Abstract
The hygroscopic nature of wood limits the use of wood thermoplastic composites (WTC) in outdoor industrial and domestic applications. To reduce this tendency, red balau saw dust was heat treated at 180 and 200°C for 1 h and compounded with Low Density Poly(ethylene) (LDPE) into 20 and 37 wt% and then molded into test specimens by injection molding. Samples were immersed in distilled water at room temperature for 4 months. Heat-treated wood composites showed remarkable water resistance relative to untreated ones. Wood composites made from wood treated at 180 and 200°C exhibited almost similar water absorption pattern. Reduced water absorption of heat-treated wood composites relative to untreated ones indicates that heat treatment has resulted in a degree of modification of the wood. Most of the composites displayed the Fickian mode of water absorption with
Keywords
Introduction
One of the shortcomings of using wood in thermoplastic composites production is the hygroscopic nature of wood. This leads to swelling and shrinkage due to moisture absorption and desorption, 1,2 resulting in poor mechanical properties of the resultant product. 3 As a result, the use of wood thermoplastic composites (WTC) in industrial and domestic applications is limited. In certain use, the environmental conditions can be so unfavorable that the performance of these composites is negatively affected. 4 Several treatment methods have been employed to checkmate these limitations and enhance better moisture resistance. 5,6 Heat treatment is one of the wood modification methods. In heat treatment, wood is subjected to higher temperatures (160–250°C) than drying. This results in the degradation of hemicellulose which has the lowest molecular weight among the wood constituents. Hemicellulose degradation results in the reduction of the OH groups and the formation of O-acetyl groups. 1 Thermal softening of cell wall matrix, mainly lignin, also sets in with cross-linking occurring between carbohydrate polymers and/or between lignin and carbohydrate polymers, resulting in an increase in the crystallinity of amorphous cellulose with consequent improvement in dimensional stability and decreased hygroscopicity of wood. Also the softened lignin flows and blocks the cell pores, thereby contributing to the reduction in moisture absorption. 7,8 Therefore, heat-treated wood is expected to become more hydrophobic with increase in treatment temperature.
It has been observed that beech wood became more water resistant after subjecting it to temperatures between 130°C and 160°C. The authors reported that the heat treatment resulted in the modification of the conformational arrangement of wood biopolymers due to the plasticization of lignin. 9 Wood panels produced by heat treatment at 180°C for 1 h has also been described as possessing lower water absorption rate after soaking for 24 h than the untreated ones. This was attributed to the degradation of hemicellulose, the most hydrophilic polymer in wood, and the chemical modification on the wood components during heat treatment. 10 Similarly, Aydemir et al. 11 compounded nylon 6 with red maple and eastern white pine treated at 212°C for 8 h. The authors reported an improvement in the mechanical, thermal, and rheological properties of the resultant composites as a result of degradation of the hemicellulose and an enhancement in the crystalline structure of cellulose. Consequently, compounding heat-treated wood with thermoplastic polymer should, in principle, decrease the water absorption tendency of the composite.
There have been several reports on the ability of filler treatment to reduce the water absorption tendencies in WTC. 12 –14 However, moisture absorption in WTC is still a major concern especially for their outdoor applications. It has been observed that the absorption of water by nonpolar polymers containing fillers depends mainly on the nature of the filler. 15 Therefore, for hydrophilic fillers such as wood, increase in water absorption should be expected.
Many polymer matrices absorb moisture reversibly by Fickian diffusion. This is found to depend on the polarity of the molecular structure (presence of chemical groups that has the ability to form hydrogen bonds with water), degree of cross-linking, presence of residual monomer, or other water attracting species and degree of crystallinity of the polymer (higher crystallinity leads to reduced water absorption). The presence of adventitious impurities is also a factor to reckon with. Consequently, non-Fickian processes may occur that may not necessarily lead to reversible effects. More so, the presence of aggregates of a second phase, such as natural filler, may complicate this phenomenon. Such been the case, the presence of a filler may modify the response of the resin in the presence of water, particularly, when the two components each has appreciably different responses. 16
Apart from their susceptibility to moisture, another very important concern in the use of natural filler as reinforcement in WTC is the negative effects moisture has on physical and mechanical properties. Reports have shown that the mechanical properties of WTC decreased with water absorption. 5,15 Therefore, it is necessary that this problem is tackled so that natural fillers can be a viable reinforcement material in WTC.
To the authors’ knowledge, research on the effects of heat treatment of wood on the water absorption behavior of WTC is rather limited. An assessment of the water absorption behavior of WTC made from heat-treated wood flour and its effects on the mechanical properties will enable the determination of suitable applications. Therefore, this article is aimed at modifying red balau saw dust with heat treatment, compounding with Low Density Poly(ethylene) (LDPE) and studying the moisture diffusion processes using equations from microscopic mass balances for diffusion in solids to predict absorbed moisture. Tensile and flexural properties were also evaluated to determine the effects of heat treatment on the mechanical properties of water-soaked WTC.
Experimental
Materials
Red balau (
Fourier transform infrared analysis
Fourier transform infrared (FTIR) spectra of heat-treated and untreated wood flour were recorded using the FTIR spectrophotometer (Spotlight 400, Perkin Elmer, Waltham, MA, USA) combined with a universal Attenuated Total Reflectance (ATR) accessory at a resolution of 4 cm−1 for 64 scans in the range of 650–4000 cm−1.
Processing
Wood pretreatment
Untreated wood saw dust was dried in an oven at 60°C for 48 h to a moisture content of less than 2% and stored in sealed plastic bags over silica gel in desiccators for not more than 24 h prior to compounding. Wood flour was subjected to 180 and 200°C in a vacuum oven for 1-h effective treatment time. The weight loss after heat treatment was 1.6 and 4.0% for 180 and 200°C treated wood flour, respectively.
Compounding
LDPE and wood flour were pre-mixed in different compositions in 200 g portions and compounded in a twin screw co-rotating extruder (Brabender KETSE 20/40 Lab Compounder, Germany) with screw diameter and L/D ratio of 20 mm and 40, respectively. The barrel temperatures were set between 150°C and 155°C along the barrel zones and screw speed was 250 r min−1. The melt pressure varied between 34 and 39 bars depending on the wood content, while the die temperature was between 164°C and 178°C. Vacuum venting was used to ventilate out the volatile compounds. The samples were extruded out through a circular die of 3 mm in diameter. The extruded strand was cooled in a water bath and pelletized to a length of about 3 mm for injection molding. Extruded pellets were oven dried at 80°C for 24 h and stored in sealed plastic bags over dried silica gel in desiccators for injection molding. Composites were prepared at two different wood flour loadings of 20 and 37% by weight as shown in Table 1.
Formulations of the composites.
Injection molding
The pellets were injection molded into tensile test pieces according to ASTM D638
16
using the BOY 55M injection molding machine at a barrel temperature range of 150–155°C, an injection pressure of 100–120 bars, and mold temperature of 25°
Water absorption
Water absorption tests were performed on samples cut from tensile test stripes according to ASTM D570.
17
Samples of 50 mm × 13 mm × 3.2 mm dimensions were cut from the central portion of the tensile test specimens and the edges smoothened with fine sand paper then dried in a vacuum oven at 90°
Water uptake,
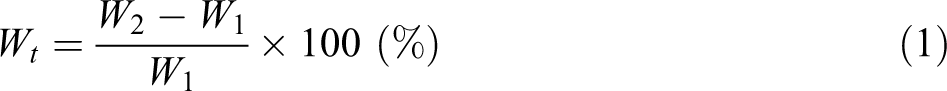
where
Kinetics of water absorption
Theoretical basis
Diffusion is the process through which matter is transferred from one part of a system to another as a result of random molecular movements. There are three different mechanisms through which moisture is absorbed into composites. The main process is the diffusion of water molecules into the microgaps between the polymer chains. Other prevalent mechanisms are movement of water molecules by capillary action into the gaps and flaws at the polymer–filler interface due to incomplete wettability and transport of water molecules by micro cracks formed during the extrusion process. 18
Generally, diffusion in glassy amorphous polymers can be classified by the relative mobility of the diffusing substance and the polymer chains. There are three categories of diffusion behavior 19 ; first (case 1), in which the rate of diffusion is much less than that of the polymer chain mobility. The equilibrium inside the polymer is readily reached and maintained with time. This is referred to as the Fickian diffusion. Second (case 2), the mobility of the diffusing substance is much higher than other relaxation processes within the polymer chain. In this case, there is the development of a boundary between the swollen outer part and the inner glassy core of the polymer. The boundary advances at a constant velocity and the core diminishes in size until an equilibrium concentration of the diffusing substance is reached in the whole polymer. Third, non-Fickian or anomalous diffusion presents an intermediate property between cases 1 and 2. It occurs when the mobility of the diffusing substance and the polymer chain relaxation are similar.
Theoretically, these three categories can be distinguished by the shape of the sorption curve represented by the following equation:

where
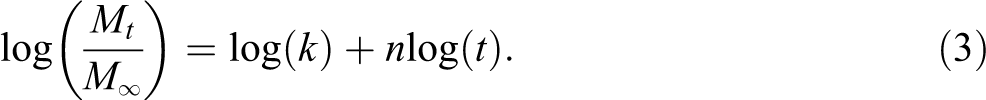
The value of the coefficient
Diffusion coefficients
Diffusion coefficients,


where
Mechanical properties
Tensile testing
Tensile tests were carried out using a universal testing machine (Instron 5569, Norwood, MA, USA) equipped with a load cell of 50 kN with a mechanical extensometer according to ASTM D638. 16 Dry and water-soaked specimens were tested at ambient conditions after equilibrium moisture content was reached at a crosshead speed of 5 mm min−1. A zero span of 50 mm was chosen for the extensometer. Values recorded are the average of at least five reproducible results.
Flexural testing
The same instrument used for tensile testing was used for the flexural testing but in three point bending mode according to ASTM D790. 21 Dry and moisture saturated tensile test specimens were tested at ambient conditions with a span of 50 mm. Samples were tested to a maximum deflection of 30 mm at a crosshead speed of 1.28 mm min−1. Reported values are the average of at least five reproducible results.
Scanning electron microscopy
The fractured surfaces of the tensile test specimens were observed using the Leica S440 (England) scanning electron microscope under an acceleration voltage of 10 kV after gold sputtering to a thickness of about 0.014 µm.
Results and discussion
FTIR analysis
The FTIR spectra of heat-treated and untreated wood flour are presented in Figure 1.
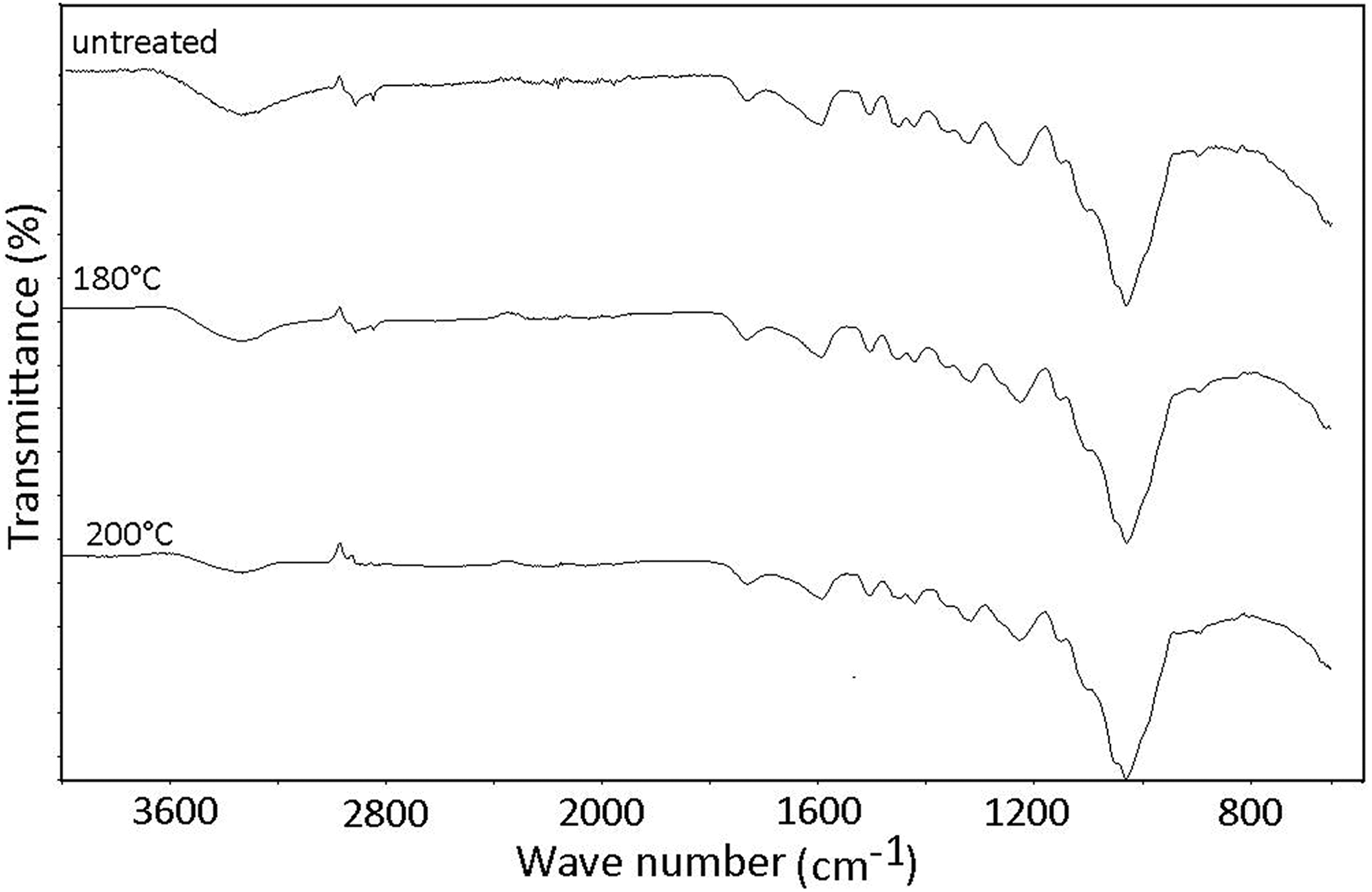
FTIR spectra of untreated and treated red balau saw dust.
The spectra look similar as all the samples exhibit characteristic signals of lignocellulosics. A broad peak of cellulose hydroxyl groups (O–H) appears at 3335 cm−1 and another peak at 1733 cm−1, which is indicative of C=O aldehyde groups of hemicelluloses and lignin. 22 Despite the similarity in the spectra, the intensity of the absorption band due to O–H stretching vibration at 3335 cm−1 is observed to decrease with treatment temperature, with the wood sample treated at 200°C showing a profound decrease. However, the absorptions due to C–H stretching vibrations (2860–2970 cm−1) remain intense. This is reported to show that there is dehydration and rearrangement of molecules leading to alkyl-carboxyl structures mostly of the ketonic and aldehydic types. 23
Water absorption behavior
A plot of moisture content against time for different treatment temperatures and wood flour loadings is presented in Figure 2.
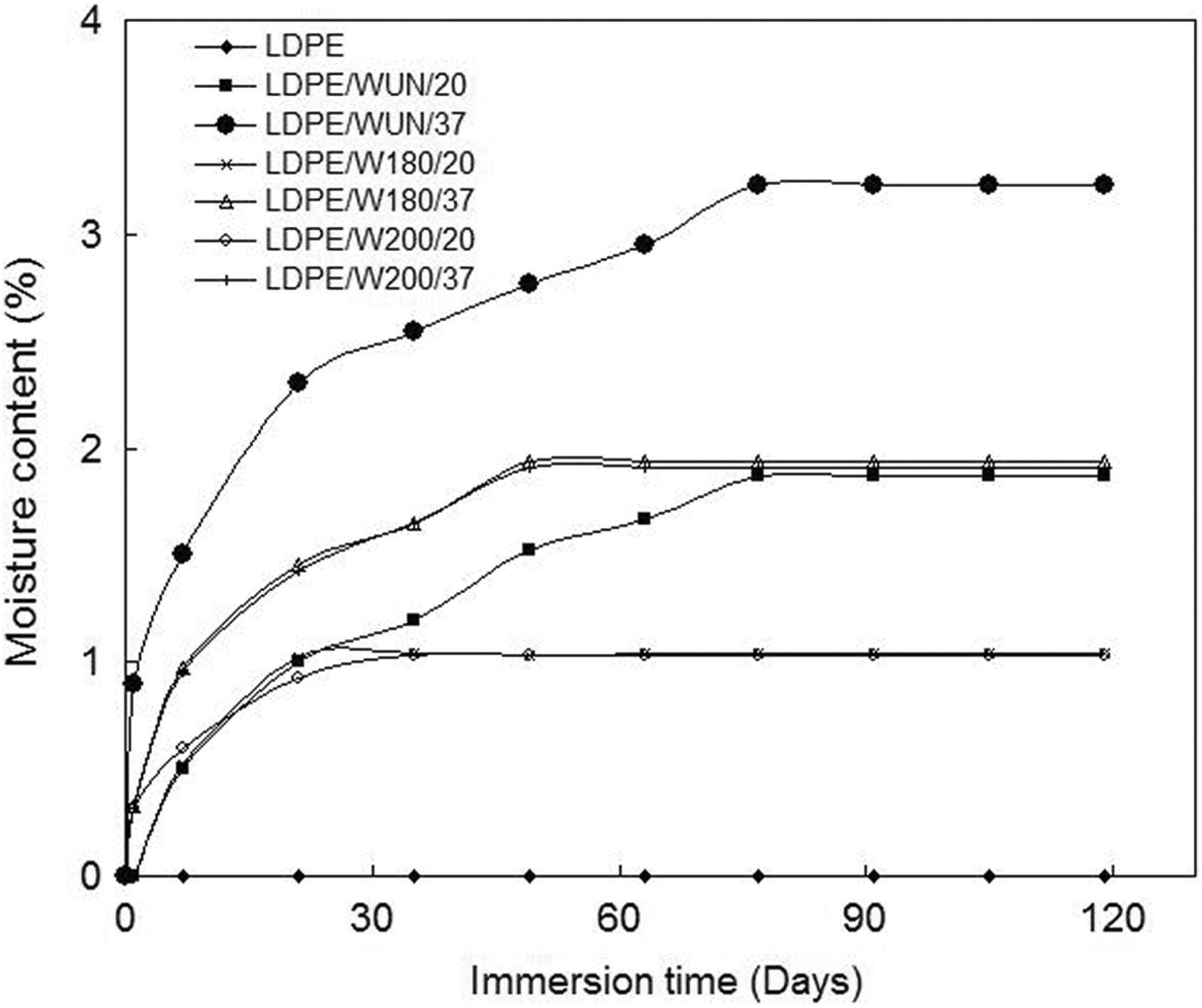
Water absorption curves of red balau/LDPE composites.
The neat LDPE shows no water absorption as no weight increase is observed over the period of study. This is because neat LDPE is hydrophobic and has no tendency of absorbing water. However, both the untreated and the treated wood composites absorbed water to various extents. As neat LDPE did not absorb water, it can be assumed that the wood flour is responsible for all the moisture absorbed by the composites. This is expected due to the hydrophilic nature of wood flour. It is sometimes believed that moisture is not an issue with WTC because wood is totally encapsulated by the matrix. If that is the case, the matrix would shield the wood from moisture because they are hydrophobic. Nevertheless, composites still exhibits a degree of moisture absorption tendency. 24,25 This seems to indicate that wood must have been exposed on the surface of the samples which may be responsible for various degrees of water absorption exhibited by the composites. Also, it can be seen that water absorption increases with time of immersion, reaching a certain value (the equilibrium point), when the water content of the composite remained constant. LDPE/W180/20 and LDPE/W200/20 reached equilibrium (1.03 and 1.04%, respectively) at 35 days, while LDPE/W180/37 and LDPE/W200/37 attained equilibrium point (1.94% and 1.91%, respectively) on the 49th day of immersion. This may be due to the presence of more O–H groups on the surface of the composites with higher filler content, leading to more ability to absorb water. 26 However, LDPE/WUN/20 and LDPE/WUN/37 exhibit a saturation point of 1.87 and 3.23%, respectively, on the 77th day of immersion. This is likely because of the higher number of free O–H groups on the surfaces of untreated wood composites which interact with water via hydrogen bonding (Figure 1). It is also possible that at 37 wt%, the wood flour is not continuously distributed and most of the wood particles may probably have contacted one another directly, in which case, complete encapsulation is not achieved (Figure 3). This could result in the higher water absorption values recorded for composites at 37 wt% wood content. Composites containing wood flour treated at 180 and 200°C exhibit similar equilibrium moisture contents. As previously reported, 27 it is possible that a 3% change in the hemicellulose of wood flour treated at 180°C relative to that treated at 200°C may not effect an appreciable difference in their water absorption behaviors.

Fractured surface of untreated wood composites after moisture saturation showing flaws and gaps.
Water absorption in WTC has been shown to not only depend on the filler particles and matrix structure but also on the filler–matrix interface. 28 Hydrophilic fillers lead to lower filler–matrix interaction resulting in poor interface, which facilitates the absorption process. This is supported by the SEM micrographs presented in Figure 3.
It has been said that moisture can be absorbed into the composite by the dissolution of water into the polymer network, moisture sorption into the free volume, if present, in the glassy structure, and by hydrogen bonding between the hydrophilic group of water and the components of the composite. Micro-cracks can also allow moisture transportation involving flow and storage of water within the cracks. 28 Untreated wood composites generally show higher water absorption than treated ones. LDPE/WUN/20 absorbed 55% more water compared to LDPE/W180/20 and LDPE/W200/20, while LDPE/WUN/37 absorbed about 40% water higher than LDPE/W180/37 and LDPE/W200/37. This could be attributed to the removal of the water sensitive components, hemicelluloses, in wood flour by heat treatment, thereby rendering the composites more water resistant. Reduction of hemicelluloses in heat-treated wood flour was reported in our previous studies. 27 In addition, research has shown that the free hydroxyl groups of the cellulose are reduced with heat treatment, leading to a decrease in the water absorption of wood 29 and consequently, the composites. The authors suggested that heat treatment depolymerizes the carbohydrates, especially the hemicelluloses. The cleaved products are leached and/or caramelized during treatment, thereby indicating a decrease in the total amount of free OH groups. Follrich et al. 30 have also reported that at 200°C, hydrophilicity of spruce wood decreased appreciably. This was observed as an important result for WTC compounding and inferred that it is an alternative way of increasing the adhesion between wood and hydrophobic thermoplastics without the use of compatibilizer. Composites containing 20 wt% wood flour absorbed water and reached saturation faster than that made from 37 wt% wood flour content, which attained saturation more gradually (Table 2). The fact that higher filler content means higher ability to absorb water may have resulted in delayed equilibrium moisture content attainment for both treated and untreated composites. Increase in water absorption of untreated wood composites relative to the treated ones is due to the hydrophilicity of wood imparted by the free O–H groups in the wood structure which has a high tendency for hydrogen bonding with water (Figure 1). It has also been reported in our previous publication that the untreated wood has higher hemicellulose content, 27 which is responsible for moisture absorption. Reports revealed that the absorption of water by non polar polymers containing fillers depends mainly on the nature and the amount of the fillers. Furthermore, the amount of accessible O–H groups that can form hydrogen bonds with water molecules is another factor responsible for the degree of water absorption by composites. The higher the amount of the OH groups, the higher is the initial rate as well as the level of water uptake, 22 explaining the observed trend.
Values of equilibrium moisture content,
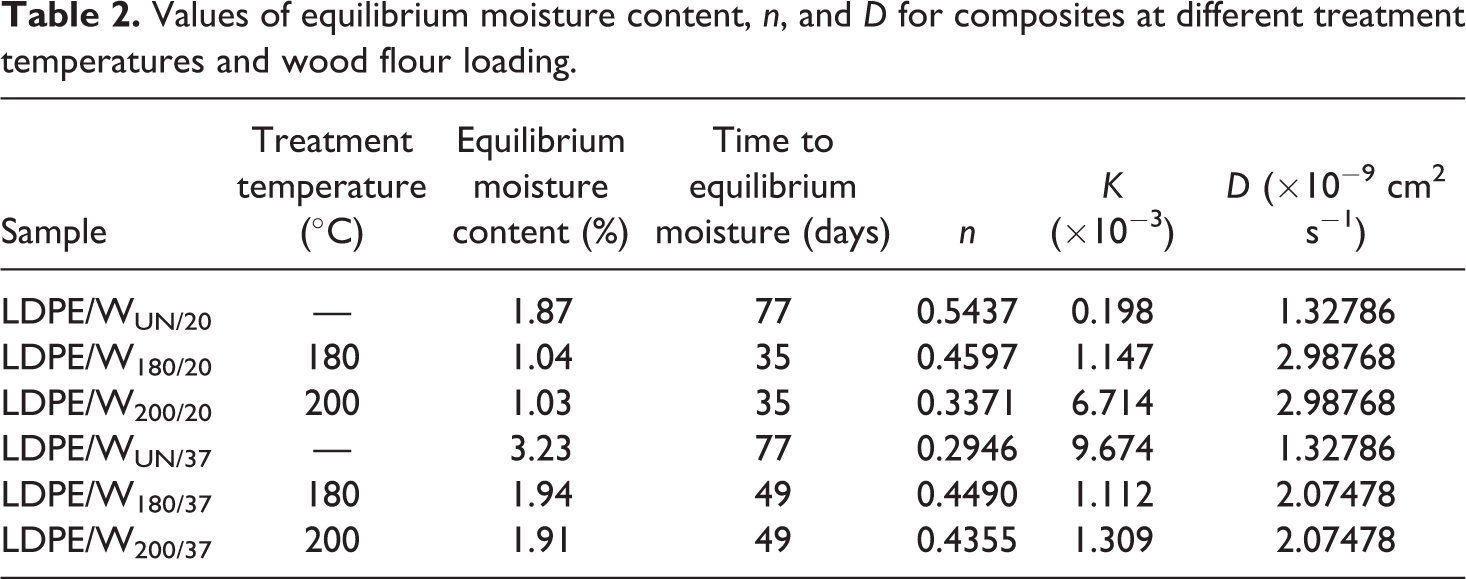
Water absorption kinetics
The values of
As stated earlier,
Effect of water absorption on mechanical properties
Tensile properties
Figures 4, 6, and 7 show the tensile properties of dry and wet specimens at equilibrium moisture content for WTC immersed in water. Tensile modulus of the composites was generally found to decrease with water immersion (Figure 4).
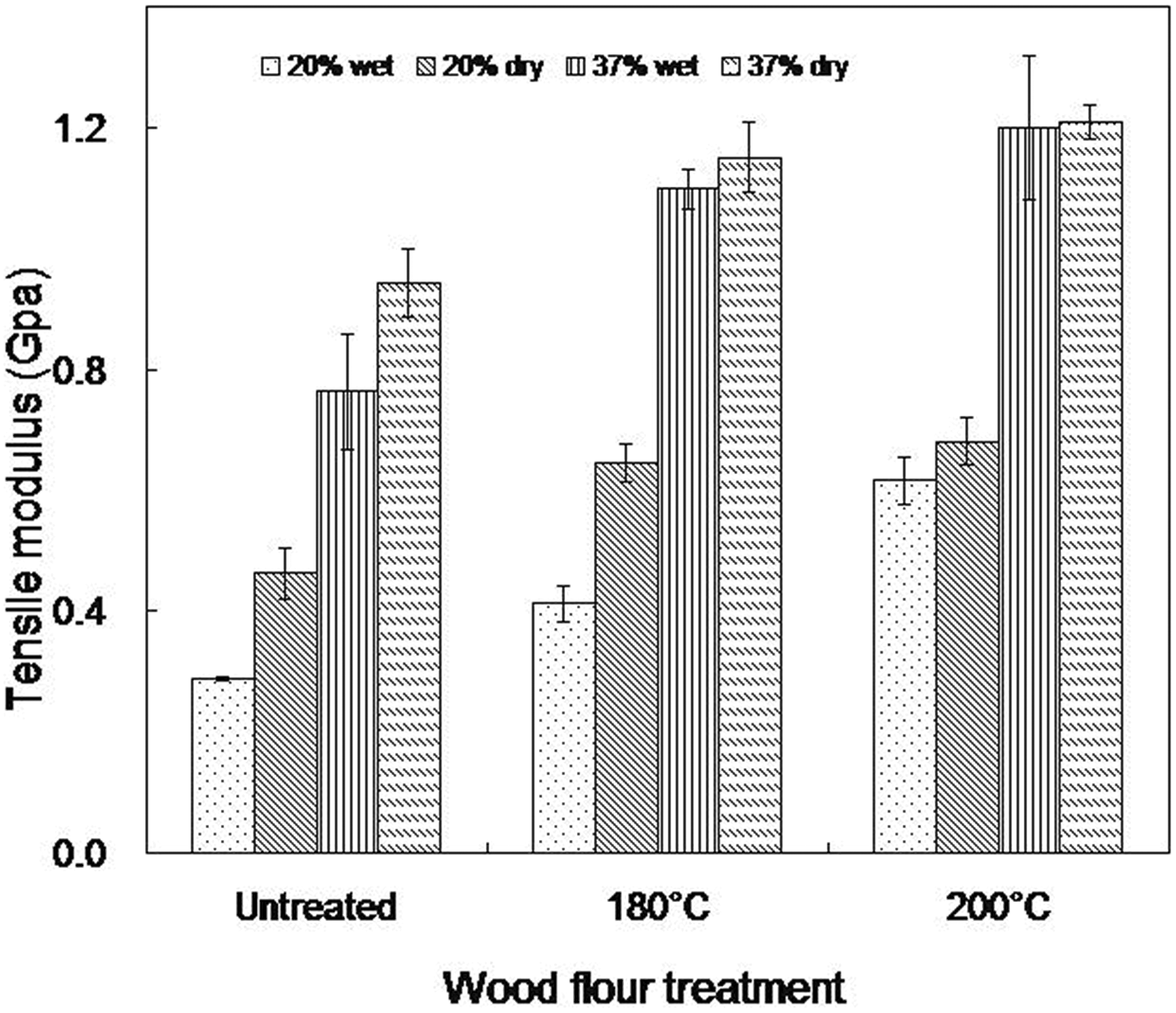
Tensile modulus of wet and dry as molded composites (20 and 37 wt% wood contents).
The relative extent of decrease is greater in untreated wood composites relative to the heat-treated ones. Composites containing 20 wt% and 37 wt% untreated wood flour present the highest reduction (38% and 23%, respectively) in tensile modulus after immersion in water compared to their counterparts made from wood flour treated at 180°C (36% and 5%, respectively) and those containing wood flour treated at 200°C (10.0% and 0.1%, respectively). The deteriorating effects of water molecules interacting with the wood at the composites interface due to the inherent incompatibility between the polar wood and the non polar matrix (Figure 3) may be responsible for this observation. As a result of the hydrophilic nature of wood, when WTC is exposed to water, the wood particles take up water resulting in the swelling of the wood cell wall, leading to changes in the dimensional stability of the composites and consequently, reduced mechanical properties. Also, water molecules present at the interface reduce the interaction between the filler and the matrix which can lead to poor mechanical properties. 34 The slight reduction in tensile modulus of LDPE/W180/37 with water absorption may indicate that reduced hydrophilicity in heat-treated wood enhanced better compatibility between the filler and the matrix. It has been reported that water molecules can act as a plasticizers to soften the wood cell wall polymers. 35 Also, the tensile modulus of a composite is determined by the stiffness of the filler and the matrix. In addition, it is a filler sensitive property in composites that is affected by moisture absorption. 36 Therefore, the lower the water uptake (Table 2), the lower is the amount of water molecules available to act as plasticizer to affect the tensile modulus. 13 However, composites from heat-treated wood at 200°C exhibit the lowest decrease in tensile modulus with water immersion (Figure 4). The better wetting shown by these composites (Figure 5(b)) may mean that the amount of water absorbed at equilibrium moisture content has minimal effect on the stress transfer capability. This can be seen from the fractured surface of moisture saturated composites containing 37 wt% wood flour treated at 200°C, with portions of the matrix adhering to the surface of the wood particles. In addition, the matrix of composites made from 200°C wood flour shows a more brittle failure (Figure 5(b)) than those containing untreated and 180°C treated wood flour (Figures 3 and 5(a)). On the other hand, LDPE/W180/20 shows a significant drop in tensile modulus compared to the other heat-treated composites.

Fractured surfaces of moisture saturated composites containing wood flour treated at (a) 180°C and (b) 200°C showing better encapsulation and lesser flaws.
Tensile strength decreases after immersion in water in the untreated wood composites (Figure 6). The tensile strength of samples made from 20 wt% untreated wood flour reduced from 8.01 MPa in the dry as molded samples to 6.08 MPa upon moisture saturation, while those formulated from 37 wt% untreated wood flour exhibit a 15% decrease in tensile strength. Again, the plasticization effect of water on WTC may be accountable for this observation and is in agreement with literature. 3,31,37 More so, in the presence of moisture, cellulose at the filler–matrix interface tends to swell. This results in a shear stress at the interface which favors ultimate debonding of the fillers, leading to a reduction in the tensile strength. 5 However, the tensile strength of composites made from heat-treated wood did not show a significant decrease. Samples containing 20 wt% wood flour treated at 180 and 200°C exhibit a marginal reduction from 8.91 MPa to 8.81 MPa and 9.92 MPa to 9.81 MPa, respectively, whereas, a decrement of about 8 and 4% are recorded in 37 wt% composites made from 180°C and 200°C, respectively. This could mean that the better interfacial adhesion in these composites due to heat treatment becomes predominant relative to the induced shear stress. The tensile strain of the wet composites decreased generally for all the composites studied relative to the dry ones (Figure 7). This is anticipated because of the notching effects of wood particles. In addition, excessive absorption of water could soften the wood particles resulting in low strain.
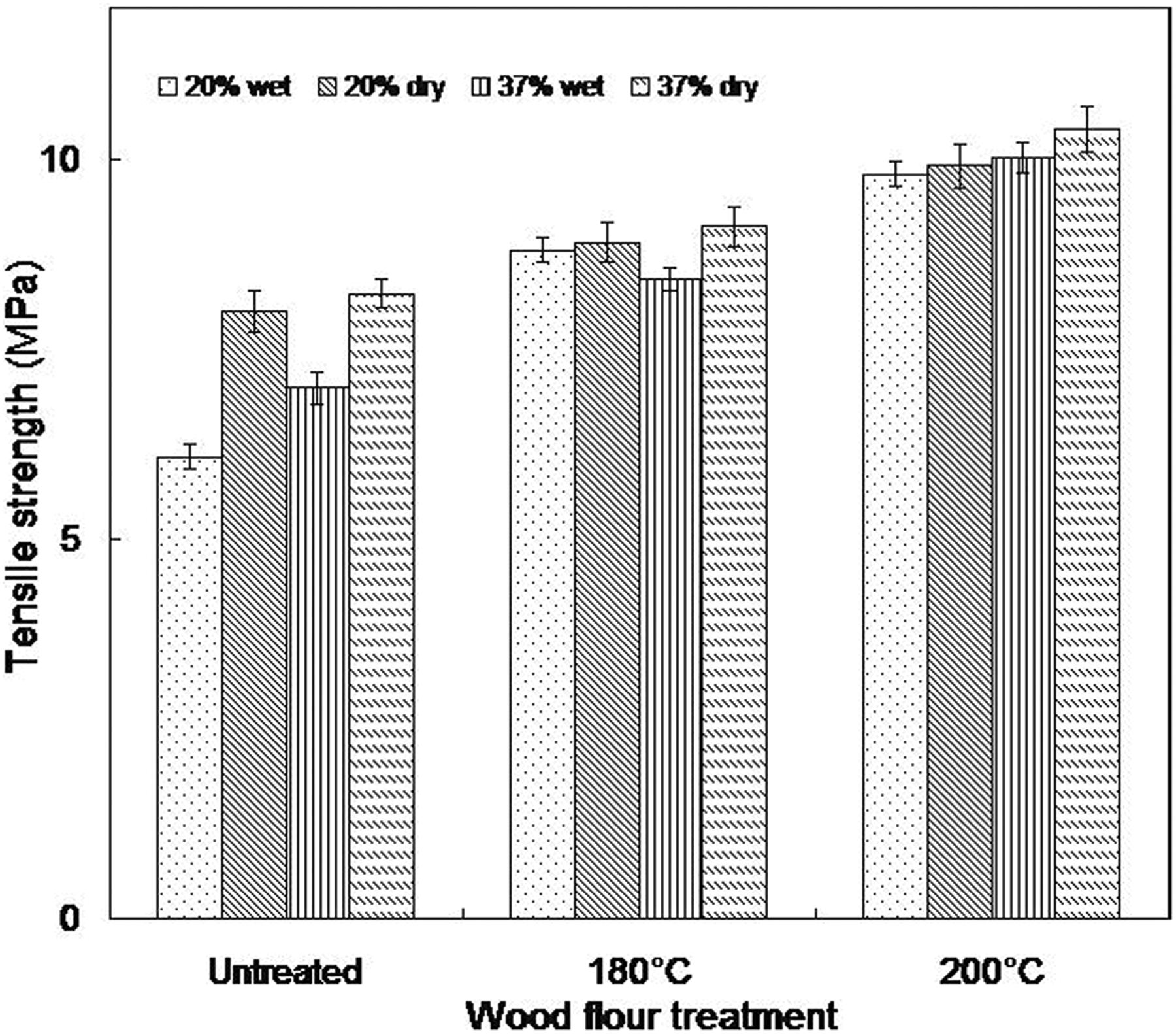
Tensile strength of wet and dry as molded composites (20 and 37 wt% wood contents).
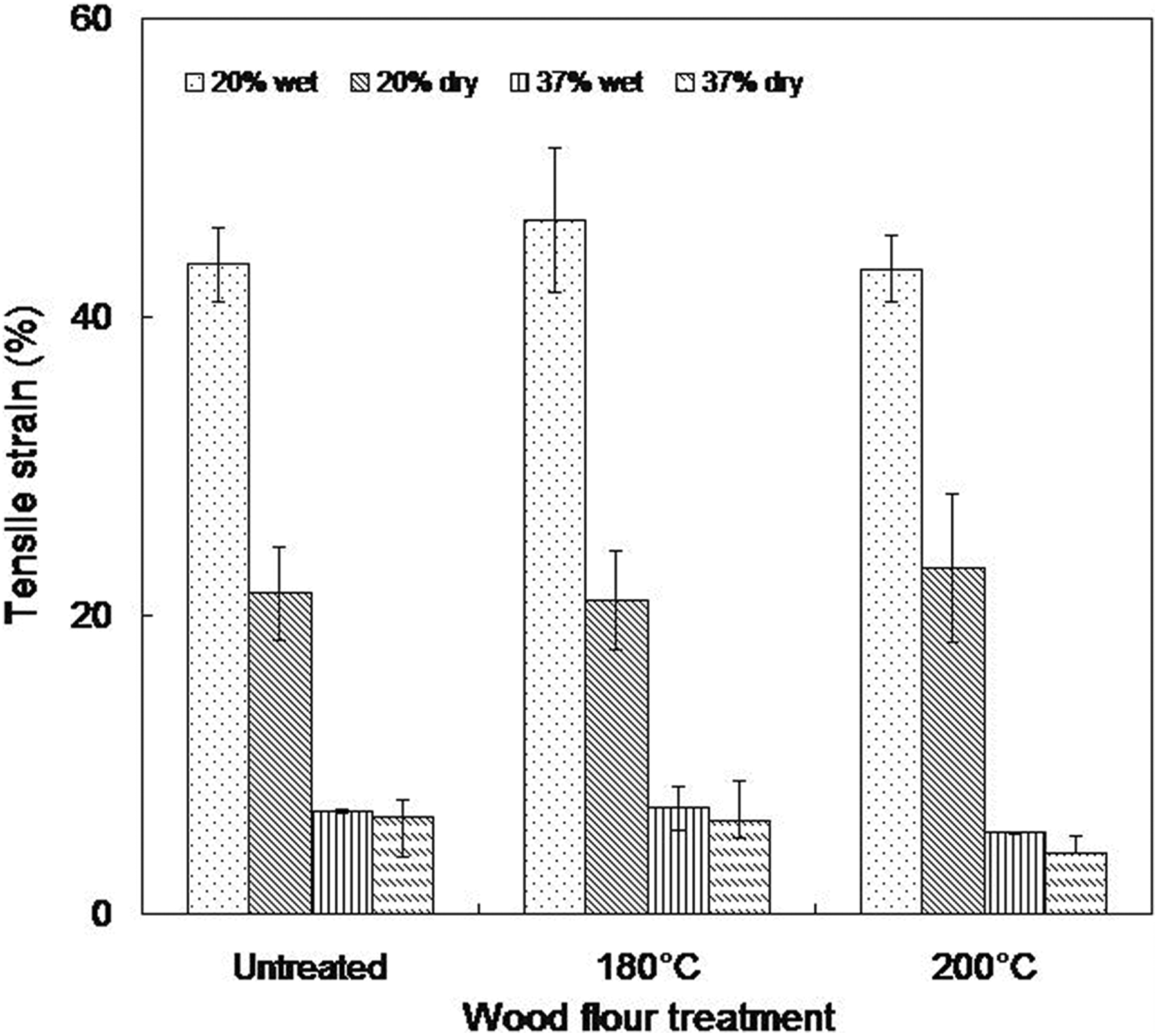
Tensile strain of wet and dry as molded composites (20 and 37 wt% wood contents).
Flexural properties
Figures 8 to 10 present the flexural properties of water soaked and dry as molded composites.
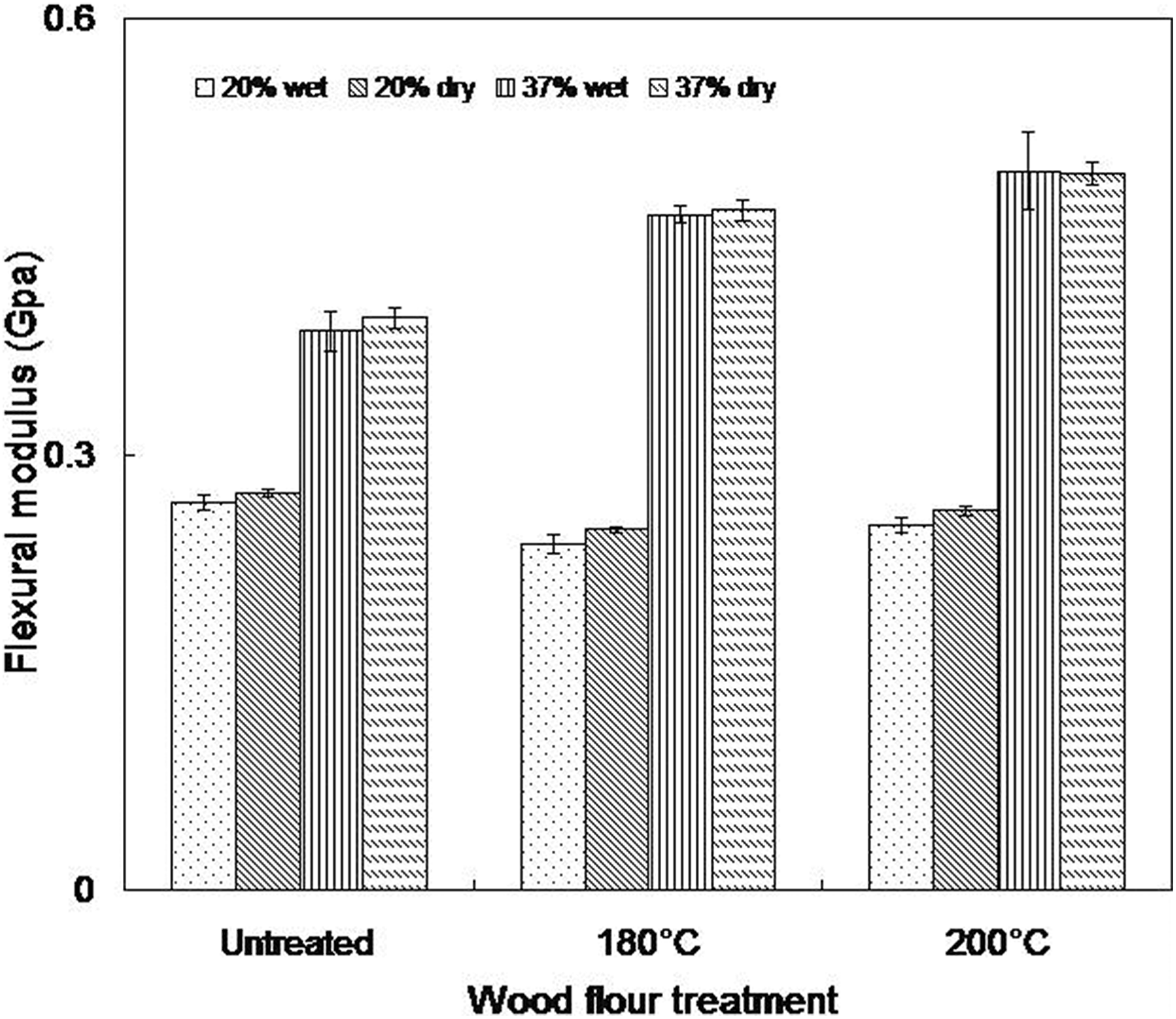
Flexural modulus of wet and dry as molded composites (20 and 37 wt% wood contents).
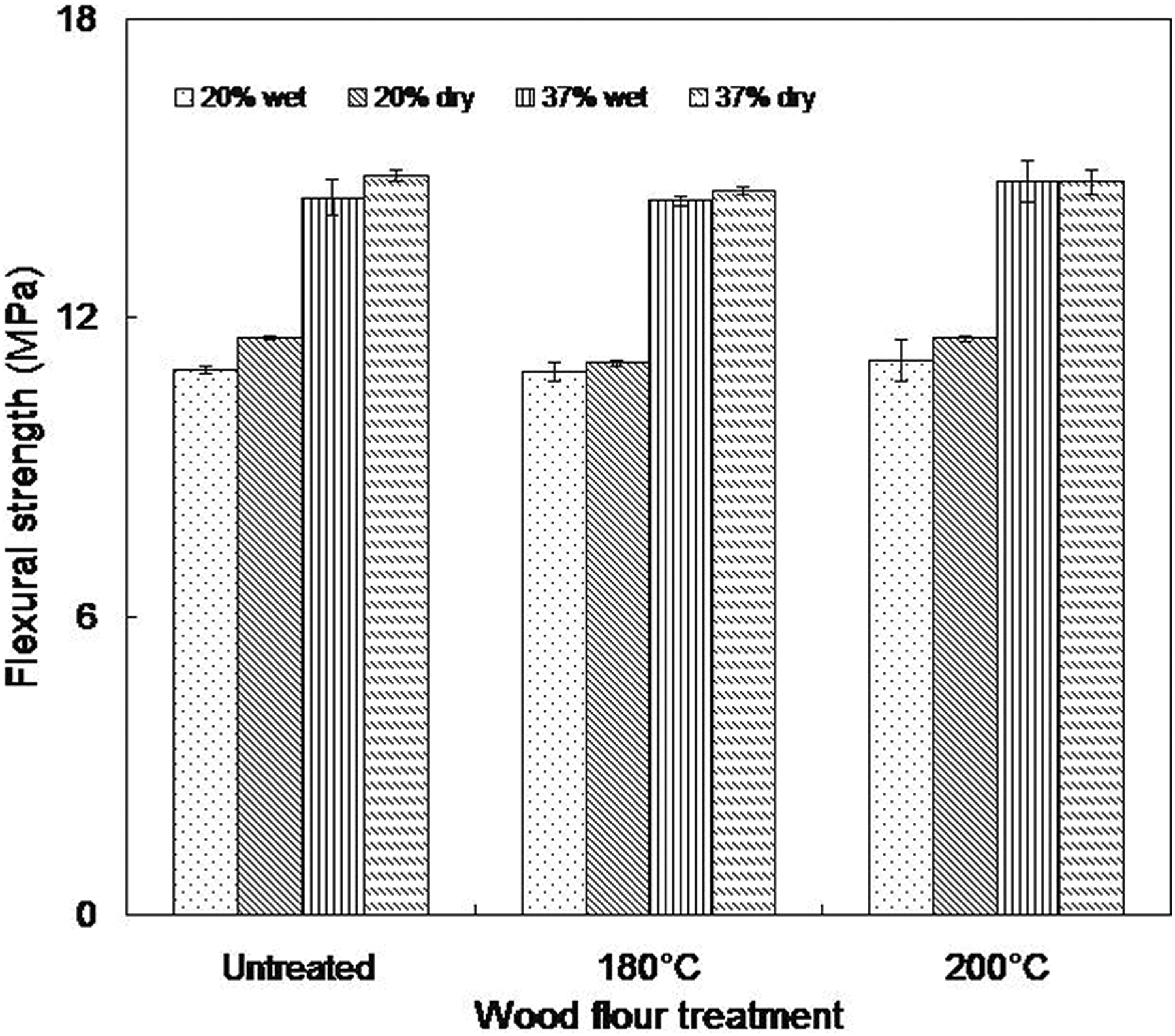
Flexural strength of wet and dry as molded composites (20 and 37 wt% wood contents).
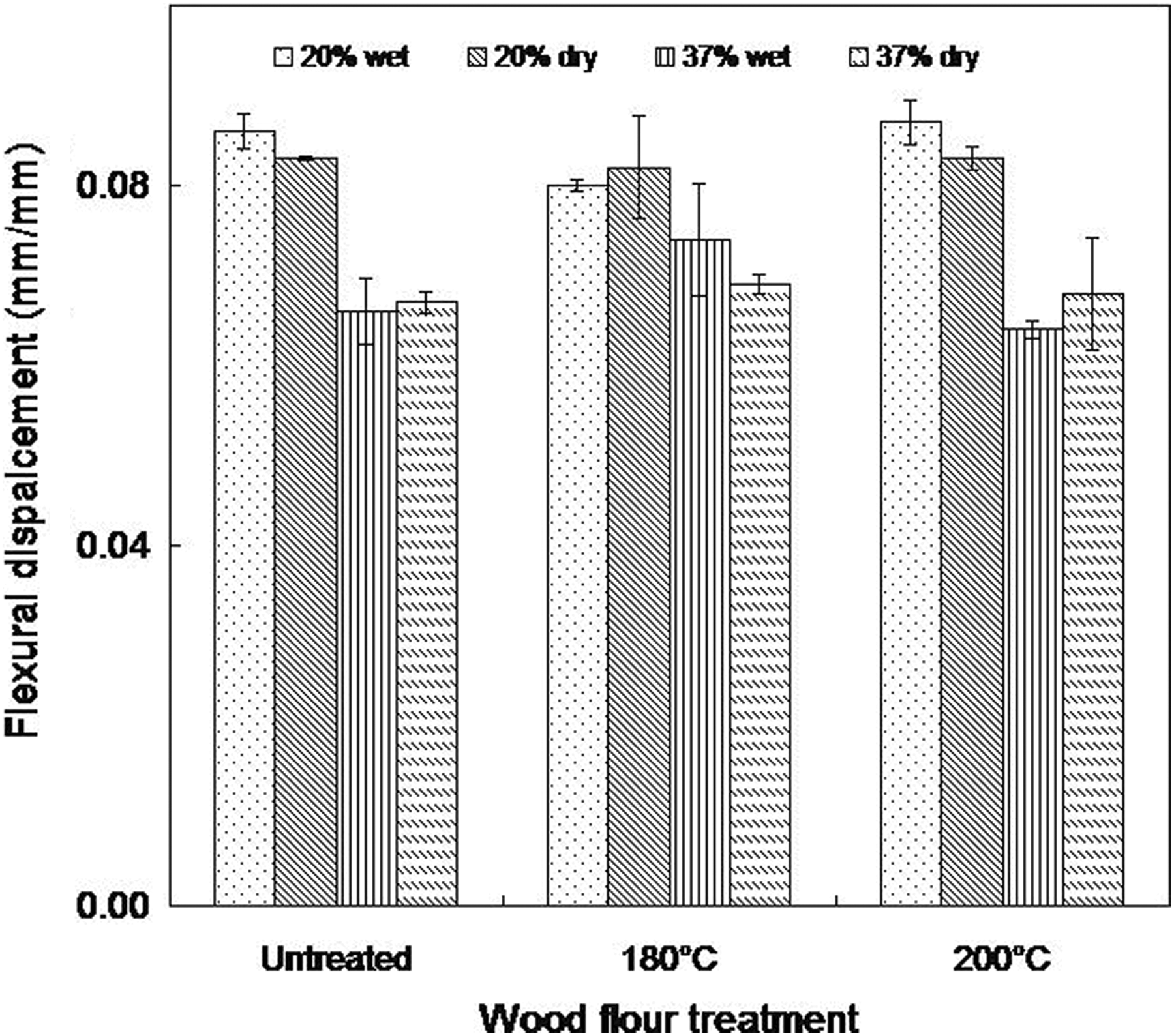
Flexural displacement of wet and dry as molded composites (20 and 37 wt% wood contents).
Generally, water absorption seems to be of no significant influence on the flexural modulus in all the composites studied. A 2% reduction in flexural modulus was observed for water-soaked composites made from untreated wood flour relative to that recorded in the samples containing heat-treated wood flour (less than 1%). Also, the flexural strength decreased marginally in wet samples relative to the dry as molded ones. Again, the rate of decrease is higher in composites made from untreated wood flour (Figure 8). However, no particular trend is observed in the flexural displacement with moisture absorption (Figure 10).
Conclusion
All the composites were found to absorb moisture to various extents due to the hydrophilic nature of wood. However, composites from heat-treated wood showed lower water absorption tendency because of the reduced hydrophilicity imparted to the wood by heat treatment. Most of the composites exhibited the Fickian mode of water absorption with
Supplemental Material
Suplementary_materials - Water absorption behavior of heat-treated and untreated red balau saw dust/LDPE composites: Its kinetics and effects on mechanical properties
Suplementary_materials for Water absorption behavior of heat-treated and untreated red balau saw dust/LDPE composites: Its kinetics and effects on mechanical properties by Ruth A Lafia-Araga, Aziz Hassan, R Yahya, N Abd Rahman and Fauzani Md Salleh in Journal of Thermoplastic Composite Materials
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the University of Malaya through grant numbers PS347/2009C and RG150/11AFR. Ruth acknowleges the financial support for her PhD studies by the Tertiary Education Trust Fund, Federal Ministry of Education, Nigeria.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
