Abstract
Nitrile butadiene rubber (NBR) particles as impact modifier mixed with treated ceramic fillers (aluminum oxide, yttria-stabilized zirconia, and silicon dioxide) were used to reinforce poly(methyl methacrylate) (PMMA) denture base material. The powder components are PMMA, benzoyl peroxide, NBR (5, 7.5, and 10 wt%), and ceramic fillers (5 wt%) treated with silane. The liquid components are 90% methyl methacrylate and 10% ethylene glycol dimethacryate. Fourier transform infrared spectroscopy and energy dispersive X-ray spectroscopy analyses confirmed that the ceramic fillers were successful. The morphology of fracture surfaces of specimens was characterized using field emission scanning electron microscopy. The impact strength (IS) and fracture toughness (
Keywords
Introduction
Poly(methyl methacrylate) (PMMA) material is the most commonly used denture base material in prosthetic dentistry. Although discovered in 1937, Walter Wright introduced PMMA material; it demonstrated excellent properties and surpassed other denture base materials such as wood, bone, ivory, tortoise shell, gutta-percha, vulcanite, cheoplastic, rose pearl, aluminum, and celluloid. 1 –3 Even in the 20th century, PMMA heat curing method is still far better than denture based made of self-cure acrylic resin, epoxy resin, polystyrene, nylon, vinyl resin, polycarbonates, visible light curing acrylic, and high-impact acrylic. 4 PMMA offers remarkable physical and mechanical properties, compatible with oral tissue, aesthetics, ease of repair, and low cost. 5 It is in context, further improvement in PMMA denture base materials is very necessary.
The fracture of prosthesis or fracture of composite restorations is a very common clinical problem.
6
The geometry of denture base is complex, and stresses can be concentrated in flaws on the surface or in frenum notch, especially lingual and labial frenum in the mandible, crack can occur in the denture base.
7
Most of upper denture base (maxilla denture) fractures are caused by the fatigue and impact, whereas about 80% of fractures in the lower denture base (mandibular denture) are due to impact
8
and low fracture toughness (
One of the primary problems of PMMA is the impact failure and fatigue resistance when the denture is accidentally dropped on a hard surface or by fatigue failure when the denture base deforms repeatedly through occlusal forces.
7,8
Vojdani et al.
11
found that 68% of PMMA dentures break within a few years after fabrication. It can be easily broken during accidents or when the patient applies high mastication force on the denture base that occurs between maxilla and mandible jaws.
12
Basically, adequate impact strength (IS) and
Several studies have shown that PMMA mechanical properties’ deficiencies were overcome using various types of fillers such as glass fiber, polyethylene, aramid, carbon fiber, polylactic fiber, ceramic particle (e.g. barium titanate (BaTiO3), zirconium dioxide (ZrO2), silicon dioxide (SiO2), hydroxyapatite (HA), titanium dioxide (TiO2), and calcium carbonate), and metal plates or wires.
7,14
–19
Many studies have been focused on the treatment of filler to improve the mechanical properties of the filler–matrix interface,
20,21
and treatment with silane is very effective in promoting adhesion for silica-based materials.
22
It was reported that silane (3-(methacryloxy) propyl trimethoxysilane (γ-MPS))-treated HA helped reduce the water uptake. This is due to the ability of γ-MPS to react with hydroxyl groups in the HA filler by introducing a hydrophobic layer on surface of ceramic particles and improved its bonding strength.
20
Furthermore, mixing harder inorganic materials like treated SiO2 increased the hardness of the PMMA denture base material.
17
In another study, Asar et al.
7
found that by using ZrO2, Al2O3, and TiO2, the
The positive effects of alumina (both micro- and nanosize)-reinforcing fillers in resin composites for dental applications have been studied. 24 Alsharif et al. 25 reported that the effects of silane-treated Al2O3 particles for dental resin composite increased the hardness and flexural properties. Other researchers attempted to improve the strength of PMMA denture base by the addition of metal wires and metal plates. 11 They found that the fracture resistance has increased in both metal wires and plates. However, the main disadvantages of metal wire are poor aesthetics and adhesion between the wire and PMMA. In the case of metal plate, it is prone to corrode and expensive. Alternatively, various fibers were used in PMMA denture base such as work done 26 using polyethylene, glass, and carbon fibers, which showed better IS. However, the IS reduced when glass woven and silk fibers were used. The negative effects of fibers as reinforcement materials are mainly on the aesthetic appearance, biocompatibility, and desire of the patient. 27 Another important approach to improve the PMMA denture base composites is through incorporation of rubber particles. According to Dowling, 28 modifying a low-ductility polymer with ductile particles such as rubber increased its fracture toughness. PMMA denture base was modified by butadiene–styrene copolymer rubber and improving the IS. 29 However, another type of rubber, nitrile butadiene rubber (NBR) has never been reported being used as PMMA denture base reinforcement. NBR has various advantages that may improve the properties of the PMMA denture base such as no allergic reaction upon contact with oral tissue of prostheses- nontoxic, 30 besides acts as impact modifier to increase the IS.
In this research, the effect of nitrile rubber (NBR) particles as impact modifier together with three different treated ceramic fillers (Al2O3, YSZ, and SiO2) in a PMMA matrix was investigated for its mechanical properties. The powder component consisted of PMMA powder, 0.5 of benzoyl peroxide (BPO), NBR particles (5, 7.5, and 10 wt%), and ceramic fillers (fixed at 5 wt%).The combination of NBR with treated ceramic fillers in PMMA denture base has never been previously studied.
Materials and experimental
The materials used in this research are PMMA powder (weight-average molecular weight (
The chemical structure and function of materials that used.
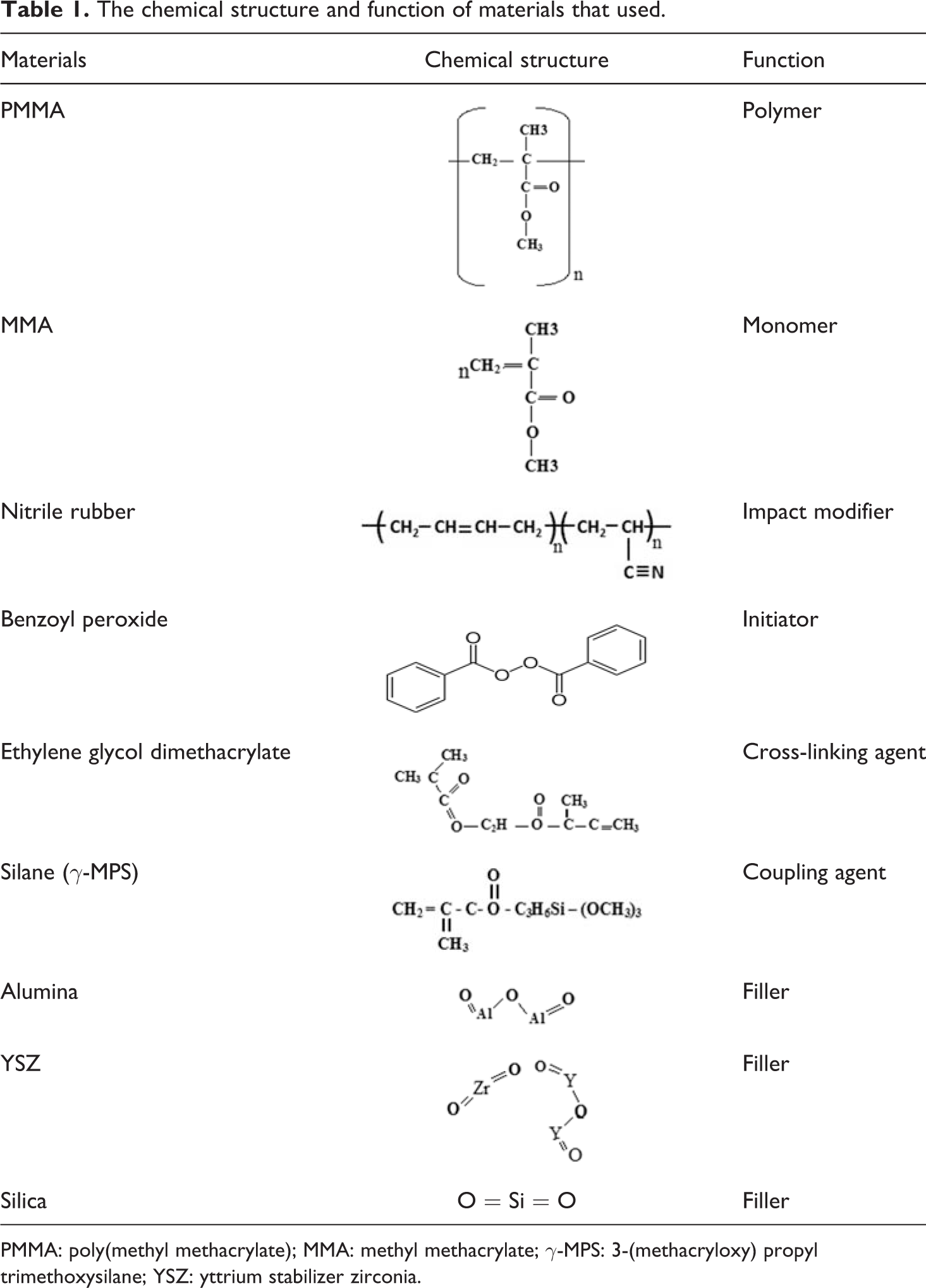
PMMA: poly(methyl methacrylate); MMA: methyl methacrylate; γ-MPS: 3-(methacryloxy) propyl trimethoxysilane; YSZ: yttrium stabilizer zirconia.
Treatment process of powder filler
Silane treatment of Al2O3, YSZ, and SiO2 composite filler
Filler treatment involved the use of 10 g of each filler powder (Al2O3, YSZ, and SiO2) with 200 ml of toluene. The procedure commenced with the dispersion of powder filler in toluene. Then, to the solution was added 10 wt% of silane at room temperature and was continuously stirred at 150 r min−1 for 15 h. Next, the solution was filtered in order to collect the modified powder. Subsequently, the powder was washed with 300 ml of fresh toluene in a Soxhlet apparatus for 24 h. The final product was then dried in an oven at 110°C for 3 h under vacuum. This treatment method also was previously described. 31
Preparation of NBR particles
NBR gloves were passed through a two-roll rubber mixing mill (Shangha Rubber Machine, China) for 5 min at room temperature to obtain strips of nitrile rubber. These strips were ground to obtain fine particles using a table-type pulverizing machine (RongTsong Precision Tech. Co. Ltd, Taiwan). The particles were sieved to pass through 150 μm. The particle size and distribution were analyzed by particle size analyzer (Mastersizer Instrument, UK).
Preparation of the PMMA denture base composite
The PMMA denture base material was prepared using the powder components mixed with liquid component. The powder components were comprised of PMMA powder, NBR particles as impact modifier, BPO as initiator (fixed at 0.5 wt%), and ceramic fillers as reinforcement particles were fixed at 5 wt% (Al2O3, YSZ, and SiO2, respectively). In total, 12 different mixtures were used in this study as shown in Table 2. Each mixture was mixed for 30 min by using the internal mixer (599957-K model, MS Instruments, Malaysia). The rotor speed was 50 r min−1 and heated at 65°C. The mixing chamber was cleaned before next mixing process to avoid contamination. The liquid medium was comprised of 10% EGDMA as cross-linking agent and 90% MMA as an activator. 18
The powder mixture components’ ratios (wt%) between nitrile rubber (NBR) and treated ceramic (fixed at 5 wt%) particles as fillers in PMMA denture base.
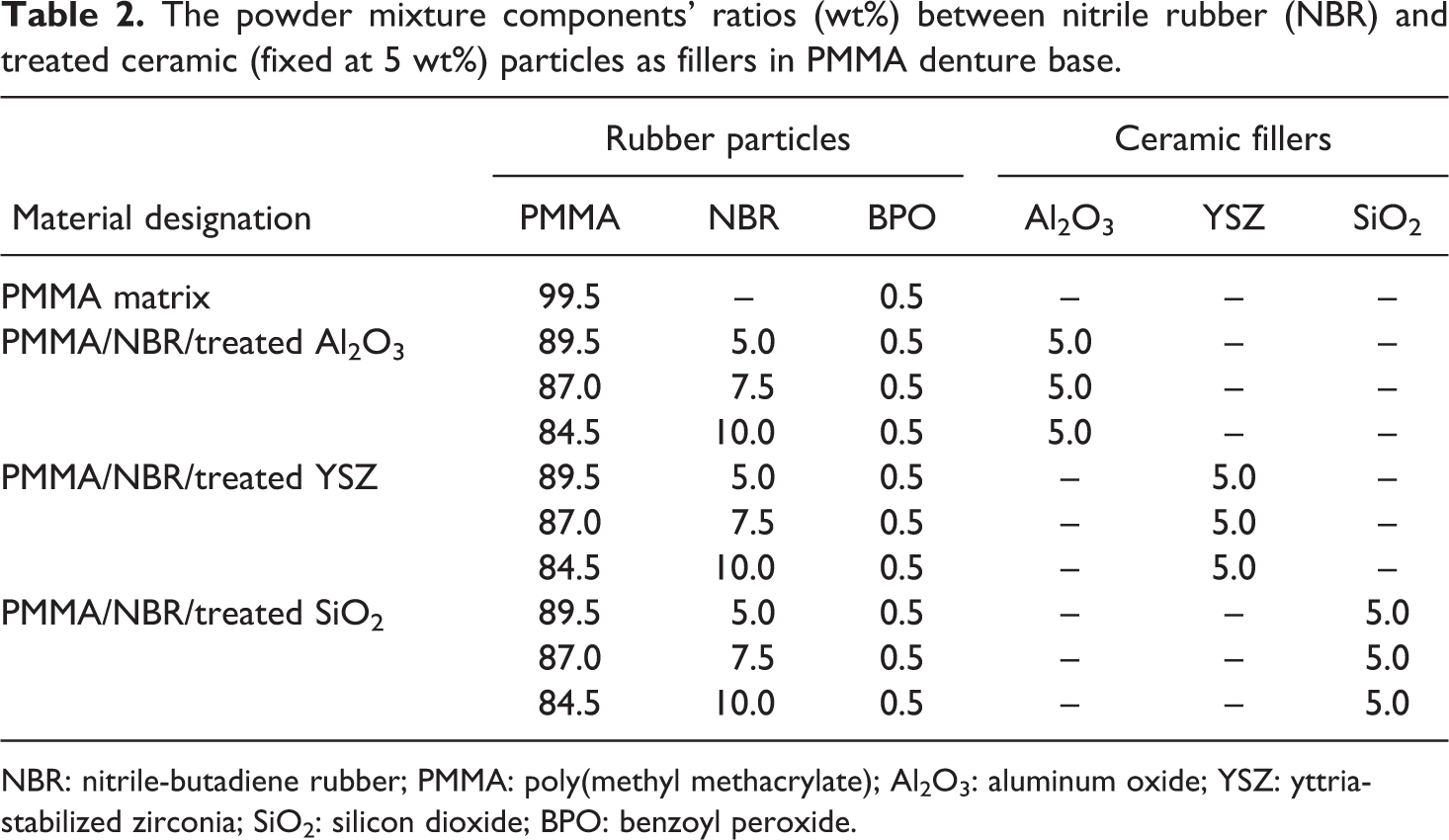
NBR: nitrile-butadiene rubber; PMMA: poly(methyl methacrylate); Al2O3: aluminum oxide; YSZ: yttria-stabilized zirconia; SiO2: silicon dioxide; BPO: benzoyl peroxide.
Each of the powder mixture components were mixed using hand mixing in liquid component. The mixing of powder mixture to liquid component
Characterization of the PMMA denture base composite
Fourier transforms infrared spectral measurements
The nitrile rubber particles were compacted into thin pellets and characterized using Universal ATR Sampling Accessory (Perkin-Elmer FTIR Spectrometer, Version 10.03.06; Waltham, Massachusetts, USA) for nitrile rubber particles. The resolution was over the wave number scanning range of 400–4000 cm−1 with resolution of 4 cm−1 in the transmission mode and performing 32 scans.
Mechanical tests
The impact test, Charpy V-notch impact test, was measured using a Zwick pendulum impact tester (Germany). The prepared samples were kept at 37°C for 24 h prior to the IS test. The dimensions of samples were 80 × 10 × 4 mm3, the width under notch

where
The fracture toughness test was determined using the single edge span notch bending test and the specimens were prepared according to ISO 13586:2000 standard. The test specimens were formed in a molded plate. The dimensions of the specimens were fixed as follows: 100 × 20 × 4 mm3 (length × width × thickness), having notch length of 4 mm and support span length of 64 mm. A crack was made on the specimens by tapping a new razor blade placed at the notch on the specimen. The specimens were tested using Instron (3366, 10 kN; Norwood, Massachusetts, USA) at a crosshead speed of 1.00 mm min−1. The specimens were kept in a humidifier at 37°C for 24 h prior to the fracture toughness test and at least six specimens for each formulation were prepared. In this case, the fracture toughness of the critical intensity factor (
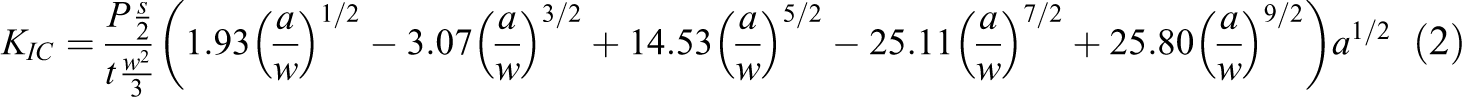
where
Morphological characterization
The morphology of the sample fracture surface of composite was studied using field-emission scanning electron microscope (FESEM; Zeiss Supra Model 35VP, Germany). The samples were mounted on aluminum stud with double-sided carbon tape, and all surfaces of samples were coated using platinum (by Bio-Rad E5150 Sputter Coater, England, UK) to eliminate the electrostatic charging effect and to enhance the image resolution in a Bio-Rad Polaron sputter coater. Energy dispersive X-ray spectroscopy (EDS) is a chemical microanalysis technique performed in conjunction with FESEM. EDS was applied to the treated filler in order to establish the composition of the filler after silane treatment.
Statistical analyses of data
The calculated values of results for tests were performed with statistical analyses (Statistical Package for the Social Sciences SPSS, statistics version 20). The data were subjected to one-way analysis of variance followed by Tukey’s post hoc test. The results of the tests were used to identify different groups and considered significant at
Results and discussion
Fourier transform infrared spectroscopy
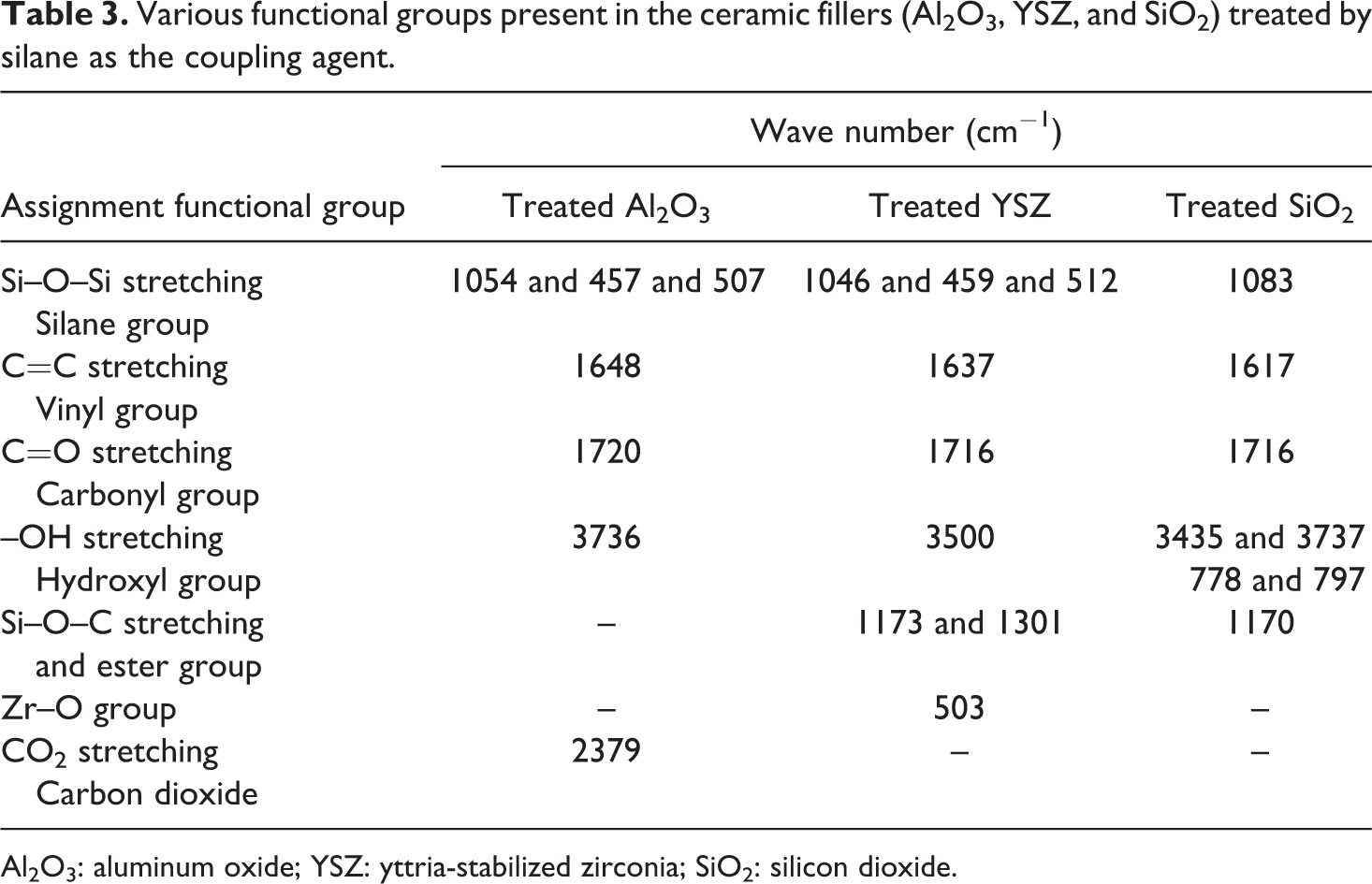
Figures 1 to 3 show the Fourier transform infrared spectroscopy (FTIR) spectra for ceramic particles before and after being treated with silane as the coupling agent (γ-MPS). The results of this finding are summarized in Table 3. The main characteristic peaks are obtained at 1040–1100 cm−1, (1617–1648 cm−1, 1716–1728 cm−1, and (3435–3800 cm−1 for Si–O–Si, C=C, C=O, and –OH groups, respectively. The findings clearly identify the presence of the siloxane (Si–O–Si) on the surface of Al2O3, which is similar to the one described by Alsharif et al. 25 who reported the presence of complex siloxane network (Si–O–Si) on the surface at 1040–1100 cm−1. According to Turner et al., 33 the Si–O–Si group observed at 1110–1053 cm−1 region indicates the availability of the coupling agent on the surface. However, Guo et al. 34 reported the presence of Si–O–Si vibration group at 1089 cm−1 for the SiO2 surface. Kang et al. 35 showed that the peaks at 1090 cm−1 attributed to Si–O–Si group on the SiO2 surface. Cervantes-Uc et al. 36 reported that the peak at 1084 cm−1 stretching indicated the presence of Si–OSi group on the SiO2 surface. The band at 503 cm−1 is attributed to the presence of Zr–O group on the surface of the filler. Otsuka and Chujo 35 reported that the peak around 500–850 cm−1 was due to Zr–O band.
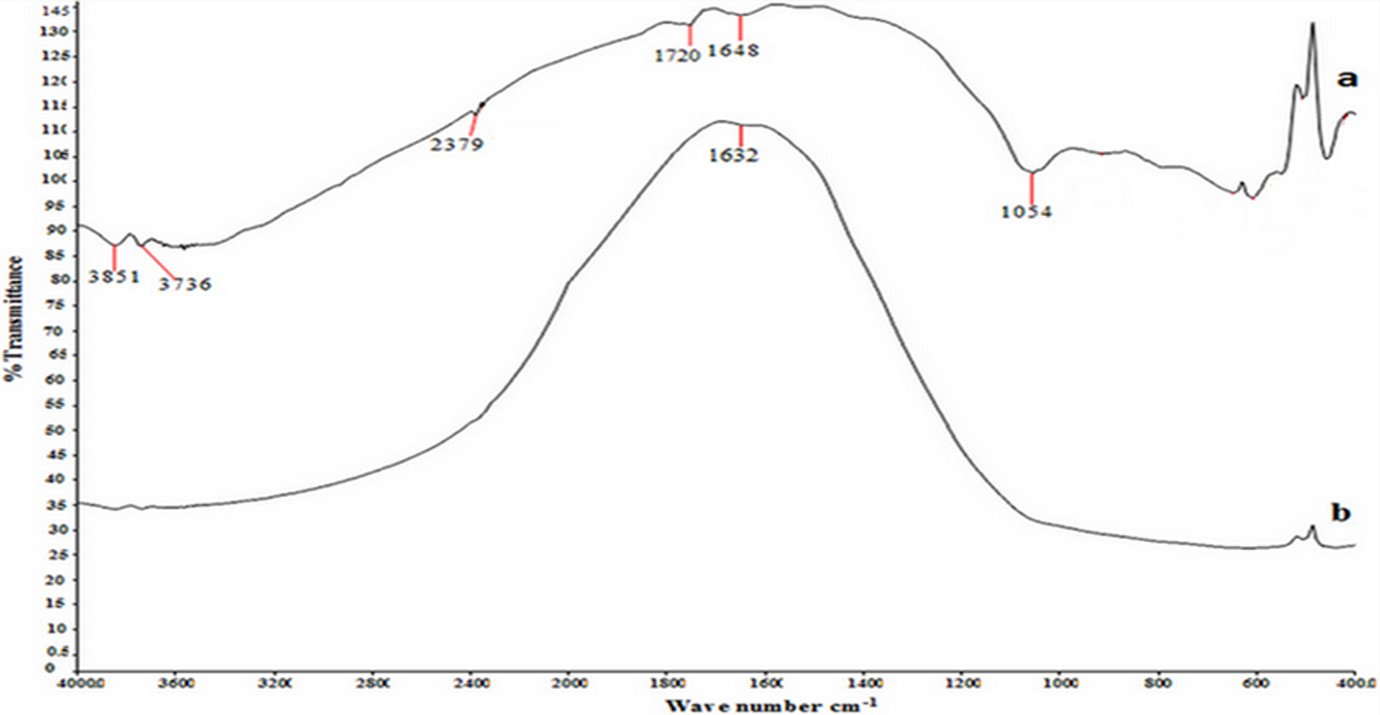
FTIR spectra of Al2O3 particles: (a) treated and (b) untreated by silane treatment. FTIR: Fourier transform infrared; Al2O3: aluminum oxide.
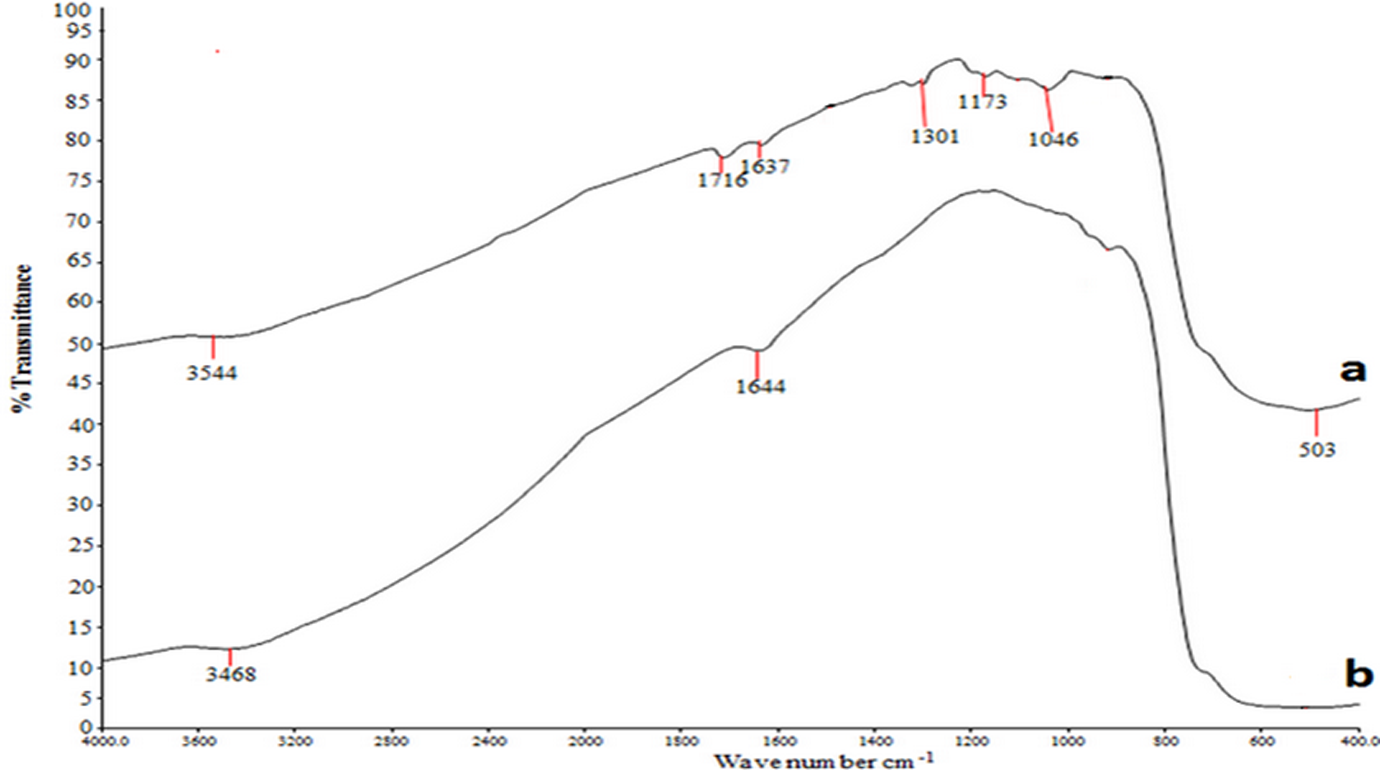
FTIR spectra of YSZ particles: (a) treated and (b) untreated by silane treatment. FTIR: Fourier transform infrared; YSZ: yttria-stabilized zirconia.
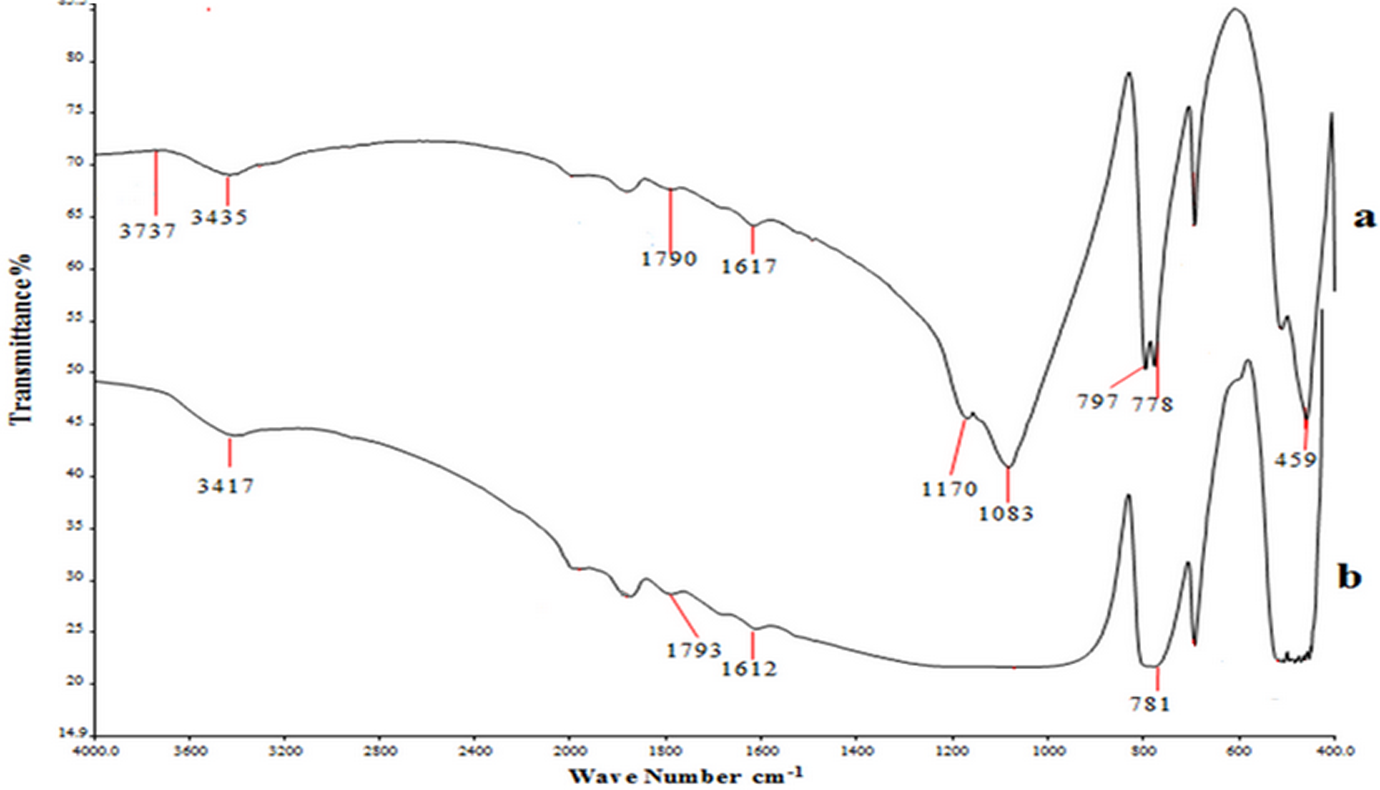
FTIR spectra of SiO2 particles: (a) treated and (b) untreated by silane treatment. FTIR: Fourier transform infrared; SiO2: silicon dioxide.
Various functional groups present in the ceramic fillers (Al2O3, YSZ, and SiO2) treated by silane as the coupling agent.
Al2O3: aluminum oxide; YSZ: yttria-stabilized zirconia; SiO2: silicon dioxide.
Table 3 refers to peak between 1617 and 1648 cm−1 attributable to vinyl C=C stretching, whereas 1716–1720 peak reflects the presence of carbonyl C=O group. Otsuka and Chujo, 37 also identified the vinyl C=C and carbonyl C=O stretching at 1637 cm−1 and 1720 cm−1, respectively. Besides, Abboud et al. 38 showed that the peak at 1719 cm−1 in treated Al2O3 can be attributed to the presence of γ-MPS on the surface. These findings are also in agreement with Wang et al. 39 who observed the carbonyl group stretching at 1716–1728 cm−1, indicating the presence of different carbonyl groups arising from the acrylic acid and the silane coupling agent.
On the other hand, the peaks at 3736, 3737, 3500, 3435, 797, and 778 cm−1 (refer Table 3) can be referred to hydroxyl groups (–OH). This is similar to the findings of Karabela and Sideridou 40 who identified the peaks for (–OH) group at 3740 cm−1. A group of peaks at 3700–3800 cm−1 is due to hydroxyl group –OH stretches on the surface of the filler. The absorption band at 3500 cm−1 and 3494 cm−1 is the evidence of hydroxyl groups as indicated by the –OH stretching band. 34,38 The broad absorption band at 3440 cm−1 was due to the –OH absorption band, which is caused by physically adsorbed water. 34,41 Kang et al. 35 reported that the peak at 3423 cm−1 is ascribed to the –OH vibrations stretching on the SiO2 surface. Jafariet al. 41 showed that the peaks at 788 cm−1 were related to the network asymmetric (–OH) bond stretching vibration of the SiO2 surface. The peak at 2379 cm−1 observed at Al2O3 is attributed to carbon dioxide (CO2). According to Guo et al., 34 the peaks around 2376 cm−1 and 2295 cm−1 corresponded to CO2 absorbed from the atmosphere on Al2O3 surface.
From Table 3, the peaks at 1301 cm−1 can be identified with Si–O–C group, whereas, 1173 cm−1 and 1170 cm−1 are attributed to ester band on the YSZ and SiO2 surfaces, respectively. Turner et al. 33 showed the presence of ester group at 1302 cm−1 on the ZrO2surface. This indicated that some of the hydroxyl groups that do not react with silanol groups (from silane coupling) were still presents on the outer surface of the SiO2 particles. 42 Silane is very effective in promoting adhesion for SiO2-based materials. 22 According to Liu et al., 43 γ-MPS can be adsorbed on the surface of SiO2 from solvent in three possible forms, (1) through the hydrogen bonding between silanol groups of γ-MPS and SiO2, (2) through hydrogen bonding of C=O and O–H group from the filler, and (3) forming multilayer in between γ-MPS molecule through hydrogen bonding.
Figure 4 shows the possible mechanism of silane as the coupling agent reaction on different ceramic fillers with addition of NBR particles in PMMA composites. Silane treatment of these fillers helps to introduce chemically active sites on the ceramic fillers surfaces, thus establishing a chemical bridge between the filler and PMMA matrix. The surface modification of fillers leads to improvement in the compatibility and interfacial bonding between the fillers and PMMA matrix. The effectiveness of silane coupling agent improved the bond between filler and PMMA matrix. This permits the modification of mechanical properties of the composite by silane bonding on the surface of oxide powders. 31 The addition of silane as the coupling agent in HA-reinforced PMMA could improve the mechanical properties by strengthening the chemical bonding and increasing the mechanical interlocking between HA and PMMA. 22
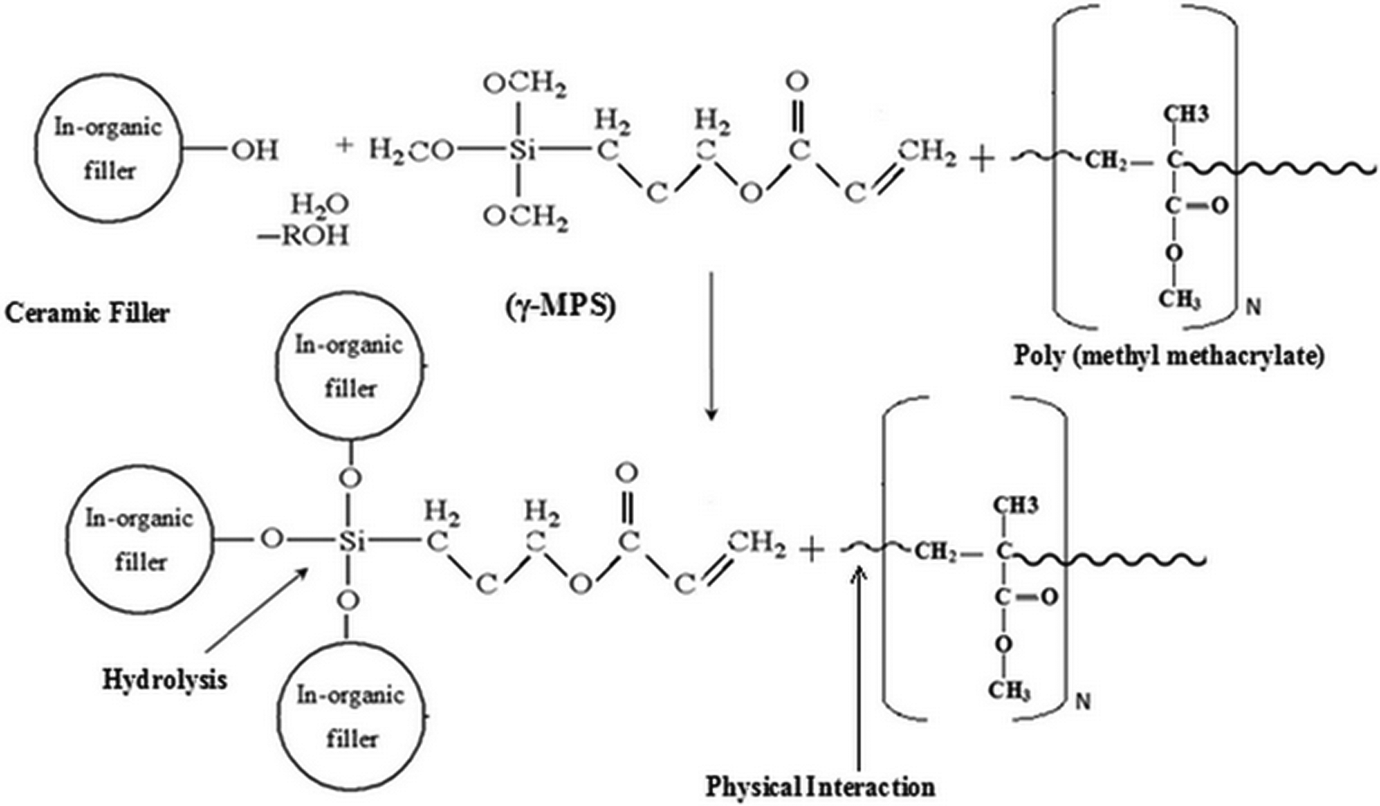
Schematic diagram representing the possible interaction mechanism reaction of silane coupling agent interlinked with inorganic ceramic surface treatment and PMMA matrix. PMMA: poly(methyl methacrylate).
FTIR spectral analysis of nitrile rubber particles
The FTIR spectrum of NBR particles is shown in Figure 5. It has various peaks that are normally present in the nitrile rubber and has good agreement with the spectrum reported. 44 The most important peak is detected at around 2237.49 cm−1, confirming the presence of C≡N group of nitrile. The band peak at 967.04 cm−1 is attributed to the –C–H stretching vibrations of butadiene double bond, which is typically present in the NBR. 45 The study by Marković et al. 46 also confirmed that the FTIR results were having C≡N group of nitrile peak at 2238 cm−1 and the – C–H group of butadiene peak starching at 969 cm−1.
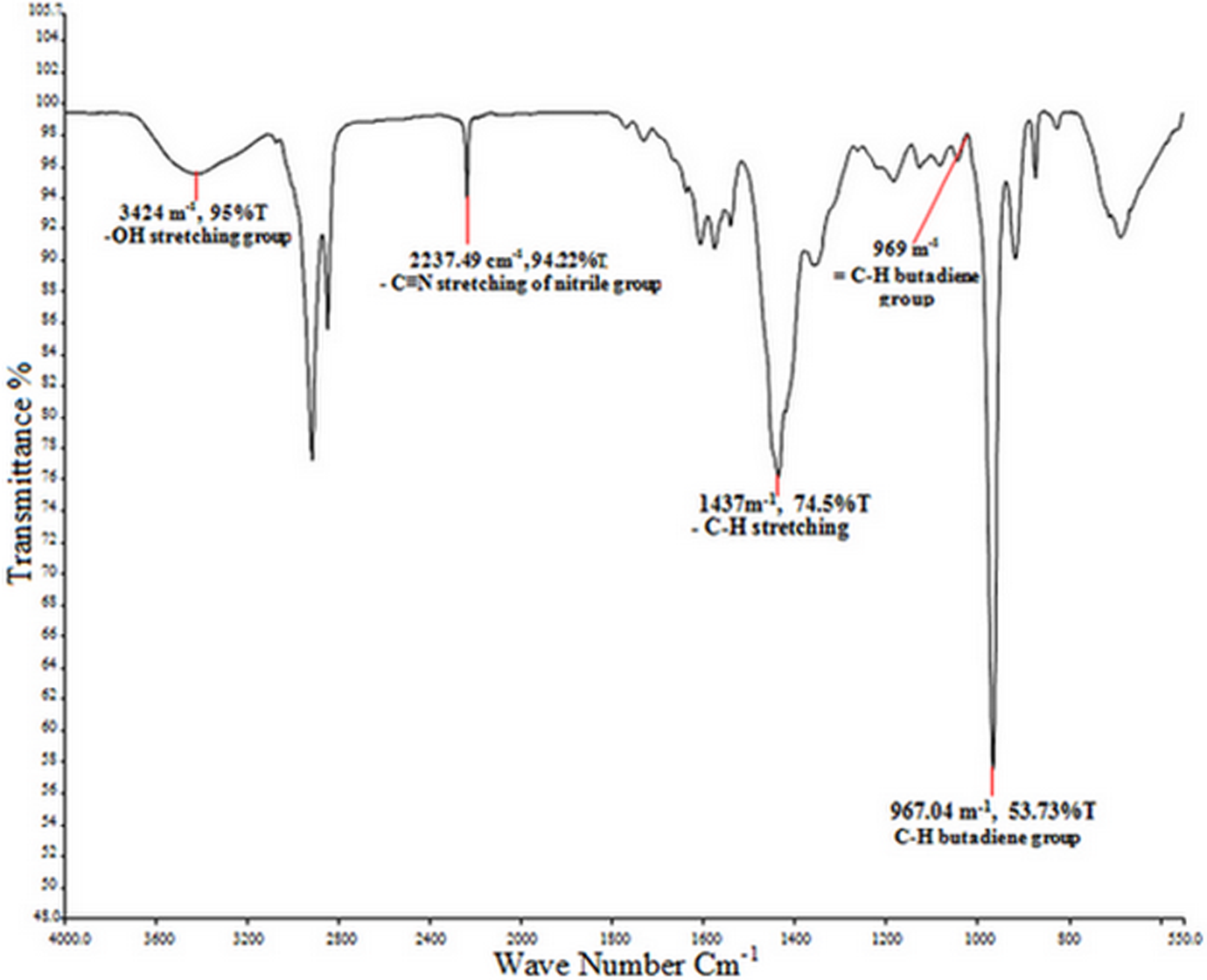
FTIR spectra shown of NBR gloves particles analyzed. FTIR: Fourier transform infrared; NBR: nitrile-butadiene rubber.
Mechanical properties of PMMA denture base composite
The PMMA matrix has been reinforced with three different treated ceramic fillers, respectively, together with NBR particles. The effects of these reinforcements on their mechanical properties are shown in Figures 6 and 7. The addition of these fillers produced higher IS and
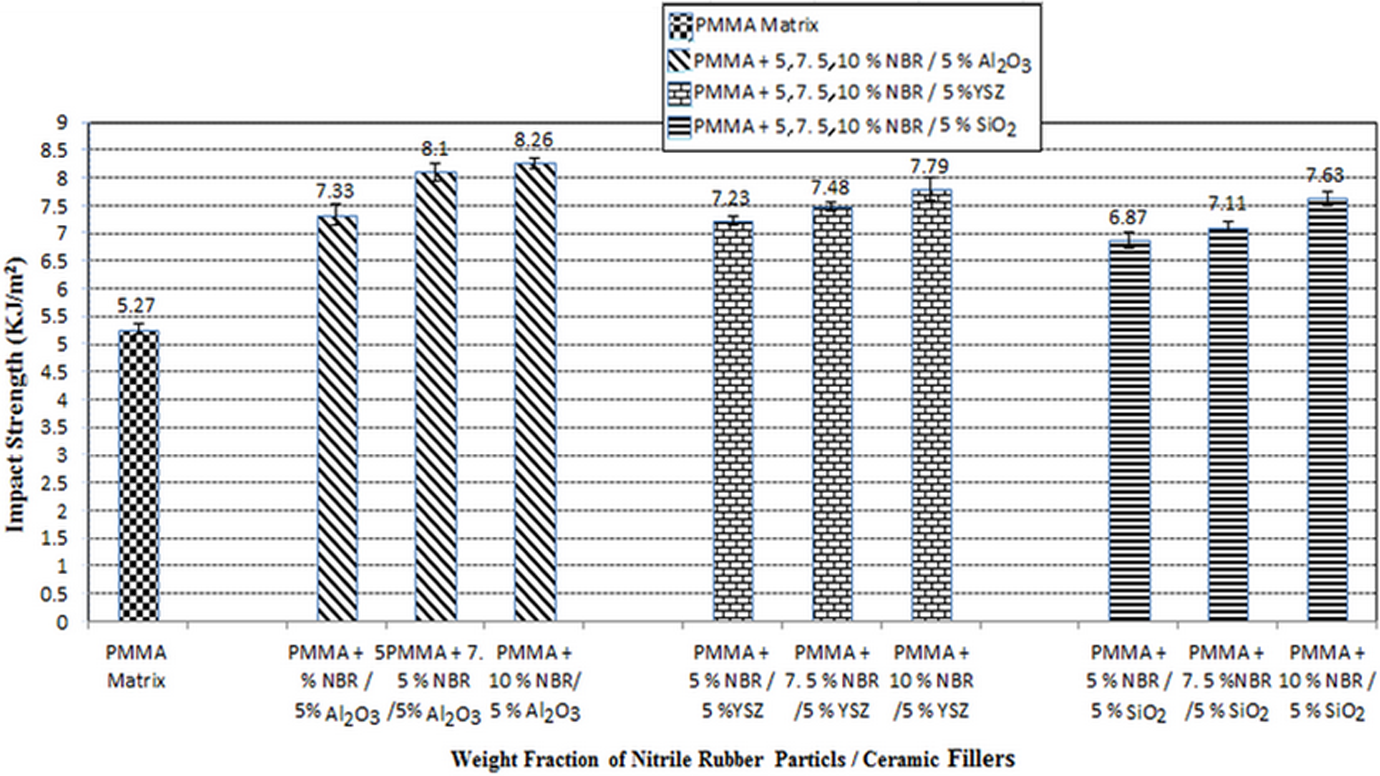
The effect of the NBR together with three different treated ceramic fillers (Al2O3, YSZ, and SiO2) in PMMA composites on the IS (kJ/m2). NBR: nitrile-butadiene rubber; Al2O3: aluminum oxide; YSZ: yttria-stabilized zirconia; SiO2: silicon dioxide; PMMA: poly(methyl methacrylate); IS: impact strength.
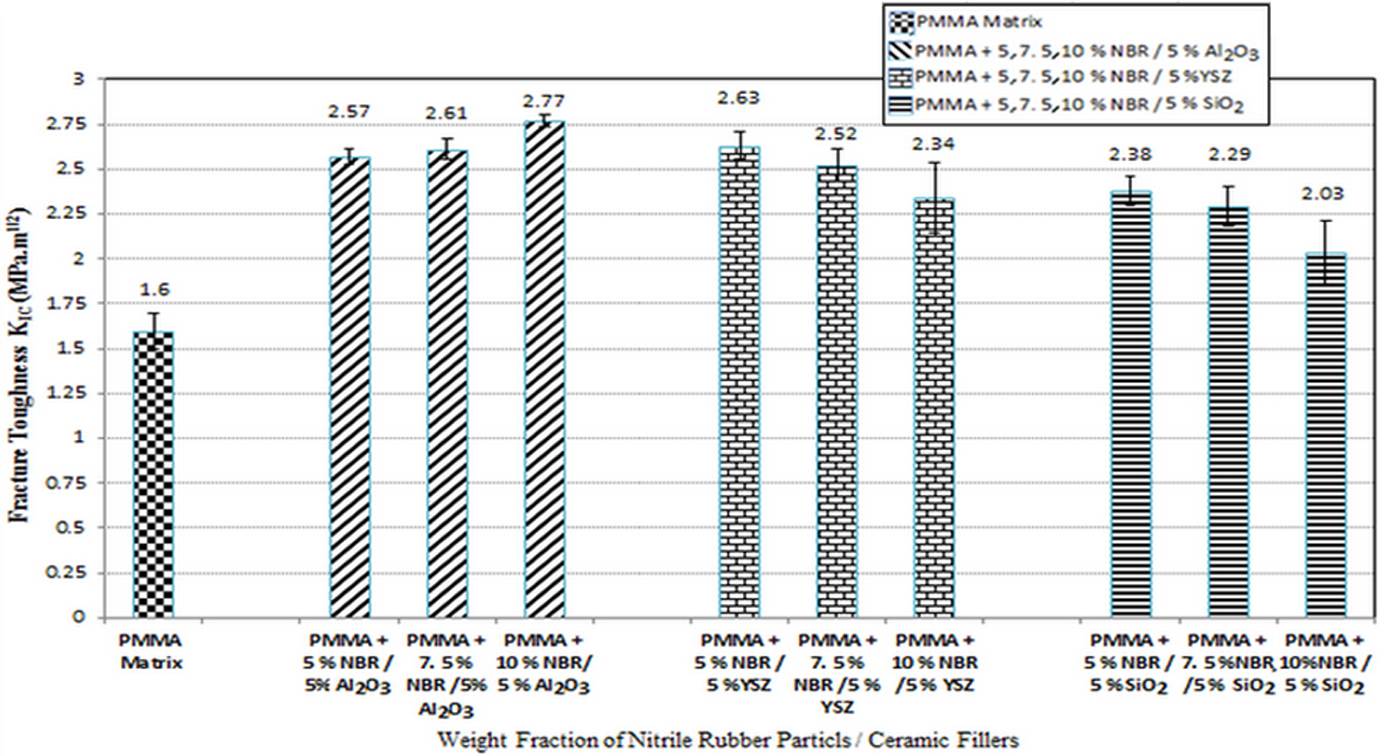
The effect of the NBR together with three different treated ceramic fillers (Al2O3, YSZ, and SiO2) in PMMA composites on the
Effect of different ratios of nitrile rubber particles with three different types of ceramic fillers treated with silane on the properties of PMMA denture base composite.a
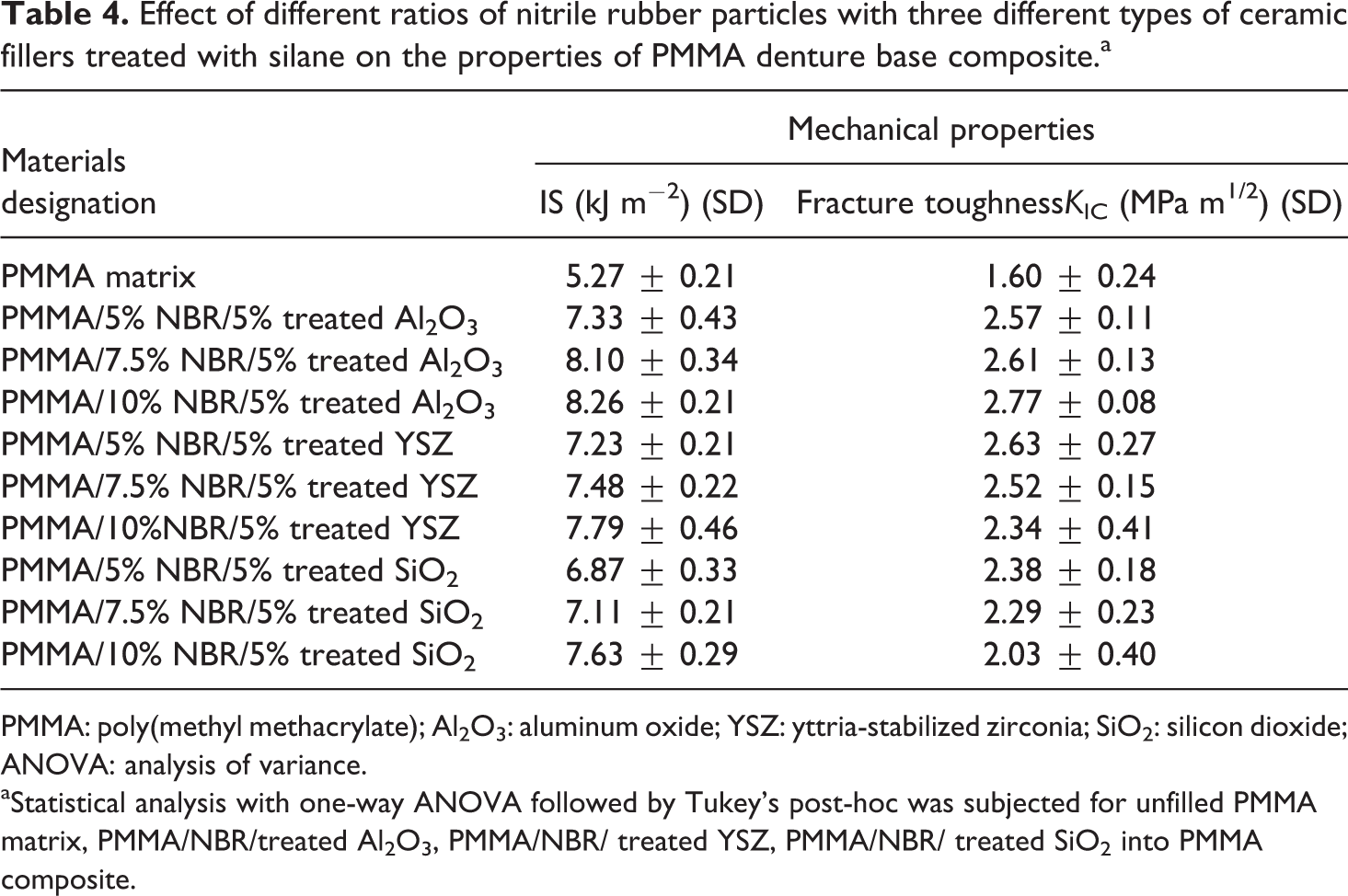
PMMA: poly(methyl methacrylate); Al2O3: aluminum oxide; YSZ: yttria-stabilized zirconia; SiO2: silicon dioxide; ANOVA: analysis of variance.
aStatistical analysis with one-way ANOVA followed by Tukey’s post-hoc was subjected for unfilled PMMA matrix, PMMA/NBR/treated Al2O3, PMMA/NBR/ treated YSZ, PMMA/NBR/ treated SiO2 into PMMA composite.
The improvement in the IS attributes to the impact resistance and excellent energy absorption capability by NBR along with the ceramic particles can help resist the occurrence of fracture to in PMMA denture base. These results are in agreement with the finding reported by Safarabadi et al. 48 who reported that IS of PMMA matrix increased significantly when reinforced by addition of 6 wt% Al2O3 particles. This could be attributed to the high interfacial shear strength that develops between the filler and matrix via the formation of cross-links or supra molecular bonding. This covers or shields the particles filler, which in turn prevents propagation of cracks. The crack propagation can be changed by good bonding between filler and resin matrix. 13,23 Several studies have also postulated that the strength of PMMA depends on the proportion of the cross-linking agent such as ethylene glycol dimethacrylate bead polymer added into the acrylic resin. 49 The addition of rubber on PMMA denture base produces better IS and facilitates dispersion of the cracks. 50 Furthermore, incorporation of PMMA reinforced by ceramic particles enhances the mechanical properties. 11,14 This result is in agreement with the finding of Ihab and Moudhaffa, 23 who reported that the addition of 2–5 wt% ZrO2 significantly improved IS performance when compared to PMMA reinforced with 7 wt% ZrO2 due to better distribution of ZrO2 particles in the PMMA matrix. Similar observation was also reported. 48 The IS of PMMA denture base material was modified by butadiene rubber and was doubled in value due to dispersion of rubber particles in the polymer. The addition of rubber particles transformed the characteristic of PMMA denture base material from brittle to ductile material with increase in rubber. 1,8 Another report by Liu et al. 43 found that incorporated PMMA matrix by treated silica filler by silane (γ-MPS) significantly affect the mechanical properties. This is due to the presence of silane on the filler surface in the composite and to reduction in debonding at the reinforced filler and matrix interface.
Figure 7 shows that the
The distribution pattern of filler (Al2O3, YSZ, and SiO2) in the PMMA matrix also plays an important role in the enhancement of

Composite particles NBR (○) and ceramic fillers (Al2O3, YSZ, and SiO2 (Δ). The filler was distributed better in the PMMA matrix composite. SEM micrographs of the fracture surface in the unreinforced and reinforced PMMA matrix, PMMA matrix (a), 5% NBR-5% Al2O3 (b), 7.5% NBR- 5% Al2O3 (c), 10% NBR-5% Al2O3 (d), 5% NBR-5% YSZ (e), 7.5% NBR-5% YSZ (f), 10% NBR-5% YSZ (g); and 5% NBR-5% SiO2 (h), 7.5% NBR-5% SiO2 (i), and 10% NBR-5% SiO2 (j). NBR: nitrile-butadiene rubber; Al2O3: aluminum oxide; YSZ: yttria-stabilized zirconia; SiO2: silicon dioxide; PMMA: poly(methyl methacrylate); SEM: scanning electron microscopy.
The role of NBR to improve
FESEM of fracture surface morphology
Figure 8 shows the FESEM micrographs of the fracture surface of unreinforced PMMA matrix and reinforced PMMA prepared using different ratios of NBR particles and treated ceramic fillers. The figure shows that addition of fillers results in the formation of microcracks in the microstructure. The trend is that the higher filler provides more cracks and this reflected in their
The fracture surfaces of the reinforced PMMA denture base by different ratios of NBR from 5 wt% to 10 wt% with 5 wt% of treated ceramic fillers (Al2O3, YSZ, and SiO2, respectively) are depicted in Figure 8(b) to (k). For the combination between Al2O3 with NBR particles (Figure 8(b) to (d)), the particles are very well distributed in the matrix and the surface displays more crack and are rough. This is similar to the observation.
51,52
They examined the development of crack accelerations in the PMMA; the fracture resistance increased with the dispersion of the rubber particles in the PMMA matrix. The NBR particles are not seen detached and pulled out of the PMMA matrix. This fact leads to an increase of the absorbed energy and explains the rise of the
Micrographs of PMMA matrix filled with NBR and YSZ fillers shows homogeneous distribution of fillers as seen in Figure 8(e) to (g). The morphology was rough with small cracks on the surface. The homogenous filler dispersion enhanced the ability of the composite to distribute the stresses uniformly. Therefore, this led to higher resistance of fracture force and absorbs greater amounts of energy at a higher strain rate before fracture. 53 The ability to resist crack propagation can be augmented by the addition of reinforcing filler. 18 The treated YSZ filler improves the interfacial interactions between filler and PMMA matrix and improves the mechanical properties. Similar observations were made. 21
For treated SiO2 particles with NBR particles, the cracks size is slightly bigger on the surface compared to other fillers as shown in Figure 8(h) to (j). This is due to incompatibility between SiO2 filler and NBR particles as shown by the surface contact between SiO2 and matrix. However, the stress at which such a ductile fracture mechanism takes place is reduced when the rubber particles increased. As the amount of NBR is increased (from 5 wt% to 10 wt%) the
Figure 9(a) to (c) shows the FESEM micrographs and EDS analysis of PMMA composite with NBR particles and ceramic fillers (Al2O3, YSZ, and SiO2) as the reinforcement particles. The ceramic fillers were treated with silane coupling agent (γ-MPS). EDS analysis detected the presence of silicon element at the interphase of each ceramic filler in the NBR/PMMA composite. This observation has confirmed the presence of silane as the coupling agent on the ceramic fillers in PMMA composites. The properties of the polymer composites provide adhesion between the polymer and filler particles. 56
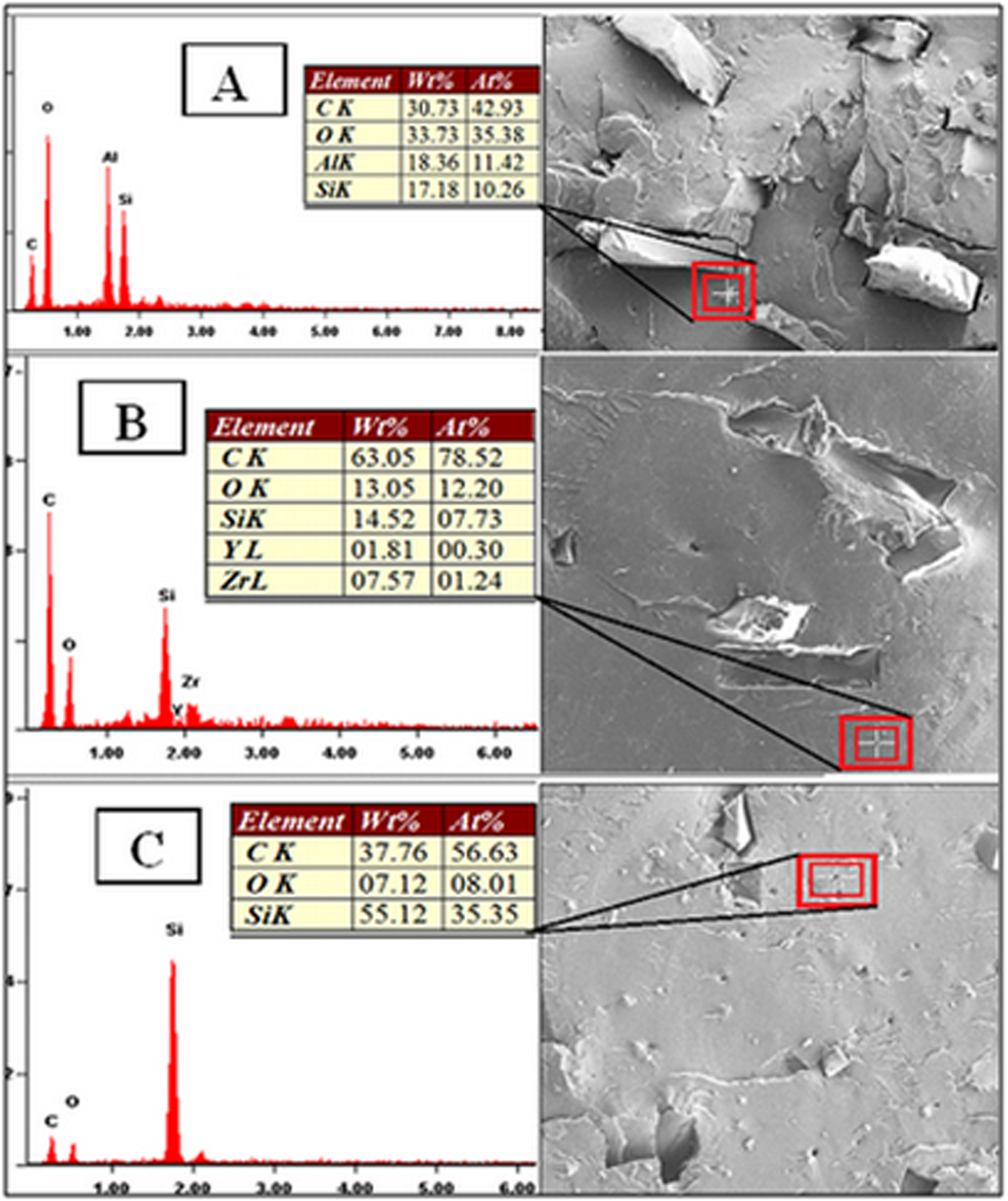
(Confirmation of silane treatment by EDX: (a) Al2O3 particles, (b) YSZ particles, and (c) SiO2 particles in the NBR/PMMA composite. EDX: energy-dispersive X-ray spectroscopy; Al2O3: aluminum oxide; YSZ: yttria-stabilized zirconia; SiO2: silicon dioxide.
Conclusion
In this work, the effect of nitrile rubber (NBR) particles as impact modifier together with three different ceramic fillers (Al2O3, YSZ, and SiO2) in PMMA matrix was investigated for its mechanical properties. Both the IS and the
Footnotes
Acknowledgement
The authors gratefully acknowledge the financial support from the School of Materials and Mineral Resources Engineering, USM under the polymer Composite Research Cluster Grant 1001/PKT/8640013 and Almorgab University, Libya.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
