Abstract
Solution-blended hybrid composites with carbon black (CB) as the second conducting filler in polyethersulfone (PES)-graphite have been prepared by first dissolving PES in dichloro methane. Addition of 1 wt% CB to solution-blended PES-7 wt% graphite results in decrease in the direct current electrical resistivity by four orders compared to the addition of 0 wt% CB. The particle size of graphite is reduced from few micron to nano level as evidenced by transmission electron microscopy analysis resulting in better dispersion. Without the addition of CB, the binary composite namely PES-7 wt% graphite exhibits finite conductivity due to increase in the contact between graphite particles as a result of reduction in the particle size. Comparison of electrical conductivity of PES-7 wt% graphite-2 wt% CB composites prepared by both solution-blending and powder-mixing routes proves the above-mentioned point. The alternating current behaviour, both conductance and effective dielectric constant studies shows that solution-blended composites exhibit higher value of conductance and effective dielectric constant at 0.01 Hz. This can only be attributed to better graphite dispersion in solution-blended composites. The enhancement is due to only particle size reduction of graphite that decreases the interparticular distance as the solution-blended binary composites with CB act as insulators up to 10 wt%. The charge transport at low concentrations of CB in solution-blended hybrid composites is dominated by the interfacial barriers and capacitance effects, while at higher concentrations it is mainly of hopping type at room temperature. The interfacial capacitance increases from 37.6 pF with 0 wt% CB addition to 96 pF with 2 wt% CB addition in PES-7 wt% graphite. Differential scanning calorimetry result suggests that more than 10°C enhancement in the glass transition temperature of PES is obtained for PES-7 wt% graphite-1 wt% CB.
Keywords
Introduction
Conducting polymer composites (CPCs) have been in focus in recent years for their potential applications in the areas such as fuel cells, 1 sensors and actuators, 2,3 electromagnetic interference (EMI) shielding devices, 4 and so on, depending on the level of conductivity required for a specific application. These composites mainly consist of polymer–matrix and conducting fillers, such as metal particles, graphite, carbon black (CB) etc., which lead to a new class of materials with good electrical and mechanical properties. These properties cannot be achieved completely with pure components alone. The electrical conductivity depends on the weight fraction, dispersion, aspect ratio, orientation of fillers, and processing routes for binary composites. For many applications it is required that the filler content should be as minimum as possible without affecting the electrical properties. In this regard, nanofillers like carbon nanotubes, 5 carbon nanofibers, 6 and graphene 7 have received much attention.
The electrical conductivity of conducting polymer composites (CPCs) vary nonlinearly and can be understood through percolation theory,
8
according to which there exists a threshold concentration (
The frequency-dependent behavior of CPCs needs to be understood for EMI shielding and antistatic applications. In this regard, polymer-CB composites have been studied extensively.
10,11
With the addition of the conducting filler, the onset frequency of increase in alternating current (AC) conductivity shifts to higher value. The AC conductivity varies with frequency as given in Equation (1).
where
There are reports in the literature regarding polymer-based hybrid composites. In our group, 14 we have shown that the addition of small amount of CB in polyethersulfone (PES)-graphite system enhances electrical conductivity significantly. Sumfleth et al. 15 have proved the synergistic effect in titania-doped multiwalled carbon nanotube (MWCNT)-epoxy composites. Raja et al. 16 investigated epoxy-graphite nanoplatelet-CB hybrid composites, and they showed that the thermal conductivity of these composites is significantly higher than that of pure epoxy. Zhang et al. 17 investigated the synergistic effect in polypropylene (PP)-MWCNT-CB hybrid composites. They showed enhanced electrical conductivity when two conducting fillers were used. Few reports are found for conducting hybrid composites especially polyphenylene sulfide (PPS)–graphite 18 and PP-graphite 19 based, and very high loading of graphite and CB have been employed to make the composites highly conducting to be used for bipolar plates. Ramanujam et al. 20 have studied impedance analysis of PPS-graphite–expanded graphite hybrid composites.
Though work related to polymer-based hybrid composites can be found in the literature, no systematic studies have been carried out to comment on the AC behavior of PES-graphite hybrid composites. Thus, the objective of the present work is to understand the effect of addition of CB in solution-blended PES-7 wt% graphite on the electrical conductivity by studying its DC, AC behavior especially impedance, and dielectric behaviour. Differential scanning calorimetry (DSC) analysis of PES-graphite-based hybrid system is also reported. The formation of nanographite due to solution blending is addressed.
Experimental
Materials
PES (3600P) powder was procured from Gharda chemicals, Mumbai, India (now sold by Solvay Advaned Polymers). The viscosity of this solution grade is 275 cP. Graphite is a natural flake graphite powder (200 mesh) supplied by Carbon enterprises, Pune, India. CB is a conducting grade supplied by Carbon enterprises (Pune, India).
Composite preparation
The composites were prepared by solution-blending method. In this method, a known quantity of PES was dissolved in 100 ml dichloro methane (DCM) and stirred well until all polymers got dissolved. As per weight fraction calculation corresponding to 10 g of hybrid composite, a known amount of CB was added with the dissolved polymer in a stoppered conical flask and stirred continuously for 2 h. Then the required amount of graphite was added and the stirring continued for another 12 h. After stirring, the hybrid composites were poured in a petri dish and vacuum dried. The resulting cake was crushed, sieved (100 mesh), and the powder was used for making pellets. Pellets were made by taking 1 g of the composite by applying 3 ton load for 3 min at room temperature. Powder-mixed composites were prepared by first mixing sieved PES (100 mesh) with the required amount of CB for 15 min. To this mixture, the calculated amount of graphite was added and the mixing was continued for another 15 min in a mortar with the help of a pestle.
Measuring electrical properties
DC resistance of different hybrid composites prepared was measured using programmable electrometer (Model: 6514; Keithley Instruments, Cleveland, Ohio, USA) by applying a constant load (8 kg/cm2) to reduce the contact resistance. Platinum electrodes of 1 cm2 area were used as electrodes. AC measurements (impedance and dielectric studies) were done on samples in a specially constructed cell with platinum electrodes of area 1 cm2 as contact electrodes using frequency response analyzer SI1255 (Solartron Analytical, Hampshire, UK) with dielectric interface 1296. The impedance plots were fit with ZVIEW® software.
Sample preparation for TEM and SEM studies
A small amount of PES-graphite composite was put in DCM to dissolve the polymer and stirred for a minute. A drop of the solution was transferred to copper grid and mounted in transmission electron microscopy (TEM) Model JEOL-1200 EX (JEOL, Tokyo, Japan). Scanning electron microscopy (SEM) pictures of pure graphite powder were taken using SEM Leica-440.
Thermal analysis
DSC studies were carried out with Q-10 model from TA instruments (New Castle, Delaware, USA) in nitrogen atmosphere. Nitrogen flow rate was 50 ml/min and the heating rate was 10°C/min.
Results and discussion
DC conductivity studies
Figure 1(a) depicts the variation in through thickness resistance of solution-blended PES-CB, PES-graphite composites. The electrode area and thickness of the sample were kept the same, that is, 1 cm2 and 5 mm, respectively. It is clear from the figure that the resistances of PES-CB composites are higher than that of PES-graphite composites. The electrical conductivity of graphite is higher than that of CB, and CB particles might have formed agglomeration in DCM. Further the percolation threshold of PES-CB composites can be identified at 10 wt% after which the resistance starts decreasing; whereas for PES-graphite composites, it is between 5–10 wt%. The solution-blended PES-10 wt% graphite exhibits a resistance of approximately 13.1 MΩ, whereas PES-5 wt% graphite exhibits a value of 4.3 × 1010 Ω which is in the insulating range. So it is logical to think of hybrid composites to improve the level of conductivity, keeping the percolation threshold as low as possible and hence we have used CB as the second conducting filler. Very small amount of CB is sufficient to cause higher conductivity in the PES-graphite composites. Further the density of CB is much lesser than that of natural graphite (1.8–2.1 g/cm3). In order to understand the effect of CB, the variation in resistance was measured by keeping the graphite concentration same. Figure 1(b) depicts the variation in through thickness resistance (5 mm thick and 1 cm2 area) of PES-
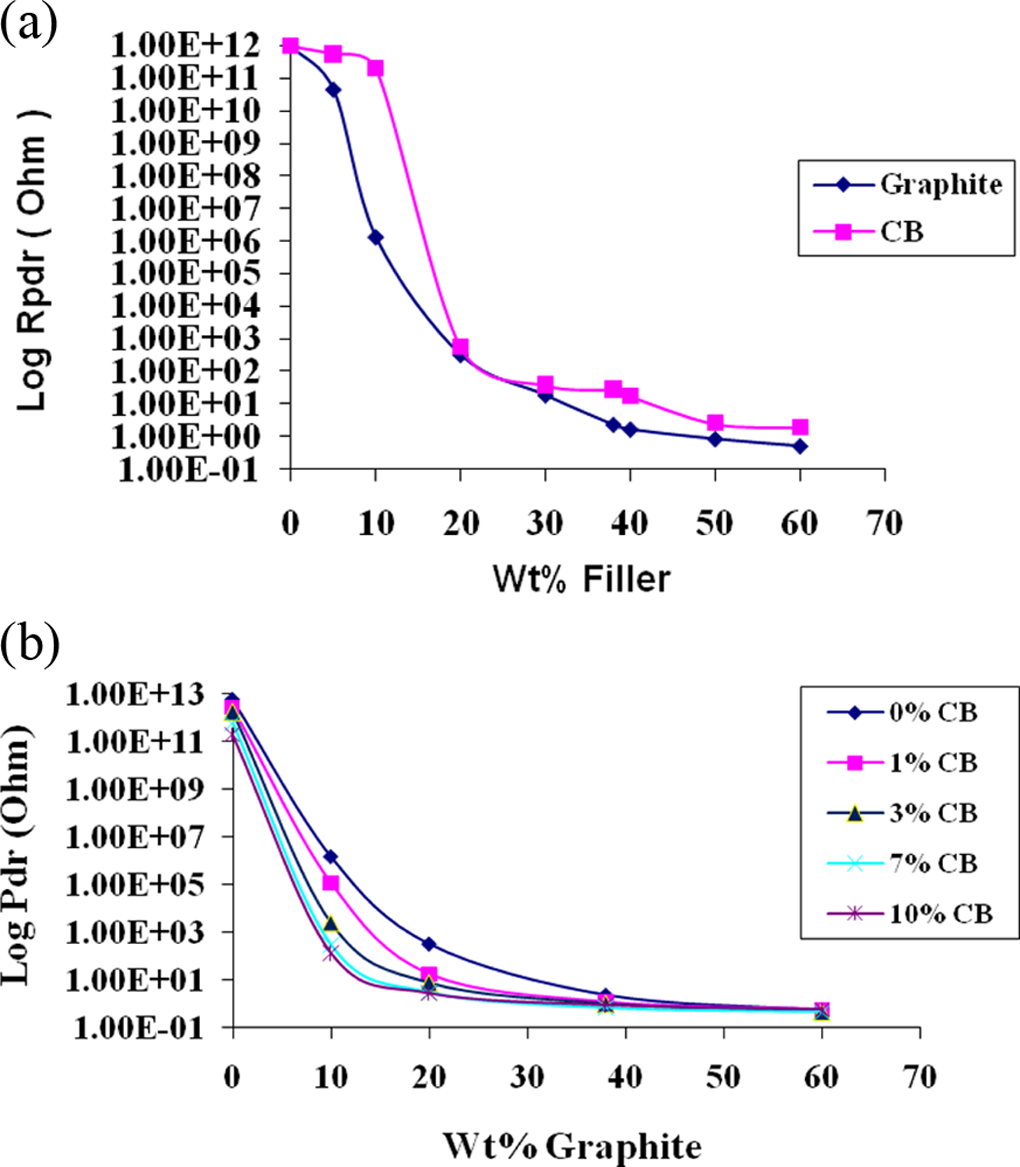
DC through thickness resistance (sample thickness: 5 mm; area: 1 cm2) variation in solution-blended (a) PES-CB and PES-graphite binary composites (b) PES-graphite-CB hybrid composites. DC: direct current; PES: polyethersulfone; CB: carbon black. CB: carbon black.

(a) SEM picture of graphite (b) and (c) TEM pictures of PES-7 wt% graphite solution-blended sample. SEM: scanning electron microscopy; TEM: transmission electron microscopy; PES: polyethersulfone.
Frequency-dependent conductance
Figure 3(a) shows the variation in log conductance with log frequency (log
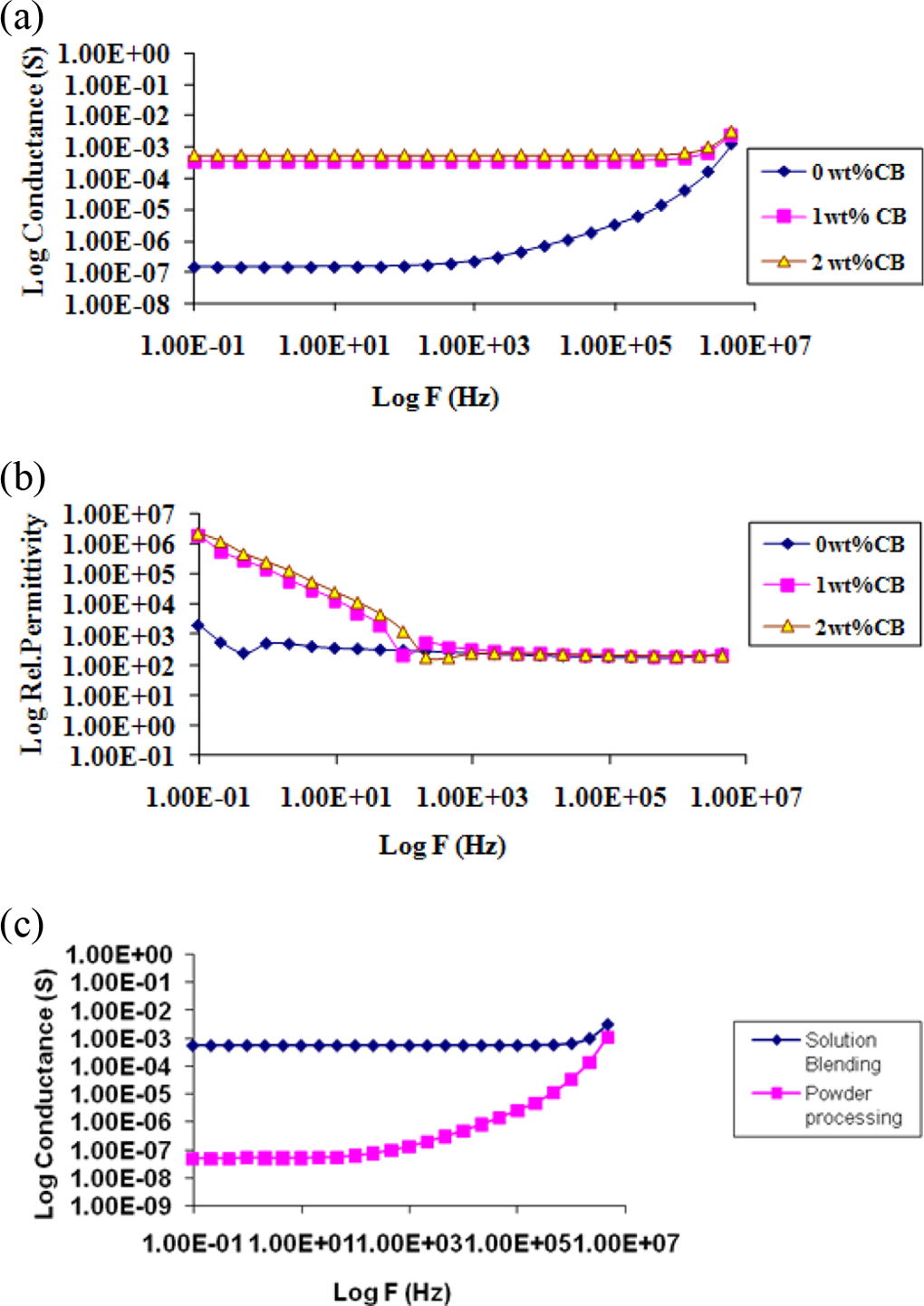
(a) AC conductance (b) effective dielectric constant variation in PES-7 wt% graphite-
Exponent “

Figure 4(a) and 4(b) depicts a model that helps to understand the enhancement in the conductance of hybrid composites. The second conducting component forms network between graphite particles so that the barrier is decreased for the electrons to jump from one particle to the other. If our model is true then the interfacial capacitance should increase because of the occupation of CB particles in the interspace of graphite particles as this will lead to decrease in the interparticular distance for the charge transport. Considering conducting particle-polymer-conducting particle configuration as parallel plate capacitors, decrease in the interparticular distance will lead to increase in the interfacial capacitance. Through impedance measurements, it is possible to evaluate interfacial capacitance that is explained in the following section. The validity of this model can be checked by studying the dielectric properties of the hybrid composites as the interfacial capacitance is related to effective dielectric constant (
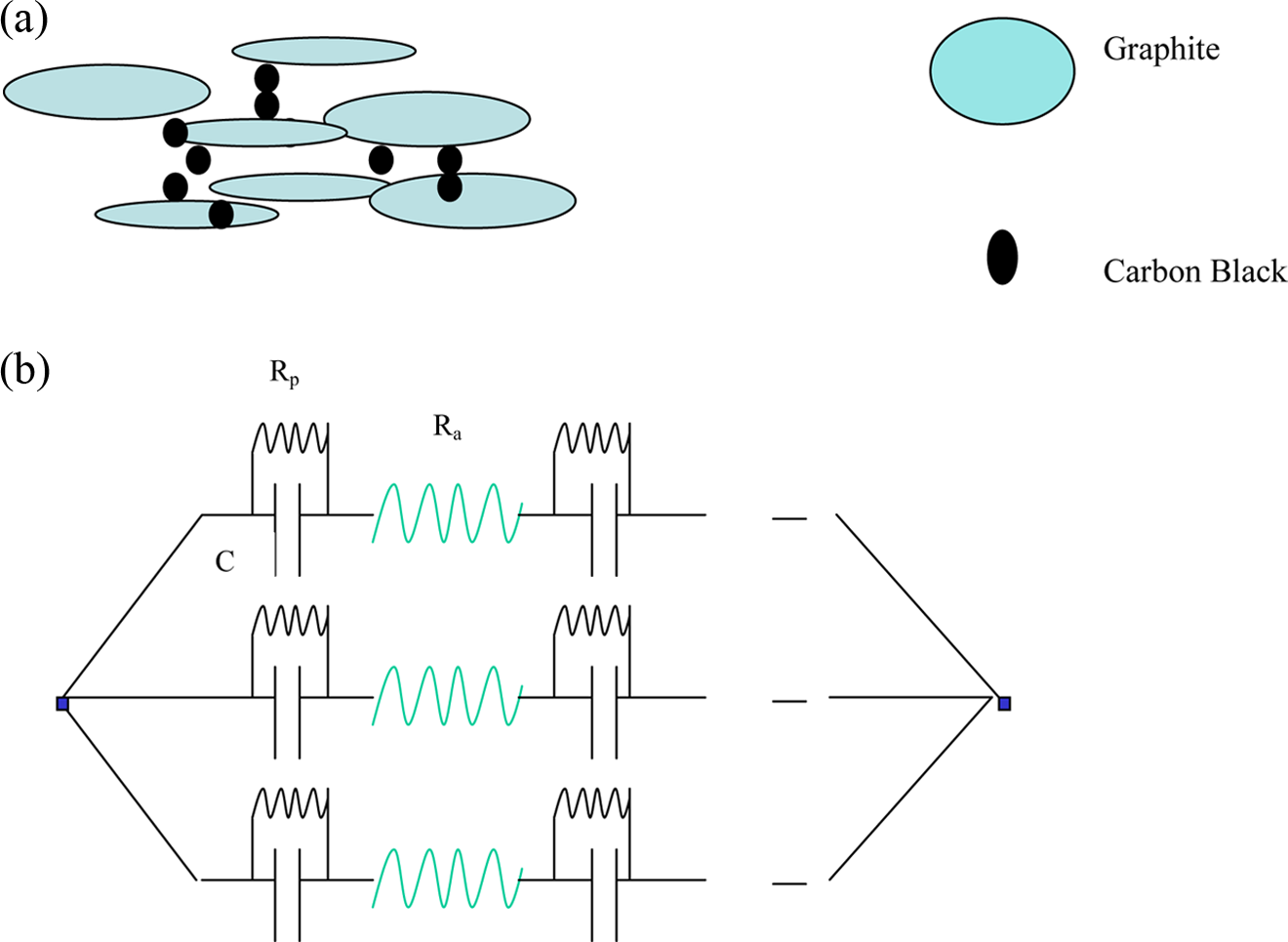
(a) Model of hybrid composites (b) equivalent circuit diagram of conducting polymer composites.
where
Dielectric studies
Figure 3(b) depicts the variation in
Impedance analysis
Impedance spectroscopy is used to understand corrosion,
23
degree of dispersion of filler,
24
and so on, and it is a nondestructive technique. The details of impedance spectroscopy measurements have been described elsewhere.
25
The parameters, which are useful to comment on the conduction process, are aggregate resistance (
Figure 5(a) and 5(b) shows the room temperature impedance plots of PES-7 wt% graphite and PES-7 wt% graphite-1 wt% CB hybrid composite prepared by solution-blending route. Various model parameters extracted from the impedance plots in Figure 5 are shown in Table 2. It is clear that with the addition of 1 wt% CB in solution-blended PES-7 wt% graphite increases
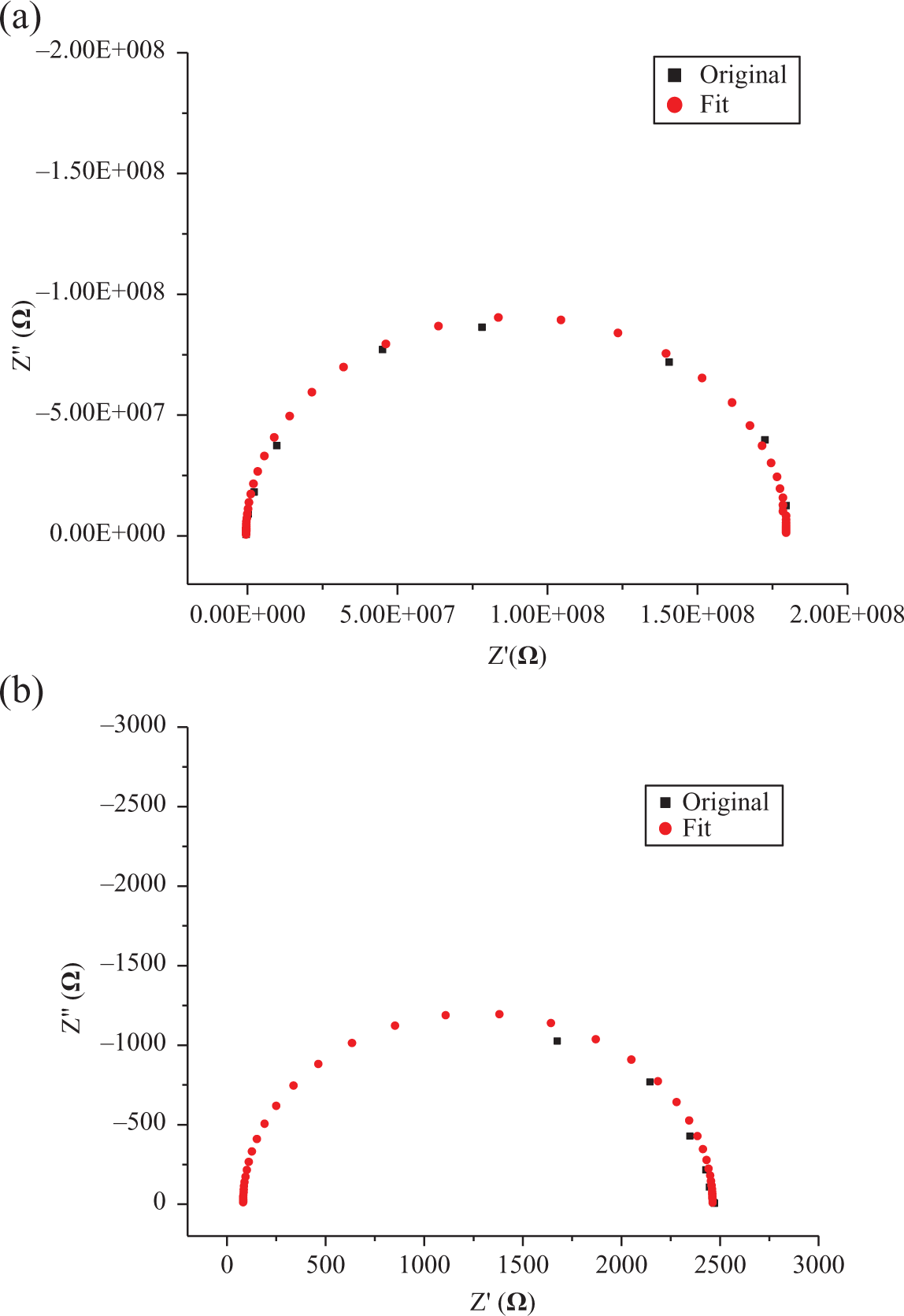
Impedance plots of (a) PES-7 wt% graphite (b) PES-7 wt% graphite-1 wt% CB solution-blended composites. PES: polyethersulfone; CB: carbon black.
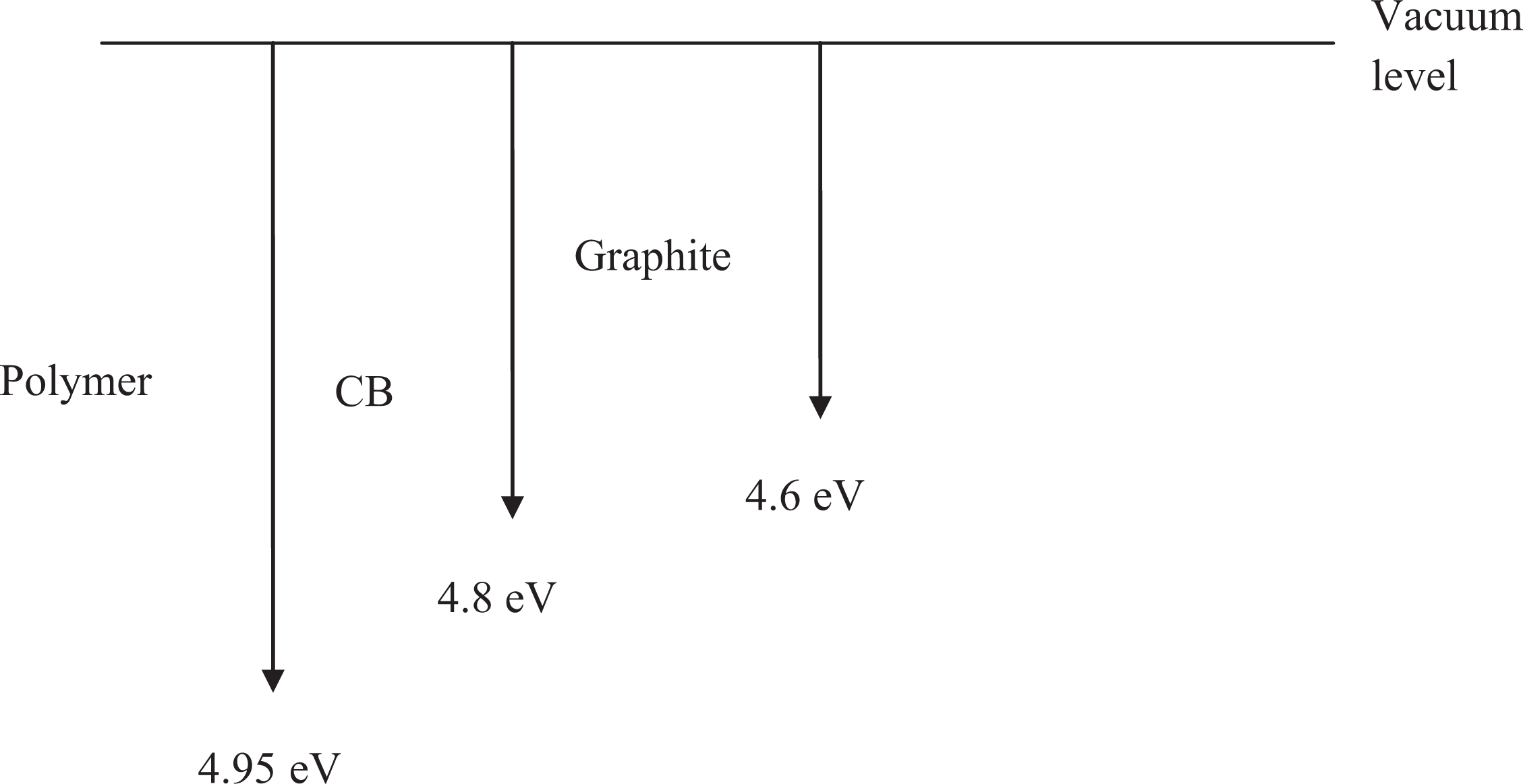
Work function diagram of PES-graphite-CB hybrid composites. PES: polyethersulfone; CB: carbon black.
Model parameters extracted from impedance measurement.

PES: polyethersulfone; CB: carbon black;
DSC analysis
DSC plots of pure PES and solution blended PES-7 wt% graphite- 1 wt% CB are shown in Figure 7(a) and 7(b) respectively. It is clear that the glass transition temperature (
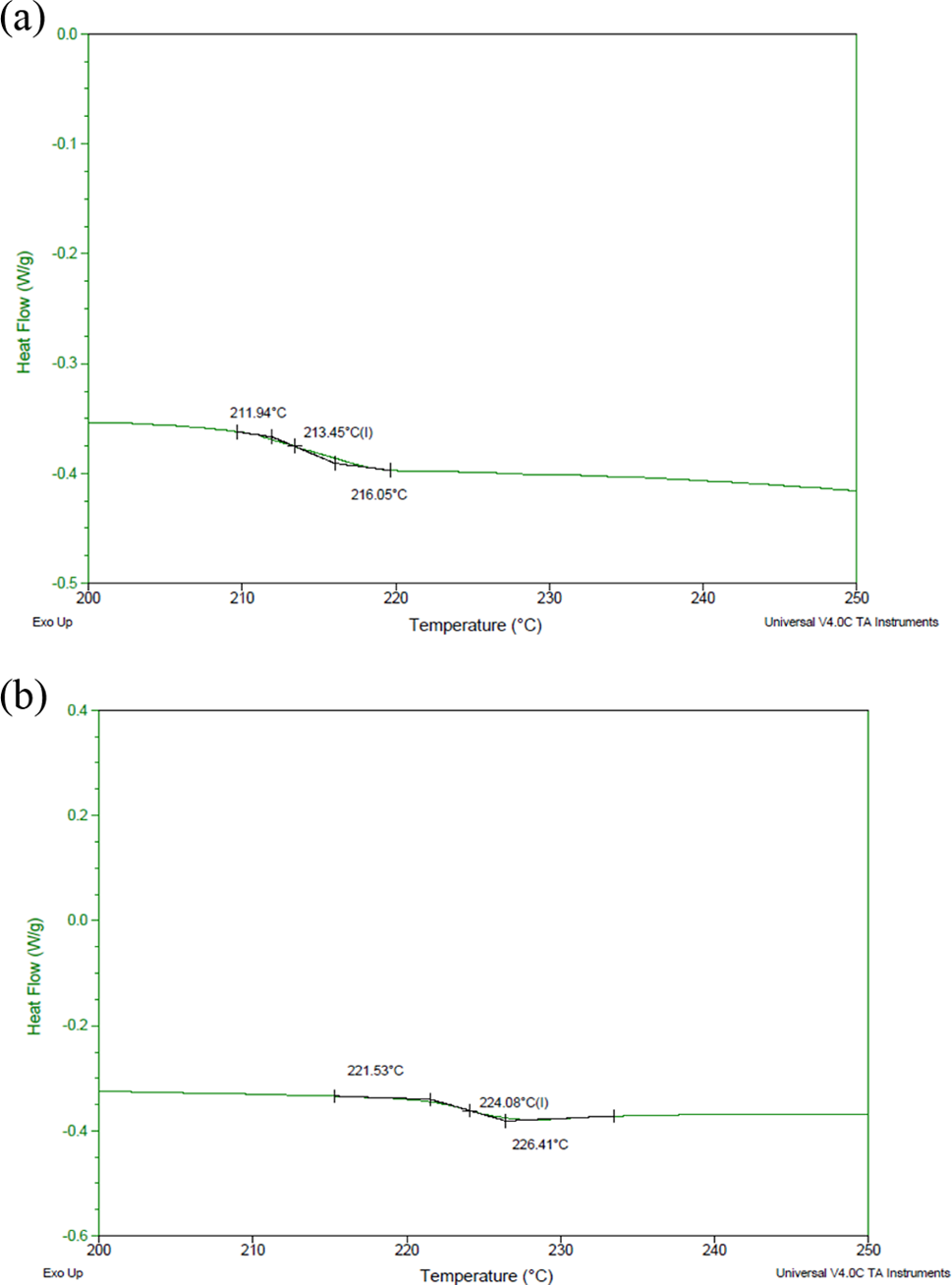
DSC curves of (a) pure PES and (b) PES-7 wt% graphite-1 wt% CB solution-blended composite. DSC: differential scanning calorimetry; PES: polyethersulfone; CB: carbon black.
Conclusions
The electrical properties of PES-graphite-CB hybrid composites prepared by solution-blending method have been studied in detail. It has been experimentally observed that the percolation threshold of PES-graphite composites lies between 5 and 10 wt%; whereas for PES-CB composites, it is at 10 wt%. One particular composition, PES-7 wt% graphite, nearer to the percolation threshold has been chosen to study the effect of addition of CB. With the addition of CB, the DC resistance of solution-blended sample decreases. In the solution-blended samples, graphite particle size is reduced to nano level as suggested by TEM results. The AC conductance studies support the DC results and the charge transport occurs by hopping mechanism for CB loading greater than 2 wt% in PES-7 wt% graphite solution-blended sample. For lesser loading, the capacitance effect dominates.
A model has been proposed in which graphite-polymer-CB type parallel plate capacitor configuration dominates. The validity of the model has been checked by dielectric measurements, which show that the effective dielectric constant increases at low frequency, implying increase in the
It is worth mentioning here that the value of conductivity obtained for PES-7 wt% graphite- 1 wt% CB hybrid composite prepared by solution blending route will match the value obtained for binary composites when the loading of graphite lies between 10-20 wt%. Thus, the hybrid composites are so effective in bringing down the percolation threshold and also in this case, the density of the composite shall be reduced if special applications like bipolar plates for fuel cells are to be developed where the weight, cost, and performance of the plates are so critical for commercialization.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
