Abstract
The aim of the research was to utilize cocoa pod husk (CPH) in polypropylene (PP) biocomposites. Maleated polypropylene (MAPP) was used as coupling agent to improve the properties of PP/CPH biocomposites. The addition of MAPP had increased the stabilization torque of PP/CPH biocomposites. The tensile strength and modulus of PP/CPH with MAPP were higher compared to PP/CPH biocomposites without MAPP, except the elongation at break decreased. The crystallinity and thermal stability of PP/CPH biocomposites with MAPP increased. These improvements were due to the enhanced interfacial bonding between CPH and PP matrix, which were proved by SEM analysis.
Introduction
Cocoa (
Currently, there are numerous combinations of agricultural by-products (such as coconut shell, 4 –7 palm kernel shell, 8 corn cob, 9 durian seed, 10 rice husk, 11 banana fiber, 12 rapeseed, 13 sunflower stalk, 14 and sunflower seed cake 15 ) and thermoplastic materials had been developed by researchers to produce biocomposites. Moreover, some of the biocomposites already marketed in Malaysia, like Melsom Biodegradable Enterprise made eco-friendly tableware from rice husk-based thermoplastic biocomposites material. 7 In our previous studies, coconut shell and corn cob was introduced to polylactic acid thermoplastic to produce ecopackaging and ecotableware materials. 6,9 Currently, the developments of polypropylene (PP)/CPH biocomposites have the potential to replace forest product, such as wooden fittings, fixtures, deck, and furniture. This will reduce the forest consumption in cutting trees and give benefit to environment.
Generally, the poor adhesion between natural filler and thermoplastic matrix due to the polarity difference are common issue among researchers and industries. Therefore, without coupling agents, biocomposites with good filler dispersion, filler–matrix adhesion, and mechanical strength are hard to achieve. Usually, maleated polymers (such as maleated polypropylene (MAPP) and maleated polyethylene (MAPE)) are used as coupling agents in the production of natural filler-based biocomposites. During compounding process at temperature above 170°C, maleated polymer is active and then the maleic anhydride group is chemically reacting with natural filler via esterification. 16 The long polymer chains covalently bond to the filler surface provide more efficient interfacial bonding with matrix. Thus, many researchers reported the biocomposites properties were remarkably improved by adding maleated polymer. 10,14 –21
Nowadays, there is no research on CPH-filled PP biocomposites. Against this background, the present research has been undertaken, with the aim to utilize CPH as filler in PP biocomposites. The present research is to investigate the effect of filler content and MAPP on processing torque, mechanical properties, thermal properties, and morphology of PP/CPH biocomposites.
Methodology
Materials
CPH was collected from cocoa plantations, Perak, Malaysia. First, the CPH was dried in oven at 80°C for 24 h. The dried CPH was crush into small pieces and further ground into fine powder. The CPH powder was sieved and average particle size of CPH powder was 22 µm, which was measured by Malvern Particle Size Analyzer Instrument (Italy). PP type co-polymer, grade SM 340 used as matrix, was supplied by Titan Petchem (M) Sdn. Bhd (Malaysia). MAPP used was obtained from Sigma Aldrich (Penang, Malaysia).
Melt compounding and molding procedures
The PP/CPH biocomposites with and without MAPP were compounded using Brabender® plastograph intermixer, Model EC PLUS (Germany) at a counterrotating mode of 180°C and rotor speed of 50 r min−1 (Table 1). The mixing procedures involved (i) PP transferred into mixing chamber for 3 min until it melted homogeneously and (ii) CPH was added to molten PP and mixed continuously for 5 min. The total time for compounding was 8 min. All the compounds were molded into 1 mm thickness sheet using hot press, model GT 7014A (Taiwan) at 180°C. The compression sequences involved (i) preheat compound for 4 min; (ii) compress under pressure of 100 kgf cm−2 for 1 min; and (iii) cool under the same pressure for 5 min. The PP/CPH biocomposites sheet was cut into tensile bar using dumbbell cutter with the dimension referring to ASTM D638 type IV.
Formulation of PP/CPH biocomposites.a
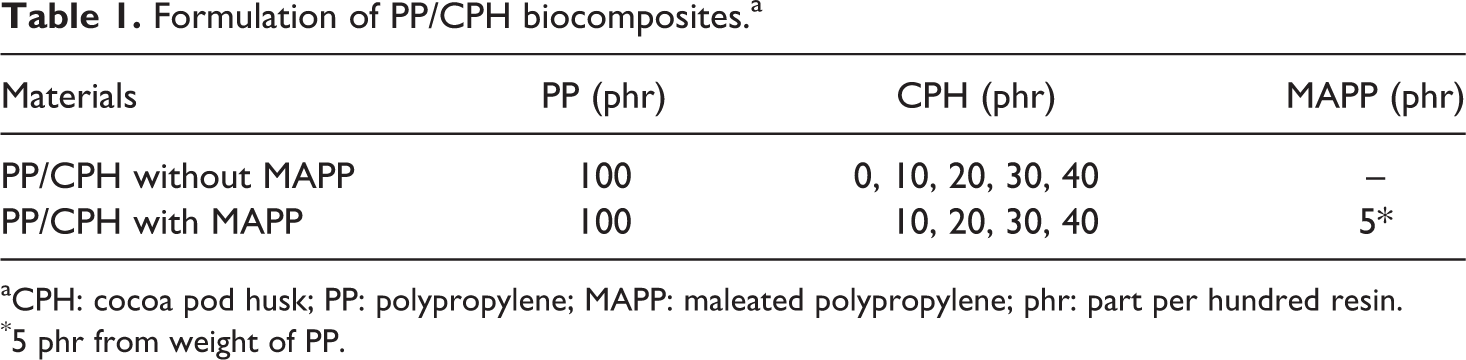
aCPH: cocoa pod husk; PP: polypropylene; MAPP: maleated polypropylene; phr: part per hundred resin.
*5 phr from weight of PP.
Processing torque measurement
The processing torque was measured during the compounding of PP/CPH biocomposites by using Brabender plastrograph internal mixer. The torque changes with time in biocomposites were recorded, and the torques versus time curves were plotted by computer. The torque values at the end of processing time were taken as stabilization torque.
Tensile testing
The tensile tests were carried out by Instron Universal Testing Machine, model 5569 (Massachusetts, USA). The load cell selected was 50 kN and the cross-head speed used was 30 mm min−1. The test was performed at 25 ± 2°C condition.
Morphological analysis
The tensile fracture surface of PP/CPH biocomposites were analyzed using SEM, model JEOL JSM-6460 LA (Japan). The samples were coated with a thin layer of palladium for conductive purpose and analyzed at 5 keV.
Fourier transform infrared (FTIR) spectroscopy
FTIR analysis of neat PP, PP/CPH biocomposites with and without MAPP was carried out by PerkinElmer Spectrum FTIR, Model Paragon 1000 (Germany) and the attenuated total reflectance (ATR) method was applied. The sample was recorded with 4 scans in the frequency range of 4000–650 cm−1 with a resolution of 4 cm−1.
Differential scanning calorimetry analysis
Differential scanning calorimetry (DSC) analysis was evaluated using DSC Q10, Research Instrument (California, USA). The sample was cut into small pieces and placed into closed aluminum pan with sample weight in range of 7 ± 2 mg. The specimen was heated from 30 to 200°C with a heating rate of 10°C min−1 under nitrogen atmosphere. The nitrogen gas flow rate was 50 ml min−1. The degree of crystallinity of biocomposite (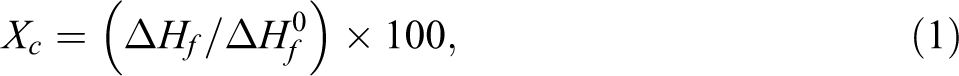
where
Thermogravimetric analysis
Thermogravimetric analysis (TGA) was carried out using TGA Pyris Diamond Perkin Elmer apparatus (California, USA). The sample was about 7 ± 2 mg in weight and was placed into platinum crucible. Then, the sample was heated from 30 to 700°C at a heating rate of 10°C min−1 under nitrogen atmospheric condition with the nitrogen flow rate of 50 ml min−1.
Results & discussion
Processing torque
The processing torque verses time curves for PP/CPH biocomposites with and without MAPP are shown in Figure 1. The first processing torque increased rapidly, while the PP pellets transferred into the mixing chamber. This was due to the shearing action from the solid PP pellets. The processing torque was reduced gradually with the change in viscosity as the PP pellets was melted at high temperature and under shearing. For PP/CPH biocomposites, a second processing torque was increased at time after 3 min. This is due to the fact that dispersive resistance from CPH particles increased the viscosity of PP matrix. Furthermore, the processing torque gradually decreased and achieved the stabilization torque after PP and CPH were homogenously mixed. This was a common trend that was also followed in our previous research and by other researchers. 22,23 Figure 2 shows the stabilization torque of PP/CPH biocomposites with and without MAPP. The stabilization torque of both biocomposites increased with increasing of CPH content. This was because the dispersed CPH particles in molten PP restricted the polymer chain mobility. Thus, the disperse resistance tend to rise the stabilization torque at higher filler content. Moreover, the presence of MAPP increased the stabilization torque of PP/CPH biocomposites. This might be the MAPP enhanced the dispersion and filler–matrix adhesion of PP/CPH biocomposites, which contributed to higher viscosity and resulting higher stabilization torque compared to PP/CPH biocomposites without MAPP.
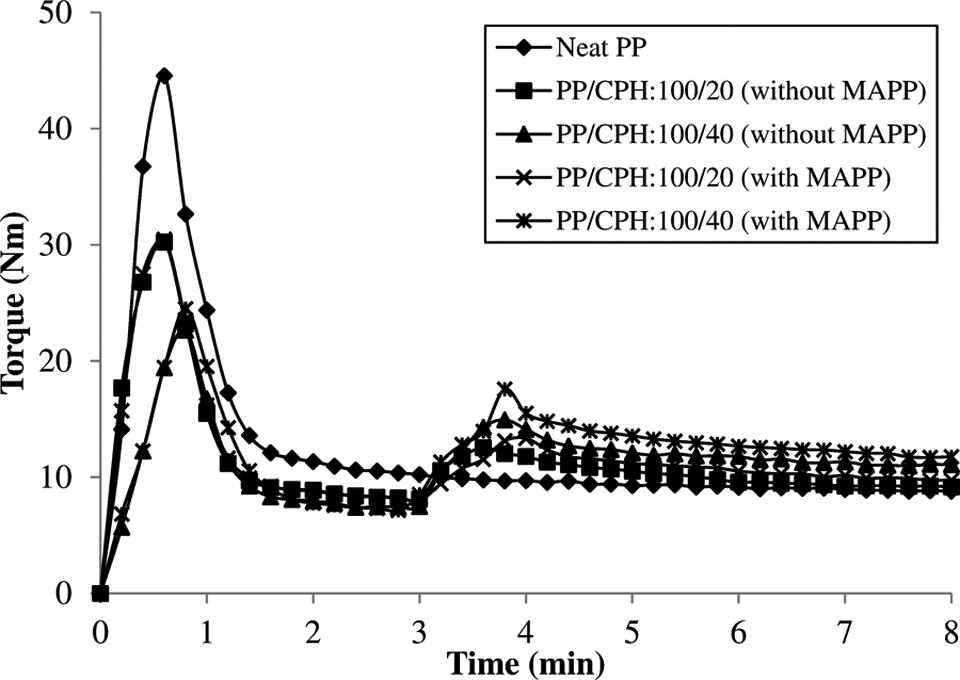
The torque versus time curves of PP/CPH biocomposites with and without MAPP at different filler contents. CPH: cocoa pod husk; PP: polypropylene; MAPP: maleated polypropylene.
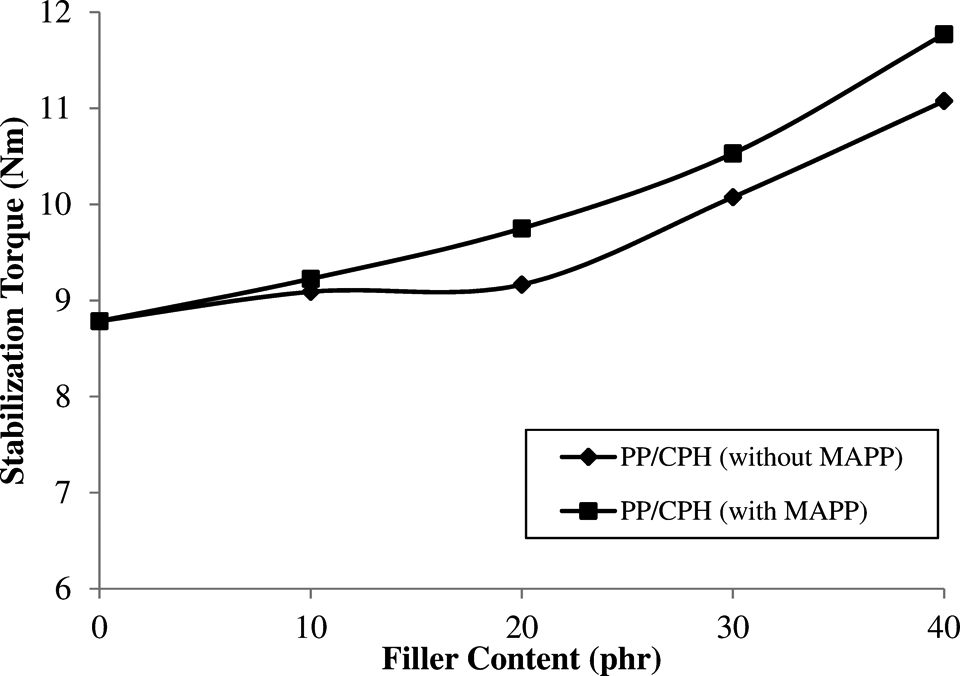
Stabilization torque of PP/CPH biocomposites with and without MAPP at different filler content. CPH: cocoa pod husk; PP: polypropylene; MAPP: maleated polypropylene.
Tensile properties
Figure 3(a) shows the effect of CPH content on tensile strength of PP/CPH biocomposites with and without MAPP. The tensile strength of both PP/CPH biocomposites decreased with increasing CPH content. The decrement in tensile strength might be attributed the low ability of stress transfer by CPH particle with low aspect ratio. Poor wet ability, filler dispersion, and interfacial bonding between hydrophilic CPH and hydrophobic PP matrix also contributed to a weak interfacial bonding. Therefore, the efficiency of stress transfer was reduced by the weak interfacial bonding between filler and matrix. The tensile strength of PP/CPH biocomposites with MAPP was higher than PP/CPH biocomposites without MAPP. However, the presence of MAPP in the biocomposites does not bring much change compared to neat PP. This was because the maleic anhydride group from MAPP reacted with hydroxyl group on the CPH filler surface via esterification. The PP chain from MAPP covalently bond on the CPH particles provided a better wettability and enhanced the filler–matrix interaction. This statement was agreed by many other researchers 10,14 –24
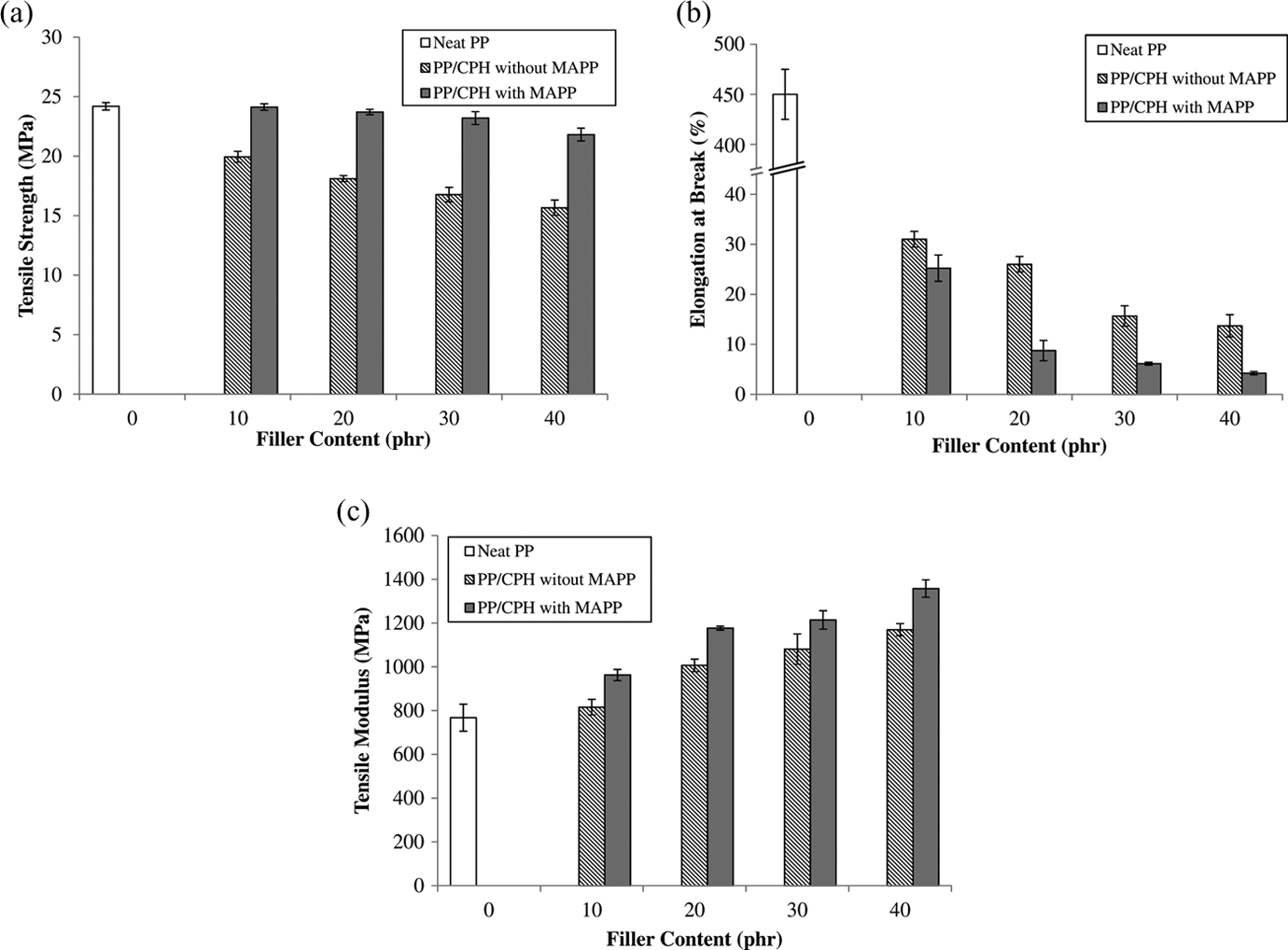
Effect of filler content on (a) tensile strength, (b) elongation at break, and (c) tensile modulus of PP/CPH biocomposites with and without MAPP. CPH: cocoa pod husk; PP: polypropylene; MAPP: maleated polypropylene.
The elongation at break of PP/CPH biocomposites with and without MAPP is shown in Figure 3(b). The result indicated the elongation at break of PP matrix was abruptly reduced by the addition of CPH. It is possible that the presence of rigid interface between CPH particle and PP matrix decreased the deformability of PP matrix. This led to more rigid and stiffer biocomposites. The PP biocomposites with coconut shell, 7 rattan, 22 rice husk, 23 wood flour, 24,25 and chitosan 26,27 also showed a similar trend on elongation at break. Furthermore, the PP/CPH biocomposites with MAPP exhibit lower elongation at break than PP/CPH biocomposites without MAPP. This was because the MAPP enhanced the interfacial interaction between CPH and PP matrix, and it generated a stronger interfacial bonding due to the reduction of molecular chain flexibility. This was a general observation that was also found by other researchers. 24,25
Figure 3(c) illustrated the tensile modulus of PP/CPH biocomposites with and without MAPP increased with the CPH content. Both biocomposites showed increased trend on tensile modulus because the CPH particles were rigid than the PP matrix. Therefore, the stiffness of biocomposites increased with the additional of CPH as expected. The increase in tensile modulus was also supported by the increased surface crystallization over bulk crystallization of PP matrix. The presence of CPH promoted transcrystalline formed around the filler surface. As a result, the crystalline region in PP matrix increased, and it might increase the tensile modulus of biocomposites. The tensile modulus of PP/CPH biocomposites was significantly higher with the addition of MAPP. The increase in tensile modulus of biocomposites was due to the improvement in interfacial bonding between the CPH and the PP matrix. A strong interfacial bonding also enhanced the nucleating effect of CPH on PP matrix, and it yields stiffer biocomposites. Some other researchers also found that the addition of natural filler increased the tensile modulus of PP biocomposites and the tensile modulus was further increased by MAPP. 10,14 –24
Morphological study
Figure 4(a) and (b) displays the SEM micrograph of tensile fracture surface of PP/CPH biocomposites without MAPP at 20 and 40 phr CPH content. The SEM micrographs show poor filler dispersion and agglomeration of CPH particles. This indicated the incompatibility between hydrophilic CPH and hydrophobic PP matrix. Figure 4(a) and (b) also shows fibrils, indicating plastic deformation of PP matrix. This was because of the poor filler–matrix interaction and the PP matrix can deform independently until CPH particles inherent in the deformation. The poor filler–matrix interaction also assigned to the holes present and detached CPH particle. In contrast, SEM micrographs of PP/CPH biocomposites with MAPP exhibit brittle fracture surface, and the CPH particles were embedded as well as coated by the PP matrix (as shown in Figure 5(a) and (b)). It can be explained as due to the incorporation of MAPP, which enhanced the interfacial interaction between the CPH particles and the PP matrix, increasing the stiffness of biocomposites and inducing brittle fracture behavior. The SEM micrographs also show the CPH particles were well dispersed and the agglomeration was not observed, because the MAPP improved the compatibility between CPH and PP matrix.

SEM micrograph of PP/CPH biocomposites without MAPP at (a) 20 phr and (b) 40 phr of CPH content. CPH: cocoa pod husk; PP: polypropylene; MAPP: maleated polypropylene.

SEM micrograph of PP/CPH biocomposites with MAPP at (a) 20 phr and (b) 40 phr of CPH content. CPH: cocoa pod husk; PP: polypropylene; MAPP: maleated polypropylene.
FTIR analysis
The FTIR spectrums of neat PP, PP/CPH biocomposites with and without MAPP are shown in Figure 6. The main characteristic peak of neat PP and PP/CPH biocomposites is listed in Table 2. The peaks at 3000–2800 cm−1 were contributed by C–H stretching vibrations in PP chains. The peaks found at 1457 and 1376 cm−1 were assigned to –CH2 and –CH3 bending vibration in PP. Moreover, 3 small peaks found at 1167, 998, and 973 cm–1 were due to –CH3 symmetric deformation vibration and –CH3 rocking vibration of PP. The broad peak of around 3300 cm−1 on PP/CPH biocomposites was attributed by –OH group from CPH. Furthermore, the absorption peak at 1601 cm−1 assigned to C=C stretching from hemicelluloses and the broad absorption peak at 1043 cm−1 was exhibited at the C–O–C and C–O groups from the main carbohydrates of cellulose and lignin. Regarding the FTIR spectrum, a new peak at 1737 cm−1 on PP/CPH biocomposites with MAPP was found. This evidenced the presence of ester linkage (C–O) between CPH and MAPP. The schematic reaction of MAPP and CPH is illustrated in Figure 7.
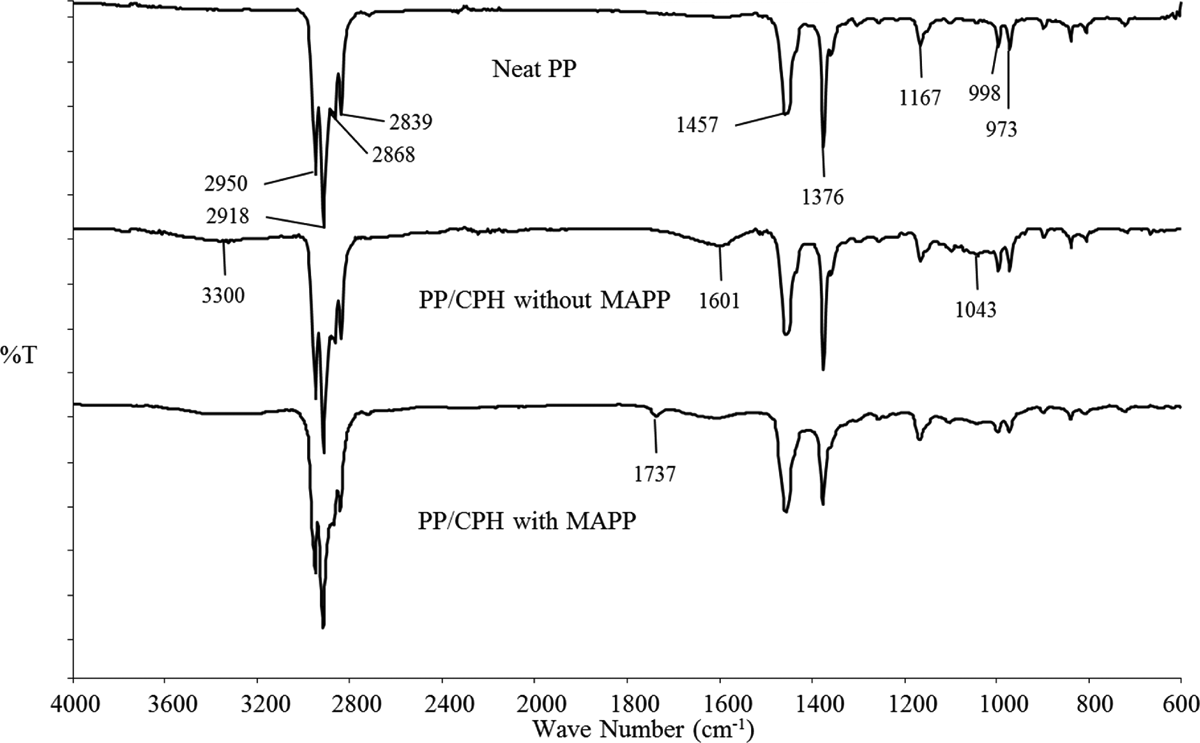
FTIR spectrums of neat PP, PP/CPH biocomposites with and without MAPP. CPH: cocoa pod husk; PP: polypropylene; MAPP: maleated polypropylene; FTIR: Fourier transform infrared.
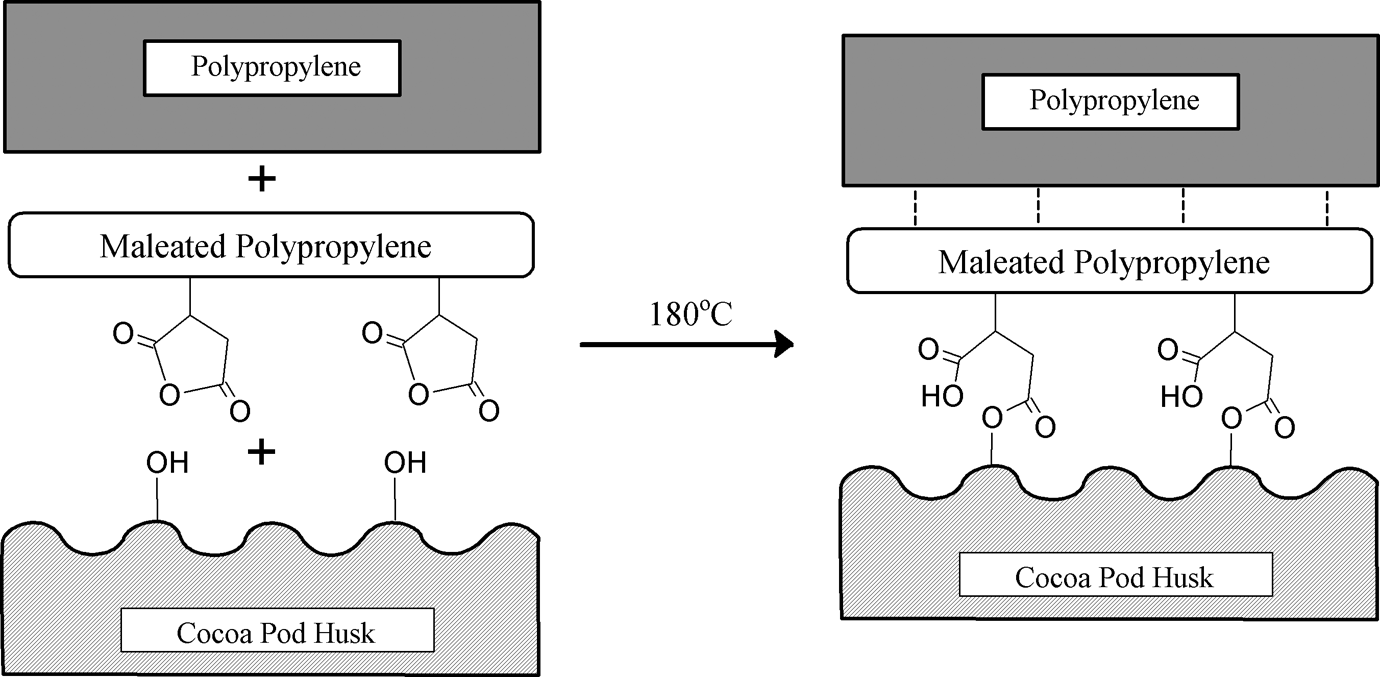
Schematic reaction between MAPP, CPH, and PP matrix. CPH: cocoa pod husk; PP: polypropylene; MAPP: maleated polypropylene.
Functional groups of neat PP and PP/CPH biocomposites.a
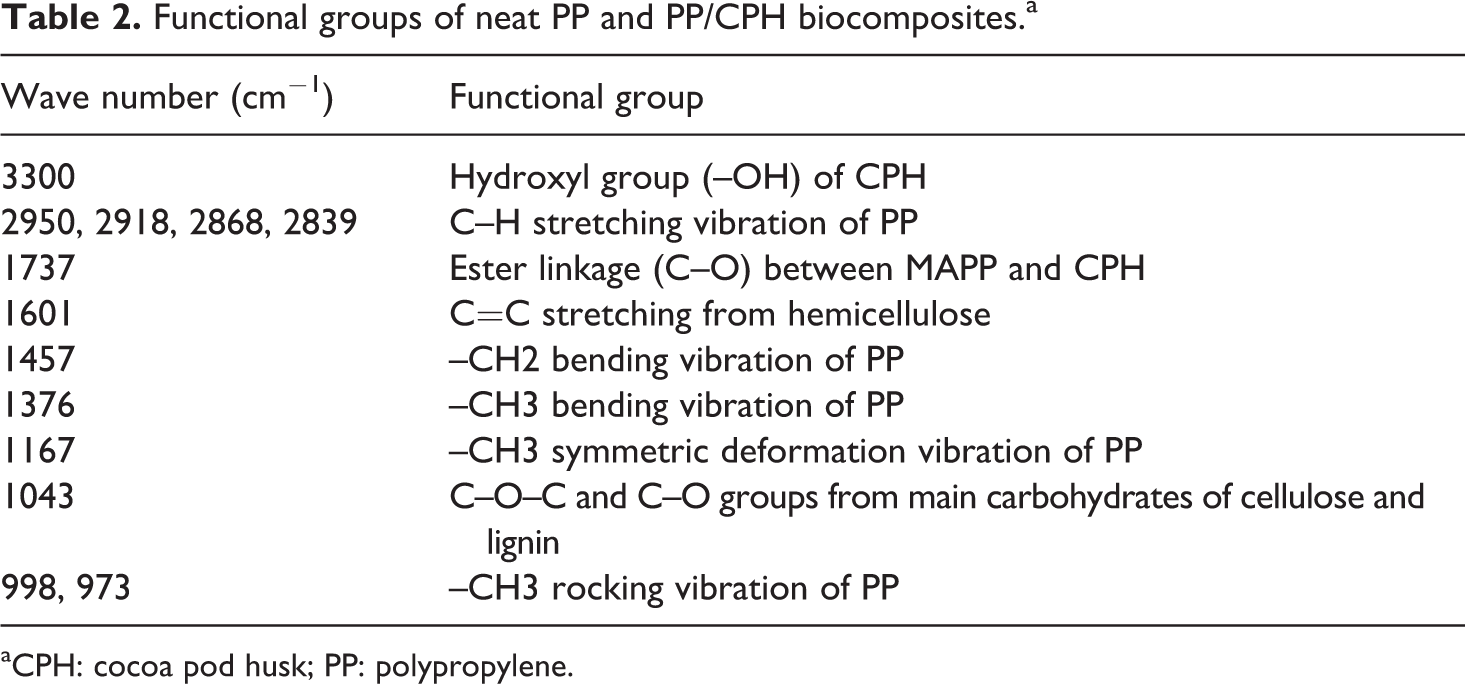
aCPH: cocoa pod husk; PP: polypropylene.
DSC properties
The DSC curves of neat PP, PP/CPH biocomposites with and without MAPP are illustrated in Figure 8. The DSC data of neat PP and both biocomposites were summarized in Table 3. The neat PP exhibited a melting temperature (
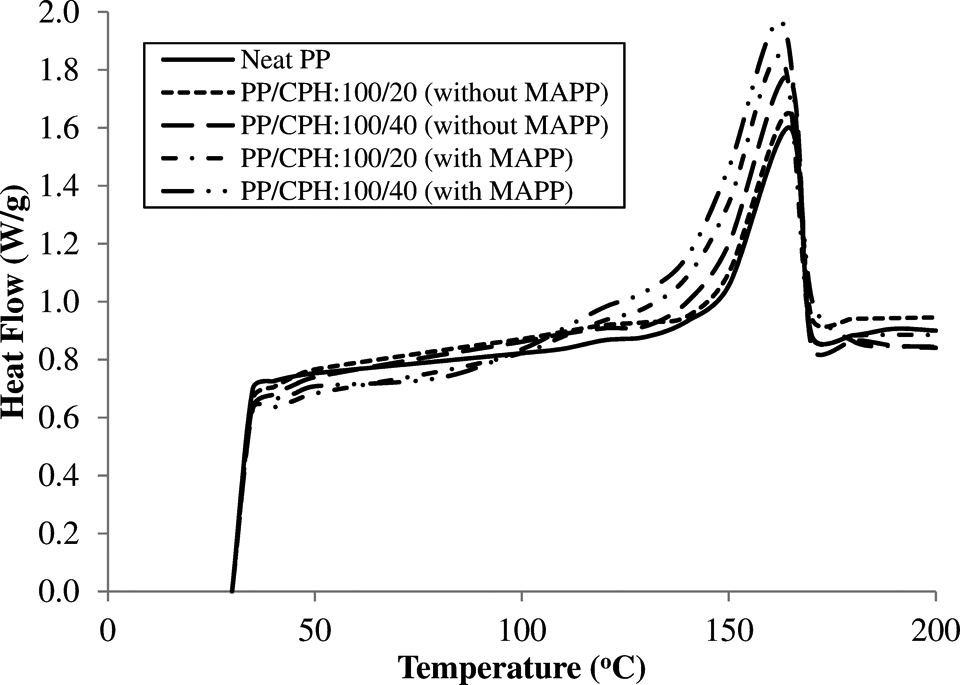
DSC curves of PP/CPH biocomposites with and without MAPP. CPH: cocoa pod husk; PP: polypropylene; MAPP: maleated polypropylene; DSC: differential scanning calorimetry.
DSC data of PP/CPH biocomposites with and without MAPP.a

aCPH: cocoa pod husk; PP: polypropylene; MAPP: maleated polypropylene;
Properties of TGA
The derivative thermogravimetry (DTG) and TGA curves of neat PP, PP/CPH biocomposites with and without MAPP are illustrated in Figure 9 and 10. The TGA data are summarized in Table 4. Figure 10 shows, the neat PP decomposed in single step at temperatures above 300°C. The CPH decomposed in 3 steps, included (i) evaporation of moisture in CPH at a temperature of 30–100°C; (ii) decomposition of hemicelluloses at a temperature of 200–350°C; and (iii) decomposition of lignin and cellulose at a temperature above 350°C. All PP/CPH biocomposites show an early thermal decomposition compared to neat PP as it can be observed through the temperature at 5% weight loss (
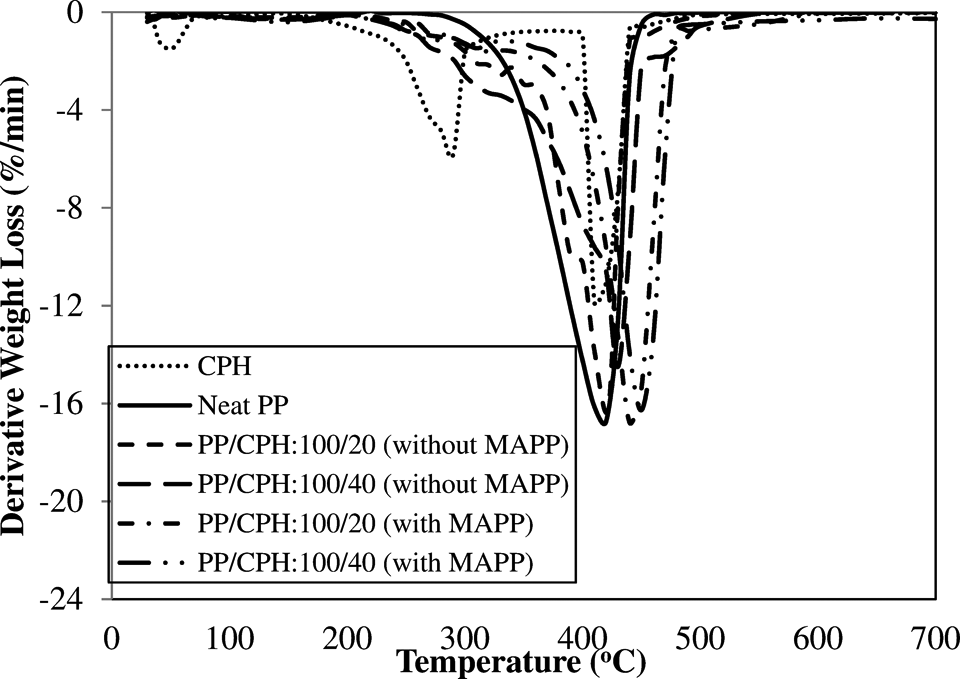
DTG curves of PP/CPH biocomposites with and without MAPP. CPH: cocoa pod husk; PP: polypropylene; MAPP: maleated polypropylene; DTG: derivative thermogravimetry.
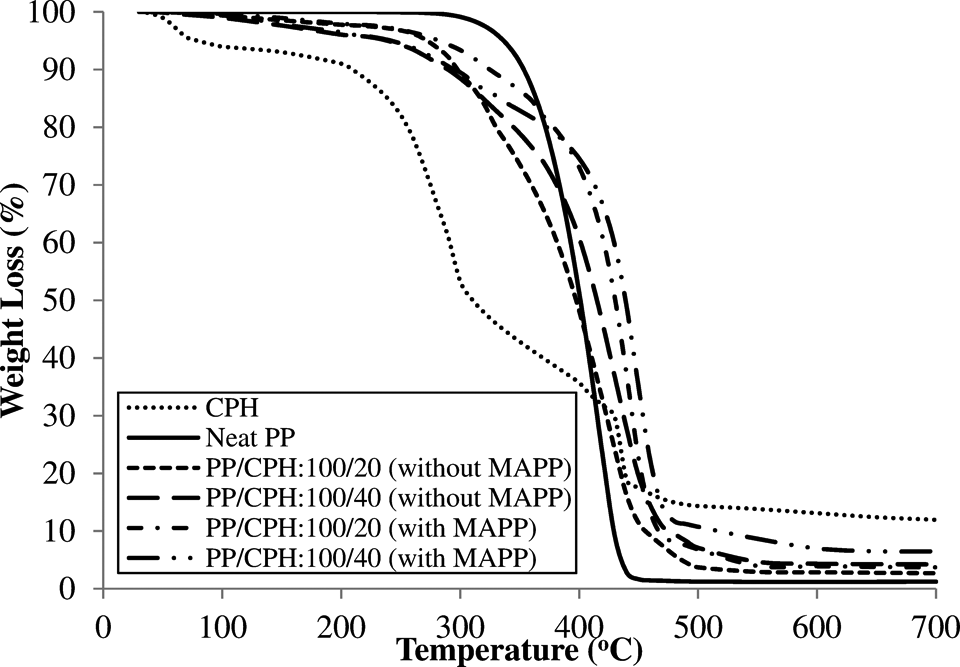
TGA curves of PP/CPH biocomposites with and without MAPP. CPH: cocoa pod husk; PP: polypropylene; MAPP: maleated polypropylene; TGA: thermogravimetric analysis.
TGA data of PP/CPH biocomposites with and without MAPP.a
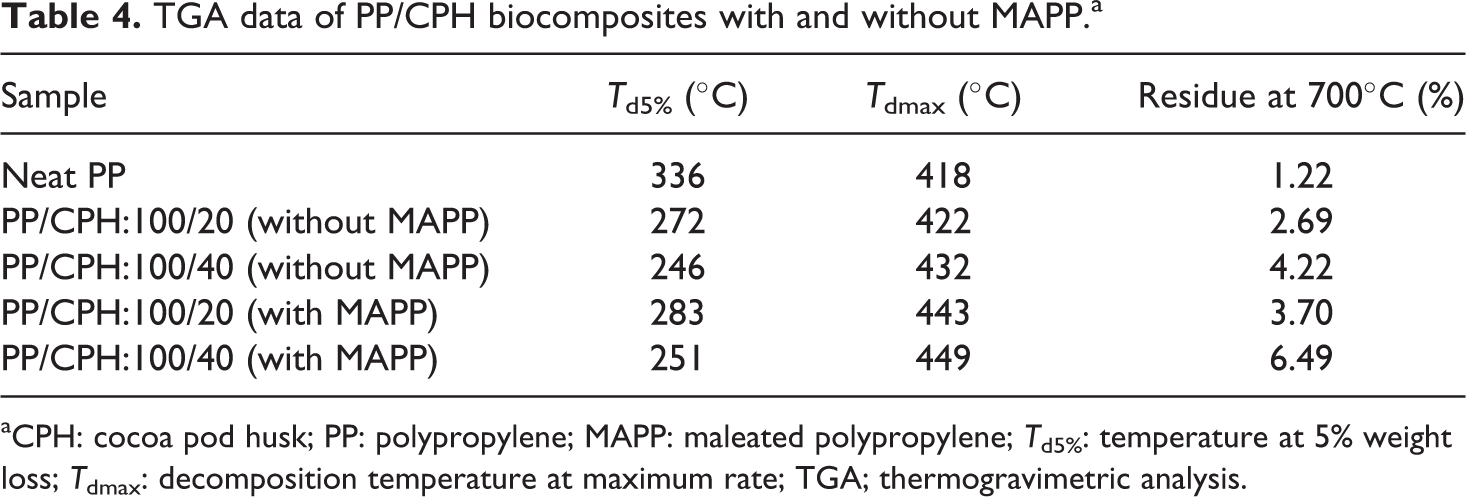
aCPH: cocoa pod husk; PP: polypropylene; MAPP: maleated polypropylene;
Conclusions
The following conclusions were drawn from the above studies: The processing torque of PP/CPH biocomposites increased with increasing CPH content. The torque of PP/CPH biocomposites with MAPP were higher compared to PP/CPH biocomposites without MAPP. The increase in CPH content decreased the tensile strength and the elongation at break of PP/CPH biocomposites but increased the tensile modulus. The addition of MAPP improved the tensile strength and tensile modulus of PP/CPH biocomposites. The PP/CPH biocomposites without MAPP exhibit poor filler dispersion, incompatibility and interfacial interaction between CPH and PP matrix. The SEM micrograph had proved the filler–matrix interaction of PP/CPH biocomposites was improved by MAPP. The crystallinity of PP matrix increased with the presence of CPH content, and it increased with a change in CPH content. However, the The PP/CPH biocomposites exhibit an early thermal decomposition, but they showed higher thermal stability at higher temperature. The thermal stability of PP/CPH biocomposites with MAPP improved as the
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
