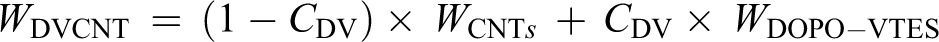Abstract
The multi-walled carbon nanotubes (CNTs) were modified using phosphaphenanthrene compounds (9,10-dihydro-9-oxa-10-phosphaphenanthrene-10-oxide (DOPO)) and vinyl triethoxy silane (VTES) by covalent bond. The DOPO- and VTES-treated CNTs were named DVCNT, and the structure was characterized by Fourier transform infrared, thermogravimetric analysis, and transmission electron microscopy. Nanocomposites were prepared by adding CNTs or DVCNT to poly(lactic acid) (PLA), and the flame retardancy was examined by determination of limiting oxygen index (LOI), vertical burning (UL94), and cone calorimetry. Results reveal that the LOI of PLA/DVCNT_4% composites was increased to 26.6 and prevents dripping of PLA in some level, and DVCNT can significantly reduce heat release rate and mass loss rate during combustion, which indicate that DVCNT can improve the dispersibility of the CNTs in the polymer composites and hence enhance the flame retardancy simultaneously.
Introduction
Due to the growing concern of environmental pollution and nondegradable shortage of many petrochemicals, a transition from fossil-based polymers to bio-based polymers would be appropriate. Poly(lactic acid) (PLA) as bio-sourced polymeric material has received more extensive attentions and has many advantages such as biodegradability, abundant renewable resources, excellent mechanical properties including high strength and high stiffness, appropriate thermal properties, and electrical properties. 1 –4 PLA represents one of the most promising bio-sourced polymers and can be widely used in electronic products, packaging field, and disposable products. Nevertheless, PLA resin is a highly flammable material with serious melt dripping during combustion, which severely limits its potential applications. Therefore, the research on flame-retardant and dripping-resistant PLA is indispensable. 5
In recent years, many studies have been conducted to reduce the flammability of PLA. Several systems revealed better flame-retardant performance, such as phosphorus-containing silsesquioxane, 6,7 intumescent flame retardants, 8 –10 and nitrogen phosphorus flame retardants. 11,12 In addition to organic flame retardants, inorganic flame retardants including zinc/aluminum-layered double hydroxide, 13 carbon nanotubes (CNTs), 14 –16 sepiolite nanoclays, 17 nanosized carbon black combined with nickel sesquioxide (Ni2O3), 18 and inorganic nanoparticles 19,20 can effectively enhance the flame-retardant properties of PLA. However, PLA often needs to blend with higher dosage flame retardants to obtain excellent flame retardancy. Therefore, a superefficient flame-retardant PLA system that maintains the balance between flame retardancy and thermal/mechanical properties at the same time still needs to be explored. 21 Recently, 9,10-dihydro-9-oxa-10-phosphaphenanthrene-10-oxide (DOPO) has demonstrated great potential on flame retardancy, 22 –25 intrinsically through the reactivity of its P–H bond that can either perform a direct nucleophilic attack on the epoxide ring or construct various derivatives bearing nucleophile group like phenol, amine, or anhydride. 26 –29 CNTs can be used to improve the flame retardancy of polymer composites. It has good effect to improve the heat release rate (HRR) and mass loss rate (MLR) of the polymer composites and provides a new approach for halogen-free flame retardant. 30 However, the CNTs have a large van der Waals force and high surface energy, so it should be modified to avoid aggregation in the composites. 31 If it can disperse in polymer materials well, the flame retardancy of polymer composites can be improved greatly. 32
In this work, the carboxyl-containing CNTs were modified using phosphaphenanthrene compounds (DOPO) and vinyl triethoxy silane (VTES) by covalent bond. The structure and properties of CNTs modified with DOPO-VTES (DVCNT) were systematically characterized, and nanocomposites were prepared by adding CNTs or DVCNT to PLA. The combustion behaviors of PLA containing DVCNT were studied and the flame-retardant effects of DVCNT were also analyzed.
Experiment
Materials
PLA (3801X, melt index of 2.99 g/10 min) was purchased from NatureWorks LLC, Blair, NE, USA. Carboxyl-containing CNTs (–COOH content, 3.86 wt%) with a diameter of less than 8 nm were purchased from Chengdu Institute of Organic Chemistry, China. DOPO was purchased from Jiangsu Hanfeng Industrial Co. Ltd (China). VTES was purchased from Beijing HWRK Chem Co., Ltd (China).
Synthesis of intermediates DOPO-VTES
The synthesis of DOPO-VTES could be achieved by the following procedures: DOPO (108.0 g, 0.5 mol) and 80 ml of toluene added to the 500 ml three-necked flask under magnetic stirring and heated to 80°C until DOPO completely dissolved, then an excessive solution of VTES (130 ml, 0.6 mol) was placed in the constant pressure dropping funnel and added dropwise into the three-necked flask in 3 h. Subsequently, the reaction was maintained at 115°C for 15 h at reflux and then 25 ml of 10% sodium hydroxide solution was added to the flask and allowed to react for 8 h. After the reaction, unreacted VTES and small molecules were removed through reduced pressure vacuum distillation. Finally, heating was stopped and the mixture was allowed to cool naturally, which results in a slightly yellowish transparent product of 150.8 g, DOPO-VTES, with a yield of 93.7%. The synthetic route is shown in Figure 1(a).
The synthetic route of DOPO-VTES (a) and DVCNT (b).
The surface modification of CNTs with DOPO-VTES
64.4 g of DOPO-VTES was mixed with 50 g of carboxyl-containing CNTs in a 500 ml three-necked flask and placed in an ultrasonic bath for 8 h at 80°C. Subsequently, the two functional groups undergo esterification reaction effectively, and carboxyl-containing CNTs were evenly dispersed in the DOPO-VTES, and then the mixture was allowed to cool naturally to room temperature. Finally, the unreacted DOPO-VTES was removed using a lot of ethanol through Polytetrafluoroethylene (PTFE) microporous membrane (aperture 0.2 microns) filter. The resulting product was dried in a vacuum at 80°C for 12 h to a constant weight to get the DVCNT. The synthesis route of DVCNT is shown in Figure 1(b).
Preparation of flame-retardant PLA composites
The flame-retardant PLA composites were prepared by mixing CNTs or DVCNT into PLA with different mass ratios, designated as PLA/CNTs_x% or PLA/DVCNT_x% (x = 1, 2, 3, 4; x denotes the wt% of CNTs or DVCNT). The PLA and DVCNT were blended in a torque rheometer (Shanghai Kechuang Rubber Plastic Mechanical Equipment Corporation, Shanghai, China) at 190°C for 6.5 min. Then, the flame-retardant PLA composites were transferred to a mold, preheated at 190°C for 5 min, pressed at 190°C for 6 min, and then cooled to room temperature. The specimens with standard dimensions for further testing were obtained. The CNTs were blended into PLA to prepare flame-retardant PLA composites using the same procedure.
Characterization
Fourier transform infrared (FTIR) spectra were detected on a Nicolet iN10MX type spectrometer (Madison, WI, USA).
The morphology of DVCNT was observed with a Talos F200 S transmission electron microscope (TEM) at an accelerating voltage of 200 kV.
Thermogravimetric analysis (TGA) was performed using a TA Instrument Q500 IR thermal gravimetric analyzer (China) at a heating rate of 20°C min−1 from 50°C to 700°C under nitrogen (N2) atmosphere.
The TGA-FTIR was carried out on a STA 8000 simultaneous thermal analyzer with a FTIR spectrophotometer (PerkinElmer, Waltham, MA, USA). The samples were placed in an alumina crucible and heated from 50°C to 700°C at a linear heating rate of 20°C min−1 under N2 atmosphere. The infrared cell and transfer tube were kept at 280°C to avoid the condensation of the pyrolysis products.
The limiting oxygen index (LOI) values were obtained using a FTT Dynisco LOI instrument (Fire Testing Technology, UK) according to ASTM D2863-97 with a sample size of 130 × 6.5 × 3.2 mm3.
The vertical burning test (UL94) was performed on FTT0082 instrument according to ASTM D3801 testing procedure with sheet dimensions of 130 × 13 × 3 mm3.
The fire behavior was characterized on a FTT cone calorimeter according to ISO 5660 under an external heat flux of 50 kW m−2 with a sample size of 100 × 100 ×3 mm3.
The dispersion of CNTs and DVCNT and the micromorphology of the residual char from the cone calorimeter test were examined using a FP 2032/14 Quanta 250 FEG scanning electron microscope (SEM) (Waltham, MA, USA) under high vacuum at a voltage of 20 kV.
Results and discussion
Characterization of DVCNT
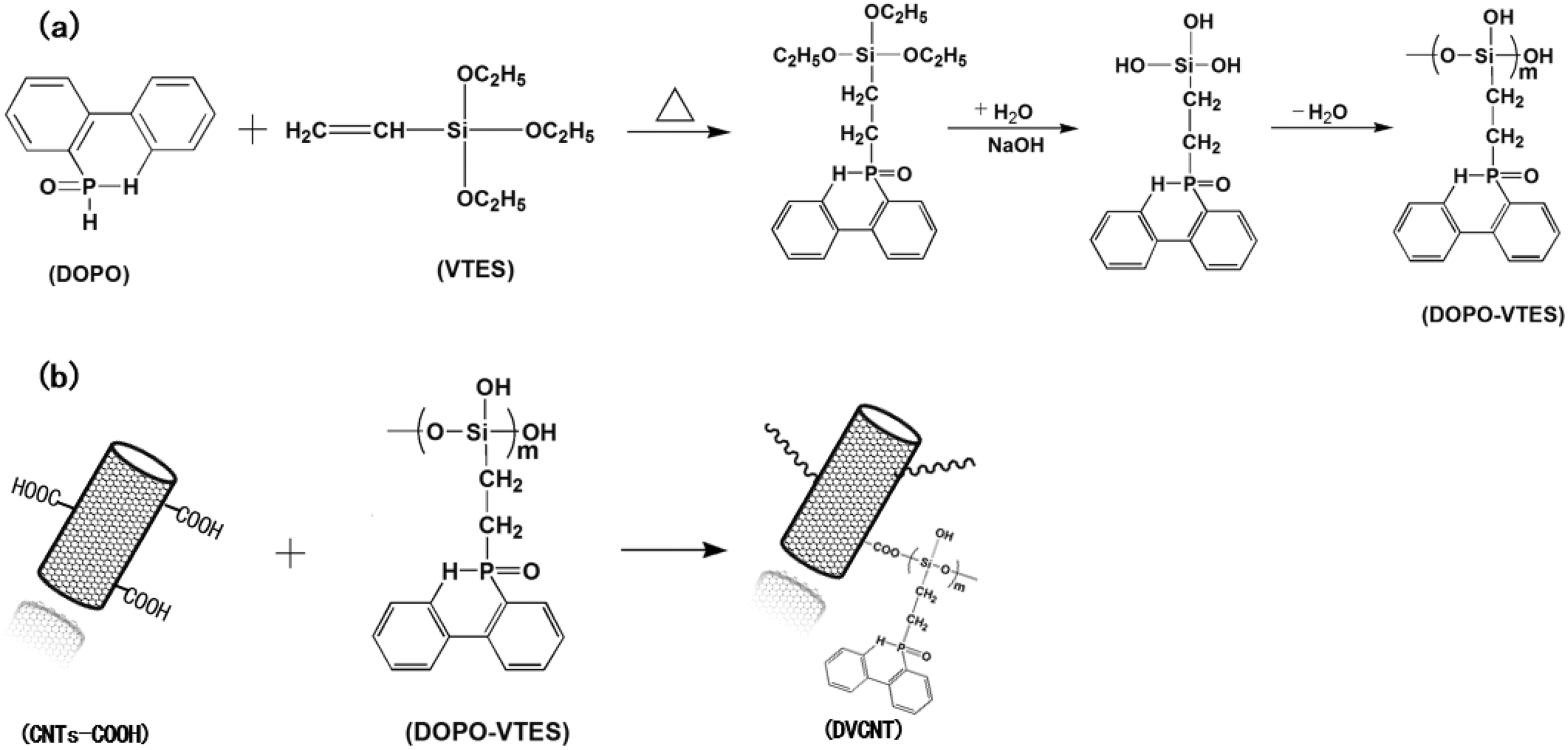
FTIR spectra of DOPO, VTES, and DOPO-VTES were presented in Figure 2. The FTIR spectrum of the DOPO showed that there were absorption peaks at 3359 cm−1 (C–H in phenyl group), 2436 cm−1 (P–H), 1594 cm−1 (phenyl group), 1478 cm−1 (P–phenyl), 1199 cm−1 (P=O), and 903 cm−1 (P–O–phenyl). The FTIR spectrum of the VTES showed that there were absorption peaks at 2885–2975 cm−1 (–CH2, –CH3), 1080 cm−1 (Si–O–C), and 1632 cm−1 (=CH2). The FTIR spectrum of the DOPO-VTES showed that the absorption peak at 2436 cm−1 (P–H) disappeared, and the absorption peak at 1477 cm–1 (P–Ph) indicates the DOPO has reacted with VTES. Meanwhile, these characteristic peaks in the spectrum of DOPO also appear in the spectrum of DOPO-VTES, which implied that the resulting DOPO-VTES contained the cyclic DOPO structure. Moreover, the absorption peaks at 3100–3600 cm−1 (Si–OH) and 1005–1118 cm−1 (Si–O–Si) indicate the siloxane hydrolytic polymerization. The results of FTIR clearly verified that DOPO-VTES was successfully synthesized.
The FTIR spectra of DOPO, VTES, and DOPO-VTES.
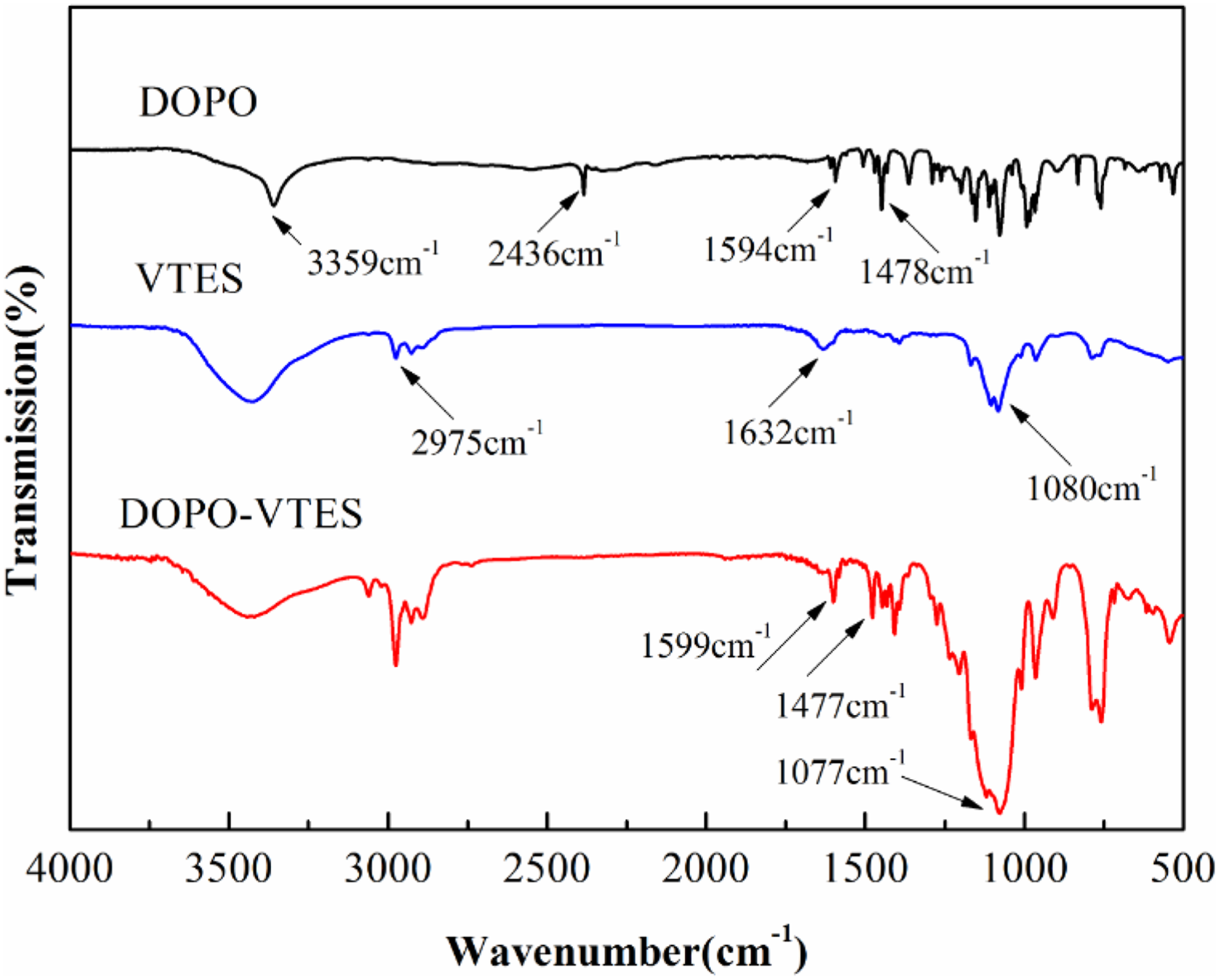
Figure 3 shows the FTIR spectra of CNTs and DVCNT. For CNTs, they have few identifiable functional groups. The peak at 1579 cm−1 is attributed to an in-plane E1u mode of monocrystalline graphite. Furthermore, those similar characteristic peaks at 1568 cm−1 (phenyl group) and 1005–1118 cm−1 (Si–O–Si) in the spectrum of DOPO-VTES also appear in the spectrum of DVCNT, demonstrating that the hydroxyl groups in DOPO-VTES have reacted with carboxyl groups and that DOPO-VTES has wrapped on the surface of CNTs.
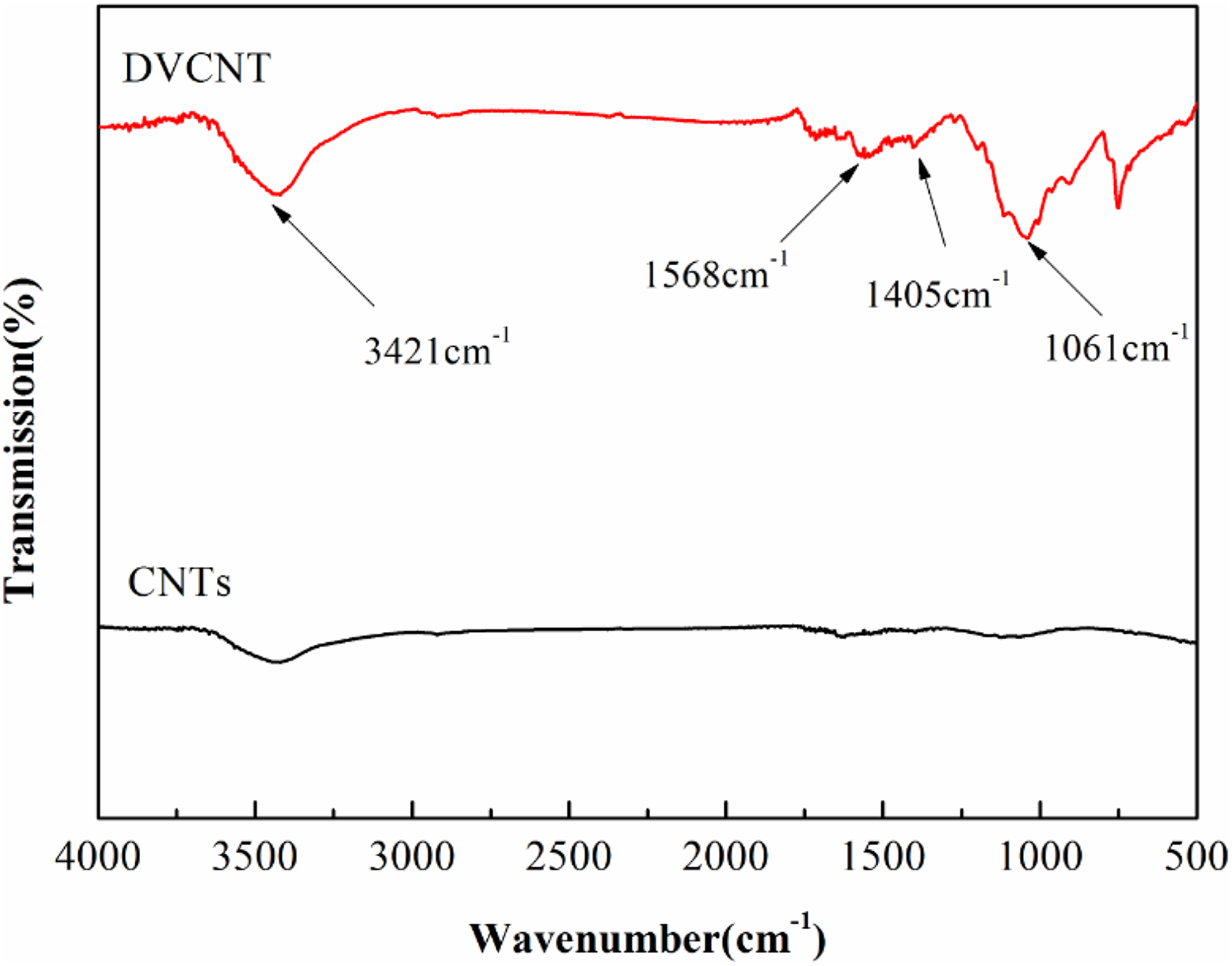
The FTIR spectra of CNTs and DVCNT.
In order to get the morphology of evidence, the comparison of TEM images of CNTs and DVCNT is presented in Figure 4. Figure 4(a) displays a typical TEM image of CNTs, showing very smooth surface and clear tubes. From the TEM image of DVCNT in Figure 4(b), the surface of CNTs became obscure and rough because the DOPO-VTES was wrapped on the external surface of CNTs. 33 In addition, the evident increase in diameters of CNTs is discerned from DVCNT in Figure 4(b). The DOPO-VTES is tightly wrapped on the surface of CNTs, because there is a strong covalent bond and π–π stacking interaction between DOPO-VTES and the walls of CNTs. Undoubtedly, the TEM images provide a further direct evidence of the modification of DVCNT.
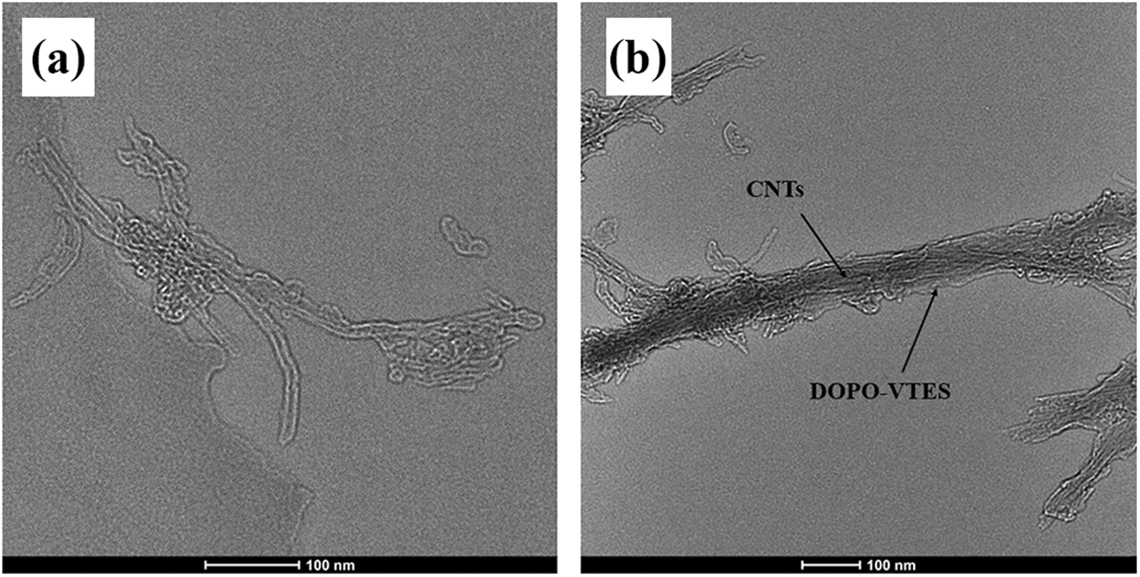
TEM images of (a) CNTs and (b) DVCNT.
Figure 5(a) shows the TGA curves of DOPO, DOPO-VTES, CNTs, and DVCNT, which were measured from 50°C to 800°C at a heating rate of 20°C min−1 under N2 atmosphere. Compared with DOPO, the initial degradation temperature of DOPO-VTES decreased, but the residue increased. This is due to the large Si–O–Si bond in DOPO-VTES, which produces silicon dioxide (SiO2) in thermal decomposition, which greatly increases the quality of residual carbon. Obviously, CNTs presented good thermal stability under N2 atmosphere. When the temperature was increased up to 800°C, there was no evident decomposition in CNTs, with 93.9 wt% of residue at that temperature. The synthesized DOPO-VTES started to decompose at 145.7°C (weight loss of 1 wt%) and about 43.5 wt% of residue was left at 800°C, indicating that DOPO-VTES had a good char-forming capability. For DVCNT, about 76.5 wt% of residues were left at 800°C. By comparing the differences in the weight loss between CNTs, DOPO-VTES, and DVCNT, the content of the DOPO-VTES in DVCNT was roughly estimated using the following equation
where W CNTs, W DOPO-VTES, and W DVCNT are the residue weight fractions of CNTs, DOPO-VTES, and DVCNT at 800°C, respectively, and C DV, percent grafting, denotes the content of the DOPO-VTES in DVCNT.
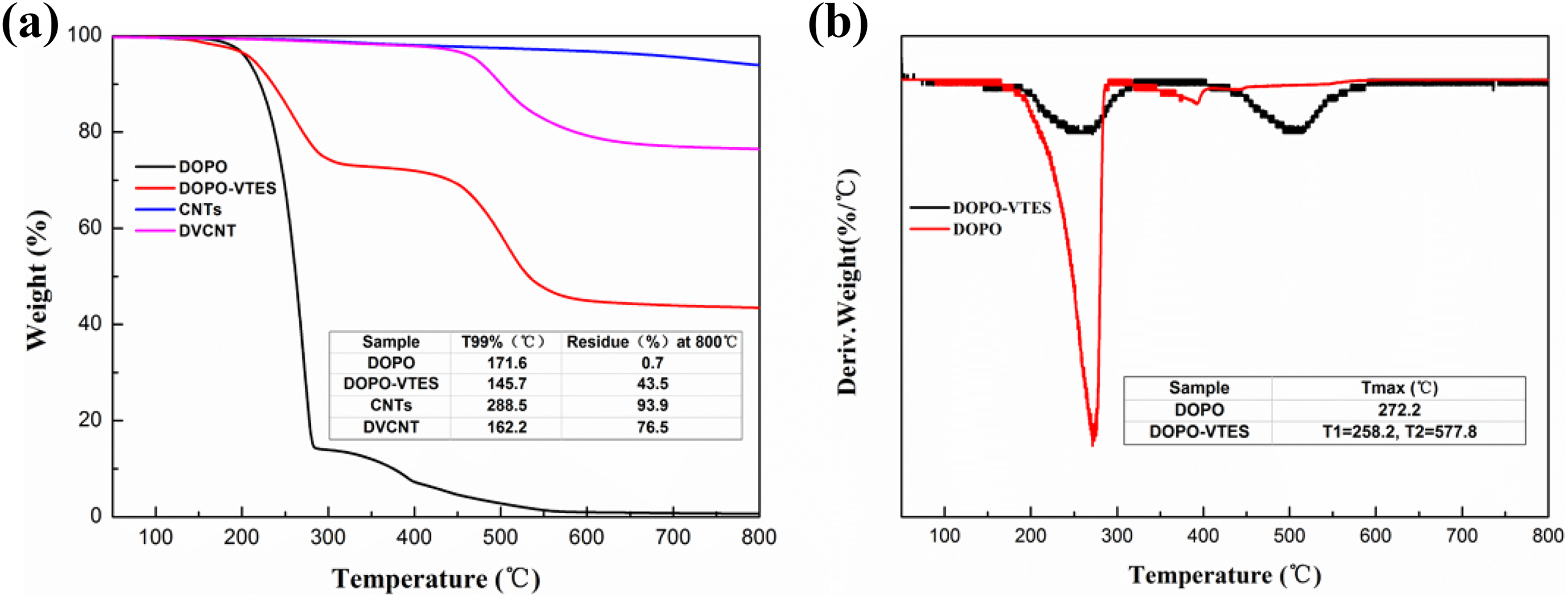
TGA curves of DOPO, DOPO-VTES, CNTs, and DVCNT (a), and DTG curves of DOPO and DOPO-VTES (b).
Considering that the mutual influence on thermal degradation between DOPO-VTES and CNTs is possible, the C DV is only approximate but not accurate. The calculated C DV is 34.5%. Moreover, the residue of DVCNT is decreased by 17.4% compared with CNTs, which can be inferred that the surface of CNTs is modified. This combined with the analysis results of FTIR can further indicate that the modification of CNTs surface is successful.
The decomposition of DOPO-VTES is divided into two stages and appear two maximum decomposition temperature (T max), the first phase is 150–350°C, the mass loss of 26.1%, a difference of 14°C of the T max between DOPO and DOPO-VTES in the temperature range. The stage of mass loss is phosphorus containing organic groups of DOPO-VTES decomposed into low molecular organic phosphoric acid and decomposition of organic silicon alcohols oligomer. The second phase is 350–700°C and mass loss of 28.8%; about 11.4% of the mass loss is from the decomposition of organic phosphoric acid, while 17.4% comes from the damaged Si–O–Si bonds produced by SiO2 and released gas product.
In order to employ the flame-retardant mechanism of DOPO-VTES in the gas phase, the TGA-FTIR analysis was adopted to explore pyrolysis products evolved during the thermal degradation. Figure 6 exhibits the FTIR spectra of TGA gas products of DOPO-VTES. The absorption peaks of gas products from heated DOPO-VTES are mainly at 2891 cm−1 (C–H), 1740 cm−1 (C=O), 2364 and 2335 cm−1 (CO2), and 2186 cm−1 (CO). The FTIR spectra of gas products from heated DOPO-VTES exhibit an additional peak at 1079 cm−1 in Figure 6, which is the absorption of phosphaphenanthrene fragments (PO). It confirms that there are some phosphorus-containing free radicals with quenching effect in gas product resulting from DOPO-VTES. Another change in Figure 6 is that the relative intensities of CO2 and CO peaks increased. The phenomenon could be caused by the full decomposition of ester bond COO, which was catalyzed or induced by acid products of DOPO-VTES.
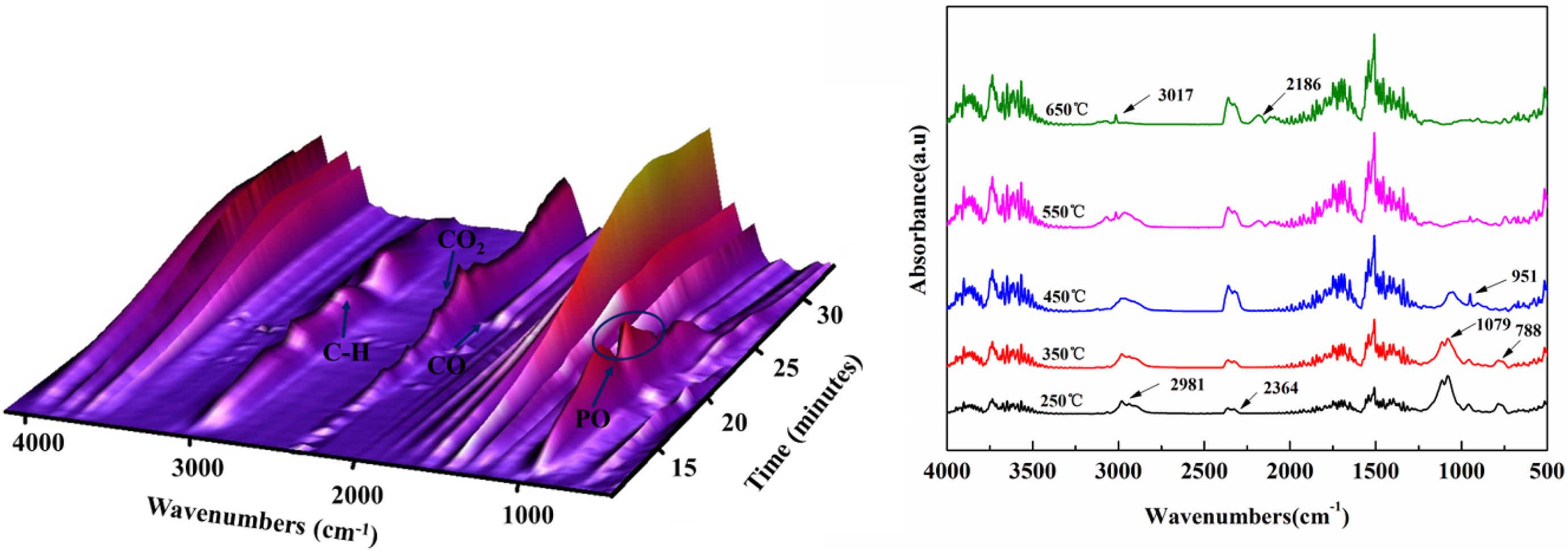
3-D TG-FTIR spectra of pyrolysis products for DOPO-VTES and the corresponding FTIR spectra at different temperatures.
Structure and morphology of PLA nanocomposites
The reinforcement potential of DVCNT is remarkably dependent on the distribution of DVCNT in the PLA matrix. Therefore, it is necessary to determine the dispersibility of DVCNT in the PLA matrix. As shown in Figure 7, the dispersion of CNTs and DVCNT was examined by SEM. It is more obvious that many large CNT aggregates are present in the PLA nanocomposite under higher magnification. In contrast to the case with CNTs, we observed that DVCNT was pulled out from the fractured PLA matrix surface, indicating that the interfacial adhesion between DVCNT and the matrix was improved. Moreover, it is not difficult to see that DVCNT uniformly dispersed in the PLA matrix, and the DVCNT is apparently form a network structure compared with pristine CNTs. This indicates that the DVCNT could present better dispersion in PLA matrix and improve the interfacial interactions than CNTs.
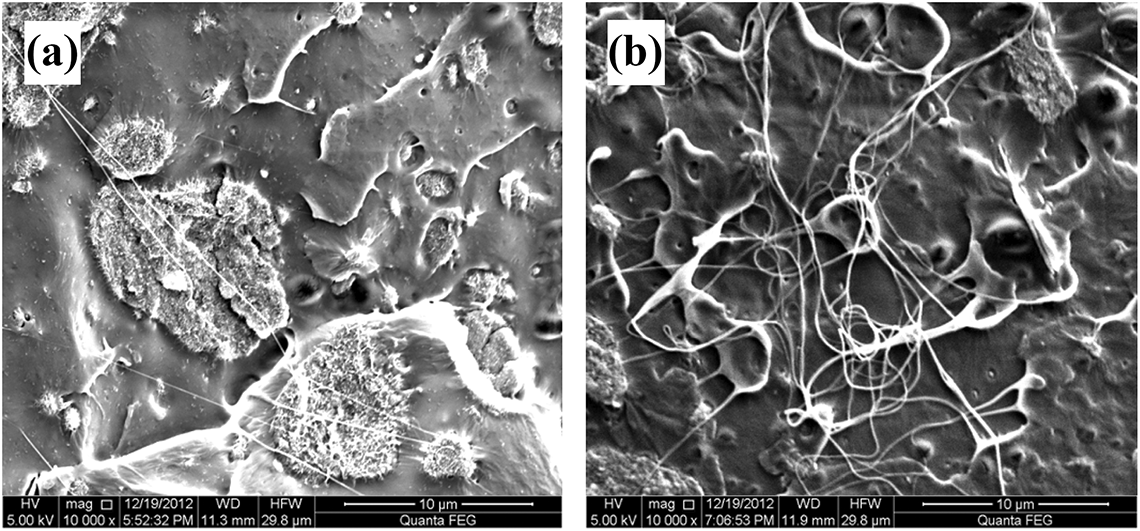
SEM images of fracture surfaces of (a) PLA/CNTs_4% and (b) PLA/DVCNT_4%.
LOI measurement and UL94 vertical burning tests
The flame retardancy of the PLA composites was detected using LOI and UL94 vertical burning tests; the corresponding results and dripping behavior of all the specimens are presented in Table 1. From LOI value, it can be seen that pristine PLA had a LOI of only 19.5% and is easily ignited, accompanied by heavy melt dripping. The presence of CNTs increased the LOI value of PLA only slightly. When the mass fractions of CNTs were 2 wt% and 4 wt%, the LOI values of PLA/CNTs only increased from 19.5% to 19.8% and 20.8%, respectively, and still caused dripping. It shows that CNTs merely exhibited flame retardancy at a low level to PLA, which can be deduced that the heavily aggregation in the composites. However, the modified CNTs significantly improved the LOI values of PLA matrix, the LOI value of PLA/DVCNT increased to 26.6% when the mass fraction of DVCNT was only 4 wt%. Moreover, when the content of DVCNT is 3 wt%, PLA/DVCNT samples can achieve UL94 V-1 level, but it can’t achieve UL94 V-0 level when the adding amount of DVCNT increase. The above results have proved that DVCNT can enhance the flame-retardant properties of PLA resin. In addition, the hydroxyl groups on the surface of DVCNT can promote PLA resin thermal degradation, decrease the molecular weight, and increase liquidity in the process of heating, 34 although the dropping rate of flame-retardant PLA composites improved but not very obvious.
The LOI value and UL94 rating of PLA and its composites.

LOI: limiting oxygen index; UL94: vertical burning; PLA: poly(lactic acid); CNTs: carbon nanotubes; DVCNT: DOPO and VTES treated carbon nanotubes; VTES: vinyl triethoxy saline; DOPO: 9,10-dihydro-9-oxa-10-phosphaphenanthrene-10-oxide.
Thermogravimetric analysis
The mass loss curve characterizes the reduction of mass of the material during decomposition. Figure 8 shows the mass loss curves of the pristine PLA, PLA/CNTs, and PLA/DVCNT composites. With the increased addition of CNTs or DVCNT, the increased char formation was observed at the end of combustion (700°C), which is evident in the TGA results. DVCNT decomposed earlier when composites were heated as the onset degradation temperature of DVCNT is lower than that of PLA. The acid products of decomposed DOPO-VTES promoted the early degradation of PLA at lower temperature. Pure PLA is found to show only one step of decomposition at about 300°C, leaving negligible char above 400°C. However, the PLA/DVCNT composites exhibit certain amount of residues above 400°C, suggesting that the thermal stability of PLA has been enhanced at higher temperature. When the addition of DVCNT was 4 wt%, the residue was 10.2 wt% and improved 96.9% compared with pristine PLA, which present better charring. Moreover, phosphorous-containing DOPO-VTES promotes dehydration/carbonization of polymer on the thermal decomposition, so that the thermal stability of PLA/DVCNT nanocomposites is better.
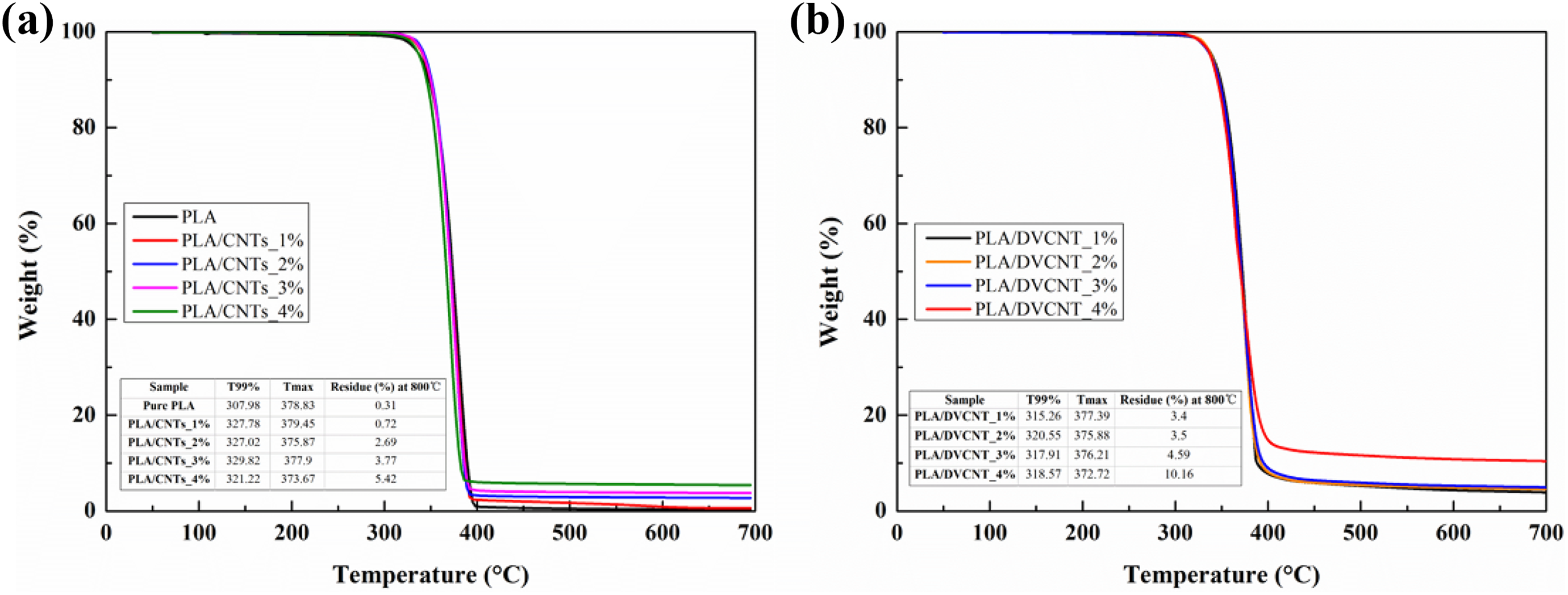
Normalized mass loss curves of pristine PLA, PLA/CNTs (a), and PLA/DVCNT composite (b) from the TGA under N2 atmosphere.
The differences in flame retardancy mechanism between PLA/CNTs_4% and PLA/DVCNT_4% are further compared. In condensed phase, the charring and barrier effects of PLA/DVCNT_4% are stronger than PLA/CNTs_4%, which just accord with the characteristics of char yield in Figure 9. The reason is that the interaction of the products from the pyrolysis of the DVCNT promoted charring of the PLA matrix and enhanced the barrier effect of the char. Thus, the reinforced char of PLA/DVCNT_4% is more effective to hinder the heat and mass transport compared with that of PLA/CNTs_4%. Further evidences in detail will be provided in the following discussion.
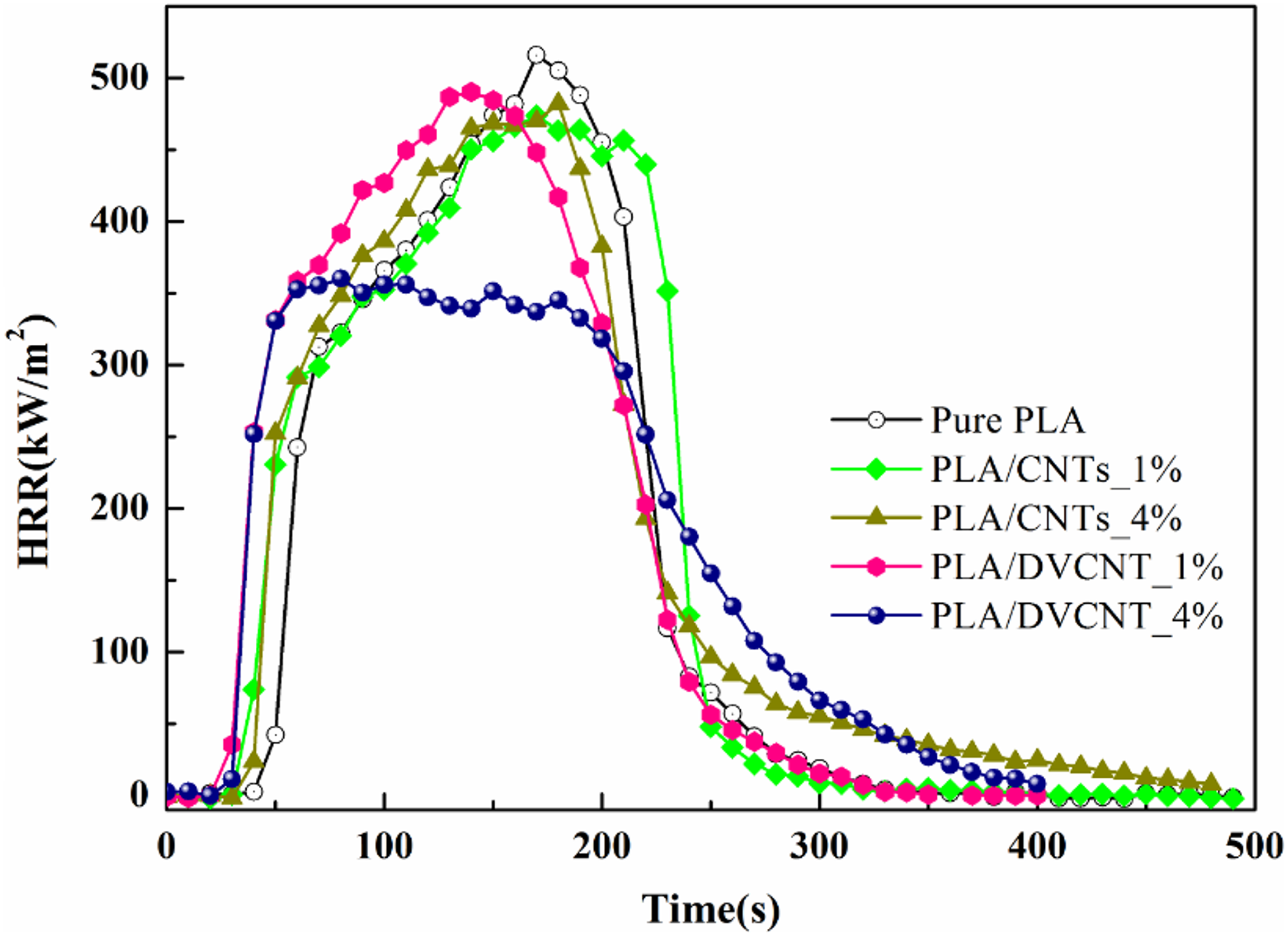
HRR curves of PLA, PLA/CNTs, and PLA/DVCNT composites.
Cone calorimetry test
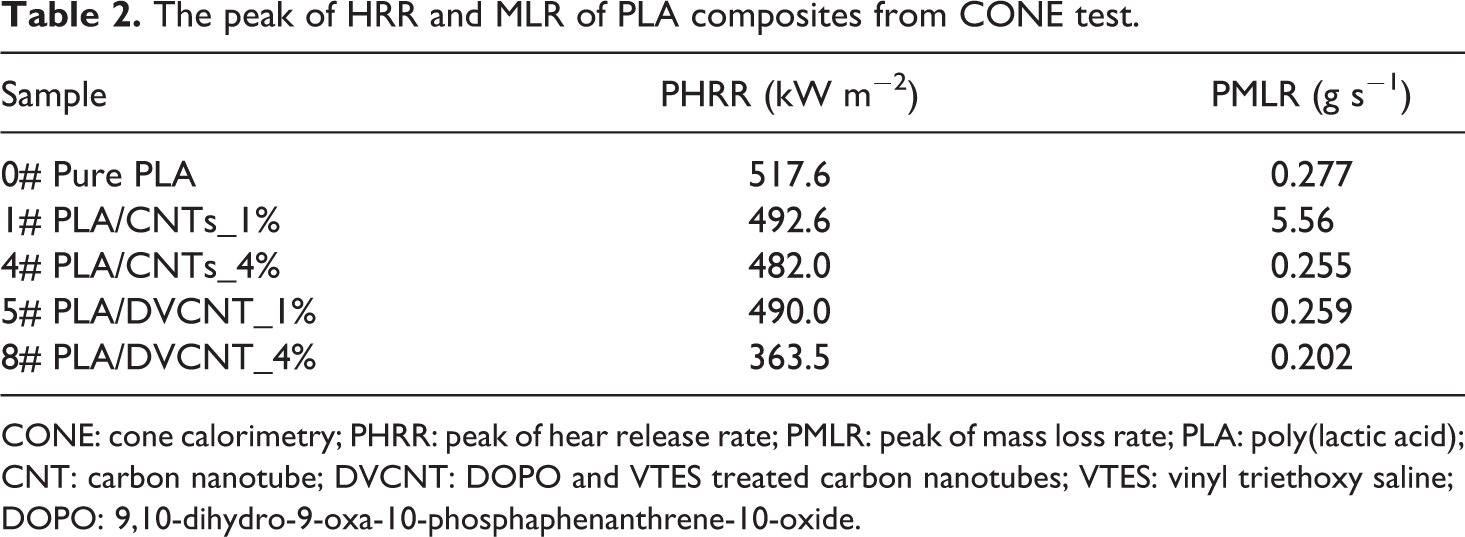
The flame-retardant effects of incorporating CNTs individually or DVCNT were evaluated by cone calorimeter. The HRR curves for pristine PLA, PLA/CNTs, and PLA/DVCNT composites are presented in Figure 9. Meanwhile, the detailed data are listed in Table 2.
The peak of HRR and MLR of PLA composites from CONE test.
CONE: cone calorimetry; PHRR: peak of hear release rate; PMLR: peak of mass loss rate; PLA: poly(lactic acid); CNT: carbon nanotube; DVCNT: DOPO and VTES treated carbon nanotubes; VTES: vinyl triethoxy saline; DOPO: 9,10-dihydro-9-oxa-10-phosphaphenanthrene-10-oxide.
To sum up, adding CNTs and DVCNTs to the PLA has reduced the burning effect of materials to a certain extent, but the effect of DVCNT is even more obvious. It can be seen that pristine PLA burned very fast after ignition and the peak HRR (PHRR) value appeared at 517.6 kW m−2. When the weight ratio of CNTs was 4 wt%, the PHRR value was 482 kW m−2, only decreased by 6.9%. The PHRR values also decreased with increasing loading of DVCNT, and the PHRR decreased to 363.5 kW m−2 when the weight ratio of DVCNT was 4 wt%.
In addition, the peak MLR (PMLR) values also show the same trend as the PHRR (Table 2). It is noteworthy that the PMLR of PLA/CNTs composites reduces by 7.9% at most, while the PMLR of PLA/DVCNT_4% composites reduces by 27.1% compared to pristine PLA. These results suggest that the DVCNT may lead to the formation of higher quality char layer during combustion. As analyzed above, the compact char layer prevents the transfer of combustible gases and heat during the combustion process. It also protects the underlying resin from further degradation. As a result, the heat evolved in tests reduces drastically.
Morphology analysis of char
The condensed-phase flame-retardant effects of the PLA/CNTs_4% and PLA/DVCNT_4% were further investigated using a digital camera and SEM. The macro-morphology images of the residues after the cone calorimeter test are shown in Figure 10. As shown in Figure 10(a), the residue from pure PLA sample reserved only tiny amounts of badly broken char, indicating that PLA almost totally decomposed during combustion. The residue of PLA/CNTs_4% in Figure 10(b) shows the local destruction and the char residue increase was not obvious, resulting in a fragile and cracked char after combustion. However, in Figure 10(c), the residue of PLA/DVCNT_4% was compact and complete, a preferable increasing char, which effectively hampered the heat and mass transfer, so it can improve the flame retardancy. This better cohesion of the combustion residue of the composite based on DVCNT could be explained by the more uniform dispersion of modified CNTs and the introduction of phosphorus and silicon with the charring ability from DOPO-VTES.

Digital photos of cone calorimeter residues of (a) pure PLA, (b) PLA/CNTs_4%, and (c) PLA/DVCNT_4%.
In order to further explore the charring and barrier effects, the microstructures of the combustion residues were observed and compared for PLA/CNTs_4% and PLA/DVCNT_4% by SEM, the images of cone calorimeter residues are shown in Figure 11. From direct observations, the residue of PLA/CNTs_4% in Figure 11(a) had a lot of open holes with different sizes, because the undispersed network char layer created by the decomposition of CNTs was brittle and easy to be damaged, which provided channels for the combustible volatiles from the inner matrix and heat feedback from the flame. Obviously, the residue of PLA/DVCNT_4% in Figure 11(b) was very dense, and few open holes were found. This can be explained by the fact that the modified CNTs formed a continuous network-structured protective layer. 30,35 Therefore, this residue structure can effectively seal the combustible volatiles in the matrix and decrease the combustion intensity through avoiding the exchange of heat and gaseous fluid, so it can improve the flame retardancy of polymer materials.

SEM analysis of cone calorimeter residues of (a) PLA/CNTs_4% and (b) PLA/DVCNT_4%.
Conclusions
The CNTs have been modified by the synthesized flame-retardant DOPO-VTES and the DVCNT was obtained successfully, which is confirmed by the evidence from the results of FTIR, TGA, and TEM measurements. The flame-retardant PLA composites were prepared and the thermal stability and flame retardance of the composites were comprehensively investigated. When the content of DVCNT is 4 wt%, the LOI value of PLA/DVCNT composites reach 26.6. PLA/DVCNT_3% can achieve UL94 V-1 level, but it can’t achieve UL94 V-0 when the percent of DVCNT increase, in other words, DVCNT can prevent dripping of PLA during the combustion in some level, but the effects were not obvious. Furthermore, DVCNT can significantly reduce PHRR and PMLR of PLA composites during combustion. It is proposed that the modification with this DOPO derivative flame retardant can improve the dispersibility of the CNTs and hence enhance the flame retardancy simultaneously.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Nature Science Foundation of China (no. 51403007).