioxidant properties, indicating antioxidant compounds in gum combinations. By adding the S. officinalis essential oil to the films, the antioxidant activity of the films significantly increased, which suggests that the plant has strong antioxidant compounds. Also the evaluation of mechanical properties showed that the addition of essential oil of salvia and Zedo gum caused a significant decrease in the elongation and tensile strength of the films compared to the control sample.