Abstract
Nanocellulose (NC)-reinforced carboxymethyl cellulose (CMC) films were prepared by solvent casting technique. CMC solution in water premixed with NC dispersion at different proportions (i.e. at different NC contents) was used so as to determine the maximum amount of NC that can be incorporated in CMC so as to achieve a film with an optimum combination of mechanical and barrier properties normally sought for packaging applications. The composite films after thorough drying were inspected in respect of its mechanical, thermal, morphological, and water vapor transmission rate characteristics. Incorporation of up to 70 wt% of NC was found to give a practically usable film. A further increase in NC proportion however led to a fragile one. A remarkable increase in tensile strength (in relation to that of CMC) with workable elongation was observed at this maximum dose of NC. Superior thermal and barrier properties of such transparent composite films were also achieved.
Introduction
Carboxymethyl cellulose (CMC) is a semisynthetic, high molecular weight water soluble heteropolysaccharide in which carboxymethyl groups are substituted on the glucose units of the cellulose chain through an ether linkage. CMC is a very unique material in many of the following respects: it forms moderately strong and clear films; 1 it is a highly absorbent polymeric material; 2 and it has the potential for excellent material properties in combination with appropriate fillers. Moreover, it is renewable and nontoxic material. 1 CMC is an anionic linear polysaccharide derived from cellulose.
The good mechanical properties of cellulose fibers and their potential as a reinforcing agent in composites were recognized long ago. Recent focus has been directed to cellulose nanocrystals, also called nanocelluloses (NCs).
Cellulose nanocrystals are rigid molecular rods and can impart significant strength and directional stability to a composite. The Young’s modulus along the chain axis for perfect crystalline NCs is estimated to be up to 167.5 GPa, 3 and the tensile strength (TS) of the crystal structure is assumed to be approximately 0.8–10 GPa. 4 The extremely good strength properties and good thermal stability of the NCs make them suitable for use as reinforcement in bio-nanocomposite. In the present work, NC was derived from sugarcane bagasse and the resulting NC particles had the dimensions 170 nm × 35 nm. 5
The high crystalline characteristics of NC, in combination with the ability of the dried fibrils to form a dense network held together by strong interfibrillar bonds, suggest a remarkable barrier and strength properties inherent in NC. Thus, NC may be an interesting alternative to conventional fillers for mechanically weak plastics materials.
The combination of CMC and NC is used in a variety of applications from viscosity modifiers to medical implants and drug delivery. 6 The numerous hydroxyl and carboxylic groups in CMC impart high water binding and moisture sorption properties. CMC is also known to improve the mechanical and barrier properties of cellulose-based films. 7 A film coated on the fibers by CMC gel tends to enhance the physical properties of paper for printing purposes. 8 In addition, water soluble polysaccharides, such as CMC, can be used to modify the suspension rheology and also to introduce functional groups to cellulosic surfaces. 9 –12
Polysaccharides are generally hydrophilic resulting in poor water vapor and gas barrier properties. This is attributed to the presence of large number of hydroxyl groups. These two drawbacks of CMC may be expected to be overcome by NC as it can be expected to mitigate the influence of hydroxyl groups of CMC by hydrogen bonding through interaction with its own hydroxyl groups.
The reports on NC-reinforced CMC biocomposite films are scarcely found. 13 CMC is a cellulose ether that forms excellent films due to its polymeric structure and high molecular weight. It would be expected that CMC would be readily compatible with NC due to its chemical similarity. 14
The objective of the present work is twofolds. First, an attempt has been made to explore the modifying influence of NC derived from sugarcane bagasse on CMC over a wide range of its incorporation in CMC in terms of reduction in its moisture absorption characteristics which poses a great threat in packaging industries. The consequent influence on the mechanical properties of the composite films governing its flexibility and machinability (capacity to withstand stress/strain) would also be investigated. This work may be an indicator for the synthesis of nano-bio-composite film for packaging industries now suffering from disposal problems for packaging films.
Experiment
Materials
Sodium CMC was purchased from Loba Chemie Pvt Ltd, Mumbai, India. The chemical reagent was used without any further purification processes. NC used was synthesized from waste sugarcane bagasse in our laboratory.
Methods
Isolation of NC
The NC suspensions were obtained by acid hydrolysis of cellulose isolated from sugarcane bagasse according to a method described in our previous work. 5 Briefly, the delignified and hemicellulose free cellulose was hydrolyzed with 60 wt% sulfuric acid at 50°C for 5 h under strong agitation. The resulting suspension was cooled to room temperature and washed with distilled water by successive centrifugations until pH 7 was achieved. Finally, the suspension was homogenized by sonication using an ultrasonic processor with probe (UP-500, E-Chrom Tech Co Ltd, Taiwan) operating at 30 kHz for 5 min in an ice bath to avoid overheating. The suspension was kept refrigerated until use. The concentration of NCs in the final dispersion was determined gravimetrically.
Preparation of NC/CMC film forming solutions
To prepare NC/CMC film forming solutions, the CMC solution was first prepared by dissolving CMC in distilled water to a concentration of 2.2 wt%, and stirred at ambient temperature for 30 min using a magnetic stirrer. Varying proportions of NC suspension with known solid content of 1.5 wt% were added to the CMC solution to adjust the NC concentration to 90, 80, 70, and 60 wt% (of the weight of the total solid content of the mix), respectively, and stirred for 10 min. The process for making blends of these ratios is being given here. CMC solution was prepared at a definite concentration of CMC in 100 ml of water. With the known solid content of CMC when a definite amount of solution is taken, the amount of CMC present in it can be easily calculated out. Once weight of the solid CMC is known, the requisite quantity of NC to make a definite percentage of the solid CMC is calculated. Now as the percentage solid content in the NC dispersion is 1.5, the amount of the said dispersion containing the requisite amount of NC is calculated out. That exact amount of NC dispersion is then added to the CMC solution to get the different ratios. The mixtures were then homogenized by sonication for 2 min. The temperature of the suspensions rose to around 55°C during this time. To remove the entrapped air bubbles during mixing, the film forming solutions were deaerated under vacuum with the aid of a vacuum pump. Then required amount of distilled water was added in all the four mixes and mixed uniformly in order to make up the volume loss.
Preparation of NC/CMC nanocomposite films
To cast the films, the final different suspensions of NC/CMC (of various composition) film forming solutions were transferred into the leveled glass plate covered with a thin polyester sheet and kept at room temperature until the films were dried and subsequently kept in vacuum oven at 75°C for 24 h to ensure complete removal of water. The resulting composite films were then peeled off from the glass plate and allowed to equilibriate at room temperature subsequently. Thicknesses of films were measured to the nearest 0.03 mm with a micrometer (QUICKmini, Mitutoyo Co., Japan) and the average was taken.
Characterization
Fold endurance test
Fold endurance test of the film samples with thickness of about 0.03 mm was performed manually. It is defined as the logarithm of the number of double folds (a process of folding a sample first backward and then forward about the same line, i.e. one complete oscillation) that are required to make a test piece break at the conditions of the laboratory. Fold endurance is expressed by the following equation
where F is fold endurance and d is the number of double folds.
Fold endurance measures the durability and ductility of sample when repeatedly folded under load (manually), determining how many times the sample can be folded until it breaks.
Fourier transform infrared spectroscopy
The Fourier transform infrared (FTIR) spectra of CMC, NC, and NC/CMC (70:30, 60:40, and 50:50) composite dry films were recorded with a spectrophotometer (Jasco FTIR 6300, UK) equipped with an attenuated total reflectance device using a tri-glycine sulfate detector. The spectrum for each sample was recorded in the region of 500–4000 cm−1 at a resolution of 4 cm−1. The resulting FTIR spectra were compared to evaluate the effects of NC filling in the NC/CMC nanocomposite films based on the intensity and shift of vibrational bands.
Mechanical properties
The mechanical behavior (TS, % elongation at break (% EB), elastic modulus, and toughness) and pictorial representation of stress–strain behavior of different specimens undergoing tensile deformation were determined using an Instron H50KT (Tinius Olsen Ltd, UK), tensile testing equipment. The maximum force of the cell used in the tensile tests on Instron machine is 100 N. Tensile deformation was determined at a crosshead speed of 2.5 mm min−1. The tests were carried out at room temperature 25°C. The dimensions of the test samples according to the standard test method American Society for Testing and Materials (ASTM) D638 were as follows: length 50 mm, width 15 mm, and thickness approximately 0.03 mm. TS, % EB, elastic modulus, and toughness were calculated on the basis of initial sample dimensions, and the results were presented as the averages of five measurements. 15
Thermal properties
Thermogravimetric analysis
The thermogravimetric analysis (TGA) studies of the films were carried out on a Shimadzu instrument (TGA-50, Japan). Approximately 3 mg of each sample was heated from room temperature to 700°C at the heating rate of 10°C min−1. All of the measurements were performed under a nitrogen atmosphere with gas flow rate of 20 cm3 min−1.
Differential scanning calorimetry
Differential scanning calorimetry (DSC) thermograms were performed with a Shimadzu instrument (DSC-60, Japan). The scanning temperature was from room temperature to 300°C using a heating rate of 10°C min−1 under nitrogen atmosphere with gas flow rate of 20 cm3 min−1.
X-ray diffraction
Wide-angle X-ray diffraction (XRD) patterns from the NC/CMC nanocomposite and the individual polymer film samples were studied with an X’Pert Pro Panalytical X-ray diffractometer (Panalytical Ltd, Cambridge, UK) in order to ascertain the crystalline nature of the prepared films. The generator operated at 40 kV and 30 mA. The samples were scanned in the range 2θ = 5° and 40° at the rate of 3° min−1 with a nickel-filtered Cu Kα beam (wavelength 1.5406 Å).
Moisture vapor transmission rate
Moisture vapor transmission rate (MVTR) tests of the film samples were conducted gravimetrically using an ASTM procedure by the Permeation And Transmission Rate Analysis (PATRA) method. 16 Individually, CMC film and NC/CMC (70:30, 60:40, and 50:50) composite films were mechanically sealed onto Patra dish containing 5 g of anhydrous calcium chloride. The Patra dishes were initially weighed and placed in a controlled humidity chamber maintained at 35°C and 100% Relative Humidity (RH) for 24 h. The amount of moisture vapor transferred through the film and absorbed by the desiccant was determined from the weight gain of the Patradish. The assemblies were weighed initially and after every 24 h for all samples and continued till a constant weight was reached. Changes in weight of the Patra dish were recorded. The test was continued until an equilibrium was reached and there was no further change in weight. The MVTR was calculated using the following equation
where Δm is the weight increase (g), Δt is the exposure time in the chamber (d), (Δm/Δt) is the slope of the straight line (weight loss per unit time) (g/d), and A is the test area of the film (m2).
The influence of NC loading on MVTR was investigated in NC/CMC composite films. Determination of MVTR under the envisaged storage conditions was required to evaluate the effective use of the material in real application.
Morphological studies
Scanning electron microscopy
Scanning electron microscopy (SEM) was used to investigate the surface morphology of the CMC, NC, and NC/CMC nanocomposite films by using an SEM apparatus from CARL ZEISS EVO 18, Special Edition (Germany) with an accelerating voltage of 15 kV. In this case, the samples were sputter-coated with a thin layer of gold to prevent the buildup of an electrostatic charge.
Results and discussion
Fold endurance test
This test is quite significant and has tremendous relevance pertaining to films meant for packaging industries. The NC film (neat) and various NC-reinforced CMC films were subjected to fold endurance test manually as shown in Figure 1.
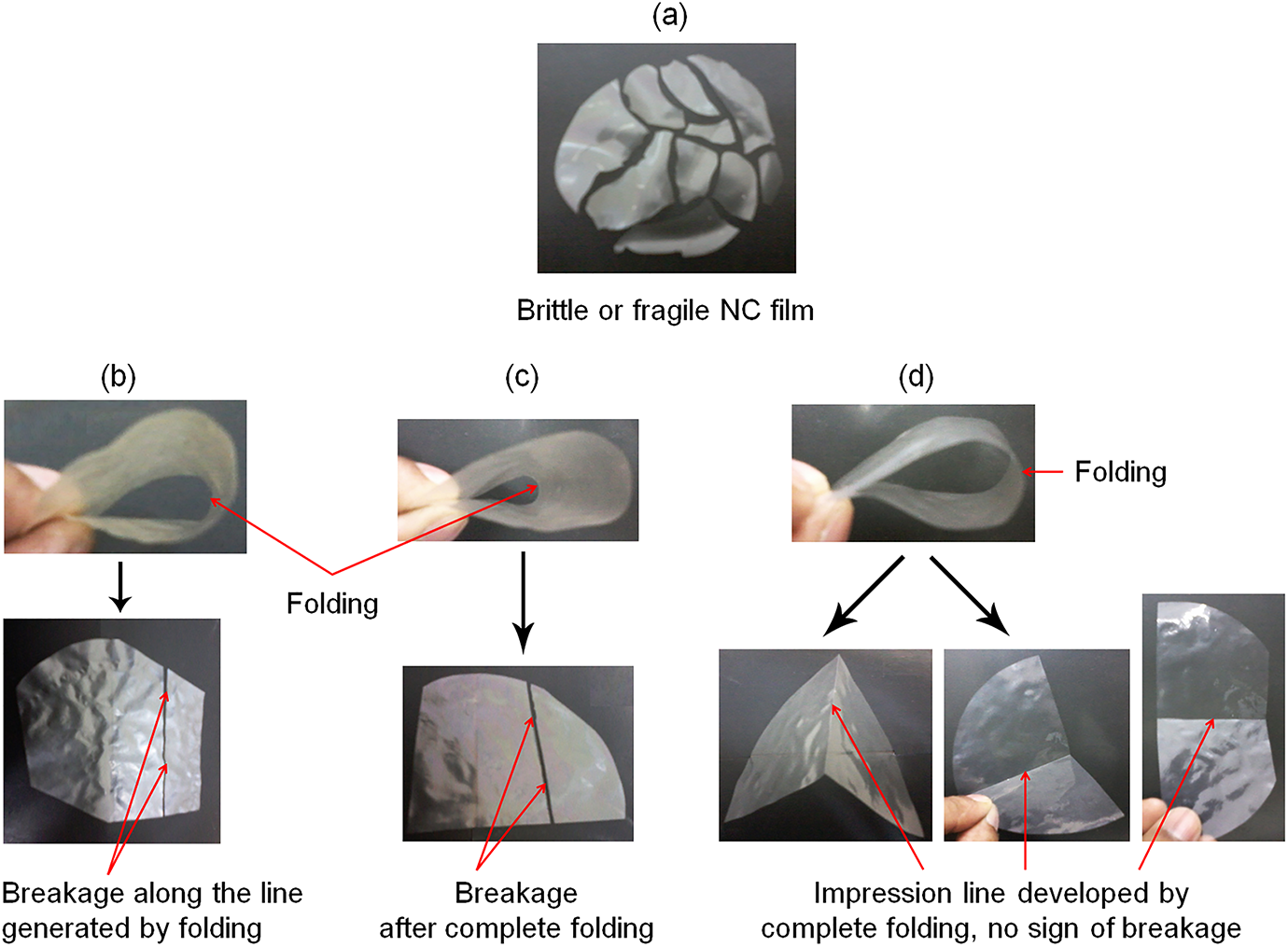
Fold endurance test of (a) NC film, (b) NC/CMC (90:10), (c) NC/CMC (80:20), and (d) NC/CMC (70:30) blend films. NC: nanocellulose; CMC: carboxymethyl cellulose.
While attempting to fold the NC (neat) film, it broke off immediately and it was not possible to fold the film (even for a single occasion) along a fixed line at all as shown in Figure 1(a). It was found that the films from 90:10 and 80:20 blends of NC/CMC could not also withstand the stress of folding and broke off immediately along the line of fold as shown in in Figure 1(b) and (c), respectively. It was only the 70:30 blend film (70% NC and 30% CMC; Figure 1(d)) that could withstand the force of folding and did not break even after repeated flexing along the same line of fold. Thus, it can possibly be commented that CMC can accommodate a maximum of 70% of its weight of NC while still maintaining a reasonably good flexibility with an enhanced strength. So, the improvement in flexibility with increasing proportion of CMC (40 and 50 wt%) is quite evident, and it can be concluded that NC can be incorporated in CMC to the maximum extent of 70 wt% to get an integrated flexible and workable film. In contrast to the normal practice of investigating the influence of a filler in a matrix from lower to higher doses, an attempt has been made to do the same in a reverse manner to find the optimum quantity of highly crystalline NC that can be incorporated in flexible CMC such that the composite film just attains high TS and good pliability.
FTIR spectroscopy analysis
The FTIR spectrum of NC is shown in Figure 2. A broad absorption band at 3278 cm−1 is evident and is attributed to the stretching frequency of the –OH group. A band at 2905 cm−1 is attributed to C–H stretching vibration. The bands around 1425 and 1337 cm−1 are assigned to –CH2– bending in plane and C–OH bending vibration, respectively. The band at 1020 cm−1 is due to C–O–C asymmetric bridge stretching.
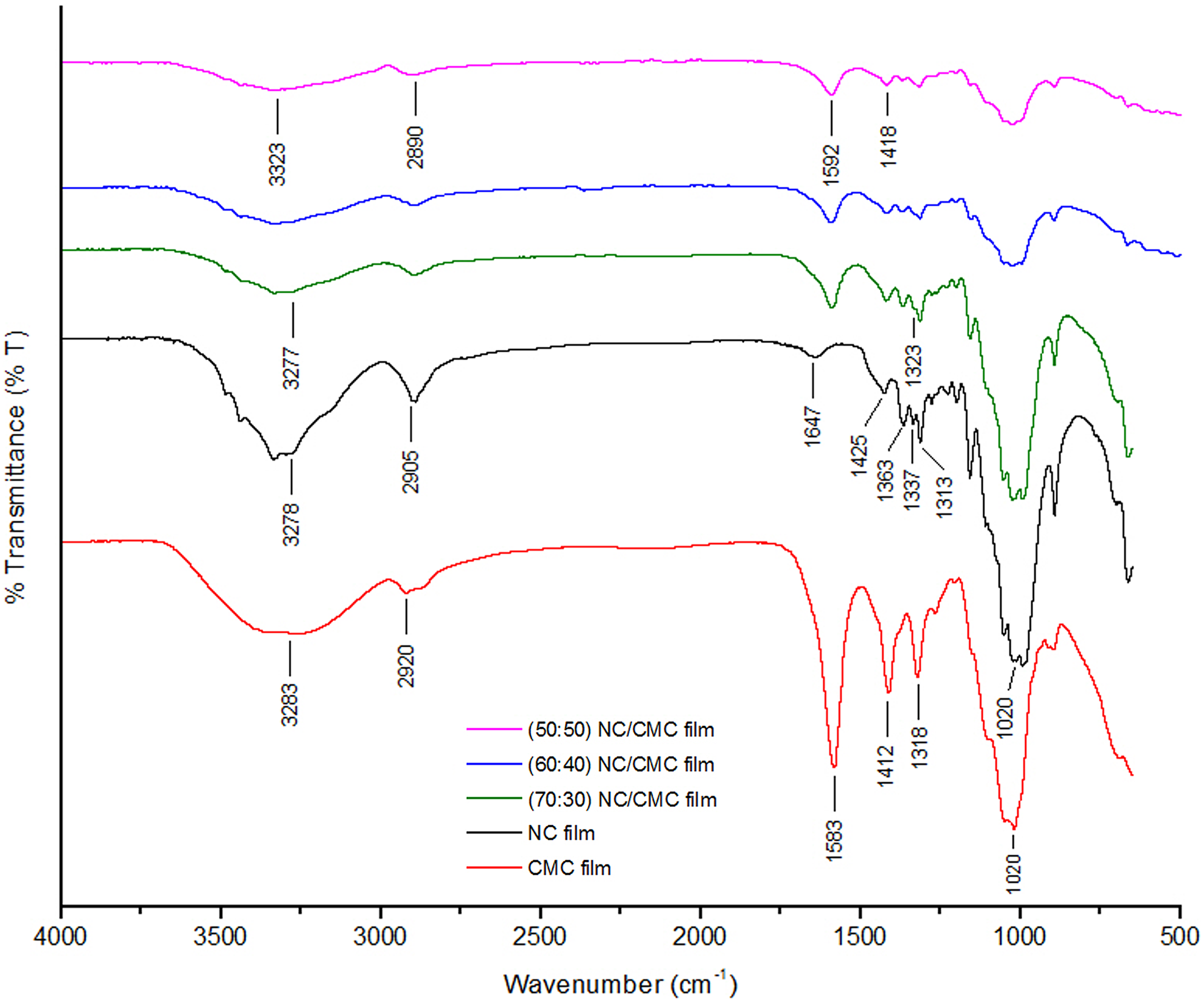
FTIR spectra of CMC film, NC film, and their different composite films of various ratios (NC/CMC ratio is 70:30, 60:40, and 50:50). FTIR: Fourier transform infrared; NC: nanocellulose; CMC: carboxymethyl cellulose.
The FTIR spectrum of a CMC film is shown in Figure 2. The broad absorption band at 3283 cm−1 is due to the stretching frequency of the –COO– group which overlaps with the –OH stretching region at 3480–3440 cm−1. The band at 2920 cm−1 is due to the C–H stretching vibration. The bands around 1412 and 1318 cm−1 are assigned to –CH2– scissoring and –OH bending vibration, respectively. The band at 1020 cm−1 is due to CH–O–CH2 stretching. A spectrum of CMC film shows peaks around 1583 cm−1 and is attributed to a symmetric vibration of COO– groups.
The blending of CMC with NC results in films with similar characteristics in the FTIR spectra as that of NC which has almost a similar chemical structure to that of CMC. The spectra of different blends containing different proportions of NC (under study) are shown in the same Figure 2. This analysis was attempted to characterize the chemical influence of incorporation of CMC in NC and distinguish the FTIR bands and vibrations shifts related to such NC incorporations. Blending CMC with NC (NC/CMC is 70:30) causes minimal shifting (1583–1592 cm−1) excepting the increased absorption in the COO– band around 1592 cm−1 in composite films. This indicates that the asymmetric and symmetric vibrations of C=O and C–O bonds are enhanced. It is interesting to note that for 60:40 and 50:50 NC/CMC films, the absorption in the COO– band around 1592 cm−1 is quite low with respect to that of neat CMC film. This is due to the presence of free –COOH groups which are not participating in the hydrogen bonding. The water absorption band of NC at 1647 cm−1 disappears upon addition of CMC, that is, in NC/CMC composite films of different compositions. The broadband located at around 3278 cm−1 appears in all films and is attributed to O–H stretching and intra- or intermolecular hydrogen bonds. The O–H stretching vibrations for 70:30, 60:40, and 50:50 NC/CMC blends films occur at 3277, 3292, and 3323 cm−1, respectively. By blending CMC with NC, the O–H stretching of the resulting films shifts to higher wavenumbers. This shift indicates that the hydrogen bonds involving the –OH group in the blends are weaker as the NC decreases from 70:30 to 50:50 NC/CMC composite films compared to the neat polymer films. 17,18 The band of C–OH bending of NC film that appears at 1337 cm−1 is shifted to 1323 cm−1 with CMC addition.
Mechanical properties of NC/CMC nanocomposite films
The mechanical properties, such as TS, % EB, elastic modulus, and toughness of neat CMC film and its composite films with various NC loadings are shown in Figure 3(a) to (d), respectively. Figure 3(e) shows the typical stress–strain curves of all the films under study.
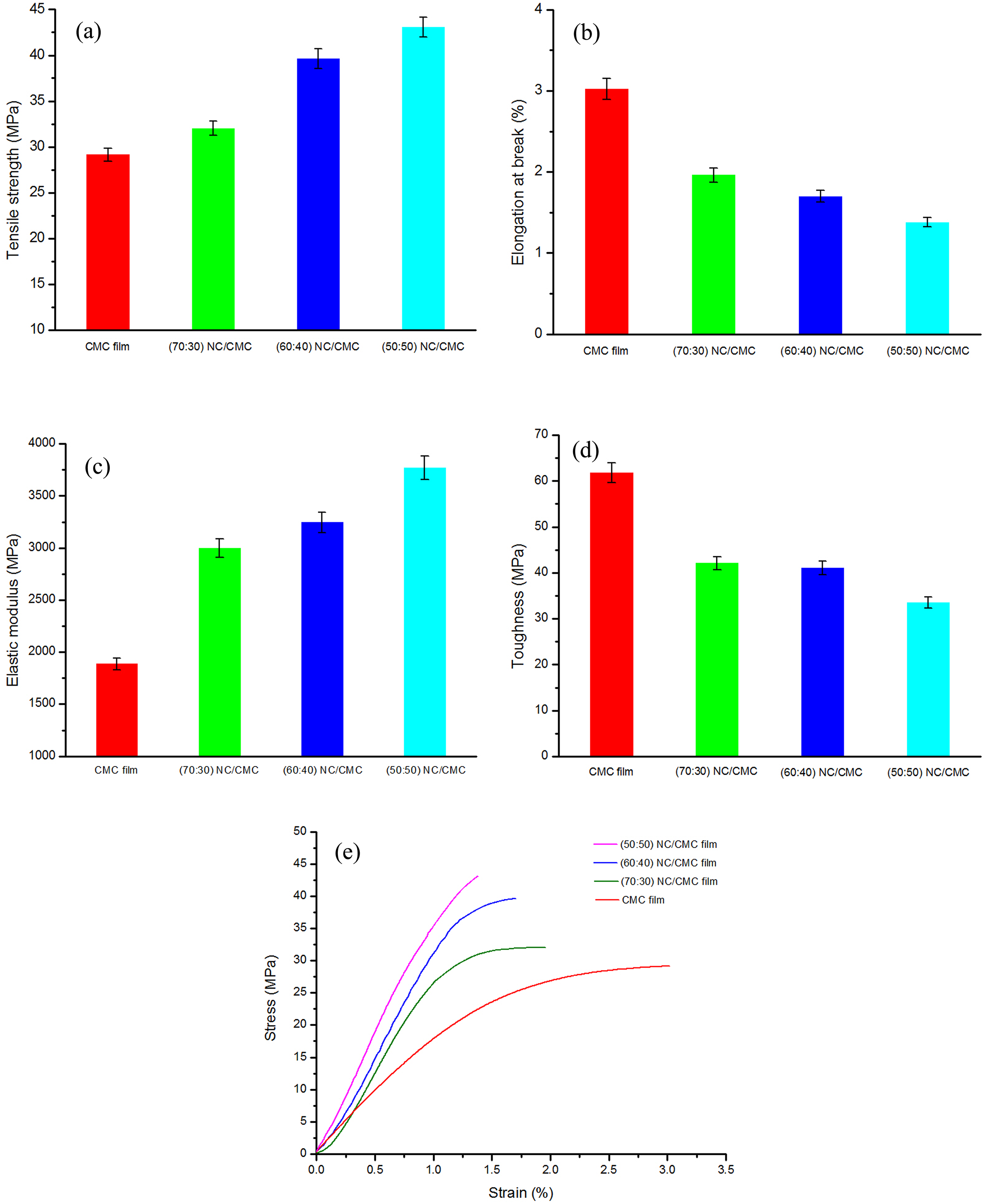
Effect of NC on the (a) tensile strength, (b) % elongation at break, (c) elastic modulus, and (d) toughness of NC/CMC (70:30, 60:40, and 50:50) composite films. (e) Stress–strain curves of CMC film and NC/CMC (70:30, 60:40, and 50:50) composite films. NC: nanocellulose; CMC: carboxymethyl cellulose.
It will possibly be worth mentioning here that although films with neat NC could be fabricated, it was not possible to carry out tensile testing with it, as it was too brittle in nature as has been revealed by fold endurance test (Figure 1(a)). It was, however, observed that a maximum of 70 wt% of NC could be incorporated in CMC so as to obtain a processable and workable film that can be handled easily.
As can be seen from Figure 3(a) and (b), the TS and % EB for CMC films were 29.14 MPa and 3.02%, respectively. Incorporation of NC causes a significant increase in TS as expected, and a consequent decrease in % EB with respect to neat CMC. The increasing TS of the NC-reinforced CMC nanocomposite films is likely to be attributed to the formation of intermolecular interaction between NC and CMC (no ester formation). 19 During the processing and drying the composite films immediately after casting, the inherent hydrogen bonds (both intra- and intermolecular) normally present in NC nanoparticles are likely to be replaced by new intermolecular hydrogen bonds formed between the hydroxyl groups in NC molecules and the carboxyl groups, and free hydroxyl groups in CMC. 20 This intermolecular interaction between NC and CMC appears to result in increase in TS 19 which is quite apparent in the bar diagram. This finding corroborates the FTIR results reported. Moreover, with increasing proportions of NC particles, the highly active nanoparticles might be expected to extend strong adsorptive forces on the CMC surface and enable it to offer greater resistive forces while attempts are made to deform the film.
The NC films are normally found to be brittle and disintegrate into pieces when dry and cannot be subjected to any mechanical testing as such. However, NC suspensions have strong interfibrillar interactions and high shearing forces are needed to break the flocks formed of fibrils due to high surface energy of the nanoparticles. Another purpose of incorporation of CMC, besides enabling NC to form a pliable film is to reduce the fibrillar interactions, the hypothesis being that the resulting ionic repulsion between fibrils (due to the presence of COO– ion) would make the fibril orientation possible with moderate shearing. 21
Incorporation of NC causes a significant increase in TS. With 70:30 of NC/CMC, the TS of the composite films increases by almost 10%. On the other hand, 60:40 and 50:50 NC/CMC contents raise the TS of the films by 36% and 48%, respectively. The increasing trend in TS with decreasing proportions of NC in the range of concentration under study is quite noteworthy. The possibility of intramolecular strong association of nanoparticles reduces quite effectively the number of free NC particles which can be accessible for interaction with CMC when the concentration of nanoparticles is high as can be expected in NC/CMC::70/30 ratio. Thus, the availability of free NC particles as its concentration goes down may be accounted for the observed increase in TS with reduction in NC content. With an increasing NC percentage, there is every possibility of agglomeration of the particles amongst itself. The NC particles being highly surface active they undergo coalescent very easily and the possibility of its interaction with CMC gets reduced. When the concentration of NC in the system is in the lower range of concentration under study, the CMC particles have the opportunity of availing the free spaces between the NC particles and get positioned in between them such that the highly active nanoparticles cannot aggregate amongst itself. The NC particles thus become more effective in modifying the properties of the ultimate composite. Thus, the interparticle interaction between CMC and NC increases which leads to enhancement in TS.
The neat CMC film exhibits an elastic modulus of 1.88 GPa. For NC/CMC composite films, a significant increase in elastic modulus after the addition of NC into CMC polymer is observed. This can be attributed to the same reason as explained earlier. The elastic modulus of the NC/CMC composites increases by about 100% (from 1.88 GPa to 3.77 GPa) when 50:50 ratio of NC/CMC is taken. Here again, like TS, the elastic modulus also undergoes an increase with decreasing proportion of NC, an observation which corroborates our earlier finding.
The stress–strain behavior of all the samples under study has been summarized in Figure 3(e) which represents more and more brittle characteristics of the composites with decreasing proportions of NC within the range studied. As the neat CMC film has the lowest TS and highest % EB, it may possibly be commented that the composites with 60–70% of NC give an optimum combination of strength and toughness.
Thermal properties
Thermogravimetric analysis
Figure 4 shows the TGA curves of the neat NC, neat CMC film samples, and their different blend films where the NC/CMC ratio varies from 70:30 to 50:50.
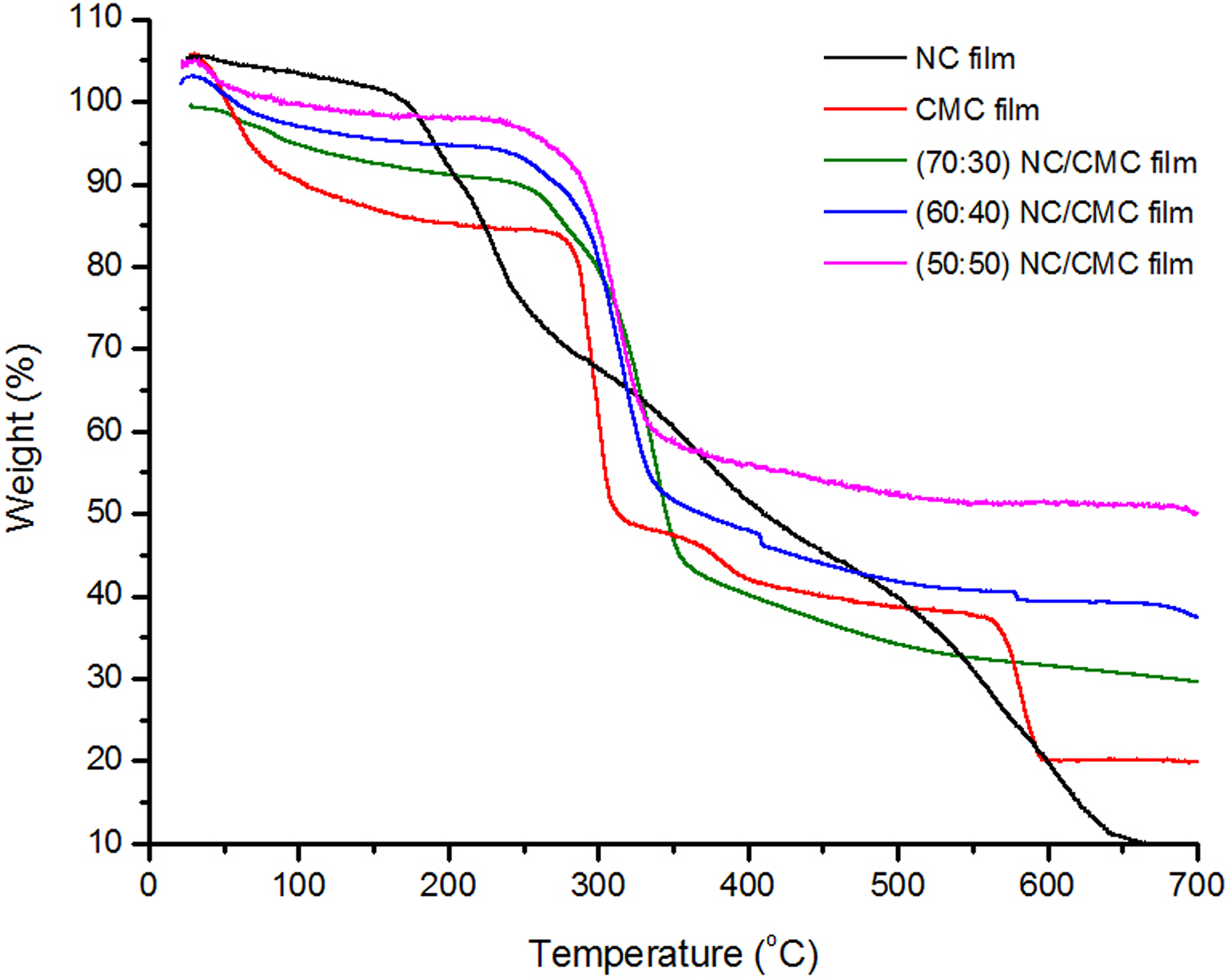
TGA thermogram of CMC film, NC film, and NC/CMC (70:30, 60:40, and 50:50) composite films. TGA: thermogravimetric analysis; NC: nanocellulose; CMC: carboxymethyl cellulose.
The early mass loss due to the evaporation of moisture as the temperature rises for all the samples under investigation is explicitly revealed in the figure. The mode of moisture loss is also confirmed in the DSC studies discussed in the subsequent section, NC shows the least amount of moisture loss due to greater extent of hydrogen bonding and tightly held water molecules, and CMC the highest one while the blends display intermediate behavior. The CMC being much less crystalline and having more space to retain trapped water molecules (due to more hydrophilic character) shows higher weight losses in the early stages of degradation. CMC also shows the fastest rate of degradation after the onset has taken place (beyond moisture loss). At temperatures 280–300°C, about 50% loss in weight occurs for CMC because of the emanation of carbon dioxide and acetic acid. 22 Subsequently, a secondary degradation occurs at a very slow rate as the temperature increases from 300°C to 600°C. In this region, it may be expected that after decarboxylation, CMC undergoes changes in structure and tends to be crystalline as that of NC and thus acquiring higher thermal stability. Then, it of course follows the degradation pattern of NC although retaining a little higher percentage of residues than NC. NC on the other hand undergoes slow and continuous degradation with increase in temperature over the entire range of temperature under study. On blending CMC with NC (30:70, 40:60, and 50:50), the rate of degradation becomes somewhat less than that of CMC itself. However, the aforesaid blends still degrade at a reasonably faster rate than the neat NC. The blends among themselves show very little differences in their courses of degradation.
It is interesting to note that toward the last step of degradation, that is, in the temperature range of 500–650°C, the thermal stabilities of the blends are much higher than either neat NC or neat CMC. The blends exhibit about 30, 40, and 50% of residues for 70:30, 60:40, and 50:50 NC/CMC composite films, respectively, in contrast to approximately 10 wt% of neat NC and 20 wt% of CMC. This again corroborates the earlier observations of the possibility of more and more hydrogen bonding of CMC with NC particles as its content decreases within the studied range, as otherwise there is more and more association within the NC particles as the content of NC increases. The increased hydrogen bonding between NC and CMC may be considered as the formation of pseudo crosslinks among the particles whereby the rigidity of the blended film is enhanced. Thus, this apparent crosslinks lead to the insensitivity toward heat within the range of temperature under study. This may possibly be attributed to the formation of increased proportion of residues.
DSC study
The DSC thermograms of neat NC, neat CMC film, and CMC film reinforced with various loading of NC (70:30, 60:40, and 50:50 NC/CMC compositions) are shown in Figure 5.
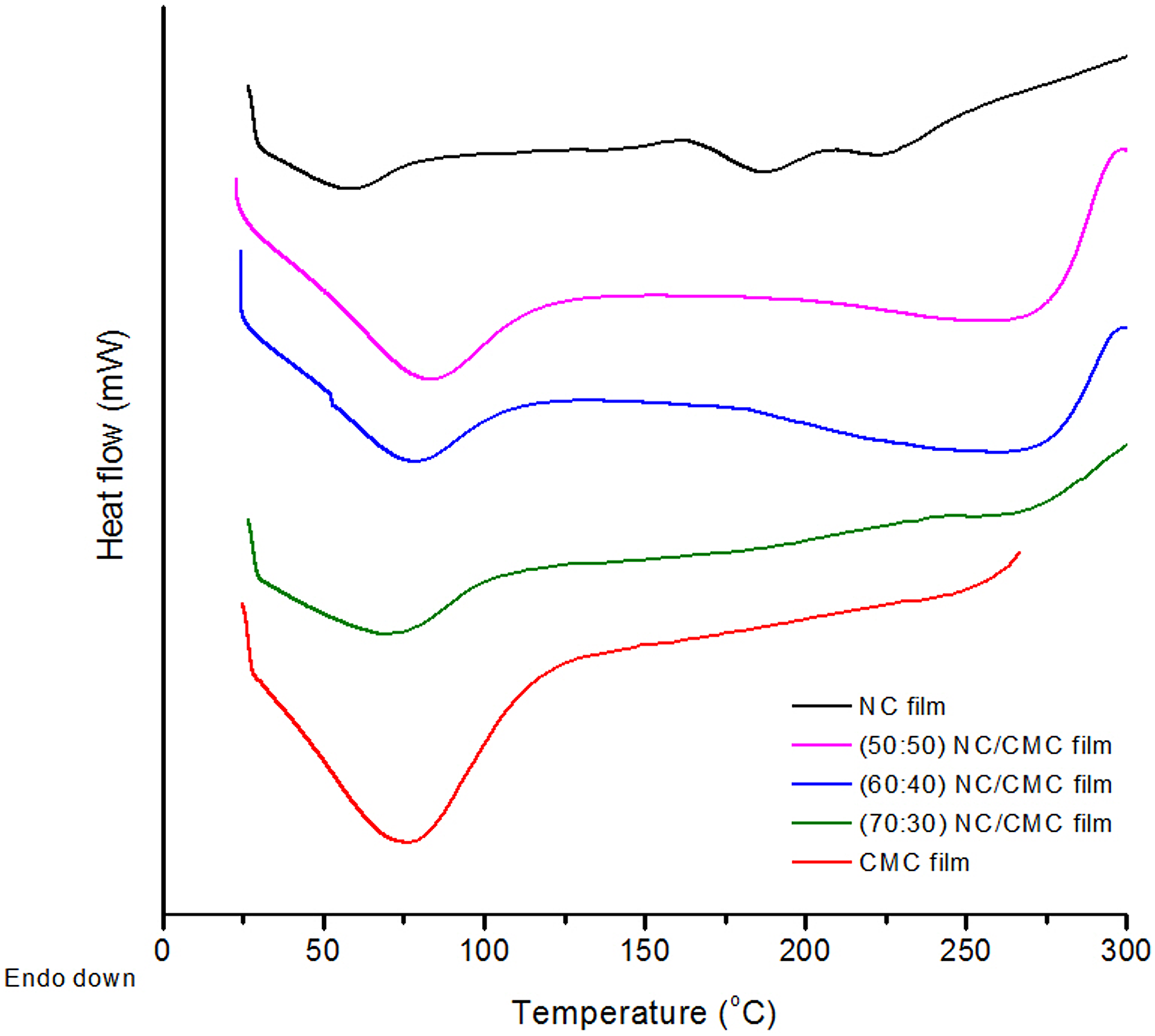
DSC thermogram of CMC film, NC film, and their different composite films (NC/CMC ratio is 70:30, 60:40, and 50:50). DSC: differential scanning calorimetry; NC: nanocellulose; CMC: carboxymethyl cellulose.
The initial endotherm, which is obtained in all the five cases at temperatures lower than 100°C, accounts for the loss of moisture due to evaporation. This mode of moisture loss is also supported by the TGA studies of the corresponding samples. The temperature ranges, as indicated by the width of the endotherms, however, vary in all the five cases.
In CMC, the presence of both hydroxyl and carboxylate anions enhances the affinity toward absorption of moisture. The adsorptive forces retaining the water molecules also undergo tremendous variation. This probably causes the maximum width of the endothermic peak characteristic of CMC among the different films under study. In the case of NC film, the incipience of moisture loss occurs early and the width is relatively narrower with respect to that of CMC. NC film (brittle in nature) attracts water molecules through the surface hydroxyl groups only and hence exerts relatively homogeneous adsorptive forces in holding them, leading to a shallow endotherm. On infusing NC into the CMC film (to the extent of 70 wt%) so that a soft and pliable film can be obtained, we can find an intermediate behavior of the endotherms in the resulting blends. The endotherm of NC now broadens as expected.
Toward the higher range of temperatures under study, the NC film shows two successive small endotherms. These two endotherms differ very little in their temperature ranges and indicate the heats of fusion of two crystal sizes which might have differed in their crystal sizes and orientation. These are sometimes attributed to cellulose II and cellulose I developed during the synthesis of NC by acid hydrolysis. They, however, differ in their crystal planes. The crystalline melting ranges as were observed with neat NC, however, almost disappear when blended with amorphous CMC. The reinforcement of CMC with NC also increases the resistance to thermal degradation as indicated by the absence of any exotherm (typical of CMC) of the resulting blend which exhibits an insignificant endotherm in the neighborhood of 250°C.
XRD studies
In order to analyze the crystallinity as well as the crystallite size of the NC films, CMC films, and NC/CMC (70:30, 60:40, and 50:50) composite films, X-ray diffractometry was carried out. The results (Figure 6) from films containing CMC show complete amorphosity compared to NC.
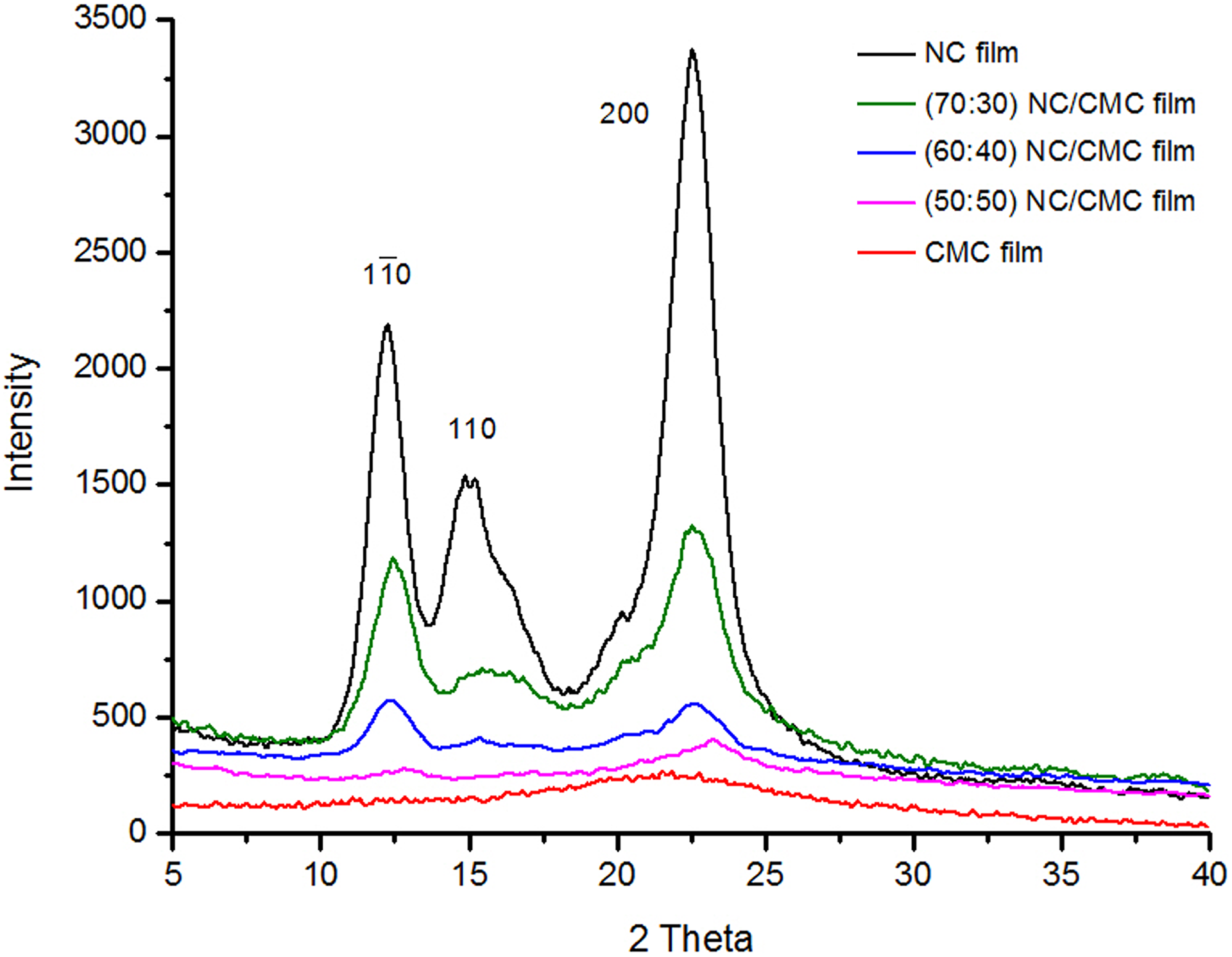
XRD patterns of CMC film, NC film, and NC/CMC composite films of various ratios (70:30, 60:40, and 50:50). XRD: X-ray diffraction; NC: nanocellulose; CMC: carboxymethyl cellulose.
The XRD patterns were considered within the 2θ range of 5°–40°. The XRD spectrum of neat NC film shows a characteristics strong and sharp peak at 2θ = 22.5° (200 plane), corresponding to a basal spacing (d-spacing) and crystallite dimension of 3.94 Å and 11.85 nm, respectively. After loading NC (70 wt%) in the CMC matrix (i.e. NC/CMC composite film), the intensity of the strong peak of NC at 22.5° (3.93 Å) has decreased as expected. Simultaneously, the intensity of the other diffraction peaks corresponding to other crystal planes of NC was also reduced to a great extent with almost no change in their 2θ values. It can be clearly seen from Figure 6 that a broadened peak at 21.56° indicates the existence of an amorphous structure corresponding to CMC film. The orientation of the films may be due to relaxation phenomena during drying of the films, because it has been shown that relaxation after shearing depends on particle size. 23 The crystallinity and crystallite size of NC, CMC, and NC/CMC composite films are analyzed by XRD and have been shown in Table 1.
CI, crystallite size, and d-spacing of CMC, NC, and NC/CMC (70:30, 60:40, and 50:50) composite films.
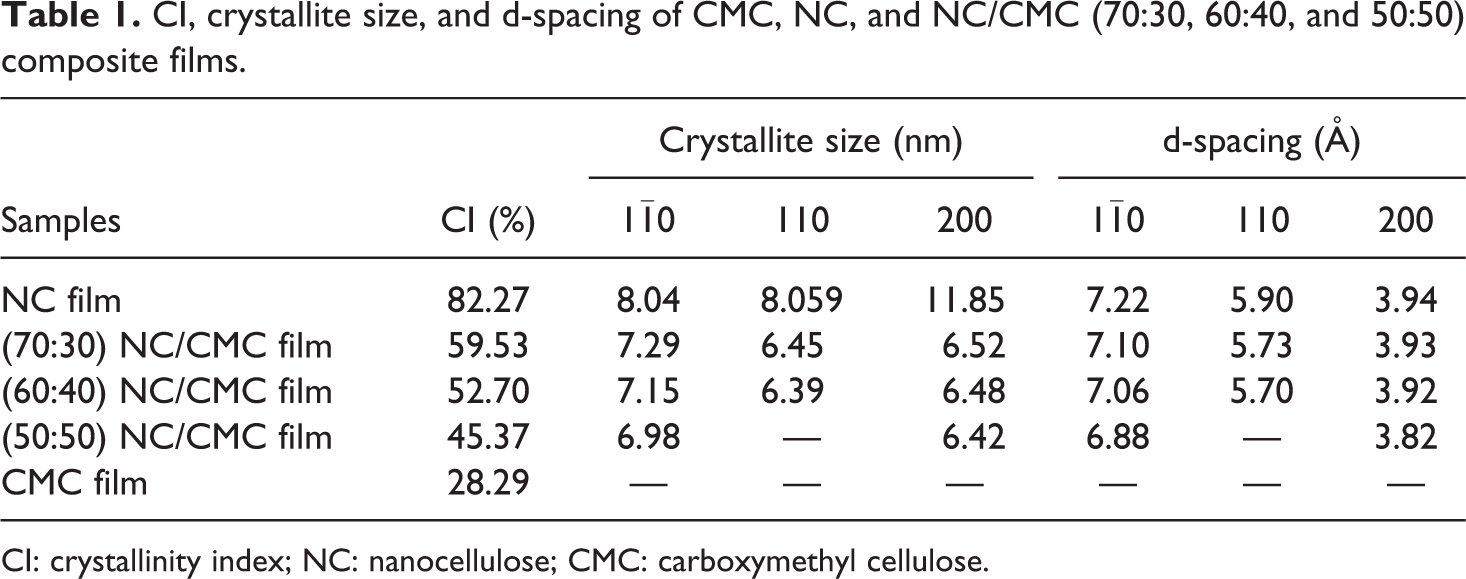
CI: crystallinity index; NC: nanocellulose; CMC: carboxymethyl cellulose.
The result of XRD analysis shows that crystallinity undergoes sharp decrease in NC/CMC composite films. CMC is an amorphous material with a % crystallinity index (CI) of 28.29 only. NC (% CI 82.27) shows three distinct crystal planes
MVTR studies
Figure 7(a) shows the moisture absorption characteristics of neat CMC film and NC/CMC (70:30, 60:40, and 50:50) composite films. The study was undertaken by the PATRA method. 16
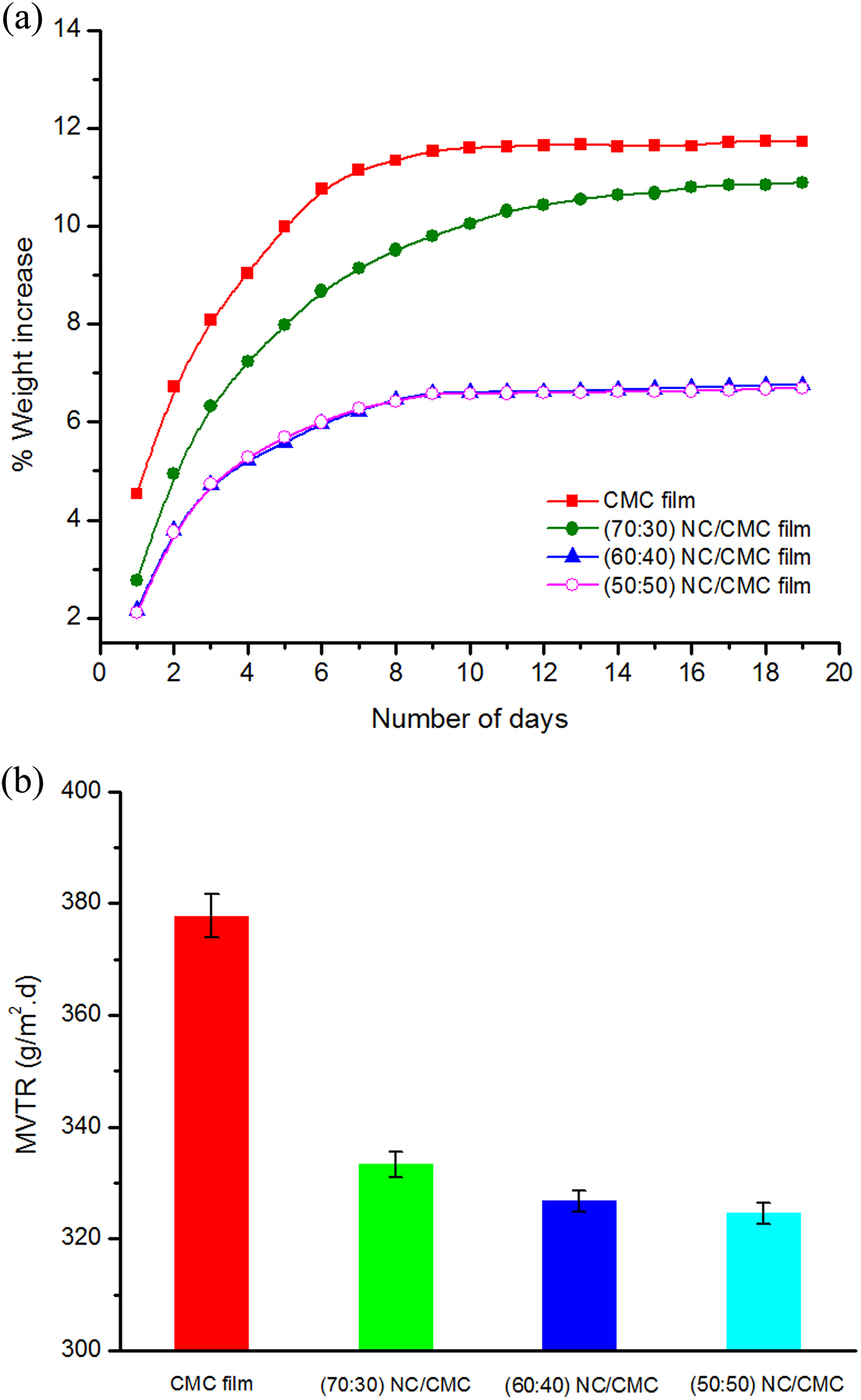
(a) Moisture absorption characteristics of CMC and NC/CMC composite films. (b) MVTR analysis of CMC film and NC/CMC (70:30, 60:40, and 50:50) composite films. MVTR: moisture vapor transmission rate; NC: nanocellulose; CMC: carboxymethyl cellulose.
The investigation was carried out over a period of 20 days. The moisture uptake by the different samples has been expressed in the ordinate as wt% increase while the abscissa denotes the number of days. The rate of moisture absorption has been calculated from the slope of the linear part of the individual plot while all of the curves exhibit a tendency to become asymptotic beyond the optimum amount of moisture uptake.
From Figure 7(b), it is quite apparent that the values of MVTR decrease sharply with the presence of NC (70 wt%) in the films of CMC. Indeed, the MVTR value of neat CMC film (without NC) was 378 g m−2·d at 100% RH. On the other hand, MVTR value of linear NC/CMC composite film (NC: CMC::70:30) was 333 g m−2·d at the same RH. Thus, an approximately 14% reduction of MVTR was obtained with 70 wt% NC in NC/CMC composite films. CMC is a hydrophilic material, and it strongly interacts with water molecules and gets saturated and thus causing an increase in the moisture vapor transmission through the film subsequently. Reduction in the MVTR with the incorporation of NC results in an improvement of the functional properties of these films particularly in those cases where barrier characteristics are of major importance.
However, NC particles possessing high specific surface area and surface activity might have been involved in interaction either through surface adsorption or hydrogen bonding with CMC. This arrangement enhances the tortuosity of the path of moisture mobility in the NC/CMC films, leading to slower diffusion process and hence to a lower transmission rate. 24 In addition, the interactions of NC with CMC films as well as the interactions among the nanofibrils may have enhanced the moisture vapor barrier properties. 25
From Figure 7(a), it is observed that the NC/CMC composite film allows free accessible carboxyl groups of CMC being involved in extensive secondary valence forces like hydrogen bonding with the neighboring NC particles or within itself and thus offering resistances to the diffusion of moisture. It is observed from Figure 7(a) and (b), respectively, that the % weight increase due to moisture absorption decreases substantially in going from 70:30:: NC: CMC composite film to 50:50:: NC: CMC composite film. This may possibly be attributed to the fact that the lower percentage of NC might offer a greater surface area (because of lower extent of association) for interaction with CMC as observed earlier. However, the 60:40 and 50:50 ratios of NC/CMC composite films differ very little in their moisture absorption characteristics.
Morphological studies
SEM analysis
The surface morphologies of the fractured surfaces of neat CMC, neat NC, and representative NC/CMC composite films have been shown in Figure 8(a) to (c), respectively.

Scanning electron microscopic images of (a) CMC film, (b) NC film, and (c) (70:30) NC/CMC composite film. NC: nanocellulose; CMC: carboxymethyl cellulose.
The micrograph of neat CMC demonstrates a homogeneous and uniform surface devoid of any structural defects. Neat NC on the other hand depicts an irregularly fractured surface probably because of its extreme brittle nature. A random distribution of cracks and crevices is observed sometimes along with pores of different shapes and sizes to pervade the entire surface. Some phase separated aggregates of NC are quite apparent. However, the fibrillar NC particles of different aspect ratios are also quite apparent. In a combination of 70% NC and 30% of CMC, we can find a well-balanced combination of characteristics pertaining to neat CMC and neat NC, respectively. From the three different SEM images, some idea of agglomeration can be made. In the SEM image of neat NC (Figure 8(b)), there is clear evidence of phase separation of clustered NC particles (white phase). In the case of composite 70% NC and 30% CMC (Figure 8(c)), this extent of agglomeration is drastically reduced. This proves that decreased proportion of NC leads to better interaction and hence the enhanced mechanical properties. It will possibly be worth mentioning here that it was not possible to achieve a reasonably flexible and workable film below 30% of CMC. The ductile behavior of 70 wt% NC/CMC composite film is quite distinguishable when compared to the SEM image of neat NC film. The brittle fracture along the fibrillar length and the asperities developed on the surface of NC in the forms of voids and cracks are no longer largely apparent. On the other hand, the fractured surface of NC/CMC film is relatively more smooth and devoid of irregularities on its surface. Thus, the flexibilization of NC by incorporation of CMC is also ensured by its morphology.
Conclusions
NC-reinforced CMC film was synthesized incorporating the optimum (threshold) concentration of NC such that the ultimate composite film could still retain its flexibility and simultaneously offering reasonable strength. This threshold concentration was found to be within the range of 60–70 wt% of NC (solid basis). The film manifested good mechanical, thermal, and barrier properties which can be considered as an indicator for its application in packaging industries. The resulting film may be considered as a tough protecting film for packaging industries. The commercially available conventional synthetic packaging films are nonbiodegradable while this film is obviously susceptible to undergo biodegradation. However, no test on biodegradation has yet been made. These films as they allow the permeation of moisture in a very control manner compared to the synthetic films will be a better alternative for packaging the products which remain fresh and active in a controlled environment of moisture and oxygen. No comparison of the mechanical properties of these films with the synthetic packaging films has been made.
Footnotes
Acknowledgement
The authors hereby acknowledge the assistance from The Calcutta University by the way of research fellowship and various other infrastructures required for the research work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by research fellowship from Calcutta University.


