Abstract
Poly(methyl)methacrylate (PMMA) composites with various contents of titanium dioxide (TiO2) were prepared. The effects of fiber content and modification on the mechanical properties and thermal behavior of PMMA composite film were studied. The use of organosolv lignin and acrylic acid together as a binary modifying agent for TiO2 filler increased both the strength and the ultimate strain of treated PMMA composites as compared to the untreated ones. The higher temperature dramatically increased the weight loss of all three sample categories. The organosolv lignin and acrylic acid mixture was more effective than a single modifying agent in reducing the thermal decomposition of PMMA/TiO2 composites.
Introduction
Polymers are becoming progressively attractive for a growing number of industries due to their low cost, ease and simplicity of manufacture and processing, low weight as well as the attractive look they furnish to any surface. 1 –3 However, abrasion and scratching are by and large the causes of the loss of their optical and tribological performances. 4 The surfaces of polymeric materials are predominantly sensitive to scratches, which obstruct their use in high-performance optical devices. In other state of affairs, these materials are used for moving mechanical parts, where friction and wear properties are important. 5 That is why there is a strong call for to search out improved scratch resistance of polymers for automotive and other applications that claim elevated surface excellence. 6
Plastics and polymers, such as poly(methyl)methacrylate (PMMA), are used as replacements for conventional materials (e.g. wood, metals, glass, etc.) in numerous domains such as electrical appliances, light diffusers, optical fibers, furniture, transport, hygiene and health, and so on. Indeed, these widespread kinds of synthetic compounds, with good technical, mechanical, chemical, optical performances, and so on, are easily manufactured for a moderate cost and can be used in a wide range of applications. 7,8 Thus, compounds made of PMMA can be used in numerous applications as signs and signboards (illuminated panels, three-dimensional (3D) lettering, indicator panels, etc.), point-of-sale advertising (display stands, testers, notice boards, etc.), interior design (shop fitting, furniture, projection screens, glazing, etc), transport (deflectors, sun visors, registration plates, ship portholes and windows, etc.), or industrial (machine guards, dials, precision parts, etc.) devices. In spite of these various uses linked to numerous advantages, plastics (highly combustible materials) constitute a grave danger (human injuries and deaths) in case of fire, as under the right conditions, they readily ignite and burn vigorously. 9
As a reference fuel material used in a cone calorimeter, solid acrylic PMMA polymer has been widely used—with or without filler—during previous studies for assessing polymer flammability and characterizing the mass loss rate during combustion processes. 10
Although about 200 years have passed since the discovery of TiO2 and 40 years since the revelation of photocatalytic activity of nano-TiO2, 11 –13 a limited research work is reported about the proper distribution of nano-TiO2 in cement media and the effect of nano-TiO2 dispersion on its photocatalytic activity. Most research works have reported on the primary particle size of nano-TiO2 and its mixing procedure with water and cement. They have not however concentrated on the final size and particle size distribution of nano-TiO2 in alkaline medium of cement paste.
Based on more detailed studies, when nano-TiO2 is used, it is inclined to strong agglomeration. 14 When nanoparticle powders are dispersed in solutions, their hydrodynamic size is larger than their primary size. After distribution in solution, nanoparticles tend to remain as agglomerates or aggregates, surrounded by an electrical double layer that includes both the inner and outer layers.
In this present study, our research has laid more stress on the mechanical properties and thermal behavior of the PMMA/TiO2 film, in which TiO2 particles were modified.
Experimental
Materials and the preparation process
Tetraethyl orthosilicate (TEOS, Aldrich, St Louis, Missouri, USA) was purified by distillation prior to use. Benzoyl peroxide (BPO) was purchased from Aldrich and recrystallized in chloroform and methanol. 3-(Trimethoxysilyl)propyl methacrylate (MSMA, Aldrich) and methyl methacrylate (MMA, Aldrich) were purified by the standard procedures and distilled under a reduced pressure prior to use. Pure commercial TiO2 (Degussa P-25, Sigma-Aldrich, St Louis, Missouri, USA) particles were also used.
Hydrochloric acid (37%, Riedel-De Haen, Germany), n-hexane (85%; Tedia, Fairfield, Ohio, USA), and toluene (99.8%; Mallinckrodt, St Louis, Missouri, USA) were used as received without further purification.
Synthesis of PMMA
A typical procedure to prepare PMMA is given as follows: 22.44 g (20.4 mmol) of MMA, 2.48 g (10.2 mmol) ofBPO, and 64 mL toluene were put into a three-necked-round bottomed flask connected to a condenser, thermometer, and nitrogen gas inlet/outlet. Under magnetic stirring, the solution was heated at 75°C for 3 h. The reaction mixture solution was then poured into 800 mL of n-hexane to precipitate the polymer. After filtration, the polymer was dissolved in 50 mL toluene followed by reprecipitating in 800 mL of n-hexane. The purification procedure was repeated at least twice, and the purified polymer was dried under vacuum at room temperature for 48 h.
Synthesis of sol–gel precursors (copolymers)
Sol–gel precursors were prepared in high yields by performing free radical copolymerizations of MSMA with MMA using BPO as the initiator. A typical procedure to prepare poly(MMA-co-MSMA) is given as follows:
First, 10.22 g (102 mmol) of MMA, 6.34 g (25 mmol) MSMA, 1.50 g of BPO, and 40 mL of toluene were put into a three necked-round bottomed flask connected to a condenser, thermometer, and nitrogen gas inlet/outlet. Under magnetic stirring, the solution was heated at 75°C for 3 h. The reaction mixture solution was then poured into 800 mL of n-hexane to precipitate the as-prepared copolymer. After filtration, the copolymer was dissolved in 50 mL of toluene followed by reprecipitating in 800 mL of n-hexane. The purification procedure was repeated at least twice, and the purified copolymer was dried under vacuum at room temperature for 48 h.
Different weight percentages of TEOS were mixed with BA (a solution followed by adding benzoxazine-functionalized silane coupling agent).
Results and discussion
Tensile properties
A tensile test was conducted to determine the strength of PMMA/TiO2 composites when the force is given in tension. Figure 1 shows stress versus strain traces recorded from tensile testing for neat PMMA, untreated PMMA composite, and treated PMMA composite containing 20 php of TiO2 filler. All traces exhibited a similar character; however, the treated PMMA/TiO2 composites showed higher strength and strain compared with the neat PMMA and the untreated composite. The presence of TiO2 as filler significantly influenced the strength of PMMA composites. Additionally, the chemical treatment did not affect the stress versus strain trace character of the treated PMMA/TiO2 composites, but it influenced the measured strength at failure. This outcome revealed that the use of organosolv lignin and acrylic acid together as a binary modifying agent for TiO2 filler increased both the strength and the ultimate strain of treated PMMA composites as compared to the untreated ones.
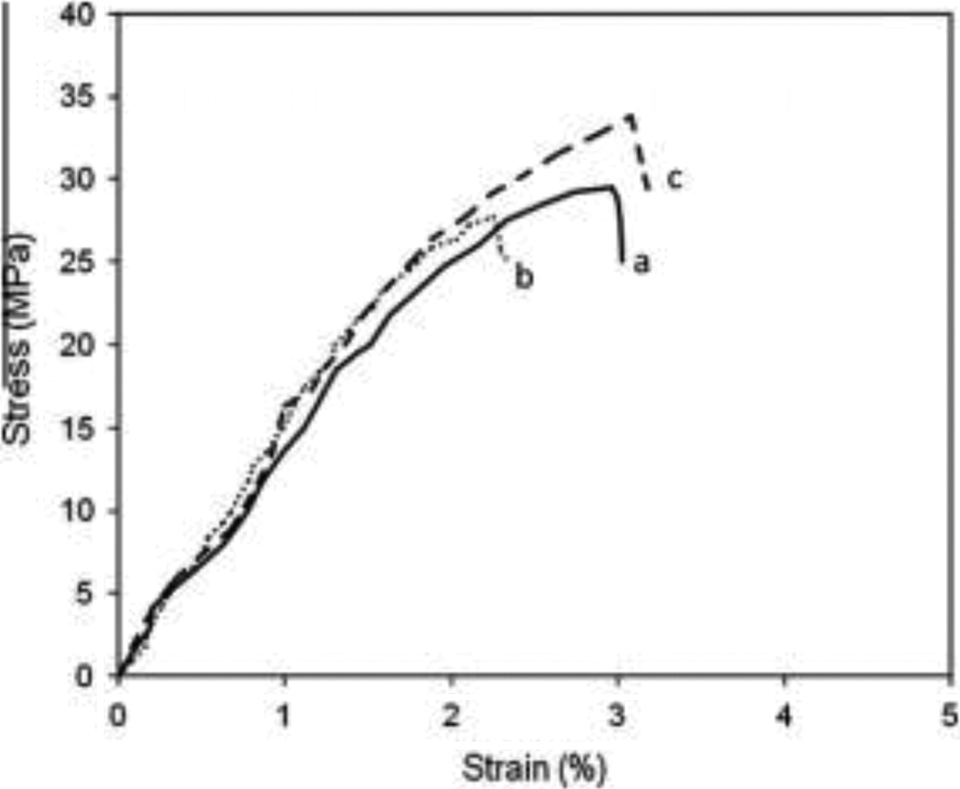
Stress versus strain traces of (a) neat PMMA; (b) untreated PMMA/TiO2 composites; and (c) treated PMMA/TiO2 composites. PMMA: poly(methyl methacrylate); TiO2: titanium dioxide.
The tensile strength of PMMA/TiO2 composites as a function of filler loading is depicted in Figure 2. The tensile strength of the composites decreased upon increasing filler loading, which is a consequence for lack of interaction at the TiO2 and PMMA matrix interface. The different affinities diminish filler–matrix interfacial adhesion. Hence, as the TiO2 loading increased, thereby increasing the interfacial area, the interfacial interaction between the filler and the matrix degraded and caused decreased tensile strength. However, due to the presence of organosolv lignin and acrylic acid on the TiO2 surface, the tensile strength of treated PMMA/TiO2 composites had higher values than the untreated composites of the same filler loading. The organosolv lignin was expected to react with the acrylic acid through a nucleophilic addition reaction between the aromatic group of organosolv lignin and the carbon–carbon double bond of acrylic acid. This product then attached to the TiO2 surface through the formation of ester linkage between the hydroxyl group of TiO2 and the carboxylic group of acrylic acid. The formation of this bonding decreased the hydrophilic character of the TiO2 leading to enhanced interfacial adhesion with the PMMA matrix. The highest tensile strength was found at 10 php filler loading, considerably due to the reason that at this concentration the filler was likely well wetted and dispersed. Upon increasing the filler loading, physical networks of filler agglomerated leading to the decreased tensile strength. In addition, the chemical modification of TiO2 using binary organosolv lignin and acrylic acid was comparable to single acrylic acid. The tensile strength was 1.82% lower on average than the tensile strength results obtained from previous work, thereby making the organosolv lignin a potential reagent to partially replace the fully synthetic counterpart. On the other hand, it was also found that the tensile strength results of the composites treated with binary modifying agent increased by 12.11% and 3.81% in comparison with the tensile strength results of composites treated with 3-aminopropyl-triethoxysilane (3-APE) and composites treated with sodium dodecyl sulfate, respectively. The increased tensile strength clearly indicated that better interfacial bonding between the filler and the matrix occurred upon chemical modification using a binary modifying agent.
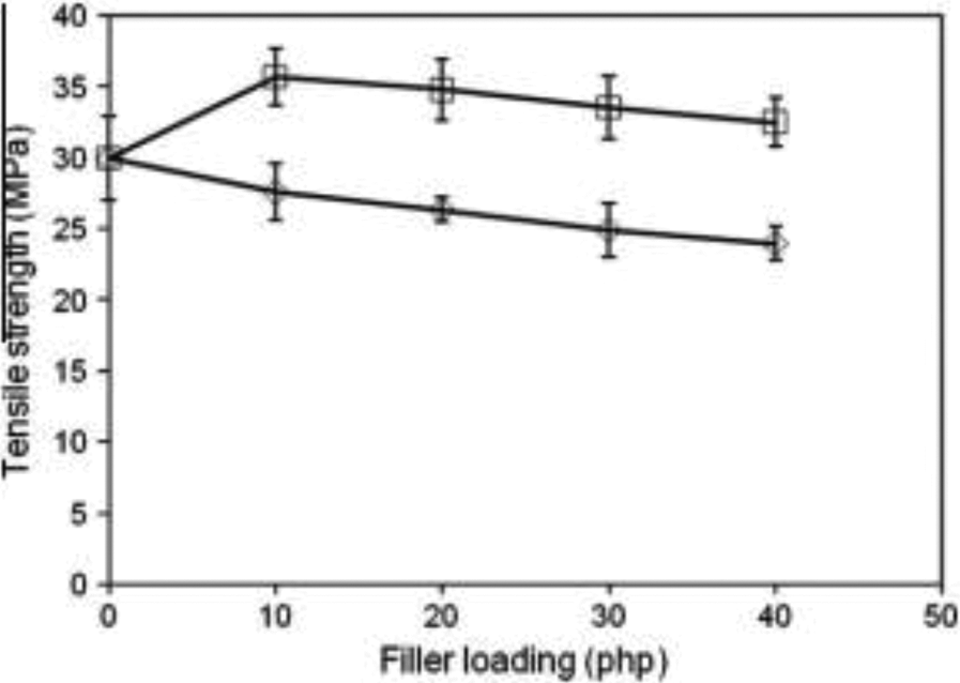
Effect of TiO2 loading on tensile strength of PMMA/TiO2 composite. PMMA: poly(methyl methacrylate); TiO2: titanium dioxide.
Figure 3 shows the Young’s modulus of PMMA/TiO2 composites at different levels of filler loading. Larger filler loading significantly increased the Young’s modulus of PMMA/TiO2 composites. The incorporation of TiO2 into the PMMA matrix decreased the deformability and therefore improved the rigidity of PMMA composites as a result of constricted molecular motion of the PMMA chain. Furthermore, the Young’s modulus of treated PMMA/TiO2 composites was significantly higher than untreated composites. This is because the presence of the aromatic group from the organosolv lignin considerably restricted the PMMA chain movements. In addition, improvement in the rigidity of the composites could be a consequence of chemical bonds between the polymer matrix and the surface of the TiO2 filler, which is enriched with organosolv lignin and acrylic acid.
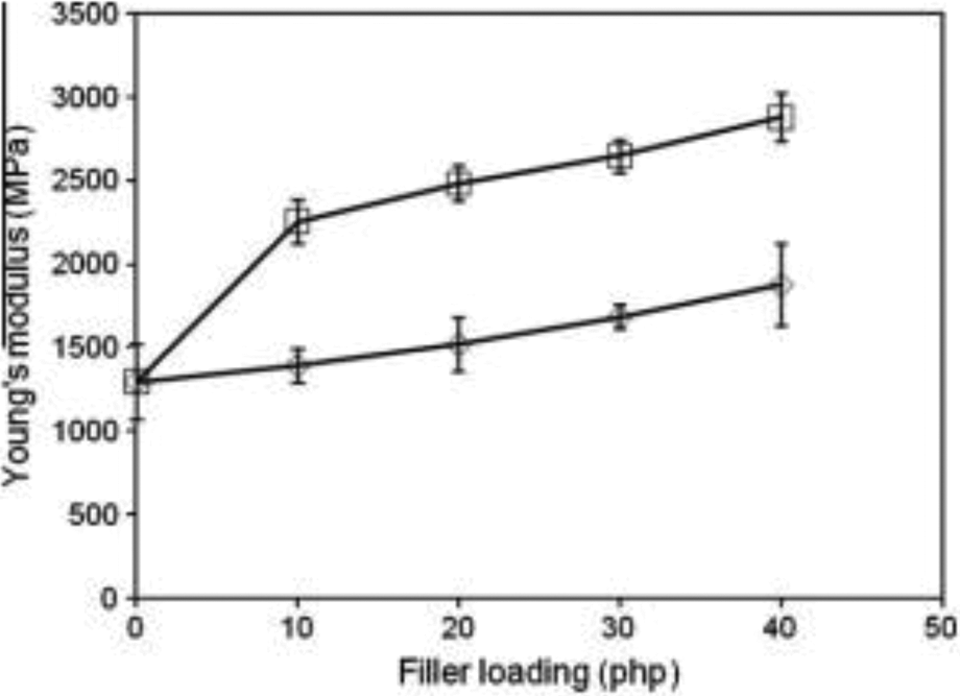
Variation of Young’s modulus of PMMA/TiO2 composite with loading. PMMA: poly(methyl methacrylate); TiO2: titanium dioxide.
Impact strength
The impact strength is an important parameter to determine the ability of polymeric materials to resist a sudden impact, because the polymers are subjected to this type of requirement in a large number of applications. Figure 4 shows impact strength of PMMA/TiO2 composites at different filler loading. Impact strength of the PMMA/TiO2 composites remarkably increases with an increase of filler loading, reaches its maximum at 30 php filler loading and then decreases at 40 php. This result clearly indicates that TiO2 has a positive contribution in improving the impact strength of the PMMA composites at filler loading of 10, 20, and 30 php, and then it declines at 40 php. The decreased impact strength of the PMMA/TiO2 composites at 40 php may be as a consequence of interaction between filler–filler that provides points of stress concentrations, thus imparting the fracture process. As a result, energy dissipation is lower and hence impact strength decreases. However, the treated PMMA/TiO2 composites are found to have higher impact strength than the untreated PMMA/TiO2 composites, indicating that better interfacial bonding between the matrix and the filler has occurred upon the presence of acrylic acid and organosolv lignin. The treated PMMA/TiO2 composites are capable of absorbing higher amount of energy to stop crack propagation compared with the untreated PMMA/TiO2 composites. In addition, it is observed from Figure 4 that the treated PMMA/TiO2 composites have higher impact strength of approximately 6.35%, 3.42%, and 8.32% as compared to PMMA/TiO2 composites treated with 3-APE, PMMA/TiO2 composites treated with sodium dodecyl sulfate and PMMA/TiO2 composites treated with acrylic acid, respectively. This outcome implies that the organosolv lignin and acrylic acid mixture is more effective than a single modifying agent in improving the fracture resistance of the PMMA/TiO2 composites.
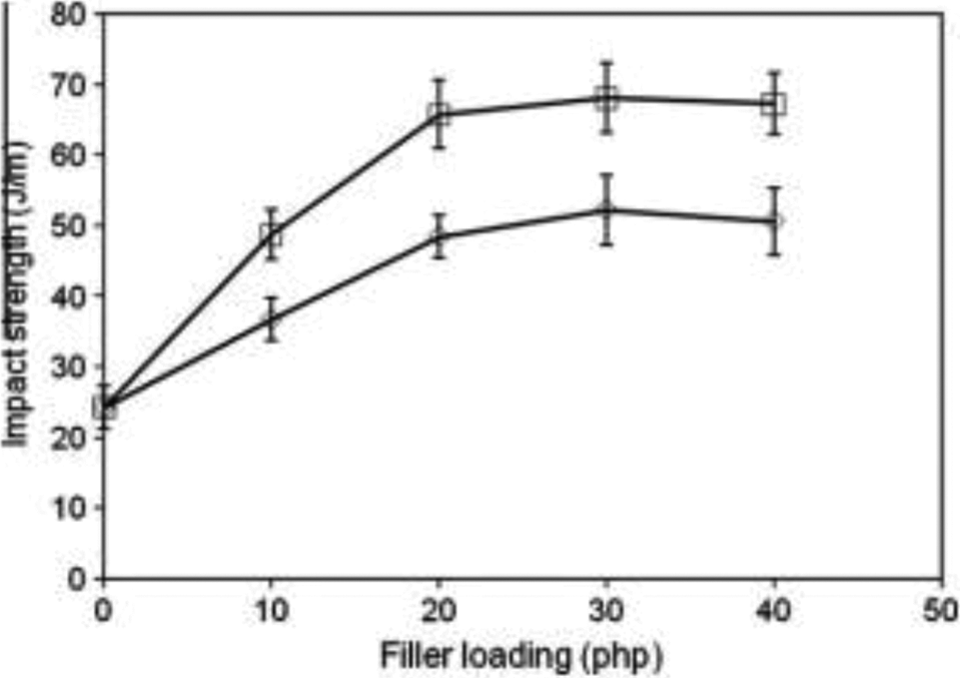
Impact strength of PMMA/TiO2 composite. PMMA: poly(methyl methacrylate); TiO2: titanium dioxide.
Thermal properties
Figure 5(a) and (b) presents the thermogravimetric analysis (TGA) and derivative TG (DTG) curves of neat PMMA, untreated PMMA/TiO2 composites, and treated PMMA/TiO2 composites. The higher temperature dramatically increased the weight loss of all three sample categories. The thermal degradation of PMMA occurred in one stage between 300 and 415°C, resulting in gaseous products. This confirmed that the degradation/depolymerization takes place at the weak sites of the PMMA chain. Alternatively, the thermal degradation of the PMMA/TiO2 composites occurred over two stages; from 50 to 300°C corresponding to the release of typical strong hydrogen-bonded water and the breakage of chemical bonds and from 300 to 450°C associated with decomposition and depolymerization of the TiO2 filler and modifying agents. This outcome indicates that the chemical modification apparently did not alter the thermal degradation mechanism of the PMMA/TiO2 composites. In addition, there was another degradation stage occurring from 470 to 600°C that was attributable to the decomposition of char residue formed from the second stage. Overall, the treated PMMA/TiO2 composites had better thermal stability than untreated composites of the same filler loading. The presence of organosolv lignin on the surface of TiO2 acted as a protective barrier against thermal degradation.
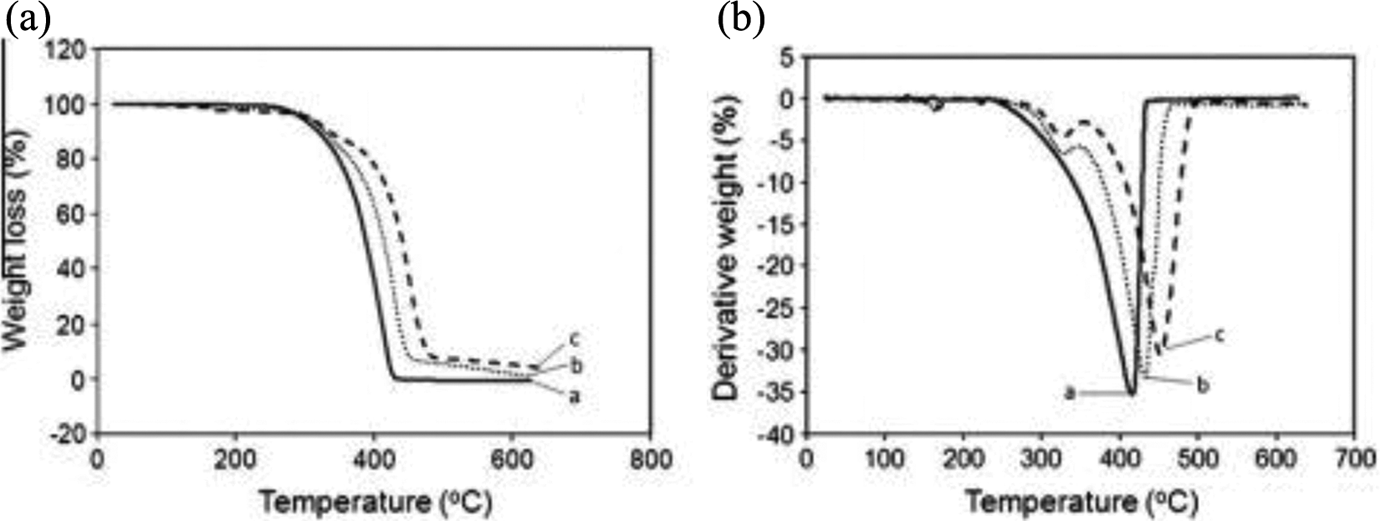
Effect of TiO2 addition and modification on the thermogravimetric properties of PMMA/TiO2 composite. TiO2 content: 20 php. (a) Weight loss versus temperature curves and (b) derivative thermogravimetric curves. The curves with alphabets c, d, and e correspond to neat PMMA, untreated PMMA/TiO2 composites, and treated PMMA/TiO2 composites, respectively. PMMA: poly(methyl methacrylate); TiO2: titanium dioxide.
Organosolv lignin has a 3D aromatic polymer structure capable of reducing the thermal decomposition rate of polymeric materials. This ability combined with the heterogeneity of the chemical bonds in the cross-linked structure is the main defensive effort to degradation. As a result, the thermal stability of the PMMA/TiO2 composites treated with binary organosolv lignin and acrylic acid were higher than the PMMA/TiO2 composites treated with only acrylic acid. This suggests that the organosolv lignin and acrylic acid mixture was more effective than a single modifying agent in reducing the thermal decomposition of PMMA/TiO2 composites.
TG and DTG curves of the studied samples are shown in Figure 6. TiO2 presents three peaks of weight mass and an onset degradation temperature at 150°C. The first peak below 110°C was the evaporation of moisture and was around 9%. The peaks at 260 and 315°C were caused by lignin and cellulose degradation, respectively. Figure 6(b) shows four degradation peaks related to PMMA. The first peak corresponds to the water loss in the resin polymerization reaction and the second can be attributed to the onset degradation temperature at 220°C. In the initial stage, decomposition starts to the loss of some small end groups, namely –CH2OH. The second and third stages may correspond to the loss of small groups and weaker bonds in the chains, such as –OH and –CH2–. The fourth peak corresponds to the loss of benzene rings.
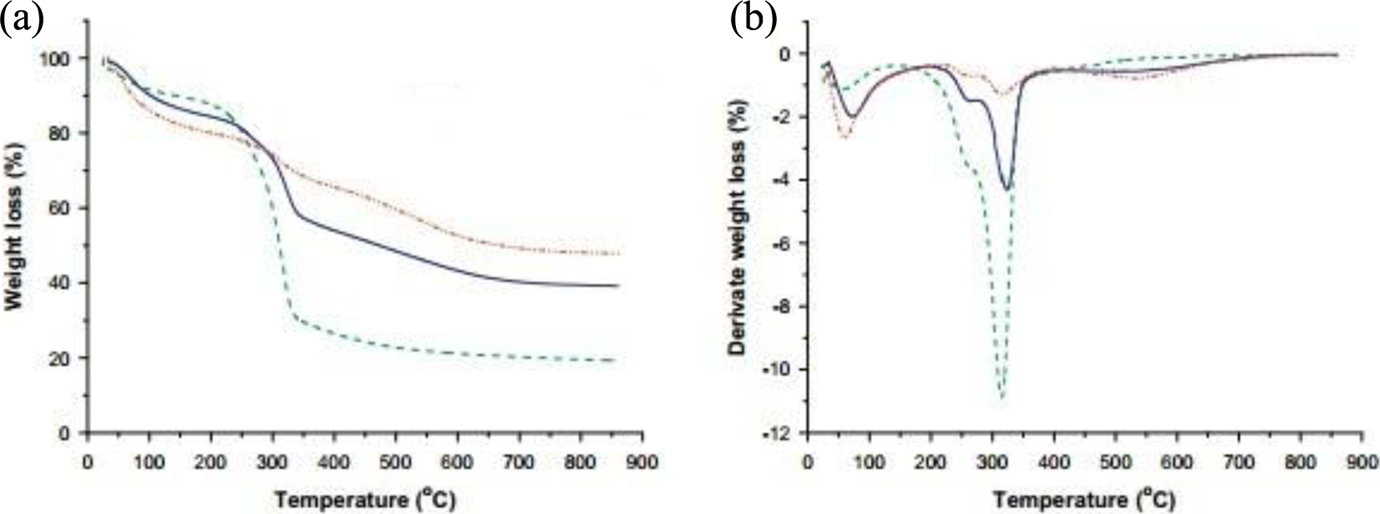
(a) Thermogravimetric curves and (b) derivative thermogravimetric curves.
Conclusions
The use of organosolv lignin and acrylic acid together as a binary modifying agent for TiO2 filler increased both the strength and the ultimate strain of treated PMMA composites as compared to untreated ones. The thermal degradation of the PMMA/TiO2 composites occurred in two stages; from 50 to 300°C corresponding to the release of typical strong hydrogen-bonded water and the breakage of chemical bonds. In conclusion, the mechanical properties of the PMMA/TiO2 composites with treatment were higher than that with the untreated one, although their overall behavior was broadly similar.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present work was financially supported by the National Science Foundation of China (51205359).
