Abstract
Magnesium hydroxide (MH) was added into high-density polyethylene (HDPE)/ethylene vinyl-acetate (EVA) copolymer blends with various MH contents (30–60 wt%) to improve the flame retardancy of HDPE/EVA blends. The flammability, morphology of charred residues, thermal stability, crystallization, and mechanical properties of HDPE/EVA/MH composites were investigated by UL-94 test, limiting oxygen index (LOI), cone calorimeter test (CCT), thermogravimetric analysis (TGA), differential scanning calorimetry (DSC), scanning electron microscopy (SEM), and tensile test. The data obtained from LOI, UL-94 test, and CCT revealed that the addition of MH provided improvements in flame retardancy by increasing the LOI values, UL-94 rating, and reducing heat release, carbon monoxide and carbon dioxide emissions along with delayed ignition with increasing the content of MH. The UL-94 V-0 rating and high LOI value were achieved with the incorporation of MH at a loading level higher than 50 wt% in HDPE/EVA blends, which suggested that the formation of intact, consolidated, and thick residue structures on the surfaces of MH-filled composites prevented the underlying polymer materials from burning. DSC results showed that the crystallinity of HDPE/EVA blends increased with increasing the content of MH, resulting in the enhancement of the strength of HDPE/EVA/MH composites. The thermal stability of HDPE/EVA blends decreased due to the addition of MH.
Introduction
Polyethylene (PE) has been used in many fields because of its balanced mechanical properties, chemical resistance, low cost, and good processability. 1,2 Because its chemical constitution consists of carbon and hydrogen, it is easily flammable. In addition, low environmental stress-cracking resistance and poor compatibility with various additives also restrict its further applications in some fields. 3 In order to overcome the above limitations, some attempts have been directed toward blending PE with other polymers, such as ethylene vinyl-acetate copolymer (EVA), polyamide-6 (PA6), and so on. The presence of polar ester groups in EVA chemical structure can provide higher filler loading capacity for PE. 4 In addition, EVA can also improve flexibility, toughness, and transparency of PE. 5 Therefore, the PE/EVA blends have become much demanded for various application fields that require high toughness, environmental stress crack resistance, and so on, such as wire and cable, foam production, packaging films, and heat shrinkable tubes. 6,7
There are a number of publications dealing with various characteristics of polymer blends containing various inorganic fillers. 8,9 For example, Sun et al. 10 studied the thermal stability, flammability, and mechanical properties of the high-density polyethylene (HDPE)/EVA/expandable graphite ternary composites. Ishidi and his coauthors 11 reported the flammability and thermal properties for groundnut shell-HDPE composites. Liu et al. 12 investigated the thermal stability and flame retardancy of HDPE/EVA/fullerene (C60) nanocomposites. Lu et al. 13,14 also reported the influence of clay on the flame retardancy, morphology, and dispersion of polystyrene/PA6 blends. It was concluded that the ternary composites had better flame retardance properties and thermal stability than the conventional one due to well-dispersed inorganic particles in the ternary composites.
Recently, halogen flame retardants are being gradually replaced by halogen-free flame retardants due to the increasing concern about the health and environmental risks. Metal hydroxyl, acting as a halogen-free additive, produces an endothermic reaction during the combustion, which decreases the surface temperature of polymer. In addition, free water can dilute the combustible gas, and the produced metal oxide cuts off sources of heat between the matrix and the surface. 15 But the biggest problem for metal hydroxyl is that its flame retardance efficiency is low. More than 60% loading level is required for acceptable combustion resistance. This directly leads to a sharp decrease of the mechanical properties because of the poor compatibility between inorganic and organic materials. 16 Chen and coauthors 17 tried a silane coupling agent and dimethyl silicone oil to polypropylene/magnesium hydroxide (PP/MH) composites, respectively, and the effects of the surface modifiers on the final properties of the flame-retardant PP/MH composites were investigated and discussed further. Yen et al. 18 studied the synergistic flame retardance effect of the metal hydroxyl in combination with nanoclays for the EVA blends to improve the thermal stability, flame retardancy, and mechanical properties of materials.
Aluminum hydroxide (ATH) and MH have become the focus because of their nontoxic and smoke suppressing properties, low cost, and so on. 18 –21 The decomposition temperature of ATH within a polymer begins near 240°C, and consumes 1300 J/g of ATH, for MH, however, it begins at somewhat higher temperature about 350°C, and consumes 1400 J/g of MH. 22 Therefore, the use of ATH is limited to polymers due to low decomposition temperature. MH decomposes thermally to produce magnesium oxide (MgO) and water vapor at higher temperatures. The formation of MgO during combustion provides a heat shielding effect and inhibits the liberation of the flammable volatile organic compounds into the gas phase while water vapor evolution also contributes positively in terms of diluting fuel in the gas phase. 23,24 This not only acted as a barrier but also acted as an additive to aid the dispersion of MH particles in polymer matrix.
To the best of our knowledge, few works have been published on HDPE/EVA blends with inorganic particles, especially MH. Furthermore, a detailed study of the influence of MH on the HDPE/EVA blends is still lacking. In this work, our aim was to explore concentrations of MH, EVA, and compatibilizer used to improve the flame retardancy and mechanical properties of HDPE/EVA blends. The MH content was varied from 30 wt% to 60 wt% in order to carry out a comprehensive study on the flame retardancy, thermal stability, crystallization behaviors, and mechanical properties of HDPE/EVA/MH composites.
Experimental
Materials
HDPE (5000 S, Melt flow rate (MFR) = 0.923 g/10 min) was obtained from Lanzhou Petrochemical Company, China Petroleum. EVA copolymer (7240 M, VA = 15 wt%, MFR = 1.5 g/10 min) was purchased from TAISOX (Taiwan, China). MH (average particle size = 1.5 μm) was supplied by Yantai FR Chemical Technology Co., Ltd (China). The Maleic anhydride-grafted PE (PE-g-MAH) and silane coupling agent are commercial products. Maleic anhydride-grafted PE is supplied by Nanjing Sutai Polymer Science and Technology Co., Ltd (China). Silane coupling agent is obtained from Nanjing Shuguang Chemical Industry Co., Ltd.
Preparation of composites
The dried HDPE, EVA, and MH powders were absolutely premixed according to the formulation, and then compounded at 170°C using a twin-screw extruder (Model HFB-150/3300, made in Nanjing Ruiya Polymer Processing Equipment Co., Ltd., China). The extrusion was cut into pellets and all the samples were hot-pressed at 170°C into sheets of suitable thickness for tests and characterization. The formulations of the HDPE/EVA/MH composites with various MH are listed in Table 1.
The formulations and flammability of HDPE/EVA/MH composites.

HDPE: high-density polyethylene; EVA: ethylene vinyl-acetate; MH: magnesium hydroxide; PE-g-MAH: Maleic anhydride-grafted polyethylene; LOI: limited oxygen index.
a t 1 was the average flaming burning time of 5 specimens after the 1st flame application.
b t 2 was the average flaming burning time of 5 specimens after the 2nd flame application.
Measurements and characterization
UL-94 test
The UL-94 vertical burning test was performed with a vertical burning instrument (Jiangning, China). The specimens for test were of dimensions of 127 × 12.7 × 3 mm3 according to the ASTM D635-77 standard.
Limiting oxygen index
Limiting oxygen index (LOI) was measured using a JF-4 oxygen index instrument (Jiangning Analysis Instrument Company, China) on the specimen of 120 × 6.5 × 3 mm3 according to the ASTM D2863-77 standard.
Thermogravimetric analysis
The thermal properties of the composites were examined by thermogravimetric analysis (TGA) using NETZSCH TG209F1 thermal analyzer (Germany). In each case, about 8–10 mg specimens were heated from room temperature to 700°C with a linear heating rate of 10°C min−1 under nitrogen (N2) atmosphere.
Morphology analysis
The fractured surfaces of the specimens were investigated using a scanning electron microscope (Model JSM-7500F, Japan). Gold sputter-coated samples were examined using a Cambridge Stereoscan 250 with an accelerating voltage of 20 kV Cambridge (UK).
Cone calorimeter test
The cone calorimeter tests (CCT; Stanton Redcroft, UK) were carried out according to ISO 5660 standard procedures. Each sample of dimensions (100 × 100 × 3 mm3) was wrapped in aluminum foil and exposed horizontally to an external heat flux of 35 kW/m2.
DSC analysis
Differential scanning calorimetry (DSC) thermograms were recorded using a TA instrument Q10 (New Castle, Delaware, USA) at a heating rate of 10oC min−1 under N2 atmosphere. The percent crystallinity was determined by dividing the heat of fusion value by 293 J/g, the heat of fusion of 100% crystalline PE.
Mechanical properties tests
Tensile properties were measured at room temperature by a tensile testing machine AGS-J (Shimadzu Company, Suzhou, China) according to ASTM D412-1998a standard. The tests were performed at a crosshead speed of 50 mm min−1.
Results and discussion
Flammability: UL-94 and LOI tests
The UL-94 and LOI tests are usually used to evaluate the flammability of materials, especially for screening the flame-retardant formulations of the materials. 25 The UL-94 ratings and LOI values of HDPE/EVA/MH composites with different loading level of MH are listed in Table 1, and the burning processes of the composites in the UL-94 test are shown in Figure 1. It was found from Figure 1 and Table 1 that the burning process was significantly different for the matrix and the flame-retardant composites. The HDPE/EVA (HEM0) burned sharply, dripping severely, which could be dangerous and cause “secondary ignition”. The excessive dripping in HDPE/EVA was attributed to the poor flame retardancy. However, the dripping was not evident when MH was added into HDPE/EVA, as shown in Figure 1 (HEM30–II50). In addition, the burning gradually became weak with increasing the MH content. The addition of 50 wt% of MH (samples HEM50, I50, and II50) into HDPE/EVA improved the flame retardancy to UL-94 V-1 rating and further enhanced UL-94 V-0 rating at 60 wt% MH content (sample HEM60).
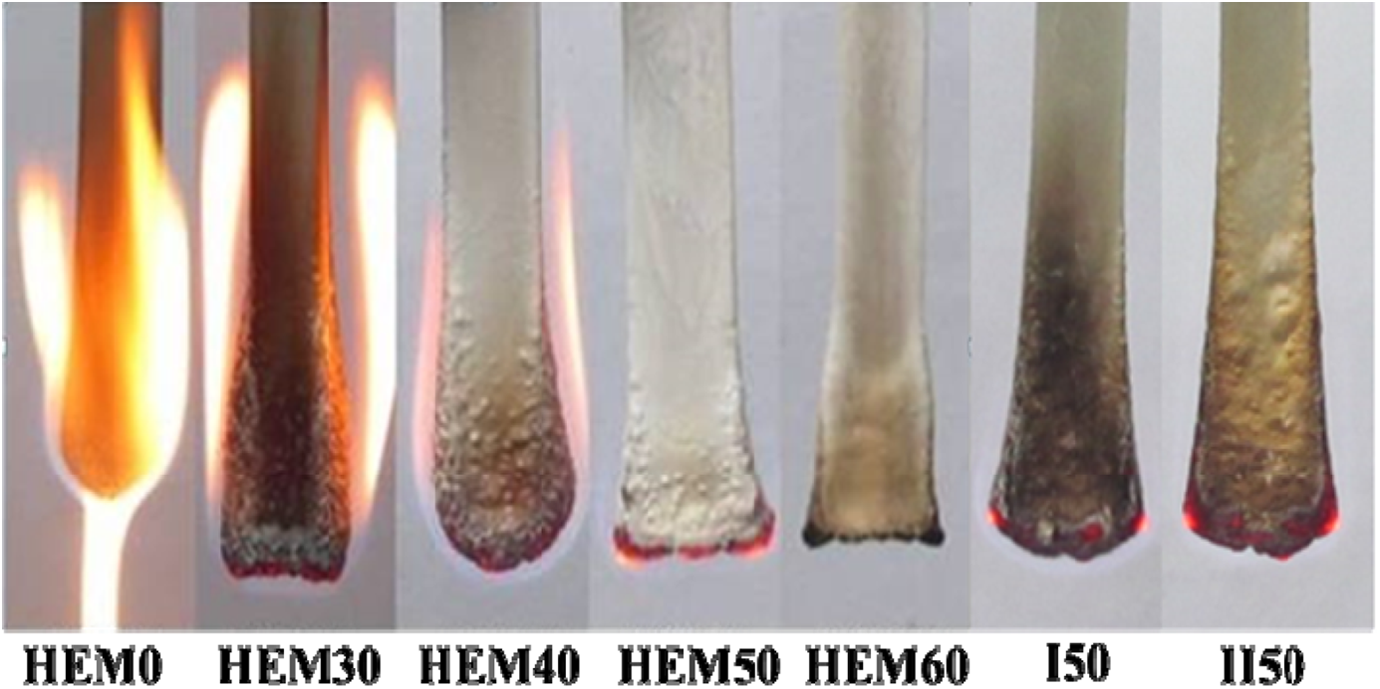
Digital photographs of HDPE/EVA composites with different MH content (formulation: HDPE/EVA = 80/20) during burning in UL-94 test.
It is seen from Table 1 that the LOI value of HDPE/EVA was only 18.9, which indicated that it was easily flammable. The LOI values of HDPE/EVA/MH composites increased with increasing content of MH. The LOI value of HEM30 with 30 wt% MH loading level increased to 26.2, and the LOI value increased to 33.6 for HEM60 with 60 wt% MH content. The LOI values were improved with increasing level of MH owing to the flame retardance effect of MH. Additionally, it is observed from Table 1 that the presence of EVA and PE-g-MAH has a little effect on the LOI values of the HDPE/EVA composites, as presented in the three samples, that is, HEM50, I50, and II50.
Dynamic flammability: CCT
The heat release rate (HRR) curves of HDPE/EVA and HDPE/EVA/MH composites with various MH content are shown in Figure 2. Combustion behaviors of HDPE/EVA were altered by the addition of MH. The HDPE/EVA (HEM0) burned quickly after ignition and a sharp HRR peak appeared at the range of 200–400 s with a peak HRR (PHRR) value of 628.6 kW/m2. The HRR values of HDPE/EVA/MH composites decreased rapidly with the addition of MH, and decreased gradually with increasing content of MH (listed in Table 2). The HRR curve of HDPE/EVA had only a single peak, whereas, there were two peaks for HDPE/EVA/MH composites with 30–60 wt% loading level. The first peak was assigned to flaming before the development of the MgO protective layer. The remaining one was attributed to the destruction of the protective layer with the release of combustible gases when the composites were continuously exposed to the heat. The second peak at the end of burning was commonly observed in CCT and corresponded to the stage where so little polymer remained. This indicated that the decomposition and combustion of polymer matrix were delayed. This was in agreement with the previous studies. 26 As a result, the flame retardance performance of HDPE/EVA was improved, as shown in Table 1. In addition, it was found that the suppression of the heat release was pronounced for MH contents from 30 wt% to 60 wt%, which exhibited a significantly longer burning time and an apparent HRR plateau preceding the peak toward the end of combustion.
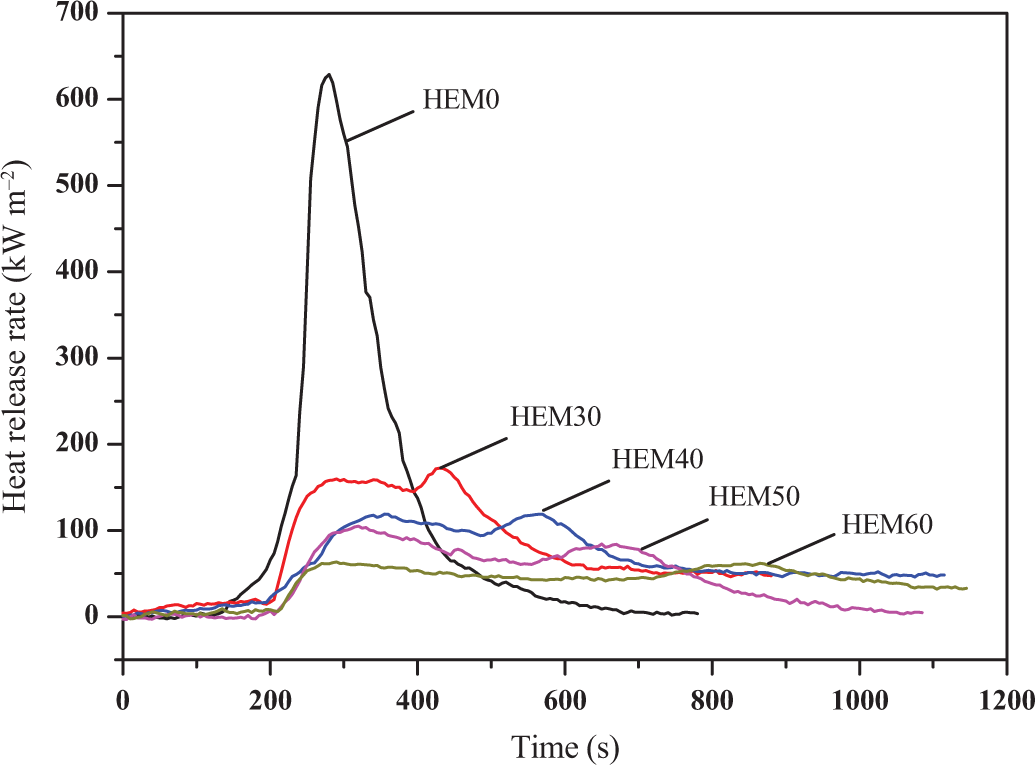
HRR curves of HDPE/EVA and HDPE/EVA/MH composites with different MH content (formulation: HDPE/EVA = 80/20).
Cone calorimetry testing results of HDPE/EVA/MH composites.

FGI: fire growth index; HRR: heat release rate; HDPE: high-density polyethylene; EVA: ethylene vinyl-acetate; MH: magnesium hydroxide; TTI: time to ignition; PHRR: peak HRR; THR: total heat release.
aTime to ignition.
bPeak of HRR, expressing the intensity of a fire.
cFGI, the ratio of PHRR to TTI.
The total heat release (THR) curves versus time for the HDPE/EVA and HDPE/EVA/MH composites are shown in Figure 3. It was found that the changes of THR curves were similar to the HRR curves and their values significantly decreased for the HDPE/EVA/MH composites with 30–60 wt% MH loading level compared with HDPE/EVA. It was proposed that the premature pyrolysis of MH resulted in the formation of residue consisted of MgO, which insulated the polymer matrix inside from the heat and O2. 27
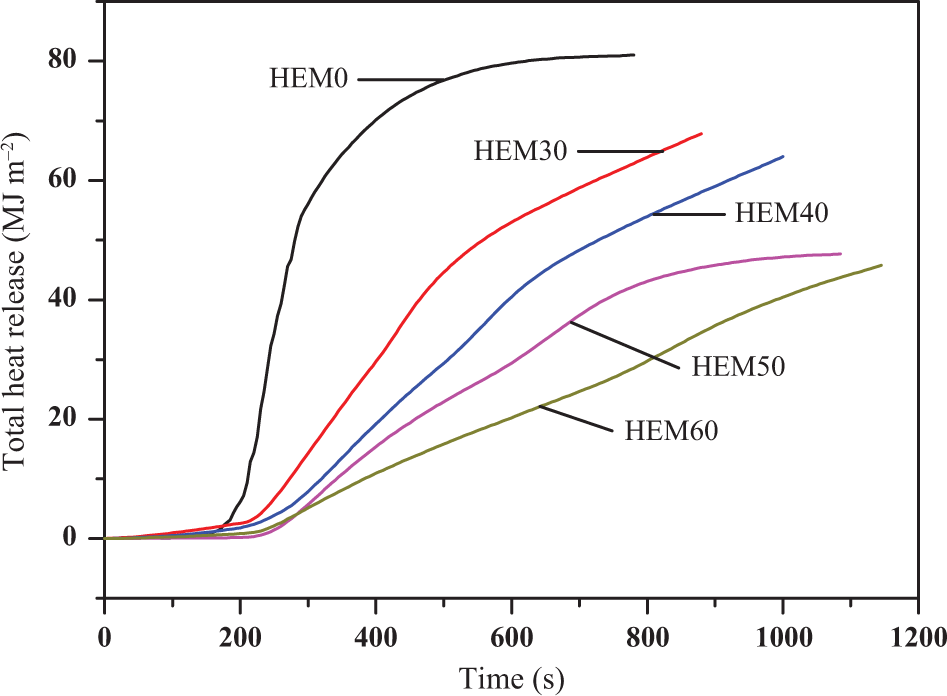
THR curves of HDPE/EVA and HDPE/EVA/MH composites with different MH content (formulation: HDPE/EVA = 80/20).
Figure 4 displays the dynamic mass loss curves versus time for HDPE/EVA and HDPE/EVA/MH composites. It was observed that HDPE/EVA lost its mass quickly after ignition and a dramatic decrease of mass occurred in a short time, whereas the mass losses of HDPE/EVA/MH composites were lower than that of HDPE/EVA, and the mass decreased slowly during the whole combustion, as shown in Figure 4. The mass loss curves of HDPE/EVA/MH composites showed that the flame-retardant additive predominantly acted in such a way that the pyrolysis was suppressed. At the same time, HDPE/EVA left nothing after combustion. However, the residues of HDPE/EVA/MH composites were enhanced due to the incorporation of MH and increased gradually with increasing the content of MH. For example, the residues of HEM30 with 30 wt% MH and HEM60 with 60 wt% MH were 27.8% and 57.5%, respectively. The fire resistance could also be evaluated from the residues after pyrolysis of the materials. Increasing residues delay the release of combustible carbon-containing gases and decreased the exothermicity induced by pyrolysis reactions as well as the thermal conductivity of the surface of burning materials. 28 The residue formation and its compact residues occurred on the surface of the burning materials providing a physical protective barrier.
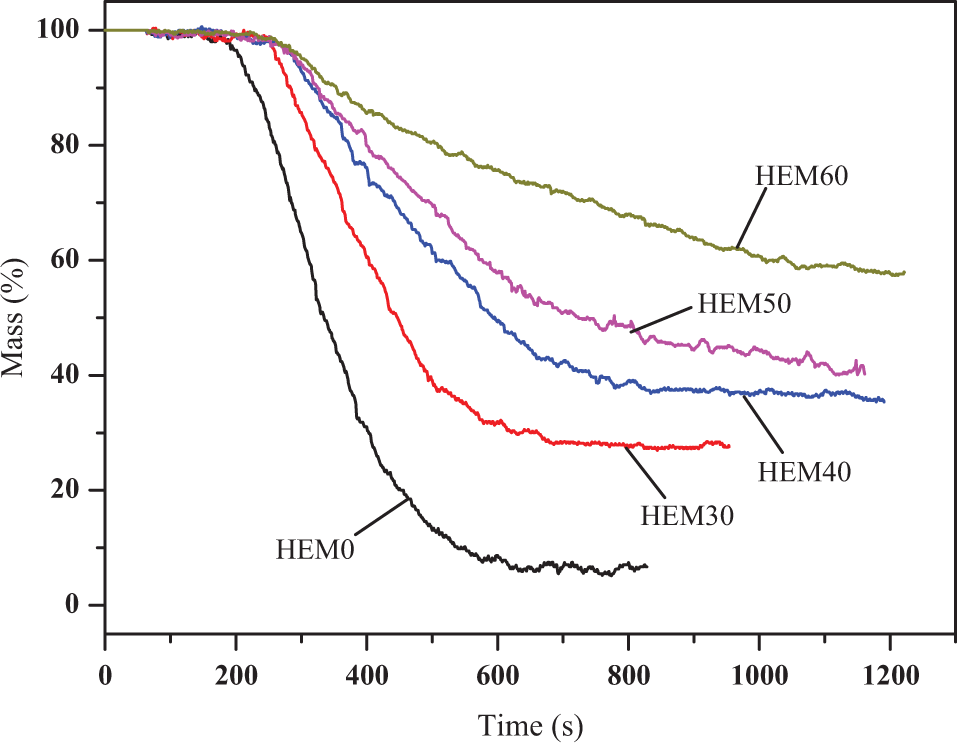
Mass curves versus time of HDPE/EVA and HDPE/EVA/MH composites with different MH content (formulation: HDPE/EVA = 80/20).
The evaluated fire performances of HDPE/EVA and HDPE/EVA/MH composites in Table 2 presented that the PHRR and THR values decreased with increasing the content of MH. The evolved water vapor which diluted the combustible vapor phase also reduced the amount of heat liberated. The time to ignition (TTI) values revealed that HDPE/EVA ignited earlier than HDPE/EVA/MH composites. The increment of TTI showed that the flame retardance effect was imposed by MH in the HDPE/EVA/MH composites. This indicated that the flame-retardant content imparted significant alterations on the combustion of HDPE/EVA. Fire growth index (FGI) was defined as the ratio of PHRR to TTI, which was generally used as an effective parameter to assess the degree of flame spread in fire hazard. 29 The lower the FGI, the better the fire resistance was obtained. It is seen from Table 2 that the addition of MH in HDPE/EVA significantly reduced the FGI owing to decreased PHRR and delayed TTI. In addition, FGI value decreased with increasing the MH content. That was because the gas phase fuel dilution mechanism provided by decomposition of MH during burning.
Figures 5 and 6 present the carbon monoxide (CO) and carbon dioxide (CO2) emission curves of HDPE/EVA/MH composites with various MH contents, respectively. It was clearly observed from Figures 5 and 6 that the variation trends of CO and CO2 emission of samples were very similar to HRR curves. Compared with HDPE/EVA, the emission of CO and CO2 of the HDPE/EVA/MH composites (HEM30∼HEM60) drastically decreased, which was attributed to the gas phase fuel dilution mechanism provided by the decomposition of MH during flammability. In addition, the peak emissions of CO and CO2 also reduced gradually with increasing the content of MH. And the appearances of peaks for CO and CO2 productions were delayed. This suggested that the flame retardance performance of HDPE/EVA was enhanced owing to the incorporation of MH.
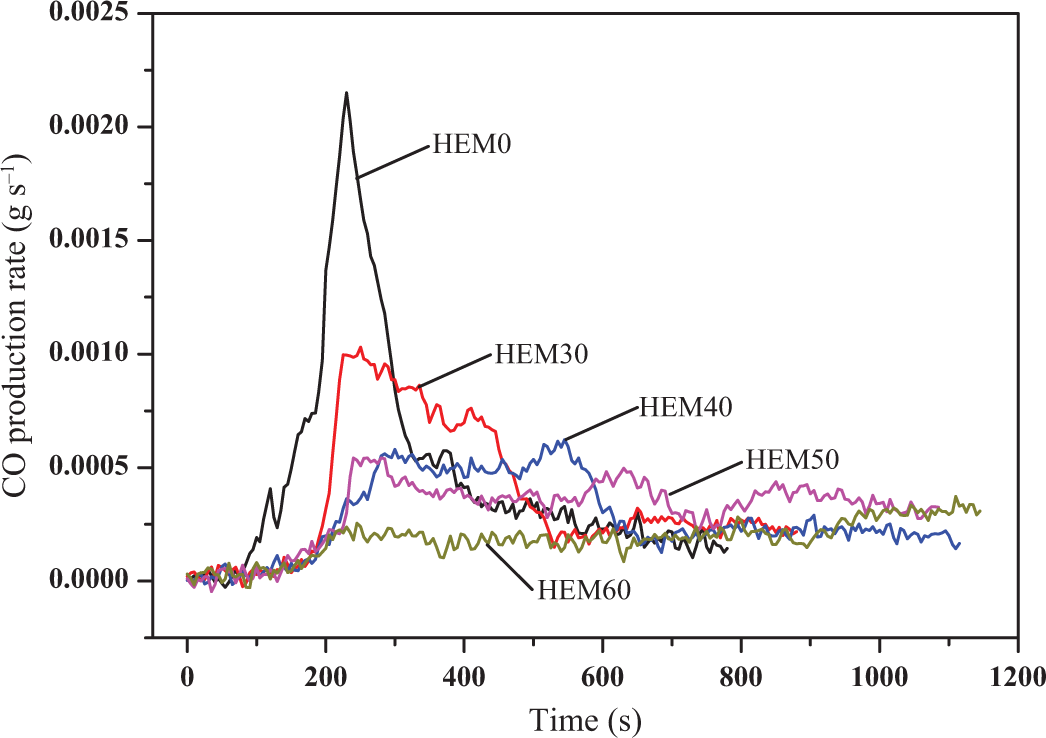
CO production of HDPE/EVA and HDPE/EVA/MH composites with different MH content (formulation: HDPE/EVA = 80/20).
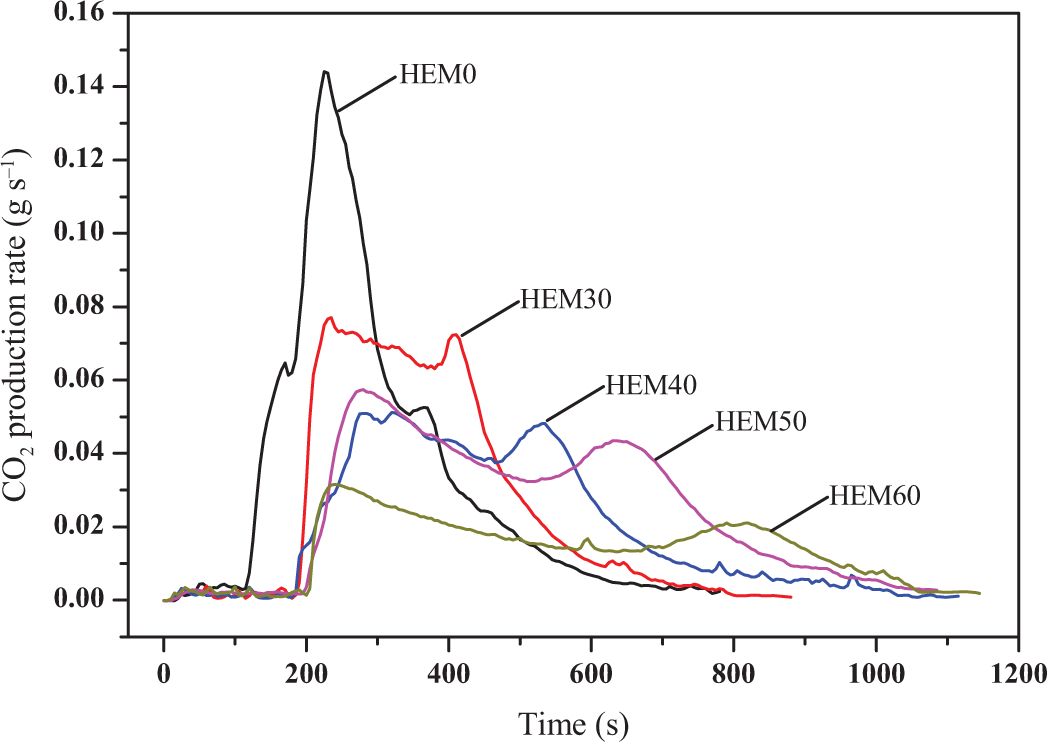
CO2 production of HDPE/EVA and HDPE/EVA/MH composites with different MH content (formulation: HDPE/EVA = 80/20).
Based on UL-94 test, LOI, and CCT, it could be noted that higher loading level of MH in HDPE/EVA was necessary to achieve satisfactory flame retardancy. In addition, the results also provided promising evidence on the prospects of MH to be used as halogen-free flame-retardant alternative for HDPE/EVA. In order to elucidate how the formation of the residues affects the combustion, the residues left after UL-94 burning tests were examined for changes in char appearance by taking photographs with a digital camera, and the results are shown in Figures 7 and 8. The melted-form residues of HDPE/EVA were improved by the incorporation of MH. There was not any charred residue left for HEM0 after combustion. The composites with loading 30–60 wt% MH were observed to resist ignition and subsequent combustion.
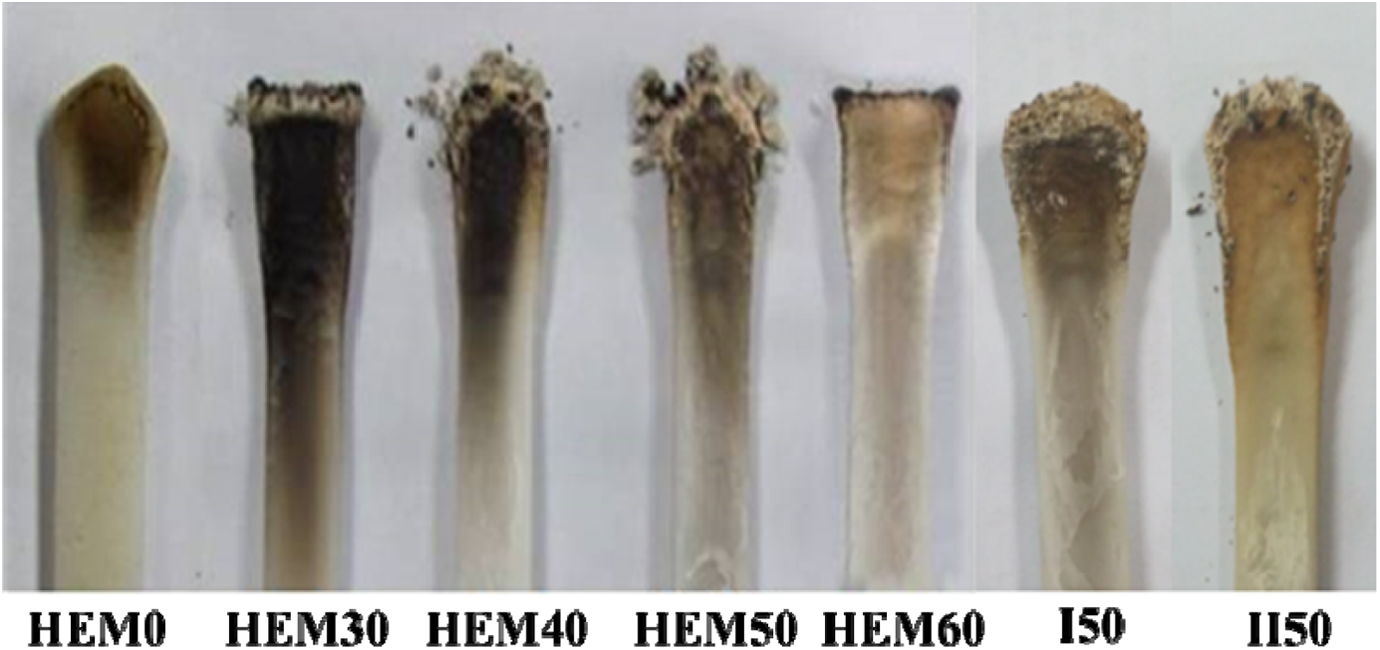
Digital photographs of HDPE/EVA composites with different MH content (formulation: HDPE/EVA = 80/20) after UL-94 test.

The charred residues formed by combustion of HDPE/EVA/MH composites with different MH content (formulation: HDPE/EVA = 80/20): (A) HEM30; (B) HEM40; (C) HEM50; (D) HEM60; (E) I50; (F) II50.
Figure 8 shows the surface images of residues of HDPE/EVA/MH composites after burning. HDPE/EVA/MH composites with 30 wt% MH (sample HEM30) had a little residue composed of discrete MgO on the surface. The residues increased with increasing content of MH. The discontinuous fire residues demonstrated by 30 wt% and 40 wt% MH in HDPE/EVA were seen. However, the consolidated, continuous, and thicker residue barriers were formed for 50 wt% and 60 wt% MH containing HDPE/EVA/MH composites (Figure 8(c) and (d)), resulting in good flame retardancy. Moreover, little difference was observed from Figure 8(c, e, and f). It was believed that the formation of MgO layer upon decomposition of MH hindered the evolution of decomposition products into the gas phase during combustion. In addition, the interaction between the oxygen in the ester group (C=O) existed in EVA, PE-g-MAH, and the decomposition product of HDPE and magnesium ion was beneficial for improving flame retardancy of the composites. 30
Thermal stability
TGA and differential thermogravimetry (DTG) curves of pure HDPE, EVA, MH, I50, and II50 are shown in Figure 9. The TGA curves of pure HDPE and MH only displayed one-step thermal degradation processes, however, EVA showed the two-step degradation processes. At the same time, it was noted that the decomposition temperature of HDPE was higher than those of EVA and MH. For HDPE and EVA, there had hardly residue left after degradation, whereas high residues of MgO were left for MH.
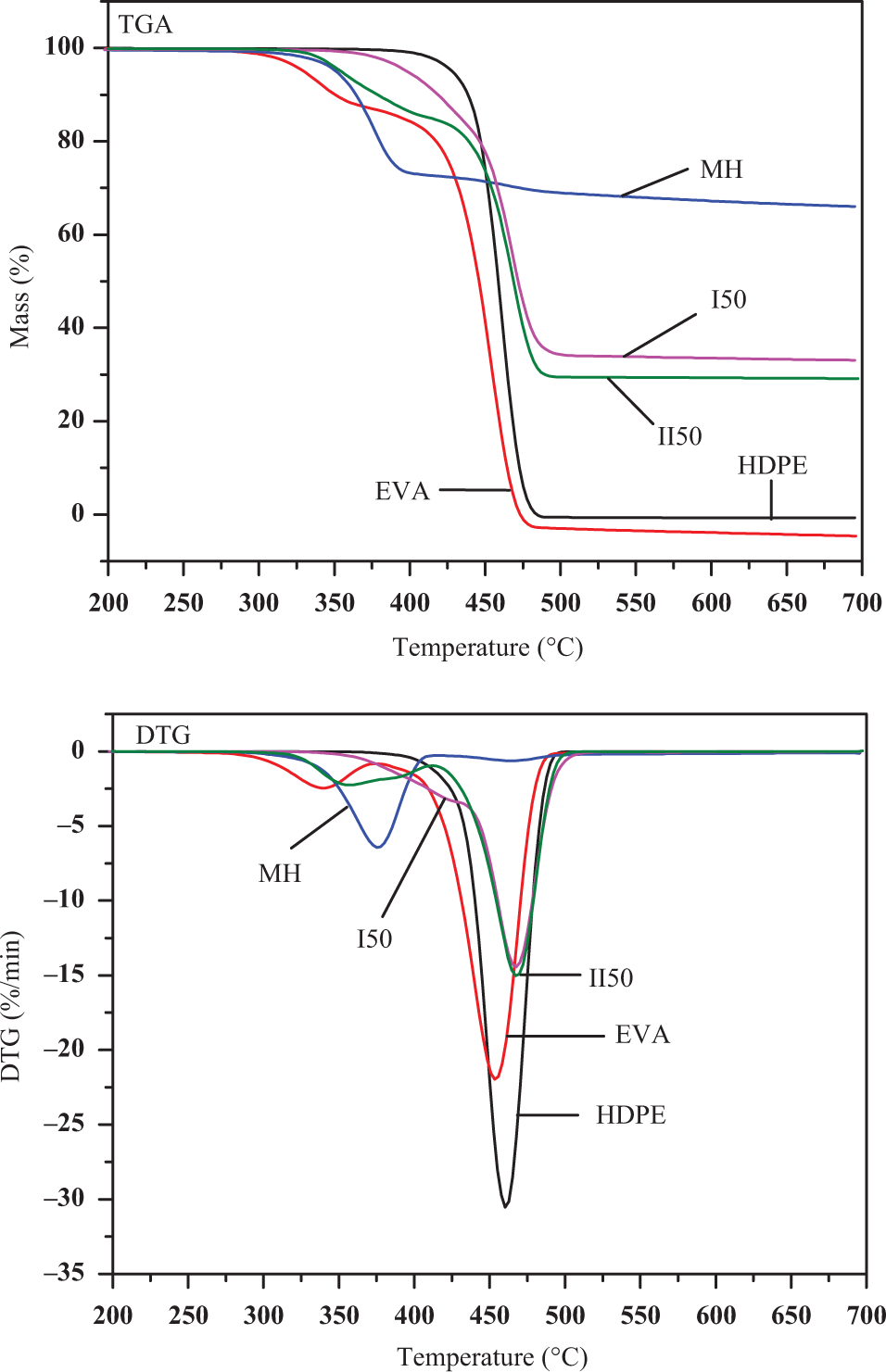
TGA and derivative TG (DTG) curves of HDPE, EVA, Mg(OH)2, I50 and II50 at a heating rate of 10oC min−1 in N2.
Figure 10 shows the TGA and DTG curves of HDPE/EVA/MH composites with different contents of MH, and the correlative data are listed in Table 3. The initial decomposition temperature was characterized by the temperatures at 5 wt% weight loss occurred, referred to as T 5 wt% (Table 3). The weight loss curves of HDPE/EVA/MH composites took place in a two-step process. The addition of MH into HDPE/EVA decreased the thermal stability by lowering the T 5 wt% and T 20 wt% values, which were attributed to the loss of acetic acid from EVA and the dehydration of MH. 25,31
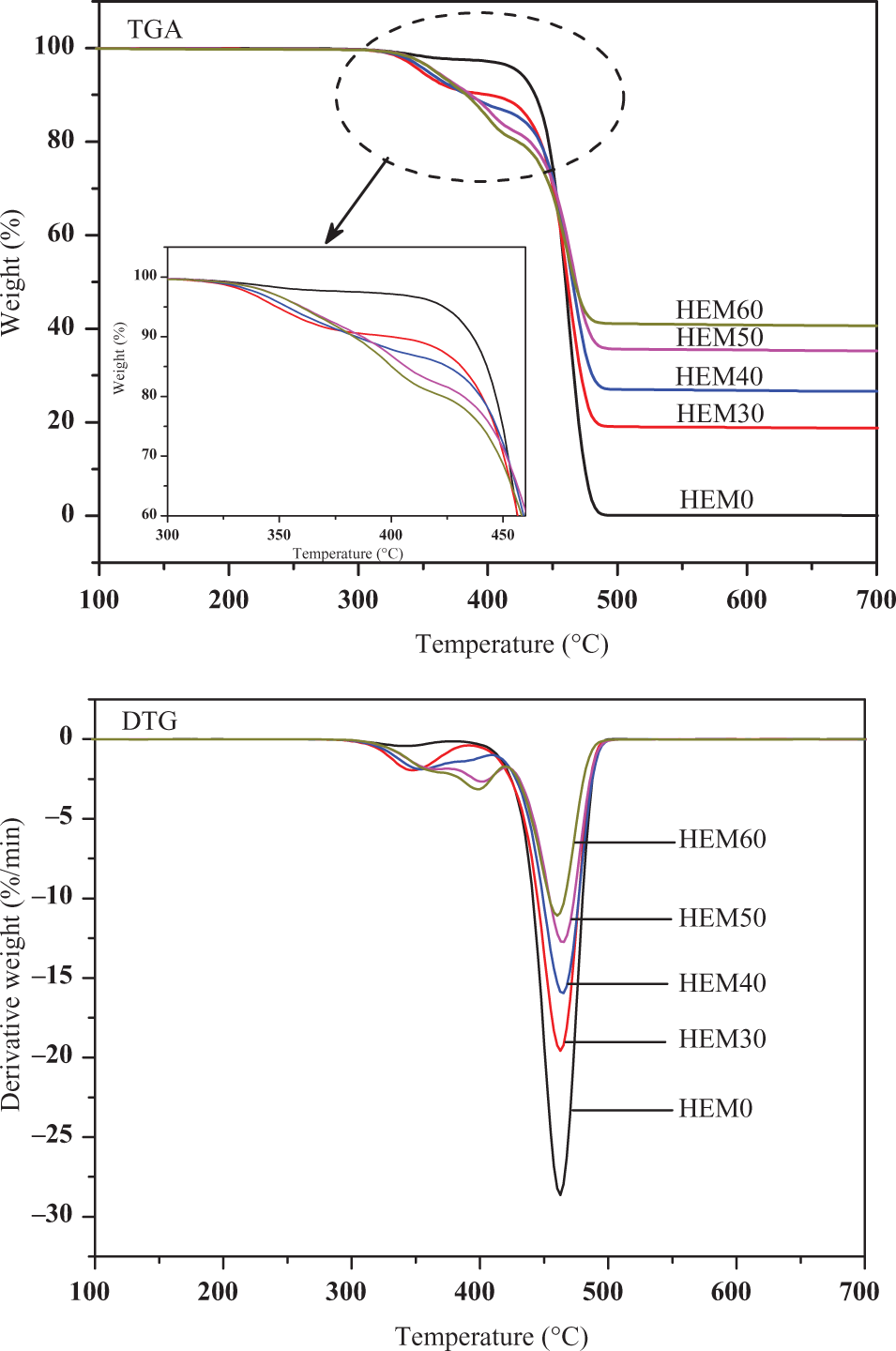
TGA and DTG of HDPE/EVA/MH composites with different MH content (formulation: HDPE/EVA = 80/20) at a heating rate of 10°C min−1 in N2.
Detailed data obtained from TGA and DTG of HDPE/EVA/MH composites.
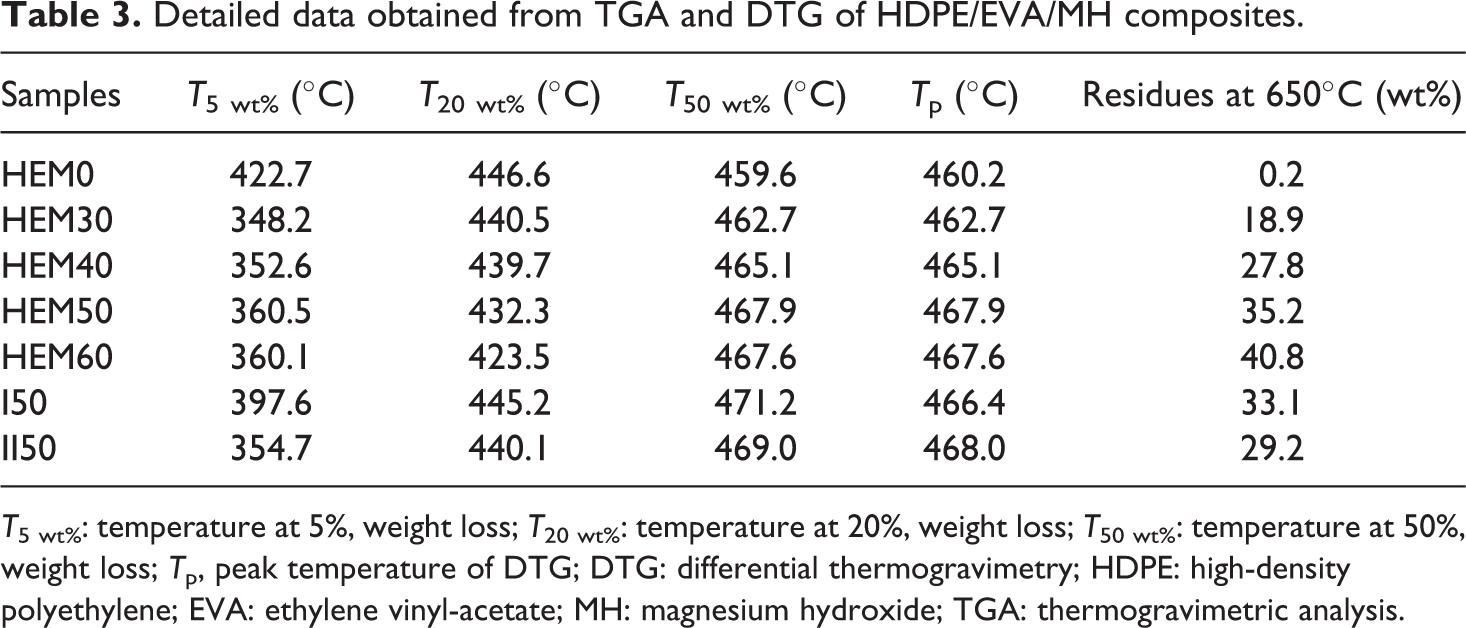
T 5 wt%: temperature at 5%, weight loss; T 20 wt%: temperature at 20%, weight loss; T 50 wt%: temperature at 50%, weight loss; T p, peak temperature of DTG; DTG: differential thermogravimetry; HDPE: high-density polyethylene; EVA: ethylene vinyl-acetate; MH: magnesium hydroxide; TGA: thermogravimetric analysis.
The initial thermal stability could be described in two sections. Firstly, the T 5 wt% values of HDPE/EVA/MH composites decreased between 310°C and 380°C. Compared with HEM50 with EVA, the T 5 wt% value of I50 sample without EVA increased from 360.5°C to 397.6°C, which indicated that this section was dominated by the decomposition of EVA. Secondly, between 420°C and 450°C, the T 20 wt% values of HDPE/EVA composites decreased with increasing MH loading compared with that of HDPE/EVA blend. As demonstrated from DTG curves in Table 3, the decomposition of MH was attributed to two steps. The first step started at 280°C and reached its maximal weight loss at 375°C and stopped at about 416°C. The earlier decomposition of MH also contributed more to the lowering of T 20 wt% values in HDPE/EVA/MH composites. The early primary decomposition of HDPE/EVA/MH composites was a consequence of endothermic decomposition of MH to MgO and water at about 380°C. 32 This temperature was favorable for the flame retardance properties due to absorbing heat and diluting gas. In addition, it was believed that the formation of MgO layer upon decomposition of MH hindered the evolution of decomposition products into the gas phase during thermal degradation. So the more MH, the denser the MgO layers were obtained. This result was in agreement with the UL-94 and LOI.
The second step occurred from 420°C to 470°C, and HDPE decomposed in the range from 370°C to 500°C. At the same time, the second degradation step of EVA occurred when the temperature ranged from 370°C to 520°C. This step might be a sign to the degradation of the HDPE main chains and the back bone of the EVA which formed in the first step and the formation of transvinylenes accompanied by main chain scission in the second step. 33,34 The residues of HDPE/EVA/MH composites with various MH contents at 650°C are also presented in Table 3. The remaining residue was attributed to the formation of MgO as a dehydration product of MH. It was observed that the amount of the residue increased with increasing the loading level of MH. The increase of residue at high temperature indicated that the flame retardancy of the composites was improved with increasing the content of MH, which was in agreement with CCT.
Crystallization behaviors
The DSC crystallization and melting curves of HDPE/EVA and HDPE/EVA/MH composites with different MH contents are shown in Figures 11 and 12, respectively. The melting and crystallization temperatures (T m and T c), the degree of crystallinity (X c), and enthalpies (ΔH m and ΔH c) are listed in Table 4. It is found from Figure 11 that only one exothermic peak was observed for each curve between 40°C and 160°C. T c for HDPE/EVA was observed at about 117.2°C. The T c slightly shifted toward a lower temperature due to the incorporation of MH.
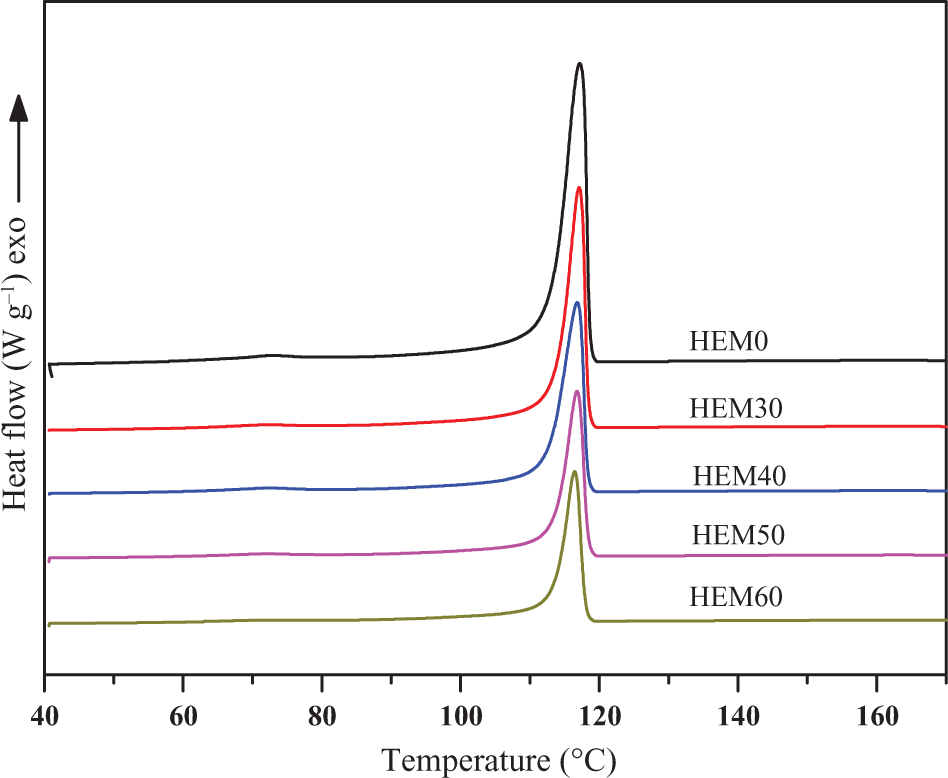
DSC crystallization exotherms of HDPE/EVA blends and HDPE/EVA/MH composites with different MH content (formulation: HDPE/EVA = 80/20).
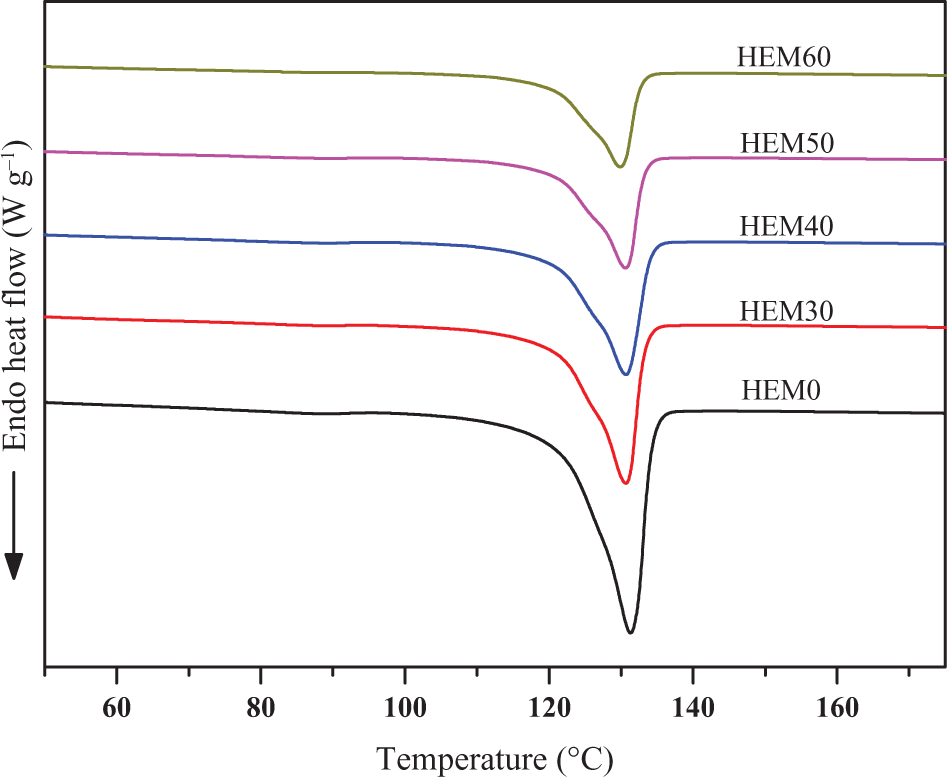
DSC melting endotherms of HDPE/EVA blends and HDPE/EVA/MH composites with different MH content (formulation: HDPE/EVA = 80/20).
Melting and crystallization characteristics of HDPE/EVA/MH composites.
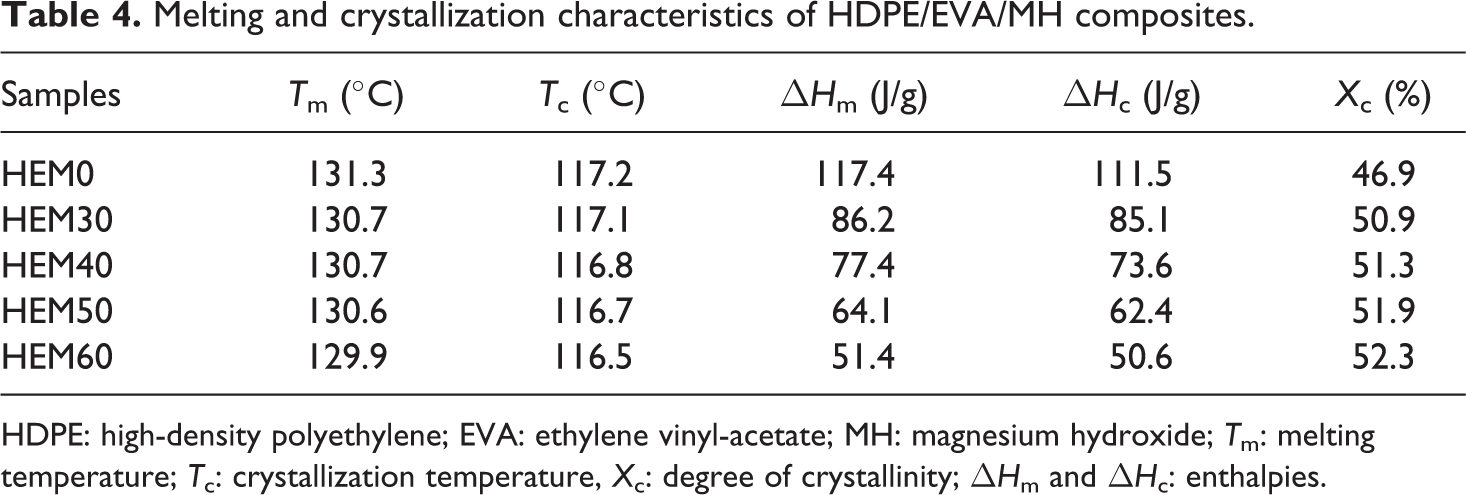
HDPE: high-density polyethylene; EVA: ethylene vinyl-acetate; MH: magnesium hydroxide; T m: melting temperature; T c: crystallization temperature, X c: degree of crystallinity; ΔH m and ΔH c: enthalpies.
Analysis of the data in Table 4 indicated the decrease of melting point of HDPE/EVA/MH composites could be discussed in terms of the systems viscosities. The inorganic particles increased the viscosity of the composites, which in turn negatively affected the crystallization growth stage. Thus, the more defective crystals would have lower T m. But the X c of the composites increased with increasing the content of MH, which might be attributed to heterogeneous nucleation effects that the inorganic particles MH acted as effective nucleating agents in the HDPE/EVA blends.
Figure 12 shows the melting endotherms for HDPE/EVA and HDPE/EVA/MH composites. All the samples also showed only one melting endotherm. This indicated that high-temperature melting peak corresponds to the melting of crystallized crystallites formed during a heating scan. The addition of MH has no essential effect on the melting temperature of HDPE/EVA, however, a slight shift to low temperature of melting peak temperature (T m) was observed. This suggested that the higher content of MH in HDPE/EVA formed defective crystallization, resulting in poor crystalline perfection.
Mechanical properties
The stress–strain curves of HDPE/EVA/MH composites with different amounts of MH are shown in Figure 13. It was clear that the tensile behaviors of HDPE/EVA/MH composites were strongly dependent on the incorporation of MH. The tensile stress of HDPE/EVA/MH composites increased with increasing the content of MH, and a significant improvement was observed when the MH content was >40 wt%. However, a great decrease in tensile strain of the composites was noted with increasing MH content. The decrease of tensile strain was attributed to the high loading of MH, which caused the poorest compatibility between the MH particles and the polymer matrix. Moreover, the high crystallinity of HDPE/EVA/MH composites might be the reason for the improvement of tensile stress and the drop of tensile strain.
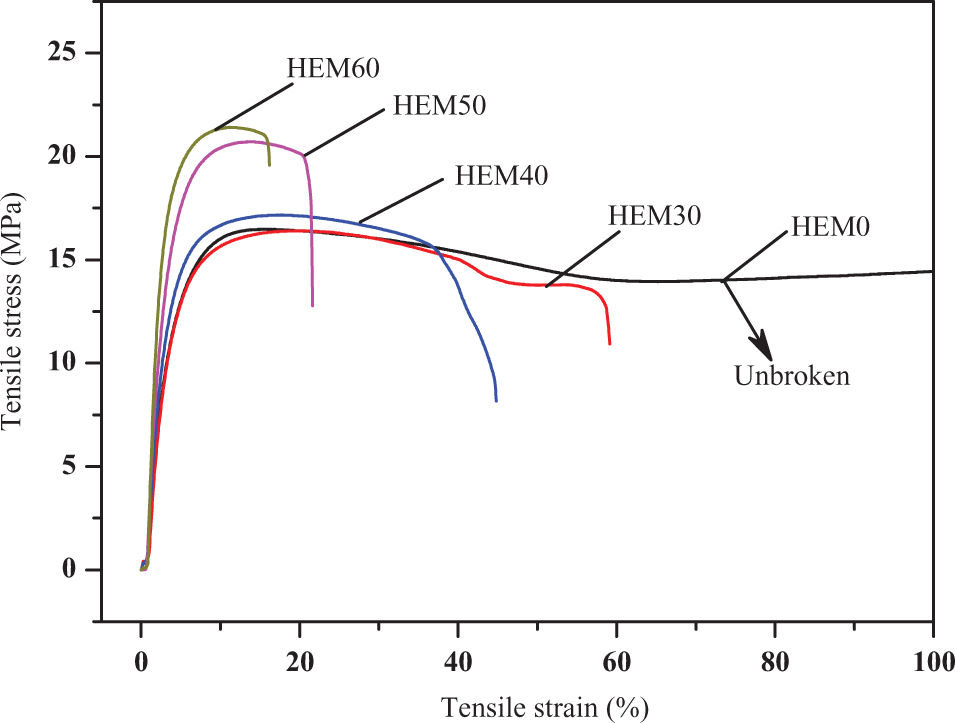
Stress–strain curves of HDPE/EVA composites with different MH content (formulation: HDPE/EVA = 80/20).
The scanning electron microscopy (SEM) morphologies of the fractured surfaces of HDPE/EVA/MH composites with various MH contents are shown Figure 14. Some differences between these SEM micrographs were clearly observed. When the loading level was 30 wt% and 40 wt%, more MH particles were embedded in the matrix and less aggregated MH particles were exposed on the surface, showing the more homogeneous dispersion and compatibility of MH in HDPE/EVA, as shown in Figure 14(a) and (b). In addition, a number of fibrous and flake-like substances were also found. The reasons might be that the formation of such a fiber or flake-like particle was caused by the softening EVA under the condition of shear of twin-screw extruder during processing or remnants of the cold-drawn HDPE filaments between the rigid MH particles. With increasing the content of MH, more aggregated MH particles could be observed on the fractured surfaces, resulting in cracks between the MH particles (Figures 14(c) and (d)). Such an interface would surely deteriorate the elongation at break, resulting in the decrease of tensile strain.
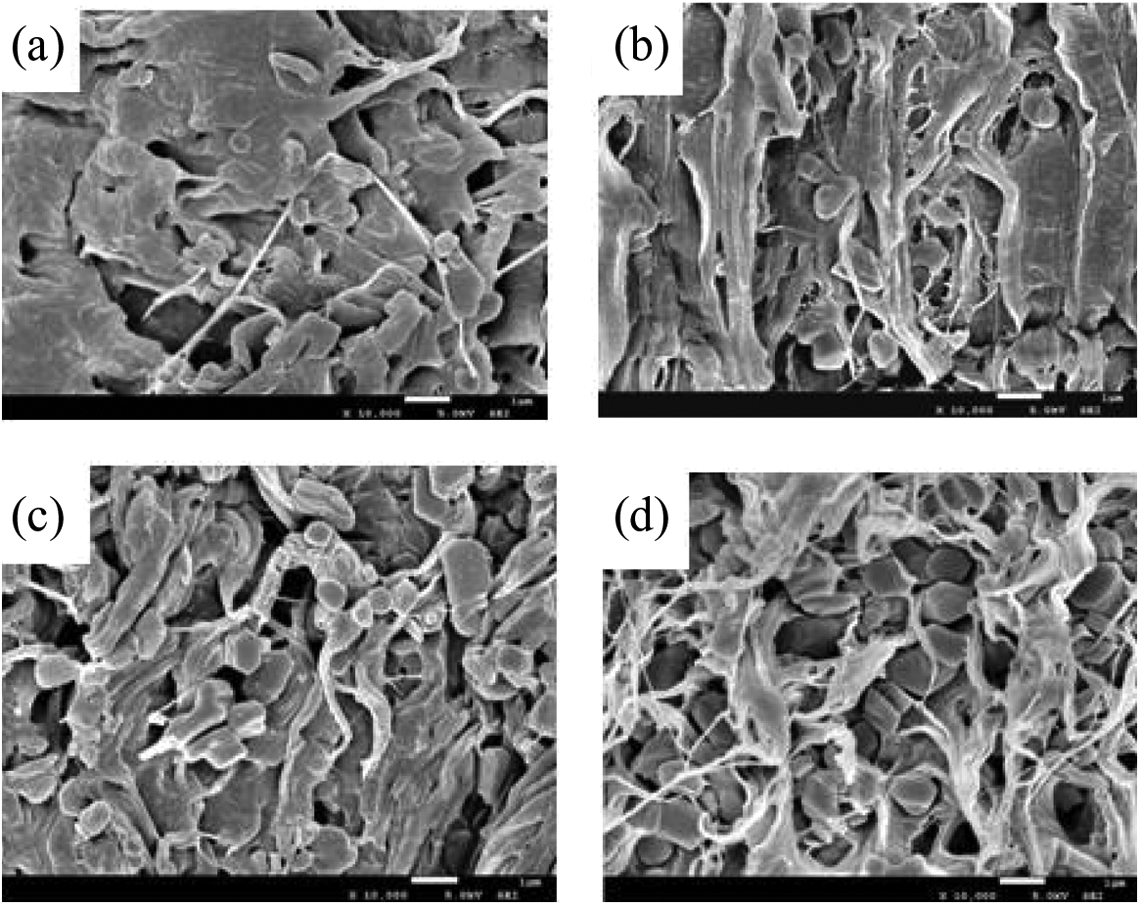
SEM photos of HDPE/EVA/MH composites with different MH content (formulation: HDPE/EVA = 80/20): (a) 30 wt% MH; (b) 40 wt% MH; (c) 50 wt% MH; (d) 60 wt% MH.
Conclusions
HDPE/EVA/MH flame-retardant composites with different MH contents (30–60 wt%) were prepared by melting blend in a twin-screwed extruder. The fire retardancy, thermal stability, morphological, crystallization, and tensile behaviors of MH flame retarded HDPE/EVA composites were characterized. The HDPE/EVA/MH composites exhibited excellent flame retardancy with increasing MH content: increased LOI, higher UL-94 rating, reduced heat release, decreased gas productions, prolonged ignition time, and higher charred residues. It was concluded that the incorporation of MH into HDPE/EVA blends enhanced the flame retardancy of composites due to the formation of intact, consolidated, and thick residue structures on the surface of MH-filled composites. The interaction between magnesium ion and the group (C=O) was in favor for improvement of flame retardancy. The crystallinity of HDPE/EVA/MH composites increased with increasing the content of MH. The tensile test indicated that the strength of HDPE/EVA/MH composites was enhanced, while the tensile strain was deteriorated.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are grateful to the National Natural Science Foundation of China (51003088 and 21266005), the Fundamental Research Funds for the Central Universities (SWJTU12CX009), 2013 national college student innovation experiment program (201310613004), and Sishi Star Foundations of Southwest Jiao tong University (2011) for financial support of this work.
