Abstract
In this article, the flammable behaviors and synergistic effects of modified expanded graphite (MEG) with zinc borate (ZB) on flame-retardant high-density polyethylene/ethylene vinyl acetate (HDPE/EVA) composites containing magnesium hydroxide (MH) and aluminum hydroxide (ATH) are investigated by the Underwriters Laboratories-94 (UL-94) test, limiting oxygen index (LOI), cone calorimeter test (CCT), thermal gravimetric analysis (TGA), Fourier transform infrared (FTIR), differential scanning calorimetry, and tensile tests. The LOI, UL-94, and CCT results show that the synergistic effect of MEG and ZB can improve the flame retardancy of the composites. With the addition of ZB and MEG, the LOI value increases, and the UL-94 reaches the V-0 rating. The heat release rate and total heat release decrease, respectively. The data obtained from the TGA indicate that the synergistic effects of ZB with MEG increase the decomposition temperature when 2 phr ZB and 8 phr MEG are added into the composites. The data from FTIR show that HMEG8 and HMEG10 composites produce phosphate at high temperatures, which promotes the formation of stable and compact charred layer. All the results show that ZB and MEG have positive synergistic effects on HDPE/EVA composites containing MH and ATH. However, ZB and MEG play a negative role in the tensile properties of the HDPE/EVA composites.
Introduction
High-density polyethylene (HDPE) is widely used in many fields today. 1 In the wire and cable industries, HDPE is used as an excellent insulating material with good mechanical and process properties.2–4 However, HDPE is flammable, producing a lot of heat and amounts of gas and smoke when burning,5–7 which restricts its application greatly. It is urgent to find a kind of flame retardant with low smoke and nontoxic gas. Metal hydroxides, especially magnesium hydroxide (MH) and aluminum hydroxide (ATH), do not produce poisonous gases when burning as environmentally friendly flame retardants.8,9 The decomposition temperatures of MH and ATH are higher than 350°C and 250°C,10,11 respectively. MH and ATH endothermically decompose to reduce the temperature of the composites and generate steam and solid oxides due to the dilute concentration of oxygen around the polymers and obstruction of the exchange of oxygen and combustible materials.12–14
However, MH and ATH have some shortcomings, such as the poor adhesion with polymer, poor flame-retardant efficiency, and high addition.15,16 The high loadings of fillers result in the decrease of mechanical properties of polymers. 17 Previous studies18,19 reported that MH and ATH cooperating with some halogen-free flame retardants, such as zinc borate (ZB), red phosphorus, expanded graphite (EG), and amine phosphate, can increase flame retardancy of MH and ATH and reduce their loadings, effectively.20,21
Ethylene vinyl acetate (EVA) copolymer, as elastomer material, has many advantages in high impact resistance, water resistance, and processability.9,22 EVA can be used as the compatibilizer, which can enhance the interface adhesion between inorganic particles and polymer matrix. In this work, a novel method of organic modification of EG is proposed by using grafting process containing phosphorous and synergistic effect with ZB applying to the HDPE/EVA composites filled with MH/ATH. The flammability, thermal stability, crystallization behaviors, and tensile properties of HDPE/EVA composites are investigated systematically by limiting oxygen index (LOI), Underwriters Laboratories-94 (UL-94) test, cone calorimetry test (CCT), thermogravimetry analysis (TGA), differential scanning calorimetry (DSC), and the tensile test.
Experimental
Material
HDPE (5000 S) is produced by Lanzhou Petrochemical Company, China Petroleum. EVA (7240 M) with vinyl acetate content of 15 wt% is produced by Taiwan Plastic Company, China. MH (average particle size: 1.5 μm) and ATH (average particle size: 2.6 μm) are bought from Yantai Ai Weier Chemical Company, China. EG with particle size of 270 µm and carbon content of 90–99.9% is provided by Qingdao Kang Boer Graphite Products Company, China. ZB is produced by Shanghai Hongye Biotechnology Co. Ltd., China, phosphorus-containing flame retardant, 9,10-dihydro-9-oxa-10-phosphaphenanthrene-10-oxidation (DOPO), is provided by Guangdong Huizhou Shengshida Technology Company, China, and the γ-glycidyloxypropyltrimethoxysilane (silane coupling agent) is provided by Chengdu Chenbang Chemical Co., Ltd., China.
Preparation of flame-retardant composites
The modification of EG is modified by DOPO. 23 The HDPE/EVA composites filled with the desired amounts of MH, ATH, modified expanded graphite (MEG), or ZB are blended in a twin-screw extruder (TSE-20A1600-4-40, made in Nanjing, China) at 130–170°C with a rotation rate of 150 r min−1 and then the extrude granulates are pressed at 170°C, 10 MPa, and then chop into slivers with two different sizes of 120.0 × 6.0 × 3.0 mm3 and 90.0 × 10.0 × 4.0 mm3 for the evaluation of LOI and UL-94 test, respectively. The specimen formulas are listed in Table 1.
Formulations and flammability of the HDPE/EVA and its flame-retardant composites.a
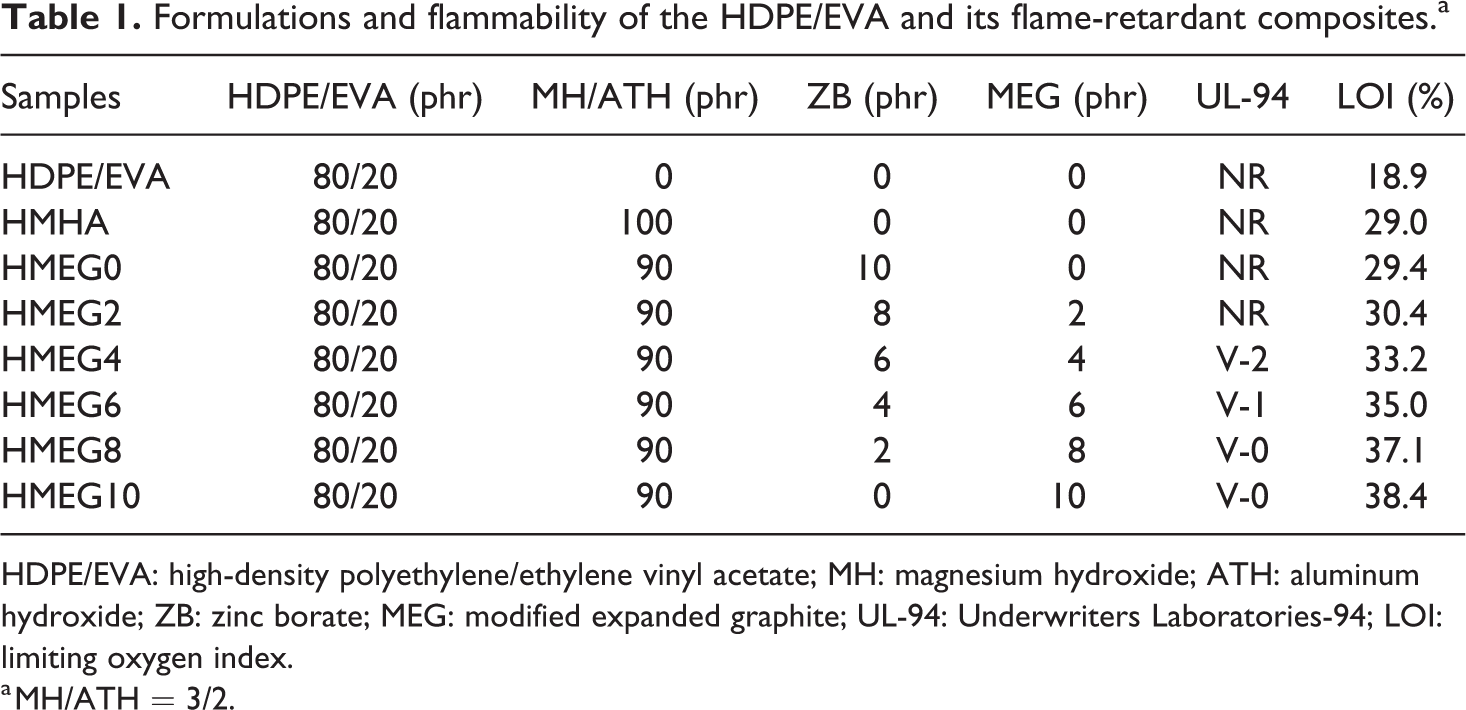
HDPE/EVA: high-density polyethylene/ethylene vinyl acetate; MH: magnesium hydroxide; ATH: aluminum hydroxide; ZB: zinc borate; MEG: modified expanded graphite; UL-94: Underwriters Laboratories-94; LOI: limiting oxygen index.
a MH/ATH = 3/2.
Measurements and characterization
The UL-94 vertical burning is a widely method for testing the flame retardancy of composites. In this article, UL-94 is carried out using CZF-1 vertical burning instrument on sheets with a size of 90.0 × 10.0 × 4.0 mm3.
LOI value is measured with JF-4TG oxygen index instrument (Nanjing, China). The LOI value is an indicator to evaluate the flammability of composites. The detection data are the average value of five samples.
CCT can comprehensively evaluate the thermal decomposition of materials during combustion. In this work, the specific size of the sample was 100 × 100 × 3 mm3 and irradiated the sample with a radiation intensity of 35 kW m−2.
TGA is usually used to analyze the thermal stability of composites on a NETZSCH TG 209F1 thermal gravimetric analyzer under nitrogen atmosphere with a flow rate of 50 mL min−1. The test process in this article is to put about 8–10 mg sample into a small crucible and then heat from room temperature to 650°C at a heating rate of 10°C min−1.
Fourier transform infrared (FTIR) spectroscopy analysis of HMEG0, HMEG8, and HMEG10 are recorded using a Nicolet MAGNA-IR 500 spectrophotometer. Powders of materials are mixed with KBr powders and then the mixture is compressed into thin slices under the force of 10 MPa for FTIR spectra analysis, and the wave number of the scanning is 400–4000 cm−1.
DSC can effectively test the crystallization properties of composites. Sample (8–10 mg) is tested under nitrogen atmosphere and then heated from room temperature to 170°C at a rate of 10°C min−1 keeping for 5 min, down to room temperature at the same rate, and finally rises to 170°C at the same rate.
The tensile properties of the HDPE/EVA composites are tested at room temperature and the tensile rate is 50 mm min−1. Each sample is tested five times, and the average value of the test samples is taken as the final experimental data.
Results and discussion
Flammability: UL-94 test and LOI
Figure 1 shows the flame shapes and the morphologies of HDPE/EVA and its flame-retardant composites during the UL-94 test. HDPE/EVA burns very violently and droplet occurs during burning. With the addition of ATH/MH, the combustion of composites becomes slower and there is no droplet phenomenon any more. The reason may be that ATH and MH decompose into water vapor at high temperature dropping down the temperature around the composites and form oxides isolating the air and heat source from exchanging. The flame retardancy of flame-retardant composites is improved when ZB and MEG are added into HDPE/EVA. Less charred layer formed by HMEG0 and HMEG2 cannot play a good role to protect the substrate. They cannot achieve UL-94 rating, either. It can be observed from Figure 1 that the flammability of HMEG4 and HMEG6 decreases obviously. And they have a small amount of charred layer formed on the surface and reach V-2 and V-1 rating, respectively. HMEG8 and HMEG10 are hardly ignited during UL-94 test and both of them reach V-0 level.

The flame shape and morphologies of HDPE/EVA and its composites during UL-94 test: (a, a′) HDPE/EVA; (b, b′) HMHA; (c, c′) HMEG0; (d, d′) HMEG2; (e, e′) HMEG4; (f, f′) HMEG6; (g, g′) HMEG8; and (h, h′) HMEG10.
The LOI and UL-94 results of HDPE/EVA and its flame-retardant composites are listed in Table 1. The LOI value of HDPE/EVA is only 18.9%. It can be also seen from Figure 1 that no charred layer is produced on the surface of HDPE/EVA. The LOI value of H/MH/ATH increases to 29% after adding MH/ATH. When the ZB and MEG cooperating with MH/ATH, the LOI values of HMEG composites increase further. Compared with HMEG0, the LOI value of HMEG2 rises to 30.4%. It can be observed from Figure 1 that the content of charred layer and worm like structure 24 formed on the surface both increase with increasing the content of MEG. Additionally, the LOI values of HMEG8 and HMEG10 increase to 37.1% and 38.4%, respectively.
Dynamic combustion behaviors
Figure 2 shows the heat release rate (HRR) curves of HDPE/EVA and its flame-retardant composites from CCT. The detailed data obtained by CCT are listed in Table 2. HDPE/EVA burns fiercely and mainly occurs between 100 s and 150 s, appearing a sharp peak of 458.3 kW m−2. In addition, the ignition time of HDPE/EVA is 18 s. However, the combustion time of the flame-retardant composites prolongs and the peak of HRR (PHRR) decreases obviously. It is surprised that in the first 500 s, the PHRRs of flame-retardant composites decrease with increasing the content of MEG, while the PHRR of HMEG0 is the lowest. After 500 s, the PHRR of HMEG0 gradually increases. The composite sample only containing MH/ATH and ZB releases the water vapor and forms the charred layer during the early combustion. The released water vapor reduces the temperature and heats around the composites. And the formed charred layer also blocks the heat into the matrix, slowing down the combustion and reducing the release rate of the heat. But after 500 s, the released water evaporates quickly due to the temperature increasing. And even the formed charred layer is damaged, which makes the internal matrix continue to burn and release heat. It is also observed from Figure 2 that the HRR values of the flame-retardant composites with MEG and ZB slow down after 500 s. The synergistic effect of MEG and ZB promotes the formation of dense charred layer, which blocks the heat source into the matrix and prevents the matrix from continuing to burn. As shown in Figure 3, the total heat release curves of the composites are similar to that of HRR.
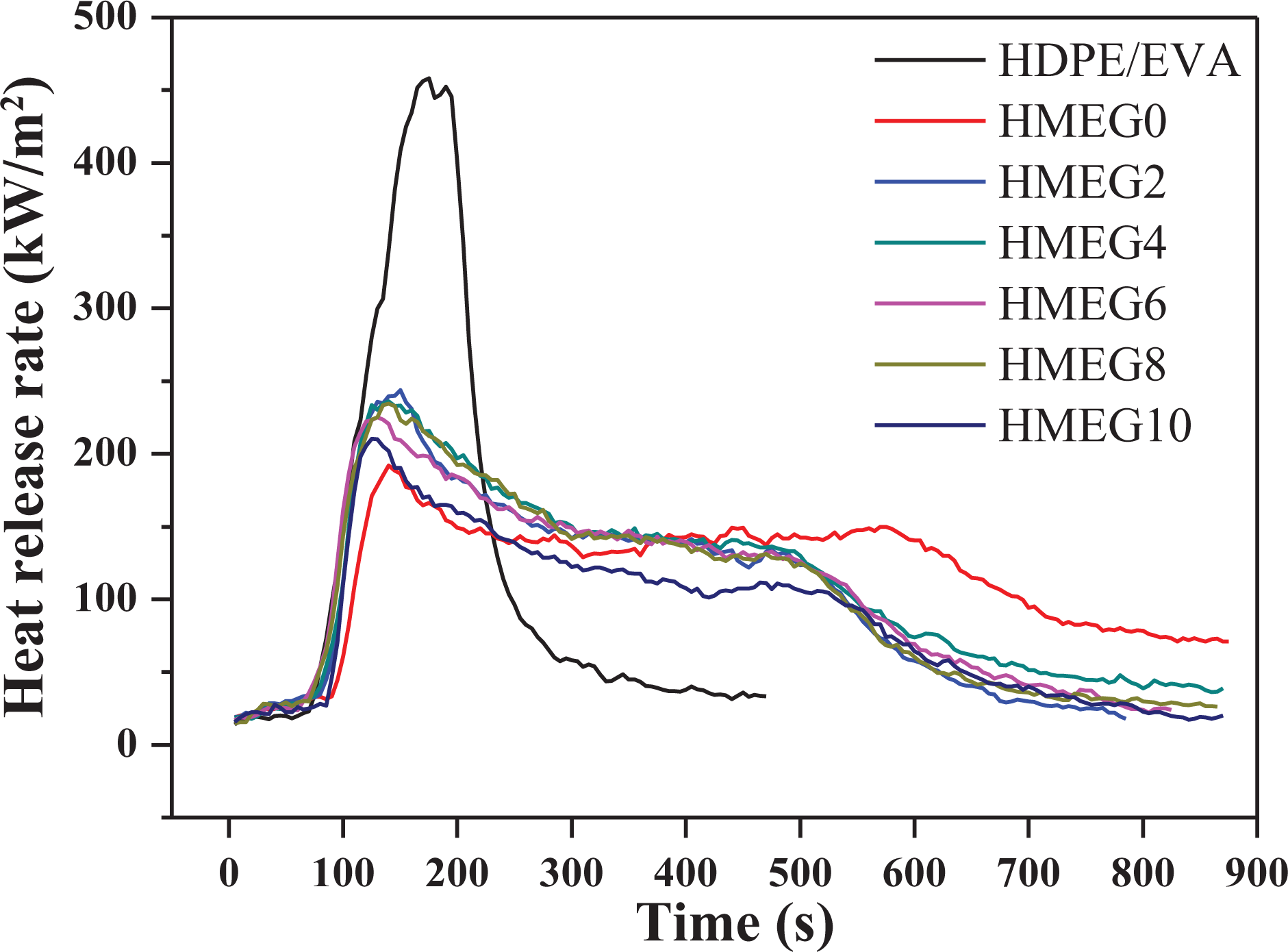
HRR curves of HDPE/EVA and its flame composites.
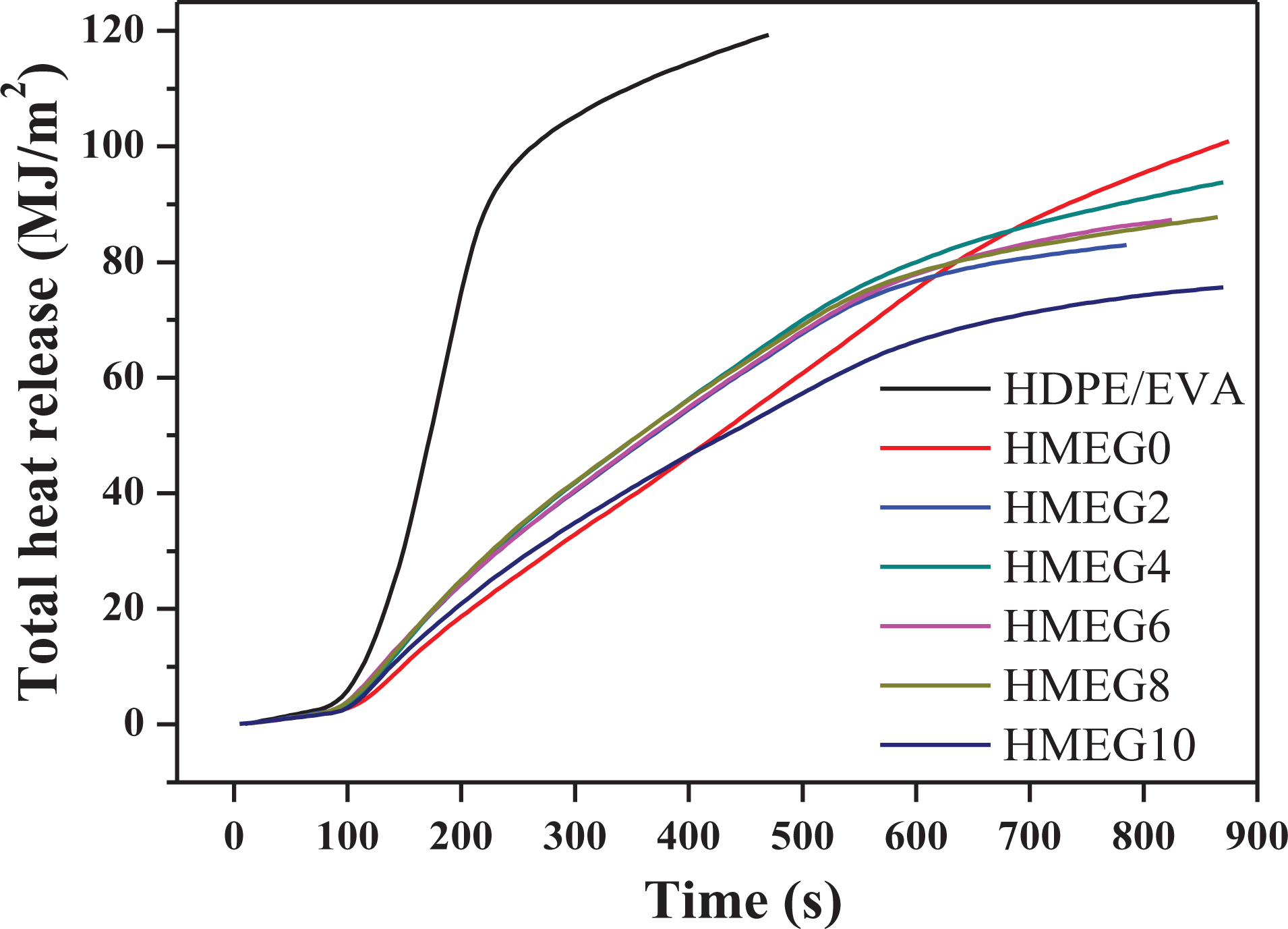
THR curves of HDPE/EVA and its flame composites.
The CCT data of HDPE/EVA and its flame-retardant composites.
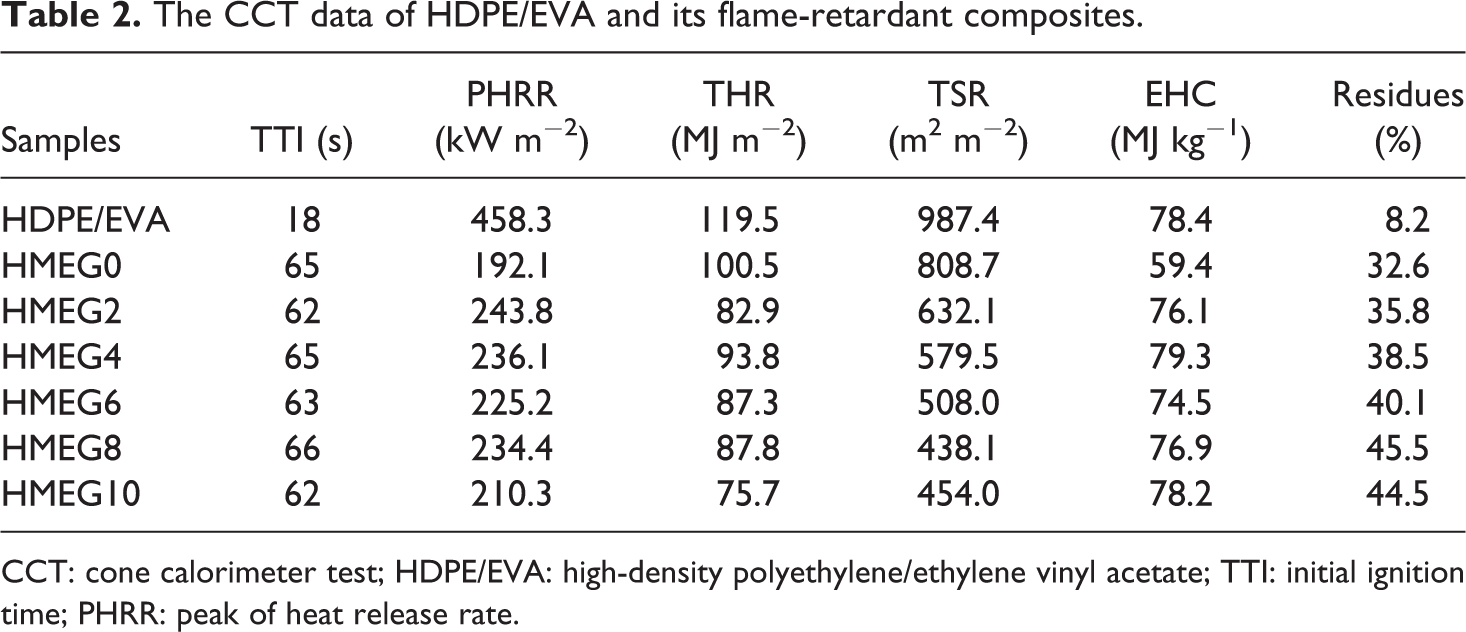
CCT: cone calorimeter test; HDPE/EVA: high-density polyethylene/ethylene vinyl acetate; TTI: initial ignition time; PHRR: peak of heat release rate.
Figures 4 and 5 show the emissions of carbon monoxide (CO) and carbon dioxide (CO2) for HDPE/EVA and its flame-retardant composites during the CCT, respectively. The HDPE/EVA releases a large number of CO and CO2 during the combustion process. However, the gas release of the flame-retardant composites greatly reduces. In addition, the release amount of CO2 of HMEG0 is the lowest before 600 s. But after 600 s, it begins to increase. It may be the reason that the quality of the charred layer formed by HMEG0 is not dense enough. Because of this, the polymer matrix exposes in the air and then burns. When the oxygen is abundant, the release of CO2 increases. However, for other flame-retardant composites, the release of CO2 is always in a state of reduction, which is the reason of the formation of the dense charred layers.
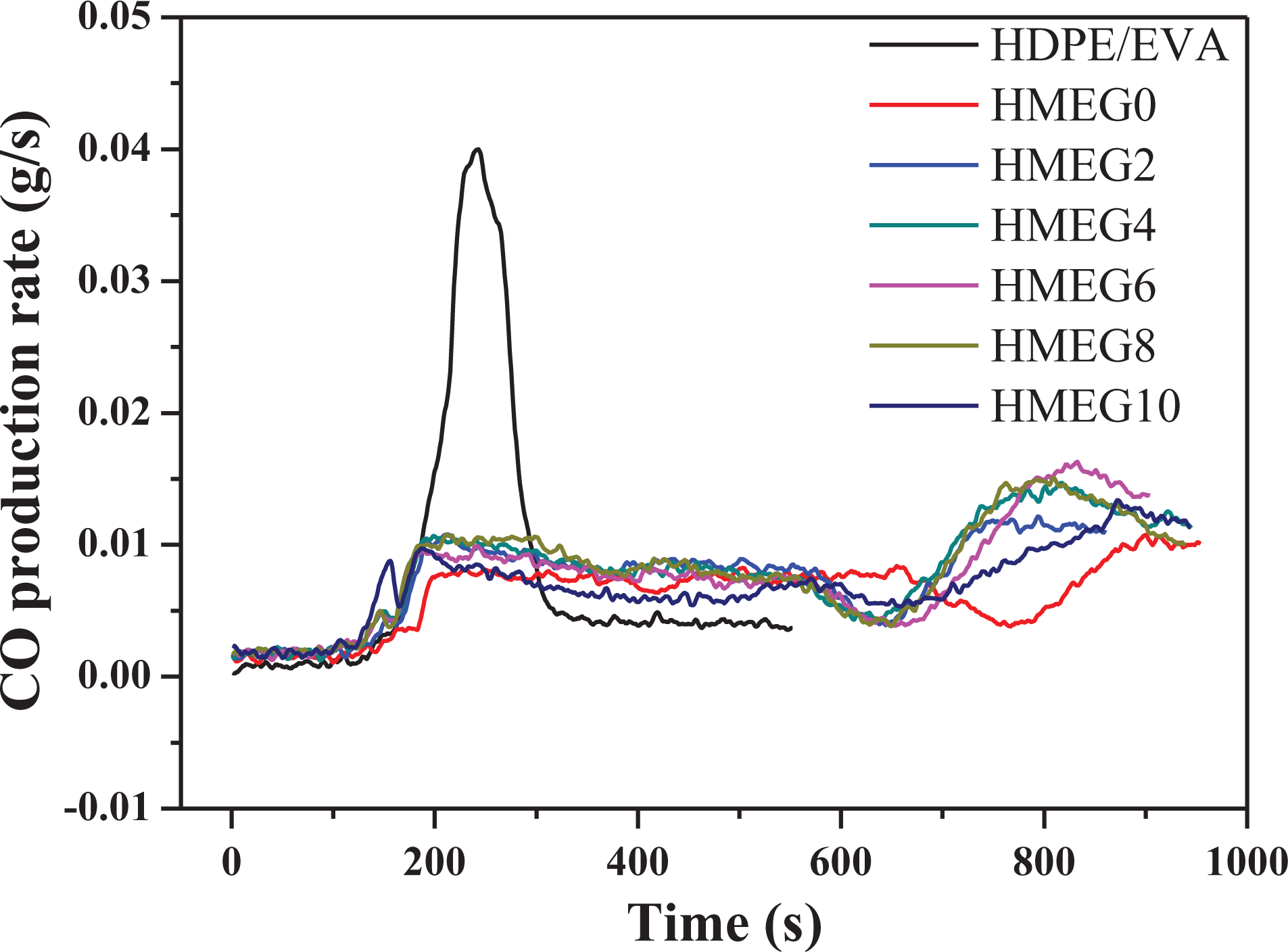
CO production rate curves of HDPE/EVA and its flame composites.
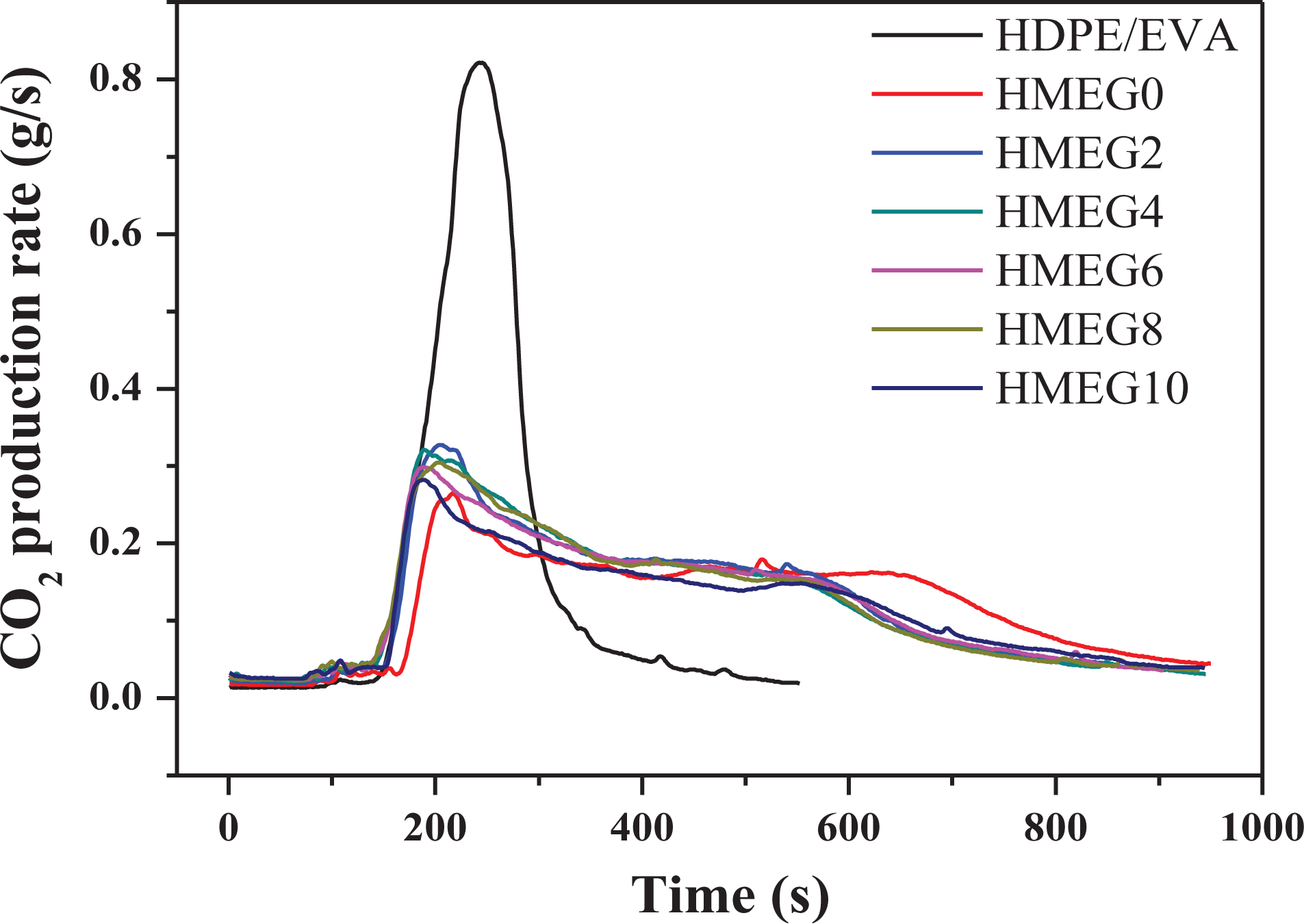
CO2 production rate curves of HDPE/EVA and its flame composites.
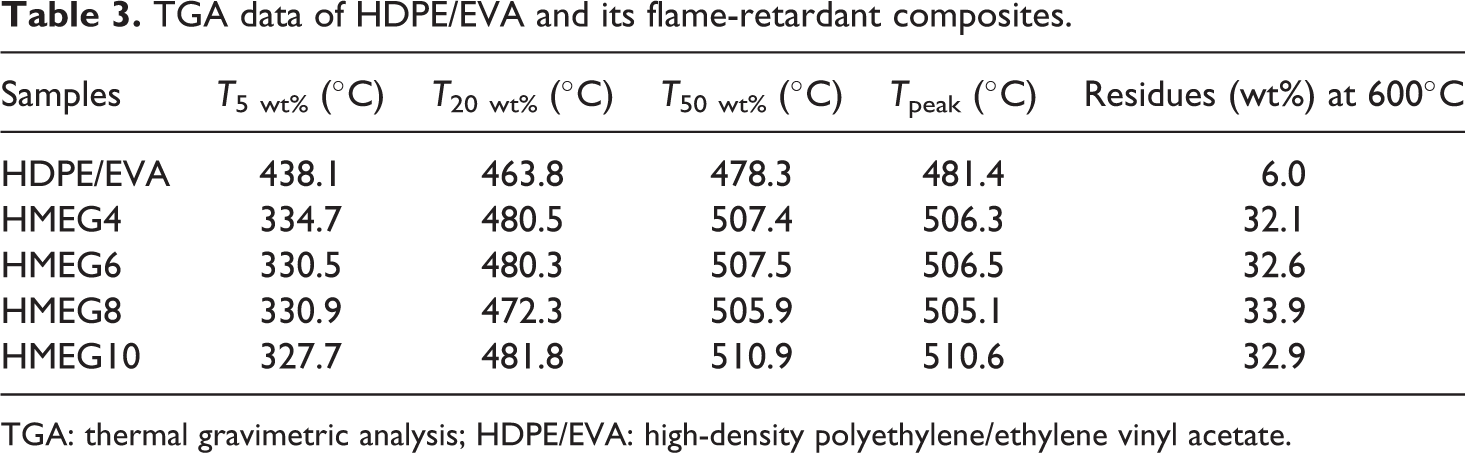
It is clearly found that the TSR data listed in Table 3 are affected greatly due to the synergistic flame retardancy between metal hydroxides and MEG. The TSR values of HDPE/EVA composites significantly decrease with the addition of MEG. It is decreased from 987.4 m2 m−2 of HDPE/EVA to 431.3 m2 m−2 of HMEG8 sample with a reduction of 56.3%. In addition, it is lower than that of HMEG0 and HMEG10. The initial ignition time (TTI) reflects the thermal conductivity or absorption of different polymer composites to be ignited in the beginning. It can be observed from Table 3 that the TTI value of HDPE/EVA is only 18 s; however, as the incorporation of compound flame retardants, the TTI values of the flame-retardant HDPE/EVA composites increase greatly. This is mainly due to low thermal stability of the graphite dispersed on the surface energy of polymer matrix. All these indicate the better flame-retardant properties of HDPE/EVA composites with MEG.
TGA data of HDPE/EVA and its flame-retardant composites.
TGA: thermal gravimetric analysis; HDPE/EVA: high-density polyethylene/ethylene vinyl acetate.
Figure 6 exhibits the charred layer morphologies of HDPE/EVA and its flame-retardant composites obtained from the CCT. It is clearly seen from Figure 6 that there is no charred residue left for HDPE/EVA. But a certain amount of charred layer is formed for the flame-retardant composites. The surface of the charred layer formed for HMEG0 and HMEG2 has been depressed. It may be that the content of MEG is low and the formed charred layer is not enough dense to block the heat into the internal material. With increasing the content of MEG, the formed charred layer has no breakage and more compact, which can effectively cut off the heat exchange between the air and the matrix.
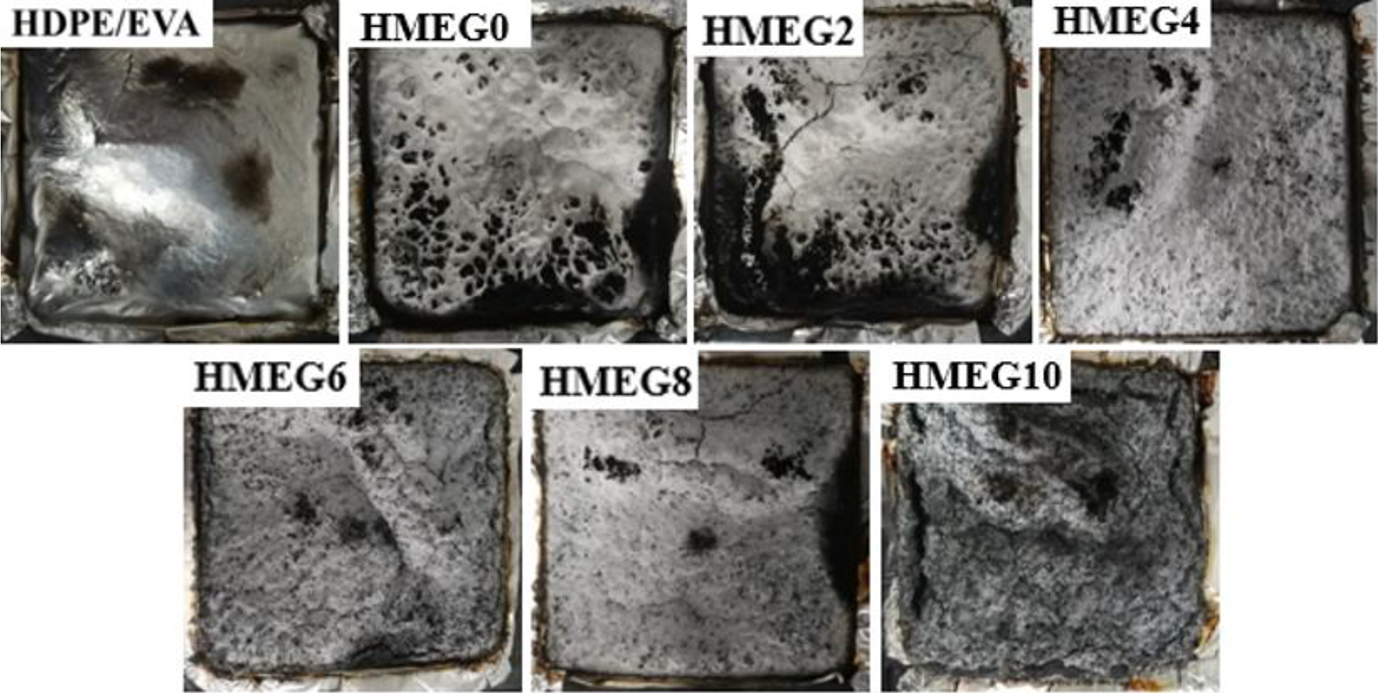
Carbon layer morphology HDPE/EVA and its flame-retardant composites.
Thermal stability and thermo-oxidative degradation of the composites
The TGA and derivative thermogravimetry curves of HDPE/EVA and its flame-retardant composites are shown in Figure 7, and the detailed data are presented in Table 3. It is obviously observed from Figure 7 that there are two different degradation regions in the whole thermal degradation process of HDPE/EVA. The first step is the degradation of EVA at 300–370°C and the second one is the degradation of the C–C bond of the HDPE/EVA at 400–500°C. However, the thermal decomposition of the flame-retardant composites mainly consists of three steps. The decomposition temperature in the first part is between 300°C and 350°C, which is the decomposition of ATH and MEG. The second part is mainly the thermal decomposition of MH. And the final degradation is C–C of polymer matrix. Compared with HDPE/EVA, the degradation of flame-retardant composites shifts to a high temperature, indicating the formation of the protection charred layers and improvement of the thermal stability of the flame-retardant composites.
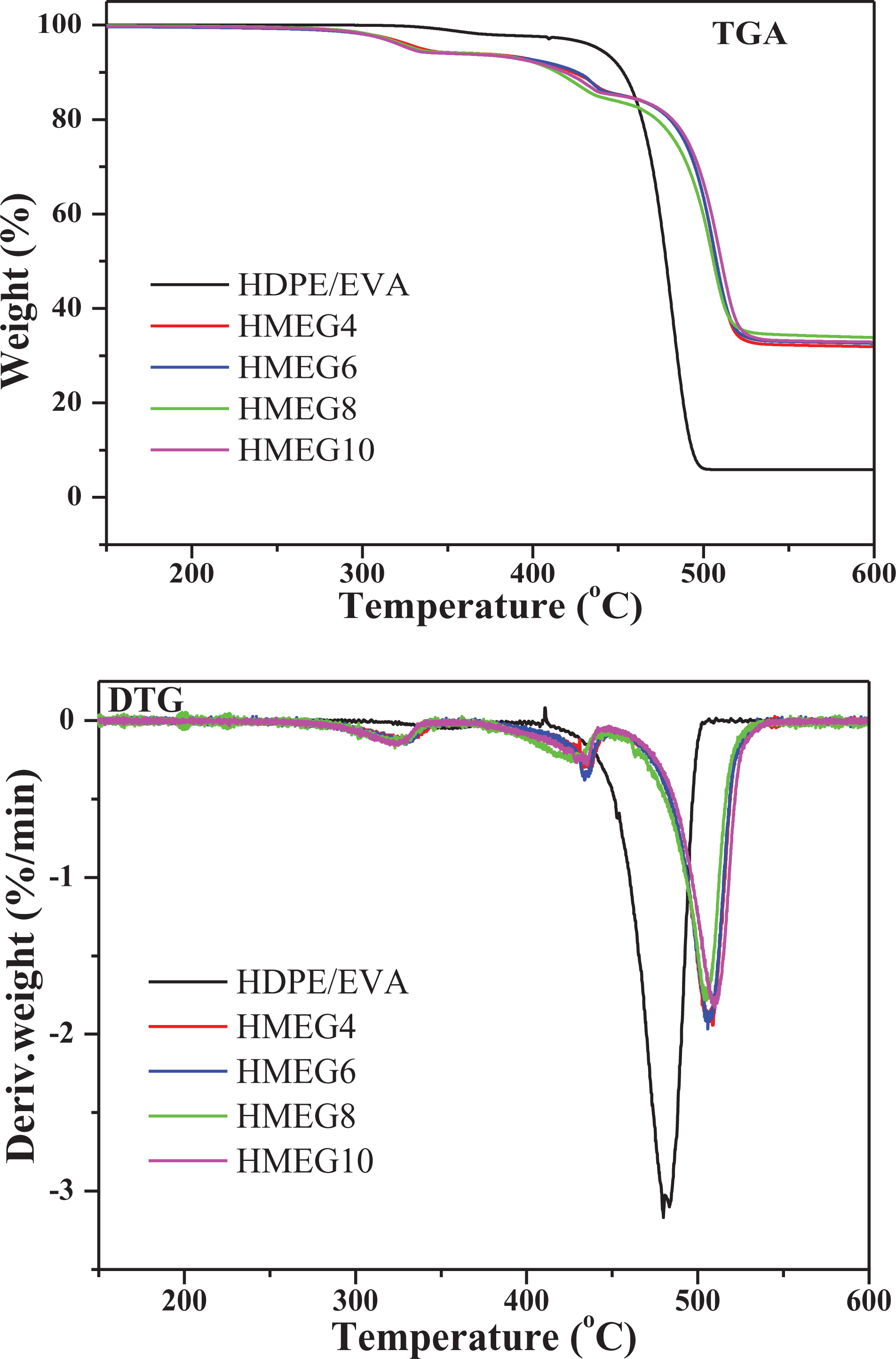
TGA and DTG curves of HDPE/EVA and its flame-retardant composites.
It is found from Table 3 that the initial decomposition temperature of HDPE/EVA is higher than all the flame-retardant composites. The initial decomposition temperature corresponding to a 5% weight loss (
FTIR spectra of the pyrolysis products for HMEG0, HMEG8, and HMEG10 at different pyrolysis temperatures are exhibited in Figure 8. The intensity of the absorption peak at 350 cm−1 decreases gradually during the thermo-oxidative degradation. This reason is due to the thermal decomposition of Mg(OH)2, resulting in a decrease of –OH content. It is clearly observed from Figure 8(a) that the peaks at 3525 and 3456 cm−1 are the vibration peaks of CH2 or CH3; 25 the peaks at 2920, 2846, and 1470 cm−1 are assigning to C–C stretching vibration; 26 the peak at 3621 cm−1 corresponds to –OH stretching vibration; 27 and the peaks at 1743, 1239, and 1029 cm−1 correspond to C=O and C–O, 28 respectively, which are the characteristic absorption peaks of EVA. These characteristic peaks have little change below 350°C, indicating HDPE/EVA may not degrade. While the temperature rises to 400°C, the characteristic peaks disappear almost because of the thermal degradation of HDPE/EVA.
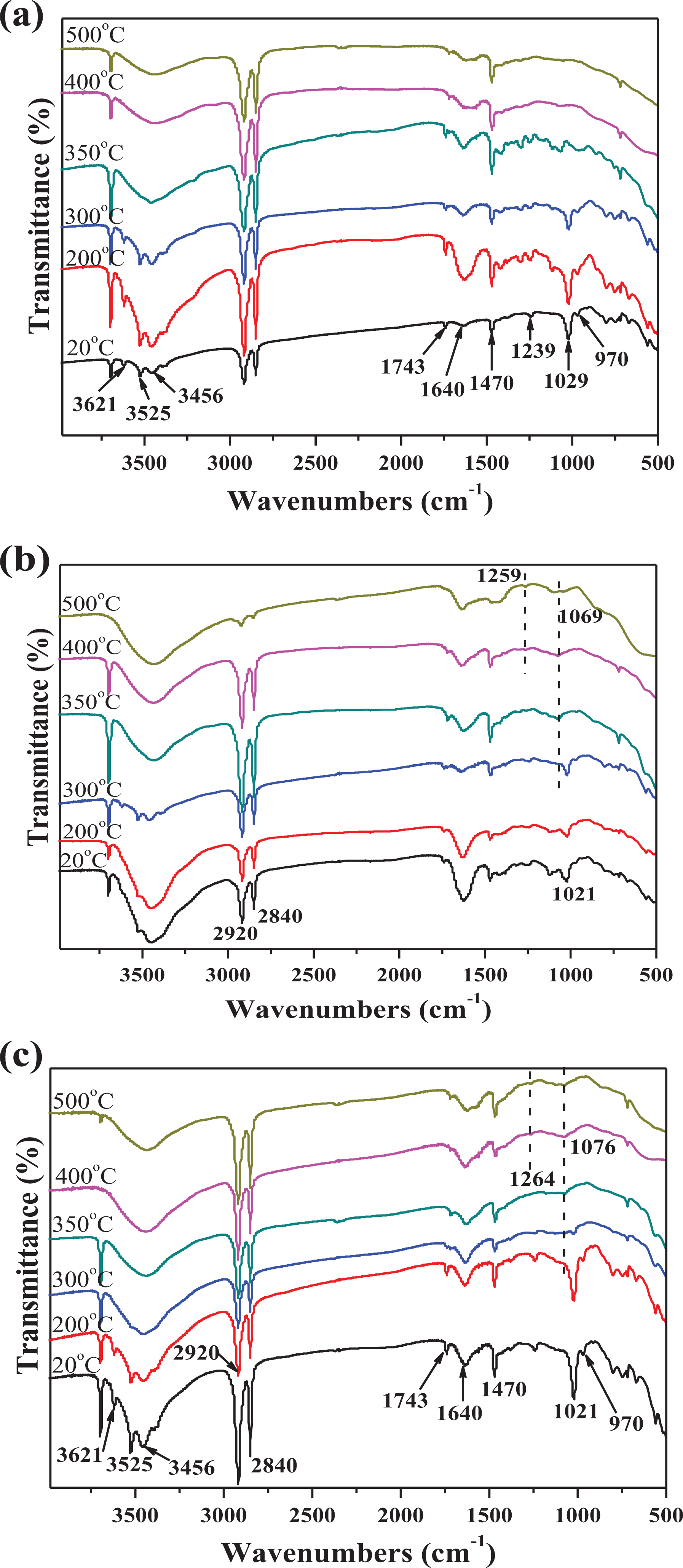
FTIR spectra of pyrolysis products at different pyrolysis temperatures for (a) HMEG0, (b) HMEG8, and (c) HMEG10.
Compared with HMEG0, the thermal decomposition processes of HMEG8 and HMEG10 are more complex. Figure 8(b) and (c) shows that the peaks of HMEG8 and HMEG10 are similar to that of HMEG0 at lower temperatures. While the temperature rises to 350°C, there are two new peaks appearing. The new peaks may be P=O and P–O of phosphate ester29,30 decomposed by MEG and it is not existing in Figure 8(a). Due to the presence of phosphate, a compact charred layer is formed to improve the thermal stability of the composites. For HMEG0 without MEG, the metal oxides and water vapor will produce at high pyrolysis temperature, which absorbs a lot of heat and dilutes the combustion gases. However, for HMEG8, the worm-like expandable charred layers are also formed when the temperature is over than 200°C. The synergistic effect between metal oxides and expandable charred layers will enhance the flame retardancy of HDPE/EVA composites. This is also confirmed by the results from LOI, UL-94, and CCT.
When HDPE/EVA matrix is exposed to high temperature, alkyl macroradicals are formed, which reacts with oxygen to form alkyl peroxide radicals. The alkyl peroxide radical abstract hydrogen forms the backbones of the polymer chains, and hydroperoxides and alkyl macroradicals are formed. The decomposition of hydroperoxides results in the production of ketones, alcohol, carboxylic acids, and so on.
Crystallization and melt behaviors
The crystallization and melting curves of HDPE/EVA and its flame-retardant composites are shown in Figure 9, and the detailed data are listed in Table 4. It is found from Figure 9 and Table 4 that the crystallization temperature (
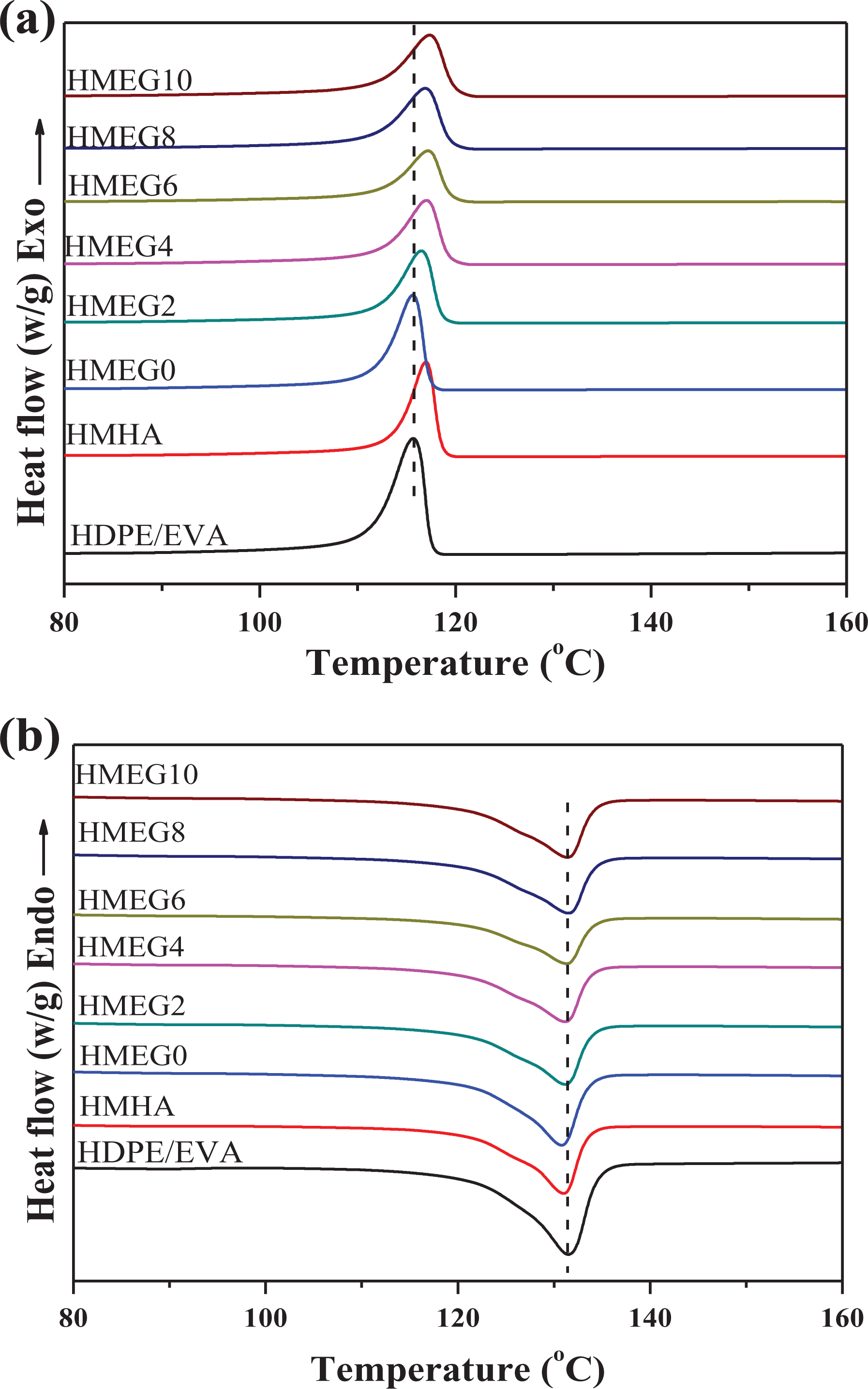
(a) DSC crystallization and (b) melting (b) curves of HDPE/EVA and its flame-retardant composites.
DSC results for HDPE/EVA and its flame-retardant composites.

DSC: differential scanning calorimetry; HDPE/EVA: high-density polyethylene/ethylene vinyl acetate.
Analysis of the data in Table 4 can be obtained that MEG plays a role of hindering the movement of molecular chains and reduces the crystallinity of HMEG systems. With the addition of MH/ATH, the
Tensile properties
The tensile properties of HDPE/EVA and its flame-retardant composites are generalized in Table 5. The tensile behavior is closely related to the content of MEG. The tensile strengths of HMHA and HMEG0 decrease significantly, and even the tensile strength of HMEG system diminishes with increasing the content of MEG. Elongation at break directly depends on the addition of MEG and decreases with increasing the MEG content. Compared with HMHA, the elongation at break of HMEG0 increases slightly. Previous study 31 has shown that ZB can enhance the elongation at break of the flame-retardant composites. The elongation at break of the HMEG containing MEG decreases with increasing the content of MEG, which indicates that MEG has a negative effect on elongation at break of materials.
Tensile properties of HDPE/EVA and its flame-retardant composites.

HDPE/EVA: high-density polyethylene/ethylene vinyl acetate.
Conclusions
In this work, the synergistic effects of ZB and MEG on the combustion behavior, thermal stability, crystallization, and tensile properties of HDPE/EVA composites with different flame retardants are characterized and discussed in detail. The flame retardancy and thermal stability of HDPE/EVA/MH/ATH composites greatly depend on the content of compound flame retardants, especially MEG. At the same time, MEG also has a negative effect on the crystallization of the flame-retardant HDPE/EVA composites. The elongation at break decreases with increasing the content of MEG. But with the incorporation MEG, the tensile strength of HDPE/EVA composites increases.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (51763002), the International Science and Technology Cooperation Project of Sichuan (2019YFH0047), the International Science and Technology Cooperation Project of Chengdu (2017-GH02-00051-HZ and 2019-GH02-00026-HZ), and Guizhou Provincial Higher Education Engineering Research Center [Qian Jiao He KY (2017)021].
