Abstract
Poly(lactic acid) (PLA) nanocomposites film reinforced with cellulose nanocrystals (CNCs) extracted from sugarcane bagasse fibre (SBF) was prepared by solvent casting method. The CNCs were obtained through alkaline treatment followed by sulphuric acid hydrolysis or phosphoric acid hydrolysis. The aim of this work was to evaluate the feasibility of utilizing phosphoric acid (mild acid) to extract CNC from the SBF. The properties of the CNC were investigated by transmission electron microscopy (TEM), X-ray photoelectron spectroscopy (XPS) and Fourier transform infrared (FTIR) spectroscopy. The thermal properties of the PLA/CNC nanocomposites were characterized using thermogravimetric analyser (TGA). TEM results demonstrated that the CNC having diameter of 5.5–6.2 nm and length of 235–300 nm. Results from XPS characterization have confirmed the existence of both sulphate group and phosphate group in the H2SO4-treated CNC (S-CNC) and H3PO4-treated CNC (P-CNC). FTIR results indicated that the presence of hydrogen bonding and chemical interaction between PLA and CNC. The thermal stability of PLA/P-CNC-10 is higher than that of PLA/S-CNC-10 nanocomposites attributed to the char formation of P-CNC in the PLA matrix.
Introduction
Nowadays, biodegradable polymers derived from natural resources are receiving more and more attention due to increasing environmental awareness. The development of biodegradable polymer originated from the natural resources can be a sustainable alternative to lessen the reliance on petroleum-derived plastics and disposal of plastic waste. Among all the biodegradable aliphatic polyesters, polycaprolactone and poly(lactic acid) (PLA) have successfully reached commercial scale production. 1 –5 The building block of PLA (i.e. lactic acid) can be synthesized from renewable sources through fermentation of agricultural products. 6 The advantages of PLA include good mechanical properties (i.e. high modulus and strength), biocompatibility, high transparency, low toxicity and good processability. 7 However, the major drawback for PLA was its brittleness and low thermal stability which restricted them to be used in some application. 8 The properties of PLA can be improved by incorporation of micro- and /or nanofillers, for example, nanoclay, nano-calcium carbonate, halloysite nanotube and nanocellulose. 9
Cellulose fibre with nanometric dimension has been recommended as a potential load bearing nanofiller for biopolymer. 10 Cellulose nanocrystals (CNC) can be isolated from different plant source or agriculture residue including sugarcane bagasse (SCB), cotton, flax and jute. 11,12 Sugarcane plays an important role as the main source to obtain sugar and this makes SCB to become a residue that is produced in large quantities every year by the sugar and alcohol industries. 13,14 Adequate use of SCB as the sources of CNC gives an added value for this material and at the same time it is a solution for this abundant agriculture waste. 15,16 The incorporation of CNC into PLA matrix could produce a wholly organic bionanocomposite. 17 By the incorporation of nanocellulose, both the thermal and mechanical properties of PLA could be enhanced and eventually optimizing the cost performance of this green material. 9
The most dominant method for production of CNC from SCB was using strong acid hydrolysis. 18 Sulphuric acid is one of the most widely employed acids because of its ability to provide certain degree of grafting for the negatively charged sulphate group onto the surface of CNC that can produce a stable aqueous nanocrystal suspension. However, the presence of this sulphate groups will tend to diminish the thermostability of the CNC. It was found that the higher the content of acid sulphate, the lower the thermal degradation temperature of the CNC. Therefore, the aim of this study was to investigate the feasibility of using H3PO4 (mild acid) to synthesis CNC from SCB, and further the thermal properties of the PLA/CNC nanocomposites were evaluated.
Materials and methods
Materials
PLA (Ingeo™ 3051D) was purchased from Nature Works LLC, Minnetonka, Minnesota, USA. The melt flow index of the PLA was 25 g 10 min−1 (2.16 kg load, 210°C). SCB was obtained from local market (Penang, Malaysia). Sodium hydroxide, acetic acid (glacial), sulphuric acid 95–97%, phosphoric acid 85%, chloroform and sodium chlorite were supplied by Merck (Germany).
Delignification of sugarcane bagasse fibre
Grounded and dried sugarcane bagasse fibre (SBF) was digested at 80°C in a 4% sodium hydroxide solution for 4 h. In order to obtain pH 4, the solution was adjusted by adding 10–15 drops of acetic acid. The bleached cellulose was washed repeatedly, initially with a 5% aqueous sodium hydroxide and subsequently deionized water in order to attain a neutral pH. The obtained cellulose was allowed to dry at room temperature and grounded to get fine powder.
Acid hydrolysis of cellulose by sulphuric acid and phosphoric acid
The delignified and hemicellulose free cellulose was acid hydrolyzed using 6 M of sulphuric acid and 11 M of phosphoric acid, respectively, for 30 min at 55°C under agitation (approximately 175 r min−1) using a mechanical mixer (IKA®, RW 20 Digital Mixer, Wilmington, North Carolina, USA). The resulting mixture was cooled to room temperature and centrifuged. The fractions were continuously washed by the addition of distilled water and centrifuged with 3000 r min−1. The centrifugation process was stopped after at least five washings, while the supernatant liquor became turbid, that is, becoming a colloidal suspension. The colloidal suspension was then allowed to undergo dialysis process using ultrapure water. The dialysis process was carried until the colloidal suspension of pH 4 was achieved. 19
PLA/CNC composites preparation using solvent casting
The obtained CNC were freeze-dried using freeze dryer (Labconco Freeze Dryer System, Kansas City, Missouri, USA). CNC and PLA were dissolved in chloroform in two different beakers. After CNC and PLA were completely dissolved, both solutions were mixed together and poured onto the petri dishes. The PLA/CNC was then dried for 24 h under room temperature. The PLA/CNC composite was prepared by adding 10 wt% of CNC into PLA. The material code PLA/H2SO4-treated CNC (S-CNC)-10 denoted that 10 wt% S-CNC was introduced into PLA, whereas PLA/P-CNC-10 represented that the 10 wt% of H3PO4-treated CNC (P-CNC) was added into the PLA.
Characterization
Transmission electron microscopy
The morphology of P-CNC was examined using energy-filtered transmission electron microscope (EFTEM, Libra 120-Carl Zeiss, Thornwood, New York, USA). A droplet of cellulose diluted by water suspension (0.1% (w/w)) was deposited on 400 square mesh copper grid and allowed to dry for 3 min. The specimens were then negatively stained with 1% uranyl acetate (UO2(CH3COO)2) for 1 min and examined using TEM.
Fourier transform infrared spectroscopy
Fourier transform infrared (FTIR) spectroscopy of the samples were obtained on FTIR spectrometer, Waltham, Massachusetts (Perkin Elmer, USA). The spectra were recorded in a range of 550–4000 cm−1 with a resolution of 4 cm−1. Samples of SBF, extracted sugarcane bagasse microcrystalline celluloses (SB-MCC), CNC obtained from both type of acid and PLA/CNC composites were analysed.
X-ray photoelectron spectroscopy
X-ray photoelectron spectroscopy (XPS) measurements were performed to investigate the surface properties of SB-MCC and CNC. The XPS wide and narrow scan spectra were acquired using X-ray photoelectron spectrometer (Kratos, AXIS Ultra DLD, Manchester, UK) equipped with a aluminium K α X-ray source (1486.6 eV) at 10 mA, 15 kV, analysing a 300 µm × 700 µm area under 4.8 × 10−9 torr ultra-vacuum environment inside the sample analyse chamber using the multichannel plate and delay line detector.
Thermogravimetric analysis
Thermal decomposition behaviour of SBF, SB-MCC, CNC and PLA/CNC nanocomposites (approximately 10 mg) were characterized using thermogravimetric analyser (TGA, Waltham, Massachusetts ) (Perkin Elmer, model Pyris 6, USA) in the temperature range of 30–600°C at a heating rate of 20°C min−1 (under an inert atmosphere of nitrogen gas).
Results and discussion
Morphology analysis
Figure 1 shows the TEM image of P-CNC. Both needle-like CNC and agglomerate can be revealed from the TEM. The needle-like CNC have diameter of 5.5–6.2 nm and length of 235–300 nm. The aspect ratio of these needle-like was about 37–55. This finding is in line with the research done by Teixeira et al. 20 In their work, it was found that the length and diameter of nanowhisker isolated from SCB in the range of 255 ± 55 nm and 4 ± 2 nm, respectively. Nevertheless, the CNC agglomerate revealed from the TEM image could be associated to the intermolecular hydrogen bonding among the cellulose.

TEM picture of P-CNC. TEM: transmission electron microscope; P-CNC: H3PO4-treated cellulose nanocrystals.
FTIR analysis
Figure 2 shows the FTIR spectrum of SBF, alkaline-treated sugarcane bagasse microcrystalline cellulose (SB-MCC) and cellulose nanocrystal isolated by H2SO4 and H3PO4 (S-CNC and P-CNC). By overview of all the peaks for SBF, SB-MCC, S-CNC and P-CNC, it can be seen that all the samples were having similar spectra bands at the range of 897–899 cm−1, 2891–2922 cm−1 and 3337–3341 cm−1. The 3337–3341 cm−1 peaks showed O–H stretching intramolecular hydrogen bonds for cellulose I and 2891–2922 cm−1 was due to the symmetrical and asymmetrical stretching of CH2 and CH. 21,22,23 In addition, the peak at the range of 897–899 cm−1 associated with the β-glycosidic linkages between glucose units in cellulose was found gradually increasing in both cellulose and nanocellulose. 24 However, all samples except SBF have the peak value at 1643 cm−1. This peak value is attributed to water adsorbed in the cellulose molecules. 25
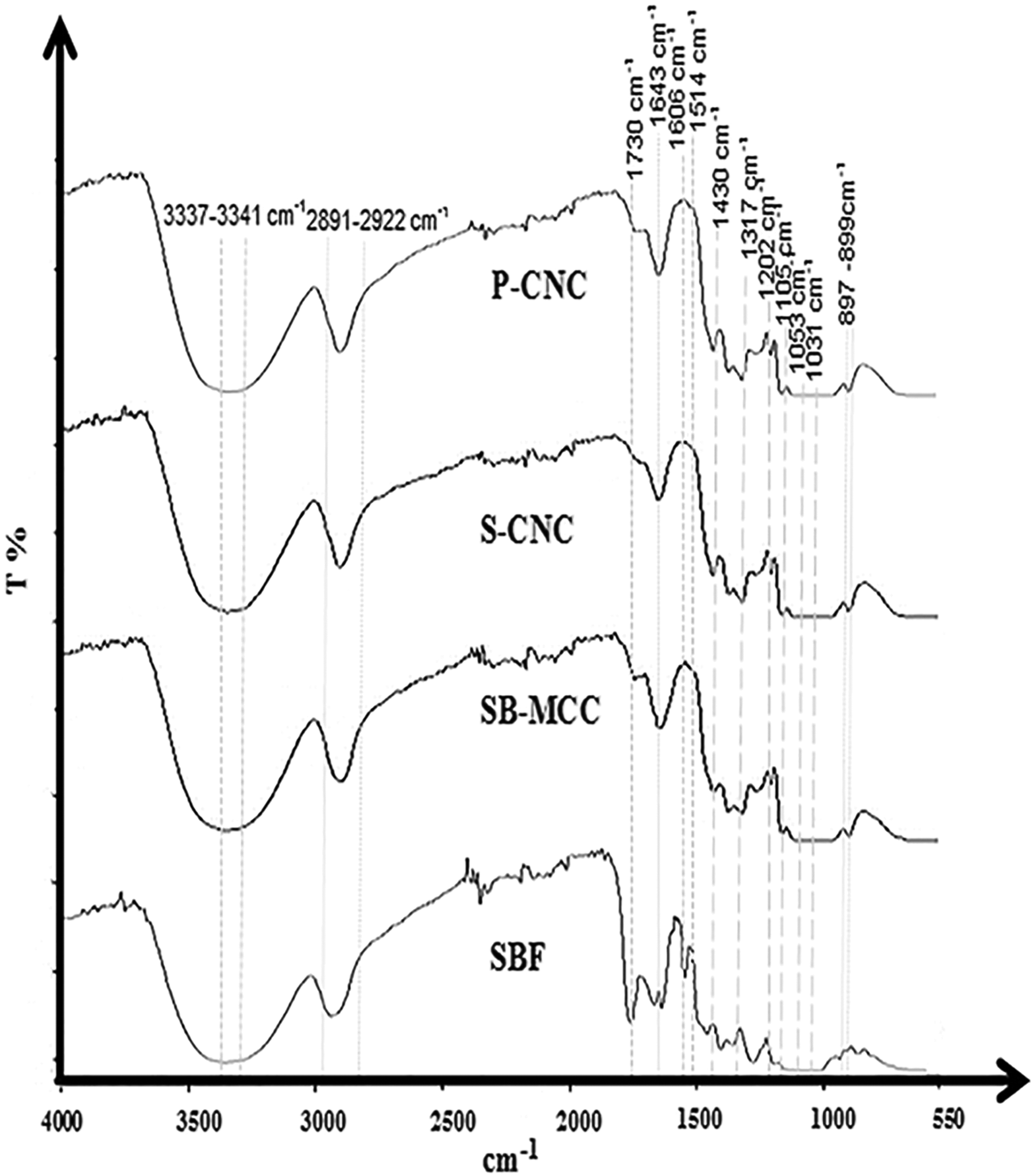
FTIR spectrum of SBF, SB-MCC and CNC hydrolyzed by both sulphuric acid (S-CNC) and phosphoric acid (P-CNC). FTIR: Fourier transform infrared; SBF: sugarcane bagasse fibre; SB-MCC: sugarcane bagasse microcrystalline FTIR peak quantification of sulphate and phosphate functional groups.cellulose; CNC: cellulose nanocrystals.
Based on the FTIR spectrum, there are several peaks in SBF that could not be found in the spectrum of both SB-MCC and acid-isolated CNCs. The disappearance of a few peak in the range of 1514–1606 cm−1 was related to the removal of lignin after the SBF undergoes alkaline treatment and acid hydrolysis processes. This observation was in line with the studies done by Pereira et al., 26 who have found the disappearance of the peak at 1512 cm−1 when the cellulose was extracted from SBF. According to Morán et al, 27 the lignin can be identified by the peaks within the range of 1500–1600 cm−1 corresponding to the aromatic skeletal vibration. Therefore, the observed spectra at 1514 cm−1 was associated to the aromatic C=C in lignin. In addition, due to the presence of functional groups in lignin such as methoxyl –O–CH3, C–O–C and aromatic C=C, absorbance in the region between 1830 cm−1 and 1730 cm−1 also can be observed . The peak 1730 cm−1 corresponds to the C–O stretching vibration for the acetyl and ester linkages in lignin. Besides lignin, the peak at 1730 cm−1 also belongs to hemicellulose. According to Zhang et al, 23 the peak in the range of 1715–1765 cm−1 was corresponding to the C=O stretching vibration for the ketone and carbonyl groups in the hemicellulose. Meanwhile, the peak at 1241 cm−1 corresponded to the stretching vibration mode of the acyl-oxygen and CO–OR is associated with the hemicelluloses. 24 The band observed at 3386 cm−1 seems to be the characteristic of OH groups present in lignin and carbohydrates. 26
The disappearance of peaks between 1500 and 1600 cm−1 in SB-MCC indicated that lignin was successfully removed after undergoing alkaline pre-treatment using sodium hydroxide. Similar finding was reported by Bensah and Mensah 28 The alkaline treatment was used to degrade glycosidic side chains and ester of the SCB, leading to the disruption of the lignin structure, swelling of cellulose and decrystallization. The FTIR results proved that the pretreatment was successful as there was no remaining lignin in the obtained celluloses.
There were many inherent FTIR peaks of CNC observed from the result obtained. From Figure 2, it can be seen that both S-CNC and P-CNC were having same peak values at 1430, 1317, 1053, 1105, 1202 and 1031 cm−1. The peak 1430 cm−1 represented CH2 bending of the intermolecular hydrogen attraction at the C6 group, 25 1317 cm−1 denoted –CH2 wagging, 13 1053 cm−1 was due to the stretching vibration of C–O–C pyranose ring, 1105 cm−1 corresponded for C–O–C glycosidic ether band, 1202 cm−1 associated to –C–OH in plane bending at C6 and 1031 cm−1 was due to the in plane C–OH stretching at C6. 29 Since two different types of acids are used to obtain CNC, the FTIR spectra bands of the CNC were compared in order to identify any significant changes in chemical structure of CNCs due to the substitution of different chemical moieties. The peaks used in FTIR quantification for both sulphuric and phosphoric compound were shown in the Table 1.
FTIR peak quantification of sulphate and phosphate functional groups.
FTIR: Fourier transform infrared.
From Table 1, it can be seen that the peaks used to characterize the presence of both sulphate and phosphate group were actually overlapping. Therefore, it was hard to justify which functional groups were existing in the S-CNC or P-CNC, because the results of FTIR obtained from these two CNCs were having the same peak values in the range of 558–661 cm−1 . 30
The chemical structure analysis of pure PLA and PLA/CNC composite was also carried out in order to study the effect of the introduction of S-CNC and P-CNC into PLA. The FTIR spectra for pure PLA, PLA/S-CNC-10 and PLA/P-CNC-10 were shown in Figure 3. Based on Figure 3, the pure PLA can be characterized by few peaks at 1748, 1455, 1366, 1183, 1129, 1043 and 946 cm−1. The peaks at 1748, 1455 and 1366 cm−1 represented C–O (carbonyl) stretching, asymmetric vibrations of CH3 and C–O–C asymmetric stretching, respectively. Meanwhile the peak 1183 cm−1 represented C–O–C symmetric stretching, 1129 cm−1 was due to the C–OH side group vibrations, 1043 cm−1 was corresponding to the C–H stretching and 946 cm−1 was the C–C vibrations. However, the carbonyl peak for the PLA at 1748 cm−1 shifted to 1753 cm−1 for the PLA/S-CNC-10 and to 1751 cm−1 for PLA/P-CNC-10. The shifting of these peaks indicates the presence of hydrogen bond interactions. 19,31
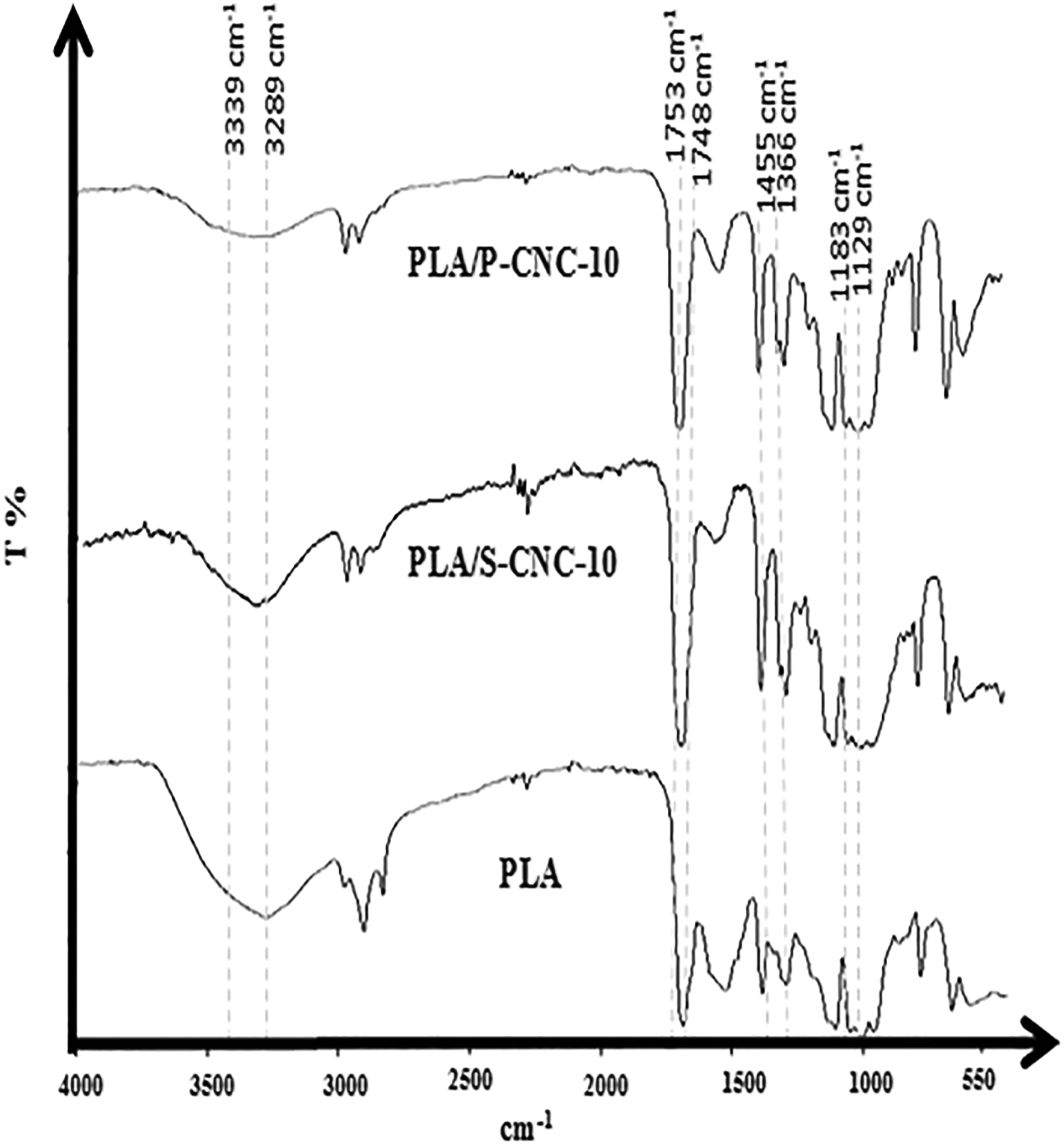
FTIR spectra for pure PLA, PLA/S-CNC-10 and PLA/P-CNC-10 composite. FTIR: Fourier transform infrared; PLA: poly(lactic acid); S-CNC: H2SO4-treated cellulose nanocrystals; P-CNC: H3PO4-treated cellulose nanocrystals.
XPS analysis
XPS measurement was used to investigate the surface chemical analysis of alkaline-treated SB-MCC, S-CNC and P-CNC. The surface compositions of three samples were listed in Table 2. Atomic concentrations of carbon, oxygen, sulphur and phosphate were determined from the XPS spectra of SB-MCC and acid-derived CNCs in order to determine the effect of treatment on the chemical components of the samples. The XPS results showed that the SB-MCC have only the element of C and O on the surface of the cellulose, whereas there is one extra element, which are S and P in both S-CNC and P-CNC, respectively. Since the FTIR result could not justify clearly the existence of both sulphate group and phosphate group, the presence of sulphate group and phosphate group could be evidenced through the XPS result. 32,33 This XPS analysis also indicated successful introduction of sulphate group and phosphate group on the surface of nanocellulose through acid hydrolysis.
XPS analysis of SB-MCC, S-CNC and P-CNC.
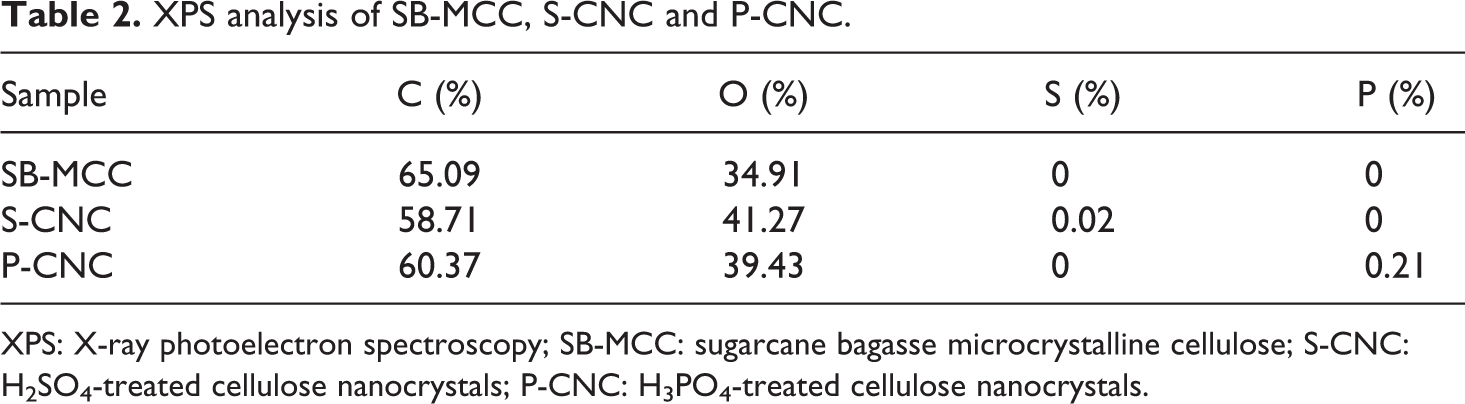
XPS: X-ray photoelectron spectroscopy; SB-MCC: sugarcane bagasse microcrystalline cellulose; S-CNC: H2SO4-treated cellulose nanocrystals; P-CNC: H3PO4-treated cellulose nanocrystals.
Thermogravimetric analysis
The thermal properties of all samples were studied in order to compare the thermal stability of the materials. The onset thermal decomposition (T onset) indicates the beginning of degradation temperature (T d) and the temperature at maximum decomposition (T max) refers to the temperature recorded at the maximum rate of degradation, which are tabulated in both Tables 3 and 4.
TGA thermal characteristics of SBF, SB-MCC, S-CNC and P-CNC.
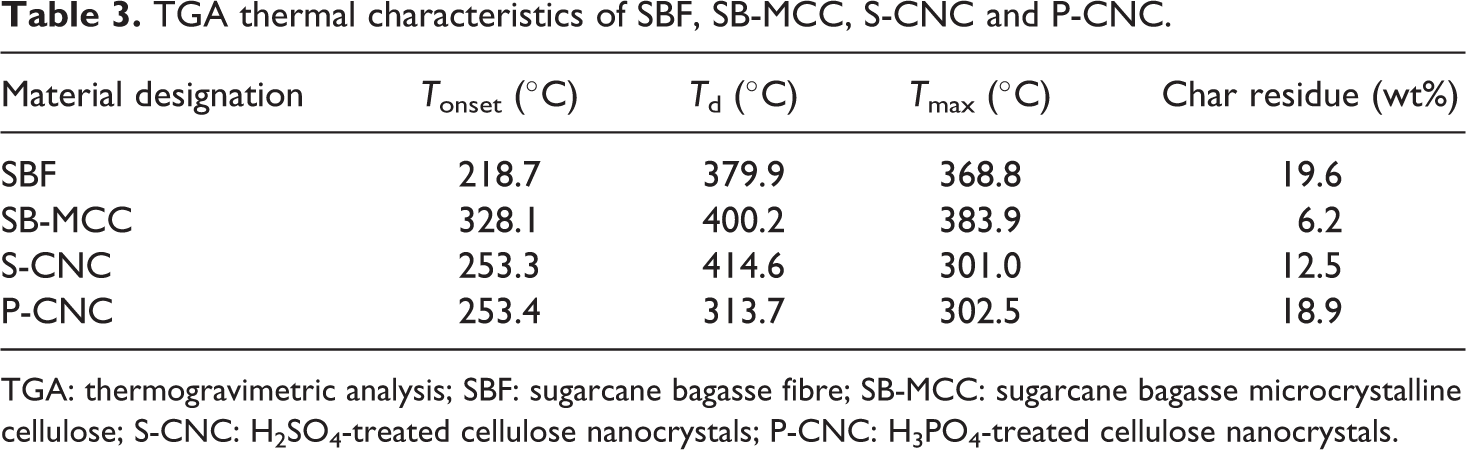
TGA: thermogravimetric analysis; SBF: sugarcane bagasse fibre; SB-MCC: sugarcane bagasse microcrystalline cellulose; S-CNC: H2SO4-treated cellulose nanocrystals; P-CNC: H3PO4-treated cellulose nanocrystals.
TGA thermal characteristic of PLA, PLA/S-CNC-10 and PLA/P-CNC-10 composites.
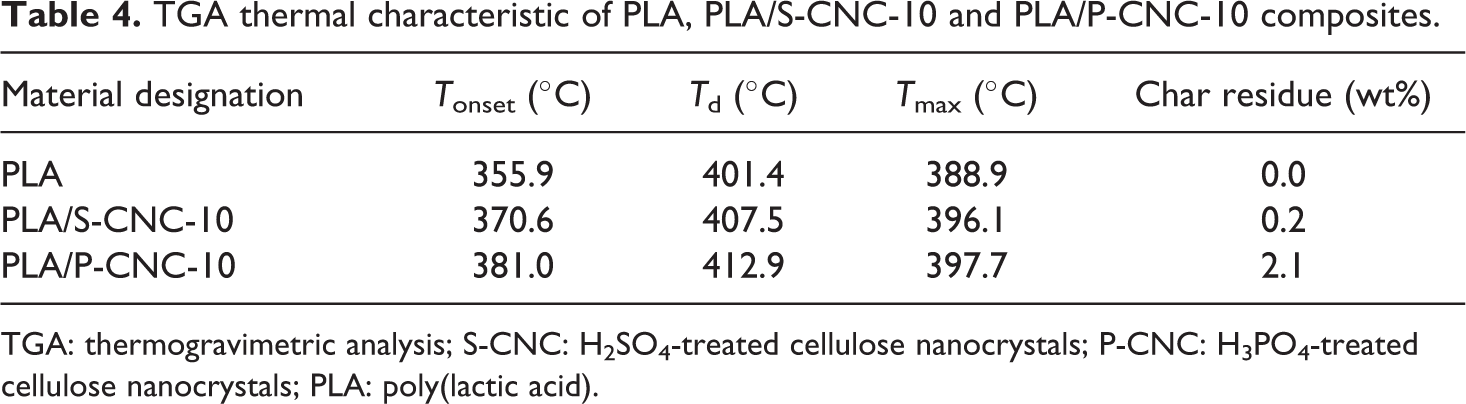
TGA: thermogravimetric analysis; S-CNC: H2SO4-treated cellulose nanocrystals; P-CNC: H3PO4-treated cellulose nanocrystals; PLA: poly(lactic acid).
Figure 4(a) and (b) showed the TGA and Derivative Thermogravimetric (DTG) curves for the SBF, SB-MCC, S-CNC and P-CNC, respectively. By observing TGA curves at Figure 4(a), all of the samples show small weight losses at the temperature range of 60–110°C and this phenomenon was probably due to the evaporation of the water. From Figure 4(a), it can be seen that the T onset of the SBF was recorded at 218.7°C. In Figure 4(b), the SBF was having another three weight loss peak after the water evaporation peak and it showed a main decomposition peak at the temperature of 368.8°C. SCB was having three major components which are lignin, hemicellulose and cellulose, and these three compounds were responsible for these weight loss peaks.
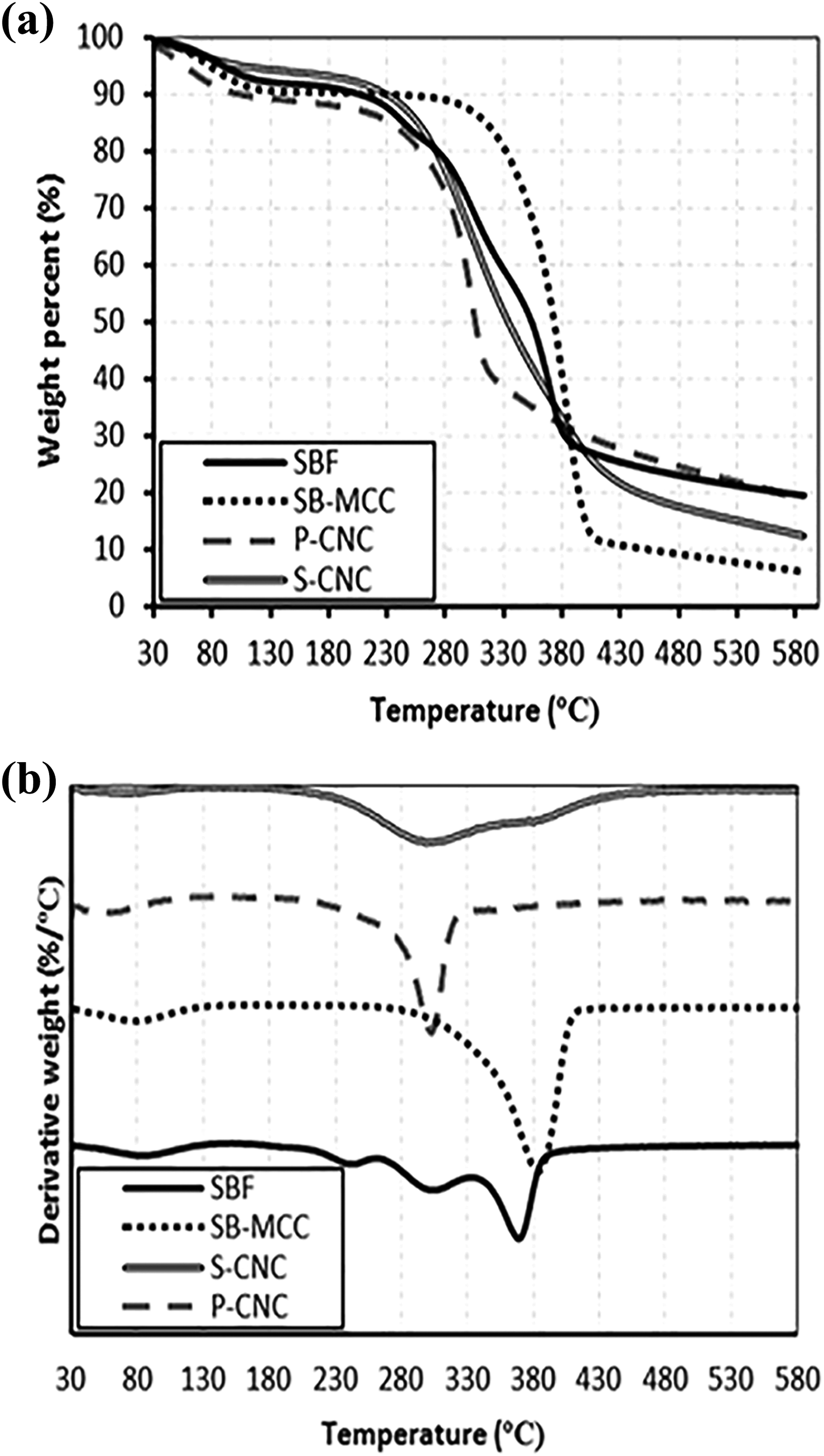
(a) TGA curves and (b) DTG curves of SBF, SB-MCC, S-CNC and P-CNC. DTG: Derivative Thermogravimetric; TGA: thermogravimetric analysis; SB-MCC: sugarcane bagasse microcrystalline cellulose; S-CNC: H2SO4-treated cellulose nanocrystals; P-CNC: H3PO4-treated cellulose nanocrystals.
According to Saelee et al, 13 lignin, hemicellulose and cellulose were having different chemical structures and these had caused them to decompose at different temperatures. The presence of the acetyl groups had made the hemicellulose to have low thermal stability; therefore, hemicellulose will decompose first followed by lignin and cellulose. From Figure 4(b), the second weight loss peak of the DTG graph belonged to hemicellulose. Based on the DTG graph, it showed that the hemicellulose began to degrade at the temperature of 222°C and end at 256.9°C and this observation matched with the result of Abraham et al, 34 who had reported that the hemicellulose was likely to decompose at the temperature of 220°C and continued up to 300°C. It was found that lignin began to decompose at 273.6°C and end at 324.5°C. The cellulose decomposition began at the temperature of 337°C and continued up to 386.2°C. This observation is in line with the finding from Yang et al, 35 who reported that cellulose was having a decomposition range from 310°C to 400°C. Overall, the decomposition of the SB-MCC has few stages, namely depolymerization, dehydration and decomposition of glucosyl units followed by the formation of charred residue. 36
By referring to the TGA curve in Figure 4(a), the SB-MCC was found to have higher T onset than SBF and this was due to the removal of the lignin and hemicellulose by the alkaline treatment using NaOH. According to Kumar et al., 21 the lignin and hemicellulose in the fibre will tend to lower the thermal stability of the SBF. By comparing the DTG curve of SBF and SB-MCC, there was absence of the shoulder peak of hemicellulose and lignin. This again evidenced that the alkaline treatment successfully removed both the lignin and hemicellulose in the SBF and this finding was matched with the FTIR result. Moreover, the absence of lignin and hemicellulose in the SB-MCC also can be reflected by the char residue of SBF. The high char residue of SBF was believed to be contributed by the lignin and hemicellulose.
Based on the TGA curve in Figure 4(a), it was found that both the S-CNC and P-CNC showed a lower T onset compared to SB-MCC. This effect might be due to the acid hydrolysis steps which introduce sulphate and phosphate groups on the surface of the CNC which lowers their thermal stability. 37 The sulphate or phosphate anhydro-glucose unit required lesser energy to breakdown and this led to lower temperatures during the degradation process. Another reason for the lower stability of the CNC was because acid hydrolysis had removed amorphous region of the fibre, decreased the particle size of the fibre which in turn led to increment of the surface area and caused the lower thermal degradation temperature. The higher surface area of the CNCs had exposed greater number of the free ends of the chains which promotes decomposition at a lower temperature. 21
Besides that, both the S-CNC and P-CNC was having higher percentage of remaining char residues than SB-MCC. This could be explained by the crystalline region of the CNCs that would act as a charring agent at high temperature. This observation was similar to the result reported by Chen et al., 24 who reported that higher crystallinity of the nanocellulose would function as a flame retardant at high temperature. These flame retardant would perform as a barrier from the burning surface to the attached polymeric chains in nanocellulose. This highly crystalline region of CNC causes more proportion of carbon, thus formation of char residue as the content of carbon increases. Besides having more crystalline region, the char residue of both S-CNC and P-CNC may be attributed by the dehydrating mechanism performed by sulphuric acid and phosphoric acid. Both the sulphuric acid and phosphoric acid would promote the decomposition of cellulose by eliminating some of the –OH groups by esterification or catalysis mechanism. The presence of the H+ ion would facilitate the production of char residue because the elimination of the oxygen in the form of H2O which causes dehydration process to be occurred. This process was favourable for the production of anhydrocellulose and the decomposition of anhydrocellulose lead to char formation. 21
Apart from that, in the DTG curve of S-CNC, it showed that there was a broad hump in close proximity at the range of 300–400°C in which lower degradation temperature may be attributed to the highly sulphated regions, whereas higher degradation temperature corresponded to the unsulphated part of the material. 25 The sulphated part was the main factor that causes the lowest T onset for S-CNCs compared to SB-MCC. This is because the sulphated anhydroglucose units will act as catalyst that increases the decomposition of the S-CNC. Aside from imparting catalytic effect on the thermal degradation of cellulose, the replacement of hydroxyl groups by sulphates which leads to the reduction of activation energy could be one of the reasons that decreases the thermal stabilities of S-CNC. 38 Apart from that, the T d of the S-CNCs was the highest among all the samples. This could be attributed to the exothermic effect produced by the burning of the charred residue. 36
Based on the Table 3, it can be seen that although both of the CNC was synthesized by the same method, P-CNCs have the highest T max among all the samples and have higher char residue compared to S-CNC. The main difference between the P-CNC and S-CNC was the substituted functional moieties on the surface of the CNC. All the cellulosic backbone of the CNCs are similar, this suggested that the presence of the anionic groups on the surface of acid-derived CNC is responsible for the variation in thermal stability.
According to the study reported by Sirviö et al., 39 the main product in degradation of cellulose was levoglucosan. The highest T max of the P-CNCs may be explained by the good interaction of the phosphoric acid with the primary hydroxyl group of cellulose which prevents the formation of levoglucosan. The phosphoric acid tends to act as catalyst to promote dehydration and desaturation of pyranose ring at temperature lesser than 400°C. This could cause the formation of lesser amount of flammable volatiles which led to higher char formation. It is known that phosphorus can perform as a flame retardant in different kind of phase. In condense phase, the phosphorus can produce more char which led to the yielding of intumescence whereas in gas phase, the phosphorus group is able to inhibit the flame. 40 Accordingly, in this study, it is reasonable to say that the char produced from the phosphate group had made the cellulose to have lesser exposure to the heat source which could subsequently enhance the thermal stability of the P-CNC.
The thermal properties of PLA and its nanocomposite were studied and compared as shown in both Figure 5(a) and (b). The thermal characteristics of the PLA composites were summarized in Table 4. Based on the TGA and DTG curve obtained, it can be seen that the thermal stability of PLA was increased when there was an introduction of CNC into the composite. The higher thermal stability of the PLA/CNC composite compared to the neat PLA was mainly due to the highly crystalline structure in the CNC. The cellulose chain in the CNC is packed into more oriented compact system through the hydrogen bonding’s stabilization. These highly oriented crystalline regions are highly thermal stable because the inter-chain hydrogen bonds are difficult to break down by high temperature which protects the cellulose from melting. 41 Therefore, the incorporation of CNC into PLA would enhance their thermal stability and eventually more resistant to heat energy because the CNC could hinder the heat flow to the composite.
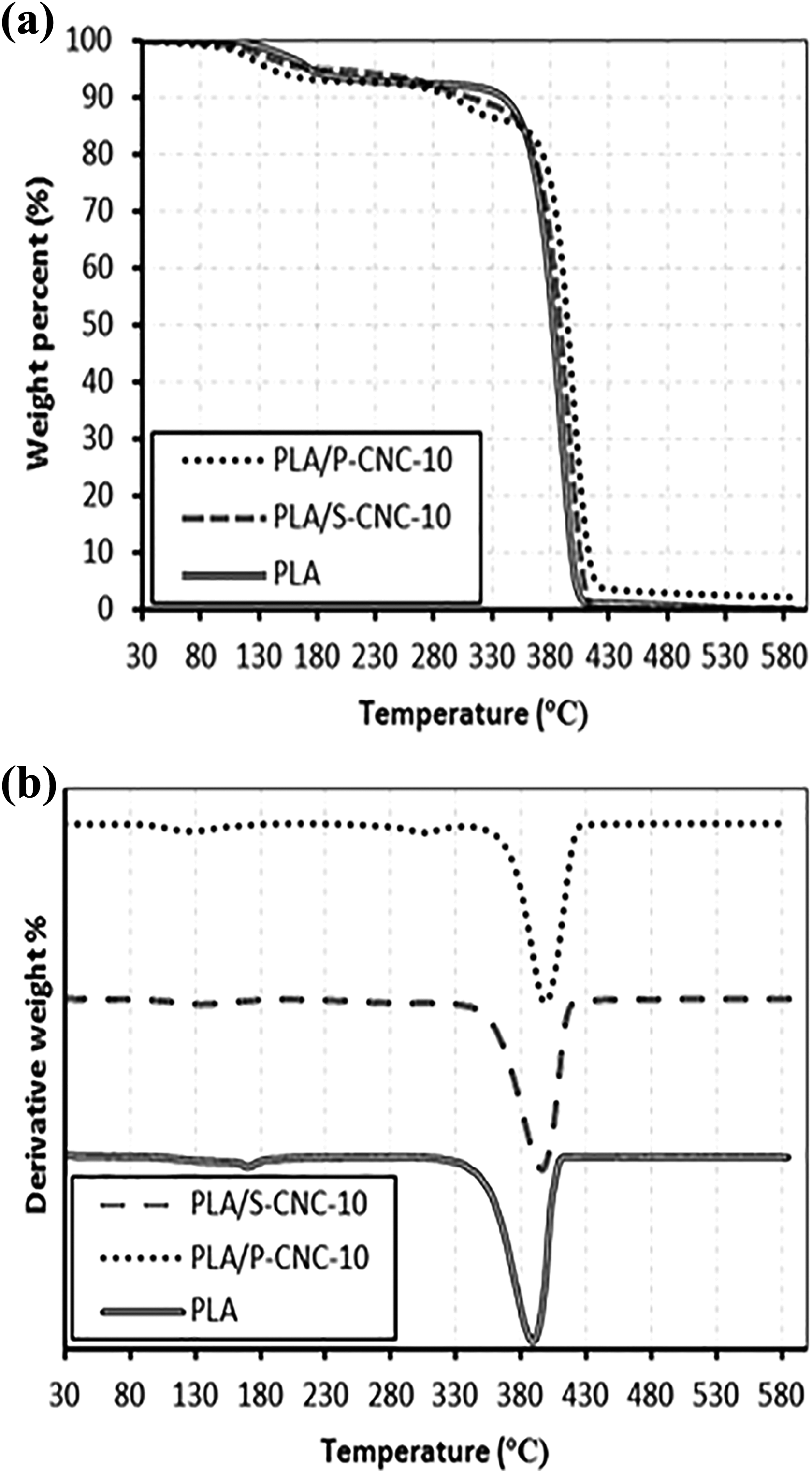
(a) TGA curves and (b) DTG curves of PLA, PLA/S-CNC-10 and PLA/P-CNC-10 composites. TGA: thermogravimetric analysis; PLA: poly(lactic acid); S-CNC: H2SO4-treated cellulose nanocrystals; P-CNC: H3PO4-treated cellulose nanocrystals.
By comparing the PLA/CNC composite, it can be seen that PLA/P-CNC-10 was having higher thermal stability than the PLA/S-CNC-10 composite. The phosphorous acid and its salt had been proven as a flame retardant due to its ability to have char formation. The high residue weight of the PLA/P-CNC-10 indicated that the ability of P-CNCs to form char protects the PLA from exposing to the heat source. Similar observation was reported by Sirviö et al., 39 who mentioned that the backbone of the cellulose is made up of high percentage of carbon and these carbon could be contributed as a source of intumescent flame retardant. In this case, the flame retardant would be phosphorous acid and the reaction of the phosphorous acid and carbon source will cause the formation of char layer. These char layer will acts as an insulator to prevent the heat from spreading further and thus enhance the thermal stability of the PLA/P-CNC-10 composite.
Conclusions
In this study, CNC were successfully extracted from SBF. TEM results demonstrated that the needle-like CNC has diameter of 5.5–6.2 nm and length of 235–300 nm. The XPS results confirmed the existence of both sulphate group and phosphate group in the S-CNC and P-CNC. FTIR results indicated the presence of hydrogen bonding and chemical interaction between PLA and CNC. The TGA results had shown that PLA/P-CNC-10 exhibited higher thermal stability than PLA/S-CNC-10 nanocomposites. The higher thermal properties corresponded to the high char residue of P-CNC. Overall, it is feasible to use phosphoric acid (mild acid) to extract CNC from the SBF, and the P-CNC could enhance the thermal stability of PLA nanocomposites.
Footnotes
Acknowledgement
The authors would like to express their appreciation to Universiti Sains Malaysia for Research University Grant.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed the receipt of following financial support for the research, authorship, and/or publication of this article: This work was supported by the Universiti Sains Malaysia for Research University Grant (grant number: 1001/PBAHAN/8014024).
