Abstract
In the present work, a different green coupling agent (GCA) was developed from virgin coconut oil (GCA-C). The GCA-C were a kind of glycidyl fatty acid ester which are reactive to natural filler. A comparison between GCA made from palm oil (GCA-P) and coconut oil (GCA-C) was studied. The results indicated the tensile strength and tensile modulus of composites increased with increasing the GCA content, and the 3 wt% of GCA content was the optimum content to achieve maximum improvement. Meanwhile, the increased GCA content slightly reduced the elongation at break of composites, but the elongation at break increased at 5 wt% of GCA content. The GCA-C had better performance in improving interfacial adhesion compared to GCA-P due to different fatty acid content. The micrographs of scanning electron microscope evidenced that the modified cocoa pod husk (CPH) with 3 wt% GCA had better filler–matrix adhesion with polypropylene matrix.
Introduction
To date, agricultural by-product has become popular choice of alternative filler in plastic industry due to the fact of sustainable features, economic factor, and accumulation of agricultural by-product as waste material. 1 –3 Generally, agricultural by-product is the best choice of filler as locally available, abundance, inexpensive, and benefit to waste management. Nowadays, numerous combinations of agricultural by-product and thermoplastic materials have been made into composite materials and already marketed by industry. The well-known example of composites based on agricultural by-product in Malaysia was eco-friendly tableware made from rice husk–thermoplastic composites that produced by Melsom Biodegradable Enterprise. 3 –5 Currently, this research focused on using cocoa pod husk (CPH) as filler in polypropylene (PP)-based composites because the CPH is the main by-product of cocoa industry which is abundant and widely available in Malaysia. Commonly, the CPH accounts up to 76% of weight of fresh cocoa pod. 6,7 However, most of the CPH is burned as the ways to dispose the waste. Thus, the use of CPH to produce composite material as value-added outcome which can benefit to environment, economy, and technology.
However, the hydrophilic nature of natural filler was the main drawback in production of composites. 8 –11 The plenty of polarized hydroxyl groups in natural filler leads to weak interfacial adhesion and poor wettability with nonpolar polymer matrix. 9,12 –14 Therefore, the coupling agent is usually applied to impart bonding and adhesion between natural filler and matrix. The silane, 6,12,15 –17 maleic anhydride grafted polymer, 5,13,18,19 fatty acid, and its derivative 2,3,6,13,20 –22 are usually used as coupling agent which shows remarkable improvement in natural filler-filled thermoplastic composites. In present work, a green coupling agent (GCA) made from coconut oil was initially developed in our laboratory. Ideally, this GCA is made from renewable resources, reactive to natural filler, and inexpensive compared to commercial coupling agent that is commonly used. The GCA is a kind of glycidyl fatty acid ester produced by reacting sodium fatty acid from plant oil and glycidyl chloride. The chemical structure of GCA consists of reactive epoxide group and nonpolar hydrocarbon chains and it can chemically bond on natural filler surface by reacting with hydroxyl group. This will lead organophilic properties to natural filler that improve wetting of natural filler by thermoplastic, thereby enhancing the filler–matrix adhesion.
The previous study was focused on the development of GCA made from palm oil (GCA-P) for PP/CPH composites. 23 This work described the effect of GCA-C content on tensile, water absorption, and morphological properties of PP/CPH composites. A comparison on tensile properties between PP/CPH composites with GCA-P and GCA-C was studied.
Methodology
Materials
The discarded CPH was collected from cocoa plantation, Perak (Malaysia). The CPH was dried in oven at 80°C (24 h). Then, dried CPH was fine ground in a miniature grinder (RT-34, Taiwan). The mean particle size of fine CPH was 22 µm, which was analyzed by Malvem particle size analyzer instrument (Italy). PP (type copolymer) was supplied by Titan Petchem (M) Sdn. Bhd. Glycidyl chloride was obtained from Sigma Aldrich (St Louis, Missouri, USA) and ethanol was obtained from Fluka (Switzerland). The virgin coconut oil used in this experiment was produced from fresh coconut milk according to the method noted by Chun and Husseinsyah. 2 The GCA from coconut oil (GCA-C) was prepared by reacting glycidyl chloride and sodium fatty acid that produced from virgin coconut oil via saponification (Figure 1). The preparation method of GCA-C was similar to previous study. 23
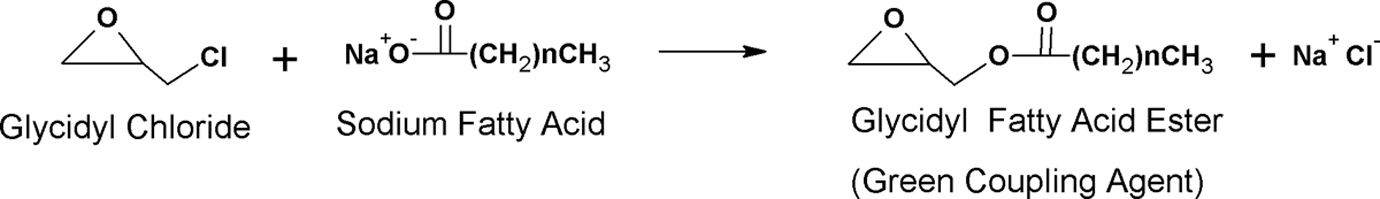
Schematic reaction of glycidyl chloride and sodium fatty acid from coconut oil to produce green coupling agent.
Filler modification
The GCA-C/ethanol solutions were prepared according to different concentrations of GCA-C following the formulation listed in Table 1. The CPH was modified by immersing them in GCA-C/ethanol solution under constant stirring for 1 h. Then, the CPH was soaked in GCA-C/ethanol solution for 12 h. The modified CPH was filtered and dried in an oven at 80°C for 24 h to completely remove the ethanol.
Formulation of PP/CPH composites.

CPH: cocoa pod husk; GCA: green coupling agent; PP: polypropylene; phr: part per hundred resin.
aBased on weight of CPH.
Preparation of composites
All composites were compounded in a Brabender Plastrograph intermixer (Model EC PLUS, Germany) in counter rotating mode at 180°C (8 min) and a rotor speed of 50 r min−1. The PP/CPH composites was prepared according to Table 1. The compounding procedures involved were as follows: (i) the PP pellets were added into the mixing chamber for 3 min until it melted homogeneously and (ii) the unmodified or modified CPH was incorporated to the melted PP and continuously mixed for 5 min. Finally, the composites compound was collected from mixing chamber. The composite compounds was molded into 1-mm thickness sheet at 180°C using a compression molding machine (model GT 7014A). The molding sequences involved were as follows: (i) preheat the compound for 4 min; (ii) compress the compound under a pressure of 100 kgf cm−2 for 1 min; and (iii) cooling under the same pressure for 5 min. Composite sheets were cut into tensile bar using a dumbbell cutter with dimension according to ASTM D638 Type IV standard. 24
Tensile test
The tensile specimens were tested for their tensile properties using an Instron Testing Machine (model 5569, Norwood, Massachusetts, USA) following ASTM D638 standard. 24 A crosshead speed of 30 mm min−1 was selected and the load cell used was 50 kN. A minimum of five specimens were measured for each composites.
Morphological analysis
The fracture surface of tensile specimen was examined for morphological properties using scanning electron microscope (SEM; model JSM-6460 LA, JEOL, Japan). The specimen was coated with a thin layer of palladium before putting into the SEM chamber.
Fourier transmission infrared analysis
The modified CPH was characterized for chemical functional groups by PerkinElmer Paragon 1000 Fourier transmission infrared (FTIR) spectrometer (Waltham, Massachusetts, USA). The attenuated total reflectance method was used. The sample was recorded with sixteen scans in the frequency range 4000–600 cm−1 with a resolution of 4 cm−1.
Results and discussion
Characterization of modified CPH
The FTIR spectrums of unmodified and modified CPH were shown in Figure 2. The main characteristic peaks of unmodified CPH were listed in Table 2. For the modified CPH, the peaks’ intensity at 2924, 2849, and 1734 cm−1 increased along with the increasing of GCA-C content. The change of peak intensity at 1734 cm−1 was related to the presence of ester bonding between GCA-C and CPH. Moreover, the peaks at 2924 and 2849 cm−1 evidenced the nonpolar hydrocarbon chains from GCA-C attached on CPH via ester bonding. Thus, the intensity of the peaks at 2924, 2849, and 1734 cm−1 changed with more GCA-C content. This indicated that the amount of GCA-C bonded on CPH surface was increased. The schematic hypothetical reaction of GCA-C and CPH was illustrated in Figure 3.
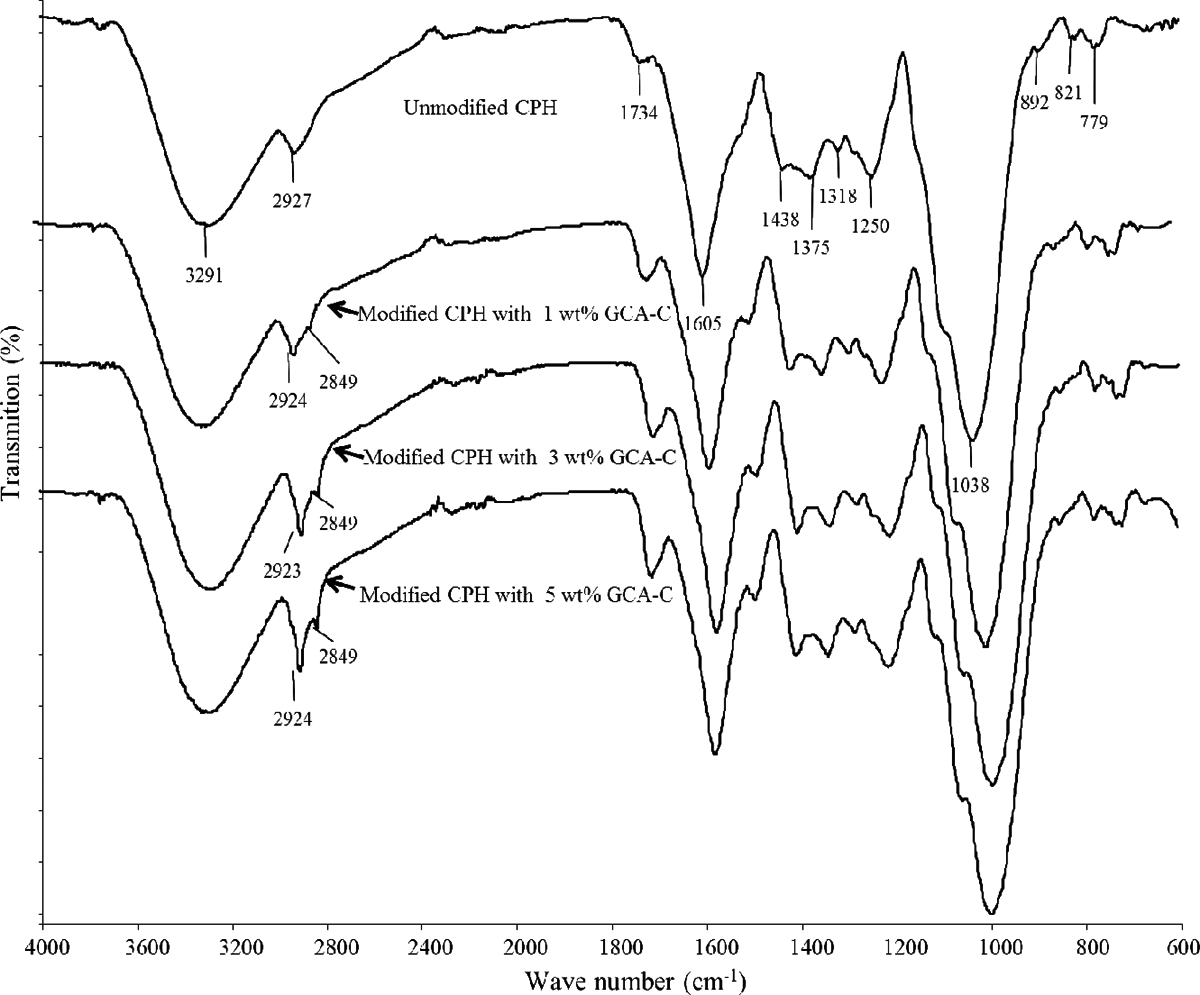
FTIR spectrums of unmodified and modified CPH with different GCA-C content.
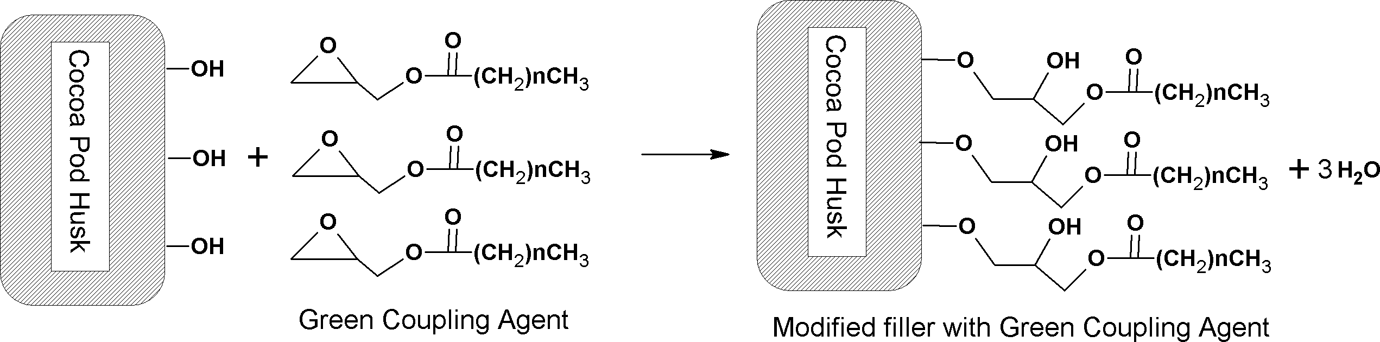
Schematic reaction between cocoa pod husk and GCA-C.
Characteristic peaks of unmodified cocoa pod husk. 4

CPH: cocoa pod husk.
Tensile properties
Figure 4 shows the effect of various GCA content on tensile strength of PP/CPH composites with at 20 part per hundred resin of CPH content. The tensile strength of PP/CPH composites increased with the increase of GCA content from 1 to 3 wt%. At 3 wt% of GCA content, the modified PP/CPH composites with GCA-P or GCA-C show highest tensile strength. This indicated that the 3 wt% of GCA content was the optimum content for this composites system. Nevertheless, the tensile strength of PP/CPH biocomposites reduced at 5 wt% of GCA content. This observation was also reported by Danyadi et al. 13 and Demjen and Pukanszky. 17 A reasonable explanation for the observations was related to the surface coverage of GCA and wettability between PP and CPH. At 1 wt% of GCA content, the CPH surface was slightly covered by GCA layers. The PP matrix was partially wetting the CPH surface as there are more hydroxyl groups that remain on CPG surface (as shown in Figure 5(a)). Thus, there is a little improvement in filler–matrix adhesion. Hence, the modified PP/CPH with 1 wt% GCA exhibited a little increment of tensile strength. Moreover, the GCA is widely covers the CPH surface at a concentration of 3 wt%, which leads to better wetting of PP matrix on CPH surface (as illustrated in Figure 5(b)). Both the modified PP/CPH biocomposites with 3 wt% of GCA content achieved a highest tensile strength because of stronger adhesion between filler and matrix. According to Figure 5(c), the modified CPH with 5 wt% of GCA content had larger surface coverage of GCA and it leads to poor filler–matrix adhesion. At this concentration, the formation of tightly packed coupling agent layer on CPH surface might increase the difficulty of CPH wetting by PP matrix and decrease the occurred interfacial adhesion. Therefore, the tensile strength of PP/CPH biocomposites dropped rapidly at 5 wt% of GCA content.
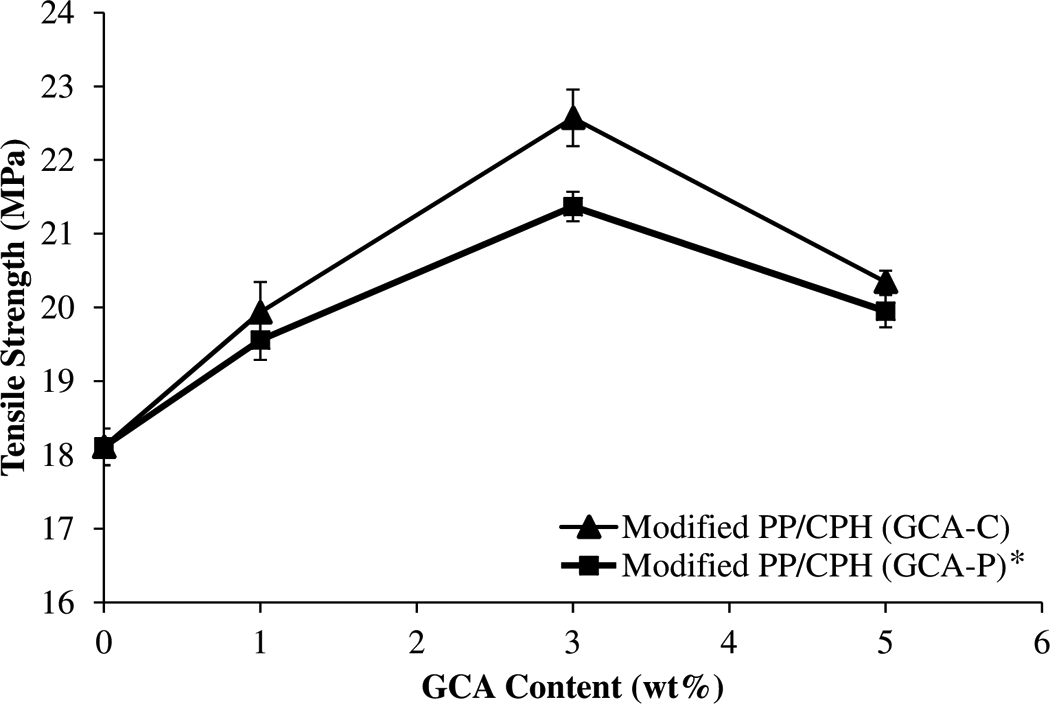
Effect of various GCA content on tensile strength of PP/CPH composites.

Different idealized surface coverage of GCA and wettability of matrix on CPH surface at various GCA content.
The effect of various GCA content on the elongation at break of PP/CPH biocomposites was demonstrated in Figure 6. The increase in GCA content from 1 to 3 wt% slightly decreased the elongation at break of PP/CPH biocomposites. This is because of the addition of GCA that enhanced the filler–matrix adhesion and reduced the chains’ mobility of PP matrix. The modified PP/CPH biocomposites with GCA were more rigid than unmodified PP/CPH biocomposites. Alternatively, the elongation at break of modified PP/CPH biocomposites increased as the GCA content at 5 wt%. This probably because the filler was fully covered by the GCA layers and it causes lubricating effect on the PP/CPH biocomposites. Demjen and Pukanszky 17 reported that the filler surface completely covered at high concentration of coupling agent will lead to decrease in the strength of adhesion but increase in the elongation at break.
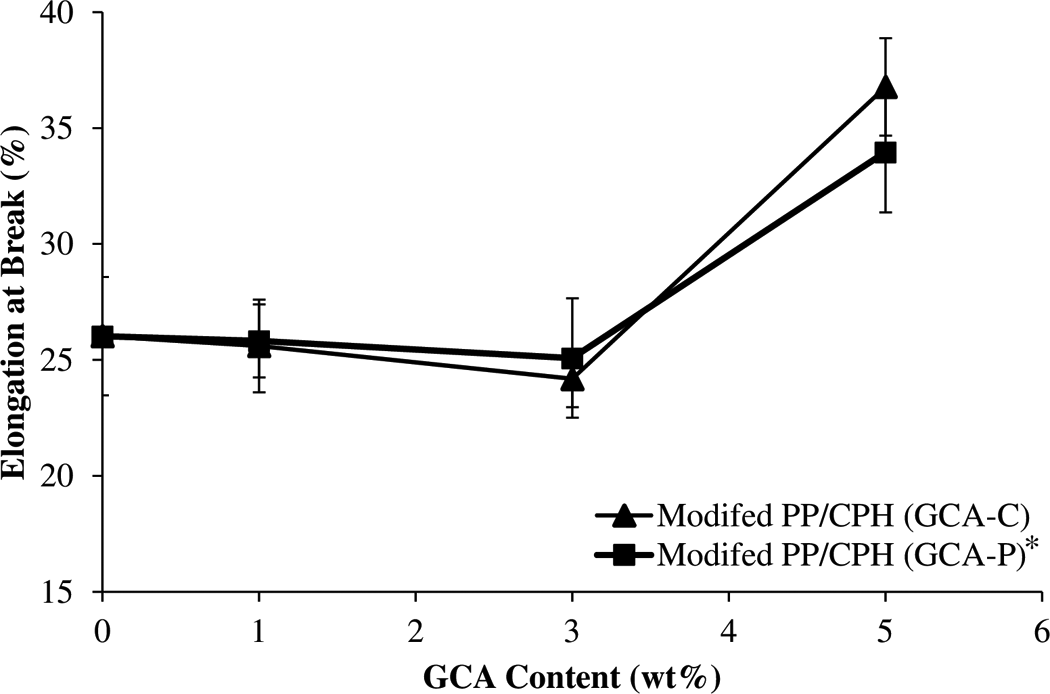
Effect of various GCA content on elongation at break of PP/CPH composites.
The effect of various GCA content on tensile modulus of PP/CPH biocomposites is shown in Figure 7. The trend of tensile modulus was similar with tensile strength of PP/CPH biocomposites as a function of GCA content. The modified PP/CPH biocomposites with 3 wt% of GCA content also show highest tensile modulus. After concentration of 3 wt%, the tensile modulus of PP/CPH biocomposites was promptly decreased. This can be explained by the modified CPH with 1 and 3 wt% of GCA exhibited a small surface coverage of GCA on CPH surface, which leads to better wetting of CPH surface by PP matrix. As a result, the filler–matrix adhesion was enhanced, which causes the tensile modulus to increase. Alternatively, a larger surface coverage of GCA was found in modified CPH with 5 wt% of GCA. This might lead to the difficulty of CPH wetting by PP matrix. Thus, a decreased tensile modulus due to the weak interfacial adhesion can be observed.
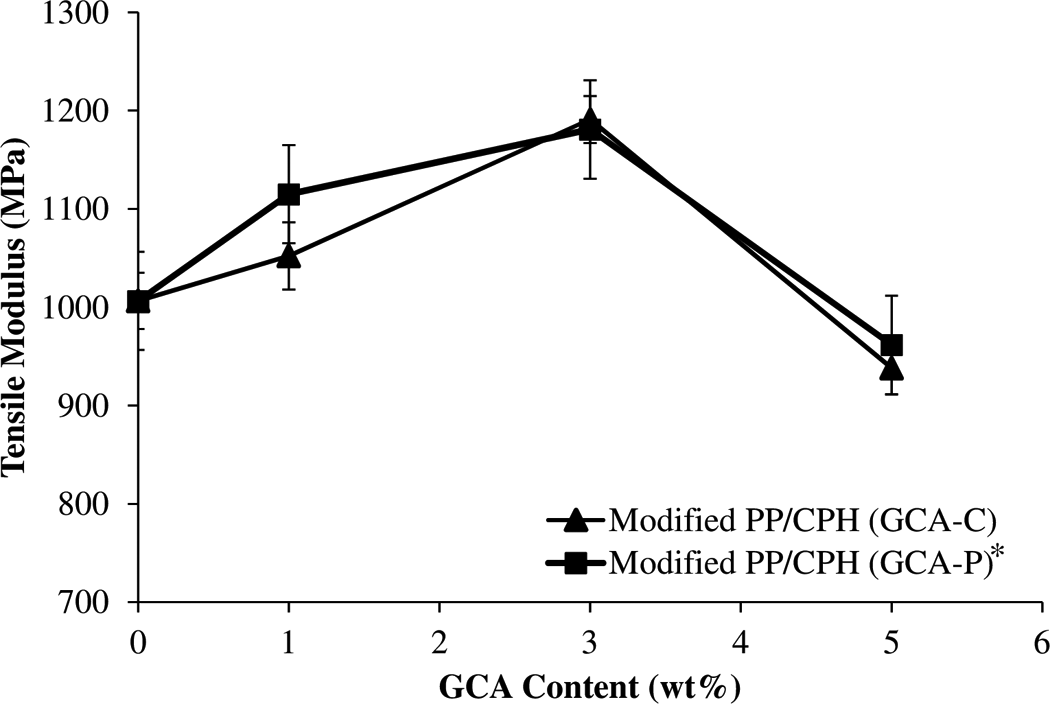
Effect of various GCA content on tensile modulus of PP/CPH composites.
Furthermore, the tensile strength of modified PP/CPH composites with GCA-C shows an average of 3.17% higher tensile strength compared to modified PP/CPH composites with GCA-P. The efficient between GCA-C and GCA-P in improving interfacial adhesion was different. This is probably due to the different fatty acid content in both GCA structures. The GCA-C has higher amount of straight alkyl chains in the structure because of the high saturated fatty acid content. From Figure 8(a), the straight alkyl chains from GCA-C are perpendicularly attached on filler surface, and it might facilitate the wetting of matrix on filler surface to achieve strong interfacial adhesion. In the other way, the GCA-P consists of certain amount of unsaturated fatty acid. Thus, some bended alkyl chains (because of the cis double) present in the structure of GCA-P might influence the wetting of matrix on filler (as shown in Figure 8(b)). In short, the GCA-C with more saturated fatty acid content shows better efficiency in enhancing interfacial adhesion between filler and matrix. Therefore, the modified PP/CPH with GCA-C achieved higher tensile strength.
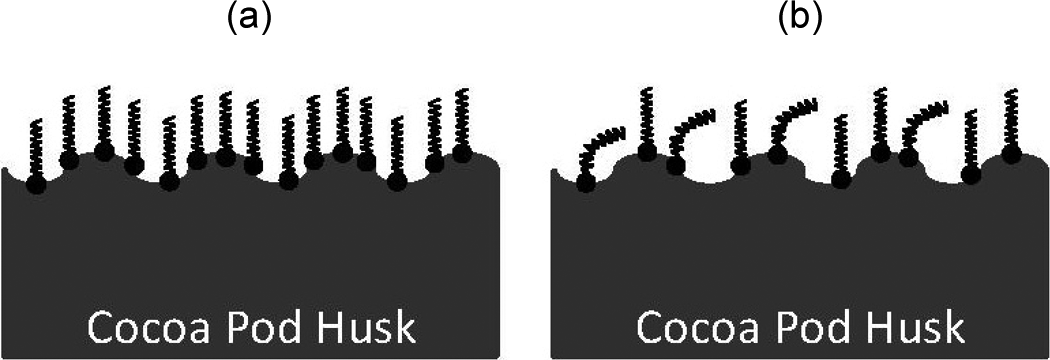
Proposed different orientation of (a) GCA-C and (b) GCA-P on CPH surface.
Morphological properties
Figure 9(a) to (d) displayed SEM micrographs on tensile fracture surface of PP/CPH composites with and without different GCA content. Figure 9(a) shows that filler was poorly dispersed and a number of holes related to the detachment of filler indicates poor interfacial adhesion. At 1 wt% of GCA content, the number of detachment of filler was reduced (as shown in Figure 9(b)). This is because the 1 wt% of GCA slightly improved the adhesion between filler and matrix. According to Figure 9(c), the composite with 3 wt% of GCA shows most of the fillers were embedded and well covered by PP matrix that evidenced good interfacial adhesion (as shown in Figure 9(b)). However, a matrix tearing can be observed in SEM micrograph of PP/CPH composite with 5 wt% of GCA (Figure 9(d)), which indicated a ductile fracture behavior due to the lubricating effect. Figure 9(d) shows that the fillers still embedded in PP matrix, but there are gaps at interface region. This evidence shows that the modified CPH with 5 wt% of GCA was unable to achieve a good adhesion with PP matrix due to poor wetting of PP matrix on CPH surface that was fully covered by a GCA layer.

Tensile fracture surface of (a) unmodified PP/CPH composites and modified PP/CPH composites with (b) 1 wt%, (b) 3 wt%, and (d) 5 wt% of GCA-C.
Conclusion
The FTIR result exhibited that the addition of GCA-C was chemically bonded with CPH via ester bonding. The tensile strength and tensile modulus of PP/CPH composites show a significant increment with 3 wt% of GCA. However, the elongation at the break of PP/CPH composites with 3 wt% GCA was reduced. This indicated that the 3 wt% of GCA content was the optimum content for this composites system. After this concentration, the tensile strength and tensile modulus of PP/CPH composites were reduced, but the elongation at break was increased. The GCA-C exhibited a better performance in enhancing interfacial adhesion between PP and CPH compared to GCA-P. The SEM micrograph also evidences the good adhesion between modified CPH with 3 wt% GCA-C and PP matrix. The use of GCA-C shows a remarkable improvement of tensile properties on PP/CPH composites. Thus, the GCA is a potential coupling agent for thermoplastic composites containing natural filler.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
