Abstract
The effect of organoclay as well as controlled dicumyl peroxide (DCP) curing on the morphology and physicomechanical properties of linear low-density polyethylene (LLDPE)/ethylene-co-methylacrylate (EMA) blends was explored in this article. The nancomposites were melt-compounded in an internal mixer, HAAKE RHEOMIX, at 140°C and a rotor speed of 60 r min−1. The nanocomposites were then cured in a compression mold at 170°C for 12 min, and their morphology and physicomechanical properties were evaluated. Exfoliated morphology was observed for DCP-treated nanocomposites. DCP initiated grafting between polymer and clay platelets resulted in high degree of reinforcement in all nanoclay-filled samples through exfoliation of clay platelets as well as interfacial cross-linking and controlled cross-linking in the two polymer phases. Reprocessability studies showed that LLDPE/EMA/Cloisite®20A nanocomposites cured with DCP up to 0.5 wt% can easily be reprocessed.
Introduction
Blending of existing polymer is an easiest and most widely used technique to develop new materials with desired properties. 1 –4 Blend of rubber and plastic commonly called thermoplastic elastomer is an example of polymer blend which combines the functional properties of conventional elastomeric materials and can be processed with a thermoplastic processing machine. 5 –11 A more recent technique for modification of polymers is incorporation of organically modified layered silicate to get polymer nanocomposites. Montmorillonite is the most commonly used layered silicate in producing polymer nanocomposites. 4 Incorporation of small amounts of organoclay (about 5 wt%) and its exfoliated dispersion in polymer matrices significantly improve the dimensional stability, mechanical, thermal, optical, electrical, and gas barrier properties of the neat polymer. This happens due to the large contact area between polymer and clay on a nanoscale range. 12 –20
Another well-known approach for modification of polymer is to incorporate cross-linker to get thermoset polymer. In recent decades, much research work has been done to establish the better curing system and compatibilization of rubber/plastic blends. 21 –24 Thermochemical cross-linking involving organic peroxides is most widely used for its control decomposition rate, minimum side products, and economic process. 25 Moreover, depending on the molecular structure of the particular peroxide and its solubility or miscibility in polymeric materials, a certain distribution of peroxide in matrix and dispersed phase might be expected. Therefore, the use of peroxide could provide the possibility of targeting reaction in the desired phase. 26
Several articles are quoted in the literature on peroxide curing of rubber plastic blends. Moly et al. studied the effect of dicumyl peroxide (DCP) cross-linking on linear low-density polyethylene (LLDPE)/ethylene vinyl acetate (EVA) blend and reported that DCP-cross-linking is more effective in EVA phase than in the LLDPE phase. 27 Borah et al. recently have studied the effect of DCP curing on LLDPE/ethylene methacrylate (EMA) blends and reported that EMA exhibits superior cross-linking as compared to LLDPE. 28 At low peroxide content (0.3 wt%), mutual cross-linking of the elastomeric phase (EMA) as well as interface with low degree of modification of plastic phase (LLDPE) has been demonstrated. It has also been reported that at 0.3 wt% of DCP loading, the gel fraction of pristine LLDPE is 18%, whereas for pristine EMA (30% methacrylate content), it is 49.89%.
More recently, the research in polymer nanocomposites has derived toward the combination of the techniques of polymer blends, nanocomposites, and vulcanizates to get thermoplastic vulcanizates (TPV)-based nanocomposites. Recently, Munusamy et al. studied the effect of sulfur and peroxide vulcanization on EVA/NR blend and reported that peroxide system showed better enhancement in mechanical properties and resistance toward swelling compared to the sulfur system. 29 Bahattab and his coworkers showed that addition of organic peroxide in EVA/LDPE/metal hydroxide nancomposites improved thermal stability and good retention of mechanical properties after thermal aging. 30 However, to date, there is very little information available regarding TPV-based nanocomposites and much to be learned about the mechanism how organic peroxide influences the morphology and properties of polymer nanocomposites.
In this article, we have studied the effect of organic peroxide on morphology and physicomechanical properties of LLDPE/EMA/clay nanocomposites. The aim of this research is to modify the properties of the LLDPE/EMA/clay nanocomposites by selectively strengthening the weaker EMA phase and interface without much affecting the plastic phase so that reprocessability can be maintained at a tolerable level. Hence, we have chosen very low level of peroxide loading (0.3 and 0.5 wt%) at which only the EMA phase along with the interface is likely to be modified appreciably.
Experimental
Materials
LLDPE (LLT12) having a density of 0.926 g cm−3 and melt flow index (MFI) 3.7 g per 10 min was obtained from Haldia Petrochemicals Ltd., India. Commercial grade of EMA, Elvaloy 1330 (with 30 wt% of methyl acrylate and an MFI of 3.0 g per 10 min; DuPont, Wilmington, Delaware, USA), was supplied by NICCO Corporation Ltd., India. DCP (Perkadox-BC-40B-PD) having an active peroxide content of 40% was obtained from Akzo Nobel Chemical Company, The Netherlands.
The compatibilizer used for this study is maleic anhydride–grafted LLDPE (LLDPE-g-MA). LLDPE-g-MA was prepared by melt blending LLDPE (100 g) with maleic anhydride (5 g) and DCP (40% activity; 0.5 g). The melt mixing was carried out in an internal mixer at 180°C and 60 r min−1 for 8 min. The grafted MA content on LLDPE was observed to be 0.78 wt%, as reported in our earlier publication. 6
The organoclay Cloisite®20A was purchased from Southern Clay Products Inc. Cloisite®20A is also a natural montmorillonite that has been ion exchanged with dimethyl, dehydrogenated tallow and quaternary ammonium chloride to form an organoclay.
Preparation of nanocomposite vulcanizates
Prior to mixing, LLDPE, EMA, and clay were dried at 80, 50, and 70°C, respectively, for 12 h in a vacuum oven. The melt blending was carried out in a HAAKE Rheomix OS (Germany) at 140°C and a rotor speed of 60 r min−1 by the variation of two different mixing sequences.
In sequence M1, LLDPE and LLDPE-g-MA were first allowed to melt for 2 min; followed by EMA (4 min) and nanoclay (4 min). Then, DCP was added and mixed for 3 min. The total mixing time was 13 min. In sequence M2, first LLDPE and LLDPE-g-MA were allowed to melt for 2 min, followed by nanoclay for 4 min. Then, EMA was added to this mixture and mixed for another 4 min and this was followed by DCP for 3 min. The mixes so obtained were sheeted out in a two-roll mill set at 2 mm nip gap.
The sheeted material was then cured in an electrically heated hydraulic press (Moore Presses, George E Moore & Sons Birmingham Ltd., United Kingdom) at 170°C for 12 min under a pressure of 5 MPa. The mold was allowed to cool under pressure till ambient temperature is attained before removing the rectangular sheet from the mold. The details of the sample and their appropriate designations are given in Table 1.
Sample designations.
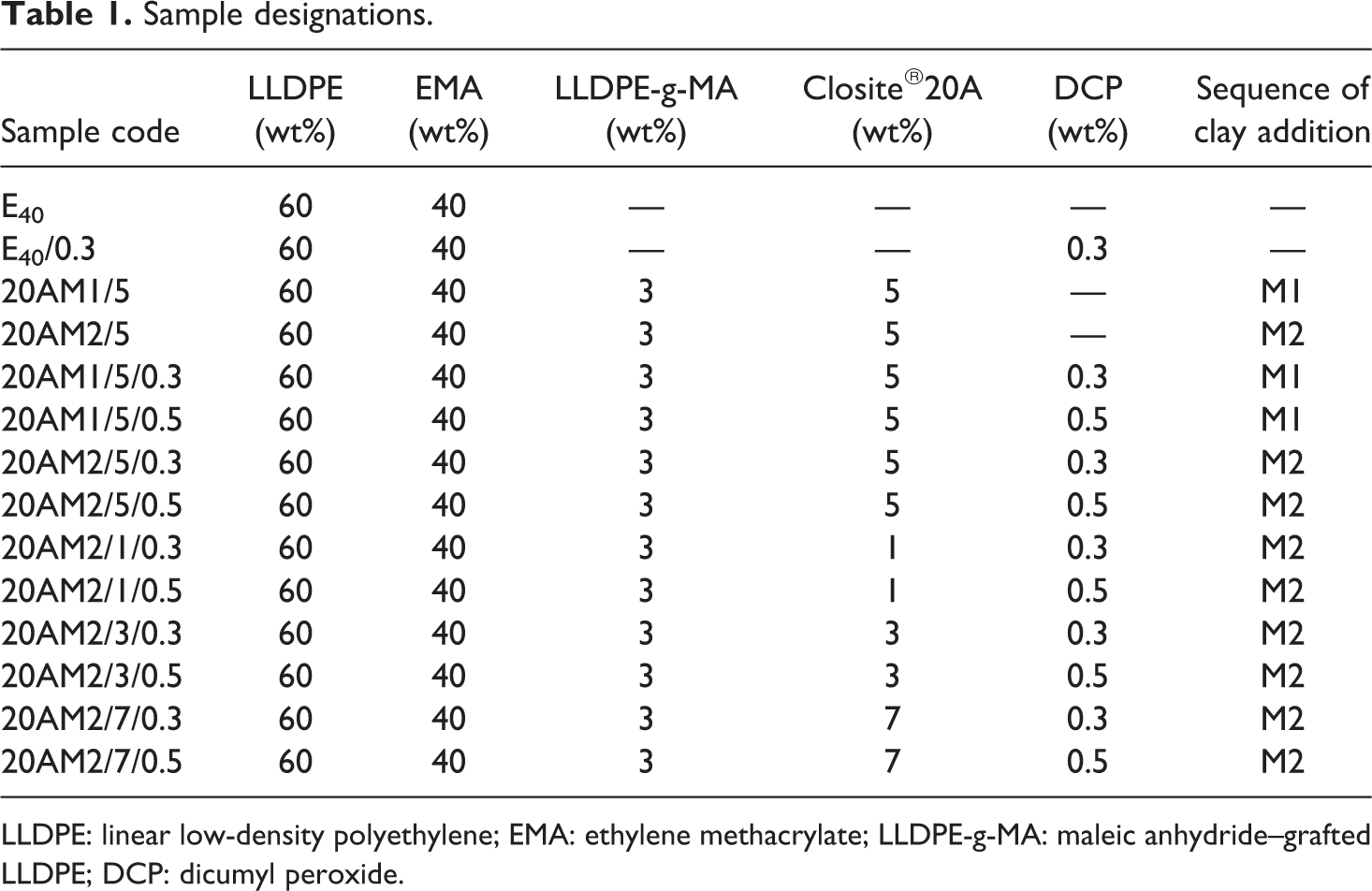
LLDPE: linear low-density polyethylene; EMA: ethylene methacrylate; LLDPE-g-MA: maleic anhydride–grafted LLDPE; DCP: dicumyl peroxide.
Morphology characterization
In order to study the dispersion and location of the organoclays in the LLDPE/EMA/Cloisite®20A nancomposites and in their vulcanizates, the samples were examined by transmission electron microscopy (TEM). For TEM observations, in compression-molded samples, the EMA phase was stained with aqueous osmium tetroxide solution for 15 days. The specimens were dried in a vacuum oven overnight. Then, the specimens were cut into 50-nm-thick sections using a Leica ULTRACUT UCT (Austria) microtome equipped with a diamond knife. The ultrathin sections were mounted on 300-mesh copper grids and dried in desiccators and examined using a high-resolution TEM instrument (JEOL JEM 2100, Japan) at 200 kV.
Mechanical properties
The tensile testing of the blends was carried out in a Hioks–Hounsfield Universal Testing Machine (Test Equipment, Ltd, Surrey, England) according to ASTM D 412-98a test method using dumb-bell shaped specimen at a cross-head speed of 500 mm min−1 at room temperature (25 ± 2°C). Impact strength was determined using a tensile impact tester, CEAST type 6545/0000 using a load of 7 kg as per DIN53448.
The tension sets were measured using similar tensile sample by stretching it to 100% elongation for 10 min, and the percentage set was measured after equilibrating (release of load) them for another 10 min.
Dynamic mechanical analysis
Dynamic mechanical analysis of the nanocomposites was carried out on a DMA 2820 (TA Instrument, USA). The experiment was performed in tension mode at a frequency of 1.0 Hz. The temperature range of the testing was −80°C to +100°C with a programmed heating rate of +2°C min−1.
X-ray analysis
X-ray diffraction (XRD) was performed with a high-resolution X-ray diffractometer (model X’Pert PRO, Philips PANalytical B.V., Almelo, The Netherlands) with Cu K α (λ = 1.54 Å) radiation source operated at voltage of 40 kV and 30 mA electric current. The samples were scanned from 2° to 10° (2θ) at a scan rate of 2° min−1.
The interplanar distance (d) or d-spacing was calculated using the Bragg’s law

Gel fraction analysis
The gel content of the cross-linked EMA phase was determined gravimetrically using a 72-h Soxhlet extraction with chloroform as the solvent at 50°C. After the extraction, the sample was vacuum dried to a constant weight. The gel fraction was calculated as the percentage ratio of the final weight of the polymer to its initial weight.
Fourier transform infrared spectroscopy
A Perkin-Elmer SPECTRUM RX 1 Fourier transform infrared (FTIR, USA) spectrometer was used to obtain the infrared spectra of the samples. Thin films were used for analysis. The spectral resolution was kept at 4 cm−1 for all samples and the scan was taken at room temperature.
Reprocessability studies
The reprocessing characteristics were measured by repeated mixing and molding the samples at the same processing conditions of their preparation, followed by measuring their mechanical properties. The experiments were carried out for five processing cycle for each sample.
Results and discussion
Wide-angle X-ray diffraction
Figure 1 shows the XRD patterns of the pristine organoclay (Cloisite®20A), LLDPE/EMA/Cloisite®20A nanocomposites, and LLDPE/EMA/Cloisite®20A nanocomposite vulcanizates. The 2θ and corresponding d-spacing value for the clay (001) peaks are summarized in Table 2. It is seen that the diffraction peak (001) for Cloisite®20A appears at 2θ = 3.27° (d 001 = 2.70 nm), while that for LLDPE/EMA/Cloisite®20A nanocomposites shifted to lower angles suggesting an intercalated structure. The characteristic diffraction peak (001) of Cloisite®20A disappears for the 0.3 and 0.5 wt% DCP-cured nanocomposites suggesting exfoliated morphology. This may probably be due to the grafting of clay platelets in the polymer matrix. Although the DCP may not have decomposed completely under the nanocomposite preparation conditions, there would have been enough decomposition to supply free radicals to the already mixed LLDPE/EMA/clay nanocomposites. These free radicals abstract hydrogen from the EMA copolymer chain and hydrogenated tallow of the organic modifier present in Cloisite®20A, followed by grafting between polymer chains and clay platelets. The probable reaction mechanism for grafting reaction is given in Figure 2. Here, we have considered the grafting of clay platelets with EMA molecules only, since irrespective of the blending sequence Cloisite®20A mostly located in the EMA phase, only few tactoids are present in the LLDPE phase (Figure 6). But similar grafting reaction with LLDPE is also possible. A schematic representation of the grafted clay particle is shown in Figure 3.
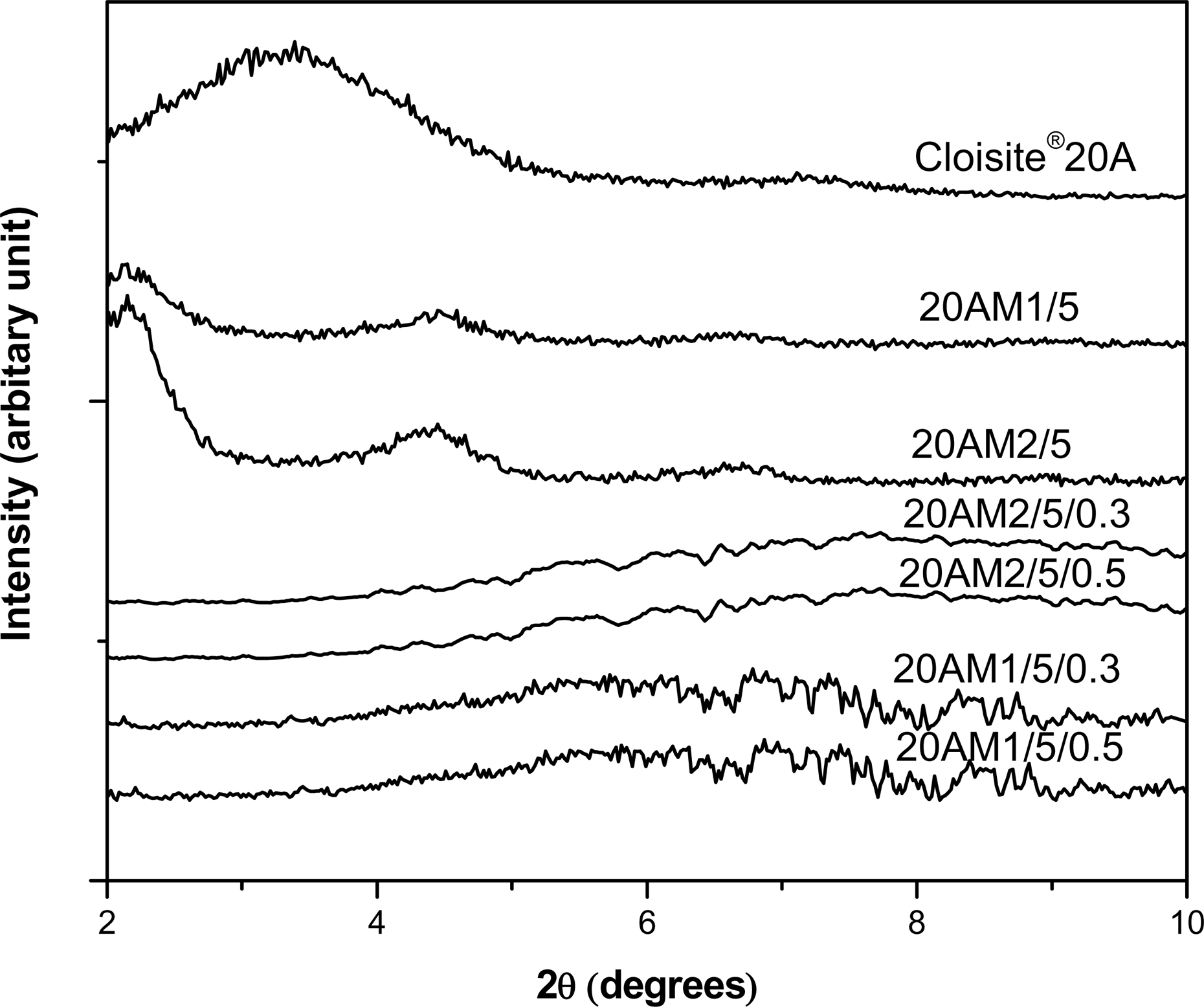
WAXD pattern of Cloisite®20A, LLDPE/EMA/Cloisite®20A uncured, and DCP-cured nanocomposites.
Organoclay interlayer spacing obtained by WAXD.

WAXD: wide-angle X-ray diffraction.
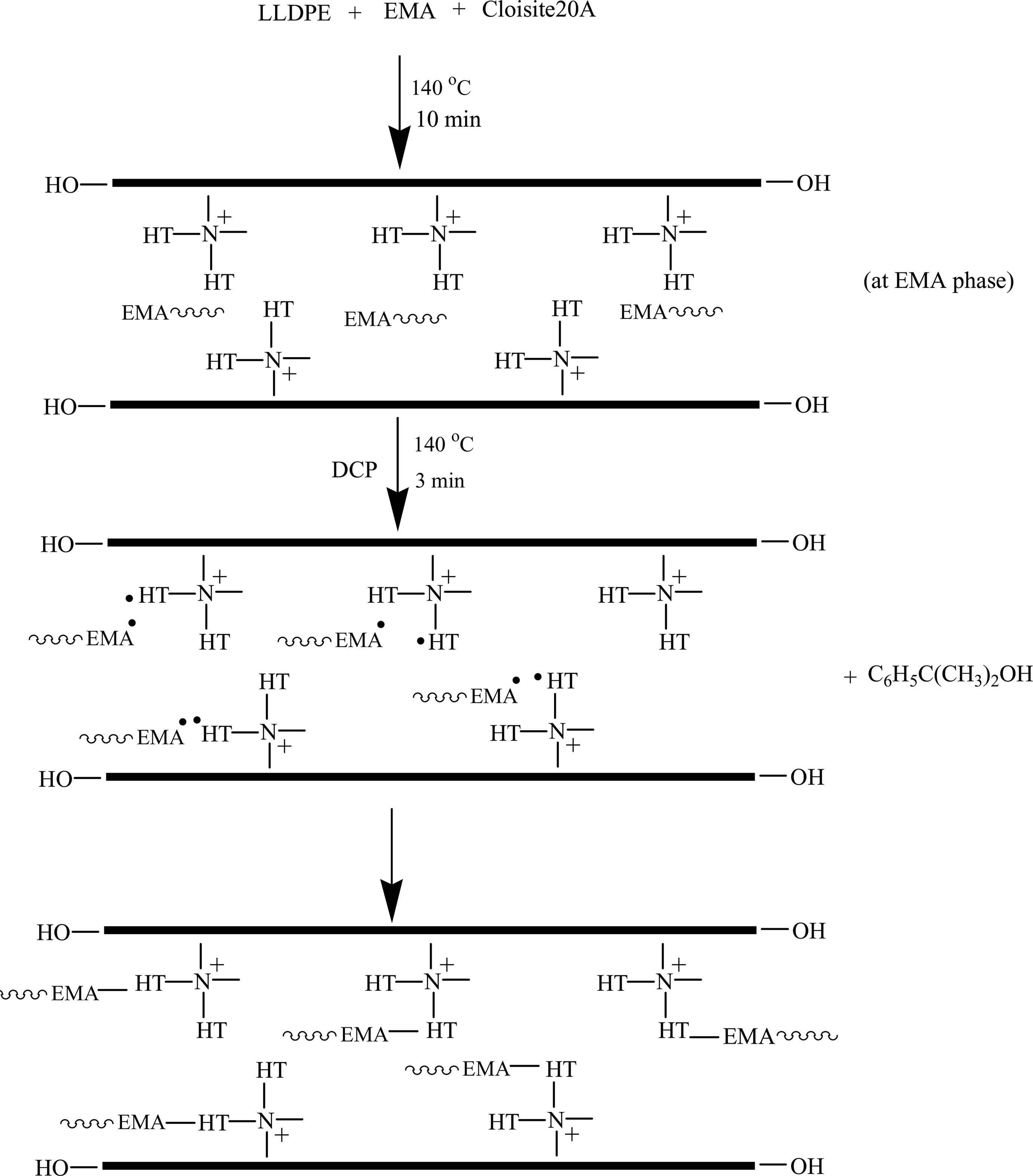
Free radical grafting of EMA on to Cloisite®20A.
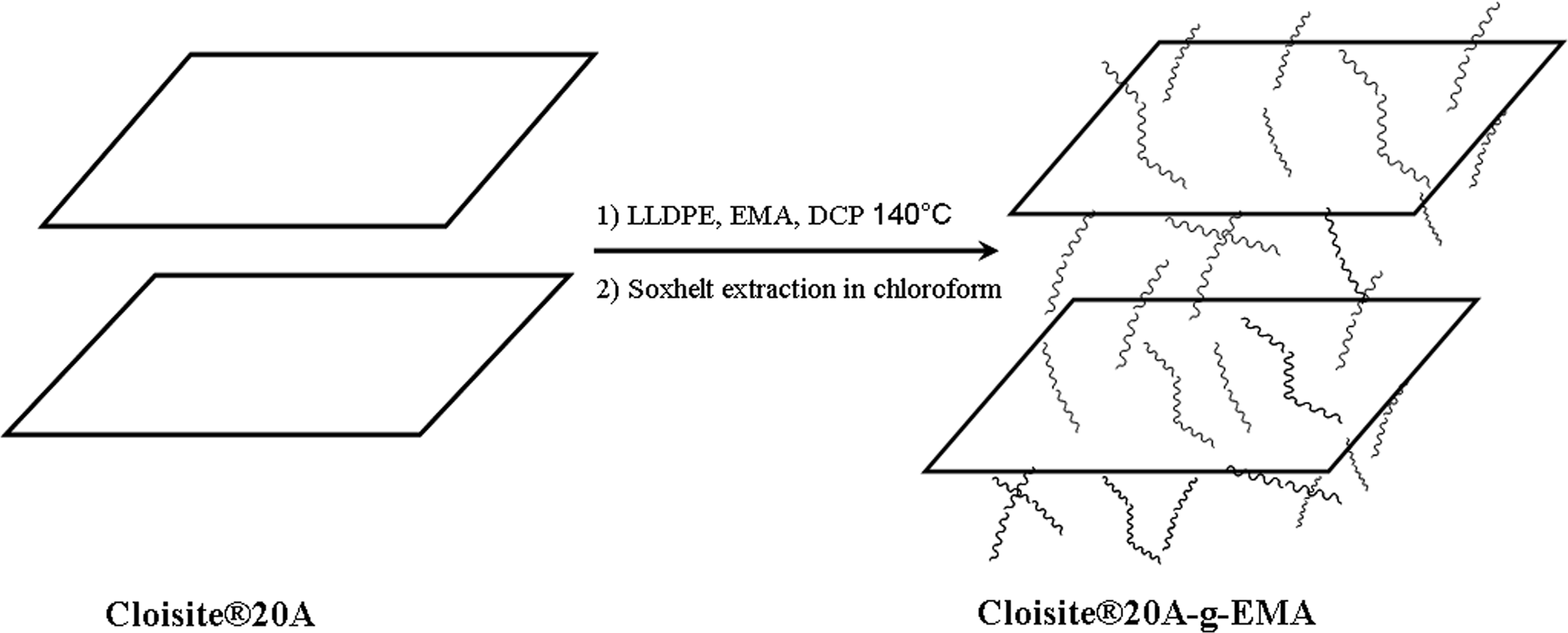
Schematic representation of grafting of EMA on to Cloisite®20A.
Figure 4 shows the XRD pattern of LLDPE/EMA/Cloisite®20A nanocomposites prepared by mixing sequence M2, with various concentrations of clay and 0.3 wt% DCP. It can be seen that up to 5 wt% clay loading, the (001) diffraction peak disappears for the DCP-cured nanocomposites. However, a small diffraction peak is observed for 7 wt% clay loading suggesting partial exfoliation of the clay platelets. The absence of complete exfoliation is the result of increasing clay/DCP ratios, giving rise to reduced interaction between the polymer chain and clay platelets. Similar behavior has been reported previously for EVA/Cloisite®20A nanocomposite vulcanizates by Mishra and Luyt. 31
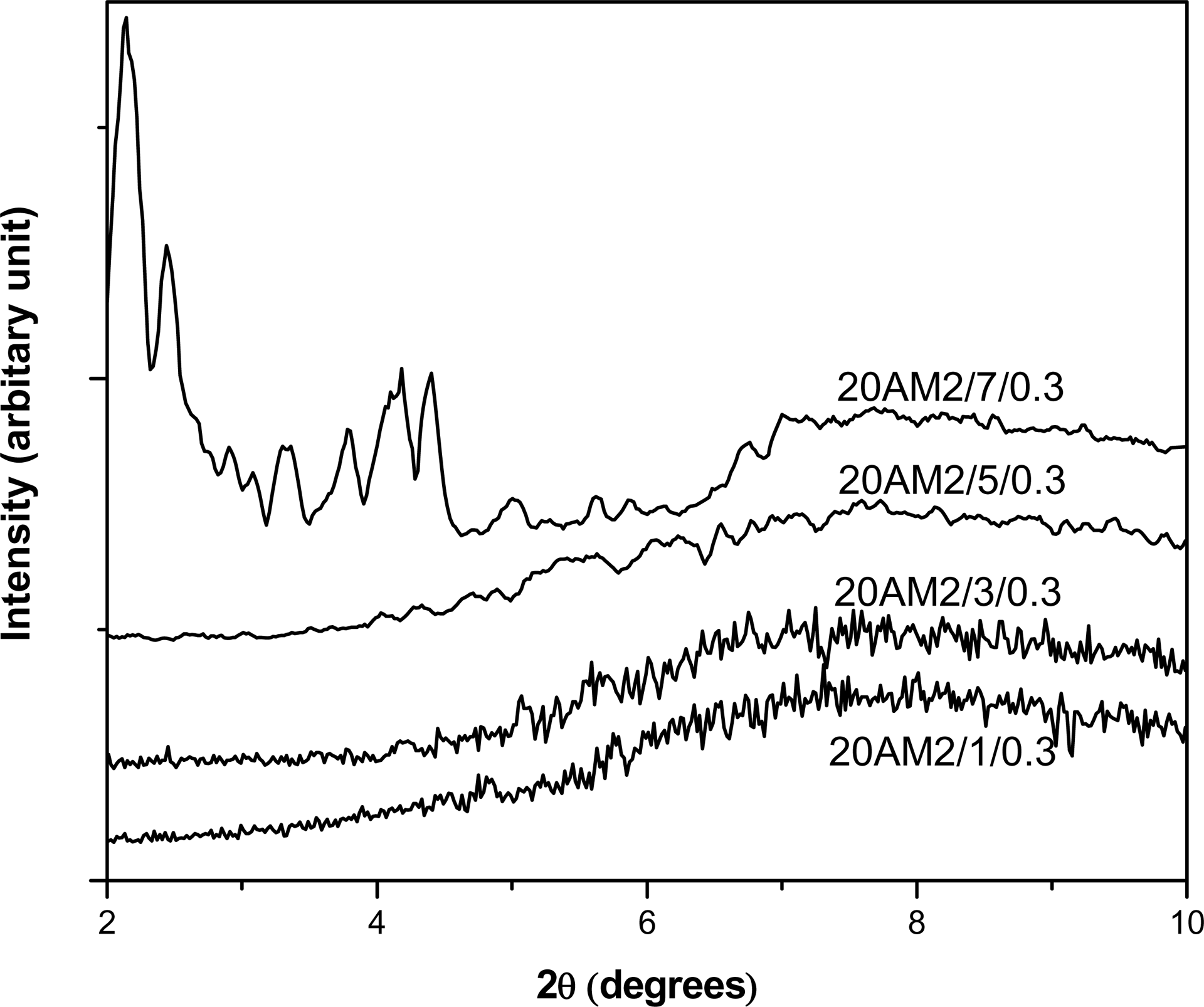
WAXD pattern of DCP-cured LLDPE/EMA/Cloisite®20A nanocomposite at different concentrations of clay.
FTIR study
In order to demonstrate the occurrence of grafting reaction by conventional analytical method, the unreacted copolymer must be separated from the grafted organoclay particles. For that, after the mixing process, the product was Soxhlet-extracted using chloroform to remove the unreacted EMA molecules. The complete removal of the unreacted EMA copolymer was confirmed by mixing EMA and Cloisite®20A without DCP. After 72 h of Soxhlet extraction, the absorption band was the same as for Cloisite®20A and the absorption band related to EMA was not observed. Figure 5 shows the FTIR spectra of Cloisite®20A, EMA, and EMA-grafted Cloisite®20A. Cloisite®20A shows characteristic peak at 3624 cm−1, 1045 cm−1, and 528 cm−1 due to the –OH stretching of structural hydroxyl group, Si–O stretching, and Al–O–Si deformation, respectively. EMA shows a characteristic peak at 1736 cm−1 due to the C=O stretching vibration of acrylate group. The presence of C=O stretching peak at 1736 cm−1 in grafted Cloisite®20A confirmed that EMA molecule was grafted in to the clay platelets during melt mixing of LLDPE/EMA/Cloisite®20A in presence of DCP.
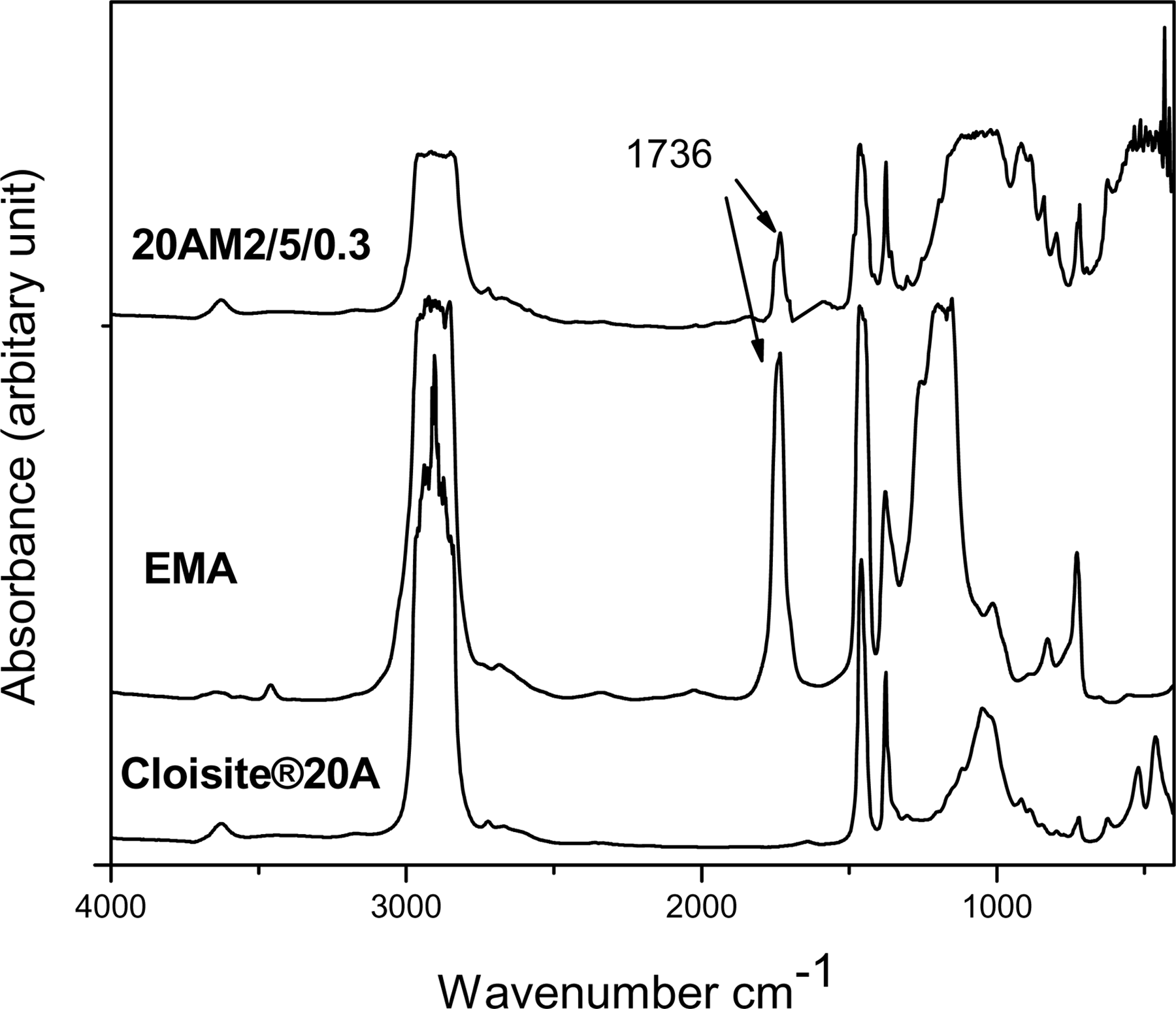
FTIR spectra of Cloisite®20A, EMA, and Cloisite®20A-g-EMA.
TEM analysis
TEM analysis was performed to investigate the state of dispersion of clay particles in the bicomponent polymer matrix. All the samples are stained with osmium tetroxide which combines with EMA 32 and thus provides higher scattering cross sections to the incident electrons. Thus, the rubbery EMA phases of LLDPE/EMA blend systems appear as dark areas in the TEM bright-field images. The dark rubbery phase appears as the dispersed phase in the continuous LLDPE matrix. In all cases, the intensity of the black color varies among the dispersed particles. The change in the intensity of the black color is attributed to the presence of clay particles (which have more electronic contrast as are likely to absorb osmium tetroxide) in the EMA phase. Figure 6 shows the TEM micrographs of uncured and DCP-cured nanocomposites. As observed in our earlier studies, 14 irrespective of the blending sequence, the organoclay, Cloisite®20A, preferentially located in the EMA phase. Thus, the weaker EMA phase (dispersed) becomes reinforced with nanoclay and contributed to the improvement of material toughness. However, for the mixing sequence M2, quite a few are visible in the amorphous portion of the semicrystalline LLDPE phase, which helps in improvement of materials’ rigidity. 14 Another important observation is that incorporation of DCP helps in the dispersion of clay particles in the polymer matrix. Figure 6(c) and (d) shows exfoliation clay platelets, 5 wt% clay loading, in DCP-treated nanocomposites. At higher loading level of clay (7 wt%), exfoliated platelets coexist with lactoids of larger size (Figure 6(e)). This agreed well with the results obtained from wide-angle X-ray diffraction (WAXD) analysis.
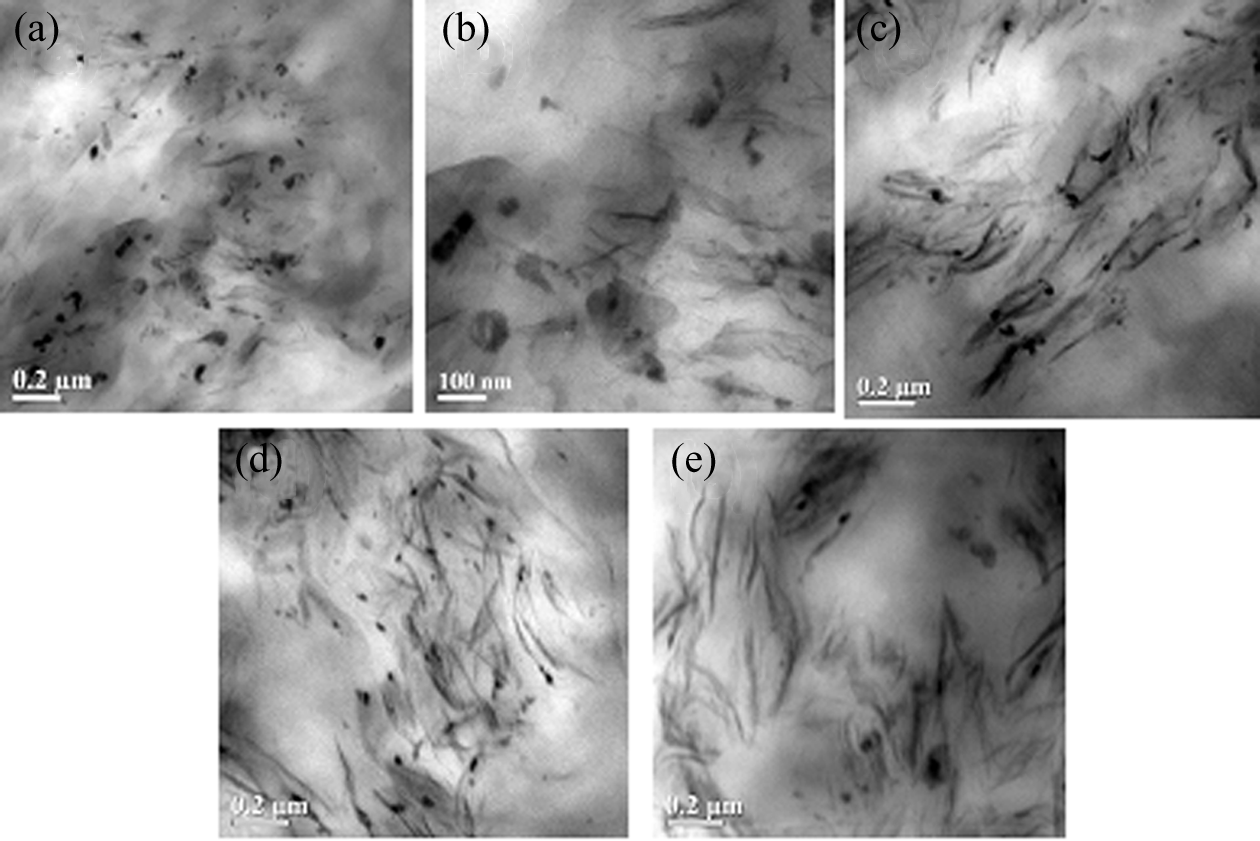
TEM micrographs of (a) 20AM1/5, (b) 20AM2/5, (c) 20AM1/5/0.3 (d) 20AM2/5/0.3, and (e) 20AM2/7/0.3 at a magnification of ×15,000.
Gel content analysis
In order to follow the extent of cross-linking reaction on the elastomeric EMA phase during tailoring of TPV nanocomposites, the gel content of the nanocomposite vulcanizates was measured directly in chloroforms at 50°C. Then, gel content of EMA was calculated from the LLDPE/EMA/clay ratio. Before the gel content measurement, the solubility of pristine LLDPE and EMA is checked. Chloroform is a good solvent at 50°C for EMA 30% and poor solvent for LLDPE. Therefore, it was decided to determine the gel content of EMA in chloroform at 50°C.
The gel content results for LLDPE/EMA blends cross-linked by DCP in the presence and absence of nanoclay are summarized in Table 3. The gel content results show the formation of gel when DCP was incorporated in the nanocomposites, although extent of gelation is much lower than expected. This confirms the possibility of cross-linking between polymers. However, at constant DCP loading, % gel content decreases with increasing clay loading probably due to the more number of peroxide free radical takes part in grafting (of clay platelets in to the polymer chain) reaction instead of cross-linking process.
Percent gel content of the EMA phase for DCP-cured sample.
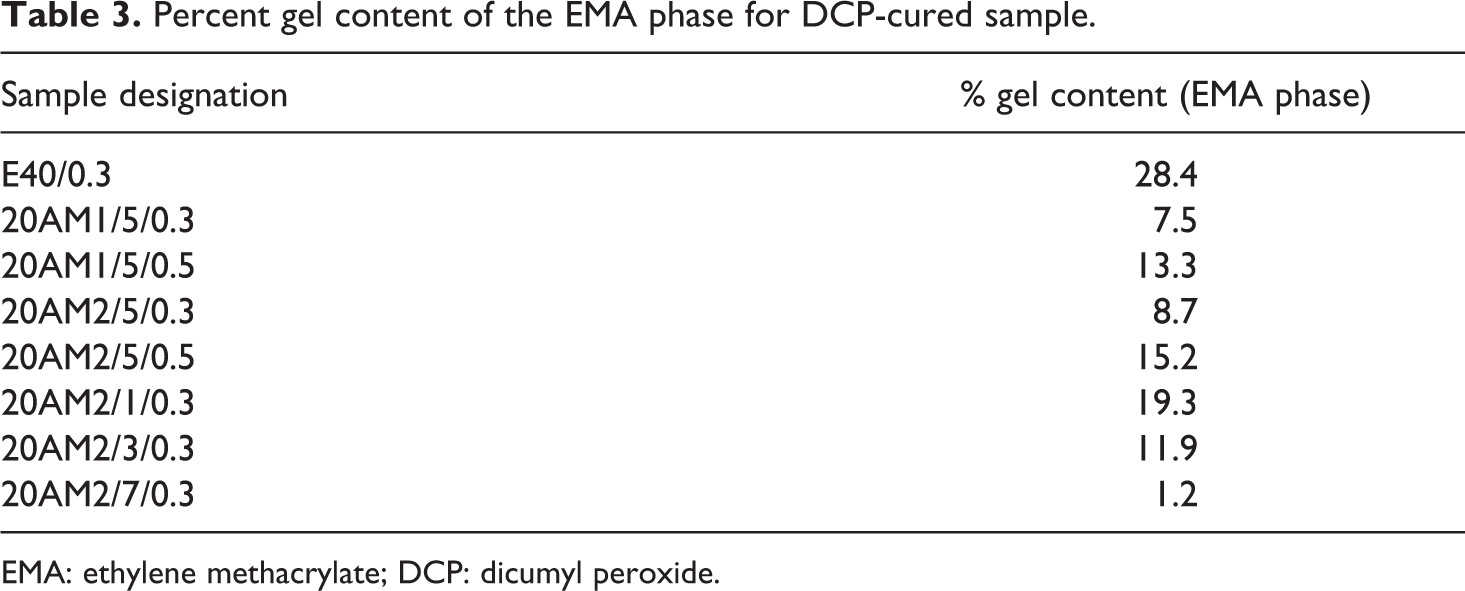
EMA: ethylene methacrylate; DCP: dicumyl peroxide.
Mechanical properties
The effect of processing sequence and filler loading on the mechanical properties of unvulcanized samples was discussed in our previous work where the marginal reinforcement effect of organoclay on LLDPE/EMA-based Thermoplastic Elastomer (TPE) system was observed. 14 Table 4 shows the effect of DCP curing on the mechanical properties of LLDPE/EMA/Cloisite®20A nanocomposites. Significant improvement in mechanical properties (tensile strength, modulus, and yield stress) was observed for cured sample as compared to uncured sample. This is due to the exfoliation of clay particles (Figure 6(c) and (d)) as well as the selective reinforcement of the weak rubbery (EMA) phase (Table 3) and the interface in the presence of DCP. However, the improvement in properties like tensile modulus, yield stress at 0.3 DCP loading is more for 20AM2/5 than 20AM1/5. This is due to the presence of small concentration of clay platelets in the LLDPE continuous phase which improves the material rigidity, as discussed in our previous work. Also, the percent gel content for the dispersed rubbery EMA phase is slightly more in 20AM2 as compared to 20AM1 (Table 3). Most importantly, the elongation at break is improved in all cured samples including the control one which indicates improvement in interfacial strength (due to peroxide cross-linking) without sacrificing the elastic property of rubber. 28 However, as compared to the control sample, impact strength increases and tension set decreases significantly for DCP-treated sample where clay platelets are exfoliated in the dispersed EMA phase as well as the EMA is reinforced with partial cross-linked. From Table 4, it can also be seen that at constant DCP loading, mechanical properties increase with increasing clay content up to 5 wt%. At higher level of loading (7 wt%), clay platelets are not fully exfoliated (see Figure 6(e)), also the percent gel content for the EMA rubbery phase is negligible (Table 3). As a result, mechanical property decreases at higher clay loading. The mechanical properties of the nanocomposite increased with increasing DCP concentration from 0.3 wt% to 05 wt%, probably due to the exfoliation of clay platelets as well as higher degree of cross-linking in the weaker EMA phase as well as the interface.
Mechanical properties of the uncured and DCP-cured nanocomposites.
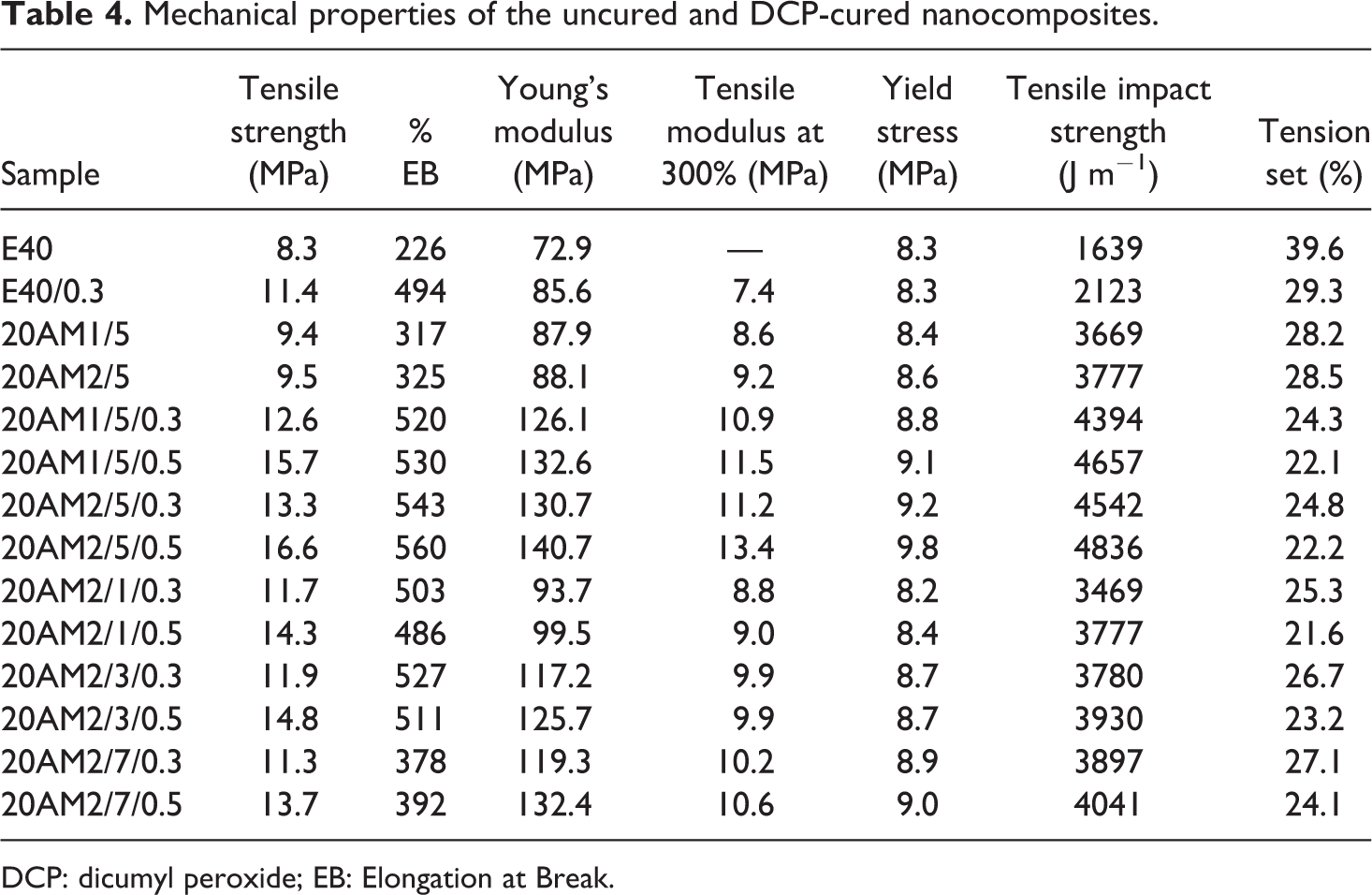
DCP: dicumyl peroxide; EB: Elongation at Break.
Dynamic mechanical analysis
Figure 7 shows the variation of storage modulus as a function of temperature for uncured and DCP-cured LLDPE/EMA/Cloisite®20A nanocomposites. It can be seen that DCP-cured sample has higher storage modulus than the uncured one. This is due to the exfoliation of clay platelets in DCP-cured sample as compared to the uncured one. The exfoliation results high polymer–filler interaction by increasing the active surface area of the filler. However, the nanocomposite vulcanizate prepared by mixing sequence M2 has higher storage modulus compared to M1. These results are in agreement with the results obtained from XRD, TEM analysis as well as from physicomechanical properties.
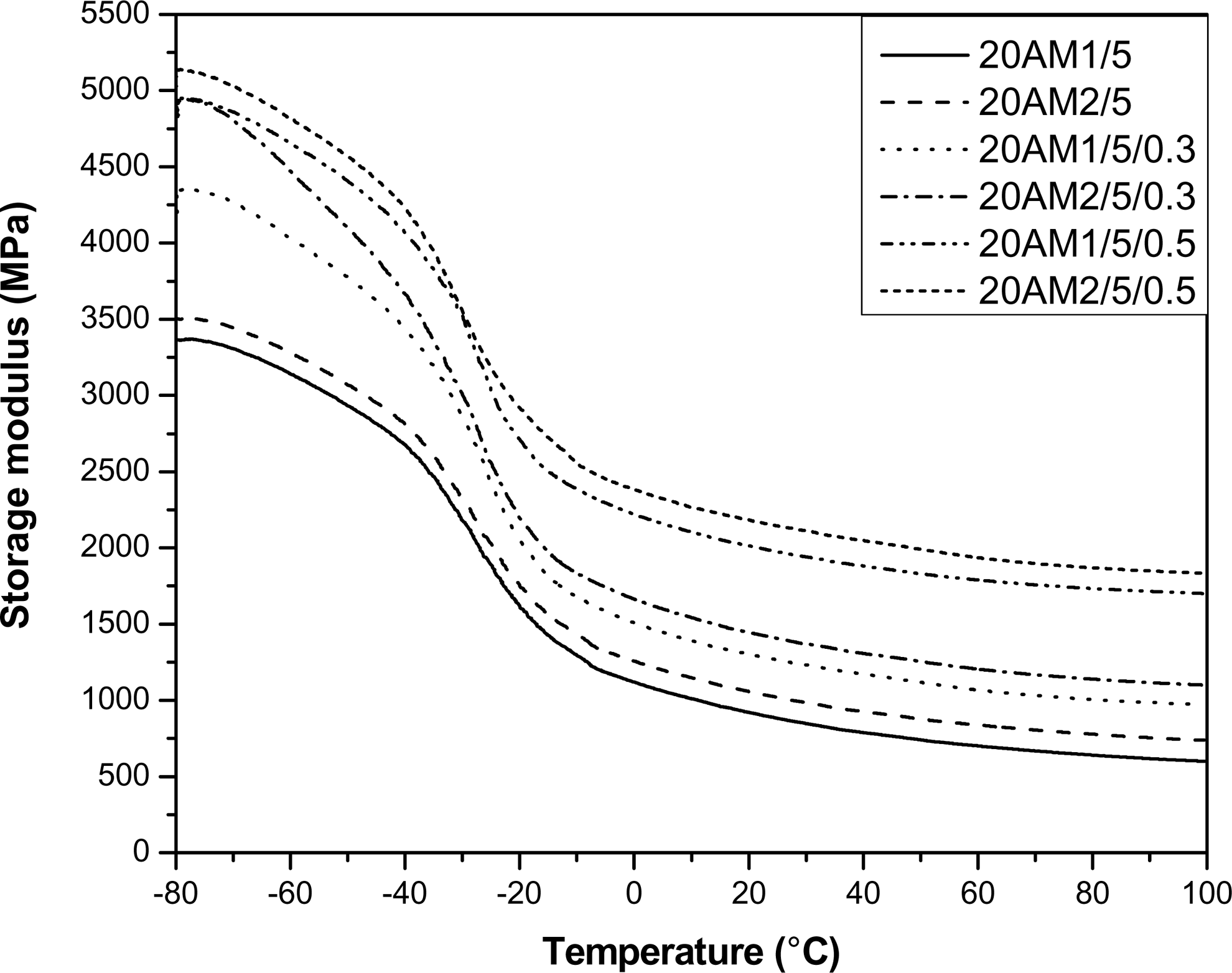
Variation of storage modulus as a function of temperature for uncured and DCP-cured LDPE/EMA/Cloisite®20A nanocomposites.
Figure 8 shows the temperature dependence of tanδ for LLDPE/EMA/Cloisite®20A nanocomposites and their vulcanizates. It is observed that β transition for cured nanocomposite occurs at high temperature as compared to the controlled sample. The shift in β transition can be explained by the degree of dispersion of the clay in the nanocomposites. It appears that the sheets of nanoclay in the polymer matrix restrict the segmental motion of polymer chains near the organic–inorganic interface, due to the increased adhesion between the polymer and organoclay surface, as a result, β transition shifted to higher temperature for nanocomposites vulcanizates. Recently, Nouri et al. also reported similar modulus and damping (tanδ) behavior for peroxide cured ethylene–octene copolymer/clay nanocomposites. 33
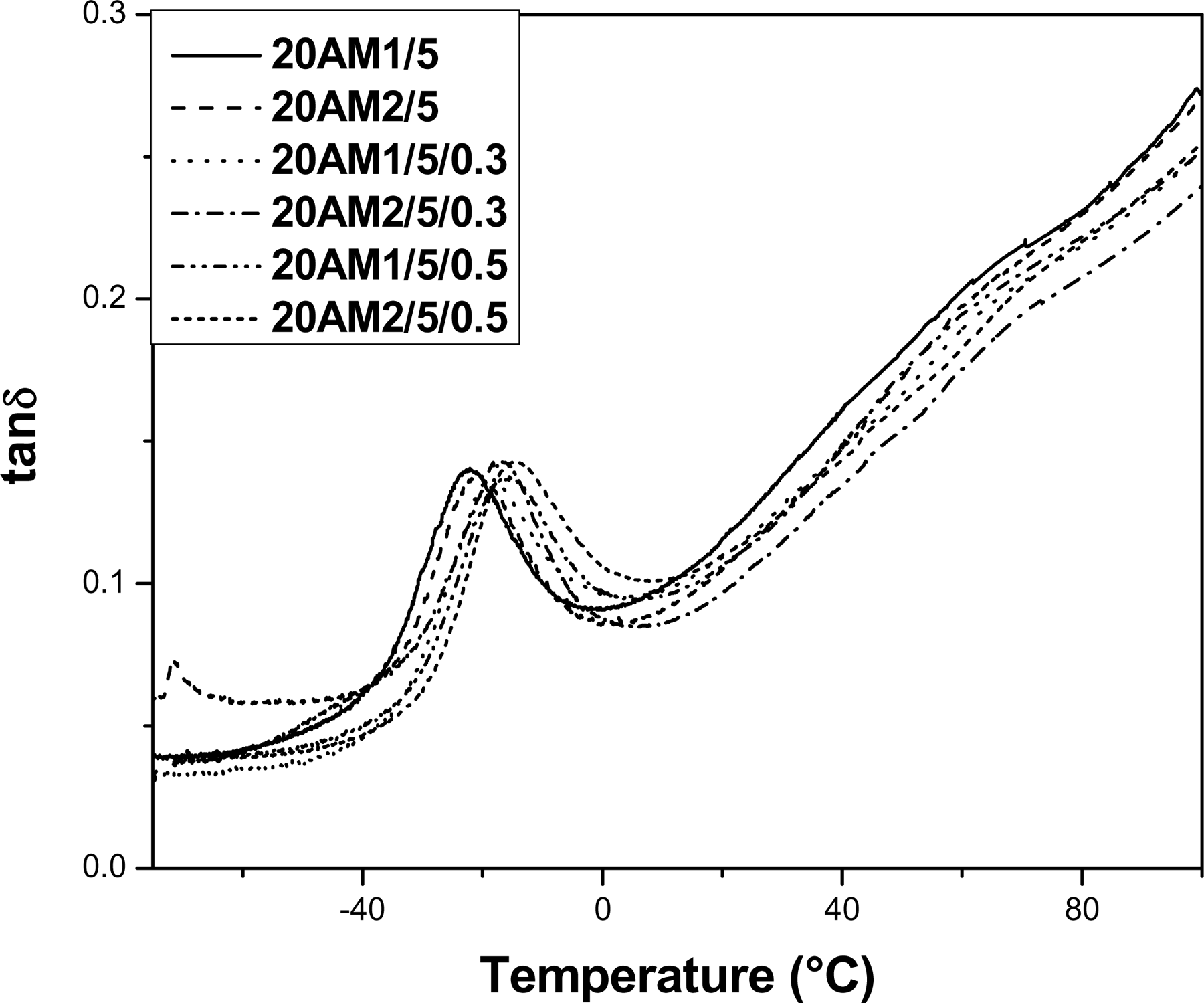
Temperature versus tanδ curves for LLDPE/EMA/Cloisite®20A uncured and DCP-cured nanocomposites.
Reprocessability studies
To establish the thermoplastic elastomeric behavior of cured nanocomposites, reprocessability studies at five processing cycles were carried out. Reprocessability studies were performed by repeated mixing and molding the samples and evaluating their mechanical properties. The results are given in Table 5. It can be seen that even in the fifth processing cycle, mechanical property changes marginally for control blend having 0.3 wt % DCP content. Beyond that level of DCP (i.e. 0.5 wt% or more), samples are not good for further processing. They are weak and show melt fracture and deteriorations of properties. However, for cured nanocomposite, all the peroxide free radical formed during processing does not take part in the cross-linking reaction. Most of the free radical takes part in the grafting reaction with polymer chain. As a result, extent of cross-linking is less for the cured nanocomposites and can be reprocessed up to 0.5 wt% DCP loading with much effect in its properties.
Reprocessability studies of DCP-cured LLDPE/EMA/Closite®20A nanocomposites.
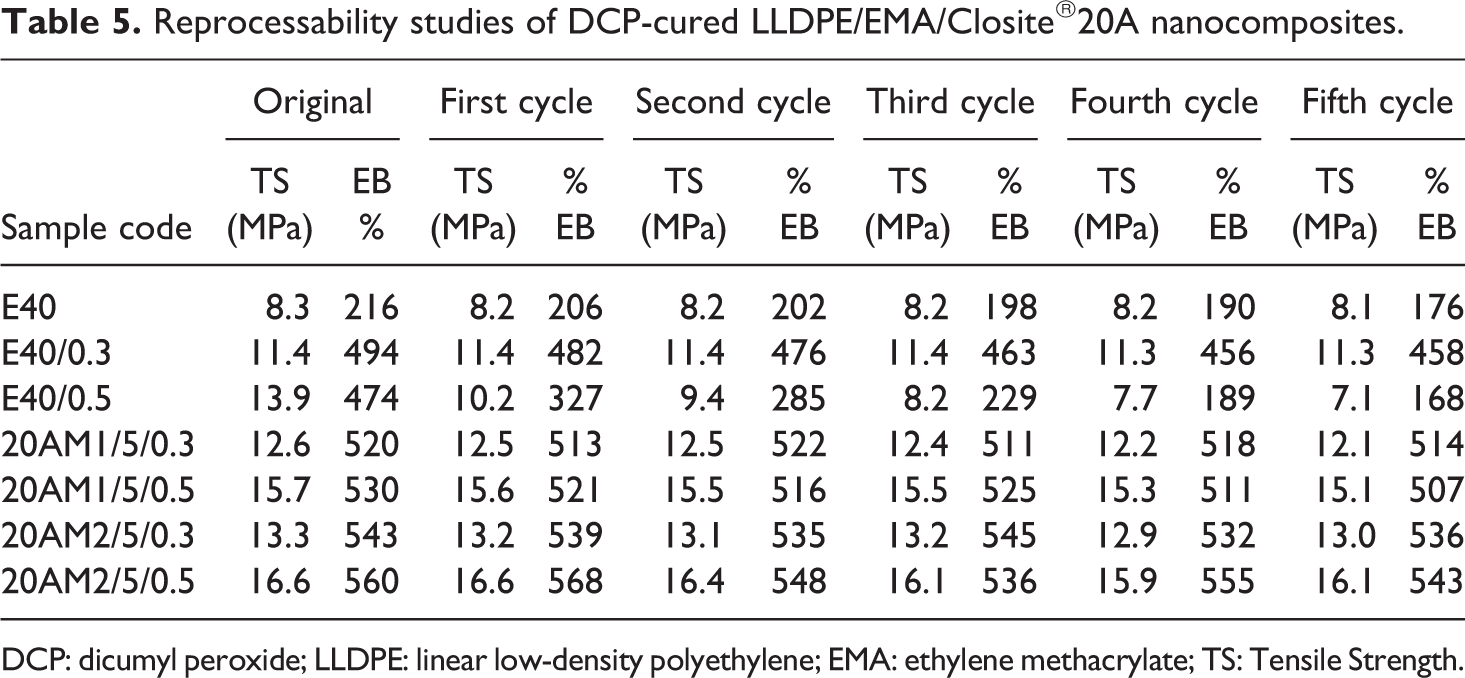
DCP: dicumyl peroxide; LLDPE: linear low-density polyethylene; EMA: ethylene methacrylate; TS: Tensile Strength.
Conclusion
Controlled DCP (0.3 and 0.5 wt% DCP) curing stabilizes the micro- and nanoscale morphology of LLDPE/EMA/Cloisite®20A nanocomposites. DCP initiates free radical grafting between polyolefins and clay modifier resulting in an exfoliated morphology. Significant improvements in mechanical and dynamic mechanical properties have been observed for DCP-cured nanocomposites. The set properties are also improved. Gel content studies show the occurrence of cross-linking in the nanocomposite vulcanizates, although extent of gelation is much less than that of the unfilled vulcanizates. LLDPE/EMA/Cloisite®20A nanocomposites cured with 0.5 wt% DCP or lower can be reprocessed. Therefore, using nanoclay reinforcement followed by controlled DCP curing (low level of DCP) can be applied to LLDPE/EMA systems to improve their technical properties as well as to stabilize their micro- and nanoscale morphology.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
