Abstract
Poly(lactic acid)/halloysite nanoclay (PLA/HNC) nanocomposites with N,N′-ethylenebis(stearamide) (EBS) were produced by melt mixing. Water absorption behaviors of the PLA nanocomposites were studied at three different temperatures, that is, 30, 40, and 50°C. The water absorption kinetics of PLA/HNC nanocomposites conform to Fickian diffusion behavior at immersion temperatures of 30 and 40°C due to the diffusional exponent (n) values that were close to 0.5 for all specimens. However, the hydrolysis of PLA occurred for longer time deviations at 50°C. Activation energy of water diffusion (E a) for PLA nanocomposites were found to be affected by the HNC and EBS contents. The glass transition temperature (T g), cold crystallization temperature (T cc), and melting temperature (T m) of the PLA sample were shifted to lower temperature after subjected to immersion temperature of 50°C. The carbonyl index of all PLA specimens increased after water absorption at 40 and 50°C due to the formation of higher amount of carboxylic acid end groups during the hydrolysis process.
Introduction
Poly(lactic acid) (PLA) that can be produced from renewable resources has recently gained growing attention due to its biodegradability, biocompatibility, transparency, high modulus, and strength. PLA promises to be nearly carbon dioxide neutral and reduces dependency on oil for the production of polymeric materials. The industrial use of PLA is targeted toward the food packaging, automotive, and medical application. 1 –4 However, there is some factors limiting the application of PLA, such as low toughness, low temperature resistance, and low moisture resistance.
Polymer nanocomposites have received attention in recent years attributed to their improved mechanical, thermal, and barrier properties. Biodegradable composites of PLA with montmorillonite nanoclay, silica, kaolinite, bentonite, hectorite, nano-calcium carbonate, graphene, carbon nanotube, titanium dioxide, halloysite nanotube were prepared and studied recently. 5 –19
Halloysite nanoclay (HNC) is a type of mineral clay with the chemical composition Al2(OH)4Si2O5(2H2O). In general, HNC has a hollow, nanotubular shape with high aspect ratio. HNC can be used in various applications, for example, in biomimetic nanoreactors, drug delivery systems, and as reinforcing fillers for polymeric materials. It has been documented that the addition of HNC improved the mechanical and thermal properties of poly(vinyl alcohol), poly(vinyl chloride), polyamide, polystyrene, polypropylene, and PLA. 20 –26 In our previous work, it was found that the addition of HNC improved the thermal stability of PLA but reduced the impact strength of the resultant composites. Thus, N,N′-ethylenebis(stearamide) (EBS) was introduced into PLA/HNC nanocomposites, and it was found that the impact strength of PLA/HNC increased significantly with the addition of EBS.
Similar to other aliphatic polyesters, PLA is sensitive to hydrolysis in the presence of water. The nature of hydrolytic degradation of PLA is due the diffusion of soluble oligomers from the surface. Consequently, it will lead to a higher chain mobility (via plasticization) and chain scissions, followed by fast degradation. It is generally known that combination of hydrolysis (due to water) and heat (due to temperature) could affect mechanical and physical properties of a polymeric material. For a biodegradable polymer (e.g. PLA), hydrolysis could increase the biodegradation rate. However, this kind of hydrolysis process needs to be controlled during the service life of a PLA product, especially for the one that may contact with moisture and water.
Leu and Chow 8 reported that the water absorption of neat PLA leveled at approximately 1%. The hydrolytic degradation process was strongly dependent on the time and temperature of the medium, especially the temperature of the medium was above the glass transition temperature (T g) of a polymer. 27 The hydrolysis of PLA occurs at the ester linkages and produces lactic acid oligomers. The formation of lactic acid oligomers increases the carboxylic acid end groups in medium and subsequently accelerates the hydrolytic degradation reaction. 28,29 Roy et al. 27 reported that the hydrolysis of PLA/montmorillonite and PLA/HNC proceeds at slower rate during prolonged hydrolysis compared to neat PLA at initial stages. It was revealed that the incorporation of clays could provide higher water barrier and reduce the hydrolysis rate for PLA composites.
In this study, water absorption behaviors of PLA/HNC nanocomposites were investigated at three different temperatures, that is, 30, 40, 50°C. Note that the selected immersion temperature is below the T g of PLA. The equilibrium water absorption (M m), diffusion coefficient (D), diffusional exponent (n), and activation energy (E a) of the composites were determined. The impact strength and molecular weight changes of the PLA composites subjected to water absorption are reported. The thermal properties and functional groups changes of PLA nanocomposites were analyzed using differential scanning calorimetric (DSC) and Fourier transform infrared (FTIR) techniques.
Experimental
Materials
PLA (Ingeo™ 3051D, NatureWorks LLC®, Blair, Nebraska, USA) was selected in this study. The specific gravity and melt flow index of the PLA were 1.25 and 25 g/10 min (2.16 kg load, 210°C). HNC was supplied by Sigma-Aldrich (Malaysia). The diameter of the HNC was in the range of 30–70 nm, while its length was 1–3 µm. EBS with particle size approximately 840 µm and melting point of 140°C was supplied by Sigma-Aldrich (St Louis, Missouri, USA).
Preparation of PLA/HNC nanocomposites
Melt compounding was carried out using an internal mixer (Haake PolyDrive R600, Germany) at 180°C for 10 min. The rotor speed was set at 50 r min−1. Prior to compounding, PLA, EBS, and HNC were dehumidified in a vacuum oven at 80°C for 24 h. The compression molding was performed at 185°C using a hot press machine (Go Tech, Taiwan). The preheating, compression molding, and cooling times were 7 min, 3 min, and 3 min, respectively. Table 1 shows the materials designations and compositions of the PLA/HNC composites.
Materials designations and compositions of PLA nanocomposites.
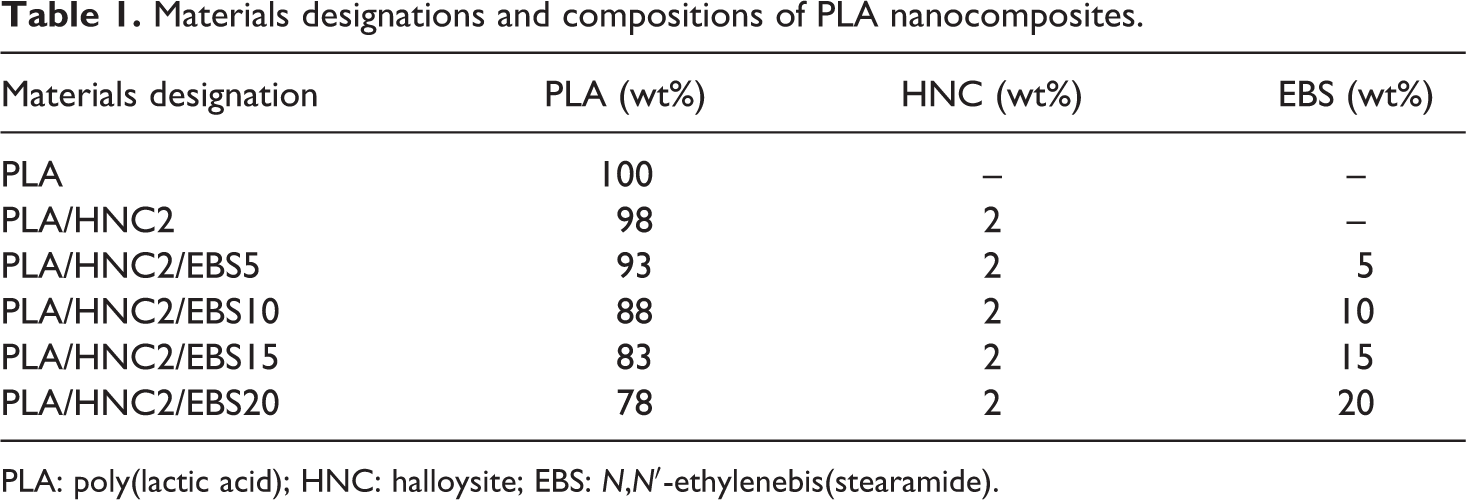
PLA: poly(lactic acid); HNC: halloysite; EBS: N,N′-ethylenebis(stearamide).
Characterization of PLA nanocomposites
Water absorption and hygrothermal aging tests
The specimens (impact bars; dimension: 65 × 13 × 3 mm3) were dried at 50°C in vacuum oven until a constant weight was attained. Then they were immersed in water in a thermostated stainless steel water bath at 30, 40, and 50°C. Weight changes were recorded by periodic removal of the specimens from the water bath and weighed on a balance with a precision of 1 mg. The percentage change at any time t, (Mt ) as a result of water absorption was determined using equation (1):

where W d and W w denote the weight of dry material (the initial weight) and weight of materials after exposure to water absorption, respectively. The percentage at M m was calculated as the average value of several consecutive measurements that showed no appreciable additional absorption. The weight gain, resulting from moisture absorption, can be expressed in terms of two parameters, the D and the M m, using equation (2).

where h is the thickness of the specimens. The D value can be calculated after rearranging equation (2) into equation (3), as follows:

where
The activation energies of water diffusion for the PLA nanocomposites were determined by using Arrhenius equation (see equation (4)).

where D is the diffusion coefficient of the sample; D 0 is the initial diffusion coefficient of the sample; E a is the activation energy of water diffusion; R is gas constant (8.314 J K−1 mol−1), and T is the temperature. By plotting the graph of ln (diffusion coefficient), ln (D) of the samples versus reciprocal of temperature (1/T), the value of E a of the water diffusion can be obtained (cf. equation (5)).

pH Measurement and molecular weight determination
The pH of the water (after the water absorption and hygrothermal aging tests) was measured using a pH510 meter (EUTECH instruments, Singapore). The molecular weight of PLA was determined by gel permeation chromatography (GPC) analysis. GPC analysis was performed with GPC system from Waters (Milford, Massachusetts, USA) equipped with a refractive index detector (RID) and columns. The calibration of the columns was carried out using a polystyrene standard of known molecular weight and polydispersity. Chloroform was used as the eluent with a flow rate of 1 ml min−1, and the injected sample volume was 20 µl. The samples were dissolved in chloroform, followed by filtration to eliminate contaminants.
DSC measurement
The melting and crystallization behavior of the composites were scanned under nitrogen atmosphere by DSC 1 (Mettler Toledo STARe, Columbus, Ohio, USA) using 10 mg samples sealed in aluminum pans. The specimens were scanned from 30°C to 200°C at a heating rate of 10°C min−1. The T g, melting temperature (T m), cold crystallization temperature (T cc), and degree of crystallinity (χ c) of the PLA composites were determined. The value of χ c of PLA nanocomposites was calculated using equation (6).

where ΔH m is the enthalpy of melting, ΔH c is the entalphy of crystallization (due to EBS), ΔH f is the enthalpy for 100% crystalline PLA, and w PLA is the net weight fraction of the PLA. The heat of fusion for 100% crystalline PLA is approximately 93.6 J g−1. 12
FTIR measurement
The chemical changes due to hygrothermal aging were monitored by FTIR (Spectrum One spectrophotometer, Perkin Elmer, Waltham, Massachusetts, USA). The spectrum of FTIR was determined from 4000 cm−1 to 400 cm−1 at 32 scans with a resolution of 4 cm−1.
Results and discussion
Kinetics of water absorption
Figure 1 shows the percentage water absorption (M t) of PLA nanocomposites as a function of t 1/2 at immersion temperature of 30, 40, and 50°C. From Figure 1(a) and (b), an initial linear relationship between M t and t 1/2 was observed in each case, followed by saturation. Table 2 shows the n value of PLA/HNC nanocomposites. The water transport behavior in polymeric materials can be divided into three types, namely, Fickian diffusion, relaxation controlled, and non-Fickian. The water diffusion behavior can be theoretically differentiated by the shape of the sorption curve (cf. equations (7) and (8)):
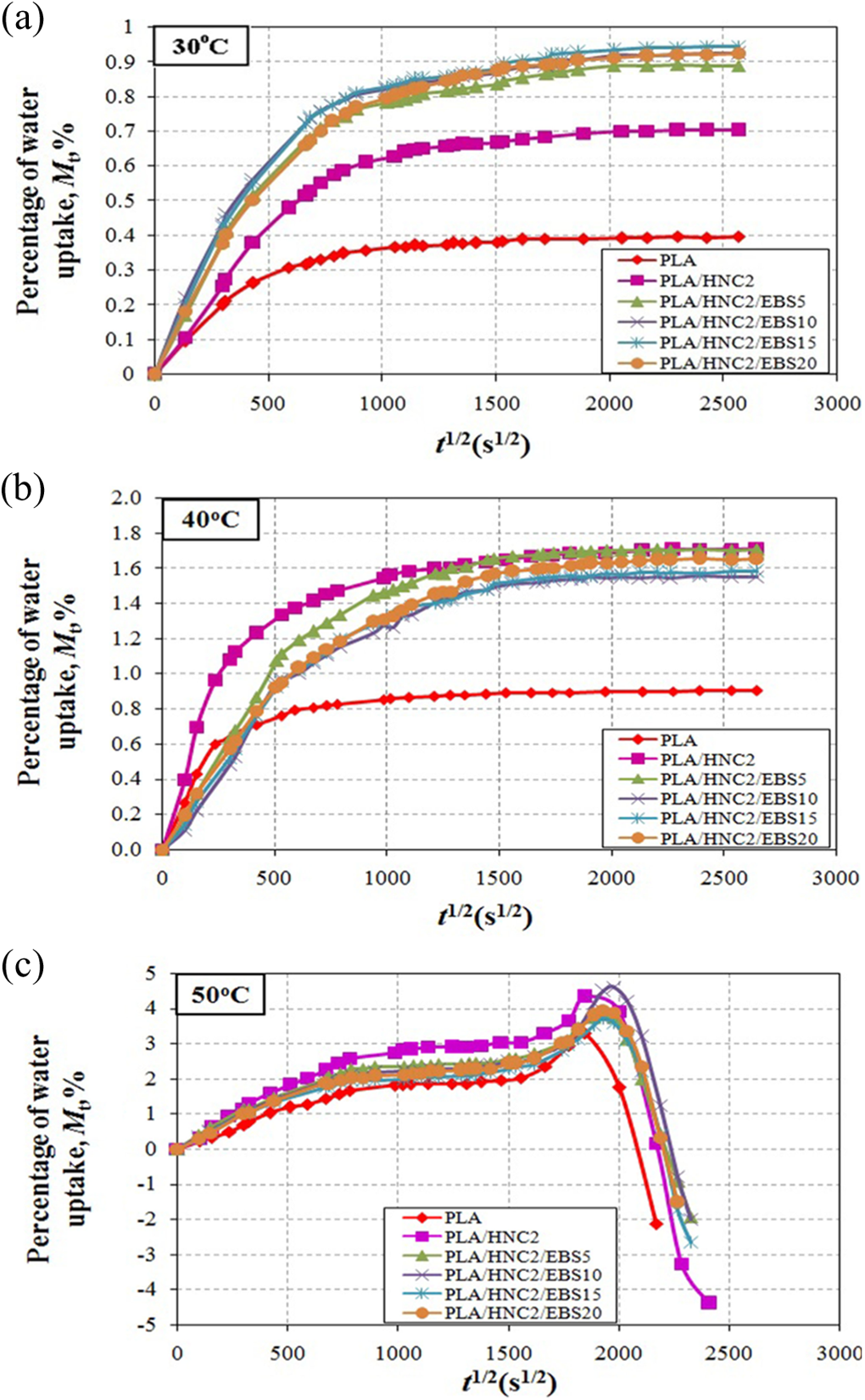
Water absorption of PLA nanocomposites at immersion temperatures of (a) 30°C, (b) 40°C, and (c) 50°C. PLA: poly(lactic acid).
Equilibrium water absorption (M m) and diffusional exponent (n) of PLA/HNC nanocomposites.
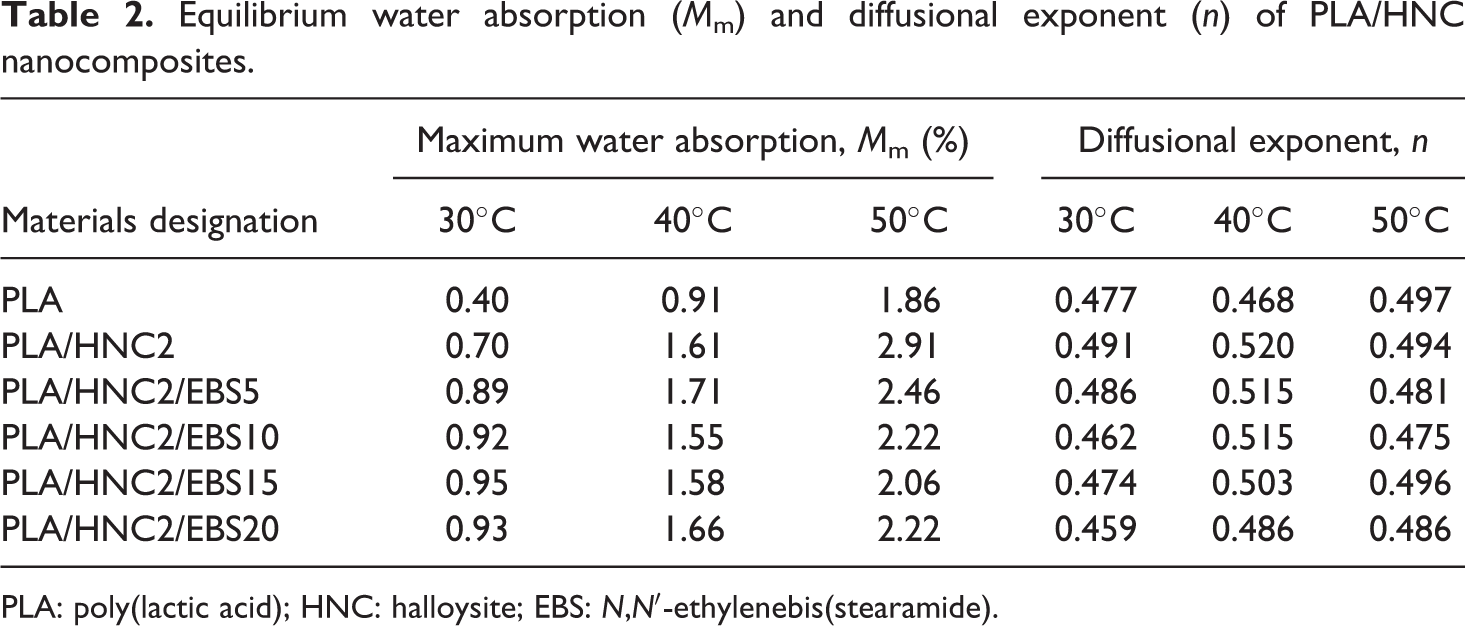
PLA: poly(lactic acid); HNC: halloysite; EBS: N,N′-ethylenebis(stearamide).


where M t is the water content at specific time t, M max is the maximum water absorption. The value of n shows different behaviors between three cases of diffusion: Fickian diffusion (n = 0.5), relaxation (n > 1), and anomalous transport (0.5 < n < 1.0). 30 The n values were determined from the slope of M t/M max versus t in the log plot (see Figure 2). Table 2 shows that the n values were close to 0.5 for all the PLA nanocomposites. This confirms that the water transportation of PLA/HNC composites is well fitted to Fickian diffusion behavior.
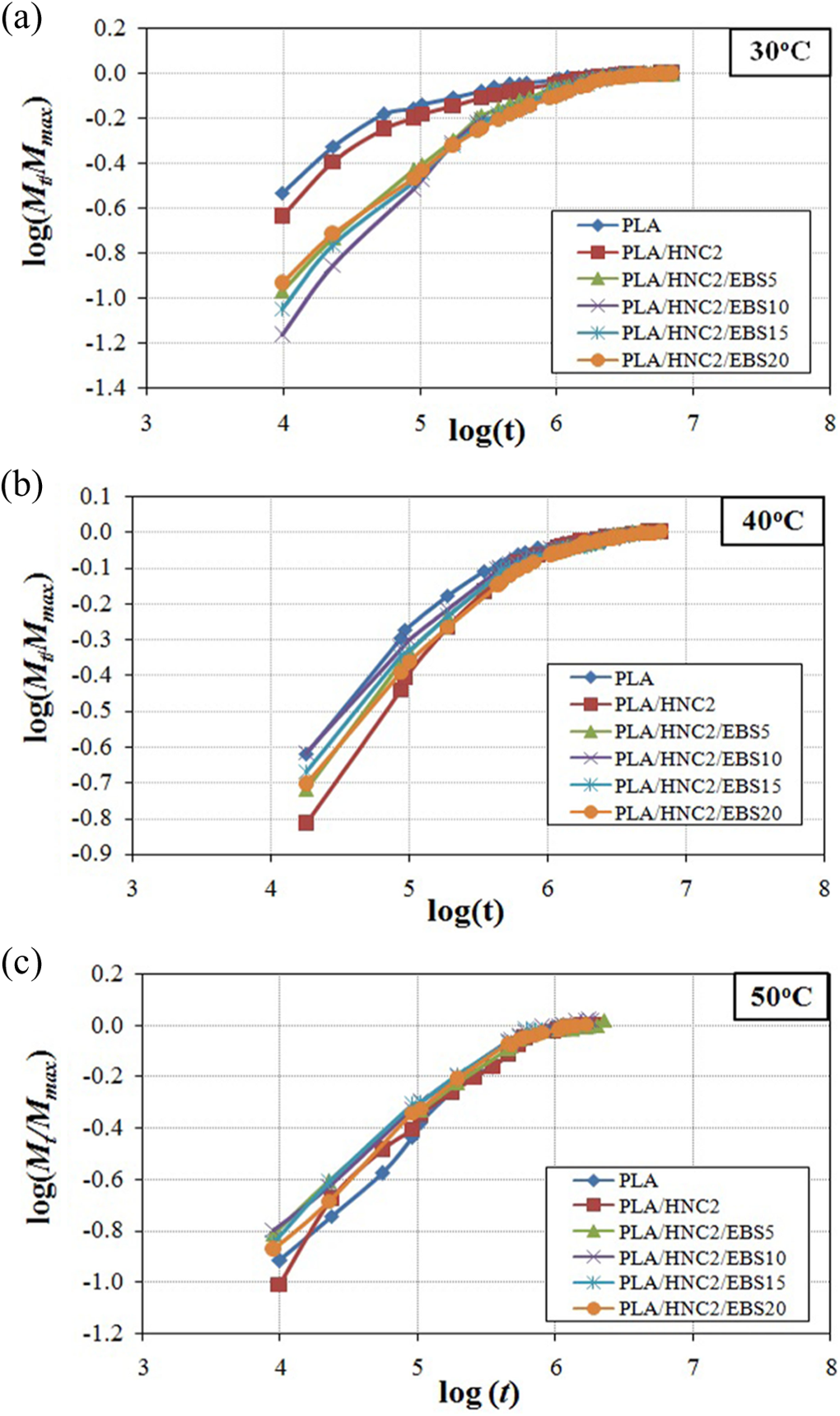
Plot log(Mt /M max) versus log(t) for PLA/HNC nanocomposites at immersion temperature of (a) 30°C, (b) 40°C, and (c) 50°C. M t: water content at specific time t, M max: maximum water absorption; PLA: poly(lactic acid); HNC: halloysite.
The M m and D values are summarized in Table 2. It can be seen that the M m of PLA/HNC2 nanocomposite is approximately 0.7% while the PLA is 0.4%. The weight gain of PLA/HNC2 nanocomposite is attributed to the hydrophilicity of HNC. The M m of PLA/HNC2/EBS is higher than that of both PLA and PLA/HNC2 nanocomposites. This due to the presence of amide group (–CONH–) in EBS that interacts with water molecules. In general, the water uptakes of polymer is governed by two theories: (i) the free volume theory (which is controlled by the moisture diffusion through substrates) and (ii) the interaction theory (which is controlled by the chemical bonding, e.g. H-bond at the polar sites). 28,30
Figure 1(a) and (b) shows the M m of PLA nanocomposites achieved at about 30 days (t 1/2 = 1500 s1/2) when subjected to water absorption at 30 and 40°C. However, Figure 1(c) shows that the PLA nanocomposites recorded M m at 11 days (t 1/2 = 1000 s1/2) when immersed in 50°C. This is due to the fact that at higher immersion temperature, water molecules gaining higher energy for the diffusion process. In other words, higher temperature increased the mobility of water molecules providing fast access of water into polymer segments. 31,32
It is worth mentioning that for the specimens which were exposed to 50°C (see Figure 1(c)), two stages of water uptake were observed. In the first stage, the water absorption obeys Fickian behavior, followed by saturation in between t 1/2 = 1000 s1/2 to t 1/2 = 1500 s1/2. In the second stage, the water uptake increased rapidly and reached a maximum point, followed by remarkable weight loss. This indicates the occurrence of hydrolytic degradation of PLA at 50°C as the immersion duration prolonged to t 1/2 > 1500 s1/2. The PLA chains become more flexible at this temperature (near to the T g), thus creating more free volume and allowing more water molecule to penetrate into the composite.
Table 3 shows the D and E a of PLA nanocomposites. It is obvious that the addition of HNC decreased the D value of PLA. The reduction of D value may be attributed to a tortuosity effect by the HNC, which restricts water molecule diffusion into the PLA. 33,34 Note that the D value of PLA/HNC2 < PLA/HNC2/EBS < PLA. It is worth mentioning that the D value of PLA/HNC2/EBS is governed by the EBS loading. The higher the EBS loading, the lower the D value is. At lower loading, the EBS was unable to restrict the water molecule diffusion because of its short hydrocarbon chain and small droplet dispersion in PLA. However, at higher loading, the EBS forms agglomerate (i.e., bigger droplets in PLA matrix) and becomes obstacles for the water molecule diffusion.
The values of D and E a of PLA nanocomposites at immersion temperature of 30°C, 40°C, and 50°C.
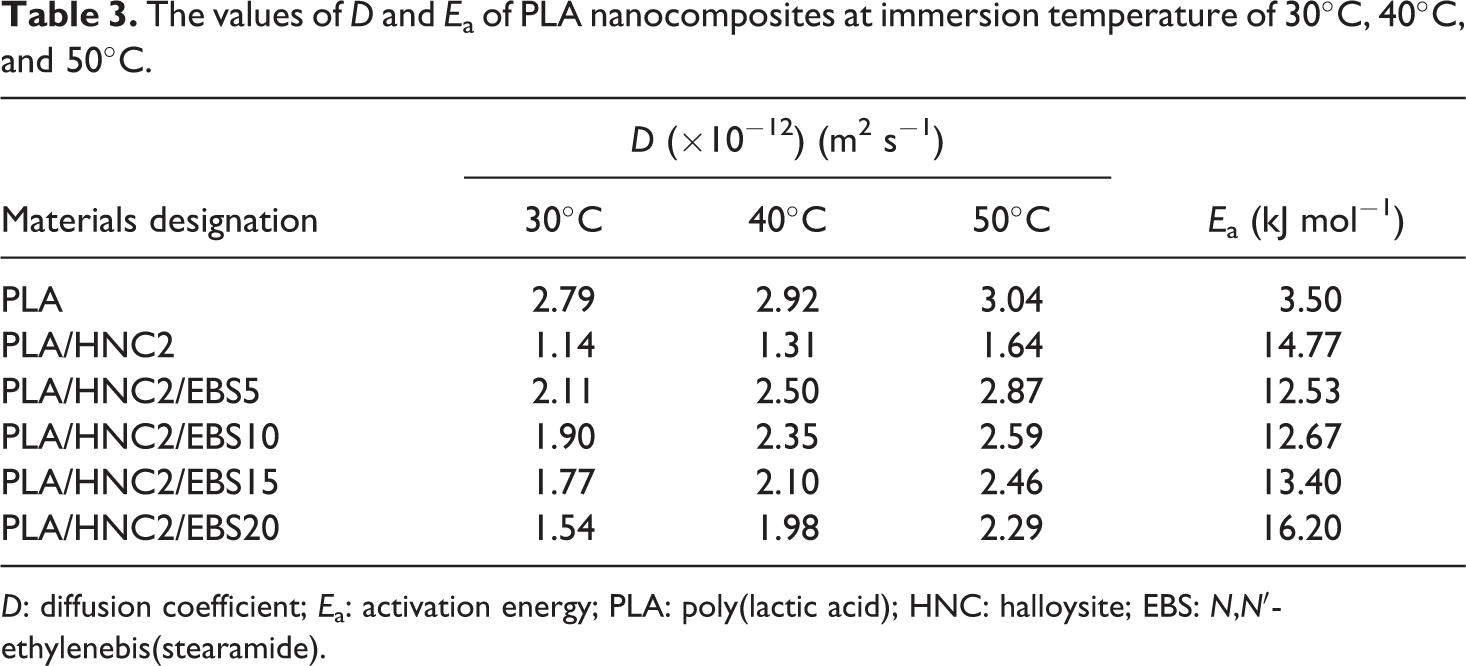
D: diffusion coefficient; E a: activation energy; PLA: poly(lactic acid); HNC: halloysite; EBS: N,N′-ethylenebis(stearamide).
Figure 3 shows the plot of ln D versus 1/T for PLA nanocomposites. The E a of water diffusion for PLA is 3.50 kJ mol−1. The E a of PLA/HNC2 (14.77 kJ mol−1) is higher than that of PLA. In general, E a is an energy barrier of a material that has to be overcome against diffusion of water. Table 3 presents the results of E a of PLA < PLA/HNC2/EBS < PLA/HNC2. The increment of E a values indicates that water molecules need more energy to diffuse into PLA matrix due to the barrier effect by HNC and EBS.
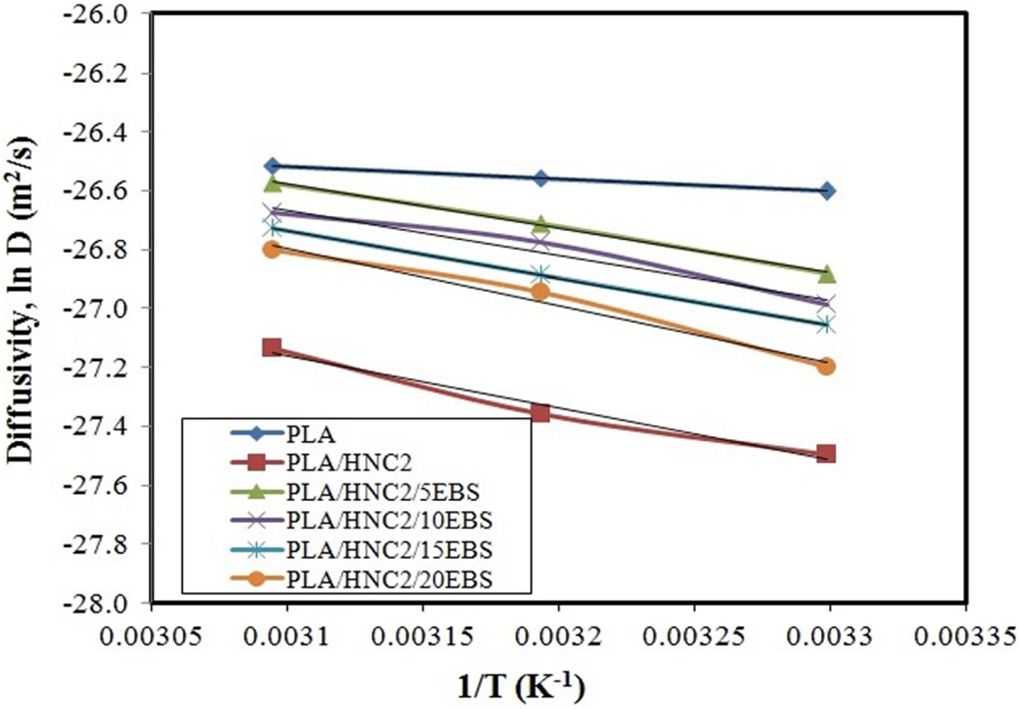
Plot of ln (D) versus 1/T for PLA nanocomposites. D: diffusion coefficient; T: temperature; PLA: poly(lactic acid).
pH and Molecular weight changes
The pH of water was examined after the PLA specimens immersed at 30, 40, and 50°C. The pH of control distilled water is 7.1. It was noted that in Table 4, the pH of water for PLA specimens is in the range of pH 6 to 7 at 30°C and 40°C. However, the pH values decrease extensively to pH 2 at an immersion temperature of 50°C. This indicates that the hydrolytic degradation of PLA induces the formation of carboxyl end group, thereby creating an acidic environment.
pH of water after the PLA samples immersed at different temperatures.a
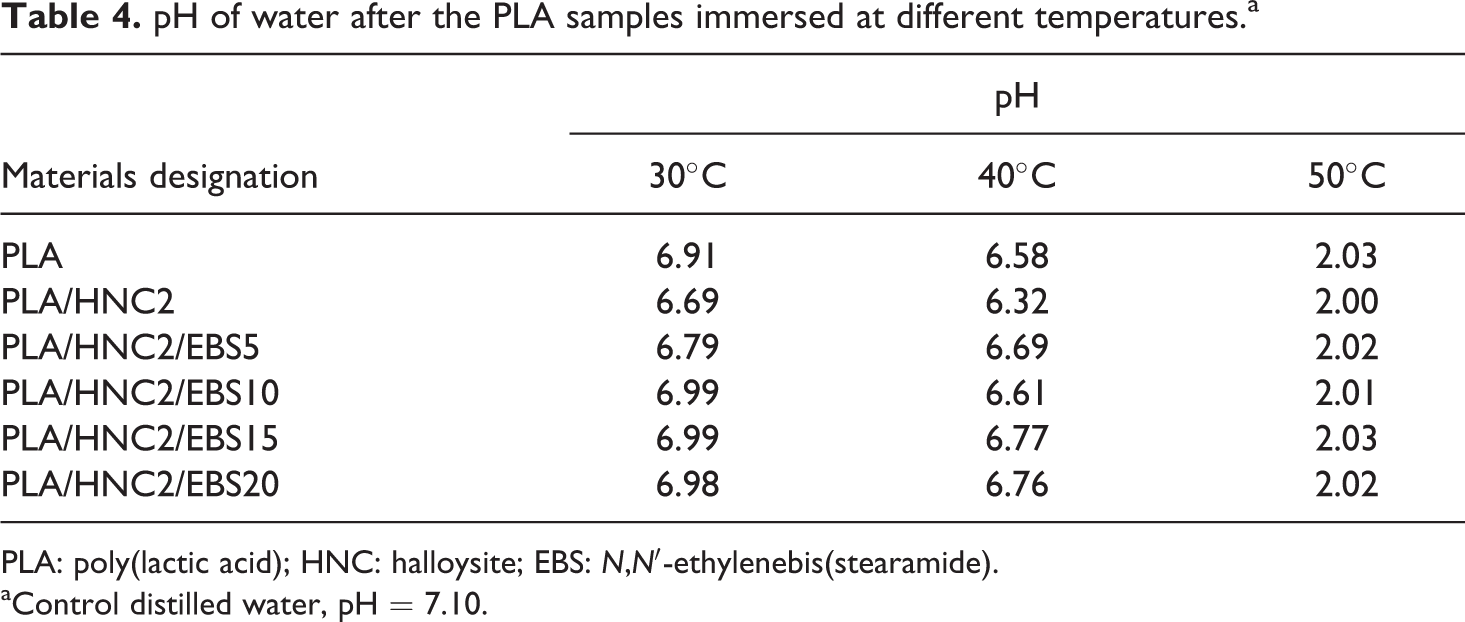
PLA: poly(lactic acid); HNC: halloysite; EBS: N,N′-ethylenebis(stearamide).
aControl distilled water, pH = 7.10.
Table 5 shows the number-average molecular weight (M n) of PLA nanocomposite after being subjected to water absorption at an immersion temperature of 50°C. The M n values for all PLA specimens showed in Table 5 are comparable before exposure to water absorption. The GPC data obtained show that a significant reduction of M n after subjected to water absorption at 50°C for 60 days. These changes can be related to the cleavage of PLA chains to shorter fragments which is caused by the hydrolysis of the ester linkage, followed by interchain transesterification and depolymerization by back-biting (intramolecular transesterification), leading to the formation of cyclic oligomers. 35
M n of PLA nanocomposite after being subjected to water absorption at immersion temperature of 50°C.
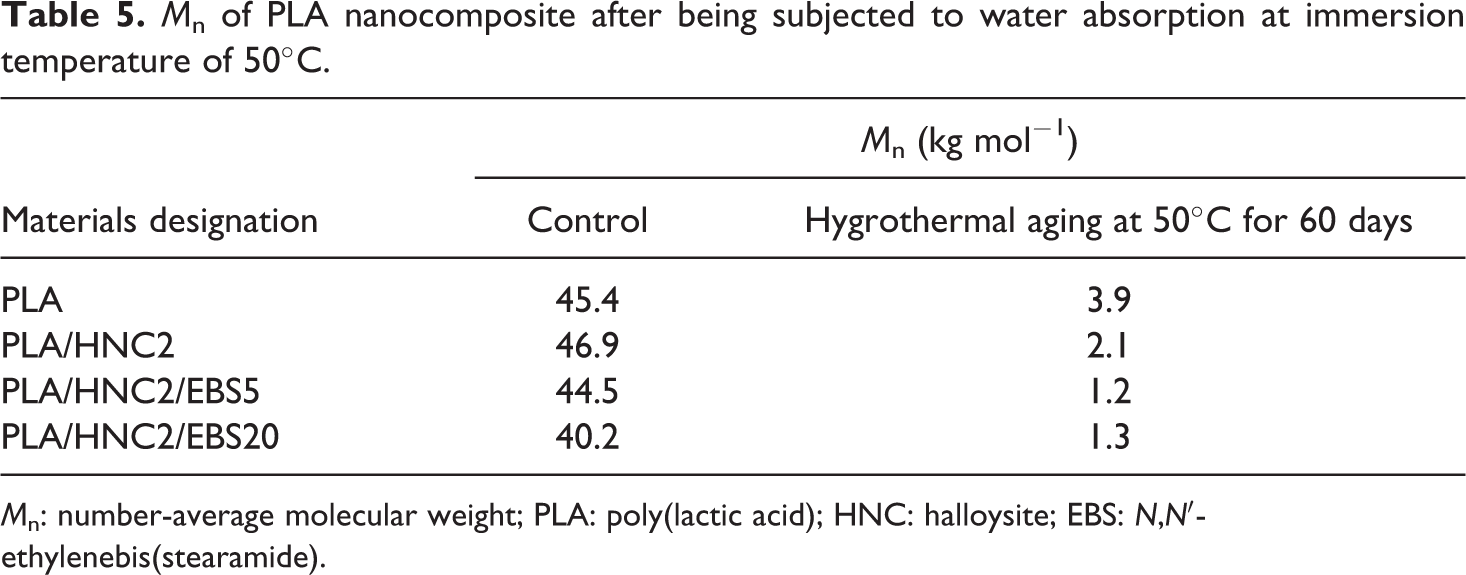
M n: number-average molecular weight; PLA: poly(lactic acid); HNC: halloysite; EBS: N,N′-ethylenebis(stearamide).
DSC measurement
Figure 4 shows the DSC curves of PLA nanocomposites before and after being subjected to water absorption. The thermal characteristics of PLA and its nanocomposites were tabulated in Table 6. At immersion temperatures of 30 and 40°C, it can be seen that the T g, T m, and T cc of PLA nanocomposites did not change much. However, a dramatic reduction of T g, T m, and T cc was observed for all PLA formulations after exposure to water absorption at 50°C. The reduction of T g is due to the plasticization effect of water, which increases the polymer chain mobility. 36 The decrease of T m can be related to the hydrolysis of PLA at 50°C. Molecular weight degradation during water absorption is attributed to the cleavage of the chain at ester group and breaking down the long macromolecular chains by hydrolysis process. 37,38 The decrease of polymer molecular weight may reduce the order of crystalline zones, owing to the disorder introduced by chain ends, and consequently shifted T m to a lower temperature. According to Fukushima et al., 39 shorter polymer chains with higher mobility show higher rate of crystallization leading to a lower T cc.
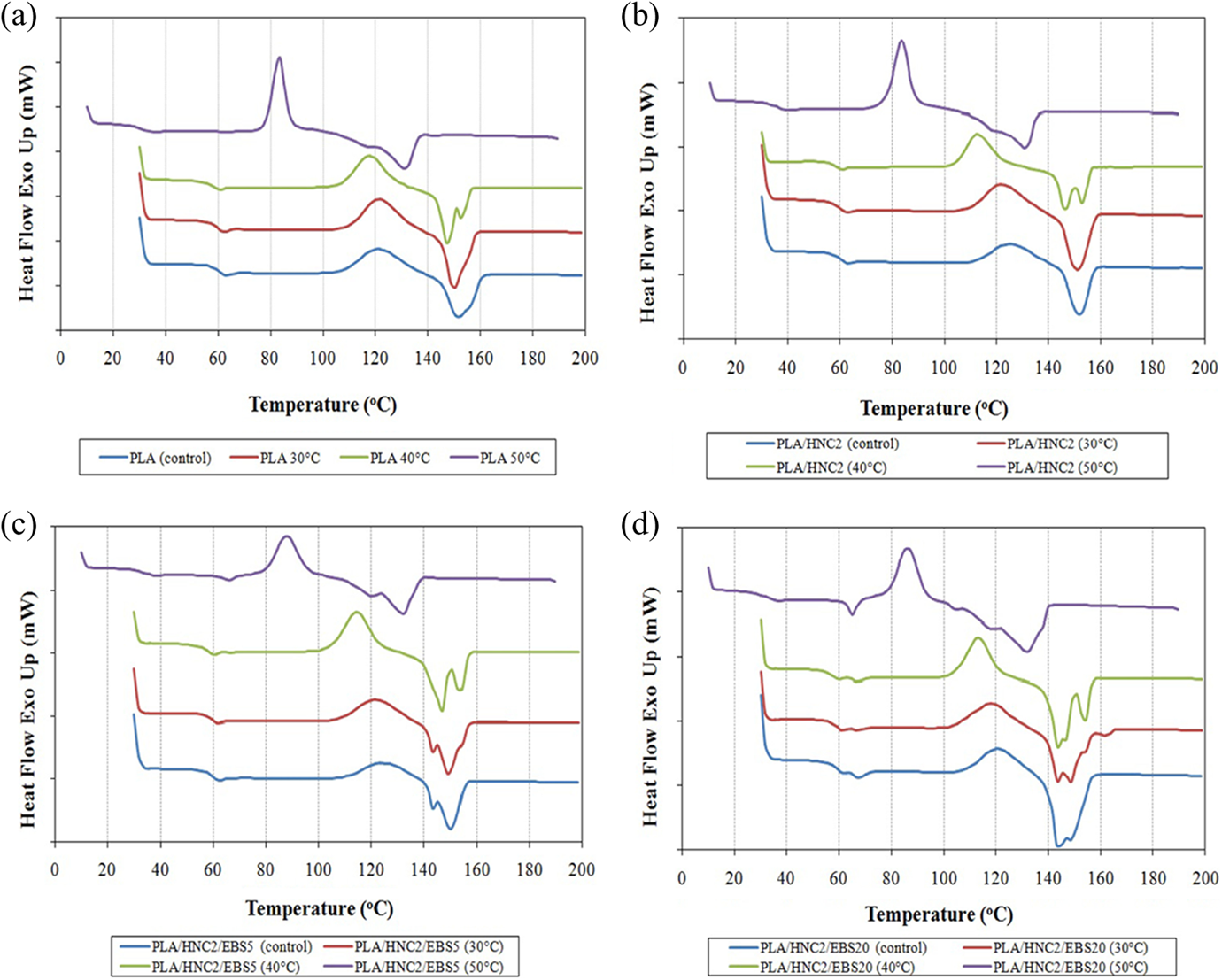
DSC curves of (a) PLA, (b) PLA/HNC2, (c) PLA/HNC2/EBS5, and (d) PLA/HNC2/EBS20 after being subjected to water absorption at three different temperatures. DSC: differential scanning calorimetry; PLA: poly(lactic acid); HNC: halloysite; EBS: N,N′-ethylenebis(stearamide).
Thermal characteristics of PLA/HNC composites before and after being subjected to water absorption.
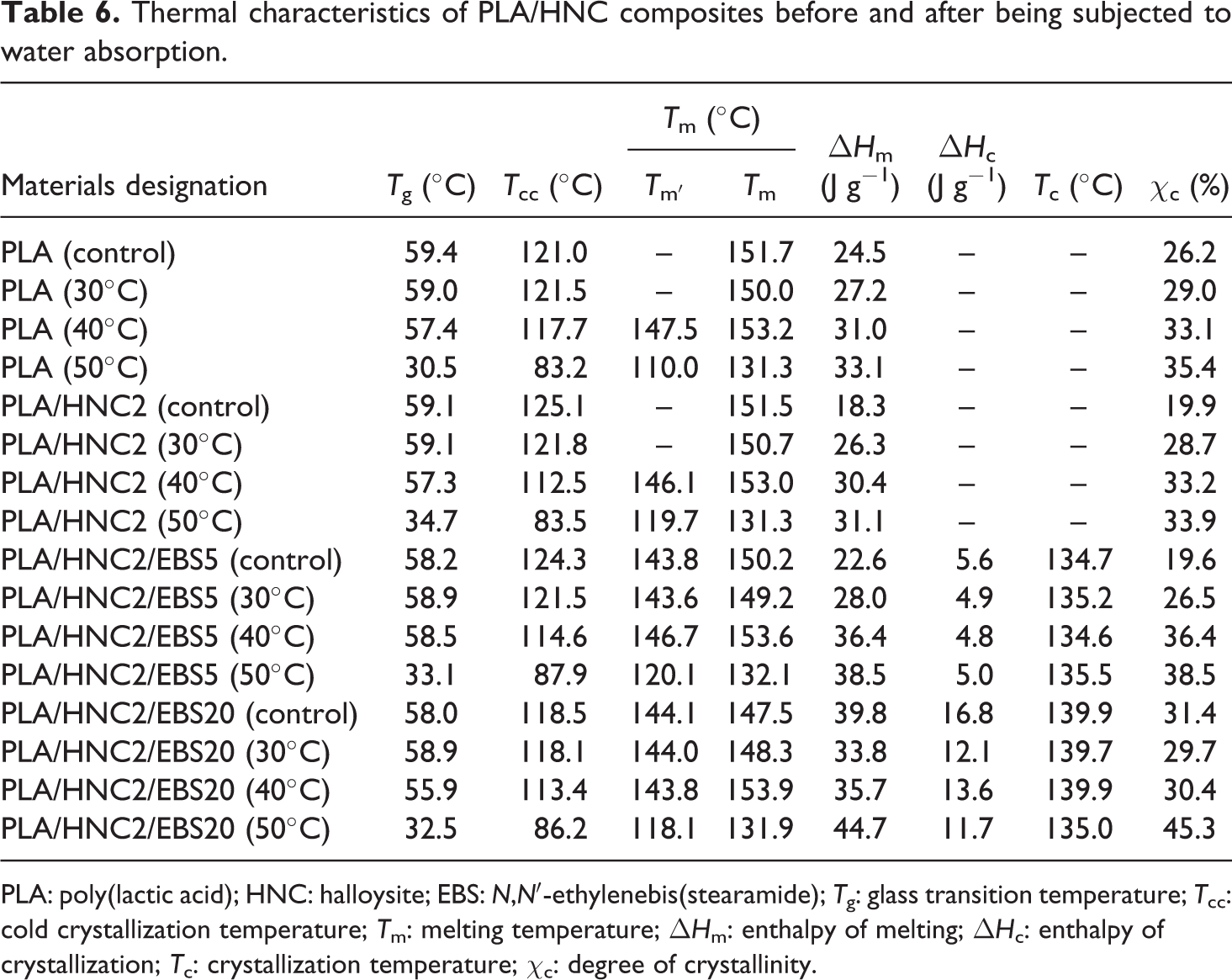
PLA: poly(lactic acid); HNC: halloysite; EBS: N,N′-ethylenebis(stearamide); T g: glass transition temperature; T cc: cold crystallization temperature; T m: melting temperature; ΔH m: enthalpy of melting; ΔH c: enthalpy of crystallization; T c: crystallization temperature; χ c: degree of crystallinity.
Double melting endotherms were observed for PLA and PLA/HNC2 nanocomposites after being subjected to water absorption at 40 and 50°C (see Table 6). This is attributed to the melting of crystalline structure domains of different sizes. 37 The formation of different size of crystal in PLA suggested significant chain orientations and morphological rearrangement occurred when molecular weight was decreased during the hydrolytic degradation process. 40 From Table 6, the χ c of PLA nanocomposites increased as the immersion temperature (i.e. 50°C) increases. The crystallization is mainly caused by the reduction of molecular weight and plasticization of PLA by water through chain scissions as well as by shorter polymer chains, that is, lactic acid oligomers during hydrolysis. 41,42 It was found that the χ c value of 20 wt% EBS-filled PLA/HNC2 nanocomposite increases dramatically compared to neat PLA as the increasing temperature.
FTIR analysis—CI measurement
Figure 5 shows the FTIR spectra of PLA nanocomposites before and after being exposed to water absorption. The characteristic functional group of PLA and PLA/HNC2 nanocomposites was reported in our previous work. 43 As shown in Figure 5(c) and (d), new bands appear at 3300 and 1560 cm−1 for PLA/HNC2/EBS nanocomposites, which can be assigned to the N–H stretching of amide group from EBS. The band intensities of 2920 and 2850 cm−1 were corresponded to C–H stretching and the spectra band at 1638 cm−1 was attributed to C=O stretching. These spectra bands were more noticeable when the loading of EBS increases in PLA nanocomposites.
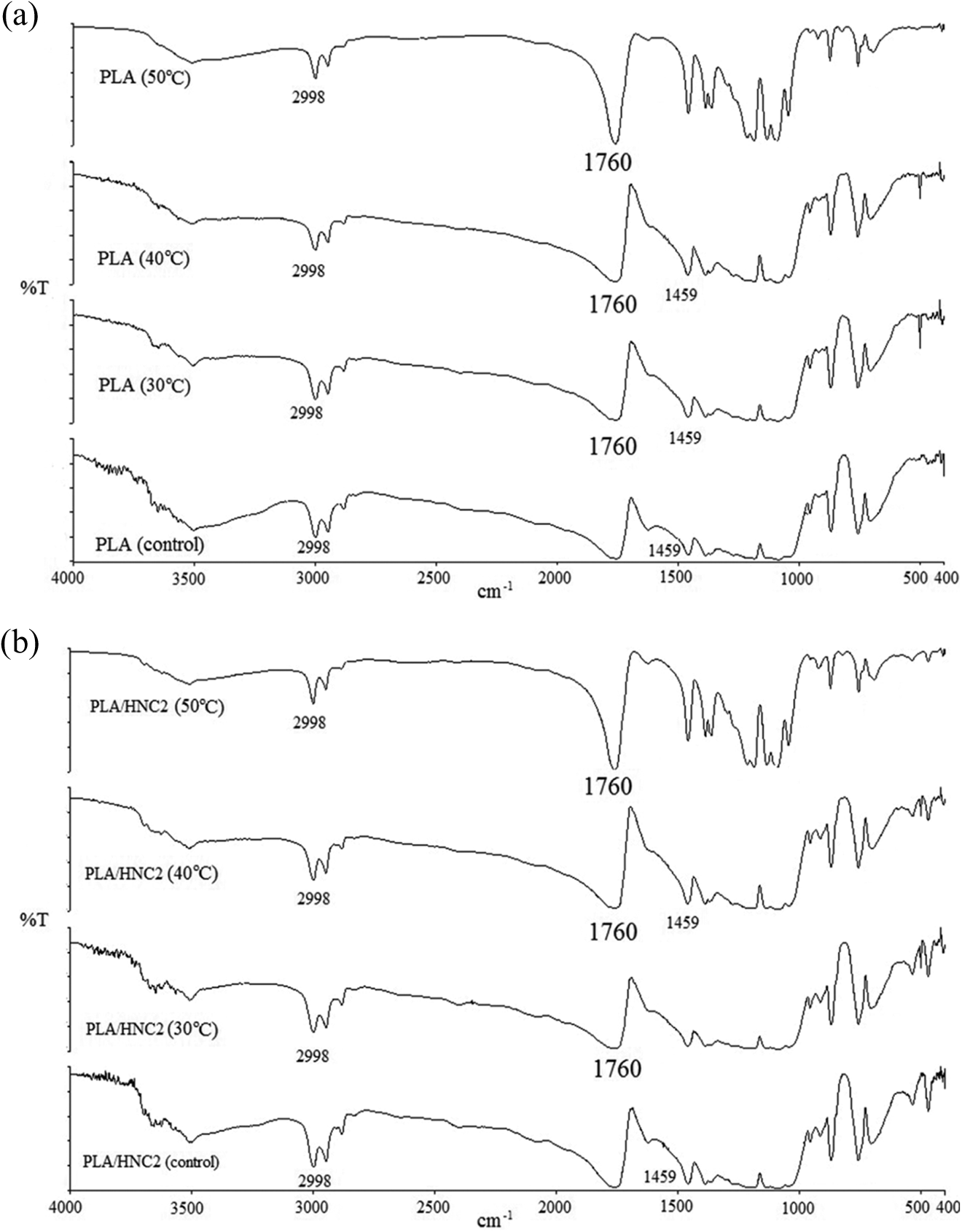
FTIR spectra of (a) PLA, (b) PLA/HNC2, (c) PLA/HNC2/EBS5, and (d) PLA/HNC2/EBS20 before and after being subjected to water absorption at three different temperatures (i.e., 30, 40, and 50°C). FTIR: Fourier transform infrared; PLA: poly(lactic acid); HNC: halloysite; EBS: N,N′-ethylenebis(stearamide).
From Figure 5, higher band intensity of 1800–1100 cm−1 for PLA nanocomposites was observed after the samples were exposed to water absorption at 50°C in comparison with the control sample. The hydrolysis of PLA backbones through its ester bond leads to the formation of carboxyl group (–COOH). It was believed that the carbonyl contents of PLA will be affected after the hydrolysis process. Therefore, carbonyl index (CI) of the PLA samples was calculated using equation (9).

The band at 3000–2998 cm−1 is chosen as reference peak, assuming that the –CH– bond does not undergo hydrolytic degradation during water absorption. The I 3000–2998 is the peak intensity of C–H band, while the I 1760–1758 represents the peak intensity of carbonyl group (–C=O). The CI values of PLA nanocomposites after being subjected to water absorption is shown in Table 7.
CI of PLA samples after being subjected to water absorption.
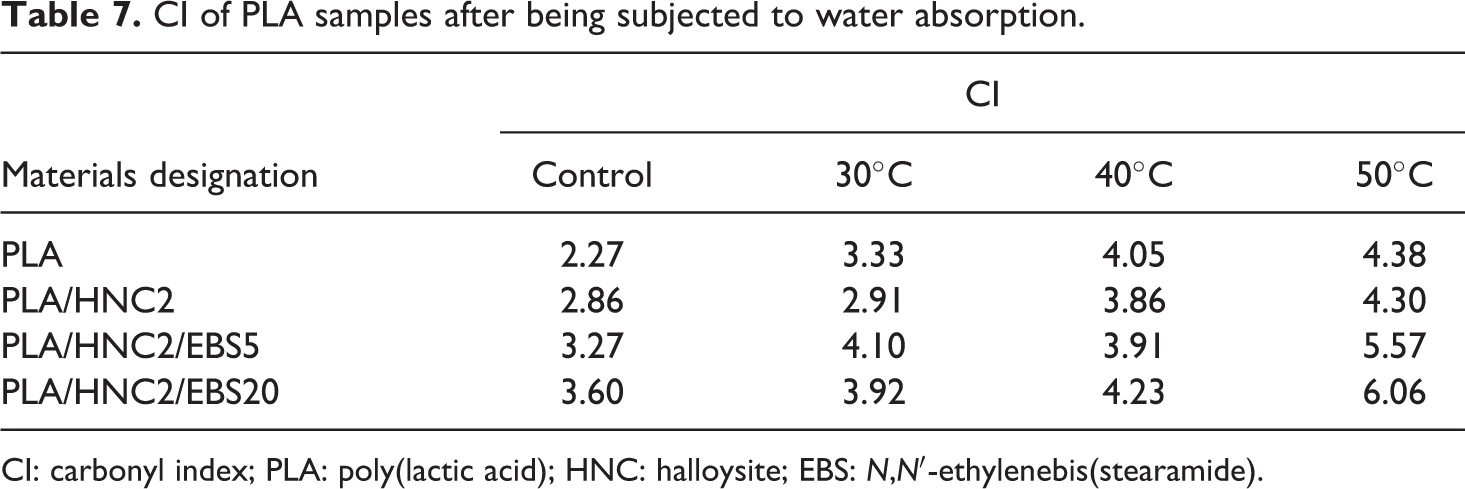
CI: carbonyl index; PLA: poly(lactic acid); HNC: halloysite; EBS: N,N′-ethylenebis(stearamide).
Note that the CI values of all PLA specimens increased after exposure to water absorption at 30, 40, and 50°C. The CI values increased dramatically at 50°C, which indicates that the increase of immersion temperature up to 50°C provokes hydrolytic degradation of PLA. The increase in CI values can be attributed to the formation of higher amount of carboxylic acid end group during hydrolysis process. The formation of lactic acid oligomers from PLA chain scission increased the carboxylic acid end groups concentration in the water medium that can induce the degradation reactions rapidly. 43 From Table 7, it can be seen that the CI values of PLA/HNC2/EBS were higher compared to that of PLA and PLA/HNC2 nanocomposites at 50°C. This can be due to the higher water absorption in the presence of EBS. EBS-containing amide group could absorb water, thereby accelerating hydrolytic degradation of PLA at higher immersion temperature. Thus, as more PLA chain scission occurs, higher amount of carboxylic acid end groups are formed and subsequently increasing the CI values.
Conclusions
The kinetics of water absorption of PLA/HNC nanocomposite fitted Fickian diffusion behavior at immersion temperature of 30 and 40°C, which can be confirmed by the n values that are close to 0.5. The E a of water diffusion for PLA nanocomposites increased with the incorporation of HNC and EBS. The T g, T cc, and T m of the PLA sample were shifted to lower temperatures and the effect was more pronounced at 50°C. This can be attributed to the fragmentation and chain scission of polymer under the water absorption environment (especially at immersion temperature, T = 50°C) owing to the molecular weight reduction. The CI values of all PLA samples increased after water absorption due to the formation of higher amount of carboxylic acid end groups during the hydrolysis process. This can be confirmed by the remarkable reduction in pH value (i.e. from pH 7 to pH 2), which suggests an acidic environment after the water absorption test.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Universiti Sains Malaysia Research University Grant (grant nos. 814070 and 814199), USM Incentive Grant (grant no. 8021013), and Ministry of Higher Education Malaysia MyPHD Scholarship Programme.
