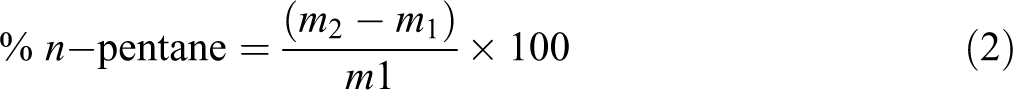Abstract
The foaming process involves three important steps: nucleation, bubble growth, and stability. In the present work, the foaming dynamics of styrene–methyl methacrylate (St-MMA) copolymer/n-pentane batch foaming system was studied at nonpressurized condition using rapid temperature increasing method. Synthesized copolymers with different compositions were characterized by proton nuclear magnetic resonance analysis and rheological measurements. An in situ foaming observation apparatus was designed, and the foaming dynamics (nucleation and bubble growth behavior) was studied. Dissolved n-pentane contents in all synthesized copolymer compositions at saturation temperatures, near their glass transition temperatures, were determined. The effects of foaming temperature, copolymer composition, and n-pentane content on the foaming dynamics of St-MMA copolymer/n-pentane system were investigated. The foaming results revealed that the nucleation rate was increased with the increasing foaming temperature, while two other aforementioned parameters were kept constant. The nucleation and bubble growth rate were decreased with increasing MMA units in copolymer composition and increased with dissolved n-pentane content increment in copolymer matrix.
Introduction
Polymeric foams have a porous structure composed of gaseous cells surrounded by a polymeric matrix. The low density and high cell density of these materials are more effective compared to the other properties. 1 –3 Generating foams using a physical blowing agent consists of saturating the polymer matrix with a blowing agent at a certain sorption temperature and pressure. The saturated polymer matrix is then subjected to a sudden thermodynamic instability (temperature increase or pressure drop), which causes the saturated gas to escape. Then, dissolved gas molecules in the polymer matrix diffuse into the nucleated bubbles and take them to grow up. In general, the foaming process involves three important steps: bubbles nucleation, growth, and stability. 4 –6 It is well-known that the cell density, cell size, cell size distribution, and foam density have important effects on the final property of prepared foam. Subsequently, a basic understanding of the relationships between the processing conditions and microstructure during foaming process (the number of nucleated bubbles and bubble size distribution) remains important to control the final microstructure of foams and consequently producing foams with high useful properties. 7 –10
Rapid pressure drop method is mostly used to study the foaming process, 11 –14 but some researchers such Iliev et al. 15 and Emami et al. 16 used temperature increasing method at nonpressurized conditions to examine the foaming dynamics. Generally, the rapid pressure drop method has been used more than temperature increasing method to study the foaming dynamics. Among processing parameters, sorption pressure and pressure drop rate have defined effect in each foaming system process, in which the nucleation and bubble growth rate increase with increasing sorption pressure and suddenly pressure drop, but temperature has no similar effect on the foaming dynamics in different foaming systems. 17
Some researchers have used supercritical CO2 (ScCo2) and ScN2 as physical blowing agents to study the in situ observation of the foaming dynamics (nucleation and bubble growth), 18 –22 but a few researchers such as Salejova and Kosek, 23 Azimi et al., 6 and Kasiri et al. 7 have studied the foaming dynamics of impregnated particles using pentane as a blowing agent. It is noted that nucleation phenomenon and bubble growth behavior have not been investigated deeply in the foaming system using pentane as the blowing agent compared to ScCO2 and ScN2. Since sorption pressure in batch foaming system using n-pentane as the blowing agent is not too high compared to ScCO2 and ScN2, pressure drop rate will not be too high. So, according to classical nucleation theory, nucleation rate is low, and the prepared foams by this foaming system have lower cell density compared to the high-pressure foaming systems. 6,7,23
To increase the cell density of prepared foams by n-pentane, temperature increment method at nonpressurized condition was used in this work. So, styrene–methyl methacrylate (St-MMA) copolymers were synthesized by suspension copolymerization process to investigate their foaming dynamics (bubbles nucleation and growth behavior) with n-pentane as the blowing agent via a batch foaming visualization apparatus. The effect of foaming temperature, n-pentane gas content, and copolymer composition on the foaming dynamics was also examined, which has not been investigated before.
Experimental
Materials
MMA and St monomers (Dae-Jung Chemicals & Metals Co., Ltd., Gyeonggi-do, South Korea) were used to synthesize St-MMA copolymer. Benzoyl peroxide (BPO) as a radical initiator; polyvinyl alcohol (PVA) with a molecular weight of 72,000 g mol−1, hydroxyethyl cellulose (HEC), and polyvinylpyrolidone (PVP) with a molecular weight of 360,000 g mol−1 as drop stabilizers; and potassium persulfate (K2S2O8) as an aqueous phase initiator were supplied by Merck (Darmstadt, Germany). Tricalcium phosphate (TCP) and sodium dodecylbenzene sulfonate (DBSNa) as suspending agents were supplied by Sigma-Aldrich (Sigma-Aldrich Chemie Gmbh, Munich, Germany). Physical blowing agent in foaming experiments was n-pentane with 99% purity (CARLO ERBA Reagents, Italy).
Suspension copolymerization
The St–MMA particles were synthesized in three copolymer compositions using a batch suspension polymerization according to our previous works. 1,5 –8,24 To identify the synthesized copolymers with different compositions, Co1, Co2, and Co3 abbreviations were used for St–MMA with 70/30, 53/47, and 30/70 feed ratios (wt%), respectively. First, the mixture of PVA, TCP, PVP, HEC, DBSNa, and K2S2O8 with distilled deionized water in a certain amount was prepared in the reactor under stirring rate of 280 r min−1 for at least a few hours to produce a homogeneous mixture. 1,5 –8,24 Then, the mixture of monomers which involved St and MMA monomers and BPO at a required amount 6,24 was stirred for 30 min and transferred to the reactor. Then, the reactor was heated to the reaction temperature (80°C) and held for 7 h. After completion of the reaction, the reactor was cooled to 35°C, and the reaction mixture was brought out, washed with water, dried, and sieved.
Copolymer characterization
Composition of the synthesized copolymers was determined using the proton nuclear magnetic resonance (H-NMR) analysis of the samples obtained with a Bruker (Avance DPX) NMR spectrometer working at 500 MHz according to our previous studies. The molar percentage of monomer incorporated to the particles was determined using equation (1): 6,8,24
where m 1 and m 2 are the mole fractions of St and MMA units in copolymer, respectively. A 1 is the integrated area of the phenyl protons of St unit at 6.5–7.5 ppm, and A 2 is the integrated area of all the protons of the copolymer. 8,24
In this work, the viscoelastic and rheological measurements were used to provide information on molecular weight (M w) and molecular weight distribution of synthesized copolymers according to our previous works. 5,6,24 The rheological measurements of synthesized copolymers were conducted at T = 200°C, angular frequencies (0.04–625 s−1), and strain of 1% under nitrogen atmosphere using a Physica MCR 301 dynamic rheometer (Anton Paar, Graz, Austria) with parallel-plate geometry.
Foaming procedure
A batch foaming observation system which was designed and explained clearly in our previous work was used to conduct foaming experiments. 5 –7 Copolymer sheets with a thickness of 200 μm were prepared by Brabender Polystat 200 T hot press (Brabender Co., Germany) and were placed into the designed chamber (Figure 1(a)). The samples were heated up to the required temperature, and subsequently, n-pentane blowing agent was inserted into sealed chamber from pentane flask. The copolymer sheet was impregnated by n-pentane in certain saturation pressure, temperature, and time. After impregnation period, the temperature of system was decreased quickly. When the temperature reached below the boiling temperature of n-pentane, the sample was taken out from the chamber.
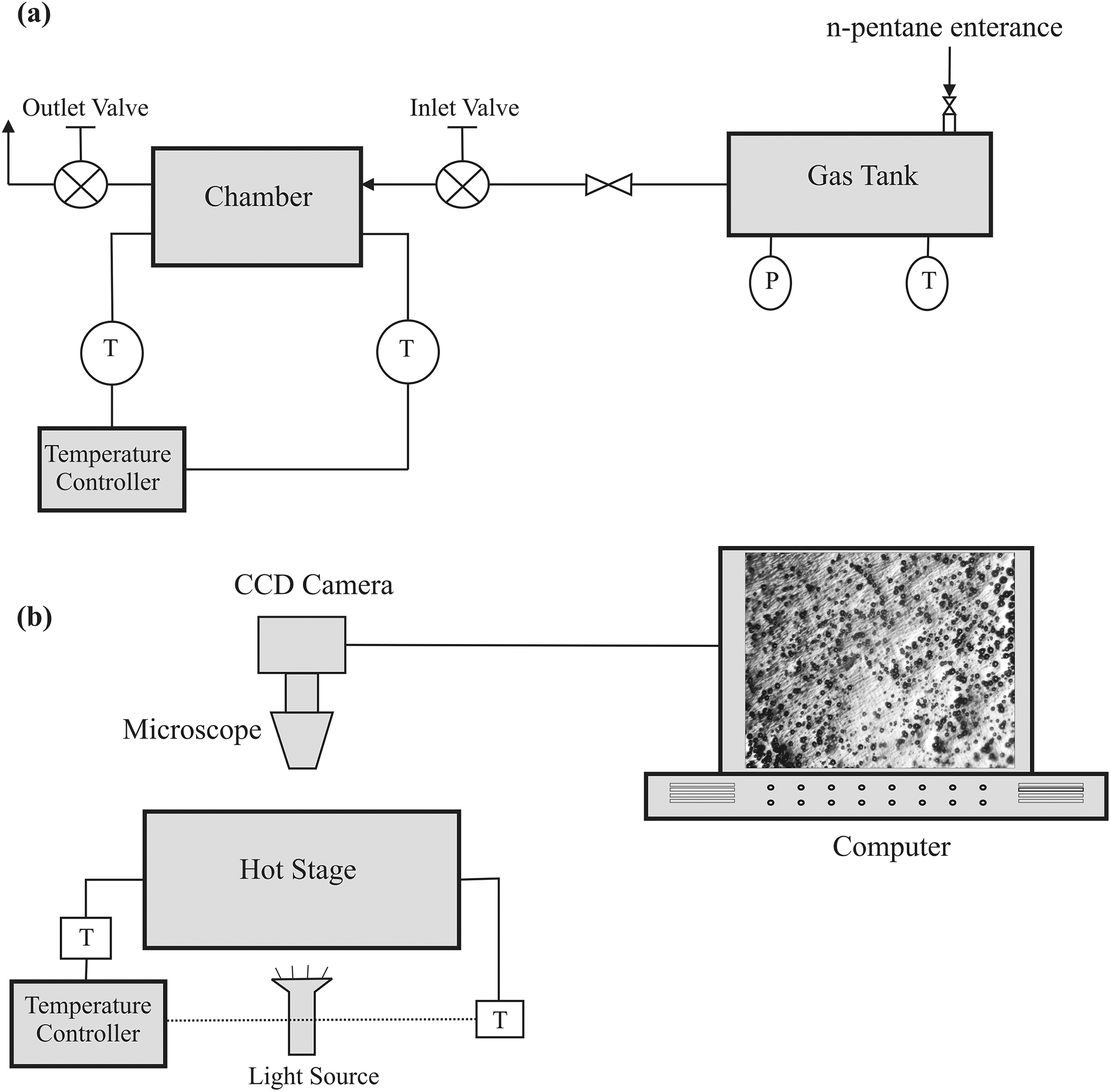
The schematic view of the designed (a) batch impregnation system and (b) foaming visualization system.
At the next step, the prepared disk with a diameter of 5 mm through impregnated and cooled sheets was placed into hot stage with a certain temperature, and the foaming process was started and recorded by a digital camera attached to the stereo microscope (Figure 1(b)). Each foaming experiment was carried out more than three times to examine the repeatability of the results. The resulting in situ visualization data of such enumeration of nucleated bubbles and their radius were analyzed using Bell View software (Bell, Italy) to obtain the bubble density and bubble growth profiles at each experimental condition.
Dissolved gas content measurement
The designed chamber according to Figure 1(a) was also used to measure dissolved n-pentane content in copolymers. The copolymer sheets with a thickness of 200 μm were prepared and weighted (m 1). After certain time (more than 3 h), they were impregnated by n-pentane at the specified temperature and sorption pressure. Then, the chamber was cooled quickly, and as soon as the temperature decreased below the boiling temperature of n-pentane, the sheets were brought out from the chamber and weighted (m 2). The dissolved content of n-pentane in sample was calculated using equation (2). 1,5
Results and discussion
Characterization of synthesized copolymers
Figure 2(a) to (c) shows typical H-NMR spectra of St-MMA copolymers at different feeding ratios for Co1, Co2, and Co3 samples. Peaks between 6.5–7.3 ppm and 3.6 ppm corresponds to C6H5 and OCH3 groups, respectively. The peak occurring at 2.8 ppm was assigned to the OCH3 group of poly(methyl methacrylate) bonded to the polystyrene segment, which was absent in the H-NMR spectra of both relevant homopolymers. 8,24 Compositions of the copolymers were evaluated using H-NMR spectroscopy and are presented in Table 1. As it can be seen, the calculated copolymer compositions are close to the monomer feed ratios.
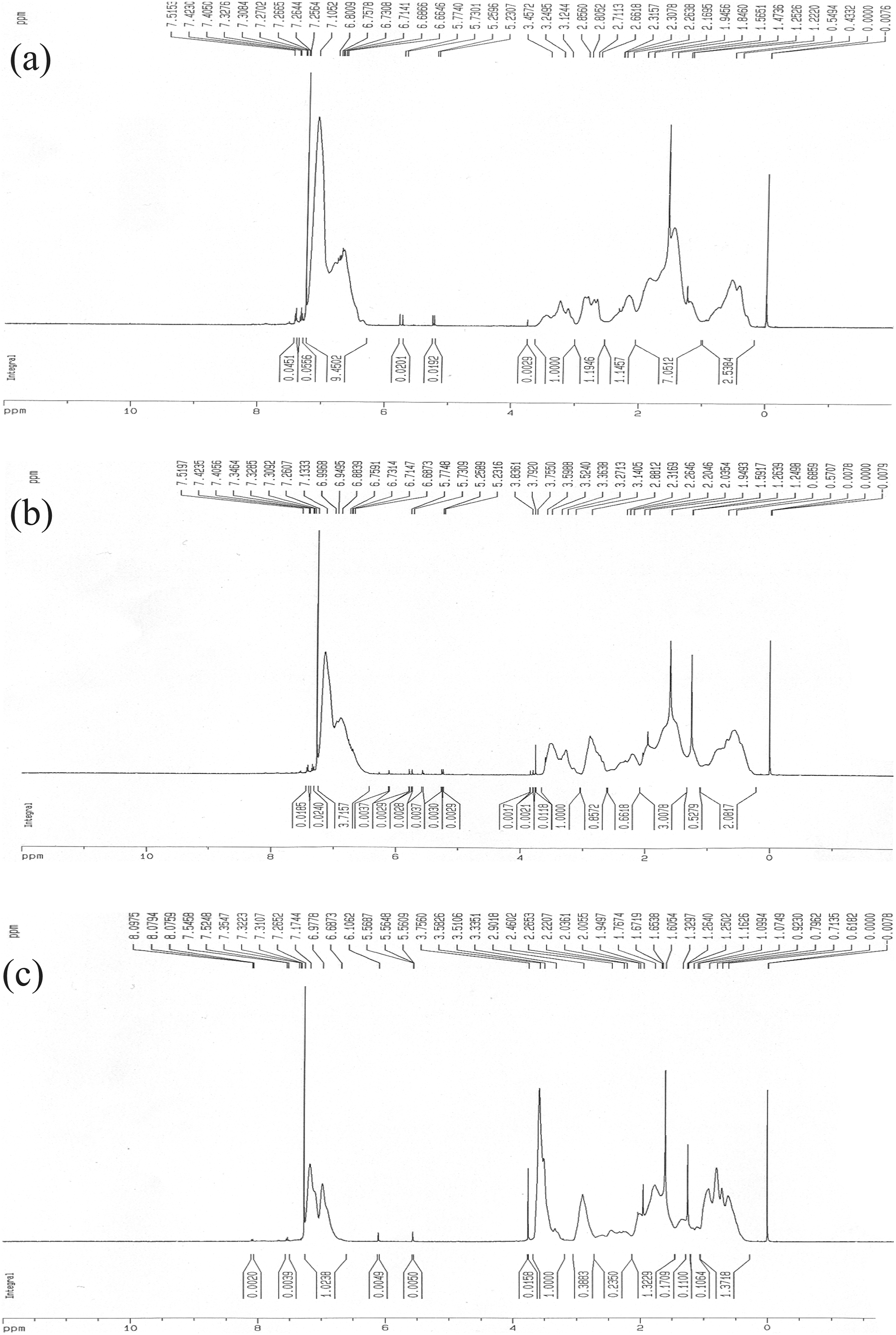
H-NMR spectra of the synthesized St-MMA copolymer which St-MMA weight ratio in feed is (a) 70/30, (b) 53/47, and (c) 30/70.
Compositions of synthesized copolymers calculated from H-NMR spectra.
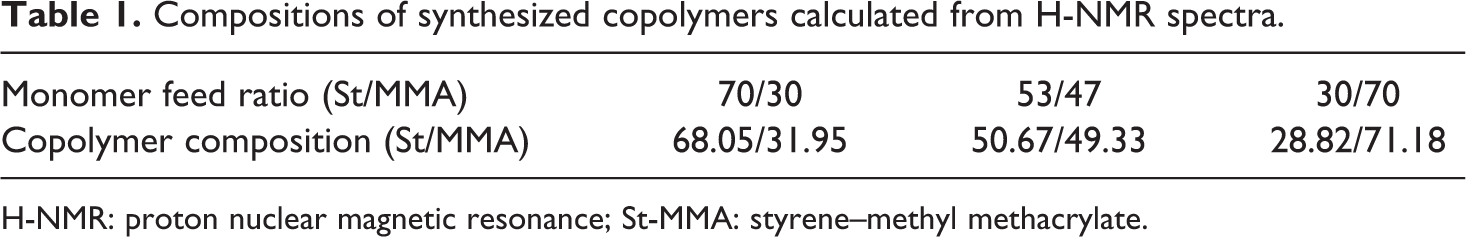
H-NMR: proton nuclear magnetic resonance; St-MMA: styrene–methyl methacrylate.
Complex viscosity (η*) and storage modulus (G′) of synthesized copolymers versus angular frequency are shown in Figures 3 and 4, respectively. Corresponding molecular weight and its distribution determined by rheological experiment are presented in Table 2. As it is clear, the complex viscosity and storage modulus of copolymer are increased with increasing MMA content, which could be attributed to the higher molecular weight of copolymers containing higher amount of MMA (Table 2). The zero-shear rate viscosity (η 0) for all copolymer compositions was calculated through rheological experiments using Carreau–Yasuda model and Cox–Merz rule, 25,26 which was 8.6 × 103, 8 × 104, and 7.8 × 105 for Co1, Co2, and Co3, respectively.
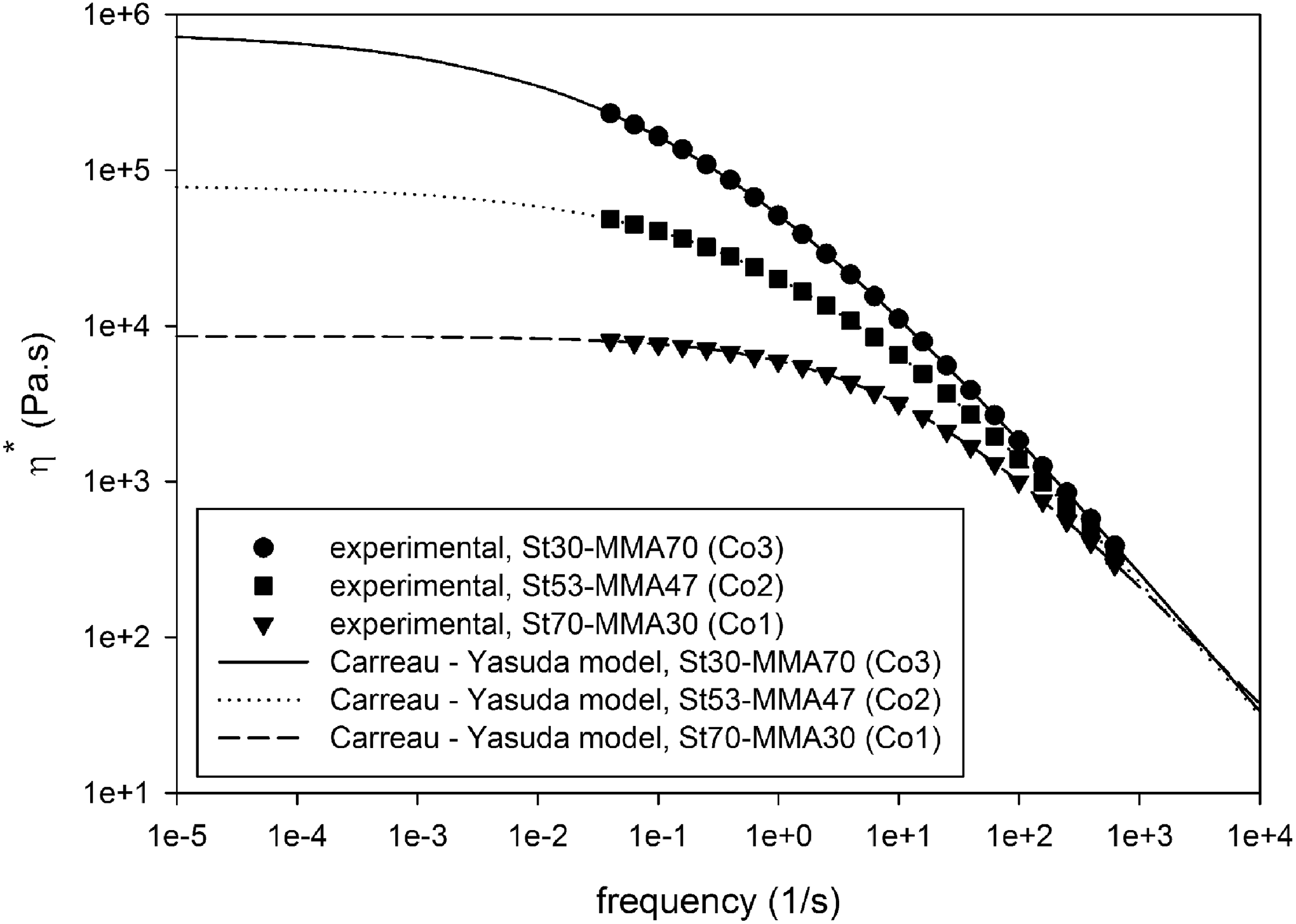
Complex viscosity (η*) of synthesized St-MMA copolymers versus angular frequency at 200°C. The lines represent nonlinear regression using the Carreau–Yasuda model.
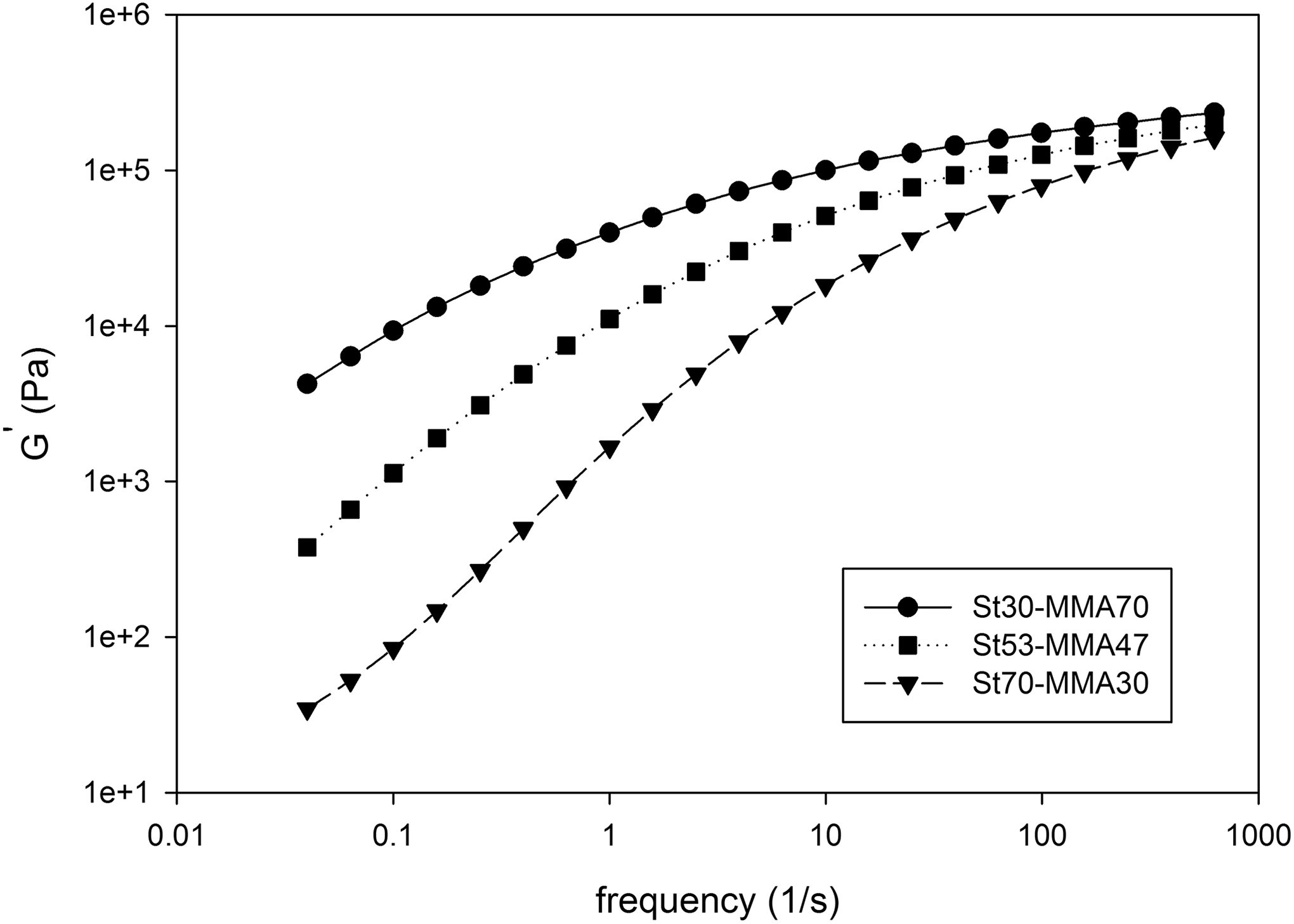
Storage modulus (G′) of synthesized St-MMA copolymers versus angular frequency at 200°C. The lines are drawn to guide the eyes.
Molecular weights and polydispersity index of St-MMA copolymers.
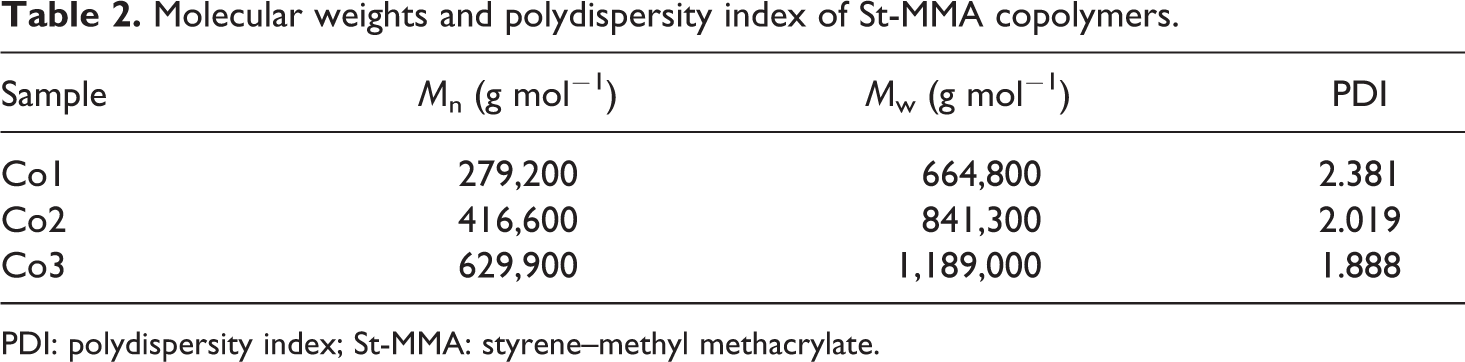
PDI: polydispersity index; St-MMA: styrene–methyl methacrylate.
Solubility measurements of n-pentane in St-MMA copolymers
Figure 5 shows the amount of impregnated n-pentane into copolymer sheets versus impregnation time at 100°C and sorption pressure of 4 bar. Since all samples were almost saturated after approximately 180 min, the impregnation time at the present study was taken more than 180 min for all samples and experiments. Moreover, it was found that the samples with higher St content were saturated more quickly than the other copolymers with lower St content. This finding depicts that the mass diffusivity of n-pentane increases with increasing St content in St–MMA copolymer.
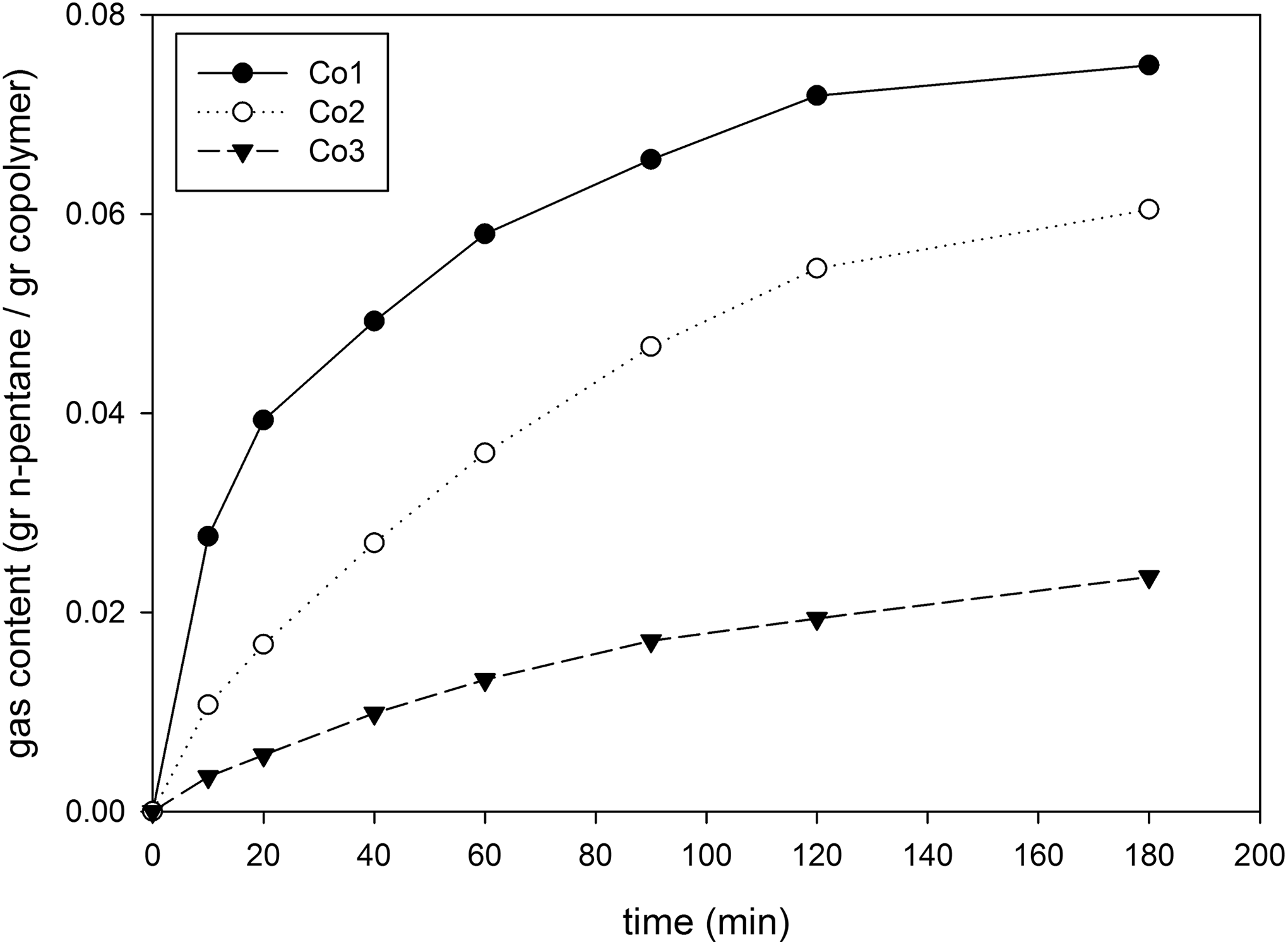
Sorption of n-pentane in copolymers versus impregnation time at T = 100°C and sorption pressure of 4 bar.
The dissolved amounts of n-pentane in St-MMA copolymers at different copolymer compositions, temperatures, and sorption pressures are shown in Figure 6(a) to (c). As it can be seen, the dissolved amount of n-pentane in all samples was increased with increasing sorption pressure and decreased with increasing temperature. As the gas concentration in the impregnation chamber (surrounding the sample) was increased with sorption pressure, the diffusion driving force was increased, which led to the higher solubility of n-pentane in St-MMA copolymer. Furthermore, the mobility of gas molecules was increased at elevated temperatures suppressing diffusion of gas into the polymer matrix. Comparison among Figure 6(a) to (c) shows that the amount of dissolved gas decreased with increasing MMA units in copolymer composition, which could be attributed to the polarity of MMA units compared with n-pentane and St units in copolymers.
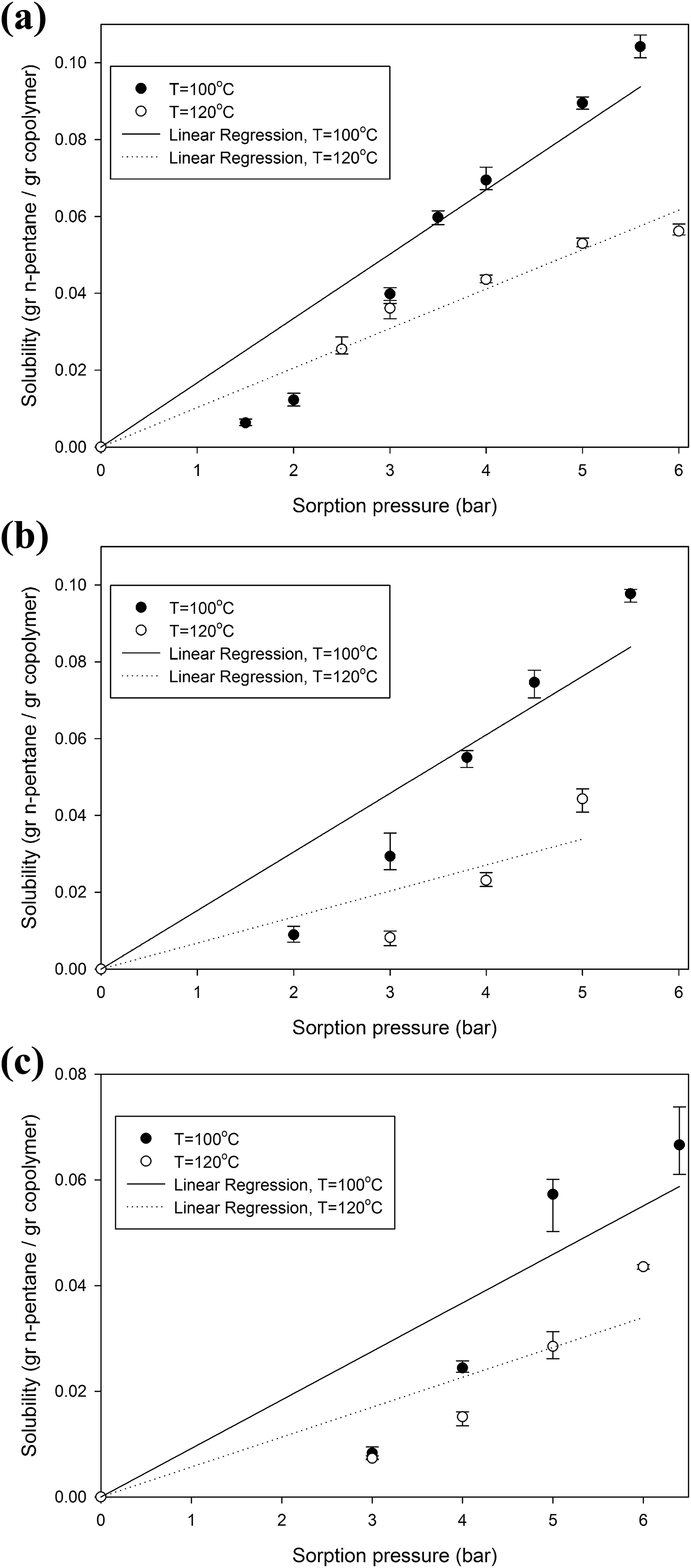
Dissolved contents of n-pentane in (a) Co1, (b) Co2, and (c) Co3 at different temperatures versus sorption pressure. Symbols indicate the average value of the solubility of four repeated experiments, the asymmetric error bars signify the minimum and maximum of data, and the solid lines represent linear regression.
Foaming dynamics of St-MMA/n-pentane system
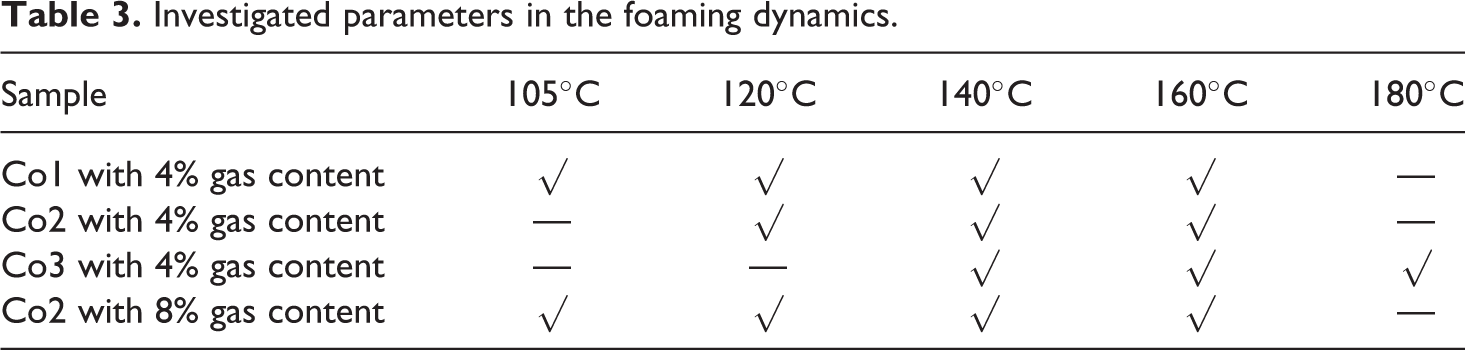
The main purpose of the present study is to focus on the investigation of the effect of copolymer composition, foaming temperature, and gas content on the foaming dynamics. Therefore, the investigated parameters in the foaming dynamics are listed according to Table 3, and all copolymer samples were impregnated by n-pentane based on conditions in Table 4. These conditions were justified using Figure 6 to reach the solubility of 4% n-pentane in St–MMA copolymers.
Investigated parameters in the foaming dynamics.
Saturation conditions to receive 4% dissolved content of n-pentane in copolymer sheets.

Figures 7 to 9 represent a sequence of optical micrographs of the foaming process of Co1, Co2, and Co3 samples, respectively, saturated with 4% gas content at different foaming temperatures. No bubbles were observed in the foaming temperature below 105°C for Co1, 120°C for Co2, and 140°C for Co3. Since melt strength (Figures 3 and 4) and glass transition temperature (T g) are increased with increasing MMA content in copolymers, minimum foaming temperature would increase as well. 1,6 Moreover, it can be observed that foaming process occurred earlier at higher foaming temperatures because of the reduction of copolymer matrix melt strength.

Visualization of Co1/n-pentane foaming process versus foaming time at nonpressurized condition with 4% dissolved n-pentane content and foaming temperature of (a) 105°C, (b) 120°C, (c) 140°C, and (d) 160°C. All images are 1336 µm in height and 1670 µm in width.
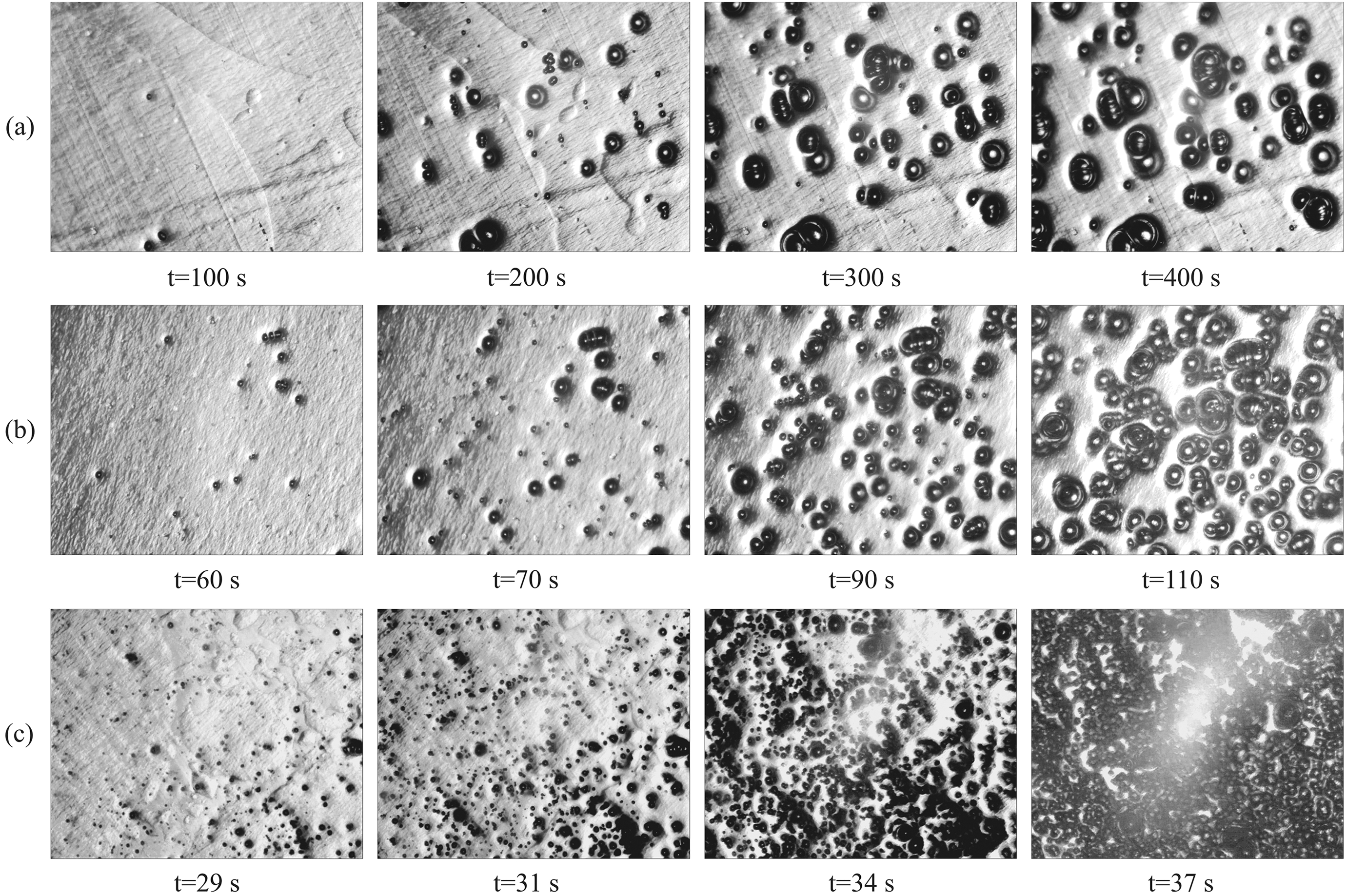
Visualization of Co2/n-pentane foaming process versus foaming time at nonpressurized condition with 4% dissolved n-pentane content and foaming temperature of (a) 120°C, (b) 140°C, and (c) 160°C. All images are 1336 µm in height and 1670 µm in width.

Visualization of Co3/n-pentane foaming process versus foaming time at nonpressurized condition with 4% dissolved n-pentane content and foaming temperature of (a) 140°C, (b) 160°C, and (c) 180°C. All images are 1336 µm in height and 1670 µm in width.
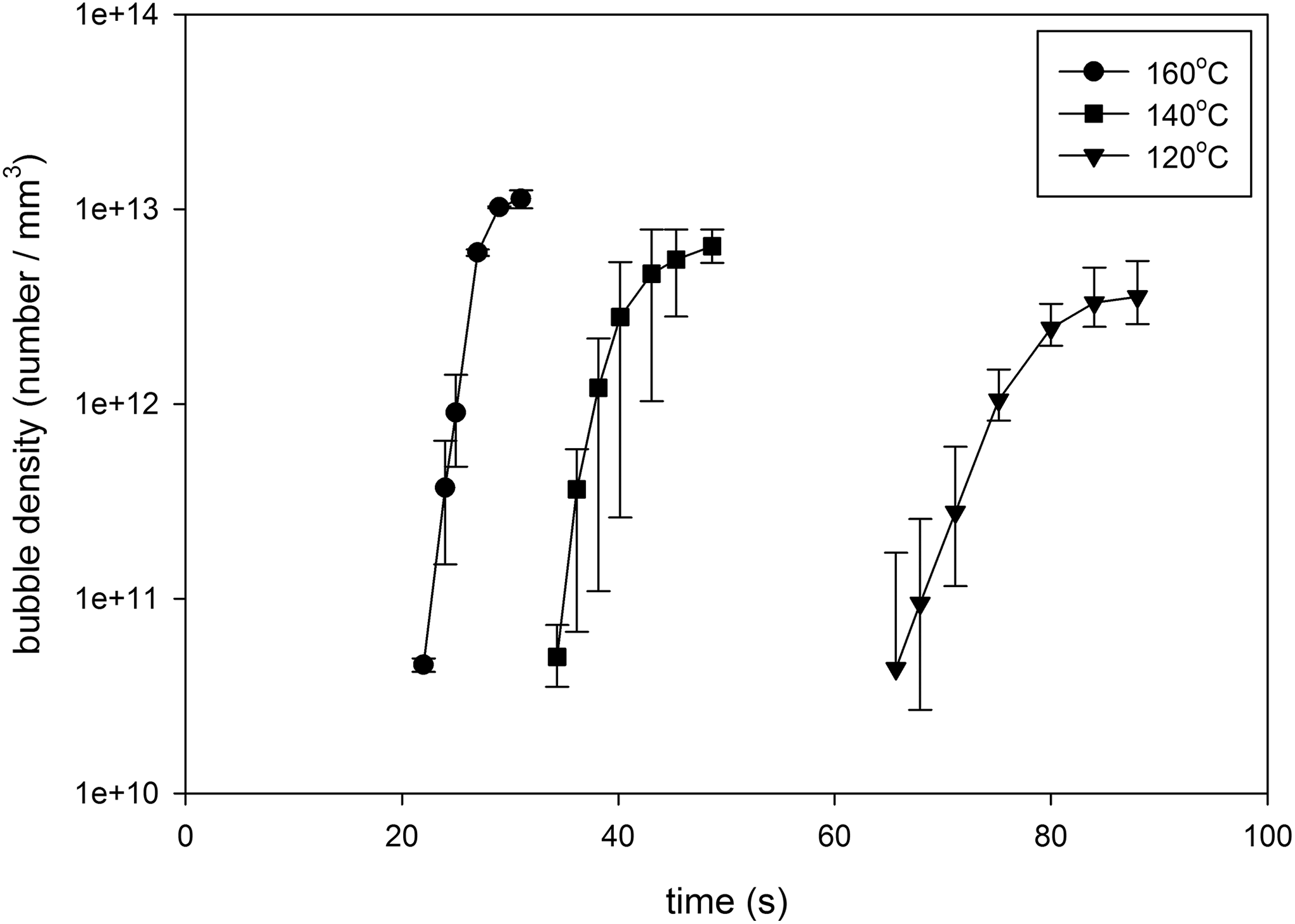
The number density of the observed bubbles versus foaming time for Co1, Co2, and Co3 samples with 4% n-pentane content is depicted in Figure 10(a) to (c). In this figure, each point represents the average bubble density of three experiments, and asymmetric error bars signify the minimum and maximum values of bubble density. Figures 10 and 11 also represent the effect of foaming temperature on the observed bubble density during the foaming process (nucleation rate) and the final bubble density at constant gas content and copolymer composition. The results reveal that increasing foaming temperature increase the nucleation rate and final bubble density. These figures also show that nucleation rate and final bubble density are decreased, and the foaming time is increased with increasing MMA content in copolymer composition. As the melt strength of the matrix is high and diffusivity of n-pentane is low in copolymers with high MMA content, both the nucleation and bubble growth rate are decreased.
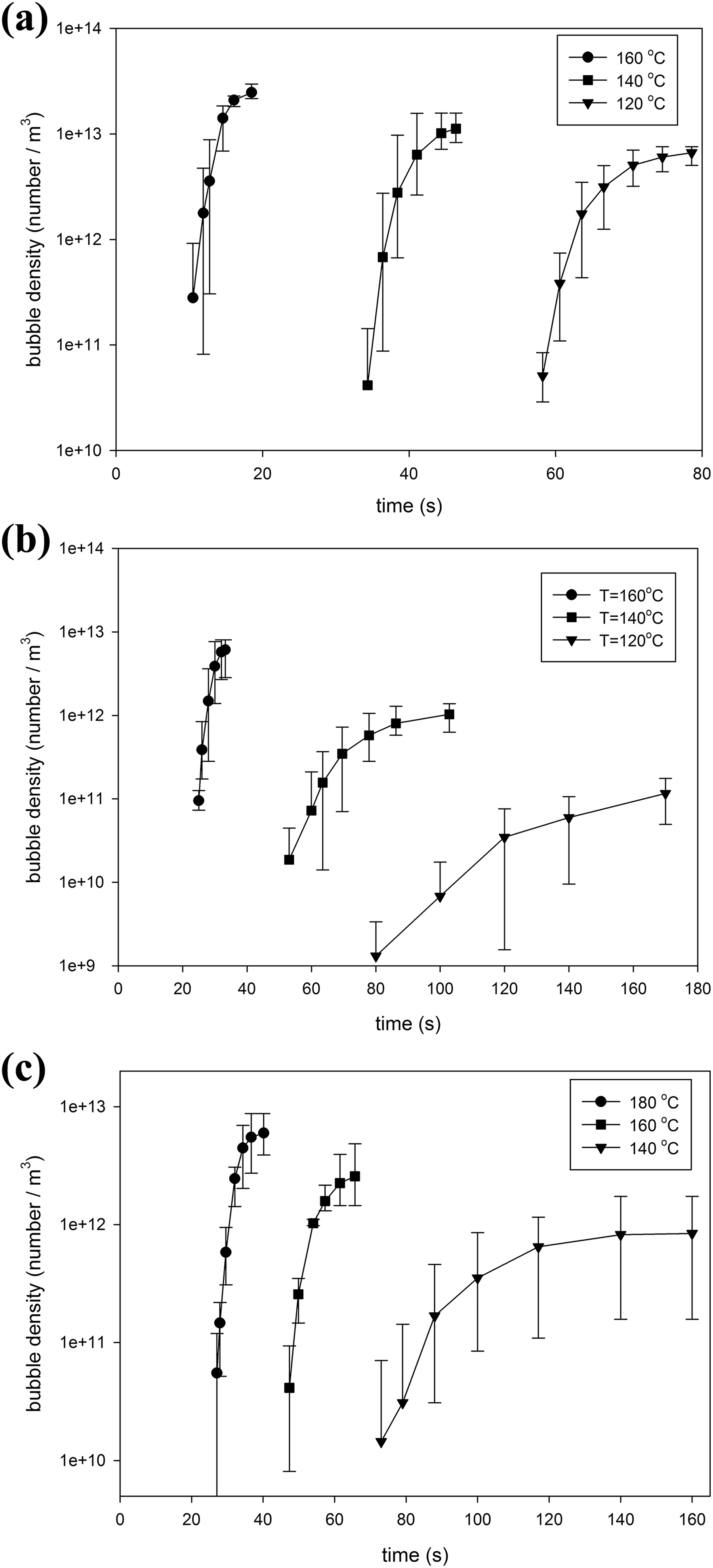
Number density of observed bubbles versus unfoamed volume respect to time in St-MMA copolymer/n-pentane foaming process at nonpressurized condition and different foaming temperatures with 4% dissolved n-pentane content: (a) Co1, (b) Co2, and (c) Co3.
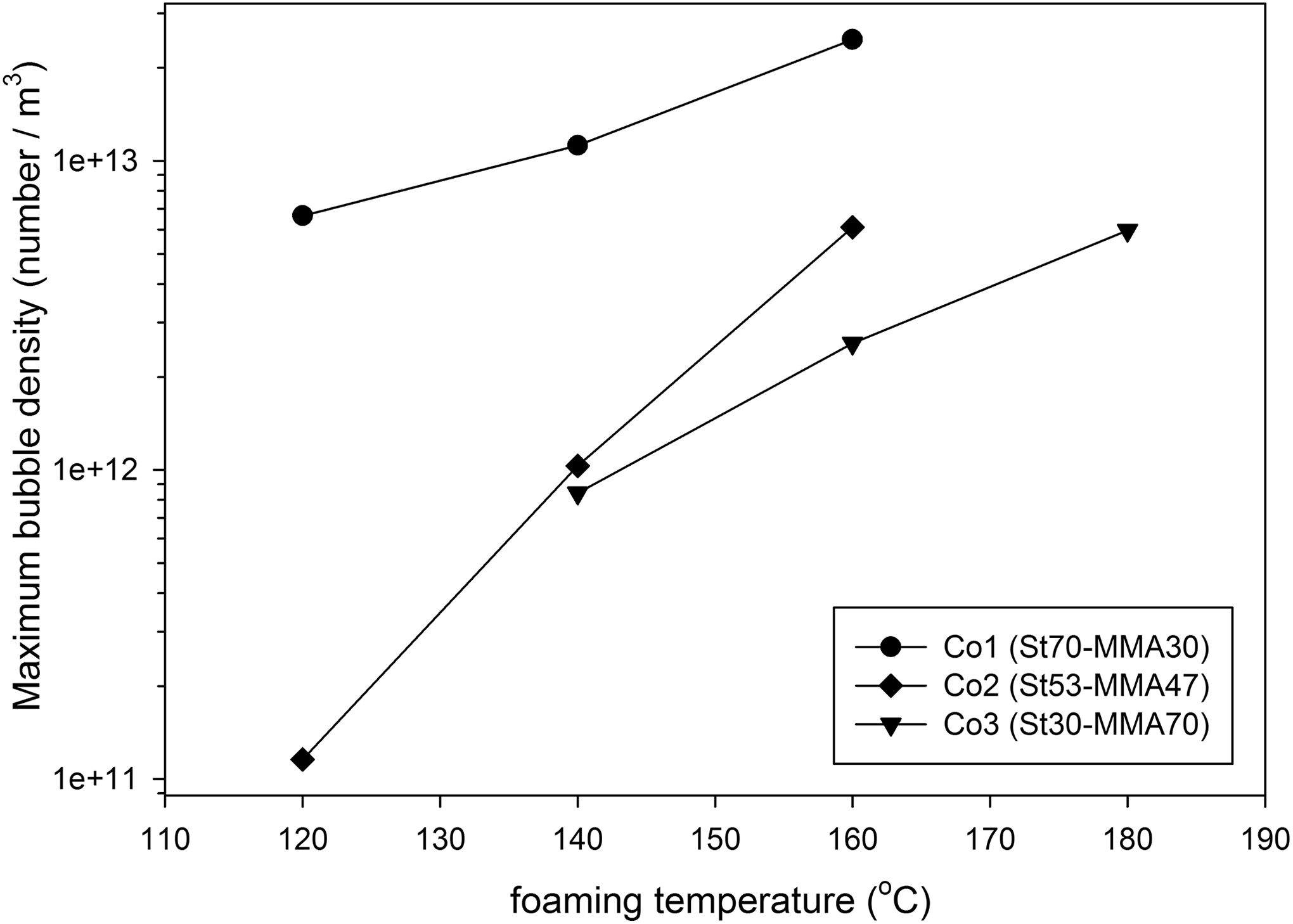
Maximum bubble density versus foaming temperature for different copolymer samples with 4% dissolved n-pentane content.
The average diameters of observed bubbles are presented in Figure 12(a) to (c) for Co1, Co2, and Co3 copolymers with 4% n-pentane content, respectively. The bubble expansion is a mass transfer-controlled process; therefore, increasing the mass diffusion coefficient accelerates the bubble growth rate. 5,27 As it can be seen, in all copolymer compositions, the higher expansion rate is attributed to higher foaming temperature because of the higher gas diffusivity and lower melt strength in those samples. Comparing the bubble growth behavior (Figure 12) shows that bubbles grow more slowly with MMA content increment in copolymers. This finding could be attributed to the reduction of n-pentane diffusivity and an increase in the melt strength of the copolymer matrix. Figure 12 also depicts that the final average size of bubbles in all copolymers is decreased as foaming temperature increases. Since the observed bubble density is increased at higher foaming temperatures, shell thickness around the bubbles becomes thinner, indicating that the available n-pentane for each growing bubble is decreased. Although the average bubble sizes are small, they grow up faster due to higher gas diffusivity at higher foaming temperature. It should be noted that while Co3 sample had lower nucleation rate (i.e. higher available n-pentane for each growing bubble), bigger bubbles are not observed for this sample, especially when compared to Co2 sample. In the case of Co3 sample, it can be concluded that the effect of lower diffusivity of n-pentane and higher melt strength of this copolymer matrix overcomes the effect of higher available gas content, which consequently led to smaller bubble size.
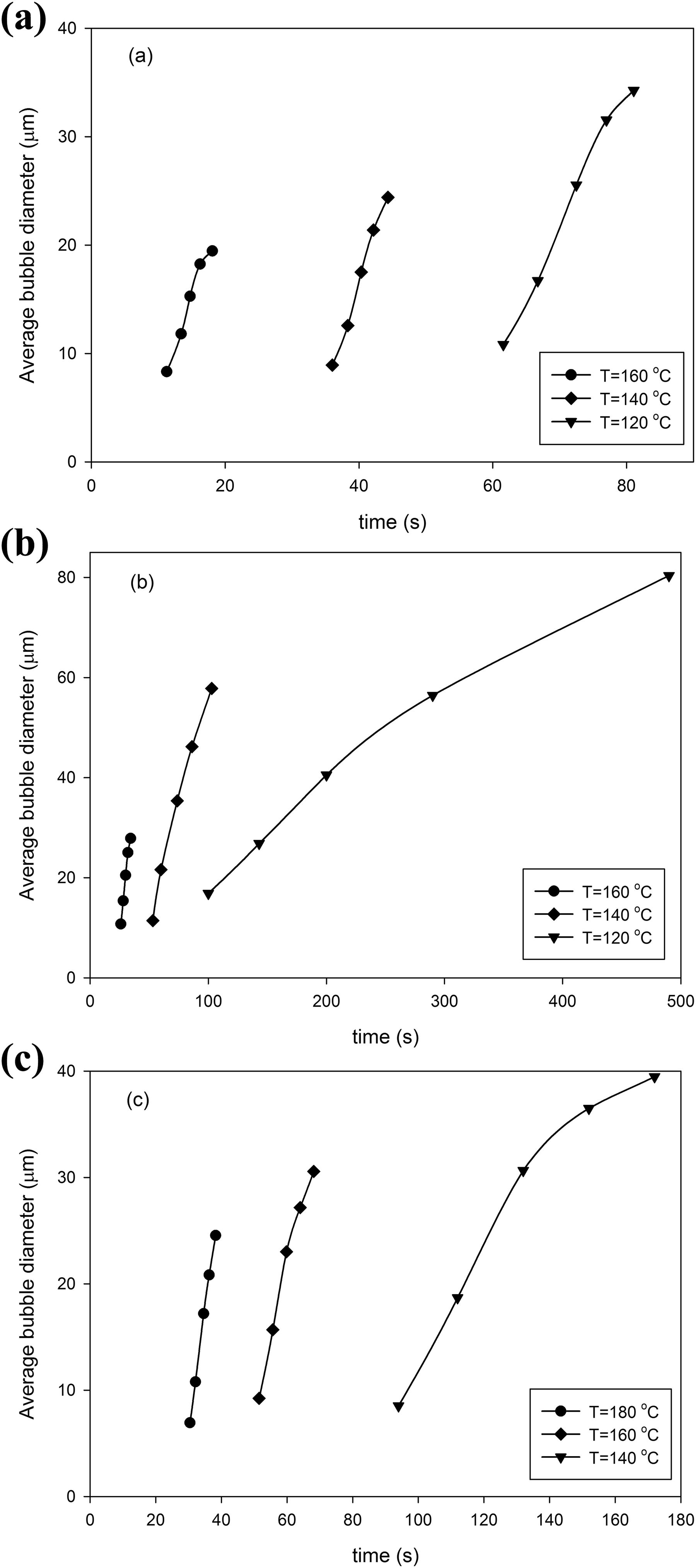
Average bubble diameter respect to time in St-MMA copolymer/n-pentane foaming process at nonpressurized condition and different foaming temperatures with 4% dissolved n-pentane content: (a) Co1, (b) Co2, and (c) Co3.
To investigate the effect of the available gas content on the foaming process, Co2 sample impregnated with n-pentane at 100°C and 4.7 bar for 4 h to attain the solubility of about 8% (Figure 6(b)). Then, the impregnated samples are foamed at different temperatures (100–160°C). The optical microscopy images of the foaming process are presented in Figure 13. As it can be noted, Co2 sample with 4% n-pentane is not foamed at a foaming temperature of 100°C; however, the sample with 8% n-pentane is expanded at 100°C. Higher amount of n-pentane in this copolymer matrix reduces the T g of the matrix, which makes it possible to be softer and expandable.

Visualization of Co2/n-pentane foaming process versus foaming time at nonpressurized condition and different foaming temperatures with 8% dissolved n-pentane content and foaming temperature of (a) 105°C, (b) 120°C, (c) 140°C, and (d) 160°C. All images are 1336 µm in height and 1670 µm in width.
The number density of observed bubbles during foaming process and final bubble density are presented in Figures 14 and 15, respectively. Figure 15 and comparison of Figure 10(b) with Figure 14 reveal that the nucleation rate and the final number density of observed bubbles are increased as the dissolved n-pentane content increases. It can be related to increasing saturation pressure with increasing available gas content, which decreases the critical bubble radius according to classical nucleation theory. Therefore, more bubbles have been passed from the nucleation barrier energy with increasing gas content. Figure 16 represents the average diameter of the observed bubble, and comparison of Figure 16 with Figure 12(b) demonstrate that the average bubbles diameter is decreased with increasing n-pentane content, which could be related to the increased bubble density, whereas the shell thickness around the bubbles would decrease.
Number density of observed bubbles versus unfoamed volume respect to time in Co2/n-pentane foaming process at nonpressurized condition and different foaming temperatures with 8% dissolved n-pentane content.
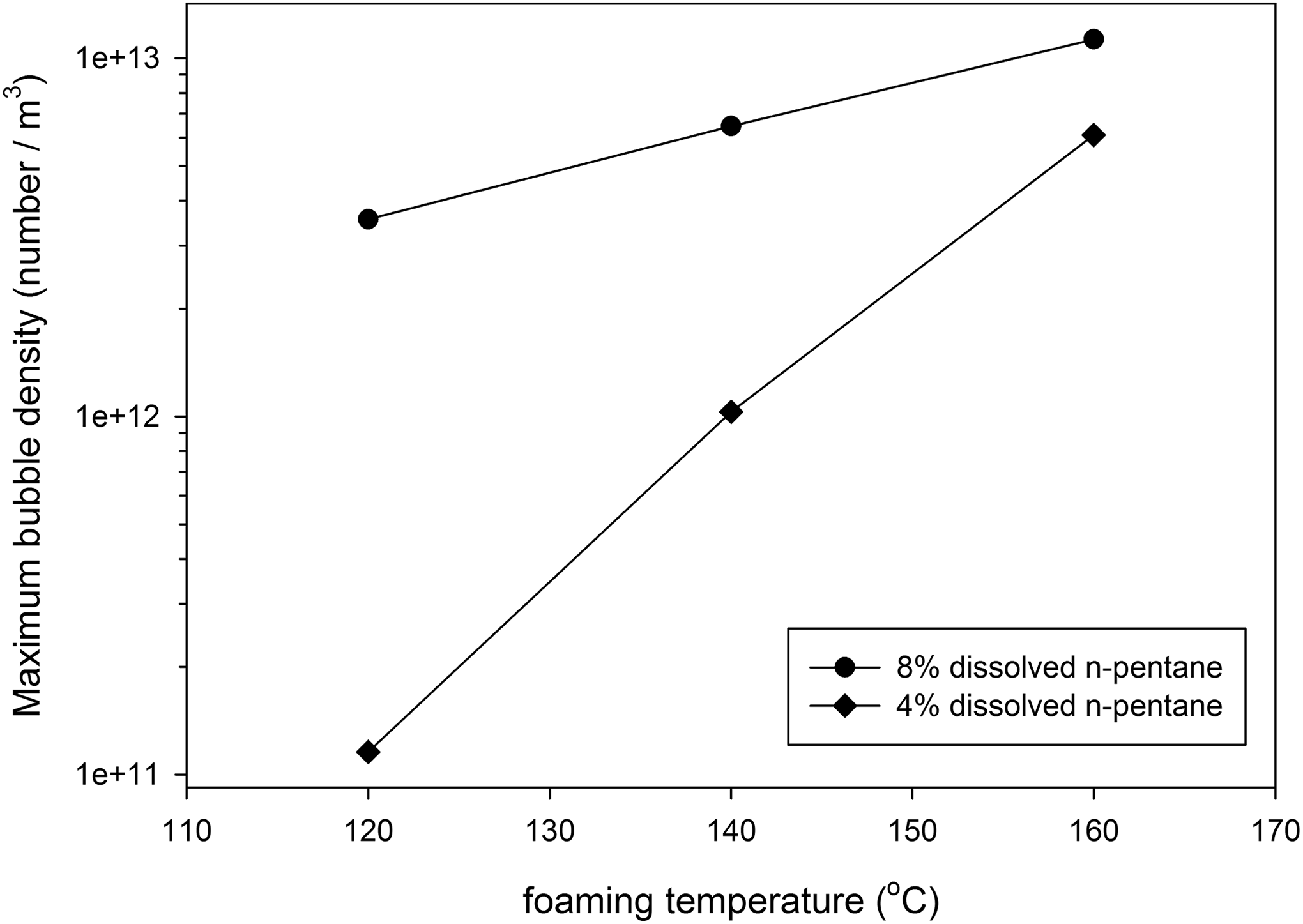
Maximum bubble density versus foaming temperature for Co2 (St53-MMA47) sample with 4% and 8% dissolved n-pentane content.
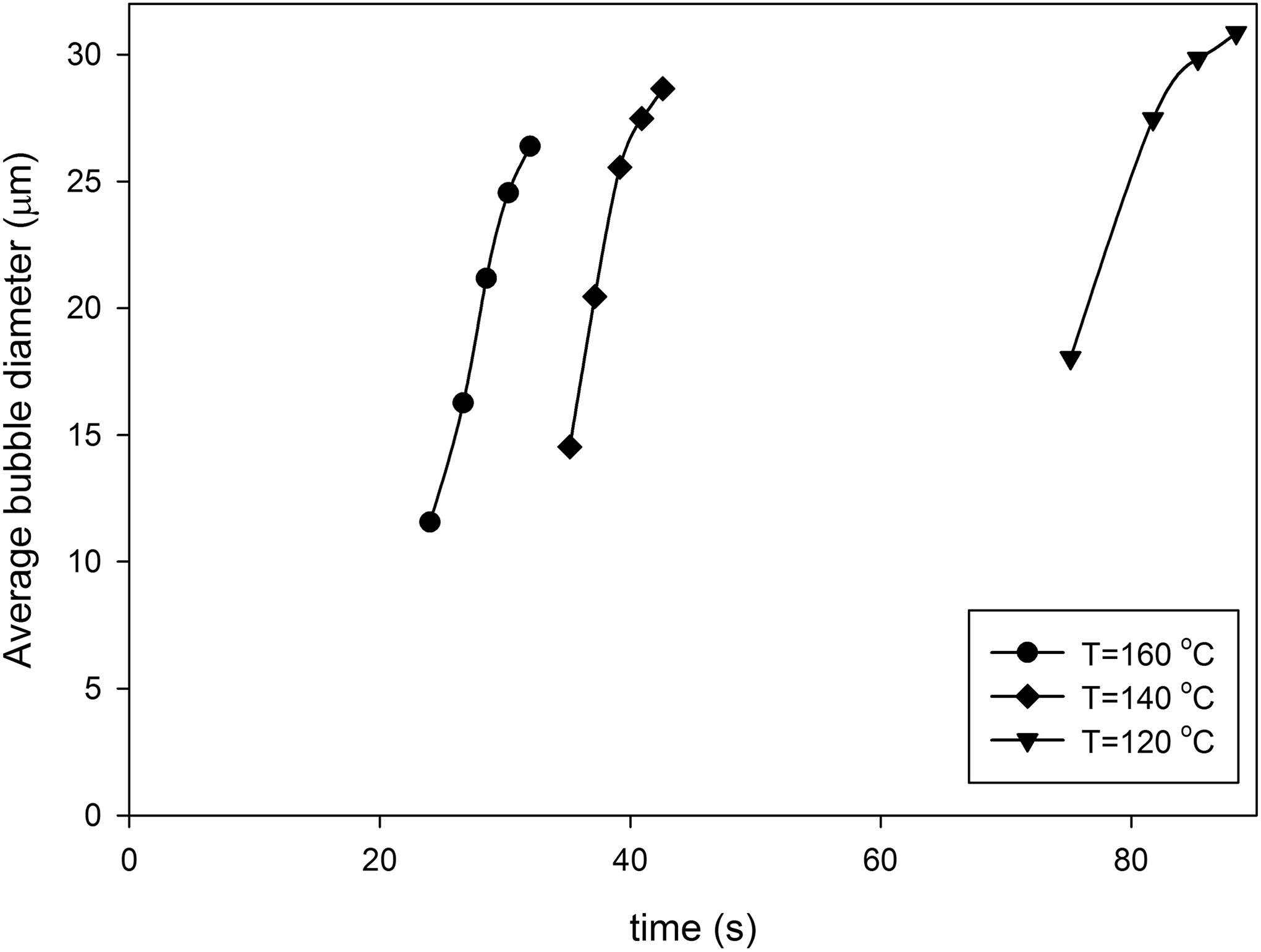
Average bubble diameter respect to time Co2/n-pentane foaming process at nonpressurized condition and different foaming temperatures with 8% dissolved n-pentane content.
Conclusion
In the present study, the foaming dynamics of the synthesized St-MMA copolymer/n-pentane foaming system was studied at nonpressurized condition. Synthesized copolymers were characterized using H-NMR spectroscopy and rheological measurements. It was found that the compositions of synthesized copolymers were close to the monomer feed ratios, and the copolymer matrix melt strength and average molecular weight were increased with increasing MMA content in copolymer compositions. The dissolved content of n-pentane in copolymers was determined at 100°C and 120° at different sorption pressures using designed batch foaming system. The dissolved content of n-pentane increased with increasing sorption pressure and declined with increasing temperature and MMA content. The effects of foaming temperature, copolymer composition, and n-pentane content on the foaming dynamics (observed bubble density and average bubble diameter) were investigated using obtained micrograph images through the foaming process. It was found that bubble nucleation and growth rate increased with increasing the foaming temperature and n-pentane content but decreased as MMA content increased.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.