Abstract
In this work, styrene/methyl methacrylate (St/MMA) copolymer particles were synthesized by suspension polymerization process with different copolymer compositions to study the visual batch foaming dynamics. The visualization system consisted of the self-sealing observation cell equipped to the pressure and temperature controller. The synthesized copolymer particles were impregnated by n-pentane, followed by recording of particle expansion. The cell structure of foams was studied by scanning electron microscopy. The effect of different foaming conditions on the expansion behavior of copolymers was examined. It was concluded that sorption pressure and temperature have contradictory effects on the foaming ratio of the synthesized copolymers at lower and higher sorption pressures, and the results were confirmed with the foams’ cell structure. Furthermore, it was shown that, at different temperatures and pressures, the expansion behavior change dramatically with increasing of MMA content in the copolymer.
Introduction
Thermoplastic foam products have become very popular in recent years. In this foam, the porous structure is achieved through the expansion of a blowing agent dissolved in a thermoplastic matrix by a batch or continuous process. 1 –7 A typical foaming process has three stages in which the first step is polymer/blowing agent dissolution, the second one is the cell nucleation, and the last stage is cell growth. 8 The first step is achieved by saturating a polymer with a blowing agent, typically above its melting or glass transition temperature (T g). This step is called the saturation period in which after that, with a rapid pressure drop or temperature increment, thermodynamic instability is introduced in the polymer/gas mixture and cell nucleation is started. Then, the gas diffuses from the polymer matrix into the cells and cell growth begins. The final properties of polymeric foams in batch systems are affected by the type and amount of the dissolved blowing agent as well as by foaming parameters, such as temperature, saturation pressure, time, and pressure release rate.
Gent and Tompkins developed a visual observation apparatus to observe foaming of the elastomer/carbon dioxide (CO2) mixture and reported the change in bubble radius with time. 9 Villamizar et al. used visual observation of structural foaming in injection molding of a mixture of polyethylene and chemical blowing agent. 10 Yoo and Han designed a visual observation apparatus and compared the experimental data and the numerical modeling of foaming behavior. 11 Actually, a few foaming experiments have focused on in situ visual foaming dynamics. Kentaro Taki et al. used visual observation of batch and continuous foaming using CO2 as the physical foaming agent to track the bubble nucleation and growth behaviors. 12 Leung et al. used the batch visualization data obtained from the experimental system and investigated the bubble growth dynamics and verified the results with theoretical model. 13 Salejova and Kosek studied the initial foaming stage through a batch visual system and discussed about the effects of different parameters on the foaming dynamics of polystyrene. 14 Recently, Taki analyzed the effect of pressure release rate on the bubble nucleation and growth via the batch foaming experiments, in which the flux of blowing agent from the polymer matrix to the bubble increased with increasing the pressure release rate. 15 Some researchers have previously analyzed bubble growth phenomena experimentally in different systems. 16 –20 Also solubility of different blowing agents in molten polymers was influenced by temperature and pressure. These parameters are the fundamental properties for all processes and were investigated by several authors. 21 –24
The suspension polymerization is commonly used for producing a wide variety of commercially important polymers such as polystyrene and its copolymers, polymethyl methacrylate (PMMA), polyvinyl acetate, and polyvinyl chloride. In our previous work, suspension copolymerization of styrene/methyl methacrylate (St/MMA) copolymer particles was conducted and the effects of different synthesis parameters were investigated. 1 Subsequently, the non-isothermal degradation kinetics of synthesized St/MMA copolymer foams was investigated under nitrogen and oxygen atmospheres. 25,26 But there were no appreciable studies about the foaming dynamics of this copolymer, thus we investigated the foaming condition in one composition of St/MMA copolymer in our previous work. 27 In this study, we synthesized St/MMA copolymer particles in three compositions by suspension polymerization process to investigate the foaming dynamics via visual batch foaming apparatus and examine the effect of different foaming conditions, like temperature, sorption pressure, and MMA content in copolymer on the foaming ratio of the synthesized copolymer. The rheological behavior of neat copolymers was also investigated to compare the effect of copolymer viscoelastic properties with different compositions on the foaming dynamics of the sample. Furthermore, the cellular structure of foams was investigated under different conditions by scanning electron microscopy (SEM) and the results were compared with foaming dynamics data.
Experimental
Materials
MMA and St monomers (Merck, Germany) were washed twice with 5 wt% aqueous solution of sodium hydroxide, followed by washing twice with distilled water to eliminate the inhibitor. Benzoyl peroxide (BPO; active content 75% and water 25%) as radical initiator with half-life (t 1/2) = 75 min at 90°C, poly(vinyl alcohol) (PVA) with a degree of hydrolysis of 72.5% and molecular weight of 72,000 g mol−1, hydroxyethyl cellulose (HEC), and poly(vinyl pyrolidone) (PVP, 360,000 g mol−1) as drop stabilizers, and potassium persulfate (K2S2O8) as an aqueous phase initiator were supplied by Merck. Tricalcium phosphate (TCP) and sodium dodcyl benzene sulfonate (DBSNa) as suspending agents were supplied by Fluka (Germany) and were used as received. n-Pentane (Merck) was used as blowing agent and polyethylene wax (PW-1% of monomers mixture weight) as a cell structure modifying aid were supplied by Tabriz Petrochemical Company (Iran). Silicone oil (Therm 420) was supplied by Lauda (Germany) and was used as received for heating the polymerization reactor.
Suspension copolymerization
The St/MMA copolymer particles were synthesized by a batch suspension copolymerization in a laboratory-scale reactor. A stainless steel reactor (1000 ml) with chilled water, silicone oil jacket, a nitrogen inlet and outlet for purging, and one baffle was used for copolymerization. The system was equipped with a proportional–integral–derivative controller to control the temperature inside the reactor by adjusting the temperature of circulating silicone oil. The required amount of PVA, TCP, PVP, HEC, DBSNa, and K2S2O8 were fed into the reactor containing pre-weighed amount of distilled deionized water. The mixture was stirred at ambient temperature under moderate stirring rate (280 r min−1) for at least a few hours to produce a homogeneous mixture. A monomer emulsion was made by adding a mixture of St and MMA monomers, free radical initiator (BPO), and cell structure improver (PW).
After the mixture was being stirred for 30 min, the content of monomer emulsion tank was transferred to the reactor. After all the reaction mixture was added and holding for 30 min, the reactor was heated to the reaction temperature (80°C) at a rate of 0.5°C min−1 and held for 7 h. After completion of the reaction, the reactor was cooled to 35°C at a rate of 5°C min−1. The reaction mixture was transferred out to a batch tank without washing residual material into the tank. The reaction mixture containing the St/MMA copolymer was acidified to a pH of 1.6–2.0 and after holding for 15 min, the residue was washed with water, dried, and sieved. More details of the copolymerization procedure have been published in our previous works. 1,27,28
Copolymer characterization
Particles composition was determined by proton nuclear magnetic resonance (1H NMR) spectroscopy. The particles were dissolved in deuterated chloroform, the spectra of the samples were obtained using a Bruker Avance DPX NMR spectrometer (Billerica, Massachusetts, USA) working at 500 MHz. The molar percentage of monomers incorporated to the particles was determined from the peak of OCH3 group (at 3.6 ppm) and the peaks correlated to the aromatic ring (between 6.5 ppm and 7.5 ppm). The peaks correlated to the CH2 and CH groups were used to verify the integration error. 27
Differential scanning calorimetry (DSC) endotherms were recorded using a Netzsch-DSC200 F3 Maia (Germany) for T g determination and was calibrated with indium standard at a heating rate of 1°C min−1 under nitrogen purge. All the experiments were carried out under nitrogen purge at a flow rate of 50 ml min−1. Five milligrams of the synthesized beads was taken in standard 40 µl aluminum crucibles and the temperature scanning was conducted from room temperature to 180°C at a heating rate of 1°C min−1. In DSC endotherms of St/MMA copolymers, in the curves, the first inflection of heat flow versus temperature was selected as a T g of the sample. 28
SEM (VEGA series, TESCAN, Czech Republic) was used to study the cell structure of foamed copolymer particles. All the samples were coated with a thin layer of gold before SEM examination.
Copolymer molecular weight was measured using Maxima 820 gel permeation chromatography (GPC) instrument (Waters Corporation; Milford, Massachusetts, USA).
The rheological measurements (using a Physica MCR 301 rheometer, Anton Paar, Austria) were conducted at temperature (T) = 160°C, angular frequencies (0.1–1000 s−1), and strain 1% under nitrogen atmosphere. The degradation of the samples was not suspected by measurement of viscosity as a function of time. By performing strain sweeps on the disentangled and fully entangled samples, the linear viscoelastic region was obtained. Due to high sample stiffness and ease of loading of high-molecular-weight samples, parallel plates were used instead of the cone and plate geometry with a disk diameter of 25 mm and a sample thickness of 1 mm. Rheological samples were compression molded at temperature 160°C and pressure approximately 1000 psi.
Foaming procedure
In this study, we designed a batch foaming dynamics examination system (Figure 1). This visual batch foaming system consisted of the self-sealing observation cell equipped with two glass windows and a Bell stereo microscope (STMDLX-T-POL (Italy), maximum magnification: 65×) attached with a Bell high-speed digital camera (DV 1300, speed: 12 frame s−1) as well as pressure and digital temperature controllers. The St/MMA copolymer particles were placed into the observation chamber on the metal grid between the two glass windows that were illuminated by a light source either from the top or from the bottom and were heated with electrical elements. The digital camera was connected to the computer equipped with digital image processing software and all changes in the expanding particle size were detected. The setting of the microscope was kept constant during the experiments. The synthesized copolymer particles impregnated by n-pentane and then foaming dynamics were recorded as the sequence of images of expanding particles located in the pressure cell. The selected sorption pressure for each experiment was supplied by the heating of the n-pentane inside of its flask by means of an element. There are two pressure gauges, one in the board of n-pentane flask in which when we heated the n-pentane, its vapor pressure increased and was shown by that gauge and the second gauge in observation cell entrance that shows the exact pressure of n-pentane. After reaching to a suitable pressure, we opened the valve between chamber and flask (valve 1) and the vapor of n-pentane went inside of the observation cell. The temperature inside of the cell was kept constant by a temperature controller. In this state, when the selected vapor pressure of n-pentane was shown by the second gauge, the valve 2 was closed and the sorption period was started. After this period of time, the valve 3 was opened and the expansion of copolymer particle was recorded in the atmospheric condition. The sealing of our observation cell is strongly isolated from the outside and previously this one was checked by nitrogen gas (valves 4 and 5). We did not see any leakage in the observation cell. But even if the pressure inside the cell slightly decreased, we could open the valves 1 and 2 at the same time and let the vapor of n-pentane stream into the cell during the experiments.
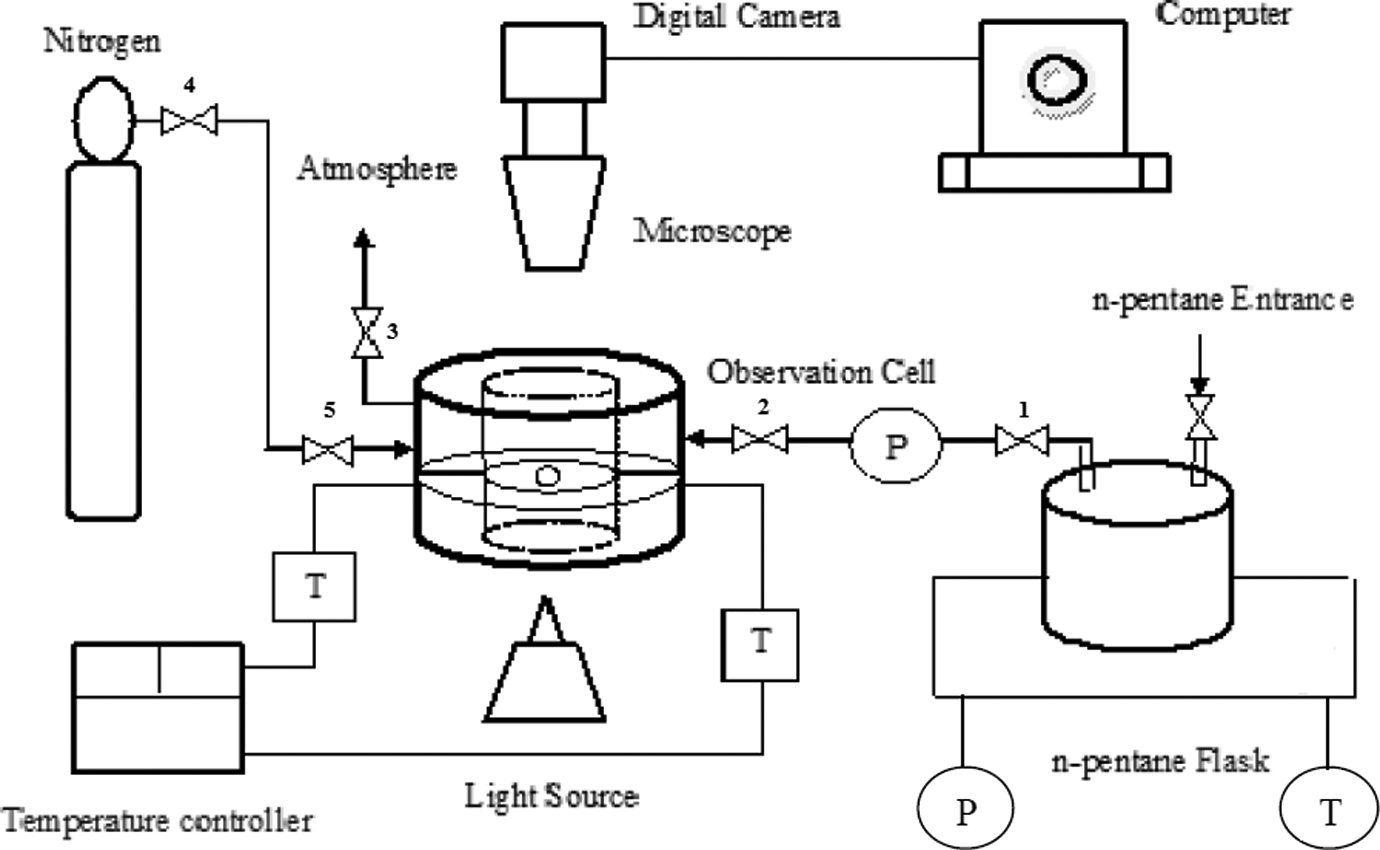
The schematic view of the designed visual batch foaming system.
All the experiments were run at constant temperature of the observation cell. The St/MMA copolymer particle was inserted into the observation cell and heated up to the required temperature. Then the sorption of n-pentane in particles was conducted at different pressures of n-pentane vapor for a specified period of sorption time (t impreg) and finally the pressure was quickly released and the foaming procedure of the particles was recorded at regular time intervals of 10 s. In these experiments, the foaming ratio (the ratio of particle diameter in each time to the initial particle diameter) was recorded versus time.
Results and discussion
Copolymer composition and structure
The 1H NMR spectra of the St/MMA copolymers were taken and the different copolymer compositions were determined as St/MMA:70/30 (wt/wt), St/MMA:53/47 (wt/wt), and St/MMA:30/70 (wt/wt). The peaks at 7.1 and 3.6 ppm correspond to –C6H5 and –OCH3 groups, respectively. The peak at 2.8 ppm, absent in the 1H NMR spectra of both relevant homopolymers, 29 was observed in the copolymers spectrum. This has been assigned to the –OCH3 group of MMA bonded to St sequence. 30 Opresnik et al. 31 also showed that this peak could be much more clearly seen in the 1H NMR spectra of a St/MMA random copolymer rather than that of a St/MMA block copolymer.
DSC endotherms of St/MMA:70/30 (wt/wt), St/MMA:53/47 (wt/wt), and St/MMA:30/70 (wt/wt) copolymers are shown in Figure 2. For comparison, also the polystyrene (PS) and PMMA endotherms are added in this figure.
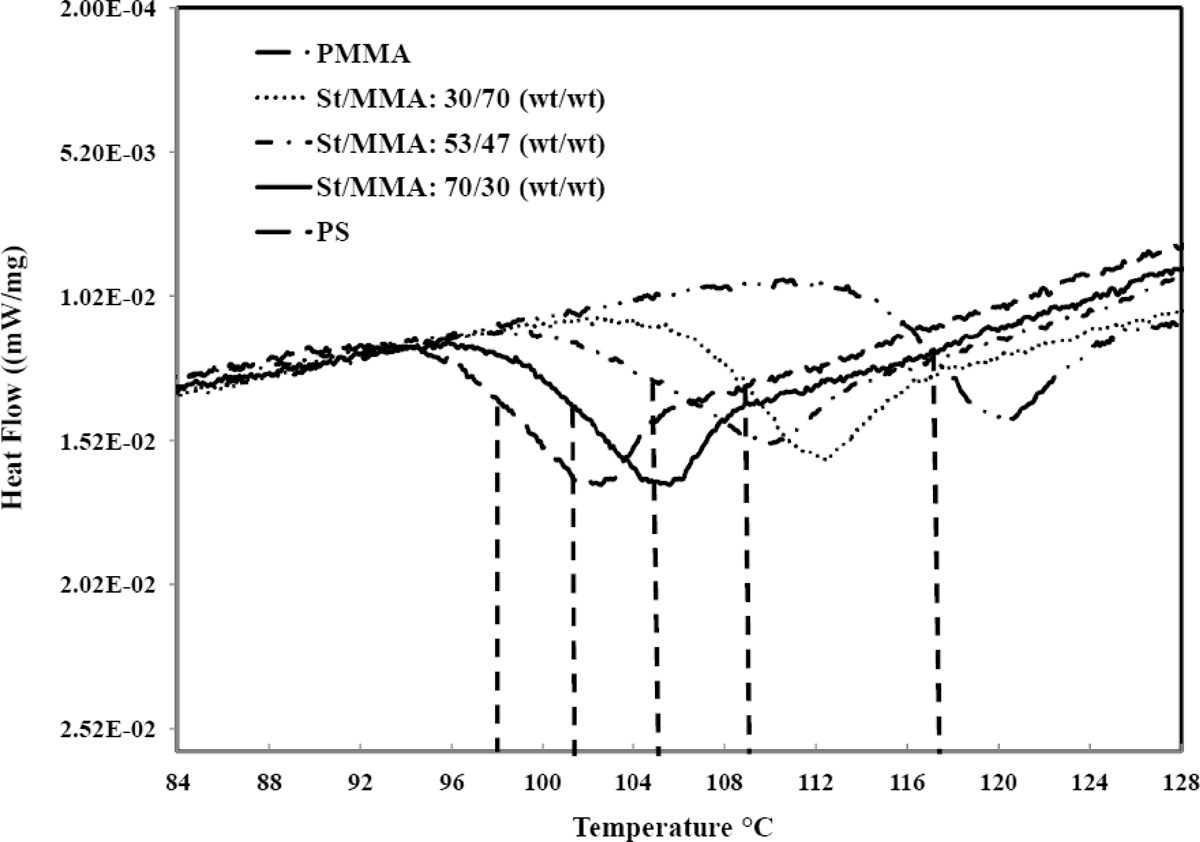
The DSC endotherms of St/MMA copolymers and neat polymers.
It is clear that the sorption of n-pentane could decrease the T g of samples due to the plasticizing effect of n-pentane. Thus in this study the amount of reduction in T g for pentane-impregnated samples before expansion was also measured. The reduced T g values for n-pentane-impregnated samples are as follows: PS: 86°C, St70/MMA30: 89°C, St53/MMA47: 92°C, St30/MMA70: 95°C, and PMMA: 106°C. When the content of St in copolymer is high, due to n-pentane high tendency to St, the sorption of n-pentane in copolymer is increased and consequently T g of sample is greatly decreased. So the reduced T g values are almost around 10–15°C lower than their values for neat samples (without n-pentane) depending on their St content in copolymer.
Average molecular weights (number-average molecular weight and weight-average molecular weight) and polydispersity index measured from GPC for copolymers and neat polymers are listed in Table 1.
Molecular weights and PDI of St/MMA copolymers and neat polymers.
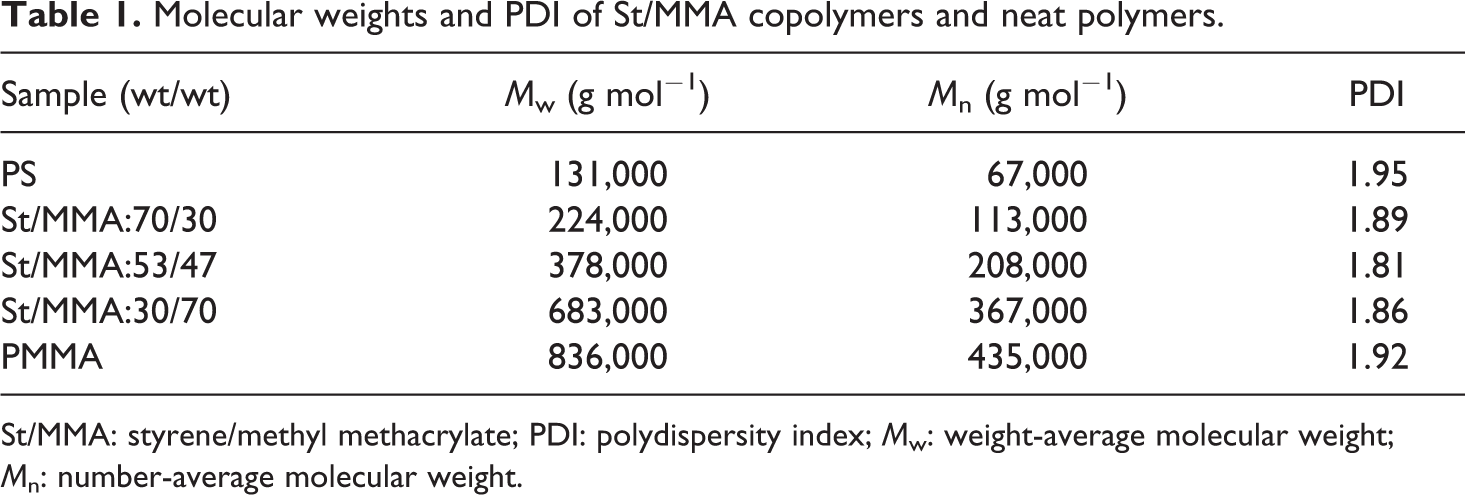
St/MMA: styrene/methyl methacrylate; PDI: polydispersity index; M w: weight-average molecular weight; M n: number-average molecular weight.
Variation of rheological properties (complex viscosity and storage modulus) versus frequency for St/MMA copolymer with different compositions at T = 160°C is shown in Figure 3.
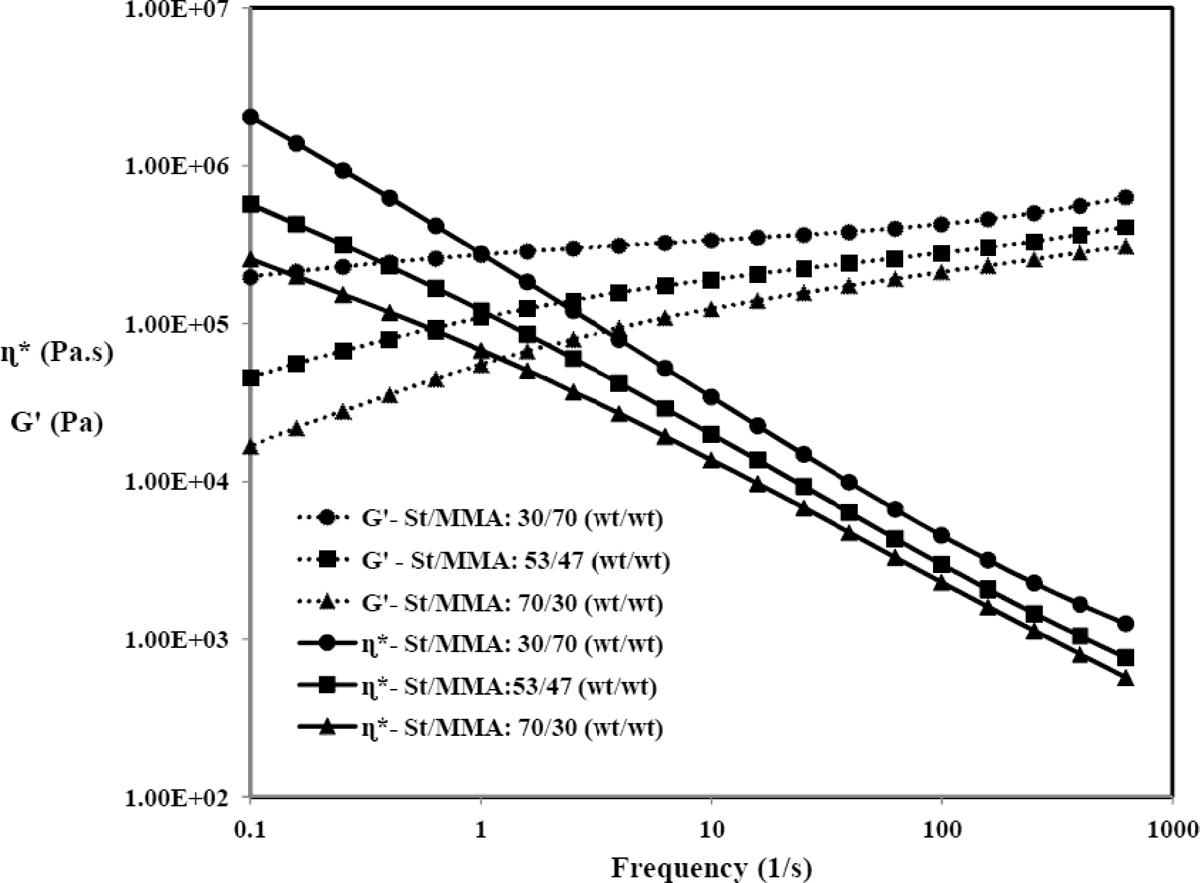
Variation of complex viscosity and storage modulus versus frequency for St/MMA copolymers at T = 160°C.
As it is clear, the complex viscosity and storage modulus of copolymer increase with MMA content increment. These behaviors are attributed to the molecular weight and the complex viscosity and storage modulus (elasticity) increment with increase in MMA concentration in copolymer. This behavior will dramatically change the foaming dynamics in expansion of St/MMA:30/70 (wt/wt) copolymer particles. Due to some technical restrictions the rheological behavior of n-pentane-impregnated samples could not be measured in this study and consequently the rheological data at T = 160°C were used just for comparison of melt viscosity and dynamic moduli of different copolymers.
Foaming experiments
In this study, the effect of different expansion conditions like temperature, sorption time, sorption pressure, and copolymer composition on the foaming dynamics of St/MMA copolymer was investigated. In all experiments, the observation cell was charged by n-pentane in determined time of pentane sorption into the copolymer particles with size of 0.17–0.24 mm. The same size was used in all foaming experiments to eliminate the effect of initial particle size on the foaming dynamics. After sorption period and rapid pressure release, the particles were expanded in atmospheric pressure on the timescale of several minutes and their size was reached to a maximum size. In all experiments, the foaming and sorption temperatures were the same. The foaming procedures of St/MMA copolymer particles with different compositions are shown in Figure 4. In these experiments, the selected sorption time, pressure, and temperature for St/MMA:70/30 (wt/wt) and St/MMA:53/47 (wt/wt) were 30 min, 3 bar, and 85°C, respectively, but these parameters for St/MMA:30/70 (wt/wt) were 60 min, 6 bar, and 120°C because for this copolymer no expansion was observed at lower temperatures and pressures. These conditions are the base state for observing the expansion process for each copolymer. Primarily it was tried to compare the foaming dynamics of all copolymers at the same condition. But by considering the different compositions of St and MMA in samples, the results of foaming dynamics, pentane content, and viscoelastic properties were being affected. So selecting the same condition to compare the foaming dynamics for all samples was not possible. Therefore, we chose different conditions (T, pressure (p), and sorption time (t sorpt)) for comparison, so the expansion in particles was not observed in temperatures and pressures lower than these base conditions. On the other hand, the base conditions are the primary and suitable conditions that the foaming of all samples happening and going on without any collapsing states.

The foaming sequence of St/MMA copolymer particles after rapid pressure release at 85°C for (a) St/MMA:70/30 (wt/wt) and (b) St/MMA:53/47 (wt/wt) and at 120°C for (c) St/MMA:30/70 (wt/wt), scale bar: 0.26 mm. St/MMA: styrene/methyl methacrylate.
As it is shown in Figure 4, it is clear that the expansion behavior for St/MMA:30/70 (wt/wt) is more uniform than two other compositions. This behavior is attributed to the higher content of MMA units in this copolymer, which leads to the higher complex viscosity and storage modulus (elasticity) and consequently more uniform expansion process in St/MMA:30/70 (wt/wt) copolymer particles compared to the other copolymer compositions. The expansion of particles is nearly complete after 130 s, regardless of the sorption time preceding the foaming. The attempts to foam the impregnated particle below 80°C and pressure of 3 bar, for St/MMA:70/30 (wt/wt) and St/MMA:53/47 (wt/wt) and below 120°C and pressure of 6 bar for St/MMA:30/70 (wt/wt), by rapid pressure release were not successful.
The SEM images of the copolymers expanded in the base conditions are shown in Figure 5. For St/MMA:53/47 (wt/wt), the cellular structure is observed in particles’ shell. For St30/MMA70 the cellular structures are so tiny compared to other samples in the base condition, which can be attributed to the higher molecular weight, complex viscosity, and storage modulus (elasticity) of this copolymer (see Figure 3). Furthermore, the expansion behavior of these copolymers in the base conditions is shown in Figure 6, in which with increasing of the MMA content in copolymers, the final values of foaming ratio were decreased. Confirming of light microscopy images shown in Figure 4, it is clear that the final foaming ratio for the St30/MMA70 particle in Figure 6 is also lower than two other copolymers.
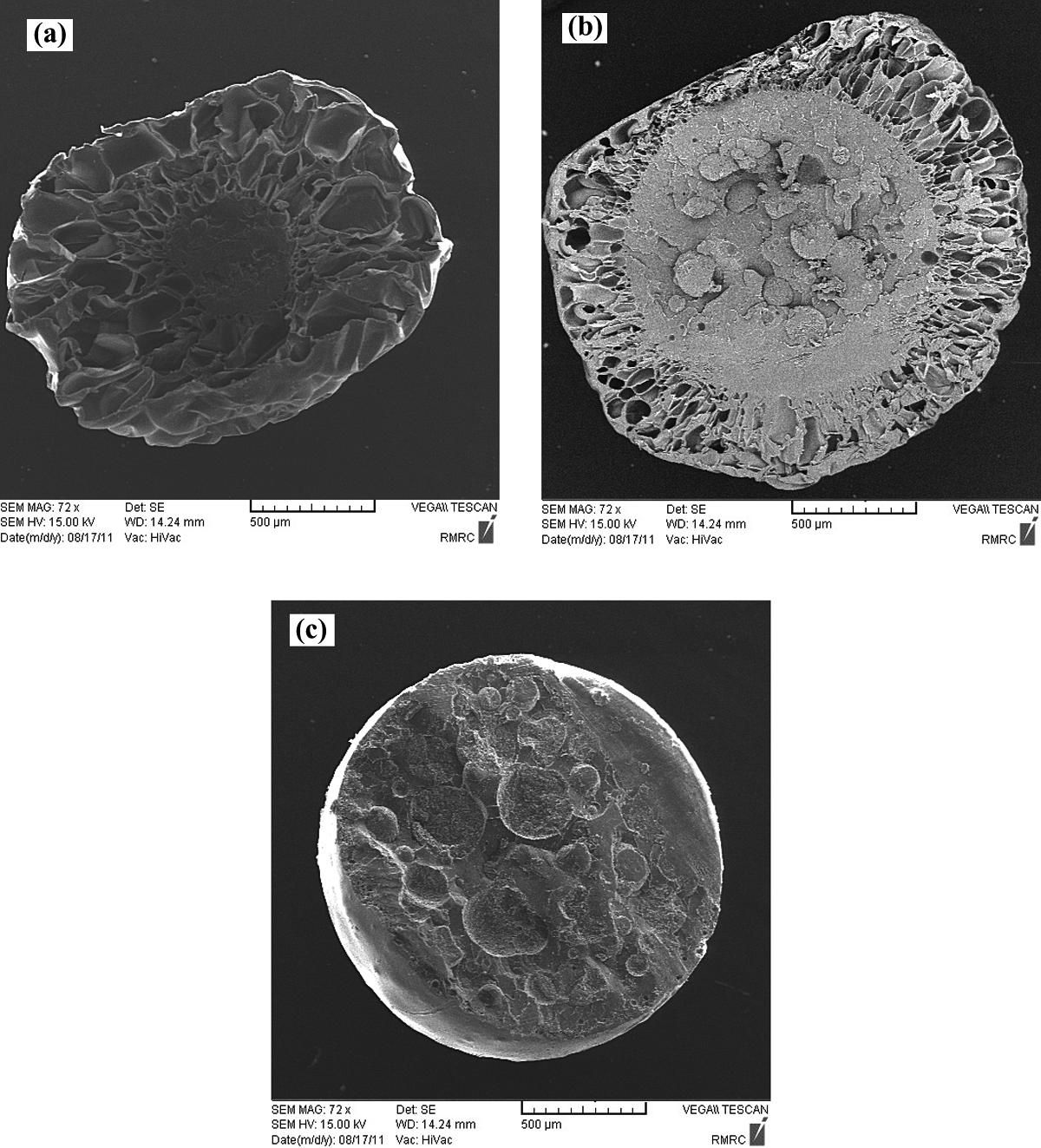
SEM images of the St/MMA copolymers: (a) St/MMA:70/30 (wt/wt), (b) St/MMA:53/47 (wt/wt), and (c) St/MMA:30/70 (wt/wt) in the base conditions. SEM: scanning electron microscopy; St/MMA: styrene/methyl methacrylate.
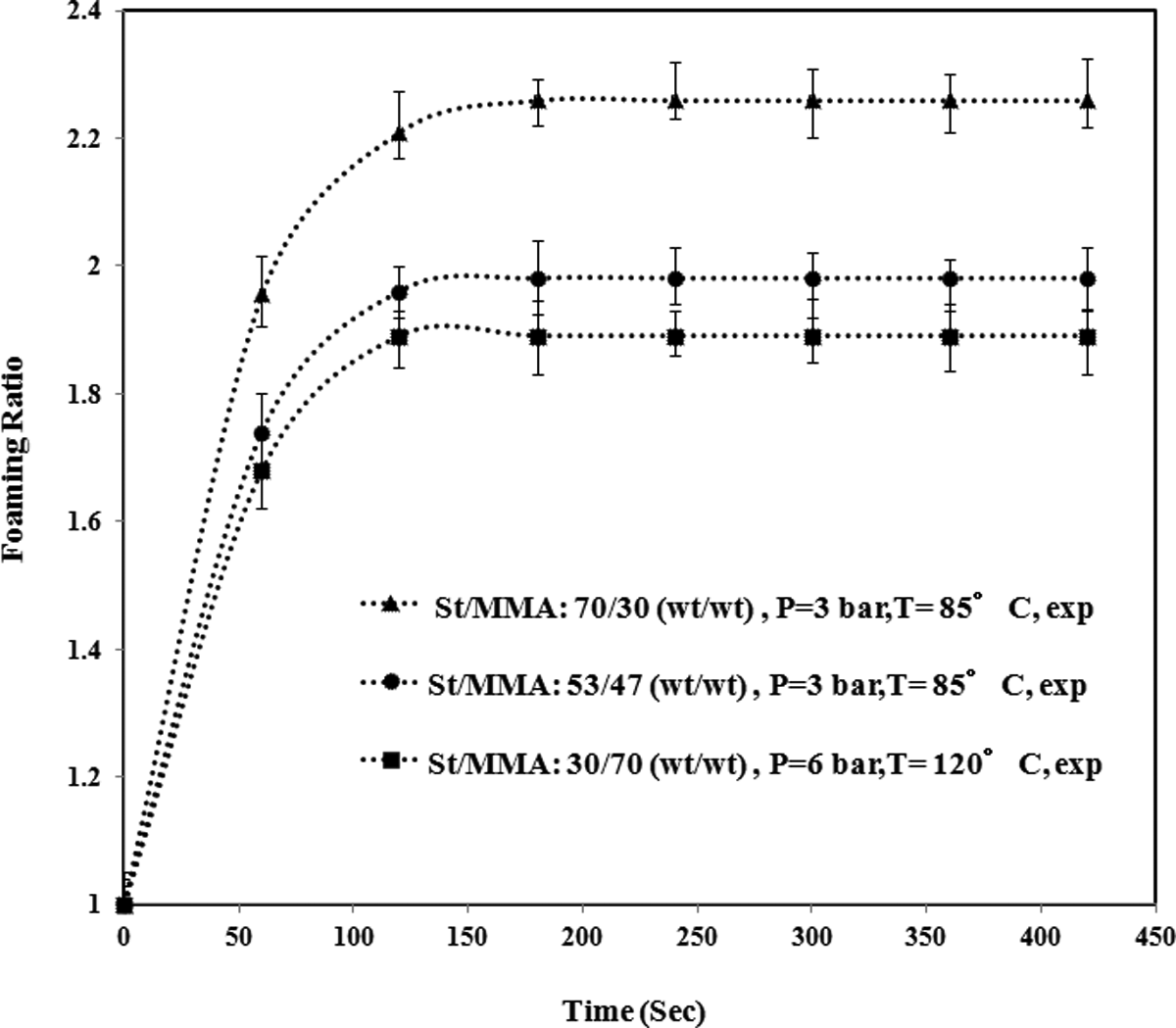
The expansion behavior of three copolymers at the base conditions.
For St/MMA:70/30 (wt/wt) expanded copolymer, it is clear that the cellular structure is constituted of more than other copolymers, so in this case, the selected sorption time and temperature are suitable.
The effect of sorption time on the cellular structure of St/MMA:53/47 (wt/wt) and St/MMA:30/70 (wt/wt) copolymers is shown in Figures 5(b), 7, and 8, respectively. With increase in the sorption time for both copolymers, the cellular structures are extended, which can be confirmed that with increase in the n-pentane sorption time in samples the saturation state as a key parameter in cellular structure constitution could be reached in these samples.
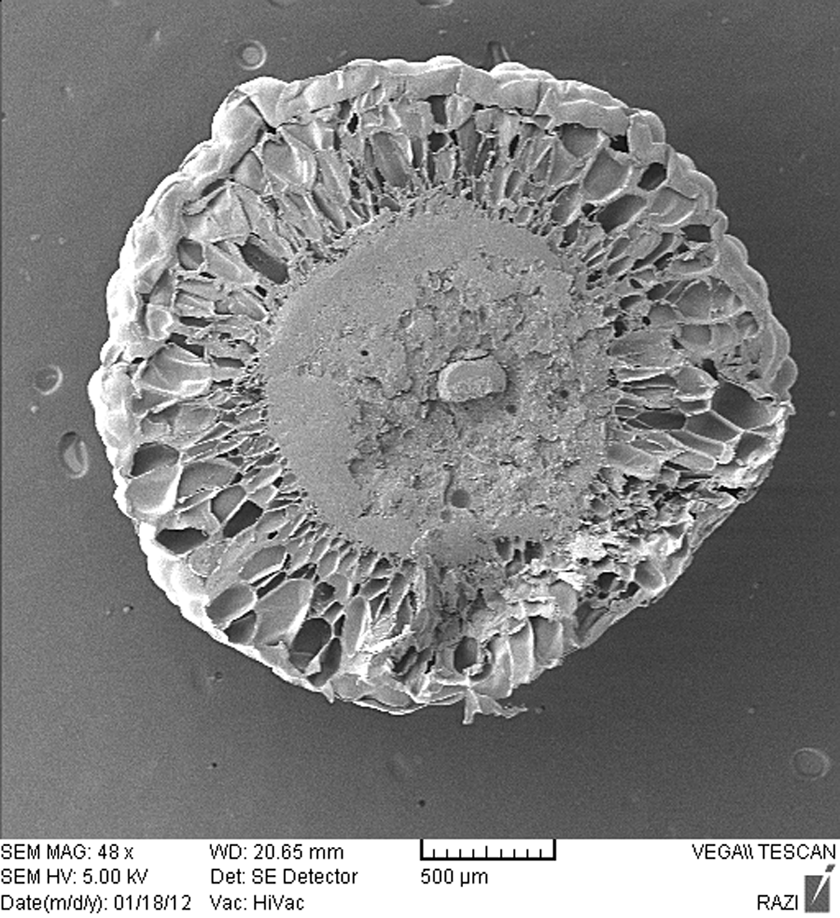
SEM image of the St/MMA:53/47 (wt/wt) copolymer at T = 85°C, p = 3 bar, and t sorpt = 120 min. SEM: scanning electron microscopy; St/MMA: styrene/methyl methacrylate.
SEM images of the St/MMA:30/70 (wt/wt) copolymer at T = 120°C, p = 8 bar: (a) t sorpt = 60 min and (b) t sorpt = 180 min. SEM: scanning electron microscopy; St/MMA: styrene/methyl methacrylate.
The selected conditions for SEM micrograph samples (Figures 7 and 8) are T = 85°C, p = 3 bar, t sorpt = 30 and 120 min for St/MMA:53/47 (wt/wt) and T = 120°C, p = 8 bar, t sorpt = 120 and 180 min for St/MMA:30/70 (wt/wt) copolymer. As it is clear in SEM images, with increase in the sorption time, more cellular structures are observed. For St/MMA:30/70 (wt/wt) copolymer, when the sorption pressure is increased from the base condition (6 bar) to 8 bar, more cells are observed because in high sorption pressures the higher amount of n-pentane is diffused into the nucleated cells and more cells grow up during expansion period. Therefore, with increment of MMA content in copolymer in lower pressures, the diffusion of n-pentane into the copolymer is more restricted and consequently a tiny cellular structure is observed.
Actually, while copolymer particles are charged with n-pentane at different pressures, two distinct phenomena are dominant. The first one is related to pentane sorption and diffusion in St/MMA copolymer matrix and the second one is attributed to the diffusion of great amounts of pentane into the nucleated cells in softened copolymer matrix. It is concluded that the changes in the mentioned phenomena are exactly related to the applied sorption pressure, temperature, and time.
The variation of foaming ratio versus time at different temperatures and pressures for all St/MMA copolymer compositions is shown in Figures 9 to 11.
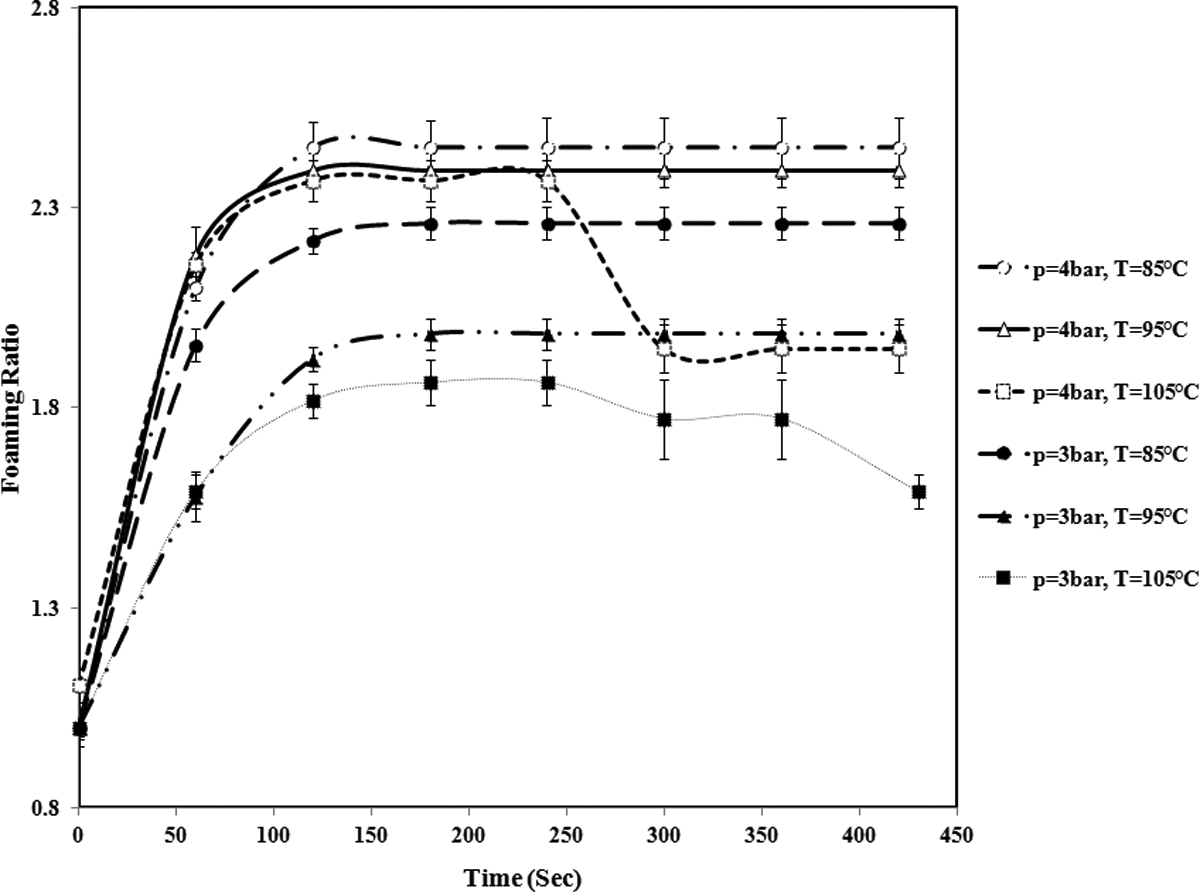
The variation of foaming ratio of St/MMA:70/30 (wt/wt) copolymer particle versus time at different temperatures and pressures (The lines are used to guide eyes.). St/MMA: styrene/methyl methacrylate.
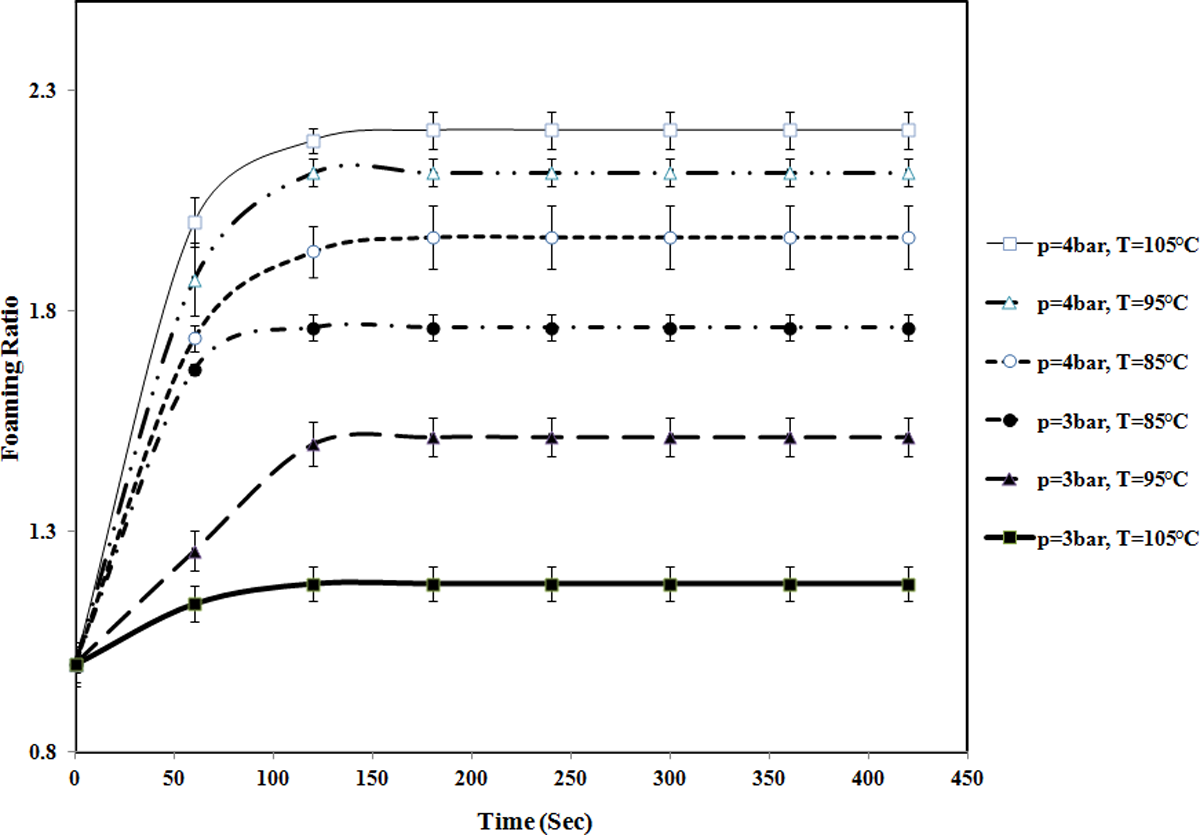
The variation of foaming ratio of St/MMA: 53/47 (wt/wt) copolymer particle versus time at different temperatures and pressures. (The lines are used to guide eyes.). St/MMA: styrene/methyl methacrylate.
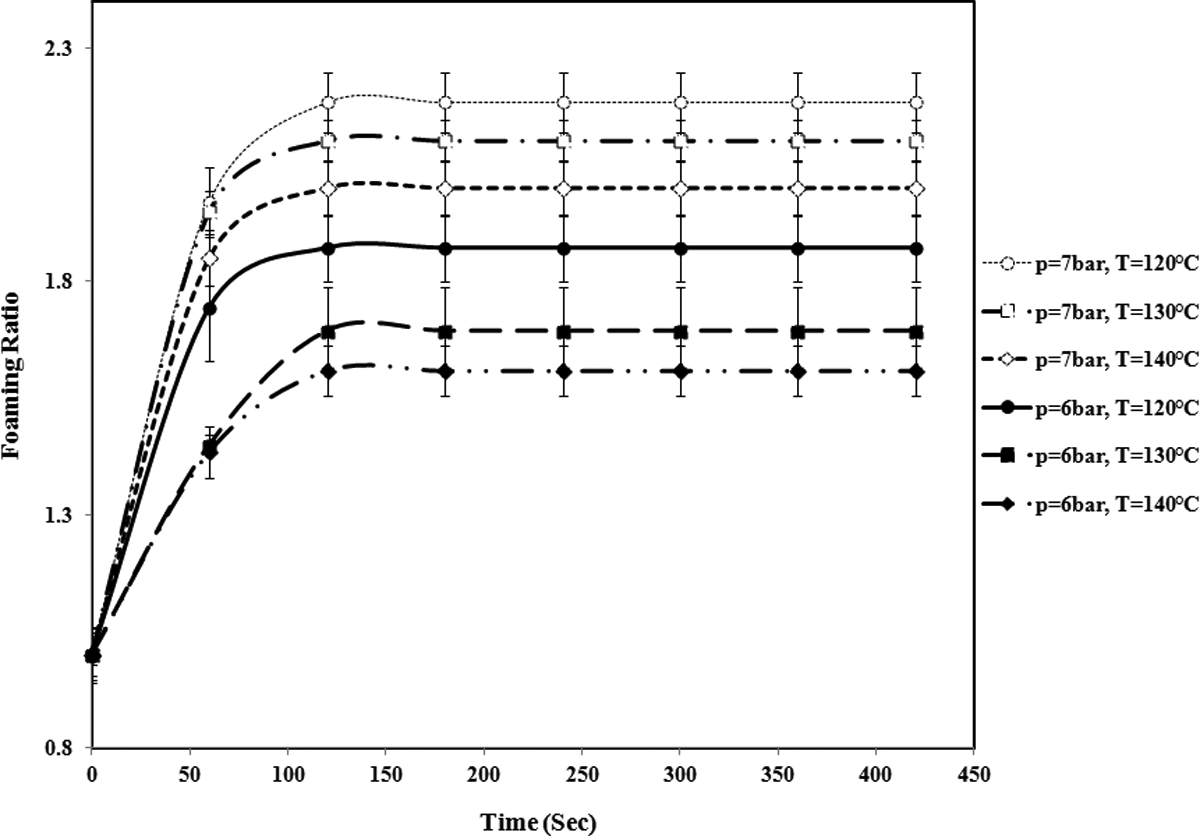
The variation of foaming ratio of St/MMA: 30/70 (wt/wt) copolymer particle versus time at different temperatures and pressures. (The lines are used to guide eyes.). St/MMA: styrene/methyl methacrylate.
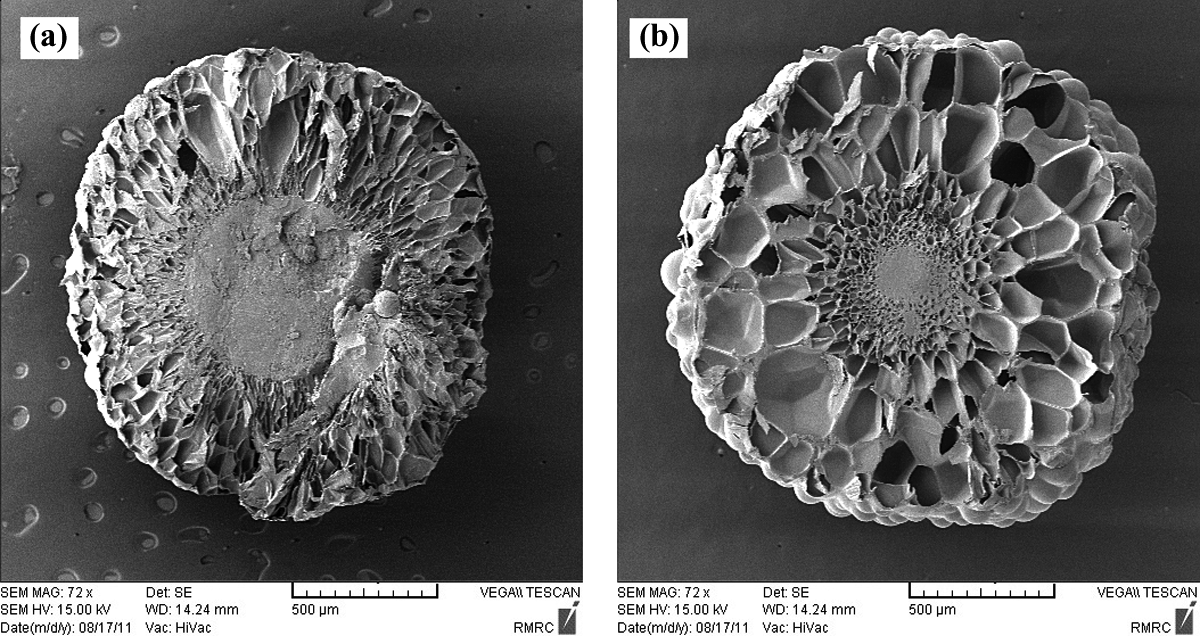
As it is observed in these figures, the foaming ratio of St/MMA copolymer particles is increased with increasing pressure in all compositions and temperatures. At higher pressures, the n-pentane sorption in the St/MMA copolymer matrix and diffusion into the cells are improved, and at the same time, the pressure difference as a driving force for expansion of particles is increased. The foaming dynamics behavior of St/MMA:30/70 (wt/wt) copolymer at T = 120°C, t sorpt = 60 min, and p = 6 and 8 bar is confirmed by SEM images in Figures 5(c) and 8(a), in which with sorption pressure increment, more cells are observed in this copolymer.
The temperature dependency of foaming ratio at different copolymer compositions is contradictory. As it is shown in Figures 9 and 10, for St/MMA:70/30 (wt/wt) and St/MMA:53/47 (wt/wt) copolymers, while the sorption pressure is lower than 4 bar, with rising temperature, the n-pentane sorption mostly decreases and a significant fraction of n-pentane diffuses out of the copolymer matrix and ultimately small amount of n-pentane as blowing agent enters into the nucleated cells and contributes to the expansion process. So in low pressures, the temperature dependency of foaming dynamics of St/MMA:70/30 (wt/wt) and St/MMA:53/47 (wt/wt) copolymers is the same.
For instance, in St/MMA:70/30 (wt/wt) copolymer, as it is obvious from SEM images in Figures 5(a) and 12, with rising temperature from 85°C to 105°C, at the same sorption pressure (p = 3 bar) and time (t sorpt = 30 min), the cell structure is disappeared, which can be attributed to n-pentane sorption decrement and its less contribution in cell growth with rising temperature. As well as for St/MMA:53/47 (wt/wt) copolymer, this pressure (p < 4 bar) for n-pentane diffusion is not high enough, consequently, the expansion process in pressures lower than 4 bar is slightly affected by pentane limited sorption and diffusion. These conditions lead to the less foaming ratio for St/MMA:70/30 (wt/wt) and St/MMA:53/47 (wt/wt) samples at low pressures and high temperatures.
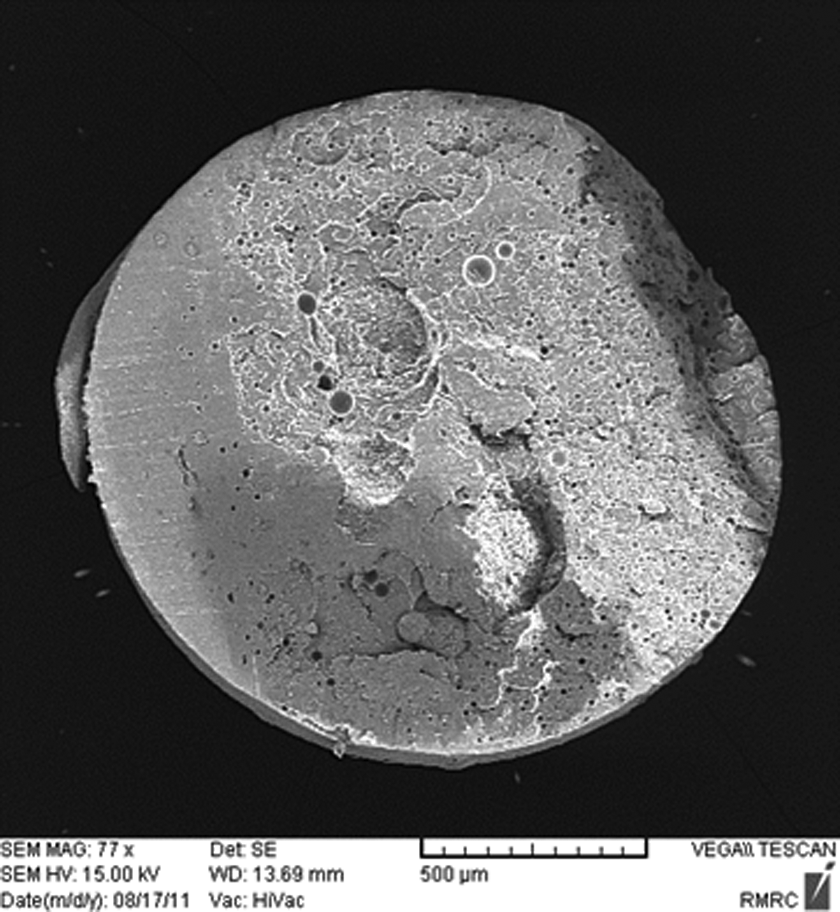
SEM image of the St/MMA:70/30 (wt/wt) copolymer at p = 3 bar, t sorpt = 30 min, and T = 105°C. SEM: scanning electron microscopy; St/MMA: styrene/methyl methacrylate.
While the pressure is going up to greater than 4 bar, the foaming ratio increment with rising temperature is observed for St/MMA:53/47 (wt/wt) particles. In this case, the diffusion of n-pentane into the copolymer matrix is conducted and with rapid pressure release at the early stage of foaming process due to diffusion of great amounts of pentane into the nucleated cells in softened copolymer matrix, the volume of the particles increases. The construction of more cellular structures is observed in SEM images of St/MMA:53/47 (wt/wt) copolymer expanded in p = 6 bar and t sorpt = 120 min (Figure 13). In this copolymer, whereas the complex viscosity and elasticity are high enough to keep the diffused n-pentane inside the cells at high pressures (p = 6 bar), more cells are observed with temperature increment. With increase in the temperature, the sorption amount of n-pentane decreases, and it tends to run out of the matrix, but due to high enough amount of copolymer complex viscosity and elasticity, it diffuses into the nucleated cell and enlarges them and finally the volume of the particles are increased as well. As it is clear, in high pressures, with rising temperature to 105°C, the driving force for expansion is increased and the cell walls are thin, thus the particles slightly collapse at high pressures and temperatures. Furthermore, the sorption time is high enough for diffusing n-pentane into the matrix. This observation revealed that the temperature is not the unique determining parameter for collapsing of the particles and if the sorption pressure and time are selected high enough, the collapsing of the cells can be observed in all copolymer compositions as well.
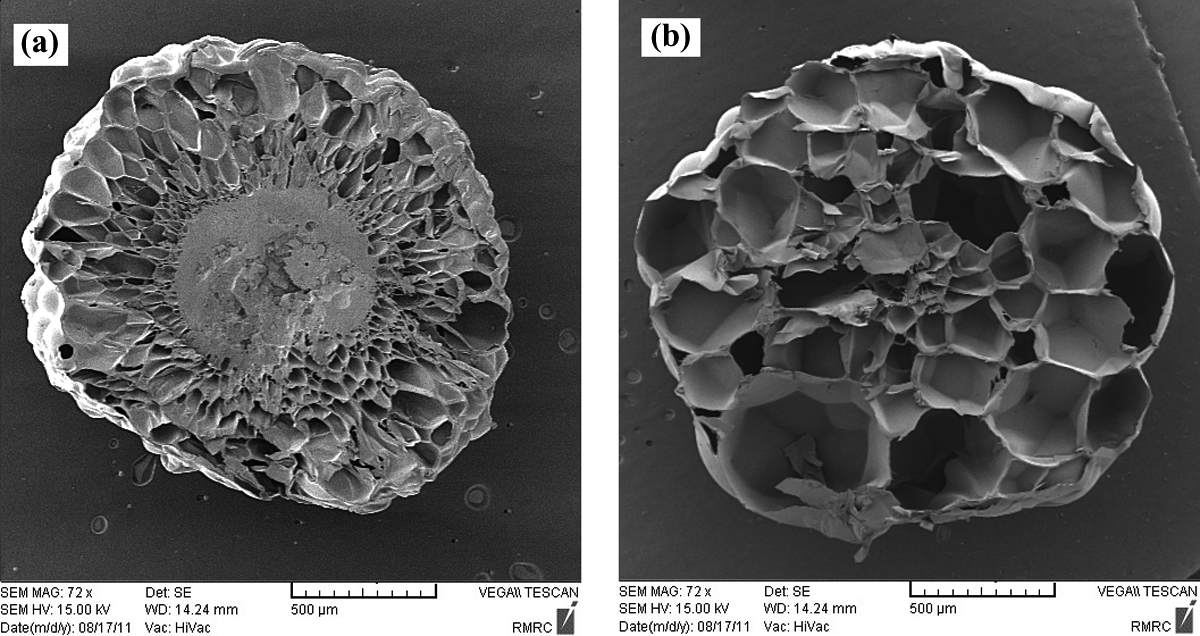
SEM images of the St/MMA:53/47 (wt/wt) copolymer at p = 6 bar, t sorpt = 120 min: (a) T = 85°C and (b) T = 105°C. SEM: scanning electron microscopy; St/MMA: styrene/methyl methacrylate.
For St/MMA:70/30 (wt/wt) copolymer, because of the lower complex viscosity and storage modulus (elasticity) and molecular weight compared to St/MMA: 53/47 (wt/wt), the conditions are as the same as lower pressures (p < 4 bar). In this composition, it seems that in pressures higher than 3 bar, the copolymer complex viscosity and storage modulus (elasticity) are not high enough to keep the n-pentane inside the cells and n-pentane cannot contribute to expansion process and diffuses out of the softened matrix. As well as due to the lower complex viscosity and storage modulus (elasticity) at T = 105°C and p = 3 and 4 bar, the expansion behavior changes after 220 s, and as it is shown in Figure 9, collapsing of the cells is observed. In Figure 14, the collapsing of St/MMA:70/30 (wt/wt) at p = 3 and T = 105°C is shown. It is clear that the final expanded particle size is decreased with time evolution.

The foaming sequence of St/MMA:70/30 (wt/wt) copolymer particles after rapid pressure release at 105°C and p = 3 bar, scale bar: 0.26 mm. SEM: scanning electron microscopy; St/MMA: styrene/methyl methacrylate.
In St/MMA:30/70 (wt/wt) copolymer (Figure 11), the foaming conditions dramatically change in which the minimum sorption temperature and pressure for expansion are 120°C and 6 bar. In this composition, due to high content of MMA units in copolymer and high amount of complex viscosity and storage modulus (elasticity) (see Figure 3), the expansion behavior changes because the applied sorption pressures and time are not high enough for n-pentane to dissolve and diffuse into the bulk of softened copolymer matrix and only diffuses to the particles shell. Upon expansion of this copolymer with increasing temperature, n-pentane goes out of the shell without any contribution to expansion and consequently the foaming ratio of the particles decreases.
As it is clear in Figures 9 and 10, the maximum foaming ratio for St/ MMA:53/47 (wt/wt) is significantly lower than St/MMA:70/30 (wt/wt) at all temperatures and pressures. With increment in MMA content in the copolymer, due to the higher tendency of n-pentane to St units than MMA units in copolymers, the diffusion of n-pentane into the bulk of copolymer decreases. Therefore, the cellular structure decreases and is limited to the shell of the copolymer particles, which leads to lower foaming ratio in these samples.
In the case of St/MMA 53/47, three different parameters are in competition with each other. The first one is the sorption pressure, the second one is temperature, and third one is copolymer composition. As well as due to nonpolarity of n-pentane and St groups, n-pentane is dissolved in St units of copolymer. When the sorption pressure is lower than 4 bar, the driving force for pentane solubility is not good enough and the less amount of pentane could dissolve in St units of copolymer. In this condition, with temperature increment, the dissolved pentane molecules run out of the copolymer and the amount of n-pentane is decreased. When the sorption pressure is higher than 4 bar, the driving force for pentane solubility is high enough and the higher amount of pentane dissolve in St units of copolymer. In this condition, with temperature increment, the MMA units may not allow n-pentane molecules to go out of copolymer matrix and keep the diffused pentane inside the sample. Also with temperature increment, the diffusivity of pentane molecules is increased and they can diffuse more and more to the bulk of the copolymer matrix. The construction of cellular structures is different with temperature increment.
The expansion rates for St/MMA:53/47 (wt/wt) and St/MMA:70/30 (wt/wt), at different pressures and T = 95°C, are compared in Figure 15. It is shown that with increment in the MMA content at constant temperature and pressure, the rate of expansion is reduced. Because with rising of MMA content in the copolymer, pentane diffusion into the cells is limited due to higher polarity of MMA units compared to St units and expansion of particles is conducted more slowly. For St/MMA:30/70 copolymer, at p = 3 and 4 bar and T = 95°C, no expansion was observed, and as it was mentioned before, the foaming conditions for this copolymer are dramatically changed and cannot be compared with two other copolymer compositions.
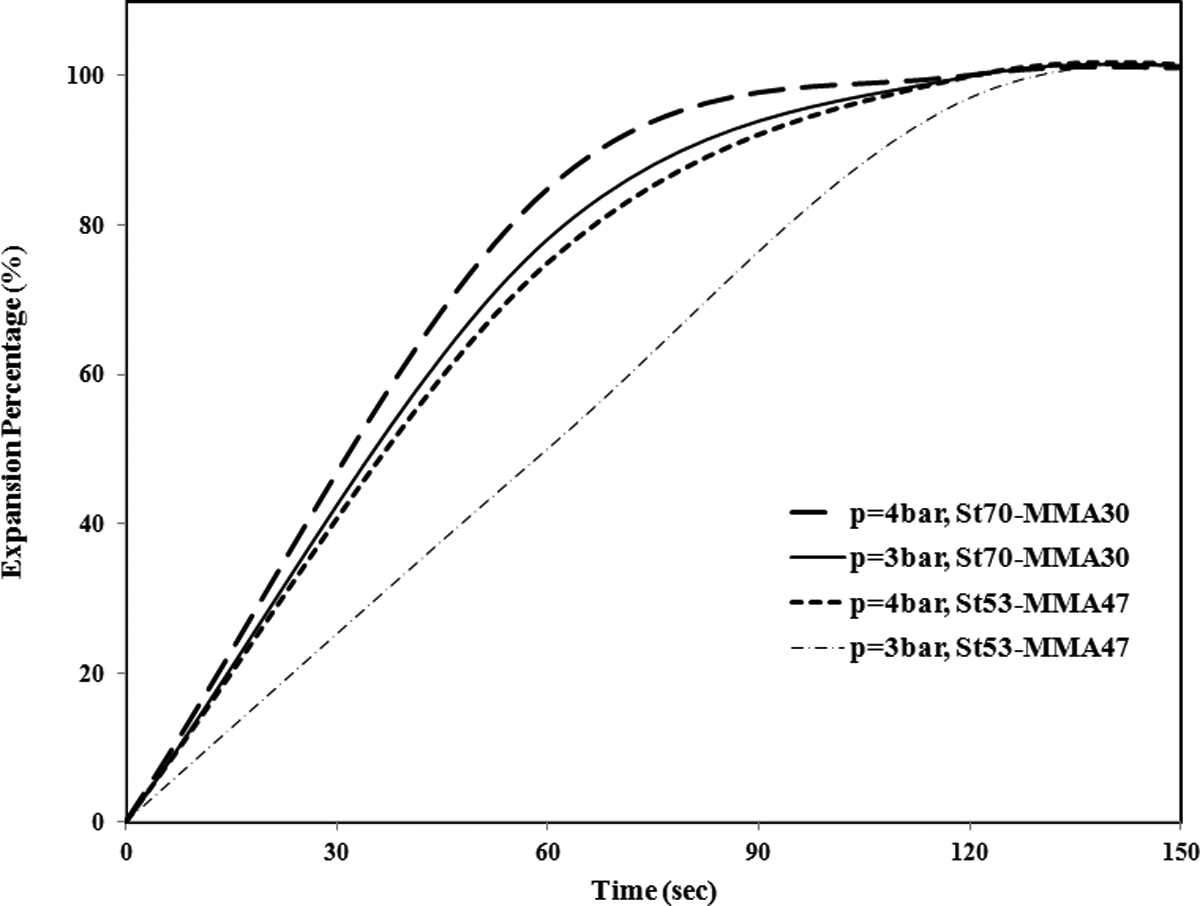
The rate of expansion of St/MMA copolymers at different pressures and T = 95°C. St/MMA: styrene/methyl methacrylate.
Conclusion
In this study, it was tried to investigate the foaming dynamics of St/MMA copolymers at different compositions via visual batch foaming process. It was concluded that two distinct phenomena are observed at different sorption pressures. The first one that occurred in pressures lower than 3 bar was related to pentane sorption in St/MMA:70/30 (wt/wt) and St/MMA:53/47 (wt/wt) copolymers, which was reduced with temperature increment, and the second one took place in pressures higher than 3 bar for St/MMA:53/47 (wt/wt) with great amounts of pentane diffused into the copolymer bulk filling the nucleated cells after rapid pressure release, therebyleading to higher foaming ratio. Furthermore in St/MMA:30/70 (wt/wt) copolymer due to high content of MMA, the diffusion of the n-pentane to the matrix was difficult. The SEM images confirmed that the cellular structures were decreased and limited to the shell of the copolymer matrix with MMA content increment in copolymer. Furthermore, the cell structure extension was improved by increasing sorption pressure and time due to rising pentane sorption and diffusion into the cells, and it was reduced with temperature increment due to declining pentane sorption. Ultimately, it was concluded that with MMA content increment, the rate of expansion was declined.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
