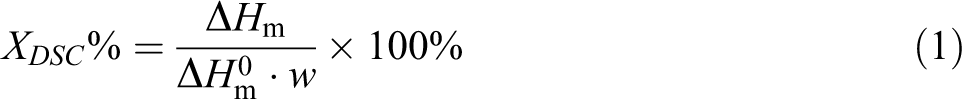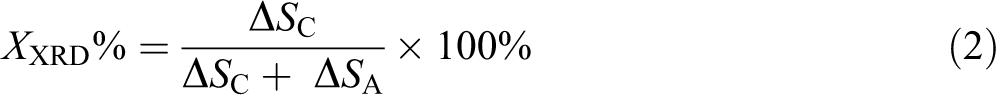Abstract
Ultrahigh molecular weight polyethylene (UHMWPE) is an excellent property polymer, but its poor processability due to high melt viscosity limits the more wide-ranging application. To improve the processability of UHMWPE composites filled by calcium carbonate (CaCO3), a novel method is reported to modify CaCO3. PE wax coated on the surface of CaCO3 (PEW@CaCO3) can be prepared by acrylic acid–modified CaCO3 (AA-CaCO3) and PE wax. The crystallization and melting behavior of UHMWPE and its composites filled by CaCO3, AA-CaCO3, and PEW@CaCO3 was compared by differential scanning calorimetry. The processability of UHMWPE composites was characterized by torque rheometer. The effect of CaCO3, AA-CaCO3, and PEW@CaCO3 on the crystallization and processability of UHMWPE was discussed.
Introduction
Compared with other thermoplastic plastics, ultrahigh molecular weight polyethylene (UHMWPE) is an outstanding performance linear crystalline polymer. Due to its excellent mechanical property, high wear resistance, low friction coefficient, outstanding chemical resistance, and self-lubrication, it is widely used in chemical engineering, medicine, metallurgy, mining, power sector, textile, and so on.
Although UHMWPE has many excellent performances which attract the attention of the researchers, some drawbacks restrict its wide-ranging application, such as its poor processability, weak creep resistant, and yield strength. To improve these drawbacks of UHMWPE, its blends with other polymers, 1 –5 composites reinforced with micro- and nanoparticles 6 –19 and all-polyolefin composites 20 –23 have been investigated.
Blending with fillers is a convenient method and suitable for industrial manufacture because of the low cost and convenient treatment of fillers. Some fillers including calcium carbonate (CaCO3), carbon nanotube (CNT), carbon fiber (CF), silicon dioxide, alumina, montmorillonite (MMT) are generally used to prepare UHMWPE composites.
For UHMWPE composites reinforced with multi-walled CNTs (MWCNTs), Enqvist et al. 6 manufactured the UHMWPE composites with 0.5 and 1.0 wt% MWCNT and found that rotational speed rather than mixing time is important for dispersing MWCNT using planetary ball milling (BM). Tensile test showed a slight decrease for the MWCNT content of 1 wt%, suggesting that this amount is the threshold for a satisfactory distribution of the filler in the matrix. Maksimkin et al. 7 observed that UHMWPE composite containing 1 wt% MWCNT showed an increase in ultimate strength four times and an increase of yield strength by 40% compared to neat UHMWPE. Tribological tests demonstrated a significant decrease in friction coefficient. Maksimkin et al. 8 and Kharitonov et al. 9 used a multistage process to prepare the MWCNT/UHMWPE composites and found that hot pressing and orientation stretching resulted in UHMWPE tensile strength increase from 21 to 132 MPa or by a factor of 6.3 as compared with pristine bulk UHMWPE. Fluorinated MWCNT was more efficient in reinforcement than pristine one. Fluorinated MWCNT did not influence composite thermal stability below 250°C and can act as crystallization centers and improve the adhesion to polymer matrix as compared with pristine MWCNT.
Khasraghi and Rezaei 10 investigated the rheological, thermal, morphological, and mechanical properties of the nanocomposite of UHMWPE/HDPE blends reinforced with 1 wt% MWCNT. MWCNT was well distributed and dispersed in HDPE matrix. The addition of HDPE can reduce the melt viscosity and improve the processability of UHMWPE. Drop in viscosity and storage modulus as well as tensile strength of nanocomposites compared to their blends was attributed to adsorption of higher molar mass polyethylene chains onto MWCNT surface. Baena and Peng 11 studied the effect of dispersion state of MWCNT on the tribological and mechanical response of UHMWPE composites. Re-agglomeration of MWCNT was evidenced in the composites filled with 1.0 wt% MWCNT. Pang et al. 12 prepared super-tough conducting CNT/UHMWPE composites. A very small amount of HDPE was used as the percolated polymer phase to load the CNT. Unique conductive networks were formed by a combination of the typical segregated and double-percolated structures, in which the fully percolated CNT/carrier polymer layers were localized at the interfaces between UHMWPE granules. Owing to the synergistic effect of the segregated and double-percolated structures, only 0.3 wt% CNT can make the composite very conductive. More interestingly, after the addition of only 2.7 wt% HDPE, the ultimate strain, tear strength, and impact strength reached 478%, 35.3 N, and 58.1 kJ/m2, respectively; these corresponded to remarkable increases of 265%, 61.9%, and 167% in these properties compared with the conventional segregated materials. These results were ascribed to the intensified interfacial adhesion between UHMWPE granules, which resulted from the strong interdiffusion and heat-sealing between the HDPE and UHMWPE molecules.
CF is also used to prepare reinforced UHMWPE composites. Chukov et al. 13,14 and Stepashkin et al. 15 investigated the effect of thermal and chemical oxidation of CF on the structural, mechanical, and tribological properties of CF/UHMWPE composites. It was found that the thermal oxidation of CF by air oxygen at 500°C can significantly enhance the interfacial interaction between the polymer matrix and CF, and Young’s modulus of composites reinforced with modified CF was significantly higher than that of composites reinforced with nonmodified CF. Interaction of matrix with a modified surface of CF results in a formation of bonds with strength higher than the yield strength of the polymer.
In order to lower the high viscosity of UHMWPE nanocomposites, Visco et al. 16 and Yousef et al. 17 prepared nanocomposites based on UHMWPE, carbon nanofiller (CNF), and paraffin oil (PO) by different mixing methods: magnetic stirring, BM, ultrasonic and minilab extruder (EX). It is found that the nanocomposites produced by EX and BM exhibit the best dispersion, good filler matrix interaction and had significantly improved mechanical properties compared to pure UHMWPE. The EX improved stiffness while the BM produced better ductility, melting temperature (T m), and the crystalline degree of the nanocomposites. The addition of 0.5–2.0 wt% CNF and 2 wt% PO can improve the wear resistance of UHMWPE using BM and extrusion techniques. The BM mixed nanocomposites with a filler load of 1.0 wt% exhibited the best wear resistance. Wood et al. 18 prepared CNF/UHMWPE composites by melt-mixing assisted by swelling with PO. The paraffin-assisted melt-mixing resulted in improved distribution and dispersion of filler at low content. However, the dispersion at higher loading was initially unsatisfactory. The addition of 1 wt% CNF resulted in further enhanced wear properties of UHMWPE. Sui et al. 19 investigated the effects of untreated and pretreated CNF on the crystallization behavior, friction behavior, and mechanical properties of UHMWPE/HDPE nanocomposites. The addition of CNF impacted the temperature of crystallization, but had no significant effect on the crystalline structure of the UHMWPE/HDPE blends. The degree of crystallinity, and the tensile strength and modulus of the UHMWPE/HDPE blends exhibited an increasing trend initially with the addition of CNF, followed by a decrease at higher contents. With the increase of untreated CNF content, the friction coefficient of UHMWPE/HDPE was decreased and displayed less change in the process of friction. The degree of crystallinity of nanocomposites with the pretreated CNF exhibited a decrease due to better interface adhesion compared to that in the nanocomposites with the same loading untreated CNFs. The enhancement in the tensile strength of nanocomposites containing 0.5 wt% treated CNF was four times higher than that of the nanocomposites containing untreated CNF.
For the inorganic particles–filled UHMWPE composites, Zhang et al. 24 investigated the interfacial adhesion and mechanical properties of silicon dioxide (SiO2)/UHMWPE nanocomposite fibers. The nano-SiO2 can be trapped on the surface of fiber to form rough surface for SiO2/UHMWPE fibers and diffused into the inner of fiber to induce the lower crystal size and higher crystallinity of UHMWPE. The resulting SiO2/UHMWPE fibers exhibit a dramatic enhancement in the adhesive properties. The mechanical properties of SiO2/UHMWPE nanocomposite fibers are enhanced simultaneously because of the influence of nano-SiO2 on the structure of UHMWPE crystalline region and fibril. Liu et al. 25 studied the rheological behavior of a novel antibacterial UHMWPE/chlorhexidine acetate–montmorillonite (CA-MMT) composite. It is found that MMT increased the viscosity of UHMWPE and CA can act as a plasticizer in the composites to bring down the T m of UHMWPE and the complex viscosity of composites. Compared with CA/UHMWPE, CA-MMT/UHMWPE had lower complex viscosity, storage modulus, and loss modulus. UHMWPE-based composites filled with aluminum oxide, tungsten, boron carbide, zinc oxide, and bronze powder have also been reported. 26
Although CaCO3 particles are widely used in LDPE and LDPE composites, 27 –34 the CaCO3-filled UHMWPE composites are little reported. Han et al. 35 investigated the processability of UHMWPE filled with different size distribution CaCO3 particles modified by stearic acid. It is observed that UHMWPE filled with different size distribution CaCO3 particles showed the largest decrease in torque compared with the composites filled with single size CaCO3 particles. The combination of different size distribution CaCO3 particles significantly increased the flow activation energy and flow activation entropy of UHMWPE. Suwanprateeb 36 prepared the CaCO3/UHMWPE/HDPE composites and found that increasing CaCO3 content increased tensile modulus, but decreased tensile strength, strain at break, and impact resistance. The addition of UHMWPE can increase the strain at break and impact resistance of composites moderately without decreasing modulus or strength. However, the effect of modified CaCO3 particles on the processability and crystallization behavior of UHMWPE is little reported.
In this article, a novel modified CaCO3 as filler was prepared. CaCO3 is firstly treated by acrylic acid (AA), then AA-modified CaCO3 (AA-CaCO3) formed by chemical reaction between AA and CaCO3 is melt-mixed with PE wax under initiator to obtain the long chain modified calcium carbonate (PEW@CaCO3), and the effect of PEW@CaCO3 on the crystallization and processability of UHMWPE has been investigated.
Experiment
Materials
UHMWPE (M w 2.2 × 106) was purchased from Mitsui Chemicals, Japan. PE wax (PEW; Product no. 1020, M w about 2000–5000, and melt point at 116°C) was provided by SCG, Thailand. Micro-CaCO3 was provided by Keynes nanomaterial (Lianzhou, China). Benzoyl peroxide (BPO) and AA were purchased from Damao Chemical (Tianjin, China).
Sample preparation
CaCO3 of 500 g was added in 2.5 L ethyl alcohol at room temperature, then 40 g AA was added and stirred 20 min by mechanical stirring so that CaCO3 reacted with AA thoroughly. After reaction, solvent was volatilized at room temperature, and the product of AA-CaCO3 was dried at 80°C for 1 h.
The AA-CaCO3, PEW (mass ratio 8:2), and 0.5 wt.% BPO (relative to CaCO3) as initiator were mixed in a mixer chamber at 150°C for 10 min. Modified CaCO3 coated by the long chain PE wax (PEW@CaCO3) was prepared due to graft reaction between PEW and AA. Composites of UHMWPE filled by different contents of PEW@CaCO3 were prepared in the mixer chamber at 190°C for 10 min.
Sample characterization
The melting and crystallization behavior of UHMWPE composites was measured by differential scanning calorimetry (DSC) at nitrogen atmosphere with the DSC-8500 (PE, USA). About 2–3 mg sample sealed in aluminum crucible was firstly heated up to 180°C at the rate of 180°C/min and kept this temperature for 3 min so that the thermal history of samples was eliminated and the sample was melt thoroughly. After that, the sample was cooled at the rate of 10°C/min from 180°C to 60°C in order to obtain the crystallization curves. At last, the sample was reheated up to 180°C at the rate of 10°C/min to obtain the melting curves. The crystallinity of samples (X DSC) is calculated by the following equation
where
X-ray diffraction (XRD) was used to investigate the crystal form of UHMWPE composites. Slice samples scanned from 5° to 40° (2θ) at the rate of 4°/min by D-MAX 2200 VPC (Rigaku, Japan) using copper Kα (λ = 0.154 nm) as radiation. The condition of treated samples was in accordance with DSC. The crystallinity of samples (X XRD) is calculated by the following equation
where
Torque testing was conducted with torque rheometer provided by PolyLab QC (Thermo Scientific, USA). Each sample of 40 g was melted at 190°C for 10 min at the rotor of 50 r/min. The torque of composites was detected through the sensor and finally the data of the variation of torque and energy with time.
Results and discussion
DSC characterization
The crystallization and melting behavior is an indispensable index for the performance of composite material. Figure 1 shows the DSC crystallization and melting curves of UHMWPE and its composites, and the data of all samples are shown in Table 1. It can be seen that the addition of PEW@CaCO3 and increasing of PEW@CaCO3 content do not change the crystallization temperatures (T
cs) of UHMWPE obviously. However, the addition of 5wt% PEW@CaCO3 can increase the T
m, crystallinity (X
DSC), and degree of cooling (
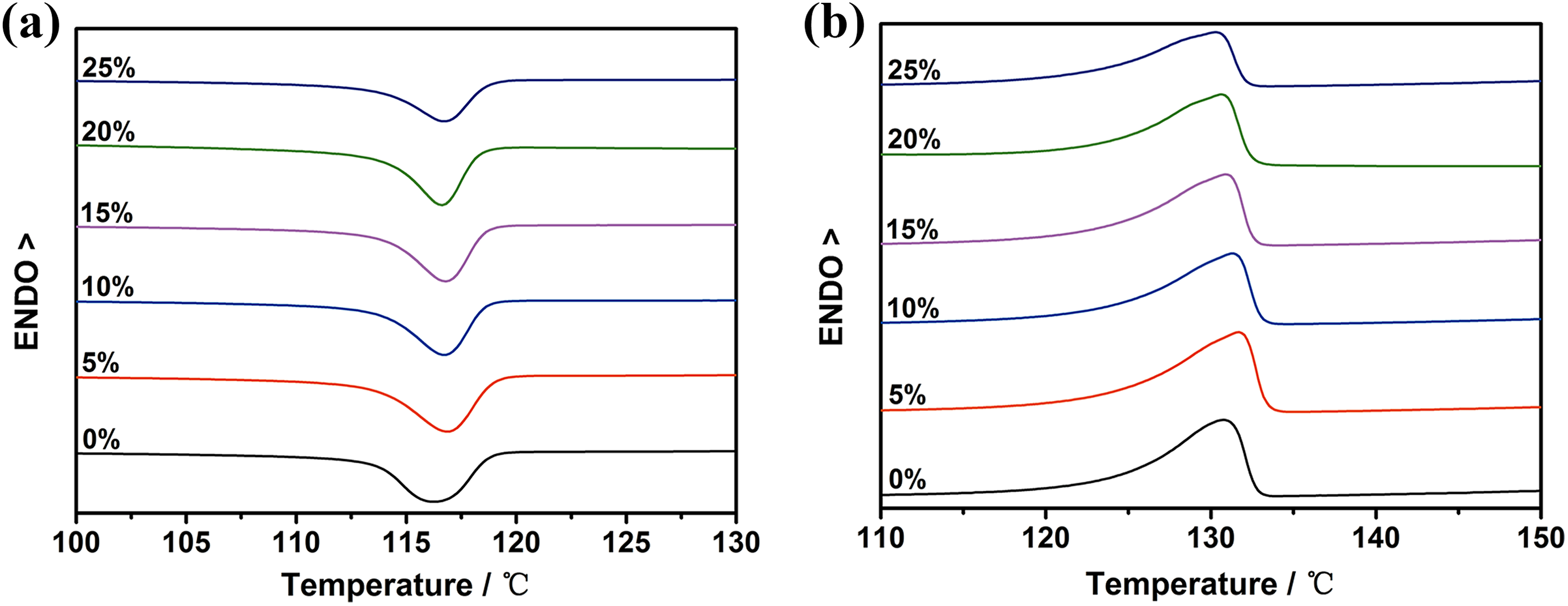
DSC crystallization and melting curves of UHMWPE composites filled by different content of filler.
DSC date of UHMWPE composites filled by different content of filler.
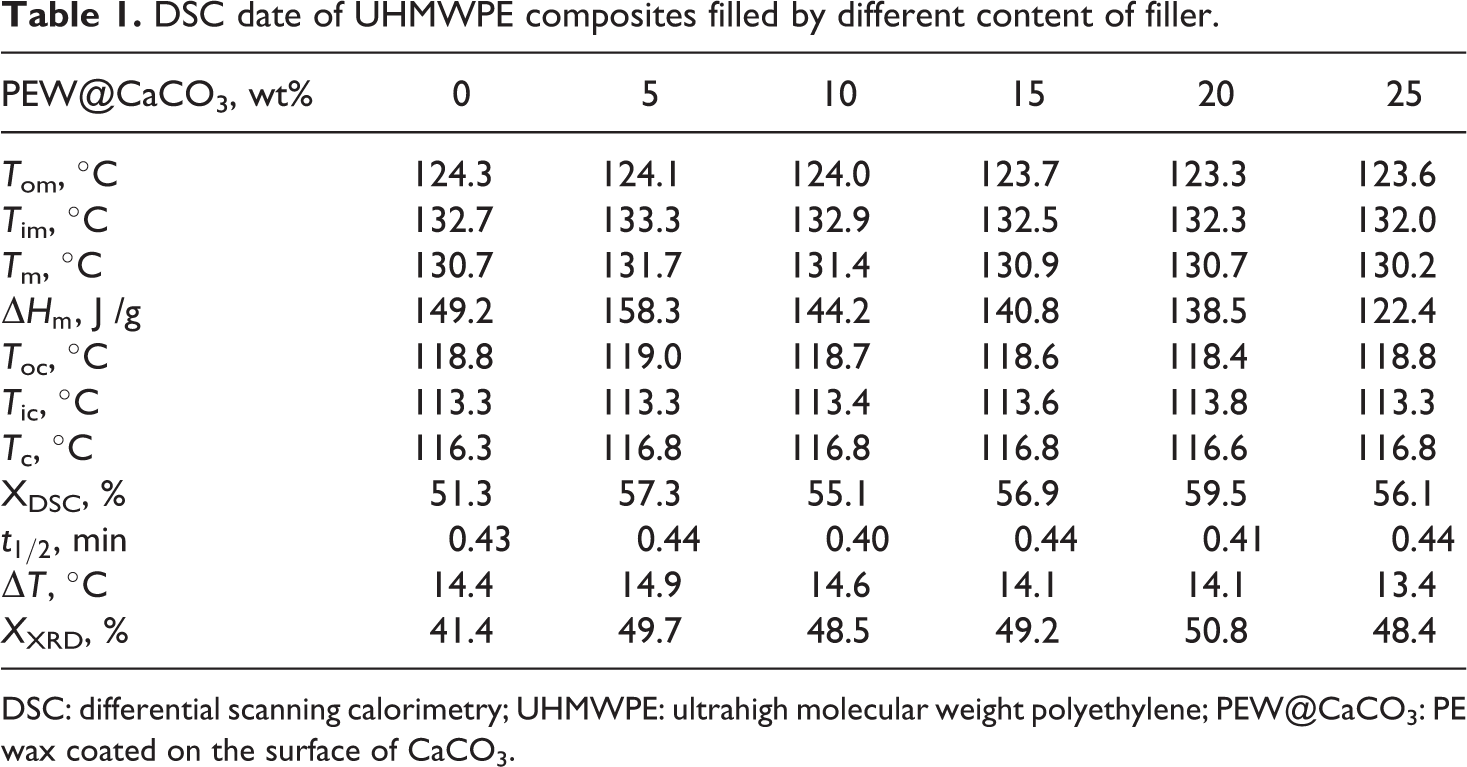
DSC: differential scanning calorimetry; UHMWPE: ultrahigh molecular weight polyethylene; PEW@CaCO3: PE wax coated on the surface of CaCO3.
Meanwhile, the crystallization and melting behavior of UHMWPE composites filled by CaCO3 modified with a different method was investigated. It can be observed from Figure 2 and Table 2 that the T c, melting point (T m), melting enthalpy (ΔH c), and crystallinity (X DSC) of UHMWPE composites filled by PEW@CaCO3, CaCO3, and AA-CaCO3 are higher than that of UHMWPE. It is indicated that PEW@CaCO3, CaCO3, and AA-CaCO3 particles show the heterogeneous nucleation for UHMWPE crystallization. Although the T cs and melting point of UHMWPE composites filled by PEW@CaCO3, CaCO3, and AA-CaCO3 have little difference, it can be observed that the order of melting enthalpy and crystallinity of UHMWPE composites is CaCO3/UHMWPE > AA-CaCO3/UHMWPE > PEW@CaCO3/UHMWPE.
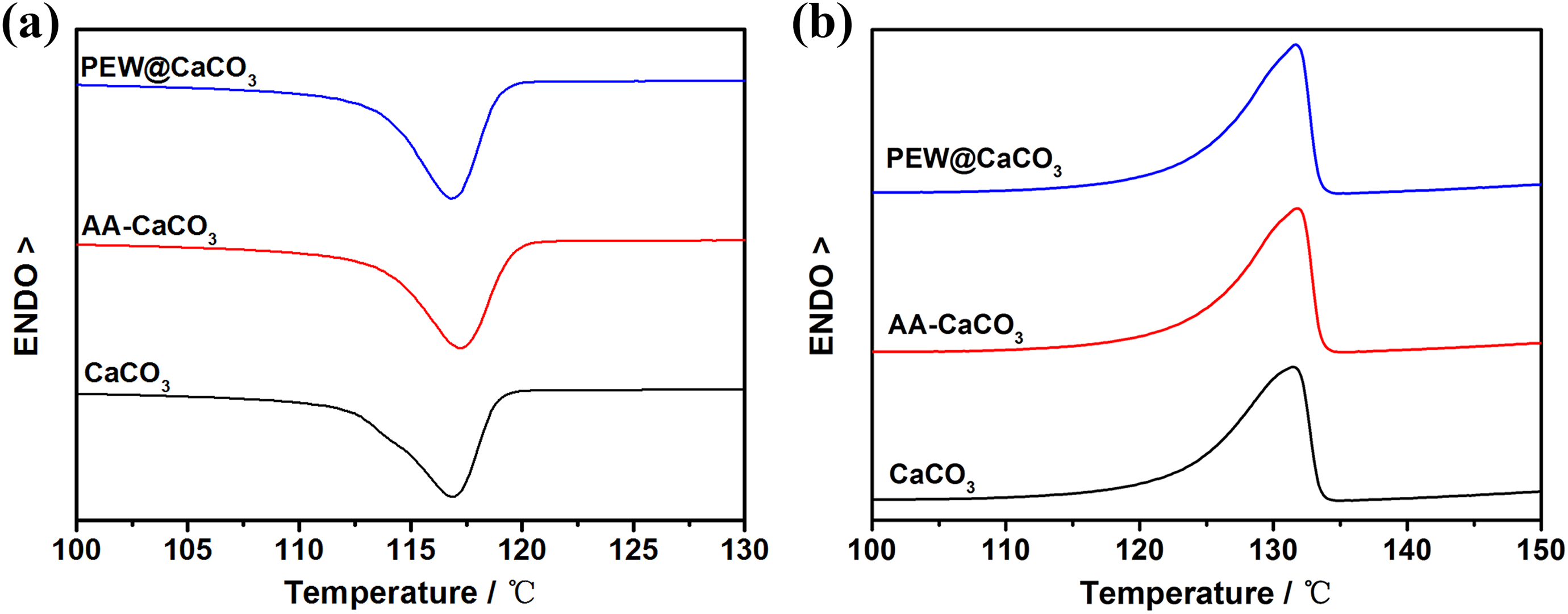
DSC crystallization and melting curves of UHMWPE composites filled by 10% different modified filler.
DSC date of UHMWPE composites filled by 10% different modified filler.
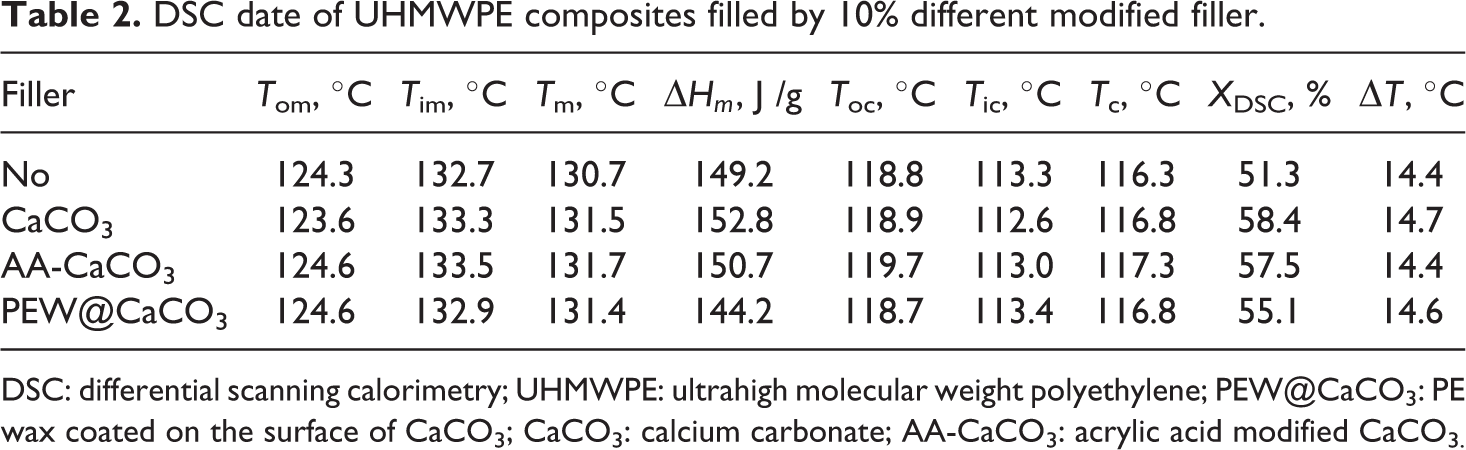
DSC: differential scanning calorimetry; UHMWPE: ultrahigh molecular weight polyethylene; PEW@CaCO3: PE wax coated on the surface of CaCO3; CaCO3: calcium carbonate; AA-CaCO3: acrylic acid modified CaCO3.
The crystallinity of PEW@CaCO3/UHMWPE is lower than the other two samples. It is suggested that the PEW coated on the CaCO3 surface can decrease the heterogeneous nucleation of CaCO3 resulting in the decreased melting enthalpy and crystallinity of UHMWPE. It is also indicated that the PEW on the surface of PEW@CaCO3 can be compatible with UHMWPE to improve the processability of UHMWPE, which is confirmed by the section “Torque characterization.”
Torque characterization
In order to study the effect of PEW@CaCO3 on the melt viscosity of UHMWPE, the torque is measured by torque rheometer. Figure 3 shows the torque curves of UHMWPE and its composites. All curves present a tendency which firstly goes up, then falls down, and finally keeps stable. The maximum torque and the stable torque of UHMWPE and its composites are shown in Table 3. The decreased torque with the increasing of PEW@CaCO3 content indicated that our prepared modified CaCO3 can decrease the melt viscosity of UHMWPE melt. It is suggested that the PEW macromolecular chain of PEW@CaCO3 can take place the interaction with UHMWPE melt due to the same chain structure between PEW -and UHMWPE. This interaction can reduce the effective entanglement density of UHMWPE so that the melt viscosity is decreased. On the other hand, the PEW macromolecular chain can also reduce the friction of UHMWPE segment so the flow of UHMWPE melt becomes easy. Increasing the PEW@CaCO3, this interaction between PEW macromolecular chain of PEW@CaCO3 and UHMWPE is more and more obvious. Therefore, the melt viscosity of UHMWPE further decreases with increasing the content of PEW@CaCO3.
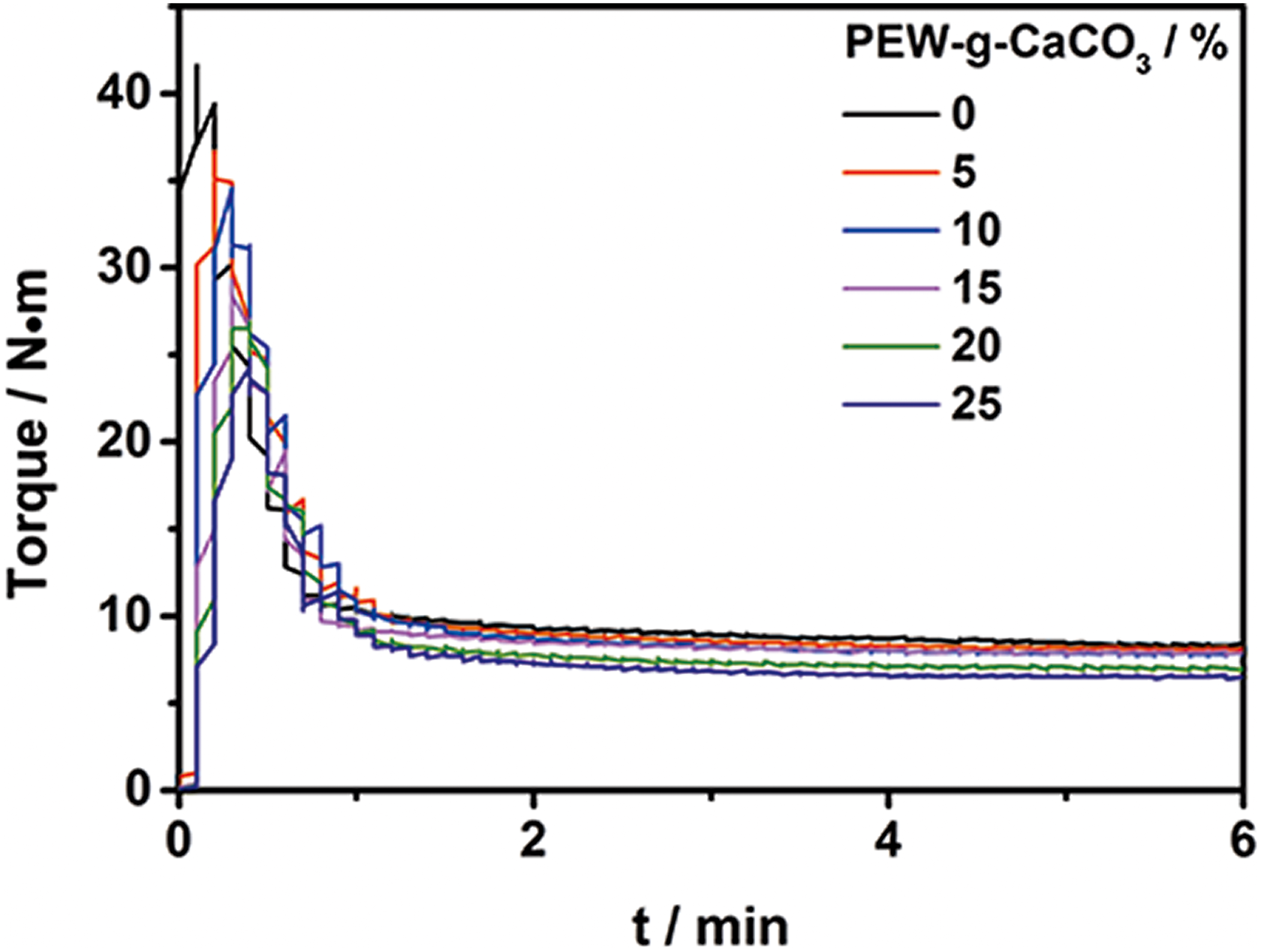
The torque curves of UHMWPE composites filled by different content of filler.
The torque of UHMWPE composites filled by different content of filler.
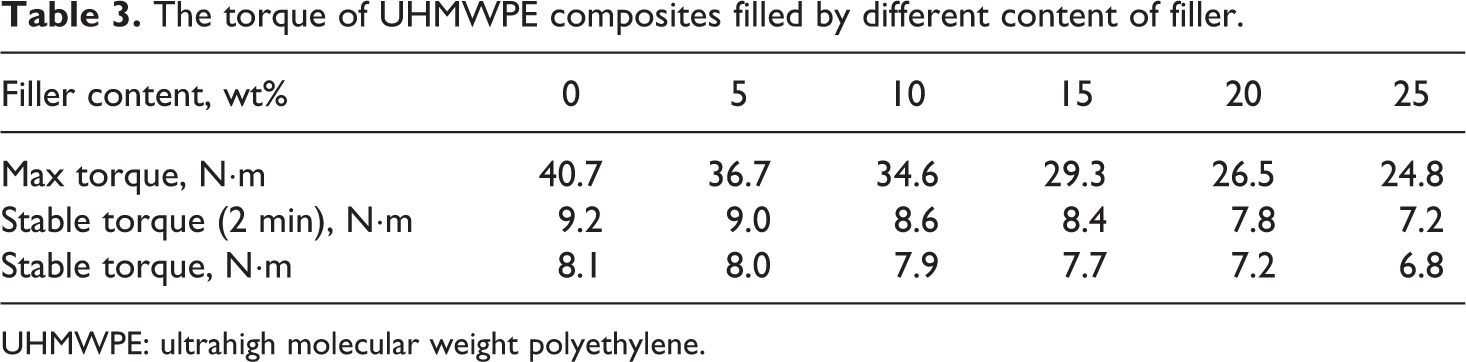
UHMWPE: ultrahigh molecular weight polyethylene.
Besides, the torque of UHMWPE composites filled by CaCO3 modified with a different method is investigated as well. It can be observed from Figure 4 and Table 4 that the CaCO3/UHMWPE composite has the highest max torque and stable torque. The torque in 2 min and stable torque of AA-CaCO3/UHMWPE composite are lower than that of CaCO3/UHMWPE composite but higher than that of the PEW@CaCO3/UHMWPE composite. It is indicated that the prepared PEW@CaCO3 can improve the processability of UHMWPE more compared to the AA-CaCO3. It is attributed to the higher compatibilization between PEW macromolecular chain of PEW@CaCO3 and UHMWPE than that between AA-CaCO3 and UHMWPE.
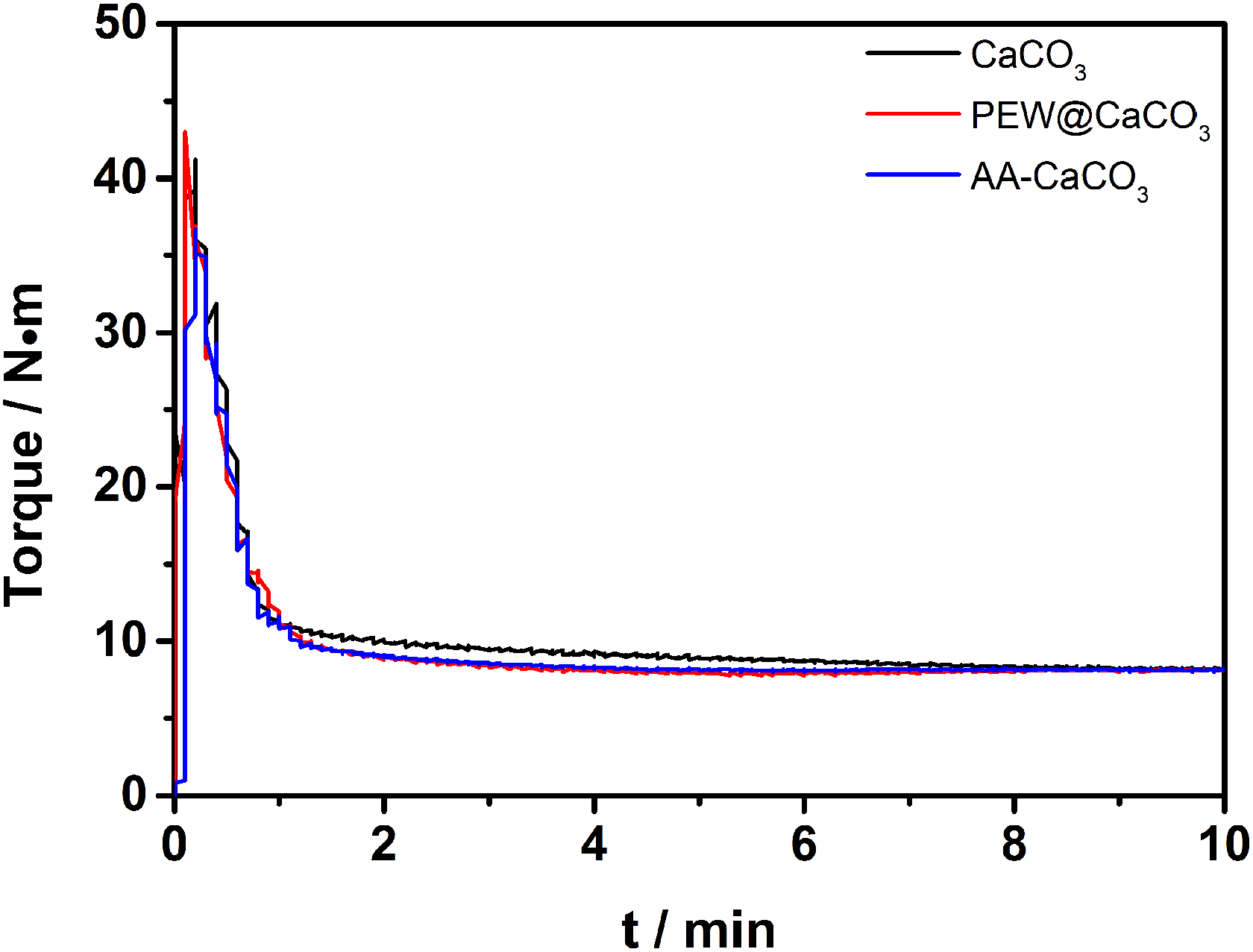
The torque curves of UHMWPE composites filled by 10 wt% different modified filler.
The torque of UHMWPE composites filled by 10 wt% different modified filler.

UHMWPE: ultrahigh molecular weight polyethylene; PE wax coated on the surface of CaCO3; CaCO3: calcium carbonate; AA-CaCO3: acrylic acid modified CaCO3.
X-ray diffraction
Figure 5 shows the XRD curves of UHMWPE and its composites. Two distinct diffraction peaks of all samples were observed at about 21.2° and 23.7°, which corresponds to (110) and (200) crystal planes of UHMWPE, respectively. The peaks of all samples have no distinct shift and no new diffraction peak appears, suggesting that the addition of PEW@CaCO3 does not change the crystal form of UHMWPE. The crystallinity (X XRD) of UHMWPE and its composites is shown in Table 1. It can also be observed that the addition of PEW@CaCO3 can increase the crystallinity of UHMWPE.
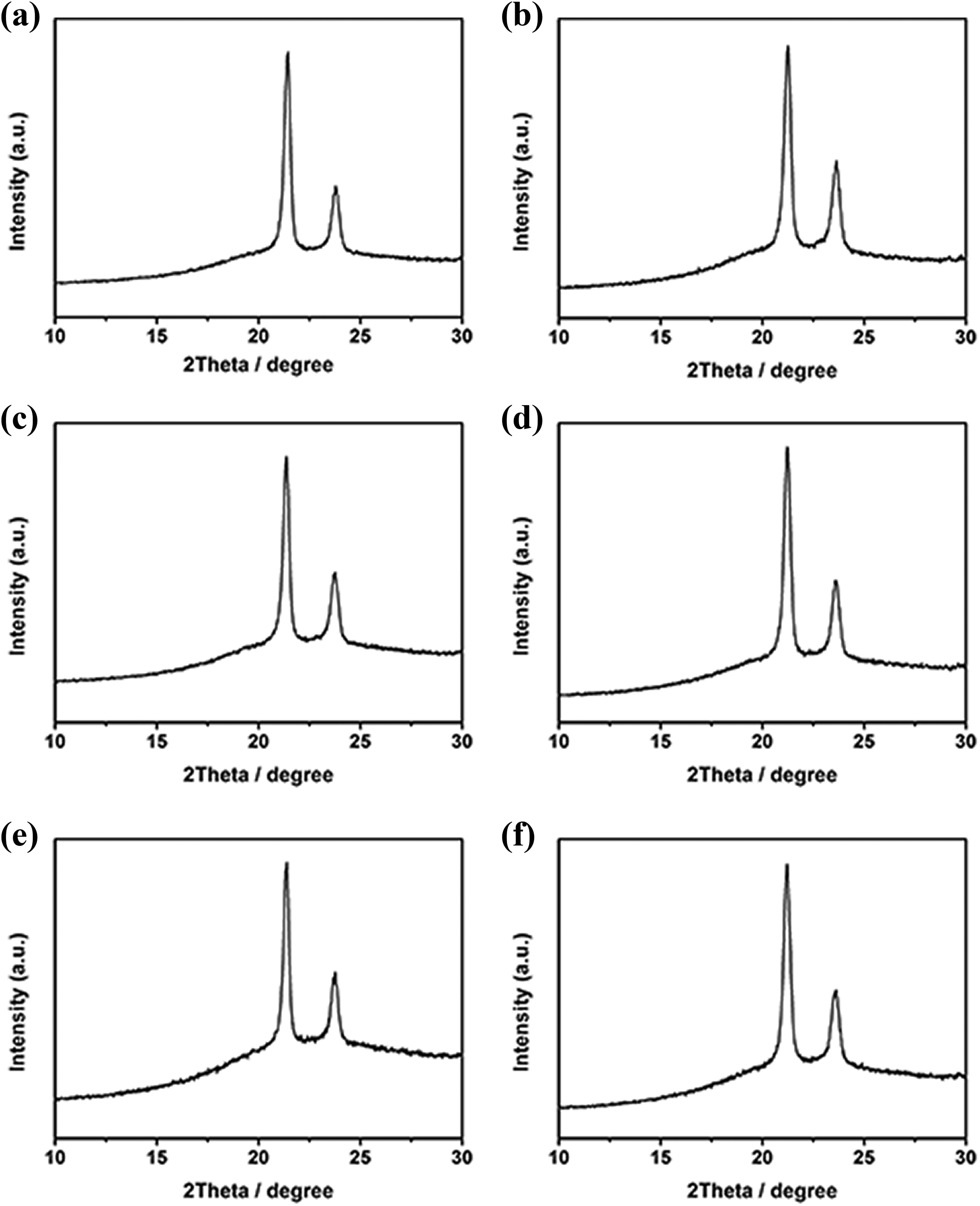
The XRD curves of UHMWPE and its composites PEW@CaCO3 content: (a) 0%, (b) 5%, (c) 10%, (d) 15%, (e) 20%, and (f) 25%.
Conclusion
In order to improve the processability of UHMWPE composites filled by CaCO3, a novel method is reported to modify CaCO3. CaCO3 is firstly treated by AA, and then AA-CaCO3 is coated by PEW. The long chain modified calcium carbonate (PEW@CaCO3) was obtained. The addition of CaCO3, AA-CaCO3, and PEW@CaCO3 increases the T cs and crystallinity of UHMWPE due to the heterogeneous nucleation of fillers. The PEW@CaCO3 improved the processability of UHMWPE more significant than that of AA-CaCO3. It is attributed to the higher compatibilization between PEW macromolecular chain of PEW@CaCO3 and UHMWPE than that between AA-CaCO3 and UHMWPE.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key Research and Development Program of China (Grant No. 2016YFB0302302) and the Natural Science Foundations of China (51573213).