Abstract
Two kinds of multiwalled carbon nanotubes/silica nanohybrids (CNTs/SiO2) were synthesized via a sol–gel method by coating SiO2 on the surfaces of CNTs that had been functionalized with poly(sodium-p-styrenesulfonate) (PSS). The influence of the nanohybrids on the thermal properties of poly(vinyl alcohol) (PVA) and polyurethane (PU) composites was investigated. Characterization of CNTs/SiO2 nanohybrids, elucidated with scanning electron microscopy and X-ray diffraction, showed that PSS played a key role in the final morphology and resulted in a “core/shell” and “candied haws on a stick”-like structure. The thermal degradation and thermo-oxidation of PVA and PU composites evaluated by thermogravimetic analyses suggested that the thermal properties of PVA/(CNTs/SiO2) composites were improved at relative high temperature range when compared with pure PVA, and the improvement mainly depended on the loading fraction of the nanohybrids, while for PU/(CNTs/SiO2) composites, a small fraction of the nanohybrids increased the materials thermal properties, but the improvements were influenced by the type of CNTs/SiO2 nanohybrids.
Introduction
Carbon nanotubes (CNTs) have attracted considerable interest in polymeric composites since they can impart the materials a unique combination of mechanical-, thermal-, and flame-retardant properties. 1 –5 However, it is difficult to make CNTs disperse homogenously within the polymeric matrix due to the intrinsic attractive van der Waals forces among CNTs, which have become the main obstacle of their applications. 6,7
There are two strategies commonly employed to functionalized CNTs to enhance the compatibility between CNTs and polymeric matrix. 8 –11 One is the chemical functionalization, which enhances the solubility of CNTs by providing covalent linkage between the sidewalls and/or endcaps of CNTs and functional groups. 12 –14 Another way is adding polymer or surfactant to form non-covalent polymer wrapping and non-covalent surfactant-encapsulating CNTs. 15 –17 Besides these two approaches, inorganic oxides (IOs) such as silica (SiO2), and metal oxides (MO) such as titanium dioxide and zinc oxide have been employed to fabricate CNTs/IO and CNTs/MO nanohybrids to surface modify CNTs, which holds high promise for new potential applications perhaps in polymeric materials. 18,19
As far as it is known, previous research studies mainly focus on the effect of covalent and/or non-covalent-functionalized CNTs on the improvement in physicochemical properties of polymeric nanocomposites, such as polystyrene, poly(methyl methacrylate), poly(vinyl alcohol) (PVA), and polyurethane (PU), but the influence of CNTs/IO and CNTs/MO nanohybrids on the mechanical, thermal properties, and flammability performance of polymeric materials are not well known. 19 –23
In the present work, two kinds of nanohybrids of multiwalled CNTs with different diameters encapsulated by SiO2 (CNTs/SiO2) were synthesized via a sol–gel method by encapsulating SiO2 on non-covalent–modified CNTs with the aid of poly(sodium-p-styrenesulfonate) (PSS). Their corresponding PVA and PU composites were prepared by solution blending. Scanning electron microscopy and thermogravimetric analyses (TGA) were employed to study the morphology of CNTs/SiO2 nanohybrids and the thermal properties of these polymeric materials, and the emphasis is to investigate how the nanohybrids influence the thermal degradation and thermo-oxidative behavior of the materials.
Experimental
Materials
Waterborne PU emulsion (PU, trade mark PU80, solid content 30 ± 1%) was purchased from Hefei Anke Fine Chemicals Co., Ltd (China). Multiwalled CNTs (CNT20 with a diameter of approximately 10–30 nm and CNT60 with a diameter of approximately 50–70 nm, trade mark HCNTs20 and HCNTs60, respectively) were provided by Shenzhen Susn Sinotech New Materials Co., Ltd (China). PSS ((C8H7NaO3S) n , average molecular weight 70,000 was obtained from J&K Scientific Ltd (Beijing, China). PVA (1750 ± 50, average molecular weight 70,000), tetraethyl orthosilicate (TEOS, C8H20O4Si), ammonium hydroxide (approximately 25–28% solution in water), and ethanol were purchased from Sinopharm Chemical Reagent Co., Ltd (China). All chemicals were used as received.
Synthesis of CNTs/SiO2 nanohybrids
Nanohybrids of CNT20 and CNT60 encapsulated by SiO2, which were denoted as CNT20/SiO2 and CNT60/SiO2, respectively, were synthesized by a sol–gel process. In a typical procedure, 50 mg CNTs dispersion in 150 ml of 0.5 wt% PSS solution consisting of 75 ml deionized water and 75 ml ethanol were ultrasonic agitated for about 2 h and transferred into a three-necked round-bottomed flask. Then, 13.5 ml ammonium hydroxide was added into the above mixture. Subsequently, a TEOS solution (1 ml TEOS/13 ml ethanol) was dripped, followed by mechanical stirring for 24 h at 32°C. The precipitates were filtered using 0.45 μm microporous membrane, washed with deionized water and ethanol, and then dried in an oven at 60°C.
Preparation of PVA/(CNTs/SiO2) and PU/(CNTs/SiO2) composites
PVA composites with CNTs/SiO2 nanohybrids were prepared by solution mixing method. A 10 wt% PVA aqueous solution (5 g PVA/45 g deionized water) was firstly prepared by stirring at 90°C. The desired amounts of CNTs/SiO2 nanohybrids were dispersed in 10 ml deionized water with ultrasonication for 10 min, followed by introduction into the PVA aqueous solution with vigorous stirring for 6 h. The obtained solutions were poured into a case to get approximately 0.3 mm thick films and air dried at room temperature before a further vacuum drying at 60°C for 12 h. The formulations are given in Table 1.
Formulations and the TGA data of PVA/(CNTs/SiO2) composites.
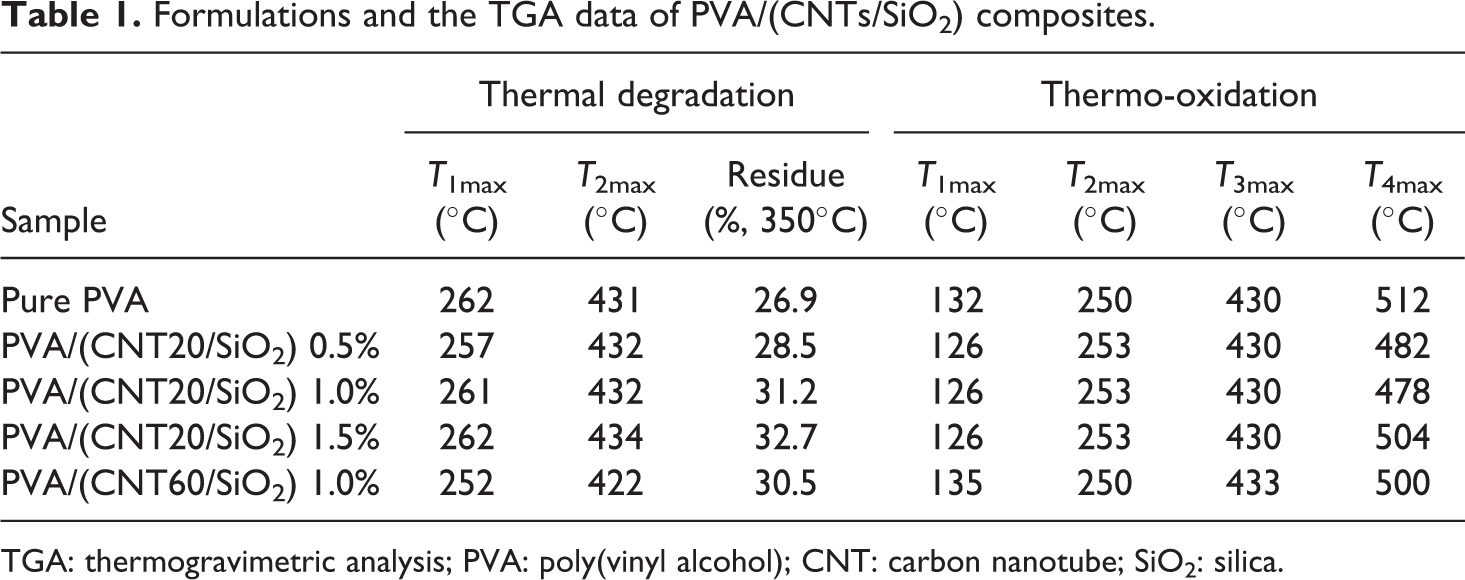
TGA: thermogravimetric analysis; PVA: poly(vinyl alcohol); CNT: carbon nanotube; SiO2: silica.
To obtain PU/(CNTs/SiO2) composites, 20 mg CNTs/SiO2 nanohybrids were firstly ultrasonic agitated in 10 ml of 0.5 wt% PSS solution for 10 min, followed by centrifugation at 2000 r min−1 for 4 min, and the supernatants were then added into 5 g PU emulsion with magnetic stirring for 4 h at room temperature. The PU/(CNTs/SiO2) films were obtained according to the above method.
Characterization
The X-ray diffraction (XRD) patterns were measured on a D/max-TTRIII X-ray diffractometer equipped with a copper K α radiation (λ = 1.5418 Å) at room temperature. Morphology of the products was observed on a Sirion 200 field-emission scanning electron microscopy (FESEM; FEI, Hillsboro, Oregon, USA). Thermal degradation and thermo-oxidative behaviors of the materials were investigated by TGA, which were conducted with a TA Q5000 thermoanalyzer instrument (TA Instruments, New Castle, Delaware, USA). In each case, approximately 5–10 mg specimens were heated from 30°C to 700°C at a heating rate of 10°C min−1 under nitrogen or in air atmosphere at a flow rate of 40 ml min−1.
Results and discussion
Thermal properties of CNTs/SiO2 nanohybrids
Thermal properties of the selected samples of CNT20s and CNT20/SiO2 nanohybrids are shown in Figure 1. It is clearly seen that CNT20s exhibit high thermal degradation stability under nitrogen atmosphere. Although they show high 5% weight loss temperature, also referred as onset temperature (T 5%) at about 600°C during thermo-oxidation, CNT20s lose a weight of about 61.3 wt% in the temperature range of approximately 600–700°C.
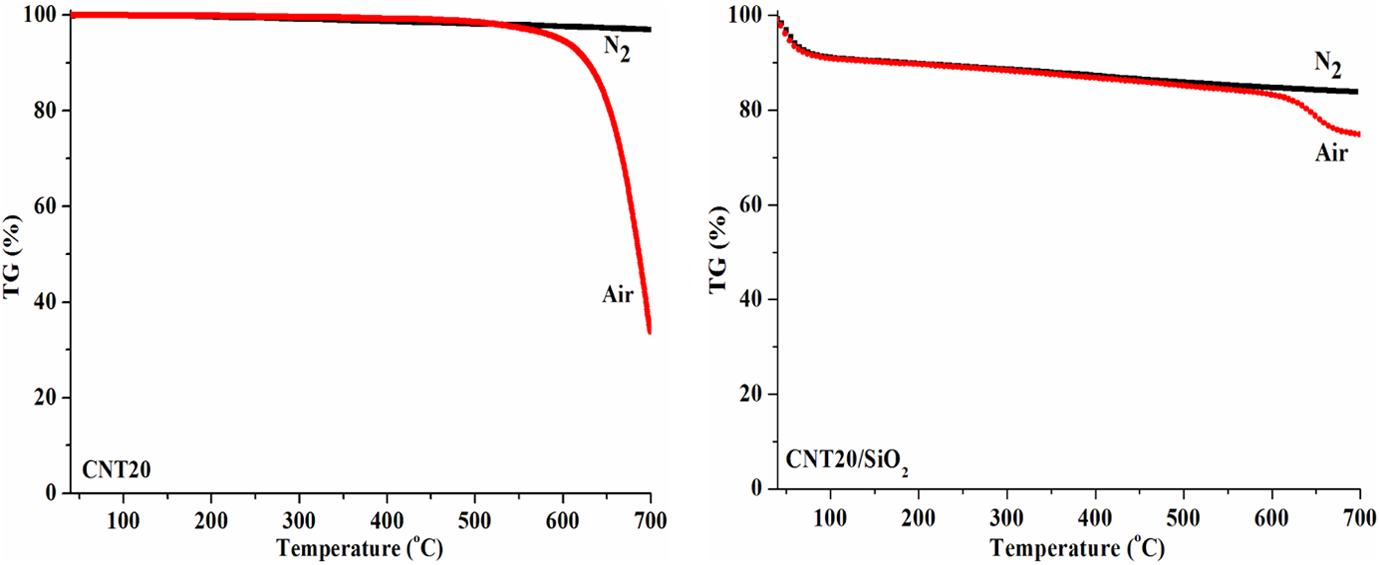
TGA curves of CNT20s and CNT20/SiO2 nanohybrids tested under nitrogen and in air atmospheres.
Compared to pristine CNT20s, both thermal degradation and thermo-oxidation of CNT20/SiO2 nanohybrids start earlier due to the loss of absorbed water and low-molecular-weight residues from PSS and exhibit faster weight loss rate in the temperature range of approximately 100–600°C (Figure 1, right). However, there is only a weight loss of about 8.4 wt% of CNT20/SiO2 nanohybrids in the temperature range of approximately 600–700°C, which is mainly caused by the condensation of SiO2 precursor and the oxidation of CNT20s; meanwhile, it also suggests that the coating of SiO2 on CNT20 surfaces enhances the thermo-oxidative stability of the nanohybrids.
Morphologies of CNTs/SiO2 nanohybrids
Figure 2 shows the FESEM images of CNT20s and CNT20/SiO2 nanohybrids. CNT20s entangle each other and randomly form an interconnecting structure (Figure 2(a)). Figure 2(b) shows that after coating, a “core/shell” structure was formed in CNT20/SiO2 nanohybrids in which the SiO2 layer was uniformly coated on the surfaces of CNT20s and resulted in a smoother surface with an increased diameter of around 100 nm. Meanwhile, a “candied haws on a stick”-like structure in which CNT20s pierced through SiO2 nanoparticles is also found in the insert. It is postulated that CNTs were firstly non-covalently wrapped by PSS via π–π interactions, which was favorable to improve the solubility of CNTs in water/ethanol, and SiO2 precursor was subsequently attached to the surfaces of CNTs through hydrogen bonds, which led to the formation of two different kinds of nanostructures.

FESEM images of (a) CNT20s, (b) CNT20/SiO2 nanohybrids, (c) CNT20/SiO2 nanohybrids calcined at 300°C for 1 h, and (d) CNT20/SiO2 nanohybrids calcined at 600°C for 3 h.
Figure 2(c) and (d) shows the CNT20/SiO2 nanohybrids calcined in air atmosphere at two different temperatures, 300°C for 1 h and 600°C for 3 h inside a muffle furnace, respectively. Figure 2(c) demonstrates that at relatively low calcination temperature, the nanohybrids remain in the structure of the uncalcined counterpart. Calcinations at 600°C led to the formation of a gray-colored powder. Figure 2(d) shows that SiO2 nanoparticles attached with irregular smaller particles can be observed, but it is hard to find the CNTs, which may indicate that CNT20s have been cut due to the air oxidation.
Figure 3 shows the FESEM images of CNT60s and CNT60/SiO2 nanohybrids. Similar to the observations in CNT20/SiO2 nanohybrids, SiO2 layer and/or SiO2 nanoparticles are found to be coated on the surface of CNT60; meanwhile, the high-temperature calcinations destroyed this structure.

FESEM images of (a) CNT60s, (b) CNT60/SiO2 nanohybrids, (c) CNT60/SiO2 nanohybrids calcined at 300°C for 1 h, and (d) CNT60/SiO2 nanohybrids calcined at 600°C for 3 h.
XRD studies of CNTs/SiO2 nanohybirds
Figure 4 shows the XRD patterns of CNT20/SiO2 and CNT60/SiO2 nanohybrids calcined at different temperatures. The peaks at 24.6° and 43.1° of CNT20s shown in the insert in Figure 5 (left) correspond to the (002) and (101) diffraction reflections of graphite layers, respectively. 24 A weak and broad peak centered at 22° assigned to a typical reflection of amorphous SiO2 appears in CNT20/SiO2 nanohybrids, which is concurrent with the weak (002) diffraction peak of CNT20. Calcinations of CNT20/SiO2 nanohybrids at 300°C leave the similar XRD pattern to that of the uncalcined counterpart. When the nanohybrids are calcined at 600°C, the peak at 22° becomes sharp, but the typical reflection at 24.6° still exists. According to FESEM observation, it is therefore deduced that during high-temperature calcinations, CNTs were oxidized to become shorter but still remained in their graphite-layered structure. As expected, CNT60/SiO2 nanohybrids showed the similar XRD features to those of CNT20/SiO2 nanohybrids.
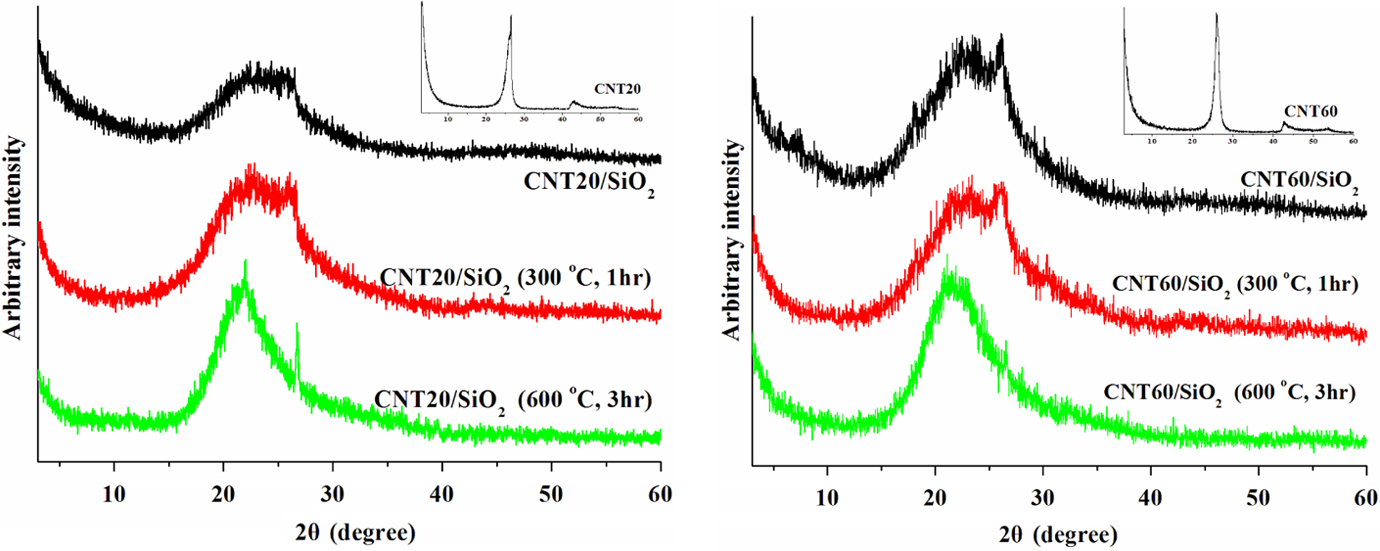
XRD patterns of CNT20/SiO2 and CNT60/SiO2 nanohybrids calcined at different temperatures in an air atmosphere.
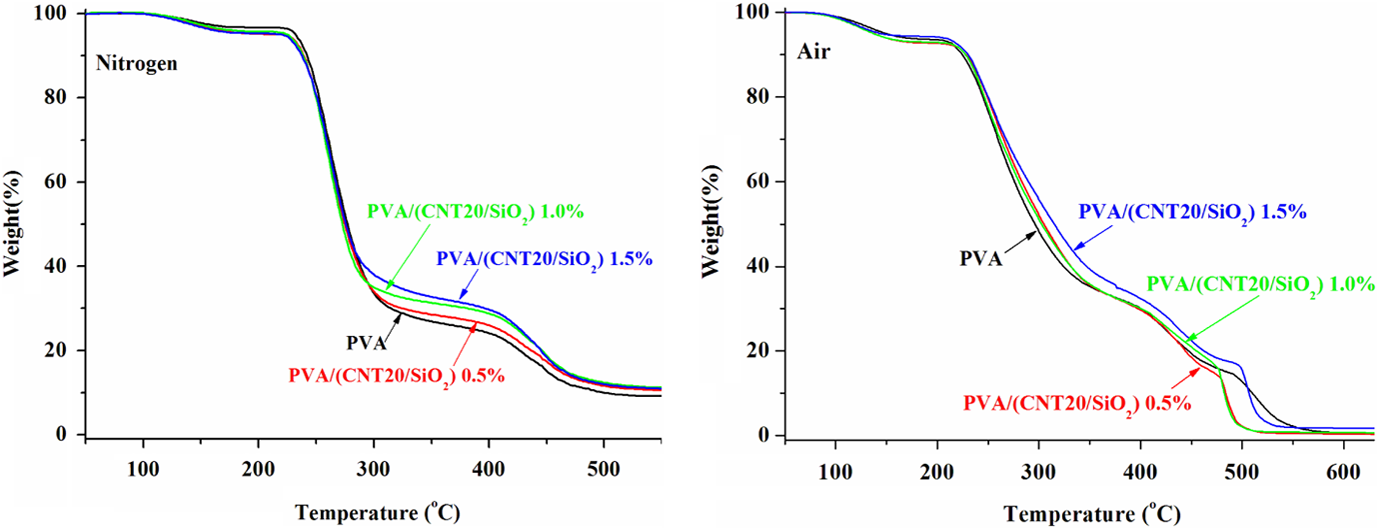
TGA curves of PVA/(CNT20/SiO2) composites tested under nitrogen and in air atmospheres.
Thermal properties of PVA/(CNTs/SiO2) composites
The influence of CNT20/SiO2 nanohybrids on thermal behavior of PVA was investigated by TGA. The TGA data, including T 5% and maximum mass loss rate temperature (T max; the temperature obtained from the derivative TGA curve (DTG)), and the residue remaining at 350°C are listed in Table 1.
Thermal degradation profiles of PVA/(CNT20/SiO2) composites are shown in Figure 5 (left). The pure PVA undergoes a two-stage degradation, where the first maximum of the degradation occurs at 262°C (T 1max), which is mainly due to the elimination of residual acetate groups, followed by the second maximum at 431°C (T 2max) due to the cleavage of the PVA backbone degradation. 25 An early weight loss process in the temperature range of approximately 100–160°C is attributed to the loss of absorbed moisture.
When CNT20/SiO2 nanohybrids are presented, all PVA/(CNT20/SiO2) composites keep two weight loss stages as pure PVA. Although the incorporation of CNT20/SiO2 nanohybrids does not change the thermal degradation feature of PVA at relatively low temperatures, the composites exhibit increased thermal degradation stability with elevating temperature above 290°C. Noticeably, the higher the loading of the CNT20/SiO2 nanohybrids, the higher is the char formation of the composites, which is evident that the residues at 350°C are 28.5, 31.2, and 32.7 wt% for PVA composites with 0.5, 1.0, and 1.5 wt% CNT20/SiO2 nanohybrids, respectively.
The thermo-oxidation of PVA/(CNT20/SiO2) composites in air atmosphere occurs as four successive processes (Figure 5, right): the loss of absorbed water, partial dehydration of PVA, degradation of polyene, and the thermo-oxidation of carbonized residue. It is noted that the incorporation of 0.5 and 1.0 wt% CNT20/SiO2 nanohybrids imparts enhancement of PVA composites in stability in the temperature range of approximately 220–470°C slightly, while the presence of 1.5 wt% enhances the stability of PVA in the temperature range of approximately 220–500°C obviously. Unexpectedly, the carbonized residues from all composites exhibit worse thermo-oxidative stability, and they show a decrease of approximately 8–35°C in terms of T 4max (the temperature at the maximum of degradation of char) compared to pure PVA, which is possibly related to the destruction of CNT20/SiO2 nanohybirds. The improvements in thermal behaviors of PVA/(CNT20/SiO2) composites are postulated to be due to the covalent bonds (–Si–O–C–) formed between hydroxyl groups of PVA and hydroxyl groups on SiO2 coated on CNTs surfaces, which help to increase the interactions between PAV and CNT20/SiO2 and restrict the polymer chain motion.
Figure 6 demonstrates that thermal behavior of PVA is not obviously influenced by the types of CNTs since PVA/(CNT60/SiO2) exhibits the similar TGA profiles to PVA/(CNT20/SiO2) at the same 1.0 wt% loading, no matter whatever atmosphere is employed.
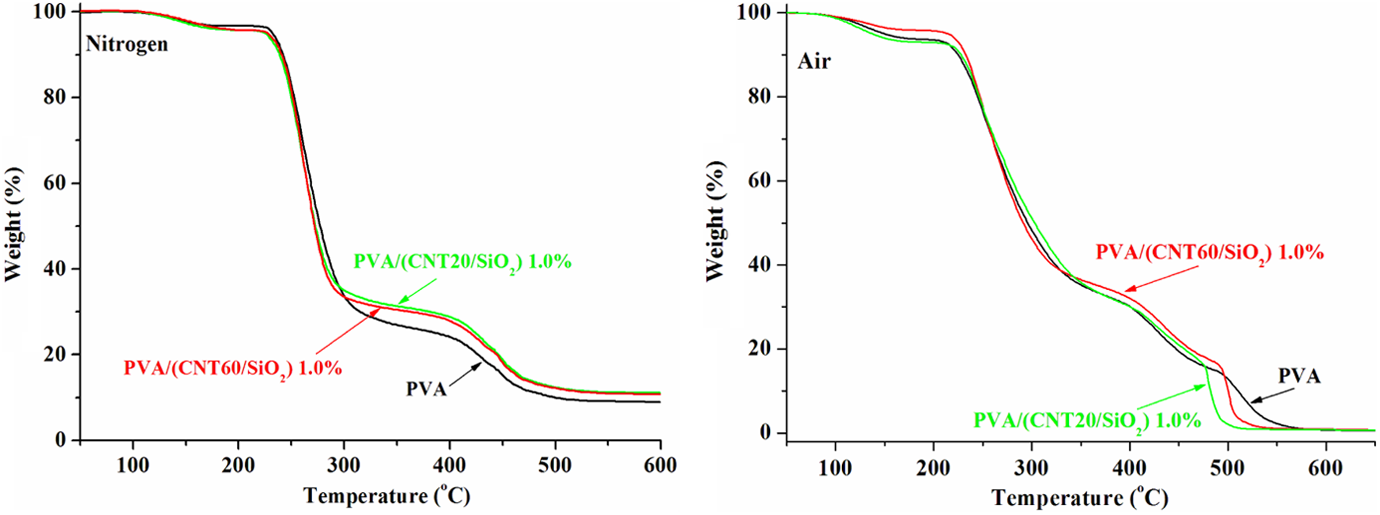
TGA curves of PVA/(CNT60/SiO2) composites tested under nitrogen and in air atmospheres.
Thermal properties of PU/(CNTs/SiO2) composites
PU is widely used in various industries because of its excellent physical and chemical properties. Comparisons of thermal degradation and thermo-oxidative stability of PU with PU/(CNTs/SiO2) composite are shown in Figures 7 and 8. The interactions between the surface hydroxyl groups on SiO2 and the –NH and –COO groups of PU are expected to improve the thermal stability of the PU/(CNTs/SiO2) composites.
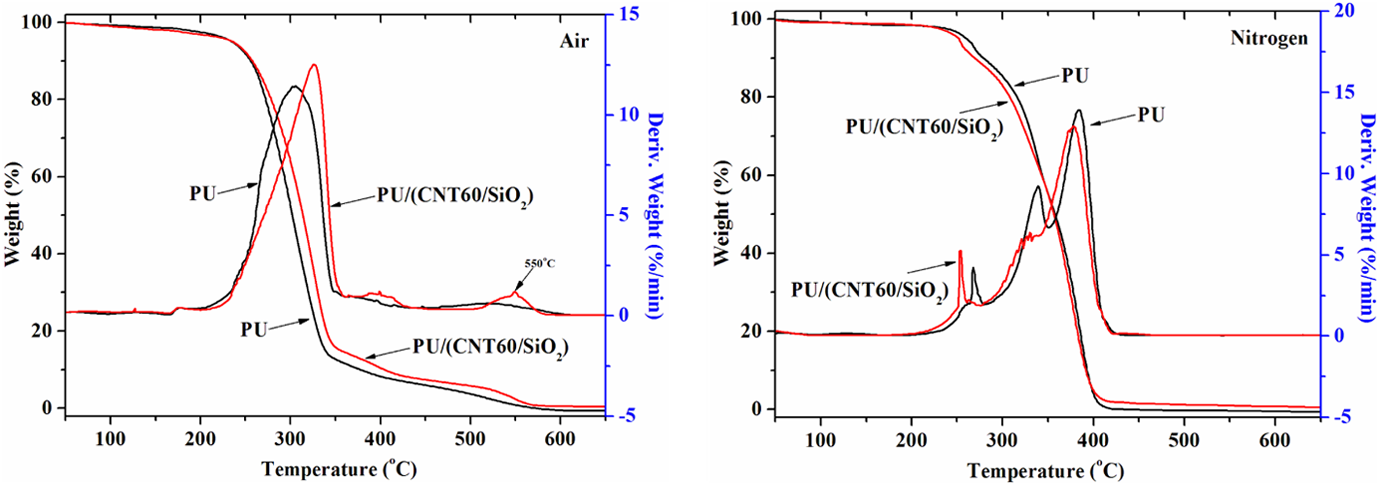
TGA and DTG curves of PU and PU/(CNT20/SiO2) composites tested under nitrogen and in air atmospheres.
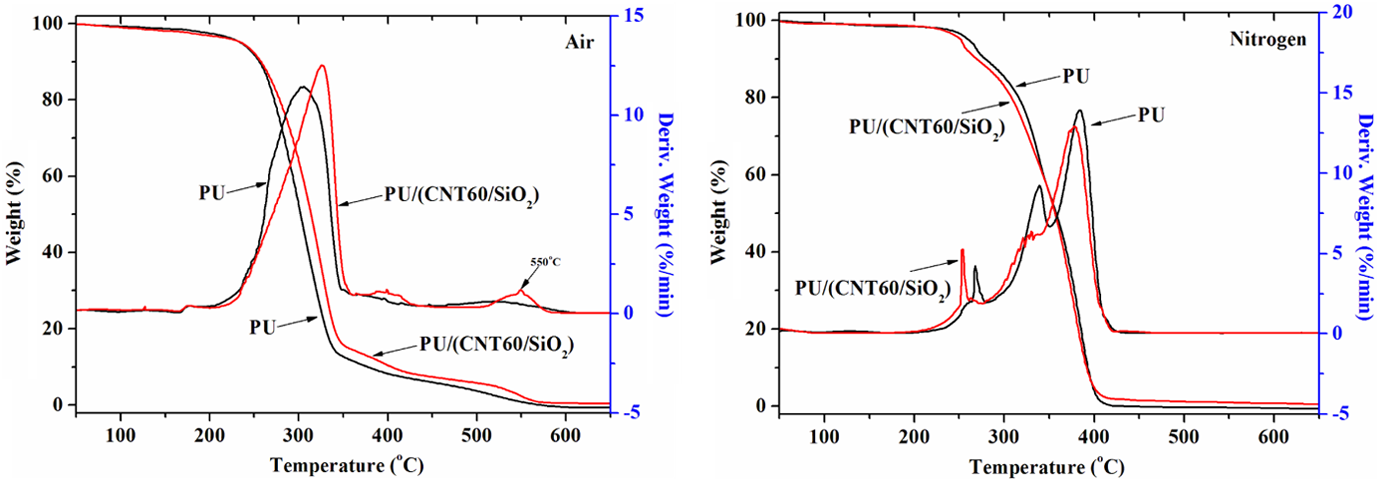
TGA and DTG curves of PU and PU/(CNT60/SiO2) composites tested under nitrogen and in air atmospheres.
Figure 7 (left) shows that when tested in an air atmosphere, the PU/(CNT20/SiO2) composite exhibits enhanced thermo-oxidative stability above 340°C compared to pure PU. A broad and weak peak centered at 522°C (T 2max) caused by the thermo-oxidation of the carbonized residue from PU can be found from the DTG curve, while for PU/(CNT20/SiO2) composite, it leaves much more residual char and the carbonized residue exhibits 23°C increase in thermo-oxidative stability (Table 2). As shown in Figure 7 (right), the thermal degradation of all samples occurs through three successive processes. PU/(CNT20/SiO2) composite starts to thermal degrade earlier compared to pure PU, but with elevating temperature above 300°C it exhibits an approximate increase of 5–15°C in thermal stability and leaves greater residual char.
TGA data of PU/(CNTs/SiO2) composites.
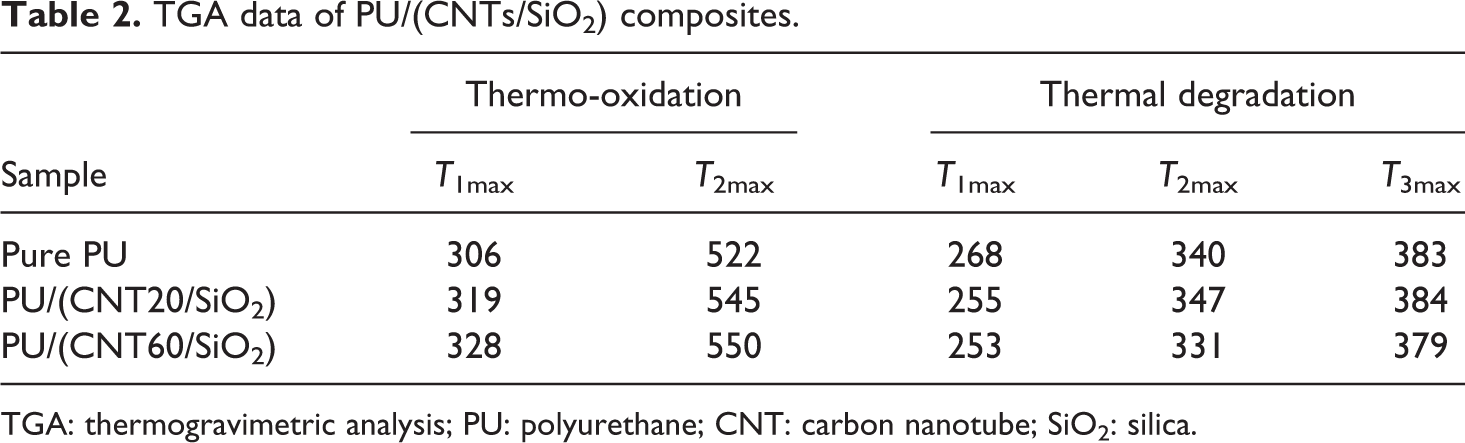
TGA: thermogravimetric analysis; PU: polyurethane; CNT: carbon nanotube; SiO2: silica.
Figure 8 shows the influence of CNT60/SiO2 nanohybirds on the thermal behavior of PU. Figure 8 (left) demonstrates that CNT60/SiO2 nanohybirds are more efficient than CNT20/SiO2 in improving the thermo-oxidative stability of PU, which evidences that T 1max of PU/(CNT60/SiO2) increases from 319°C to 328°C and the residual char is more stable than that from PU/(CNT20/SiO2). However, the presence of CNT60/SiO2 does not change the thermal degradation profile of pure PU, further deteriorating its thermal degradation stability. The differences in thermal behaviors of PU composites derived from the introducing of CNT20/SiO2 and CNT60/SiO2 may come from the differences in diameters of CNTs.
Conclusions
The influence of the CNTs/SiO2 nanohybrids with nanostructures of core/shell and candied haws on a stick-like structures on PVA and PU composites were investigated. It is noteworthy that the types of CNTs, the loading of the nanohybrids, and the interaction between the nanohybrids and the polymer matrices play important roles in the thermal stability of the materials. The incorporation of CNTs/SiO2 nanohybrids into the PVA matrix led to an increase in both thermal degradation and thermo-oxidative stability to some extent due to the formation of covalent bonds, but the improvements depend on the loading fraction of CNTs/SiO2. In the case of PU composites, the improvements in thermal properties were strongly influenced by the type of CNTs/SiO2 nanohybrids, and CNT20/SiO2 nanohybrids were more efficient than CNT60/SiO2 nanohybrids to enhance the thermal degradation stability of PU, but the tendency was contrary during thermo-oxidation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by National Natural Science Foundation of China (grant nos. 51276054 and 51403048), Program for Excellent Young Talents in University of Anhui Province, National Undergraduate Innovation and Entrepreneurship Training Programs (grant nos. 201311059005 and 201311059006), and DAAD (Deutscher Akademischer Austausch Dienst).
