Abstract
Polyphenylene sulfide (PPS)-expanded graphite (ExGr) conducting nanocomposites have been prepared by powder mixing and in situ polymerization routes after sonicating ExGr particles in acetone. Synthesized PPS has been used to make powder mixed composites. The powder mixed composites exhibit a percolation threshold of 3 wt% due to the formation of graphite nanosheets. When PPS-ExGr composites are prepared by in situ polymerization route, very low electrical percolation threshold less than 0.5 wt% ExGr is obtained. The low percolation threshold obtained is attributed to better dispersion of ExGr nanosheets in the polymer matrix when compared to powder mixed composites. The synthesized PPS has been characterized by X-ray diffraction, differential scanning calorimetry, and infrared spectroscopy. The formation of graphite nanosheets has been confirmed by transmission and scanning electron microcopy analysis.
Keywords
Introduction
Applications such as fuel cells, 1 –3 Electromagnetic Interference (EMI) Shielding devices, 4 –6 anticorrosion coatings, 7 –9 and so on employ conducting polymer composites (CPCs) due to easy synthesis and processing. Each application demands certain level of electrical conductivity of CPCs. In binary conducting composites, polymer matrix being insulator, required level of electrical conductivity is obtained by proper selection of fillers, processing routes, and so on. The minimum concentration of fillers in polymer matrix at which the electrical conductivity starts increasing to many orders is known as electrical percolation threshold. The percolation threshold depends on filler particle size, aspect ratio, processing routes, orientation, and so on. In order to reduce the percolation threshold, nanofillers such as carbon nanotubes (CNTs), 10 –12 carbon nanofiber, 13 –15 graphene 16 –18 etc. are being focused. Reducing the percolation threshold helps in easy processing of CPCs. Conducting hybrid composites have also been focused in which nanofillers are used as the second conducting component. 19 In hybrid composites, the nanofillers will be used along with the conventional cheap fillers, and hence percolation threshold can be reduced compared to binary composites with micron-sized filler alone. It has been shown for polyethersulfone (PES)-graphite-carbon black (CB) hybrid composites solution blending process results in graphite particle size reduction which results in better dispersion when compared to powder mixed hybrid composites. 20 Hence the electrical percolation threshold is reduced in the case of solution-blended PES-graphite-CB hybrid composites compared to powder mixed ones. Novel material such as expanded graphite obtained by acid intercalation in graphite layers, and subsequent heat treatment 21 –23 has been given much attention because of easy synthesis of graphite nanosheets.
The percolation threshold is also dependent on the insulating polymer characteristics. Thermoplastic polymers such as polypropylene, polyethylene, polystyrene, and so on have been focused to synthesize CPCs. Polyphenylene sulfide (PPS) is a high-temperature thermoplastic that retains structural stability at elevated temperatures. PPS is inherently flame retardant and resistant to oxidation and hydrolysis. Compared to high-temperature polyesters, PPS offers excellent dimensional stability, abrasion, and creep resistance. Its importance can be gauged from the fact that it is being used in electronic and automotive industries due to high temperature stability.
Ramanujam and Radhakrishnan 24 have studied charge transport and impedance characteristics of PPS-expanded graphite (ExGr) hybrid composites in which commercial grade PPS has been used. With polyether sulfone, conducting composites were made and basic understanding of charge transport has been reported. 25 Goyal and Kadam 26 have reported low percolation threshold ∼2 wt% in PPS-ExGr composites. They have also studied dielectric constant and dissipation factor of PPS-ExGr nanocomposites. Commercial PPS has been used by them. However, the authors have not reported the effect of different processing routes on the electrical percolation threshold. However, there are reports on the effect of processing routes for different system such as SWCNT-PET nanocomposites. The melt-blended nanocomposites exhibits higher percolation than the one obtained for in situ polymerized samples as reported. 27 The reason for higher percolation threshold in melt mixed composites is due to poor dispersion of filler particles in the polymer matrix. Zhao et al. 28 have studied electrical, mechanical, and thermal properties of PPS-ExGr binary composites. They have reported less than 1 wt% as percolation threshold for the binary composites, when sonicated ExGr is mixed with PPS. The authors have employed melt compounding method (Haake rheomix) for making conducting composites. For 4 wt% sonicated ExGr, the crystallization temperature of PPS was shown to increase by 11°C. Since, in rheomix due to high shear, better breaking and dispersion of ExGr nanosheets could have been obtained. However, they have also not reported the effect of synthesis routes such as in situ polymerization on the electrical percolation threshold.
Since there are no reports in the literature regarding synthesis of PPS-ExGr composites by in situ polymerization technique to the best of our knowledge, an attempt has been made to understand the electrical percolation threshold in PPS-ExGr binary composites synthesized by powder mixing and in situ polymerization routes.
Experimental
Materials
PPS has been procured from (Ryton P4 powder) Chevron Philips (Singapore), whose MFI is 75 g/10 min determined through ASTM D1283 standard and ExGr was procured from ARCI (Hyderabad, Telangana, India) as free sample with expansion ratio ∼100.
Composite preparation
Powder mixing route
Required amount of ExGr is sonicated in 50–100 ml of acetone for 10 min and then mixed with synthesized PPS powder in 50 ml acetone as per the calculation. The resultant mixture is sonicated for 3 min and then poured in a petri dish. Sonication is done in the ultrasonic bath (170–245 VAC 50 Hz single phase) from Analab Scientific Instruments Ltd (Vadodara, Gujarat, India) at a frequency 33 KHz. After acetone evaporation, the resultant composite has been dried and mixed in a mortar with the help of a pestle for 20 min manually. Pellets of diameter 13 mm are made by taking 0.5 g of the sieved composite (100 mesh) and applying 3 ton load for 3 min at room temperature.
In situ polymerization route
PPS is synthesized by making DBB in
First PPS is synthesized at 180°C by dissolving required amounts of p-DBB in NMP and sodium sulfide in methanol taken in a round-bottomed flask kept in an oil bath, and the reaction has been carried out for 18 h. The round-bottomed flask is attached to a reflux condenser to make sure that the vaporized solvent molecules are condensed back into the flask so that the chemical reaction is continued. The dissolved sodium sulfide in methanol is added slowly to dissolved p-DBB in NMP while the stirring is on. The temperature has been slowly increased to the required temperature. After 18 h of chemical reaction the solid polymer has been separated and washed with methanol repeatedly till the odor of NMP is subsided and vacuum dried. From the yield of that reaction, the required amount of ExGr corresponding to different weight fraction is added after sonicating in NMP when in situ polymerization has been carried out. In in situ polymerization route, first the ExGr particles are soaked in NMP overnight into which already 1,4-dibromobenzene of required amount has been dissolved. Then the ExGr particles in NMP are ultrasonicated using a sonicator as mentioned in the composites preparation section for 10 min after which the polymerization is carried out by adding dissolved sodium sulfide in methanol and increasing the temperature slowly to 180°C. Stirring of the mixture of dissolved 1,4-dibromobenzene in NMP, sodium sulfide in methanol and sonicated ExGr has been continued. Once the reaction has been completed, the product is washed with methanol and vacuum dried. The mixture is then ground in the mortar and pellets of diameter 13 mm are made by applying 3 ton load for 3 min on 0.5 g of composites at room temperature.
Measuring electrical properties
Direct current (DC) resistance of different composites has been measured with Keithley system electrometer (model: 6514) as reported elsewhere. 24 The conductivity data presented are the average of data collected for each composition from three different pellets.
Structural analysis
Structural characterization of synthesized composites has been carried out in powder x-ray diffractometer model xpert pro from Pananalytical with copper
TEM and SEM studies
Cross section of powder mixed PPS-ExGr composite after etching in presence of alpha chloronaphthalene vapors is seen under scanning electron microscope (SEM) Leica-440 model (UK). Thin section of in situ polymerized PPS-ExGr composite has been cut using Leica UCT microtome at room temperature and transferred to copper grid which has been mounted in transmission electron microscope (TEM) Model Joel – 1200 EX (Japan) to see nanostructures.
Thermal analysis
With DSC Q10 model from TA Instruments (New Castle, Delaware, USA), differential scanning calorimetric studies have been carried out keeping nitrogen flow rate 50 ml/min and the heating rate at 10°C min−1. Data corresponding to second heating–cooling cycle are presented, as the first cycle is used to destroy variations due to previous history of the sample. The mass taken for DSC analysis is 5 mg.
IR analysis
The powder specimens both synthesized PPS and commercial PPS are mixed with KBr and then loaded in Schimadzu FTIR 8300 spectrometer and analyzed in the range of 625–2500 cm−1. The IR pattern of synthesized PPS is compared with that of commercial Ryton P4 powder.
Results and discussion
DC electrical conductivity studies
Figure 1 depicts dc electrical conductivity variation of PPS-ExGr composites prepared by both powder mixing and in situ polymerization routes. For the synthesis of powder mixed composites, synthesized PPS has been used. The percolation threshold of PPS-ExGr composites synthesized by powder mixing route (3 wt%) is higher than that of composites prepared by in situ polymerization route. Percolation threshold less than 0.5 wt% is obtained for the composites prepared by the later route. The reason for low electrical percolation threshold obtained for composites prepared by in situ polymerization route is due to better dispersion of graphite nanosheets whose agglomeration is avoided in that process unlike the case of powder mixing route. It should be remembered that in in situ polymerization route, first ExGr has been soaked in dissolved 1,4-dibromobenzene overnight and then sonicated for 10 min using an ultrasonicator, and then the polymerization is carried out. Ultrasonication results in the formation of graphite nanosheets as evidenced through TEM analysis. Dissolved DBB molecules could have been very well adsorbed on to the graphite sheets. When the polymerization is started after sonication, the polymer, PPS, penetrates better into the agglomerated nanostructure and separates them well. This results in better dispersion of graphite nanosheets and hence the particle–particle contact is improved at lower loading itself. As a consequence, the percolation threshold is reduced in composites synthesized by in situ polymerization technique. In fact TEM study reveals very clearly few layers graphene formation in that process. XRD, DSC, and IR results clearly demonstrate that the synthesized product is PPS only.
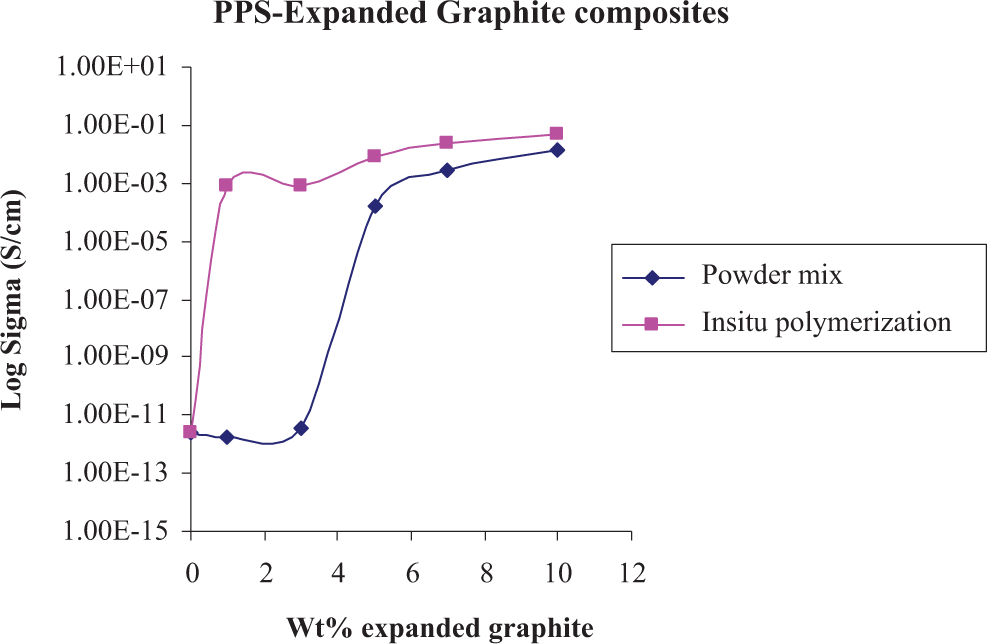
Comparison of DC electrical conductivity variation of powder mixed and in-situ polymerized PPS-ExGr nanocomposites.
Structural analysis
The structural characterization of synthesized PPS has been done through XRD. Figure 2 shows the X-ray diffractogram of synthesized PPS and commercial grade Ryton PPS. PPS has orthorhombic structure with the following four major reflections at different 2
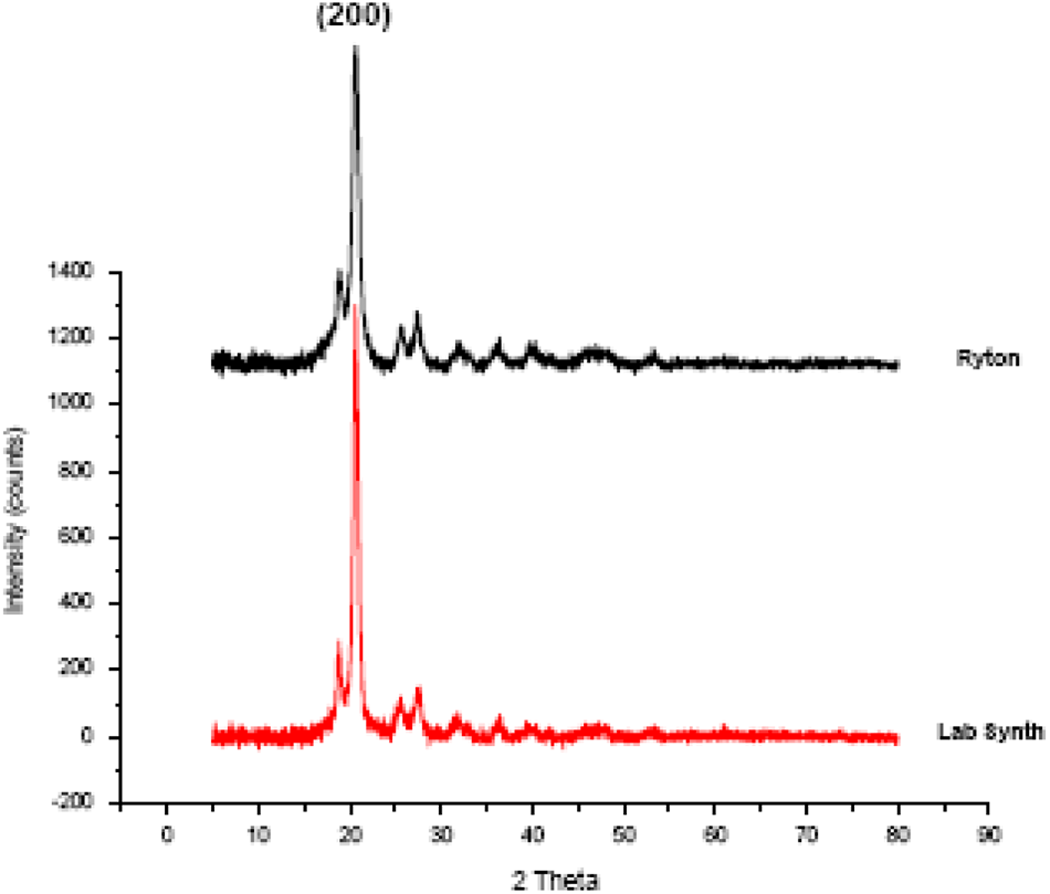
XRD patterns of laboratory synthesized and commercial Ryton PPS.
DSC analysis
In order to confirm that the synthesized product is indeed PPS, the melting temperature of synthesized PPS has been compared with that of commercial high molecular weight Ryton PPS as shown in Figure 3(b) and (a), respectively. Commercial Ryton PPS exhibits melting at ∼280.9°C and the synthesized PPS melts at 271°C. The melting temperature depends on the molecular weight of the polymer. The synthesized PPS might be of low molecular weight polymer when compared to Ryton commercial PPS. In this work, for making powder mixed composites, synthesized PPS has been used and the electrical percolation is compared with that of in situ polymerized PPS-ExGr composites. Since in both routes, PPS has been synthesized, there will not be any effect which could be correlated to molecular weight of the polymer.
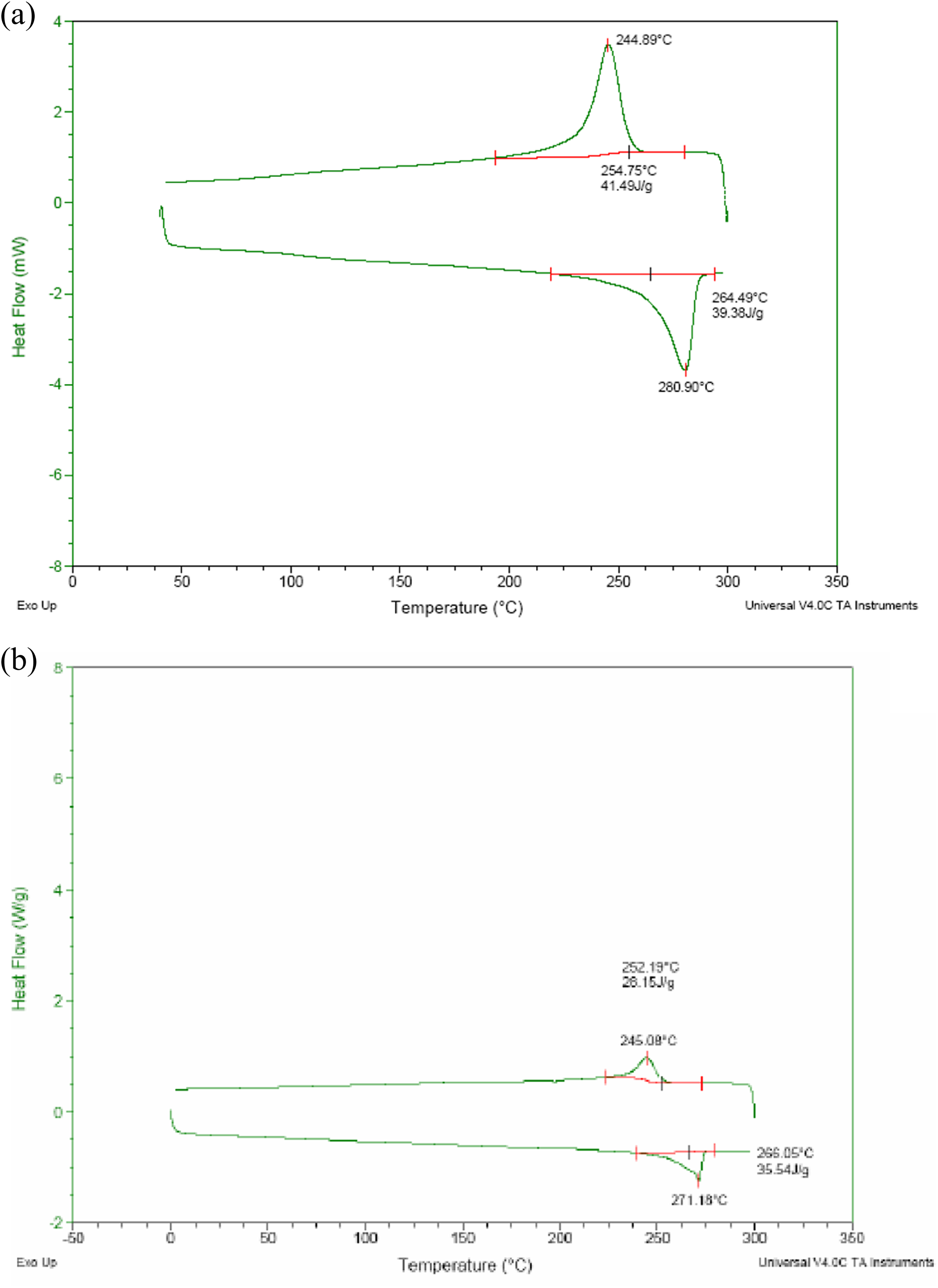
DSC curve of (a) Ryton PPS (b) laboratory synthesized PPS.
IR analysis
The Fourier transform infrared (FTIR) spectrum of synthesized and commercial Ryton PPS match very well, as shown in Figure 4. The characteristic peak at 1470 cm−1 signifying the stretching vibration of C=C in benzene and deformation vibration of =C–H in 1,4 place of benzene at 818 cm−1 confirms that the synthesized product is indeed PPS as reported elsewhere. 30 Those two characteristic peaks are indicated by arrows in Figure 4. All wave numbers are not assigned the kind of vibration, as IR is taken just to compare the position of major peaks. The FTIR pattern of both synthesized and commercial Ryton PPS match very well, and hence the synthesized polymer is confirmed to be PPS.
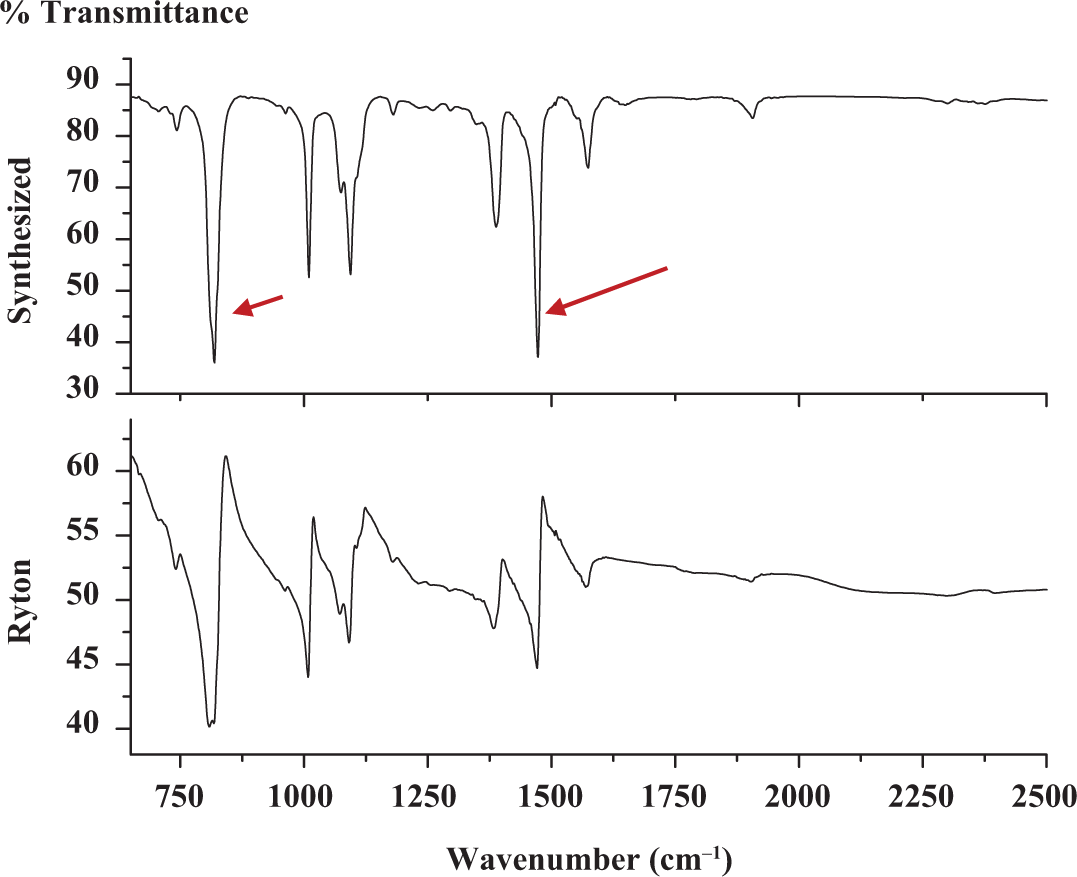
FTIR patterns of commercial Ryton PPS and laboratory synthesized PPS.
TEM and SEM analyses
In order to understand the origin of very low electrical percolation threshold in in situ polymerized PPS-ExGr composites, SEM and TEM analyses are carried out. Figure 5(a) shows the SEM picture of as received ExGr particles. The pore structure of ExGr particles can be clearly seen. When sonicated, the pore structure of ExGr particles is brokened which resulted in the formation of graphite nanosheets. The room temperature ultramicrotome section of in situ polymerized PPS-3 wt% ExGr is observed in HRTEM and shown in Figure 5(b) and (c). It is clear from TEM pictures that very thin graphite nanosheets are formed. Graphitic planes are clearly observable at high resolution and also few layer graphene can be very clearly seen as indicated by the arrows. Figure 5(d) shows thin graphite nanosheets as one could see the interplanar spacing of graphite. Figure 5(e) shows the cross section of SEM picture of powder mixed PPS-3 wt% ExGr composite. The agglomerated graphite nanosheets whose thickness is much higher compared to what has been obtained for in situ polymerized sample can be clearly seen. Agglomerated graphite sheets are buried in the polymer matrix as indicated by the arrows. As very thin graphite nanosheets are obtained when composites are synthesized by in situ polymerization route compared to powder mixing route, at low concentration of fillers, better contact exists which eventually results in lesser percolation threshold. The reason for the formation of very thin graphite nanosheets in in situ polymerized PPS-ExGr composites is that the polymer can penetrate better into the agglomerated graphite nanosheets and separate them as compared to powder mixed composites.
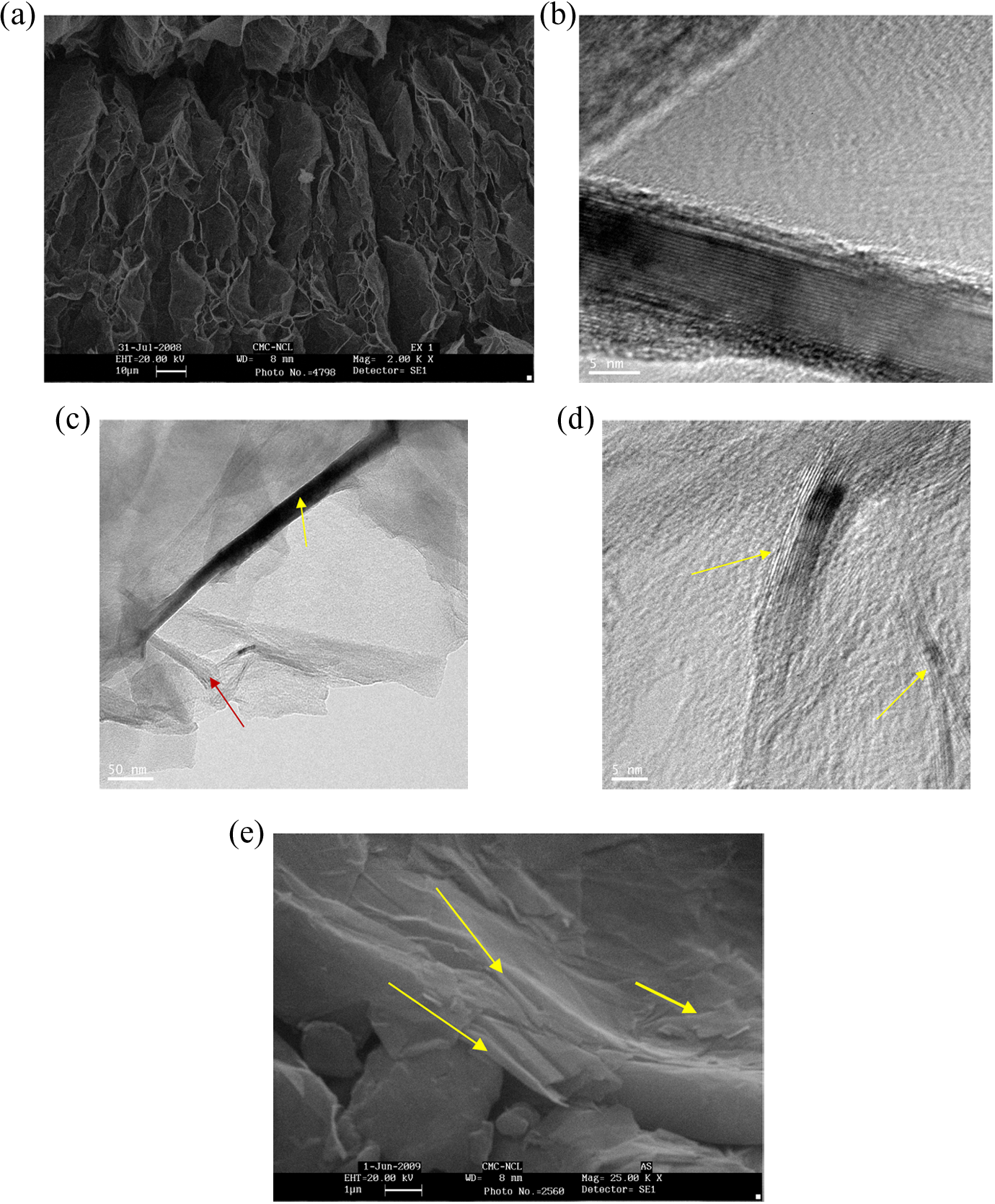
(a) SEM picture of as received ExGr particles (b), (c), and (d) HRTEM picture of ultramicrotome section of in situ polymerized PPS-3 wt% ExGr. (e) SEM picture of cross section of powder mixed PPS-3 wt% ExGr composite.
Conclusion
PPS-ExGr nanocomposites have been prepared by powder mixing and in situ polymerization routes after sonicating ExGr particles in appropriate solvents. Ultrasonication of ExGr particles results in the formation of graphite nanosheets. The electrical percolation threshold has been found to be less than 0.5 wt% for the composites synthesized by the later route due to better penetration of polymer into the agglomerated graphite platelets. This eventually results in better dispersion of graphite nanosheets and hence better contact between the high aspect ratio nanofillers results. The electrical percolation threshold for powder mixed PPS-ExGr composites is at 3 wt% ExGr. SEM study reveals that agglomeration of graphite nanosheets occurs in powder mixed PPS-3 wt% ExGr as compared to in situ polymerized sample resulting in higher percolation threshold. Thus the processing route affects the electrical percolation to a greater extent. The formation of graphite nanosheets has been proved through TEM analysis.
Footnotes
Acknowledgement
The authors sincerely thank the Director, NCL, for allowing us to use the facilities.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
