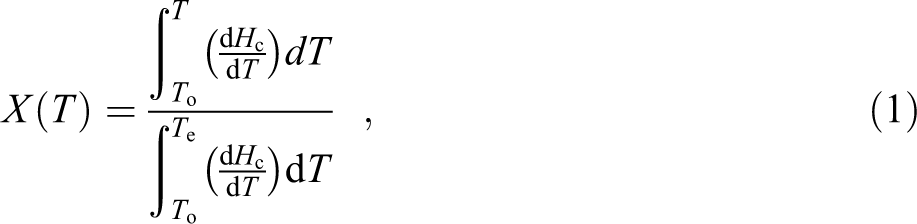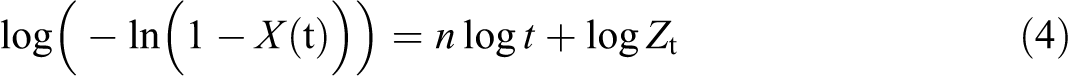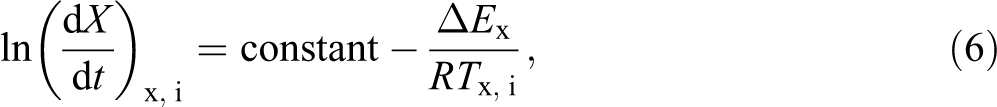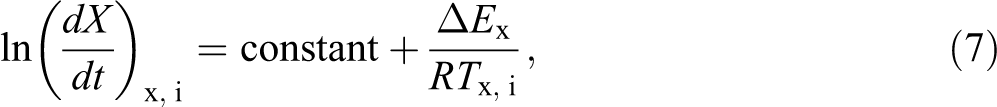Abstract
Kinetics of non-isothermal crystallization of isotactic polypropylene (iPP)/nucleating agent blends was studied for the nucleation mechanism. Avrami, Jeziorny, and correctional Friedman methods were applied to analyze the non-isothermal crystallization of the iPP/aryl heterocylic aluminum phosphate/sodium laurate (iPP/AHP-Al/L-Na) blends. For a given cooling rate of 10°C min−1, the onset temperature (To) and crystallization temperature (Tc) of the iPP/AHP-Al/L-Na blends increased by 4.19°C and 3.57°C, respectively, compared to iPP/L-Na. The To and Tc of iPP/AHP-Al/L-Na blends increased by 2.13°C and 1.79°C, respectively, compared to iPP/AHP-Al, when the AHP-Al mass fraction reached 1.2‰. The effective activation barrier (ΔE) for non-isothermal crystallization was the least when the AHP-Al mass fraction was 0.8‰. The value of the initial slope of the exotherm increased with the addition of AHP-Al monotonically. All these findings indicated that AHP-Al could improve the initial crystallization rate and also that the addition of AHP-Al/L-Na reduced the ΔE for non-isothermal crystallization. It is speculated that an ion exchange reaction between AHP-Al and L-Na occurs during the mixing process and could be possible that this results in the generation of a more efficient nucleating agent.
Keywords
Introduction
Polypropylene is a very important commodity polymer. Plastics used in automobiles, household appliances, and in construction industry are required to be stiff and tough, similar to engineering plastics. 1 However, crystallization is a critical phenomenon that affects the morphology and the properties of a semicrystalline polymer, like polypropylene. 2 In order to optimize the crystallization of polypropylene, numerous studies have been carried out. 3 –15 Polypropylene has been modified by many nucleating agents, such as talc, 12,16,17 sorbitol-based nucleating agents, 18 –20 and organic acid derivatives. 21 –23 Moreover the addition of a nucleating agent can significantly accelerate crystallization rate, shorten the processing cycle, and improve the properties of the material.
Xin et al. 24 studied the synergistic effects of aluminum organic phosphate and sodium carboxylate composite nucleating agents on the improvement of the mechanical and optical properties of isotactic polypropylene (iPP). They found that the performances of the composite nucleating agents were better than any of the individual ingredients when the mass fraction of aluminum organic phosphate in the composite was 60%. In addition, the mechanism of the organic phosphate and alkyl carboxylic acid salt were studied and applied widely. 25 –30 However, the synergistic effects via the non-isothermal crystallization kinetics method have been rarely investigated.
In this article, the non-isothermal crystallization kinetics of iPP/aryl heterocylic aluminum phosphate (AHP-Al)/sodium laurate (L-Na) blends were investigated by differential scanning calorimetry (DSC) to give an insight into the nucleation mechanism of a composite nucleating agent.
Theoretical background
In DSC, the energy released during a non-isothermal crystallization process is a function of temperature. Hence, the relative crystallinity as a function of temperature (X(T)) can be formulated as:
where, To, Te, and T represent the onset, end, and an arbitrary temperature, respectively. dHc is the enthalpy of crystallization released during an infinitesimal temperature range (dT) and the denominator represents the total enthalpy of crystallization for a specific cooling condition.
In order to use equation (1) for analyzing the non-isothermal crystallization data obtained by DSC, it is assumed that both, the sample and the DSC furnace, have the same temperature. This can only be realized when the difference between the temperatures of the sample and the furnace is minimal. If this condition is valid, the relation between the crystallization time t and the sample temperature T can be written as:
where, T0 is an initial temperature and D is the cooling rate. According to equation (2), the horizontal temperature axis observed in a DSC thermogram for non-isothermal crystallization data can then be transformed into time domain.
Avrami’s equation is widely applied for non-isothermal crystallization of polymers. 31,32 It assumes that the relative degree of crystallinity develops with the crystallization time t,
where, exponent n is a mechanism constant, which depends on the type of nucleation and growth process, Xt is the relative degree of crystallinity at time t, and Zt is a composite rate constant involving both nucleation and growth rate parameters. Taking logarithms on both sides of equation (3) gives the following equation:
Considering the non-isothermal nature of the process investigated, Jeziorny pointed out that the value of rate parameter Zt should be adequately corrected. 33 Assuming constant or approximately constant D, the final form of the parameter characterizing the kinetics of equation for non-isothermal crystallization is as follows:
Apart from the parameter Zc, it is also interesting to evaluate the effective energy barrier, ΔE, for the non-isothermal crystallization process of polymer. The differential isoconversional method of Friedman 4,34 is one of the most appropriate methods for evaluating the effective energy barrier:
where, dX/dt is the instantaneous crystallization rate as a function of time for a given conversion X. According to this method, the Xt function, obtained from the integration of the experimentally measured crystallization rates, is initially differentiated with respect to time, in order to obtain the instantaneous crystallization rate, dX/dt. Referring to equation (2), equation (6) can be also written as following:
where, dT is the infinitesimal temperature range.
Experimental
Materials
iPP T30S, a commercial product of Lanzhou Petroleum Chemical (China) with a tacticity of 98% and a melt flow rate of 2.6 g/10 min, was used in the study. The number- and weight-average molecular weights of this resin were 211 and 734 kg mol−1, respectively. Industrial-grade L-Na was provided by Shanghai Ao keep industrial Co., LTD (China). AHP-Al was prepared according to our previously published paper. 35 Anhydrous ethanol, aluminum chloride, and sodium chloride were all of analytical grade and were used as received.
Composite nucleating agent
AHP-Al and L-Na were used to prepare the composite nucleating agent according to the formula in Table 1.
Formula of the composite nucleating agent.
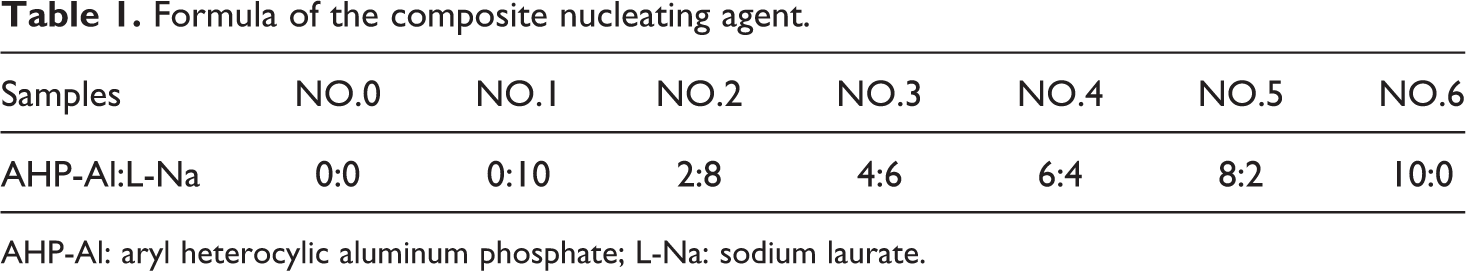
AHP-Al: aryl heterocylic aluminum phosphate; L-Na: sodium laurate.
Preparation of iPP/AHP-Al/L-Na blends
IPP/AHP-Al/L-Na blends with mass fraction of the composite nucleating agent (AHP-Al/L-Na) of 0.2% were mixed together and extruded from a corotating twin-screw extruder, having a length–diameter of 20. The barrel temperatures were maintained at 175, 180, 185, 200, 210, and 220°C from the hopper to the die, and the screw speed was set at 203 r min−1. After pelletizing and drying, the extruded iPP/composite nucleating agent blends were dried at above 100°C for minimum of 12 h.
DSC analysis
The non-isothermal crystallization behaviors of the samples were investigated by DSC (TA Instruments, Q10; New Castle, Delaware, USA). The experiments were carried out using 5–7 mg of each sample, sealed in aluminum pans under dry ultrapure nitrogen with a flow rate of 20 mL min−1. Samples were first heated from 40°C to 210°C at a rate of 30°C min−1 and held at this temperature for 5 min to erase any previous thermal history. They were then cooled to 70°C at different rates (D = 5, 10, 15, 20°C min−1) and the endothermic curves were recorded.
Results and discussion
Non-isothermal crystallization behavior
Figure 1 shows typical DSC curves for heat flow as a function of temperature at a cooling rate of 10°C min−1 for virgin iPP and iPP/AHP-Al/L-Na blends. It can be seen from Figure 1 that virgin iPP has a single peak at about 116.8°C, whereas the peak in iPP/AHP-Al/L-Na blends appears at around 124.6°C or above. Distinctly, both AHP-Al and L-Na play the role of a nucleating agent. However, the nucleation ability of L-Na is less compared to that of AHP-Al, with regard to the To and crystallization peak temperature (Tc), which are shown in Figure 2. Obviously, both To and Tc of iPP/AHP-Al/L-Na blend were the highest when the AHP-Al mass fraction was 1.2‰. Moreover, the To and Tc increased by 4.19°C and 3.57°C, respectively, compared to iPP/L-Na. Additionally, the To and Tc increased by 2.13°C and 1.79°C, respectively, compared to iPP/AHP-Al. This demonstrates the role of AHP-Al and L-Na in synergistic nucleation of iPP. It can be speculated that the ion exchange reaction between AHP-Al and L-Na probably occurs during the mixing process, and it is likely that a more efficient nucleating agent is generated.
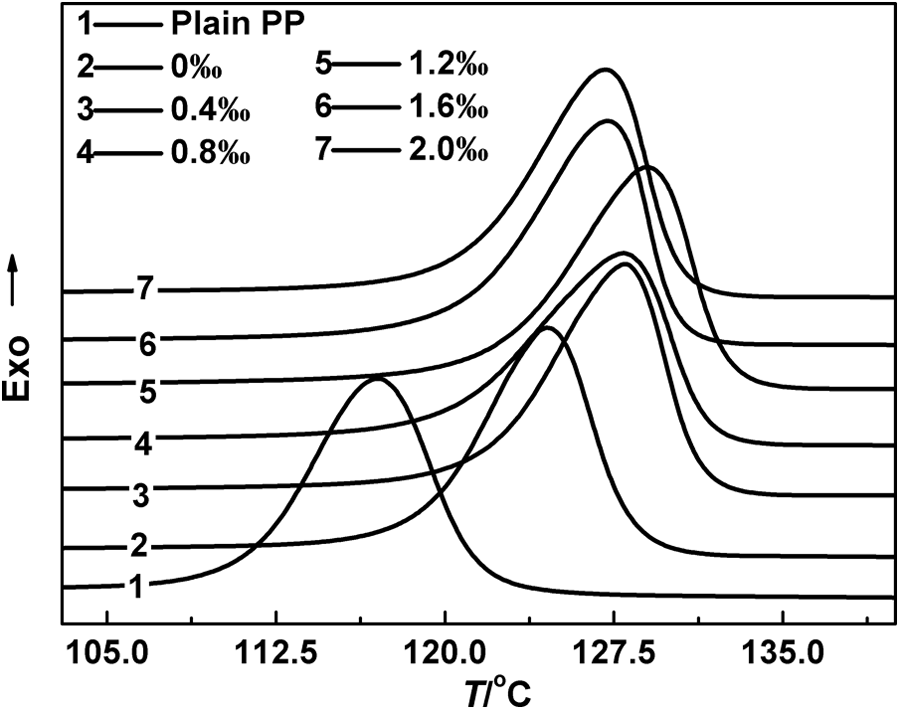
DSC curves of non-isothermal crystallization for the plain iPP and iPP/AHP-Al/L-Na blends at the cooling rate of 10°C min−1. DSC: differential scanning calorimetry; iPP: isotactic polypropylene; AHP-Al: aryl heterocylic aluminum phosphate; L-Na: sodium laurate.
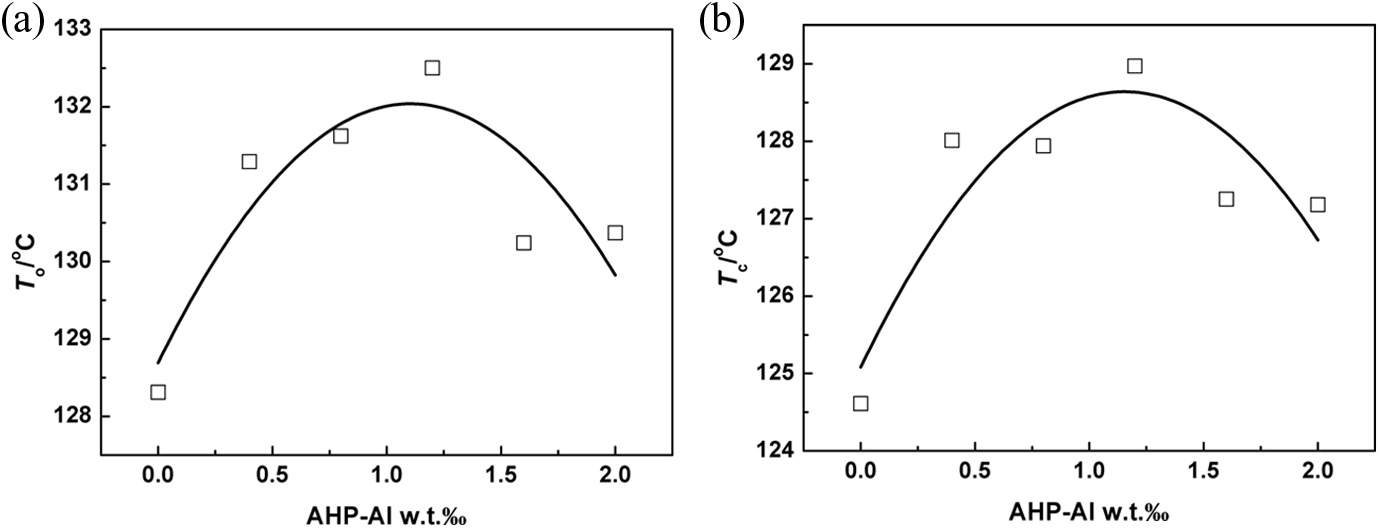
The To and Tc versus the mass fraction of AHP-Al at the cooling rate of 10°C min−1. To: onset crystallization temperature; Tc: crystallization peak temperature; AHP-Al: aryl heterocylic aluminum phosphate.
Figure 3 shows the schematic representation of the method for determination of various parameters from the DSC crystallization exotherm peak. 36 Initial slope of the exotherm (Si) is the slope of the high temperature side of the exotherm. The value of Si is an indication of the rate of nucleation. Figure 4 shows that addition of AHP-Al increases the Si of the crystallization exotherm of iPP. In this way, AHP-Al plays an important role in improving the rate of nucleation, which can be explained by the kinetics of the nucleation of iPP with AHP-Al/L-Na.
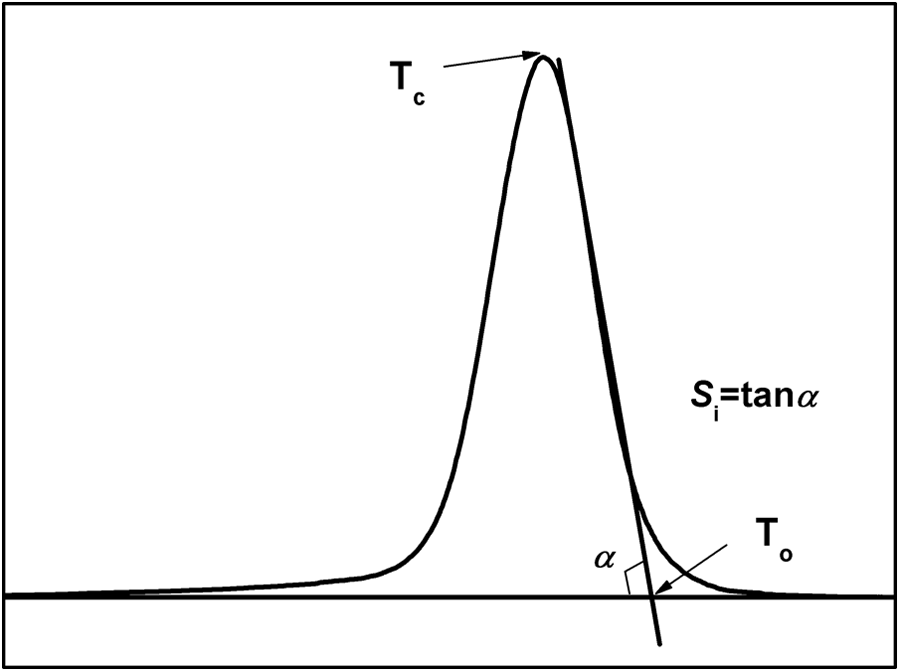
Schematic representation of the method of determination of the various parameters from DSC crystallization exotherm peak. DSC: differential scanning calorimetry.
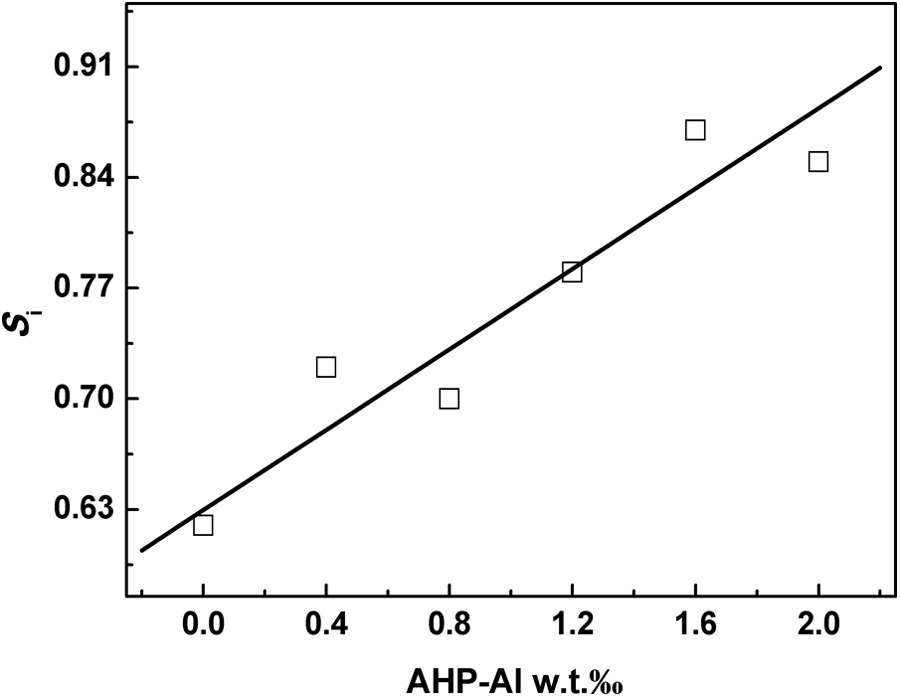
The Si versus the mass fraction of AHP-Al at the cooling rate of 10°C min−1. Si: initial slope of the exotherm; AHP-Al: aryl heterocylic aluminum phosphate.
Figure 5 shows the crystallization time t versus mass fraction of AHP-Al for different relative crystallinities at the cooling rate of 10°C min−1. It can be seen that the crystallization time t does not change to a greater extent or regularly with the addition of AHP-Al, which implies that the crystallization time for virgin iPP and iPP/AHP-Al/L-Na blends at different relative crystallinities is not affected by the addition of AHP-Al/L-Na.
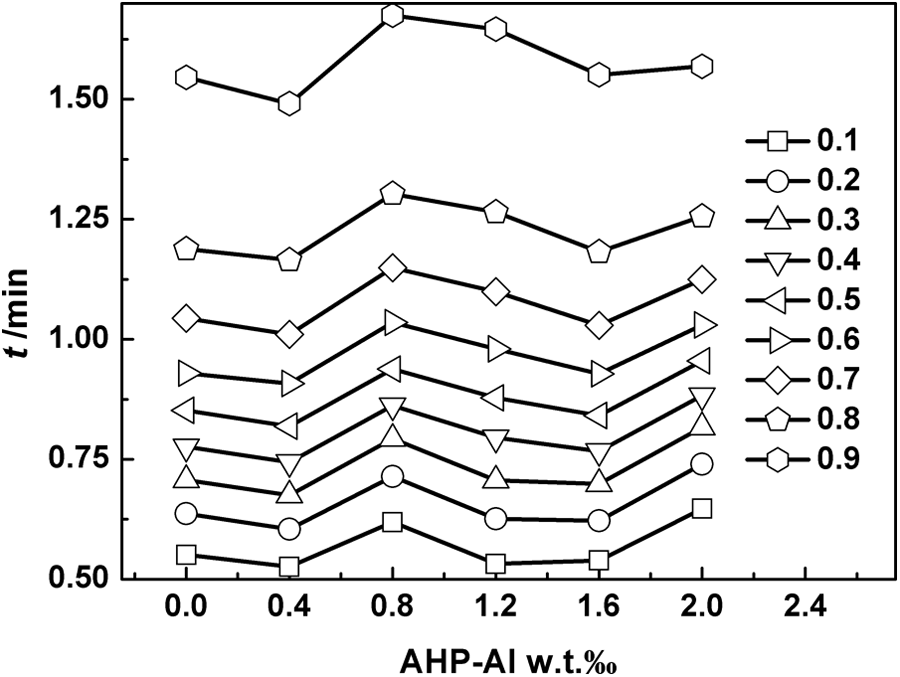
The crystalline time t versus the mass fraction of AHP-Al for different relative crystallinity at the cooling rate of 10°C min−1. AHP-Al: aryl heterocylic aluminum phosphate.
Non-isothermal analysis of crystallization kinetics by Jeziorny method
The plots of log(−ln(1 − Xt)) versus log t, using equation (4), are shown in Figure 6. It shows straight lines for all samples (correlation coefficient R > 0.98) at a cooling rate of 10°C min−1. The values for n, log Zt, and Zc, determined from the slope and intercept of the initial linear portion in Figure 6, are listed in Table 2. The values of the Avrami exponent for virgin iPP and iPP/AHP-Al/L-Na blends are 4.3, 3.4, 2.7, 2.6, 2.8, 2.8, and 2.8, respectively. The values of the rate parameter Zc increase with an increase in the addition of AHP-Al. This implies the crystallization rate of blends is accelerated by addition of AHP-Al. The results are in accordance with the analyses of Si, and both results support the crystallization kinetics involved in the nucleation of iPP with AHP-Al/L-Na.
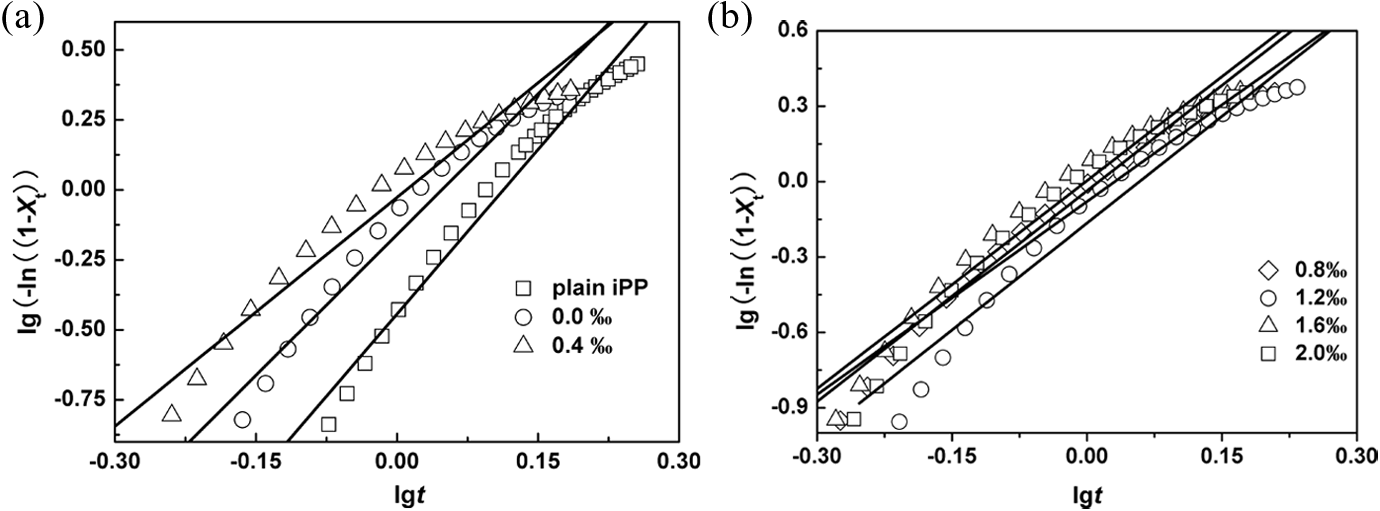
Plots of log(−ln(1 − X(t))) versus log t for non-isothermal crystallization of samples at the cooling rate of 10°C min−1.
Results of the Avrami method and Jeziorny method analysis for non-isothermal crystallization of plain iPP and iPP/AHP-Al/L-Na blends at the cooling rate of 10°C min−1.
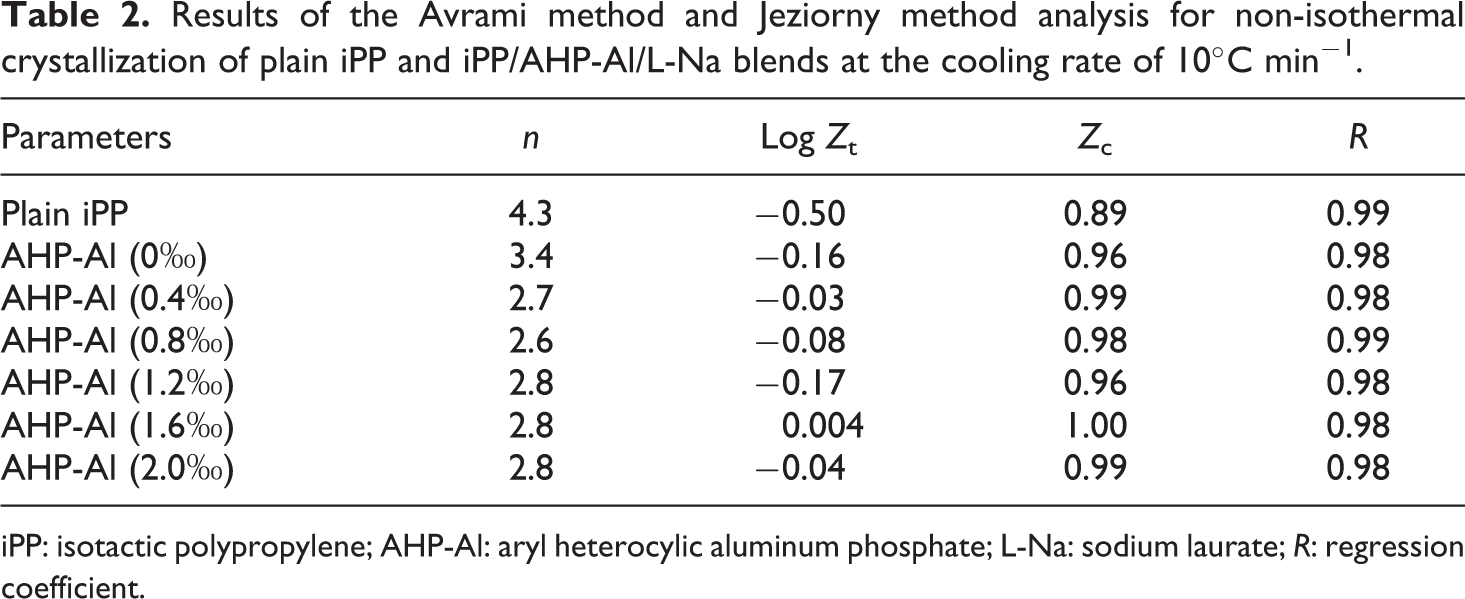
iPP: isotactic polypropylene; AHP-Al: aryl heterocylic aluminum phosphate; L-Na: sodium laurate; R: regression coefficient.
Effective energy barrier of non-isothermal crystallization
Plots of ln(dX/dt)x,i versus 1/(RTx,i) for all iPP/AHP-Al/L-Na blends are shown in Figure 7. It can be seen that straight lines are obtained from each and every sample (correlation coefficient > 0.98). The values of ΔE for virgin iPP and the blends can be calculated from the slopes of these lines. It can be seen from Figure 8 that the ΔE value of the iPP/AHP-Al/L-Na blend with mass fraction of AHP-Al of 0.8‰ is less than the other iPP/AHP-Al/L-Na blend with relative crystallinities of 0.2, 0.4, and 0.6 at a cooling rate of 10°C min−1, which are in turn, lesser compared to virgin iPP (16,354, 11,934, and 9122 J mol−1). These results suggest a possible ion exchange reaction between AHP-Al and L-Na occurring during the mixing process, with the possibility of a more efficient nucleating agent being generated. Therefore, AHP-Al and L-Na synergistically affect the nucleation of iPP, which kinetically proves the mechanism of nucleation of iPP with AHP-Al and L-Na.
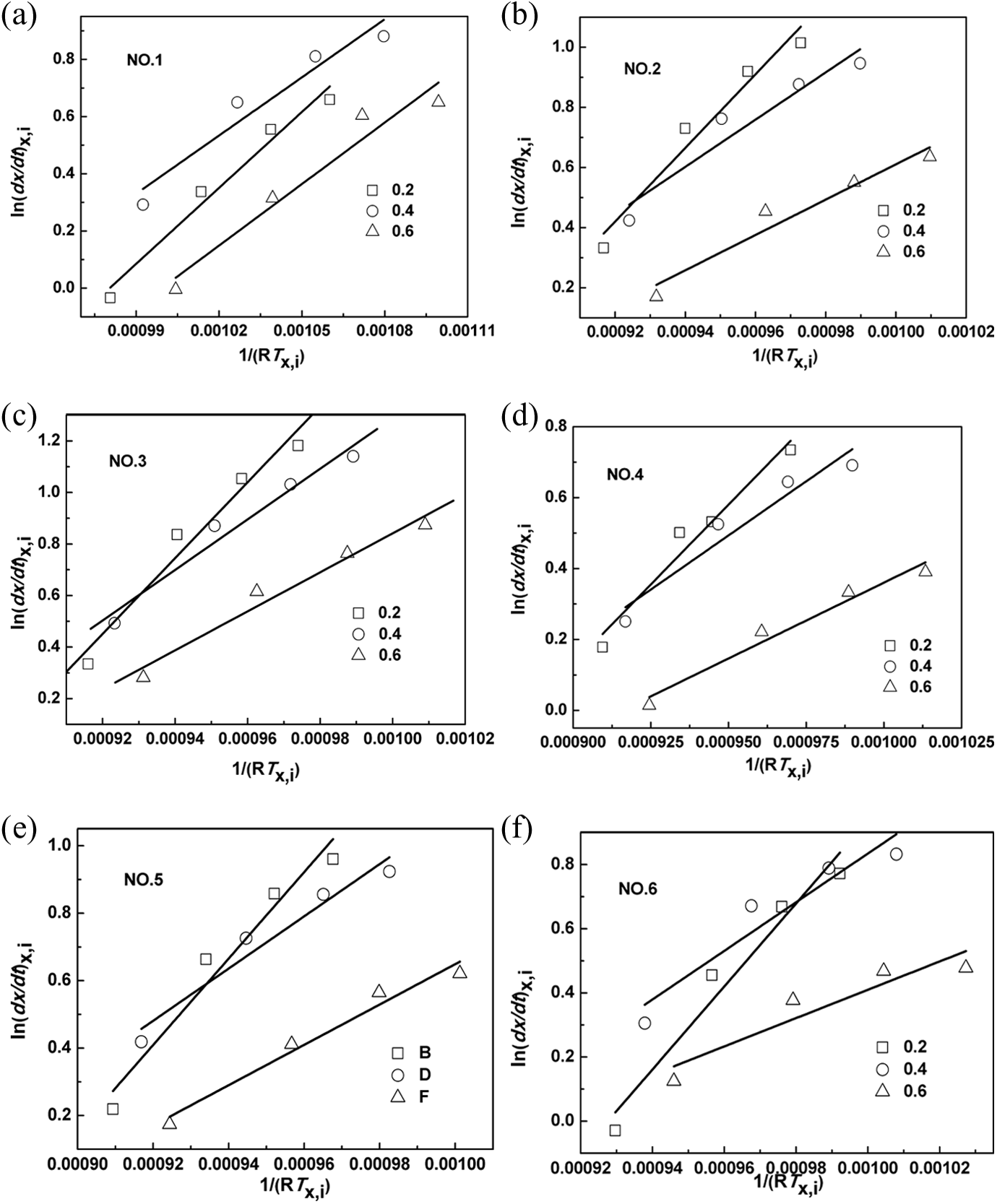
Plots of ln(dX/dT)x,i versus 1/(RTx,i) for non-isothermal crystallization of samples at the cooling rate of 10°C min−1.
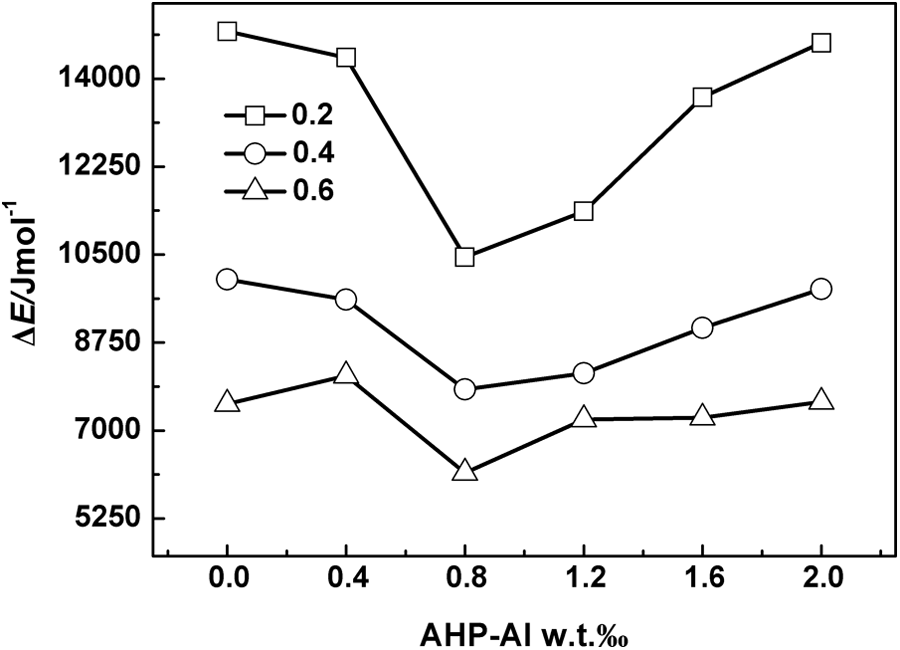
The effective energy barrier (ΔE) versus the mass fraction of AHP-Al for different relative crystallinity at the cooling rate of 10°C min−1. AHP-Al: aryl heterocylic aluminum phosphate.
It should be noted that this study has only examined the non-isothermal crystallization kinetics of iPP/AHP-Al/L-Na blends. Other properties such as nucleating optical pictures for blends and the characteristic properties of blends are reported in the other article. 37 Notwithstanding its limitation, this study does suggest that the non-isothermal crystallization of iPP has improved with the addition of composite nucleating agent to it. It is a great significance for us and others in researching the mechanism of iPP and crystallization of other polymers nucleated by composite nucleating agent.
Conclusions
The non-isothermal crystallization kinetics of iPP/AHP-Al/L-Na blends were determined by DSC. The Avrami equation, Jeziorny method, and a correctional Friedman method were successfully employed to describe the non-isothermal crystallization behavior of iPP/AHP-Al/L-Na blends. The Tc of iPP/AHP-Al/L-Na blends with AHP-Al fraction of 0.12‰ was found to be the highest, indicating that the crystallization rate was the highest with this proportion of the composite nucleating agent. Particularly, the value of Si increases monotonically with the addition of AHP-Al, thereby proving the role of AHP-Al in improving the nucleation rate. Finally, the analysis of the effective energy barrier of crystallization of iPP/AHP-Al/L-Na blends showed that the ΔE value of iPP/AHP-Al/L-Na blends reduces with an increase in the content of AHP-Al. With an AHP-Al proportion of 0.8‰, the value of ΔE was found to be the least.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is financially supported by International S&T Cooperation Program of Guizhou Province ([2012]7029).